Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.113366
Revised: September 30, 2025
Accepted: January 8, 2026
Published online: March 27, 2026
Processing time: 215 Days and 15.2 Hours
Chronic hepatitis C virus (HCV) infection is a significant global health concern, often associated with fatigue that influences patients' quality of life.
To assess prevalence of significant or severe fatigue in cases with HCV-related chronic liver disease and to detect factors independently related to fatigue using the multidimensional fatigue inventory (MFI-20).
A cross-sectional study was conducted at the National Liver Institute, Menoufia University, Egypt, including 300 patients with chronic HCV infection. Participants with significant comorbidities or conditions impacting fatigue were excluded. Recruited participants completed a structured questionnaire on demographics, clinical and laboratory data with assistance from qualified physicians. Fatigue was evaluated in five domains (physical, general, reduced activity, decreased moti
Significant/severe fatigue was prevalent in 27.3% of chronic HCV patients. A higher MELD score, presence of a history of gastrointestinal bleeding, ascites, lack of antiviral treatment response, hepatic encephalopathy, antiviral treatment within the past year, and longer HCV infection were significantly associated with significant/severe fatigue. Decompensated liver disease, inter
Fatigue is highly prevalent among HCV-related chronic liver disease patients. Using the MFI-20, we revealed that fatigue severity is independently associated with liver disease severity, antiviral treatment, and infection duration. This highlights the necessity of regular fatigue assessment in clinical practice.
Core Tip: Chronic hepatitis C virus (HCV) infection is often associated with fatigue. We recruited 300 chronic HCV patients. Fatigue has been evaluated with multidimensional fatigue inventory-20 (validated tool to assess different dimensions of fatigue in this population. Significant/severe fatigue was prevalent in 27.3% of HCV patients. Decompensated liver disease, intermediate/high-risk fibrosis, < 1 year duration of last dose of antiviral treatment, and HCV infection duration were independent risk factors for fatigue. Fatigue is a widespread and complex symptom in cases with chronic HCV, closely linked to liver disease severity, antiviral treatment, and infection duration.
- Citation: Tahoon MA, Elkhadry SW, Abdelsameea E, Ashour R. Fatigue assessment in Egyptian patients with hepatitis C virus-related chronic liver disease: Single center study. World J Hepatol 2026; 18(3): 113366
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/113366.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.113366
Chronic liver disease (CLD) affects over 1.5 billion people worldwide[1]. According to the World Health Organization, 304 million people worldwide have viral hepatitis B and C in 2022[2]. According to estimates, 1.3 million people died from viral hepatitis in 2022, with hepatitis C virus (HCV) accounting for 17% of these fatalities. There were 2.2 million new viral hepatitis infections in 2022, including nearly 1.0 million new HCV infections[2]. About 160 million individuals worldwide have been estimated to be chronically infected with HCV, however, the majority remain naïve regarding their condition[3]. HCV infection represents a major public health challenge affecting both developed and developing coun
Patients with CLD frequently complain of fatigue, which affects 50% to 85% of them. It mostly interferes with physical activity, family life, and the ability to work[10]. HCV infection may have an impact on the brain, according to rising evidence. Regardless of their liver disease stage or virus replication rate, roughly half of HCV-infected individuals report having persistent fatigue[11]. About one-third of patients still experience fatigue even after receiving effective antiviral medication. Additionally, a lot of patients experience memory, focus, and concentration problems, some even express depression. Deficits in verbal learning capacity and attention were also identified even in those with normal liver function[10,11]. A generalized sense of exhaustion, weakness, persistent tiredness, and reduced energy are among the most common psychosomatic symptoms of cancer and chronic illness patients. In chronic HCV patients, fatigue represents the most common clinical manifestation, significantly impairing quality of life and limiting daily activities[12,13].
Fatigue is a complex, multidimensional and non-specific phenomenon characterized by self-reported tiredness or lack of energy, with a relevant impact on daily activities, social life and work performance[14,15]. Patients often describe fatigue as sleepiness or an uncontrollable need to sleep. While the fatigue severity scale (FSS), which is a common tool used to evaluates the degree of fatigue and how it affects daily functioning[16], fatigue is documented as a multidimensional concept and instruments assessing fatigue should therefore cover several different dimensions. The 20-item multidimensional fatigue inventory (MFI-20) evaluates five distinct dimensions: General fatigue, physical fatigue, reduced activity, reduced motivation, and mental fatigue[17,18]. This is arguably the most widely used fatigue qu
Although psychosomatic symptoms have been described in other chronic illnesses, in HCV patients these symptoms substantially impair daily functioning and quality of life, emphasizing the importance of comprehensive fatigue ass
Therefore, our study was aimed to assess the prevalence of significant or severe fatigue in several dimensions in Egyptian patients with HCV-related CLD using the validated Arabic version of the MFI-20[20], and to identify inde
This cross-sectional investigation has been performed at the National Liver Institute (NLI), Menoufia University, Egypt. We aimed to assess the prevalence of significant or severe fatigue in patients with HCV-related CLD using the validated Arabic version of the MFI-20[20]. Also identify independent factors associated with fatigue and investigate its correlation with disease severity, as assessed by fibrosis (FIB) stage, Child-Pugh score, and MELD score. Additionally, investigated any associations between fatigue and treatment with direct-acting antiviral (DAA) drugs was done.
Participants: Our study included 300 cases diagnosed with chronic hepatitis C infection (termed as positive serology by at least a second-generation enzyme linked immunosorbent assay test). The inclusion criteria were adults aged eighteen and older, diagnosed with chronic HCV infection for at least six months, and willing to participate with informed consent. Exclusion criteria were presence of hepatocellular carcinoma, co-infections with hepatitis B (definite by the presence of hepatitis B surface antigen) or D virus, or human immunodeficiency virus; evidence of autoimmune hepatitis; heavy alcohol use; pregnancy; prior interferon treatment; significant comorbidities such as heart failure or chronic kidney disease; and psychiatric or cognitive disorders that could impair understanding of the questionnaire.
Sample size calculation: A sample size of approximately 217 patients has been estimated depend on 95% percent con
Eligible participants were randomly selected from the outpatient clinics at the NLI. After providing informed consent, they completed a structured questionnaire covering demographic information incorporating (gender, age, employment status, level of educational, smoking, and residence), clinical data [duration of HCV infection, treatment history, comorbid conditions such as diabetes and hypertension, presence of ascites, history of hepatic encephalopathy and history of gastrointestinal tract (GIT) bleeding], and laboratory data [total bilirubin (mg/dL), serum albumin (g/dL), international normalized ratio (INR), platelet count, serum creatinine (mg/dL), alanine aminotransferase (ALT) and aspartate aminotransferase (AST)]. The questionnaire was fulfilled by the aid of the trained physicians.
Liver dysfunction has been categorized based on the Child-Pugh (Child A, B, and C) and model for end stage liver disease (MELD) scores[22]. The MELD score has been computed using the following equation: MELD score = 3.78 × log (bilirubin, milligrams per deciliter) + 11.2 × log [INR + 9.57 × log (creatinine, milligrams per deciliter)] + 6.
The extent of liver FIB has been evaluated using the FIB-4 index[23]. FIB-4 = age (years) × AST (U/L)/{[platelets (PLT) 109/L] × ALT1/2 (U/L)}. The FIB-4 score is classified into various ranges to assess the severity of liver FIB: (1) FIB-4 < 1.45: Minimal risk of significant FIB (usually F0-F1); (2) FIB-4 1.45-3.25: Middle risk of significant FIB (possible F2-F3); and (3) FIB-4 > 3.25: Great risk of advanced cirrhosis or FIB (usually F4).
Fatigue severity has been assessed using subscales from the MFI[18]. The MFI-20 is a confirmed self-report instrument intended to assess fatigue through five subscales: General fatigue (items 1, 5, 12, 16), physical fatigue (items 2, 8, 14, 20), reduced activity (items 3, 6, 10, 17), reduced motivation (items 4, 9, 15, 18) and mental fatigue (items 7, 11, 13, 19). The domain of general fatigue is composed of the general statements about fatigue and reduced functioning, covering physical as well as psychological aspects of fatigue. Physical fatigue concerns physical feelings related to fatigue. Mental fatigue relates to cognitive functioning, such as concentration difficulties. Reduced activity subscale evaluates the impact of psychological and physical factors on the activity level. The lack of motivation to start an activity is reflected by the subscale of reduced motivation. The scale has an acceptable level of validity and internal consistency, and the Arabic version validity and reliability has been proved[20].
Scoring of MFI-20: Each subscale comprises four items with responses available on a five-point Likert scale (1 = yes, that is true; 5 = no, this is not true). The total score varies from 4 to 20 for each subscale and from 20 to 100 for the overall fatigue score, with a higher score signifying greater fatigue.
The participants were divided into non fatigue and significant or severe fatigue groups. Severe/significant fatigue as defined by the MFI-20 is typically characterized by high scores (≥ 75 percentile) reflecting significant impairment in general, physical, activity-related, motivational, and mental aspects of fatigue[24,25].
Ethical consideration: The NLI ethics committee approved the investigation under protocol number (IRB number 00632/2024) (NLI IRB 00014014). Each participant had to sign an informed consent form before they could begin. They were all made aware of the goals of the investigation. We assured them that the information we collected from them would be kept private and they can withdraw from the study without obligation at any time.
Descriptive statistics have been utilized to describe the demographic and clinical attributes of the subjects. Continuous variables have been presented as mean ± SD for normally distributed data and as median with interquartile range for non-normally distributed information, while categorical variables have been reported as n (%). Comparative analyses have been conducted to discover parameters related to elevated fatigue levels. The Mann-Whitney test was utilized for continuous variables, whereas χ2 tests have been utilized for categorical ones. A multivariate linear regression analysis has been performed to identify the independent determinants of fatigue across various dimensions, while controlling for relevant confounders. CI set to 95% with odds ratios (OR) were used to express multivariable studies. The Spearman correlation test has been utilized to assess the association among two continuous variables. The significance criterion for all tests was established at P < 0.05[26].
A total of 300 chronic HCV cases have been involved in the investigation. The mean age of the participants was 57.3 years (SD ± 10.6), with a median age of 59 years. The age distribution was as follows: 22% were under 45 years, 36.7% were between 45 years and 59 years, 41.3% were 60 years or more. Most participants were male (63.3%), married (86%) and had secondary education (38.7%). About 56% of the participants resided in rural areas, and 72% were non-smokers. Half of the participants (50%) had comorbidities, including diabetes mellitus and/or hypertension (Table 1).
| Variables | |
| Age (years) | 57.3 ± 10.6, 59.0 (15.0) |
| < 45 years | 66 (22.0) |
| 45-59 years | 110 (36.7) |
| ≥ 60 years | 124 (41.3) |
| Gender | |
| Male | 190 (63.3) |
| Female | 110 (36.7) |
| Marital status | |
| Single | 6 (2.0) |
| Married | 258 (86.0) |
| Divorced | 30 (10.0) |
| Widow | 6 (2.0) |
| Level of education | |
| Illiterate and primary education | 96 (32.0) |
| Secondary education | 116 (38.7) |
| Higher education | 88 (29.3) |
| Residence | |
| Rural | 168 (56.0) |
| Urban | 132 (44.0) |
| Smoking | |
| No | 216 (72.0) |
| Yes | 58 (19.3) |
| Ex smoker | 26 (8.7) |
| Co-morbidities (yes)1 | 150 (50.0) |
| Clinical stage | |
| Chronic liver disease | 52 (17.3) |
| Compensated | 174 (58.0) |
| Decompensated | 72 (24.0) |
| Child score (n = 248) | |
| A | 170 (68.5) |
| B | 28 (11.3) |
| C | 50 (20.2) |
| Ascites (yes) | 79 (26.3) |
| History of GIT bleeding (yes) | 56 (18.7) |
| History of hepatic encephalopathy (yes) | 51 (17) |
| HCV infection duration | |
| < 10 years | 159 (53.0) |
| ≥ 10 years | 141 (47.0) |
| Antiviral treatment (yes) | 262 (87.3) |
| The duration of last dose of antiviral treatment (n = 262) | |
| < 1 year | 6 (2.0) |
| ≥ 1 year | 256 (97.7) |
| Antiviral treatment response (n = 262) | |
| Response | 247 (94.3) |
| Failure | 15 (5.7) |
| MELD score | 9.2 (3.1) |
| ALT | 29.0 (19.3) |
| AST | 32.0 (24.8) |
| Total bilirubin (mg/dL) | 1.0 (0.6) |
| Albumin (g/dL) | 3.8 (0.7) |
| Creatinine (mg/dL) | 0.90 (0.3) |
| INR | 1.1 (0.1) |
| Platelet | 155.0 (140.0) |
Regarding the clinical stage, 58% had compensated liver disease, while 24% had decompensated liver disease and 17.3% of them presented with CLD. About 68.5% of them were classified as Child A, 11.3% as Child B, and 20.2% as Child C. Ascites was present in 26.3% of the participants and a history of gastrointestinal hemorrhage and hepatic encephalopathy has been noted in 18.7% and 17.0% of the participants, respectively. The median MELD score was 9.2 (interquartile range = 3.1) (Table 1).
The MFI-20 revealed that the total fatigue score for the study population was 62.5 (SD ± 3.8), with the following subscale scores: General fatigue (12.3 ± 1.13), physical fatigue (12.9 ± 1.7), decreased activity (12.6 ± 1.4), decreased motivation (12.9 ± 1.5), and mental fatigue (13.2 ± 1.1) (Table 2).
| MFI-20 | mean ± SD/median, interquartile range |
| General fatigue | 12.3 ± 1.13/12.0, 1.0 |
| Physical fatigue | 12.9 ± 1.7/13.0, 2.0 |
| Reduced activity | 12.6 ± 1.4/13.0, 2.0 |
| Reduced motivation | 12.9 ± 1.5/13.0, 2.0 |
| Mental fatigue | 13.2 ± 1.1/12.0, 1.0 |
| Total MFI score | 62.5 ± 3.8/63.0, 4.0 |
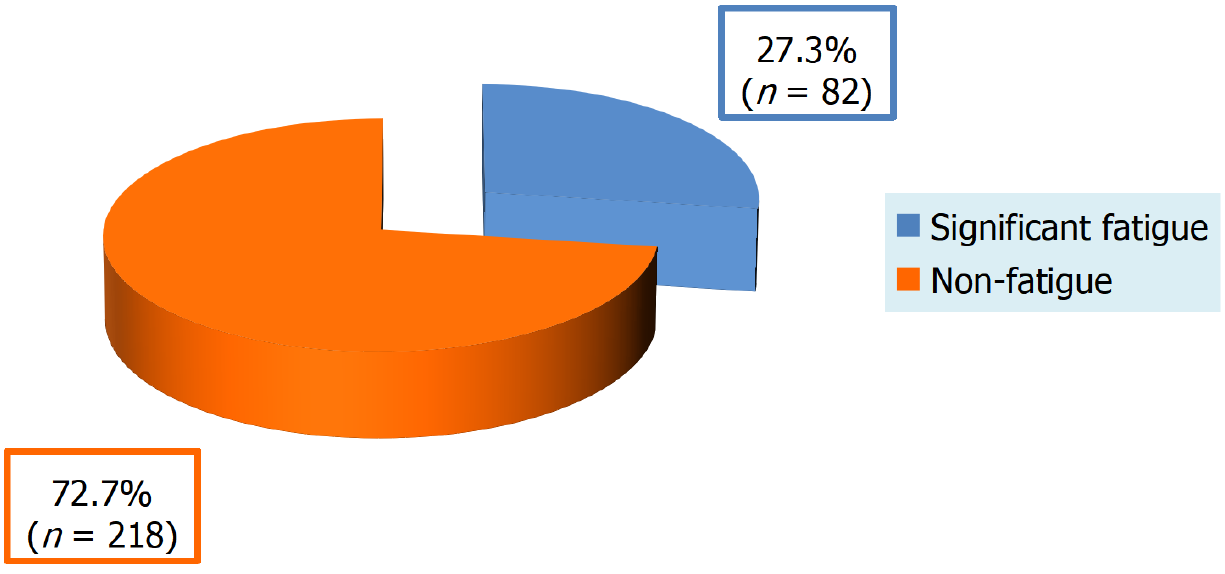
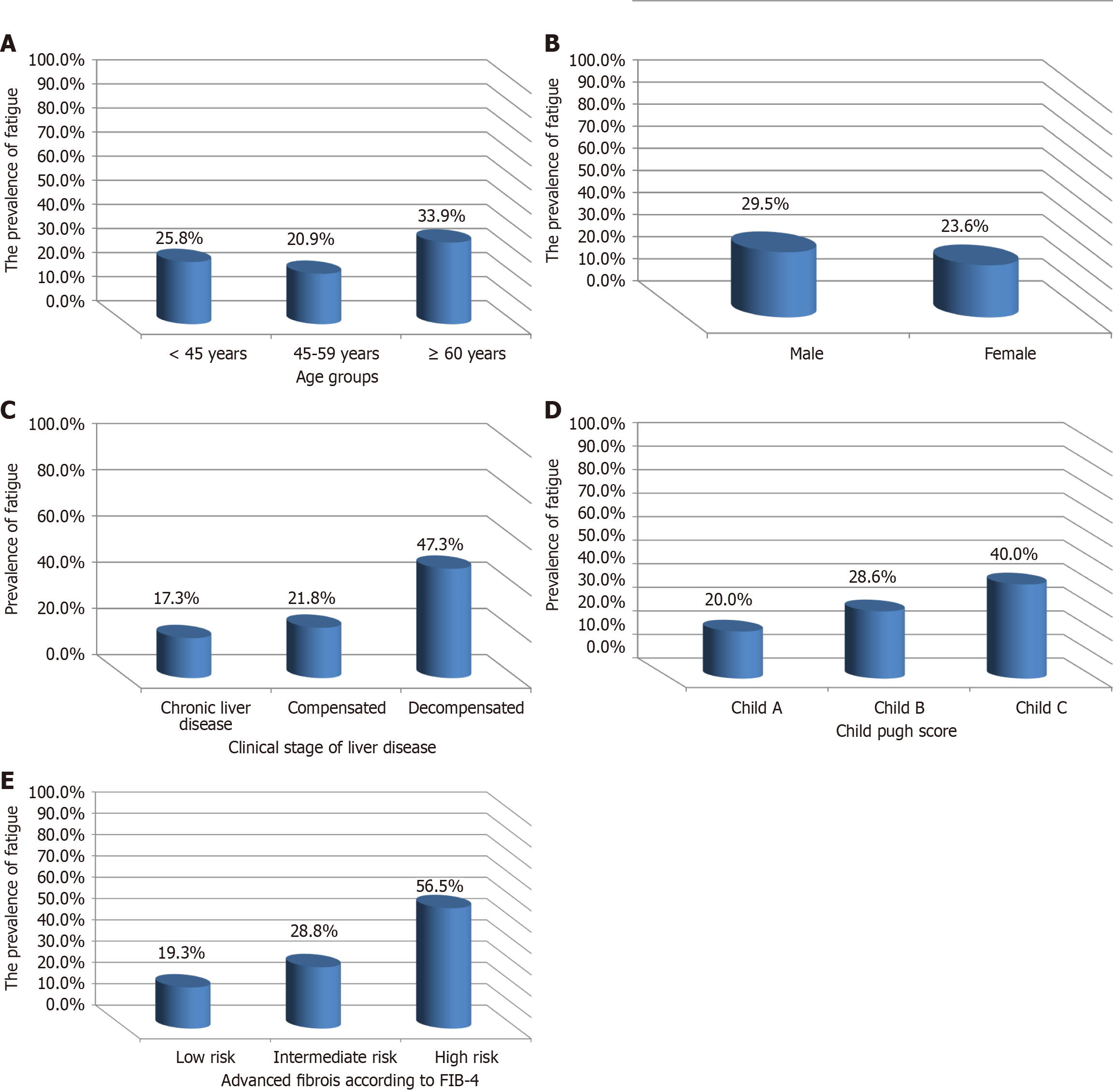
The prevalence of significant fatigue among chronic HCV disease infected patients was 27.3% (n = 82) as shown in Figure 1. Regarding age, prevalence of significant fatigue was higher in patient's ≥ 60 years (33.9%) followed by patients < 45 years (25.9%) then patients between 45-59 years (20.9%) (Figure 2A). As regard their gender, 29.5% of male participants experienced significant fatigue while 23.6% of female participants had significant fatigue (Figure 2B). Regarding the clinical stage of liver disease among our studied population, prevalence was higher among decompensated group as 47.3% of decompensated patients suffered from significant fatigue; on the other hand, only 17.3% of cases with chronic liver disorder had significant fatigue (Figure 2C). Regarding child Pugh score, the prevalence of significant fatigue was predominated among Child C group where 40% of Child C patients suffered from significant fatigue (Figure 2D). Ad
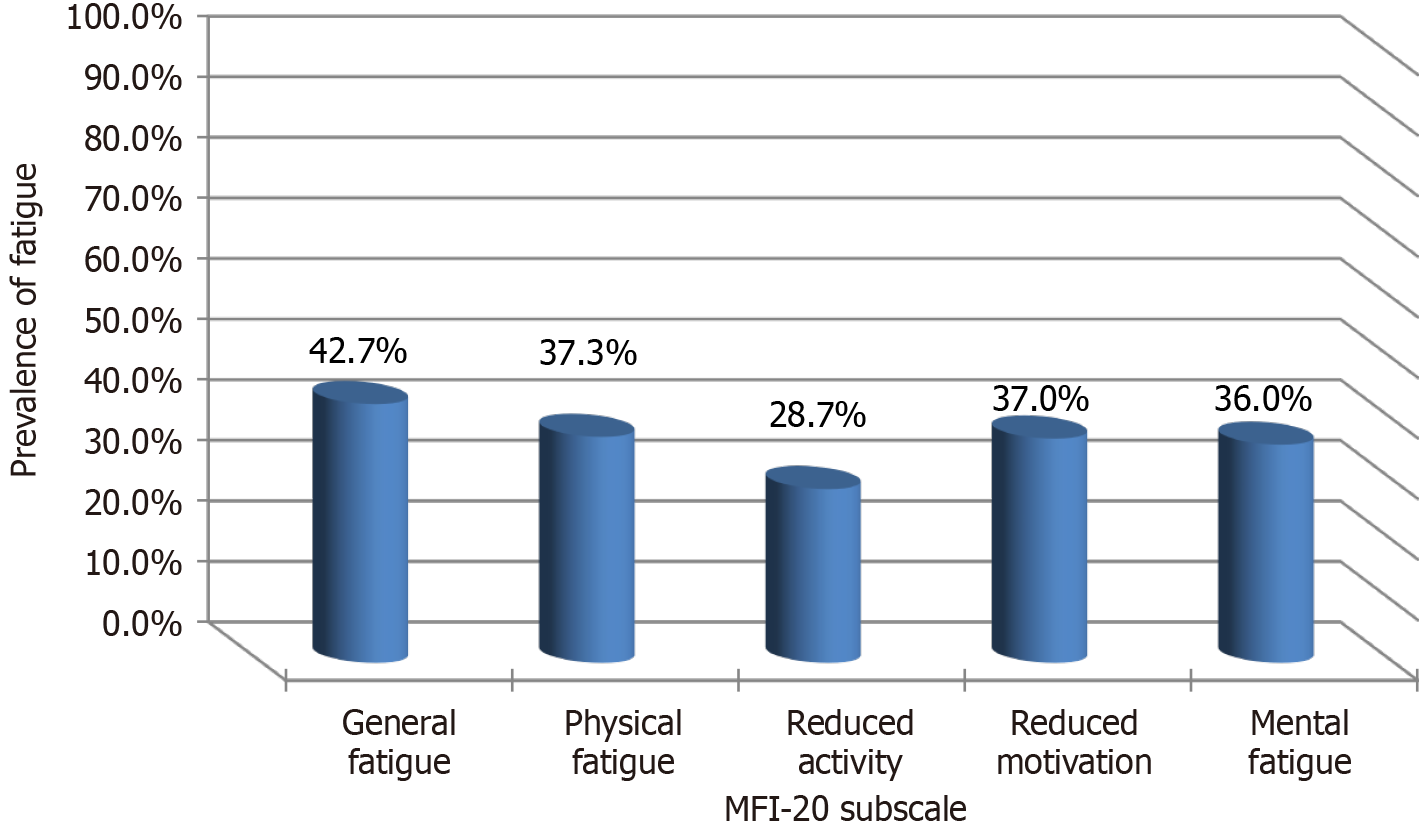
Regarding MFI-20 subscales, 42.7% of the participants experienced significant general fatigue, 37.3% of them with significant physical fatigue, 28.7% had significant reduced activity, and 37% with significant reduced motivation and 36% of them had significant mental fatigue (Figure 3).
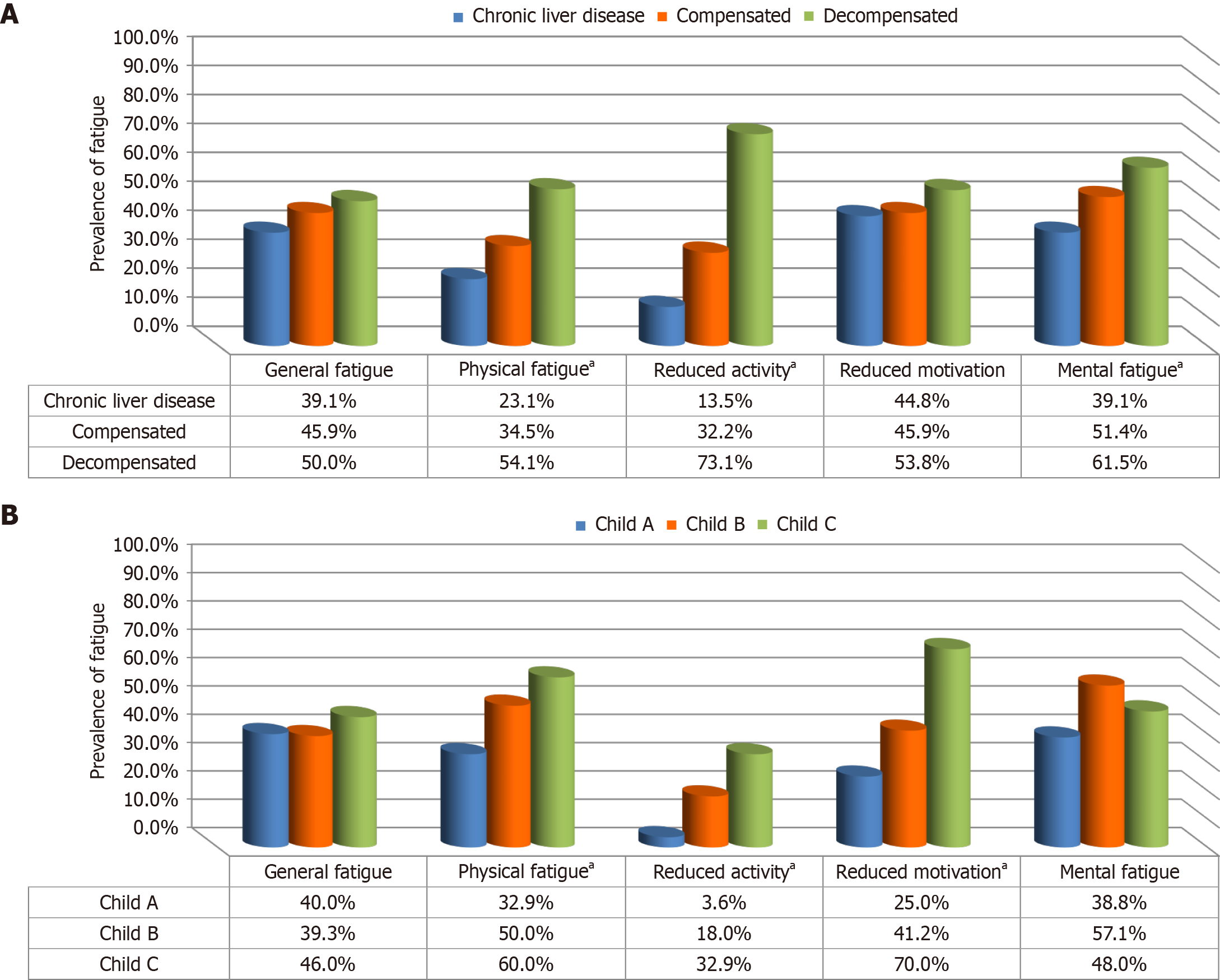
Figure 4 illustrated prevalence of significant fatigue in different MFI-20 subscales and other factors, Panel (a) presents data stratifying significant fatigue levels subscales according to the different clinical stages of liver illness. There is a corresponding increase in the occurrence of significant fatigue in distinctive MFI-20 subscales among decompensated patients than compensated and CLD patients (Figure 4A). Panel (b) further stratifies the prevalence of fatigue according to the Child classification. The findings reveal the prevalence of significant fatigue in different MFI-20 subscales were higher among Child C patients than Child B and A (Figure 4B).
Table 3 illustrates the factors associated with significant fatigue among chronic HCV patients. The data reveals those residing in urban areas, and those with decompensated liver disease, higher Child scores (B and C), and higher FIB-4 scores showed significantly higher fatigue levels. Additionally, a higher MELD score, presence of history of GIT hemorrhage, ascites, lack of antiviral treatment response, hepatic encephalopathy, antiviral treatment within the past year and longer HCV infection were significantly associated with significant fatigue. However, there was non-statistical significance difference between significant fatigue and non-fatigue group regarding their gender, age, marital status, smoking and presence of comorbidities (P > 0.05) (Table 3).
| Variables | Non-fatigue group (n = 218) | Significant fatigue group (n = 82) | P value |
| Age groups | 0.081 | ||
| < 45 years | 49 (74.2) | 17 (25.8) | |
| 45-59 years | 87 (79.1) | 23 (20.9) | |
| ≥ 60 years | 82 (66.1) | 42 (33.9) | |
| Gender | 0.274 | ||
| Male | 134 (70.5) | 56 (29.5) | |
| Female | 84 (76.4) | 26 (23.6) | |
| Marital status | 0.347 | ||
| Single1 | 28 (66.7) | 14 (33.3) | |
| Married | 190 (73.6) | 68 (26.4) | |
| Residence | 0.039 | ||
| Rural | 130 (77.4) | 38 (22.6) | |
| Urban | 88 (66.7) | 44 (33.3) | |
| Smoking | 0.543 | ||
| No | 174 (71.9) | 68 (28.1) | |
| Yes | 44 (75.9) | 14 (24.1) | |
| Co-morbidities (DM and/HTN) | 0.195 | ||
| No | 114 (76.0) | 36 (24.0) | |
| Yes | 104 (69.3) | 46 (30.7) | |
| Clinical stage | 0.001 | ||
| Chronic liver disease | 43 (82.7) | 9 (17.3) | |
| Compensated liver disease | 136 (78.2) | 38 (21.8) | |
| Decompensated liver disease | 39 (52.7) | 35 (47.3) | |
| Child score (n = 248) | 0.015 | ||
| A | 136 (80.0) | 34 (20.0) | |
| B | 20 (71.4) | 8 (28.6) | |
| C | 30 (60.0) | 20 (40.0) | |
| FIB-4 fibrosis classes | 0.001 | ||
| Low risk (FIB-4 < 1.45) | 146 (80.7) | 35 (19.3) | |
| Intermediate risk (FIB from 1.45-3.25) | 52 (28.8) | 21 (28.8) | |
| High risk (FIB-4 > 3.25) | 20 (43.5) | 26 (56.5) | |
| MELD score | 0.001 | ||
| < 15 | 211 (78.1) | 59 (21.9) | |
| ≥ 15 | 7 (23.3) | 23 (76.7) | |
| Ascites | 0.001 | ||
| No | 173 (78.3) | 48 (21.7) | |
| Yes | 45 (57.0) | 34 (43.0) | |
| History of GIT bleeding | 0.026 | ||
| No | 184 (75.4) | 60 (24.6) | |
| Yes | 34 (60.7) | 22 (39.3) | |
| History of hepatic encephalopathy | 0.001 | ||
| No | 195 (78.3) | 54 (21.7) | |
| Yes | 23 (45.1) | 28 (54.9) | |
| Antiviral treatment | 0.003 | ||
| No | 20 (52.6) | 18 (47.4) | |
| Yes | 198 (75.6) | 64 (24.4) | |
| The duration of last dose of antiviral treatment (n = 262) | 0.033 | ||
| < 1 year | 2 (33.3) | 4 (66.7) | |
| ≥ 1 year | 196 (76.6) | 60 (23.4) | |
| Antiviral treatment response (n = 262) | 0.059 | ||
| Response | 190 (76.9) | 57 (23.1) | |
| Failure | 8 (53.3) | 7 (46.7) | |
| HCV infection duration2 | 0.001 | ||
| < 10 years | 138 (86.8) | 21 (13.2) | |
| ≥ 10 years | 80 (56.7) | 61 (43.3) | |
Univariate and multivariate analyses have been carried out to define the association among fatigue and several factors (Table 4). Univariate analysis showed that decompensated liver disease (OR = 3.3, 95%CI: 1.7-6.6, P value = 0.001), Child B and C scores (OR = 2.2, 95%CI: 1.2-4.1, P value = 0.008), higher FIB-4 scores (OR = 3.7, 95%CI: 1.7-7.8, P value = 0.001), MELD scores ≥ 15 (OR = 11.7, 95%CI: 4.8-28.7, P value = 0.001), presence of ascites (OR = 1.6, 95%CI: 1.2-2.1, P value = 0.001), history of gastrointestinal bleeding (OR = 1.4, 95%CI. 1.04-1.9, P value = 0.028), and hepatic encephalopathy (OR = 2.09, 95%CI: 1.5-2.87, P value = 0.001) were significantly associated with fatigue. Additionally, antiviral treatment within the past year and longer HCV infection duration (≥ 10 years) were significant predictors of fatigue (OR = 8.6, 95%CI: 1.27-58.3, P value = 0.027; OR = 1.2, 95%CI: 1.13-1.34, P = 0.001, respectively). Further multivariate analysis indicated that decompensated liver disease (OR = 3.3, 95%CI: 1.7-6.6, P value = 0.001), higher FIB-4 scores (OR = 3.7, 95%CI: 1.7-7.8, P value = 0.001), antiviral treatment within the past year (OR = 8.6, 95%CI: 1.2-58.3, P value = 0.027) and longer HCV infection (OR = 1.2, 95%CI: 1.13-1.34, P value = 0.001) were independent risk factors for fatigue.
| Variables1 | Univariate | Multivariate analysis | ||
| OR (95%CI) | P value | OR (95%CI) | P value | |
| Age (< 45 years/≥ 45 years) | 1.67 (0.822-3.44) | 0.155 | ||
| Gender (male/female) | 1.35 (0.74-2.3) | 0.275 | ||
| Clinical stage of liver disease (chronic liver disease/compensated/decompensated) | 3.4 (1.9-5.9) | 0.001 | 3.3 (1.7-6.6) | 0.001 |
| Child score (Child A/Child B and C) | 2.2 (1.2-4.1) | 0.008 | ||
| FIB-4 fibrosis classes (low risk/intermediate/high risk) | 2.7 (1.6-4.5) | 0.001 | 3.7 (1.7-7.8) | 0.001 |
| MELD score (< 15/≥ 15) | 11.7 (4.8-28.7) | 0.001 | ||
| Ascites (no/yes) | 1.6 (1.2-2.1) | 0.001 | ||
| History of GIT bleeding (no/yes) | 1.4 (1.04-1.9) | 0.028 | ||
| History of hepatic encephalopathy (no/yes) | 2.09 (1.5-2.87) | 0.001 | ||
| Antiviral treatment (no/yes) | 2.7 (1.3-5.5) | 0.004 | ||
| The duration of last dose of antiviral treatment (n = 262) (< 1 year/≥ 1 year) | 6.5 (1.5-36.5) | 0.033 | 8.6 (1.27-58.3) | 0.027 |
| Antiviral treatment response (n = 262) (response/failure) | 2.9 (1.04-8.3) | 0.047 | ||
| HCV infection duration (< 10 years/≥ 10 years) | 1.19 (1.12-1.22) | 0.003 | 1.2 (1.13-1.34) | 0.001 |
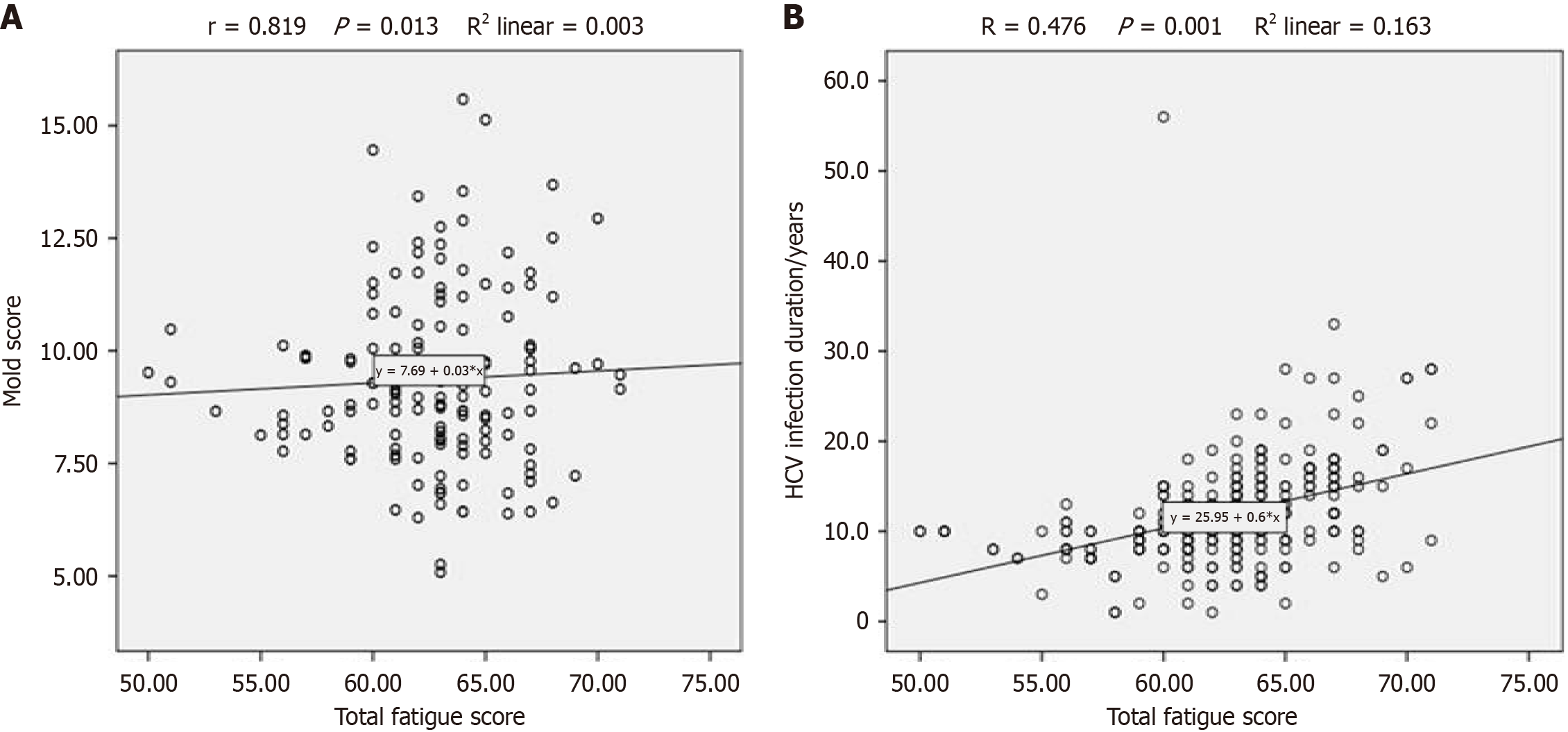
Correlation analyses exhibited a significant positive association among the total MFI-20 fatigue score and MELD score (r = 0.819, P value = 0.013) and the duration of HCV infection (r = 0.476, P value = 0.001) (Figure 5).
Fatigue is the most prevalent extra-hepatic sign observed in chronic hepatitis C cases[27]. The putative causal reasons of fatigue in hepatitis C remain unclear[28]. The FSS is frequently utilized to evaluate fatigue in hepatitis C cases by assessing its severity and impact on daily activities[16]; nevertheless, fatigue is considered a multidimensional concept, requiring that assessment tools involve various dimensions. The multidimensional weariness inventory assesses five distinct characteristics of fatigue[17]. Consequently, our research aimed to evaluate the prevalence of severe or significant fatigue in cases with hepatitis C-related CLD utilizing the MFI-20. Additionally, find independent variables correlated with fatigue.
In our analysis, the occurrence of substantial or severe fatigue in the study population was 27.3%, aligning with other research that demonstrates fatigue as a common and disabling symptom in chronic hepatitis C cases[13,29]. Similarly, prior literature indicates that fatigue is a predominant symptom of hepatitis C infection, with one in five patients reporting significant fatigue at enrollment[30]. Our findings align with those of Poynard et al[21], who reported that 53% of cases had fatigue (95%CI: 51-56), with seventeen percent (95%CI: 15-19) suffering from severe fatigue that impeded their activities[21].
The mean total MFI-20 score of 62.5 (SD ± 3.8) and the high scores in subscales such as mental fatigue (13.2 ± 1.1) and physical fatigue (12.9 ± 1.7) highlight the multidimensional nature of fatigue in this population.
Our study found that decompensated liver disease, higher Child and MELD scores, presence of history of gastro
Cases who received antiviral medicine within the past year were more likely to report significant fatigue. This could be due to the side effects of antiviral medications or the lingering effects of the virus on the body. Also, those with a longer duration since the last dose of antiviral treatment had lower fatigue levels, suggesting a potential recovery over time post-treatment. Similarly, there are many studies detected a statistically significant decrease in perceived fatigue level following the end of therapy[28,35]. Sharma et al[34] stated that post-DAA treatment, significant score improvements have been observed in both the mild fatigue group (28.34 + 3.19 vs 21.22 + 5.16; P < 0.05) and the severe fatigue group (41.82 + 4.34 vs 32.82 + 6.51; P < 0.05)[34].
This investigation has several limitations. The cross-sectional design restricts the capacity to infer causality among the factors identified & fatigue. Additionally, self-reported measures of fatigue may be subject to recall bias and subjective interpretation. Future longitudinal investigations are needed to better understand the causal relationships and the long-term impact of antiviral treatment on fatigue.
Fatigue is a prevalent, multidimensional symptom in chronic HCV patients with 27.3% experiencing significant or severe fatigue. Using the MFI-20, we identified that fatigue severity significantly impacts patient quality of life and significantly correlated with the severity of liver disease, antiviral treatment, and longer duration of HCV infection. These findings underscore the need for comprehensive management strategies that address both the clinical and psychosocial aspects of fatigue to improve patient outcomes. Further research should explore the underlying mechanisms of patient outcomes. Further research should explore the underlying mechanisms of fatigue in chronic HCV and assess the potential benefits of targeted interventions.
The research team expresses gratitude to the data recruiters and the research participants for their significant contributions to this investigation.
| 1. | Moon AM, Singal AG, Tapper EB. Contemporary Epidemiology of Chronic Liver Disease and Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:2650-2666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 923] [Cited by in RCA: 836] [Article Influence: 139.3] [Reference Citation Analysis (6)] |
| 2. | Burki T. WHO's 2024 global hepatitis report. Lancet Infect Dis. 2024;24:e362-e363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 51] [Article Influence: 25.5] [Reference Citation Analysis (1)] |
| 3. | Lavanchy D. Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect. 2011;17:107-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 983] [Cited by in RCA: 914] [Article Influence: 60.9] [Reference Citation Analysis (1)] |
| 4. | Mohd Hanafiah K, Groeger J, Flaxman AD, Wiersma ST. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57:1333-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1926] [Cited by in RCA: 1807] [Article Influence: 139.0] [Reference Citation Analysis (2)] |
| 5. | Anwar WA, El Gaafary M, Girgis SA, Rafik M, Hussein WM, Sos D, Mossad IM, Fontanet A, Temime L. Hepatitis C virus infection and risk factors among patients and health-care workers of Ain Shams University hospitals, Cairo, Egypt. PLoS One. 2021;16:e0246836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 6. | Naga IS, Kamel AAF, Ooda SA, Elbab HMF, El-sharkawy RM. Effect of directly acting anti-viral agents on immunological imprints in chronic HCV-4a patients: interleukin-10 and vascular endothelial growth factor genes expression level. Egypt Liver J. 2021;11:35. [RCA] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 7. | El-Zanaty F, Way A. Egypt demographic and health survey 2008. [cited 19 November 2025]. Available from: https://www.dhsprogram.com/pubs/pdf/FR220/FR220.pdf. |
| 8. | Egypt Health Issue Survey. [cited 19 November 2025]. Available from: https://dhsprogram.com/pubs/pdf/FR313/FR313.pdf. |
| 9. | Cornberg M, Razavi HA, Alberti A, Bernasconi E, Buti M, Cooper C, Dalgard O, Dillion JF, Flisiak R, Forns X, Frankova S, Goldis A, Goulis I, Halota W, Hunyady B, Lagging M, Largen A, Makara M, Manolakopoulos S, Marcellin P, Marinho RT, Pol S, Poynard T, Puoti M, Sagalova O, Sibbel S, Simon K, Wallace C, Young K, Yurdaydin C, Zuckerman E, Negro F, Zeuzem S. A systematic review of hepatitis C virus epidemiology in Europe, Canada and Israel. Liver Int. 2011;31 Suppl 2:30-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 306] [Cited by in RCA: 281] [Article Influence: 18.7] [Reference Citation Analysis (1)] |
| 10. | Kośnik A, Wójcicki M. Fatigue in chronic liver disease patients: prevalence, pathophysiology, and management. Prz Gastroenterol. 2022;17:21-27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (4)] |
| 11. | Weissenborn K, Tryc AB, Heeren M, Worthmann H, Pflugrad H, Berding G, Bokemeyer M, Tillmann HL, Goldbecker A. Hepatitis C virus infection and the brain. Metab Brain Dis. 2009;24:197-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 64] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 12. | Karaivazoglou K, Iconomou G, Triantos C, Hyphantis T, Thomopoulos K, Lagadinou M, Gogos C, Labropoulou-Karatza C, Assimakopoulos K. Fatigue and depressive symptoms associated with chronic viral hepatitis patients. health-related quality of life (HRQOL). Ann Hepatol. 2010;9:419-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 41] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 13. | Foster GR, Goldin RD, Thomas HC. Chronic hepatitis C virus infection causes a significant reduction in quality of life in the absence of cirrhosis. Hepatology. 1998;27:209-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 375] [Article Influence: 13.4] [Reference Citation Analysis (1)] |
| 14. | Pennisi M, Malaguarnera G, Di Bartolo G, Lanza G, Bella R, Chisari EM, Cauli O, Vicari E, Malaguarnera M. Decrease in Serum Vitamin D Level of Older Patients with Fatigue. Nutrients. 2019;11:2531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 15. | Yennurajalingam S, Bruera E. Palliative management of fatigue at the close of life: "it feels like my body is just worn out". JAMA. 2007;297:295-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 44] [Article Influence: 2.3] [Reference Citation Analysis (4)] |
| 16. | Rosa K, Fu M, Gilles L, Cerri K, Peeters M, Bubb J, Scott J. Validation of the Fatigue Severity Scale in chronic hepatitis C. Health Qual Life Outcomes. 2014;12:90. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 17. | Ericsson A, Mannerkorpi K. Assessment of fatigue in patients with fibromyalgia and chronic widespread pain. Reliability and validity of the Swedish version of the MFI-20. Disabil Rehabil. 2007;29:1665-1670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 66] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 18. | Smets EM, Garssen B, Bonke B, De Haes JC. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res. 1995;39:315-325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2933] [Cited by in RCA: 2484] [Article Influence: 80.1] [Reference Citation Analysis (1)] |
| 19. | Wondie Y, Hinz A. Application of the Multidimensional Fatigue Inventory to Ethiopian Cancer Patients. Front Psychol. 2021;12:687994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 20. | Haggag WE, Soliman H. The Arabic Version of h'lultidimensional Fatigue Inventory: Reliability, Validity and Findings in Three Groups. J Psychiat. 1997;20:85-96. |
| 21. | Poynard T, Cacoub P, Ratziu V, Myers RP, Dezailles MH, Mercadier A, Ghillani P, Charlotte F, Piette JC, Moussalli J; Multivirc group. Fatigue in patients with chronic hepatitis C. J Viral Hepat. 2002;9:295-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 159] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 22. | Singal AK, Kamath PS. Model for End-stage Liver Disease. J Clin Exp Hepatol. 2013;3:50-60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 132] [Article Influence: 10.2] [Reference Citation Analysis (2)] |
| 23. | Sterling RK, Lissen E, Clumeck N, Sola R, Correa MC, Montaner J, S Sulkowski M, Torriani FJ, Dieterich DT, Thomas DL, Messinger D, Nelson M; APRICOT Clinical Investigators. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43:1317-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4179] [Cited by in RCA: 3909] [Article Influence: 195.5] [Reference Citation Analysis (7)] |
| 24. | Singer S, Kuhnt S, Zwerenz R, Eckert K, Hofmeister D, Dietz A, Giesinger J, Hauss J, Papsdorf K, Briest S, Brown A. Age- and sex-standardised prevalence rates of fatigue in a large hospital-based sample of cancer patients. Br J Cancer. 2011;105:445-451. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 70] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 25. | Arbuckle JL. IBM® SPSS® Amos™ 26. [cited 19 November 2025]. Available from: https://www.ibm.com/docs/en/SSLVMB_26.0.0/pdf/amos/IBM_SPSS_Amos_User_Guide.pdf. |
| 26. | Field AP. Discovering statistics using IBM SPSS statistics. [cited 3 March 2025]. Available from: https://docs.edtechhub.org/lib/CFJAF8HE. |
| 27. | Gumber SC, Chopra S. Hepatitis C: a multifaceted disease. Review of extrahepatic manifestations. Ann Intern Med. 1995;123:615-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 290] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 28. | Sarkar S, Jiang Z, Evon DM, Wahed AS, Hoofnagle JH. Fatigue before, during and after antiviral therapy of chronic hepatitis C: results from the Virahep-C study. J Hepatol. 2012;57:946-952. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 29. | Kallman J, O'Neil MM, Larive B, Boparai N, Calabrese L, Younossi ZM. Fatigue and health-related quality of life (HRQL) in chronic hepatitis C virus infection. Dig Dis Sci. 2007;52:2531-2539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 119] [Article Influence: 6.3] [Reference Citation Analysis (1)] |
| 30. | Faccioli J, Nardelli S, Gioia S, Riggio O, Ridola L. Neurological and psychiatric effects of hepatitis C virus infection. World J Gastroenterol. 2021;27:4846-4861. [PubMed] [DOI] [Full Text] |
| 31. | Friedman SL. Cellular networks in hepatic fibrosis. Digestion. 1998;59:368-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (2)] |
| 32. | Hassoun Z, Willems B, Deslauriers J, Nguyen BN, Huet PM. Assessment of fatigue in patients with chronic hepatitis C using the Fatigue Impact Scale. Dig Dis Sci. 2002;47:2674-2681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 46] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 33. | Bajaj JS, Thacker LR, Wade JB, Sanyal AJ, Heuman DM, Sterling RK, Gibson DP, Stravitz RT, Puri P, Fuchs M, Luketic V, Noble N, White M, Bell D, Revicki DA. PROMIS computerised adaptive tests are dynamic instruments to measure health-related quality of life in patients with cirrhosis. Aliment Pharmacol Ther. 2011;34:1123-1132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 34. | Sharma M, Talele R, Chaudhary M. 60. Fatigue improves after successful antiviral therapy in chronic hepatitis C patients. J Clin Exp Hepatol. 2018;8:S132. [DOI] [Full Text] |
| 35. | Ware JE Jr, Bayliss MS, Mannocchia M, Davis GL. Health-related quality of life in chronic hepatitis C: impact of disease and treatment response. The Interventional Therapy Group. Hepatology. 1999;30:550-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 231] [Cited by in RCA: 205] [Article Influence: 7.6] [Reference Citation Analysis (1)] |