Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.115456
Revised: November 23, 2025
Accepted: February 5, 2026
Published online: April 27, 2026
Processing time: 186 Days and 19.3 Hours
Non-alcoholic fatty liver disease (NAFLD) is the most common chronic liver disease worldwide, as well as a growing public health concern. Erectile dys
To systematically review and narrative synthesize the existing evidence on the association between NAFLD and ED, and to elucidate potential underlying me
We conducted a comprehensive search of various electronic databases to identify original observational studies investigating the association between NAFLD and ED. However, due to significant heterogeneity among the included studies and the limited number of eligible investigations, we opted for a narrative synthesis instead of a meta-analysis.
A total of seven observational studies, involving 4442 participants (2043 of whom had NAFLD), were included in the analysis. The evidence consistently indicates a significant positive association between NAFLD and ED. Key influencing factors identified across the studies included age, components of metabolic syndrome, insulin resistance, serum testosterone levels, and psychological factors such as anxiety.
Existing evidence indicates a strong correlation between NAFLD and ED. The strength of this association is influenced by various factors, including age, insulin resistance, testosterone levels, and anxiety. NAFLD is an important risk factor for ED. Further high-quality studies are needed to confirm this relationship and explain its underlying mechanisms.
Core Tip: Non-alcoholic fatty liver disease (NAFLD) is the most common chronic liver condition worldwide. It often occurs alongside metabolic disorders, such as obesity and type 2 diabetes. Erectile dysfunction (ED) is a common sexual health issue in men. However, there is limited high-quality evidence systematically examining the relationship between ED and NAFLD. This systematic narrative review summarizes the existing research and confirms that NAFLD is a significant risk factor for ED. The strength of this association depends on various factors, including age, insulin resistance, testosterone levels, and anxiety. This study provides evidence-based support for the early identification of ED risk in NAFLD patients, which is significant for advancing comprehensive metabolic interventions, both socially and clinically.
- Citation: He YF, Bian N, Wang HZ, Hu XD, Liu JQ, Lu SF, Wu N, He QT, Li HM. Correlation between non-alcoholic fatty liver disease and erectile dysfunction: A systematic narrative review. World J Hepatol 2026; 18(4): 115456
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/115456.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.115456
Non-alcoholic fatty liver disease (NAFLD) is an umbrella term for various liver conditions that affect approximately 25% to 30% of adults worldwide[1]. This spectrum ranges from simple steatosis to non-alcoholic steatohepatitis, which can progress to liver fibrosis, cirrhosis, and hepatocellular carcinoma. NAFLD is not only a liver disease, but also the primary hepatic manifestation of metabolic syndrome[2]. NAFLD often occurs alongside metabolic disorders such as obesity, type 2 diabetes, and insulin resistance, presenting an increasingly significant public health challenge.
Erectile dysfunction (ED) is defined as the consistent inability to achieve or maintain an erection sufficient for satisfactory sexual intercourse. It is one of the most common sexual dysfunctions in men[3]. Its prevalence increases with age, affecting approximately 52% of men aged 40 to 70[4]. ED can severely impact patients’ quality of life and their relationships with partners. ED is also recognized as an early warning sign of cardiovascular disease. ED has a mul
Both NAFLD and ED share a pathophysiological basis involving insulin resistance and chronic, low-grade inflammation[6]. Research also suggests the existence of liver-specific pathways that promote ED development independently[7], including gender hormone metabolism disorders and impaired detoxification function[8,9]. However, clinical studies directly investigating the association between NAFLD and ED are limited. This study aims to synthesize existing evidence systematically using a narrative review methodology. The review will qualitatively examine the association between NAFLD and ED, their potential mechanisms, and clinical implications. The results will inform clinical practice and guide future research.
What is the association between NAFLD and ED?
This study conducted a systematic search of multiple Chinese and English databases, including PubMed, EMBASE, MEDLINE, Google Scholar, Scopus, Wiley, Web of Science, Cochrane, ScienceDirect, and other English-language resources, as well as Chinese databases such as CBM and CNKI. With the help of research librarian Wang J, who played a key role in designing and refining the search strategy, the team developed a core retrieval strategy. To comprehensively cover unpublished gray literature, the study searched the two major preprint platforms, medRxiv and bioRxiv, simultaneously. Additionally, manually tracing the reference lists of the included studies supplemented and refined the comprehensiveness of the retrieval results.
The search process used Boolean logic operators to combine keywords. The core search terms were “non-alcoholic fatty liver disease”, “sexual dysfunction”, “erectile dysfunction”, “erection”, and “impotence”, as well as their synonyms and related terms. This approach balanced recall and precision. There were no restrictions based on publication date or language, and the last update was conducted on October 10, 2025.
We included original studies published online that directly examined the association between NAFLD and ED. We excluded news reports, case reports, animal studies, and studies with missing or unobtainable key data. We only considered studies that provided sufficient key information in their abstracts for data extraction and analysis.
Due to significant methodological heterogeneity among the included studies and the limited number of eligible investigations, we opted for a narrative synthesis approach instead of a quantitative meta-analysis. This approach better addresses the diversity of the current clinical and methodological evidence base. Two researchers conducted an independent screening study. The subjects were limited to adult males with confirmed NAFLD. The outcome was the incidence of ED or related assessment metrics. In cases of disagreement, consensus was reached through discussion. If resolution remained elusive, a third researcher was consulted for arbitration.
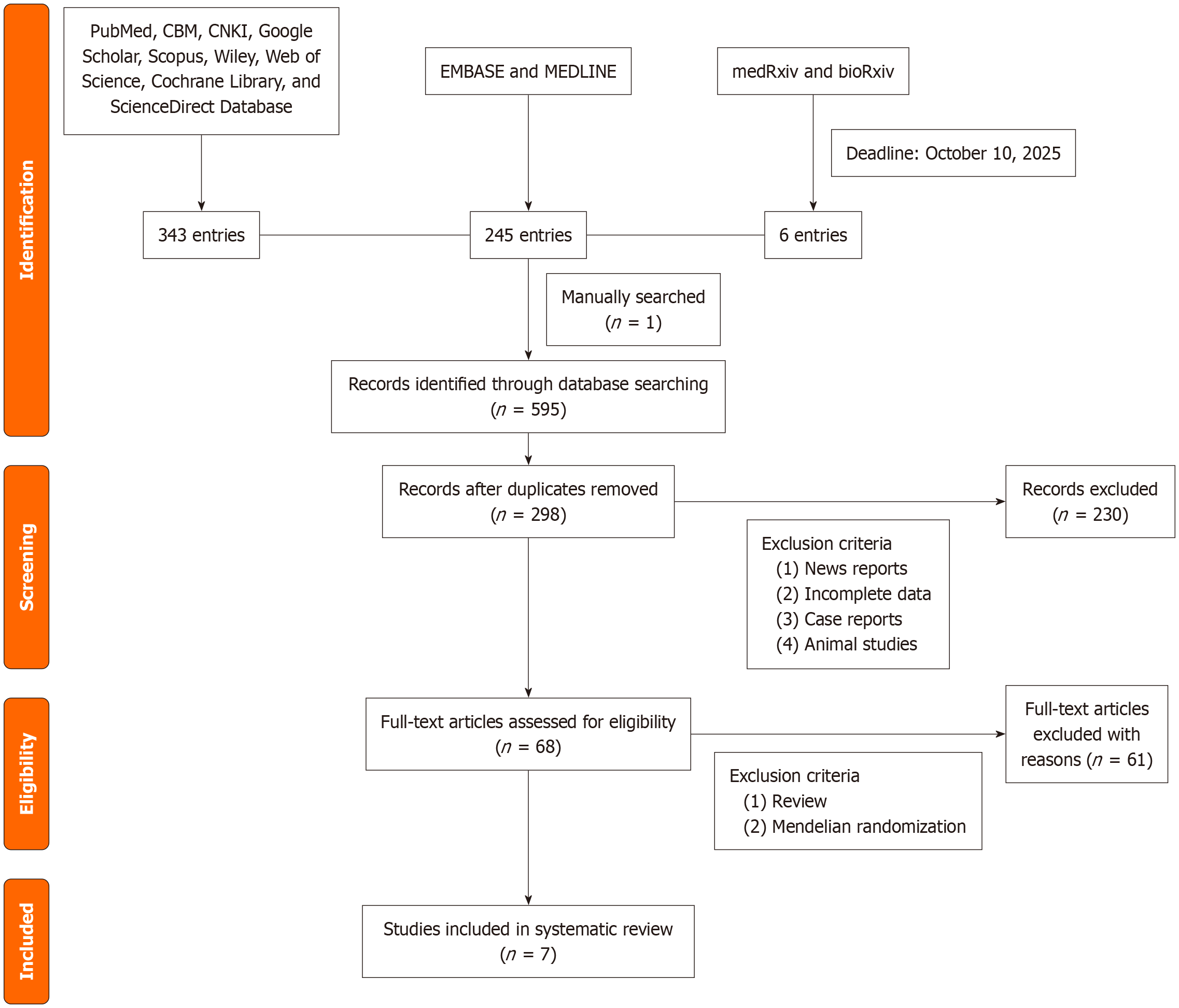
The initial search yielded 298 publications, and one additional record was identified manually. After removing duplicates and screening the titles and abstracts of the remaining publications, 68 full-text articles were assessed for eligibility. Ultimately, seven studies were included in this systematic review[3,10-15]. Figure 1 illustrates the process of visualizing literature retrieval. Two researchers independently extracted data using a standardized form. Key elements included the characteristics of the studies, demographics of the participants, methods used to diagnose NAFLD, tools used to assess ED, and main findings. The extracted data were systematically organized and summarized in Table 1.
| Ref. | Country of origin | Study design | Time period | Participants (n) | NAFLD patients (n) | Mean age, years | BMI (kg/m2) | ED risk | Population | Diagnostic methods for NAFLD | Assessment tools for ED | Main findings |
| Tarik Kani et al[3], 2021 | Turkey | Retrospective study | From 2017 to 2018 | 1362 | 684 | 48.65 ± 11.83 | 30.98 ± 6.43 | 68 (50.0) | Patients were diagnosed with NAFLD at a single gastroenterology outpatient clinic | Ultrasonography | IIEF-5 | ED is common among patients with NAFLD. Age, obesity, and hypertension are also associated with ED in these patients |
| 03 | 685 | 42.63 ± 10.52 | 29.02 ± 3.55 | |||||||||
| Youcheng et al[10], 2025 | China | Cross-sectional study | From 2001 to 2004 | 14982 | 4834 | 51.5 ± 18.6 | 28.0 ± 5.6 | 483 (32.2) | Adult males who participated in the 2001-2004 National Health and Nutrition Examination Survey in the United States | US-FLI score | Answer the questions | Participants with NAFLD had a significantly increased risk of developing ED compared with those without fatty liver disease |
| 22653 | 10155 | |||||||||||
| Duman et al[11], 2016 | Turkey | Prospective study | 20151 | 402 | 134 | 38.3 ± 11.2 | 27.88 ± 3.13 | 13 (32.5) | Male NAFLD patients aged 18 years to 75 years | Biopsy | IIEF-5 | There is a significant association between NAFLD and ED |
| 03 | 275 | 41.0 ± 9.8 | 28.62 ± 3.66 | |||||||||
| Yilmaz et al[12], 2021 | Turkey | Cross-sectional study | From February 2018 to January 2019 | 1062 | 744 | 51.47 ± 10.34 | 28.51 ± 3.57 | 74 (69.8) | Male patients presenting to the urology outpatient clinic with symptoms of erectile dysfunction | Ultrasonography | IIEF-5 | NAFLD is a significant risk factor for ED. Patients with NAFLD are 292 times more likely to experience ED than those without NAFLD |
| 403 | 325 | |||||||||||
| Eren and Horsanali[13], 2019 | Turkey | Retrospective study | From January 2016 to March 2018 | 2562 | NA4 | 61.02 ± 9.27 | 27.4 ± 3.5 | NA | Male patients are diagnosed with lower urinary tract symptoms or benign prostatic hyperplasia | Ultrasonography | IIEF-5 | NAFLD is an independent predictor of IIEF-5 scores. The severity of NAFLD is negatively correlated with IIEF-5 scores |
| 1003 | NA5 | 29.67 ± 4.16 | ||||||||||
| Hasanain et al[14], 2017 | Egypt | Prospective study | From April 2013 to February 2015 | 1922 | 884 | 42.4 ± 7.7 | NA | 88 (45.8) | A series of male patients with NAFLD were admitted across multiple disciplines | Ultrasonography | IIEF-5 | ED is a common condition among male NAFLD patients. Insulin resistance and low serum testosterone levels jointly contribute to its development |
| 03 | 1045 | |||||||||||
| Ragab et al[15], 2024 | Egypt | Observational study | From November 2021 to November 2022 | 712 | 414 | 38.9 ± 10.3 | 26.91 ± 3.12 | 41 (57.7) | NAFLD patients with normal liver function and NAFLD patients with abnormal liver function | Ultrasonography | IIEF-5 | Abnormal levels of nitric oxide and adropin may play a potential role in the development and progression of erectile dysfunction in patients with nonalcoholic fatty liver disease |
| 943 | 305 | 39.1 ± 10.7 | 27.26 ± 3.63 |
We conducted a systematic assessment of the methodological quality of the seven included studies[3,10-15]. We evaluated the cohort and case-control studies using the Newcastle-Ottawa Scale, and we applied the Agency for Healthcare Research and Quality’s criteria to the cross-sectional studies. The results of the assessment indicated that the studies’ overall quality was moderate (Table 2).
| Ref. | Study design | Assessment tools | Assessment results | Interpretation of results |
| Tarik Kani et al[3], 2021 | Retrospective study | NOS | 7 stars | The maximum total score is nine stars. A score of seven or more stars generally indicates high-quality research, while a score of five or six stars indicates medium quality, and a score of less than five stars indicates low quality |
| Youcheng et al[10], 2025 | Prospective study | NOS | 6 stars | |
| Duman et al[11], 2016 | Retrospective study | NOS | 6 stars | |
| Yilmaz et al[12], 2021 | Prospective study | NOS | 7 stars | |
| Eren and Horsanali[13], 2019 | Cross-sectional study | AHRQ | 8 points | This standard comprises 11 items, each of which is answered with “Yes”, “No”, or “Uncertain”. A “Yes” response earns 1 point. A total score of 0-3 indicates low quality, 4-7 indicates medium quality, and 8-11 indicates high quality |
| Hasanain et al[14], 2017 | Cross-sectional study | AHRQ | 7 points | |
| Ragab et al[15], 2024 | Observational study | AHRQ | 6 points |
Throughout the analysis, we thoroughly considered potential biases. Since this study employed a narrative synthesis rather than a meta-analysis and did not quantitatively pool results, statistical methods such as Begg’s test were not used to assess publication bias quantitatively. We acknowledge the possibility of publication bias and discuss it qualitatively in the limitations section, clarifying its potential impact on our conclusions.
A total of seven studies[3,10-15] were included in the final analysis. These studies had a combined sample size of 4442 participants, 2043 of whom had NAFLD. The study designs included two prospective studies[11,14] and five observational studies[3,10,12,13,15]. No randomized controlled trials were included. Of the included studies, four were from Turkey[3,11-13], two were from Egypt[14,15], and one was from China[10]. All of the studies spanned the period from 2001 to 2022. Regarding diagnostic methods for NAFLD, five studies used ultrasound examinations[3,12-15], one study used a liver biopsy[11], and one study used the ultrasonographic fatty liver index score[10]. ED was primarily assessed using the International Index of Erectile Function (IIEF-5) questionnaire.
A narrative synthesis of seven studies[3,10-15] revealed consistent evidence supporting an association between NAFLD and ED. Table 3 summarizes the factors associated with ED in NAFLD patients identified across the included studies. Four studies identified NAFLD severity scores as significant factors associated with ED[10-13]. Age was independently associated with ED in four studies[3,12-14]. Other significant factors included hypertension, insulin resistance, anxiety scores [generalized anxiety disorder-7 (GAD-7)], serum testosterone levels, adropin levels, and creatinine levels.
| Ref. | Influencing factors | Conclusions |
| Tarik Kani et al[3], 2021 | Age, obesity, hypertension | Older age, obesity, and hypertension are associated with ED in the NAFLD cohort |
| Youcheng et al[10], 2025 | US-FLI | NAFLD is positively correlated with ED |
| Duman et al[11], 2016 | NAFLD score | There is a significant association between NAFLD and ED |
| Yilmaz et al[12], 2021 | Age, MetS, NAFLD score | As NAFLD-related liver damage progresses, symptoms of ED show a significant worsening trend |
| Eren and Horsanali[13], 2019 | Age, MetS, NAFLD score | The higher the NAFLD grade, the lower the IIEF-5 score |
| Hasanain et al[14], 2017 | Age ≥ 40 years, IR, low serum testosterone | Age, insulin resistance, and low serum testosterone levels are independently associated with ED |
| Ragab et al[15], 2024 | Serum adropin, creatinine, GAD-7 score | GAD-7 scale scores, creatinine levels, and adropin were the only strong independent predictors of IIEF-5 scores |
Significant clinical and methodological heterogeneity was observed across studies, particularly about the following four factors: (1) Participant characteristics (e.g., age distribution and metabolic comorbidities); (2) Diagnostic criteria for NAFLD, including the use of imaging vs biochemical markers; (3) ED assessment methods; and (4) Control of potential confounding factors (e.g., diabetes status and cardiovascular risk factors). These variations limited the feasibility of meaningful quantitative synthesis.
This systematic review summarizes the results of seven studies[3,10-15] examining the relationship between NAFLD and ED. Despite their methodological limitations and heterogeneity, the studies consistently indicate a significant association between the two conditions. Specifically, multiple studies provide quantitative evidence of this association. For example, Yilmaz et al[12] found that patients with NAFLD were approximately 2.92 times more likely to experience ED than those without NAFLD. A cross-sectional study based on the National Health and Nutrition Examination Survey showed a positive correlation between a high fatty liver index score and ED[10]. However, caution should be exercised when interpreting the relationship between the two conditions. A mendelian randomization study failed to confirm a direct causal effect of NAFLD on ED at the genetic level[16]. This suggests that the overall causal effect of NAFLD on ED may be limited in the general population, and that the significant associations observed in observational studies may be influenced by confounding factors.
The link between NAFLD and ED stems from various mechanisms and multifactorial interactions. A thorough understanding of these mechanisms is essential for the early clinical identification and treatment of ED.
Insulin resistance and vascular endothelial dysfunction: Insulin resistance suppresses endothelial nitric oxide synthase (eNOS) activity, thereby reducing nitric oxide production[17,18]. Nitric oxide is a key mediator of smooth muscle relaxation in the corpus cavernosum[19]. Additionally, proinflammatory factors, such as tumor necrosis factor-α and interleukin-6, released from visceral fat tissue further inhibit eNOS, thereby exacerbating vascular endothelial dys
Synergistic effects of shared metabolic risk factors: Both NAFLD and ED originate from metabolic syndrome. Obesity, diabetes, dyslipidemia, and hypertension promote atherosclerosis and cavernosal fibrosis synergistically[21,22]. NAFLD is a manifestation of metabolic dysfunction in the liver, whereas ED is an early manifestation of metabolic dysfunction in the penile vasculature.
The role of hepatocyte-specific pathways and hepatocyte-derived factors: One study suggests that patients with NAFLD are at a significantly higher risk of ED, even if they do not have diabetes[11]. However, a mendelian randomization analysis by Chen et al[16] failed to detect a significant genetic causal association between NAFLD and ED. This implies that the contribution of specific subgroups or intrahepatic pathways cannot be ruled out entirely in certain contexts. These findings suggest that NAFLD may cause ED via liver-specific pathways, independently of diabetes-related factors[23]. These mechanisms include hepatic dysregulation of sex hormone metabolism[8], impaired detoxification function[24], and the secretion of hepatocyte factors, such as fetuin-B[25]. Once in the bloodstream, these liver-derived factors directly impair systemic vascular endothelial function (including that of the penile cavernosal arteries) by inhibiting nitric oxide production, inducing oxidative stress, and promoting endothelial inflammation. Together, these processes establish the “liver-vascular-erectile dysfunction axis” as a potential pathological pathway.
Our synthesis revealed several significant factors associated with ED in populations with NAFLD.
Age: Of the seven studies included in this systematic review, four explicitly identified age as an independent predictor of ED in patients with NAFLD[3,12-14]. Epidemiological investigations indicate that ED prevalence increases significantly with advancing age[26]. Mechanistically, aging is associated with a progressive decline in vascular endothelial function, worsening of atherosclerosis, decreased testosterone levels, and heightened chronic inflammation. These factors collectively contribute to the development of ED[27].
Metabolic syndrome and insulin resistance: Of the seven studies included in this systematic review, two indicated that metabolic syndrome is a risk factor for ED[12,13]. Metabolic syndrome is characterized by obesity, abnormal blood glucose levels, dyslipidemia, and hypertension. It shares a core pathophysiological basis with NAFLD and ED. Insulin resistance inhibits eNOS activity, reducing nitric oxide production and directly impairing penile cavernosal vasodilation. Additionally, insulin resistance promotes chronic inflammation and oxidative stress, which further exacerbates vascular endothelial dysfunction[27].
Serum testosterone level: NAFLD is often associated with hypogonadism, which creates a cycle of low testosterone levels[28]. Visceral obesity increases aromatase activity, promoting the conversion of testosterone to estrogen. This suppresses the hypothalamic-pituitary-gonadal axis further[29]. Low testosterone levels affect libido and directly impair eNOS activity and cavernous vasodilation capacity[30].
Psychological factors and anxiety states: The GAD-7 scale is commonly used to assess anxiety. Anxiety can exacerbate endothelial dysfunction and inhibit nitric oxide signaling pathways by activating the hypothalamic-pituitary-adrenal axis and increasing cortisol release[31]. Studies indicate a negative correlation between GAD-7 scores and IIEF-5 scores[15,32]. This suggests that anxiety may exacerbate the inflammatory responses and insulin resistance associated with NAFLD[32].
Serum creatinine level: An elevated serum creatinine level reliably indicates impaired renal function. It is also associated with NAFLD and ED. Patients with NAFLD have significantly higher creatinine levels than individuals without NAFLD, and these levels are independently associated with the degree of insulin resistance[33]. Lower IIEF-5 scores are associated with decreased creatinine clearance. Together, these findings suggest that renal impairment may contribute to ED through mechanisms such as uremic toxin accumulation, increased oxidative stress, and vascular calcification[34].
Adropin: This study presents evidence that patients with NAFLD have significantly lower serum adropin levels[15]. Adropin exerts protective effects by improving insulin resistance and enhancing endothelial nitric oxide bioavailability, and suppressing inflammation. Decreased adropin levels may exacerbate insulin resistance and ED, thereby increasing the risk of ED[35]. However, direct mechanistic evidence is needed.
To our knowledge, this is one of the few systematic reviews to comprehensively collate and integrate evidence of the link between NAFLD and ED. The review confirms the significant association between the two conditions, providing an in-depth explanation of the underlying pathophysiological mechanisms. It also sheds new light on this relationship within the cross-metabolic-vascular-endocrine axis.
However, this study also has several limitations. Firstly, we acknowledge that the original studies included in the analysis were predominantly based on cross-sectional or retrospective observational designs. The methodological characteristics of these designs make it difficult to control for confounding factors, thus precluding the establishment of a causal relationship between NAFLD and ED. Furthermore, a mendelian randomization study found no genetic-level causal association between the two conditions[16], suggesting that the observational findings may be influenced by residual confounding factors. Secondly, significant clinical and methodological heterogeneity was observed among the included studies regarding NAFLD diagnostic methods, ED assessment tools, and control of key confounders (e.g., diabetes status). Therefore, a quantitative synthesis (i.e., a meta-analysis) could not be performed, so a narrative synthesis was used instead. Consequently, precise pooled effect estimates could not be provided. Thirdly, the review carries a potential risk of bias. In addition to the selection and information biases inherent in observational studies, publication bias may also be present, whereby positive results are more likely to be published. Notably, most of the included studies failed to exclude diabetic patients or conduct subgroup analyses on non-diabetic NAFLD populations. This is a significant potential source of confounding bias. Finally, the included studies were geographically concentrated in the Middle East and North Africa, particularly in Turkey and Egypt. Relatively little evidence came from East Asia (e.g., China). Given the known variations in NAFLD prevalence, metabolic characteristics, and cultural lifestyles across different populations, this geographic limitation may affect how generalizable the study’s conclusions are to other populations worldwide, including Eur
Despite the limitations, these findings are clinically significant. In clinical practice, the strong correlation between NAFLD and ED suggests that ED may indicate vascular problems at an early stage in individuals with NAFLD. Therefore, ED screening should be incorporated into routine assessments for patients with NAFLD, particularly focusing on early identification in individuals aged 40 years and over. The clinical management strategy should aim to improve insulin resistance by encouraging lifestyle changes and, if necessary, prescribing metabolic regulators such as glucagon-like peptide-1 receptor agonists. Patients with central obesity or low testosterone should undergo an assessment of their hormone levels and psychological status. A multidisciplinary collaboration model integrating hepatology, metabolism, andrology, and psychology should be established to achieve comprehensive intervention and enhance both liver and sexual health.
This systematic narrative review identified consistent evidence of a link between NAFLD and ED, mediated via complex metabolic, hormonal, and inflammatory pathways. The strength of this association appears to be influenced by various factors, including age, insulin resistance, testosterone levels, and psychological state. Future research should focus on designing rigorously designed prospective cohort studies and exploring the clinical utility of integrated management strategies for these interrelated conditions.
The authors would like to thank Wang J for his expertise in developing and refining the literature search strategy.
| 1. | Powell EE, Wong VW, Rinella M. Non-alcoholic fatty liver disease. Lancet. 2021;397:2212-2224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2350] [Cited by in RCA: 2058] [Article Influence: 411.6] [Reference Citation Analysis (3)] |
| 2. | Haas JT, Francque S, Staels B. Pathophysiology and Mechanisms of Nonalcoholic Fatty Liver Disease. Annu Rev Physiol. 2016;78:181-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 319] [Article Influence: 29.0] [Reference Citation Analysis (0)] |
| 3. | Tarik Kani H, Emre Sener T, Emre Aykut U, Ozer Demirtas C, Keklikkiran C, Ergenc I, Fatih Demirci A, Kamil Cam H, Celikel C, Akbal C, Duman D. Causes of erectile dysfunction in non-alcoholic fatty liver disease. Hepatol Forum. 2021;2:60-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 4. | Wang W, Fan J, Huang G, Zhu X, Tian Y, Tan H, Su L. Meta-Analysis of Prevalence of Erectile Dysfunction in Mainland China: Evidence Based on Epidemiological Surveys. Sex Med. 2017;5:e19-e30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 48] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Celik O, Ipekci T, Akarken I, Ekin G, Koksal T. To evaluate the etiology of erectile dysfunction: What should we know currently? Arch Ital Urol Androl. 2014;86:197-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 6. | Tarantino G, Savastano S, Colao A. Hepatic steatosis, low-grade chronic inflammation and hormone/growth factor/adipokine imbalance. World J Gastroenterol. 2010;16:4773-4783. [PubMed] [DOI] [Full Text] |
| 7. | Mei Y, Wang X, Li Y, Xu R, Feng X. Liver transplantation improves erectile function in patients with end-stage liver disease: a systematic review and meta-analysis. Transl Androl Urol. 2024;13:1795-1804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 8. | Zang G, Sun X, Sun Y, Zhao Y, Dong Y, Pang K, Cheng P, Wang M, Zheng Y. Chronic liver diseases and erectile dysfunction. Front Public Health. 2022;10:1092353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 9. | Wang J, Cao X, Deng S, Wang B, Feng J, Meng F, Xu H, Wang S, Zao X, Li H, Ye Y. Effect of liver cirrhosis on erectile function in rats: A study combining bioinformatics analysis and experimental research. Andrologia. 2022;54:e14352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Youcheng L, Xun W, Zhufeng C. Association between nonalcoholic fatty liver disease and erectile dysfunction among American Adults from the National Health and Nutrition Examination Survey: a cross-sectional study. Int J Impot Res. 2025;37:320-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 11. | Duman DG, Biçakci E, Çelikel ÇA, Akbal C. Nonalcoholic Fatty Liver Disease is Associated with Erectile Dysfunction: A Prospective Pilot Study. J Sex Med. 2016;13:383-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | Yilmaz M, Odabas O, Karaaslan M, Guler OF, Toprak T, Bicer S, Tonyali S. Predicting risk of erectile dysfunction in patients with nonalcoholic fatty liver disease. Andrologia. 2021;53:e14091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 13. | Eren H, Horsanali MO. The independent association of non-alcoholic fatty liver disease with lower urinary tract symptoms/benign prostatic hyperplasia and erectile function scores. BJU Int. 2019;124:329-335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Hasanain AFA, Mahdy RE, Mahran AMA, Safwat ASM, Mohamed AO, Abdel-Aal SM. Erectile dysfunction in patients with nonalcoholic fatty liver disease. Arab J Gastroenterol. 2017;18:21-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 15. | Ragab A, Fattah AMA, Sayed AR, GamalEl Din SF, Mahmoud Hassan SM, Mohamed AYM, Hamed MA. Correlation between Serum Levels of Nitric Oxide and Adropin and Erectile Dysfunction in Males with Nonalcoholic Fatty Liver Disease: An Observational Study. Reprod Sci. 2024;31:2676-2684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Chen P, Wang YB, Shen YJ. [Nonalcoholic fatty liver disease and risk of erectile dysfunction: A bidirectional two-sample mendelian randomization study]. Zhonghua Nanke Zazhi. 2023;29:705-710. [DOI] [Full Text] |
| 17. | Duplain H, Burcelin R, Sartori C, Cook S, Egli M, Lepori M, Vollenweider P, Pedrazzini T, Nicod P, Thorens B, Scherrer U. Insulin resistance, hyperlipidemia, and hypertension in mice lacking endothelial nitric oxide synthase. Circulation. 2001;104:342-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 385] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 18. | Cunningham RP, Moore MP, Dashek RJ, Meers GM, Takahashi T, Sheldon RD, Wheeler AA, Diaz-Arias A, Ibdah JA, Parks EJ, Thyfault JP, Rector RS. Critical Role for Hepatocyte-Specific eNOS in NAFLD and NASH. Diabetes. 2021;70:2476-2491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 19. | Zheng ZJ, Chen Y, Chen QX, Tang WH, Chung E, Hong K, Zhang SD, Lin HC. Near-infrared photobiomodulation therapy for age-related erectile dysfunction: molecular and physiological restoration in a mouse model. Asian J Androl. 2026;28:80-88. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Persico M, Masarone M, Damato A, Ambrosio M, Federico A, Rosato V, Bucci T, Carrizzo A, Vecchione C. "Non alcoholic fatty liver disease and eNOS dysfunction in humans". BMC Gastroenterol. 2017;17:35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 21. | Francque SM, van der Graaff D, Kwanten WJ. Non-alcoholic fatty liver disease and cardiovascular risk: Pathophysiological mechanisms and implications. J Hepatol. 2016;65:425-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 437] [Cited by in RCA: 395] [Article Influence: 39.5] [Reference Citation Analysis (2)] |
| 22. | Nozaki Y, Fujita K, Wada K, Yoneda M, Shinohara Y, Imajo K, Ogawa Y, Kessoku T, Nakamuta M, Saito S, Masaki N, Nagashima Y, Terauchi Y, Nakajima A. Deficiency of eNOS exacerbates early-stage NAFLD pathogenesis by changing the fat distribution. BMC Gastroenterol. 2015;15:177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 23. | Xu H, Wang L. The Role of Notch Signaling Pathway in Non-Alcoholic Fatty Liver Disease. Front Mol Biosci. 2021;8:792667. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 24. | Xu S, Ilyas I, Little PJ, Li H, Kamato D, Zheng X, Luo S, Li Z, Liu P, Han J, Harding IC, Ebong EE, Cameron SJ, Stewart AG, Weng J. Endothelial Dysfunction in Atherosclerotic Cardiovascular Diseases and Beyond: From Mechanism to Pharmacotherapies. Pharmacol Rev. 2021;73:924-967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 623] [Cited by in RCA: 819] [Article Influence: 163.8] [Reference Citation Analysis (3)] |
| 25. | Meex RC, Hoy AJ, Morris A, Brown RD, Lo JC, Burke M, Goode RJ, Kingwell BA, Kraakman MJ, Febbraio MA, Greve JW, Rensen SS, Molloy MP, Lancaster GI, Bruce CR, Watt MJ. Fetuin B Is a Secreted Hepatocyte Factor Linking Steatosis to Impaired Glucose Metabolism. Cell Metab. 2015;22:1078-1089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 192] [Article Influence: 17.5] [Reference Citation Analysis (3)] |
| 26. | Johannes CB, Araujo AB, Feldman HA, Derby CA, Kleinman KP, McKinlay JB. Incidence of erectile dysfunction in men 40 to 69 years old: longitudinal results from the Massachusetts male aging study. J Urol. 2000;163:460-463. [PubMed] |
| 27. | Kim JA, Montagnani M, Koh KK, Quon MJ. Reciprocal relationships between insulin resistance and endothelial dysfunction: molecular and pathophysiological mechanisms. Circulation. 2006;113:1888-1904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1328] [Cited by in RCA: 1210] [Article Influence: 60.5] [Reference Citation Analysis (0)] |
| 28. | Papadimitriou K, Mousiolis AC, Mintziori G, Tarenidou C, Polyzos SA, Goulis DG. Hypogonadism and nonalcoholic fatty liver disease. Endocrine. 2024;86:28-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 29. | Colleluori G, Chen R, Turin CG, Vigevano F, Qualls C, Johnson B, Mediwala S, Villareal DT, Armamento-Villareal R. Aromatase Inhibitors Plus Weight Loss Improves the Hormonal Profile of Obese Hypogonadal Men Without Causing Major Side Effects. Front Endocrinol (Lausanne). 2020;11:277. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 30. | Marino L, Jornayvaz FR. Endocrine causes of nonalcoholic fatty liver disease. World J Gastroenterol. 2015;21:11053-11076. [PubMed] [DOI] [Full Text] |
| 31. | Demirtaş Şahin T, Yazir Y, Utkan T, Gacar G, Halbutoğulları ZS, Gocmez SS. Depression induced by chronic stress leads to penile cavernosal dysfunction: protective effect of anti-TNF-α treatment. Can J Physiol Pharmacol. 2018;96:933-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 32. | Ragab A, Ahmed MH, Reda Sayed A, EldinAbdelbary DAK, GamalEl Din SF. Serum nesfatin-1 level in men with diabetes and erectile dysfunction correlates with generalized anxiety disorder-7: A prospective comparative study. Andrology. 2023;11:307-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Yildirim U, Karakayali M, Uslu M, Ezer M, Erihan IB, Artac I, Omar T, Karabag Y, Rencuzogullari I. Association between international index of erectile function-5 scores and circadian patterns of newly diagnosed hypertension in erectile dysfunction patients. Andrologia. 2022;54:e14622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 34. | Cordeiro AC, Mizzaci CC, Fernandes RM, Araujo-Junior AG, Cardoso PO, Dutra LV, Sousa AG, Amodeo C. Simplified International Index of Erectile Function (IIEF-5) and coronary artery disease in hypertensive patients. Arq Bras Cardiol. 2012;99:924-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 35. | Celik HT, Bilen M, Kazancı F, Yildirim ME, İncebay İB, Erdamar H. Serum adropin as a predictive biomarker of erectile dysfunction in coronary artery disease patients. Cent European J Urol. 2019;72:302-306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |