Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.116153
Revised: December 8, 2025
Accepted: February 6, 2026
Published online: April 27, 2026
Processing time: 168 Days and 19.2 Hours
The role of fecal microbiota transplantation (FMT) in the treatment of minimal hepatic encephalopathy (MHE) is unclear.
To compare the efficacy and safety of FMT with oral lactulose in the treatment of MHE in cirrhosis.
In this randomized-controlled trial, 130 patients with cirrhosis and MHE were randomized to receive either 2 endoscopic infusions of donor stool one-month apart or daily oral lactulose for 3 months. The primary outcome was MHE resolution at 3 months (assessed by psychometric hepatic encephalopathy score > -5). We performed microbiome analysis using next-generation sequencing and evaluated 16 antimicrobial-resistance gene profiles using gene-specific oligonucleotides.
Among 122/130 (93.8%) patients eligible for analysis [FMT: 60; lactulose: 62; age: 44.3 ± 8.9 years; 96.7% male; child A/B/C: 32.1%/53.3%/14.6%; median model for end-stage liver disease: 13 (10-17)], rates of MHE resolution were similar in both groups [41/60 (68.3%) vs 41/62 (66.1%); absolute risk-difference: 2.2% (95% confidence interval: -14.4% to 18.9%); P = 0.80] at 3 months. Both groups exhibited similar improvements in psychometric hepatic encephalopathy score (intergroup difference: -0.12 ± 0.31; P = 0.71) and 36-item short form survey scores over this time with similar risk of progression to overt encephalopathy [2 (3.3%) vs 1 (1.6%); P = 1.0], serious adverse events, and mortality. At 3 months, microbial diversity remained unchanged in both groups; the FMT group showed only a modest, non-significant increase in Observed and Chao1 indices. FMT responders showed reduced proinflammatory taxa, ethanol-producing microbiota, and antimicrobial-resistance genes, indicating favourable gut microbiome modulation.
FMT and lactulose have comparable efficacy in improving MHE and health-related quality of life in patients with cirrhosis.
Core Tip: Minimal hepatic encephalopathy (MHE) is common in cirrhosis. Gut dysbiosis is a key pathogenic player of MHE. Lactulose has been demonstrated to reverse MHE and prevent progression to overt hepatic encephalopathy. This randomised trial compared two endoscopic fecal microbiota transplantations (1 month apart) with daily lactulose over 3 months for MHE reversal. We found fecal microbiota transplantation and lactulose to have similar efficacy in improving MHE, supporting microbiota-targeted therapy as a viable alternative to standard care for MHE. These findings justify larger trials to assess long-term outcomes and optimal microbiota-based strategies.
- Citation: Agarwal A, Agrawal S, Talukdar D, Kaur M, Biswas S, Swaroop S, Golla R, Kandiyal B, Jana P, Tanwar S, Aggarwal A, Singh HC, Agarwal A, Mahajan M, Tiwari B, Nayak B, Goel A, Das B, Shalimar. Impact of two-session fecal microbiota transplantation on minimal hepatic encephalopathy in cirrhosis. World J Hepatol 2026; 18(4): 116153
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/116153.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.116153
Minimal hepatic encephalopathy (MHE) is a covert form of HE found in 30%-70% patients with cirrhosis, characterized by subtle motor and cognitive deficits detected on specialized psychometric tests[1]. It significantly impairs health-related quality of life (HRQoL) and is associated with sleep disturbances, motor incoordination, and increased risk of falls[2,3]. Over half of the patients with MHE progress to overt hepatic encephalopathy (OHE) within 3 years, and it is associated with poorer 5-year survival and higher hospitalization risk compared to those without MHE[4,5]. Despite this, MHE remains underrecognized in clinical practice and is not systematically treated in most patients[6-8]. Emerging evidence implicates gut dysbiosis as a key pathogenic player in the development of MHE, primarily through disruption of the gut-liver-brain axis. This dysregulation contributes to hyperammonemia, systemic inflammation, neuroinflammation, and endotoxemia[9,10].
The European Society for the Study of the Liver and the American Association for the Study of Liver Diseases recommend non-absorbable disaccharides such as lactulose (with adjunctive rifaximin use in selected patients) as the primary therapeutic modality for management of MHE[11,12]. Lactulose improves cognition through ammonia reduction and its osmotic effects, but its impact on the gut microbiome is inconsistent and does not reliably reverse cirrhosis-associated dysbiosis[13,14]. Rifaximin, although clinically beneficial, functions mainly as a non-absorbable antibiotic and acts by altering microbial metabolism rather than improving microbial richness or diversity[15].
Fecal microbiota transplantation (FMT), which has the potential to restore gut eubiosis, is effective in recurrent Clostridioides difficile infection and inflammatory bowel disease[16,17]. It has been shown to be safe in cirrhosis and is associated with a reduced recurrence of HE, decreased inflammatory cytokines, enhanced gut microbial diversity, and improved cognition[18,19]. Despite its promising results in patients with OHE, the role of FMT in the treatment of MHE has not been systematically evaluated. This randomized trial compared two endoscopic FMT infusions, administered one month apart, to daily lactulose therapy over three months for MHE resolution and HRQoL improvement.
This investigator-initiated, open-label, parallel-group randomized controlled trial was conducted at a single tertiary care hospital in India. The protocol was approved by the Institutional Ethics Committee (No. IEC-753/02.09.2022, RP-37/2022) and prospectively registered at the Clinical Trials Registry of India (No. CTRI/2023/02/050008). All procedures were performed according to the Good Clinical Practice guidelines and the Declaration of Helsinki.
Consecutive adult patients (age ≥ 18 years) with any etiology of cirrhosis who presented between February 2023 and April 2024 were screened for MHE using the psychometric hepatic encephalopathy score (PHES) (Supplementary material). Those who tested positive for MHE (PHES ≤ -5) were diagnosed with MHE and considered for inclusion in the study. The presence of cirrhosis was confirmed on hematological and biochemical parameters supplemented by imaging findings (ultrasound or computerized tomography). Patients with prior OHE, recent variceal bleed (within 1 month), recent alcohol use (within the last 1 month), recent illicit drug abuse/psychoactive medicine use (within the last 1 month), known psychiatric or neurologic illnesses, and prior transjugular intrahepatic portosystemic shunt placement or balloon occluded retrograde transvenous obliteration were excluded. Detailed eligibility criteria are provided in the Supple
Eligible patients were randomly assigned (1:1) to receive either FMT or lactulose using a computer-generated sequence (simple randomization). Allocation was concealed using sequentially numbered, opaque sealed envelopes prepared by a research assistant who was unaware of study outcomes. Patients were aware of the allocated treatment group. Masking was not done; however, the data analyst was blinded to group allocation at the time of analysis.
FMT arm: Patients in this group received two FMT sessions (day 0 and one month later). Details of the procedure are provided in the Supplementary material. Briefly, fresh stool from healthy donors at each study visit was processed within 6 hours of defecation. Fifty grams of donor stool was mixed with 250 mL of preservative-free normal saline and blended for 3-4 minutes. The suspension was sieved to prevent clogging during infusion. A total of 200 mL of fecal suspension was infused into the distal duodenum/jejunum via esophagogastroduodenoscopy. We utilized two healthy stool donors for the study, with the same donor for both sessions in a given patient. The demographic details of the 2 healthy donors are shown in the Supplementary material.
Lactulose arm: Patients received 30 mL lactulose once to thrice daily for 3 months, titrated to achieve 2-3 soft stools per day. An overview of the study protocol is provided in Supplementary Figure 1.
Patients were followed up on an outpatient basis after allocation of treatment groups. Visits were scheduled monthly (1 month, 2 months, and 3 months from randomization), with weekly telephonic monitoring to assess adverse events (AE) and in the lactulose group, to monitor stool frequency and ensure adherence. Baseline and follow-up assessments included PHES, hematological, and biochemical parameters. at every visit. Liver stiffness measurement (transient elastography- Fibroscan® 630 Expert), serum ammonia, Child-Turcotte-Pugh (CTP) score, model for end-stage liver disease (MELD) scores, stool samples for microbiome analysis, and HRQoL [assessed using the 36-item short form survey (SF-36) assessments][21] were obtained at baseline and 3-months. AEs were recorded and graded using CTCAE v5.0[22].
The primary outcome was the resolution of MHE at the end of 3 months, defined as PHES score > -5. Secondary outcomes included the development of OHE (as per the West Haven criteria)[12], changes in the CTP, MELD, SF-36 score, serum ammonia, and numerical PHES score/individual components at 3 months. In addition, the resolution of MHE was compared between treatment groups at 1- and 2-month follow-up.
As exploratory outcomes, changes in fecal microbiome diversity, composition, engraftment (through comparison with donor microbiome), and functional pathways were compared between treatment groups, as well as between subjects with MHE resolution and those with persistent MHE. We also explored whether different microbial genera were associated with specific clinical outcomes, and if there was donor-specific variation in microbiota and clinical outcomes. Safety outcomes included overall AE, serious AE (defined as CTCAE v5.0 Grade 3 or more), and infections.
Bacterial metagenomic DNA isolation was performed using the methodology established at the Biotechnology Research and Innovation Council-Translational Health Science and Technology Institute[23]. Approximately 200 mg of adult stool samples were collected into a microcentrifuge tube, resuspended in TE buffer (10 mmol/L Tris-Cl, 1 mmol/L ethylenediamine tetraacetic acid, pH 7-8), and the samples underwent enzymatic treatment with a cocktail of bacterial cell-wall lysing enzymes - lysozyme, mutanolysin, and lysostaphin. Following lysis, the samples were washed with TENP solution. The nucleic acid was precipitated with isopropanol and pelleted by centrifugation at 14000 rpm for 15 minutes. The pellet was washed twice with 75% chilled ethanol at 4 °C, air-dried, and finally dissolved in 10 mmol/L Tris-Cl (pH 8.0). To check the abundance of the leading bacterial population in the sample, 16S ribosomal RNA was targeted for metagenomic sequencing. The V3-V4 regions of the bacterial 16S rRNA gene were targeted for amplicon-based metagenomic sequencing. Polymerase chain reaction amplification was performed using the KAPA HiFi Hotstart Readymix kit (Roche, Switzerland), and libraries were prepared using the Nextera XT V2 dual index kit (Illumina, CA, United States). Quality control was performed using TapeStation 4200, and libraries were quantified and normalized to 4 nM using Qubit dsDNA HS kit (Invitrogen, Thermo Fisher Scientific, MA, United States). Sequencing was carried out on the Illumina NextSeq 2000 platform with 600-cycle chemistry (2 × 300 bp reads) (Illumina, CA, United States). The raw reads were preprocessed using the DADA2 pipeline. This process inferred precise amplicon sequence variants based on 100% sequence similarity using the make sequence table function. Chimeric sequences were discarded, and taxonomy was assigned to each amplicon sequence variant using the assign taxonomy function, which compared the representative sequences to the SILVA Reference database (Version 138.2) for the 16S rRNA gene, with taxonomic classification made at the genus level[24]. Further, the functional potential of the bacterial communities in the gut microbiome was inferred from 16S rRNA gene amplicon data using the Phylogenetic Investigation of Communities by Reconstruction of Unobserved States software (v2.4.2)[25] (The Huttenhower Lab, Harvard T.H. Chan School of Public Health). Core taxa were identified using eulerr, microbiome, and microbiomeutilities.
The normality of the dataset was tested using the Shapiro-Wilk test. Alpha diversity was assessed with phyloseq, and group differences were tested using a nonparametric Tukey-HSD test for normally distributed data, whereas the Mann-Whitney U test was used for non-parametric data. Low-abundance taxa were filtered out, and microbial composition comparisons were conducted using ampvis2. The Spearman correlation analysis between microbial taxa and PHES score was performed using linear regression. Donor engraftment was quantitatively assessed using the Fast expectation-maximization for microbial source tracking algorithm[26].
In stool microbiome analysis, alpha diversity represents the microbial diversity within a single stool sample, while beta diversity compares the diversity between different stool samples. In essence, alpha diversity is a measure of the “richness” (number of species) and “evenness” (relative abundance) within a sample, whereas beta diversity assesses how different the microbial composition is between two or more samples. Several indices are used to determine microbial diversity. Of those used in this study, the Observed and Chao1 indices provide an estimate of species richness but may underestimate the species diversity, whereas the Shannon index provides an assessment of the abundance of species, while Simpson’s index emphasises the evenness or relative abundance. Polymerase chain reaction detection of antimicrobial resistance (AMR) genes generated binary data (presence/absence). Comparison between responders and non-responders was performed using gene prevalence and analysed with Fisher’s Exact Test.
Previous studies on the use of lactulose for treatment of MHE have shown MHE improvement in 50%-65%[27-29] patients with lactulose and a spontaneous improvement rate of 10%-25% with placebo[27,30,31]. There is, however, limited data on the role of FMT in the resolution of MHE. We assumed that lactulose would improve MHE in 50% and FMT in 25% (similar to placebo). We estimated that 58 participants in each arm would be required to detect a statistically significant difference with 80% power and a two-sided alpha of 0.05. Accounting for an attrition rate of 10%, the total sample size was adjusted to 130 participants (65 per arm). The modified intention-to-treat analysis included all randomized patients who received at least 1 dose of the allocated intervention and had at least one follow-up PHES assessment. For study participants with missing data of the primary outcome on follow-up, the last observation was carried forward, and the same was indicated in the study results.
Resolution of MHE (primary outcome) was compared between groups by estimating the unadjusted group differences (absolute risk differences) and its 95% confidence intervals. Overall, intergroup PHES scores, HRQoL, and their components were compared using a linear mixed effects model, using patient ID as a random effect and allocated group (FMT/Lactulose) and time of measurement as fixed effects. The use of such a linear mixed-effects model allowed the assessment of repeated measures of numerical endpoints while adjusting for within-subject variations. Both intergroup (i.e., between FMT/Lactulose arms) and intragroup (i.e., within allocated treatment arm over time) comparisons were made. A P < 0.05 was considered statistically significant. Computed P-values were adjusted for multiple comparisons.
We expressed baseline/follow-up continuous data as mean with standard deviations or median and interquartile range, depending on the normality of the underlying data distribution. Nominal/ordinal data were presented as proportions with a 95% confidence interval. All data was entered in Microsoft Excel version 16.62 and was analyzed using RStudio Version 2024.09.1+394 (R Studio, Inc., Boston, MA, United States). Besides base packages in R and those listed for microbiome analysis, tidyverse, readxl, ggradar, tableone, emmeans, interpretCI, and lme4 packages were used.
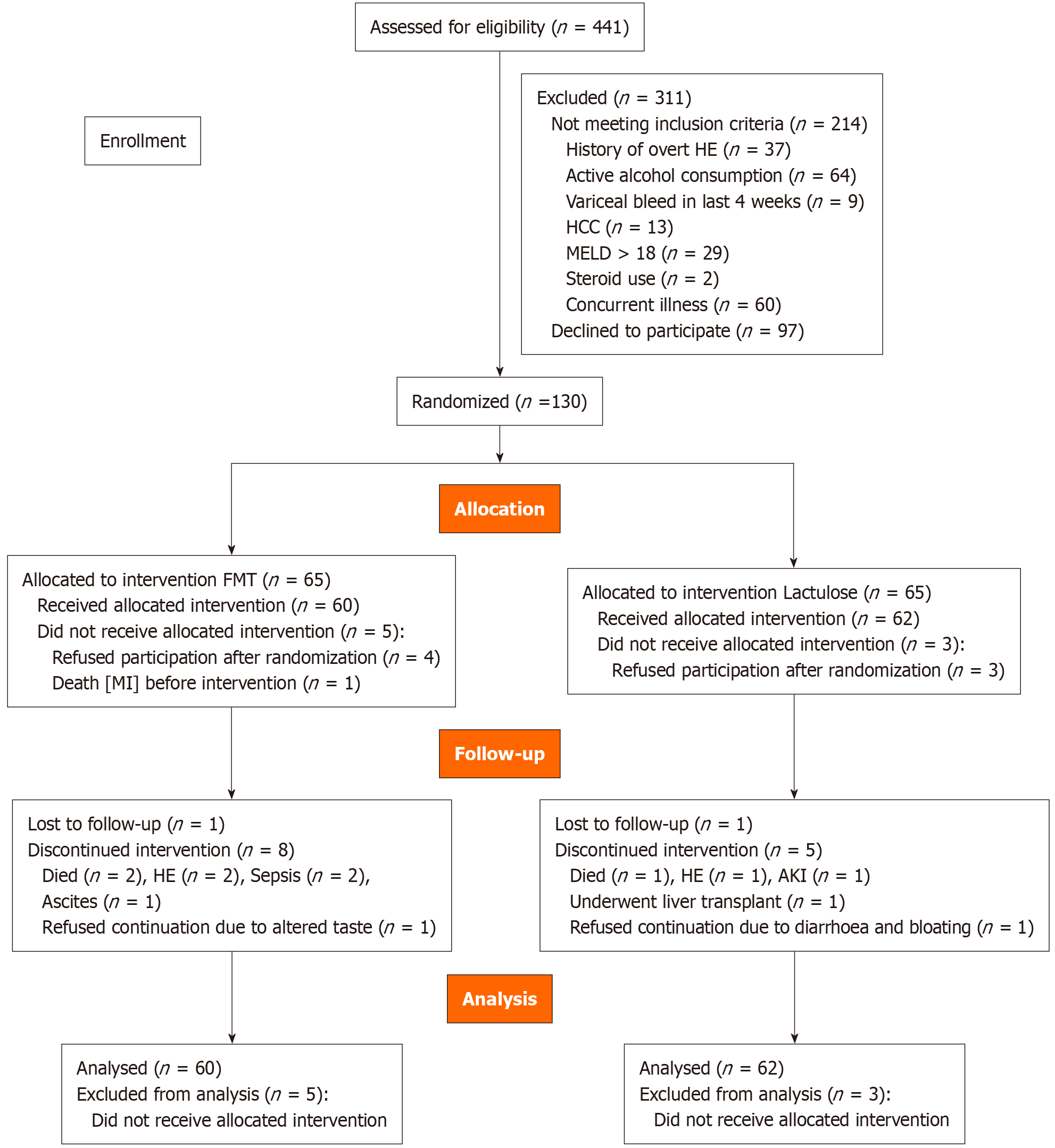
Between February 2023 and April 2024, 441 patients with cirrhosis were screened for MHE. A total of 130 eligible patients with MHE were randomized to receive either FMT or lactulose. The final analysis included 122 patients (FMT: 60; lactulose: 62) (Figure 1). The mean age (SD) of the cohort was 44.3 ± 8.9 years, and the majority were male (96.7%). The predominant etiologies of cirrhosis were alcohol-use (54.9%) and viral infections (27.1%). The median CTP and MELD scores were 7 [interquartile range (IQR): 6-9] and 13 (IQR: 10-17), respectively. Overall baseline characteristics are summarized in Table 1.
| Characteristics | Overall population (n = 122) | FMT (n = 60) | Lactulose (n = 62) |
| Age (years) | 44.3 ± 8.9 | 43.7 ± 9.1 | 44.8 ± 8.9 |
| Sex (male) | 118 (96.7) | 57 (95.0) | 61 (98.4) |
| BMI (kg/m2) | 23.6 (21.9-25.8) | 23.4 (21.2-25.9) | 23.8 (22.1-25.8) |
| Education (formal education in years) | 11.4 ± 3.4 | 11.3 ± 3.1 | 11.5 ± 3.7 |
| Etiology | |||
| Alcohol | 67 (54.9) | 32 (53.3) | 35 (56.4) |
| MASLD | 22 (18.0) | 9 (15.0) | 13 (21.0) |
| Hepatitis B | 25 (20.5) | 15 (25.0) | 10 (16.1) |
| Hepatitis C | 8 (6.6) | 4 (6.7) | 4 (6.5) |
| Hemoglobin (g/dL) | 11.1 (9.4-12.6) | 11.5 (10.12-12.85) | 10.8 (9.12-12.1) |
| TLC (mm3) | 5500 (4400-7350) | 6030 (5105-8655) | 5290 (4090-6150) |
| Platelet count (× 103/mm3) | 98 (66-128) | 99 (68-137) | 97 (65-118) |
| Total bilirubin (mg/dL) | 2.0 (1.3-2.6) | 2.1 (1.3-2.9) | 1.7 (1.1-2.4) |
| AST (U/L) | 64 (45-98) | 66 (47-106) | 64 (41-92) |
| ALT (U/L) | 35 (25-49) | 36 (24-51) | 33 (26-45) |
| ALP (U/L) | 139 (107-186) | 140 (109-186) | 139 (107-208) |
| Total protein (g/dL) | 7.9 (7.4-8.2) | 7.9 (7.3-8.2) | 7.9 (7.5-8.3) |
| Albumin (g/dL) | 3.5 ± 0.7 | 3.4 ± 0.7 | 3.6 ± 0.7 |
| Urea (mg/dL) | 22 (16-32) | 25 (16-34) | 20 (16-28) |
| Creatinine (mg/dL) | 0.9 ± 0.2 | 0.9 ± 0.2 | 0.9 ± 0.2 |
| Sodium (mEq/L) | 136.0 ± 4.6 | 136.1 ± 4.2 | 135.9 ± 4.9 |
| Potassium (mEq/L) | 4.4 ± 0.5 | 4.3 ± 0.6 | 4.5 ± 0.5 |
| INR | 1.5 (1.3-1.8) | 1.6 (1.3-1.8) | 1.4 (1.3-1.8) |
| AFP | 4.3 (2.7-5.8) | 4.5 (2.7-5.8) | 3.7 (2.68-6.0) |
| CTP class | |||
| A | 39 (32.0) | 15 (25.0) | 24 (38.7) |
| B | 65 (53.3) | 33 (55.0) | 32 (51.6) |
| C | 18 (14.6) | 12 (20.0) | 6 (9.7) |
| ANT | 12.4 ± 2.3 | 12.7 ± 2.2 | 12.0 ± 2.4 |
| LSM (kPa) | 40.1 (26.6-60.2) | 37.3 (28.0-58.6) | 43.3 (26.0-64.7) |
| CAP (dB/m) | 224 (193-249) | 223 (196-242) | 226 (190-259) |
| Ammonia | 86 (72-107) | 87 (66-107) | 84 (74-105) |
| CTP score | 7.0 (6.0-9.0) | 7.0 (6.5-9.0) | 7.0 (6.0-9.0) |
| MELD | 13 (10-17) | 14 (11-17) | 13 (10-16) |
| MELD-Na | 15.4 ± 4.5 | 15.7 ± 4.5 | 15.2 ± 4.5 |
At 3-month follow-up, MHE resolution was observed in 41 (68.3%) patients in the FMT arm and 41 (66.1%) in the lactulose arm [absolute risk-difference: 2.2% (95% confidence interval: -14.4% to 18.9%); P = 0.80]. Both groups showed progressive MHE improvement at 1 month and 2 months, without significant inter-group differences (Supplementary Table 1). No significant differences in MHE resolution were observed between the recipients of two FMT donors (Supplementary Table 2).
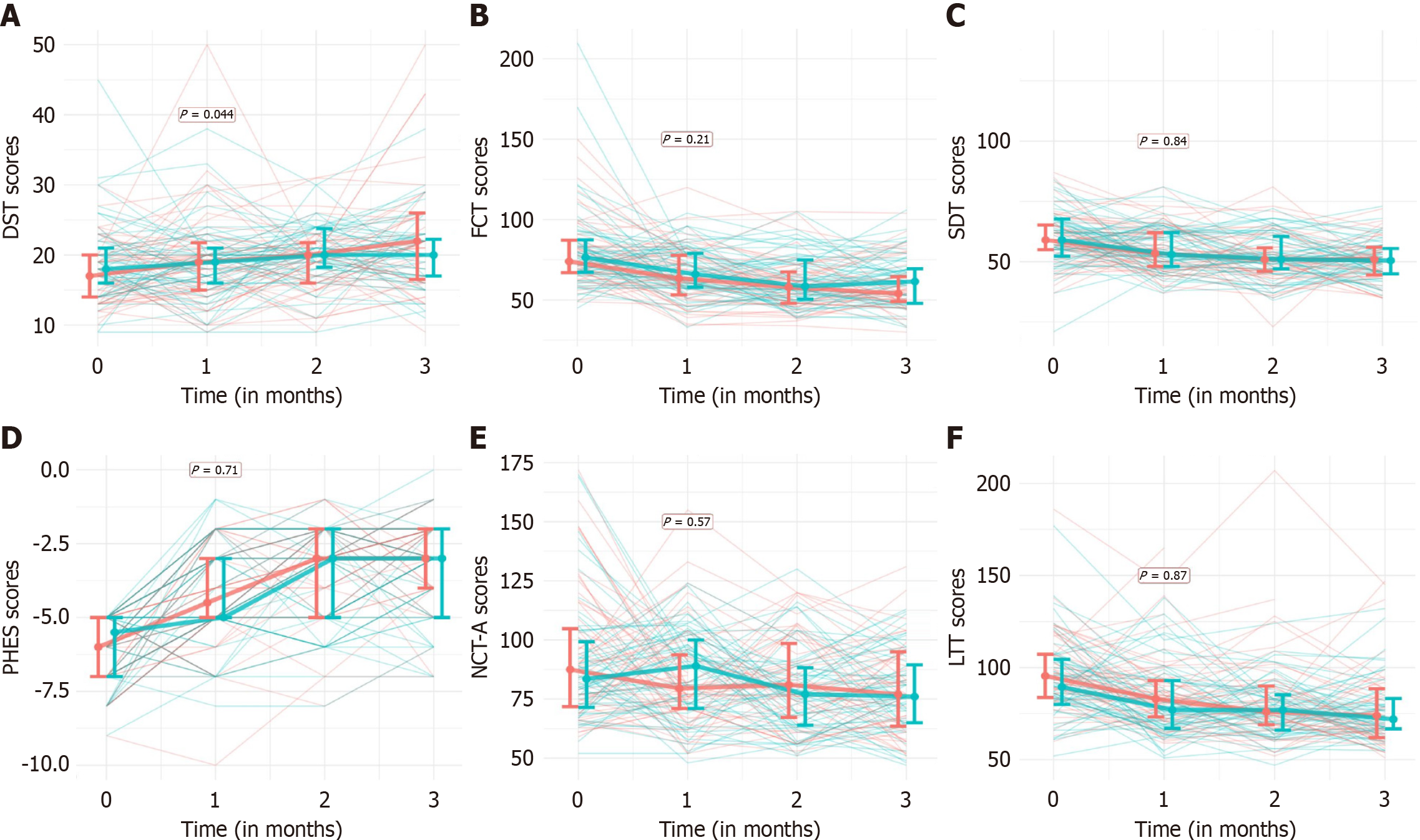
Cognitive changes: Both groups exhibited significant improvements in overall PHES scores and individual test components at 3 months (Figure 2; Table 2). No significant between-group differences were observed, except for a marginally greater improvement in the digit symbol test in the FMT group (adjusted intergroup difference -2.520 ± 1.237; P = 0.044).
| Outcomes | FMT baseline (n = 60) | FMT 3 months | P valuea,1 | Lactulose baseline (n = 62) | Lactulose 3-month (n = 62) | P value2 | Intergroup difference3 (adjusted mean difference ± standard error) [adjusted risk difference (95% confidence interval)] | P value4 |
| Primary outcome | ||||||||
| Resolution of MHE | - | 41 (68.3%) | - | - | 41 (66.1%) | - | 2.2% (-14.4% to 18.9%) | 0.800 |
| Secondary outcomes | ||||||||
| Overt HE | - | 2 (3.3%) | - | - | 1 (1.6%) | - | -1.7% (-7.2% to 3.8%) | 1.000 |
| MELD-Na score | 15.7 ± 4.5 | 14.9 ± 4.3 | 0.18 | 15.2 ± 4.5 | 13.7 ± 4.3 | 0.06 | -0.89 ± 0.35 | 0.250 |
| CTP score | 7.9 ± 1.5 | 7.2 ± 1.5 | < 0.001 | 7.4 ± 1.4 | 7.2 ± 1.5 | 0.22 | 0.32 ± 0.22 | 0.150 |
| Ammonia | 89 ± 30 | 85 ± 27 | 0.351 | 89 ± 26 | 82 ± 22 | 0.109 | -0.9 ± 12.5 | 0.950 |
| PHES score | -6.1 ± 1.2 | -3.3 ± 1.8 | < 0.001 | -6.1 ± 1.2 | -3.2 ± 1.5 | < 0.001a | -0.12 ± 0.31 | 0.710 |
| PHES individual components | ||||||||
| NCT-A | 89.6 ± 25.4 | 78.0 ± 17.1 | 0.065 | 93.4 ± 28.3 | 80.1 ± 20.0 | 0.009a | -2.08 ± 3.61 | 0.570 |
| FCT | 79.2 ± 20.7 | 62.4 ± 15.8 | < 0.001 | 79.2 ± 20.7 | 58.4 ± 15.7 | < 0.001a | 3.76 ± 2.99 | 0.210 |
| SDT | 61.0 ± 10.5 | 50.9 ± 7.8 | < 0.001 | 61.0 ± 10.5 | 50.6 ± 9.4 | < 0.001a | 0.34 ± 1.66 | 0.840 |
| DST | 17.4 ± 4.3 | 22.3 ± 7.8 | < 0.001 | 17.4 ± 4.3 | 20.2 ± 5.0 | 0.044a | -2.52 ± 1.24 | 0.044a |
| LTT | 97.7 ± 20.7 | 76.9 ± 16.9 | < 0.001 | 97.7 ± 20.7 | 78.3 ± 22.4 | < 0.001a | -0.60 ± 3.73 | 0.870 |
| Health-related Quality of Life (SF-36) domains | ||||||||
| Physical functioning | 657 ± 183 | 783 ± 145 | 0.001 | 637 ± 193 | 798 ± 136 | < 0.001a | 15.07 ± 27.69 | 0.590 |
| Role limitation physical health | 193 ± 140 | 290 ± 134 | 0.005 | 161 ± 130 | 302 ± 124 | < 0.001a | 7.36 ± 25.69 | 0.780 |
| Role limitation and mental health | 237 ± 84 | 280 ± 46 | 0.007 | 242 ± 82 | 260 ± 78 | 0.250 | -19.64 ± 12.85 | 0.130 |
| Energy/fatigue | 188 ± 40 | 217 ± 42 | 0.002 | 185 ± 39 | 206 ± 42 | 0.005a | -11.23 ± 8.27 | 0.180 |
| Emotional well-being | 345 ± 33 | 363 ± 25 | 0.003 | 338 ± 38 | 359 ± 31 | 0.004a | -4.76 ± 5.62 | 0.400 |
| Social functioning | 164 ± 33 | 186 ± 23 | 0.001 | 166 ± 33 | 179 ± 25 | 0.018a | -7.58 ± 4.76 | 0.120 |
| Pain | 150 ± 29 | 183 ± 22 | < 0.001 | 148 ± 24 | 175 ± 25 | < 0.001a | -8.86 ± 4.64 | 0.059 |
| General health | 241 ± 47 | 276 ± 41 | 0.001 | 229 ± 39 | 284 ± 36 | < 0.001a | 7.84 ± 7.72 | 0.312 |
HRQoL: Significant improvement was observed in most SF-36 domains in both groups over 3 months. No significant inter-group differences were observed after adjusting for baseline values (Supplementary Table 3).
OHE and liver functions: Two patients in the FMT group and one in the lactulose group developed OHE, all triggered by sepsis (pneumonia and urinary tract infection in FMT; cellulitis in lactulose). All were treated with antibiotics and a lactulose-rifaximin combination. None of the patients developed spontaneous bacterial peritonitis during the study duration.
Over three months, there were no significant differences in the serum ammonia levels or MELD-Na scores between the two groups (Table 2 and Supplementary Figure 2). Although CTP scores improved significantly from baseline in the FMT group, between-group differences did not reach statistical significance after adjusting for baseline values (Table 2).
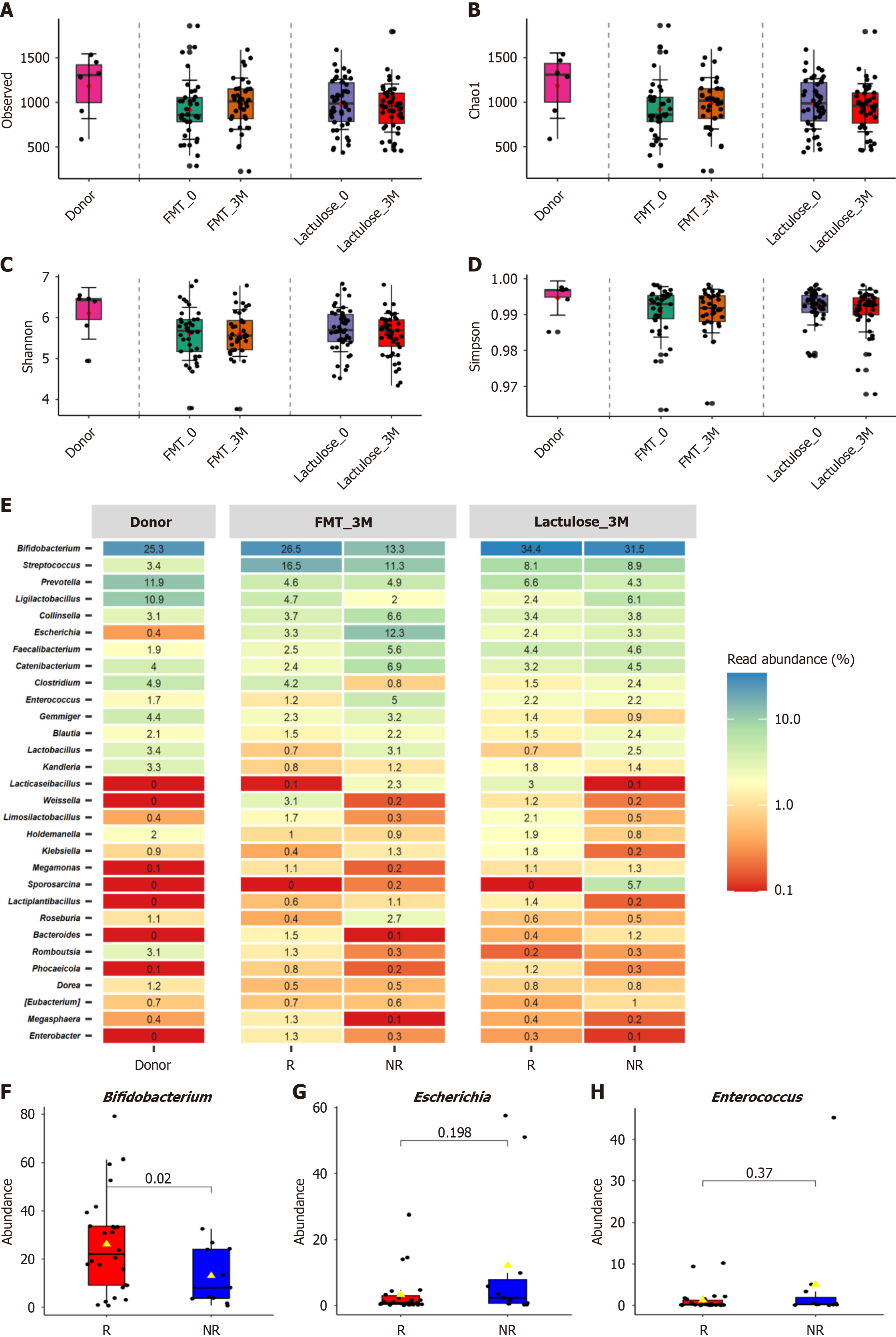
We analyzed 172 fecal samples, including paired samples available from 37/60 (61.7%) FMT and 46/62 (74.2%) lactulose recipients, along with 6 samples from 2 healthy donors (Supplementary Table 4). Post-FMT, microbial diversity showed a modest, non-significant increase at the Observed (917.5 ± 331.7 to 985.9 ± 287.9) and Chao1 (918.7 ± 332.3 to 987.6 ± 288.8) index levels. No changes were detected in Shannon (5.6 ± 0.6 to 5.6 ± 0.6) or Simpson (1.0 ± 0.0 to 1.0 ± 0.0) indices in patients receiving FMT, indicating stability in overall community evenness and richness. Similarly, microbial diversity remained stable in lactulose-treated patients as well, with a slight, non-significant decline observed in the Observed (978.6 ± 281.8 to 936.5 ± 269.7) and Chao1 (980.9 ± 282.6 to 937.8 ± 269.9) indices. Shannon (5.7 ± 0.5 to 5.6 ± 0.5) and Simpson (1.0 ± 0.0 to 1.0 ± 0.0) indices showed no change, indicating preserved community structure and evenness (Figure 3A-D). Although both Observed and Chao1 diversity indices increased three months post-FMT compared to the lactulose group, the differences were not statistically significant after adjusting for baseline diversity (P = 0.15). The results of the normality test (Shapiro-Wilk test) are provided in the Supplementary material. The details of the core taxa and abundance with each treatment are shown in Supplementary Figures 3 and 4.
Donor-derived bacterial engraftment (> 50%) was observed in 19/37 FMT recipients. Among those with MHE resolution (n = 26), 58% had > 50% engraftment rate vs 36% in non-responders (n = 11) (Supplementary Table 5). This indicates a potential association between higher engraftment levels and positive clinical response following FMT, although it did not reach statistical significance. Bifidobacterium abundance was significantly higher in the MHE responder group (P = 0.02), while Escherichia (P = 0.20) and Enterococcus (P = 0.37) were lower, although not significant. Correlation analysis between Bifidobacterium abundance and PHES scores at 3 months post-FMT revealed a weak positive correlation (Spearman’s rho = 0.196, P-value = 0.244), suggesting that a higher abundance of Bifidobacterium may be associated with better cognitive performance (Supplementary Figure 5). A slight statistically insignificant decrease in Escherichia was also noted in the lactulose group (Figure 3E-H). The associations between PHES scores and individual bacterial genera, including Bifidobacterium, Escherichia, and Enterococcus, were post-hoc and exploratory, and the findings should be interpreted as hypothesis-generating.
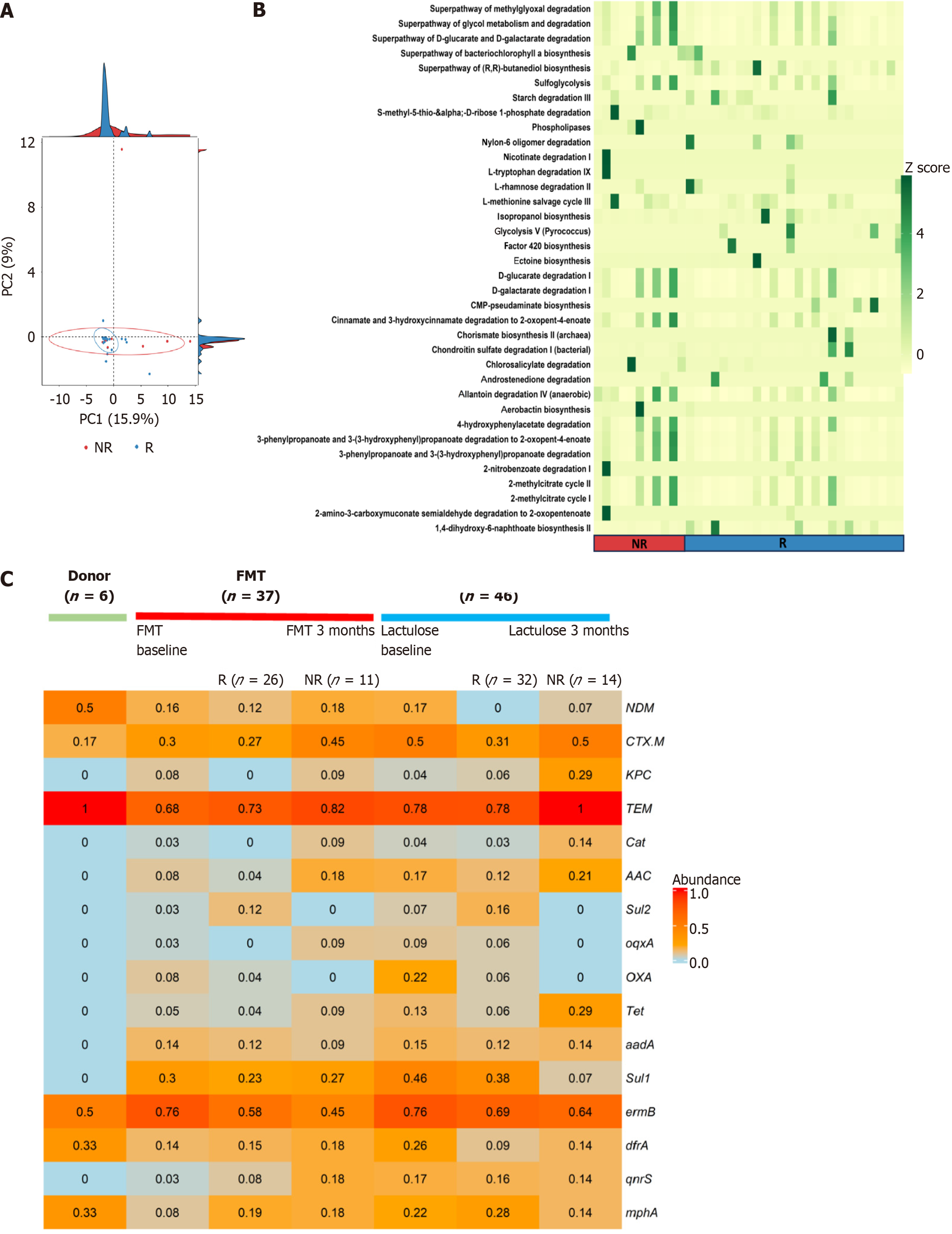
Principal component analysis (PCA) of Phylogenetic Investigation of Communities by Reconstruction of Unobserved States-predicted microbial functions showed substantial overlap between responders and non-responders in the FMT group; however, responders formed a tighter cluster, whereas non-responders were widely dispersed, indicating greater functional heterogeneity (PC1: 15.9%, PC2: 9%; Figure 4A). In the lactulose group, a similar pattern was observed: Responders clustered more tightly, whereas non-responders were more dispersed, indicating higher functional variability (PC1: 12.2%, PC2: 11.4%; Supplementary Figure 6A). We identified 36 differentially enriched pathways (P-adjusted < 0.05, represented by Z score) between MHE responders and non-responders in the FMT arm at 3 months (Supplementary Table 6, Figure 4B). Notably, the super pathways relevant to the gut-liver-brain axis, including (R, R)-butanediol degradation (P-adjust = 0.03) and chondroitin sulfate degradation (bacteria) (P-adjust = 0.01), were significantly enriched in MHE responders in the FMT arm. In contrast, the super pathway of methylglyoxal degradation (P-adjust = 0.01) was more active in the non-responders after FMT, suggesting a higher burden of methylglyoxal or related toxic metabolites in these patients. Additionally, pathways like D-galactarate degradation I (P-adjust = 0.04), allantoin degradation IV (anaerobic) pathway (P-adjust = 0.03), and the super pathway of D-glucarate and D-galactarate degradation (P-adjust = 0.03) were elevated in MHE non-responders. Furthermore, in the lactulose arm, we identified 68 differentially enriched pathways when stratified based on MHE response (Supplementary Table 7, Supplementary Figure 6B). Pathways such as creatinine degradation I (P-adjust < 0.001) and creatinine degradation II (P-adjust = 0.01) were higher in MHE responders, suggesting enhanced nitrogen metabolism and detoxification.
AMR genes carrriage was higher in MHE non-responders across both groups (Figure 4C). In the FMT group: Cefotaxime-Munich (CTX-M): Non-responders (5/11) vs responders (7/26), Klebsiella pneumoniae carbapenemase (KPC): Non-responders (1/11) vs responders (0/26), temoniera (TEM): Non-responders (9/11) vs responders (19/26), aminoglycoside acetyltransferase (AAC): Non-responders (2/11) vs responders (1/26). In the lactulose group: CTX-M: Non-responders (7/14) vs responders (10/32), KPC: Non-responders (4/14) vs responders (2/32), TEM: Non-responders (14/14) vs responders (25/32), AAC: Non-responders (3/14) vs responders (4/36). However, in both the FMT and lactulose groups, no AMR gene showed a significant difference in prevalence between responder and non-responder after false discovery rate correction. Although a few genes in the Lactulose group had lower unadjusted P-values, none reached statistical significance (Supplementary Tables 8 and 9). These detectable differences may be partly attributable to the modest sample size, which reduces statistical power. Compared to baseline, the prevalence of CTX-M, KPC, and AAC remained high or unchanged in the non-responder groups of both arms, suggesting that lack of clinical response is associated with persistent carriage of key AMR genes. Further comparison between FMT and lactulose responders showed that, after 3 months, the prevalence of AMR genes, including AAC, sul1, and ermB, was lower in the FMT group than in the lactulose group. Interestingly, KPC, Cat, and oqxA were absent in FMT responders, indicating a potential role of FMT in suppressing or eliminating certain resistance determinants. Additionally, none of these AMR genes (KPC, Cat, AAC, sul2, oqxA, OXA, tet, aadA, sul1) were detected in the donor stool, highlighting the low AMR burden in the donor microbiota.
Overall, 47.5% of patients experienced at least one AE, with no significant difference between groups (FMT: 53.3% vs lactulose: 41.9%; P = 0.21). Bloating was significantly more frequent in the lactulose group (P < 0.01), while constipation occurred more commonly in the FMT group (P < 0.01). Fever was observed more frequently in patients receiving FMT compared to those in the lactulose group (20.0% vs 8.1%, P = 0.06). Other AE were comparable between the two groups (Table 3).
| Overall (n = 122) | FMT (n = 60) | Lactulose (n = 62) | P value | |
| Any adverse events1 | 58 (47.5) | 32 (53.3) | 26 (41.9) | 0.21 |
| Bloating | 18 (14.8) | 3 (5.0) | 15 (24.2) | < 0.01 |
| Abdominal pain | 16 (13.1) | 7 (11.7) | 9 (14.5) | 0.64 |
| Diarrhoea | 11 (9.0) | 6 (10.0) | 5 (8.1) | 0.71 |
| Vomiting | 4 (3.3) | 3 (5.0) | 1 (1.6) | 0.36 |
| Malaise | 9 (7.4) | 7 (11.7) | 2 (3.2) | 0.09 |
| Fever | 17 (13.9) | 12 (20.0) | 5 (8.1) | 0.06 |
| Cough | 3 (2.5) | 3 (5.0) | 0 | 0.12 |
| Constipation | 7 (5.7) | 7 (11.7) | 0 | < 0.01 |
| Altered taste | 1 (0.8) | 1 (1.7) | 0 | 0.49 |
| Death | 3 (2.5) | 2 (3.3) | 1 (1.6) | 0.62 |
| Overt HE | 3 (2.5) | 2 (3.3) | 1 (1.6) | 1.00 |
| Sepsis | 7 (5.7) | 5 (8.3) | 2 (3.2) | 0.13 |
| Pneumonia | 2 (1.6) | 2 (3.3) | 0 | |
| Urosepsis | 2 (1.6) | 1 (1.7) | 1 (1.6) | |
| Cellulitis | 1 (0.8) | 0 | 1 (1.6) | |
| Gastrointestinal | 2 (1.6) | 2 (3.3) | 0 |
Seven patients required hospitalisation during the study (4 in the FMT group, 3 in the lactulose group). Hospitalizations in the FMT group were due to OHE and sepsis (2 patients each), whereas in the lactulose group, hospitalizations were due to OHE, acute kidney injury, and acute on chronic liver failure (1 each). Three deaths occurred during the study period. In the FMT group, one patient died from progressive liver failure and another from acute respiratory distress syndrome secondary to pneumonia. One death occurred in the lactulose group due to worsening liver disease. Recidivism was noted in two patients, one in each group.
Our randomized controlled trial demonstrates that two sessions of FMT led to MHE resolution in approximately two-thirds of patients with improvement in cognitive function and HRQoL, comparable to three months of daily lactulose therapy. FMT was associated with a positive impact on gut microbiota with increased diversity, and those with MHE resolution had a reduction in pathogenic bacteria and AMR resistance genes, which may be important intermediate outcomes in patients with cirrhosis. FMT was safe, well-tolerated, and was not associated with a higher incidence of AE or liver-related events, supporting its utility in this patient population and indicating potential for further exploration and optimization.
Non-absorbable disaccharides like lactulose are well-established for MHE treatment[12], and MHE reversal, cognitive improvement, and quality of life (SF-36) improvements seen with lactulose in our trial align with prior studies[27-29]. However, lactulose response is variable, optimal duration is unclear, and long-term compliance is often a challenge due to adverse effects[27]. Our results indicate that two sessions of FMT, a month apart, were comparable to three months of lactulose therapy, with sustained benefit. While the clinical benefits of FMT over lactulose were limited, we believe that FMT may be a useful alternative therapeutic option, especially among patients with MHE who do not tolerate or respond to lactulose therapy.
Among FMT recipients, microbial diversity indices (Operational taxonomic unit and Chao1) showed a modest, non-significant upward trend, while evenness (Shannon and Simpson indices) remained largely unchanged compared with lactulose. Although exploratory, these patterns suggest that FMT may induce subtle, potentially beneficial changes in the gut microbial community composition rather than a complete restoration of eubiosis. While not statistically significant, these changes may still be biologically relevant in cirrhosis, where microbial networks are often fragile. Thus, FMT may carry significant potential among patients with cirrhosis and warrants further exploration and optimization to further enhance this ecological shift.
Patients in both treatment arms had reductions in serum ammonia, but the change was not statistically significant compared to baseline values. Pathway analysis in MHE responders revealed enhanced nitrogen metabolism and detoxification activity, higher mean engraftment rate, and significantly greater relative abundance of Bifidobacterium, a genus linked to gut homeostasis and short-chain fatty acid production[18]. Bifidobacterium bifidum, linked to upregulation of tight junction proteins, may improve MHE by reducing gut permeability to bacteria/bacterial endotoxins and systemic inflammation[32,33].
In our study, on functional metagenomic analysis of 16S rRNA gene amplicon data, we identified 36 differentially enriched microbial pathways (P-adjusted < 0.05, represented by Z score) between MHE responders and non-responders in the FMT arm at 3 months. Notably, pathways relevant to the gut-liver-brain axis, including (R, R)-butanediol de
AE occurred in nearly half the patients in both groups, with no statistically significant difference overall. However, infectious events, including fever (20.0% vs 8.1%) and sepsis (8.3% vs 3.3%), were higher in the FMT group and approached significance. Although our sample size was underpowered to detect definitive differences, these trends raise safety concerns. It is important to note that these AE in FMT may be non-trivial, especially considering that cirrhosis is associated with altered homeostasis and an enhanced inflammatory response to otherwise well-tolerated antigens. FMT could be leading to transient bacteremia/translocation of lipopolysaccharides or other pathogen-associated molecular patterns, which could be responsible for the systemic inflammatory response. Unfortunately, we did not perform longitudinal assessments of inflammatory cytokines, endotoxin levels, or other immune markers that could have clarified whether these events were related to FMT-related immune modulation or incidental infections.
There are several limitations of this study. First, the absence of a placebo or sham arm limits our ability to distinguish the true biological effect of FMT or lactulose from placebo responses. Second, we did not use direct shotgun metagenomic sequencing to analyze the gut microbiota and relied on predicted metagenomic functions using Phylogenetic Investigation of Communities by Reconstruction of Unobserved States 2. This may be particularly important as patients with cirrhosis often have significant gut dysbiosis where strain-level functions may be increasingly relevant, and they may also harbor novel and poorly characterized species, which may not be adequately picked up using the current study methodology, and that could have limited our ability to assess the gut microbial changes. Third, the study design involved only two upper gastrointestinal infusions of FMT compared to continuous lactulose therapy over 3 months, creating a potential treatment exposure imbalance. The variability in the lactulose titration and adherence may have influenced outcomes despite close monitoring. In addition, 22% of eligible patients denied participation, often due to lack of symptoms or concerns about FMT, introducing potential selection bias. The study was also open-label, and that could be an additional source of bias in outcome assessment, even though the chosen outcomes were objective, and we made all possible attempts to reduce bias. Although the sample size in this study was larger than that of most previous studies on MHE, it still limits the power to detect smaller differences between treatment groups, including differences in AE. Finally, no formal cost-effectiveness analysis was performed, which would have helped contextualize the clinical relevance and feasibility of FMT in practice.
In conclusion, in this study, the effect of FMT on MHE resolutions seems to be comparable to that of daily oral lactulose. In addition, FMT was also effective in preventing the development of OHE, improving cognitive function, and improving quality of life. By restoring gut eubiosis, FMT has the potential to further reduce the risk of disease recurrence and mitigate complications associated with gut dysbiosis. Larger, multicenter studies with longer follow-up are required to validate our findings and optimise FMT protocols for clinical use.
| 1. | Pisarek W. Minimal hepatic encephalopathy - diagnosis and treatment. Prz Gastroenterol. 2021;16:311-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 2. | Ridola L, Nardelli S, Gioia S, Riggio O. Quality of life in patients with minimal hepatic encephalopathy. World J Gastroenterol. 2018;24:5446-5453. [PubMed] [DOI] [Full Text] |
| 3. | Bajaj JS, Duarte-Rojo A, Xie JJ, Acharya C, Wade JB, Robles C, Thacker LR, Flud C, Fagan A, Garcia-Saenz-de-Sicilia M, White MB, Kelly M, Nguyen V, Gavis EA, Vargas HE. Minimal Hepatic Encephalopathy and Mild Cognitive Impairment Worsen Quality of Life in Elderly Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:3008-3016.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 4. | Faccioli J, Nardelli S, Gioia S, Riggio O, Ridola L. Minimal Hepatic Encephalopathy Affects Daily Life of Cirrhotic Patients: A Viewpoint on Clinical Consequences and Therapeutic Opportunities. J Clin Med. 2022;11:7246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Ampuero J, Montoliú C, Simón-Talero M, Aguilera V, Millán R, Márquez C, Jover R, Rico MC, Sendra C, Serra MÁ, Romero-Gómez M. Minimal hepatic encephalopathy identifies patients at risk of faster cirrhosis progression. J Gastroenterol Hepatol. 2018;33:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 6. | Gairing SJ, Mangini C, Zarantonello L, Gioia S, Nielsen EJ, Danneberg S, Gabriel M, Ehrenbauer AF, Bloom PP, Ripoll C, Sultanik P, Galle PR, Labenz J, Thabut D, Zipprich A, Lok AS, Weissenborn K, Marquardt JU, Lauridsen MM, Nardelli S, Montagnese S, Labenz C. Prevalence of Minimal Hepatic Encephalopathy in Patients With Liver Cirrhosis: A Multicenter Study. Am J Gastroenterol. 2023;118:2191-2200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 7. | Rathi S, Chopra M, Chouduri G, Sharma P, Madan K, Chhabra M, Rai RR, Govil A, Konar A, Goenka M, Agarwal M, Mukherjee J, Thorat V, Salunkhe S, Abraham P, Nagral A, Jhaveri A, Bhat N, Varghese J, R S A, Ravishankar, Reddy DC, Dhiman RK. Prevalence of Minimal Hepatic Encephalopathy in Patients With Liver Cirrhosis: A Cross-Sectional, Clinicoepidemiological, Multicenter, Nationwide Study in India: The PREDICT Study. J Clin Exp Hepatol. 2019;9:476-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 8. | Lv XH, Lu Q, Deng K, Yang JL, Yang L. Prevalence and Characteristics of Covert/Minimal Hepatic Encephalopathy in Patients With Liver Cirrhosis: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2024;119:690-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 9. | Kang DJ, Betrapally NS, Ghosh SA, Sartor RB, Hylemon PB, Gillevet PM, Sanyal AJ, Heuman DM, Carl D, Zhou H, Liu R, Wang X, Yang J, Jiao C, Herzog J, Lippman HR, Sikaroodi M, Brown RR, Bajaj JS. Gut microbiota drive the development of neuroinflammatory response in cirrhosis in mice. Hepatology. 2016;64:1232-1248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 90] [Article Influence: 9.0] [Reference Citation Analysis (2)] |
| 10. | Ahluwalia V, Betrapally NS, Hylemon PB, White MB, Gillevet PM, Unser AB, Fagan A, Daita K, Heuman DM, Zhou H, Sikaroodi M, Bajaj JS. Impaired Gut-Liver-Brain Axis in Patients with Cirrhosis. Sci Rep. 2016;6:26800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 189] [Cited by in RCA: 183] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 11. | Vilstrup H, Amodio P, Bajaj J, Cordoba J, Ferenci P, Mullen KD, Weissenborn K, Wong P. Hepatic encephalopathy in chronic liver disease: 2014 Practice Guideline by the American Association for the Study of Liver Diseases and the European Association for the Study of the Liver. Hepatology. 2014;60:715-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1758] [Cited by in RCA: 1514] [Article Influence: 126.2] [Reference Citation Analysis (5)] |
| 12. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatic encephalopathy. J Hepatol. 2022;77:807-824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 362] [Cited by in RCA: 322] [Article Influence: 80.5] [Reference Citation Analysis (5)] |
| 13. | Lunia MK, Sharma BC, Sharma P, Sachdeva S, Srivastava S. Probiotics prevent hepatic encephalopathy in patients with cirrhosis: a randomized controlled trial. Clin Gastroenterol Hepatol. 2014;12:1003-8.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 165] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 14. | Bajaj JS, Ridlon JM, Hylemon PB, Thacker LR, Heuman DM, Smith S, Sikaroodi M, Gillevet PM. Linkage of gut microbiome with cognition in hepatic encephalopathy. Am J Physiol Gastrointest Liver Physiol. 2012;302:G168-G175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 462] [Cited by in RCA: 438] [Article Influence: 31.3] [Reference Citation Analysis (5)] |
| 15. | Bajaj JS, Heuman DM, Sanyal AJ, Hylemon PB, Sterling RK, Stravitz RT, Fuchs M, Ridlon JM, Daita K, Monteith P, Noble NA, White MB, Fisher A, Sikaroodi M, Rangwala H, Gillevet PM. Modulation of the metabiome by rifaximin in patients with cirrhosis and minimal hepatic encephalopathy. PLoS One. 2013;8:e60042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 362] [Cited by in RCA: 350] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 16. | Baunwall SMD, Lee MM, Eriksen MK, Mullish BH, Marchesi JR, Dahlerup JF, Hvas CL. Faecal microbiota transplantation for recurrent Clostridioides difficile infection: An updated systematic review and meta-analysis. EClinicalMedicine. 2020;29-30:100642. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 207] [Cited by in RCA: 195] [Article Influence: 32.5] [Reference Citation Analysis (1)] |
| 17. | Caldeira LF, Borba HH, Tonin FS, Wiens A, Fernandez-Llimos F, Pontarolo R. Fecal microbiota transplantation in inflammatory bowel disease patients: A systematic review and meta-analysis. PLoS One. 2020;15:e0238910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 18. | Bajaj JS, Fagan A, Gavis EA, Sterling RK, Gallagher ML, Lee H, Matherly SC, Siddiqui MS, Bartels A, Mousel T, Davis BC, Puri P, Fuchs M, Moutsoglou DM, Thacker LR, Sikaroodi M, Gillevet PM, Khoruts A. Microbiota transplant for hepatic encephalopathy in cirrhosis: The THEMATIC trial. J Hepatol. 2025;83:81-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 34] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 19. | Bloom PP, Donlan J, Torres Soto M, Daidone M, Hohmann E, Chung RT. Fecal microbiota transplant improves cognition in hepatic encephalopathy and its effect varies by donor and recipient. Hepatol Commun. 2022;6:2079-2089. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 20. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2259] [Cited by in RCA: 2068] [Article Influence: 258.5] [Reference Citation Analysis (6)] |
| 21. | Lins L, Carvalho FM. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016;4:2050312116671725. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 876] [Cited by in RCA: 730] [Article Influence: 73.0] [Reference Citation Analysis (1)] |
| 22. | Freites-Martinez A, Santana N, Arias-Santiago S, Viera A. Using the Common Terminology Criteria for Adverse Events (CTCAE - Version 5.0) to Evaluate the Severity of Adverse Events of Anticancer Therapies. Actas Dermosifiliogr (Engl Ed). 2021;112:90-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 705] [Cited by in RCA: 631] [Article Influence: 126.2] [Reference Citation Analysis (1)] |
| 23. | Bag S, Saha B, Mehta O, Anbumani D, Kumar N, Dayal M, Pant A, Kumar P, Saxena S, Allin KH, Hansen T, Arumugam M, Vestergaard H, Pedersen O, Pereira V, Abraham P, Tripathi R, Wadhwa N, Bhatnagar S, Prakash VG, Radha V, Anjana RM, Mohan V, Takeda K, Kurakawa T, Nair GB, Das B. An Improved Method for High Quality Metagenomics DNA Extraction from Human and Environmental Samples. Sci Rep. 2016;6:26775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 130] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 24. | Quast C, Pruesse E, Yilmaz P, Gerken J, Schweer T, Yarza P, Peplies J, Glöckner FO. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 2013;41:D590-D596. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27806] [Cited by in RCA: 20134] [Article Influence: 1548.8] [Reference Citation Analysis (8)] |
| 25. | Douglas GM, Maffei VJ, Zaneveld JR, Yurgel SN, Brown JR, Taylor CM, Huttenhower C, Langille MGI. PICRUSt2 for prediction of metagenome functions. Nat Biotechnol. 2020;38:685-688. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5471] [Cited by in RCA: 3972] [Article Influence: 662.0] [Reference Citation Analysis (5)] |
| 26. | Shenhav L, Thompson M, Joseph TA, Briscoe L, Furman O, Bogumil D, Mizrahi I, Pe'er I, Halperin E. FEAST: fast expectation-maximization for microbial source tracking. Nat Methods. 2019;16:627-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 397] [Article Influence: 56.7] [Reference Citation Analysis (0)] |
| 27. | Prasad S, Dhiman RK, Duseja A, Chawla YK, Sharma A, Agarwal R. Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy. Hepatology. 2007;45:549-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 485] [Cited by in RCA: 398] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 28. | Sidhu SS, Goyal O, Parker RA, Kishore H, Sood A. Rifaximin vs. lactulose in treatment of minimal hepatic encephalopathy. Liver Int. 2016;36:378-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 29. | Mittal VV, Sharma BC, Sharma P, Sarin SK. A randomized controlled trial comparing lactulose, probiotics, and L-ornithine L-aspartate in treatment of minimal hepatic encephalopathy. Eur J Gastroenterol Hepatol. 2011;23:725-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 164] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 30. | Sidhu SS, Goyal O, Mishra BP, Sood A, Chhina RS, Soni RK. Rifaximin improves psychometric performance and health-related quality of life in patients with minimal hepatic encephalopathy (the RIME Trial). Am J Gastroenterol. 2011;106:307-316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 187] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 31. | Bajaj JS, Heuman DM, Wade JB, Gibson DP, Saeian K, Wegelin JA, Hafeezullah M, Bell DE, Sterling RK, Stravitz RT, Fuchs M, Luketic V, Sanyal AJ. Rifaximin improves driving simulator performance in a randomized trial of patients with minimal hepatic encephalopathy. Gastroenterology. 2011;140:478-487.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 194] [Cited by in RCA: 179] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 32. | Al-Sadi R, Dharmaprakash V, Nighot P, Guo S, Nighot M, Do T, Ma TY. Bifidobacterium bifidum Enhances the Intestinal Epithelial Tight Junction Barrier and Protects against Intestinal Inflammation by Targeting the Toll-like Receptor-2 Pathway in an NF-κB-Independent Manner. Int J Mol Sci. 2021;22:8070. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 33. | Fukuda S, Toh H, Hase K, Oshima K, Nakanishi Y, Yoshimura K, Tobe T, Clarke JM, Topping DL, Suzuki T, Taylor TD, Itoh K, Kikuchi J, Morita H, Hattori M, Ohno H. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature. 2011;469:543-547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2050] [Cited by in RCA: 1759] [Article Influence: 117.3] [Reference Citation Analysis (4)] |
| 34. | Wang K, Qi L, Zhao L, Liu J, Guo Y, Zhang C. Degradation of chondroitin sulfate: Mechanism of degradation, influence factors, structure-bioactivity relationship and application. Carbohydr Polym. 2023;301:120361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 35. | Dodd D, Spitzer MH, Van Treuren W, Merrill BD, Hryckowian AJ, Higginbottom SK, Le A, Cowan TM, Nolan GP, Fischbach MA, Sonnenburg JL. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature. 2017;551:648-652. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1203] [Cited by in RCA: 1051] [Article Influence: 116.8] [Reference Citation Analysis (0)] |
| 36. | Egea J, García AG, Verges J, Montell E, López MG. Antioxidant, antiinflammatory and neuroprotective actions of chondroitin sulfate and proteoglycans. Osteoarthritis Cartilage. 2010;18 Suppl 1:S24-S27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 79] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 37. | de Bari L, Scirè A, Minnelli C, Cianfruglia L, Kalapos MP, Armeni T. Interplay among Oxidative Stress, Methylglyoxal Pathway and S-Glutathionylation. Antioxidants (Basel). 2020;10:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 38. | Shamsaddini A, Gillevet PM, Acharya C, Fagan A, Gavis E, Sikaroodi M, McGeorge S, Khoruts A, Albhaisi S, Fuchs M, Sterling RK, Bajaj JS. Impact of Antibiotic Resistance Genes in Gut Microbiome of Patients With Cirrhosis. Gastroenterology. 2021;161:508-521.e7. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 52] [Article Influence: 10.4] [Reference Citation Analysis (0)] |