Published online Mar 27, 2026. doi: 10.4254/wjh.v18.i3.112934
Revised: September 25, 2025
Accepted: January 8, 2026
Published online: March 27, 2026
Processing time: 228 Days and 18.2 Hours
Liver transplantation (LT) is the most effective treatment for the advanced stages of primary sclerosing cholangitis (PSC). However, up to 30% of patients develop recurrence of PSC (rPSC), which negatively affects graft and patient outcomes. Given the heterogeneous nature of PSC, patients undergo LT either due to end-stage liver disease (ESLD) or to symptoms that significantly reduce quality of life (non-ESLD), such as recurrent bacterial cholangitis or refractory pruritus. How
To compare post-LT outcomes between PSC recipients with ESLD and non-ESLD indications, and identify rPSC and graft failure risk factors.
This single-center retrospective study comprised 131 adult LT recipients for PSC (including PSC/autoimmune overlap). Patients were grouped by listing in
No significant difference was found in the incidence of rPSC, graft survival, or overall survival between patients indicated for LT for ESLD and those with non-advanced symptomatic disease. Cytomegalovirus infection (hazard ratio [HR] = 2.16; 95% confidence interval [CI]: 1.05-4.46), acute cellular rejection (ACR) (HR = 3.95; 95%CI: 1.44-10.8), and length of hospitalization after LT (HR = 1.02; 95%CI: 1.01-1.04) were significantly associated with risk of rPSC. In addition, multiple episodes of ACR (HR = 4.93; 95%CI: 1.22-19.9) and the length of hospitalization after LT (HR = 1.04; 95%CI: 1.01-1.06) were significantly associated with graft failure.
Patients with PSC with advanced liver cirrhosis before LT did not have worse post-transplant outcomes than those without ESLD. Cytomegalovirus infection, ACR, and prolonged hospitalization after LT were associated with worse out
Core Tip: This study demonstrated the outcomes of liver transplantation (LT) recipients with primary sclerosing cholangitis (PSC) and risk factors for recurrence of PSC (rPSC) and graft failure. Patients with PSC undergoing LT for end-stage liver disease have similar rPSC rates, graft failure rates, and overall survival comparable to those undergoing LT for non-end-stage liver disease indications (e.g., recurrent bacterial cholangitis). Acute cellular rejection, cytomegalovirus infection, and longer post
- Citation: Hlavaty M, Brezina J, Wohl P, Modos I, Bajer L, Fabian O, Vajsova A, Hucl T, Drastich P. Patients with primary sclerosing cholangitis with and without end-stage liver disease have similar outcomes after liver transplantation. World J Hepatol 2026; 18(3): 112934
- URL: https://www.wjgnet.com/1948-5182/full/v18/i3/112934.htm
- DOI: https://dx.doi.org/10.4254/wjh.v18.i3.112934
Primary sclerosing cholangitis (PSC) is a chronic liver disease of unknown etiology that is associated with inflammatory bowel disease (IBD) and characterized by progressive inflammation and fibrosis of extra- and intrahepatic bile ducts[1,2]. Biliary fibrosis leads to the development of multifocal strictures that result in episodes of recurrent cholangitis, secondary biliary cirrhosis, cholangiocarcinoma, and ultimately death in some patients[1,3]. As the disease slowly progresses despite conservative treatment, liver transplantation (LT) is the only definitive treatment[4,5].
Unfortunately, recurrence of PSC (rPSC) is a common long-term complication of LT, which significantly decreases both graft survival and overall patient survival[6-8]. The reported incidence of rPSC is about 25%[6,7,9-11]. The etiology of rPSC has been extensively studied, and although it is known to be multifactorial, it is not entirely understood. In a meta-analysis of 14 studies, Steenstraten et al[12] identified several factors associated with an increased risk of rPSC, including the presence of IBD or cholangiocarcinoma, donor age, one or multiple occurrences of acute cellular rejection (ACR), and an increased model for end-stage liver disease (ESLD) (MELD) score. The association between an increased MELD score and rPSC suggests that patients with PSC and advanced liver cirrhosis are at an increased risk of worse outcomes after LT compared with those without advanced cirrhosis, although this has not yet been confirmed. Despite these valuable insights, results across studies remain inconsistent. Further research is needed to improve our understanding of recurrent disease, and eventually, to develop clinical recommendations for this rare condition.
The primary study objective was to evaluate whether patients with PSC and ESLD indicated for LT had higher rPSC rates and lower graft and overall survival than those indicated for LT for non-ESLD indications. The secondary objectives were to assess the overall outcomes of LT and to identify factors associated with an increased risk of rPSC and graft failure in this cohort.
This single-center retrospective study included patients who underwent LT for PSC at our transplant center between January 1, 1999 and December 31, 2019. Patients with different diagnoses confirmed on liver explant and those who underwent liver retransplantation were excluded from the study. Patient data through October 2022 were retrieved retrospectively from digital and paper medical records. Collected data included demographic variables, PSC-related data, PSC phenotype, the presence of overlap features or liver cirrhosis, history of decompensation, and pre-LT Child-Pugh and MELD scores. Furthermore, LT-related data were collected, including age at LT, graft type (full/reduced/split), donor criteria, donor age, donor sex, ABO incompatibility, immunosuppression regimen, type of biliary anastomosis, length of transplant-associated hospitalization, history of ACR or cytomegalovirus (CMV) infection, and IBD-related data. The diagnosis of rPSC was based on the criteria proposed by Graziadei et al[13]. PSC was confirmed by cholangiography before LT and liver explant evaluation. Magnetic resonance cholangiopancreatography was performed cross-sectionally before data collection to assess the prevalence of rPSC. Hepatic artery thrombosis was excluded by Doppler ultrasound and/or computed tomography angiography in patients with suspected rPSC. Liver biopsies were performed per protocol in most of the patients to exclude chronic rejection or ductopenia. ACR was defined as biopsy-proven and graded according to the Banff criteria, and “multiple episodes of ACR” was defined as ≥ 2 discrete episodes requiring anti-rejection therapy[14]. Graft failure was defined as either retransplantation or death related to graft failure.
As this was a retrospective study, no specific protocol was followed. However, all included patients had been routinely followed by our transplant center at 3-month intervals. After enrollment, the patients were categorized based on the severity of liver dysfunction before LT. The first group consisted of patients who underwent LT for ESLD, defined as ESLD = (MELD score ≥ 15 and Child-Pugh score ≥ 8) or ≥ 1 sign of decompensated cirrhosis (history of variceal bleeding, ascites, or hepatic encephalopathy).
Jaundice was excluded as a sign of decompensated cirrhosis because it could not be reliably distinguished from obstructive jaundice, which is common in patients with PSC. The presence of liver cirrhosis was evaluated using imaging methods such as ultrasound, computed tomography, and magnetic resonance imaging, and was further confirmed by histology of the liver explant. The second group included patients indicated for LT because of low quality of life in the absence of ESLD, with or without compensated liver cirrhosis. These patients presented with recurrent episodes of acute cholangitis, pruritus refractory to treatment, or progressive jaundice despite endoscopic treatment. At the time of this study, cholangiocarcinoma was not considered an indication for LT at our center.
Data were entered into Microsoft Excel and analyzed using R statistics. Kaplan-Meier analysis was used to estimate recurrence rates, graft survival, and patient survival, and log-rank tests were used to determine the significance of differences between groups. Factors associated with the risk of rPSC and graft failure were assessed using Kaplan-Meier and Cox proportional hazard models with baseline and time-updated covariates. The statistical analysis was reviewed by a biomedical statistician.
A total of 1771 LTs were performed between 1999 and 2019 at the Institute of Clinical and Experimental Medicine (Prague, Czech Republic), of which 186 were performed for PSC. Liver retransplantation recipients (n = 16) were excluded from the final analysis, as well as 10 patients whose follow-up was conducted at another center, 14 patients with insufficient data, 7 patients with different diagnoses on liver explant, 7 patients with ABO-incompatible grafts, and 1 patient with extrahepatic malignancy. The remaining 131 patients met the eligibility criteria and were included in the study.
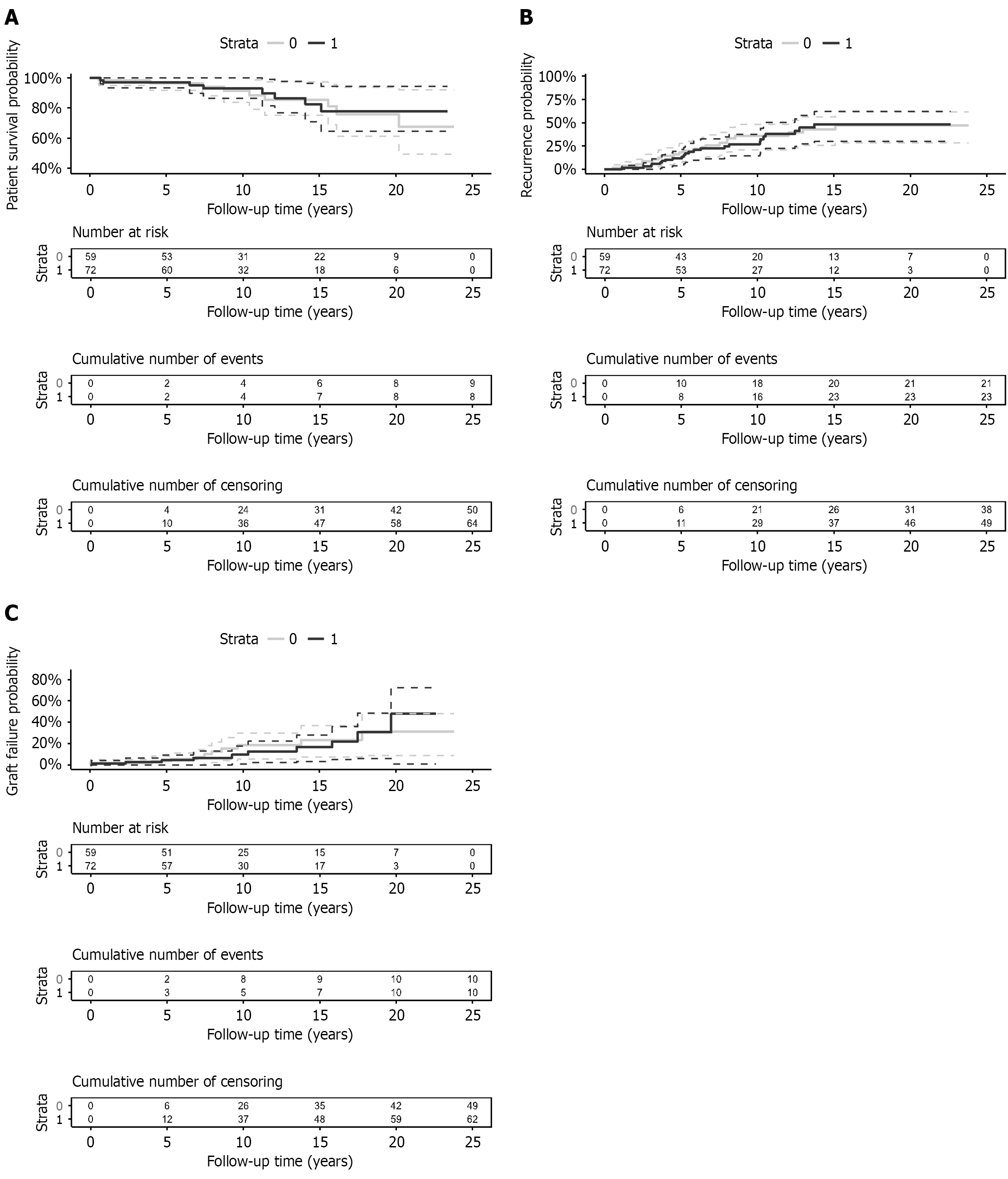
The characteristics of all the included patients are summarized in Table 1. As expected, most patients were male (n = 96, 73.3%), and IBD was present in 93 patients (71.0%), including 86 with ulcerative colitis and 7 with Crohn’s disease. The median age at LT was 38 (interquartile range [IQR]: 13-69) years, and the median follow-up period was 7.85 (IQR: 4.57-12.98) years. The overall survival, graft survival, and recurrence rates are displayed in Figure 1A.
| Characteristic | Total (n = 131) | ESLD (n = 72) | nESLD (n = 59) | P value |
| Sex | ||||
| Male | 96 (73.3) | 49 (68.1) | 47 (79.7) | - |
| Female | 35 (26.7) | 23 (31.9) | 12 (20.3) | - |
| Age at LT, median (IQR) | 38 (30, 47) | 40 (33, 51) | 37 (28, 45) | NS |
| MELD at LT, median (IQR) | 15 (12, 19) | 17 (13, 21) | 13 (11, 15) | - |
| PSC phenotype | ||||
| Large duct | 130 (99.2) | 72 (98.6) | 59 (100.0) | NS |
| Small duct | 1 (0.8) | 1 (1.4) | 0 (0.0) | - |
| PSC/AIH overlap | 33 (25.2) | 24 (33.3) | 9 (15.3) | 0.025 |
| IBD | ||||
| UC | 86 (65.6) | 42 (58.3) | 44 (74.6) | - |
| CD | 7 (5.3) | 6 (8.3) | 1 (1.7) | - |
| No IBD | 38 (29.0) | 24 (33.4) | 14 (23.7) | NS |
| Colectomy | ||||
| Before LT | 6 (4.6) | 3 (4.2) | 3 (5.1) | NS |
| After LT | 14 (10.7) | 5 (6.9) | 9 (15.3) | - |
| rPSC | ||||
| Present | 44 (33.6) | 23 (31.9) | 21 (35.6) | NS |
| No rPSC | 87 (66.4) | 49 (68.1) | 38 (64.4) | - |
| Immunosuppression | ||||
| Tacrolimus | 113 (86.3) | 63 (87.5) | 50 (84.7) | NS |
| Cyclosporine | 18 (13.7) | 9 (12.5) | 9 (15.3) | - |
| Donor type | ||||
| DBD | 131 (100.0) | 72 (100.0) | 59 (100.0) | NS |
| DCD | 0 (0.0) | 0 (0.0) | 0 (0.0) | - |
| Anastomosis | ||||
| Roux-en-Y | 78 (59.5) | 39 (54.2) | 39 (66.1) | NS |
| Duct-to-duct | 53 (40.5) | 33 (45.8) | 20 (33.9) | - |
| Follow-up, median (minimum, maximum) | 7.85 (4.57, 12.98) | 7.81 (5.11, 13.03) | 8.20 (4.57, 12.59) | NS |
During follow-up, 17 patients (13.0%) died. The main causes of death were infections (n = 7), malignancies (n = 7), graft failure (n = 1), severe autoimmune hemolytic anemia (n = 1), and unknown cause (n = 1).
The most frequent indication for LT in our cohort was ESLD, present in 72 patients (54.9%). The remaining 59 patients (45.1%) had a non-ESLD indication, namely recurrent cholangitis in 35 (59.3%), refractory pruritus in 3 (5.1%), or progressive jaundice despite endoscopic treatment in 21 patients (35.6%). The characteristics of the study groups are listed in Table 1. Notably, the ESLD group included a significantly higher number of patients with PSC/autoimmune hepatitis (AIH) overlap syndrome (P = 0.03). After LT, the unadjusted overall survival in the ESLD group was 97% at 5 years, 93% at 10 years, and 83% at 15 years. The corresponding survival rates in the non-ESLD group were 97%, 92%, and 85% respectively, with no significant intergroup differences (P = 0.8; Figure 1A). The cumulative incidence of rPSC in the ESLD group was 12% at 5 years, 27% at 10 years, and 48% at 15 years. There was no significant difference in the incidence of rPSC when compared with the non-ESLD group, with rPSC rates of 18% at 5 years, 36% at 10 years, and 43% at 15 years (Figure 1B). Similarly, the cumulative incidence of graft failure in the ESLD group was 4.5%, 9.6%, and 17% at 5 years, 10 years, and 15 years, with no significant difference when compared with the non-ESLD group (3.5%, 19%, and 23% at 5 years, 10 years, and 15 years, respectively; Figure 1C). In total, 10 patients in the ESLD group and 10 in the non-ESLD group underwent retransplantation, with no significant difference between the groups.
A total of 33 patients (25.2%) with PSC/AIH overlap syndrome were included in the study. Compared with the PSC-only group, these patients had significantly higher Child-Pugh scores at the time of LT (P = 0.0079) and a more frequent history of decompensation before LT (P = 0.0144), resulting in a greater proportion being classified in the ESLD group (P = 0.025). They were also less likely to have concomitant PSC-IBD (P = 0.0052). In addition, these patients underwent duct-to-duct biliary anastomoses more frequently (P = 0.0008) and received grafts from donors after brain death with expanded donor criteria more often (P = 0.0092; Supplementary Table 1).
In our cohort, 57 patients (43.5%) were diagnosed with biliary strictures after LT, including 34 (26.0%) with non-anastomotic biliary strictures, 13 (9.9%) with anastomotic strictures, and 10 (7.6%) with both anastomotic and non-anastomotic strictures. was diagnosed in 44 patients (33.6%), with a median time to diagnosis of rPSC of 5.39 (IQR: 3.62-8.26) years.
In the univariate Cox proportional hazards model, CMV infection at any time after LT was associated with a two-fold increased risk of rPSC (Table 2). In the same model, multiple episodes of ACR significantly increased the risk of rPSC, whereas a single episode of ACR was not significantly associated with rPSC. The MELD score, cyclosporine-based immunosuppression, presence of IBD or cholangiocarcinoma, and other previously known risk factors for rPSC were not found to be significantly associated with rPSC risk in these patients. When all parameters were included in a multivariate Cox regression model (Table 2), CMV infection (hazard ratio [HR] = 2.16; 95% confidence interval [CI]: 1.05-4.46) and multiple episodes of ACR (HR = 3.95; 95%CI: 1.44-10.8) occurring any time after LT were significant independent risk factors for the development of rPSC. Longer hospitalization after LT was also associated with an increased risk (HR = 1.02; 95%CI: 1.01-1.04).
| Characteristic | Univariable | Multivariable | |||||
| n | HR | 95%CI | P value | HR | 95%CI | P value | |
| Age at LT | 131 | 0.99 | 0.96-1.01 | 0.4 | 0.99 | 0.96-1.02 | 0.5 |
| Sex | 131 | - | - | - | - | - | - |
| Female | - | - | - | - | - | - | - |
| Male | - | 1.17 | 0.59-2.31 | 0.7 | 1.23 | 0.54-2.78 | 0.6 |
| Overlap PSC/AIH | 131 | 1.54 | 0.80-2.95 | 0.2 | 2.13 | 0.87-5.21 | 0.1 |
| IBD before LT | 131 | 1 | 0.52-1.91 | > 0.9 | 1.05 | 0.50-2.17 | > 0.9 |
| Liver cirrhosis | 131 | 1.78 | 0.70-4.52 | 0.2 | 2.34 | 0.82-6.69 | 0.11 |
| Decompensation of liver cirrhosis | 131 | 0.96 | 0.53-1.74 | 0.9 | 0.84 | 0.32-2.17 | 0.7 |
| Child-Pugh score prior to LT | 131 | 1.09 | 0.94-1.26 | 0.3 | - | - | - |
| MELD prior to LT | 131 | 1.03 | 0.98-1.09 | 0.2 | 1 | 0.93-1.07 | > 0.9 |
| End-stage liver disease indication | 131 | 0.92 | 0.51-1.66 | 0.8 | 1.04 | 0.39-2.73 | > 0.9 |
| Split or reduced liver graft | 131 | 2.42 | 0.93-6.26 | 0.07 | 5 | 1.62-15.4 | 0.005 |
| Donor type | 131 | - | - | - | - | - | - |
| DBD-ECD | - | - | - | - | - | - | - |
| DBD-SCD | - | 0.62 | 0.31-1.25 | 0.2 | 1.06 | 0.45-2.49 | 0.9 |
| Donor-recipient sex missmatch | 131 | 1.37 | 0.76-2.49 | 0.3 | 1.65 | 0.78-3.52 | 0.2 |
| Cyclosporine | 131 | 0.52 | 0.20-1.33 | 0.2 | 0.4 | 0.13-1.27 | 0.12 |
| Duct-to-duct anastomosis | 131 | 1.32 | 0.70-2.47 | 0.4 | 0.91 | 0.41-2.03 | 0.8 |
| Episode of ACR | 131 | 1.19 | 0.66-2.15 | 0.6 | 1.08 | 0.52-2.26 | 0.8 |
| Multiple episodes of ACR | 131 | 2.28 | 1.06-4.91 | 0.036 | 3.95 | 1.44-10.8 | 0.008 |
| CMV infection | 131 | 2.1 | 1.14-3.89 | 0.018 | 2.16 | 1.05-4.46 | 0.037 |
| Length of hospitalization | 131 | 1.01 | 1.00-1.03 | 0.12 | 1.02 | 1.01-1.04 | 0.009 |
Across the entire cohort, 20 patients (15.2%) experienced graft failure. The most common cause was rPSC in 17 patients (85%), followed by hepatic artery thrombosis in 1 patient (5%), primary graft dysfunction in 1 patient (5%), and acute-on-chronic liver failure due to hemorrhagic shock in 1 patient (5%). Among patients with graft loss because of rPSC, the median time to retransplantation was 8.6 (IQR: 6.8-13.8) years. In addition, 3 patients underwent a second liver retransplantation, and 1 patient underwent a third liver retransplantation. In univariate analysis, the most significant risk factor for graft failure was multiple episodes of ACR (HR = 3.48; 95%CI: 1.32-9.14). In addition, the length of hospitalization after LT was associated with a higher risk of graft failure (HR = 1.02; 95%CI: 1.0-1.04). In multivariate analysis, both length of hospitalization after LT (HR = 1.04; 95%CI: 1.01-1.06) and multiple episodes of ACR (HR = 4.93; 95%CI: 1.22-19.9) were significantly associated with graft failure (Table 3).
| Characteristic | Univariable | Multivariable | |||||
| n | HR | 95%CI | P value | HR | 95%CI | P value | |
| Age at LT | 131 | 0.98 | 0.94-1.01 | 0.2 | 0.97 | 0.92-1.02 | 0.2 |
| Sex | 131 | - | - | - | - | - | - |
| Female | - | - | - | - | - | - | - |
| Male | - | 1.69 | 0.56-5.09 | 0.3 | 2.67 | 0.61-11.7 | 0.2 |
| Overlap PSC/AIH | 131 | 1.53 | 0.55-4.29 | 0.4 | 4.07 | 0.93-17.9 | 0.063 |
| IBD before LT | 131 | 0.77 | 0.31-1.93 | 0.6 | 1.28 | 0.39-4.15 | 0.7 |
| Liver cirrhosis | 131 | 1.33 | 0.39-4.55 | 0.7 | 1.34 | 0.29-6.18 | 0.7 |
| Decompensation of liver cirrhosis | 131 | 1.02 | 0.42-2.48 | > 0.9 | 0.69 | 0.17-2.77 | 0.6 |
| Child-Pugh score prior to LT | 131 | 1.06 | 0.83-1.36 | 0.6 | - | - | - |
| MELD prior to LT | 131 | 1.01 | 0.93-1.10 | 0.8 | 0.95 | 0.82-1.10 | 0.5 |
| End-stage liver disease indication | 131 | 0.93 | 0.39-2.25 | 0.9 | 1.73 | 0.36-8.38 | 0.5 |
| Split or reduced graft | 131 | 1.55 | 0.20-12.2 | 0.7 | 4.76 | 0.46-48.8 | 0.2 |
| Donor type | 131 | - | - | - | - | - | - |
| DBD-ECD | - | - | - | - | - | - | - |
| DBD-SCD | - | 0.81 | 0.22-2.90 | 0.7 | 2.29 | 0.45-11.5 | 0.3 |
| Donor-recipient sex missmatch | 131 | 1.22 | 0.50-2.96 | 0.7 | 2.21 | 0.63-7.80 | 0.2 |
| Cyclosporine | 131 | 0.54 | 0.17-1.76 | 0.3 | 0.21 | 0.04-1.06 | 0.059 |
| Duct-to-duct anastomosis | 131 | 0.76 | 0.25-2.36 | 0.6 | 0.58 | 0.14-2.46 | 0.5 |
| Episode of ACR | 131 | 1.51 | 0.62-3.72 | 0.4 | 1.36 | 0.38-4.81 | 0.6 |
| Multiple episodes of ACR | 131 | 3.48 | 1.32-9.14 | 0.011 | 4.93 | 1.22-19.9 | 0.025 |
| CMV infection | 131 | 2.14 | 0.87-5.24 | 0.10 | 2.51 | 0.77-8.19 | 0.13 |
| Length of hospitalization | 131 | 1.02 | 1.00-1.04 | 0.029 | 1.04 | 1.01-1.06 | 0.001 |
As rPSC was the most common cause of graft failure, we performed a subgroup analysis including only patients with rPSC, comparing those who experienced graft failure with those who did not (Supplementary Table 2). Roux-en-Y reconstruction was significantly more common in the group with rPSC who experienced graft failure (P = 0.03), and multiple episodes of ACR were associated with both rPSC and graft failure.
In this retrospective study from a high-volume LT center, which included 131 LT recipients for PSC, post-LT outcomes and risk factors for rPSC and graft failure were evaluated. The post-LT outcomes of patients with an ESLD indication were compared with those of patients with non-ESLD indications. No significant differences were observed in the prevalence of rPSC, graft failure, or overall survival between the two groups. Several independent risk factors for both rPSC and graft failure were identified, such as ACR, CMV infection, and prolonged postoperative hospitalization.
The methodology applied in this study was similar to that used in other retrospective studies of rPSC, in that rPSC was analyzed as a time-dependent variable. The basic demographic characteristics, such as age at LT, sex distribution, and presence of IBD, were also comparable with those reported in other studies[6,7,9-11,13,15-21]. During the study period, the rPSC rate in our cohort was 33.6%, nearly double the 16.7% rate reported in a recent large European multicenter study[22]. The prevalence of rPSC in our cohort was assessed using standard criteria originally proposed by Graziadei et al[13]. This finding may suggest that rPSC is generally underdiagnosed.
Although the prevalence of rPSC in our cohort was relatively high, the mortality and graft failure rates were comparable with those reported in European registry data[12]. In contrast, the number of colectomies performed before LT was relatively low and was therefore not included in the analysis of risk factors. Notably, the proportion of patients with PSC/AIH overlap syndrome in our cohort was considerably higher than in other published studies (25% vs 6%-14%)[23]. Although patients with overlap syndrome presented with more advanced disease and could represent a potential confounder, incorporation of overlap status into both univariate and multivariate Cox analyses did not identify it as an independent predictor of rPSC or graft failure in our cohort. However, given the limited sample size, a modest effect cannot be excluded, and these results should therefore be interpreted with caution.
This study provides insights into post-LT outcomes of patients with PSC by comparing those with ESLD and those without ESLD before LT. The association between the development of non-anastomotic biliary strictures and a high MELD score before LT has been described before, even in patients without PSC[16,19,24]. Previous studies did not agree on whether this association resulted from hepatic dysfunction or a more complicated operative course with an increased risk of ischemia-reperfusion injury that subsequently contributes to rPSC. Hildebrand et al[19] suggested that a higher MELD score may be associated with a more aggressive disease course. Given that patients with PSC with more advanced liver disease are at higher risk of rPSC, these findings could potentially favor early listing of patients with PSC.
In this study, careful patient selection was performed, using signs of decompensated liver disease and a combination of two scoring systems (Child-Pugh and MELD) to differentiate patients with hepatic dysfunction. Therefore, hypoalbuminemia, ascites, and hepatic encephalopathy were included in the selection criteria. Based on our results, the presence of ESLD before LT had no significant impact on rPSC rate, graft failure, or patient survival (Figure 1). However, Visseren et al[25] demonstrated that patients indicated for LT due to recurrent cholangitis were at increased risk of recurrent disease. Although their study methods differed only slightly from ours, the results are not entirely consistent.
CMV infection is a common infectious complication after LT[26]. Based on our results, CMV infection after LT was a significant independent risk factor for rPSC. Consistent with our findings, Egawa et al[16] reported CMV infection as a significant risk factor for biliary strictures and rPSC in a small cohort of patients. A newer study by Dekkers et al[27] demonstrated an association between intercurrent CMV viremia and rPSC. The underlying mechanism of biliary damage remains unclear, and several hypotheses have been proposed. Gotthardt et al[28] detected CMV DNA in bile and suggested that chronic CMV infection of the bile ducts contributes to the development of non-anastomotic biliary strictures. It can be hypothesized that the tissue damage caused by CMV infection of liver grafts could reactivate an immune response against bile duct epithelial cells in genetically predisposed individuals. Other infections, particularly hepatotropic viruses, may also elicit an immune response; however, these were not evaluated in this study due to the higher risk of bias associated with missing data.
Multiple studies have demonstrated that ACR, particularly multiple episodes of ACR, significantly increases the risk of rPSC[10,15,17,20,21]. In addition, Uemura et al[29] reported an increased incidence of ACR in patients with PSC. Our findings are consistent with these reports, demonstrating that multiple episodes of ACR were a significant risk factor for rPSC. Moreover, we showed that multiple episodes of ACR were also associated with graft failure, a finding that was confirmed in the subgroup analysis of patients with rPSC only. The pathogenesis of biliary stricture formation in patients with ACR is not fully understood. It has been suggested that ACR causes damage to the biliary epithelium, thereby triggering immune-mediated injury directed against newly exposed antigens[20].
Prolonged post-LT hospitalization was associated with an increased risk of rPSC and graft failure. However, this association may be attributable to early post-transplant events, such as ACR, CMV infection, other infectious complications, or surgical complications, which could negatively influence long-term outcomes. In the rPSC subgroup, Roux-en-Y biliary anastomosis was associated with worse graft outcomes, which could be related to the endoscopic inaccessibility of this reconstruction and its potential negative effects on the bile ducts.
Many other risk factors for rPSC have been previously reported, such as the presence of IBD before LT[11,12,19,30], younger age at LT[7,12,20], male recipient[30], cyclosporine-based immunosuppression[7], younger age at diagnosis of PSC[6], steroid-resistant ACR[10,15], and the protective effect of colectomy before LT[6,9]. None of these associations were confirmed in our cohort. However, many of the studies included larger cohorts, and it must be emphasized that very few pre-LT colectomies were performed in our center compared with others. As PSC is a rare disease, additional studies, especially those involving larger cohorts, are required to confirm the potential risk factors for rPSC and graft failure.
The limitations of this study were its retrospective design and the previous lack of routine magnetic resonance cholangiopancreatography, colonoscopy, and liver biopsy in the post-transplant follow-up. Moreover, the study presents a cohort with a long-term follow-up period, during which different immunosuppressants were used. Due to the retrospective design, several potential factors associated with rPSC, such as the microbiome, bile acid composition, or IBD activity, could not be evaluated. Nevertheless, as our transplant center performs more than 90% of LTs in the Czech Republic, this study largely represents the national PSC transplant population. This study is an extension of a previously published study of patients with PSC who underwent LT by applying stricter inclusion and exclusion criteria[31]. We aimed to present a larger cohort and to reevaluate the risk factors of rPSC and the outcomes of carefully selected groups based on their indication for LT.
Through this study, we compared the outcomes of patients with PSC and ESLD and those without ESLD, using clearly defined criteria to distinguish between the two groups. ESLD was not found to be a risk factor for rPSC, graft failure, or mortality. The prevalence of rPSC in this carefully screened cohort was higher than that reported in multicenter studies and was frequently observed even in asymptomatic patients. CMV infection and ACR were significant risk factors for rPSC and graft failure. PSC recurrence is a serious complication associated with increased mortality and morbidity in LT recipients, which has attracted increasing attention from clinicians and researchers in recent years. Prospective studies are needed to confirm our results and to better understand the pathogenesis of this condition.
| 1. | Chapman RW, Arborgh BA, Rhodes JM, Summerfield JA, Dick R, Scheuer PJ, Sherlock S. Primary sclerosing cholangitis: a review of its clinical features, cholangiography, and hepatic histology. Gut. 1980;21:870-877. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 574] [Cited by in RCA: 517] [Article Influence: 11.2] [Reference Citation Analysis (1)] |
| 2. | Thorpe ME, Scheuer PJ, Sherlock S. Primary sclerosing cholangitis, the biliary tree, and ulcerative colitis. Gut. 1967;8:435-448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 133] [Cited by in RCA: 125] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 3. | Wee A, Ludwig J, Coffey RJ Jr, LaRusso NF, Wiesner RH. Hepatobiliary carcinoma associated with primary sclerosing cholangitis and chronic ulcerative colitis. Hum Pathol. 1985;16:719-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 120] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 4. | Karlsen TH, Folseraas T, Thorburn D, Vesterhus M. Primary sclerosing cholangitis - a comprehensive review. J Hepatol. 2017;67:1298-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 758] [Cited by in RCA: 655] [Article Influence: 72.8] [Reference Citation Analysis (2)] |
| 5. | LaRusso NF, Shneider BL, Black D, Gores GJ, James SP, Doo E, Hoofnagle JH. Primary sclerosing cholangitis: summary of a workshop. Hepatology. 2006;44:746-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 168] [Article Influence: 8.4] [Reference Citation Analysis (1)] |
| 6. | Lindström L, Jørgensen KK, Boberg KM, Castedal M, Rasmussen A, Rostved AA, Isoniemi H, Bottai M, Bergquist A. Risk factors and prognosis for recurrent primary sclerosing cholangitis after liver transplantation: a Nordic Multicentre Study. Scand J Gastroenterol. 2018;53:297-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (1)] |
| 7. | Ravikumar R, Tsochatzis E, Jose S, Allison M, Athale A, Creamer F, Gunson B, Iyer V, Madanur M, Manas D, Monaco A, Mirza D, Owen N, Roberts K, Sen G, Srinivasan P, Wigmore S, Fusai G, Fernando B, Burroughs A. Risk factors for recurrent primary sclerosing cholangitis after liver transplantation. J Hepatol. 2015;63:1139-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 116] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 8. | Gautam M, Cheruvattath R, Balan V. Recurrence of autoimmune liver disease after liver transplantation: a systematic review. Liver Transpl. 2006;12:1813-1824. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 171] [Article Influence: 8.6] [Reference Citation Analysis (2)] |
| 9. | Alabraba E, Nightingale P, Gunson B, Hubscher S, Olliff S, Mirza D, Neuberger J. A re-evaluation of the risk factors for the recurrence of primary sclerosing cholangitis in liver allografts. Liver Transpl. 2009;15:330-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 209] [Article Influence: 12.3] [Reference Citation Analysis (2)] |
| 10. | Alexander J, Lord JD, Yeh MM, Cuevas C, Bakthavatsalam R, Kowdley KV. Risk factors for recurrence of primary sclerosing cholangitis after liver transplantation. Liver Transpl. 2008;14:245-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 99] [Article Influence: 5.5] [Reference Citation Analysis (2)] |
| 11. | Cholongitas E, Shusang V, Papatheodoridis GV, Marelli L, Manousou P, Rolando N, Patch D, Rolles K, Davidson B, Burroughs AK. Risk factors for recurrence of primary sclerosing cholangitis after liver transplantation. Liver Transpl. 2008;14:138-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 125] [Article Influence: 6.9] [Reference Citation Analysis (1)] |
| 12. | Steenstraten IC, Sebib Korkmaz K, Trivedi PJ, Inderson A, van Hoek B, Rodriguez Girondo MDM, Maljaars PWJ. Systematic review with meta-analysis: risk factors for recurrent primary sclerosing cholangitis after liver transplantation. Aliment Pharmacol Ther. 2019;49:636-643. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 99] [Article Influence: 14.1] [Reference Citation Analysis (1)] |
| 13. | Graziadei IW, Wiesner RH, Batts KP, Marotta PJ, LaRusso NF, Porayko MK, Hay JE, Gores GJ, Charlton MR, Ludwig J, Poterucha JJ, Steers JL, Krom RA. Recurrence of primary sclerosing cholangitis following liver transplantation. Hepatology. 1999;29:1050-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 235] [Article Influence: 8.7] [Reference Citation Analysis (3)] |
| 14. | Demetris AJ, Bellamy C, Hübscher SG, O'Leary J, Randhawa PS, Feng S, Neil D, Colvin RB, McCaughan G, Fung JJ, Del Bello A, Reinholt FP, Haga H, Adeyi O, Czaja AJ, Schiano T, Fiel MI, Smith ML, Sebagh M, Tanigawa RY, Yilmaz F, Alexander G, Baiocchi L, Balasubramanian M, Batal I, Bhan AK, Bucuvalas J, Cerski CTS, Charlotte F, de Vera ME, ElMonayeri M, Fontes P, Furth EE, Gouw ASH, Hafezi-Bakhtiari S, Hart J, Honsova E, Ismail W, Itoh T, Jhala NC, Khettry U, Klintmalm GB, Knechtle S, Koshiba T, Kozlowski T, Lassman CR, Lerut J, Levitsky J, Licini L, Liotta R, Mazariegos G, Minervini MI, Misdraji J, Mohanakumar T, Mölne J, Nasser I, Neuberger J, O'Neil M, Pappo O, Petrovic L, Ruiz P, Sağol Ö, Sanchez Fueyo A, Sasatomi E, Shaked A, Shiller M, Shimizu T, Sis B, Sonzogni A, Stevenson HL, Thung SN, Tisone G, Tsamandas AC, Wernerson A, Wu T, Zeevi A, Zen Y. 2016 Comprehensive Update of the Banff Working Group on Liver Allograft Pathology: Introduction of Antibody-Mediated Rejection. Am J Transplant. 2016;16:2816-2835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 571] [Cited by in RCA: 472] [Article Influence: 47.2] [Reference Citation Analysis (2)] |
| 15. | Brandsaeter B, Schrumpf E, Bentdal O, Brabrand K, Smith HJ, Abildgaard A, Clausen OP, Bjoro K. Recurrent primary sclerosing cholangitis after liver transplantation: a magnetic resonance cholangiography study with analyses of predictive factors. Liver Transpl. 2005;11:1361-1369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 69] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 16. | Egawa H, Ueda Y, Ichida T, Teramukai S, Nakanuma Y, Onishi S, Tsubouchi H. Risk factors for recurrence of primary sclerosing cholangitis after living donor liver transplantation in Japanese registry. Am J Transplant. 2011;11:518-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 17. | Gelley F, Zádori G, Görög D, Kóbori L, Fehérvári I, Gámán G, Gerlei Z, Nagy P, Sárváry E, Nemes B. Recurrence of primary sclerosing cholangitis after liver transplantation - The Hungarian experience. Interv Med Appl Sci. 2014;6:16-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 18. | Gordon FD, Goldberg DS, Goodrich NP, Lok AS, Verna EC, Selzner N, Stravitz RT, Merion RM. Recurrent primary sclerosing cholangitis in the Adult-to-Adult Living Donor Liver Transplantation Cohort Study: Comparison of risk factors between living and deceased donor recipients. Liver Transpl. 2016;22:1214-1222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 74] [Cited by in RCA: 60] [Article Influence: 6.0] [Reference Citation Analysis (3)] |
| 19. | Hildebrand T, Pannicke N, Dechene A, Gotthardt DN, Kirchner G, Reiter FP, Sterneck M, Herzer K, Lenzen H, Rupp C, Barg-Hock H, de Leuw P, Teufel A, Zimmer V, Lammert F, Sarrazin C, Spengler U, Rust C, Manns MP, Strassburg CP, Schramm C, Weismüller TJ; German PSC Study Group. Biliary strictures and recurrence after liver transplantation for primary sclerosing cholangitis: A retrospective multicenter analysis. Liver Transpl. 2016;22:42-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 114] [Article Influence: 11.4] [Reference Citation Analysis (2)] |
| 20. | Jeyarajah DR, Netto GJ, Lee SP, Testa G, Abbasoglu O, Husberg BS, Levy MF, Goldstein RM, Gonwa TA, Tillery GW, Crippin JS, Klintmalm GB. Recurrent primary sclerosing cholangitis after orthotopic liver transplantation: is chronic rejection part of the disease process? Transplantation. 1998;66:1300-1306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 90] [Article Influence: 3.2] [Reference Citation Analysis (2)] |
| 21. | Moncrief KJ, Savu A, Ma MM, Bain VG, Wong WW, Tandon P. The natural history of inflammatory bowel disease and primary sclerosing cholangitis after liver transplantation--a single-centre experience. Can J Gastroenterol. 2010;24:40-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 71] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 22. | Visseren T, Erler NS, Polak WG, Adam R, Karam V, Vondran FWR, Ericzon BG, Thorburn D, IJzermans JNM, Paul A, van der Heide F, Taimr P, Nemec P, Pirenne J, Romagnoli R, Metselaar HJ, Darwish Murad S; European Liver and Intestine Transplantation Association (ELITA). Recurrence of primary sclerosing cholangitis after liver transplantation - analysing the European Liver Transplant Registry and beyond. Transpl Int. 2021;34:1455-1467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 57] [Cited by in RCA: 55] [Article Influence: 11.0] [Reference Citation Analysis (1)] |
| 23. | Ricciuto A, Kamath BM, Hirschfield GM, Trivedi PJ. Primary sclerosing cholangitis and overlap features of autoimmune hepatitis: A coming of age or an age-ist problem? J Hepatol. 2023;79:567-575. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (1)] |
| 24. | Howell JA, Gow PJ, Angus PW, Jones RM, Wang BZ, Bailey M, Fink MA. Early-onset versus late-onset nonanastomotic biliary strictures post liver transplantation: risk factors reflect different pathogenesis. Transpl Int. 2012;25:765-775. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 25. | Visseren T, Erler NS, Heimbach JK, Eaton JE, Selzner N, Gulamhusein A, van der Heide F, Porte RJ, van Hoek B, Alwayn IPJ, Metselaar HJ, IJzermans JNM, Darwish Murad S. Inflammatory conditions play a role in recurrence of PSC after liver transplantation: An international multicentre study. JHEP Rep. 2022;4:100599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 26. | Yadav SK, Saigal S, Choudhary NS, Saha S, Kumar N, Soin AS. Cytomegalovirus Infection in Liver Transplant Recipients: Current Approach to Diagnosis and Management. J Clin Exp Hepatol. 2017;7:144-151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 29] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 27. | Dekkers N, Westerouen van Meeteren M, Wolterbeek R, Farina Sarasqueta A, Laleman W, Inderson A, Desschans B, van Hoek B, Sebib Korkmaz K, Vermeire S, Maljaars J. Does mucosal inflammation drive recurrence of primary sclerosing cholangitis in liver transplantion recipients with ulcerative colitis? Dig Liver Dis. 2020;52:528-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 28. | Gotthardt DN, Senft J, Sauer P, Weiss KH, Flechtenmacher C, Eckerle I, Schaefer Y, Schirmacher P, Stremmel W, Schemmer P, Schnitzler P. Occult cytomegalovirus cholangitis as a potential cause of cholestatic complications after orthotopic liver transplantation? A study of cytomegalovirus DNA in bile. Liver Transpl. 2013;19:1142-1150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 29. | Uemura T, Ikegami T, Sanchez EQ, Jennings LW, Narasimhan G, McKenna GJ, Randall HB, Chinnakotla S, Levy MF, Goldstein RM, Klintmalm GB. Late acute rejection after liver transplantation impacts patient survival. Clin Transplant. 2008;22:316-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 67] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 30. | Vera A, Moledina S, Gunson B, Hubscher S, Mirza D, Olliff S, Neuberger J. Risk factors for recurrence of primary sclerosing cholangitis of liver allograft. Lancet. 2002;360:1943-1944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 136] [Article Influence: 5.7] [Reference Citation Analysis (1)] |
| 31. | Bajer L, Slavcev A, Macinga P, Sticova E, Brezina J, Roder M, Janousek R, Trunecka P, Spicak J, Drastich P. Risk of recurrence of primary sclerosing cholangitis after liver transplantation is associated with de novo inflammatory bowel disease. World J Gastroenterol. 2018;24:4939-4949. [PubMed] [DOI] [Full Text] |