Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.113284
Revised: September 23, 2025
Accepted: January 9, 2026
Published online: March 27, 2026
Processing time: 217 Days and 23.3 Hours
Metabolically associated fatty liver disease, recently redefined from non-alcoholic fatty liver disease, has emerged as the most common chronic liver disease wor
Core Tip: Metabolically associated fatty liver disease (MAFLD) is the new term replacing non-alcoholic fatty liver disease. Today, it is the most common chronic liver disease worldwide, affecting approximately 38% of the world's population. This comprehensive review offers insight into the various aspects of MAFLD, focusing on terminology, epidemiology, new findings on pathophysiology, key clinical factors, and current treatment options.
- Citation: Plakida A, Iushkovska O, Sierpińska LE. Metabolically associated fatty liver disease: What hepatologists need to know about this systemic disease. World J Hepatol 2026; 18(3): 113284
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/113284.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.113284
Metabolically associated fatty liver disease (MAFLD) is now recognized as the most common chronic liver disease worldwide, affecting nearly 40% of the global population[1,2]. It encompasses a broad clinical spectrum, ranging from isolated hepatic steatosis (HS) to metabolic dysfunction-associated steatohepatitis (MASH), which can progress to fibrosis, cirrhosis, and hepatocellular carcinoma (HCC)[3]. The increasing prevalence of obesity and type 2 diabetes has accelerated the global burden of MAFLD, making it not only a leading cause of chronic liver disease but also a significant contributor to extrahepatic morbidity and mortality[4].
In 2020, an international consensus proposed replacing the long-used term non-alcoholic fatty liver disease (NAFLD) with MAFLD, and in 2023 further refinements introduced the term metabolic dysfunction-associated steatotic liver disease (MASLD)[5,6]. This nomenclature shift reflects the central role of metabolic dysfunction in the disease process, offers clearer diagnostic criteria, and aligns clinical research with the metabolic nature of the condition. However, it also raises new challenges: How to stratify risk more accurately, identify patients who do not fully meet the new criteria, and integrate emerging therapies into clinical practice[7,8].
Given these rapid developments, a timely synthesis is required. This review aims to summarize the evolving ter
Current epidemiological data suggest that metabolic dysfunction-related fatty liver disease is present in roughly 25% of the global adult population. It seriously threatens the health and economy of all societies[9-11]. The high prevalence of this disease is due to the rapid increase in sedentary lifestyles, low levels of physical activity, and excess caloric intake compared with expenditure due to an unbalanced and unhealthy diet[12]. At the same time, impaired metabolic health is highly prevalent among adults in high-income countries, including individuals who do not meet criteria for overweight or obesity[13,14]. The lack of a precise nomenclature of liver diseases not associated with alcohol consumption, along with the lack of clear clinical criteria for a “positive” diagnosis of this disease, determine the relevance of this problem.
The first descriptions of lipid accumulation in hepatocytes of street drinkers who consume excessive amounts of alcoholic beverages date back to the 19th century, and the term “fatty liver” was first used in 1836[15]. In 1839, it was suggested that fat accumulation in the liver could be the cause of cirrhosis[16]. Forty years later, a higher incidence of fatty liver degeneration was noted in patients with diabetes and obesity[16,17]. However, the similarity of histopathological changes in the liver in patients who abuse alcohol and in patients with obesity and/or diabetes was described much later, in the 1950s-1970s[16].
The concepts of NAFLD and non-alcoholic steatohepatitis (NASH) were introduced in the early 1980s to characterize a progressive liver condition that closely resembles alcoholic steatohepatitis on histological examination, yet occurs in individuals without significant alcohol consumption[18]. Liver steatosis is defined as an initial stage with subsequent development of NASH, fibrosis, and adverse outcomes: Cardiovascular complications, cirrhosis, or HCC; at the same time, the transition from steatosis to NASH is not always observed.
NAFLD has traditionally been diagnosed when HS is detected by imaging or histology in the absence of other identifiable causes of liver injury, including significant alcohol intake, exposure to hepatotoxic drugs or toxins, viral hepatitis, or inherited liver disorders[19].
However, the use of negative, exclusion-based terminology has important clinical and communicative limitations. Disease names that define a condition by what it is not may be confusing for patients and may fail to convey the underlying mechanisms or clinical relevance to healthcare providers. In particular, the term NAFLD emphasizes the absence of alcohol consumption rather than the central role of metabolic dysfunction, thereby underrepresenting its strong association with cardiometabolic risk and systemic complications.
In the following 40 years, the presence of “non-alcoholic” in the definition emphasized this feature as the leading one. The diagnosis of this nosology was based on excluding other possible causes of steatosis. Along with the presence of excess lipids in the liver, the criteria for diagnosis were the absence of a history of chronic alcohol consumption in toxic doses, as well as the exclusion of other causes of fatty liver infiltration: Hepatotropic viruses, genetically determined diseases, and drug-induced liver injury (DILI)[15,16,18].
Between 2002 and 2017, a diagnostic approach (diagnosis by exclusion) based on exclusion criteria became widespread. Over time, experts began to justify the use of criteria to confirm this diagnosis. In 2005, Loria et al[20] suggested in
The proposed approach was based on the fact that NAFLD is primarily a consequence of systemic metabolic dys
During the 2020 international discussion involving experts from 22 countries, a consensus was reached on revising the NAFLD definition. The review resulted in a proposal to change the nomenclature, introduce a new term, MAFLD instead of NAFLD, and change the diagnostic criteria, taking into account the importance of metabolic factors[28]. These changes were intended to facilitate a clear assessment of the contribution of etiologic factors to liver damage (dietary habits, alcohol consumption, microbiota, genetics, epigenetics, and individual metabolic imbalances).
Similar to NAFLD, MAFLD represents the hepatic component of a broader multisystem disorder characterized by substantial heterogeneity in etiological factors, clinical presentation, disease trajectory, and long-term outcomes[29]. Owing to the complexity of its underlying mechanisms, the development of a single, definitive diagnostic test is unlikely. Instead, MAFLD will probably require operational diagnostic frameworks based on combinations of clinical, metabolic, and laboratory criteria, analogous to the evolving definitions used for metabolic syndrome[30-32].
Under the updated nomenclature, MAFLD is diagnosed based on documented HS, confirmed by imaging modalities, serum-based biomarkers, or histological assessment[33] in combination with overweight/obesity or type 2 diabetes. In a patient with normal body weight and without type 2 diabetes, HS must be combined with the presence of two or more signs of metabolic dysfunction.
Metabolic dysregulation is defined by the presence of one or more of the following features: (1) Increased waist circumference (≥ 102 cm in men and ≥ 88 cm in women of European ancestry; ≥ 90 cm in Asian men and ≥ 80 cm in Asian women); (2) Elevated blood pressure (≥ 130/85 mmHg) or current use of antihypertensive medication; (3) Hypertriglyceridemia [serum triglycerides (TG) ≥ 1.7 mmol/L (150 mg/dL)] or ongoing lipid-lowering treatment; (4) Reduced high-density lipoprotein cholesterol (HDL-C) concentrations [≤ 1.0 mmol/L (40 mg/dL) in men and ≤ 1.3 mmol/L (50 mg/dL) in women] or treatment for dyslipidemia; (5) Impaired glucose metabolism, including prediabetes, defined by elevated fasting plasma glucose, abnormal glucose tolerance, or increased glycated hemoglobin levels; and (6) Additional markers of metabolic dysfunction include an elevated insulin resistance (IR) index, as assessed by the homeostatic model ass
An essential feature of MAFLD is the exclusion of the fact of abuse of hepatotoxic doses of alcohol, as well as other etiological variants of chronic liver diseases. According to the new nomenclature, simple steatosis and NASH were not two different forms of the disease. However, they were considered a single pathological process, the severity of which depends on the activity of inflammation and the stage of liver fibrosis[28].
The main limitations of the NAFLD and NASH terms are their dependence on the need to formulate a diagnosis by exclusion and the use of potentially stigmatizing formulations[34]. It should be noted that the new MAFLD term also has several shortcomings. The prerequisites for the next stage of revision of the terms were, in particular, the lack of consideration of the role of alcohol, which accelerates the course of the disease[35], the use of HOMA-IR and hsCRP as diagnostic criteria, which are often not assessed in routine clinical practice, and the lack of inclusion of patients with a normal body mass index (BMI)[33].
The revision aims to increase awareness among healthcare workers, society, and patients of the disease, its course, treatment, and outcomes, as well as combat stigma by focusing on the initial cardiometabolic etiological factor. A more precise assessment of risk factors for disease progression, validation of non-invasive diagnostics, an indirect positive effect on the search for new drugs, and the ability to provide personalized medical care to this group of patients are planned[35].
In June 2023, the European Association for the Study of the Liver (EASL) addressed the practical implications and clinical application of the updated terminology during its annual congress[36] and in September 2023 during a summit devoted to updating the NAFLD/MAFLD nomenclature[35]. A total of 236 experts from 56 countries took part in the discussions[34]. The Working Committee consisted of 34 experts from 7 scientific societies and patient advocacy organizations, selected based on the size of the societies or organizations (EASL: 29%, American Association for the Study of Liver: 27%, Asian Pacific Association for the Study of the Liver: 13%, Latin American Association for the Study of the Liver: 12%, other societies: 7%, patient advocacy organizations: 11%) and divided into six working groups.
During these discussions, a substantial proportion of respondents reported that the descriptors “non-alcoholic” and “fatty” were perceived as stigmatizing, with 61% and 66% expressing concern, respectively[34].
According to the accepted nomenclature, the term steatotic liver disease (SLD) was introduced; steatosis is confirmed by imaging methods or biopsy, which, in turn, includes MASLD and SLD, in which metabolic dysfunction (Met) is combined with alcoholic liver disease (ALD).
When diagnosing Met is combined with ALD (MetALD), the amount of ethyl alcohol consumed is taken into account. For women, the diagnostic alcohol intake range is defined as 140-210 g per week (approximately 20-50 g/day), while for men it is 350-420 g per week (approximately 30-60 g/day). Within this spectrum, lower intake thresholds (≥ 140 g/week in women and ≥ 350 g/week in men) support a diagnosis of MetALD with a predominant metabolic component. Conversely, higher intake levels (≥ 210 g/week in women and ≥ 420 g/week in men) indicate MetALD with a dominant contribution of alcohol-related liver injury.
In addition to these categories, the spectrum of SLDs includes ALD; SLD of defined etiology, such as DILI or monogenic disorders (including lysosomal acid lipase deficiency, Wilson disease, hypobetalipoproteinemia, and other inborn errors of metabolism); mixed-etiology SLD associated with conditions such as chronic hepatitis C (CHC) infection, malnutrition, or celiac disease; as well as SLD of indeterminate origin.
Following new approaches to diagnostics, the main principle for determining the form of SLD is the presence or absence of the cardiometabolic criterion[34,35].
Cardiometabolic risk in adults is defined by the presence of one or more of the following abnormalities: (1) Excess adiposity, reflected by a BMI ≥ 25 kg/m2 (≥ 23 kg/m2 in Asian populations) or increased waist circumference (> 94 cm in men and > 80 cm in women, with ethnicity-specific thresholds applied where appropriate); (2) Impaired glucose homeostasis, including elevated fasting or post-load plasma glucose levels, increased glycated hemoglobin, established type 2 diabetes mellitus (T2DM), or ongoing antidiabetic treatment; (3) Elevated blood pressure (≥ 130/85 mmHg) or current antihypertensive therapy; (4) Hypertriglyceridemia [plasma TG ≥ 1.7 mmol/L (150 mg/dL)] or lipid-lowering treatment; and (5) Reduced HDL-C concentrations [≤ 1.0 mmol/L (40 mg/dL) in men and ≤ 1.3 mmol/L (50 mg/dL) in women] or therapy for dyslipidemia[34,35].
For children, cardiometabolic criteria include: In pediatric populations, cardiometabolic dysfunction is defined by age- and sex-specific criteria, including: (1) Excess adiposity (BMI at or above the 85th percentile or a BMI z-score ≥ +1, increased waist circumference above the 95th percentile, or ethnicity-adjusted thresholds); (2) Abnormal glucose metabolism, reflected by elevated fasting or random plasma glucose concentrations, impaired glucose tolerance on oral testing, increased glycated hemoglobin levels, or a prior diagnosis of T2DM and its treatment; (3) Elevated blood pressure (at or above the 95th percentile for children younger than 13 years, or ≥ 130 mmHg in adolescents aged 13 years or older, whichever threshold is lower) or current antihypertensive therapy; (4) Hypertriglyceridemia [plasma TG ≥ 1.15 mmol/L (100 mg/dL) in children younger than 10 years and ≥ 1.7 mmol/L (150 mg/dL) in those aged 10 years or older] or lipid-lowering treatment; and (5) Reduced HDL-C concentrations [≤ 1.0 mmol/L (40 mg/dL)] or ongoing lipid-lowering therapy[34,35].
If the patient has at least one of the cardiometabolic criteria and no other causes of steatosis, the diagnosis of MASLD is made; if there are other causes of steatosis, the diagnosis is MetALD (with high doses of alcohol) or MASLD of other mixed etiology. Suppose there are no cardiometabolic criteria and there are other causes of steatosis. In that case, the diagnosis is SLD of other etiology (for example, ALD, DILI, and monogenic diseases). If there are no other causes of steatosis, the diagnosis is cryptogenic SLD[36]. Instead of the NASH term, the definition of "MASH" is introduced[37]. The chronology of changes in terms is presented in Table 1.
| Transcript | Years of use | Features/reasons for introduction |
| Non-alcoholic fatty liver disease | 1980-2020 | Emphasized the absence of alcohol as a cause of the disease |
| Metabolic (dysfunction) associated fatty liver disease | Since 2020 | The emphasis is on metabolic disorders |
| Metabolic dysfunction-associated steatotic liver disease | Since 2023 | A more precise definition is adopted in American Association for the Study of Liver Diseases/European Association for the Study of the Liver |
The development of MAFLD and the disease’s severity are associated with single nucleotide polymorphisms[38]. Genetic susceptibility plays a key role in interindividual variability of disease severity in MAFLD. Among identified variants, the rs738409 polymorphism in the PNPLA3 gene represents the strongest genetic determinant of HS and disease progression[39]. Additional single-nucleotide polymorphisms implicated in the development and advancement of steatohepatitis include TM6SF2 rs58542926, which influences very-low-density lipoprotein (VLDL) assembly and secretion, and MBOAT7 rs641738, involved in phospholipid acyl-chain remodeling[40,41]. The PNPLA3 rs738409 variant alters hepatic lipid handling by impairing TG remodeling and disrupting the transfer of polyunsaturated fatty acids required for phosphatidylcholine synthesis[42]. Moreover, homozygosity for this variant enhances the profibrogenic activation of hepatic stellate cells[43]. Reduced activity of enzymes responsible for xenobiotic metabolism and elimination in carriers of PNPLA3 rs738409 may further increase vulnerability to hepatotoxic injury, potentially contributing to chronic low-grade liver damage[44].
Epigenetic regulation has emerged as an important contributor to the pathogenesis of steatohepatitis and related metabolic disorders[45]. Key mechanisms include alterations in DNA methylation, post-translational histone modifications, and dysregulation of non-coding microRNAs[46]. In patients with steatohepatitis, hepatic expression of miR-122 is markedly reduced compared with simple steatosis, whereas other microRNAs, such as miR-223, demonstrate increased expression[47]. In addition, elevated methylation of mitochondrial DNA has been observed in liver tissue from patients with steatohepatitis and is thought to promote activation and transdifferentiation of hepatic stellate cells into myofibroblasts, thereby contributing to the development and progression of liver fibrosis[48].
Obesity represents one of the most prevalent and well-established risk factors for MAFLD[49]. More than half of adults diagnosed with MAFLD are affected by obesity, and the rising global burden of the disease closely parallels the worldwide increase in obesity rates[50]. Nevertheless, accumulating evidence suggests that disease risk is driven less by overall adiposity than by fat distribution. In particular, expansion of visceral adipose tissue (VAT) has been consistently linked to a higher likelihood of HS and MAFLD development, underscoring the pathogenic role of central fat accumulation[51,52]. In addition, the size of VAT is directly associated with liver inflammation and the development of fibrosis in patients with MAFLD[53,54]. This association is partly explained by the endocrine activity of VAT, which functions as a metabolically active organ and releases multiple mediators implicated in MAFLD pathogenesis, including adipokines and pro-inflammatory cytokines such as adiponectin, interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α). Circulating adiponectin, an adipokine with anti-inflammatory and anti-lipogenic properties, is reduced in patients with steatohepatitis and demonstrates an inverse relationship with visceral fat accumulation[55]. In patients with metabolically unhealthy obesity, IL-6 levels in VAT are more than a hundred times higher than those in the liver, indicating increased proinflammatory activity of VAT[56]. Approximately 30% of normal-weight individuals have metabolic obesity with a high risk of developing metabolic diseases such as MAFLD[51,52]. Lean patients with MAFLD also have the presence of visceral obesity[57] and exhibit altered gut microbiota and higher secondary bile acid levels[58].
The pathogenesis of MAFLD is highly complex and involves multiple, interrelated mechanisms. Earlier concepts described disease development using the “two-hit” model, in which an initial accumulation of hepatic lipids sensitizes hepatocytes to subsequent injurious processes, followed by oxidative stress–driven inflammation and cellular damage. However, this framework has largely been replaced by more integrative models recognizing the simultaneous contribution of metabolic, inflammatory, and oxidative pathways[59,60]. However, at present, the mechanism of MAFLD development appears to be more complex, and the "multiple hit" hypothesis is gaining increasing support[61].
The main mechanisms in the development of MAFLD are considered to be the following: (1) Excessive calorie intake and lipid metabolism disorders-excessive intake of fats and carbohydrates contributes to fat deposition in the liver; (2) IR and hyperinsulinemia-the ability of cells to absorb glucose decreases, which leads to fat accumulation in the liver; (3) Oxidative stress and inflammation-increased lipid peroxidation processes lead to damage to hepatocytes; and (4) In
Hepatic lipid overload in MAFLD results from the convergence of several metabolic pathways, including enhanced lipolysis in adipose tissue, increased de novo lipogenesis (DNL), and excessive dietary intake of fats and simple sugars. Rather than TG accumulation itself, the pathogenic trigger is the excess of lipotoxic free fatty acids (FFA), which promote hepatocellular stress and injury. In steatohepatitis, hepatocytes also exhibit elevated levels of free cholesterol, a factor known to activate inflammatory signaling and fibrogenic pathways[62,63]. This dysregulation is accompanied by alter
In parallel, liver sinusoidal endothelial cells play a critical role in disease progression. Steatosis-induced endothelial dysfunction disrupts hepatic microcirculation, and as MAFLD advances, progressive impairment of endothelial integrity contributes to inflammation, fibrogenesis, and downstream complications such as increased intrahepatic resistance and portal hypertension[66].
Oxidative stress represents a disturbance in redox homeostasis caused by an imbalance between the generation of reactive oxygen species (ROS) and the capacity of antioxidant defense systems[67]. ROS are continuously produced as by-products of cellular metabolism, beginning with the formation of superoxide anions and subsequently giving rise to other highly reactive intermediates. Excessive ROS interact with major cellular macromolecules, including proteins, lipids, and nucleic acids, resulting in oxidative modifications such as protein carbonylation, formation of lipid peroxidation products (including malondialdehyde and 4-hydroxynonenal), and oxidative DNA damage exemplified by 8-hydroxy-2′-deoxyguanosine[68].
Cellular protection against oxidative injury is mediated by a complex antioxidant network comprising enzymatic and non-enzymatic components. Enzymatic defenses include superoxide dismutase, which catalyzes the conversion of superoxide radicals into hydrogen peroxide, and catalase, which further reduces hydrogen peroxide to water. Non-enzymatic antioxidants consist of endogenous molecules such as glutathione (GSH), as well as a broad array of diet-derived compounds, including tocopherols, polyphenols, vitamins, and isoflavones; collectively, these components determine the overall antioxidant capacity of the organism.
In MAFLD, excessive oxidative stress plays a pivotal role in disease progression by promoting lipid peroxidation, impairing mitochondrial fatty acid oxidation, and stimulating the release of pro-inflammatory cytokines. Lipid peroxidation is initiated when ROS abstract hydrogen atoms from unsaturated fatty acids, triggering self-propagating chain reactions that disrupt cellular membranes and generate cytotoxic secondary metabolites[69,70]. These products activate hepatic stellate cells, enhance pro-inflammatory signaling, and may induce hepatocyte necrosis or apoptosis through pathways such as fas ligand activation, thereby contributing to fibrogenesis and progressive liver injury[71,72].
Oxidative stress has been postulated as a central process contributing to liver injury and driving the transition from simple steatosis to MAFLD[73]. Mitochondrial dysfunction in liver tissue in MAFLD has been reported to alter liver lipid balance, promote ROS production, and affect LPO and cytokine release, ultimately leading to cell death[74]. Due to impaired mitochondrial function, mitochondria in MAFLD become more susceptible to mitochondrial permeability transients, leading to cell death during ischemia and reperfusion injury[75]. Mitochondrial membrane phospholipids are oxidized, reducing fluidity and impeding GSH entry into the mitochondria, causing an imbalance between antioxidants and ROS. This causes oxidative stress, which leads to a decrease in mitochondrial ATP synthesis. Consequently, patients with MAFLD have decreased ATP stores, which makes cells more susceptible to necrosis and triggers an inflammatory response when the liver is damaged by ischemia and hypoxia[75].
In MAFLD, obligate-induced oxidative metabolism due to increased anabolism can lead to oxidative stress, in which leucine aminopeptidases (LAPs) play a significant role[76]. LAPs, cell maintenance enzymes, have multiple functions. LAP3, one of the critical members of M1 LAP, has multifunctional roles in tumor metastasis[77] and progressive mali
Today, IR is considered one of the primary pathogenetic mechanisms involved in developing MAFLD and its progression to NASH[23]. The prevalence of IR in patients with MAFLD is higher than in patients with simple steatosis, and in NASH it is even higher[83]. IR is traditionally described as a state in which target tissues exhibit a diminished biological response to insulin signaling at the receptor and post-receptor levels. This impairment leads to reduced glucose utilization and is accompanied by broader metabolic disturbances, particularly affecting insulin-sensitive organs such as adipose tissue and the liver[84]. Numerous biological links between adipose and liver tissues support glycolipid homeostasis, whose dysfunction creates a dysmetabolic context that can trigger the onset of MAFLD and contribute to its progression[85]. In addition, insulin physiologically plays an anabolic role in adipose tissue, suppressing lipolysis and enhancing lipogenesis, especially in the postprandial period. Conversely, in individuals with IR, altered regulatory pathways fuel lipolysis, releasing FFA, which reach the liver and cause fat overload in hepatocytes[86]. Hyperinflux of FFA causes mitochondrial dysfunction by inducing incomplete FFA and worsening hepato-IR; it also enhances gluconeogenesis by suppressing insulin-dependent glycogen synthesis[87]. In the liver, failure of insulin to adequately suppress hepatic glucose production promotes DNL, a key pathogenic mechanism underlying MAFLD[88]. These alterations are, at least in part, attributable to defects in intracellular signaling pathways downstream of the insulin receptor. Under conditions of IR, hepatic lipid accumulation reflects a combined disturbance in FFA uptake, synthesis, export, and oxidation. Consistent with this, the severity of HS in MAFLD correlates with increased circulating FFA concentrations, largely resulting from insufficient insulin-mediated inhibition of adipose tissue lipolysis.
Under physiological conditions, insulin suppresses lipolysis in adipocytes; however, IR within adipose tissue leads to excessive mobilization of FFAs[89,90]. As visceral fat mass expands, lipolysis from visceral depots becomes a dominant source of hepatic FFA influx[91]. Accordingly, patients with MAFLD exhibit markedly elevated circulating FFA levels, which have been identified as an independent predictor of advanced liver fibrosis[92]. Beyond their metabolic effects, increased FFAs also activate pro-inflammatory signaling pathways in the liver, including nuclear factor-κB (NF-κB), thereby amplifying hepatic inflammation and disease progression[93]. Thus, the release of fatty acids from dysfunctional and insulin-resistant adipocytes leads to lipotoxicity, accumulating toxic TG-derived metabolites in ectopic tissues, including the liver[94].
These mechanisms have also been demonstrated in MAFLD patients without obesity or overt diabetes, indicating that IR may precede and independently contribute to disease development. In this context, IR disrupts multiple metabolic processes, including hepatic glucose production, glucose utilization (glycogen synthesis and glucose oxidation), lipolysis, and lipid oxidation. Although VAT is not the primary contributor to circulating FFAs, it represents a major source of pro-inflammatory mediators delivered to the liver, as evidenced by the strong association between IL-6 and C-reactive protein concentrations in portal venous blood.
Under insulin-resistant conditions, the liver progressively loses its capacity to suppress glucose output in response to insulin, while DNL is simultaneously enhanced, in part through activation of the Notch signaling pathway. This dual disturbance accounts for the markedly increased contribution of DNL to hepatic lipid accumulation in MAFLD compared with healthy individuals and may also explain the heightened susceptibility to type 2 diabetes observed in this population.
Insulin-like growth factor-1 is a peptide hormone structurally related to insulin, predominantly synthesized by hepatocytes under growth hormone regulation and circulating bound to IGF-binding protein-3[95]. IGF-1 plays an important role in hepatocyte proliferation, differentiation, and survival[96,97]. Meta-analytic data indicate that circulating IGF-1 levels are reduced in patients with MAFLD compared with healthy controls, supporting a potential link between IGF-1 deficiency and disease progression[98]. Consistently, lower IGF-1 concentrations have been reported in patients with advanced liver fibrosis compared with those with absent or mild fibrosis[99]. Although the precise molecular pathways remain incompletely defined, emerging evidence suggests that IGF-1 may modulate liver fibrosis by regulating stress-induced premature hepatocyte senescence.
The evidence presented above has developed a classic pathogenetic model in which IR unilaterally fuels MAFLD[100]. From a modern perspective and in terms of identifying potential pharmacological targets, MAFLD and IR can be considered as “two faces of Janus”[100]. An increasing body of evidence supports a bidirectional relationship between MAFLD and IR, whereby HS not only results from impaired insulin signaling but may also exacerbate both hepatic and systemic IR[101]. This reciprocal interaction establishes a self-perpetuating cycle that drives disease persistence and progression, although the underlying molecular mechanisms remain incompletely defined. Within this context, IR is tightly interconnected with oxidative stress and chronic low-grade inflammation, forming a pathogenic axis that contributes to metabolic dysregulation and immune dysfunction[102]. Mitochondrial impairment, a hallmark of HS, plays a central role by promoting incomplete fatty acid oxidation, excessive generation of ROS, and accumulation of toxic lipid intermediates. These processes further amplify oxidative stress, trigger inflammatory signaling, and disrupt insulin-responsive pathways, thereby reinforcing disease progression[103].
In advanced stages of MAFLD, particularly in the presence of cirrhosis, IR represents a central driving force of chronic liver disease progression. At the same time, cirrhosis itself predisposes patients to IR irrespective of etiology, as structural and functional hepatic impairment profoundly alters glucose metabolism[104]. Nevertheless, the severity of IR, commonly assessed using the HOMA-IR, varies according to the underlying cause of cirrhosis and appears to be more pronounced in MAFLD- and hepatitis C virus (HCV)-related liver disease than in alcohol-associated liver disease or chronic hepatitis B (CHB)[105].
Hepatic-derived mediators may further aggravate IR in this setting. Certain hepatokines, such as selenoprotein P, have been shown to directly impair insulin signaling in peripheral tissues, particularly skeletal muscle, in patients with MAFLD[106]. In parallel, sustained necroinflammatory activity within the liver leads to increased production of pro-inflammatory cytokines, including TNF-α and IL-6, which contribute to systemic IR and metabolic dysregulation.
In the last decade, the role of gut-liver interactions in developing MAFLD has attracted increasing attention.
The human microbiota encompasses the diverse community of microorganisms residing in the body; however, because the vast majority are located within the gastrointestinal tract, the term “gut microbiota” is commonly used[107,108]. The intestinal ecosystem represents a highly complex microbial network composed of bacteria, viruses, fungi, and other microorganisms that coexist in a dynamic and tightly regulated equilibrium. Interactions within this microbial com
Under physiological conditions, the gut microbiota and the host exist in a state of mutual equilibrium. Disruption of this balance by genetic, dietary, or environmental factors leads to gut dysbiosis, which has been increasingly implicated in the pathogenesis of MAFLD through the gut-liver axis. The gut-liver axis refers to the bidirectional communication between the intestinal microbiota and the liver, mediated by epithelial, vascular, and immune pathways that link intestinal barrier function with hepatic circulation[110].
The intestinal barrier is a multilayered defense system composed of a mucus layer-characterized by an outer loose layer and an inner dense layer-followed by a monolayer of epithelial cells that constitutes the second major protective barrier. These epithelial cells are interconnected by tight junctions, multiprotein complexes primarily formed by claudins, occludin, and zonula occludens proteins, as well as junctional adhesion molecules[111]. Tight junctions are essential for preserving epithelial integrity, selectively permitting nutrient absorption while restricting the translocation of microbial products and pathogens across the intestinal epithelium[112].
In addition to the mucin layer produced by goblet cells and TJs between IECs, the immune system's involvement is equally important. Mucosal immune defense within the intestine is further supported by locally produced immunoglobulin A, which binds and neutralizes invading microorganisms at the luminal surface. In parallel, activation of interleukin-23-dependent signaling pathways stimulates group 3 innate lymphoid cells, leading to the secretion of interleukin-22. IL-22 plays a key role in enhancing epithelial defense by inducing the production of antimicrobial peptides from paneth cells and intestinal epithelial cells. Beyond the epithelial layer, the gut-vascular barrier provides an additional level of protection by limiting the translocation of bacteria, microbial antigens, and other luminal components into the systemic circulation[113].
The gut-liver axis constitutes an essential defense system that restricts the translocation of pathogens and toxic microbial products, thereby limiting systemic inflammation. This protective function, however, depends on the maintenance of microbial and barrier homeostasis. In the setting of gut dysbiosis, this integrated defense network becomes compromised. In MAFLD/MASLD, dysbiosis is characterized by pronounced alterations in gut microbial composition and reduced microbial diversity, which contribute to disruption of intestinal barrier function and enhanced hepatic exposure to microbial-derived signals[114].
Patients with MAFLD and MASH have altered gut microbiota composition and a higher prevalence of small intestinal bacterial overgrowth[115]. Disruption of the gut microbial ecosystem adversely affects the integrity of the gut-vascular barrier, resulting in increased intestinal vascular permeability, particularly in the context of a high-fat diet[116]. In patients with MAFLD, accumulating evidence demonstrates profound alterations in gut microbial composition, accompanied by a significant reduction in α-diversity, further supporting the role of dysbiosis in disease pathogenesis[117]. MAFLD patients often exhibit a predominantly Firmicutes-dominant gut microbiota, resulting in an increased Firmicutes to Bacteroidetes ratio compared to healthy controls[118-121].
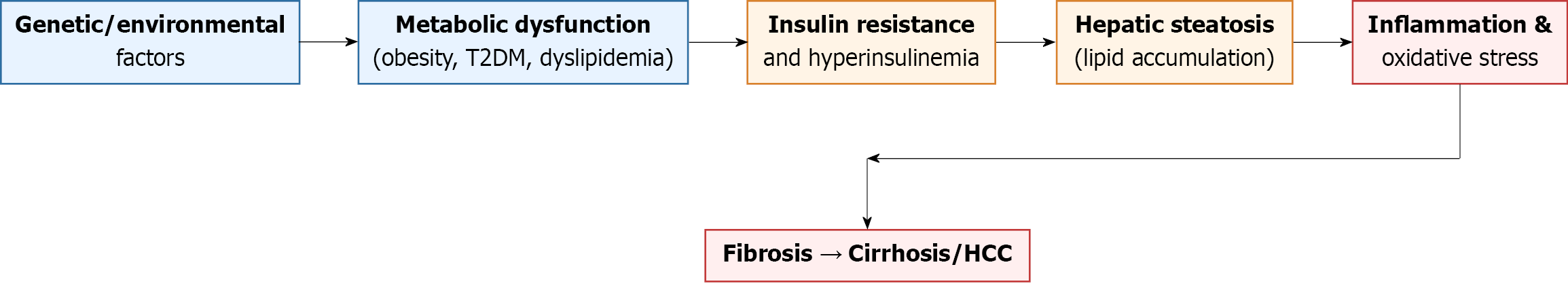
In MAFLD, enrichment of specific bacterial taxa, including Bacteroides vulgatus, Escherichia coli, and Klebsiella pneumoniae (K. pneumoniae), has been linked to adverse metabolic phenotypes such as obesity and IR. These microorganisms contribute to hepatic lipid accumulation and metabolic dysfunction through the production of lipopolysaccharides, which activate Toll-like receptor 4-dependent signaling in the liver and downstream NF-κB-mediated inflammatory pathways. Under inflammatory conditions, hepatic lipid handling is further disrupted, as reflected by reduced expression of hepatic lipase and concomitant upregulation of HMG-CoA reductase, the key regulatory enzyme of cholesterol biosynthesis[122]. This imbalance favors impaired lipid hydrolysis alongside enhanced hepatic cholesterol production, thereby exacerbating steatosis and metabolic injury[123,124]. K. pneumoniae increases oxidative stress and proinflammatory cytokine release, directly damaging hepatocytes[125]. Beneficial gut bacteria also exert protective effects against MAFLD through the production of short-chain fatty acids (SCFAs). Akkermansia muciniphila generates SCFAs that activate G protein-coupled receptors, thereby regulating host energy metabolism and conferring metabolic protection[126]. Similarly, Faecalibacterium prausnitzii produces anti-inflammatory SCFAs, particularly butyrate, which suppresses NF-κB signaling and limits the production of pro-inflammatory cytokines. Members of the genus Bifidobacterium further contribute to intestinal homeostasis by strengthening epithelial barrier function, thereby reducing endotoxin translocation into the systemic circulation. In addition, Bifidobacterium species promote immune tolerance by enhancing regulatory T cell activity and increasing the secretion of anti-inflammatory cytokines such as IL-10 and transforming growth factor-β (TGF-β), collectively attenuating hepatic and systemic inflammation[127]. The pathophysiology of the development of metabolically associated fatty liver disease can be graphically represented as follows (Figure 1).
The clinical and pathogenetic relationship between NAFLD and T2DM is beyond doubt; however, estimates of the extent of this comorbidity vary widely across studies, likely due to differences in diagnostic criteria and patient selection methods[128-130]. While the prevalence of each disease in the general population is estimated at 25% and approximately 10%, respectively[131,132], the figures are significantly higher when they coincide. According to data[133], almost half (54%) of patients with MAFLD have concomitant T2DM. However, data from individual studies are not so homogeneous: For example, in one cohort, liver steatosis in patients with T2DM was detected in 85.3%of cases[134], while in another, it was 42.6%, with NAFLD itself accounting for approximately 70% of these cases[135]. Thus, a large meta-analysis indicates that 55.48% of patients with T2DM suffer from NAFLD[136]. This wide range only highlights the complexity of epidemiological assessments and the need for standardized diagnostic protocols for this patient group. Nevertheless, the mutually aggravating influence of NAFLD and T2DM is a clinically significant fact that requires the attention of physicians.
The bidirectional nature of the MAFLD-T2DM association extends beyond mere coexistence to a mutual aggravation of clinical outcomes. While MAFLD elevates the risk of incident T2DM and its vascular complications[137], diabetes, in turn, acts as a potent accelerator of hepatic disease progression. In patients with MAFLD, the presence of T2DM is linked to a more severe histological phenotype, including NASH, advanced fibrosis, cirrhosis, and HCC[138]. This is robustly evidenced by biopsy-based studies, where T2DM conferred a 70% increased relative risk of fibrosis progression, in
Although the prevalence of the HCV is declining, CHC remains a significant cause of liver cirrhosis and HCC worldwide[143]. An estimated 58 million people worldwide suffer from CHC[144].
Chronic HCV infection represents a complex clinical entity that extends beyond purely viral liver damage, intersecting significantly with metabolic dysfunction. While the natural history of HCV infection is well-characterized-with a high rate of chronicity (60%-80%) leading to fibrosis, cirrhosis, and HCC[145-147]- its metabolic implications are of growing relevance. A striking and genotype-dependent association exists between HCV and HS[148-150]. This metabolic-viral interplay has critical clinical consequences: HCV is a leading etiology for end-stage liver disease and HCC in the West[151,152], and its numerous extrahepatic manifestations[153] further complicate management. Notably, genotype 3 infection exhibits a direct steatogenic viral effect, contributing to the remarkably high prevalence of fatty liver (40%-80%) in the HCV population[154]. Even after accounting for common metabolic confounders (e.g., obesity, diabetes), the prevalence remains elevated at approximately 40%, underscoring a multifactorial pathogenesis involving both viral and host metabolic factors[154]. Therefore, in the modern management of CHC, assessment of metabolic comorbidities is not optional but essential, particularly for risk stratification and comprehensive care in patients presenting with HS.
The revised MAFLD nomenclature introduces a critical paradigm shift in categorizing patients with HCV infection and HS. While HCV genotype 3 remains recognized as a direct viral cause of fat accumulation and is thus excluded from the standalone MAFLD diagnosis, the new criteria explicitly account for metabolic cofactors. Consequently, patients with HCV who also exhibit metabolic dysregulation now fall under the dual diagnosis of “HCV infection with concomitant MAFLD”. This creates a clear dichotomization within the HCV population: “HCV with MAFLD” vs “HCV without MAFLD” (where steatosis is presumed to be purely virally induced, typically by genotype 3).
This refined classification has direct clinical utility. By distinguishing the primary driver of steatosis-metabolic, viral, or both-it paves the way for more targeted management. The differential diagnosis should hinge on a composite assessment of metabolic risk factors, HCV genotype and viral load, and the response to antiviral therapy[155]. For instance, persistent steatosis after sustained virologic response would strongly point toward underlying metabolic disease. Thus, the new framework moves beyond mere labeling, aiming to stratify patients for combined therapeutic strategies that address both the viral infection and the metabolic syndrome where present.
The interplay between HCV and host metabolic pathways is bidirectional and central to disease pathogenesis, creating a vicious cycle that worsens outcomes. HCV actively remodels lipid metabolism to facilitate its own life cycle, impairing VLDL secretion and promoting hepatic lipogenesis, which directly contributes to HS-a common histological feature in CHC[156]. This virus-induced dysregulation manifests clinically as a distinct hypolipidemic profile (low total cholesterol, hypobetalipoproteinemia)[157].
The impact of this metabolic hijacking, however, extends beyond viral fitness to influence disease progression. Interestingly, the relationship between lipids and viral replication is complex and not merely facilitative. While lipids are essential for HCV replication, certain fatty acids and products of lipid peroxidation (a feature of NASH) can conversely inhibit it, suggesting the existence of a delicate regulatory network within a fatty liver[158,159]. Liver steatosis in CHC is a well-established accelerator of fibrosis[160] and HCC[161]. This metabolic-viral synergy culminates in significant clinical cross-talk[162,163]. HCV infection independently increases the risk of IR and type 2 diabetes[164,165], effectively creating and exacerbating the metabolic drivers of MAFLD. Furthermore, the associated metabolic milieu-particularly IR and elevated BMI is linked to a poorer response to interferon-alpha-based therapies[166]. Consequently, although a direct link to metabolic syndrome remains debated, patients with the dual burden of HCV and MAFLD face a disproportionately worse prognosis. Compared to those with HCV alone, they experience accelerated fibrogenesis, a heightened risk of HCC and atherosclerosis, and potentially suboptimal responses to certain therapies, underscoring the critical need for a combined metabolic and antiviral management approach[167].
IR emerges as a critical pathogenic nexus and a potent amplifier of liver injury in both MAFLD and CHC, with its prevalence and impact being particularly severe in the context of co-existing disease. While IR is a hallmark of MAFLD, affecting approximately 70% of patients[168], its frequency escalates dramatically to over 80% in patients with CHC and concomitant MAFLD, underscoring the synergistic role of HCV infection in disrupting carbohydrate metabolism[169]. Crucially, the severity of IR, as measured by insulin levels and the HOMA index, correlates directly with the degree of histological liver damage[170,171], positioning it not merely as a comorbidity but as a key driver of disease progression.
The mechanisms by which IR exacerbates hepatocyte damage and accelerates fibrosis are multifaceted. Hyperinsulinemia, a direct consequence of IR, promotes hepatic fibrogenesis through several pathways[172]. Furthermore, IR and the associated lipid dysregulation lead to fatty degeneration of hepatocytes and alter the lipid composition of their membranes. This cellular steatosis disrupts fundamental biochemical processes, including gluconeogenesis, rendering hepatocytes more vulnerable to injury. Against this backdrop of metabolic stress, persistent viral-driven (in CHC) and/or diet-driven inflammation creates a profibrogenic milieu, where progressive mesenchymal activation rapidly translates into collagen deposition and fibrosis[165]. Thus, IR acts as a common metabolic driver that synergizes with viral factors to fuel a self-perpetuating cycle of inflammation, hepatocyte dysfunction, and accelerated fibrogenesis.
Hepatitis B: The convergence of two major global liver epidemics-CHB and MAFLD-presents a growing clinical and public health challenge. Chronic HBV infection, affecting an estimated 248 million people worldwide, remains a leading cause of cirrhosis and HCC, accounting for nearly 680000 annual deaths[173,174].
With the parallel rise in MAFLD prevalence, co-existing disease is increasingly common. Reported prevalence rates of MAFLD in CHB cohorts vary widely, from 14% to 70%, a discrepancy largely attributable to differences in diagnostic methodologies [ultrasound (US), transient elastography, biopsy] and study population characteristics[175]. Regardless of the exact figure, this overlap is clinically significant. Both conditions independently drive chronic liver injury, progressive fibrosis, and carcinogenesis, suggesting a potential for synergistic harm. Consequently, understanding the interaction between viral and metabolic liver damage has become a priority, as their coexistence may accelerate disease progression, complicate management, and significantly threaten long-term hepatic health[176].
Effect of HBV on systemic metabolism: The relationship between HBV infection and MAFLD is complex and appears paradoxical, characterized by seemingly protective metabolic effects that contrast with synergistic harm when both diseases coexist. Epidemiological studies consistently report an inverse association between chronic HBV infection and the development of MAFLD, as evidenced by lower prevalence rates of HS, hypertriglyceridemia, and metabolic syndrome in HBsAg-positive individuals across different populations[177-179]. This protective phenotype is mechanistically linked to HBV-induced alterations in lipid metabolism, specifically a reduction in hepatic lipoprotein biosynthesis, leading to favorable serum lipid profiles (lower TG, LDL-C) and a decreased prevalence of hyperlipidemia[179-181]. Consequently, HBV-infected patients often exhibit lower intrahepatic lipid content compared to those with MAFLD alone[182]. Thus, while HBV may modestly modulate systemic lipid metabolism, likely influenced by the natural history of infection[183], it does not confer protection against the detrimental hepatic consequences of metabolic dysfunction. This inverse relationship between HBV infection and serum lipid profile may also contribute to the decreased prevalence of metabolic syndrome[184-186].
However, this apparent metabolic “benefit” does not translate into improved outcomes when MAFLD is established. On the contrary, the coexistence of MAFLD and HBV creates a high-risk phenotype. IR is more prevalent in patients with dual pathology than in those with either condition alone[187]. Critically, the presence of metabolic syndrome-a core diagnostic component of MAFLD-acts as a potent independent driver of disease progression in CHB, accelerating the risks of liver fibrosis[188], cirrhosis[189], and hepatitis B-related mortality/HCC[190], irrespective of viral load or ALT levels. The superimposition of MAFLD on CHB creates a synergistic injurious milieu where metabolic and viral insults converge, outweighing any modestly favorable lipid effects and leading to worse clinical outcomes.
Emerging evidence points to a complex immunomodulatory role of HS in the natural history of CHB, which may paradoxically suppress viral activity while potentially altering immune clearance. Clinical observations consistently reveal a negative association between HS and markers of HBV activity. This is evidenced by an increased likelihood of HBsAg seroclearance and a higher rate of transition out of the immunotolerant phase in patients with concomitant fatty liver[182,188,191,192]. Furthermore, a significant inverse correlation is noted between steatosis and HBV replication levels in humans, though the precise mechanisms remain undefined[193].
Preclinical models provide direct causal support for these clinical findings. In mouse models of co-existing MAFLD and HBV infection, HS consistently leads to a reduction in HBV DNA load and antigen levels[194,195]. Importantly, these studies suggest that the suppressive effect is unidirectional: While steatosis inhibits viral replication, HBV infection does not appear to significantly alter the underlying metabolic phenotype of MAFLD in these models[195].
The mechanisms underlying this viral suppression are likely multifactorial and may involve both direct metabolic interference and enhanced immune surveillance. Proposed pathways include: (1) Direct metabolic inhibition, where lipid accumulation within hepatocytes creates an intracellular environment unfavorable for HBV replication; (2) Activation of innate immunity by MAFLD-related lipotoxicity or inflammation, which enhances antiviral responses; and (3) MAFLD-mediated hepatocyte apoptosis, which could selectively reduce the pool of HBV-infected cells[196]. Thus, the steatotic liver microenvironment appears to exert a net restrictive pressure on HBV, adding a novel metabolic dimension to the host-virus interplay in CHB.
Obstructive sleep apnea (OSA) is increasingly recognized not merely as a sleep disorder but as a systemic metabolic stressor that contributes to the pathogenesis and progression of MAFLD through intermittent hypoxia (IH)-mediated pathways. Diagnosis, based on the apnea-hypopnea index (AHI) via polysomnography, stratifies severity from mild to severe[197-200].
Clinical evidence consistently links OSA severity with MAFLD phenotypes. Epidemiological and meta-analytic data robustly associate OSA, particularly in its moderate to severe forms, with an increased prevalence and severity of MAFLD, NASH, and liver fibrosis[201,202]. Crucially, the AHI serves as an independent predictor of significant liver fibrosis in this population, correlating with validated non-invasive markers such as fibrosis-4 index and liver stiffness measurements[203-205]. Meta-regression analyses showed that OSA was associated with steatosis, lobular inflammation, ballooning, and fibrosis independent of age, gender, BMI, and abdominal obesity[206-208].
The central mechanistic link is IH, which drives MAFLD progression via two interconnected axes: Oxidative stress and dysregulated hypoxia signaling.
Oxidative stress and mitochondrial dysfunction: The cyclical hypoxia-reoxygenation cycles of IH generate ROS, over
Hypoxia-inducible factor activation: IH potently upregulates the master transcriptional regulator hypoxia-inducible factor (HIF-1α). Elevated HIF-1α, in turn, activates a cascade of genes involved in lipogenesis, inflammation, and fibrogenesis[212,213]. This pathway integrates the hypoxic stimulus into sustained metabolic dysregulation and hepatic injury, directly promoting IR, hepatocyte damage, and the progression toward NASH and fibrosis[211,214].
In summary, OSA, through the recurrent hypoxic insult of IH, acts as a direct promoter of MAFLD by inducing a state of chronic oxidative stress and activating the HIF-1α-mediated hypoxic response, thereby accelerating steatosis, inflammation, and fibrogenesis.
Furthermore, IH and OSA critically exacerbate dyslipidemia-a hallmark of MAFLD-creating a vicious metabolic cycle. The condition is a well-established risk factor for lipid metabolism disorders, with which it shares a bidirectional relationship[215]. Mechanistically, IH enhances adipose tissue lipolysis, flooding the liver with FFA while simultaneously impairing hepatic mitochondrial β-oxidation and postprandial lipid clearance. This results in a net increase in hepatic lipid synthesis and accumulation[216]. A key mediator of this metabolic shift is HIF-2α, which is upregulated by IH and directly promotes disordered hepatic lipid metabolism. Evidence of this pathway in humans includes the significant elevation of the lipid transport molecule CD36 in the livers of OSA patients, a finding strongly correlated with AHI severity and oxygen desaturation indices[217]. Thus, the HIF-mediated hypoxic response, through both HIF-1α (inflammation, fibrosis) and HIF-2α (lipid metabolism), acts as a central conductor orchestrating multiple metabolic hits that drive MAFLD progression in the context of sleep apnea.
Ultimately, IH converges on inducing insulin and leptin resistance, a fundamental pillar in the pathogenesis of MAFLD and a critical metabolic bridge linking OSA to liver disease. Clinical data robustly position IH/OSA as an independent risk factor for the development and severity of MAFLD specifically through the induction of IR[218,219]. This is evidenced by studies where the AHI and the HOMA-IR are independently associated with MAFLD prevalence and elevated liver enzymes, even after adjustment for confounders like obesity[219,220].
Thus, the pathophysiological cascade from OSA to MAFLD can be summarized as a self-reinforcing cycle: OSA-driven IH activates hypoxic signaling (HIF-1α/2α), which simultaneously: (1) Promotes systemic and hepatic oxidative stress; (2) Dysregulates lipid metabolism promoting steatosis; and (3) Induces insulin/Leptin resistance.
The resulting hyperinsulinemia and metabolic dysfunction further accelerate hepatic lipogenesis, inflammation, and fibrogenesis, thereby fueling MAFLD progression. While the precise molecular intermediaries require further elucidation, the central role of IH-induced metabolic resistance in this pathway is unequivocal, establishing OSA not merely as a comorbidity but as an active driver of metabolic liver disease.
Emerging research points to two additional, interlinked pathways through which OSA may exacerbate MAFLD: Gut microbiota dysbiosis and the promotion of hepatocyte death.
Gut-liver axis disruption: OSA and its characteristic IH are implicated in inducing gut dysbiosis, though human data remain limited compared to animal evidence[221-224]. The proposed alterations-reduced microbial diversity, a decrease in beneficial SCFAs producers, and an increase in pro-inflammatory pathobionts like Prevotella-mirror changes observed in MAFLD[225-229]. This dysbiosis is hypothesized to increase intestinal permeability, facilitating the translocation of microbial products into the portal circulation. This, in turn, fuels hepatic inflammation and IR, creating a vicious gut-liver axis that accelerates MAFLD progression[227].
Hepatocyte death pathways: OSA-induced IH directly promotes hepatocyte injury via multiple programmed cell death mechanisms. Experimental models demonstrate that IH exacerbates apoptosis through ER and oxidative stress, effects that can be mitigated by antioxidants[230-233]. Furthermore, IH is shown to promote necroptosis, a pro-inflammatory form of cell death mediated by RIPK3/MLKL, thereby amplifying liver injury[231]. Given the crosstalk between these pathways, a holistic concept like “panoptosis”-the simultaneous activation of multiple death pathways-may best describe the hepatocyte demise in OSA-related MAFLD[234].
In summary, OSA contributes to MAFLD through a multifactorial network of interconnected insults. Systemic IH acts as the primary driver, concurrently: (1) Inducing oxidative stress and HIF activation; (2) Dysregulating lipid and glucose metabolism; (3) Altering the gut microbiota and gut-liver axis; and (4) Triggering inflammatory hepatocyte death pathways. These processes collectively create a profibrogenic and pro-steatotic hepatic microenvironment, positioning OSA as a significant modifiable risk factor in the pathogenesis and progression of metabolic liver disease.
Hypertension and MAFLD are intricately linked components of the metabolic syndrome, sharing a bidirectional relationship that significantly amplifies the risk of adverse hepatic and cardiovascular outcomes. Epidemiological data robustly support this mutual aggravation. Prospective studies demonstrate that normotensive individuals with MAFLD have a markedly higher incidence of hypertension, with risk escalating in proportion to the severity of HS[235,236]. Conversely, hypertension is an independent risk factor for the development and progression of MAFLD, including advanced fibrosis and NASH[237,238]. This synergy is evident in long-term cohorts, where MAFLD is associated with a three-fold higher incidence of hypertension, and the presence of hypertension, in turn, predicts a greater risk of liver fibrosis progression[239-242].
The pathophysiological crosstalk between MAFLD and hypertension is mediated by several interconnected me
The renin-angiotensin system: A central pathway: Dysregulation of the hepatic and systemic renin-angiotensin system (RAS) serves as a key nexus. In hypertension, angiotensin II promotes systemic inflammation and hepatic stellate cell activation, driving fibrogenesis and altering lipid metabolism to favor intrahepatic TG (IHTG) accumulation[243-245]. Reciprocally, MAFLD itself induces intrahepatic RAS overactivity, further exacerbating liver inflammation, fibrosis, and contributing to systemic vascular tension[246,247]. Pharmacological RAS inhibition in experimental models attenuates steatosis and fibrosis, highlighting its pathogenic role[247].
IR and chronic inflammation: A common soil: Shared metabolic disturbances form a critical link. IR, a hallmark of MAFLD, impairs the PI3K/Akt vasodilatory pathway in endothelium while potentiating vasoconstrictive MAPK signaling, promoting hypertension[248,249]. Concurrently, chronic low-grade inflammation in MAFLD, driven by pro-inflammatory cytokines (e.g., from activated NF-κB), not only perpetuates hepatic injury but also contributes to endothelial dysfunction and increased arterial stiffness[122,242].
Sympathetic nervous system hyperactivation and adipokine imbalance: Autonomic dysfunction bridges both con
In conclusion, the MAFLD-hypertension association is not merely coincidental but causal and bidirectional, rooted in shared pathophysiological pathways-RAS activation, IR with inflammation, and SNS dysregulation. This interplay necessitates an integrated clinical approach where the diagnosis of one condition should prompt screening and targeted management of the other to mitigate the compounded risk of hepatic and cardiovascular morbidity. The most common comorbidities associated with MAFLD are presented in Table 2.
| Condition | Mechanisms/links with MAFLD | Clinical outcomes | Ref. |
| Type 2 diabetes mellitus | Insulin resistance, chronic hyperglycemia, lipotoxicity | Faster progression to NASH and fibrosis; higher risk of CVD | Byrne et al[263] |
| Chronic hepatitis B/C | Additive hepatocellular injury, inflammation, altered lipid metabolism | Increased fibrosis progression, higher risk of HCC | Choi et al[175] |
| Arterial hypertension | Systemic inflammation, endothelial dysfunction, insulin resistance | Greater cardiovascular morbidity, worse liver outcomes | Lonardo et al[235] |
| Obstructive sleep apnea | Intermittent hypoxia, oxidative stress, systemic inflammation | Worsening fibrosis; increased CVD and metabolic complications | Zhang et al[220] |
MAFLD is now unequivocally recognized not merely as a liver-specific disorder but as a pivotal independent risk factor for cardiovascular disease (CVD), contributing significantly to both morbidity and mortality. This association extends beyond shared risk factors, positioning MAFLD as an active player in atherogenesis and cardiac dysfunction. Prospective cohort studies and meta-analyses consistently demonstrate a 1.5- to 2.5-fold increased risk of cardiovascular events in individuals with MAFLD, with risk escalating with disease severity[254-259].
Major adverse cardiovascular events: Increased incidence of coronary heart disease, myocardial infarction, and stroke[256,259].
Subclinical atherosclerosis: Strong associations with carotid plaque, increased intima-media thickness, arterial stiffness, endothelial dysfunction, and coronary artery calcification[260-272].
Mortality: CVD is a leading cause of death in the MAFLD population, with a significantly elevated all-cause and cardiovascular mortality risk[257,265,266]. Consequently, current guidelines advocate for routine cardiovascular risk assessment in all MAFLD patients[268].
The MAFLD-CVD nexus is rooted in a common soil of metabolic and inflammatory disturbances: А hallmark of MAFLD, characterized by elevated pro-inflammatory cytokines (e.g., IL-6, TNF-α), which directly promote vascular inflammation, endothelial activation, and plaque instability[273-276].
Dyslipidemia MAFLD fundamentally disrupts hepatic lipid homeostasis, generating a highly atherogenic lipid profile (the lipid triad).
Increased very-low-density lipoprotein secretion: Enhanced hepatic DNL and TG synthesis stimulate the production and secretion of VLDL[277-279].
Lipoprotein remodeling: Elevated plasma TGs promote the cholesterol ester transfer protein-mediated exchange of TGs from VLDL for cholesteryl esters from low density lipoprotein (LDL) and high-density lipoprotein (HDL). This leads to the formation of: Small, dense LDL particles: More prone to oxidation and endothelial penetration.
Dysfunctional, cholesterol-depleted HDL: With impaired atheroprotective capacity[280].
Drives hepatic DNL and systemic dyslipidemia while promoting endothelial dysfunction via impaired PI3K/Akt signaling and enhanced MAPK vasoconstrictive pathways[281-285]. Reduced adiponectin (vasoprotective) and elevated leptin levels exacerbate IR, inflammation, and vascular damage[286].
The liver’s role in synthesizing coagulation factors is dysregulated in MAFLD. Increased production of fibrinogen, factor VIII, and von Willebrand factor, alongside impaired fibrinolysis (e.g., elevated PAI-1), creates a pro-thrombotic milieu[287-290]. Fibrinogen deposition in the vessel wall facilitates LDL retention and plaque development[291-293]. In advanced disease, a fragile rebalancing occurs, often with reduced synthesis of both pro- and anti-coagulant factors, but the net effect remains an elevated risk of thrombosis[290].
The increased cardiovascular risk in MAFLD is not incidental but causal, mediated by a constellation of interrelated metabolic, inflammatory, thrombotic, and vascular insults originating from or exacerbated by the dysfunctional liver. This understanding mandates a paradigm shift in clinical management: Patients diagnosed with MAFLD must undergo comprehensive cardiovascular risk stratification and receive integrated treatment targeting both HS and cardiometabolic risk factors.
Emerging evidence positions MAFLD as a significant independent risk factor for the development and progression of chronic kidney disease (CKD), establishing a formidable liver-kidney axis within the spectrum of metabolic syndrome. This association extends beyond mere coexistence, implicating shared pathophysiology and bidirectional organ crosstalk.
Meta-analyses of large longitudinal cohorts confirm that individuals with MAFLD face a approximately 45%-80% increased risk of developing incident CKD, with the risk magnitude escalating in parallel with the severity of liver disease, particularly the stage of hepatic fibrosis[294-299]. The prevalence of CKD is substantially higher in MAFLD populations (20%-55%) compared to controls (5%-30%), and this association persists after adjustment for traditional renal and metabolic risk factors[300-308]. Notably, MAFLD appears to be more strongly associated with early-stage CKD, suggesting a role in disease initiation[308-319].
The MAFLD-CKD link is rooted in a common pathogenic milieu characterized by: (1) IR and hyperinsulinemia: A central driver that promotes glomerular hyperfiltration, endothelial dysfunction, and mesangial cell proliferation[311,316]; (2) Chronic low-grade inflammation: A hallmark of both conditions, fueled by adipokine imbalance (e.g., elevated leptin), pro-inflammatory cytokines, and oxidative stress, which directly contributes to renal tubular injury and interstitial fibrosis[315,317,319]; and (3) Dysregulated lipid metabolism: Atherogenic dyslipidemia and ectopic lipid deposition (lipotoxicity) can induce renal cellular injury via ER stress and apoptosis[311,318].
MAFLD likely contributes to CKD through multiple, intertwined systemic pathways.
Hepatokine dysregulation: The steatotic liver alters the secretion of hepatokines (e.g., fibroblast growth factor-21, fetuin-A) that can directly influence renal insulin sensitivity, inflammation, and lipid metabolism[312].
Adipokine imbalance: Adipose tissue dysfunction leads to elevated leptin (pro-fibrotic, pro-inflammatory) and reduced adiponectin, exacerbating IR and renal injury[319,320].
Dyslipidemia and HDL dysfunction: MAFLD-induced atherogenic dyslipidemia and the generation of dysfunctional, pro-inflammatory HDL particles contribute to endothelial damage and glomerulosclerosis[321,322].
RAS: Systemic and intrahepatic RAS activation, prevalent in MAFLD, promotes renal vasoconstriction, sodium retention, and fibrotic signaling, creating a direct link to hypertension and CKD progression[311,314].
Integrated stress responses: Converging pathways of ER stress, oxidative stress (mediated by Nrf2 dysregulation), and activation of transcription factors like SREBP and TGF-β create a pro-fibrotic, pro-apoptotic microenvironment in both liver and kidney cells[316-318].
In the context of advanced MAFLD progressing to cirrhosis, a distinct hepatorenal reflex becomes clinically para
The relationship between MAFLD and CKD is causal and incremental, mediated by a constellation of metabolic, inflammatory, and endocrine disturbances originating from the dysfunctional liver. The strength of association correlates with liver disease severity, particularly fibrosis stage. This evidence mandates a proactive clinical approach: Patients diagnosed with MAFLD, especially those with signs of advanced fibrosis, should be considered at high risk for CKD and warrant regular monitoring of renal function (e.g., estimated glomerular filtration rate, albuminuria). Conversely, unexplained CKD should prompt evaluation for MAFLD.
HCC represents a formidable and growing complication of MAFLD, emerging as a leading etiology of liver cancer worldwide, particularly in the absence of advanced cirrhosis[327-335]. This paradigm shift underscores MAFLD as a direct and potent oncogenic driver.
MAFLD is now recognized as the fastest-growing risk factor for HCC globally, with its attributable fraction rising dramatically, especially in Asian countries[336-338]. Projections indicate an increase in MAFLD-related HCC incidence by 47%-86% across Asia by 2030[339]. Despite the high population prevalence of MAFLD, the absolute risk of HCC in an individual patient remains low (2.4%-12.8%), highlighting the critical need to identify high-risk subgroups[340-343].
A defining and challenging feature of MAFLD-associated HCC is its frequent development in the absence of established cirrhosis, accounting for up to 50%-54% of cases[344-349]. This non-cirrhotic carcinogenesis distinguishes MAFLD from other chronic liver diseases and complicates surveillance paradigms. Key risk factors within the MAFLD spectrum include advanced liver fibrosis, T2DM (the strongest metabolic risk factor)[341,348], male gender, older age, and the presence of metabolic syndrome[346,347]. Historically, many MAFLD-driven HCC cases were misclassified as “cryptogenic”, obscuring the true burden. It is now clear that metabolic dysfunction underlies a substantial proportion of these cases[340].
Hepatocarcinogenesis in MAFLD is driven by a complex interplay of local and systemic factors.
Metabolic dysregulation and IR: Chronic hyperinsulinemia and altered hepatocyte metabolism create a pro-proliferative, anti-apoptotic microenvironment. Diabetes and metabolic syndrome are potent co-factors[347,348].
Chronic inflammation and oxidative stress: Persistent low-grade inflammation, cytokine release, and lipid peroxidation generate DNA damage and promote a tumor-promoting niche.
Gut-liver axis disruption: MAFLD-associated gut dysbiosis (e.g., reduction in Akkermansia, Bifidobacterium) increases intestinal permeability, facilitating the translocation of microbial products that fuel hepatic inflammation and carcinogenesis[350-354].
Fibrosis as a key driver: While cirrhosis is not a prerequisite, advanced fibrosis (F3-F4) is the most significant histological risk marker, acting as a surrogate for the duration and severity of liver injury and creating a pro-fibrogenic, pro-oncogenic stroma[355].
The high prevalence of non-cirrhotic HCC and patient factors like obesity create unique surveillance hurdles in MAFLD.
Limitations of standard US: While US remains the first-line surveillance tool, its sensitivity for early HCC, especially in obese patients, is suboptimal (approximately 47% in cirrhosis) and operator-dependent[356-360].
Abbreviated magnetic resonance imaging: Emerging as a highly sensitive alternative or adjunct for high-risk patients (e.g., those with advanced fibrosis, inadequate US views, or diabetes). Prospective data shows superior sensitivity of magnetic resonance imaging (MRI) over US (85.7% vs 26.7%) for early HCC detection[361-363].
Alpha-fetoprotein: Adds modest sensitivity (increasing detection by approximately 18% when combined with US) but lacks sufficient specificity or sensitivity for standalone use[360,364].
Computed tomography: Not recommended for routine surveillance due to radiation exposure and inferior contrast resolution compared to MRI for early lesions. Clinical manifestations of the most common complications of MAFLD are presented in Table 3.
| Complication | Mechanisms/links with MAFLD | Clinical outcomes | Ref. |
| Cardiovascular disease | Atherosclerosis, dyslipidemia, systemic inflammation | Myocardial infarction, stroke, heart failure | Mantovani et al[254] |
| CKD | Shared risk factors (diabetes, hypertension), systemic inflammation | Higher CKD incidence, faster progression | Agustanti et al[296] |
| HCC | Fibrosis/cirrhosis, lipotoxicity, chronic inflammation | Increased HCC risk even without cirrhosis | Tan et al[356] |
| Advanced liver disease (fibrosis, cirrhosis) | Persistent steatosis, oxidative stress, inflammation | End-stage liver disease, need for transplantation | Spahis et al[73] |
Standard treatment for MAFLD focuses on lifestyle changes leading to weight loss, which includes calorie reduction, exercise, and healthy eating[365].
Intentional weight loss through lifestyle modification remains the foundational and most effective strategy for treating MAFLD, with histological benefits directly proportional to the degree of weight reduction[366-368]. This approach targets the root cause of the disease-metabolic dysfunction.
A dose-response relationship exists between weight loss and histological improvement. Clinical evidence establishes clear therapeutic thresholds: (1) ≥ 5% body weight loss: Leads to significant improvement in HS; (2) 7%-10% body weight loss: Results in amelioration of necroinflammation (ballooning, lobular inflammation) and improvement in the NAFLD Activity Score (NAS); and (3) > 10% body weight loss: Is typically required to demonstrate regression of liver fibrosis, the most critical predictor of long-term outcomes[369,370].
The efficacy of combined dietary and exercise interventions is robust. Structured programs combining caloric restriction with moderate-intensity physical activity (e.g., ≥ 150-200 minutes per week) can achieve a mean reduction in hepatic fat content of up to 40%, with higher-intensity interventions yielding greater benefits[371-373]. The key mechanism is the reversal of positive energy balance, which reduces adipose tissue lipolysis and DNL while improving hepatic and peripheral insulin sensitivity.
Successful implementation requires a structured, behavioral framework. Effective weight loss is a chronic process best supported by intensive, behavior-based interventions that include: (1) Professional counseling (from physicians, dietitians, psychologists); (2) Self-monitoring strategies (e.g., food/activity diaries); and (3) Peer support groups and structured relapse prevention plans[374,375].
Lifestyle intervention is the first-line therapy for all patients with MAFLD. For patients with a BMI ≥ 30 kg/m2 (or ≥ 27 kg/m2 with weight-related comorbidities such as type 2 diabetes or hypertension) who fail to achieve or maintain clinically meaningful weight loss (5%-10%) through lifestyle measures alone, the addition of anti-obesity pharmacotherapy should be considered as an adjunct to enhance and sustain weight loss efforts[376].
While caloric deficit is the primary driver for reducing IHTG content, the composition of the diet plays a critical modulating role in achieving sustained metabolic and hepatic benefits. The therapeutic goal extends beyond mere weight loss to improving dietary quality and reducing systemic inflammation.
A consistent energy deficit of 500-1000 kcal/day reliably reduces IHTG and improves hepatic IR within days, with long-term adherence (e.g., 11 weeks) further enhancing peripheral glucose uptake in muscle[377-379]. This underscores that quantitative calorie control is the non-negotiable foundation of dietary therapy.
For qualitative dietary composition, the Mediterranean diet emerges as the best-supported pattern for MAFLD management. Its benefits are multifactorial.
Direct hepatic impact: Randomized trials confirm its superiority in reducing HS and improving insulin sensitivity compared to standard low-fat diets[380,381].
Anti-inflammatory and antioxidant properties: Rich in polyphenols, monounsaturated fats (olive oil), and fiber, it actively lowers systemic inflammation-a key driver of MAFLD progression. This is reflected in its favorable Dietary Inflammatory Index (DII) score[379,382].
Cardiometabolic synergy: It concurrently improves glycemic control and reduces cardiovascular risk, addressing the two leading causes of morbidity and mortality in the MAFLD population[383].
Ketogenic diet shows promise for rapid improvement in IR and steatosis due to profound carbohydrate restriction[384]. However, its long-term sustainability, safety, and effects on fibrosis and cardiovascular risk markers require further evaluation before it can be recommended as a first-line strategy.
Macrobiotic and other anti-inflammatory diets characterized by low DII scores, they share the principle of high food quality and low processed food intake, suggesting that reducing dietary inflammation is a beneficial universal principle[379].
For clinical practice, a stepwise dietary approach is recommended: (1) Establish a caloric deficit (500-1000 kcal/day deficit) tailored to the patient; (2) Implement the mediterranean diet framework as the preferred nutritional pattern to achieve this deficit, emphasizing vegetables, fruits, whole grains, nuts, and olive oil while limiting saturated fats and refined carbohydrates; and (3) Consider patient preference and adherence; while the Mediterranean diet is ideal, any sustainable diet that creates a caloric deficit and improves dietary quality (low DII) is beneficial. More restrictive regimens like ketogenic diets may be considered on a case-by-case basis with monitoring.
Regular physical activity constitutes an independent and essential therapeutic component for MAFLD, conferring metabolic and hepatic benefits that extend beyond-and often occur in the absence of-significant weight loss[385,386]. Current evidence does not establish the clear superiority of any single exercise modality (aerobic, resistance, or high-intensity interval training) for reducing HS[387]. The most significant benefit is derived from regular performance of any exercise the patient can adhere to. However, a combined approach is rational.
Aerobic exercise (e.g., brisk walking, cycling, swimming): Effectively improves cardiorespiratory fitness and whole-body insulin sensitivity.
Resistance training (weight lifting, bodyweight exercises): is crucial for preserving or increasing lean muscle mass, countering the sarcopenia often exacerbated by calorie-restricted diets in obese patients, and independently improving insulin signaling[385,386].
Minimum effective dose: Engaging in ≥ 150 minutes of moderate-intensity activity per week is associated with significant improvements in liver enzymes and metabolic health[388].
Systemic protection: Regular exercise mitigates key MAFLD risk factors, including visceral adiposity and IR, thereby lowering the risk of progression to type 2 diabetes and CVD[389,390].
Hepatic impact: Even without weight loss, sustained exercise programs (e.g., 30-60 minutes, 2-3 times per week for 6-12 weeks) reliably reduce intrahepatic lipid content by 20%-30%, as measured by MR spectroscopy[391-393]. Proposed mechanisms include reduced hepatic lipogenesis and enhanced fatty acid oxidation[392].
Modulation of the gut-liver axis via probiotics represents an emerging, though not yet firmly established, adjunctive strategy for MAFLD management, targeting underlying dysbiosis and systemic inflammation. Results from human studies using probiotic mixtures primarily Lactobacillus and Bifidobacterium strains) are promising but inconsistent.
Positive outcomes: Several randomized controlled trials report improvements in surrogate markers, including reductions in liver enzymes alanine aminotransferase, γ-glutamyltransferase, hepatic steatosis index, systemic inflammation (hs-CRP), and IR (HOMA-IR)[394-398].
Negative or neutral findings: Other well-designed studies show no significant benefit on primary liver endpoints, highlighting heterogeneity in strains, dosage, treatment duration, and patient populations[399].
Probiotics are hypothesized to benefit MAFLD by: (1) Reducing intestinal permeability and bacterial translocation; (2) Modulating immune responses and lowering systemic inflammation; and (3) Influencing host metabolism (e.g., bile acid metabolism, SCFAs production). Specific multi-strain formulations like VSL#3 have been extensively studied, showing modest improvements in metabolic and inflammatory parameters[395,397].
Research is shifting toward more targeted approaches: (1) Next-generation probiotics: Defined bacterial strains with specific metabolic functions (e.g., Akkermansia muciniphila, Faecalibacterium prausnitzii) show potent preclinical promise for improving metabolic health and reducing steatosis[400]; and (2) Precision Microbiota Therapy: Future strategies may involve personalized probiotic cocktails or fecal microbiota transplantation based on individual microbial signatures[401].
Given the current evidence, probiotics cannot be recommended as a standalone or first-line therapy for MAFLD. Their role is best viewed as a potential adjunct within a comprehensive lifestyle modification program, particularly for patients with significant dysbiosis-related symptoms (e.g., bloating, irregular bowels). Further large-scale, long-term trials with histological endpoints are required to define their precise place in the therapeutic algorithm.
Pharmacotherapy for MAFLD is adjunctive to lifestyle modification and should be tailored to the disease stage, predominant phenotype (metabolic, fibrotic), and individual patient profile, particularly the presence of T2DM and cardiovascular risk[402-424]. The goals are to reduce steatosis, resolve necroinflammation, halt fibrosis, and mitigate associated cardiometabolic risks[425-430].
GLP-1 receptor agonists: This class has emerged as a leading option due to multi-organ benefits. Beyond robust glycemic control and weight reduction (the primary driver of hepatic fat loss[424]), GLP-1 receptor agonists (GLP-1 RAs) (e.g., semaglutide, dulaglutide) demonstrate direct histological efficacy, improving steatosis, inflammation, and fibrosis in randomized trials[421-423]. Their cardiorenal protective effects solidify their position as a first-choice therapy in T2DM with MAFLD, especially with obesity or high cardiovascular risk[418,425-427].
SGLT2 inhibitors: These agents promote glucosuria, leading to weight loss, improved glycemic control, and significant reductions in IHTG and liver enzymes[428]. Their proven benefits in reducing heart failure hospitalizations and slowing CKD progression are critical, given the comorbidity profile in MAFLD[427]. While hepatic benefits are likely secondary to metabolic improvement[429], their cardiorenal protection makes them a cornerstone in T2DM management, often used in combination with GLP-1 RAs for synergistic effect[430].
Pioglitazone (PPAR-γ agonist): This insulin sensitizer has the strongest evidence for improving all key histological features of NASH, including fibrosis, in patients with and without T2DM[412,416]. It increases adiponectin and promotes fat redistribution. However, its use is limited by side effects: Weight gain, edema, and elevated fracture risk, mandating careful patient selection and monitoring[417].
Resmetirom (THR-β agonist): Representing the first FDA-approved drug specifically for NASH with moderate fibrosis, resmetirom targets hepatic metabolism. Phase 3 trials confirm its efficacy in reducing liver fat and improving NASH resolution and fibrosis[431-434]. This marks a shift towards targeted, liver-directed pharmacotherapy.
Vitamin E (α-tocopherol): As an antioxidant, high-dose vitamin E (800 IU/day) is effective in improving steatosis, inflammation, and ballooning, leading to NASH resolution in non-diabetic adults[435-438]. It does not reverse fibrosis. Safety concerns about a potential increase in all-cause mortality at high doses remain controversial but warrant caution and avoidance of long-term, indiscriminate use[439,440].
Metformin: Despite improving insulin sensitivity and offering weight loss and potential chemopreventive benefits against HCC[404,405], metformin does not lead to significant histological improvement in NASH[411]. Its role in MAFLD is primarily as a first-line glycemic control agent in T2DM, with hepatic benefits being an ancillary, not primary, outcome.
Statins: Safe and recommended in MAFLD patients for primary and secondary cardiovascular prevention, given that CVD is the leading cause of death. Some studies suggest modest improvements in liver enzymes, but this is not a treatment goal[432-434]. Concerns about hepatotoxicity are unfounded in this population.
Clinical takeaway: The pharmacological strategy should be personalized. For a diabetic patient with obesity and high CVD risk, a GLP-1 RA is optimal. For a non-diabetic with active NASH, pioglitazone or vitamin E are first-line options, with resmetirom now as a targeted alternative. All therapy rests on the foundation of lifestyle intervention. The main therapeutic approaches for MAFLD are presented in Table 4.
| Therapy | Mechanisms | Clinical outcomes/limitations | Ref. |
| Lifestyle modification | Weight loss, improved insulin sensitivity, reduced inflammation | Most effective, especially with ≥ 10% weight loss; limited adherence | Vilar-Gomez et al[369] |
| Metformin | Insulin sensitizer, improves metabolic profile | Improves liver enzymes and insulin resistance, but no histological benefit | Rena et al[403] |
| Vitamin E | Antioxidant, reduces oxidative stress | Improves steatosis and inflammation, but not fibrosis; conflicting evidence | Yakaryilmaz et al[436] |
| GLP-1 receptor agonists | Weight loss, improved insulin sensitivity, reduced inflammation | Histological improvement, promising data; long-term outcomes unknown | Hartman et al[425] |
| SGLT2 inhibitors | Improve glycemic control, promote weight loss, reduce steatosis and fibrosis | Improve metabolic and hepatic parameters; long-term outcomes lacking | Pradhan et al[429] |
For a subset of patients with severe obesity and MAFLD refractory to lifestyle and pharmacological therapy, metabolic (bariatric) surgery represents the most effective intervention, offering profound and sustained metabolic, histological, and potentially oncological benefits.
Surgery is indicated after failure of intensive conservative management. Current guidelines (e.g., ADA 2022) recommend consideration for patients with: (1) Class III obesity (BMI ≥ 40 kg/m2); or (2) Class II obesity (BMI 35.0-39.9 kg/m2) with comorbid T2DM that is not adequately controlled despite optimal medical therapy[441].
This aligns with the MAFLD population, where a significant proportion struggles to achieve and maintain the > 7%-10% weight loss required for histological improvement through non-surgical means[369]. Bariatric surgery induces transformative changes in liver histology, superior to any pharmacotherapy.
Studies demonstrate durable resolution of steatohepatitis in a majority of patients within 1 year post-surgery[442,443].
Critically, benefits extend to fibrosis, with progressive regression observed over 5 years of follow-up, addressing the key driver of long-term liver-related outcomes[443,444].
Emerging meta-analytic data suggest bariatric surgery may be associated with a reduced risk of HCC, adding a significant long-term protective dimension[445].
The safety profile in patients with compensated MAFLD-related cirrhosis appears acceptable in specialized centers, though risks are higher and require meticulous multidisciplinary assessment[446].
Roux-en-Y gastric bypass appears to offer superior metabolic and histological outcomes compared to restrictive procedures like adjustable gastric banding, likely due to more pronounced hormonal changes[447]. Less invasive endoscopic procedures offer a middle ground between pharmacotherapy and surgery: Endoscopic sleeve gastroplasty and aspiration therapy achieve significant weight loss (approximately 15%-17% total body weight loss at 12 months) and improvement in metabolic parameters[448]. Intragastric balloons have shown efficacy in improving NAS[449].
While long-term histological data are less robust than for surgery, EBMTs present a valuable option for patients with lower BMI or those seeking a less invasive approach. Metabolic surgery is not a “last resort” but a powerful, disease-modifying therapy for eligible patients. The decision should be made within a multidisciplinary team (hepatologist, endocrinologist, bariatric surgeon, dietitian) and involves weighing the proven benefits of NASH resolution and fibrosis regression against procedural risks. For those ineligible or hesitant about surgery, endoscopic bariatric therapies provide a potent intermediate step.
MAFLD is a systemic metabolic disorder with profound hepatic and extrahepatic implications. Its strong bidirectional links with type 2 diabetes, CVD, and CKD necessitate a shift from a hepatocentric to a multidisciplinary care model. Effective management is staged, with lifestyle intervention as the uncontested cornerstone. Pharmacotherapy, including novel agents like resmetirom and GLP-1 receptor agonists, should be tailored to the individual’s metabolic phenotype and fibrosis risk. For eligible patients with severe obesity, metabolic surgery offers potent disease modification. A critical clinical imperative is the bidirectional screening between MAFLD and its cardiometabolic comorbidities. Future efforts must prioritize non-invasive biomarkers for risk stratification and the development of integrated care pathways. Ultimately, addressing the MAFLD epidemic requires recognizing it as a multisystem condition, demanding collaborative management to improve long-term patient outcomes.
| 1. | Feng J, MengHuan L, TingTing Y, XueJie Y, HaiNing G. Research progress on AMPK in the pathogenesis and treatment of MASLD. Front Immunol. 2025;16:1558041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 2. | Huang DQ, Wong VWS, Rinella ME, Boursier J, Lazarus JV, Yki-Järvinen H, Loomba R. Metabolic dysfunction-associated steatotic liver disease in adults. Nat Rev Dis Primers. 2025;11:14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 161] [Article Influence: 161.0] [Reference Citation Analysis (1)] |
| 3. | Iruzubieta P, Jimenez-gonzalez C, Cabezas J, Crespo J. From NAFLD to MASLD: transforming steatotic liver disease diagnosis and management. Metab Target Organ Damage. 2025;5. [DOI] [Full Text] |
| 4. | Park Y, Ko KS, Rhee BD. Non-Alcoholic Fatty Liver Disease (NAFLD) Management in the Community. Int J Mol Sci. 2025;26:2758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 5. | Mladenova IL, Tan EF, Ng JY, Sharma P. Non-alcoholic fatty liver disease (NAFLD) and its association to cardiovascular disease: A comprehensive meta-analysis. JRSM Cardiovasc Dis. 2025;14:20480040251325929. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 6. | Pădureanu V, Forțofoiu MC, Pîrșcoveanu M, Pădureanu R, Rădulescu D, Donoiu I, Pîrșcoveanu DFV. Cardiovascular Manifestations of Patients with Non-Alcoholic Fatty Liver Disease. Metabolites. 2025;15:149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 7. | Merheb C, Gerbal-Chaloin S, Casas F, Diab-Assaf M, Daujat-Chavanieu M, Feillet-Coudray C. Omega-3 Fatty Acids, Furan Fatty Acids, and Hydroxy Fatty Acid Esters: Dietary Bioactive Lipids with Potential Benefits for MAFLD and Liver Health. Nutrients. 2025;17:1031. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 8. | Sharma S, Tiwari N, Tanwar SS. The current findings on the gut-liver axis and the molecular basis of NAFLD/NASH associated with gut microbiome dysbiosis. Naunyn Schmiedebergs Arch Pharmacol. 2025;398:11541-11579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 9. | Eslam M, Sanyal AJ, George J; International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158:1999-2014.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2794] [Cited by in RCA: 2500] [Article Influence: 416.7] [Reference Citation Analysis (5)] |
| 10. | Younossi Z, Anstee QM, Marietti M, Hardy T, Henry L, Eslam M, George J, Bugianesi E. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2018;15:11-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4585] [Cited by in RCA: 4094] [Article Influence: 511.8] [Reference Citation Analysis (5)] |
| 11. | Sarin SK, Kumar M, Eslam M, George J, Al Mahtab M, Akbar SMF, Jia J, Tian Q, Aggarwal R, Muljono DH, Omata M, Ooka Y, Han KH, Lee HW, Jafri W, Butt AS, Chong CH, Lim SG, Pwu RF, Chen DS. Liver diseases in the Asia-Pacific region: a Lancet Gastroenterology & Hepatology Commission. Lancet Gastroenterol Hepatol. 2020;5:167-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 414] [Article Influence: 69.0] [Reference Citation Analysis (1)] |
| 12. | Inoue Y, Qin B, Poti J, Sokol R, Gordon-Larsen P. Epidemiology of Obesity in Adults: Latest Trends. Curr Obes Rep. 2018;7:276-288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 238] [Cited by in RCA: 221] [Article Influence: 27.6] [Reference Citation Analysis (1)] |
| 13. | Stefan N, Schick F, Häring HU. Causes, Characteristics, and Consequences of Metabolically Unhealthy Normal Weight in Humans. Cell Metab. 2017;26:292-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 478] [Cited by in RCA: 421] [Article Influence: 46.8] [Reference Citation Analysis (2)] |
| 14. | Araújo J, Cai J, Stevens J. Prevalence of Optimal Metabolic Health in American Adults: National Health and Nutrition Examination Survey 2009-2016. Metab Syndr Relat Disord. 2019;17:46-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 15. | Pavlov CS, Teplyuk DA, Lazebnik LB, Ametov AS, Pashkova EY, Sorokoletov SM, Uspenskiy YP, Turkina SV, Ponomarenko EV, Maslakov AS. The clinician's view on the advantages and contradictions of the new nomenclature of steatotic liver disease: A review. Terapevticheskii Arkhiv. 2024;96:429-435. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 16. | Maev IV, Andreev DN, Kucheryavyy YA. Metabolically associated fatty liver disease – a disease of the 21st century: A review. Consilium Medicum. 2022;24:325-332. [RCA] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 17. | Connor CL. Fatty infiltration of the liver and the development of cirrhosis in diabetes and chronic alcoholism. Am J Pathol. 1938;14:347-364.9. [PubMed] |
| 18. | Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434-438. [PubMed] |
| 19. | Angulo P. Nonalcoholic fatty liver disease. N Engl J Med. 2002;346:1221-1231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4005] [Cited by in RCA: 3644] [Article Influence: 151.8] [Reference Citation Analysis (6)] |
| 20. | Loria P, Lonardo A, Carulli N. Should nonalcoholic fatty liver disease be renamed? Dig Dis. 2005;23:72-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 3.0] [Reference Citation Analysis (2)] |
| 21. | Kim GA, Moon JH, Kim W. Critical appraisal of metabolic dysfunction-associated steatotic liver disease: Implication of Janus-faced modernity. Clin Mol Hepatol. 2023;29:831-843. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 108] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 22. | Lim S, Kim JW, Targher G. Links between metabolic syndrome and metabolic dysfunction-associated fatty liver disease. Trends Endocrinol Metab. 2021;32:500-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 148] [Article Influence: 29.6] [Reference Citation Analysis (4)] |
| 23. | Buzzetti E, Pinzani M, Tsochatzis EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016;65:1038-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2614] [Cited by in RCA: 2359] [Article Influence: 235.9] [Reference Citation Analysis (15)] |
| 24. | Venkatesan N, Doskey LC, Malhi H. The Role of Endoplasmic Reticulum in Lipotoxicity during Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) Pathogenesis. Am J Pathol. 2023;193:1887-1899. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 25. | Vesković M, Šutulović N, Hrnčić D, Stanojlović O, Macut D, Mladenović D. The Interconnection between Hepatic Insulin Resistance and Metabolic Dysfunction-Associated Steatotic Liver Disease-The Transition from an Adipocentric to Liver-Centric Approach. Curr Issues Mol Biol. 2023;45:9084-9102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 26. | Ziamanesh F, Mohammadi M, Ebrahimpour S, Tabatabaei-Malazy O, Mosallanejad A, Larijani B. Unraveling the link between insulin resistance and Non-alcoholic fatty liver disease (or metabolic dysfunction-associated steatotic liver disease): A Narrative Review. J Diabetes Metab Disord. 2023;22:1083-1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 27. | Bellentani S, Tiribelli C. Is it time to change NAFLD and NASH nomenclature? Lancet Gastroenterol Hepatol. 2017;2:547-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 28. | Eslam M, Newsome PN, Sarin SK, Anstee QM, Targher G, Romero-Gomez M, Zelber-Sagi S, Wai-Sun Wong V, Dufour JF, Schattenberg JM, Kawaguchi T, Arrese M, Valenti L, Shiha G, Tiribelli C, Yki-Järvinen H, Fan JG, Grønbæk H, Yilmaz Y, Cortez-Pinto H, Oliveira CP, Bedossa P, Adams LA, Zheng MH, Fouad Y, Chan WK, Mendez-Sanchez N, Ahn SH, Castera L, Bugianesi E, Ratziu V, George J. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J Hepatol. 2020;73:202-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3640] [Cited by in RCA: 3337] [Article Influence: 556.2] [Reference Citation Analysis (6)] |
| 29. | Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol. 2015;62:S47-S64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2509] [Cited by in RCA: 2297] [Article Influence: 208.8] [Reference Citation Analysis (8)] |
| 30. | Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA. 2001;285:2486-2497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23048] [Cited by in RCA: 20353] [Article Influence: 814.1] [Reference Citation Analysis (17)] |
| 31. | Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC Jr, Spertus JA, Costa F; American Heart Association; National Heart, Lung, and Blood Institute. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735-2752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9267] [Cited by in RCA: 8317] [Article Influence: 396.0] [Reference Citation Analysis (4)] |
| 32. | Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Forceon Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World HeartFederation; International Atherosclerosis Society; and International Association for the Study of Obesity. Obes Metabol. 2010;7:63-65. [DOI] [Full Text] |
| 33. | De A, Bhagat N, Mehta M, Taneja S, Duseja A. Metabolic dysfunction-associated steatotic liver disease (MASLD) definition is better than MAFLD criteria for lean patients with NAFLD. J Hepatol. 2024;80:e61-e62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 107] [Article Influence: 53.5] [Reference Citation Analysis (1)] |
| 34. | Rinella ME, Lazarus JV, Ratziu V, Francque SM, Sanyal AJ, Kanwal F, Romero D, Abdelmalek MF, Anstee QM, Arab JP, Arrese M, Bataller R, Beuers U, Boursier J, Bugianesi E, Byrne CD, Castro Narro GE, Chowdhury A, Cortez-Pinto H, Cryer DR, Cusi K, El-Kassas M, Klein S, Eskridge W, Fan J, Gawrieh S, Guy CD, Harrison SA, Kim SU, Koot BG, Korenjak M, Kowdley KV, Lacaille F, Loomba R, Mitchell-Thain R, Morgan TR, Powell EE, Roden M, Romero-Gómez M, Silva M, Singh SP, Sookoian SC, Spearman CW, Tiniakos D, Valenti L, Vos MB, Wong VW, Xanthakos S, Yilmaz Y, Younossi Z, Hobbs A, Villota-Rivas M, Newsome PN; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. J Hepatol. 2023;79:1542-1556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2209] [Cited by in RCA: 1961] [Article Influence: 653.7] [Reference Citation Analysis (0)] |
| 35. | EASL SLD Summit 2023. EASL-The Home of Hepatology. Available from: https://easl.eu/event/easl-sld-summit-2023/. |
| 36. | EASL Congress 2023. EASL-The Home of Hepatology. Available from: https://easl.eu/event/easl-congress-2023/. |
| 37. | AASLD. New MASLD Nomenclature. Available from: https://www.aasld.org/new-masld-nomenclature. |
| 38. | Gatzios A, Rombaut M, Buyl K, De Kock J, Rodrigues RM, Rogiers V, Vanhaecke T, Boeckmans J. From NAFLD to MAFLD: Aligning Translational In Vitro Research to Clinical Insights. Biomedicines. 2022;10:161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 39. | Eslam M, Valenti L, Romeo S. Genetics and epigenetics of NAFLD and NASH: Clinical impact. J Hepatol. 2018;68:268-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 813] [Cited by in RCA: 738] [Article Influence: 92.3] [Reference Citation Analysis (4)] |
| 40. | Liu J, Perry RC, Ngai C, Ginsberg HN. Abstract 682: TM6SF2 is Necessary for the Late Addition of Lipid and Secretion of Fully-lipidated Very Low Density Lipoproteins From Hepg2 Cell. ATVB. 2019;39. [DOI] [Full Text] |
| 41. | Borén J, Adiels M, Björnson E, Matikainen N, Söderlund S, Rämö J, Ståhlman M, Ripatti P, Ripatti S, Palotie A, Mancina RM, Hakkarainen A, Romeo S, Packard CJ, Taskinen MR. Effects of TM6SF2 E167K on hepatic lipid and very low-density lipoprotein metabolism in humans. JCI Insight. 2020;5:e144079. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 60] [Cited by in RCA: 58] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 42. | Luukkonen PK, Nick A, Hölttä-Vuori M, Thiele C, Isokuortti E, Lallukka-Brück S, Zhou Y, Hakkarainen A, Lundbom N, Peltonen M, Orho-Melander M, Orešič M, Hyötyläinen T, Hodson L, Ikonen E, Yki-Järvinen H. Human PNPLA3-I148M variant increases hepatic retention of polyunsaturated fatty acids. JCI Insight. 2019;4:e127902. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 135] [Article Influence: 19.3] [Reference Citation Analysis (1)] |
| 43. | Bruschi FV, Claudel T, Tardelli M, Caligiuri A, Stulnig TM, Marra F, Trauner M. The PNPLA3 I148M variant modulates the fibrogenic phenotype of human hepatic stellate cells. Hepatology. 2017;65:1875-1890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 202] [Article Influence: 22.4] [Reference Citation Analysis (1)] |
| 44. | Tilson SG, Morell CM, Lenaerts AS, Park SB, Hu Z, Jenkins B, Koulman A, Liang TJ, Vallier L. Modeling PNPLA3-Associated NAFLD Using Human-Induced Pluripotent Stem Cells. Hepatology. 2021;74:2998-3017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 45. | Sodum N, Kumar G, Bojja SL, Kumar N, Rao CM. Epigenetics in NAFLD/NASH: Targets and therapy. Pharmacol Res. 2021;167:105484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 106] [Article Influence: 21.2] [Reference Citation Analysis (4)] |
| 46. | He Y, Hwang S, Cai Y, Kim SJ, Xu M, Yang D, Guillot A, Feng D, Seo W, Hou X, Gao B. MicroRNA-223 Ameliorates Nonalcoholic Steatohepatitis and Cancer by Targeting Multiple Inflammatory and Oncogenic Genes in Hepatocytes. Hepatology. 2019;70:1150-1167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 128] [Cited by in RCA: 130] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 47. | Pirola CJ, Gianotti TF, Burgueño AL, Rey-Funes M, Loidl CF, Mallardi P, Martino JS, Castaño GO, Sookoian S. Epigenetic modification of liver mitochondrial DNA is associated with histological severity of nonalcoholic fatty liver disease. Gut. 2013;62:1356-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 299] [Article Influence: 23.0] [Reference Citation Analysis (1)] |
| 48. | Page A, Paoli P, Moran Salvador E, White S, French J, Mann J. Hepatic stellate cell transdifferentiation involves genome-wide remodeling of the DNA methylation landscape. J Hepatol. 2016;64:661-673. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 123] [Cited by in RCA: 114] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 49. | Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, Harrison SA, Brunt EM, Sanyal AJ. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5962] [Cited by in RCA: 5328] [Article Influence: 666.0] [Reference Citation Analysis (5)] |
| 50. | Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9103] [Cited by in RCA: 8091] [Article Influence: 809.1] [Reference Citation Analysis (8)] |
| 51. | Wang B, Zhuang R, Luo X, Yin L, Pang C, Feng T, You H, Zhai Y, Ren Y, Zhang L, Li L, Zhao J, Hu D. Prevalence of Metabolically Healthy Obese and Metabolically Obese but Normal Weight in Adults Worldwide: A Meta-Analysis. Horm Metab Res. 2015;47:839-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 81] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 52. | Zheng Q, Lin W, Liu C, Zhou Y, Chen T, Zhang L, Zhang X, Yu S, Wu Q, Jin Z, Zhu Y. Prevalence and epidemiological determinants of metabolically obese but normal-weight in Chinese population. BMC Public Health. 2020;20:487. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 53. | van der Poorten D, Milner KL, Hui J, Hodge A, Trenell MI, Kench JG, London R, Peduto T, Chisholm DJ, George J. Visceral fat: a key mediator of steatohepatitis in metabolic liver disease. Hepatology. 2008;48:449-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 502] [Cited by in RCA: 470] [Article Influence: 26.1] [Reference Citation Analysis (1)] |
| 54. | Yu SJ, Kim W, Kim D, Yoon JH, Lee K, Kim JH, Cho EJ, Lee JH, Kim HY, Kim YJ, Kim CY. Visceral Obesity Predicts Significant Fibrosis in Patients With Nonalcoholic Fatty Liver Disease. Medicine (Baltimore). 2015;94:e2159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 94] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 55. | Leite NC, Salles GF, Cardoso CR, Villela-Nogueira CA. Serum biomarkers in type 2 diabetic patients with non-alcoholic steatohepatitis and advanced fibrosis. Hepatol Res. 2013;43:508-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 56. | Jonas MI, Kurylowicz A, Bartoszewicz Z, Lisik W, Jonas M, Wierzbicki Z, Chmura A, Pruszczyk P, Puzianowska-Kuznicka M. Interleukins 6 and 15 Levels Are Higher in Subcutaneous Adipose Tissue, but Obesity Is Associated with Their Increased Content in Visceral Fat Depots. Int J Mol Sci. 2015;16:25817-25830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 57. | Ayonrinde OT. Historical narrative from fatty liver in the nineteenth century to contemporary NAFLD - Reconciling the present with the past. JHEP Rep. 2021;3:100261. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (2)] |
| 58. | Fracanzani AL, Petta S, Lombardi R, Pisano G, Russello M, Consonni D, Di Marco V, Cammà C, Mensi L, Dongiovanni P, Valenti L, Craxì A, Fargion S. Liver and Cardiovascular Damage in Patients With Lean Nonalcoholic Fatty Liver Disease, and Association With Visceral Obesity. Clin Gastroenterol Hepatol. 2017;15:1604-1611.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 178] [Article Influence: 19.8] [Reference Citation Analysis (6)] |
| 59. | Boccatonda A, Andreetto L, D'Ardes D, Cocco G, Rossi I, Vicari S, Schiavone C, Cipollone F, Guagnano MT. From NAFLD to MAFLD: Definition, Pathophysiological Basis and Cardiovascular Implications. Biomedicines. 2023;11:883. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 79] [Article Influence: 26.3] [Reference Citation Analysis (1)] |
| 60. | Day CP, James OF. Steatohepatitis: a tale of two "hits"? Gastroenterology. 1998;114:842-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3398] [Cited by in RCA: 3120] [Article Influence: 111.4] [Reference Citation Analysis (6)] |
| 61. | Santos RD, Valenti L, Romeo S. Does nonalcoholic fatty liver disease cause cardiovascular disease? Current knowledge and gaps. Atherosclerosis. 2019;282:110-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 86] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 62. | Boeckmans J, Natale A, Buyl K, Rogiers V, De Kock J, Vanhaecke T, Rodrigues RM. Human-based systems: Mechanistic NASH modelling just around the corner? Pharmacol Res. 2018;134:257-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 63. | Caligiuri A, Gentilini A, Marra F. Molecular Pathogenesis of NASH. Int J Mol Sci. 2016;17:1575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 166] [Cited by in RCA: 151] [Article Influence: 15.1] [Reference Citation Analysis (1)] |
| 64. | Harrison SA, Ruane PJ, Freilich B, Neff G, Patil R, Behling C, Hu C, Shringarpure R, de Temple B, Fong E, Tillman EJ, Rolph T, Cheng A, Yale K. A randomized, double-blind, placebo-controlled phase IIa trial of efruxifermin for patients with compensated NASH cirrhosis. JHEP Rep. 2023;5:100563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 70] [Article Influence: 23.3] [Reference Citation Analysis (1)] |
| 65. | Ballestri S, Lonardo A, Bonapace S, Byrne CD, Loria P, Targher G. Risk of cardiovascular, cardiac and arrhythmic complications in patients with non-alcoholic fatty liver disease. World J Gastroenterol. 2014;20:1724-1745. [PubMed] [DOI] [Full Text] |
| 66. | Gracia-Sancho J, Caparrós E, Fernández-Iglesias A, Francés R. Role of liver sinusoidal endothelial cells in liver diseases. Nat Rev Gastroenterol Hepatol. 2021;18:411-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 339] [Cited by in RCA: 313] [Article Influence: 62.6] [Reference Citation Analysis (2)] |
| 67. | Sies H. Oxidative stress: from basic research to clinical application. Am J Med. 1991;91:31S-38S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 876] [Cited by in RCA: 704] [Article Influence: 20.1] [Reference Citation Analysis (1)] |
| 68. | Phaniendra A, Jestadi DB, Periyasamy L. Free radicals: properties, sources, targets, and their implication in various diseases. Indian J Clin Biochem. 2015;30:11-26. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2214] [Cited by in RCA: 1461] [Article Influence: 132.8] [Reference Citation Analysis (9)] |
| 69. | Polimeni L, Del Ben M, Baratta F, Perri L, Albanese F, Pastori D, Violi F, Angelico F. Oxidative stress: New insights on the association of non-alcoholic fatty liver disease and atherosclerosis. World J Hepatol. 2015;7:1325-1336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 167] [Article Influence: 15.2] [Reference Citation Analysis (1)] |
| 70. | Benzie IF. Lipid peroxidation: a review of causes, consequences, measurement and dietary influences. Int J Food Sci Nutr. 1996;47:233-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 175] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 71. | James OF, Day CP. Non-alcoholic steatohepatitis (NASH): a disease of emerging identity and importance. J Hepatol. 1998;29:495-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 259] [Article Influence: 9.3] [Reference Citation Analysis (3)] |
| 72. | Koek GH, Liedorp PR, Bast A. The role of oxidative stress in non-alcoholic steatohepatitis. Clin Chim Acta. 2011;412:1297-1305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 249] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 73. | Spahis S, Delvin E, Borys JM, Levy E. Oxidative Stress as a Critical Factor in Nonalcoholic Fatty Liver Disease Pathogenesis. Antioxid Redox Signal. 2017;26:519-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 325] [Cited by in RCA: 300] [Article Influence: 33.3] [Reference Citation Analysis (0)] |
| 74. | Tang SP, Mao XL, Chen YH, Yan LL, Ye LP, Li SW. Reactive Oxygen Species Induce Fatty Liver and Ischemia-Reperfusion Injury by Promoting Inflammation and Cell Death. Front Immunol. 2022;13:870239. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 100] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 75. | Reiniers MJ, van Golen RF, van Gulik TM, Heger M. Reactive oxygen and nitrogen species in steatotic hepatocytes: a molecular perspective on the pathophysiology of ischemia-reperfusion injury in the fatty liver. Antioxid Redox Signal. 2014;21:1119-1142. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 76. | Matsui M, Fowler JH, Walling LL. Leucine aminopeptidases: diversity in structure and function. Biol Chem. 2006;387:1535-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 252] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 77. | He X, Huang Q, Qiu X, Liu X, Sun G, Guo J, Ding Z, Yang L, Ban N, Tao T, Wang D. LAP3 promotes glioma progression by regulating proliferation, migration and invasion of glioma cells. Int J Biol Macromol. 2015;72:1081-1089. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 78. | Tian SY, Chen SH, Shao BF, Cai HY, Zhou Y, Zhou YL, Xu AB. Expression of leucine aminopeptidase 3 (LAP3) correlates with prognosis and malignant development of human hepatocellular carcinoma (HCC). Int J Clin Exp Pathol. 2014;7:3752-3762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 71] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 79. | Fang C, Zhang J, Yang H, Peng L, Wang K, Wang Y, Zhao X, Liu H, Dou C, Shi L, Zhao C, Liang S, Li D, Wang X. Leucine aminopeptidase 3 promotes migration and invasion of breast cancer cells through upregulation of fascin and matrix metalloproteinases-2/9 expression. J Cell Biochem. 2019;120:3611-3620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 80. | Jeong SH, Kim HB, Kim MC, Lee JM, Lee JH, Kim JH, Kim JW, Park WY, Kim SY, Kim JB, Kim H, Kim JM, Choi HS, Lim DS. Hippo-mediated suppression of IRS2/AKT signaling prevents hepatic steatosis and liver cancer. J Clin Invest. 2018;128:1010-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 148] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 81. | Didangelos A. COVID-19 Hyperinflammation: What about Neutrophils? mSphere. 2020;5:e00367-e00320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 96] [Article Influence: 16.0] [Reference Citation Analysis (1)] |
| 82. | Niu L, Geyer PE, Wewer Albrechtsen NJ, Gluud LL, Santos A, Doll S, Treit PV, Holst JJ, Knop FK, Vilsbøll T, Junker A, Sachs S, Stemmer K, Müller TD, Tschöp MH, Hofmann SM, Mann M. Plasma proteome profiling discovers novel proteins associated with non-alcoholic fatty liver disease. Mol Syst Biol. 2019;15:e8793. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 264] [Cited by in RCA: 227] [Article Influence: 32.4] [Reference Citation Analysis (5)] |
| 83. | Armandi A, Rosso C, Caviglia GP, Bugianesi E. Insulin Resistance across the Spectrum of Nonalcoholic Fatty Liver Disease. Metabolites. 2021;11:155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 84. | Palma R, Pronio A, Romeo M, Scognamiglio F, Ventriglia L, Ormando VM, Lamazza A, Pontone S, Federico A, Dallio M. The Role of Insulin Resistance in Fueling NAFLD Pathogenesis: From Molecular Mechanisms to Clinical Implications. J Clin Med. 2022;11:3649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 76] [Article Influence: 19.0] [Reference Citation Analysis (5)] |
| 85. | Yki-Järvinen H, Luukkonen PK, Hodson L, Moore JB. Dietary carbohydrates and fats in nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2021;18:770-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 200] [Article Influence: 40.0] [Reference Citation Analysis (2)] |
| 86. | Geng Y, Faber KN, de Meijer VE, Blokzijl H, Moshage H. How does hepatic lipid accumulation lead to lipotoxicity in non-alcoholic fatty liver disease? Hepatol Int. 2021;15:21-35. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 369] [Cited by in RCA: 324] [Article Influence: 64.8] [Reference Citation Analysis (1)] |
| 87. | Sekizkardes H, Chung ST, Chacko S, Haymond MW, Startzell M, Walter M, Walter PJ, Lightbourne M, Brown RJ. Free fatty acid processing diverges in human pathologic insulin resistance conditions. J Clin Invest. 2020;130:3592-3602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (1)] |
| 88. | Sakurai Y, Kubota N, Yamauchi T, Kadowaki T. Role of Insulin Resistance in MAFLD. Int J Mol Sci. 2021;22:4156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 376] [Cited by in RCA: 311] [Article Influence: 62.2] [Reference Citation Analysis (1)] |
| 89. | Londos C, Honnor RC, Dhillon GS. cAMP-dependent protein kinase and lipolysis in rat adipocytes. III. Multiple modes of insulin regulation of lipolysis and regulation of insulin responses by adenylate cyclase regulators. J Biol Chem. 1985;260:15139-15145. [PubMed] |
| 90. | Softic S, Boucher J, Solheim MH, Fujisaka S, Haering MF, Homan EP, Winnay J, Perez-Atayde AR, Kahn CR. Lipodystrophy Due to Adipose Tissue-Specific Insulin Receptor Knockout Results in Progressive NAFLD. Diabetes. 2016;65:2187-2200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 111] [Cited by in RCA: 101] [Article Influence: 10.1] [Reference Citation Analysis (1)] |
| 91. | Nielsen S, Guo Z, Johnson CM, Hensrud DD, Jensen MD. Splanchnic lipolysis in human obesity. J Clin Invest. 2004;113:1582-1588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 735] [Cited by in RCA: 640] [Article Influence: 29.1] [Reference Citation Analysis (1)] |
| 92. | Zhang J, Zhao Y, Xu C, Hong Y, Lu H, Wu J, Chen Y. Association between serum free fatty acid levels and nonalcoholic fatty liver disease: a cross-sectional study. Sci Rep. 2014;4:5832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 143] [Article Influence: 11.9] [Reference Citation Analysis (3)] |
| 93. | Boden G, She P, Mozzoli M, Cheung P, Gumireddy K, Reddy P, Xiang X, Luo Z, Ruderman N. Free fatty acids produce insulin resistance and activate the proinflammatory nuclear factor-kappaB pathway in rat liver. Diabetes. 2005;54:3458-3465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 399] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 94. | Cusi K. Role of obesity and lipotoxicity in the development of nonalcoholic steatohepatitis: pathophysiology and clinical implications. Gastroenterology. 2012;142:711-725.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 735] [Cited by in RCA: 683] [Article Influence: 48.8] [Reference Citation Analysis (5)] |
| 95. | McMillan DE. Increased levels of acute-phase serum proteins in diabetes. Metabolism. 1989;38:1042-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 117] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 96. | Donnelly KL, Smith CI, Schwarzenberg SJ, Jessurun J, Boldt MD, Parks EJ. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J Clin Invest. 2005;115:1343-1351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2984] [Cited by in RCA: 2684] [Article Influence: 127.8] [Reference Citation Analysis (1)] |
| 97. | Yao Y, Miao X, Zhu D, Li D, Zhang Y, Song C, Liu K. Insulin-like growth factor-1 and non-alcoholic fatty liver disease: a systemic review and meta-analysis. Endocrine. 2019;65:227-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 98. | Bou Matar D, Zhra M, Nassar WK, Altemyatt H, Naureen A, Abotouk N, Elahi MA, Aljada A. Adipose tissue dysfunction disrupts metabolic homeostasis: mechanisms linking fat dysregulation to disease. Front Endocrinol (Lausanne). 2025;16:1592683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 35.0] [Reference Citation Analysis (4)] |
| 99. | Hagström H, Stål P, Hultcrantz R, Brismar K, Ansurudeen I. IGFBP-1 and IGF-I as markers for advanced fibrosis in NAFLD - a pilot study. Scand J Gastroenterol. 2017;52:1427-1434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 100. | Bugianesi E, Zannoni C, Vanni E, Marzocchi R, Marchesini G. Non-alcoholic fatty liver and insulin resistance: a cause-effect relationship? Dig Liver Dis. 2004;36:165-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 66] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 101. | Yki-Järvinen H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014;2:901-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1082] [Cited by in RCA: 982] [Article Influence: 81.8] [Reference Citation Analysis (6)] |
| 102. | Barazzoni R, Gortan Cappellari G, Ragni M, Nisoli E. Insulin resistance in obesity: an overview of fundamental alterations. Eat Weight Disord. 2018;23:149-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 242] [Article Influence: 30.3] [Reference Citation Analysis (4)] |
| 103. | Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A. Role of Interleukin-6 in Development of Insulin Resistance and Type 2 Diabetes Mellitus. Crit Rev Eukaryot Gene Expr. 2017;27:229-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 250] [Article Influence: 27.8] [Reference Citation Analysis (1)] |
| 104. | Goral V, Atalay R, Kucukoner M. Insulin resistance in liver cirrhosis. Hepatogastroenterology. 2010;57:309-315. [PubMed] |
| 105. | Goswami A, Bhargava N, Dadhich S, Kulamarva G. Insulin resistance in euglycemic cirrhosis. Ann Gastroenterol. 2014;27:237-243. [PubMed] |
| 106. | Misu H. Identification of hepatokines involved in pathology of type 2 diabetes and obesity. Endocr J. 2019;66:659-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 107. | Vallianou NG, Kounatidis D, Psallida S, Vythoulkas-Biotis N, Adamou A, Zachariadou T, Kargioti S, Karampela I, Dalamaga M. NAFLD/MASLD and the Gut-Liver Axis: From Pathogenesis to Treatment Options. Metabolites. 2024;14:366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 59] [Article Influence: 29.5] [Reference Citation Analysis (3)] |
| 108. | Fay KT, Ford ML, Coopersmith CM. The intestinal microenvironment in sepsis. Biochim Biophys Acta Mol Basis Dis. 2017;1863:2574-2583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 136] [Article Influence: 15.1] [Reference Citation Analysis (2)] |
| 109. | Breuninger TA, Wawro N, Breuninger J, Reitmeier S, Clavel T, Six-Merker J, Pestoni G, Rohrmann S, Rathmann W, Peters A, Grallert H, Meisinger C, Haller D, Linseisen J. Associations between habitual diet, metabolic disease, and the gut microbiota using latent Dirichlet allocation. Microbiome. 2021;9:61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 110. | Ohtani N, Kamiya T, Kawada N. Recent updates on the role of the gut-liver axis in the pathogenesis of NAFLD/NASH, HCC, and beyond. Hepatol Commun. 2023;7:e0241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (1)] |
| 111. | DiGuilio KM, Del Rio EA, Harty RN, Mullin JM. Micronutrients at Supplemental Levels, Tight Junctions and Epithelial Barrier Function: A Narrative Review. Int J Mol Sci. 2024;25:3452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 112. | Benedé-Ubieto R, Cubero FJ, Nevzorova YA. Breaking the barriers: the role of gut homeostasis in Metabolic-Associated Steatotic Liver Disease (MASLD). Gut Microbes. 2024;16:2331460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 149] [Article Influence: 74.5] [Reference Citation Analysis (1)] |
| 113. | Noval Rivas M, Wakita D, Franklin MK, Carvalho TT, Abolhesn A, Gomez AC, Fishbein MC, Chen S, Lehman TJ, Sato K, Shibuya A, Fasano A, Kiyono H, Abe M, Tatsumoto N, Yamashita M, Crother TR, Shimada K, Arditi M. Intestinal Permeability and IgA Provoke Immune Vasculitis Linked to Cardiovascular Inflammation. Immunity. 2019;51:508-521.e6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 118] [Article Influence: 16.9] [Reference Citation Analysis (1)] |
| 114. | Younossi ZM, Stepanova M, Ong J, Trimble G, AlQahtani S, Younossi I, Ahmed A, Racila A, Henry L. Nonalcoholic Steatohepatitis Is the Most Rapidly Increasing Indication for Liver Transplantation in the United States. Clin Gastroenterol Hepatol. 2021;19:580-589.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 436] [Article Influence: 87.2] [Reference Citation Analysis (2)] |
| 115. | Wijarnpreecha K, Lou S, Watthanasuntorn K, Kroner PT, Cheungpasitporn W, Lukens FJ, Pungpapong S, Keaveny AP, Ungprasert P. Small intestinal bacterial overgrowth and nonalcoholic fatty liver disease: a systematic review and meta-analysis. Eur J Gastroenterol Hepatol. 2020;32:601-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 44] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 116. | Mouries J, Brescia P, Silvestri A, Spadoni I, Sorribas M, Wiest R, Mileti E, Galbiati M, Invernizzi P, Adorini L, Penna G, Rescigno M. Microbiota-driven gut vascular barrier disruption is a prerequisite for non-alcoholic steatohepatitis development. J Hepatol. 2019;71:1216-1228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 588] [Cited by in RCA: 541] [Article Influence: 77.3] [Reference Citation Analysis (2)] |
| 117. | Fang J, Yu CH, Li XJ, Yao JM, Fang ZY, Yoon SH, Yu WY. Gut dysbiosis in nonalcoholic fatty liver disease: pathogenesis, diagnosis, and therapeutic implications. Front Cell Infect Microbiol. 2022;12:997018. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 183] [Cited by in RCA: 151] [Article Influence: 37.8] [Reference Citation Analysis (1)] |
| 118. | Hrncir T, Hrncirova L, Kverka M, Hromadka R, Machova V, Trckova E, Kostovcikova K, Kralickova P, Krejsek J, Tlaskalova-Hogenova H. Gut Microbiota and NAFLD: Pathogenetic Mechanisms, Microbiota Signatures, and Therapeutic Interventions. Microorganisms. 2021;9:957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 136] [Article Influence: 27.2] [Reference Citation Analysis (5)] |
| 119. | Aron-Wisnewsky J, Vigliotti C, Witjes J, Le P, Holleboom AG, Verheij J, Nieuwdorp M, Clément K. Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat Rev Gastroenterol Hepatol. 2020;17:279-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 937] [Cited by in RCA: 815] [Article Influence: 135.8] [Reference Citation Analysis (5)] |
| 120. | Tokuhara D. Role of the Gut Microbiota in Regulating Non-alcoholic Fatty Liver Disease in Children and Adolescents. Front Nutr. 2021;8:700058. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 67] [Article Influence: 13.4] [Reference Citation Analysis (2)] |
| 121. | Jasirwan COM, Muradi A, Hasan I, Simadibrata M, Rinaldi I. Correlation of gut Firmicutes/Bacteroidetes ratio with fibrosis and steatosis stratified by body mass index in patients with non-alcoholic fatty liver disease. Biosci Microbiota Food Health. 2021;40:50-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (2)] |
| 122. | Cai D, Yuan M, Frantz DF, Melendez PA, Hansen L, Lee J, Shoelson SE. Local and systemic insulin resistance resulting from hepatic activation of IKK-beta and NF-kappaB. Nat Med. 2005;11:183-190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1866] [Cited by in RCA: 1742] [Article Influence: 83.0] [Reference Citation Analysis (5)] |
| 123. | Feingold KR, Hardardottir I, Memon R, Krul EJ, Moser AH, Taylor JM, Grunfeld C. Effect of endotoxin on cholesterol biosynthesis and distribution in serum lipoproteins in Syrian hamsters. J Lipid Res. 1993;34:2147-2158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 130] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 124. | Feingold KR, Memon RA, Moser AH, Shigenaga JK, Grunfeld C. Endotoxin and interleukin-1 decrease hepatic lipase mRNA levels. Atherosclerosis. 1999;142:379-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 44] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 125. | Kimura I, Inoue D, Hirano K, Tsujimoto G. The SCFA Receptor GPR43 and Energy Metabolism. Front Endocrinol (Lausanne). 2014;5:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 212] [Article Influence: 17.7] [Reference Citation Analysis (4)] |
| 126. | Miquel S, Martín R, Rossi O, Bermúdez-Humarán LG, Chatel JM, Sokol H, Thomas M, Wells JM, Langella P. Faecalibacterium prausnitzii and human intestinal health. Curr Opin Microbiol. 2013;16:255-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 891] [Article Influence: 68.5] [Reference Citation Analysis (0)] |
| 127. | Kwon HK, Lee CG, So JS, Chae CS, Hwang JS, Sahoo A, Nam JH, Rhee JH, Hwang KC, Im SH. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci U S A. 2010;107:2159-2164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 523] [Cited by in RCA: 466] [Article Influence: 29.1] [Reference Citation Analysis (5)] |
| 128. | Petit JM, Cercueil JP, Loffroy R, Denimal D, Bouillet B, Fourmont C, Chevallier O, Duvillard L, Vergès B. Effect of Liraglutide Therapy on Liver Fat Content in Patients With Inadequately Controlled Type 2 Diabetes: The Lira-NAFLD Study. J Clin Endocrinol Metab. 2017;102:407-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 86] [Article Influence: 9.6] [Reference Citation Analysis (7)] |
| 129. | Younossi ZM. Non-alcoholic fatty liver disease - A global public health perspective. J Hepatol. 2019;70:531-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1844] [Cited by in RCA: 1614] [Article Influence: 230.6] [Reference Citation Analysis (6)] |
| 130. | Qi Y, Fan L, Ran D, Xu J, Wang Y, Wu J, Zhang Z. Main Risk Factors of Type 2 Diabetes Mellitus with Nonalcoholic Fatty Liver Disease and Hepatocellular Carcinoma. J Oncol. 2021;2021:7764817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 131. | Riazi K, Azhari H, Charette JH, Underwood FE, King JA, Afshar EE, Swain MG, Congly SE, Kaplan GG, Shaheen AA. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2022;7:851-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1837] [Cited by in RCA: 1585] [Article Influence: 396.3] [Reference Citation Analysis (6)] |
| 133. | Amiri Dash Atan N, Koushki M, Motedayen M, Dousti M, Sayehmiri F, Vafaee R, Norouzinia M, Gholami R. Type 2 diabetes mellitus and non-alcoholic fatty liver disease: a systematic review and meta-analysis. Gastroenterol Hepatol Bed Bench. 2017;10:S1-S7. [PubMed] |
| 134. | Targher G, Bertolini L, Padovani R, Rodella S, Tessari R, Zenari L, Day C, Arcaro G. Prevalence of nonalcoholic fatty liver disease and its association with cardiovascular disease among type 2 diabetic patients. Diabetes Care. 2007;30:1212-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 795] [Cited by in RCA: 698] [Article Influence: 36.7] [Reference Citation Analysis (4)] |
| 135. | Williamson RM, Price JF, Glancy S, Perry E, Nee LD, Hayes PC, Frier BM, Van Look LA, Johnston GI, Reynolds RM, Strachan MW; Edinburgh Type 2 Diabetes Study Investigators. Prevalence of and risk factors for hepatic steatosis and nonalcoholic Fatty liver disease in people with type 2 diabetes: the Edinburgh Type 2 Diabetes Study. Diabetes Care. 2011;34:1139-1144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 300] [Article Influence: 20.0] [Reference Citation Analysis (1)] |
| 136. | Younossi ZM, Golabi P, de Avila L, Paik JM, Srishord M, Fukui N, Qiu Y, Burns L, Afendy A, Nader F. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J Hepatol. 2019;71:793-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1890] [Cited by in RCA: 1685] [Article Influence: 240.7] [Reference Citation Analysis (4)] |
| 137. | Mantovani A, Dalbeni A, Beatrice G, Cappelli D, Gomez-Peralta F. Non-Alcoholic Fatty Liver Disease and Risk of Macro- and Microvascular Complications in Patients with Type 2 Diabetes. J Clin Med. 2022;11:968. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 37] [Article Influence: 9.3] [Reference Citation Analysis (1)] |
| 138. | Zhang X, Yip TC, Tse YK, Hui VW, Li G, Lin H, Liang LY, Lai JC, Chan HL, Chan SL, Kong AP, Wong GL, Wong VW. Duration of type 2 diabetes and liver-related events in nonalcoholic fatty liver disease: A landmark analysis. Hepatology. 2023;78:1816-1827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 139. | Huang DQ, Wilson LA, Behling C, Kleiner DE, Kowdley KV, Dasarathy S, Amangurbanova M, Terrault NA, Diehl AM, Chalasani N, Neuschwander-Tetri BA, Sanyal AJ, Tonascia J; NASH Clinical Research Network, Loomba R. Fibrosis Progression Rate in Biopsy-Proven Nonalcoholic Fatty Liver Disease Among People With Diabetes Versus People Without Diabetes: A Multicenter Study. Gastroenterology. 2023;165:463-472.e5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 143] [Article Influence: 47.7] [Reference Citation Analysis (1)] |
| 140. | Castera L, Laouenan C, Vallet-Pichard A, Vidal-Trécan T, Manchon P, Paradis V, Roulot D, Gault N, Boitard C, Terris B, Bihan H, Julla JB, Radu A, Poynard T, Brzustowsky A, Larger E, Czernichow S, Pol S, Bedossa P, Valla D, Gautier JF; QUID-NASH investigators. High Prevalence of NASH and Advanced Fibrosis in Type 2 Diabetes: A Prospective Study of 330 Outpatients Undergoing Liver Biopsies for Elevated ALT, Using a Low Threshold. Diabetes Care. 2023;46:1354-1362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 98] [Article Influence: 32.7] [Reference Citation Analysis (1)] |
| 141. | Taylor RS, Taylor RJ, Bayliss S, Hagström H, Nasr P, Schattenberg JM, Ishigami M, Toyoda H, Wai-Sun Wong V, Peleg N, Shlomai A, Sebastiani G, Seko Y, Bhala N, Younossi ZM, Anstee QM, McPherson S, Newsome PN. Association Between Fibrosis Stage and Outcomes of Patients With Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-Analysis. Gastroenterology. 2020;158:1611-1625.e12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 899] [Cited by in RCA: 823] [Article Influence: 137.2] [Reference Citation Analysis (4)] |
| 142. | Zoppini G, Fedeli U, Gennaro N, Saugo M, Targher G, Bonora E. Mortality from chronic liver diseases in diabetes. Am J Gastroenterol. 2014;109:1020-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 125] [Article Influence: 10.4] [Reference Citation Analysis (1)] |
| 143. | European Association for the Study of the Liver; Clinical Practice Guidelines Panel: Chair:; EASL Governing Board representative:; Panel members:. EASL recommendations on treatment of hepatitis C: Final update of the series(☆). J Hepatol. 2020;73:1170-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 999] [Cited by in RCA: 890] [Article Influence: 148.3] [Reference Citation Analysis (2)] |
| 144. | World Health Organization. Updated recommendations on treatment of adolescents and children with chronic HCV infection, and HCV simplified service delivery and diagnostics. Geneva: World Health Organization, 2022. |
| 145. | Samuel S, Abulawi A, Malik R. Hepatitis C and Nonalcoholic Steatohepatitis in the 21st Century: Impact on Liver Disease and Liver Transplantation. Gastroenterol Insights. 2023;14:249-270. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 146. | Bang BR, Elmasry S, Saito T. Organ system view of the hepatic innate immunity in HCV infection. J Med Virol. 2016;88:2025-2037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 147. | Stanaway JD, Flaxman AD, Naghavi M, Fitzmaurice C, Vos T, Abubakar I, Abu-Raddad LJ, Assadi R, Bhala N, Cowie B, Forouzanfour MH, Groeger J, Hanafiah KM, Jacobsen KH, James SL, MacLachlan J, Malekzadeh R, Martin NK, Mokdad AA, Mokdad AH, Murray CJL, Plass D, Rana S, Rein DB, Richardus JH, Sanabria J, Saylan M, Shahraz S, So S, Vlassov VV, Weiderpass E, Wiersma ST, Younis M, Yu C, El Sayed Zaki M, Cooke GS. The global burden of viral hepatitis from 1990 to 2013: findings from the Global Burden of Disease Study 2013. Lancet. 2016;388:1081-1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1101] [Cited by in RCA: 1002] [Article Influence: 100.2] [Reference Citation Analysis (2)] |
| 148. | Smith DB, Bukh J, Kuiken C, Muerhoff AS, Rice CM, Stapleton JT, Simmonds P. Expanded classification of hepatitis C virus into 7 genotypes and 67 subtypes: updated criteria and genotype assignment web resource. Hepatology. 2014;59:318-327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1098] [Cited by in RCA: 976] [Article Influence: 81.3] [Reference Citation Analysis (0)] |
| 149. | Rosen HR. Clinical practice. Chronic hepatitis C infection. N Engl J Med. 2011;364:2429-2438. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 365] [Cited by in RCA: 311] [Article Influence: 20.7] [Reference Citation Analysis (4)] |
| 150. | Polaris Observatory HCV Collaborators. Global prevalence and genotype distribution of hepatitis C virus infection in 2015: a modelling study. Lancet Gastroenterol Hepatol. 2017;2:161-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1292] [Reference Citation Analysis (0)] |
| 151. | Modi AA, Liang TJ. Hepatitis C: a clinical review. Oral Dis. 2008;14:10-14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 152. | AASLD-IDSA HCV Guidance Panel. Hepatitis C Guidance 2018 Update: AASLD-IDSA Recommendations for Testing, Managing, and Treating Hepatitis C Virus Infection. Clin Infect Dis. 2018;67:1477-1492. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 525] [Cited by in RCA: 496] [Article Influence: 62.0] [Reference Citation Analysis (27)] |
| 153. | El-Zayadi AR, Anis M. Hepatitis C virus induced insulin resistance impairs response to anti viral therapy. World J Gastroenterol. 2012;18:212-224. [PubMed] [DOI] [Full Text] |
| 154. | Wang X, Xie Q. Metabolic Dysfunction-associated Fatty Liver Disease (MAFLD) and Viral Hepatitis. J Clin Transl Hepatol. 2022;10:128-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 155. | Negro F. Facts and fictions of HCV and comorbidities: steatosis, diabetes mellitus, and cardiovascular diseases. J Hepatol. 2014;61:S69-S78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 136] [Article Influence: 11.3] [Reference Citation Analysis (3)] |
| 156. | Negro F. Abnormalities of lipid metabolism in hepatitis C virus infection. Gut. 2010;59:1279-1287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 157. | Schaefer EA, Chung RT. HCV and host lipids: an intimate connection. Semin Liver Dis. 2013;33:358-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 158. | Yamane D, McGivern DR, Wauthier E, Yi M, Madden VJ, Welsch C, Antes I, Wen Y, Chugh PE, McGee CE, Widman DG, Misumi I, Bandyopadhyay S, Kim S, Shimakami T, Oikawa T, Whitmire JK, Heise MT, Dittmer DP, Kao CC, Pitson SM, Merrill AH Jr, Reid LM, Lemon SM. Regulation of the hepatitis C virus RNA replicase by endogenous lipid peroxidation. Nat Med. 2014;20:927-935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 130] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 159. | Hofmann S, Krajewski M, Scherer C, Scholz V, Mordhorst V, Truschow P, Schöbel A, Reimer R, Schwudke D, Herker E. Complex lipid metabolic remodeling is required for efficient hepatitis C virus replication. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863:1041-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 160. | Fartoux L, Chazouillères O, Wendum D, Poupon R, Serfaty L. Impact of steatosis on progression of fibrosis in patients with mild hepatitis C. Hepatology. 2005;41:82-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 143] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 161. | Ohata K, Hamasaki K, Toriyama K, Matsumoto K, Saeki A, Yanagi K, Abiru S, Nakagawa Y, Shigeno M, Miyazoe S, Ichikawa T, Ishikawa H, Nakao K, Eguchi K. Hepatic steatosis is a risk factor for hepatocellular carcinoma in patients with chronic hepatitis C virus infection. Cancer. 2003;97:3036-3043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 238] [Article Influence: 10.3] [Reference Citation Analysis (1)] |
| 162. | Poynard T, Ratziu V, McHutchison J, Manns M, Goodman Z, Zeuzem S, Younossi Z, Albrecht J. Effect of treatment with peginterferon or interferon alfa-2b and ribavirin on steatosis in patients infected with hepatitis C. Hepatology. 2003;38:75-85. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 473] [Cited by in RCA: 426] [Article Influence: 18.5] [Reference Citation Analysis (2)] |
| 163. | Rubbia-Brandt L, Giostra E, Mentha G, Quadri R, Negro F. Expression of liver steatosis in hepatitis C virus infection and pattern of response to alpha-interferon. J Hepatol. 2001;35:307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 164. | White DL, Ratziu V, El-Serag HB. Hepatitis C infection and risk of diabetes: a systematic review and meta-analysis. J Hepatol. 2008;49:831-844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 339] [Cited by in RCA: 306] [Article Influence: 17.0] [Reference Citation Analysis (1)] |
| 165. | Hui JM, Sud A, Farrell GC, Bandara P, Byth K, Kench JG, McCaughan GW, George J. Insulin resistance is associated with chronic hepatitis C virus infection and fibrosis progression [corrected]. Gastroenterology. 2003;125:1695-1704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 517] [Article Influence: 22.5] [Reference Citation Analysis (2)] |
| 166. | Romero-Gómez M, Del Mar Viloria M, Andrade RJ, Salmerón J, Diago M, Fernández-Rodríguez CM, Corpas R, Cruz M, Grande L, Vázquez L, Muñoz-De-Rueda P, López-Serrano P, Gila A, Gutiérrez ML, Pérez C, Ruiz-Extremera A, Suárez E, Castillo J. Insulin resistance impairs sustained response rate to peginterferon plus ribavirin in chronic hepatitis C patients. Gastroenterology. 2005;128:636-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 575] [Cited by in RCA: 522] [Article Influence: 24.9] [Reference Citation Analysis (2)] |
| 167. | Wang CC, Cheng PN, Kao JH. Systematic review: chronic viral hepatitis and metabolic derangement. Aliment Pharmacol Ther. 2020;51:216-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 91] [Article Influence: 15.2] [Reference Citation Analysis (1)] |
| 168. | Zabolotna IB, Gushcha SG, Vladimir LM. Non-alcoholic fatty liver disease and mineral waters of Ukraine – opportunities of application (experimental-clinical studies). Balneo. 2018;9:270-276. [DOI] [Full Text] |
| 169. | Izha G, Dragomiretska N, Gushcha S, Sierpińska LE, Plakida A. Features of clinical, biochemical, and sonographic parameters in patients with chronic viral hepatitis C with concomitant non-alcoholic fatty liver disease. Ann Agric Environ Med. 2023;30:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (1)] |
| 170. | Miyauchi S, Miyake T, Miyazaki M, Eguchi T, Niiya T, Yamamoto S, Senba H, Furukawa S, Matsuura B, Hiasa Y. Insulin-like growth factor-1 is inversely associated with liver fibrotic markers in patients with type 2 diabetes mellitus. J Diabetes Investig. 2019;10:1083-1091. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 171. | Nakahara T, Hyogo H, Yoneda M, Sumida Y, Eguchi Y, Fujii H, Ono M, Kawaguchi T, Imajo K, Aikata H, Tanaka S, Kanemasa K, Fujimoto K, Anzai K, Saibara T, Sata M, Nakajima A, Itoh Y, Chayama K, Okanoue T; Japan Study Group of Nonalcoholic Fatty Liver Disease. Type 2 diabetes mellitus is associated with the fibrosis severity in patients with nonalcoholic fatty liver disease in a large retrospective cohort of Japanese patients. J Gastroenterol. 2014;49:1477-1484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 122] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 172. | Paradis V, Perlemuter G, Bonvoust F, Dargere D, Parfait B, Vidaud M, Conti M, Huet S, Ba N, Buffet C, Bedossa P. High glucose and hyperinsulinemia stimulate connective tissue growth factor expression: a potential mechanism involved in progression to fibrosis in nonalcoholic steatohepatitis. Hepatology. 2001;34:738-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 404] [Cited by in RCA: 392] [Article Influence: 15.7] [Reference Citation Analysis (1)] |
| 173. | Terrault NA, Lok ASF, McMahon BJ, Chang KM, Hwang JP, Jonas MM, Brown RS Jr, Bzowej NH, Wong JB. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology. 2018;67:1560-1599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3558] [Cited by in RCA: 3190] [Article Influence: 398.8] [Reference Citation Analysis (6)] |
| 174. | Schweitzer A, Horn J, Mikolajczyk RT, Krause G, Ott JJ. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. Lancet. 2015;386:1546-1555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2262] [Cited by in RCA: 2057] [Article Influence: 187.0] [Reference Citation Analysis (4)] |
| 175. | Choi HSJ, Brouwer WP, Zanjir WMR, de Man RA, Feld JJ, Hansen BE, Janssen HLA, Patel K. Nonalcoholic Steatohepatitis Is Associated With Liver-Related Outcomes and All-Cause Mortality in Chronic Hepatitis B. Hepatology. 2020;71:539-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 181] [Article Influence: 30.2] [Reference Citation Analysis (1)] |
| 176. | Chan AW, Wong GL, Chan HY, Tong JH, Yu YH, Choi PC, Chan HL, To KF, Wong VW. Concurrent fatty liver increases risk of hepatocellular carcinoma among patients with chronic hepatitis B. J Gastroenterol Hepatol. 2017;32:667-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 172] [Article Influence: 19.1] [Reference Citation Analysis (1)] |
| 177. | Wong VW, Wong GL, Chu WC, Chim AM, Ong A, Yeung DK, Yiu KK, Chu SH, Chan HY, Woo J, Chan FK, Chan HL. Hepatitis B virus infection and fatty liver in the general population. J Hepatol. 2012;56:533-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 199] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 178. | Joo EJ, Chang Y, Yeom JS, Ryu S. Hepatitis B virus infection and decreased risk of nonalcoholic fatty liver disease: A cohort study. Hepatology. 2017;65:828-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 129] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 179. | Thomopoulos KC, Arvaniti V, Tsamantas AC, Dimitropoulou D, Gogos CA, Siagris D, Theocharis GJ, Labropoulou-Karatza C. Prevalence of liver steatosis in patients with chronic hepatitis B: a study of associated factors and of relationship with fibrosis. Eur J Gastroenterol Hepatol. 2006;18:233-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 105] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 180. | Arain SQ, Talpur FN, Channa NA, Ali MS, Afridi HI. Serum lipid profile as a marker of liver impairment in hepatitis B Cirrhosis patients. Lipids Health Dis. 2017;16:51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 181. | Liu PT, Hwang AC, Chen JD. Combined effects of hepatitis B virus infection and elevated alanine aminotransferase levels on dyslipidemia. Metabolism. 2013;62:220-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 182. | Li H, Xu QY, Xie Y, Luo JJ, Cao HX, Pan Q. Effects of chronic HBV infection on lipid metabolism in non-alcoholic fatty liver disease: A lipidomic analysis. Ann Hepatol. 2021;24:100316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 183. | McMahon BJ. The natural history of chronic hepatitis B virus infection. Hepatology. 2009;49:S45-S55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 592] [Cited by in RCA: 554] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 184. | Huang CY, Lu CW, Liu YL, Chiang CH, Lee LT, Huang KC. Relationship between chronic hepatitis B and metabolic syndrome: A structural equation modeling approach. Obesity (Silver Spring). 2016;24:483-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 185. | Zhao X, Shah D, Sambamoorthi U. Association between chronic hepatitis B infection and metabolic syndrome. J Diabetes Metab Disord. 2018;17:223-232. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 186. | Simental-Mendía LE, Rodríguez-Morán M, Gómez-Díaz R, Wacher NH, Rodríguez-Hernández H, Guerrero-Romero F. Insulin resistance is associated with elevated transaminases and low aspartate aminotransferase/alanine aminotransferase ratio in young adults with normal weight. Eur J Gastroenterol Hepatol. 2017;29:435-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 187. | Ye J, Hu X, Wu T, Wu Y, Shao C, Li F, Lin Y, Feng S, Wang W, Zhong B. Insulin resistance exhibits varied metabolic abnormalities in nonalcoholic fatty liver disease, chronic hepatitis B and the combination of the two: a cross-sectional study. Diabetol Metab Syndr. 2019;11:45. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 188. | Mak LY, Hui RW, Fung J, Liu F, Wong DK, Cheung KS, Yuen MF, Seto WK. Diverse effects of hepatic steatosis on fibrosis progression and functional cure in virologically quiescent chronic hepatitis B. J Hepatol. 2020;73:800-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 124] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 189. | Wong GL, Wong VW, Choi PC, Chan AW, Chim AM, Yiu KK, Chan HY, Chan FK, Sung JJ, Chan HL. Metabolic syndrome increases the risk of liver cirrhosis in chronic hepatitis B. Gut. 2009;58:111-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 190] [Article Influence: 11.2] [Reference Citation Analysis (4)] |
| 190. | Yu MW, Shih WL, Lin CL, Liu CJ, Jian JW, Tsai KS, Chen CJ. Body-mass index and progression of hepatitis B: a population-based cohort study in men. J Clin Oncol. 2008;26:5576-5582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 102] [Article Influence: 5.7] [Reference Citation Analysis (4)] |
| 191. | Cheng YL, Wang YJ, Kao WY, Chen PH, Huo TI, Huang YH, Lan KH, Su CW, Chan WL, Lin HC, Lee FY, Wu JC. Inverse association between hepatitis B virus infection and fatty liver disease: a large-scale study in populations seeking for check-up. PLoS One. 2013;8:e72049. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 192. | Lee HA, Lee HW, Kim IH, Park SY, Sinn DH, Yu JH, Seo YS, Um SH, Lee JI, Lee KS, Lee CH, Tak WY, Kweon YO, Kang W, Paik YH, Lee JW, Suh SJ, Jung YK, Kim BK, Park JY, Kim DY, Ahn SH, Han KH, Yim HJ, Kim SU. Extremely low risk of hepatocellular carcinoma development in patients with chronic hepatitis B in immune-tolerant phase. Aliment Pharmacol Ther. 2020;52:196-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 193. | Hu D, Wang H, Wang H, Wang Y, Wan X, Yan W, Luo X, Ning Q. Non-alcoholic hepatic steatosis attenuates hepatitis B virus replication in an HBV-immunocompetent mouse model. Hepatol Int. 2018;12:438-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 194. | Zhang Z, Pan Q, Duan XY, Liu Q, Mo GY, Rao GR, Fan JG. Fatty liver reduces hepatitis B virus replication in a genotype B hepatitis B virus transgenic mice model. J Gastroenterol Hepatol. 2012;27:1858-1864. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (2)] |
| 195. | Pan Q, Zhang RN, Wang YQ, Zheng RD, Mi YQ, Liu WB, Shen F, Chen GY, Lu JF, Zhu CY, Zhang SY, Chen YM, Sun WL, Fan JG. Linked PNPLA3 polymorphisms confer susceptibility to nonalcoholic steatohepatitis and decreased viral load in chronic hepatitis B. World J Gastroenterol. 2015;21:8605-8614. [PubMed] [DOI] [Full Text] |
| 196. | Zhang J, Lin S, Jiang D, Li M, Chen Y, Li J, Fan J. Chronic hepatitis B and non-alcoholic fatty liver disease: Conspirators or competitors? Liver Int. 2020;40:496-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 197. | Lévy P, Kohler M, McNicholas WT, Barbé F, McEvoy RD, Somers VK, Lavie L, Pépin JL. Obstructive sleep apnoea syndrome. Nat Rev Dis Primers. 2015;1:15015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 886] [Cited by in RCA: 774] [Article Influence: 70.4] [Reference Citation Analysis (1)] |
| 198. | Drager LF, Togeiro SM, Polotsky VY, Lorenzi-Filho G. Obstructive sleep apnea: a cardiometabolic risk in obesity and the metabolic syndrome. J Am Coll Cardiol. 2013;62:569-576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 728] [Cited by in RCA: 628] [Article Influence: 48.3] [Reference Citation Analysis (1)] |
| 199. | Malhotra RK, Kirsch DB, Kristo DA, Olson EJ, Aurora RN, Carden KA, Chervin RD, Martin JL, Ramar K, Rosen CL, Rowley JA, Rosen IM; American Academy of Sleep Medicine Board of Directors. Polysomnography for Obstructive Sleep Apnea Should Include Arousal-Based Scoring: An American Academy of Sleep Medicine Position Statement. J Clin Sleep Med. 2018;14:1245-1247. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 142] [Cited by in RCA: 118] [Article Influence: 14.8] [Reference Citation Analysis (1)] |
| 200. | Gottlieb DJ, Punjabi NM. Diagnosis and Management of Obstructive Sleep Apnea: A Review. JAMA. 2020;323:1389-1400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1225] [Cited by in RCA: 979] [Article Influence: 163.2] [Reference Citation Analysis (2)] |
| 201. | Tang H, Lv F, Zhang P, Liu J, Mao J. The impact of obstructive sleep apnea on nonalcoholic fatty liver disease. Front Endocrinol (Lausanne). 2023;14:1254459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 202. | Benotti P, Wood GC, Argyropoulos G, Pack A, Keenan BT, Gao X, Gerhard G, Still C. The impact of obstructive sleep apnea on nonalcoholic fatty liver disease in patients with severe obesity. Obesity (Silver Spring). 2016;24:871-877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 203. | Shah AG, Lydecker A, Murray K, Tetri BN, Contos MJ, Sanyal AJ; Nash Clinical Research Network. Comparison of noninvasive markers of fibrosis in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2009;7:1104-1112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1340] [Cited by in RCA: 1261] [Article Influence: 74.2] [Reference Citation Analysis (4)] |
| 204. | Agrawal S, Duseja A, Aggarwal A, Das A, Mehta M, Dhiman RK, Chawla Y. Obstructive sleep apnea is an important predictor of hepatic fibrosis in patients with nonalcoholic fatty liver disease in a tertiary care center. Hepatol Int. 2015;9:283-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 205. | Trzepizur W, Boursier J, Le Vaillant M, Ducluzeau PH, Dubois S, Henni S, Abraham P, Aubé C, Calès P, Gagnadoux F; on the behalf of the METABOL group. Increased liver stiffness in patients with severe sleep apnoea and metabolic comorbidities. Eur Respir J. 2018;51:1800601. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 5.4] [Reference Citation Analysis (2)] |
| 206. | Jin S, Jiang S, Hu A. Association between obstructive sleep apnea and non-alcoholic fatty liver disease: a systematic review and meta-analysis. Sleep Breath. 2018;22:841-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (1)] |
| 207. | Musso G, Cassader M, Olivetti C, Rosina F, Carbone G, Gambino R. Association of obstructive sleep apnoea with the presence and severity of non-alcoholic fatty liver disease. A systematic review and meta-analysis. Obes Rev. 2013;14:417-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 199] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 208. | Chung GE, Cho EJ, Yoo JJ, Chang Y, Cho Y, Park SH, Shin DW, Han K, Yu SJ. Nonalcoholic fatty liver disease is associated with the development of obstructive sleep apnea. Sci Rep. 2021;11:13473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 209. | Stanek A, Brożyna-Tkaczyk K, Myśliński W. Oxidative Stress Markers among Obstructive Sleep Apnea Patients. Oxid Med Cell Longev. 2021;2021:9681595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 77] [Article Influence: 15.4] [Reference Citation Analysis (1)] |
| 210. | Gaucher J, Vial G, Montellier E, Guellerin M, Bouyon S, Lemarie E, Pelloux V, Bertrand A, Pernet-Gallay K, Lamarche F, Borel AL, Arnaud C, Belaidi E, Clément K, Godin Ribuot D, Aron-Wisnewsky J, Pépin JL. Intermittent Hypoxia Rewires the Liver Transcriptome and Fires up Fatty Acids Usage for Mitochondrial Respiration. Front Med (Lausanne). 2022;9:829979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 211. | Holzner LMW, Murray AJ. Hypoxia-Inducible Factors as Key Players in the Pathogenesis of Non-alcoholic Fatty Liver Disease and Non-alcoholic Steatohepatitis. Front Med (Lausanne). 2021;8:753268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 212. | Wang X, de Carvalho Ribeiro M, Iracheta-Vellve A, Lowe P, Ambade A, Satishchandran A, Bukong T, Catalano D, Kodys K, Szabo G. Macrophage-Specific Hypoxia-Inducible Factor-1α Contributes to Impaired Autophagic Flux in Nonalcoholic Steatohepatitis. Hepatology. 2019;69:545-563. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 127] [Article Influence: 18.1] [Reference Citation Analysis (1)] |
| 213. | Gabryelska A, Szmyd B, Szemraj J, Stawski R, Sochal M, Białasiewicz P. Patients with obstructive sleep apnea present with chronic upregulation of serum HIF-1α protein. J Clin Sleep Med. 2020;16:1761-1768. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 80] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 214. | da Rosa DP, Forgiarini LF, Baronio D, Feijó CA, Martinez D, Marroni NP. Simulating sleep apnea by exposure to intermittent hypoxia induces inflammation in the lung and liver. Mediators Inflamm. 2012;2012:879419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 52] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 215. | Wei Z, Chen Y, Upender RP. Sleep Disturbance and Metabolic Dysfunction: The Roles of Adipokines. Int J Mol Sci. 2022;23:1706. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 216. | Barros D, García-Río F. Obstructive sleep apnea and dyslipidemia: from animal models to clinical evidence. Sleep. 2019;42:zsy236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 99] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 217. | Rey E, Del Pozo-Maroto E, Marañón P, Beeler B, García-García Y, Landete P, Isaza SC, Farré R, García-Monzón C, Almendros I, González-Rodríguez Á. Intrahepatic Expression of Fatty Acid Translocase CD36 Is Increased in Obstructive Sleep Apnea. Front Med (Lausanne). 2020;7:450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 218. | Polyzos SA, Kountouras J, Mantzoros CS. Leptin in nonalcoholic fatty liver disease: a narrative review. Metabolism. 2015;64:60-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 196] [Cited by in RCA: 178] [Article Influence: 16.2] [Reference Citation Analysis (1)] |
| 219. | Ding H, Huang JF, Xie HS, Wang BY, Lin T, Zhao JM, Lin QC. The association between glycometabolism and nonalcoholic fatty liver disease in patients with obstructive sleep apnea. Sleep Breath. 2019;23:373-378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 220. | Zhang L, Zhang X, Meng H, Li Y, Han T, Wang C. Obstructive sleep apnea and liver injury in severely obese patients with nonalcoholic fatty liver disease. Sleep Breath. 2020;24:1515-1521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 221. | Moreno-Indias I, Torres M, Montserrat JM, Sanchez-Alcoholado L, Cardona F, Tinahones FJ, Gozal D, Poroyko VA, Navajas D, Queipo-Ortuño MI, Farré R. Intermittent hypoxia alters gut microbiota diversity in a mouse model of sleep apnoea. Eur Respir J. 2015;45:1055-1065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 224] [Article Influence: 20.4] [Reference Citation Analysis (2)] |
| 222. | Zhang Y, Xie B, Chen X, Zhang J, Yuan S. A key role of gut microbiota-vagus nerve/spleen axis in sleep deprivation-mediated aggravation of systemic inflammation after LPS administration. Life Sci. 2021;265:118736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 56] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 223. | Wu J, Lu Y, Cai X, Chen Y, Shen Z, Lyv Q. Gut microbiota dysbiosis in 4- to 6-year-old children with obstructive sleep apnea-hypopnea syndrome. Pediatr Pulmonol. 2022;57:2012-2022. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 224. | Valentini F, Evangelisti M, Arpinelli M, Di Nardo G, Borro M, Simmaco M, Villa MP. Gut microbiota composition in children with obstructive sleep apnoea syndrome: a pilot study. Sleep Med. 2020;76:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 83] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 225. | Ko CY, Liu QQ, Su HZ, Zhang HP, Fan JM, Yang JH, Hu AK, Liu YQ, Chou D, Zeng YM. Gut microbiota in obstructive sleep apnea-hypopnea syndrome: disease-related dysbiosis and metabolic comorbidities. Clin Sci (Lond). 2019;133:905-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 129] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 226. | Benedict C, Vogel H, Jonas W, Woting A, Blaut M, Schürmann A, Cedernaes J. Gut microbiota and glucometabolic alterations in response to recurrent partial sleep deprivation in normal-weight young individuals. Mol Metab. 2016;5:1175-1186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 283] [Cited by in RCA: 245] [Article Influence: 24.5] [Reference Citation Analysis (5)] |
| 227. | Nobili V, Alisi A, Cutrera R, Carpino G, De Stefanis C, D'Oria V, De Vito R, Cucchiara S, Gaudio E, Musso G. Altered gut-liver axis and hepatic adiponectin expression in OSAS: novel mediators of liver injury in paediatric non-alcoholic fatty liver. Thorax. 2015;70:769-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 228. | Tang SS, Liang CH, Liu YL, Wei W, Deng XR, Shi XY, Wang LM, Zhang LJ, Yuan HJ. Intermittent hypoxia is involved in gut microbial dysbiosis in type 2 diabetes mellitus and obstructive sleep apnea-hypopnea syndrome. World J Gastroenterol. 2022;28:2320-2333. [PubMed] [DOI] [Full Text] |
| 229. | Ko CY, Fan JM, Hu AK, Su HZ, Yang JH, Huang LM, Yan FR, Zhang HP, Zeng YM. Disruption of sleep architecture in Prevotella enterotype of patients with obstructive sleep apnea-hypopnea syndrome. Brain Behav. 2019;9:e01287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 43] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 230. | Hernández A, Geng Y, Sepúlveda R, Solís N, Torres J, Arab JP, Barrera F, Cabrera D, Moshage H, Arrese M. Chemical hypoxia induces pro-inflammatory signals in fat-laden hepatocytes and contributes to cellular crosstalk with Kupffer cells through extracellular vesicles. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 8.5] [Reference Citation Analysis (4)] |
| 231. | Liu Q, Liu Y, Li F, Gu Z, Liu M, Shao T, Zhang L, Zhou G, Pan C, He L, Cai J, Zhang X, Barve S, McClain CJ, Chen Y, Feng W. Probiotic culture supernatant improves metabolic function through FGF21-adiponectin pathway in mice. J Nutr Biochem. 2020;75:108256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 232. | Zhang H, Zhou L, Zhou Y, Wang L, Jiang W, Liu L, Yue S, Zheng P, Liu H. Intermittent hypoxia aggravates non-alcoholic fatty liver disease via RIPK3-dependent necroptosis-modulated Nrf2/NFκB signaling pathway. Life Sci. 2021;285:119963. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 233. | Xin L, Fan W, Tingting D, Zuoming S, Qiang Z. 4-phenylbutyric acid attenuates endoplasmic reticulum stress-mediated apoptosis and protects the hepatocytes from intermittent hypoxia-induced injury. Sleep Breath. 2019;23:711-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 234. | Christgen S, Tweedell RE, Kanneganti TD. Programming inflammatory cell death for therapy. Pharmacol Ther. 2022;232:108010. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 253] [Cited by in RCA: 217] [Article Influence: 54.3] [Reference Citation Analysis (0)] |
| 235. | Lonardo A, Nascimbeni F, Mantovani A, Targher G. Hypertension, diabetes, atherosclerosis and NASH: Cause or consequence? J Hepatol. 2018;68:335-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 582] [Article Influence: 72.8] [Reference Citation Analysis (1)] |
| 236. | Ryoo JH, Suh YJ, Shin HC, Cho YK, Choi JM, Park SK. Clinical association between non-alcoholic fatty liver disease and the development of hypertension. J Gastroenterol Hepatol. 2014;29:1926-1931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 101] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 237. | Zhang Y, Zhang T, Zhang C, Tang F, Zhong N, Li H, Song X, Lin H, Liu Y, Xue F. Identification of reciprocal causality between non-alcoholic fatty liver disease and metabolic syndrome by a simplified Bayesian network in a Chinese population. BMJ Open. 2015;5:e008204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 238. | Singh S, Allen AM, Wang Z, Prokop LJ, Murad MH, Loomba R. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol. 2015;13:643-54.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1438] [Cited by in RCA: 1321] [Article Influence: 120.1] [Reference Citation Analysis (6)] |
| 239. | Aneni EC, Oni ET, Martin SS, Blaha MJ, Agatston AS, Feldman T, Veledar E, Conçeicao RD, Carvalho JA, Santos RD, Nasir K. Blood pressure is associated with the presence and severity of nonalcoholic fatty liver disease across the spectrum of cardiometabolic risk. J Hypertens. 2015;33:1207-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 108] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 240. | Yang S, Kwak S, Lee JH, Kang S, Lee SP. Nonalcoholic fatty liver disease is an early predictor of metabolic diseases in a metabolically healthy population. PLoS One. 2019;14:e0224626. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 241. | Ryoo JH, Ham WT, Choi JM, Kang MA, An SH, Lee JK, Shin HC, Park SK. Clinical significance of non-alcoholic fatty liver disease as a risk factor for prehypertension. J Korean Med Sci. 2014;29:973-979. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 242. | Lau K, Lorbeer R, Haring R, Schmidt CO, Wallaschofski H, Nauck M, John U, Baumeister SE, Völzke H. The association between fatty liver disease and blood pressure in a population-based prospective longitudinal study. J Hypertens. 2010;28:1829-1835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 3.7] [Reference Citation Analysis (2)] |
| 243. | Shim KY, Eom YW, Kim MY, Kang SH, Baik SK. Role of the renin-angiotensin system in hepatic fibrosis and portal hypertension. Korean J Intern Med. 2018;33:453-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 75] [Cited by in RCA: 66] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 244. | Kuzminova NV, Gribenyuk OV, Osovska NY, Knyazkova II. Arterial hypertension, obesity and non-alcoholic fatty liver disease: is there any connection? Arterial Hypertens. 2016;20:216-227. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 245. | Matthew Morris E, Fletcher JA, Thyfault JP, Rector RS. The role of angiotensin II in nonalcoholic steatohepatitis. Mol Cell Endocrinol. 2013;378:29-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 246. | Munshi MK, Uddin MN, Glaser SS. The role of the renin-angiotensin system in liver fibrosis. Exp Biol Med (Maywood). 2011;236:557-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 55] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 247. | Kato J, Koda M, Kishina M, Tokunaga S, Matono T, Sugihara T, Ueki M, Murawaki Y. Therapeutic effects of angiotensin II type 1 receptor blocker, irbesartan, on non-alcoholic steatohepatitis using FLS-ob/ob male mice. Int J Mol Med. 2012;30:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 22] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 248. | Aizawa-Abe M, Ogawa Y, Masuzaki H, Ebihara K, Satoh N, Iwai H, Matsuoka N, Hayashi T, Hosoda K, Inoue G, Yoshimasa Y, Nakao K. Pathophysiological role of leptin in obesity-related hypertension. J Clin Invest. 2000;105:1243-1252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 376] [Cited by in RCA: 337] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 249. | Artunc F, Schleicher E, Weigert C, Fritsche A, Stefan N, Häring HU. The impact of insulin resistance on the kidney and vasculature. Nat Rev Nephrol. 2016;12:721-737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 386] [Cited by in RCA: 360] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 250. | DiBona GF, Esler M. Translational medicine: the antihypertensive effect of renal denervation. Am J Physiol Regul Integr Comp Physiol. 2010;298:R245-R253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 350] [Cited by in RCA: 304] [Article Influence: 19.0] [Reference Citation Analysis (1)] |
| 251. | Sabath E, Báez-Ruiz A, Buijs RM. Non-alcoholic fatty liver disease as a consequence of autonomic imbalance and circadian desynchronization. Obes Rev. 2015;16:871-882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 33] [Article Influence: 3.0] [Reference Citation Analysis (4)] |
| 252. | Sun W, Zhang D, Sun J, Xu B, Sun K, Wang T, Ren C, Li J, Chen Y, Xu M, Bi Y, Xu Q, Wang W, Gu Y, Ning G. Association between non-alcoholic fatty liver disease and autonomic dysfunction in a Chinese population. QJM. 2015;108:617-624. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 253. | Sigala B, McKee C, Soeda J, Pazienza V, Morgan M, Lin CI, Selden C, Vander Borght S, Mazzoccoli G, Roskams T, Vinciguerra M, Oben JA. Sympathetic nervous system catecholamines and neuropeptide Y neurotransmitters are upregulated in human NAFLD and modulate the fibrogenic function of hepatic stellate cells. PLoS One. 2013;8:e72928. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 254. | Mantovani A, Csermely A, Petracca G, Beatrice G, Corey KE, Simon TG, Byrne CD, Targher G. Non-alcoholic fatty liver disease and risk of fatal and non-fatal cardiovascular events: an updated systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2021;6:903-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 521] [Cited by in RCA: 461] [Article Influence: 92.2] [Reference Citation Analysis (2)] |
| 255. | Fracanzani AL, Tiraboschi S, Pisano G, Consonni D, Baragetti A, Bertelli C, Norata D, Valenti L, Grigore L, Porzio M, Catapano A, Fargion S. Progression of carotid vascular damage and cardiovascular events in non-alcoholic fatty liver disease patients compared to the general population during 10 years of follow-up. Atherosclerosis. 2016;246:208-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 75] [Article Influence: 7.5] [Reference Citation Analysis (1)] |
| 256. | Zeb I, Li D, Budoff MJ, Katz R, Lloyd-Jones D, Agatston A, Blumenthal RS, Blaha MJ, Blankstein R, Carr J, Nasir K. Nonalcoholic Fatty Liver Disease and Incident Cardiac Events: The Multi-Ethnic Study of Atherosclerosis. J Am Coll Cardiol. 2016;67:1965-1966. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 257. | Kouvari M, Chrysohoou C, Skoumas J, Pitsavos C, Panagiotakos DB, Mantzoros CS; ATTICA study Investigators. The presence of NAFLD influences the transition of metabolically healthy to metabolically unhealthy obesity and the ten-year cardiovascular disease risk: A population-based cohort study. Metabolism. 2022;128:154893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 258. | Park JG, Jung J, Verma KK, Kang MK, Madamba E, Lopez S, Qas Yonan A, Liu A, Bettencourt R, Sirlin C, Loomba R. Liver stiffness by magnetic resonance elastography is associated with increased risk of cardiovascular disease in patients with non-alcoholic fatty liver disease. Aliment Pharmacol Ther. 2021;53:1030-1037. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 259. | Ma W, Wu W, Wen W, Xu F, Han D, Lyu J, Huang Y. Association of NAFLD with cardiovascular disease and all-cause mortality: a large-scale prospective cohort study based on UK Biobank. Ther Adv Chronic Dis. 2022;13:20406223221122478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 260. | Pais R, Giral P, Khan JF, Rosenbaum D, Housset C, Poynard T, Ratziu V; LIDO Study Group. Fatty liver is an independent predictor of early carotid atherosclerosis. J Hepatol. 2016;65:95-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 88] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 261. | Mantovani A, Dauriz M, Sandri D, Bonapace S, Zoppini G, Tilg H, Byrne CD, Targher G. Association between non-alcoholic fatty liver disease and risk of atrial fibrillation in adult individuals: An updated meta-analysis. Liver Int. 2019;39:758-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 102] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 262. | Targher G, Byrne CD, Lonardo A, Zoppini G, Barbui C. Non-alcoholic fatty liver disease and risk of incident cardiovascular disease: A meta-analysis. J Hepatol. 2016;65:589-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1182] [Cited by in RCA: 1077] [Article Influence: 107.7] [Reference Citation Analysis (1)] |
| 263. | Byrne CD, Targher G. Non-alcoholic fatty liver disease-related risk of cardiovascular disease and other cardiac complications. Diabetes Obes Metab. 2022;24 Suppl 2:28-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 74] [Article Influence: 18.5] [Reference Citation Analysis (1)] |
| 264. | Meyersohn NM, Mayrhofer T, Corey KE, Bittner DO, Staziaki PV, Szilveszter B, Hallett T, Lu MT, Puchner SB, Simon TG, Foldyna B, Voora D, Ginsburg GS, Douglas PS, Hoffmann U, Ferencik M. Association of Hepatic Steatosis With Major Adverse Cardiovascular Events, Independent of Coronary Artery Disease. Clin Gastroenterol Hepatol. 2021;19:1480-1488.e14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 265. | Mantovani A, Scorletti E, Mosca A, Alisi A, Byrne CD, Targher G. Complications, morbidity and mortality of nonalcoholic fatty liver disease. Metabolism. 2020;111S:154170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 424] [Cited by in RCA: 393] [Article Influence: 65.5] [Reference Citation Analysis (1)] |
| 266. | Paik JM, Henry L, De Avila L, Younossi E, Racila A, Younossi ZM. Mortality Related to Nonalcoholic Fatty Liver Disease Is Increasing in the United States. Hepatol Commun. 2019;3:1459-1471. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 201] [Cited by in RCA: 185] [Article Influence: 26.4] [Reference Citation Analysis (1)] |
| 267. | Bonci E, Chiesa C, Versacci P, Anania C, Silvestri L, Pacifico L. Association of Nonalcoholic Fatty Liver Disease with Subclinical Cardiovascular Changes: A Systematic Review and Meta-Analysis. Biomed Res Int. 2015;2015:213737. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 268. | Wu S, Wu F, Ding Y, Hou J, Bi J, Zhang Z. Association of non-alcoholic fatty liver disease with major adverse cardiovascular events: A systematic review and meta-analysis. Sci Rep. 2016;6:33386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 251] [Cited by in RCA: 239] [Article Influence: 23.9] [Reference Citation Analysis (1)] |
| 269. | Oni ET, Agatston AS, Blaha MJ, Fialkow J, Cury R, Sposito A, Erbel R, Blankstein R, Feldman T, Al-Mallah MH, Santos RD, Budoff MJ, Nasir K. A systematic review: burden and severity of subclinical cardiovascular disease among those with nonalcoholic fatty liver; should we care? Atherosclerosis. 2013;230:258-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 309] [Cited by in RCA: 290] [Article Influence: 22.3] [Reference Citation Analysis (5)] |
| 270. | Fracanzani AL, Burdick L, Raselli S, Pedotti P, Grigore L, Santorelli G, Valenti L, Maraschi A, Catapano A, Fargion S. Carotid artery intima-media thickness in nonalcoholic fatty liver disease. Am J Med. 2008;121:72-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 166] [Article Influence: 9.2] [Reference Citation Analysis (1)] |
| 271. | Bonapace S, Perseghin G, Molon G, Canali G, Bertolini L, Zoppini G, Barbieri E, Targher G. Nonalcoholic fatty liver disease is associated with left ventricular diastolic dysfunction in patients with type 2 diabetes. Diabetes Care. 2012;35:389-395. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 143] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 272. | Moon SH, Noh TS, Cho YS, Hong SP, Hyun SH, Choi JY, Kim BT, Lee KH. Association between nonalcoholic fatty liver disease and carotid artery inflammation evaluated by 18F-fluorodeoxyglucose positron emission tomography. Angiology. 2015;66:472-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 273. | Ismaiel A, Dumitraşcu DL. Cardiovascular Risk in Fatty Liver Disease: The Liver-Heart Axis-Literature Review. Front Med (Lausanne). 2019;6:202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 139] [Article Influence: 19.9] [Reference Citation Analysis (1)] |
| 274. | D'Ardes D, Boccatonda A, Cocco G, Fabiani S, Rossi I, Bucci M, Guagnano MT, Schiavone C, Cipollone F. Impaired coagulation, liver dysfunction and COVID-19: Discovering an intriguing relationship. World J Gastroenterol. 2022;28:1102-1112. [PubMed] [DOI] [Full Text] |
| 275. | Ference BA, Ginsberg HN, Graham I, Ray KK, Packard CJ, Bruckert E, Hegele RA, Krauss RM, Raal FJ, Schunkert H, Watts GF, Borén J, Fazio S, Horton JD, Masana L, Nicholls SJ, Nordestgaard BG, van de Sluis B, Taskinen MR, Tokgözoglu L, Landmesser U, Laufs U, Wiklund O, Stock JK, Chapman MJ, Catapano AL. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38:2459-2472. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3226] [Cited by in RCA: 2768] [Article Influence: 307.6] [Reference Citation Analysis (6)] |
| 276. | Heeren J, Scheja L. Metabolic-associated fatty liver disease and lipoprotein metabolism. Mol Metab. 2021;50:101238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 537] [Cited by in RCA: 474] [Article Influence: 94.8] [Reference Citation Analysis (1)] |
| 277. | Bence KK, Birnbaum MJ. Metabolic drivers of non-alcoholic fatty liver disease. Mol Metab. 2021;50:101143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 172] [Article Influence: 34.4] [Reference Citation Analysis (1)] |
| 278. | Alves-Bezerra M, Cohen DE. Triglyceride Metabolism in the Liver. Compr Physiol. 2017;8:1-8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 321] [Cited by in RCA: 466] [Article Influence: 51.8] [Reference Citation Analysis (1)] |
| 279. | Deng KQ, Huang X, Lei F, Zhang XJ, Zhang P, She ZG, Cai J, Ji YX, Li H. Role of hepatic lipid species in the progression of nonalcoholic fatty liver disease. Am J Physiol Cell Physiol. 2022;323:C630-C639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 280. | Katsiki N, Mikhailidis DP, Mantzoros CS. Non-alcoholic fatty liver disease and dyslipidemia: An update. Metabolism. 2016;65:1109-1123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 484] [Cited by in RCA: 439] [Article Influence: 43.9] [Reference Citation Analysis (4)] |
| 281. | Dongiovanni P, Rametta R, Meroni M, Valenti L. The role of insulin resistance in nonalcoholic steatohepatitis and liver disease development--a potential therapeutic target? Expert Rev Gastroenterol Hepatol. 2016;10:229-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 282. | Postic C, Girard J. Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: lessons from genetically engineered mice. J Clin Invest. 2008;118:829-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1014] [Cited by in RCA: 938] [Article Influence: 52.1] [Reference Citation Analysis (3)] |
| 283. | Smith GI, Shankaran M, Yoshino M, Schweitzer GG, Chondronikola M, Beals JW, Okunade AL, Patterson BW, Nyangau E, Field T, Sirlin CB, Talukdar S, Hellerstein MK, Klein S. Insulin resistance drives hepatic de novo lipogenesis in nonalcoholic fatty liver disease. J Clin Invest. 2020;130:1453-1460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 646] [Cited by in RCA: 612] [Article Influence: 102.0] [Reference Citation Analysis (4)] |
| 284. | Duran EK, Aday AW, Cook NR, Buring JE, Ridker PM, Pradhan AD. Triglyceride-Rich Lipoprotein Cholesterol, Small Dense LDL Cholesterol, and Incident Cardiovascular Disease. J Am Coll Cardiol. 2020;75:2122-2135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 287] [Cited by in RCA: 256] [Article Influence: 42.7] [Reference Citation Analysis (6)] |
| 285. | Ghosh A, Gao L, Thakur A, Siu PM, Lai CWK. Role of free fatty acids in endothelial dysfunction. J Biomed Sci. 2017;24:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 336] [Cited by in RCA: 313] [Article Influence: 34.8] [Reference Citation Analysis (5)] |
| 286. | Sasso FC, Pafundi PC, Marfella R, Calabrò P, Piscione F, Furbatto F, Esposito G, Galiero R, Gragnano F, Rinaldi L, Salvatore T, D'Amico M, Adinolfi LE, Sardu C. Adiponectin and insulin resistance are related to restenosis and overall new PCI in subjects with normal glucose tolerance: the prospective AIRE Study. Cardiovasc Diabetol. 2019;18:24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (1)] |
| 287. | Vilar R, Fish RJ, Casini A, Neerman-Arbez M. Fibrin(ogen) in human disease: both friend and foe. Haematologica. 2020;105:284-296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 175] [Article Influence: 29.2] [Reference Citation Analysis (5)] |
| 288. | Ogresta D, Mrzljak A, Cigrovski Berkovic M, Bilic-Curcic I, Stojsavljevic-Shapeski S, Virovic-Jukic L. Coagulation and Endothelial Dysfunction Associated with NAFLD: Current Status and Therapeutic Implications. J Clin Transl Hepatol. 2022;10:339-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 289. | Tripodi A, Lombardi R, Primignani M, La Mura V, Peyvandi F, Fracanzani AL. Hypercoagulability in Patients with Non-Alcoholic Fatty Liver Disease (NAFLD): Causes and Consequences. Biomedicines. 2022;10:249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 290. | Virović-Jukić L, Stojsavljević-Shapeski S, Forgač J, Kukla M, Mikolašević I. Non-alcoholic fatty liver disease - a procoagulant condition? Croat Med J. 2021;62:25-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 291. | Surma S, Banach M. Fibrinogen and Atherosclerotic Cardiovascular Diseases-Review of the Literature and Clinical Studies. Int J Mol Sci. 2021;23:193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 97] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 292. | Kryczka KE, Kruk M, Demkow M, Lubiszewska B. Fibrinogen and a Triad of Thrombosis, Inflammation, and the Renin-Angiotensin System in Premature Coronary Artery Disease in Women: A New Insight into Sex-Related Differences in the Pathogenesis of the Disease. Biomolecules. 2021;11:1036. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 293. | Ciavarella A, Gnocchi D, Custodero C, Lenato GM, Fiore G, Sabbà C, Mazzocca A. Translational insight into prothrombotic state and hypercoagulation in nonalcoholic fatty liver disease. Thromb Res. 2021;198:139-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 294. | Cao Y, Deng Y, Wang J, Zhao H, Zhang J, Xie W. The association between NAFLD and risk of chronic kidney disease: a cross-sectional study. Ther Adv Chronic Dis. 2021;12:20406223211048649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 33] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 295. | Wattacheril J. Extrahepatic Manifestations of Nonalcoholic Fatty Liver Disease. Gastroenterol Clin North Am. 2020;49:141-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 296. | Agustanti N, Soetedjo NNM, Damara FA, Iryaningrum MR, Permana H, Bestari MB, Supriyadi R. The association between metabolic dysfunction-associated fatty liver disease and chronic kidney disease: A systematic review and meta-analysis. Diabetes Metab Syndr. 2023;17:102780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 297. | Mantovani A, Zaza G, Byrne CD, Lonardo A, Zoppini G, Bonora E, Targher G. Nonalcoholic fatty liver disease increases risk of incident chronic kidney disease: A systematic review and meta-analysis. Metabolism. 2018;79:64-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 259] [Article Influence: 32.4] [Reference Citation Analysis (1)] |
| 298. | Mantovani A, Petracca G, Beatrice G, Csermely A, Lonardo A, Schattenberg JM, Tilg H, Byrne CD, Targher G. Non-alcoholic fatty liver disease and risk of incident chronic kidney disease: an updated meta-analysis. Gut. 2022;71:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 292] [Article Influence: 73.0] [Reference Citation Analysis (1)] |
| 299. | Ayada I, van Kleef LA, Alferink LJM, Li P, de Knegt RJ, Pan Q. Systematically comparing epidemiological and clinical features of MAFLD and NAFLD by meta-analysis: Focusing on the non-overlap groups. Liver Int. 2022;42:277-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (1)] |
| 300. | Tanaka M, Mori K, Takahashi S, Higashiura Y, Ohnishi H, Hanawa N, Furuhashi M. Metabolic dysfunction-associated fatty liver disease predicts new onset of chronic kidney disease better than fatty liver or nonalcoholic fatty liver disease. Nephrol Dial Transplant. 2023;38:700-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (1)] |
| 301. | Zhang Y, Zhang T, Liu Y, Bai S, Jiang J, Zhou H, Luan J, Cao L, Lv Y, Zhang Q, Liu L, Sun S, Wang X, Zhou M, Jia Q, Song K, Zhang H, Chang Q, Fan X, Ding Y, Chen L, Zhao Y, Niu K, Xia Y. Adherence to healthy lifestyle was associated with an attenuation of the risk of chronic kidney disease from metabolic dysfunction-associated fatty liver disease: Results from two prospective cohorts. Diabetes Metab Syndr. 2023;17:102873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 302. | Paik J, Golabi P, Younoszai Z, Mishra A, Trimble G, Younossi ZM. Chronic kidney disease is independently associated with increased mortality in patients with nonalcoholic fatty liver disease. Liver Int. 2019;39:342-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (1)] |
| 303. | Li G, Shi W, Hug H, Chen Y, Liu L, Yin D. Nonalcoholic fatty liver disease associated with impairment of kidney function in nondiabetes population. Biochem Med (Zagreb). 2012;22:92-99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 304. | Targher G, Chonchol MB, Byrne CD. CKD and nonalcoholic fatty liver disease. Am J Kidney Dis. 2014;64:638-652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 166] [Article Influence: 13.8] [Reference Citation Analysis (2)] |
| 305. | Targher G, Pichiri I, Zoppini G, Trombetta M, Bonora E. Increased prevalence of chronic kidney disease in patients with Type 1 diabetes and non-alcoholic fatty liver. Diabet Med. 2012;29:220-226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 60] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 306. | Sun K, Lin D, Li F, Qi Y, Feng W, Yan L, Chen C, Ren M, Liu D. Fatty liver index, albuminuria and the association with chronic kidney disease: a population-based study in China. BMJ Open. 2018;8:e019097. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 307. | Yasui K, Sumida Y, Mori Y, Mitsuyoshi H, Minami M, Itoh Y, Kanemasa K, Matsubara H, Okanoue T, Yoshikawa T. Nonalcoholic steatohepatitis and increased risk of chronic kidney disease. Metabolism. 2011;60:735-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 125] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 308. | Zhang M, Lin S, Wang MF, Huang JF, Liu SY, Wu SM, Zhang HY, Wu ZM, Liu WY, Zhang DC, Hao CM, Zhu YY, Zheng MH, Wang XZ. Association between NAFLD and risk of prevalent chronic kidney disease: why there is a difference between east and west? BMC Gastroenterol. 2020;20:139. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 26] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 309. | Arase Y, Suzuki F, Kobayashi M, Suzuki Y, Kawamura Y, Matsumoto N, Akuta N, Kobayashi M, Sezaki H, Saito S, Hosaka T, Ikeda K, Kumada H, Ohmoto Y, Amakawa K, Tsuji H, Hsieh SD, Kato K, Tanabe M, Ogawa K, Hara S, Kobayashi T. The development of chronic kidney disease in Japanese patients with non-alcoholic fatty liver disease. Intern Med. 2011;50:1081-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 310. | Evans M, Lewis RD, Morgan AR, Whyte MB, Hanif W, Bain SC, Davies S, Dashora U, Yousef Z, Patel DC, Strain WD. A Narrative Review of Chronic Kidney Disease in Clinical Practice: Current Challenges and Future Perspectives. Adv Ther. 2022;39:33-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 245] [Cited by in RCA: 184] [Article Influence: 46.0] [Reference Citation Analysis (1)] |
| 311. | Matsuda S, Kobayashi M, Kitagishi Y. Roles for PI3K/AKT/PTEN Pathway in Cell Signaling of Nonalcoholic Fatty Liver Disease. ISRN Endocrinol. 2013;2013:472432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 131] [Cited by in RCA: 136] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 312. | Wang TY, Wang RF, Bu ZY, Targher G, Byrne CD, Sun DQ, Zheng MH. Association of metabolic dysfunction-associated fatty liver disease with kidney disease. Nat Rev Nephrol. 2022;18:259-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 166] [Article Influence: 41.5] [Reference Citation Analysis (1)] |
| 313. | Cheung A, Ahmed A. Nonalcoholic Fatty Liver Disease and Chronic Kidney Disease: A Review of Links and Risks. Clin Exp Gastroenterol. 2021;14:457-465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 314. | Targher G, Byrne CD. Non-alcoholic fatty liver disease: an emerging driving force in chronic kidney disease. Nat Rev Nephrol. 2017;13:297-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 240] [Article Influence: 26.7] [Reference Citation Analysis (2)] |
| 315. | Masenga SK, Kabwe LS, Chakulya M, Kirabo A. Mechanisms of Oxidative Stress in Metabolic Syndrome. Int J Mol Sci. 2023;24:7898. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 553] [Cited by in RCA: 417] [Article Influence: 139.0] [Reference Citation Analysis (1)] |
| 316. | Ruan X, Guan Y. Metabolic syndrome and chronic kidney disease. J Diabetes. 2009;1:236-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 317. | Stenvinkel P, Chertow GM, Devarajan P, Levin A, Andreoli SP, Bangalore S, Warady BA. Chronic Inflammation in Chronic Kidney Disease Progression: Role of Nrf2. Kidney Int Rep. 2021;6:1775-1787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 260] [Cited by in RCA: 224] [Article Influence: 44.8] [Reference Citation Analysis (1)] |
| 318. | Dorotea D, Koya D, Ha H. Recent Insights Into SREBP as a Direct Mediator of Kidney Fibrosis via Lipid-Independent Pathways. Front Pharmacol. 2020;11:265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 71] [Article Influence: 11.8] [Reference Citation Analysis (1)] |
| 319. | Polyzos SA, Aronis KN, Kountouras J, Raptis DD, Vasiloglou MF, Mantzoros CS. Circulating leptin in non-alcoholic fatty liver disease: a systematic review and meta-analysis. Diabetologia. 2016;59:30-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 192] [Article Influence: 19.2] [Reference Citation Analysis (4)] |
| 320. | Hashemi L, Hsiung JT, Arif Y, Soohoo M, Jackson N, Gosmanova EO, Budoff M, Kovesdy CP, Kalantar-Zadeh K, Streja E. Serum Low-Density Lipoprotein Cholesterol and Cardiovascular Disease Risk Across Chronic Kidney Disease Stages (Data from 1.9 Million United States Veterans). Am J Cardiol. 2022;170:47-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 321. | Baragetti A, Norata GD, Sarcina C, Rastelli F, Grigore L, Garlaschelli K, Uboldi P, Baragetti I, Pozzi C, Catapano AL. High density lipoprotein cholesterol levels are an independent predictor of the progression of chronic kidney disease. J Intern Med. 2013;274:252-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 322. | Baragetti A, Ossoli A, Strazzella A, Simonelli S, Baragetti I, Grigore L, Pellegatta F, Catapano AL, Norata GD, Calabresi L. Low Plasma Lecithin: Cholesterol Acyltransferase (LCAT) Concentration Predicts Chronic Kidney Disease. J Clin Med. 2020;9:2289. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (5)] |
| 323. | Adebayo D, Neong SF, Wong F. Ascites and Hepatorenal Syndrome. Clin Liver Dis. 2019;23:659-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 324. | Ng CK, Chan MH, Tai MH, Lam CW. Hepatorenal syndrome. Clin Biochem Rev. 2007;28:11-17. [PubMed] |
| 325. | Francoz C, Durand F, Kahn JA, Genyk YS, Nadim MK. Hepatorenal Syndrome. Clin J Am Soc Nephrol. 2019;14:774-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 91] [Article Influence: 13.0] [Reference Citation Analysis (1)] |
| 326. | Iwakiri Y, Shah V, Rockey DC. Vascular pathobiology in chronic liver disease and cirrhosis - current status and future directions. J Hepatol. 2014;61:912-924. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 266] [Cited by in RCA: 245] [Article Influence: 20.4] [Reference Citation Analysis (2)] |
| 327. | Mantovani A, Petracca G, Beatrice G, Csermely A, Tilg H, Byrne CD, Targher G. Non-alcoholic fatty liver disease and increased risk of incident extrahepatic cancers: a meta-analysis of observational cohort studies. Gut. 2022;71:778-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 255] [Article Influence: 63.8] [Reference Citation Analysis (1)] |
| 328. | Wang Z, Zhao X, Chen S, Wang Y, Cao L, Liao W, Sun Y, Wang X, Zheng Y, Wu S, Wang L. Associations Between Nonalcoholic Fatty Liver Disease and Cancers in a Large Cohort in China. Clin Gastroenterol Hepatol. 2021;19:788-796.e4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 329. | Kim GA, Lee HC, Choe J, Kim MJ, Lee MJ, Chang HS, Bae IY, Kim HK, An J, Shim JH, Kim KM, Lim YS. Association between non-alcoholic fatty liver disease and cancer incidence rate. J Hepatol. 2017;S0168-8278(17)32294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 211] [Article Influence: 23.4] [Reference Citation Analysis (1)] |
| 330. | Allen AM, Hicks SB, Mara KC, Larson JJ, Therneau TM. The risk of incident extrahepatic cancers is higher in non-alcoholic fatty liver disease than obesity - A longitudinal cohort study. J Hepatol. 2019;71:1229-1236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 280] [Cited by in RCA: 260] [Article Influence: 37.1] [Reference Citation Analysis (1)] |
| 331. | Mantovani A, Dauriz M, Byrne CD, Lonardo A, Zoppini G, Bonora E, Targher G. Association between nonalcoholic fatty liver disease and colorectal tumours in asymptomatic adults undergoing screening colonoscopy: a systematic review and meta-analysis. Metabolism. 2018;87:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 95] [Article Influence: 11.9] [Reference Citation Analysis (1)] |
| 332. | Kanwal F, Kramer JR, Mapakshi S, Natarajan Y, Chayanupatkul M, Richardson PA, Li L, Desiderio R, Thrift AP, Asch SM, Chu J, El-Serag HB. Risk of Hepatocellular Cancer in Patients With Non-Alcoholic Fatty Liver Disease. Gastroenterology. 2018;155:1828-1837.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 668] [Cited by in RCA: 626] [Article Influence: 78.3] [Reference Citation Analysis (6)] |
| 333. | Kwak MS, Yim JY, Yi A, Chung GE, Yang JI, Kim D, Kim JS, Noh DY. Nonalcoholic fatty liver disease is associated with breast cancer in nonobese women. Dig Liver Dis. 2019;51:1030-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 334. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76817] [Cited by in RCA: 69700] [Article Influence: 13940.0] [Reference Citation Analysis (45)] |
| 335. | Moon AM, Singal AG, Tapper EB. Contemporary Epidemiology of Chronic Liver Disease and Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:2650-2666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 923] [Cited by in RCA: 836] [Article Influence: 139.3] [Reference Citation Analysis (6)] |
| 336. | Singal AG, Kanwal F, Llovet JM. Global trends in hepatocellular carcinoma epidemiology: implications for screening, prevention and therapy. Nat Rev Clin Oncol. 2023;20:864-884. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 609] [Article Influence: 203.0] [Reference Citation Analysis (2)] |
| 337. | Tateishi R, Uchino K, Fujiwara N, Takehara T, Okanoue T, Seike M, Yoshiji H, Yatsuhashi H, Shimizu M, Torimura T, Moriyama M, Sakaida I, Okada H, Chiba T, Chuma M, Nakao K, Isomoto H, Sasaki Y, Kaneko S, Masaki T, Chayama K, Koike K. A nationwide survey on non-B, non-C hepatocellular carcinoma in Japan: 2011-2015 update. J Gastroenterol. 2019;54:367-376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 181] [Cited by in RCA: 172] [Article Influence: 24.6] [Reference Citation Analysis (1)] |
| 338. | Estes C, Anstee QM, Arias-Loste MT, Bantel H, Bellentani S, Caballeria J, Colombo M, Craxi A, Crespo J, Day CP, Eguchi Y, Geier A, Kondili LA, Kroy DC, Lazarus JV, Loomba R, Manns MP, Marchesini G, Nakajima A, Negro F, Petta S, Ratziu V, Romero-Gomez M, Sanyal A, Schattenberg JM, Tacke F, Tanaka J, Trautwein C, Wei L, Zeuzem S, Razavi H. Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016-2030. J Hepatol. 2018;69:896-904. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1680] [Cited by in RCA: 1432] [Article Influence: 179.0] [Reference Citation Analysis (5)] |
| 339. | Estes C, Chan HLY, Chien RN, Chuang WL, Fung J, Goh GB, Hu TH, Huang JF, Jang BK, Jun DW, Kao JH, Lee JW, Lin HC, Razavi-Shearer K, Seto WK, Wong GL, Wong VW, Razavi H. Modelling NAFLD disease burden in four Asian regions-2019-2030. Aliment Pharmacol Ther. 2020;51:801-811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 132] [Article Influence: 22.0] [Reference Citation Analysis (5)] |
| 340. | White DL, Kanwal F, El-Serag HB. Association between nonalcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin Gastroenterol Hepatol. 2012;10:1342-1359.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 609] [Cited by in RCA: 592] [Article Influence: 42.3] [Reference Citation Analysis (1)] |
| 341. | Kanwal F, Kramer JR, Li L, Dai J, Natarajan Y, Yu X, Asch SM, El-Serag HB. Effect of Metabolic Traits on the Risk of Cirrhosis and Hepatocellular Cancer in Nonalcoholic Fatty Liver Disease. Hepatology. 2020;71:808-819. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 244] [Article Influence: 40.7] [Reference Citation Analysis (4)] |
| 342. | El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557-2576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4288] [Cited by in RCA: 4170] [Article Influence: 219.5] [Reference Citation Analysis (4)] |
| 343. | Liangpunsakul S, Haber P, McCaughan GW. Alcoholic Liver Disease in Asia, Europe, and North America. Gastroenterology. 2016;150:1786-1797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 144] [Article Influence: 14.4] [Reference Citation Analysis (6)] |
| 344. | Baffy G, Brunt EM, Caldwell SH. Hepatocellular carcinoma in non-alcoholic fatty liver disease: an emerging menace. J Hepatol. 2012;56:1384-1391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 712] [Cited by in RCA: 659] [Article Influence: 47.1] [Reference Citation Analysis (4)] |
| 345. | Piscaglia F, Svegliati-Baroni G, Barchetti A, Pecorelli A, Marinelli S, Tiribelli C, Bellentani S; HCC-NAFLD Italian Study Group. Clinical patterns of hepatocellular carcinoma in nonalcoholic fatty liver disease: A multicenter prospective study. Hepatology. 2016;63:827-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 485] [Article Influence: 48.5] [Reference Citation Analysis (4)] |
| 346. | Yip TC, Lee HW, Chan WK, Wong GL, Wong VW. Asian perspective on NAFLD-associated HCC. J Hepatol. 2022;76:726-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 110] [Article Influence: 27.5] [Reference Citation Analysis (4)] |
| 347. | Ren H, Wang J, Gao Y, Yang F, Huang W. Metabolic syndrome and liver-related events: a systematic review and meta-analysis. BMC Endocr Disord. 2019;19:40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 348. | Yang JD, Ahmed F, Mara KC, Addissie BD, Allen AM, Gores GJ, Roberts LR. Diabetes Is Associated With Increased Risk of Hepatocellular Carcinoma in Patients With Cirrhosis From Nonalcoholic Fatty Liver Disease. Hepatology. 2020;71:907-916. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 173] [Cited by in RCA: 162] [Article Influence: 27.0] [Reference Citation Analysis (1)] |
| 349. | Puoti C. New insights on hepatocellular carcinoma: epidemiology and clinical aspects. Hepatoma Res. 2018;4:57. [DOI] [Full Text] |
| 350. | El-Serag HB, Kanwal F. Epidemiology of hepatocellular carcinoma in the United States: where are we? Hepatology. 2014;60:1767-1775. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 540] [Cited by in RCA: 509] [Article Influence: 42.4] [Reference Citation Analysis (5)] |
| 351. | Pal Chaudhary S, Reyes S, Chase ML, Govindan A, Zhao L, Luther J, Bhan I, Bethea E, Franses JW, Paige Walsh E, Anne Dageford L, Kimura S, Elias N, Yeh H, Markman J, Bozorgzadeh A, Tanabe K, Ferrone C, Zhu AX, Andersson K, Thiim M, Antonio Catalano O, Kambadakone A, Vagefi PA, Qadan M, Pratt D, Hashemi N, Corey KE, Misdraji J, Goyal L, Clark JW. Resection of NAFLD/NASH-related Hepatocellular Carcinoma (HCC): Clinical Features and Outcomes Compared with HCC Due to Other Etiologies. Oncologist. 2023;28:341-350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (7)] |
| 352. | Ponziani FR, Bhoori S, Castelli C, Putignani L, Rivoltini L, Del Chierico F, Sanguinetti M, Morelli D, Paroni Sterbini F, Petito V, Reddel S, Calvani R, Camisaschi C, Picca A, Tuccitto A, Gasbarrini A, Pompili M, Mazzaferro V. Hepatocellular Carcinoma Is Associated With Gut Microbiota Profile and Inflammation in Nonalcoholic Fatty Liver Disease. Hepatology. 2019;69:107-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 580] [Cited by in RCA: 544] [Article Influence: 77.7] [Reference Citation Analysis (5)] |
| 353. | Ferguson D, Finck BN. Emerging therapeutic approaches for the treatment of NAFLD and type 2 diabetes mellitus. Nat Rev Endocrinol. 2021;17:484-495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 409] [Cited by in RCA: 380] [Article Influence: 76.0] [Reference Citation Analysis (7)] |
| 354. | Geh D, Anstee QM, Reeves HL. NAFLD-Associated HCC: Progress and Opportunities. J Hepatocell Carcinoma. 2021;8:223-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 355. | Yasui K, Hashimoto E, Komorizono Y, Koike K, Arii S, Imai Y, Shima T, Kanbara Y, Saibara T, Mori T, Kawata S, Uto H, Takami S, Sumida Y, Takamura T, Kawanaka M, Okanoue T; Japan NASH Study Group, Ministry of Health, Labour, and Welfare of Japan. Characteristics of patients with nonalcoholic steatohepatitis who develop hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2011;9:428-33; quiz e50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 347] [Cited by in RCA: 325] [Article Influence: 21.7] [Reference Citation Analysis (4)] |
| 356. | Tan DJH, Ng CH, Lin SY, Pan XH, Tay P, Lim WH, Teng M, Syn N, Lim G, Yong JN, Quek J, Xiao J, Dan YY, Siddiqui MS, Sanyal AJ, Muthiah MD, Loomba R, Huang DQ. Clinical characteristics, surveillance, treatment allocation, and outcomes of non-alcoholic fatty liver disease-related hepatocellular carcinoma: a systematic review and meta-analysis. Lancet Oncol. 2022;23:521-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 250] [Article Influence: 62.5] [Reference Citation Analysis (0)] |
| 357. | Fujiwara N, Kubota N, Crouchet E, Koneru B, Marquez CA, Jajoriya AK, Panda G, Qian T, Zhu S, Goossens N, Wang X, Liang S, Zhong Z, Lewis S, Taouli B, Schwartz ME, Fiel MI, Singal AG, Marrero JA, Fobar AJ, Parikh ND, Raman I, Li QZ, Taguri M, Ono A, Aikata H, Nakahara T, Nakagawa H, Matsushita Y, Tateishi R, Koike K, Kobayashi M, Higashi T, Nakagawa S, Yamashita YI, Beppu T, Baba H, Kumada H, Chayama K, Baumert TF, Hoshida Y. Molecular signatures of long-term hepatocellular carcinoma risk in nonalcoholic fatty liver disease. Sci Transl Med. 2022;14:eabo4474. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 91] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 358. | Loomba R, Lim JK, Patton H, El-Serag HB. AGA Clinical Practice Update on Screening and Surveillance for Hepatocellular Carcinoma in Patients With Nonalcoholic Fatty Liver Disease: Expert Review. Gastroenterology. 2020;158:1822-1830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 303] [Cited by in RCA: 291] [Article Influence: 48.5] [Reference Citation Analysis (1)] |
| 359. | Schoenberger H, Chong N, Fetzer DT, Rich NE, Yokoo T, Khatri G, Olivares J, Parikh ND, Yopp AC, Marrero JA, Singal AG. Dynamic Changes in Ultrasound Quality for Hepatocellular Carcinoma Screening in Patients With Cirrhosis. Clin Gastroenterol Hepatol. 2022;20:1561-1569.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 106] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 360. | Tzartzeva K, Obi J, Rich NE, Parikh ND, Marrero JA, Yopp A, Waljee AK, Singal AG. Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients With Cirrhosis: A Meta-analysis. Gastroenterology. 2018;154:1706-1718.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 975] [Cited by in RCA: 931] [Article Influence: 116.4] [Reference Citation Analysis (6)] |
| 361. | Park HJ, Kim SY, Singal AG, Lee SJ, Won HJ, Byun JH, Choi SH, Yokoo T, Kim MJ, Lim YS. Abbreviated magnetic resonance imaging vs ultrasound for surveillance of hepatocellular carcinoma in high-risk patients. Liver Int. 2022;42:2080-2092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 11.3] [Reference Citation Analysis (1)] |
| 362. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J Hepatol. 2025;82:315-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 606] [Cited by in RCA: 524] [Article Influence: 524.0] [Reference Citation Analysis (9)] |
| 363. | Huang DQ, Fowler KJ, Liau J, Cunha GM, Louie AL, An JY, Bettencourt R, Jung J, Gitto Z, Hernandez C, Lopez SJ, Gupta H, Sirlin CB, Marks RM, Loomba R. Comparative efficacy of an optimal exam between ultrasound versus abbreviated MRI for HCC screening in NAFLD cirrhosis: A prospective study. Aliment Pharmacol Ther. 2022;55:820-827. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 50] [Article Influence: 12.5] [Reference Citation Analysis (1)] |
| 364. | Schütte K, Schulz C, Link A, Malfertheiner P. Current biomarkers for hepatocellular carcinoma: Surveillance, diagnosis and prediction of prognosis. World J Hepatol. 2015;7:139-149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (6)] |
| 365. | Bischoff SC, Bernal W, Dasarathy S, Merli M, Plank LD, Schütz T, Plauth M. ESPEN practical guideline: Clinical nutrition in liver disease. Clin Nutr. 2020;39:3533-3562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 305] [Cited by in RCA: 242] [Article Influence: 40.3] [Reference Citation Analysis (5)] |
| 366. | Zelber-Sagi S, Ratziu V, Oren R. Nutrition and physical activity in NAFLD: an overview of the epidemiological evidence. World J Gastroenterol. 2011;17:3377-3389. [PubMed] [DOI] [Full Text] |
| 367. | Koutoukidis DA, Astbury NM, Tudor KE, Morris E, Henry JA, Noreik M, Jebb SA, Aveyard P. Association of Weight Loss Interventions With Changes in Biomarkers of Nonalcoholic Fatty Liver Disease: A Systematic Review and Meta-analysis. JAMA Intern Med. 2019;179:1262-1271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 210] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 368. | Koutoukidis DA, Koshiaris C, Henry JA, Noreik M, Morris E, Manoharan I, Tudor K, Bodenham E, Dunnigan A, Jebb SA, Aveyard P. The effect of the magnitude of weight loss on non-alcoholic fatty liver disease: A systematic review and meta-analysis. Metabolism. 2021;115:154455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 369. | Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L, Torres-Gonzalez A, Gra-Oramas B, Gonzalez-Fabian L, Friedman SL, Diago M, Romero-Gomez M. Weight Loss Through Lifestyle Modification Significantly Reduces Features of Nonalcoholic Steatohepatitis. Gastroenterology. 2015;149:367-78.e5; quiz e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2098] [Cited by in RCA: 1840] [Article Influence: 167.3] [Reference Citation Analysis (5)] |
| 370. | Promrat K, Kleiner DE, Niemeier HM, Jackvony E, Kearns M, Wands JR, Fava JL, Wing RR. Randomized controlled trial testing the effects of weight loss on nonalcoholic steatohepatitis. Hepatology. 2010;51:121-129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1116] [Cited by in RCA: 1016] [Article Influence: 63.5] [Reference Citation Analysis (6)] |
| 371. | Tiikkainen M, Bergholm R, Vehkavaara S, Rissanen A, Häkkinen AM, Tamminen M, Teramo K, Yki-Järvinen H. Effects of identical weight loss on body composition and features of insulin resistance in obese women with high and low liver fat content. Diabetes. 2003;52:701-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 194] [Article Influence: 8.4] [Reference Citation Analysis (2)] |
| 372. | Tamura Y, Tanaka Y, Sato F, Choi JB, Watada H, Niwa M, Kinoshita J, Ooka A, Kumashiro N, Igarashi Y, Kyogoku S, Maehara T, Kawasumi M, Hirose T, Kawamori R. Effects of diet and exercise on muscle and liver intracellular lipid contents and insulin sensitivity in type 2 diabetic patients. J Clin Endocrinol Metab. 2005;90:3191-3196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 272] [Cited by in RCA: 239] [Article Influence: 11.4] [Reference Citation Analysis (5)] |
| 373. | Lazo M, Solga SF, Horska A, Bonekamp S, Diehl AM, Brancati FL, Wagenknecht LE, Pi-Sunyer FX, Kahn SE, Clark JM; Fatty Liver Subgroup of the Look AHEAD Research Group. Effect of a 12-month intensive lifestyle intervention on hepatic steatosis in adults with type 2 diabetes. Diabetes Care. 2010;33:2156-2163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 269] [Article Influence: 16.8] [Reference Citation Analysis (5)] |
| 374. | Behavioral Weight Loss Interventions to Prevent Obesity-Related Morbidity and Mortality in Adults: Recommendation Statement. Am Fam Physician. 2019;99:Online. [PubMed] |
| 375. | Durrer Schutz D, Busetto L, Dicker D, Farpour-Lambert N, Pryke R, Toplak H, Widmer D, Yumuk V, Schutz Y. European Practical and Patient-Centred Guidelines for Adult Obesity Management in Primary Care. Obes Facts. 2019;12:40-66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 404] [Cited by in RCA: 303] [Article Influence: 43.3] [Reference Citation Analysis (1)] |
| 376. | Garvey WT, Mechanick JI, Brett EM, Garber AJ, Hurley DL, Jastreboff AM, Nadolsky K, Pessah-Pollack R, Plodkowski R; Reviewers of the AACE/ACE Obesity Clinical Practice Guidelines. American association of clinical endocrinologists and american college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract. 2016;22 Suppl 3:1-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1481] [Cited by in RCA: 1208] [Article Influence: 120.8] [Reference Citation Analysis (4)] |
| 377. | Eslam M, Sarin SK, Wong VW, Fan JG, Kawaguchi T, Ahn SH, Zheng MH, Shiha G, Yilmaz Y, Gani R, Alam S, Dan YY, Kao JH, Hamid S, Cua IH, Chan WK, Payawal D, Tan SS, Tanwandee T, Adams LA, Kumar M, Omata M, George J. The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol Int. 2020;14:889-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 691] [Cited by in RCA: 634] [Article Influence: 105.7] [Reference Citation Analysis (4)] |
| 378. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388-1402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3686] [Cited by in RCA: 3319] [Article Influence: 331.9] [Reference Citation Analysis (7)] |
| 379. | Belančić A, Kenđel Jovanović G, Klobučar Majanović S. Obesity-related low-grade chronic inflammation: implementation of the dietary inflammatory index in clinical practice is the milestone? Med Flum. 2018;54:373-378. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 380. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1461] [Cited by in RCA: 1258] [Article Influence: 629.0] [Reference Citation Analysis (2)] |
| 381. | Ryan MC, Itsiopoulos C, Thodis T, Ward G, Trost N, Hofferberth S, O'Dea K, Desmond PV, Johnson NA, Wilson AM. The Mediterranean diet improves hepatic steatosis and insulin sensitivity in individuals with non-alcoholic fatty liver disease. J Hepatol. 2013;59:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 638] [Cited by in RCA: 574] [Article Influence: 44.2] [Reference Citation Analysis (4)] |
| 382. | Kawaguchi T, Charlton M, Kawaguchi A, Yamamura S, Nakano D, Tsutsumi T, Zafer M, Torimura T. Effects of Mediterranean Diet in Patients with Nonalcoholic Fatty Liver Disease: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Randomized Controlled Trials. Semin Liver Dis. 2021;41:225-234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (2)] |
| 383. | Esposito K, Maiorino MI, Ceriello A, Giugliano D. Prevention and control of type 2 diabetes by Mediterranean diet: a systematic review. Diabetes Res Clin Pract. 2010;89:97-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 141] [Article Influence: 8.8] [Reference Citation Analysis (1)] |
| 384. | Luukkonen PK, Dufour S, Lyu K, Zhang XM, Hakkarainen A, Lehtimäki TE, Cline GW, Petersen KF, Shulman GI, Yki-Järvinen H. Effect of a ketogenic diet on hepatic steatosis and hepatic mitochondrial metabolism in nonalcoholic fatty liver disease. Proc Natl Acad Sci U S A. 2020;117:7347-7354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 194] [Article Influence: 32.3] [Reference Citation Analysis (1)] |
| 385. | Boccatonda A, Tripaldi R, Davì G, Santilli F. Oxidative Stress Modulation Through Habitual Physical Activity. Curr Pharm Des. 2016;22:3648-3680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 386. | Hashida R, Kawaguchi T, Bekki M, Omoto M, Matsuse H, Nago T, Takano Y, Ueno T, Koga H, George J, Shiba N, Torimura T. Aerobic vs. resistance exercise in non-alcoholic fatty liver disease: A systematic review. J Hepatol. 2017;66:142-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 437] [Cited by in RCA: 365] [Article Influence: 40.6] [Reference Citation Analysis (4)] |
| 387. | Zhang HJ, He J, Pan LL, Ma ZM, Han CK, Chen CS, Chen Z, Han HW, Chen S, Sun Q, Zhang JF, Li ZB, Yang SY, Li XJ, Li XY. Effects of Moderate and Vigorous Exercise on Nonalcoholic Fatty Liver Disease: A Randomized Clinical Trial. JAMA Intern Med. 2016;176:1074-1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 220] [Article Influence: 22.0] [Reference Citation Analysis (3)] |
| 388. | St George A, Bauman A, Johnston A, Farrell G, Chey T, George J. Independent effects of physical activity in patients with nonalcoholic fatty liver disease. Hepatology. 2009;50:68-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 221] [Article Influence: 13.0] [Reference Citation Analysis (4)] |
| 389. | Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM; Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346:393-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14659] [Cited by in RCA: 12454] [Article Influence: 518.9] [Reference Citation Analysis (5)] |
| 390. | Ismail I, Keating SE, Baker MK, Johnson NA. A systematic review and meta-analysis of the effect of aerobic vs. resistance exercise training on visceral fat. Obes Rev. 2012;13:68-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 220] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 391. | Keating SE, Hackett DA, George J, Johnson NA. Exercise and non-alcoholic fatty liver disease: a systematic review and meta-analysis. J Hepatol. 2012;57:157-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 380] [Article Influence: 27.1] [Reference Citation Analysis (4)] |
| 392. | Rector RS, Thyfault JP, Morris RT, Laye MJ, Borengasser SJ, Booth FW, Ibdah JA. Daily exercise increases hepatic fatty acid oxidation and prevents steatosis in Otsuka Long-Evans Tokushima Fatty rats. Am J Physiol Gastrointest Liver Physiol. 2008;294:G619-G626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 240] [Article Influence: 13.3] [Reference Citation Analysis (3)] |
| 393. | van der Heijden GJ, Wang ZJ, Chu ZD, Sauer PJ, Haymond MW, Rodriguez LM, Sunehag AL. A 12-week aerobic exercise program reduces hepatic fat accumulation and insulin resistance in obese, Hispanic adolescents. Obesity (Silver Spring). 2010;18:384-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 170] [Article Influence: 10.6] [Reference Citation Analysis (3)] |
| 394. | Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen S, Calder PC, Sanders ME. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8109] [Cited by in RCA: 6147] [Article Influence: 512.3] [Reference Citation Analysis (13)] |
| 395. | Velayudham A, Dolganiuc A, Ellis M, Petrasek J, Kodys K, Mandrekar P, Szabo G. VSL#3 probiotic treatment attenuates fibrosis without changes in steatohepatitis in a diet-induced nonalcoholic steatohepatitis model in mice. Hepatology. 2009;49:989-997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 218] [Cited by in RCA: 207] [Article Influence: 12.2] [Reference Citation Analysis (1)] |
| 396. | Derosa G, Guasti L, D'Angelo A, Martinotti C, Valentino MC, Di Matteo S, Bruno GM, Maresca AM, Gaudio GV, Maffioli P. Probiotic Therapy With VSL#3(®) in Patients With NAFLD: A Randomized Clinical Trial. Front Nutr. 2022;9:846873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 397. | Chong PL, Laight D, Aspinall RJ, Higginson A, Cummings MH. A randomised placebo controlled trial of VSL#3(®) probiotic on biomarkers of cardiovascular risk and liver injury in non-alcoholic fatty liver disease. BMC Gastroenterol. 2021;21:144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 398. | Duseja A, Acharya SK, Mehta M, Chhabra S; Shalimar, Rana S, Das A, Dattagupta S, Dhiman RK, Chawla YK. High potency multistrain probiotic improves liver histology in non-alcoholic fatty liver disease (NAFLD): a randomised, double-blind, proof of concept study. BMJ Open Gastroenterol. 2019;6:e000315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 121] [Cited by in RCA: 105] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 399. | Silva-Sperb AS, Moraes HA, Barcelos STA, de Moura BC, Longo L, Michalczuk MT, Cerski CTS, Uribe-Cruz C, da Silveira TR, Álvares-da-Silva MR, Dall'Alba V. Probiotic supplementation for 24 weeks in patients with non-alcoholic steatohepatitis: the PROBILIVER randomized clinical trial. Front Nutr. 2024;11:1362694. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 400. | Arellano-García L, Portillo MP, Martínez JA, Milton-Laskibar I. Usefulness of Probiotics in the Management of NAFLD: Evidence and Involved Mechanisms of Action from Preclinical and Human Models. Int J Mol Sci. 2022;23:3167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 401. | Vallianou NG, Kounatidis D, Tsilingiris D, Panagopoulos F, Christodoulatos GS, Evangelopoulos A, Karampela I, Dalamaga M. The Role of Next-Generation Probiotics in Obesity and Obesity-Associated Disorders: Current Knowledge and Future Perspectives. Int J Mol Sci. 2023;24:6755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 86] [Article Influence: 28.7] [Reference Citation Analysis (2)] |
| 402. | American Diabetes Association Professional Practice Committee. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022;45:S125-S143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 678] [Cited by in RCA: 530] [Article Influence: 132.5] [Reference Citation Analysis (4)] |
| 403. | Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia. 2017;60:1577-1585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1875] [Cited by in RCA: 1586] [Article Influence: 176.2] [Reference Citation Analysis (6)] |
| 404. | Schulte L, Scheiner B, Voigtländer T, Koch S, Schweitzer N, Marhenke S, Ivanyi P, Manns MP, Rodt T, Hinrichs JB, Weinmann A, Pinter M, Vogel A, Kirstein MM. Treatment with metformin is associated with a prolonged survival in patients with hepatocellular carcinoma. Liver Int. 2019;39:714-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 50] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 405. | Li Q, Xu H, Sui C, Zhang H. Impact of metformin use on risk and mortality of hepatocellular carcinoma in diabetes mellitus. Clin Res Hepatol Gastroenterol. 2022;46:101781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 406. | Minassian C, Tarpin S, Mithieux G. Role of glucose-6 phosphatase, glucokinase, and glucose-6 phosphate in liver insulin resistance and its correction by metformin. Biochem Pharmacol. 1998;55:1213-1219. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 41] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 407. | Li Y, Liu L, Wang B, Wang J, Chen D. Metformin in non-alcoholic fatty liver disease: A systematic review and meta-analysis. Biomed Rep. 2013;1:57-64. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 230] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 408. | Musso G, Gambino R, Cassader M, Pagano G. A meta-analysis of randomized trials for the treatment of nonalcoholic fatty liver disease. Hepatology. 2010;52:79-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 471] [Cited by in RCA: 431] [Article Influence: 26.9] [Reference Citation Analysis (5)] |
| 409. | Loomba R, Lutchman G, Kleiner DE, Ricks M, Feld JJ, Borg BB, Modi A, Nagabhyru P, Sumner AE, Liang TJ, Hoofnagle JH. Clinical trial: pilot study of metformin for the treatment of non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2009;29:172-182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 245] [Cited by in RCA: 218] [Article Influence: 12.8] [Reference Citation Analysis (1)] |
| 410. | Shields WW, Thompson KE, Grice GA, Harrison SA, Coyle WJ. The Effect of Metformin and Standard Therapy versus Standard Therapy alone in Nondiabetic Patients with Insulin Resistance and Nonalcoholic Steatohepatitis (NASH): A Pilot Trial. Therap Adv Gastroenterol. 2009;2:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 113] [Article Influence: 6.6] [Reference Citation Analysis (4)] |
| 411. | Uygun A, Kadayifci A, Isik AT, Ozgurtas T, Deveci S, Tuzun A, Yesilova Z, Gulsen M, Dagalp K. Metformin in the treatment of patients with non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2004;19:537-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 272] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 412. | Chang CH, Lin JW, Wu LC, Lai MS, Chuang LM, Chan KA. Association of thiazolidinediones with liver cancer and colorectal cancer in type 2 diabetes mellitus. Hepatology. 2012;55:1462-1472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 147] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 413. | Lai SW, Lin CL, Liao KF. Association of hepatocellular carcinoma with thiazolidinediones use: A population-based case-control study. Medicine (Baltimore). 2020;99:e19833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 414. | Ratziu V, Giral P, Jacqueminet S, Charlotte F, Hartemann-Heurtier A, Serfaty L, Podevin P, Lacorte JM, Bernhardt C, Bruckert E, Grimaldi A, Poynard T; LIDO Study Group. Rosiglitazone for nonalcoholic steatohepatitis: one-year results of the randomized placebo-controlled Fatty Liver Improvement with Rosiglitazone Therapy (FLIRT) Trial. Gastroenterology. 2008;135:100-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 531] [Cited by in RCA: 475] [Article Influence: 26.4] [Reference Citation Analysis (3)] |
| 415. | Belfort R, Harrison SA, Brown K, Darland C, Finch J, Hardies J, Balas B, Gastaldelli A, Tio F, Pulcini J, Berria R, Ma JZ, Dwivedi S, Havranek R, Fincke C, DeFronzo R, Bannayan GA, Schenker S, Cusi K. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N Engl J Med. 2006;355:2297-2307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1535] [Cited by in RCA: 1349] [Article Influence: 67.5] [Reference Citation Analysis (4)] |
| 416. | Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, Bass NM, Neuschwander-Tetri BA, Lavine JE, Tonascia J, Unalp A, Van Natta M, Clark J, Brunt EM, Kleiner DE, Hoofnagle JH, Robuck PR; NASH CRN. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010;362:1675-1685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2814] [Cited by in RCA: 2559] [Article Influence: 159.9] [Reference Citation Analysis (3)] |
| 417. | Yen FS, Wei JC, Chiu LT, Hsu CC, Hou MC, Hwu CM. Thiazolidinediones were associated with higher risk of cardiovascular events in patients with type 2 diabetes and cirrhosis. Liver Int. 2021;41:110-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 418. | Gimeno RE, Briere DA, Seeley RJ. Leveraging the Gut to Treat Metabolic Disease. Cell Metab. 2020;31:679-698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 419. | Armstrong MJ, Gaunt P, Aithal GP, Barton D, Hull D, Parker R, Hazlehurst JM, Guo K; LEAN trial team, Abouda G, Aldersley MA, Stocken D, Gough SC, Tomlinson JW, Brown RM, Hübscher SG, Newsome PN. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387:679-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1837] [Cited by in RCA: 1650] [Article Influence: 165.0] [Reference Citation Analysis (6)] |
| 420. | Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, McGowan BM, Rosenstock J, Tran MTD, Wadden TA, Wharton S, Yokote K, Zeuthen N, Kushner RF; STEP 1 Study Group. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384:989-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3803] [Cited by in RCA: 3175] [Article Influence: 635.0] [Reference Citation Analysis (7)] |
| 421. | Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, Sanyal AJ, Sejling AS, Harrison SA; NN9931-4296 Investigators. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N Engl J Med. 2021;384:1113-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1693] [Cited by in RCA: 1492] [Article Influence: 298.4] [Reference Citation Analysis (7)] |
| 422. | O'Neil PM, Birkenfeld AL, McGowan B, Mosenzon O, Pedersen SD, Wharton S, Carson CG, Jepsen CH, Kabisch M, Wilding JPH. Efficacy and safety of semaglutide compared with liraglutide and placebo for weight loss in patients with obesity: a randomised, double-blind, placebo and active controlled, dose-ranging, phase 2 trial. Lancet. 2018;392:637-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 673] [Cited by in RCA: 589] [Article Influence: 73.6] [Reference Citation Analysis (4)] |
| 423. | Kuchay MS, Krishan S, Mishra SK, Choudhary NS, Singh MK, Wasir JS, Kaur P, Gill HK, Bano T, Farooqui KJ, Mithal A. Effect of dulaglutide on liver fat in patients with type 2 diabetes and NAFLD: randomised controlled trial (D-LIFT trial). Diabetologia. 2020;63:2434-2445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 188] [Article Influence: 31.3] [Reference Citation Analysis (1)] |
| 424. | Ghosh-Swaby OR, Goodman SG, Leiter LA, Cheng A, Connelly KA, Fitchett D, Jüni P, Farkouh ME, Udell JA. Glucose-lowering drugs or strategies, atherosclerotic cardiovascular events, and heart failure in people with or at risk of type 2 diabetes: an updated systematic review and meta-analysis of randomised cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2020;8:418-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 116] [Article Influence: 19.3] [Reference Citation Analysis (4)] |
| 425. | Hartman ML, Sanyal AJ, Loomba R, Wilson JM, Nikooienejad A, Bray R, Karanikas CA, Duffin KL, Robins DA, Haupt A. Effects of Novel Dual GIP and GLP-1 Receptor Agonist Tirzepatide on Biomarkers of Nonalcoholic Steatohepatitis in Patients With Type 2 Diabetes. Diabetes Care. 2020;43:1352-1355. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 297] [Cited by in RCA: 290] [Article Influence: 48.3] [Reference Citation Analysis (5)] |
| 426. | Hendryx M, Dong Y, Ndeke JM, Luo J. Sodium-glucose cotransporter 2 (SGLT2) inhibitor initiation and hepatocellular carcinoma prognosis. PLoS One. 2022;17:e0274519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (1)] |
| 427. | Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Furtado RHM, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Sabatine MS. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2383] [Cited by in RCA: 2074] [Article Influence: 296.3] [Reference Citation Analysis (4)] |
| 428. | Kahl S, Gancheva S, Straßburger K, Herder C, Machann J, Katsuyama H, Kabisch S, Henkel E, Kopf S, Lagerpusch M, Kantartzis K, Kupriyanova Y, Markgraf D, van Gemert T, Knebel B, Wolkersdorfer MF, Kuss O, Hwang JH, Bornstein SR, Kasperk C, Stefan N, Pfeiffer A, Birkenfeld AL, Roden M. Empagliflozin Effectively Lowers Liver Fat Content in Well-Controlled Type 2 Diabetes: A Randomized, Double-Blind, Phase 4, Placebo-Controlled Trial. Diabetes Care. 2020;43:298-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 265] [Article Influence: 44.2] [Reference Citation Analysis (1)] |
| 429. | Pradhan R, Yin H, Yu O, Azoulay L. Glucagon-Like Peptide 1 Receptor Agonists and Sodium-Glucose Cotransporter 2 Inhibitors and Risk of Nonalcoholic Fatty Liver Disease Among Patients With Type 2 Diabetes. Diabetes Care. 2022;45:819-829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 430. | Gastaldelli A, Repetto E, Guja C, Hardy E, Han J, Jabbour SA, Ferrannini E. Exenatide and dapagliflozin combination improves markers of liver steatosis and fibrosis in patients with type 2 diabetes. Diabetes Obes Metab. 2020;22:393-403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (1)] |
| 431. | Harrison SA, Bedossa P, Guy CD, Schattenberg JM, Loomba R, Taub R, Labriola D, Moussa SE, Neff GW, Rinella ME, Anstee QM, Abdelmalek MF, Younossi Z, Baum SJ, Francque S, Charlton MR, Newsome PN, Lanthier N, Schiefke I, Mangia A, Pericàs JM, Patil R, Sanyal AJ, Noureddin M, Bansal MB, Alkhouri N, Castera L, Rudraraju M, Ratziu V; MAESTRO-NASH Investigators. A Phase 3, Randomized, Controlled Trial of Resmetirom in NASH with Liver Fibrosis. N Engl J Med. 2024;390:497-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1457] [Cited by in RCA: 1320] [Article Influence: 660.0] [Reference Citation Analysis (2)] |
| 432. | Browning JD. Statins and hepatic steatosis: perspectives from the Dallas Heart Study. Hepatology. 2006;44:466-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 115] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 433. | Foster T, Budoff MJ, Saab S, Ahmadi N, Gordon C, Guerci AD. Atorvastatin and antioxidants for the treatment of nonalcoholic fatty liver disease: the St Francis Heart Study randomized clinical trial. Am J Gastroenterol. 2011;106:71-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 204] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 434. | Athyros VG, Tziomalos K, Gossios TD, Griva T, Anagnostis P, Kargiotis K, Pagourelias ED, Theocharidou E, Karagiannis A, Mikhailidis DP; GREACE Study Collaborative Group. Safety and efficacy of long-term statin treatment for cardiovascular events in patients with coronary heart disease and abnormal liver tests in the Greek Atorvastatin and Coronary Heart Disease Evaluation (GREACE) Study: a post-hoc analysis. Lancet. 2010;376:1916-1922. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 540] [Cited by in RCA: 518] [Article Influence: 32.4] [Reference Citation Analysis (2)] |
| 435. | Dufour JF, Oneta CM, Gonvers JJ, Bihl F, Cerny A, Cereda JM, Zala JF, Helbling B, Steuerwald M, Zimmermann A; Swiss Association for the Study of the Liver. Randomized placebo-controlled trial of ursodeoxycholic acid with vitamin e in nonalcoholic steatohepatitis. Clin Gastroenterol Hepatol. 2006;4:1537-1543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 253] [Article Influence: 12.7] [Reference Citation Analysis (4)] |
| 436. | Yakaryilmaz F, Guliter S, Savas B, Erdem O, Ersoy R, Erden E, Akyol G, Bozkaya H, Ozenirler S. Effects of vitamin E treatment on peroxisome proliferator-activated receptor-alpha expression and insulin resistance in patients with non-alcoholic steatohepatitis: results of a pilot study. Intern Med J. 2007;37:229-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 437. | Bjelakovic G, Gluud LL, Nikolova D, Bjelakovic M, Nagorni A, Gluud C. Meta-analysis: antioxidant supplements for liver diseases - the Cochrane Hepato-Biliary Group. Aliment Pharmacol Ther. 2010;32:356-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 438. | Lincoff AM, Wolski K, Nicholls SJ, Nissen SE. Pioglitazone and risk of cardiovascular events in patients with type 2 diabetes mellitus: a meta-analysis of randomized trials. JAMA. 2007;298:1180-1188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1062] [Cited by in RCA: 900] [Article Influence: 47.4] [Reference Citation Analysis (3)] |
| 439. | Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142:37-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2065] [Cited by in RCA: 1618] [Article Influence: 77.0] [Reference Citation Analysis (3)] |
| 440. | Berry D, Wathen JK, Newell M. Bayesian model averaging in meta-analysis: vitamin E supplementation and mortality. Clin Trials. 2009;6:28-41. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 441. | American Diabetes Association Professional Practice Committee. 8. Obesity and Weight Management for the Prevention and Treatment of Type 2 Diabetes: Standards of Medical Care in Diabetes-2022. Diabetes Care. 2022;45:S113-S124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 136] [Article Influence: 34.0] [Reference Citation Analysis (3)] |
| 442. | Laursen TL, Hagemann CA, Wei C, Kazankov K, Thomsen KL, Knop FK, Grønbæk H. Bariatric surgery in patients with non-alcoholic fatty liver disease - from pathophysiology to clinical effects. World J Hepatol. 2019;11:138-149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 129] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 443. | Lassailly G, Caiazzo R, Ntandja-Wandji LC, Gnemmi V, Baud G, Verkindt H, Ningarhari M, Louvet A, Leteurtre E, Raverdy V, Dharancy S, Pattou F, Mathurin P. Bariatric Surgery Provides Long-term Resolution of Nonalcoholic Steatohepatitis and Regression of Fibrosis. Gastroenterology. 2020;159:1290-1301.e5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 520] [Cited by in RCA: 455] [Article Influence: 75.8] [Reference Citation Analysis (5)] |
| 444. | Bazerbachi F, Vargas EJ, Rizk M, Maselli DB, Mounajjed T, Venkatesh SK, Watt KD, Port JD, Basu R, Acosta A, Hanouneh I, Gara N, Shah M, Mundi M, Clark M, Grothe K, Storm AC, Levy MJ, Abu Dayyeh BK. Intragastric Balloon Placement Induces Significant Metabolic and Histologic Improvement in Patients With Nonalcoholic Steatohepatitis. Clin Gastroenterol Hepatol. 2021;19:146-154.e4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 445. | Ramai D, Singh J, Lester J, Khan SR, Chandan S, Tartaglia N, Ambrosi A, Serviddio G, Facciorusso A. Systematic review with meta-analysis: bariatric surgery reduces the incidence of hepatocellular carcinoma. Aliment Pharmacol Ther. 2021;53:977-984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 446. | Are VS, Knapp SM, Banerjee A, Shamseddeen H, Ghabril M, Orman E, Patidar KR, Chalasani N, Desai AP. Improving Outcomes of Bariatric Surgery in Patients With Cirrhosis in the United States: A Nationwide Assessment. Am J Gastroenterol. 2020;115:1849-1856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (1)] |
| 447. | Caiazzo R, Lassailly G, Leteurtre E, Baud G, Verkindt H, Raverdy V, Buob D, Pigeyre M, Mathurin P, Pattou F. Roux-en-Y gastric bypass versus adjustable gastric banding to reduce nonalcoholic fatty liver disease: a 5-year controlled longitudinal study. Ann Surg. 2014;260:893-8; discussion 898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 187] [Article Influence: 15.6] [Reference Citation Analysis (3)] |
| 448. | Khan Z, Khan MA, Hajifathalian K, Shah S, Abdul M, Saumoy M, Aronne L, Lee W, Sharaiha RZ. Efficacy of Endoscopic Interventions for the Management of Obesity: a Meta-analysis to Compare Endoscopic Sleeve Gastroplasty, AspireAssist, and Primary Obesity Surgery Endolumenal. Obes Surg. 2019;29:2287-2298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 449. | Ren M, Zhou X, Zhang Y, Mo F, Yang J, Yu M, Ji F. Effects of Bariatric Endoscopy on Non-Alcoholic Fatty Liver Disease: A Comprehensive Systematic Review and Meta-Analysis. Front Endocrinol (Lausanne). 2022;13:931519. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |