Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.116689
Revised: December 9, 2025
Accepted: February 5, 2026
Published online: April 27, 2026
Processing time: 154 Days and 20.9 Hours
Non-alcoholic fatty liver disease (NAFLD) represents a global clinical challenge, largely due to the liver’s central role as a key immunometabolic organ. Recent research underscores the systemic immunometabolic nature of NAFLD. It has been shown that peripheral blood immune cells of NAFLD patients exist in a primed state, which aligns with the concept of long-term functional reprogram
Core Tip: Non-alcoholic fatty liver disease is a systemic immunometabolic disorder. Chronic exposure to metabolic factors such as lipotoxicity and hyperglycemia induces long-term functional reprogramming of innate immunity, underlying phenomena like priming and trained immunity. The resulting hyperactive pro-inflammatory phenotype of monocytes and macrophages serves as a key mechanism linking obesity, insulin resistance, and chronic liver inflammation, opening new avenues for therapeutic intervention.
- Citation: Kotlyarov S. Monocyte reprogramming and trained immunity: Linking metabolism to inflammation in non-alcoholic fatty liver disease. World J Hepatol 2026; 18(4): 116689
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/116689.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.116689
Non-alcoholic fatty liver disease (NAFLD) or, in the new classification, metabolic dysfunction-associated steatotic liver disease is a global health problem affecting up to 20% of the adult population and is the leading cause of end-stage liver disease, transplantation, and hepatocellular carcinoma[1-3]. Understanding of the pathogenesis of NAFLD has improved significantly in recent years and has evolved from a simple classical hepatocentric theory to the recognition of the disease as a complex multifactorial process involving insulin resistance, oxidative stress, chronic inflammation, and other immune disorders occurring at the whole-body level[4-7]. At the same time, there is a growing understanding of the importance of immune disorders and their complex cross-links with metabolism. A recent study by Zeber-Lubecka et al[8] emphasizes the systemic immune nature of NAFLD. It has been shown that even in the absence of peripheral blood mononuclear cells stimulation, adolescents with NAFLD have a transcriptional profile indicative of systemic immune activation and functional cell reprogramming. These data contribute to the understanding that NAFLD is a systemic immunometabolic disease and once again highlight the complexity of the immune system’s interactions[9]. Thus, the aim of this mini-review is to examine the role of long-term functional reprogramming of monocytes and macrophages - key innate immune cells, as central players in NAFLD pathogenesis, linking metabolic disturbances to persistent inflammation.
The key mechanism linking metabolic disorders to chronic inflammation in NAFLD and other associated conditions (such as atherosclerosis) is considered to be dysfunction of the innate immune system[10-12]. The mammalian immune system has a variety of defense mechanisms and is believed to include two evolutionarily conserved components: Innate and adaptive immunity. The innate immune system relies on phylogenetically ancient mechanisms and is inherent in all multicellular organisms, including humans. Innate immunity is characterized by a rapid and nonspecific response to pathogens, including biochemical and cellular reactions[13]. Evidence indicates that impaired mechanisms of the innate immune system, in particular a disproportionate pro-inflammatory response, underlie many chronic non-infectious diseases in humans, such as atherosclerosis[14-16].
It is believed that invertebrates have only an innate immune system and do not have an adaptive immune system, which, however, has not prevented them from being a very numerous group of animals that have successfully colonized various ecological niches. Thus, only one innate immune system can provide them with the necessary protection against pathogens. For a long time, it was believed that the innate immune system has no immunological memory and is unable to respond differentially to pathogens, while the adaptive immune system retains memory of the pathogen and can act more selectively. Data obtained in recent years have made it possible to revise some of these principles of immunology, as it has been shown that the innate immune system is also capable of retaining some immunological memory, which has been termed “trained immunity” and has been convincingly demonstrated in monocytes, macrophages, and natural killer cells, as well as in numerous experiments on invertebrates[17-19]. It is important to note that the functional repro
It has been established that trained immunity is based on epigenetic mechanisms and the switching of cellular meta
Metabolic reprogramming in trained immunity corresponds to the broader concept of immunometabolism, which involves switching metabolic pathways in immune cells between different states that provide the cell with energy or substrates for biochemical processes necessary for the immune response[25]. Thus, macrophages can demonstrate different roles in inflammation or even switch from one role to another by changing their cellular metabolism. The classic model of immunometabolism demonstrated in mice includes two key macrophage phenotypes: M1 and M2 polarization (it is important to note that this classification is greatly simplified compared to the actual, more complex picture of macrophage polarization, but this model is useful for understanding the role of metabolism in immune cell function). M1 polarization of macrophages is pro-inflammatory. In these cells, glycolysis is activated (similar to the Warburg effect in highly proliferative tumor cells) and oxidative phosphorylation activity is reduced. M2 macrophages are involved in resolving inflammation and rely heavily on oxidative phosphorylation as a key source of energy[36-38]. The disrupted tricarboxylic acid cycle in M1 phenotype macrophages contributes to the accumulation of succinate, which stabilizes hypoxia-inducible factor 1-alpha, leading to increased interleukin 1 beta production via NLR-family pyrin domain-containing protein 3 inflammasome. An anti-inflammatory mechanism is also induced through the synthesis of itaconate, which inhibits glycolysis and suppresses the production of pro-inflammatory cytokines[39-41]. Itaconate is considered one of the key metabolites involved in the formation of innate immune tolerance and exerting an indirect effect on the adaptive immune response[42].
Thus, the concept of trained immunity suggests that monocytes circulating in the bloodstream can come into contact with pathogens or danger signals, such as lipopolysaccharides of Gram-negative bacteria or oxidized low-density lipo
Evidence suggests that systemic metabolic dysregulation in NAFLD reprograms innate immune cells[48]. Monocytes circulating in an environment rich in free fatty acids and glucose can switch their metabolism from oxidative phos
The gut microbiota plays an important role in regulating immunological memory, which can be disrupted by obesity and NAFLD. For example, it has been shown that microbial dysbiosis in the neonatal period also contributes to the training of the sensitivity of innate immune cells in the intestine, bone marrow, and liver, which leads to the pro
In the context of NAFLD, functional reprogramming of innate immune cells can play a negative role, but at different stages of disease development. On the one hand, priming and trained immunity exacerbate systemic and hepatic inflammation, contributing to the progression of steatohepatitis and fibrosis. On the other hand, the formation of tolerance in resident liver macrophages (Kupffer cells) may be an adaptation to the chronic, low-intensity influx of bacterial products from the intestine in dysbiosis. At the same time, “tolerant” macrophages lose their ability to effectively phagocytose and clear bacteria. As a result, subsequent damage to hepatocytes due to lipotoxicity or acute infectious exposure can lead to a loss of tolerance and uncontrolled release of accumulated pro-inflammatory mediators, which contribute to the progression of the disease[57,58].
Thus, the innate immune system, which relies on evolutionarily conserved mechanisms, has a long-term and ambiguous effect on the pathogenesis of chronic non-infectious diseases. In this context, the immunometabolic reprogramming of monocytes and macrophages can be considered a key link in the pathogenesis of NAFLD. The concepts of trained immunity and immunometabolic reprogramming expand the understanding of NAFLD development mechanisms, shifting the focus from hepatocentric models to a systemic view of the disease, where immune cells act as integrators and long-term carriers of metabolic damage.
However, when interpreting these data clinically, it is crucial to consider the existing limitations. A significant portion of the data linking trained immunity to NAFLD has been obtained from experimental animal models or in vitro studies, and their direct translation to humans requires caution. Secondly, many observations are correlational, and establishing causal relationships necessitates further longitudinal and interventional studies. Thirdly, there is considerable inter-individual variability in the formation of immunological memory, driven by genetic factors, metabolic profile, and lifestyle characteristics. Finally, the pathogenic pathways of NAFLD are closely intertwined with those of atherosclerosis, insulin resistance, and obesity, complicating the isolation of liver-specific immunometabolic disturbances.
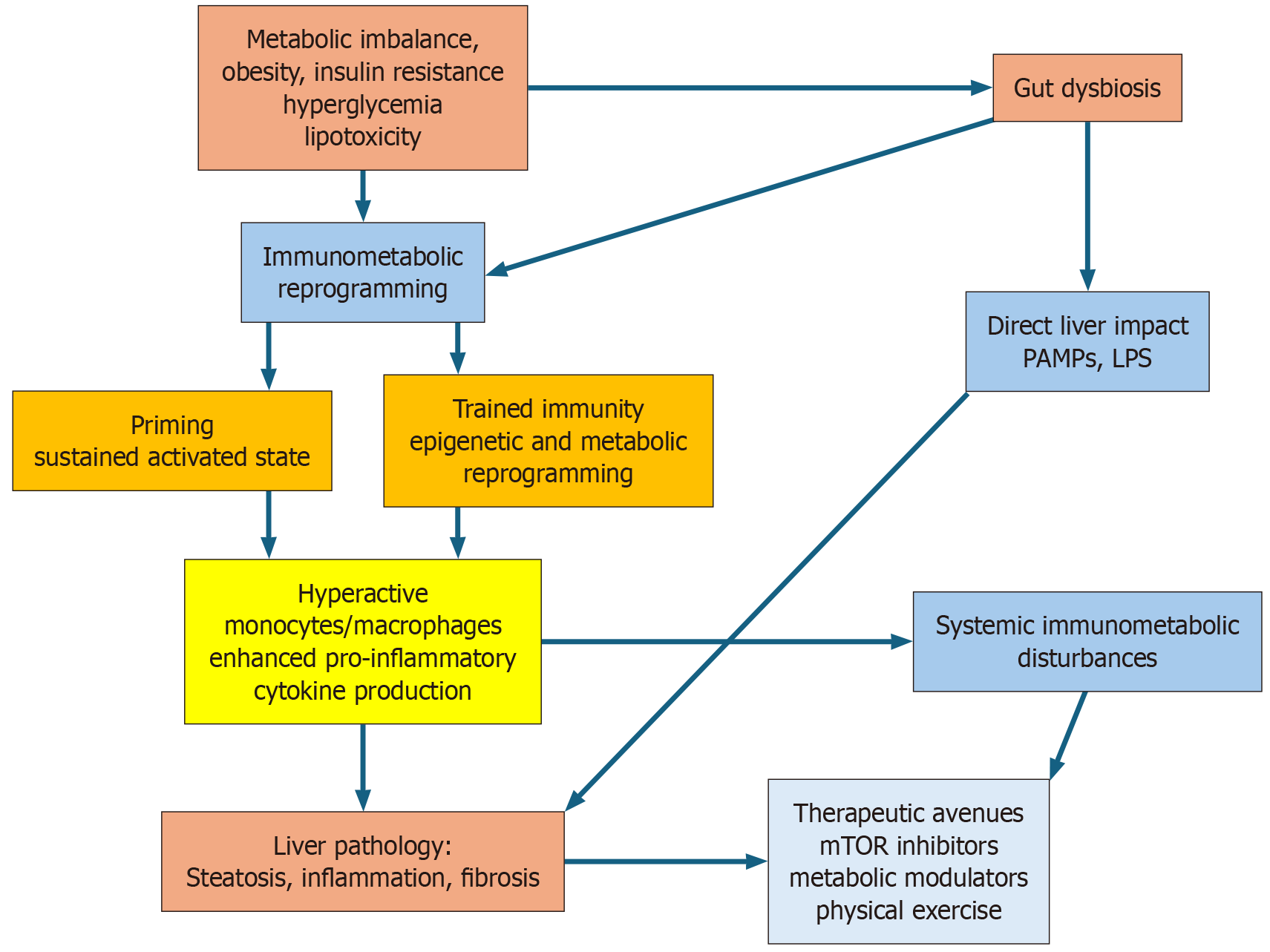
Despite these limitations, the concept of immunometabolic reprogramming opens new clinical possibilities (Figure 1). Promising directions for future research include the search for biomarkers reflecting the state of immune cell repro
Thus, trained immunity, which evolved as a mechanism to enhance the body’s defense against reinfection by pathogens or tissue damage, may be one of the mechanisms of immunometabolic diseases. The role of the innate immune system in general and trained immunity in particular in the pathogenesis of NAFLD is a promising topic for future research and will improve our understanding of the complex links between NAFLD and other diseases, thereby increasing the effectiveness of diagnosis and treatment. In this regard, the concept of trained immunity represents a paradigm shift in understanding the pathogenesis of NAFLD. It shifts the focus from a purely hepatocentric model to a systemic immunometabolic approach, in which innate immune cells, such as monocytes and macrophages, act as long-term integrators of metabolic dysfunction and inflammatory priming. Chronic functional reprogramming of these cells, based on epigenetic and metabolic changes, creates a self-sustaining cycle of inflammation that fuels the progression of the disease from steatosis to steatohepatitis and fibrosis. This new paradigm positions trained immunity as a novel therapeutic target, which is a promising avenue for future research.
| 1. | European Association for the Study of the Liver; European Association for the Study of Diabetes; European Association for the Study of Obesity. EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD): Executive Summary. Diabetologia. 2024;67:2375-2392. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 103] [Article Influence: 51.5] [Reference Citation Analysis (2)] |
| 2. | Ambartsumyan AR, Pyatibrat ED, Deryagina LE, Batskov SS, Pyatibrat AO. Features of the course of liver fatty degeneration in cholecystectomy and in non-operated patients of different age groupss. I P Pavlov Russ Med Biol Herald. 2025;33:23-35. [DOI] [Full Text] |
| 3. | Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31:S32-S50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 511] [Cited by in RCA: 497] [Article Influence: 497.0] [Reference Citation Analysis (0)] |
| 4. | Badmus OO, Hillhouse SA, Anderson CD, Hinds TD, Stec DE. Molecular mechanisms of metabolic associated fatty liver disease (MAFLD): functional analysis of lipid metabolism pathways. Clin Sci (Lond). 2022;136:1347-1366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 318] [Cited by in RCA: 268] [Article Influence: 67.0] [Reference Citation Analysis (0)] |
| 5. | Day CP, James OF. Steatohepatitis: a tale of two "hits"? Gastroenterology. 1998;114:842-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3398] [Cited by in RCA: 3117] [Article Influence: 111.3] [Reference Citation Analysis (6)] |
| 6. | Tilg H, Moschen AR. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology. 2010;52:1836-1846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2088] [Cited by in RCA: 1943] [Article Influence: 121.4] [Reference Citation Analysis (4)] |
| 7. | Buzzetti E, Pinzani M, Tsochatzis EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016;65:1038-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2614] [Cited by in RCA: 2362] [Article Influence: 236.2] [Reference Citation Analysis (15)] |
| 8. | Zeber-Lubecka N, Michalkiewicz J, Dabrowska M, Goryca K, Wierzbicka-Rucińska A, Jańczyk W, Jankowska I, Świąder-Leśniak A, Kubiszewska I, Ziemska-Legięcka J, Socha P, Ostrowski J. Transcriptome profiles of peripheral blood mononuclear cells differentiate male adolescents with non-alcoholic fatty liver disease from healthy peers. World J Hepatol. 2025;17:113359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Guo Z, Wu Q, Xie P, Wang J, Lv W. Immunomodulation in non-alcoholic fatty liver disease: exploring mechanisms and applications. Front Immunol. 2024;15:1336493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 10. | He Y, Chen Y, Qian S, van Der Merwe S, Dhar D, Brenner DA, Tacke F. Immunopathogenic mechanisms and immunoregulatory therapies in MASLD. Cell Mol Immunol. 2025;22:1159-1177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 40.0] [Reference Citation Analysis (1)] |
| 11. | Aronova A, Tosato F, Naser N, Asare Y. Innate Immune Pathways in Atherosclerosis-From Signaling to Long-Term Epigenetic Reprogramming. Cells. 2023;12:2359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 12. | Shalhoub J, Falck-Hansen MA, Davies AH, Monaco C. Innate immunity and monocyte-macrophage activation in atherosclerosis. J Inflamm (Lond). 2011;8:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 99] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 13. | Buchmann K. Evolution of Innate Immunity: Clues from Invertebrates via Fish to Mammals. Front Immunol. 2014;5:459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 309] [Cited by in RCA: 394] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 14. | Roy P, Orecchioni M, Ley K. How the immune system shapes atherosclerosis: roles of innate and adaptive immunity. Nat Rev Immunol. 2022;22:251-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 399] [Article Influence: 99.8] [Reference Citation Analysis (0)] |
| 15. | Zhong C, Yang X, Feng Y, Yu J. Trained Immunity: An Underlying Driver of Inflammatory Atherosclerosis. Front Immunol. 2020;11:284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 16. | Bäck M, Yurdagul A Jr, Tabas I, Öörni K, Kovanen PT. Inflammation and its resolution in atherosclerosis: mediators and therapeutic opportunities. Nat Rev Cardiol. 2019;16:389-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 748] [Article Influence: 124.7] [Reference Citation Analysis (0)] |
| 17. | Netea MG, Quintin J, van der Meer JW. Trained immunity: a memory for innate host defense. Cell Host Microbe. 2011;9:355-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1421] [Cited by in RCA: 1212] [Article Influence: 80.8] [Reference Citation Analysis (0)] |
| 18. | Kurtz J. Specific memory within innate immune systems. Trends Immunol. 2005;26:186-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 261] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 19. | Kloc M, Halasa M, Kubiak JZ, Ghobrial RM. Invertebrate Immunity, Natural Transplantation Immunity, Somatic and Germ Cell Parasitism, and Transposon Defense. Int J Mol Sci. 2024;25:1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 20. | Kim HY, Lee WW. Trained immunity induced by DAMPs and LAMPs in chronic inflammatory diseases. Exp Mol Med. 2025;57:2137-2147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 21. | Lajqi T, Köstlin-Gille N, Bauer R, Zarogiannis SG, Lajqi E, Ajeti V, Dietz S, Kranig SA, Rühle J, Demaj A, Hebel J, Bartosova M, Frommhold D, Hudalla H, Gille C. Training vs. Tolerance: The Yin/Yang of the Innate Immune System. Biomedicines. 2023;11:766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 43] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 22. | Ifrim DC, Quintin J, Joosten LA, Jacobs C, Jansen T, Jacobs L, Gow NA, Williams DL, van der Meer JW, Netea MG. Trained immunity or tolerance: opposing functional programs induced in human monocytes after engagement of various pattern recognition receptors. Clin Vaccine Immunol. 2014;21:534-545. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 264] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 23. | Wiesenthal AA, Legroux TM, Richter C, Junker BH, Hecksteden A, Kessler SM, Hoppstädter J, Kiemer AK. Endotoxin Tolerance Acquisition and Altered Hepatic Fatty Acid Profile in Aged Mice. Biology (Basel). 2023;12:530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 24. | Li J, Wang H, Xia S. Hematopoietic stem and progenitor cells fine-tuning the "sweet" of trained immunity. J Leukoc Biol. 2025;117:qiaf043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 25. | Zhao B, Wan J, Zhou H, Yang J, Wan H. Trained immunity in atherosclerosis: plasticity, metabolic-vascular axis, and AI-driven precision remodeling. Front Immunol. 2025;16:1669796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 26. | Netea MG, Joosten LA, Latz E, Mills KH, Natoli G, Stunnenberg HG, O'Neill LA, Xavier RJ. Trained immunity: A program of innate immune memory in health and disease. Science. 2016;352:aaf1098. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1360] [Cited by in RCA: 1965] [Article Influence: 196.5] [Reference Citation Analysis (0)] |
| 27. | Netea MG, Domínguez-Andrés J, Barreiro LB, Chavakis T, Divangahi M, Fuchs E, Joosten LAB, van der Meer JWM, Mhlanga MM, Mulder WJM, Riksen NP, Schlitzer A, Schultze JL, Stabell Benn C, Sun JC, Xavier RJ, Latz E. Defining trained immunity and its role in health and disease. Nat Rev Immunol. 2020;20:375-388. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2281] [Cited by in RCA: 1864] [Article Influence: 310.7] [Reference Citation Analysis (21)] |
| 28. | Gill PS, Ozment TR, Lewis NH, Sherwood ER, Williams DL. Trained Immunity Enhances Human Monocyte Function in Aging and Sepsis. Front Immunol. 2022;13:872652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 29. | Quintin J, Saeed S, Martens JHA, Giamarellos-Bourboulis EJ, Ifrim DC, Logie C, Jacobs L, Jansen T, Kullberg BJ, Wijmenga C, Joosten LAB, Xavier RJ, van der Meer JWM, Stunnenberg HG, Netea MG. Candida albicans infection affords protection against reinfection via functional reprogramming of monocytes. Cell Host Microbe. 2012;12:223-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 697] [Cited by in RCA: 986] [Article Influence: 75.8] [Reference Citation Analysis (0)] |
| 30. | Bekkering S, Arts RJW, Novakovic B, Kourtzelis I, van der Heijden CDCC, Li Y, Popa CD, Ter Horst R, van Tuijl J, Netea-Maier RT, van de Veerdonk FL, Chavakis T, Joosten LAB, van der Meer JWM, Stunnenberg H, Riksen NP, Netea MG. Metabolic Induction of Trained Immunity through the Mevalonate Pathway. Cell. 2018;172:135-146.e9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 574] [Article Influence: 71.8] [Reference Citation Analysis (3)] |
| 31. | Keating ST, Groh L, Thiem K, Bekkering S, Li Y, Matzaraki V, van der Heijden CDCC, van Puffelen JH, Lachmandas E, Jansen T, Oosting M, de Bree LCJ, Koeken VACM, Moorlag SJCFM, Mourits VP, van Diepen J, Strienstra R, Novakovic B, Stunnenberg HG, van Crevel R, Joosten LAB, Netea MG, Riksen NP. Rewiring of glucose metabolism defines trained immunity induced by oxidized low-density lipoprotein. J Mol Med (Berl). 2020;98:819-831. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 99] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 32. | Cheng SC, Quintin J, Cramer RA, Shepardson KM, Saeed S, Kumar V, Giamarellos-Bourboulis EJ, Martens JH, Rao NA, Aghajanirefah A, Manjeri GR, Li Y, Ifrim DC, Arts RJ, van der Veer BM, Deen PM, Logie C, O'Neill LA, Willems P, van de Veerdonk FL, van der Meer JW, Ng A, Joosten LA, Wijmenga C, Stunnenberg HG, Xavier RJ, Netea MG. mTOR- and HIF-1α-mediated aerobic glycolysis as metabolic basis for trained immunity. Science. 2014;345:1250684. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1920] [Cited by in RCA: 1758] [Article Influence: 146.5] [Reference Citation Analysis (0)] |
| 33. | Arts RJW, Joosten LAB, Netea MG. The Potential Role of Trained Immunity in Autoimmune and Autoinflammatory Disorders. Front Immunol. 2018;9:298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 143] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 34. | Arts RJW, Carvalho A, La Rocca C, Palma C, Rodrigues F, Silvestre R, Kleinnijenhuis J, Lachmandas E, Gonçalves LG, Belinha A, Cunha C, Oosting M, Joosten LAB, Matarese G, van Crevel R, Netea MG. Immunometabolic Pathways in BCG-Induced Trained Immunity. Cell Rep. 2016;17:2562-2571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 522] [Article Influence: 58.0] [Reference Citation Analysis (11)] |
| 35. | Funes SC, Rios M, Fernández-Fierro A, Di Genaro MS, Kalergis AM. Trained Immunity Contribution to Autoimmune and Inflammatory Disorders. Front Immunol. 2022;13:868343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 36. | Gauthier T, Chen W. Modulation of Macrophage Immunometabolism: A New Approach to Fight Infections. Front Immunol. 2022;13:780839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 107] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 37. | Chen S, Saeed AFUH, Liu Q, Jiang Q, Xu H, Xiao GG, Rao L, Duo Y. Macrophages in immunoregulation and therapeutics. Signal Transduct Target Ther. 2023;8:207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1715] [Cited by in RCA: 1468] [Article Influence: 489.3] [Reference Citation Analysis (0)] |
| 38. | Wang L, Wang D, Zhang T, Ma Y, Tong X, Fan H. The role of immunometabolism in macrophage polarization and its impact on acute lung injury/acute respiratory distress syndrome. Front Immunol. 2023;14:1117548. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 39. | Michelucci A, Cordes T, Ghelfi J, Pailot A, Reiling N, Goldmann O, Binz T, Wegner A, Tallam A, Rausell A, Buttini M, Linster CL, Medina E, Balling R, Hiller K. Immune-responsive gene 1 protein links metabolism to immunity by catalyzing itaconic acid production. Proc Natl Acad Sci U S A. 2013;110:7820-7825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 572] [Cited by in RCA: 965] [Article Influence: 74.2] [Reference Citation Analysis (0)] |
| 40. | Lampropoulou V, Sergushichev A, Bambouskova M, Nair S, Vincent EE, Loginicheva E, Cervantes-Barragan L, Ma X, Huang SC, Griss T, Weinheimer CJ, Khader S, Randolph GJ, Pearce EJ, Jones RG, Diwan A, Diamond MS, Artyomov MN. Itaconate Links Inhibition of Succinate Dehydrogenase with Macrophage Metabolic Remodeling and Regulation of Inflammation. Cell Metab. 2016;24:158-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1341] [Cited by in RCA: 1226] [Article Influence: 122.6] [Reference Citation Analysis (0)] |
| 41. | Tannahill GM, Curtis AM, Adamik J, Palsson-McDermott EM, McGettrick AF, Goel G, Frezza C, Bernard NJ, Kelly B, Foley NH, Zheng L, Gardet A, Tong Z, Jany SS, Corr SC, Haneklaus M, Caffrey BE, Pierce K, Walmsley S, Beasley FC, Cummins E, Nizet V, Whyte M, Taylor CT, Lin H, Masters SL, Gottlieb E, Kelly VP, Clish C, Auron PE, Xavier RJ, O'Neill LA. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature. 2013;496:238-242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3601] [Cited by in RCA: 3326] [Article Influence: 255.8] [Reference Citation Analysis (4)] |
| 42. | Domínguez-Andrés J, Novakovic B, Li Y, Scicluna BP, Gresnigt MS, Arts RJW, Oosting M, Moorlag SJCFM, Groh LA, Zwaag J, Koch RM, Ter Horst R, Joosten LAB, Wijmenga C, Michelucci A, van der Poll T, Kox M, Pickkers P, Kumar V, Stunnenberg H, Netea MG. The Itaconate Pathway Is a Central Regulatory Node Linking Innate Immune Tolerance and Trained Immunity. Cell Metab. 2019;29:211-220.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 292] [Article Influence: 41.7] [Reference Citation Analysis (2)] |
| 43. | Groh L, Keating ST, Joosten LAB, Netea MG, Riksen NP. Monocyte and macrophage immunometabolism in atherosclerosis. Semin Immunopathol. 2018;40:203-214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 189] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 44. | Flores-Gomez D, Bekkering S, Netea MG, Riksen NP. Trained Immunity in Atherosclerotic Cardiovascular Disease. Arterioscler Thromb Vasc Biol. 2021;41:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 45. | Ochando J, Mulder WJM, Madsen JC, Netea MG, Duivenvoorden R. Trained immunity - basic concepts and contributions to immunopathology. Nat Rev Nephrol. 2023;19:23-37. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 312] [Reference Citation Analysis (1)] |
| 46. | Leentjens J, Bekkering S, Joosten LAB, Netea MG, Burgner DP, Riksen NP. Trained Innate Immunity as a Novel Mechanism Linking Infection and the Development of Atherosclerosis. Circ Res. 2018;122:664-669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 115] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 47. | Bekkering S, Quintin J, Joosten LA, van der Meer JW, Netea MG, Riksen NP. Oxidized low-density lipoprotein induces long-term proinflammatory cytokine production and foam cell formation via epigenetic reprogramming of monocytes. Arterioscler Thromb Vasc Biol. 2014;34:1731-1738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 617] [Cited by in RCA: 567] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 48. | Kountouras J, Kazakos E, Polyzos SA, Papaefthymiou A, Zavos C, Tzitiridou-Chatzopoulou M, Chatzopoulos D, Vardaka E, Gatopoulou A, Kyrailidi F, Mouratidou MC, Doulberis M. Potential impact of trained innate immunity on the pathophysiology of metabolic dysfunction-associated fatty liver disease. Clin Immunol. 2023;256:109776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 49. | Li P, He K, Li J, Liu Z, Gong J. The role of Kupffer cells in hepatic diseases. Mol Immunol. 2017;85:222-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 182] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 50. | Jin G, Yao X, Liu D, Zhang J, Zhang X, Yang Y, Bi Y, Zhang H, Dong G, Tang H, Cheng S, Hong F, Si M. Inducible nitric oxide synthase accelerates nonalcoholic fatty liver disease progression by regulating macrophage autophagy. Immun Inflamm Dis. 2023;11:e1114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 51. | Luo W, Xu Q, Wang Q, Wu H, Hua J. Effect of modulation of PPAR-γ activity on Kupffer cells M1/M2 polarization in the development of non-alcoholic fatty liver disease. Sci Rep. 2017;7:44612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 244] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 52. | Moayedfard Z, Sani F, Alizadeh A, Bagheri Lankarani K, Zarei M, Azarpira N. The role of the immune system in the pathogenesis of NAFLD and potential therapeutic impacts of mesenchymal stem cell-derived extracellular vesicles. Stem Cell Res Ther. 2022;13:242. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 53. | Xu GX, Wei S, Yu C, Zhao SQ, Yang WJ, Feng YH, Pan C, Yang KX, Ma Y. Activation of Kupffer cells in NAFLD and NASH: mechanisms and therapeutic interventions. Front Cell Dev Biol. 2023;11:1199519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 54. | Jonscher KR, Abrams J, Friedman JE. Maternal Diet Alters Trained Immunity in the Pathogenesis of Pediatric NAFLD. J Cell Immunol. 2020;2:315-325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 55. | Lasaviciute G, Barz M, van der Heiden M, Arasa C, Tariq K, Quin J, Östlund Farrants AK, Sverremark-Ekström E. Gut commensal Limosilactobacillus reuteri induces atypical memory-like phenotype in human dendritic cells in vitro. Gut Microbes. 2022;14:2045046. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 56. | Lajqi T, Köstlin-Gille N, Hillmer S, Braun M, Kranig SA, Dietz S, Krause C, Rühle J, Frommhold D, Pöschl J, Gille C, Hudalla H. Gut Microbiota-Derived Small Extracellular Vesicles Endorse Memory-like Inflammatory Responses in Murine Neutrophils. Biomedicines. 2022;10:442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 57. | Dembek A, Laggai S, Kessler SM, Czepukojc B, Simon Y, Kiemer AK, Hoppstädter J. Hepatic interleukin-6 production is maintained during endotoxin tolerance and facilitates lipid accumulation. Immunobiology. 2017;222:786-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 58. | Orci LA, Kreutzfeldt M, Goossens N, Rubbia-Brandt L, Slits F, Hammad K, Delaune V, Oldani G, Negro F, Clément S, Gonelle-Gispert C, Buhler LH, Toso C, Lacotte S. Tolerogenic properties of liver macrophages in non-alcoholic steatohepatitis. Liver Int. 2020;40:609-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 59. | Chen J, Wang C, Chen S, Cai H, Wang M, Chang J, Cai X, Yang J, Cao P. Trained immunity modulators: A new frontier in immunoregulation and disease intervention. J Adv Res. 2025;S2090-1232(25)00734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 60. | Zhang H, Chen T, Ren J, Xia Y, Onuma A, Wang Y, He J, Wu J, Wang H, Hamad A, Shen C, Zhang J, Asara JM, Behbehani GK, Wen H, Deng M, Tsung A, Huang H. Pre-operative exercise therapy triggers anti-inflammatory trained immunity of Kupffer cells through metabolic reprogramming. Nat Metab. 2021;3:843-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 78] [Article Influence: 15.6] [Reference Citation Analysis (0)] |