Copyright: ©Author(s) 2026.
World J Gastrointest Oncol. Mar 15, 2026; 18(3): 115722
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115722
Published online Mar 15, 2026. doi: 10.4251/wjgo.v18.i3.115722
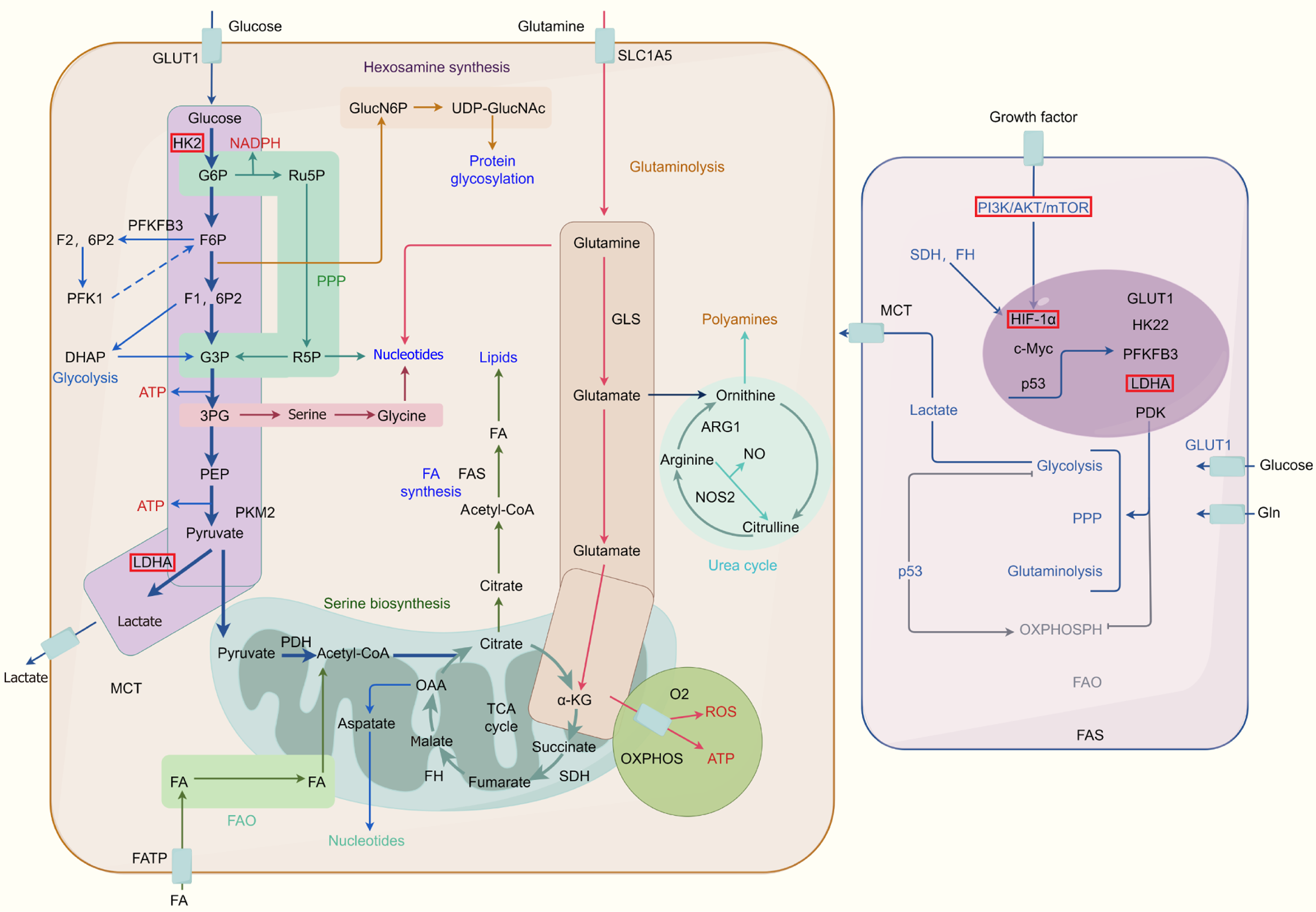
Figure 1 Metabolic reprogramming in cancer cells.
The metabolic reprogramming mechanism in cancer cells involves a series of changes in core molecules and metabolic pathways aimed at meeting their energy, nutrient, and biosynthetic material demands for rapid growth and uncontrolled proliferation. Warburg effect is a hallmark of metabolic reprogramming in cancer cells. While normal cells primarily produce energy through oxidative phosphorylation, cancer cells tend to ferment glucose into lactic acid, even in the presence of oxygen. This process allows cancer cells to generate fewer ATP but provides more biosynthetic materials needed for growth and division. Glucose metabolism: Cancer cells significantly increase their uptake and metabolism of glucose to meet their high energy demands. This includes glycolytic pathways where glucose is broken down into pyruvate and lactic acid, producing a small amount of ATP. Core molecules involved in this process include the regulation of PFK-1 and the regulation of the pentose phosphate pathway. When cancer cells are exposed to low oxygen conditions, HIF-1α protein is activated, prompting cells to use lactic acid fermentation for metabolism to enhance their survival and growth. HIF-1α activation also leads to the upregulation of glucose metabolism genes such as GLUT1 and lactate dehydrogenase. AMPK activated when energy supply is insufficient. AMPK inhibits cell growth and promotes energy production by inhibiting mTORC1, slowing down protein synthesis, and promoting glucose uptake and oxidation to restore energy balance. PI3K/Akt/mTOR pathway plays a crucial role in the metabolic reprogramming of cancer cells. Activation of Akt promotes highly enhanced glycolysis, while mTOR regulates cell growth and metabolism. p53 is a suppressive protein that regulates the cell cycle and metabolism. In some cases, the loss of p53 in cancer cells leads to metabolic reprogramming, including increased glycolysis and glucose uptake. Lipid metabolism-related core molecules include lipid synthesis enzymes such as fatty acid synthase and enzymes related to lipid breakdown. Cancer cells tend to increase lipid synthesis to meet their membrane component requirements. Cancer cells often exhibit alterations in amino acid uptake and metabolism. Core molecules include mTORC1 and GCN2, which play important roles in amino acid synthesis and metabolism. Serine metabolism-related molecules including serine synthesis enzymes and pyruvate kinase, are involved in serine metabolism, providing essential metabolic products for growth and biosynthesis. This Figure was drawn by Figdraw (Supplementary material). MCT: Monocarboxylate transporter; FA: Fatty acid; FAS: Fatty acid synthase; FATP: Fatty acid transport protein; TCA: Tricarboxylic acid; ROS: Reactive oxygen species; OXPHOS: Oxidative phosphorylation; SOD: Superoxide dismutase; SDH: Succinate dehydrogenase; FH: Fumarate hydratase; FAO: Fatty acid oxidation; OAA: Oxaloacetate.
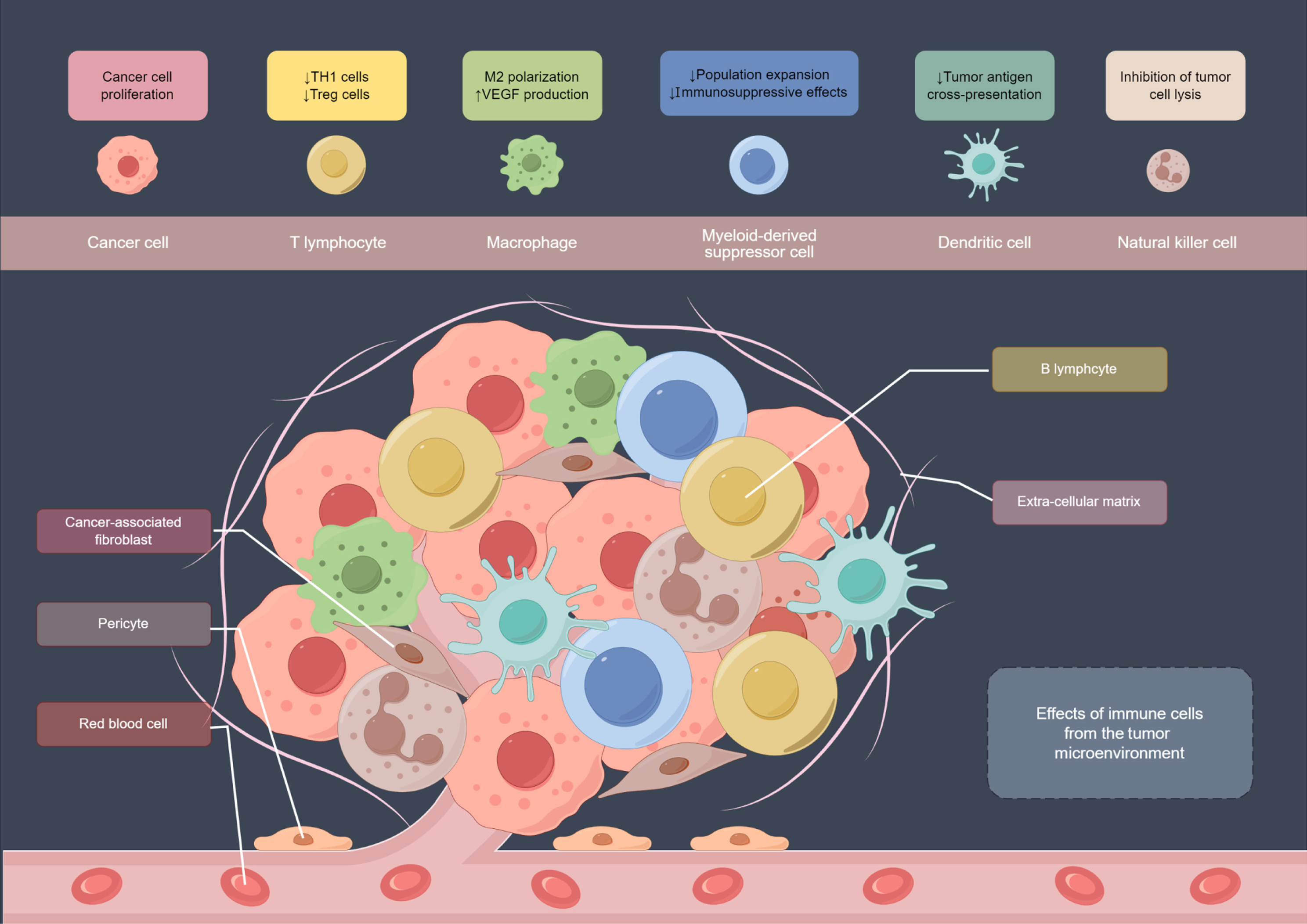
Figure 2 The effects of immune cells from the tumor microenvironment.
Different types of immune cells have various effects on the tumor microenvironment: CD4+ T cells can differentiate into different subsets, such as Th1 cells, which release interferons and activate other immune cells to enhance anti-tumor immune responses. However, Tregs common in the tumor microenvironment can suppress immune responses, contributing to immune evasion by the tumor. Macrophages have the ability to engulf tumor cells, release inflammatory factors, recruit other immune cells, clear tumor cell debris, and promote anti-tumor immune responses. However, in certain cases, tumors can activate macrophages to produce inhibitory factors, thereby suppressing their anti-tumor effects. myeloid-derived suppressor cells are a type of immunosuppressive cells that inhibit T cell activity in the tumor microenvironment, suppressing immune responses. They hinder the function of immune cells by releasing inhibitory molecules like NO and H2O2, aiding in immune evasion by the tumor. Dendritic cells are antigen-presenting cells that can recognize tumor antigens and present them to T cells, activating T cell immune responses. The presence of dendritic cells can enhance anti-tumor immune effects, but certain factors in the tumor microenvironment may inhibit dendritic cell function. Natural killer (NK) cells can rapidly recognize and kill tumor cells without the need for prior immune memory. They induce tumor cell apoptosis by releasing perforin and other cytotoxic substances. The activity of NK cells may sometimes be suppressed in the tumor microenvironment. This Figure was drawn by Figdraw (Supplementary material).
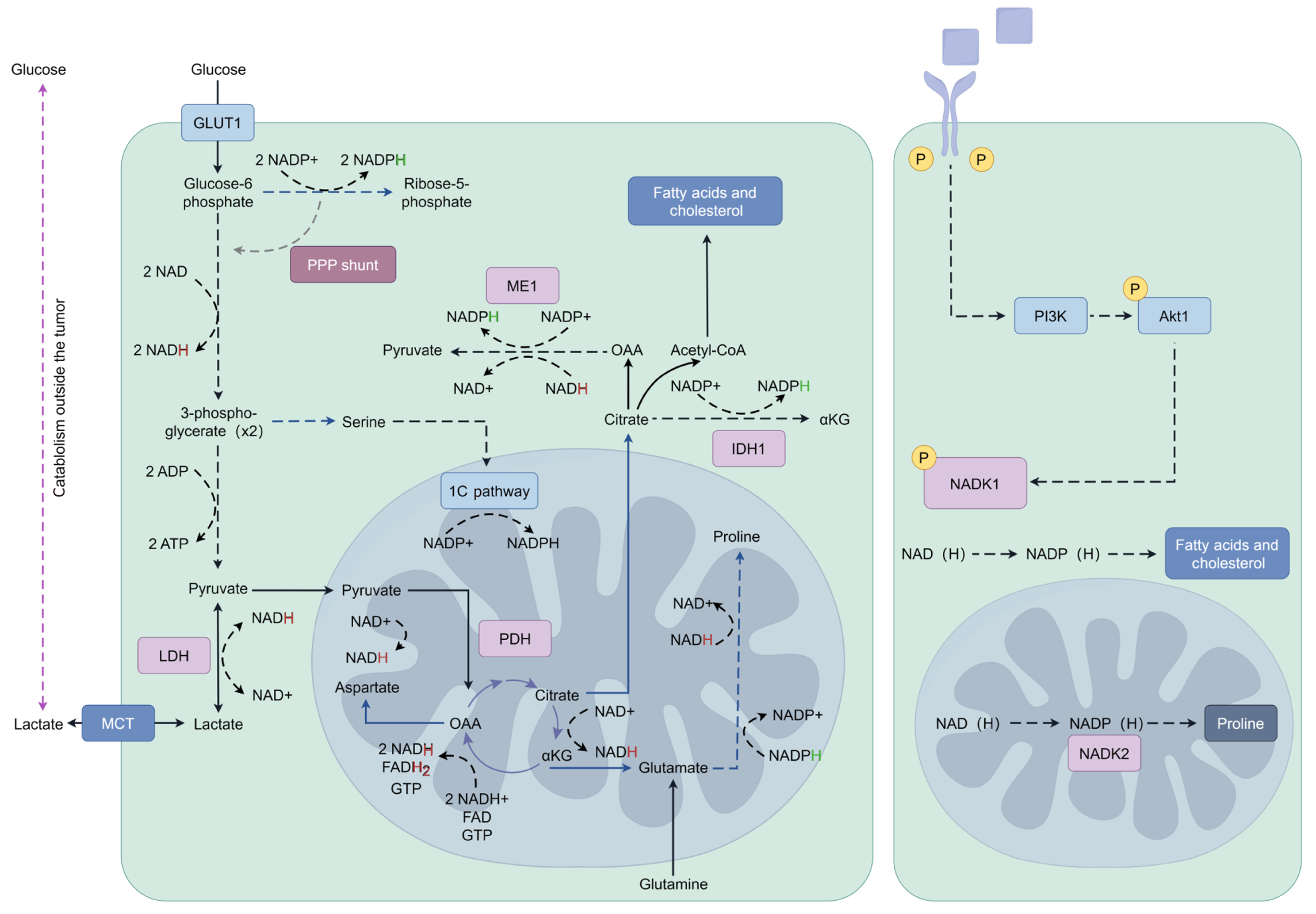
Figure 3 Mechanisms of glucose metabolism and fatty acid metabolism in tumor cell.
Glucose metabolism and fatty acid metabolism are two essential metabolic pathways in cells that provide the necessary energy and building blocks for various cellular processes. Here is a detailed description of these two metabolic processes, including the roles of key molecules: Glucose metabolism: Glycolysis is the process by which glucose is broken down into pyruvate and lactate, typically occurring in the cytoplasm. This pathway involves a series of reactions where glucose is converted into two three-carbon molecules, generating a small amount of ATP and NADH. PFK is the rate-limiting enzyme of glycolysis, and its activity increases in response to energy demands, driving glycolysis. PK promotes the conversion of pyruvate into lactate in the final step of glycolysis. Citric acid cycle (Krebs cycle) takes place in the mitochondria and involves the oxidation of pyruvate into carbon dioxide and ATP. NAD+ and FAD accept electrons during the citric acid cycle and ultimately drive ATP synthesis in the electron transport chain. The electron transport chain is located on the inner mitochondrial membrane and transfers electrons from NADH and FADH2 to oxygen, resulting in the production of a significant amount of ATP. Cytochrome c responsible for electron transfer, ultimately driving ATP synthesis. Fatty acid metabolism: Fatty acid synthesis occurs in the cytoplasm, where acetyl-CoA is used to synthesize long-chain fatty acids through a series of enzymatic reactions. Acetyl-CoA is the initial substrate for fatty acid synthesis, often derived from glucose metabolism. Fatty acid synthase is a key enzyme responsible for synthesizing fatty acids. Fatty acid oxidation occurs in the mitochondria, where long-chain fatty acids are oxidized to generate acetyl-CoA, which is used to produce ATP. This Figure was drawn by Figdraw (Supplementary material). MCT: Monocarboxylate transporter; LDH: Lactate dehydrogenase; PDH: Pyruvate dehydrogenase complex; OAA: Oxaloacetate.
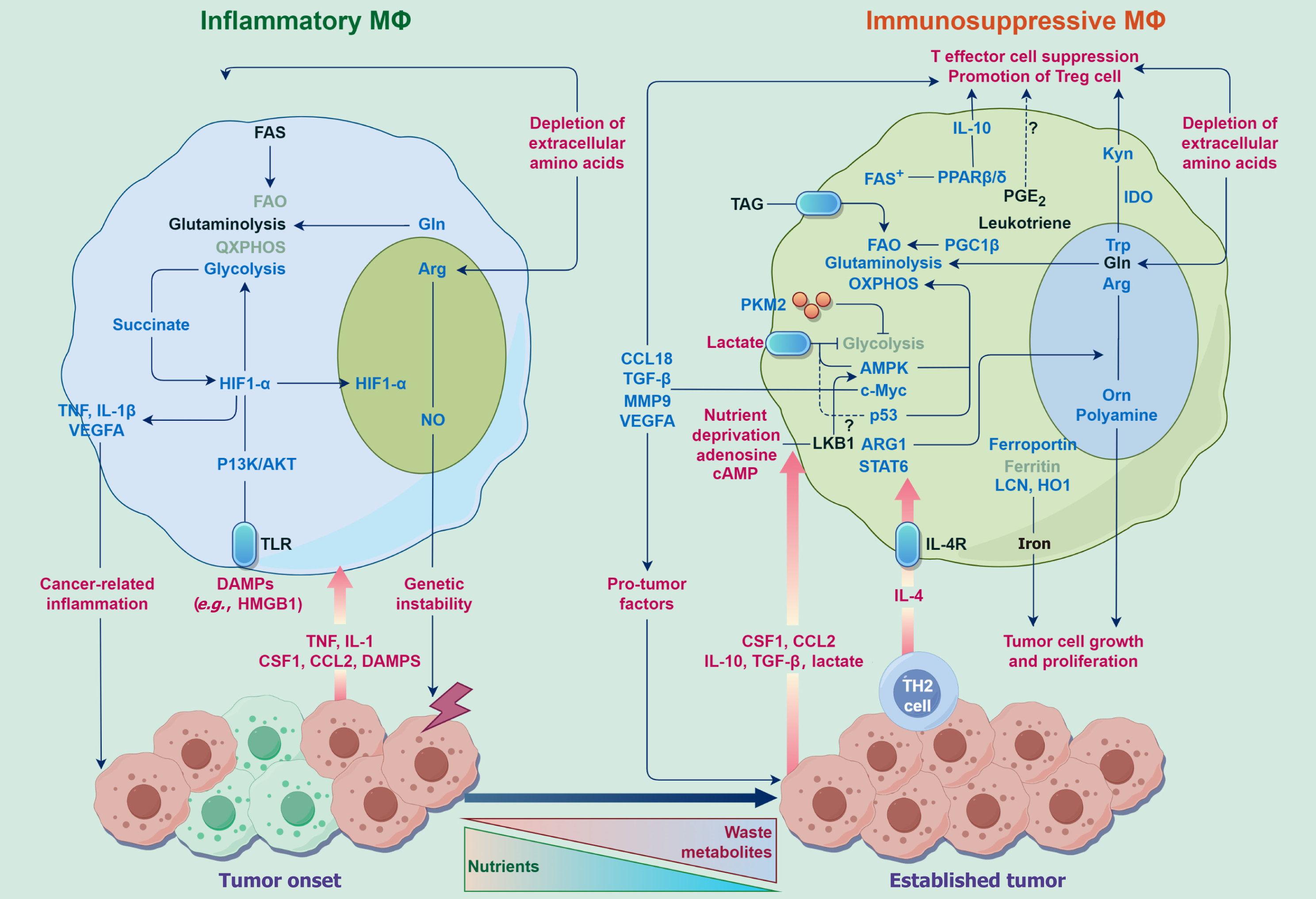
Figure 4 The roles of inflammatory macrophages and immunosuppressive macrophages in cancer.
Role of inflammatory macrophages in tumors: Inflammatory macrophages typically participate in early anti-tumor immune responses. Inflammatory macrophages can recognize and phagocytose tumor cells, eliminating potential tumor cell threats. They release inflammatory cytokines such as tumor necrosis factor alpha, interleukin (IL)-1β, and IL-12 to stimulate an inflammatory response, recruit other immune cells like T cells and natural killer cells, and assist in clearing tumor cells. Inflammatory macrophages also influence tumor development through various molecules and signaling pathways, such as NF-κB signaling pathway, iNOS, IL-12 and IL-23, which promote T cell activation and enhance the immune response. Immunosuppressive macrophages, also known as M2-type macrophages, typically play an immunosuppressive role in the tumor microenvironment, helping to maintain immune balance and reduce inflammation. Immunosuppressive macrophages release anti-inflammatory cytokines like IL-10 and transforming growth factor beta (TGF-β) to suppress the activity of immune cells, especially T cells and influence the growth and survival of tumor cells in the tumor microenvironment through various molecules and signaling pathways, such as STAT3 signaling pathway which is activated in immunosuppressive macrophages, promoting the production of anti-inflammatory factors like TGF-β. Besides, the activity of ARG1 is increased in immunosuppressive macrophages, leading to the degradation of L-arginine and the suppression of T cell activity. This Figure was drawn by Figdraw (Supplementary material). FAS: Fatty acid synthase; FAO: Fatty acid oxidation; DAMP: Damage-associated molecular pattern; IDO: Indoleamine-2,3-dioxygenase; TNF: Tumor necrosis factor; IL: Interleukin; TGF-β: Transforming growth factor-beta.
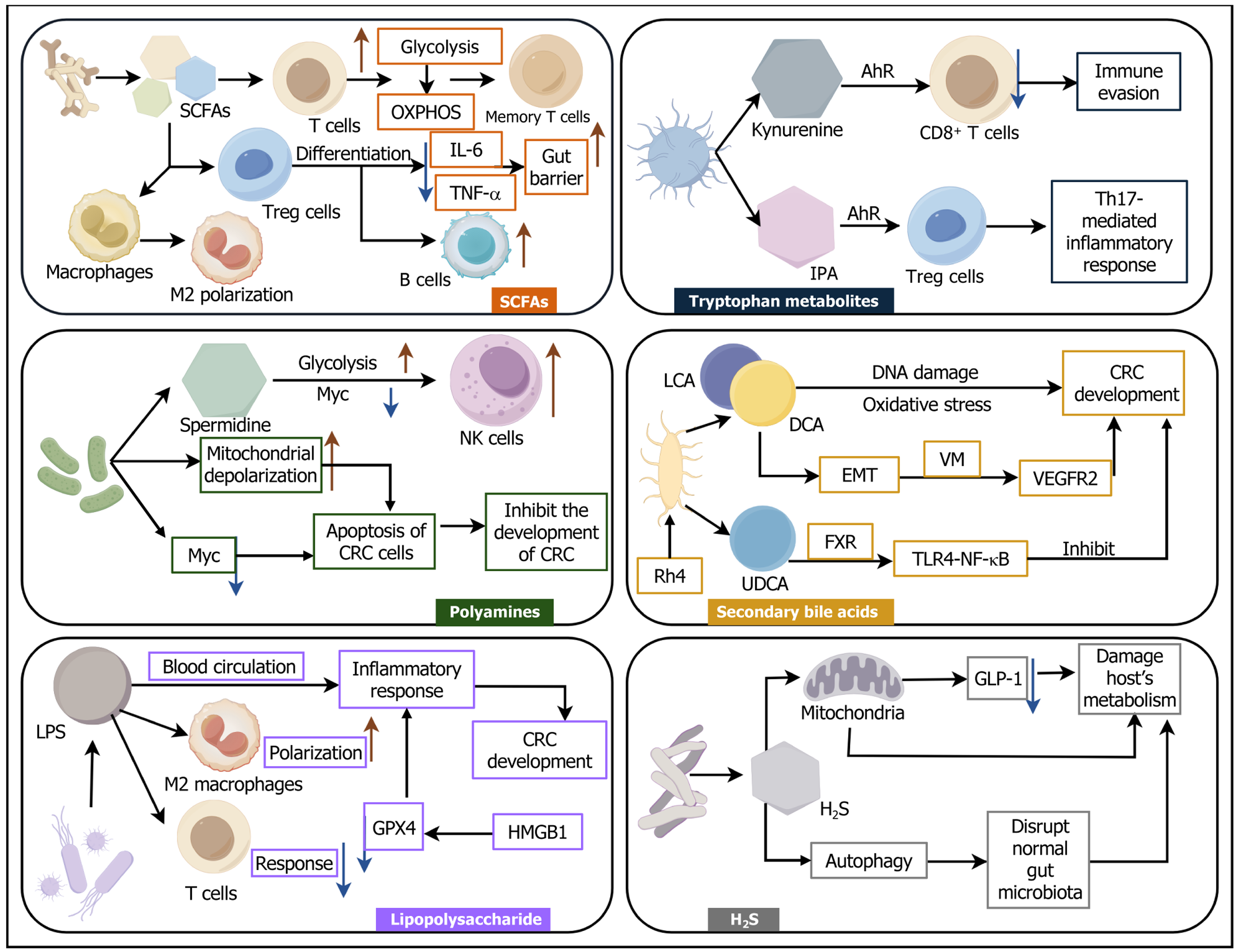
Figure 5 The impact of gut microbiota metabolites on immunometabolism and the tumor microenvironment in colorectal cancer.
This diagram systematically illustrates the impact of six major metabolites of the gut microbiota on immune cells and the tumor microenvironment. Among them, short-chain fatty acids can, on the one hand, promote the killing effect of T cells and inhibit Tregs cells, thereby suppressing the development of colorectal cancer (CRC); on the other hand, they can induce the polarization of M2 macrophages to promote the progression of CRC. Tryptophan metabolites represented by kynurenine can inhibit the activity of CD8+ T cells. Polyamine metabolites are related to the apoptosis of CRC cells, and their deficiency often indicates the formation of an immunosuppressive microenvironment. Secondary bile acids such as lithocholic acid and deoxycholic acid can induce the development of CRC by directly damaging DNA, inducing oxidative stress, and promoting epithelial-mesenchymal transition. Lipopolysaccharides promote the progression of CRC by inducing pro-inflammatory responses, while H2S affects the metabolism of normal intestinal epithelial cells by influencing mitochondrial function, thereby affecting the development of CRC. This Figure was drawn by Figdraw (Supplementary material). SCFA: Short-chain fatty acid; OXPHOS: Oxidative phosphorylation; IL: Interleukin; TNF-α: Tumor necrosis factor alpha; CRC: Colorectal cancer; LPS: Lipopolysaccharides; AhR: Aryl hydrocarbon receptor; IPA: Indolepropionic acid; LCA: Lithocholic acid; DCA: Deoxycholic acid; EMT: Epithelial-mesenchymal transition; VM: Vasculogenic mimicry; UDCA: Ursodeoxycholic acid.
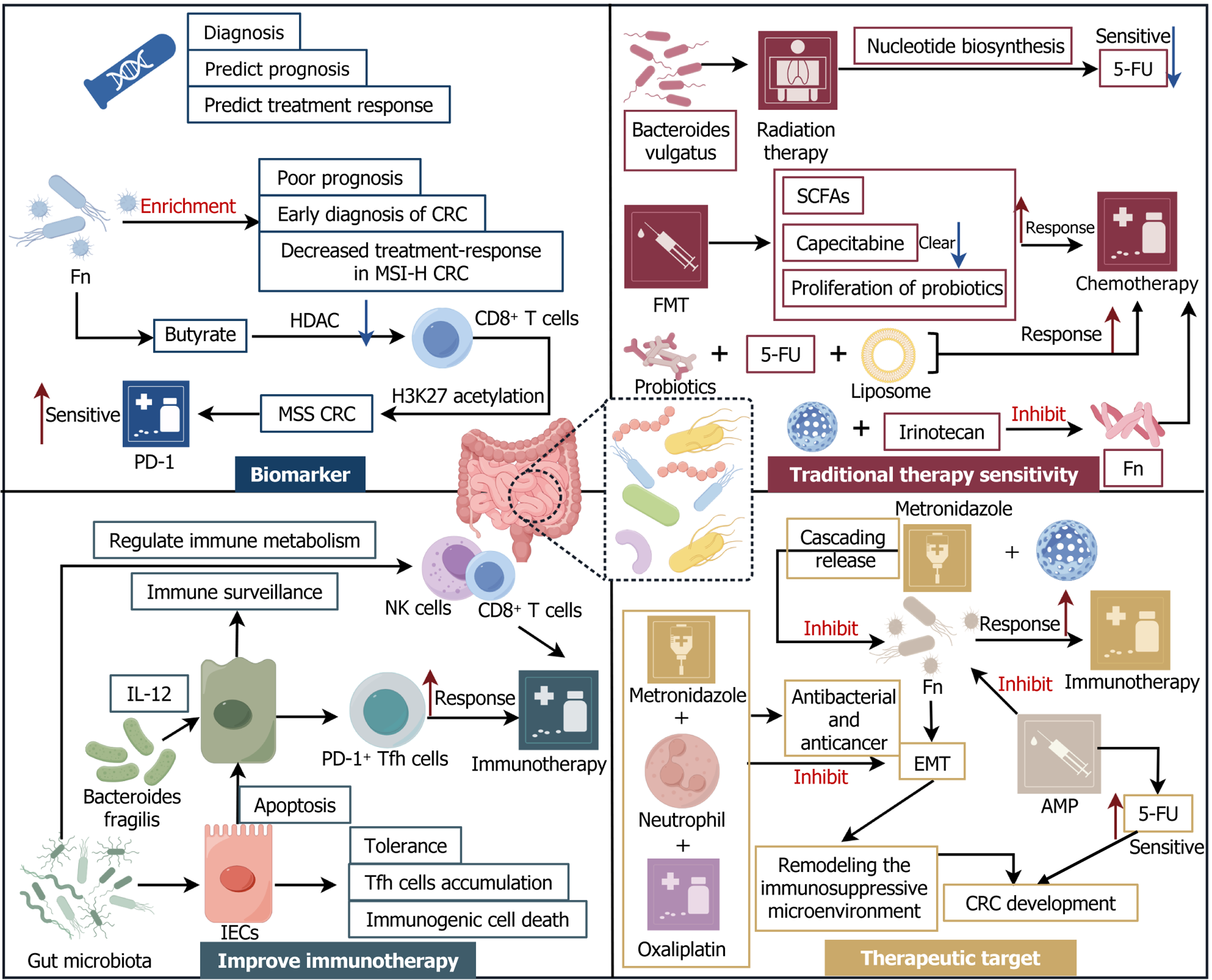
Figure 6 Clinical applications of gut microbiota in colorectal cancer.
In terms of biomarkers, the enrichment level of gut microbiota such as Fuso
- Citation: Luan WY, Zhang SP, Xu KZ, Shang YH, Hu WJ, Sun H, Miao YD. Microbiota-driven immunometabolic regulation in colorectal cancer: Mechanisms and therapeutic opportunities. World J Gastrointest Oncol 2026; 18(3): 115722
- URL: https://www.wjgnet.com/1948-5204/full/v18/i3/115722.htm
- DOI: https://dx.doi.org/10.4251/wjgo.v18.i3.115722