Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.112974
Revised: November 25, 2025
Accepted: January 4, 2026
Published online: March 15, 2026
Processing time: 213 Days and 6.2 Hours
Endoscopic ultrasound-guided radiofrequency ablation (EUS-RFA) has emerged as a promising minimally invasive treatment option for pancreatic cancer, offering precision and reduced morbidity compared to traditional surgical methods. Re
Core Tip: Endoscopic ultrasound-guided radiofrequency ablation (EUS-RFA) is a promising minimally invasive treatment for pancreatic cancer, particularly in patients with unresectable or high-risk tumors. Recent technological advancements have improved procedural precision and safety, demonstrating high technical success rates and potential symptom palliation. When combined with chemotherapy, EUS-RFA may offer enhanced tumor control and survival benefits, though long-term clinical outcomes require further investigation. Additionally, the immunomodulatory effects of EUS-RFA may augment systemic anti-tumor immunity, offering a potential synergy with immunotherapies. However, larger randomized controlled trials and standardized protocols are needed to fully establish its role in the clinical management of pancreatic cancer.
- Citation: Tong C, Hui YQ, Wang JR. Endoscopic ultrasound-guided radiofrequency ablation: A game-changer in pancreatic cancer treatment. World J Gastrointest Oncol 2026; 18(3): 112974
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/112974.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.112974
Pancreatic cancer is one of the most aggressive malignancies, with a five-year survival rate of less than 10%, making it one of the deadliest cancers worldwide[1,2]. The majority of patients present with advanced or unresectable disease at the time of diagnosis, and only a small subset of patients is eligible for curative surgical resection[1,3]. Surgical resection remains the only potentially curative treatment but is feasible in fewer than 20% of cases, highlighting the need for alternative therapies that can effectively manage or control the disease[1,3].
Over the past two decades, radiofrequency ablation (RFA) has emerged as a promising minimally invasive therapy for pancreatic cancer, evolving from intraoperative and percutaneous techniques to endoscopic ultrasound-guided RFA (EUS-RFA). EUS-RFA allows for precise targeting of pancreatic lesions using real-time imaging, minimizing damage to adjacent healthy tissues and reducing procedural morbidity[4,5]. This development has significantly expanded the potential applications of RFA, which is now used not only for treating liver tumors but also for pancreatic neuroendocrine tumors, cystic lesions, and pancreatic ductal adenocarcinoma[4,6].
Despite these advancements, the management of pancreatic cancer remains a formidable challenge. The disease's aggressive biology, dense stromal microenvironment, and resistance to conventional therapies complicate treatment efforts[7,8]. While EUS-RFA has demonstrated technical feasibility and safety in various studies, there remains a lack of consensus regarding its long-term efficacy, optimal patient selection, and its comparative effectiveness against other standard treatments[6,9,10]. Furthermore, controversies persist about the extent of survival benefits, the immunomodulatory effects of the procedure, and the adverse event profiles, with some studies reporting promising outcomes and others indicating limited impact on overall survival (OS)[6,11]. These uncertainties have led to confusion in clinical guidelines and have contributed to the underutilization of this potentially valuable therapeutic modality[3,12].
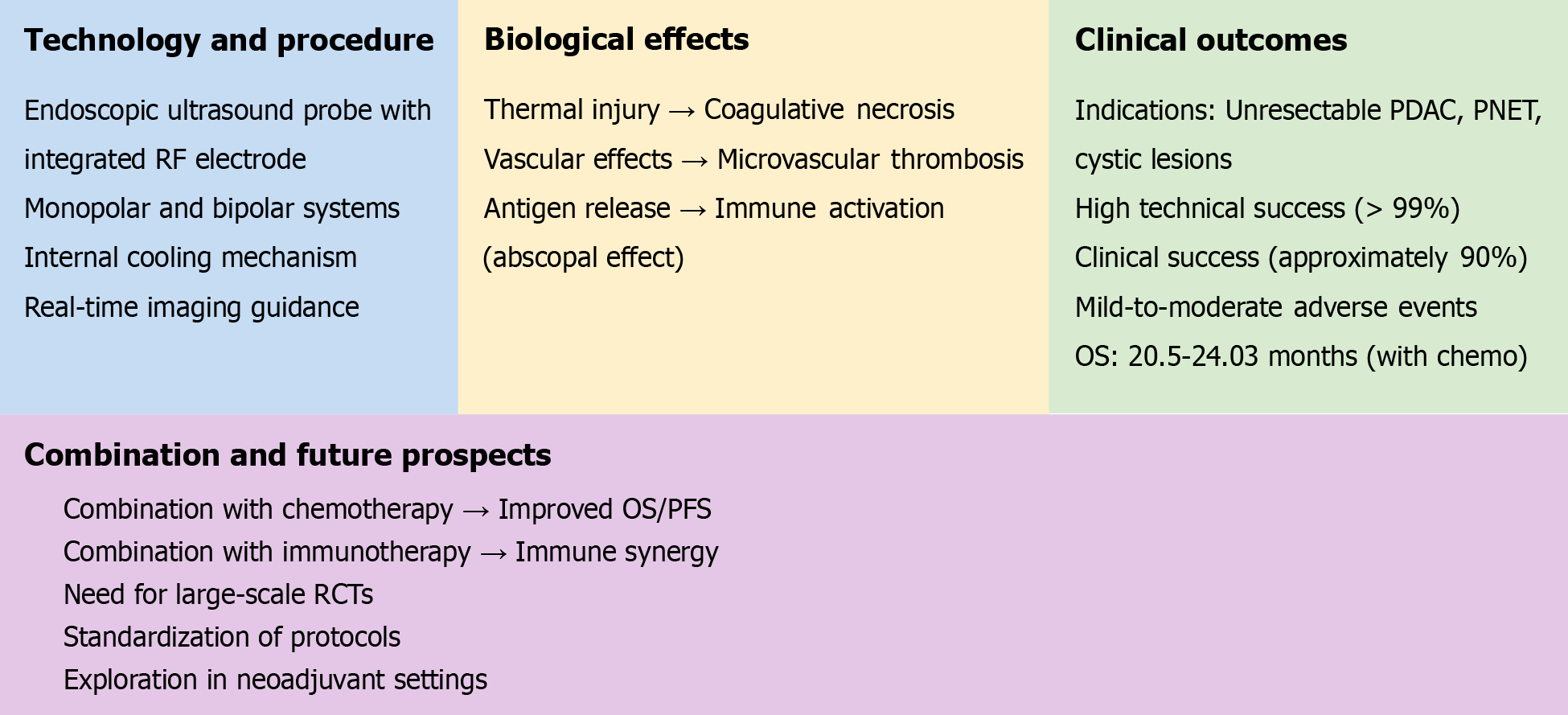
One of the primary advancements in RFA for pancreatic cancer has been the integration of EUS-RFA, which significantly enhances the precision of tumor targeting. EUS-RFA allows for real-time imaging guidance, enabling clinicians to precisely localize the tumor and ablate it with minimal impact on surrounding healthy tissues[4,6]. The overall workflow and biological effects of EUS-RFA are illustrated in Figure 1. The development of specialized monopolar and bipolar electrode systems has further improved RFA's precision and safety, making it possible to treat tumors that were previously considered inoperable or high-risk for conventional surgery[1,5]. Bipolar systems, in particular, allow for more controlled and localized ablation, reducing the risk of thermal injury to adjacent organs[13]. These technological innovations also include systems with internal cooling mechanisms, which help to prevent collateral damage and improve procedural safety[1].
Furthermore, advancements in RFA probe technology have enabled better targeting and more efficient delivery of energy. For instance, the use of probes with tailored energy settings and the development of probes with integrated cooling systems have improved the procedure's overall efficiency, reduced complications, and increased tumor necrosis rates[1,14]. These developments are critical for increasing the procedural success rates of EUS-RFA and expanding its applications to a wider range of pancreatic lesions, including not only pancreatic ductal adenocarcinoma but also pancreatic neuroendocrine tumors and cystic lesions[4,6].
Despite these advances, there is still a lack of consensus on optimal RFA protocols, including energy levels, ablation duration, and the frequency of sessions[1,6]. The variability in RFA devices and settings between different centers complicates direct comparisons of outcomes and hinders the development of standardized procedural guidelines[4]. Additionally, although the integration of cooling systems and more advanced energy modulation has reduced some of the risks associated with thermal injury, further improvements in device precision and operator training are needed to minimize adverse events, particularly in tumors located near critical structures[15,16].
Clinical studies on EUS-RFA have consistently demonstrated high technical success rates, with some studies reporting rates of up to 99%[9]. Clinical success, defined as tumor size reduction or necrosis, has been observed in approximately 85% to 90% of patients[4]. EUS-RFA has shown particular promise in providing symptom relief, especially in terms of pain management, and improving the quality of life for patients with unresectable pancreatic cancer[4,9]. These findings are significant, as pancreatic cancer is often associated with severe pain and discomfort, particularly in the advanced stages.
In addition to symptom palliation and tumor cytoreduction, emerging evidence suggests that EUS-RFA may also contribute to downstaging borderline resectable pancreatic tumors, thereby increasing the feasibility of subsequent radical surgical resection in selected patients[17]. Although current data remain preliminary and mostly derived from small cohorts or case series, several reports have shown partial tumor shrinkage and improved vascular clearance after RFA, indicating its potential role as an adjunct to neoadjuvant strategies[18].
In terms of safety, EUS-RFA has a favorable safety profile. The most commonly reported adverse events include mild to moderate abdominal pain and pancreatitis, both of which are generally self-limited and manageable[9,19]. More severe complications, such as perforation or bleeding, are rare and occur in fewer than 5% of cases[9]. This makes EUS-RFA an attractive alternative for patients who are high surgical risks or have tumors that are otherwise not amenable to surgical resection[19].
However, despite these promising results, the long-term clinical benefits of EUS-RFA remain unclear. While some studies report improved survival rates in patients treated with EUS-RFA in combination with chemotherapy[20], the evidence remains limited, and further research is needed to establish the definitive role of EUS-RFA in extending survival. The need for more randomized controlled trials with longer follow-up periods is critical to determine the long-term efficacy of EUS-RFA in pancreatic cancer treatment[1,2].
Importantly, not all studies have demonstrated favorable or meaningful clinical benefits. Several reports have shown only modest tumor response or no significant extension in OS, particularly in patients with advanced disease or large tumor burden[21]. These less favorable findings highlight that EUS-RFA is not universally effective and that its clinical benefit may depend strongly on tumor biology, stage, and procedural factors. Incorporating these results ensures a balanced perspective and underscores the need for cautious interpretation of existing evidence.
EUS-RFA has been compared with other treatment modalities, such as chemotherapy, surgery, and other locoregional therapies, to assess its effectiveness in managing pancreatic cancer. The combination of EUS-RFA with chemotherapy has shown encouraging results, particularly in terms of improving tumor control and reducing the need for pain medication. Some studies suggest that this combination may lead to improved OS and progression-free survival compared to chemotherapy alone[20]. Median OS in patients receiving EUS-RFA combined with chemotherapy has been reported to range from 20.5 months to 24.03 months[20], which is promising compared to historical controls.
Beyond survival benefits, EUS-RFA has also been investigated as a potential tool for downstaging borderline resectable pancreatic cancer[22]. By reducing tumor volume or decreasing encasement of major vascular structures, RFA may faci
In comparison to surgical resection, EUS-RFA offers a less invasive option with reduced morbidity and mortality. While surgery remains the gold standard for resectable pancreatic tumors, EUS-RFA provides a valuable option for patients who are ineligible for surgery due to comorbidities or tumor location[1,25]. However, more direct comparisons between EUS-RFA and other locoregional therapies, such as microwave ablation (MWA) or irreversible electroporation (IRE), are needed to assess its relative efficacy[15,16]. Ongoing trials like the PELICAN trial are evaluating the role of EUS-RFA combined with chemotherapy vs chemotherapy alone, which will provide more clarity on the overall effectiveness of this combination[12].
In addition to these preliminary comparisons, several studies have contrasted EUS-RFA directly with other emerging ablative modalities[26]. MWA, for instance, can achieve higher intratumoral temperatures and larger, more uniform ablation zones, but its application near major vascular structures may increase the risk of collateral injury[27]. In contrast, EUS-RFA offers more controlled and localized thermal delivery with real-time endoscopic visualization, making it potentially safer for lesions embedded within complex pancreatic anatomy[28].
IRE, another frequently compared modality, is nonthermal and therefore preserves the integrity of surrounding vasculature and ductal tissues[29]. While this makes IRE attractive for tumors encasing major vessels, it requires general anesthesia with neuromuscular blockade and is associated with higher procedural complexity[30]. EUS-RFA, by comparison, can be performed under moderate sedation or deep sedation and involves shorter procedural time, though its ablation field is generally smaller[31].
Overall, these comparative data suggest that each modality – EUS-RFA, MWA, and IRE – possesses unique strengths and limitations. EUS-RFA stands out for its minimally invasive endoscopic access, real-time visualization, and relatively low complication rates, whereas MWA and IRE may be preferable in selected anatomic or tumor-specific scenarios. Continued head-to-head research will be critical for refining optimal treatment algorithms and patient selection.
Emerging evidence suggests that EUS-RFA has potential immunomodulatory effects that may enhance systemic anti-tumor immunity. RFA induces localized tumor destruction, which results in the release of tumor antigens, thereby activating the immune system and potentially leading to an abscopal effect, where immune responses target distant tumor sites[7,32]. This is particularly relevant in pancreatic cancer, where the tumor microenvironment is highly immunosuppressive, and traditional treatments have limited success in activating the immune system.
Several studies have explored the potential synergy between EUS-RFA and immunotherapies, such as immune checkpoint inhibitors. The combination of RFA with immune modulation may help overcome resistance mechanisms in the tumor microenvironment and enhance the efficacy of systemic treatments[7,32]. However, these findings are still in the early stages, and further research is required to validate the clinical benefits of combining EUS-RFA with immunotherapies[7].
While EUS-RFA has shown promise in treating pancreatic cancer, several challenges and controversies remain. There is a need for large-scale randomized controlled trials to better define the long-term efficacy of EUS-RFA, especially in combination with other therapies such as chemotherapy and immunotherapy[1,2]. Standardization of procedural protocols and patient selection criteria remains insufficient, further contributing to uncertainty in clinical outcomes[1,12].
Another important controversy concerns the inconsistency of clinical outcomes across studies. While many reports show favorable results, others demonstrate neutral or limited benefits, particularly regarding survival outcomes and long-term disease control. These discrepancies may be related to heterogeneity in patient selection, tumor characteristics, RFA parameters, and operator experience[33]. Recognition of these neutral or less positive findings is essential to avoid overstating the efficacy of EUS-RFA and to highlight the areas where further validation is needed.
Moreover, procedural success and complication rates appear to be strongly influenced by operator expertise and institutional case volume. Centers with extensive experience in advanced endoscopic interventions tend to achieve higher technical success and lower adverse event rates, whereas outcomes in low-volume or less specialized centers may be more variable. This dependence on operator skill and institutional capacity significantly limits widespread adoption and complicates the development of universally applicable clinical guidelines.
Current clinical guidelines from major professional societies continue to position EUS-RFA as an investigational or adjunctive modality rather than a standard therapeutic option. Both the American Society for Gastrointestinal Endoscopy and the European Association for the Study of the Liver note that although early clinical data are encouraging, the evidence remains insufficient to support routine incorporation of EUS-RFA into standard care pathways for pancreatic cancer.
Guidelines emphasize that the use of EUS-RFA should be limited to carefully selected patients and performed in high-experience centers with advanced endoscopic expertise, given the procedure’s dependence on operator skill, device familiarity, and institutional volume. Additionally, current recommendations highlight the need for further prospective trials to clarify optimal patient selection, procedural parameters, and long-term clinical outcomes before broader adoption can be recommended.
These guideline perspectives reinforce that EUS-RFA, while promising, remains an evolving technology whose role in pancreatic cancer management is still being defined within evidence-based frameworks.
In light of these controversies, several future research directions have emerged. There is a need for large-scale rando
In addition, the potential immunomodulatory effects of EUS-RFA open exciting avenues for future research. Studies investigating the combination of EUS-RFA with immune checkpoint inhibitors or other immune-modulating therapies may offer a new frontier in pancreatic cancer treatment[7,32]. This approach could significantly enhance the treatment landscape for pancreatic cancer, a disease that has historically been resistant to many therapeutic strategies.
Further technological refinement – including improvements in ablation probes, real-time temperature monitoring, and energy delivery modulation – may enhance both safety and efficacy. Expanding clinical evidence from high-volume centers and developing training frameworks may also help to decrease the variability in outcomes across institutions. Such advancements will be crucial for standardizing procedure protocols and expanding clinical adoption.
In conclusion, EUS-RFA represents a significant advancement in the treatment of pancreatic cancer, particularly for patients with unresectable or high-risk tumors. While it has shown excellent technical success rates and symptom relief, its long-term survival benefits remain uncertain. The integration of EUS-RFA with chemotherapy and immunotherapies may provide additional therapeutic advantages, and future research should focus on clarifying the role of EUS-RFA in multimodal treatment strategies. Larger randomized trials with long-term follow-up are essential to establish definitive guidelines and expand the clinical indications of EUS-RFA in pancreatic cancer management.
| 1. | Gollapudi LA, Tyberg A. EUS-RFA of the pancreas: where are we and future directions. Transl Gastroenterol Hepatol. 2022;7:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 2. | Farmer W, Hannon G, Ghosh S, Prina-Mello A. Thermal ablation in pancreatic cancer: A scoping review of clinical studies. Front Oncol. 2022;12:1066990. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 3. | Lesmana CRA. Impact of endoscopic ultrasound-guided radiofrequency ablation in managing pancreatic malignancy. World J Gastrointest Surg. 2023;15:163-168. [PubMed] [DOI] [Full Text] |
| 4. | Coluccio C, Cappetta S, Romagnoli G, Di Giorgio V, Giuffrida P, Fabbri S, Fabbri C, Binda C. Endoscopic-Ultrasound-Guided Radiofrequency Ablation for Pancreatic Tumors. J Clin Med. 2025;14:495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 5. | Changela K, Patil R, Duddempudi S, Gaduputi V. Endoscopic Ultrasound-Guided Radiofrequency Ablation of the Pancreatic Tumors: A Promising Tool in Management of Pancreatic Tumors. Can J Gastroenterol Hepatol. 2016;2016:4189358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Khoury T, Sbeit W, Napoléon B. Endoscopic ultrasound guided radiofrequency ablation for pancreatic tumors: A critical review focusing on safety, efficacy and controversies. World J Gastroenterol. 2023;29:157-170. [PubMed] [DOI] [Full Text] |
| 7. | Moond V, Maniyar B, Harne PS, Bailey-Lundberg JM, Thosani NC. Harnessing endoscopic ultrasound-guided radiofrequency ablation to reshape the pancreatic ductal adenocarcinoma microenvironment and elicit systemic immunomodulation. Explor Target Antitumor Ther. 2024;5:1056-1073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 8. | De Grandis MC, Ascenti V, Lanza C, Di Paolo G, Galassi B, Ierardi AM, Carrafiello G, Facciorusso A, Ghidini M. Locoregional Therapies and Remodeling of Tumor Microenvironment in Pancreatic Cancer. Int J Mol Sci. 2023;24:12681. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 9. | Ehsan H, Masood A, Wahab A, Ahmed Z, Franco D, Rafae A, Yousaf MN. Efficacy and the safety of endoscopic ultrasound guided radiofrequency ablation of pancreatic cancer: A systematic review and meta-analysis. J Clin Oncol. 2022;40:e16276-e16276. [DOI] [Full Text] |
| 10. | Tiankanon K, Kongkam P, Luangsukrerk T, Sriuranpong V, Nantavithya C, Jantarattana T, Canones A, Angsuwatcharakon P, Ridtitid W, Kullavanijaya P, Rerknimitr R. IDDF2019-ABS-0171 EUS-guided radiofrequency ablation as adjunctive treatment for unresectable pancreatic cancer versus chemotherapy alone (ERAP). Clin Gastroenterol. 2019. [DOI] [Full Text] |
| 11. | Yousaf MN, Ehsan H, Muneeb A, Wahab A, Sana MK, Neupane K, Chaudhary FS. Role of Radiofrequency Ablation in the Management of Unresectable Pancreatic Cancer. Front Med (Lausanne). 2020;7:624997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 12. | Walma MS, Rombouts SJ, Brada LJH, Borel Rinkes IH, Bosscha K, Bruijnen RC, Busch OR, Creemers GJ, Daams F, van Dam RM, van Delden OM, Festen S, Ghorbani P, de Groot DJ, de Groot JWB, Haj Mohammad N, van Hillegersberg R, de Hingh IH, D'Hondt M, Kerver ED, van Leeuwen MS, Liem MS, van Lienden KP, Los M, de Meijer VE, Meijerink MR, Mekenkamp LJ, Nio CY, Oulad Abdennabi I, Pando E, Patijn GA, Polée MB, Pruijt JF, Roeyen G, Ropela JA, Stommel MWJ, de Vos-Geelen J, de Vries JJ, van der Waal EM, Wessels FJ, Wilmink JW, van Santvoort HC, Besselink MG, Molenaar IQ; Dutch Pancreatic Cancer Group. Radiofrequency ablation and chemotherapy versus chemotherapy alone for locally advanced pancreatic cancer (PELICAN): study protocol for a randomized controlled trial. Trials. 2021;22:313. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 13. | Scopelliti F, Pea A, Conigliaro R, Butturini G, Frigerio I, Regi P, Giardino A, Bertani H, Paini M, Pederzoli P, Girelli R. Technique, safety, and feasibility of EUS-guided radiofrequency ablation in unresectable pancreatic cancer. Surg Endosc. 2018;32:4022-4028. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 14. | Wang J, Wang Y, Zhao Y, Wu X, Zhang M, Hou W, Chen Q, Cheng B. Endoscopic ultrasound-guided radiofrequency ablation of unresectable pancreatic cancer with low ablation power and multiple applications: a preliminary study of 11 patients. Ann Palliat Med. 2021;10:1842-1850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 15. | Spiliopoulos S, Reppas L, Filippiadis D, Delvecchio A, Conticchio M, Memeo R, Inchingolo R. Irreversible electroporation for the management of pancreatic cancer: Current data and future directions. World J Gastroenterol. 2023;29:223-231. [PubMed] [DOI] [Full Text] |
| 16. | Sarkisian AM, Andalib I, Kumta NA, Sharaiha RZ. Radiofrequency ablation for pancreatobiliary disease. Curr Opin Gastroenterol. 2016;32:353-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 17. | Iancu I, Bartoș A, Cioltean CL, Breazu C, Iancu C, Bartoș D. Role of radio-ablative technique for optimizing the survival of patients with locally advanced pancreatic adenocarcinoma (Review). Exp Ther Med. 2021;22:853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Ji JH, Park SH, Lee J, Kim TW, Hong YS, Kim KP, Kim SY, Baek JY, Kang HJ, Shin SJ, Shim BY, Park YS. Prospective phase II study of neoadjuvant FOLFOX6 plus cetuximab in patients with colorectal cancer and unresectable liver-only metastasis. Cancer Chemother Pharmacol. 2013;72:223-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 19. | Spadaccini M, Di Leo M, Iannone A, von den Hoff D, Fugazza A, Galtieri PA, Pellegatta G, Maselli R, Anderloni A, Colombo M, Siersema PD, Carrara S, Repici A. Endoscopic ultrasound-guided ablation of solid pancreatic lesions: A systematic review of early outcomes with pooled analysis. World J Gastrointest Oncol. 2022;14:533-542. [PubMed] [DOI] [Full Text] |
| 20. | Thosani N, Cen P, Rowe J, Guha S, Bailey-Lundberg JM, Bhakta D, Patil P, Wray CJ. Endoscopic ultrasound-guided radiofrequency ablation (EUS-RFA) for advanced pancreatic and periampullary adenocarcinoma. Sci Rep. 2022;12:16516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 21. | Patel KS, Everson RG, Yao J, Raymond C, Goldman J, Schlossman J, Tsung J, Tan C, Pope WB, Ji MS, Nguyen NT, Lai A, Nghiemphu PL, Liau LM, Cloughesy TF, Ellingson BM. Diffusion Magnetic Resonance Imaging Phenotypes Predict Overall Survival Benefit From Bevacizumab or Surgery in Recurrent Glioblastoma With Large Tumor Burden. Neurosurgery. 2020;87:931-938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 22. | Robles-Medranda C, Del Valle R, Puga-Tejada M, Arevalo-Mora M, Cunto D, Egas-Izquierdo M, Estrada-Guevara L, Bunces-Orellana O, Moreno-Zambrano D, Alcivar-Vasquez J, Alvarado-Escobar H, Merfea RC, Barreto-Perez J, Rodriguez J, Calle-Loffredo D, Pitanga-Lukashok H, Baquerizo-Burgos J, Tabacelia D. Assessing EUS-guided radiofrequency ablation in unresectable pancreatic ductal adenocarcinoma: a single-center historic cohort study. Gastrointest Endosc. 2024;100:250-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 23. | Cha CH, Luo SD, Chiang PL, Chen WC, Tung YC, Su YY, Lin WC. Long-Term Outcomes of Radiofrequency Ablation for Treatment of Cystic Warthin Tumors versus Solid Warthin Tumors. Int J Environ Res Public Health. 2021;18:6640. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 24. | D'Onofrio M, Barbi E, Girelli R, Tinazzi Martini P, De Robertis R, Ciaravino V, Salvia R, Butturini G, Frigerio I, Milazzo T, Crosara S, Paiella S, Pederzoli P, Bassi C. Variation of tumoral marker after radiofrequency ablation of pancreatic adenocarcinoma. J Gastrointest Oncol. 2016;7:213-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 25. | Rimbaș M, Dumitru AC, Tripodi G, Larghi A. EUS-Guided Radiofrequency Ablation Therapy for Pancreatic Neoplasia. Diagnostics (Basel). 2024;14:2111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 26. | Lakhtakia S. Therapy of Pancreatic Neuroendocrine Tumors: Fine Needle Intervention including Ethanol and Radiofrequency Ablation. Clin Endosc. 2017;50:546-551. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 27. | Li F, Zhang YY, Li M, Chen SK. Microwave ablation for liver metastases from colorectal cancer: A comprehensive review of clinical efficacy and safety. World J Gastrointest Surg. 2025;17:101162. [PubMed] [DOI] [Full Text] |
| 28. | Okasha HH, Gadour E, Alyouzbaki AZ, Shaaban HE. Endoscopic ultrasound-guided radiofrequency ablation of pancreatic tumors: Current status and future perspectives. World J Methodol. 2025;15:107305. [PubMed] [DOI] [Full Text] |
| 29. | Alonso-González R, Abadal Villayandre JM, Gálvez Gonzalez E, Álvarez Perez MJ, Méndez Alonso S, de Gregorio Ariza MA. Irreversible electroporation: Beyond the limits of tumor ablation. Radiologia (Engl Ed). 2024;66:47-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 30. | Yun JH, Fang A, Khorshidi F, Habibollahi P, Kutsenko O, Etezadi V, Hunt S, Nezami N. New Developments in Image-Guided Percutaneous Irreversible Electroporation of Solid Tumors. Curr Oncol Rep. 2023;25:1213-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 31. | Rossi G, Petrone MC, Capurso G, Partelli S, Falconi M, Arcidiacono PG. Endoscopic ultrasound radiofrequency ablation of pancreatic insulinoma in elderly patients: Three case reports. World J Clin Cases. 2022;10:6514-6519. [PubMed] [DOI] [Full Text] |
| 32. | Chavan R, Thosani N, Kothari S. Role of Endoscopic Ultrasound-Guided Radiofrequency Ablation in Pancreatic Lesions: Where Are We Now and What Does the Future Hold? Cancers (Basel). 2024;16:3662. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 33. | Yin FF, Zhao LJ, Ji XY, Duan N, Wang YK, Zhou JY, Wei LH, He XJ, Wang JL, Li XP. Intra-tumor heterogeneity for endometrial cancer and its clinical significance. Chin Med J (Engl). 2019;132:1550-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |