Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.116074
Revised: December 29, 2025
Accepted: January 27, 2026
Published online: April 27, 2026
Processing time: 171 Days and 0.9 Hours
Metabolic dysfunction-associated steatotic liver disease (MASLD) is a prominent metabolic disease characterized by hepatic steatosis, inflammation, and progres
Core Tip: This review proposes a unifying framework in which a high-fat diet, gut dysbiosis, and oxidative stress form a self-reinforcing pathogenic triad driving metabolic dysfunction-associated steatotic liver disease. We synthesize evidence showing how diet-induced microbial imbalance disrupts intestinal barrier integrity, promotes endotoxemia, and amplifies hepatic oxidative stress, mitochondrial dysfunction, inflammation, and fibrosis. By positioning oxidative stress as a central mechanistic nexus linking diet and microbiota, this review highlights antioxidant-based lifestyle, nutritional, and pharmacological strategies as promising, integrative approaches for prevention and treatment.
- Citation: Zhang ZR, She L, Cao YR, Chen YW. High-fat diet, gut dysbiosis, and oxidative stress: A synergistic triangle in metabolic dysfunction-associated steatotic liver disease pathogenesis. World J Hepatol 2026; 18(4): 116074
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/116074.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.116074
Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly known as nonalcoholic fatty liver disease (NAFLD), is characterized by hepatic steatosis involving more than 5% of hepatocytes in the absence of significant alcohol consumption or other identifiable causes of hepatic fat accumulation. MASLD is further categorized into simple steatosis, which lacks hepatocellular injury, and metabolic dysfunction-associated steatohepatitis (MASH), a more severe phe
A high-fat diet (HFD) is an energy-dense dietary pattern that contributes to insulin resistance (IR), obesity, diabetes, and other metabolic disorders, particularly MASLD[4]. The primary pathogenic feature of MASLD is excessive lipid accumulation in the liver, which serves as a central organ regulating systemic lipid metabolism. Following ingestion, dietary fats are metabolized into triglycerides and assembled into chylomicrons in the intestine. While surrounding tissues such as muscle and adipose tissue can utilize or store the free fatty acids (FFAs), excessive lipid intake leads to elevated FFA levels in the portal circulation and subsequent hepatic accumulation[5]. This lipid overload renders the liver especially vulnerable to diet-induced steatosis and injury.
The gut microbiota comprises a highly complex and diverse community of microorganisms that play a crucial role in maintaining host metabolic homeostasis and other physiological functions[6]. However, this microbial balance can be disrupted by various factors, leading to dysbiosis, typically characterized by a collapse in microbial diversity and alterations in microbial composition[7]. Evidence suggests that patients with MASLD exhibit a distinct gut microbiota profile compared to healthy individuals, including shifts in the abundance of specific microbial genera[8]. This microbial imbalance is increasingly recognized as a contributor to MASLD pathogenesis and progression through mechanisms such as increased intestinal permeability, systemic inflammation, and altered metabolite production[9].
Oxidative stress (OxS) arises from a disrupted balance between oxidants and antioxidants[10]. Reactive oxygen species (ROS), which include free radicals (e.g., superoxide anion and hydroxyl radical) and non-radical species [e.g., hydrogen peroxide (H2O2)], can damage cellular structures if not adequately neutralized[11,12]. The body relies on enzymatic and non-enzymatic antioxidant systems to detoxify ROS and prevent cellular damage[13,14]. Importantly, OxS activates redox-sensitive signaling pathways such as the nuclear factor erythroid 2-related factor 2 (Nrf2), which regulates genes involved in antioxidant defense and detoxification[15]. Under physiological conditions, ROS serve as signaling molecules in various metabolic processes, immune responses, and cell proliferation[11]. However, excessive ROS under OxS conditions lead to pathological modifications of lipids, proteins, and nucleic acids, contributing to cellular damage and the development of diseases including MASLD.
The pathogenesis of MASLD involves both genetic and environmental factors and remains incompletely understood[16]. The earlier “two-hit” hypothesis proposed that hepatic fat accumulation (first hit) sensitizes the liver to OxS and inflammation (second hit)[17,18]. Although this model provided useful insights, it failed to capture the multifactorial complexity of MASLD. A more comprehensive “multiple parallel hits” hypothesis has since emerged, suggesting that MASLD results from the convergence of multiple simultaneous insults, including lipid metabolism disorders, lipotoxicity, OxS, mitochondrial and endoplasmic reticulum (ER) dysfunction, endotoxemia, cytokine alterations, and genetic predisposition[10,19]. Elucidating the interplay among these factors is critical to developing integrative strategies for MASLD prevention and management.
Although HFD, gut dysbiosis, and OxS have each been independently implicated in MASLD, a comprehensive understanding of their interconnections remains lacking. This narrative review synthesizes current evidence to elucidate the triangular and synergistic interactions among these factors in MASLD pathogenesis and to highlight antioxidant-based therapeutic strategies.
We performed a targeted literature search using PubMed/MEDLINE and Google Scholar. The search strategy combined Medical Subject Headings terms and free-text keywords, including but not limited to “MASLD”, “metabolic dysfunction-associated steatotic liver disease”, “NAFLD”, “NASH”, “high-fat diet”, “gut microbiota”, “microbiome”, “gut dysbiosis”, “oxidative stress”, “reactive oxygen species”, “lipotoxicity”, and “inflammation”. Duplicate records were removed prior to screening. Reference lists of these articles were subsequently reviewed, resulting in a total of 194 sources included in this narrative review.
Hepatic fatty acid uptake plays a central role in maintaining triglyceride homeostasis within the liver. Hepatic lipid homeostasis is maintained through the dynamic balance between lipid input, including dietary fatty acids, de novo lipogenesis (DNL), and intracellular lipid stores, and lipid output, such as mitochondrial β-oxidation and export via lipoproteins. Fat accumulation in hepatocytes occurs when lipid influx exceeds efflux[4].
The liver is particularly susceptible to diet-triggered steatosis due to its direct exposure to dietary lipids and carbohydrates via the portal vein. DNL is upregulated in response to elevated carbohydrate and insulin levels, which stimulate key lipogenic transcription factors, including carbohydrate-responsive element-binding protein (ChREBP) and sterol regulatory element-binding protein 1c (SREBP-1c), both of which are involved in gluconeogenesis and lipogenesis[20]. Mitochondrial β-oxidation is the primary pathway for lipid catabolism. While short- and medium-chain fatty acids freely diffuse into mitochondria, long-chain fatty acids require transport by carnitine palmitoyl transferase-1 (CPT-1)[16]. However, HFD significantly suppress CPT-1 messenger RNA expression, impairing fatty acid oxidation[21]. IR further disrupts DNL, adipose tissue lipolysis, and fatty acid export[20]. Collectively, when hepatic lipid accumulation overwhelms physiological regulation, it triggers a cascade of metabolic disturbances that promote the development of MASLD.
Specific dietary components play critical roles in the progression of MASLD. Diets high in fructose is also recognized as a potent driver of MASLD. Fructose metabolism strongly increases protein levels of all DNL enzymes and upregulates lipogenic transcription factors, specifically ChREBP and SREBP-1c, promoting the accumulation of intrahepatic triglycerides[22] and inhibiting fatty acid β-oxidation[23]. As the most abundant dietary antioxidants, polyphenols exert hepatoprotective effects in part via activation of adenosine 5’-monophosphate-activated protein kinase (AMPK) and Nrf2 signaling[24]. A diet deficient in polyphenols promotes the release of pro-inflammatory cytokines and exacerbating liver injury.
Saturated fatty acids (SFAs) and unsaturated fatty acids (UFAs) represent the two major classes of dietary fatty acids. SFAs have been consistently associated with IR, hepatic lipid accumulation, and pro-inflammatory responses[25]. Accumulating evidence suggests that SFAs may activate Toll-like receptor (TLR) 4[26,27], potentially due to their structural similarity to lipopolysaccharide (LPS)[28]. Trans fatty acids (TFAs) are a subclass of UFAs characterized by one or more unconjugated double bonds in the trans configuration. Most TFAs originate from industrial processing [industrial TFAs (iTFAs)], whereas a smaller proportion arises from natural fermentation in the rumen of ruminant animals [ruminant TFAs (rTFAs)][29]. However, limited preclinical evidence indicates that iTFAs, but not rTFAs, adversely affect insulin sensitivity and lipid metabolism and may promote inflammation and stress[30]. The differential biological effects of rTFAs appear to be mediated by distinct signaling pathways, such as activation of peroxisome proliferator-activated receptor-α (PPARα), and warrant further investigation[31].
The gut microbiota is intricately involved in the pathogenesis and progression of MASLD. The interplay between microbial metabolites, immune activation, and endotoxemia, primarily due to intestinal barrier dysfunction, synergistically drives inflammation and OxS in MASLD[9,32].
Gut microbes employ diverse enzymatic systems to produce a wide array of metabolites that influence host metabolism[33]. Among these, short-chain fatty acids (SCFAs) are particularly significant, serving as vital energy sources for colonocytes and playing essential roles in metabolic and immune regulation[34]. For example, butyric acid supports intestinal barrier integrity and inhibits lipogenic transcription factors such as ChREBP and SREBP-1c[35], while propionic acid may exacerbate MASLD by promoting adipogenesis and gluconeogenesis[36]. SCFAs also activate G protein-coupled receptors in the gut and adipose tissues, slowing intestinal motility and enhancing nutrient absorption, which may further contribute to MASLD progression[37].
Bile acids (BAs), the principal constituents of bile, consist of primary BAs (PBAs) synthesized in the liver and secondary BAs (SBAs) produced by gut microbial conversion. PBAs regulate microbial composition by directly suppressing pathogenic bacteria and by activating the intestinal farnesoid X receptor (FXR)[38]. FXR activation enhances glycogen synthesis and inhibits SREBP-1c expression, thereby reducing hepatic fat accumulation[39]. Conversely, a reduction in SBAs impairs FXR signaling, while excessive SBAs may induce DNA damage and ROS generation. Changes in gut microbiota and liver function can further alter BA profiles and their metabolic effects.
Choline metabolism is also closely linked to hepatic lipid regulation. Choline deficiency impairs the synthesis and secretion of very low-density lipoproteins in hepatocytes, leading to hepatic lipid accumulation[40,41]. Choline-derived metabolites such as trimethylamine-N-oxide have been implicated in hepatic inflammation and injury[42]. Other microbial byproducts, including ethanol, acetaldehyde, phenols, ammonia, and related compounds, can also contribute to hepatic inflammation and OxS.
OxS driven by ROS production and chronic inflammation, is now recognized as a central mechanism in the onset and progression of MASLD. A close interplay exists between OxS and mitochondrial dysfunction, as mitochondria are the primary source of intracellular ROS[13,43]. Studies have reported various mitochondrial abnormalities in MASLD, including ultrastructural damage, altered morphology, mitochondrial DNA (mtDNA) mutations and depletion[44], reduced electron transport chain (ETC) activity, and impaired β-oxidation. Lipid metabolic imbalance contributes significantly to mitochondrial impairment. One major fate of hepatic lipids is mitochondrial β-oxidation, which generates energy via the ETC[45]. In MASLD, excessive FFAs overload already compromised mitochondria[46]. The excessive oxidative load and elevated electron flux within the ETC lead to incomplete electron transfer, resulting in electron leakage and increased ROS generation[43,47]. Moreover, FFAs and ROS compromise mitochondrial function by altering inner membrane permeability and inhibiting key components of the ETC, including complexes I and II and adenosine triphosphate (ATP) synthase. Superoxide and H2O2, produced in the mitochondrial inner membrane, interact readily with nearby mtDNA, causing point mutations and deletions.
The ER is another key intracellular source of ROS. ER stress, marked by disruption of protein-folding homeostasis, plays a pivotal role in MASLD pathogenesis. The ER supports proper folding and assembly of nascent proteins, a process that inherently generates ROS through disulfide bond formation[48]. Under stress conditions, such as lipid overload or the accumulation of misfolded proteins, the unfolded protein response (UPR) is activated to restore homeostasis[49]. UPR initiates the expression of ATP-dependent chaperone proteins to refold misfolded proteins, thereby increasing energy demands on already dysfunctional mitochondria. The UPR is mediated by three key transmembrane sensors (PERK, IRE1, and ATF6), which regulate genes involved in antioxidant defense and inflammatory signaling. Prolonged ER stress enhances ROS production by activating pro-apoptotic pathways such as CHOP (regulated by PERK and ATF6), c-Jun N-terminal kinase (via IRE1), and nuclear factor-κB (NF-κB) (via ATF6). Furthermore, PERK-mediated inhibition of Nrf2 impairs antioxidant responses[50-53]. ER stress also disrupts calcium homeostasis, exacerbating mitochondrial dysfunction[54]. Together, these mechanisms lead to ROS-mediated cell damage and apoptosis.
Numerous studies report reduced antioxidant defenses in individuals with MASLD, including decreased levels of superoxide dismutase (SOD), catalase, and glutathione (GSH)[55]. These reductions impair enzymatic detoxification of ROS, intensifying OxS and contributing to hepatic damage and MASLD progression.
Dietary composition profoundly influences the structure and diversity of the gut microbiota. Polyphenol-rich diets have been shown to specifically restore the abundance of beneficial gut microbes such as Lactobacillus and Bifidobacterium, while inhibiting potentially harmful bacteria[56]. In contrast, the Western diet, rich in fats and fructose, reshapes microbial communities by altering nutrient availability and the intestinal environment, promoting the expansion of lipid-adapted bacteria while depleting beneficial species[57,58]. Animal studies indicate that HFD increases the relative abundance of Firmicutes, Escherichia, and Shigella, while reducing Bacteroidetes, Lactobacillus, and Bifidobacterium[59,60]. HFD is also associated with enrichment of LPS-producing bacteria[61] and taurine-metabolizing bacteria[62], thereby altering inflammation and BAs metabolism.
Beyond its effects on microbial composition, a HFD directly impairs intestinal barrier function and increases intestinal permeability[63]. The intestinal barrier, composed of epithelial cells joined by tight junctions and covered by a mucus layer, prevents luminal microbiota from entering host circulation[64]. Studies suggest that HFD compromises this barrier via several mechanisms: (1) Downregulation or aberrant expression of tight junction proteins and abnormal mucus secretion[65]; (2) Chylomicron accumulation between epithelial cells, increasing mechanical stress that disrupts tight junctions or perforates the basal membrane[66,67]; and (3) Activation of mast cells during fat absorption, releasing mediators such as histamine and prostaglandin D2 that are associated with increased intestinal permeability[68]. These disruptions promote the translocation of luminal antigens, leading to enterocyte OxS and apoptosis, and contributing to the “leaky gut” phenotype.
Gut dysbiosis further exacerbates intestinal barrier dysfunction, creating a self-reinforcing pathogenic cycle. The gastrointestinal tract harbors a range of immune cells within gut-associated lymphoid tissue, and disturbances in microbial composition can impair immune regulation and elicit inflammatory responses[69]. Chronic inflammation, mediated by immune cells and cytokines, disrupts tight junctions, injures epithelial cells, and alters the composition of the mucus layer[70-72]. The overgrowth of pathogenic microbes further compromises the protective roles of beneficial bacteria in maintaining barrier integrity[73]. As a result, increased intestinal permeability, or “leaky gut”, permits the translocation of microbial components, toxins, and metabolites into systemic circulation[74,75]. Among these microbial products, LPS, a key structural component of gram-negative bacterial cell walls, plays a pivotal role in triggering endotoxemia[61]. Once in the bloodstream, LPS activates immune cells expressing TLR4, initiating chronic hepatic inflammation[76]. Kupffer cells, which express high levels of TLR4 in the liver, are central mediators of this response, producing cytokines, chemokines, and ROS[77,78]. The LPS-TLR4 interaction activates downstream signaling cascades, including NF-κB, leading to the secretion of pro-inflammatory cytokines[79,80]. This process further damages intestinal barrier function and perpetuates MASLD progression. Tumor necrosis factor-alpha (TNF-α), a major cytokine induced by LPS-TLR4 signaling, plays a critical role in hepatotoxicity, inflammation, and IR[81]. These inflammatory and oxidative pathways form a vicious cycle that exacerbates hepatic injury, promotes gut barrier dysfunction, and sustains gut dysbiosis.
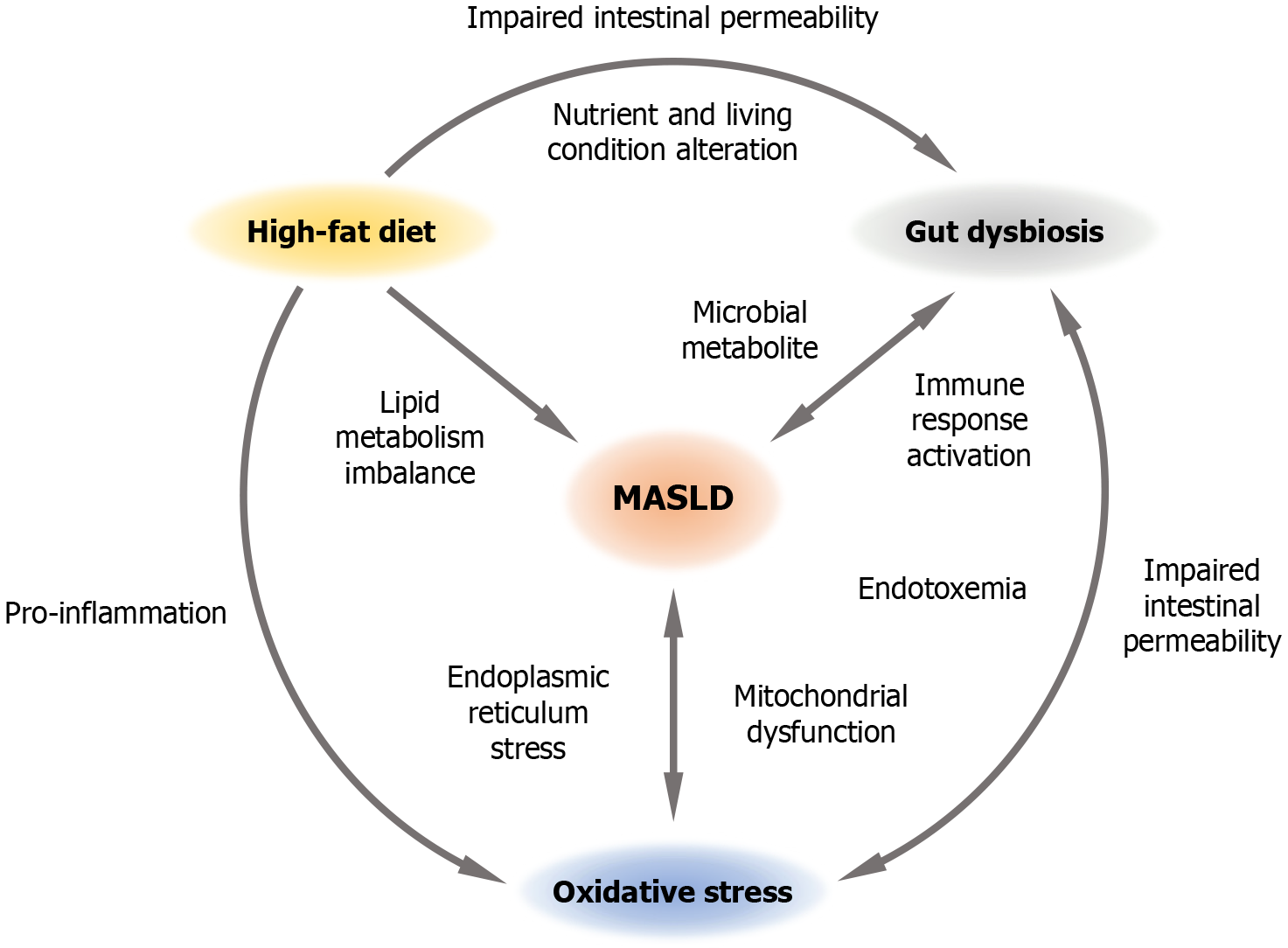
Within the liver, chronic exposure to excess FFAs, inflammatory mediators, and OxS promotes lipid overload and facilitates the accumulation of toxic lipid intermediates, including ceramides and diacylglycerols (DAGs)[16,19]. Reduced oxidative capacity further contributes to intracellular triglyceride accumulation, which is highly susceptible to ROS-induced lipid peroxidation. Lipotoxic byproducts such as malondialdehyde (MDA) and 4-hydroxy-2-nonenal (4-HNE) further exacerbate oxidative injury[82]. These lipid intermediates cause extensive damage to cellular and subcellular structures, disrupt macromolecular integrity, and impair organelle function[83]. Ceramides inhibit mitochondrial β-oxidation and enhance intramitochondrial ROS generation[84], whereas DAGs and ceramides activate pro-inflammatory and pro-apoptotic signaling pathways, including NF-κB and c-Jun N-terminal kinase[85]. In parallel, MDA and 4-HNE inhibit cytochrome c oxidase, uncouple ETC complex II, destabilize mitochondrial membranes, and induce mtDNA mutations[86]. Subsequent leakage of damaged mtDNA into the cytosol activates innate immune receptors and downstream inflammatory cascades[87]. These mitochondrial abnormalities impair β-oxidation, enhance pro-inflammatory cytokine production, exacerbate hepatocellular triglyceride accumulation, and diminish ROS clearance, thereby establishing a self-amplifying cycle of OxS and mitochondrial dysfunction that accelerates liver injury, inflammation, fibrosis, and disease progression (Figure 1).
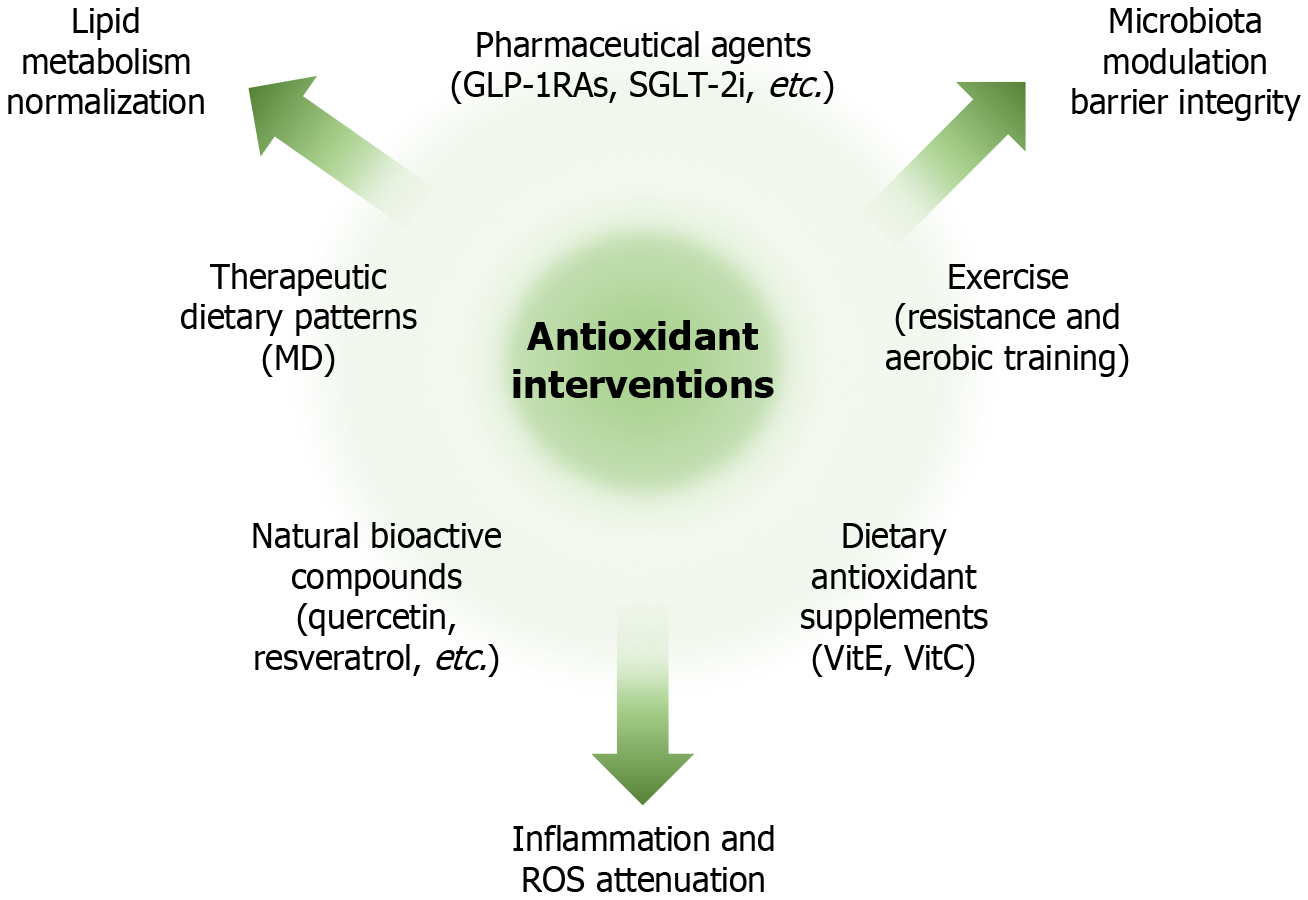
Current management strategies for MASLD primarily involve lifestyle modifications, antioxidant interventions, and combination therapies. Among these, antioxidants, including dietary supplements, natural bioactive compounds, and pharmacological agents, have shown promising potential in mitigating MASLD by targeting OxS and restoring redox homeostasis. In this section, we provide a comprehensive overview of antioxidant-based therapeutic approaches and their underlying mechanisms in the context of MASLD (Figure 2).
Lifestyle interventions, including exercise and dietary changes, remain the cornerstone of MASLD management.
Exercise: Exercise is widely recognized as a cost-effective, non-pharmacological intervention with both therapeutic and preventive benefits in metabolic diseases such as MASLD[88,89]. In addition to improving liver health, regular physical activity mitigates key risk factors for cardiovascular disease and hypertension[90]. Substantial clinical evidence supports the effectiveness of various forms of exercise, including both resistance and aerobic training. For instance, 12 weeks of high-intensity interval training or aerobic exercise has been shown to reduce serum triglyceride levels, liver enzyme activity, and biomarkers of inflammation and OxS[91,92]. Similarly, a 12-week resistance training regimen involving push-ups and squats was associated with delayed MASLD progression[93]. Current guidelines recommend that patients with MASLD engage in at least 150 minutes of moderate-intensity or 75 minutes of vigorous-intensity physical activity per week[94].
The hepatoprotective effects of exercise in MASLD are mediated through several mechanisms, including enhanced mitochondrial β-oxidation of fatty acids, suppression of lipogenesis, and reduction of intrahepatic fat accumulation[95,96]. Exercise also stimulates the activity of endogenous antioxidant enzymes and reduces OxS, inflammation, and mitochondria-mediated apoptosis[97,98]. Studies have shown that exercise activates mitogen-activated protein kinase, which in turn induces the NF-κB signaling pathway, leading to upregulation of antioxidant defense enzymes[99-101]. Moreover, exercise in combination with cold exposure has been reported to suppress the transforming growth factor-β/SMAD2 signaling pathway and activate the fibroblast growth factor (FGF) 21-β-klotho/FGF receptor 1 axis, thereby improving lipid metabolism and attenuating hepatic fibrosis and injury in MASLD models[102,103].
Therapeutic dietary patterns: A meta-analysis reported that adherence to Western dietary patterns is associated with a 56% increased risk of MASLD, underscoring the critical role of dietary habits in disease development and progression[104]. Dietary interventions that reduce carbohydrate and free sugar intake, and consequently restrict total caloric intake, have demonstrated hepatoprotective effects, making them a promising strategy for MASLD management[105]. Among these, caloric restriction remains the most widely adopted dietary approach.
Compared to diets high in saturated fats and sucrose but low in dietary fiber, the Mediterranean diet (MD), characterized by high consumption of fruits, vegetables, fish, whole grains, nuts, seeds, and olive oil, has demonstrated superior health benefits[106]. The European Association for the Study of the Liver-European Association for the Study of Diabetes-European Association for the Study of Obesity Clinical Practice Guidelines strongly recommend MD as a therapeutic option for MASLD[1]. Clinical studies have shown that MD reduces hepatic steatosis and fibrosis, alleviates IR and hyperlipidemia, promotes weight loss[107-109], and provides cardiovascular protection, thereby improving overall clinical outcomes in MASLD. Intervention studies have reported significant reductions in body weight, blood pressure, and cardiometabolic risk factors following MD adoption. Similarly, clinical trials in adolescents with obesity and MASLD have shown that 12 weeks of MD intake leads to improvements in body mass index, fat mass, hepatic steatosis, inflammatory and OxS markers, as well as reductions in serum transaminase levels[110-112].
The MD is a rich source of polyphenols and UFAs. Polyphenols are widely distributed in fruits, tea, berries, coffee, red wine, and dark chocolate and can be broadly classified into flavonoids (e.g., flavones, anthocyanins) and non-flavonoid compounds (e.g., phenolic acids, hydroxycinnamic acids, lignans, and tannins)[113]. Accumulating evidence indicates that polyphenols attenuate OxS, enhance fatty acid β-oxidation, improve IR, modulate DNL, and influence gut microbiota composition[24]. The therapeutic potential of representative flavonoids, including resveratrol, quercetin, and curcumin, in MASLD will be discussed in subsequent sections.
UFAs are structurally classified into polyunsaturated fatty acids (PUFAs) and monounsaturated fatty acids (MUFAs). The two principal PUFA families are the n-3 and n-6 classes. n-3 PUFAs, predominantly derived from marine and plant sources, exert hepatoprotective effects. Mechanistically, n-3 PUFAs alleviate hepatic steatosis by suppressing DNL while concomitantly activating PPARs to enhance fatty acid oxidation[114]. In contrast, the role of n-6 PUFAs, which are abundant in vegetable oils, is more complex, as an excessively high dietary n-6/n-3 ratio has been associated with a pro-inflammatory milieu[115,116]. MUFAs, primarily obtained from olive oil, avocados, and nuts, constitute a cornerstone of the MD. MUFAs intake has been consistently associated with improved insulin sensitivity and marked reductions in hepatic fat content[117].
In addition to MD, several emerging dietary strategies are being investigated for MASLD management. The ketogenic diet (KD), characterized by extremely low carbohydrate and protein intake but high fat content[118], has garnered attention for its effects on liver mitochondrial metabolism and redox balance[119]. However, potential cardiovascular and renal complications associated with KD, as reported in animal and clinical studies, remain a concern[120,121]. The long-term sustainability and safety of KD in individuals with MASLD require further investigation. Low-fat diets have also been shown to improve hepatic steatosis, liver enzymes, and IR in both adult and pediatric MASLD populations[122], although their benefits appear comparable or slightly inferior to those of MD. High-protein diets, whether animal- or plant-based, have demonstrated effectiveness in reducing hepatic fat accumulation, IR, and systemic inflammation[123]. Nonetheless, further long-term, controlled studies are needed to evaluate the safety, efficacy, and sustainability of these dietary interventions in MASLD.
Vitamin C (VitC) and vitamin E (VitE) are classical antioxidants known to attenuate OxS by scavenging ROS. Several studies have reported beneficial effects of VitE and VitC supplementation in patients with MASLD[124-126].
VitE, a lipid-soluble vitamin primarily stored in the liver and adipose tissue, exerts antioxidant effects through multiple mechanisms. Experimental evidence suggests that the protective actions of VitE and its homologues may be attributed to the suppression of OxS and inflammation, inhibition of hepatic DNL, activation of the Nrf2 signaling pathway, protection against cellular injury, and normalization of lipid metabolism[127-130]. The PIVENS trial demonstrated that VitE supplementation (800 IU/day) significantly improved the NAFLD activity score, liver enzyme levels, steatosis, lobular inflammation, and hepatocellular ballooning although it did not improve fibrosis when compared to pioglitazone or placebo groups[131]. Additional clinical studies have confirmed that VitE therapy reduces intrahepatic lipid accumulation, slows MASLD progression, and improves liver histology[132-134]. Importantly, large-scale prevention trials, such as the SELECT study, have demonstrated a significant increase in prostate cancer risk associated with high-dose VitE supplementation, underscoring important safety concerns regarding its therapeutic use[135].
VitC, an essential water-soluble vitamin obtained exclusively through the diet, has shown variable therapeutic effects in MASLD. Some studies suggest that VitC supplementation exerts hepatoprotective effects, including improvements in liver histology and reductions in serum transaminase levels[136,137]. The beneficial actions of VitC may involve modulation of gut microbiota and BA metabolism[138], as well as attenuation of inflammation and apoptosis, potentially mediated by upregulation of PPARα and genes involved in β-oxidation[139,140].
Accumulating evidence from preclinical models has highlighted the therapeutic potential of natural bioactive compounds in the treatment of liver diseases, including MASLD[141,142]. However, results from human studies remain inconsistent, likely owing to rapid metabolism, limited systemic bioavailability, small sample sizes, heterogeneous patient populations, and variability in treatment duration. Consequently, high-dose supplementation of natural bioactive compounds remains controversial, and well-designed, large-scale clinical trials are required to validate their efficacy and elucidate the un
Quercetin (3,3’,4’,5,6-pentahydroxyflavone), a widely distributed dietary flavonoid found in fruits and vegetables, exhibits strong antioxidant and anti-inflammatory properties[143]. Preclinical studies suggest that quercetin ameliorates MASLD by attenuating endotoxemia (via TLR4/NF-κB)[144], reducing inflammation (via TNF-α)[145], restoring redox homeostasis[146-148], and modulating lipid metabolism (via regulation of DNL and β-oxidation)[149]. Although a randomized, double-blind, placebo-controlled crossover trial demonstrated that 12 weeks of quercetin supplementation significantly reduced intrahepatic lipid content[150], some animal studies revolved possible critical safety aspects involving oral quercetin application like nephrotoxicity and tumor promotion[151].
Resveratrol (3,5,4’-trihydroxystilbene), a natural polyphenol present in grapes, berries, peanuts, and red wine, has been widely studied for its therapeutic potential in metabolic disorders[152]. In animal models of MASLD, resveratrol improved glucose and lipid metabolism, suppressed inflammation, and reduced OxS[153,154]. These effects are thought to be mediated through activation of Nrf2, silent information regulator 1 (SIRT1), and protein kinase A/AMPK/PPARα pathways[155-157]. Resveratrol has also been shown to promote mitochondrial biogenesis and enhance expression of mitochondrial respiratory chain proteins[158]. While some clinical trials in MASLD patients have reported reductions in hepatic steatosis, liver enzymes, and inflammatory markers[159,160], other studies have failed to confirm consistent therapeutic benefit[161-163].
Curcumin, a polyphenolic compound extracted from the rhizome of Curcuma longa, is well known for its potent antioxidant, anti-inflammatory, and hepatoprotective properties[164,165]. Both preclinical and clinical studies have reported that curcumin significantly improves hepatic lipid accumulation, oxidative damage, inflammation, and fibrogenesis by modulating antioxidant enzyme activity (e.g., SOD, PPARs), and regulating immune cell infiltration[166].
Silybin, the primary bioactive constituent of silymarin extracted from Silybum marianum[167], possesses antioxidant, anti-fibrotic, and anti-inflammatory effects[168]. Recent preclinical and clinical studies have demonstrated that silybin can improve hyperlipidemia, hepatic fibrosis, and inflammation in models of MASH, primarily through activation of key signaling pathways, such as SIRT1/AMPK, nicotinamide adenine dinucleotide/SIRT2, and NF-κB[169-172].
Glucagon-like peptide-1 (GLP-1) receptor agonists (GLP-1RAs), initially developed for the treatment of type 2 diabetes mellitus (T2DM), have also demonstrated considerable promise in MASLD[173]. Agents such as liraglutide and semaglutide have been shown to reduce hepatic steatosis and IR, in part through enhanced β-oxidation, and to promote histological resolution of steatohepatitis in patients with MASLD[174,175]. GLP-1 receptors are expressed on immune cells, indicating that GLP-1RAs may exert immunomodulatory effects, including upregulation of anti-inflammatory cytokines [e.g., interleukin (IL)-10] and suppression of pro-inflammatory mediators such as TNF-α, IL-6, and IL-1β[176-179]. In addition, liraglutide has been shown to activate Nrf2 signaling, thereby mitigating lipotoxicity and OxS[180]. Clinically, the LEAN trial (liraglutide 1.8 mg daily for 48 weeks) demonstrated resolution of MASH in 39% of patients receiving liraglutide compared with 9% in the placebo group, along with reduced fibrosis progression[181]. Similarly, the phase 3 ESSENCE trial reported that semaglutide (2.4 mg weekly for 72 weeks) significantly increased rates of steatohepatitis resolution and fibrosis improvement compared with placebo[182]. Based on these findings, the United States Food and Drug Administration approved semaglutide 2.4 mg for adults with non-cirrhotic MASH and moderate-to-advanced fibrosis (stages F2-F3), in combination with diet and physical activity. Furthermore, tirzepatide, a dual GLP-1/glucose-dependent insulinotropic polypeptide receptor agonist, has shown robust efficacy in MASLD/MASH, with the phase 2 SYNERGY-NASH trial demonstrating substantially higher rates of MASH resolution without worsening of fibrosis compared with placebo[183].
Sodium-glucose cotransporter-2 inhibitors (SGLT-2i) (e.g., dapagliflozin and empagliflozin) confer both metabolic and hepatic benefits in patients with T2DM and MASLD[173]. Mechanistically, SGLT-2i enhance fatty acid β-oxidation and insulin sensitivity, modulate gut microbiota, and attenuate inflammation, OxS, lipogenesis, and fibrosis[184]. Evidence from randomized controlled trials and meta-analyses demonstrates significant reductions in liver fat content, as assessed by magnetic resonance imaging-proton density fat fraction (MRI-PDFF), compared with control interventions[185]. For instance, the E-LIFT trial (empagliflozin 10 mg daily for 20 weeks) reported a significant reduction in MRI-PDFF relative to placebo[186].
The rationale for antioxidant supplementation in MASLD is supported by consistent evidence showing reduced hepatic antioxidant capacity in affected individuals[187]. GSH, a critical intracellular antioxidant, plays an essential role in redox balance and detoxification processes[188]. A pilot study in Japanese patients with MASLD reported that oral GSH administration (300 mg/day for 4 months) significantly reduced alanine aminotransferase levels and hepatic steatosis[189]. N-acetylcysteine (NAC), a thiol-containing compound and GSH precursor, exerts both antioxidant and anti-inflammatory effects by replenishing intracellular GSH levels and enhancing GSH reductase activity. Findings from both preclinical and clinical studies suggest that NAC improves lipid metabolism, reduces liver inflammation, and attenuates steatosis in MASLD[190-192].
Pirfenidone, an anti-fibrotic agent approved for the treatment of idiopathic pulmonary fibrosis and other fibrotic disorders[193], has also demonstrated therapeutic potential in experimental models of MASLD. Its hepatoprotective effects are thought to involve attenuation of OxS and inflammation through activation of Nrf2 signaling and inhibition of the NF-κB pathway[194].
The pathogenesis of MASLD is multifactorial and involves the disruption of numerous physiological homeostatic mechanisms. A HFD is a well-established risk factor, and its deleterious effects are largely mediated through alterations in gut microbiota and induction of OxS. Gut dysbiosis not only disrupts microbial metabolite profiles but also impairs intestinal barrier integrity, promotes endotoxemia, and activates pro-inflammatory signaling pathways, thereby exacerbating hepatic OxS and metabolic disturbances. OxS, in particularly, plays a central role in MASLD progression. HFD-induced lipid overload and microbial imbalance contribute to redox dysregulation and increased production of ROS and lipotoxic intermediates within hepatocytes. This oxidative burden results in cellular macromolecule damage, hepatocellular inflammation, and fibrogenesis. Together, HFD, gut dysbiosis, and OxS form a synergistic triad that drives the progression of MASLD to more advanced and clinically significant stages. Recent experimental studies have underscored the therapeutic potential of targeting OxS in MASLD. Natural compounds such as quercetin and resveratrol have shown promise due to their potent antioxidant properties. Pharmaceutical agents, including GLP-1RAs and SGLT-2i have also demonstrated beneficial effects through modulation of redox and inflammatory pathways. In addition, lifestyle in
| 1. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1278] [Article Influence: 639.0] [Reference Citation Analysis (2)] |
| 2. | Younossi ZM, Kalligeros M, Henry L. Epidemiology of metabolic dysfunction-associated steatotic liver disease. Clin Mol Hepatol. 2025;31:S32-S50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 511] [Cited by in RCA: 499] [Article Influence: 499.0] [Reference Citation Analysis (0)] |
| 3. | Chan WK, Chuah KH, Rajaram RB, Lim LL, Ratnasingam J, Vethakkan SR. Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): A State-of-the-Art Review. J Obes Metab Syndr. 2023;32:197-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 617] [Cited by in RCA: 527] [Article Influence: 175.7] [Reference Citation Analysis (10)] |
| 4. | Malesza IJ, Malesza M, Walkowiak J, Mussin N, Walkowiak D, Aringazina R, Bartkowiak-Wieczorek J, Mądry E. High-Fat, Western-Style Diet, Systemic Inflammation, and Gut Microbiota: A Narrative Review. Cells. 2021;10:3164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 648] [Cited by in RCA: 546] [Article Influence: 109.2] [Reference Citation Analysis (4)] |
| 5. | Fielding B. Tracing the fate of dietary fatty acids: metabolic studies of postprandial lipaemia in human subjects. Proc Nutr Soc. 2011;70:342-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 62] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 6. | Hooper LV, Gordon JI. Commensal host-bacterial relationships in the gut. Science. 2001;292:1115-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1872] [Cited by in RCA: 1537] [Article Influence: 61.5] [Reference Citation Analysis (3)] |
| 7. | Jiang H, Ling Z, Zhang Y, Mao H, Ma Z, Yin Y, Wang W, Tang W, Tan Z, Shi J, Li L, Ruan B. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav Immun. 2015;48:186-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1952] [Cited by in RCA: 1676] [Article Influence: 152.4] [Reference Citation Analysis (6)] |
| 8. | Boursier J, Mueller O, Barret M, Machado M, Fizanne L, Araujo-Perez F, Guy CD, Seed PC, Rawls JF, David LA, Hunault G, Oberti F, Calès P, Diehl AM. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology. 2016;63:764-775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1277] [Cited by in RCA: 1148] [Article Influence: 114.8] [Reference Citation Analysis (5)] |
| 9. | Jadhav PA, Thomas AB, Nanda RK, Chitlange SS. Unveiling the role of gut dysbiosis in non-alcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2023;35:1324-1333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 10. | Takaki A, Kawai D, Yamamoto K. Multiple hits, including oxidative stress, as pathogenesis and treatment target in non-alcoholic steatohepatitis (NASH). Int J Mol Sci. 2013;14:20704-20728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 380] [Cited by in RCA: 349] [Article Influence: 26.8] [Reference Citation Analysis (1)] |
| 11. | Forrester SJ, Kikuchi DS, Hernandes MS, Xu Q, Griendling KK. Reactive Oxygen Species in Metabolic and Inflammatory Signaling. Circ Res. 2018;122:877-902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1782] [Cited by in RCA: 1526] [Article Influence: 190.8] [Reference Citation Analysis (8)] |
| 12. | van der Vliet A, Janssen-Heininger YMW, Anathy V. Oxidative stress in chronic lung disease: From mitochondrial dysfunction to dysregulated redox signaling. Mol Aspects Med. 2018;63:59-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 128] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 13. | Figueira TR, Barros MH, Camargo AA, Castilho RF, Ferreira JC, Kowaltowski AJ, Sluse FE, Souza-Pinto NC, Vercesi AE. Mitochondria as a source of reactive oxygen and nitrogen species: from molecular mechanisms to human health. Antioxid Redox Signal. 2013;18:2029-2074. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 304] [Cited by in RCA: 332] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 14. | Zhang L, Wang X, Cueto R, Effi C, Zhang Y, Tan H, Qin X, Ji Y, Yang X, Wang H. Biochemical basis and metabolic interplay of redox regulation. Redox Biol. 2019;26:101284. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 235] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 15. | Smolková K, Mikó E, Kovács T, Leguina-Ruzzi A, Sipos A, Bai P. Nuclear Factor Erythroid 2-Related Factor 2 in Regulating Cancer Metabolism. Antioxid Redox Signal. 2020;33:966-997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 79] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 16. | Chen Z, Yu Y, Cai J, Li H. Emerging Molecular Targets for Treatment of Nonalcoholic Fatty Liver Disease. Trends Endocrinol Metab. 2019;30:903-914. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 107] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 17. | Rolo AP, Teodoro JS, Palmeira CM. Role of oxidative stress in the pathogenesis of nonalcoholic steatohepatitis. Free Radic Biol Med. 2012;52:59-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 791] [Cited by in RCA: 730] [Article Influence: 52.1] [Reference Citation Analysis (4)] |
| 18. | Serviddio G, Bellanti F, Vendemiale G. Free radical biology for medicine: learning from nonalcoholic fatty liver disease. Free Radic Biol Med. 2013;65:952-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 233] [Cited by in RCA: 214] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 19. | Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018;24:908-922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3652] [Cited by in RCA: 3330] [Article Influence: 416.3] [Reference Citation Analysis (7)] |
| 20. | Samuel VT, Shulman GI. Nonalcoholic Fatty Liver Disease as a Nexus of Metabolic and Hepatic Diseases. Cell Metab. 2018;27:22-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 467] [Cited by in RCA: 574] [Article Influence: 71.8] [Reference Citation Analysis (4)] |
| 21. | Lian CY, Zhai ZZ, Li ZF, Wang L. High fat diet-triggered non-alcoholic fatty liver disease: A review of proposed mechanisms. Chem Biol Interact. 2020;330:109199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 218] [Article Influence: 36.3] [Reference Citation Analysis (0)] |
| 22. | Softic S, Cohen DE, Kahn CR. Role of Dietary Fructose and Hepatic De Novo Lipogenesis in Fatty Liver Disease. Dig Dis Sci. 2016;61:1282-1293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 582] [Cited by in RCA: 511] [Article Influence: 51.1] [Reference Citation Analysis (4)] |
| 23. | Herman MA, Samuel VT. The Sweet Path to Metabolic Demise: Fructose and Lipid Synthesis. Trends Endocrinol Metab. 2016;27:719-730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 174] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 24. | Abenavoli L, Larussa T, Corea A, Procopio AC, Boccuto L, Dallio M, Federico A, Luzza F. Dietary Polyphenols and Non-Alcoholic Fatty Liver Disease. Nutrients. 2021;13:494. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 95] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 25. | Kennedy A, Martinez K, Chuang CC, LaPoint K, McIntosh M. Saturated fatty acid-mediated inflammation and insulin resistance in adipose tissue: mechanisms of action and implications. J Nutr. 2009;139:1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 355] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 26. | Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS. TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest. 2006;116:3015-3025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3051] [Cited by in RCA: 2818] [Article Influence: 140.9] [Reference Citation Analysis (2)] |
| 27. | Ohashi K, Shibata R, Murohara T, Ouchi N. Role of anti-inflammatory adipokines in obesity-related diseases. Trends Endocrinol Metab. 2014;25:348-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 248] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 28. | Schaeffler A, Gross P, Buettner R, Bollheimer C, Buechler C, Neumeier M, Kopp A, Schoelmerich J, Falk W. Fatty acid-induced induction of Toll-like receptor-4/nuclear factor-kappaB pathway in adipocytes links nutritional signalling with innate immunity. Immunology. 2009;126:233-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 280] [Article Influence: 15.6] [Reference Citation Analysis (3)] |
| 29. | Stender S, Astrup A, Dyerberg J. Ruminant and industrially produced trans fatty acids: health aspects. Food Nutr Res. 2008;52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 107] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 30. | Oteng AB, Kersten S. Mechanisms of Action of trans Fatty Acids. Adv Nutr. 2020;11:697-708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 167] [Article Influence: 27.8] [Reference Citation Analysis (0)] |
| 31. | Jaudszus A, Jahreis G, Schlörmann W, Fischer J, Kramer R, Degen C, Rohrer C, Roth A, Gabriel H, Barz D, Gruen M. Vaccenic acid-mediated reduction in cytokine production is independent of c9,t11-CLA in human peripheral blood mononuclear cells. Biochim Biophys Acta. 2012;1821:1316-1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 32. | Arslan N. Obesity, fatty liver disease and intestinal microbiota. World J Gastroenterol. 2014;20:16452-16463. [PubMed] [DOI] [Full Text] |
| 33. | Fang J, Yu CH, Li XJ, Yao JM, Fang ZY, Yoon SH, Yu WY. Gut dysbiosis in nonalcoholic fatty liver disease: pathogenesis, diagnosis, and therapeutic implications. Front Cell Infect Microbiol. 2022;12:997018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 151] [Article Influence: 37.8] [Reference Citation Analysis (1)] |
| 34. | Rauf A, Khalil AA, Rahman UU, Khalid A, Naz S, Shariati MA, Rebezov M, Urtecho EZ, de Albuquerque RDDG, Anwar S, Alamri A, Saini RK, Rengasamy KRR. Recent advances in the therapeutic application of short-chain fatty acids (SCFAs): An updated review. Crit Rev Food Sci Nutr. 2022;62:6034-6054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 117] [Article Influence: 23.4] [Reference Citation Analysis (6)] |
| 35. | Park JH, Kotani T, Konno T, Setiawan J, Kitamura Y, Imada S, Usui Y, Hatano N, Shinohara M, Saito Y, Murata Y, Matozaki T. Promotion of Intestinal Epithelial Cell Turnover by Commensal Bacteria: Role of Short-Chain Fatty Acids. PLoS One. 2016;11:e0156334. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 202] [Article Influence: 20.2] [Reference Citation Analysis (6)] |
| 36. | Liu W, Luo X, Tang J, Mo Q, Zhong H, Zhang H, Feng F. A bridge for short-chain fatty acids to affect inflammatory bowel disease, type 1 diabetes, and non-alcoholic fatty liver disease positively: by changing gut barrier. Eur J Nutr. 2021;60:2317-2330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (3)] |
| 37. | Lu Y, Fan C, Li P, Lu Y, Chang X, Qi K. Short Chain Fatty Acids Prevent High-fat-diet-induced Obesity in Mice by Regulating G Protein-coupled Receptors and Gut Microbiota. Sci Rep. 2016;6:37589. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 557] [Cited by in RCA: 483] [Article Influence: 48.3] [Reference Citation Analysis (4)] |
| 38. | Fiorucci S, Distrutti E. The Pharmacology of Bile Acids and Their Receptors. Handb Exp Pharmacol. 2019;256:3-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 92] [Article Influence: 13.1] [Reference Citation Analysis (3)] |
| 39. | Yang ZX, Shen W, Sun H. Effects of nuclear receptor FXR on the regulation of liver lipid metabolism in patients with non-alcoholic fatty liver disease. Hepatol Int. 2010;4:741-748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 190] [Cited by in RCA: 183] [Article Influence: 11.4] [Reference Citation Analysis (2)] |
| 40. | Ding N, Wang K, Jiang H, Yang M, Zhang L, Fan X, Zou Q, Yu J, Dong H, Cheng S, Xu Y, Liu J. AGK regulates the progression to NASH by affecting mitochondria complex I function. Theranostics. 2022;12:3237-3250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 41. | Zhang J, Zhao Y, Wang S, Li G, Xu K. CREBH alleviates mitochondrial oxidative stress through SIRT3 mediating deacetylation of MnSOD and suppression of Nlrp3 inflammasome in NASH. Free Radic Biol Med. 2022;190:28-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 42. | Li X, Su C, Jiang Z, Yang Y, Zhang Y, Yang M, Zhang X, Du Y, Zhang J, Wang L, Jiang J, Hong B. Berberine attenuates choline-induced atherosclerosis by inhibiting trimethylamine and trimethylamine-N-oxide production via manipulating the gut microbiome. NPJ Biofilms Microbiomes. 2021;7:36. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 43. | Begriche K, Massart J, Robin MA, Bonnet F, Fromenty B. Mitochondrial adaptations and dysfunctions in nonalcoholic fatty liver disease. Hepatology. 2013;58:1497-1507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 519] [Cited by in RCA: 481] [Article Influence: 37.0] [Reference Citation Analysis (0)] |
| 44. | Seki S, Kitada T, Yamada T, Sakaguchi H, Nakatani K, Wakasa K. In situ detection of lipid peroxidation and oxidative DNA damage in non-alcoholic fatty liver diseases. J Hepatol. 2002;37:56-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 441] [Cited by in RCA: 398] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 45. | Vercesi AE, Castilho RF, Kowaltowski AJ, de Oliveira HCF, de Souza-Pinto NC, Figueira TR, Busanello ENB. Mitochondrial calcium transport and the redox nature of the calcium-induced membrane permeability transition. Free Radic Biol Med. 2018;129:1-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 104] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 46. | Nguyen P, Leray V, Diez M, Serisier S, Le Bloc'h J, Siliart B, Dumon H. Liver lipid metabolism. J Anim Physiol Anim Nutr (Berl). 2008;92:272-283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 806] [Cited by in RCA: 687] [Article Influence: 38.2] [Reference Citation Analysis (1)] |
| 47. | Zorov DB, Juhaszova M, Sollott SJ. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev. 2014;94:909-950. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4705] [Cited by in RCA: 4043] [Article Influence: 336.9] [Reference Citation Analysis (0)] |
| 48. | Shimizu Y, Hendershot LM. Oxidative folding: cellular strategies for dealing with the resultant equimolar production of reactive oxygen species. Antioxid Redox Signal. 2009;11:2317-2331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 131] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 49. | Malhotra JD, Kaufman RJ. The endoplasmic reticulum and the unfolded protein response. Semin Cell Dev Biol. 2007;18:716-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 841] [Cited by in RCA: 783] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 50. | Song B, Scheuner D, Ron D, Pennathur S, Kaufman RJ. Chop deletion reduces oxidative stress, improves beta cell function, and promotes cell survival in multiple mouse models of diabetes. J Clin Invest. 2008;118:3378-3389. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 586] [Cited by in RCA: 574] [Article Influence: 31.9] [Reference Citation Analysis (4)] |
| 51. | Okada K, Warabi E, Sugimoto H, Horie M, Gotoh N, Tokushige K, Hashimoto E, Utsunomiya H, Takahashi H, Ishii T, Yamamoto M, Shoda J. Deletion of Nrf2 leads to rapid progression of steatohepatitis in mice fed atherogenic plus high-fat diet. J Gastroenterol. 2013;48:620-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 73] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 52. | van Galen P, Kreso A, Mbong N, Kent DG, Fitzmaurice T, Chambers JE, Xie S, Laurenti E, Hermans K, Eppert K, Marciniak SJ, Goodall JC, Green AR, Wouters BG, Wienholds E, Dick JE. The unfolded protein response governs integrity of the haematopoietic stem-cell pool during stress. Nature. 2014;510:268-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 298] [Article Influence: 24.8] [Reference Citation Analysis (0)] |
| 53. | Lebeaupin C, Vallée D, Hazari Y, Hetz C, Chevet E, Bailly-Maitre B. Endoplasmic reticulum stress signalling and the pathogenesis of non-alcoholic fatty liver disease. J Hepatol. 2018;69:927-947. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 819] [Cited by in RCA: 733] [Article Influence: 91.6] [Reference Citation Analysis (3)] |
| 54. | Deniaud A, Sharaf el dein O, Maillier E, Poncet D, Kroemer G, Lemaire C, Brenner C. Endoplasmic reticulum stress induces calcium-dependent permeability transition, mitochondrial outer membrane permeabilization and apoptosis. Oncogene. 2008;27:285-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 500] [Cited by in RCA: 474] [Article Influence: 26.3] [Reference Citation Analysis (3)] |
| 55. | García-Ruiz C, Morales A, Ballesta A, Rodés J, Kaplowitz N, Fernández-Checa JC. Effect of chronic ethanol feeding on glutathione and functional integrity of mitochondria in periportal and perivenous rat hepatocytes. J Clin Invest. 1994;94:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 166] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 56. | Wang X, Qi Y, Zheng H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants (Basel). 2022;11:1212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 218] [Article Influence: 54.5] [Reference Citation Analysis (0)] |
| 57. | Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30:492-506. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3319] [Cited by in RCA: 2716] [Article Influence: 452.7] [Reference Citation Analysis (6)] |
| 58. | Li Q, Rempel JD, Yang J, Minuk GY. The Effects of Pathogen-Associated Molecular Patterns on Peripheral Blood Monocytes in Patients with Non-alcoholic Fatty Liver Disease. J Clin Exp Hepatol. 2022;12:808-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 59. | Zhang M, Yang XJ. Effects of a high fat diet on intestinal microbiota and gastrointestinal diseases. World J Gastroenterol. 2016;22:8905-8909. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 127] [Cited by in RCA: 120] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 60. | Hamamah S, Iatcu OC, Covasa M. Dietary Influences on Gut Microbiota and Their Role in Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Nutrients. 2024;17:143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 25] [Article Influence: 12.5] [Reference Citation Analysis (15)] |
| 61. | Cani PD, Amar J, Iglesias MA, Poggi M, Knauf C, Bastelica D, Neyrinck AM, Fava F, Tuohy KM, Chabo C, Waget A, Delmée E, Cousin B, Sulpice T, Chamontin B, Ferrières J, Tanti JF, Gibson GR, Casteilla L, Delzenne NM, Alessi MC, Burcelin R. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56:1761-1772. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5365] [Cited by in RCA: 4782] [Article Influence: 251.7] [Reference Citation Analysis (4)] |
| 62. | Natividad JM, Lamas B, Pham HP, Michel ML, Rainteau D, Bridonneau C, da Costa G, van Hylckama Vlieg J, Sovran B, Chamignon C, Planchais J, Richard ML, Langella P, Veiga P, Sokol H. Bilophila wadsworthia aggravates high fat diet induced metabolic dysfunctions in mice. Nat Commun. 2018;9:2802. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 482] [Cited by in RCA: 419] [Article Influence: 52.4] [Reference Citation Analysis (5)] |
| 63. | Rohr MW, Narasimhulu CA, Rudeski-Rohr TA, Parthasarathy S. Negative Effects of a High-Fat Diet on Intestinal Permeability: A Review. Adv Nutr. 2020;11:77-91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 650] [Cited by in RCA: 489] [Article Influence: 81.5] [Reference Citation Analysis (7)] |
| 64. | Jayachandran M, Qu S. Non-alcoholic fatty liver disease and gut microbial dysbiosis- underlying mechanisms and gut microbiota mediated treatment strategies. Rev Endocr Metab Disord. 2023;24:1189-1204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 65. | Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, Burcelin R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57:1470-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4053] [Cited by in RCA: 3610] [Article Influence: 200.6] [Reference Citation Analysis (8)] |
| 66. | Shen L, Su L, Turner JR. Mechanisms and functional implications of intestinal barrier defects. Dig Dis. 2009;27:443-449. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 120] [Cited by in RCA: 116] [Article Influence: 6.8] [Reference Citation Analysis (4)] |
| 67. | Salim SY, Söderholm JD. Importance of disrupted intestinal barrier in inflammatory bowel diseases. Inflamm Bowel Dis. 2011;17:362-381. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 472] [Article Influence: 31.5] [Reference Citation Analysis (6)] |
| 68. | Ji Y, Sakata Y, Tso P. Nutrient-induced inflammation in the intestine. Curr Opin Clin Nutr Metab Care. 2011;14:315-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 69. | Donaldson DS, Else KJ, Mabbott NA. The Gut-Associated Lymphoid Tissues in the Small Intestine, Not the Large Intestine, Play a Major Role in Oral Prion Disease Pathogenesis. J Virol. 2015;89:9532-9547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 70. | Henao-Mejia J, Elinav E, Thaiss CA, Flavell RA. Inflammasomes and metabolic disease. Annu Rev Physiol. 2014;76:57-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 108] [Article Influence: 8.3] [Reference Citation Analysis (2)] |
| 71. | Tilg H, Cani PD, Mayer EA. Gut microbiome and liver diseases. Gut. 2016;65:2035-2044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 377] [Article Influence: 37.7] [Reference Citation Analysis (0)] |
| 72. | Volynets V, Louis S, Pretz D, Lang L, Ostaff MJ, Wehkamp J, Bischoff SC. Intestinal Barrier Function and the Gut Microbiome Are Differentially Affected in Mice Fed a Western-Style Diet or Drinking Water Supplemented with Fructose. J Nutr. 2017;147:770-780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 142] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 73. | Di Vincenzo F, Del Gaudio A, Petito V, Lopetuso LR, Scaldaferri F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern Emerg Med. 2024;19:275-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1010] [Cited by in RCA: 782] [Article Influence: 391.0] [Reference Citation Analysis (1)] |
| 74. | Brun P, Castagliuolo I, Di Leo V, Buda A, Pinzani M, Palù G, Martines D. Increased intestinal permeability in obese mice: new evidence in the pathogenesis of nonalcoholic steatohepatitis. Am J Physiol Gastrointest Liver Physiol. 2007;292:G518-G525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 670] [Cited by in RCA: 644] [Article Influence: 33.9] [Reference Citation Analysis (3)] |
| 75. | Soderborg TK, Clark SE, Mulligan CE, Janssen RC, Babcock L, Ir D, Young B, Krebs N, Lemas DJ, Johnson LK, Weir T, Lenz LL, Frank DN, Hernandez TL, Kuhn KA, D'Alessandro A, Barbour LA, El Kasmi KC, Friedman JE. The gut microbiota in infants of obese mothers increases inflammation and susceptibility to NAFLD. Nat Commun. 2018;9:4462. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 230] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 76. | An L, Wirth U, Koch D, Schirren M, Drefs M, Koliogiannis D, Nieß H, Andrassy J, Guba M, Bazhin AV, Werner J, Kühn F. The Role of Gut-Derived Lipopolysaccharides and the Intestinal Barrier in Fatty Liver Diseases. J Gastrointest Surg. 2022;26:671-683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 232] [Cited by in RCA: 198] [Article Influence: 49.5] [Reference Citation Analysis (1)] |
| 77. | Su GL, Klein RD, Aminlari A, Zhang HY, Steinstraesser L, Alarcon WH, Remick DG, Wang SC. Kupffer cell activation by lipopolysaccharide in rats: role for lipopolysaccharide binding protein and toll-like receptor 4. Hepatology. 2000;31:932-936. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 209] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 78. | Baffy G. Kupffer cells in non-alcoholic fatty liver disease: the emerging view. J Hepatol. 2009;51:212-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 389] [Cited by in RCA: 382] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 79. | Abu-Shanab A, Quigley EM. The role of the gut microbiota in nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2010;7:691-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 388] [Cited by in RCA: 354] [Article Influence: 22.1] [Reference Citation Analysis (3)] |
| 80. | Machado MV, Cortez-Pinto H. Gut microbiota and nonalcoholic fatty liver disease. Ann Hepatol. 2012;11:440-449. [PubMed] |
| 81. | Rabelo F, Oliveira CP, Faintuch J, Mazo DF, Lima VM, Stefano JT, Barbeiro HV, Soriano FG, Alves VA, Carrilho FJ. Pro- and anti-inflammatory cytokines in steatosis and steatohepatitis. Obes Surg. 2010;20:906-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 82. | Ucar F, Sezer S, Erdogan S, Akyol S, Armutcu F, Akyol O. The relationship between oxidative stress and nonalcoholic fatty liver disease: Its effects on the development of nonalcoholic steatohepatitis. Redox Rep. 2013;18:127-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 108] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 83. | Musso G, Cassader M, Paschetta E, Gambino R. Bioactive Lipid Species and Metabolic Pathways in Progression and Resolution of Nonalcoholic Steatohepatitis. Gastroenterology. 2018;155:282-302.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 267] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 84. | Fucho R, Casals N, Serra D, Herrero L. Ceramides and mitochondrial fatty acid oxidation in obesity. FASEB J. 2017;31:1263-1272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 102] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 85. | Palomer X, Pizarro-Delgado J, Barroso E, Vázquez-Carrera M. Palmitic and Oleic Acid: The Yin and Yang of Fatty Acids in Type 2 Diabetes Mellitus. Trends Endocrinol Metab. 2018;29:178-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 401] [Article Influence: 50.1] [Reference Citation Analysis (8)] |
| 86. | Gao D, Wei C, Chen L, Huang J, Yang S, Diehl AM. Oxidative DNA damage and DNA repair enzyme expression are inversely related in murine models of fatty liver disease. Am J Physiol Gastrointest Liver Physiol. 2004;287:G1070-G1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 87] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 87. | Garcia-Martinez I, Santoro N, Chen Y, Hoque R, Ouyang X, Caprio S, Shlomchik MJ, Coffman RL, Candia A, Mehal WZ. Hepatocyte mitochondrial DNA drives nonalcoholic steatohepatitis by activation of TLR9. J Clin Invest. 2016;126:859-864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 404] [Article Influence: 40.4] [Reference Citation Analysis (4)] |
| 88. | Brouwers B, Schrauwen-Hinderling VB, Jelenik T, Gemmink A, Sparks LM, Havekes B, Bruls Y, Dahlmans D, Roden M, Hesselink MKC, Schrauwen P. Exercise training reduces intrahepatic lipid content in people with and people without nonalcoholic fatty liver. Am J Physiol Endocrinol Metab. 2018;314:E165-E173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 50] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 89. | Wong VW, Wong GL, Chan RS, Shu SS, Cheung BH, Li LS, Chim AM, Chan CK, Leung JK, Chu WC, Woo J, Chan HL. Beneficial effects of lifestyle intervention in non-obese patients with non-alcoholic fatty liver disease. J Hepatol. 2018;69:1349-1356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 244] [Article Influence: 30.5] [Reference Citation Analysis (1)] |
| 90. | Jafarikhah R, Damirchi A, Rahmani Nia F, Razavi-Toosi SMT, Shafaghi A, Asadian M. Effect of functional resistance training on the structure and function of the heart and liver in patients with non-alcoholic fatty liver. Sci Rep. 2023;13:15475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 91. | O'Gorman P, Naimimohasses S, Monaghan A, Kennedy M, Melo AM, Ní Fhloinn D, Doherty DG, Beddy P, Finn SP, Moore JB, Gormley J, Norris S. Improvement in histological endpoints of MAFLD following a 12-week aerobic exercise intervention. Aliment Pharmacol Ther. 2020;52:1387-1398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 92. | Babu AF, Csader S, Männistö V, Tauriainen MM, Pentikäinen H, Savonen K, Klåvus A, Koistinen V, Hanhineva K, Schwab U. Effects of exercise on NAFLD using non-targeted metabolomics in adipose tissue, plasma, urine, and stool. Sci Rep. 2022;12:6485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 53] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 93. | Takahashi A, Abe K, Fujita M, Hayashi M, Okai K, Ohira H. Simple resistance exercise decreases cytokeratin 18 and fibroblast growth factor 21 levels in patients with nonalcoholic fatty liver disease: A retrospective clinical study. Medicine (Baltimore). 2020;99:e20399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 94. | Stine JG, Long MT, Corey KE, Sallis RE, Allen AM, Armstrong MJ, Conroy DE, Cuthbertson DJ, Duarte-Rojo A, Hallsworth K, Hickman IJ, Kappus MR, Keating SE, Pugh CJA, Rotman Y, Simon TL, Vilar-Gomez E, Wong VW, Schmitz KH. Physical Activity and Nonalcoholic Fatty Liver Disease: A Roundtable Statement from the American College of Sports Medicine. Med Sci Sports Exerc. 2023;55:1717-1726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 95. | Rector RS, Thyfault JP, Morris RT, Laye MJ, Borengasser SJ, Booth FW, Ibdah JA. Daily exercise increases hepatic fatty acid oxidation and prevents steatosis in Otsuka Long-Evans Tokushima Fatty rats. Am J Physiol Gastrointest Liver Physiol. 2008;294:G619-G626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 240] [Article Influence: 13.3] [Reference Citation Analysis (3)] |
| 96. | Jeppesen J, Kiens B. Regulation and limitations to fatty acid oxidation during exercise. J Physiol. 2012;590:1059-1068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 92] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 97. | Tung BT, Rodriguez-Bies E, Thanh HN, Le-Thi-Thu H, Navas P, Sanchez VM, López-Lluch G. Organ and tissue-dependent effect of resveratrol and exercise on antioxidant defenses of old mice. Aging Clin Exp Res. 2015;27:775-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 98. | Cui J, Guo Y, Yang W, Zhao X, Yu F, Tang W, Pang B, Su X. Effects of exercise on learning and memory, oxidative stress and nNOS expression in marginal division of striatum of ovariectomized rats. J Sports Med Phys Fitness. 2018;58:356-365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 99. | Ghareghani P, Shanaki M, Ahmadi S, Khoshdel AR, Rezvan N, Meshkani R, Delfan M, Gorgani-Firuzjaee S. Aerobic endurance training improves nonalcoholic fatty liver disease (NAFLD) features via miR-33 dependent autophagy induction in high fat diet fed mice. Obes Res Clin Pract. 2018;12:80-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 100. | Bae-Gartz I, Kasper P, Großmann N, Breuer S, Janoschek R, Kretschmer T, Appel S, Schmitz L, Vohlen C, Quaas A, Schweiger MR, Grimm C, Fischer A, Ferrari N, Graf C, Frese CK, Lang S, Demir M, Schramm C, Fink G, Goeser T, Dötsch J, Hucklenbruch-Rother E. Maternal exercise conveys protection against NAFLD in the offspring via hepatic metabolic programming. Sci Rep. 2020;10:15424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (2)] |
| 101. | Berná G, Romero-Gomez M. The role of nutrition in non-alcoholic fatty liver disease: Pathophysiology and management. Liver Int. 2020;40 Suppl 1:102-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 203] [Cited by in RCA: 176] [Article Influence: 29.3] [Reference Citation Analysis (2)] |
| 102. | Geng X, Rao Z, Zhang J, Huang P, Qu C, Wu D, Wei Q, Liu S, Zhuang X, Zhao J. Combined Cold Exposure and Exercise Improves NAFLD: Mechanistic Insights. Med Sci Sports Exerc. 2025;57:1988-2000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 103. | Zhang J, Li C, Rao Z, Geng X, Liang C, Liu S, Huang P, Wu D, Feng Y, Zhao J. Cold aerobic exercise mitigates NAFLD fibrosis through UBAP2L-regulated TGF-β/SMAD2 signaling. J Endocrinol. 2025;265:e240351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 104. | Hassani Zadeh S, Mansoori A, Hosseinzadeh M. Relationship between dietary patterns and non-alcoholic fatty liver disease: A systematic review and meta-analysis. J Gastroenterol Hepatol. 2021;36:1470-1478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (1)] |
| 105. | Risi R, Tozzi R, Watanabe M. Beyond weight loss in nonalcoholic fatty liver disease: the role of carbohydrate restriction. Curr Opin Clin Nutr Metab Care. 2021;24:349-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 106. | Plaz Torres MC, Aghemo A, Lleo A, Bodini G, Furnari M, Marabotto E, Miele L, Giannini EG. Mediterranean Diet and NAFLD: What We Know and Questions That Still Need to Be Answered. Nutrients. 2019;11:2971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 75] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 107. | Properzi C, O'Sullivan TA, Sherriff JL, Ching HL, Jeffrey GP, Buckley RF, Tibballs J, MacQuillan GC, Garas G, Adams LA. Ad Libitum Mediterranean and Low-Fat Diets Both Significantly Reduce Hepatic Steatosis: A Randomized Controlled Trial. Hepatology. 2018;68:1741-1754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 180] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 108. | Biolato M, Manca F, Marrone G, Cefalo C, Racco S, Miggiano GA, Valenza V, Gasbarrini A, Miele L, Grieco A. Intestinal permeability after Mediterranean diet and low-fat diet in non-alcoholic fatty liver disease. World J Gastroenterol. 2019;25:509-520. [PubMed] [DOI] [Full Text] |
| 109. | George ES, Reddy A, Nicoll AJ, Ryan MC, Itsiopoulos C, Abbott G, Johnson NA, Sood S, Roberts SK, Tierney AC. Impact of a Mediterranean diet on hepatic and metabolic outcomes in non-alcoholic fatty liver disease: The MEDINA randomised controlled trial. Liver Int. 2022;42:1308-1322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 110. | Kawaguchi T, Charlton M, Kawaguchi A, Yamamura S, Nakano D, Tsutsumi T, Zafer M, Torimura T. Effects of Mediterranean Diet in Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Randomized Controlled Trials. Semin Liver Dis. 2021;41:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (2)] |
| 111. | Akbulut UE, Isik IA, Atalay A, Eraslan A, Durmus E, Turkmen S, Yurttas AS. The effect of a Mediterranean diet vs. a low-fat diet on non-alcoholic fatty liver disease in children: a randomized trial. Int J Food Sci Nutr. 2022;73:357-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 112. | Yurtdaş G, Akbulut G, Baran M, Yılmaz C. The effects of Mediterranean diet on hepatic steatosis, oxidative stress, and inflammation in adolescents with non-alcoholic fatty liver disease: A randomized controlled trial. Pediatr Obes. 2022;17:e12872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 113. | Di Lorenzo C, Colombo F, Biella S, Stockley C, Restani P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients. 2021;13:273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 579] [Article Influence: 115.8] [Reference Citation Analysis (0)] |
| 114. | Monteiro J, Leslie M, Moghadasian MH, Arendt BM, Allard JP, Ma DW. The role of n - 6 and n - 3 polyunsaturated fatty acids in the manifestation of the metabolic syndrome in cardiovascular disease and non-alcoholic fatty liver disease. Food Funct. 2014;5:426-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 115. | Mäkelä TNK, Tuomainen TP, Hantunen S, Virtanen JK. Associations of serum n-3 and n-6 polyunsaturated fatty acids with prevalence and incidence of nonalcoholic fatty liver disease. Am J Clin Nutr. 2022;116:759-770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 116. | Hao L, Chen CY, Nie YH, Kaliannan K, Kang JX. Differential Interventional Effects of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on High Fat Diet-Induced Obesity and Hepatic Pathology. Int J Mol Sci. 2023;24:17261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 117. | Das A, Tang YLM, Althumiri NA, Garcia-Larsen V, Schattenberg JM, Alqahtani SA. Fatty acid composition but not quantity is an important indicator of non-alcoholic fatty liver disease: a systematic review. Eur J Clin Nutr. 2023;77:1113-1129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 118. | Watanabe M, Tozzi R, Risi R, Tuccinardi D, Mariani S, Basciani S, Spera G, Lubrano C, Gnessi L. Beneficial effects of the ketogenic diet on nonalcoholic fatty liver disease: A comprehensive review of the literature. Obes Rev. 2020;21:e13024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 189] [Cited by in RCA: 172] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 119. | Luukkonen PK, Dufour S, Lyu K, Zhang XM, Hakkarainen A, Lehtimäki TE, Cline GW, Petersen KF, Shulman GI, Yki-Järvinen H. Effect of a ketogenic diet on hepatic steatosis and hepatic mitochondrial metabolism in nonalcoholic fatty liver disease. Proc Natl Acad Sci U S A. 2020;117:7347-7354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 194] [Article Influence: 32.3] [Reference Citation Analysis (1)] |
| 120. | Anekwe CV, Chandrasekaran P, Stanford FC. Ketogenic Diet-induced Elevated Cholesterol, Elevated Liver Enzymes and Potential Non-alcoholic Fatty Liver Disease. Cureus. 2020;12:e6605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 121. | Muyyarikkandy MS, McLeod M, Maguire M, Mahar R, Kattapuram N, Zhang C, Surugihalli C, Muralidaran V, Vavilikolanu K, Mathews CE, Merritt ME, Sunny NE. Branched chain amino acids and carbohydrate restriction exacerbate ketogenesis and hepatic mitochondrial oxidative dysfunction during NAFLD. FASEB J. 2020;34:14832-14849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 122. | Crabtree CD, Kackley ML, Buga A, Fell B, LaFountain RA, Hyde PN, Sapper TN, Kraemer WJ, Scandling D, Simonetti OP, Volek JS. Comparison of Ketogenic Diets with and without Ketone Salts versus a Low-Fat Diet: Liver Fat Responses in Overweight Adults. Nutrients. 2021;13:966. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 123. | Markova M, Pivovarova O, Hornemann S, Sucher S, Frahnow T, Wegner K, Machann J, Petzke KJ, Hierholzer J, Lichtinghagen R, Herder C, Carstensen-Kirberg M, Roden M, Rudovich N, Klaus S, Thomann R, Schneeweiss R, Rohn S, Pfeiffer AF. Isocaloric Diets High in Animal or Plant Protein Reduce Liver Fat and Inflammation in Individuals With Type 2 Diabetes. Gastroenterology. 2017;152:571-585.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 242] [Cited by in RCA: 208] [Article Influence: 23.1] [Reference Citation Analysis (1)] |
| 124. | Yang JP, Shin JH, Seo SH, Kim SG, Lee SH, Shin EH. Effects of Antioxidants in Reducing Accumulation of Fat in Hepatocyte. Int J Mol Sci. 2018;19:2563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 125. | Ivancovsky-Wajcman D, Fliss-Isakov N, Salomone F, Webb M, Shibolet O, Kariv R, Zelber-Sagi S. Dietary vitamin E and C intake is inversely associated with the severity of nonalcoholic fatty liver disease. Dig Liver Dis. 2019;51:1698-1705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 126. | Poonyam P, Kritsanaviparkporn C, Chommaitree P, Soodcharoen A. The Effects of Combined Vitamin E and C for Treatment of Non-Alcoholic Fatty Liver Disease (NAFLD): A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Asian Pac J Cancer Prev. 2022;23:2891-2899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 127. | Hadzi-Petrushev N, Dimovska K, Jankulovski N, Mitrov D, Mladenov M. Supplementation with Alpha-Tocopherol and Ascorbic Acid to Nonalcoholic Fatty Liver Disease's Statin Therapy in Men. Adv Pharmacol Sci. 2018;2018:4673061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 128. | Podszun MC, Alawad AS, Lingala S, Morris N, Huang WA, Yang S, Schoenfeld M, Rolt A, Ouwerkerk R, Valdez K, Umarova R, Ma Y, Fatima SZ, Lin DD, Mahajan LS, Samala N, Violet PC, Levine M, Shamburek R, Gharib AM, Kleiner DE, Garraffo HM, Cai H, Walter PJ, Rotman Y. Vitamin E treatment in NAFLD patients demonstrates that oxidative stress drives steatosis through upregulation of de-novo lipogenesis. Redox Biol. 2020;37:101710. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 86] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 129. | Fang J, Yin H, Yang Z, Tan M, Wang F, Chen K, Zuo Z, Shu G, Cui H, Ouyang P, Guo H, Chen Z, Huang C, Geng Y, Liu W. Vitamin E protects against cadmium-induced sub-chronic liver injury associated with the inhibition of oxidative stress and activation of Nrf2 pathway. Ecotoxicol Environ Saf. 2021;208:111610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 62] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 130. | Sahin A, Demirel-Yalciner T, Sozen E, Ozer NK. Protective effect of alpha-tocopherol on lipogenesis and oxysterol production in hypercholesterolemia-induced nonalcoholic steatohepatitis. Free Radic Res. 2024;58:630-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 131. | Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, Bass NM, Neuschwander-Tetri BA, Lavine JE, Tonascia J, Unalp A, Van Natta M, Clark J, Brunt EM, Kleiner DE, Hoofnagle JH, Robuck PR; NASH CRN. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010;362:1675-1685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2814] [Cited by in RCA: 2560] [Article Influence: 160.0] [Reference Citation Analysis (3)] |
| 132. | Wang MY, Prabahar K, Găman MA, Zhang JL. Vitamin E supplementation in the treatment on nonalcoholic fatty liver disease (NAFLD): Evidence from an umbrella review of meta-analysis on randomized controlled trials. J Dig Dis. 2023;24:380-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 133. | Chee NM, Sinnanaidu RP, Chan WK. Vitamin E improves serum markers and histology in adults with metabolic dysfunction-associated steatotic liver disease: Systematic review and meta-analysis. J Gastroenterol Hepatol. 2024;39:2545-2554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 23] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 134. | Song Y, Ni W, Zheng M, Sheng H, Wang J, Xie S, Yang Y, Chi X, Chen J, He F, Fan X, Mi Y, Zhang J, Wang B, Bai L, Xie W, Zhong B, Yeo YH, Rui F, Zang S, Li J, Shi J; Chinese NAFLD Clinical Research Network (CNAFLD CRN). Vitamin E (300 mg) in the treatment of MASH: A multi-center, randomized, double-blind, placebo-controlled study. Cell Rep Med. 2025;6:101939. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 135. | Klein EA, Thompson IM Jr, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, Minasian LM, Ford LG, Parnes HL, Gaziano JM, Karp DD, Lieber MM, Walther PJ, Klotz L, Parsons JK, Chin JL, Darke AK, Lippman SM, Goodman GE, Meyskens FL Jr, Baker LH. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306:1549-1556. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1497] [Cited by in RCA: 1247] [Article Influence: 83.1] [Reference Citation Analysis (4)] |
| 136. | Zeng Q, Zhao L, Meng C, Zhao X, Liu Y, Shi R, Han X, Wang T, Li J. Prophylactic and therapeutic effects of different doses of vitamin C on high-fat-diet-induced non-alcoholic fatty liver disease in mice. Biomed Pharmacother. 2020;131:110792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 137. | He Z, Li X, Yang H, Wu P, Wang S, Cao D, Guo X, Xu Z, Gao J, Zhang W, Luo X. Effects of Oral Vitamin C Supplementation on Liver Health and Associated Parameters in Patients With Non-Alcoholic Fatty Liver Disease: A Randomized Clinical Trial. Front Nutr. 2021;8:745609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 138. | Chen Q, Zhao L, Mei L, Zhao X, Han P, Liu J, Meng C, Li R, Zhong R, Wang K, Li J. Vitamin C and vitamin D(3) alleviate metabolic-associated fatty liver disease by regulating the gut microbiota and bile acid metabolism via the gut-liver axis. Front Pharmacol. 2023;14:1163694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 139. | Ipsen DH, Tveden-Nyborg P, Lykkesfeldt J. Does vitamin C deficiency promote fatty liver disease development? Nutrients. 2014;6:5473-5499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 140. | Aghaei F, Wong A, Zargani M, Sarshin A, Feizolahi F, Derakhshan Z, Hashemi M, Arabzadeh E. Effects of swimming exercise combined with silymarin and vitamin C supplementation on hepatic inflammation, oxidative stress, and histopathology in elderly rats with high-fat diet-induced liver damage. Nutrition. 2023;115:112167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 141. | Zhang A, Sun H, Wang X. Recent advances in natural products from plants for treatment of liver diseases. Eur J Med Chem. 2013;63:570-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 159] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 142. | Simental-Mendía LE, Gamboa-Gómez CI, Guerrero-Romero F, Simental-Mendía M, Sánchez-García A, Rodríguez-Ramírez M. Beneficial Effects of Plant-Derived Natural Products on Non-alcoholic Fatty Liver Disease. Adv Exp Med Biol. 2021;1308:257-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 143. | Pisonero-Vaquero S, González-Gallego J, Sánchez-Campos S, García-Mediavilla MV. Flavonoids and Related Compounds in Non-Alcoholic Fatty Liver Disease Therapy. Curr Med Chem. 2015;22:2991-3012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 144. | Porras D, Nistal E, Martínez-Flórez S, Pisonero-Vaquero S, Olcoz JL, Jover R, González-Gallego J, García-Mediavilla MV, Sánchez-Campos S. Protective effect of quercetin on high-fat diet-induced non-alcoholic fatty liver disease in mice is mediated by modulating intestinal microbiota imbalance and related gut-liver axis activation. Free Radic Biol Med. 2017;102:188-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 467] [Cited by in RCA: 403] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 145. | Wang XC, Song L, Wang XH. Efficacy of dietary polyphenol supplement in patients with non-alcoholic fatty liver disease: a network meta-analysis. Front Nutr. 2025;12:1582861. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 146. | Vidyashankar S, Sandeep Varma R, Patki PS. Quercetin ameliorate insulin resistance and up-regulates cellular antioxidants during oleic acid induced hepatic steatosis in HepG2 cells. Toxicol In Vitro. 2013;27:945-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 147. | Yang H, Yang T, Heng C, Zhou Y, Jiang Z, Qian X, Du L, Mao S, Yin X, Lu Q. Quercetin improves nonalcoholic fatty liver by ameliorating inflammation, oxidative stress, and lipid metabolism in db/db mice. Phytother Res. 2019;33:3140-3152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 207] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 148. | Zhao X, Wang J, Deng Y, Liao L, Zhou M, Peng C, Li Y. Quercetin as a protective agent for liver diseases: A comprehensive descriptive review of the molecular mechanism. Phytother Res. 2021;35:4727-4747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 86] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 149. | Li X, Wang R, Zhou N, Wang X, Liu Q, Bai Y, Bai Y, Liu Z, Yang H, Zou J, Wang H, Shi T. Quercetin improves insulin resistance and hepatic lipid accumulation in vitro in a NAFLD cell model. Biomed Rep. 2013;1:71-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 63] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 150. | Li N, Cui C, Xu J, Mi M, Wang J, Qin Y. Quercetin intervention reduced hepatic fat deposition in patients with nonalcoholic fatty liver disease: a randomized, double-blind, placebo-controlled crossover clinical trial. Am J Clin Nutr. 2024;120:507-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 151. | Andres S, Pevny S, Ziegenhagen R, Bakhiya N, Schäfer B, Hirsch-Ernst KI, Lampen A. Safety Aspects of the Use of Quercetin as a Dietary Supplement. Mol Nutr Food Res. 2018;62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 400] [Article Influence: 44.4] [Reference Citation Analysis (2)] |
| 152. | Gambini J, Inglés M, Olaso G, Lopez-Grueso R, Bonet-Costa V, Gimeno-Mallench L, Mas-Bargues C, Abdelaziz KM, Gomez-Cabrera MC, Vina J, Borras C. Properties of Resveratrol: In Vitro and In Vivo Studies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxid Med Cell Longev. 2015;2015:837042. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 638] [Cited by in RCA: 525] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 153. | Berman AY, Motechin RA, Wiesenfeld MY, Holz MK. The therapeutic potential of resveratrol: a review of clinical trials. NPJ Precis Oncol. 2017;1:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 643] [Cited by in RCA: 558] [Article Influence: 62.0] [Reference Citation Analysis (0)] |
| 154. | Perumpail BJ, Li AA, John N, Sallam S, Shah ND, Kwong W, Cholankeril G, Kim D, Ahmed A. The Role of Vitamin E in the Treatment of NAFLD. Diseases. 2018;6:86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 97] [Article Influence: 12.1] [Reference Citation Analysis (1)] |
| 155. | Charytoniuk T, Drygalski K, Konstantynowicz-Nowicka K, Berk K, Chabowski A. Alternative treatment methods attenuate the development of NAFLD: A review of resveratrol molecular mechanisms and clinical trials. Nutrition. 2017;34:108-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 156. | Hosseini H, Teimouri M, Shabani M, Koushki M, Babaei Khorzoughi R, Namvarjah F, Izadi P, Meshkani R. Resveratrol alleviates non-alcoholic fatty liver disease through epigenetic modification of the Nrf2 signaling pathway. Int J Biochem Cell Biol. 2020;119:105667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 117] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 157. | Li Q, Zheng Y, Sun Y, Xu G. Resveratrol attenuated fatty acid synthesis through MAPK-PPAR pathway in red tilapia. Comp Biochem Physiol C Toxicol Pharmacol. 2023;268:109598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 158. | Rafiei H, Omidian K, Bandy B. Comparison of dietary polyphenols for protection against molecular mechanisms underlying nonalcoholic fatty liver disease in a cell model of steatosis. Mol Nutr Food Res. 2017;61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 42] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 159. | Faghihzadeh F, Adibi P, Hekmatdoost A. The effects of resveratrol supplementation on cardiovascular risk factors in patients with non-alcoholic fatty liver disease: a randomised, double-blind, placebo-controlled study. Br J Nutr. 2015;114:796-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 137] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 160. | Heebøll S, Kreuzfeldt M, Hamilton-Dutoit S, Kjær Poulsen M, Stødkilde-Jørgensen H, Møller HJ, Jessen N, Thorsen K, Kristina Hellberg Y, Bønløkke Pedersen S, Grønbæk H. Placebo-controlled, randomised clinical trial: high-dose resveratrol treatment for non-alcoholic fatty liver disease. Scand J Gastroenterol. 2016;51:456-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 122] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 161. | Elgebaly A, Radwan IA, AboElnas MM, Ibrahim HH, Eltoomy MF, Atta AA, Mesalam HA, Sayed AA, Othman AA. Resveratrol Supplementation in Patients with Non-Alcoholic Fatty Liver Disease: Systematic Review and Meta-analysis. J Gastrointestin Liver Dis. 2017;26:59-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 72] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 162. | Asghari S, Asghari-Jafarabadi M, Somi MH, Ghavami SM, Rafraf M. Comparison of Calorie-Restricted Diet and Resveratrol Supplementation on Anthropometric Indices, Metabolic Parameters, and Serum Sirtuin-1 Levels in Patients With Nonalcoholic Fatty Liver Disease: A Randomized Controlled Clinical Trial. J Am Coll Nutr. 2018;37:223-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 73] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 163. | Ali Sangouni A, Abdollahi S, Mozaffari-Khosravi H. Effect of resveratrol supplementation on hepatic steatosis and cardiovascular indices in overweight subjects with type 2 diabetes: a double-blind, randomized controlled trial. BMC Cardiovasc Disord. 2022;22:212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 164. | Grynkiewicz G, Ślifirski P. Curcumin and curcuminoids in quest for medicinal status. Acta Biochim Pol. 2012;59:201-212. [PubMed] |
| 165. | Priyadarsini KI. The chemistry of curcumin: from extraction to therapeutic agent. Molecules. 2014;19:20091-20112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1238] [Cited by in RCA: 858] [Article Influence: 71.5] [Reference Citation Analysis (0)] |
| 166. | Stellavato A, Pirozzi AVA, de Novellis F, Scognamiglio I, Vassallo V, Giori AM, De Rosa M, Schiraldi C. In vitro assessment of nutraceutical compounds and novel nutraceutical formulations in a liver-steatosis-based model. Lipids Health Dis. 2018;17:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 167. | Abenavoli L, Capasso R, Milic N, Capasso F. Milk thistle in liver diseases: past, present, future. Phytother Res. 2010;24:1423-1432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 481] [Cited by in RCA: 359] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 168. | Cacciapuoti F, Scognamiglio A, Palumbo R, Forte R, Cacciapuoti F. Silymarin in non alcoholic fatty liver disease. World J Hepatol. 2013;5:109-113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 85] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 169. | Salomone F, Barbagallo I, Godos J, Lembo V, Currenti W, Cinà D, Avola R, D'Orazio N, Morisco F, Galvano F, Li Volti G. Silibinin Restores NAD⁺ Levels and Induces the SIRT1/AMPK Pathway in Non-Alcoholic Fatty Liver. Nutrients. 2017;9:1086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 83] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 170. | Cui S, Pan XJ, Ge CL, Guo YT, Zhang PF, Yan TT, Zhou JY, He QX, Cheng LH, Wang GJ, Hao HP, Wang H. Silybin alleviates hepatic lipid accumulation in methionine-choline deficient diet-induced nonalcoholic fatty liver disease in mice via peroxisome proliferator-activated receptor α. Chin J Nat Med. 2021;19:401-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 171. | Zhang R, Xu D, Zhang Y, Wang R, Yang N, Lou Y, Zhao H, Aa J, Wang G, Xie Y. Silybin Restored CYP3A Expression through the Sirtuin 2/Nuclear Factor κ-B Pathway in Mouse Nonalcoholic Fatty Liver Disease. Drug Metab Dispos. 2021;49:770-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 172. | de Avelar CR, Nunes BVC, da Silva Sassaki B, Dos Santos Vasconcelos M, de Oliveira LPM, Lyra AC, Bueno AA, de Jesus RP. Efficacy of silymarin in patients with non-alcoholic fatty liver disease - the Siliver trial: a study protocol for a randomized controlled clinical trial. Trials. 2023;24:177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 173. | Mantovani A, Byrne CD, Targher G. Efficacy of peroxisome proliferator-activated receptor agonists, glucagon-like peptide-1 receptor agonists, or sodium-glucose cotransporter-2 inhibitors for treatment of non-alcoholic fatty liver disease: a systematic review. Lancet Gastroenterol Hepatol. 2022;7:367-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 189] [Article Influence: 47.3] [Reference Citation Analysis (3)] |
| 174. | Svegliati-Baroni G, Saccomanno S, Rychlicki C, Agostinelli L, De Minicis S, Candelaresi C, Faraci G, Pacetti D, Vivarelli M, Nicolini D, Garelli P, Casini A, Manco M, Mingrone G, Risaliti A, Frega GN, Benedetti A, Gastaldelli A. Glucagon-like peptide-1 receptor activation stimulates hepatic lipid oxidation and restores hepatic signalling alteration induced by a high-fat diet in nonalcoholic steatohepatitis. Liver Int. 2011;31:1285-1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 342] [Cited by in RCA: 331] [Article Influence: 22.1] [Reference Citation Analysis (3)] |
| 175. | Eguchi Y, Kitajima Y, Hyogo H, Takahashi H, Kojima M, Ono M, Araki N, Tanaka K, Yamaguchi M, Matsuda Y, Ide Y, Otsuka T, Ozaki I, Ono N, Eguchi T, Anzai K; Japan Study Group for NAFLD (JSG-NAFLD). Pilot study of liraglutide effects in non-alcoholic steatohepatitis and non-alcoholic fatty liver disease with glucose intolerance in Japanese patients (LEAN-J). Hepatol Res. 2015;45:269-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 154] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 176. | Ferreira ST. Brain insulin, insulin-like growth factor 1 and glucagon-like peptide 1 signalling in Alzheimer's disease. J Neuroendocrinol. 2021;33:e12959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 177. | Bendotti G, Montefusco L, Lunati ME, Usuelli V, Pastore I, Lazzaroni E, Assi E, Seelam AJ, El Essawy B, Jang J, Loretelli C, D'Addio F, Berra C, Ben Nasr M, Zuccotti G, Fiorina P. The anti-inflammatory and immunological properties of GLP-1 Receptor Agonists. Pharmacol Res. 2022;182:106320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 279] [Article Influence: 69.8] [Reference Citation Analysis (0)] |
| 178. | Diz-Chaves Y, Mastoor Z, Spuch C, González-Matías LC, Mallo F. Anti-Inflammatory Effects of GLP-1 Receptor Activation in the Brain in Neurodegenerative Diseases. Int J Mol Sci. 2022;23:9583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 103] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 179. | Pang J, Feng JN, Ling W, Jin T. The anti-inflammatory feature of glucagon-like peptide-1 and its based diabetes drugs-Therapeutic potential exploration in lung injury. Acta Pharm Sin B. 2022;12:4040-4055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 180. | Zhu CG, Luo Y, Wang H, Li JY, Yang J, Liu YX, Qu HQ, Wang BL, Zhu M. Liraglutide Ameliorates Lipotoxicity-Induced Oxidative Stress by Activating the NRF2 Pathway in HepG2 Cells. Horm Metab Res. 2020;52:532-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 181. | Armstrong MJ, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, Hazlehurst JM, Guo K; LEAN trial team, Abouda G, Aldersley MA, Stocken D, Gough SC, Tomlinson JW, Brown RM, Hübscher SG, Newsome PN. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387:679-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1837] [Cited by in RCA: 1653] [Article Influence: 165.3] [Reference Citation Analysis (6)] |
| 182. | Sanyal AJ, Newsome PN, Kliers I, Østergaard LH, Long MT, Kjær MS, Cali AMG, Bugianesi E, Rinella ME, Roden M, Ratziu V; ESSENCE Study Group. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis. N Engl J Med. 2025;392:2089-2099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 536] [Cited by in RCA: 501] [Article Influence: 501.0] [Reference Citation Analysis (0)] |
| 183. | Loomba R, Hartman ML, Lawitz EJ, Vuppalanchi R, Boursier J, Bugianesi E, Yoneda M, Behling C, Cummings OW, Tang Y, Brouwers B, Robins DA, Nikooie A, Bunck MC, Haupt A, Sanyal AJ; SYNERGY-NASH Investigators. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis. N Engl J Med. 2024;391:299-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 671] [Cited by in RCA: 586] [Article Influence: 293.0] [Reference Citation Analysis (1)] |
| 184. | Zhang W, Hu H. The Emerging Therapeutic Promise of SGLT2 Inhibitors in Metabolic Dysfunction-Associated Steatotic Liver Disease. Dig Dis Sci. 2025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 185. | Ge Q, Zhang F, Liu Y. Effect of SGLT2 inhibitors on liver fat content: A meta-analysis. Biomol Biomed. 2025;25:2647-2657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 186. | Kuchay MS, Krishan S, Mishra SK, Farooqui KJ, Singh MK, Wasir JS, Bansal B, Kaur P, Jevalikar G, Gill HK, Choudhary NS, Mithal A. Effect of Empagliflozin on Liver Fat in Patients With Type 2 Diabetes and Nonalcoholic Fatty Liver Disease: A Randomized Controlled Trial (E-LIFT Trial). Diabetes Care. 2018;41:1801-1808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 545] [Cited by in RCA: 502] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 187. | Hajighasem A, Farzanegi P, Mazaheri Z. Effects of combined therapy with resveratrol, continuous and interval exercises on apoptosis, oxidative stress, and inflammatory biomarkers in the liver of old rats with non-alcoholic fatty liver disease. Arch Physiol Biochem. 2019;125:142-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 188. | Sacco R, Eggenhoffner R, Giacomelli L. Glutathione in the treatment of liver diseases: insights from clinical practice. Minerva Gastroenterol Dietol. 2016;62:316-324. [PubMed] |
| 189. | Honda Y, Kessoku T, Sumida Y, Kobayashi T, Kato T, Ogawa Y, Tomeno W, Imajo K, Fujita K, Yoneda M, Kataoka K, Taguri M, Yamanaka T, Seko Y, Tanaka S, Saito S, Ono M, Oeda S, Eguchi Y, Aoi W, Sato K, Itoh Y, Nakajima A. Efficacy of glutathione for the treatment of nonalcoholic fatty liver disease: an open-label, single-arm, multicenter, pilot study. BMC Gastroenterol. 2017;17:96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 190. | Khoshbaten M, Aliasgarzadeh A, Masnadi K, Tarzamani MK, Farhang S, Babaei H, Kiani J, Zaare M, Najafipoor F. N-acetylcysteine improves liver function in patients with non-alcoholic Fatty liver disease. Hepat Mon. 2010;10:12-16. [PubMed] |
| 191. | Ding Q, Guo R, Pei L, Lai S, Li J, Yin Y, Xu T, Yang W, Song Q, Han Q, Dou X, Li S. N-Acetylcysteine alleviates high fat diet-induced hepatic steatosis and liver injury via regulating the intestinal microecology in mice. Food Funct. 2022;13:3368-3380. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 192. | Yang K, Kim HH, Shim YR, Ryu T, Kim CW. Comprehensive transcriptomic analysis and meta-analysis identify therapeutic effects of N-acetylcysteine in nonalcoholic fatty liver disease. Front Pharmacol. 2023;14:1186582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 193. | Lancaster LH, de Andrade JA, Zibrak JD, Padilla ML, Albera C, Nathan SD, Wijsenbeek MS, Stauffer JL, Kirchgaessler KU, Costabel U. Pirfenidone safety and adverse event management in idiopathic pulmonary fibrosis. Eur Respir Rev. 2017;26:170057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 215] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 194. | Xie R, Chen S, Li F, Yang L, Yu B. Pirfenidone attenuates nonalcoholic fatty liver disease through activation of the nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway. J Biochem Mol Toxicol. 2023;37:e23251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |