Published online Mar 26, 2026. doi: 10.4252/wjsc.v18.i3.115363
Revised: November 23, 2025
Accepted: January 26, 2026
Published online: March 26, 2026
Processing time: 161 Days and 4.5 Hours
The global prevalence of type 2 diabetes mellitus (T2DM) is increasing. Although globular adiponectin (gAd) shows potential in improving islet function, its clinical application is limited by rapid clearance. Given the promising prospects of adipose-derived mesenchymal stem cell exosomes (Exos) in targeted therapy, whether this nanocarrier can enhance gAd’s efficacy in improving islet function warrants significant research attention.
To develop a new synergistic therapeutic strategy based on adipose-derived mesenchymal stem cells Exos loaded with gAd (gAd-Exo).
A T2DM rat model was established using a high-fat diet and streptozotocin. Rats were randomized into control, T2DM, T2DM + gAd, T2DM + Exo, and T2DM + gAd-Exo groups, receiving respective treatments via tail vein injection for four weeks. Pancreatic tissues were subjected to histological, immunohistochemical, and biochemical analyses. Meanwhile in vitro experiments assessed the protective effects of gAd-Exo on palmitic acid-injured INS-1 cells.
gAd-Exo treatment significantly ameliorated hyperglycemia, improved pancreatic islet morphology, and reduced β-cell apoptosis compared to other groups. It enhanced insulin sensitivity and down-regulated glucagon expression. Mechanistically, gAd-Exo activated the AMP-activated protein kinase/acetyl-CoA carboxylase signaling pathway. In vitro, gAd-Exo superiorly mitigated palmitic acid-induced oxidative stress and apoptosis in INS-1 cells.
This study shows that the combination of gAd and Exo produced a significant synergistic effect. gAd-Exo can relieve type 2 diabetes by reducing blood glucose, improving hyperinsulinaemia and islet function, and at the same time reducing islet β cells apoptosis. It may be achieved by activating the AMP-activated protein kinase/acetyl-CoA carboxylase pathway. The discovery provides a new strategy with synergistic regenerative potential for diabetes treatment.
Core Tip: Researchers have developed a promising new approach to treat type 2 diabetes using exosomes (Exos) derived from adipose-derived mesenchymal stem cells loaded with globular adiponectin (gAd) - referred to as gAd-Exo. The combination of gAd and Exo produced a significant synergistic effect. In a diabetic rat model, gAd-Exo significantly lowered blood glucose, reduced pancreatic islet cell death, and improved insulin sensitivity. It also helped restore the structure and function of pancreatic islets. Mechanistically, gAd-Exo activated key metabolic pathway - AMP-activated protein kinase/acetyl-CoA carboxylase - which are crucial for energy metabolism and cell survival. This study highlights the potential of Exo-mediated delivery to enhance the efficacy of therapeutic molecules like gAd, offering a targeted and sustained treatment strategy for type 2 diabetes.
- Citation: Wang AR, Zhang YY, Liu YJ, Ren LN, Yan QH, Wang KF, Wang Y. Exosomes of adipose-derived mesenchymal stem cells loaded with globular adiponectin improve islet function for type 2 diabetes. World J Stem Cells 2026; 18(3): 115363
- URL: https://www.wjgnet.com/1948-0210/full/v18/i3/115363.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i3.115363
Diabetes mellitus, a hyperglycemic metabolic disorder, is defined by inadequate insulin secretion or diminished sensitivity to insulin[1]. The global prevalence of type 2 diabetes mellitus (T2DM) is on the rise, with estimates indicating a potential surge to 693 million individuals by 2045[2,3]. Elevated blood glucose levels worsen pancreatic cell damage, impacting T2DM progression. Therefore, preserving pancreatic cell mass and function is crucial for restoring blood glucose control, a key aspect of T2DM management treatment. Presently, oral medications are the mainstay of T2DM treatment[4]. However, their ability to regulate blood glucose is temporary, often compromised by high water solubility or rapid clearance post-metabolism.
Exosomes (Exos) are nanoscale vesicles secreted by cells that exhibit low immunogenicity and possess distinct functions. They serve as effective drug carriers for the delivery of therapeutic agents[5,6] and can also function as therapeutic agents for specific diseases[7,8]. As a result, there has been a marked increase in research focused on Exos and immune cells. Investigations have demonstrated that Exos possess considerable therapeutic capabilities through transporting cargo to target cells and modulating various signaling pathways[9,10]. Numerous studies have shown that Exos derived from adipose-derived mesenchymal stem cells (ADSC-Exo) can enhance diabetes outcomes[11-13]. In experimental diabetes models, adipose-derived mesenchymal stem cells (ADSCs) have been observed to mitigate transplant rejection, facilitate beta cell regeneration, preserve residual β-cell mass, enhance insulin sensitivity, and counteract insulin resistance by suppressing stress-induced serine kinases[14]. These results underscore the significant therapeutic promise of Exos in managing diabetes.
Adiponectin, primarily secreted by adipocytes, is a circulating plasma protein with diverse biological functions, such as enhancing insulin sensitivity, combating oxidative stress, and preventing atherosclerosis[15]. The active site of adiponectin, known as the globular adiponectin (gAd), exhibits various adiponectin-related effects, including ameliorating insulin resistance, inhibiting apoptosis, and providing protection to the liver and pancreas[16,17]. Despite its beneficial effects, adiponectin has limitations in enhancing islet function. While adiponectin levels are typically high under normal physiological conditions, they are notably reduced in individuals with diabetes. The core functional domain of adiponectin, gAd, is widely distributed in the body; however, its receptors on pancreatic β-cells are relatively scarce. Moreover, gAd is prone to recognition and clearance by macrophages during systemic circulation, resulting in reduced bioavailability in pancreatic tissue and limited therapeutic efficacy[18]. In contrast, Exos characterized by lipid bilayers, effectively shield their cargo from environmental degradation, ensuring sustained release effects[19]. None
Building on the aforementioned background, we prepared gAd-Exo and established a rat model of diabetes. Subse
Adipose tissue was aseptically harvested from euthanized SD rats, washed with phosphate buffered saline (PBS) (Solarbio, Beijing, China) minced, and digested in 0.25% collagenase I (GIBCO, Thermo Fisher Scientific, MA, United States) at 37 °C for 1 hour. The resulting digest underwent filtration, centrifugation, and the pellet was subsequently washed and resuspended. Cells were seeded at 1 × 106/mL in T25 flasks and cultured at 37 °C with 5% CO2. ADSCs were identified by morphology and flow cytometry for CD44 and CD90 (Affinity, Cincinnati, OH, United States).
ADSCs were cultured in medium for one day. The supernatant was collected and subjected to sequential centrifugation at 2000 × g and 10000 × g to eliminate debris. Subsequently, ultracentrifugation was performed at 100000 × g (Hitachi, Japan). After discarding the supernatant, this process was repeated to isolate the Exos.
The Exos suspension was applied to a copper grid for 1 minute, then blotted. After adding uranyl acetate solution, it precipitates again. They were dried at room temperature and Exos morphology was examined by transmission electron microscopy (Hitachi, Japan).
Exos size and concentration were measured using a PMX120 instrument (Particle Metrix, Germany) and ZetaViewEV software via nanoparticle tracking analysis after diluting samples in PBS.
The proteins were separated using sodium-dodecyl sulfate gel electrophoresis and subsequently transferred onto polyvinylidene fluoride membranes. Skim milk powder was used for blocking and incubated with specific primary antibodies at 4 °C for 16 hours. Following this, the bands were exposed to the secondary antibody conjugated with HRP at room temperature for 1.5 hours. Protein bands were visualized using luminescence, and their expression levels were assessed through grayscale analysis. The experiment was replicated thrice. The antibodies targeting the protein of interest are listed below: CD9 (Affinity, Cincinnati, OH, United States); CD63 (Affinity, Cincinnati, OH, United States); AMP-activated protein kinase (AMPK) mouse monoclonal antibody (Bioss, Beijing, China); phosphorylated-AMPK antibody (p-AMPK) (Abmart, Shanghai, China); acetyl-CoA carboxylase (ACC) monoclonal antibody (Proteintech, Wuhan, Hubei Province, China); phospho-ACC antibody (p-ACC) (Proteintech, Wuhan, Hubei Province, China); beta actin monoclonal antibody (Proteintech, Wuhan, Hubei Province, China); and the dilution ratio is 1:1000; ECL luminescent liquid (Abbkine, Wuhan, Hubei Province, China).
Exos were co-incubated with gAd at 25 °C for 30 minutes, followed by ultracentrifugation and enzyme-linked immuno
gAd was synthesized from Jiangsu Pubo Biotechnology Co., Ltd. Palmitic acid (PA) and streptozotocin (STZ) was obtained from MedChemExpress (MCE, NJ, United States). Cell viability was determined using a Cell Counting Kit-8 (Abbkine, Wuhan, Hubei Province, China). DAPI was purchased from Solarbio (Beijing, China). The following assay kits from Beyotime (Shanghai, China) were used according to the manufacturer’s instructions: Reactive oxygen species assay kit; mitochondrial membrane potential assay kit with JC-1; and Annexin V-FITC apoptosis detection kit; TUNEL reagent.
INS-1 cells were obtained from BaiDi Biotechnology Co., Ltd. Cells were cultured in RPMI-1640 medium (Procell, Wuhan, Hubei Province, China) under 5% CO2. They were supplemented with 10% fetal bovine serum (CellMax, Beijing, China). A PA-induced lipotoxicity model was established by treating cells with 0-550 μmol/L PA for 18 hours. To assess the protective effects, cells at 75% confluence were divided into five groups: Control, model, gAd, Exo, and gAdExo.
Approximately 5 × 104 cells/well were seeded overnight. After 18 hours of drug treatment, the cells were incubated with 10 μmol/L H2DCFDA for 20 minutes, washed with PBS, and imaged using fluorescence microscopy.
The mitochondrial membrane potential was assessed using JC-1. After seeding 5 × 104 cells/well and treating with drugs for 18 hours, cells were stained with JC-1, followed by DAPI, washed, and examined under a fluorescence microscope.
After seeding 5 × 104 cells/well overnight and treating for 18 hours, cells were stained with Annexin V-FITC and PI in binding buffer (1:2:39) for 30 minutes. Following washes and DAPI staining, samples were observed by fluorescence microscopy.
SD rats were obtained from Beijing Specific Bio-technology Co. Ltd and maintained under standard laboratory conditions. They were acclimated to a 12-hour light-dark cycle, with each phase lasting 12 hours, and were provided ad libitum access to food and water to aid their adaptation to the new environment. Thirty SD rats were acclimatized and randomized into five groups: Control, T2DM, T2DM + Exo, T2DM + gAd, and T2DM + gAd-Exo. After 8 weeks of high-fat diet, T2DM was induced by STZ (60 mg/kg) in diabetic groups, and controls received citrate buffer. Within 7 days of STZ administration, rats were given 10% sucrose water[20]. Rats with fasting glucose > 11.1 mmol/L after 3 days were regarded as a modeling success. They were injected daily for 4 weeks with PBS, Exo, gAd or gAd-Exo. Each rat was injected with Exos with 5 × 109 particles each time a day[21-23]. 2.5 μg of gAd was injected daily[24], and the contents of gAd and Exo in gAd-Exo were consistent with the above.
Blood glucose, body weight, length, and abdominal circumference were measured weekly. Serum samples collected before and after STZ intervention were analyzed for insulin using a rat enzyme-linked immunosorbent assay kit (Cloud-Clone Corp, Wuhan, Hubei Province, China).
Different drug formulations were labeled with DiR and subsequently administered via tail vein injection. The in vivo distribution of groups was tracked by imaging at 30 minutes, 60 minutes, and 120 minutes post-injection (n = 3 rats/group).
Pancreatic tissues were collected after 4 weeks of intervention, fixed in formalin, and paraffin-embedded. Sections (5 μm) were baked, deparaffinized through xylene and graded ethanol, then subjected to hematoxylin and eosin staining. Finally, these were observed through fluorescence microscopy.
Pancreatic paraffin sections were mounted on polylysine-coated slides, dried at 37 °C, and deparaffinized using xylene and graded ethanol. After antigen retrieval and blocking, the sections were incubated with primary antibodies (Insulin Antibody and Glucagon Mouse Monoclonal Antibody, Affinity, Cincinnati, OH, United States), followed by a secondary antibody, SP reaction, DAB development, hematoxylin counterstaining, dehydration, clearing, and mounting for microscopic examination.
The method is similar to the immunohistochemistry steps. The antibodies used are as follows: CD63 (Santa Cruz, 1:200, TX, United States), DyLight 488 secondary antibody (Boster, 1:100, Wuhan, Hubei Province, China).
Rat pancreas paraffin sections underwent deparaffinization and were treated with TUNEL reagent (TdT enzyme to fluorescent labeling solution ratio of 1:9) at 37 °C for 1 hour in the absence of light. Following PBS rinses, DAPI was used to stain the nuclei. The sections were then mounted using anti-fade medium, and apoptosis was observed through fluorescence microscopy.
Total RNA was extracted from pancreatic tissue using the Trizol method and reverse transcribed into cDNA according to the reverse transcription kit (Mei5bio, Beijing, China). Primer sequences were synthesized by Sangon Biotech (Table 1). A fluorescent dye was incorporated into the real-time quantitative polymerase chain reaction system during its preparation. A total reaction volume of 10 μL was prepared, and polymerase chain reaction assays were subsequently performed using Mei5bio’s PCR kit. Cycle threshold (Ct) values were determined, and the relative expression level of the target gene was quantified using β-actin as the reference gene.
| Primer ID | 5’-3’ |
| β-actin-F | ACCCAGATCATGTTTGAGACCT |
| β-actin-R | GACCAGAGGCATACAGGGACAAC |
| AMPK-F | CAGGAAGATTGTACGCAGGC |
| AMPK-R | AAAAGAGTTGGCACGTGGTC |
| PI3K-F | CTTCTGCCTTACGGTTGCATC |
| PI3K-R | CCTTGAGCCAGTGATTCAGG |
| ACC-F | GCGGCTCTGGAGGTATATGT |
| ACC-R | TTAGCGTGGGGATGTTCCCT |
| AKT-F | CAGGCACCCCTTCCTTACAG |
| AKT-R | GGTACACCACATCCGTCGAG |
| U6-F | CTCGCTTCGGCAGCACA |
| U6-R | AACGCTTCACGAATTTGCGT |
| miR-21-F | CGGCTAGCTTATCAGACTGA |
| miR-21-R | GTGCAGGGTCCGAGGT |
| RT primer | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACTCAACA |
All data are expressed as mean ± SEM. The data was initially assessed using the Shapiro-Wilk test. Parametric testing methods were subsequently employed for normally distributed data: The independent sample test for comparing two groups, one-way analysis of variance for comparing multiple groups, and the Bonferroni method for post hoc testing. The data analysis of this study was performed using GraphPad Prism 9.2 software. The threshold for statistical significance was set at P < 0.05.
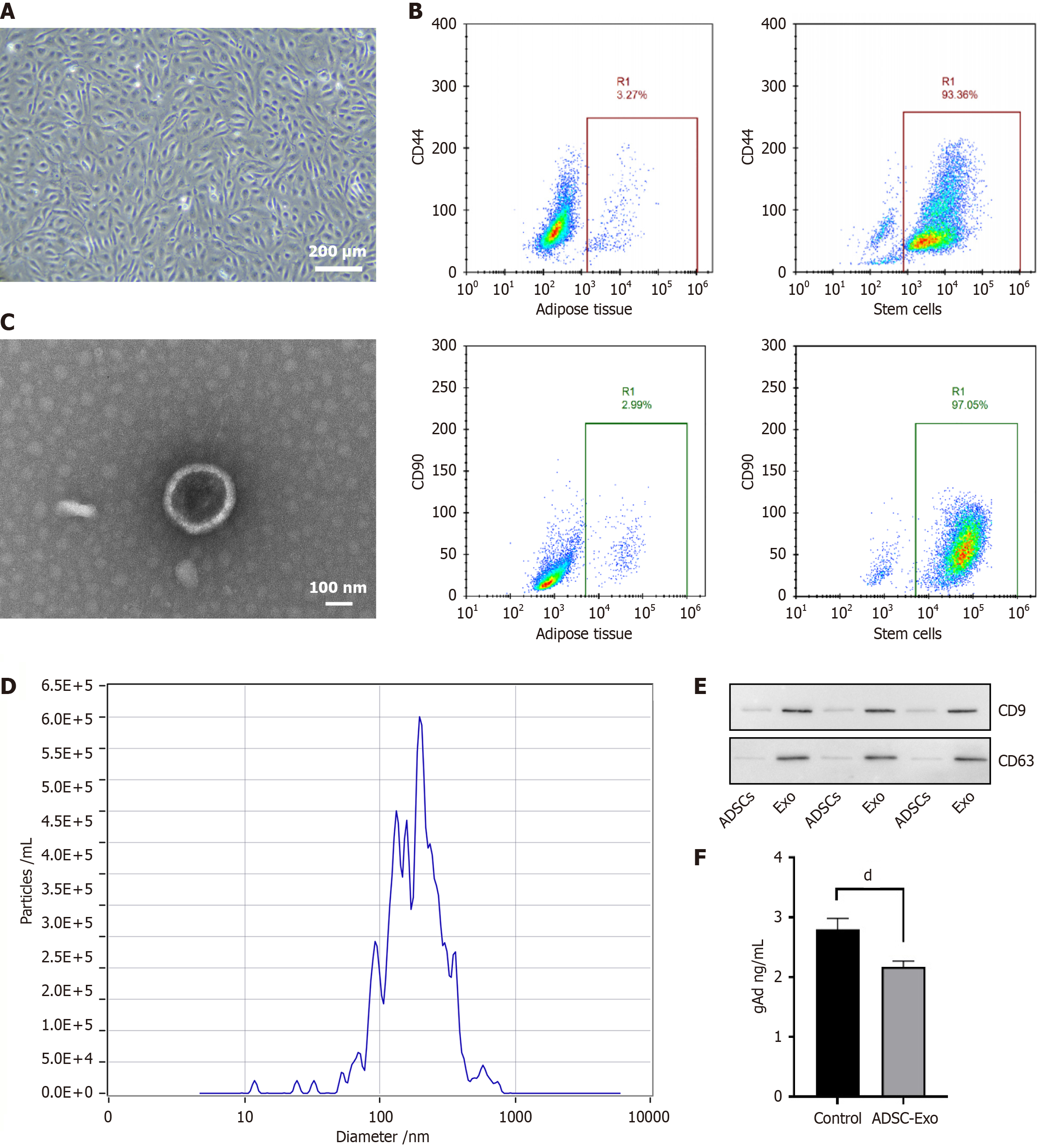
The extracted and cultured ADSCs exhibited a classical fibroblastic morphology (Figure 1A). We identified ADSCs. Flow cytometry results showed that almost all the ADSCs expressed CD44 (93.36%) and CD90 (97.05%) (Figure 1B). Under transmission electron microscopy, the ADSC-Exo exhibited a typical double-layered membranous Exos structure (Figure 1C). Nanoparticle tracking analysis results revealed the diameters of ADSC-Exo were ranging from 100-200 nm (Figure 1D). Positive expressions of CD9 and CD63 in the ADSC-Exo were detected by western blotting (Figure 1E). These results indicate that the Exo isolation was successful.
Exos were co-incubated with gAd and collected by ultracentrifugation. The concentration of gAd in the Exo super
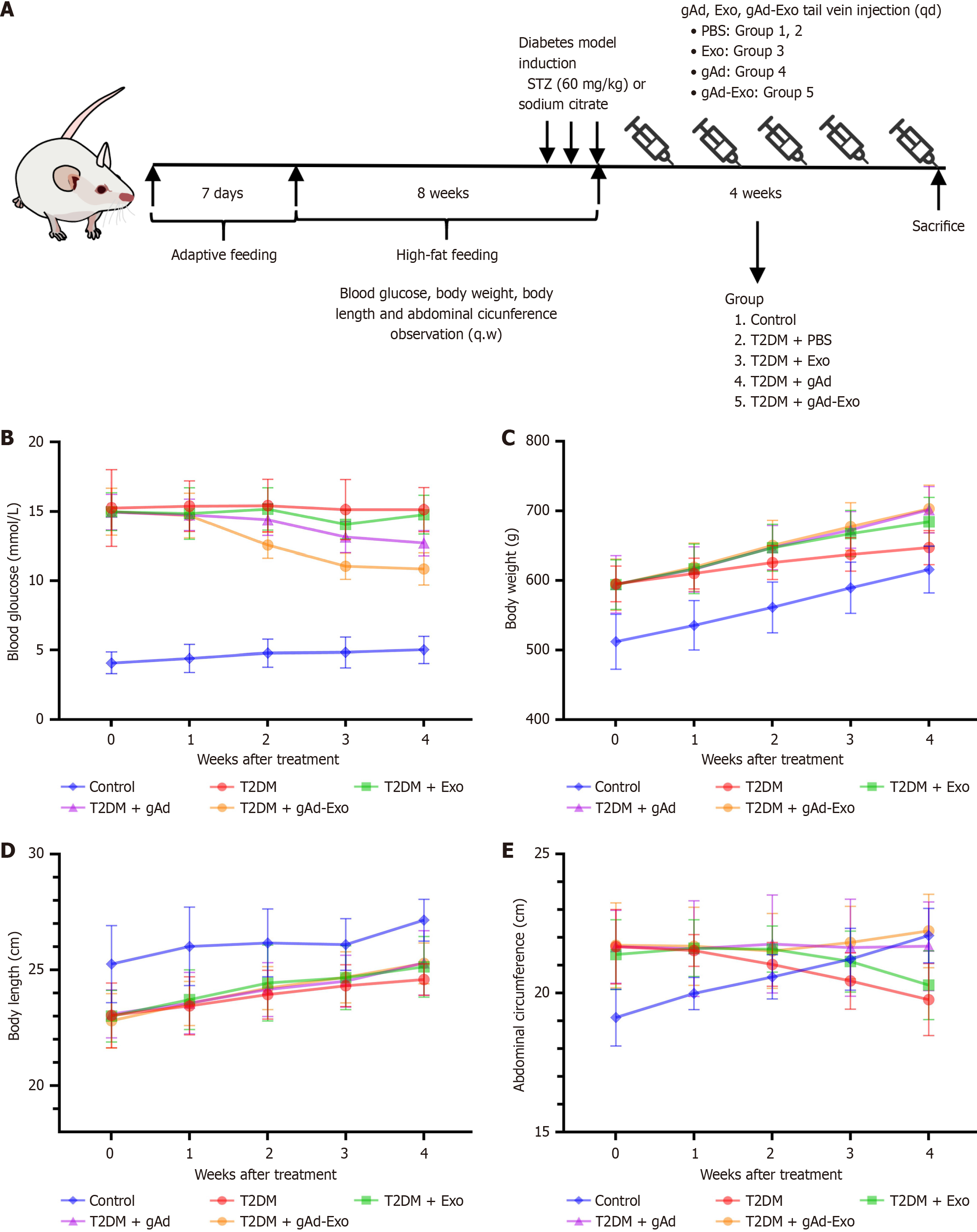
Figure 2A is the scheme of study design. To elucidate the potential therapeutic role of Exo and gAd in STZ-induced diabetic rats, blood glucose, body weight, body length, and abdominal circumference were determined. Blood glucose levels were significantly decreased in the gAd and gAd-Exo groups compared to the T2DM group (P < 0.05) (Figure 2B). Moreover, the reduction in blood glucose was more substantial in the gAd-Exo group than in the Exo group (P < 0.01). Administration of gAd alone or in conjunction with Exo led to a rise in body weight in comparison to untreated diabetic rats (Figure 2C). The body weight of the Exo group did not exhibit a significant variance from that of the T2DM group. The body length growth in the T2DM group appeared to be slower than that in the gAd group post-intervention but did not exhibit a significant difference (Figure 2D). After 4 weeks of intervention, the abdominal circumference of the gAd-Exo group significantly exceeded that of the T2DM group. The abdominal circumference of both the gAd and Exo groups did not significantly differ from that of the T2DM group (Figure 2E).
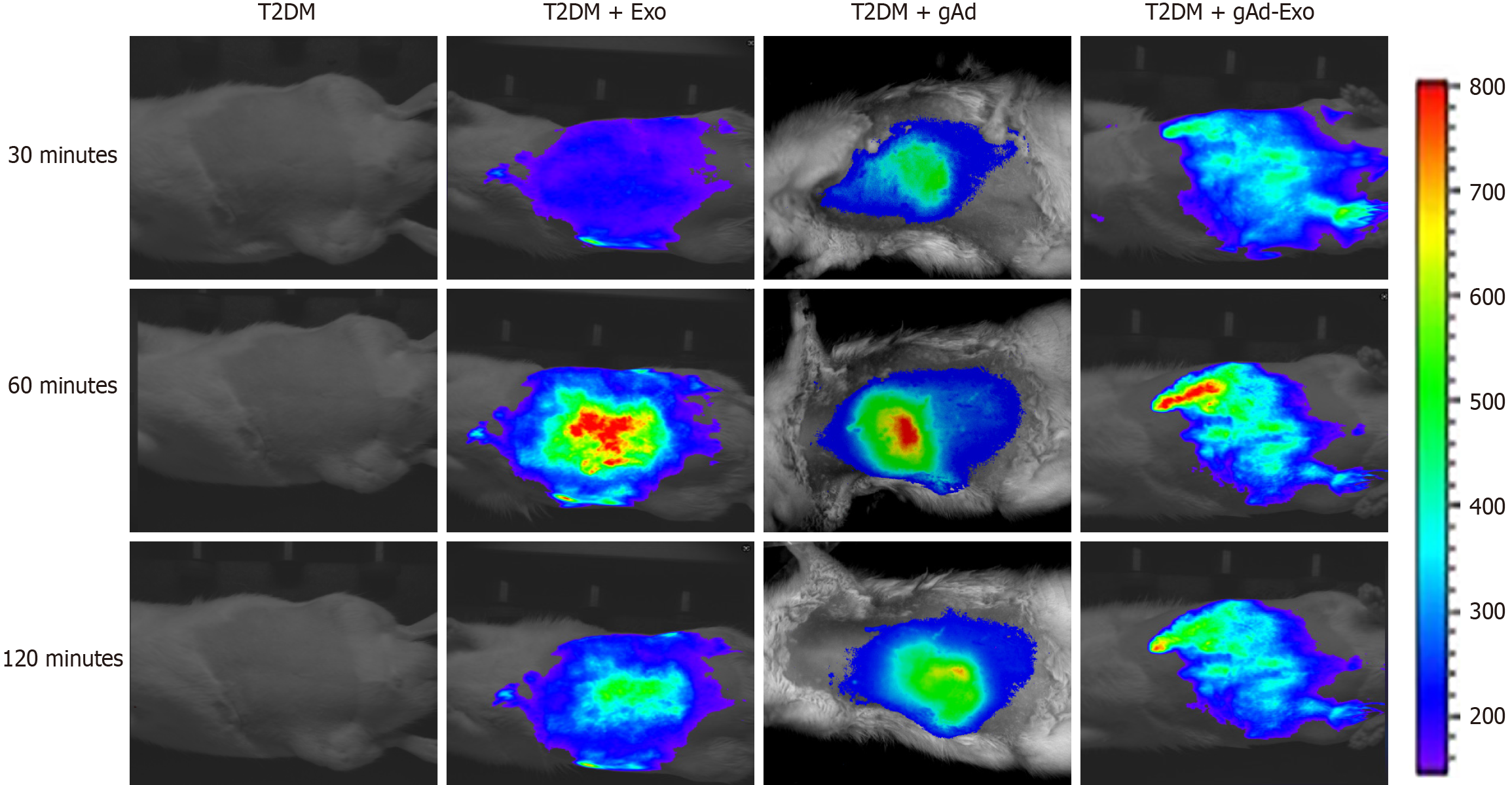
Figure 3 shows the in vivo fluorescent images of T2DM, Exo and gAd-Exo in rats of different groups. There was no fluorescent signal in the T2DM group. Fluorescent signals-appeared in the abdomen of rats 30 minutes after tail vein injection of Exo, gAd or gAd-Exo, and the fluorescence intensity was the strongest at 60 minutes and weakened at 120 minutes. The distribution of Exo and gAd-Exo in rats. In addition, immunofluorescence staining was performed on the pancreas. The expression of Exo marker CD63 in the Exo group (P < 0.0001) and the gAd-Exo group (P = 0.0079) was significantly higher than that in the gAd group (Supplementary Figure 2).
Histological examination revealed pronounced loss of lobules and cell boundaries, vacuolar degeneration, fibrosis, extensive structural impairment of islets, and a notable decrease in islet quantity in the diabetic cohort compared to the non-diabetic cohort. The islet architecture was also compromised in the Exo and gAd cohorts, albeit to a lesser degree than the T2DM cohort. In the gAd-Exo cohort, the islets remained structurally impaired, albeit less so than in the T2DM, Exo, and gAd cohorts, with an increased number of cells within the islets (Figure 4A).
Insulin expression was notably lower in the control group compared to the T2DM group (P = 0.0004). In the gAd, Exo, and gAd-Exo groups, insulin expression was down-regulated relative to the T2DM group, with the most significant decrease observed in the gAd-Exo group compared to the other two groups (Figure 4B and C). Glucagon expression was markedly reduced in the control group in comparison to the T2DM group. In the gAd, Exo, and gAd-Exo groups, glu
Before the intervention, the experimental groups exhibited significantly higher serum insulin levels compared to the control group, but there were no significant differences in serum insulin levels among the experimental groups (Figure 4F). Following the intervention, the gAd, Exo, and gAd-Exo groups showed significantly lower serum insulin levels compared to the T2DM group, with the gAd-Exo group displaying significantly lower serum insulin levels than the Exo and gAd groups (Figure 4F). Furthermore, we implemented the HOMA-IR formula, computed as fasting glucose (mmol/L) × fasting insulin (μU/mL)/22.5. This calculation revealed a notable enhancement in systemic insulin sensitivity among diabetic rats treated with gAd-Exo (P < 0.05) (Figure 4G).
The rate of apoptosis in pancreatic cells was notably lower in the control group compared to the T2DM, Exo and gAd groups. The apoptosis rate of pancreatic cells in the gAd-Exo group was significantly lower than that in the T2DM, Exo and gAd groups, and was not significantly different from that of the control group (Figure 4H and I).
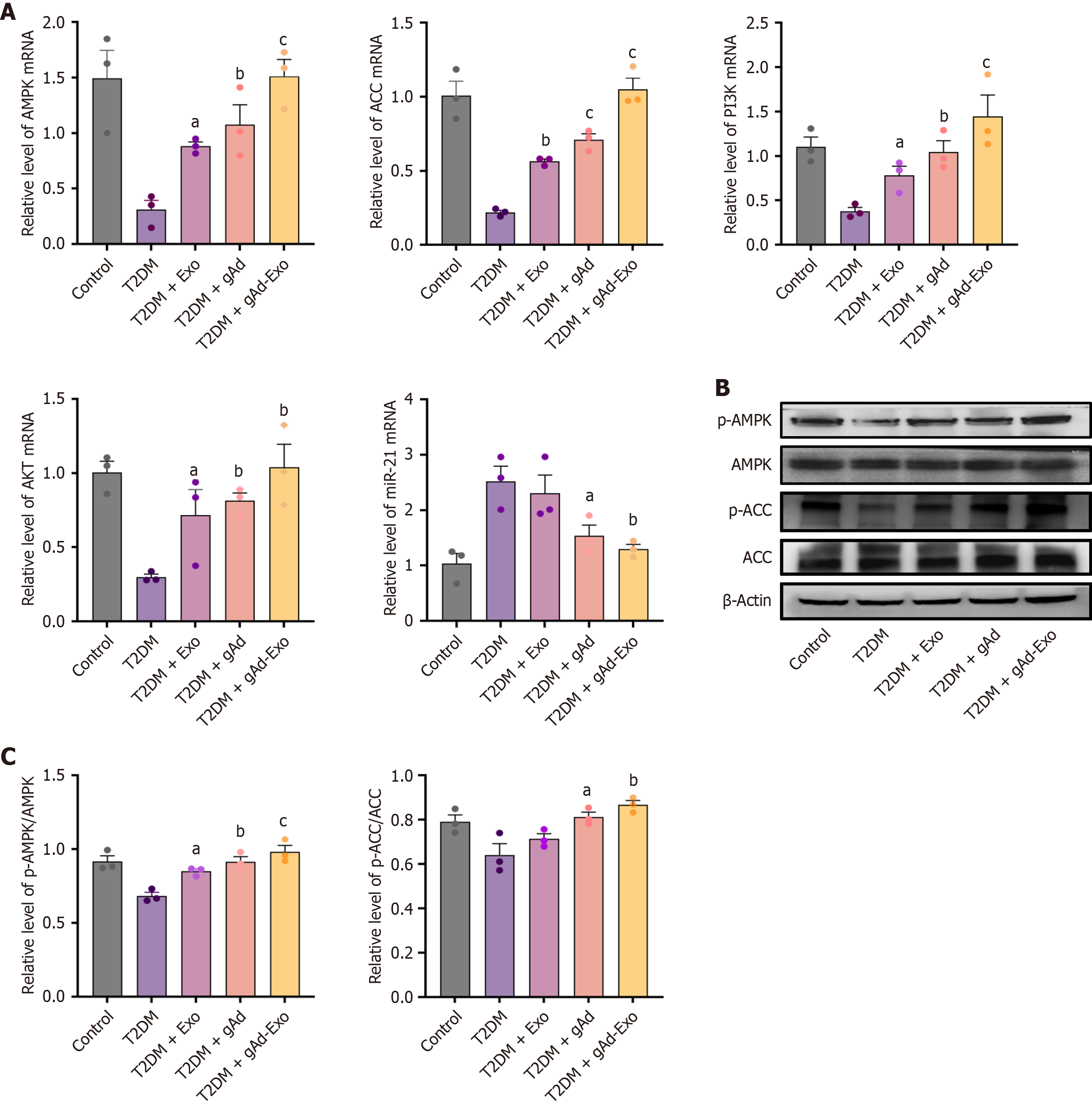
The genes expression levels of AMPK, ACC, phosphatidylinositol 3-kinase (PI3K), protein kinase B (AKT), and miRNA-21 in pancreatic tissue were assessed using fluorescent real-time quantitative polymerase chain reaction. As shown in Figure 5A, the mRNA levels of AMPK, PI3K, AKT, and ACC were notably diminished (P < 0.05), indicating a success of diabetes model. In contrast to the diabetic group, the levels of AMPK, PI3K, AKT, and ACC were elevated in the gAd group alone or in combination with gAd and Exo, with a significant increase in gAd-Exo expression (P < 0.05), indicating that the beneficial impact of gAd-Exo on pancreatic cells. miR-21 is a highly expressed microRNA in Exos. Compared with the gAd injection alone group, the Exo injection group showed higher miR-21 expression, indicating that Exos targeted the pancreas.
Protein levels of AMPK, p-AMPK, ACC and p-ACC in pancreatic tissue were analyzed western blotting. In rats with T2DM, levels of AMPK phosphorylation were found to be reduced; however, this reduction was mitigated by treatments with gAd, Exo, and gAd-Exo. Furthermore, ACC phosphorylation was assessed to gauge the impact of these treatments on insulin resistance. Both gAd and gAd-Exo-treated rats exhibited improved insulin sensitivity, characterized by an elevated p-ACC/ACC ratio (Figure 5B and C).
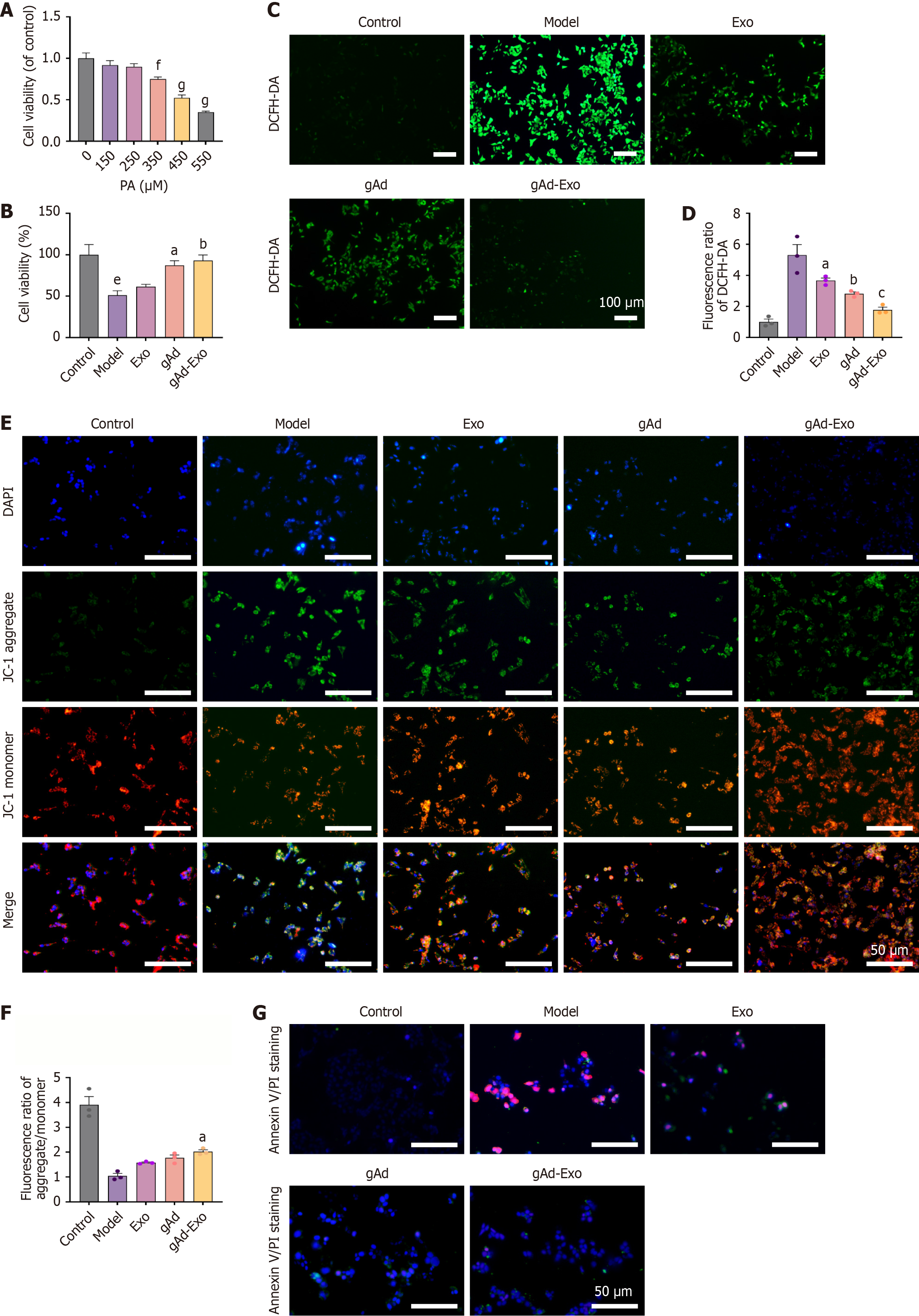
The optimal treatment concentration of PA was identified from a range of concentrations (Figure 6A). Subsequently, pancreatic β cells were treated with 350 μmol/L PA to establish an in vitro T2DM model. Results indicated increased cell viability rates in the gAd and Exo monotherapy groups compared to the model group, with the gAd-Exo combination demonstrating the most pronounced enhancement in cell viability (P = 0.0053) (Figure 6B). Moreover, the DCFH-DA expression in gAd-Exo significantly decreased (P = 0.0002) (Figure 6C and D). Compared to the gAd or Exo alone groups, the gAd-Exo group exhibited a notable decrease in mitochondrial membrane potential depolarization (P = 0.0144) (Figure 6E and F). Additionally, gAd-Exo demonstrated improved anti-apoptotic effects (Figure 6G). These results align with those observed in vivo drug administration model in diabetic rats.
Insulin resistance and pancreatic cell injury are the two main characteristics of T2DM[25]. Pancreatic cell injury, characterized by a loss of functional β-cell mass, results in insufficient insulin secretion and consequent hyperglycemia. Therefore, strategies aimed at restoring functional β-cell mass are a central focus in developing new therapies for T2DM.
gAd, a potential therapeutic agent for T2DM, can enhance insulin sensitivity and mitigate inflammation and oxidative stress, and enhance energy metabolism, thereby alleviating pancreatic injury. However, its poor target specificity and rapid clearance in vivo limit the actual efficacy of the therapy in vivo. In this study, using the innovative intervention of gAd-Exo, apoptosis and oxidative stress of pancreatic islet β cells were alleviated both in vivo and in vitro, and islet function was improved, thus enhancing the therapeutic effect of gAd on T2DM.
Compared to existing drug delivery systems, Exos are a promising delivery vehicles. Due to the restriction of lipid biomolecules, Exos can provide a barrier to protect gAd from degradation. The blood-pancreas barrier between pan
Currently, mesenchymal stem cells hold significant potential in regenerative medicine, and their secreted Exos are used in the clinical treatment of various diseases[27-29]. Exos derived from human umbilical cords alleviate CCl4-induced liver inflammation and fibrosis in mice[30]. Exos can alleviate diabetes and possess the ability to regenerate pancreatic islets[23]. In vivo experiments confirmed the therapeutic effects of gAd-Exo on the quantity and function of β-cells. gAd-Exo exhibited significantly stronger anti-apoptotic effects in pancreatic cells compared to gAd and Exo. In addition, short-term T2DM experimental results indicated that gAd-Exo can significantly ameliorate the symptoms of T2DM, including weight, HOMA-IR and blood glucose. Hematoxylin and eosin and immunohistochemical staining results demonstrated notable resistance to pancreatic cell apoptosis, and an improvement in overall islet architecture.
The upregulation of AMPK phosphorylation and its downstream target ACC indicates that gAd-Exo exerts its beneficial effects, in part, by activating the AMPK/ACC signaling pathway. Activation of the AMPK/ACC axis plays a crucial role in regulating cellular energy balance and insulin sensitivity[31]. p-AMPK promotes glucose uptake and fatty acid oxidation while suppressing lipid synthesis, leading to decreased intracellular malonyl-CoA levels and alleviating its inhibition of carnitine palmitoyltransferase 1. This metabolic shift is essential for mitigating lipid-induced insulin resistance in peripheral tissues and safeguarding pancreatic β-cells against lipoapoptosis. Additionally, by enhancing cellular energy balance and reducing lipotoxicity-induced oxidative stress, AMPK activation may help maintain β-cell mitochondrial function, thereby inhibiting the intrinsic apoptotic pathway. This unified mechanism offers a comprehensive rationale for the enhancements in systemic insulin sensitivity and preservation of pancreatic islet integrity observed in vivo, as well as the mitigation of excessive reactive oxygen species production, mitochondrial depolarization, and apoptosis in INS-1 cells induced by PA in vitro.
Although current research indicates that gAd-Exo demonstrates therapeutic potential for T2DM, several limitations warrant attention. In the future, long-term studies must be conducted to assess the persistence of metabolic improve
This study shows that the combination of gAd and Exo produced a significant synergistic effect. The gAd-Exo can effectively improve glucose-lipid metabolism, lower blood glucose levels and reduce insulin resistance by affect the pancreatic AMPK/ACC pathway. It also inhibits pancreatic cell apoptosis and promotes the recovery and regeneration of pancreatic islets, with an overall effect that is significantly better than using either ingredient alone. This biological effect stems from the complementary functions of the two. Exo serves as a carrier to enhance the stability and targeting of gAd, while gAd confers a well-defined metabolic regulatory function to Exo. The discovery provides a new strategy with synergistic regenerative potential for diabetes treatment.
| 1. | Nyenwe EA, Jerkins TW, Umpierrez GE, Kitabchi AE. Management of type 2 diabetes: evolving strategies for the treatment of patients with type 2 diabetes. Metabolism. 2011;60:1-23. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 252] [Cited by in RCA: 221] [Article Influence: 14.7] [Reference Citation Analysis (1)] |
| 2. | King H, Aubert RE, Herman WH. Global burden of diabetes, 1995-2025: prevalence, numerical estimates, and projections. Diabetes Care. 1998;21:1414-1431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3875] [Cited by in RCA: 3241] [Article Influence: 115.8] [Reference Citation Analysis (1)] |
| 3. | Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8030] [Cited by in RCA: 6302] [Article Influence: 1575.5] [Reference Citation Analysis (12)] |
| 4. | Padhi S, Nayak AK, Behera A. Type II diabetes mellitus: a review on recent drug based therapeutics. Biomed Pharmacother. 2020;131:110708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 569] [Cited by in RCA: 353] [Article Influence: 58.8] [Reference Citation Analysis (1)] |
| 5. | Tan A, Rajadas J, Seifalian AM. Exosomes as nano-theranostic delivery platforms for gene therapy. Adv Drug Deliv Rev. 2013;65:357-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 201] [Article Influence: 15.5] [Reference Citation Analysis (1)] |
| 6. | Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5374] [Cited by in RCA: 4847] [Article Influence: 403.9] [Reference Citation Analysis (4)] |
| 7. | Mathieu M, Martin-Jaular L, Lavieu G, Théry C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3270] [Cited by in RCA: 2881] [Article Influence: 411.6] [Reference Citation Analysis (5)] |
| 8. | Chulpanova DS, Kitaeva KV, James V, Rizvanov AA, Solovyeva VV. Therapeutic Prospects of Extracellular Vesicles in Cancer Treatment. Front Immunol. 2018;9:1534. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 96] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 9. | Zöller M, Zhao K, Kutlu N, Bauer N, Provaznik J, Hackert T, Schnölzer M. Immunoregulatory Effects of Myeloid-Derived Suppressor Cell Exosomes in Mouse Model of Autoimmune Alopecia Areata. Front Immunol. 2018;9:1279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (1)] |
| 10. | Kamei N, Atesok K, Ochi M. The Use of Endothelial Progenitor Cells for the Regeneration of Musculoskeletal and Neural Tissues. Stem Cells Int. 2017;2017:1960804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (1)] |
| 11. | Harrell CR, Jovicic N, Djonov V, Arsenijevic N, Volarevic V. Mesenchymal Stem Cell-Derived Exosomes and Other Extracellular Vesicles as New Remedies in the Therapy of Inflammatory Diseases. Cells. 2019;8:1605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 687] [Cited by in RCA: 620] [Article Influence: 88.6] [Reference Citation Analysis (1)] |
| 12. | Shen Z, Huang W, Liu J, Tian J, Wang S, Rui K. Effects of Mesenchymal Stem Cell-Derived Exosomes on Autoimmune Diseases. Front Immunol. 2021;12:749192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 244] [Cited by in RCA: 221] [Article Influence: 44.2] [Reference Citation Analysis (3)] |
| 13. | Thomou T, Mori MA, Dreyfuss JM, Konishi M, Sakaguchi M, Wolfrum C, Rao TN, Winnay JN, Garcia-Martin R, Grinspoon SK, Gorden P, Kahn CR. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature. 2017;542:450-455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1006] [Cited by in RCA: 1233] [Article Influence: 137.0] [Reference Citation Analysis (1)] |
| 14. | Sanap A, Bhonde R, Joshi K. Conditioned medium of adipose derived Mesenchymal Stem Cells reverse insulin resistance through downregulation of stress induced serine kinases. Eur J Pharmacol. 2020;881:173215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 15. | Yanai H, Yoshida H. Beneficial Effects of Adiponectin on Glucose and Lipid Metabolism and Atherosclerotic Progression: Mechanisms and Perspectives. Int J Mol Sci. 2019;20:1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 168] [Cited by in RCA: 343] [Article Influence: 49.0] [Reference Citation Analysis (1)] |
| 16. | Rakatzi I, Mueller H, Ritzeler O, Tennagels N, Eckel J. Adiponectin counteracts cytokine- and fatty acid-induced apoptosis in the pancreatic beta-cell line INS-1. Diabetologia. 2004;47:249-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 147] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 17. | Zhao L, Fu Z, Wu J, Aylor KW, Barrett EJ, Cao W, Liu Z. Globular adiponectin ameliorates metabolic insulin resistance via AMPK-mediated restoration of microvascular insulin responses. J Physiol. 2015;593:4067-4079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 18. | Fang H, Judd RL. Adiponectin Regulation and Function. Compr Physiol. 2018;8:1031-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 409] [Cited by in RCA: 450] [Article Influence: 56.3] [Reference Citation Analysis (0)] |
| 19. | Mehdizadeh S, Mamaghani M, Hassanikia S, Pilehvar Y, Ertas YN. Exosome-powered neuropharmaceutics: unlocking the blood-brain barrier for next-gen therapies. J Nanobiotechnology. 2025;23:329. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 53] [Reference Citation Analysis (2)] |
| 20. | Hua YY, Zhang Y, Gong WW, Ding Y, Shen JR, Li H, Chen Y, Meng GL. Dihydromyricetin Improves Endothelial Dysfunction in Diabetic Mice via Oxidative Stress Inhibition in a SIRT3-Dependent Manner. Int J Mol Sci. 2020;21:6699. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 21. | Khereldin RM, Abouelela YS, Yasin NAE, Youssef FS, Abdelhameed MI, Tohamy AF, Rizk H, Daghash SM. Comparing the therapeutic influence of bone marrow Mesenchymal stem cells versus its derived exosomes against diabetic hepatopathy in rats. Exp Cell Res. 2025;447:114436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 22. | Krishnan I, Ling MTM, Ng MH, Law JX, Yusof MRM, Thangarajah T, Mahmood Z, Uda Zahli NI, Rajamanickam S, Subramani B, Lokanathan Y. Efficacy of Fetal Wharton's Jelly Mesenchymal Stem Cells-Derived Small Extracellular Vesicles in Metabolic Syndrome. Biomolecules. 2025;15:44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 23. | Sharma R, Kumari M, Mishra S, Chaudhary DK, Kumar A, Avni B, Tiwari S. Exosomes Secreted by Umbilical Cord Blood-Derived Mesenchymal Stem Cell Attenuate Diabetes in Mice. J Diabetes Res. 2021;2021:9534574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (1)] |
| 24. | Li X, Zhang D, Vatner DF, Goedeke L, Hirabara SM, Zhang Y, Perry RJ, Shulman GI. Mechanisms by which adiponectin reverses high fat diet-induced insulin resistance in mice. Proc Natl Acad Sci U S A. 2020;117:32584-32593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 110] [Article Influence: 18.3] [Reference Citation Analysis (1)] |
| 25. | Veelen A, Erazo-Tapia E, Oscarsson J, Schrauwen P. Type 2 diabetes subgroups and potential medication strategies in relation to effects on insulin resistance and beta-cell function: A step toward personalised diabetes treatment? Mol Metab. 2021;46:101158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (5)] |
| 26. | Shao J, Zaro J, Shen Y. Advances in Exosome-Based Drug Delivery and Tumor Targeting: From Tissue Distribution to Intracellular Fate. Int J Nanomedicine. 2020;15:9355-9371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 243] [Article Influence: 40.5] [Reference Citation Analysis (1)] |
| 27. | Mehryab F, Rabbani S, Shahhosseini S, Shekari F, Fatahi Y, Baharvand H, Haeri A. Exosomes as a next-generation drug delivery system: An update on drug loading approaches, characterization, and clinical application challenges. Acta Biomater. 2020;113:42-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 147] [Article Influence: 24.5] [Reference Citation Analysis (1)] |
| 28. | Jiao YR, Chen KX, Tang X, Tang YL, Yang HL, Yin YL, Li CJ. Exosomes derived from mesenchymal stem cells in diabetes and diabetic complications. Cell Death Dis. 2024;15:271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 57] [Article Influence: 28.5] [Reference Citation Analysis (4)] |
| 29. | Gao Y, Chen Q, Wu Z, Yuan L. Regulation of pancreatic β cells by exosomes from different sources. Diabetes Res Clin Pract. 2025;224:112222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 30. | Li T, Yan Y, Wang B, Qian H, Zhang X, Shen L, Wang M, Zhou Y, Zhu W, Li W, Xu W. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis. Stem Cells Dev. 2013;22:845-854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 761] [Cited by in RCA: 707] [Article Influence: 54.4] [Reference Citation Analysis (2)] |
| 31. | Wen JP, Liu CE, Hu YT, Chen G, Lin LX. Globular adiponectin regulates energy homeostasis through AMP-activated protein kinase-acetyl-CoA carboxylase (AMPK/ACC) pathway in the hypothalamus. Mol Cell Biochem. 2010;344:109-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |