Published online Apr 27, 2026. doi: 10.4254/wjh.v18.i4.115060
Revised: November 20, 2025
Accepted: February 2, 2026
Published online: April 27, 2026
Processing time: 196 Days and 21.9 Hours
The introduction of effective direct-acting antiviral (DAA) therapy for hepatitis C virus (HCV) has changed the landscape of chronic liver disease worldwide.
To compare the trends in etiologies of liver disease and causes of liver-related hospital admissions at a single tertiary transplant centre in Australia in the eras before and after DAA therapy.
A single-centre observational study was conducted to identify patients admitted to hospital with decompensated cirrhosis in the 2012 (pre-DAA) and 2019 (post-DAA) calendar years. The primary outcome was the reason for admission. Se
One hundred and seventy-eight patients with cirrhosis were admitted in 2012 compared to 198 patients in 2019. There was no significant difference in the gender, age or model for end-stage liver disease score (all P > 0.4) between the two cohorts. Ascites remains the most common complication, however the incidence rate decreased between eras, while the incidence rate of non-variceal bleeding increased. HCV-related cirrhosis admissions decreased, while non-alcoholic steatohepatitis-related cirrhosis admissions increased.
This study demonstrates a shift in the phenotype of cirrhosis, with increasing admissions for non-variceal bleeding and fewer admissions for ascites. This may be due to a shift in etiology of liver disease, with fewer cases of HCV-related and increasing non-alcoholic steatohepatitis-related cirrhosis. Further work is required to better understand cirrhosis presentations in the context of rising steatotic liver disease cases and to plan adequate resource allocation to allow health systems to cope with an increasingly complex patient population.
Core Tip: Effective treatment is now available for chronic hepatitis C, and as a result, hospitalisation of patients with hepatitis C-related cirrhosis is decreasing, while metabolic-associated steatotic liver disease is rapidly becoming one of the most common causes of cirrhosis requiring hospital admission to a major tertiary hospital in Australia. The phenotype of patients with cirrhosis is also shifting, with a decreasing incidence rate of ascites and hepatic encephalopathy and increasing incidence of non-variceal bleeding. Information on the shifting etiologies, phenotypes and prevalence of complications of cirrhosis is helpful for planning future resource allocation and devising novel ways of managing these patients.
- Citation: Terbah R, Zhao C, Greeve T, Cannan E, Wong D, Testro A, Majumdar A, Sinclair M. Shifting cirrhosis phenotype with changes in liver disease etiology in an Australian tertiary hospital. World J Hepatol 2026; 18(4): 115060
- URL: https://www.wjgnet.com/1948-5182/full/v18/i4/115060.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i4.115060
Cirrhosis continues to be a global health burden. An estimated 1.5 billion people were affected by chronic liver disease in 2017, of whom just under 123 million were cirrhotic[1]. Cirrhosis was associated with 2.4% of global deaths in 2019[2]. Clinical sequelae of cirrhosis include ascites, spontaneous bacterial peritonitis, variceal haemorrhage, hepatic encephalopathy, hepatorenal syndrome-acute kidney injury and hepatocellular carcinoma (HCC). These complications result in significant morbidity and mortality and place a large demand on healthcare resources. The predominant etiology of underlying liver disease varies around the world, but in Western countries, the most common etiologies have tra
In 2016, the Australian government invested $1 billion to provide access to the new and highly effective direct acting antiviral agents (DAAs) for the estimated 230000 Australians living with chronic hepatitis C virus (HCV)[3], and between 2016 and 2022, an estimated 60% of Australians living with HCV received treatment[4]. Corresponding with this, the proportion of liver transplants performed for HCV-related cirrhosis in Australia and New Zealand since 2016 has significantly decreased[5]. With effective HCV treatment now widely available, MASLD is becoming the leading cause of chronic liver disease in the Western world[6,7] and is already the fastest-rising indication for liver transplantation in the United States[8].
The aim of this study was to compare the trends in the underlying etiologies of liver disease presenting to hospital and the causes of liver-related hospital admissions at a single tertiary liver centre in Melbourne, Australia in the era before and after the use of DAAs for the treatment of chronic HCV.
This was a single-centre observational study of all patients aged 18 and over diagnosed with cirrhosis who were admitted to our tertiary liver centre for management of cirrhosis-related complications during two distinct calendar years: 2012 (pre-DAA) and 2019 (post-DAA).
Health Information International Classification of Diseases coding from hospital admission and discharge summaries was retrieved to identify patients with cirrhosis admitted with the complications of interest. Patient medical records including inpatient consultation notes, discharge summaries, medication charts, pathology, radiology and endoscopy reports were then manually reviewed to confirm the diagnosis and collect the data. We used the diagnosis of non-alcoholic fatty liver disease (NAFLD) and non-alcoholic steatohepatitis (NASH) for patients as it was the accepted definition at the time of admission in each year of interest.
The diagnosis of cirrhosis was confirmed from medical records using a combination of clinical findings, pathology results and radiology reports.
Patients with cirrhosis admitted for management of the following decompensated liver events were included for analysis: Hepatic encephalopathy, hepatorenal syndrome-acute kidney injury, variceal haemorrhage, ascites and hepatic hydrothorax, spontaneous bacterial peritonitis, and biochemical decompensation/jaundice with a bilirubin of > 100 μmol/L. While not strictly considered a decompensating cirrhosis event, significant episodes of non-variceal bleeding requiring admission including gastric antral vascular ectasia, portal hypertensive gastropathy, peptic ulcer disease, other upper gastrointestinal pathology, and non-upper gastrointestinal bleeds were also included for analysis given the role of the liver in haemostasis.
Planned admissions such as for gastroscopy for variceal surveillance and for treatment of HCC (e.g., for trans-arterial chemoembolization) were excluded as the primary aim was to look for admissions for decompensating liver events.
Demographic variables recorded included age, gender, etiology of liver disease and severity of liver disease on admission as measured by Child-Pugh score and model for end-stage liver disease (MELD) score. The prospective liver transplant database maintained by the Liver Transplant Unit was accessed to identify patients undergoing liver transplantation, the etiology of liver disease and indication for transplant.
The primary outcome was the reason for and number of hospital admissions for complications of decompensated cirrhosis in 2012 and 2019 to determine the trends of complications of different etiologies of liver disease.
Secondary outcomes included total number of admissions and decompensating episodes during the year, as well as the number, indication for and etiology of patients undergoing orthotopic liver transplant (OLTx) in each year. A sub-analysis was also performed on the cohort of HCV patients to compare rates of sustained virological response, reasons for admission, and severity of liver disease (Child-Pugh class, MELD) in the pre- and post-DAA eras.
Descriptive statistics were used to present the demographic data of the patients. Quantitative data are expressed as median [interquartile range (IQR)] unless otherwise specified. Categorical variables are presented as n (%). Continuous and categorical data were compared using the Wilcoxon Rank Sum (Mann-Whitney) test and χ2 test, respectively. Incident rate ratios were calculated from count data and compared with the χ2 test for difference using the epiR package[9]. Statistical analysis was conducted using R v4.4.0[10].
Ethical approval for the study was granted by the Austin Health Research Ethics Committee (approval No. Audit/20/Austin/129) and the study was conducted in accordance with the Declaration of Helsinki and the Declaration of Istanbul.
2012 cohort: One hundred and seventy-eight patients with cirrhosis were admitted with the complications of interest. The median age of this cohort was 58 ± 11 years, and the majority were male (n = 123, 69%). The most common etiologies of underlying liver disease were alcohol (n = 65, 37%), chronic hepatitis C (n = 59, 33%), NAFLD (n = 16, 9%), autoimmune liver disease (n = 15, 8.4%), and chronic hepatitis B (n = 14, 7.9%).
Most patients had advanced liver disease, as reflected by Child-Pugh score, with 54% (n = 97) of patients being classified as Child-Pugh C, 42% as Child-Pugh B (n = 74), and only 3.9% (n = 7) as Child Pugh A. This is also reflected by a median MELD score of 17 (IQR: 13-22), and median MELD-Na score of 20 (16-26) at the time of their first admission.
2019 cohort: One hundred and ninety-eight patients were admitted with cirrhotic complications in 2019. The median age of this cohort was 59 ± 12 years, and 65% were male (n = 128). Alcohol was the most common cause of liver disease (n = 95, 48%), followed by NAFLD (n = 36, 18%), HCV (n = 30, 15%), autoimmune liver disease (n = 12, 6.1%) and chronic hepatitis B (n = 9, 4.5%).
Almost half of the patients were classified as Child Pugh C (n = 92, 46%), followed closely by Child Pugh B (n = 91, 46%), with Child Pugh A patients making up only 7.6% (n = 15) of the cohort. The median MELD score was 18 (13-22), and the median MELD-Na was 21 (15-25.5). The baseline demographics for each cohort are listed in Table 1.
| Characteristic | 2012 (n = 178) | 2019 (n = 198) | P value |
| Gender | 0.4 | ||
| Male | 123 (69) | 128 (65) | |
| Female | 55 (31) | 70 (35) | |
| Age | 57 (51, 66) | 60 (51, 67) | 0.4 |
| Etiology of liver disease | < 0.001 | ||
| Alcohol | 65 (36.5) | 95 (48) | |
| Autoimmune | 15 (8.4) | 12 (6.1) | |
| HBV | 14 (7.9) | 9 (4.5) | |
| HCV | 59 (33.1) | 30 (15.2) | |
| NASH | 16 (9) | 36 (18.2) | |
| Child-Pugh score | 0.2 | ||
| A | 7 (3.9) | 15 (7.6) | |
| B | 74 (41.6) | 91 (46) | |
| C | 97 (54.5) | 92 (46) | |
| MELD score | 17 (13, 22) | 18 (13, 22) | 0.6 |
There was a higher total number of admissions in 2012 compared to 2019 (969 vs 690), however the median number of admissions per patient was the same between the two cohorts (P = 0.2), and there was no statistically significant difference in the reason for first presentation (P = 0.4). There were a total of 1165 decompensating liver events in 2012 compared to 895 in 2019. The reasons for inpatient hospitalisation are presented in Table 2.
| 2012 | 2019 | P value | |
| Total number of admissions | 969 | 690 | |
| Median number of admissions per patient, n (IQR) | 2 (1, 4) | 2 (1, 3) | 0.2 |
| Reason for first presentation of the year | 0.4 | ||
| Ascites/hepatic hydrothorax | 72 (40) | 75 (38) | |
| Hepatic encephalopathy | 48 (27) | 54 (27) | |
| Bleeding | 25 (14) | 36 (18) | |
| Hepatorenal syndrome-acute kidney injury | 10 (5.6) | 7 (3.5) | |
| Spontaneous bacterial peritonitis | 10 (5.6) | 5 (2.5) | |
| Other | 13 (7.3) | 21 (11) | |
| Total number of decompensating liver events | Total n = 1165 | Total n = 895 | |
| Ascites | 760 | 474 | |
| Hepatic hydrothorax | 27 | 29 | |
| Hepatic encephalopathy | 194 | 139 | |
| Variceal bleeding | 47 | 40 | |
| Non-variceal bleeding | 35 | 73 | |
| Hepatorenal syndrome-acute kidney injury | 51 | 50 | |
| Spontaneous bacterial peritonitis | 28 | 45 | |
| Jaundice | 23 | 45 |
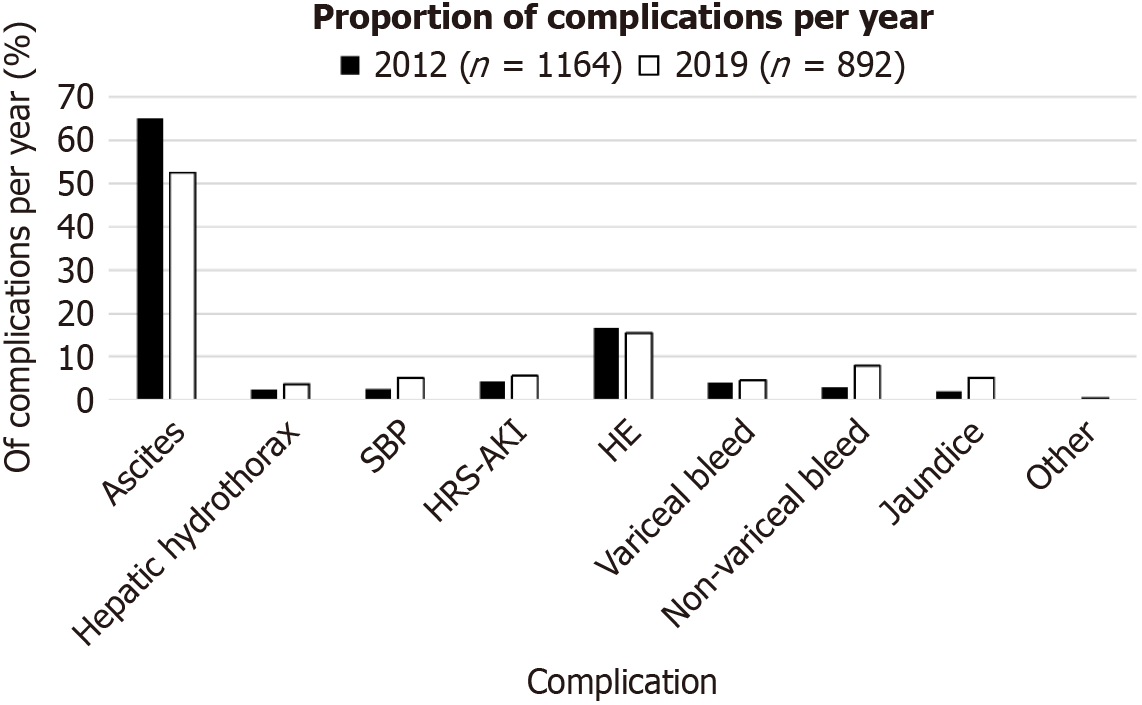
The overall incidence for each complication was different between the two cohorts, with a higher rate of admissions with ascites [incidence rate ratio (IRR): 1.80 (IQR: 1.60-2.02)] and hepatic encephalopathy [IRR: 1.56 (IQR: 1.25-1.96)] (both P < 0.0001) in 2012 compared to 2019, while the incidence of admissions with non-variceal bleeding [IRR: 0.54, (IQR: 0.35-0.83); P = 0.003] and jaundice [IRR: 0.57, (IQR 0.33-0.97); P = 0.03] was higher in 2019 compared to 2012. The incidence rate ratios for each complication are listed in Table 3. The proportion of admissions by complications are shown in Figure 1.
| Complication | Incidence rate ratio (IQR) | P value |
| Ascites | 1.80 (1.60, 2.02) | < 0.0001 |
| Hepatic hydrothorax | 1.04 (0.59, 1.83) | 0.87 |
| Hepatic encephalopathy | 1.56 (1.25, 1.96) | < 0.0001 |
| Variceal bleeding | 1.32 (0.85, 2.06) | 0.2 |
| Non-variceal bleeding | 0.54 (0.35, 0.83) | 0.003 |
| Hepatorenal syndrome-acute kidney injury | 1.14 (0.76, 1.72) | 0.5 |
| Spontaneous bacterial peritonitis | 0.70 (0.42, 1.14) | 0.13 |
| Jaundice | 0.57 (0.33, 0.97) | 0.03 |
| Other | 2.80 (0.45, 29.43) | 0.20 |
A further Poisson multivariate regression analysis was performed to assess for factors associated with increasing incidence of admissions and is included in Supplementary Table 1. There was a 35% lower incidence rate of admission in 2019 compared to 2012, and female gender was associated with a 17% lower incidence rate of admission compared to male gender.
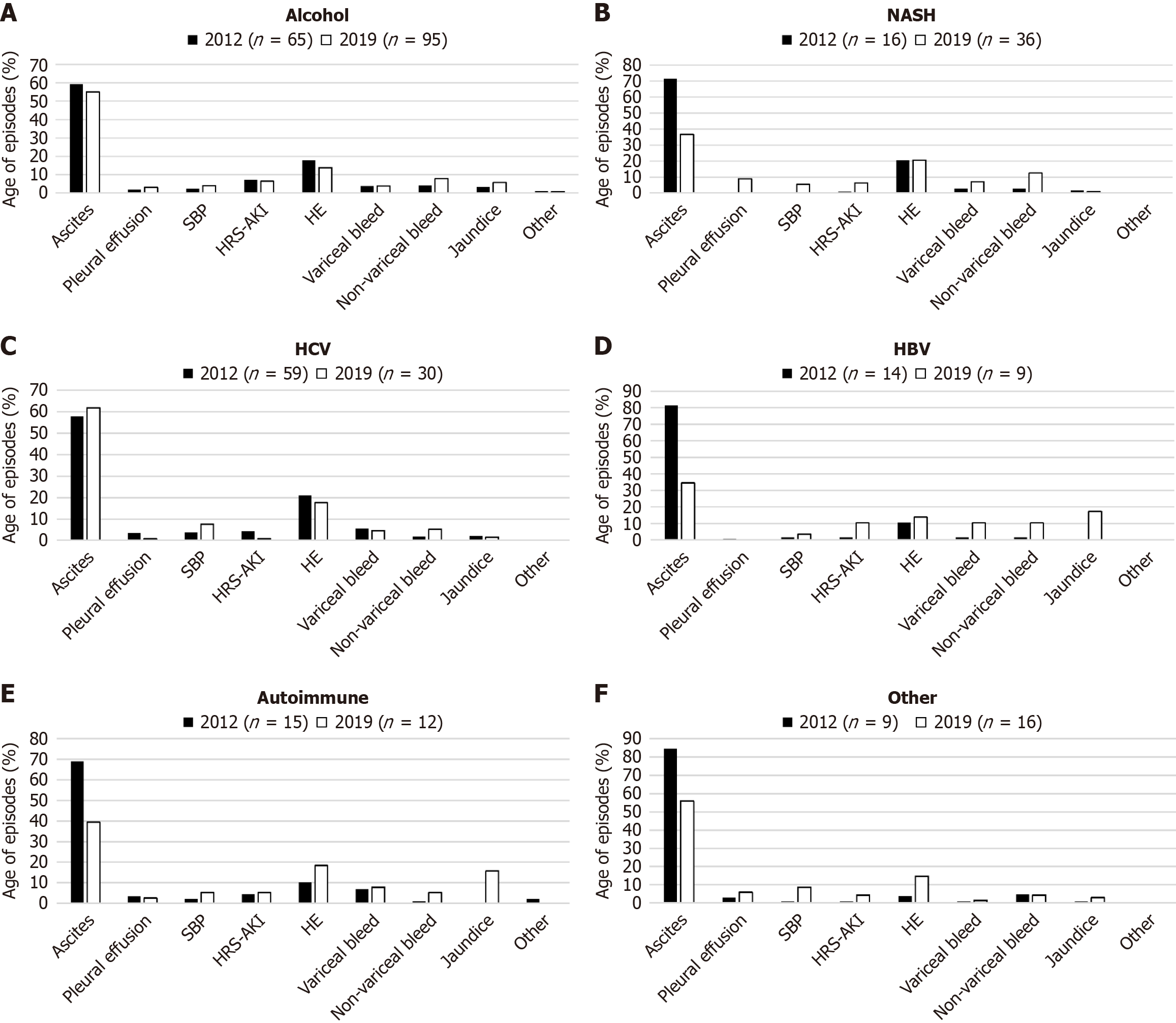
Complications by etiology: The proportion of admissions by etiology of liver disease differed between the two cohorts (P < 0.001) (Table 1), and the complications of cirrhosis of each etiology also differed (Figure 2). The proportion of episodes of ascites decreased for all etiologies except HCV, while episodes of non-variceal bleeding increased. The causes of the non-variceal bleeds are listed in Table 4. Of the twenty-three patients in 2012 and 51 patients in 2019 with non-variceal bleeds, one patient (4.3%) was taking antiplatelet or anticoagulation therapy in 2012 compared to 27.5% (n = 14) of patients in 2019.
| 2012 | 2019 | |
| Patients admitted with a significant non-variceal haemorrhage | 23 | 51 |
| Etiology of liver disease | ||
| Alcohol | 9 (39.1) | 29 (56.9) |
| Autoimmune | 1 (4.3) | 2 (3.9) |
| HBV | 2 (8.7) | 1 (2) |
| HCV | 6 (26.1) | 7 (13.7) |
| NASH | 2 (8.7) | 10 (19.6) |
| Other | 3 (13) | 2 (3.9) |
| Antiplatelet or anticoagulation therapy | ||
| Yes | 1 (4.3) | 14 (27.5) |
| No | 22 (95.7) | 37 (72.5) |
| Patients taking antiplatelet/anticoagulant therapy by etiology of liver disease | ||
| Alcohol | 0 | 4 |
| Autoimmune | 0 | 0 |
| HBV | 0 | 1 |
| HCV | 0 | 3 |
| NASH | 1 | 6 |
| Other | 0 | 0 |
| Cause of non-variceal bleeding (number of episodes) | ||
| GAVE/portal hypertensive gastropathy | 24 | 32 |
| Peptic ulcer | 5 | 11 |
| Other upper gastrointestinal pathology | 3 | 7 |
| Non-upper gastrointestinal bleed | 3 | 23 |
Transplants: Thirty patients in 2012 and 24 patients in 2019 underwent OLTx for cirrhotic non-HCC indications. The etiology of liver disease for patients receiving OLTx was significantly different between the two cohorts (P = 0.006) (Table 5).
| Etiology | 2012 (n = 30) | 2019 (n = 24) |
| Alcohol | 4 (13.3) | 11 (45.8) |
| Autoimmune | 8 (26.7) | 3 (12.5) |
| HBV | 0 (0) | 1 (4.2) |
| HCV | 13 (43.3) | 2 (8.3) |
| NASH | 3 (10) | 4 (16.7) |
| Other | 2 (6.7) | 3 (12.5) |
HCV patients by cohort: In 2012, 59 patients with HCV required inpatient admission, compared to 30 in 2019. There was no significant difference in the age of the patients admitted in 2012 (55, 51-63 years) compared to those admitted in 2019 (60, 52-66 years), P = 0.3, nor was there a significant difference in the gender of the two cohorts (P = 0.8).
Sustained virological response status: Of the 59 patients with HCV admitted in 2012, only one patient (1.7%) had achieved sustained virological response, compared to 68.6% (n = 24) of the 35 patients with HCV admitted in 2019 (P < 0.001).
Admissions: The reason for first admission was similar between the 2 cohorts, with fluid issues remaining the most common reason (42% in 2012 vs 40% in 2019), followed by hepatic encephalopathy (31% vs 30%), and bleeding (17% vs 23%), P > 0.9. Ascites remained the most common complication requiring hospitalization throughout the year in both cohorts (Figure 2).
This study describes for the first time not only the overall shift in etiology of liver disease requiring hospitalisation, but also the shift in phenotype of decompensated cirrhosis presenting to a major tertiary centre in Australia. We report that post-DAA availability we are seeing reduced rates of hospitalisation for decompensated cirrhosis due to HCV, and more cases of MASLD. We are also seeing reduced hospitalisations for fluid-related issues and a greater proportion of admissions for non-variceal gastrointestinal bleeding and hepatic encephalopathy.
Findings pertaining to etiology of liver disease are similar to that reported elsewhere and are due to widespread availability of DAA therapy. Treatment of the underlying etiology of liver disease is known to reduce rates of progression to decompensated liver disease[11], and it is therefore not surprising that the proportion of patients with chronic HCV-related cirrhotic patients being admitted to our centre with decompensated cirrhosis decreased by 54% from 33.1% in the pre-DAA era in 2012 compared to 15.4% in 2019. Similar to our findings, Alavi et al[12] also found a sharp decline in decompensated cirrhosis in patients with chronic HCV, however this group did not specify which complications of decompensation were the major causes of morbidity and mortality in this population. A significantly lower proportion of patients with HCV underwent liver transplantation at our centre in 2019 compared to 2012 (8.3% vs 43.3%), reflecting that successful treatment of HCV leads to reduced demand for this resource-limited treatment which is experiencing an overall increase in demand due to other etiologies of cirrhosis. This trend has also been noted in Europe[13] and the United States[14], and highlights the importance of diagnosis and successful treatment of patients with HCV. The incidence of both new HCV infections and HCV-related deaths are falling globally[15,16], however HCV continues to remain a significant cause of chronic liver disease[6,17] and there is still significant work to be done in order to meet the World Health Organization target of eliminating viral hepatitis by 2030.
The shift in etiology of cirrhosis seen at our centre, with the predominance of alcohol-related cirrhosis and rise of MASLD is consistent with what has been documented at other centres in Australia[12] and around the world[18,19]. Alcohol-related liver disease continues to remain the most common cause of liver disease requiring admission, with the proportion of alcohol-related liver disease actually increasing over time. This reinforces the further work that needs to be done by governments at a society-level, including education and community-support programs to help people reduce harmful levels of alcohol intake. NASH-related cirrhosis has also increased in prevalence, accounting for 18.2% of admissions in 2019 compared to 9% in 2012, and 16.7% of liver transplants compared to 10% in 2012. The estimated global prevalence of NASH is approximately 30%[20], and is projected to rise[21]. This trend is particularly pertinent given the prevalence of the metabolic syndrome all around the world and the implications for the potential significant increase in NASH-associated liver disease and resultant burden on healthcare resources. As yet, the only United States Food and Drug Administration-approved treatment for MASLD is resmetirom, which is currently contraindicated in decom
While the shift in etiology of liver disease presenting to our centre is consistent with global trends, there is only limited data regarding the phenotypes of decompensated cirrhosis. Only one other study, to our knowledge, has assessed both the trends in etiology and the admissions by complication of cirrhosis, with Gu et al[18] examining both the etiology and complications of cirrhosis of patients admitted to German hospitals between 2006 and 2018. Consistent with our findings, Gu et al[18] recorded alcohol as the most common etiology of cirrhosis requiring hospitalisation, with a significant decrease in HCV and increase in MASLD-related cirrhosis. They also noted an increase in the prevalence of ascites, infections and hepatic encephalopathy, and a decrease in the prevalence of both variceal and gastrointestinal bleeding.
Consistent with the German findings, ascites remains the most common complication of cirrhosis requiring inpatient management at our centre, however unlike in Germany, the incidence rate decreased in 2019 compared to 2012. Tr
Contrary to the trends in Germany in which rates of hepatic encephalopathy increased and gastrointestinal bleeding decreased, we found an increased incidence of non-variceal bleeding, and decreased incidence of hepatic encephalopathy. The reduction in encephalopathy may relate to improved management and increased access to therapies such as rifaximin, and is a positive finding given the poor prognosis conferred[25]. The increase in non-variceal bleeding may relate to the rise in prevalence of MASLD, which is often associated with comorbidities such as diabetes and car
The strength of this study is that it provides epidemiological information looking at trends of etiology of cirrhosis, and rates of each of the complications of cirrhosis, providing information about the clinical phenotype of different etiologies of liver disease. This is helpful in planning future resource allocation and utilisation, for example allocating day procedure units for management of large volume paracentesis, as well as providing information on a larger public health scale to identify areas of concern that would benefit from interventions at a society level. As 30-day hospital readmission rates have been documented as high as 46%[26], it also highlights the importance of devising novel ways of managing these patients in the community such as home terlipressin or the LivR Well program, which provides hospital-level care in the home setting to patients with acute on chronic liver failure, including medical, nursing and allied health care through a combination of home visits, telehealth and ambulatory clinics at the hospital[27,28]. Every effort should also be made to aggressively uptitrate and optimise medical therapy in the outpatient setting, including non-selective beta-blocker therapy to reduce portal hypertension and diuretic therapy to manage ascites and fluid overload, and to perform medication reviews and reconciliation to ensure patients are not maintained on medications unnecessarily, including antiplatelet and anticoagulant therapy. Timely access to outpatient clinics is often challenging, however further resourcing for outpatient management may reduce the need for inpatient admission.
Limitations of the study include the relatively small numbers at a single tertiary centre who were admitted predominantly for cirrhosis-related complications. However, the results are in keeping with other studies both in Australia and around the world describing falling admissions for HCV-related complications and a rising prevalence of MASLD. Despite small numbers, we were still able to show a significant difference in cirrhosis phenotype between eras. We purposefully excluded HCC-related presentations, as our centre is the state-wide liver transplant centre in our region, and many patients with HCC seen at our centre are from outside the regular hospital catchment area, artificially inflating the numbers of HCC presentations. Other hospitals in our state have also developed their own HCC services in recent years, reducing the number of referrals to our centre, and further altering the number of HCC presentations to our centre. Future studies should comprise multi-centre datasets collected prospectively to better identify the needs of the modern-day cohort of patients suffering from cirrhosis.
In conclusion, this study confirms previously reported changes in the etiology of liver disease, with falling rates of HCV since the institution of effective DAA therapy and increasing metabolic-associated steatotic liver disease. Alcohol remains a major contributor to cirrhosis. We describe for the first time in Australia that there is also a shift in the phenotype of decompensated cirrhosis, with reducing rates of ascites and hepatic encephalopathy and increasing non-variceal bleeding compared to previous years, that may be due to the shifting etiology of liver disease. Further work is required to better understand the needs of an increasingly complex population suffering decompensated cirrhosis at both a clinical and public health level to allow health systems to cope with the expected growing demand from this patient group.
| 1. | GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789-1858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10625] [Cited by in RCA: 9428] [Article Influence: 1178.5] [Reference Citation Analysis (13)] |
| 2. | Huang DQ, Terrault NA, Tacke F, Gluud LL, Arrese M, Bugianesi E, Loomba R. Global epidemiology of cirrhosis - aetiology, trends and predictions. Nat Rev Gastroenterol Hepatol. 2023;20:388-398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 666] [Cited by in RCA: 563] [Article Influence: 187.7] [Reference Citation Analysis (1)] |
| 3. | Australian Government Department of Health, Disability and Ageing. Fourth National Hepatitis C Strategy 2014-2017. [cited 3 August 2025]. Available from: http://www.health.gov.au/internet/main/publishing.nsf/content/ohp-bbvs-hepc2014. |
| 4. | Australia’s Progress towards Hepatitis C Elimination: Annual Report 2023. [cited 3 August 2025]. Available from: https://www.burnet.edu.au/media/tahd41iz/australias-progress-towards-hepatitis-c-elimination-annual-report-2023.pdf. |
| 5. | Howell J, Majumdar A, Fink MA, Byrne M, McCaughan G, Strasser SI, Crawford M, Hodgkinson P, Stuart KA, Tallis C, Chen J, Wigg A, Jones R, Jaques B, Jeffrey G, Adams L, Wallace MC, Munn S, Gane E, Thompson AJ, Gow P. Turning the Tide on Hepatitis C Virus-Related Liver Transplantation: The Return on Investment in Hepatitis C Virus Treatment in Australia and New Zealand. Liver Transpl. 2022;28:236-246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (3)] |
| 6. | Younossi ZM, Wong G, Anstee QM, Henry L. The Global Burden of Liver Disease. Clin Gastroenterol Hepatol. 2023;21:1978-1991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 335] [Article Influence: 111.7] [Reference Citation Analysis (6)] |
| 7. | Teng ML, Ng CH, Huang DQ, Chan KE, Tan DJ, Lim WH, Yang JD, Tan E, Muthiah MD. Global incidence and prevalence of nonalcoholic fatty liver disease. Clin Mol Hepatol. 2023;29:S32-S42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 576] [Cited by in RCA: 509] [Article Influence: 169.7] [Reference Citation Analysis (3)] |
| 8. | Younossi ZM, Stepanova M, Ong J, Trimble G, AlQahtani S, Younossi I, Ahmed A, Racila A, Henry L. Nonalcoholic Steatohepatitis Is the Most Rapidly Increasing Indication for Liver Transplantation in the United States. Clin Gastroenterol Hepatol. 2021;19:580-589.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 437] [Article Influence: 87.4] [Reference Citation Analysis (2)] |
| 9. | epiR: Tools for the Analysis of Epidemiological Data. [cited 3 August 2025]. Available from: https://cran.r-project.org/web/packages/epiR/. |
| 10. | The R Project for Statistical Computing. [cited 3 August 2025]. Available from: https://www.r-project.org/. |
| 11. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. J Hepatol. 2018;69:406-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2259] [Cited by in RCA: 2068] [Article Influence: 258.5] [Reference Citation Analysis (6)] |
| 12. | Alavi M, Law MG, Valerio H, Grebely J, Amin J, Hajarizadeh B, Selvey C, George J, Dore GJ. Declining hepatitis C virus-related liver disease burden in the direct-acting antiviral therapy era in New South Wales, Australia. J Hepatol. 2019;71:281-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 13. | Müller PC, Kabacam G, Vibert E, Germani G, Petrowsky H. Current status of liver transplantation in Europe. Int J Surg. 2020;82S:22-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 69] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 14. | Cholankeril G, Ahmed A. Alcoholic Liver Disease Replaces Hepatitis C Virus Infection as the Leading Indication for Liver Transplantation in the United States. Clin Gastroenterol Hepatol. 2018;16:1356-1358. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 216] [Cited by in RCA: 208] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 15. | World Health Organization. Global hepatitis report 2024: action for access in low-and middle-income countries. [cited 3 August 2025]. Available from: https://www.who.int/publications/i/item/9789240091672. |
| 16. | Cui F, Blach S, Manzengo Mingiedi C, Gonzalez MA, Sabry Alaama A, Mozalevskis A, Séguy N, Rewari BB, Chan PL, Le LV, Doherty M, Luhmann N, Easterbrook P, Dirac M, de Martel C, Nayagam S, Hallett TB, Vickerman P, Razavi H, Lesi O, Low-Beer D. Global reporting of progress towards elimination of hepatitis B and hepatitis C. Lancet Gastroenterol Hepatol. 2023;8:332-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 279] [Cited by in RCA: 228] [Article Influence: 76.0] [Reference Citation Analysis (5)] |
| 17. | Moon AM, Singal AG, Tapper EB. Contemporary Epidemiology of Chronic Liver Disease and Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:2650-2666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 923] [Cited by in RCA: 836] [Article Influence: 139.3] [Reference Citation Analysis (6)] |
| 18. | Gu W, Hortlik H, Erasmus HP, Schaaf L, Zeleke Y, Uschner FE, Ferstl P, Schulz M, Peiffer KH, Queck A, Sauerbruch T, Brol MJ, Rohde G, Sanchez C, Moreau R, Arroyo V, Zeuzem S, Welsch C, Trebicka J. Trends and the course of liver cirrhosis and its complications in Germany: Nationwide population-based study (2005 to 2018). Lancet Reg Health Eur. 2022;12:100240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (2)] |
| 19. | Devarbhavi H, Asrani SK, Arab JP, Nartey YA, Pose E, Kamath PS. Global burden of liver disease: 2023 update. J Hepatol. 2023;79:516-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1738] [Cited by in RCA: 1417] [Article Influence: 472.3] [Reference Citation Analysis (5)] |
| 20. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1273] [Article Influence: 636.5] [Reference Citation Analysis (2)] |
| 21. | Estes C, Razavi H, Loomba R, Younossi Z, Sanyal AJ. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67:123-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2053] [Cited by in RCA: 1882] [Article Influence: 235.3] [Reference Citation Analysis (7)] |
| 22. | Terbah R, Testro AG, Hoermann R, Majumdar A, Chapman B, Gow PJ, Sinclair M. Continuous home terlipressin infusion increases handgrip strength and reduces ascites-A prospective randomized crossover study. Hepatology. 2024;80:605-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 17] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 23. | Gow PJ, Sinclair M, Thwaites PA, Angus PW, Chapman B, Terbah R, Testro AG. Safety and efficacy of outpatient continuous terlipressin infusion for the treatment of portal hypertensive complications in cirrhosis. Eur J Gastroenterol Hepatol. 2022;34:206-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Gow PJ, Ardalan ZS, Vasudevan A, Testro AG, Ye B, Angus PW. Outpatient Terlipressin Infusion for the Treatment of Refractory Ascites. Am J Gastroenterol. 2016;111:1041-1042. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 25. | Bohra A, Worland T, Hui S, Terbah R, Farrell A, Robertson M. Prognostic significance of hepatic encephalopathy in patients with cirrhosis treated with current standards of care. World J Gastroenterol. 2020;26:2221-2231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 26. | Vaz K, Tan K, Chew M, Crawford J, Ma R, Grace J, Gow P, Sinclair M, Testro A. Rate of early hospital readmission amongst cirrhotic patients is high in Australia: experience from a single liver transplant centre. Intern Med J. 2022;52:2086-2095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 27. | Ngu NL, Saxby E, Worland T, Anderson P, Stothers L, Figredo A, Hunter J, Elford A, Ha P, Hartley I, Roberts A, Seah D, Tambakis G, Liew D, Rogers B, Sievert W, Bell S, Le S. A home-based, multidisciplinary liver optimisation programme for the first 28 days after an admission for acute-on-chronic liver failure (LivR well): a study protocol for a randomised controlled trial. Trials. 2022;23:744. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 28. | Ngu NLY, Saxby E, Worland T, Anderson P, Stothers L, Hunter J, Elford AT, Ha P, Hartley I, Roberts A, Seah D, Tambakis G, Connoley D, Figredo A, Ratnam D, Liew D, Rogers B, Sievert W, Bell S, Le S. A Nonrandomized Pilot Study to Investigate the Acceptability and Feasibility of LivR Well: A Multifaceted 28-Day Home-Based Liver Optimization Program for Acute-on-Chronic Liver Failure. Gastro Hep Adv. 2025;4:100567. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |