Published online Mar 26, 2026. doi: 10.4252/wjsc.v18.i3.114746
Revised: November 7, 2025
Accepted: January 22, 2026
Published online: March 26, 2026
Processing time: 179 Days and 7.2 Hours
Mesenchymal stem cells (MSCs) and their secretome offer a promising approach for treating osteoradionecrosis of the jaw (ORNJ); however, their efficacy remains controversial and the underlying molecular mechanisms require elucidation.
To investigate the role of small extracellular vesicles derived from human am
A Sprague-Dawley rat model of ORNJ received local hAMSCs-sEVs after mandi
In vivo, hAMSCs-sEVs enhanced socket healing in ORNJ rats, with micro computed tomography showing increased bone volume (bone volume/total volume). Histology revealed mature bone formation and higher expression of collagen-1, ALP, CD31, and vascular endothelial growth factor in the hAMSCs-sEVs group vs controls. In vitro, 12 Gy irradiation significantly inhibited osteogenic differentiation in BMSCs and angiogenesis in HUVECs. hAMSCs-sEVs were internalized by irradiated cells and reversed these inhibitions in a concentration-dependent manner. A concentration of 1010 particles/mL significantly upregulated osteogenic markers (collagen-1, ALP, runt-related transcription factor 2, osteopontin) in BMSCs and angiogenic markers (vascular endothelial growth factor, CD31) in HUVECs. RNA sequencing revealed that hAMSCs-sEVs reactivated the phosphatidylinositol 3-kinase/protein kinase B pathway in BMSCs and the Wnt/β-catenin pathway in HUVECs, which were inhibited by radiation. These effects were blocked by pathway inhibitors Ly294002 and MSAB, respectively.
hAMSCs-sEVs promote the repair of ORNJ by partially reversing radiation-induced inhibition of osteogenesis and angiogenesis, potentially through reactivation of the phosphatidylinositol 3-kinase/protein kinase B and Wnt/β-catenin signaling pathways.
Core Tip: The small extracellular vesicles from human amniotic mesenchymal stem cells (hAMSCs-sEVs) enhanced tooth socket healing in rats with osteoradionecrosis of the jaw, increasing bone formation and expression of osteogenic (collagen-1, alkaline phosphatase) as well as angiogenic (CD31, vascular endothelial growth factor) markers. In vitro, hAMSCs-sEVs reversed the inhibition of osteogenesis and angiogenesis by reactivating the phosphatidylinositol 3-kinase/protein kinase B and Wnt/β-catenin signaling pathways, respectively, as confirmed by pathway inhibitors. These findings indicate that hAMSCs-sEVs facilitate osteoradionecrosis of the jaw bone healing by reversing radiation-impaired osteogenesis and angiogenesis via key signaling pathways.
- Citation: Zhou HF, Xu JM, Li J, Xu F, Li JF, Yu Y, Yuan H, Bian YF, Wang YL, Du YF. Human amniotic mesenchymal stem cell-derived small extracellular vesicles repair of jaw osteoradionecrosis. World J Stem Cells 2026; 18(3): 114746
- URL: https://www.wjgnet.com/1948-0210/full/v18/i3/114746.htm
- DOI: https://dx.doi.org/10.4252/wjsc.v18.i3.114746
Osteoradionecrosis of the jaw (ORNJ) is a serious complication of radiotherapy in approximately 5% of head and neck cancer patients[1,2]. It is clinically characterized by chronic pain, orocutaneous fistula, soft tissue ulceration, progressive bone destruction and even pathological fractures, substantially impairing patients’ health and quality of life[3,4]. Despite modern advances in radiotherapy techniques, ORNJ remains a significant challenge with few effective pharmacological or surgical interventions.
The precise pathogenic mechanisms of ORNJ are incompletely elucidated. Current hypotheses implicate multiple interrelated pathological processes, including vascular injury and tissue ischemia, fibrotic tissue response, immune suppression and local infection, dysregulated cell differentiation as well as hypoxia and reactive oxygen species accumulation[5]. Collectively, these pathological and biological changes result in a vicious cycle of vascular and osteocyte damage, chronic inflammation and aberrant bone remodeling, ultimately resulting in progressive ischemic necrosis of irradiated bone.
Mesenchymal stem cells (MSCs) based therapies represented a promising alternative for bone repair and regeneration[6]. Previous investigations including ours confirmed that human amniotic MSCs (hAMSCs) significantly enhanced the osteogenic differentiation of human bone marrow MSCs (BMSCs) and the angiogenic capacity of human umbilical vein endothelial cells (HUVECs)[6-10]. Furthermore, our studies demonstrated that hAMSCs could effectively counteract the suppression of osteogenic differentiation induced by oxidative stress and inflammatory stimuli[11,12]. Notably, hAMSCs exhibited pronounced immunomodulatory properties through the secretion of bioactive factors that induce polarization of M0 macrophages toward the M2 anti-inflammatory phenotype, thereby creating a favorable microenvironment for bone defect regeneration[13]. These multifunctional properties encompassing osteogenic promotion, angiogenic sti
hAMSCs were derived from amniotic membrane, BMSCs were collected from patients undergoing sagittal split ramus osteotomy and HUVECs were obtained from fresh umbilical cords. All cells were isolated independently and identified as previously reported[7,16]. The primary antibodies are shown in Supplementary Table 1. In order to verify the mul
The 3rd generation of hAMSCs was cultured in a 15 cm dish to 70%-80%, the culture medium was discarded and the cells were washed 3-5 times with phosphate buffered saline (PBS). The culture medium was then replaced with α-MEM medium [10% sEVs-free foetal bovine serum (FBS)] containing FBS without sEVs to 90%, and the cells were then cultured in serum-free medium for 36 hours. The culture medium supernatant was collected and gradient centrifugation was performed as follows: 300 × g at 4 ℃ for 10 minutes; 2000 × g at 4 ℃ for 10 minutes; and 10000 × g at 4 ℃ for 30 minutes. The cell debris was removed and the supernatant was collected and filtered using a 0.22 μm diameter filter. Finally, the supernatant was centrifuged at 4 ℃ for 140 minutes at 100000 × g, and the deposit was collected and resuspended in
Eight-week-old Sprague-Dawley (SD) male rats were supplied by the Animal Core Facility of Nanjing Medical University and housed under specific pathogen-free conditions at a temperature of 22 ± 1 ℃, relative humidity of 50% ± 1%, and a light/dark cycle of 12/12 hours with free access to food and water. All animal studies (including the rat euthanasia procedure) were performed in compliance with the regulations and guidelines of institutional animal care for Nanjing Medical University and conducted according to the IACUC guidelines (Approval No. IACUC-2310104). SD rats were exposed to 8 Gy/day for 5 days to establish the ORNJ model[19,20]. One week later, bilateral mandibular first molars were extracted under general anaesthesia induced by intraperitoneal injection of ketamine (100 mg/kg). SD rats were euthanized by cervical dislocation 7 days and 14 days post-extraction. Bilateral mandibles were collected and fixed with 4% paraformaldehyde for at least 24 hours. To evaluate the effect of hAMSCs-sEVs on bone healing of ORNJ, the rats were divided into three groups: Control, ORNJ, and ORNJ + sEVs. Rats in the sEVs group received a socket injection of hAMSCs-sEVs (1010/mL) after tooth extraction. Rats in the control and ORNJ groups received equivalent volumes of PBS.
Mandibles were harvested for micro computed tomography analysis (Skyscan 1176, Kontich, Belgium) at a voxel resolution of 18 μm, an energy of 50 kV and a current of 456 μA at 7 days and 14 days post-extraction. Three-dimensional reconstruction of the mandible was generated and analyzed using NRecon v1.6 and CTAn v1.13.8.1 software. The region of interest was defined to encompass the extraction sockets of the mandibular first molar teeth and bone volume fraction (%) of the region of interest was statistically calculated.
Mandible samples were harvested and fixed in 4% paraformaldehyde for 24 hours. Following decalcification in 10% ethylenediaminetetraacetic acid, the samples were embedded in paraffin and sections of 4-μm thickness were prepared for hematoxylin and eosin, Masson trichrome, and immunohistochemical (IHC) staining. For IHC staining, slides were treated with 3% hydrogen peroxide for 20 minutes, rinsed twice with PBS, and blocked with normal goat serum at 37 ℃ for 40 minutes. Primary antibodies were applied and incubated at 4 ℃ overnight. The slides were subsequently treated with appropriate secondary antibodies, followed by diaminobenzidine and hematoxylin counterstaining. Primary antibodies used for IHC analysis are shown in Supplementary Table 2. Positive areas were observed under the mic
hAMSCs-sEVs were incubated with the lipophilic dye CM-DiI 1 μM (Molecular Probes, MA, United States) in a 5% CO2 and 37 ℃ incubator for 5 minutes and then centrifuged for 60 minutes in 4 ℃ at 100000 × g to remove excess dye solution. BMSCs or HUVECs were co-cultured with CM-DiI labeled hAMSCs-sEVs for 4, 6, 8, 10, and 12 hours and fixed with 4% paraformaldehyde for 30 minutes. Cells were sealed at room temperature for 1 hour with 0.5% BSA and 0.2% Triton X-100 and then incubated with ghost pen cyclic peptide dye (Biyuntian Corp., Suzhou, Jiangsu Province, China) at room temperature for 30 minutes. Finally, the cell nucleus was stained with 4’,6-diamidino-2-phenylindole staining solution for 10 minutes and images were captured under an inverted fluorescence microscope (Olympus, Japan).
Cells (BMSCs/HUVECs with or without radiation treatment) were seeded at a density of 1000 cells per well into 96-well plates, with three replicates for each group. Treatments were applied after 12, 24, 48, 72 and 96 hours of incubation with or without hAMSCs-sEVs. Subsequently, the medium was replaced with 100 μL of fresh medium containing 10 μL CCK8 reagent (Biyuntian Corp., Shanghai, China). After 2 hours of incubation, optical density was measured at 450 nm using a microplate reader (ThermoFisher, MA, United States).
After 24-hour passaging, BMSCs (with or without radiation treatment) were incubated in osteogenic medium (100 nM dexamethasone, 50 mg/mL ascorbic acid, and 5 mmol/L β-glycerophosphate; Sigma, MO, United States) with or without hAMSCs-sEVs for 14-21 days. The cells were grown in a humidified 5% CO2 incubator at 37 ℃. The osteogenic medium was refreshed every 3 days. After 14 days of osteogenic induction, the cells were treated with the NBT/BCIP staining kit (Beyotime Institute of Biotechnology, China) for alkaline phosphatase (ALP) staining as previously described[20]. Calcium deposition was assessed using Alizarin Red S solution (Biyuntian Corp., Shanghai, China) after 21 days of os
Geltrex™ matrix was prepared as previously reported[16]. HUVECs (with or without radiation treatment) were starved overnight and then seeded onto a layer of Geltrex™ matrix and incubated at 37 ℃ for 30 minutes with extracellular matrix and 10% FBS and then replaced with medium with or without hAMSCs-sEVs at 37 ℃ for 24 hours. Three images per group were captured using an inverted microscope (Leica Microsystems, Germany) and quantified evaluation of tube formation was obtained by measuring the length and number of tube-like structures[22].
A Transwell assay was used to assess migration of HUVECs (with or without radiation treatment) using a Corning TransWell Chamber (Corning, NY, United States). This was followed by trypsinization, counting and incubation in
HUVECs (with or without radiation treatment) were incubated for 24 hours, after which the 6-well culture dish was scratched using a 200 μL sterile pipette tip, resulting in three separate wounds. Cells were then stimulated with or without hAMSCs-sEVs for 24 hours. Changes in wound width were measured using ImageJ software (NIH) at 0 hour and 24 hours time points.
Western blotting analysis was performed as previously described[23]. The primary antibodies are shown in Supplementary Table 3. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) or β-actin served as an internal control. Relative densitometry analysis was carried out using ImageJ software (NIH).
RNA isolation and cDNA transcription were performed using Trizol reagent (Invitrogen, NY, United States) and a reverse transcription kit (Applied Biosystems, CA, United States). Quantitative real-time polymerase chain reaction was conducted as previously reported[21]. The primer sequences used are listed in Supplementary Table 4. Human GAPDH was used as a reference for estimating the levels of mRNAs. The fold changes in gene expression were determined by the 2-ΔΔCt method.
HUVECs or BMSCs were cultured and set as the control, radiation and radiation + sEVs groups. Clean reads were aligned to the reference genome utilizing the HISAT2 aligner, and gene expression levels were quantified using featureCounts. Differential expression analyses between the control and radiation groups as well as the radiation and radiation + sEVs groups, were performed using the DESeq2 package in R, whereby genes exhibiting an adjusted P-value < 0.05 and |log2 fold change| > 1.2 were deemed differentially expressed.
Differentially expressed genes (DEGs) underwent annotation with Gene Ontology (GO) terms via the Database for Annotation, Visualization, and Integrated Discovery tool. Subsequent enrichment analysis of GO terms aimed to unveil significantly enriched biological processes, molecular functions, and cellular components. Additionally, DEGs were mapped to the Kyoto Encyclopedia of Genes and Genomes pathways utilizing the Kyoto Encyclopedia of Genes and Genomes Orthology-Based Annotation System tool, facilitating the identification of pathways closely associated with the DEGs.
All data are expressed as mean ± SD. Statistical analyses were performed using GraphPad Prism 8.0. Comparisons between two groups were made using the t test, and comparisons between three or more subgroups were statistically analyzed using one-way ANOVA and Tukey’s test. Statistical differences were considered significant at P < 0.05.
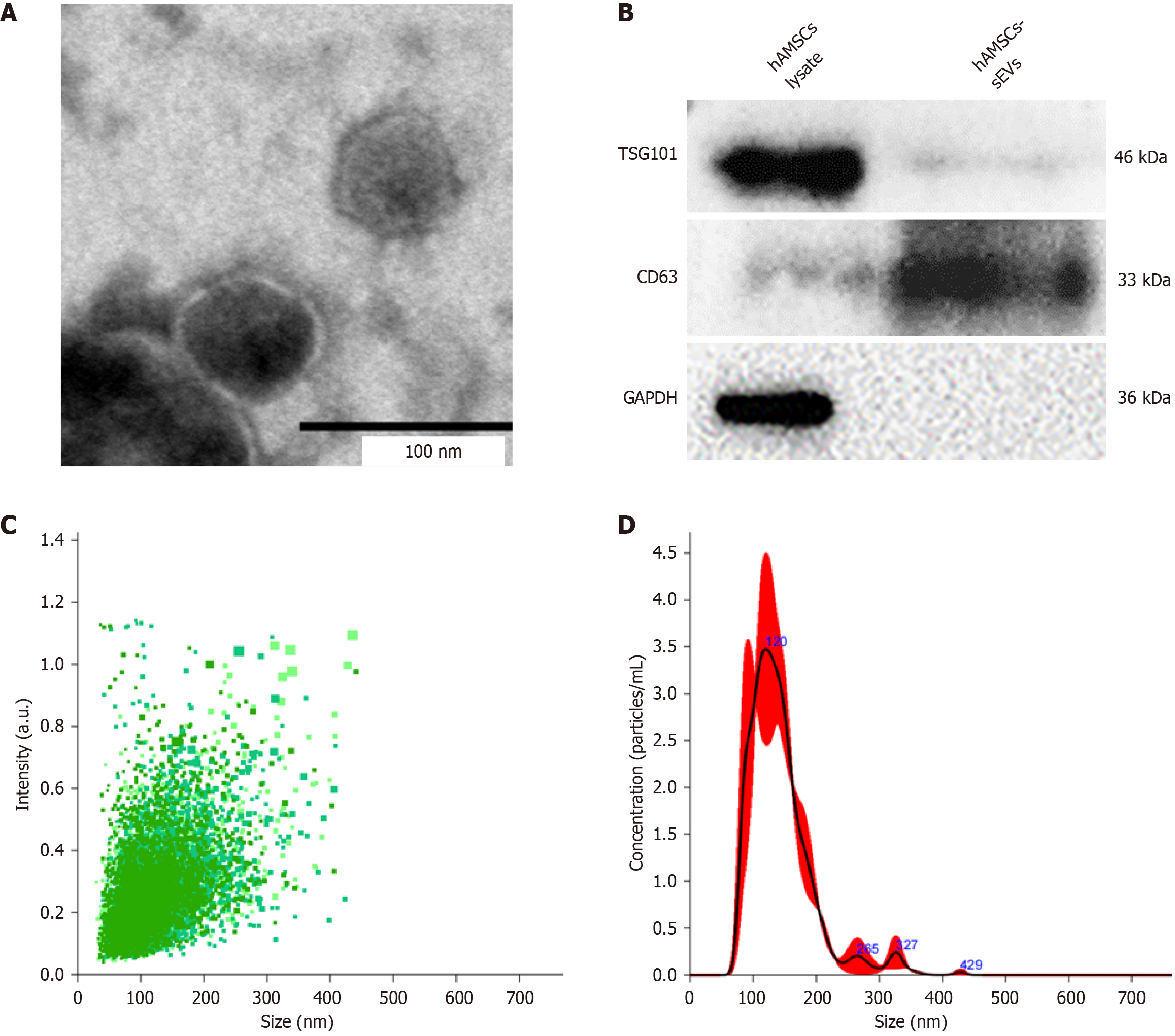
hAMSCs were obtained from the amniotic membrane and fluorescence-activated cell sorting showed that isolated 3rd generation hAMSCs were positive for the surface markers CD105, CD73 and CD90, but negative for CD14, CD45 and CD34 (Supplementary Figure 1A-H). The results of multidirectional differentiation proved that hAMSCs could differentiate into osteoblasts, adipocytes and chondrocytes (Supplementary Figure 1I-K). TEM observation revealed that hAMSCs-sEVs exhibited typical circular nanoparticles with a diameter ranging from 50 nm to 200 nm, forming a disk-like two-layer membrane structure (Figure 1A). The results of western blotting showed that compared with the cellular proteins of hAMSCs, hAMSCs-sEVs could express tumor susceptibility gene 101 and CD63, while GAPDH expression was negative (Figure 1B). The particle size distribution measured by nanoparticle tracking analysis was consistent with TEM observations, with an average particle size of 142.7 ± 6.1 nm and a peak particle size of 198.4 ± 8.6 nm. The actual concentration was 1.33 × 1011 ± 2.04 × 1010 particles/mL (Figure 1C and D).
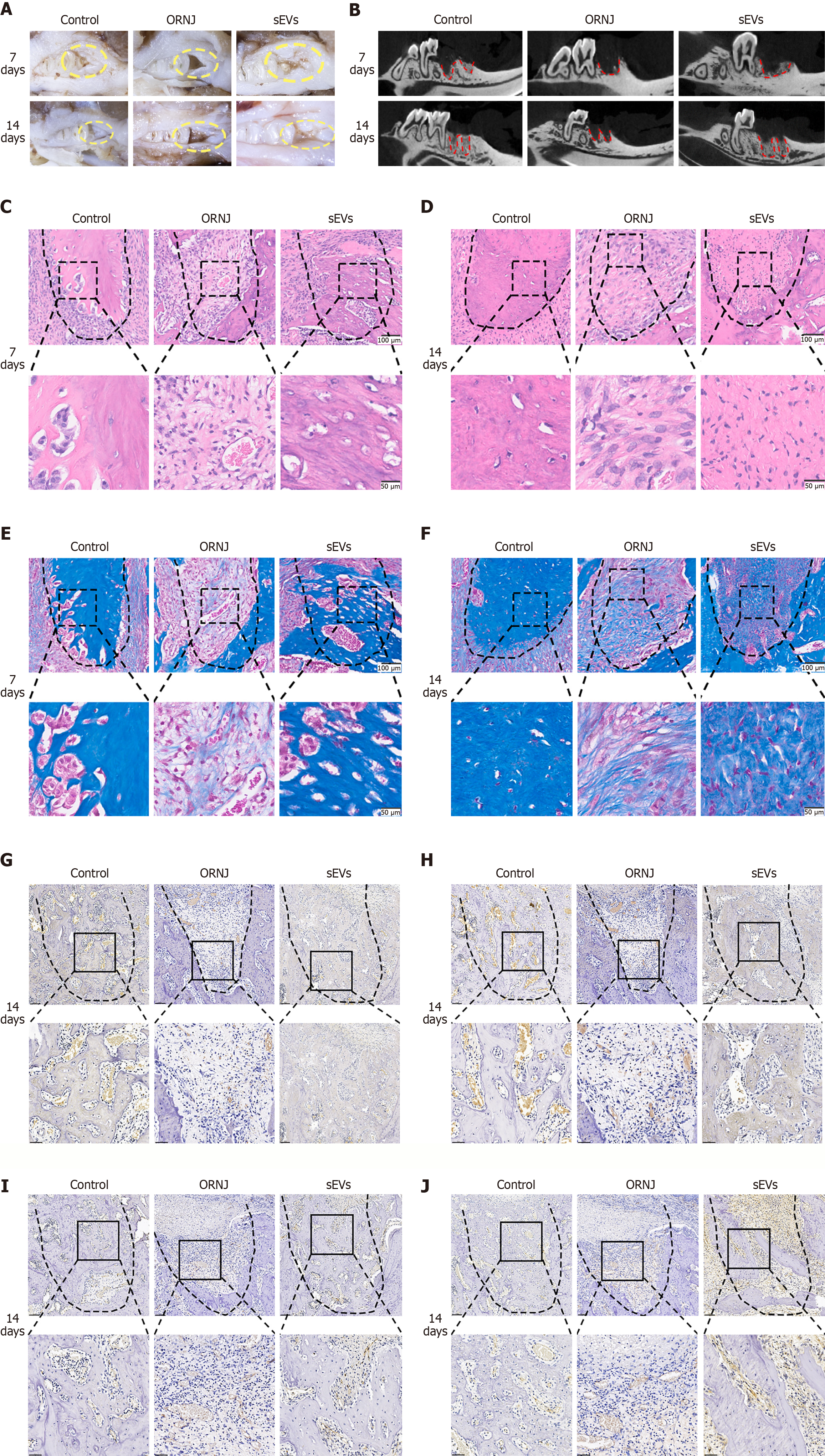
Compared to the control group, the 40 Gy group showed delayed or non-healing of the tooth socket post-extraction, while the 40 Gy + sEVs group presented with a closed healing tooth socket under microscopic observation (Figure 2A). Micro computed tomography showed new bone regeneration in the tooth socket at 7 days and 14 days post-extraction in the control and 40 Gy + sEVs groups, while less bone formation was observed in the 40 Gy group (Figure 2B). The bone volume fraction in the 40 Gy group at 14 days post-extraction was significantly reduced compared to that in the control and 40 Gy + sEVs groups (Supplementary Figure 2). Both hematoxylin and eosin and Masson staining showed mature bone matrix and trabecula in the tooth socket of the control and 40 Gy + sEVs groups, while a large amount of loose connective tissue with less mature bone trabecula was seen in the tooth socket in the 40 Gy group (Figure 2C-F). Furthermore, the results of IHC staining showed more positive staining of collagen-1 (COL-1), ALP, CD31 and vascular endothelial growth factor (VEGF) in the control and 40 Gy + sEVs groups in contrast to that in the 40 Gy group (Figure 2G-J).
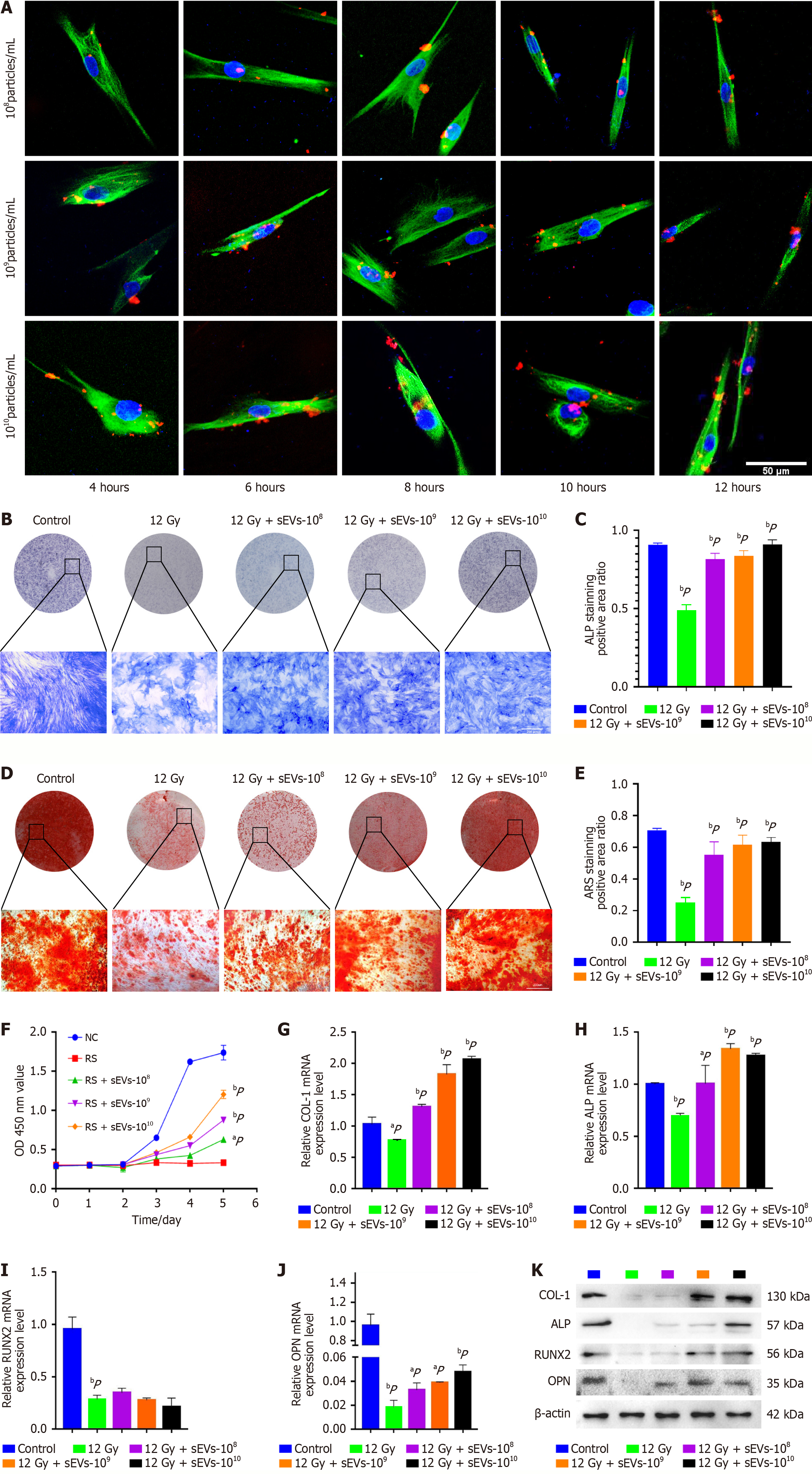
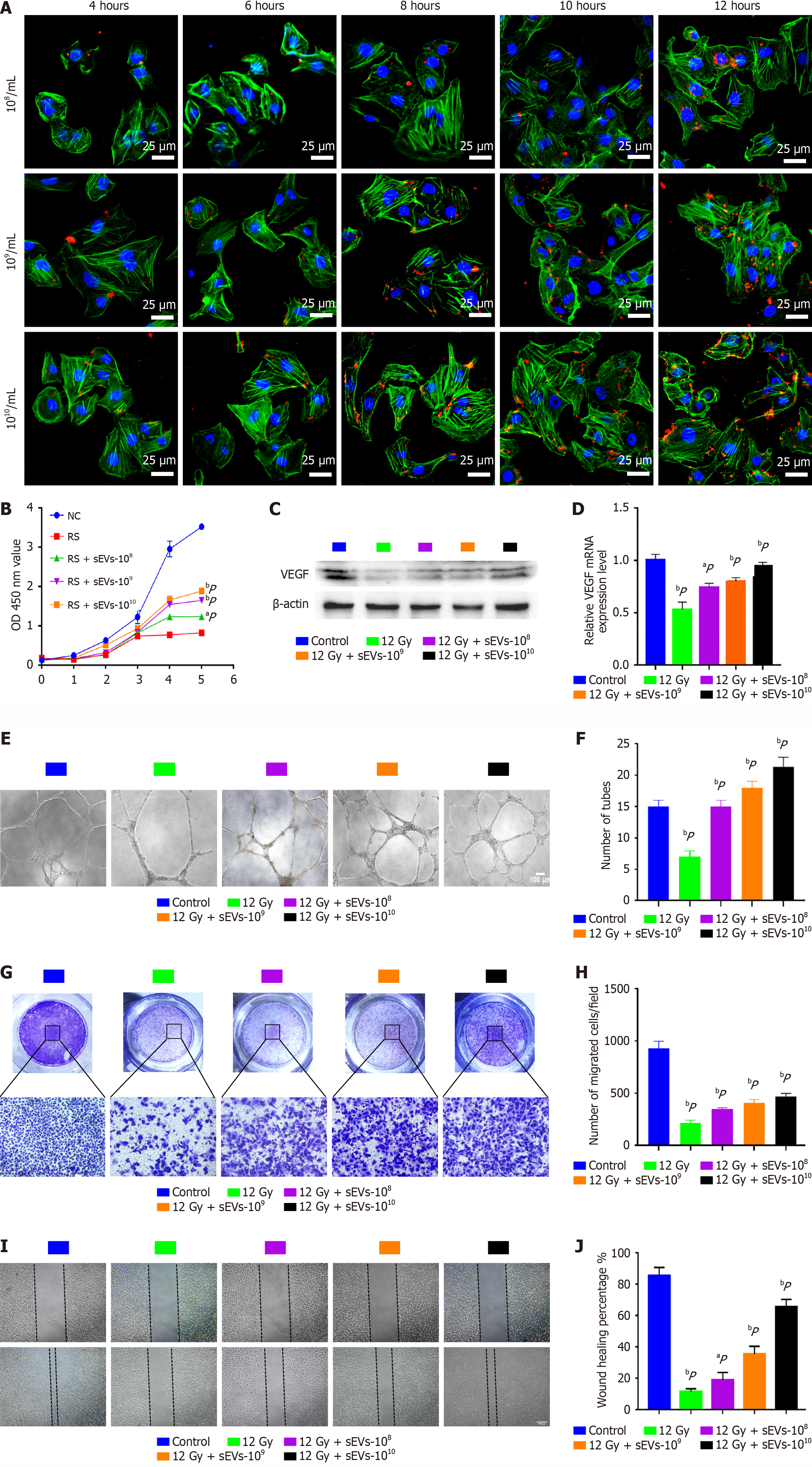
Fluorescence-activated cell sorting showed that isolated 3rd generation BMSCs were positive for the surface markers CD105, CD73 and CD90 but negative for CD14, CD45 and CD34 (Supplementary Figure 3). The dose-dependent radiation-induced BMSCs osteogenesis inhibition was verified in vitro, and 12 Gy (1.2 Gy per minute, for 10 minutes) was selected as the appropriate dose for further investigation (Supplementary Figure 4). BMSCs were co-cultured with CM-DiI-labeled hAMSCs-sEVs for 4-12 hours, and hAMSCs-sEVs uptake was further confirmed by fluorescence imaging. As shown in Figure 3A, the uptake rate was positively correlated with co-culture time and concentration of hAMSCs-sEVs.
The results of ALP and Alizarin Red assays showed that hAMSCs-sEVs alleviated 12 Gy irradiated BMSCs oste
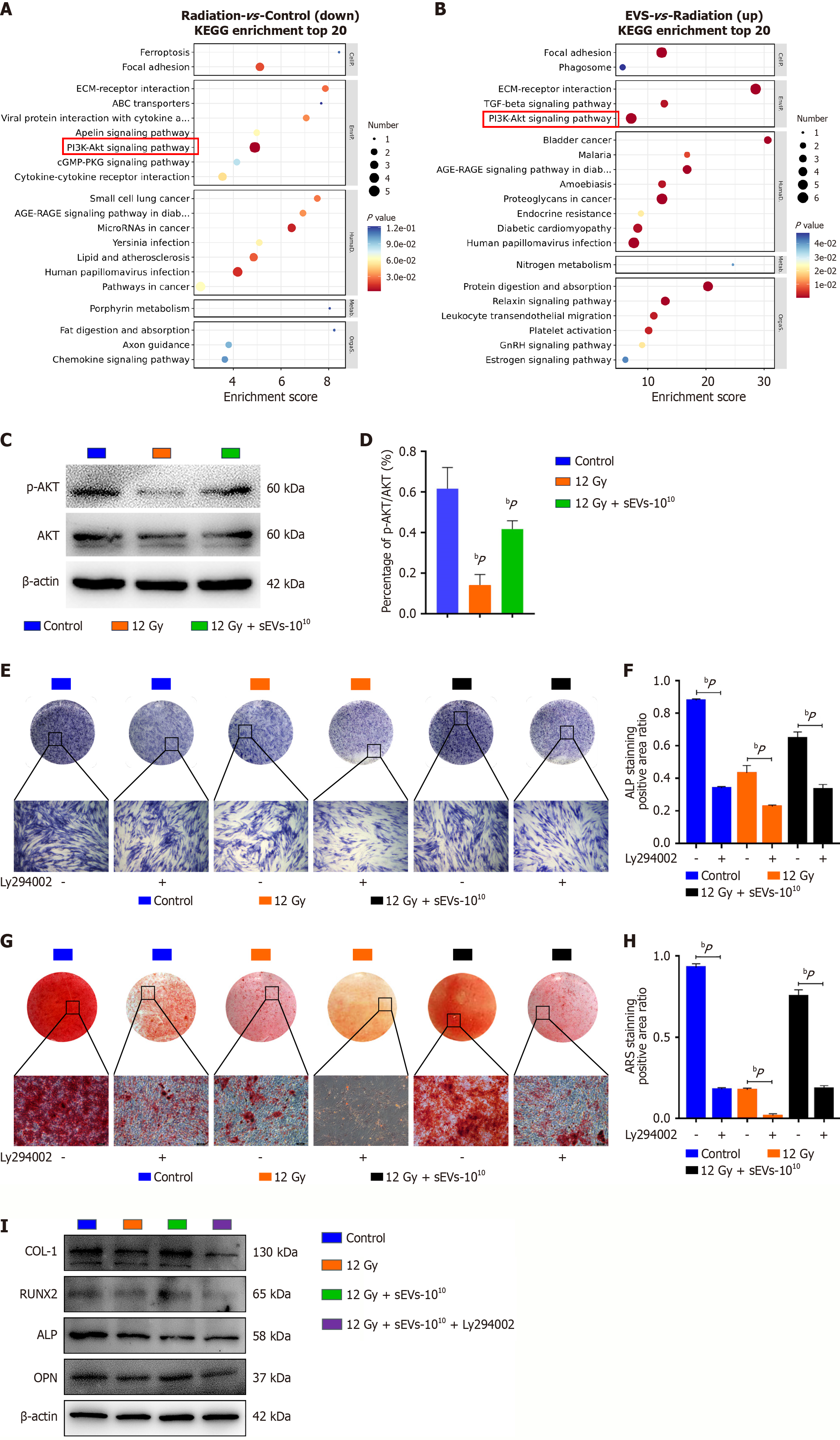
In order to further clarify how hAMSCs-sEVs affected irradiated BMSCs osteogenesis, we performed high-throughput sequencing on the RNA of BMSCs in the control, 12 Gy, and 12 Gy + sEVs-1010 groups. The sequencing data were subjected to principal component analysis. GO enrichment analysis was performed on DEGs, and it was found that the 12 Gy group could enrich the phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) signaling pathway compared to the control group (Figure 4A), and the 12 Gy + sEVs-1010 group could also enrich the PI3K/AKT signaling pathway compared to the 12 Gy group (Figure 4B). Western blotting was used to detect the expression of AKT and phospho
In order to investigate whether PI3K/AKT could play a role in osteogenesis of 12 Gy irradiated BMSCs treated with hAMSCs-sEVs, the PI3K/AKT signaling selective inhibitor Ly294002 was used. We detected the osteogenesis of BMSCs in the control, control + Ly294002, 12 Gy, 12 Gy + Ly294002, 12 Gy + sEVs-1010, and 12 Gy + sEVs-1010 + Ly294002 groups. We found that the alleviation of osteogenesis inhibition of 12 Gy irradiated BMSCs by hAMSCs-sEVs was reduced by Ly294002 (Figure 4E-I). In addition, following Ly294002 treatment of BMSCs, the expression of COL-1, ALP, RUNX2 and osteopontin proteins was downregulated (Figure 4I). These results revealed that hAMSCs-sEVs could reactivate p-AKT expression in irradiated BMSCs to promote osteogenesis.
The dose-dependent radiation-induced HUVECs angiogenesis inhibition was verified in vitro, and 12 Gy (1.2 Gy per minute, for 10 minutes) was selected as the appropriate dose for further investigation (Supplementary Figure 5). HUVECs were co-cultured with CM-DiI-labeled hAMSCs-sEVs for 4, 6, 8, 10 and 12 hours, and hAMSCs-sEVs uptake was further verified by fluorescence imaging. The uptake rate was positively correlated with co-culture time and concentration of hAMSCs-sEVs (Figure 5A). The results of the CCK8 assay showed that hAMSCs-sEVs alleviated 12 Gy-induced HUVECs proliferative inhibition in a concentration-dependent manner (Figure 5B). The expression of VEGF protein in 12 Gy-induced HUVECs was downregulated but restored by hAMSCs-sEVs (Figure 5C and D). The results of tube formation and migration assays revealed that 12 Gy-induced angiogenesis inhibition of HUVECs could be rescued by hAMSCs-sEVs in a concentration-dependent manner (Figure 5E-J). These results revealed that hAMSCs-sEVs could alleviate radiation-induced HUVECs angiogenesis inhibition.
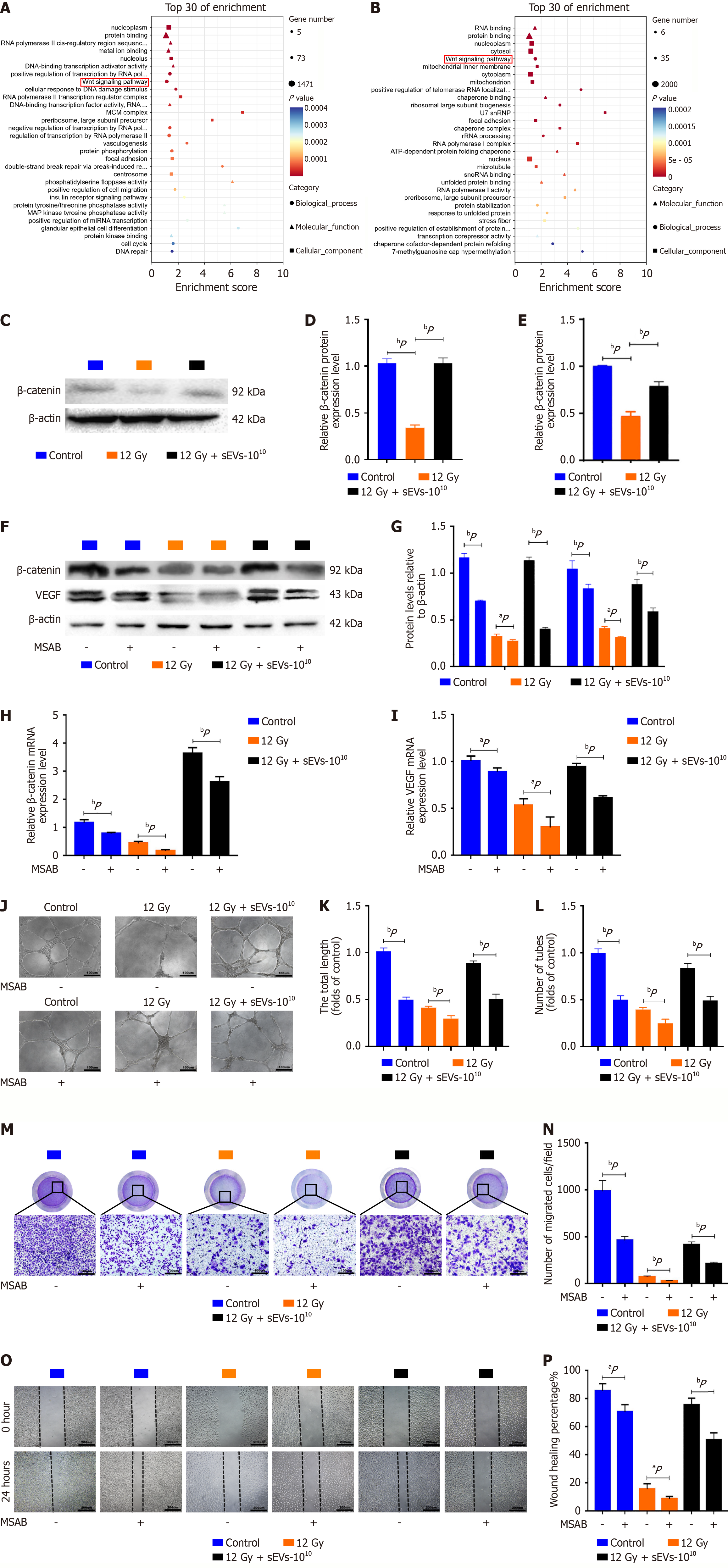
High-throughput sequencing on the RNA of HUVECs was performed in the control, 12 Gy, and 12 Gy + sEVs-1010 groups. The sequencing data were subjected to principal component analysis. GO enrichment analysis was performed on DEGs, and the Wnt/β-catenin signaling pathway was enriched between the control and 12 Gy groups as well as between the 12 Gy and 12 Gy + sEVs-1010 groups (Figure 6A and B). Western blotting and quantitative real-time polymerase chain reaction were used to detect the expression of β-catenin protein and mRNA in the three groups, and it was found that the expression of β-catenin in HUVECs was inhibited after 12 Gy irradiation, while hAMSCs-sEVs could restore the expression of β-catenin (Figure 6C-E).
In order to investigate the roles of the Wnt/β-catenin signaling pathway in the angiogenesis of HUVECs treated with hAMSCs-sEVs, the Wnt/β-catenin signaling selective inhibitor MSAB was used. It was found that the alleviation of angiogenesis inhibition in 12 Gy-HUVECs by hAMSCs-sEVs was reduced by MSAB (Figure 6F-I). Also, following MSAB treatment of HUVECs, the expression of β-catenin and VEGF proteins was downregulated (Figure 6J-P). These results suggested that hAMSCs-sEVs could activate the Wnt/β-catenin signaling pathway in radiation-induced HUVECs to promote angiogenesis.
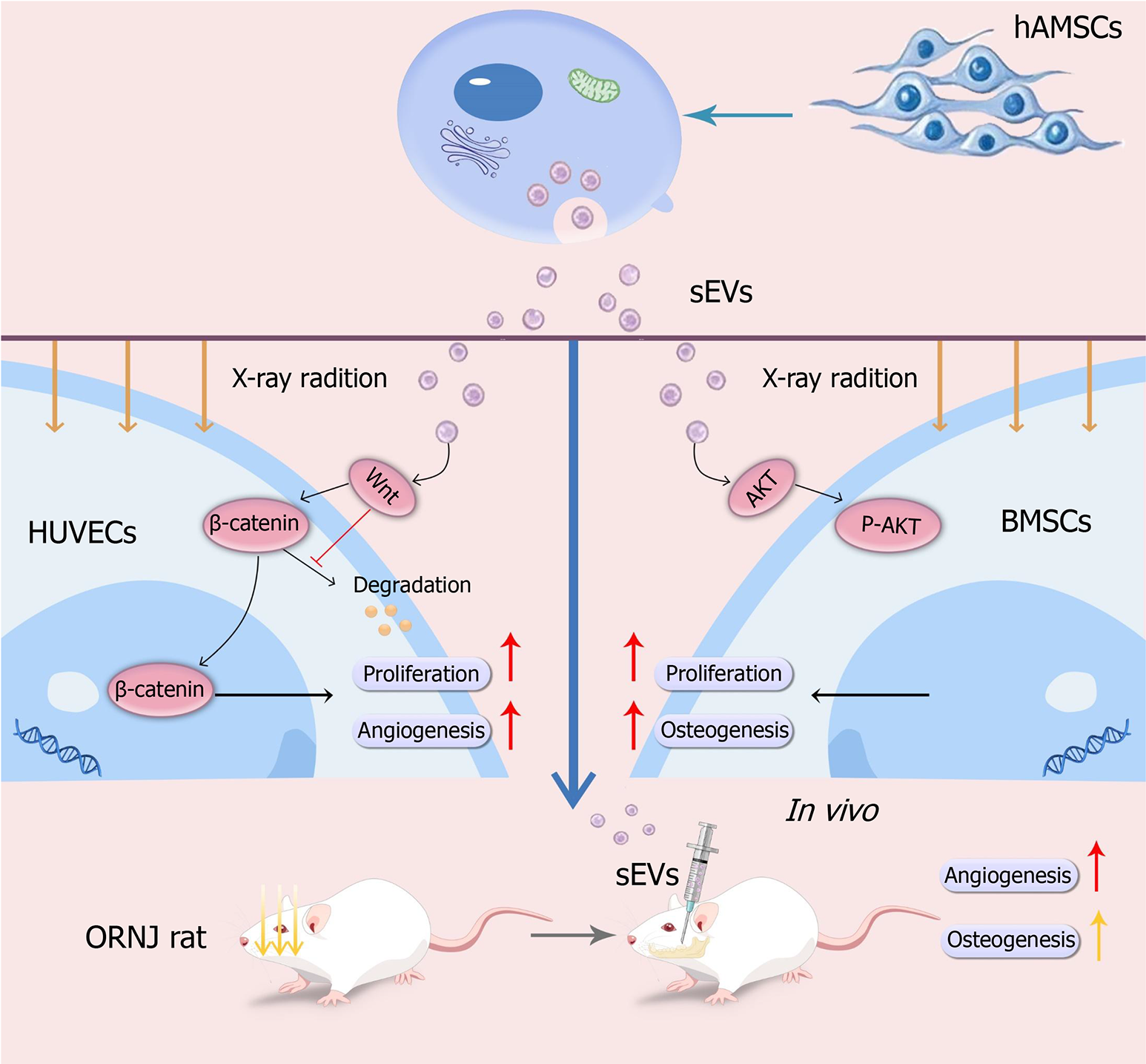
hAMSCs, recognized for their high pluripotency and potent paracrine effects, have garnered significant interest in cell therapy and regenerative medicine. They hold potential for treating diverse conditions, including premature ovarian failure, spinal cord and brain injury, Alzheimer’s disease, pulmonary and liver fibrosis, diabetic wounds, and bone diseases[24]. The therapeutic benefits of hAMSCs are largely attributed to their paracrine activity, wherein EVs serve as key mediators by transporting therapeutic cargo[18,25]. Although previous studies have established the osteoregenerative potential of hAMSCs, the application of either hAMSCs or hAMSCs-sEVs in radiation-induced bone disease remains largely unexplored. In the present study, we investigated the efficacy of hAMSCs-sEVs in mitigating radiation-induced bone damage. Our in vivo experiments demonstrated that hAMSCs-sEVs promoted the healing of tooth extraction sockets in a rat model of ORNJ by enhancing bone regeneration and angiogenesis. Furthermore, in vitro analyses revealed that hAMSCs-sEVs counteracted radiation-induced impairment of osteogenesis and angiogenesis by activating the p-AKT and Wnt/β-catenin signaling pathways, respectively. To our knowledge, this is the first study to elucidate the role and mechanism of hAMSCs-sEVs in the context of ORNJ.
Radiation-induced impairment of osteogenesis is a key pathophysiological factor in ORNJ. While low-dose irradiation (< 2 Gy) enhanced osteoclastogenesis and adipogenesis in BMSCs with limited effects on osteoblasts, high-dose irradiation (> 10 Gy) led to severe disruption of bone architecture[26]. In the study by Wang et al[27], BMSCs exposed to increasing radiation doses (0.25-10 Gy) exhibited reduced cell viability at 2 Gy, suppressed proliferation at 0.5 Gy, and a dose-dependent decline in osteogenic differentiation capacity. Previous evidence suggested that hAMSCs could mitigate inflammation-induced suppression of osteogenic differentiation in BMSCs[11,13]. In the present study, treatment of 12 Gy-irradiated BMSCs with hAMSCs-sEVs promoted osteogenic differentiation in a dose-dependent manner. Notably, a marked increase in RUNX2 protein - but not mRNA - expression was observed in the hAMSCs-sEVs group compared to the irradiation-only group (Figure 3I), indicating post-transcriptional regulation potentially involving enhanced protein stability, extended half-life, or post-translational activation[28]. Together with the in vivo findings, these results imply that hAMSCs-sEVs facilitate bone regeneration in ORNJ partially by restoring the osteogenic capability of radiation-impaired BMSCs.
RNA sequencing followed by GO enrichment analysis of DEGs in BMSCs was performed and this approach identified that the PI3K/AKT signaling pathway was significantly modulated. The PI3K/AKT signaling pathway is a canonical signal transduction cascade that regulates multiple cellular processes through phosphorylation and activation of its central effector, AKT, leading to downstream kinase activation. This pathway has been established as a key regulator of osteogenic differentiation through several mechanisms. It enhanced the Wnt/β-catenin pathway via inactivation of glycogen synthase kinase-3β, thereby promoting osteogenesis[29]. Furthermore, PI3K/AKT signaling regulated osteoblast proliferation and differentiation through interactions with bone morphogenetic proteins (BMPs), notably BMP2, BMP4, BMP7, and BMP9[30,31]. Angiogenesis, a critical supporting process for bone regeneration, was also modulated by this pathway. For example, EVs derived from human induced pluripotent stem cells promoted angiogenesis via PI3K/AKT signaling[32], and BMSCs-EVs were shown to ameliorate impaired angiogenesis and bone regeneration induced by dimethyloxaloylglycine through the AKT/mechanistic target of rapamycin axis[33]. Together, these results demonstrate that hAMSCs-sEVs partially rescue osteogenic differentiation in irradiated BMSCs by activating the PI3K/AKT signaling pathway.
Radiotherapy-induced impairment of blood circulation results in local hypoxia and nutritional deficiencies, representing a hallmark of ORNJ[34]. Clinical evidence indicated a significant reduction in vascular density and area in ORNJ-affected bone, with the extent of microvascular damage positively correlating with radiation dose[34]. Preclinical models further support this, demonstrating a 50% reduction in jawbone blood flow in miniature pigs following 15 days of irra
As the key effector of the Wnt/β-catenin pathway, β-catenin accumulates in the cytoplasm and translocates to the nucleus to drive target gene expression[40]. It played an indispensable role in vascular development, differentiation, and remodeling, thereby critically supporting angiogenesis and vascular stabilization[41]. For example, Wnt5a potentiated VEGF-mediated angiogenesis by upregulating VEGF expression[42]. Similarly, Wnt4 carried by MSCs-derived EVs activated β-catenin in endothelial cells to foster angiogenesis[43]. Furthermore, human umbilical cord MSC-EVs have been shown to promote β-catenin nuclear translocation, accelerating angiogenesis in skin wound healing - an effect abolished by the β-catenin inhibitor ICG-001[43]. In our study, hAMSCs-sEV treatment elevated both β-catenin and VEGF protein levels in irradiated HUVECs. Crucially, this pro-angiogenic effect was reversed by MSAB, a selective inhibitor of β-catenin signaling. Collectively, these findings demonstrate that activation of the Wnt/β-catenin pathway is pivotal for hAMSCs-sEVs-induced angiogenesis in the context of ORNJ.
This study validates that hAMSCs-sEVs restore radiation-induced impairments in osteogenesis and angiogenesis in a concentration-dependent manner, mechanisms mediated through activation of the PI3K/AKT and Wnt/β-catenin signaling pathways, respectively. Collectively, these actions enhanced bone regeneration in our ORNJ rat model. Our findings provide an experimental foundation for the future clinical translation of hAMSCs-sEV-based therapies for ORNJ (Figure 7).
| 1. | Aarup-Kristensen S, Hansen CR, Forner L, Brink C, Eriksen JG, Johansen J. Osteoradionecrosis of the mandible after radiotherapy for head and neck cancer: risk factors and dose-volume correlations. Acta Oncol. 2019;58:1373-1377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 138] [Article Influence: 19.7] [Reference Citation Analysis (1)] |
| 2. | Topkan E, Kucuk A, Somay E, Yilmaz B, Pehlivan B, Selek U. Review of Osteoradionecrosis of the Jaw: Radiotherapy Modality, Technique, and Dose as Risk Factors. J Clin Med. 2023;12:3025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 53] [Article Influence: 17.7] [Reference Citation Analysis (1)] |
| 3. | Chronopoulos A, Zarra T, Ehrenfeld M, Otto S. Osteoradionecrosis of the jaws: definition, epidemiology, staging and clinical and radiological findings. A concise review. Int Dent J. 2018;68:22-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 204] [Article Influence: 22.7] [Reference Citation Analysis (1)] |
| 4. | Fritz MA, Arianpour K, Liu SW, Lamarre ED, Genther DJ, Ciolek PJ, Byrne PJ, Prendes BL. Managing Mandibular Osteoradionecrosis. Otolaryngol Head Neck Surg. 2025;172:406-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (1)] |
| 5. | Chen H, Liu Z, Yue K, Ding J, He Y. Immune microenvironment: novel perspectives on bone regeneration disorder in osteoradionecrosis of the jaws. Cell Tissue Res. 2023;392:413-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (1)] |
| 6. | Li J, Zhou Z, Wen J, Jiang F, Xia Y. Human Amniotic Mesenchymal Stem Cells Promote Endogenous Bone Regeneration. Front Endocrinol (Lausanne). 2020;11:543623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 7. | Zhang C, Du Y, Yuan H, Jiang F, Shen M, Wang Y, Wang R. HAMSCs/HBMSCs coculture system ameliorates osteogenesis and angiogenesis against glucolipotoxicity. Biochimie. 2018;152:121-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 8. | Wang Y, Jiang F, Liang Y, Shen M, Chen N. Human Amnion-Derived Mesenchymal Stem Cells Promote Osteogenic Differentiation in Human Bone Marrow Mesenchymal Stem Cells by Influencing the ERK1/2 Signaling Pathway. Stem Cells Int. 2016;2016:4851081. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 9. | Zhang C, Yu L, Liu S, Wang Y. Human amnion-derived mesenchymal stem cells promote osteogenic and angiogenic differentiation of human adipose-derived stem cells. PLoS One. 2017;12:e0186253. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 10. | Liu Z, Li Y, Yang J, Huang J, Luo C, Zhang J, Yan W, Ao Y. Bone morphogenetic protein 9 enhances osteogenic and angiogenic responses of human amniotic mesenchymal stem cells cocultured with umbilical vein endothelial cells through the PI3K/AKT/m-TOR signaling pathway. Aging (Albany NY). 2021;13:24829-24849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 11. | Wang Y, Wu H, Shen M, Ding S, Miao J, Chen N. Role of human amnion-derived mesenchymal stem cells in promoting osteogenic differentiation by influencing p38 MAPK signaling in lipopolysaccharide -induced human bone marrow mesenchymal stem cells. Exp Cell Res. 2017;350:41-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 12. | Bian Y, Ma X, Wang R, Yuan H, Chen N, Du Y. Human amnion-derived mesenchymal stem cells promote osteogenesis of human bone marrow mesenchymal stem cells against glucolipotoxicity. FEBS Open Bio. 2019;9:74-81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 13. | Jiang F, Zhang W, Zhou M, Zhou Z, Shen M, Chen N, Jiang X. Human amniotic mesenchymal stromal cells promote bone regeneration via activating endogenous regeneration. Theranostics. 2020;10:6216-6230. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 35] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 14. | Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9106] [Cited by in RCA: 8000] [Article Influence: 1333.3] [Reference Citation Analysis (13)] |
| 15. | Mathieu M, Martin-Jaular L, Lavieu G, Théry C. Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol. 2019;21:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3270] [Cited by in RCA: 2880] [Article Influence: 411.4] [Reference Citation Analysis (5)] |
| 16. | Du Y, Jiang F, Liang Y, Wang Y, Zhou W, Pan Y, Xue M, Peng Y, Yuan H, Chen N, Jiang H. The angiogenic variation of skeletal site-specific human BMSCs from same alveolar cleft patients: a comparative study. J Mol Histol. 2016;47:153-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 17. | Aghaloo TL, Chaichanasakul T, Bezouglaia O, Kang B, Franco R, Dry SM, Atti E, Tetradis S. Osteogenic potential of mandibular vs. long-bone marrow stromal cells. J Dent Res. 2010;89:1293-1298. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 136] [Cited by in RCA: 126] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 18. | Song Y, Zhang T, Shi P, Gao Y, Pang X. Exosomes derived from human amniotic mesenchymal stem cells promotes angiogenesis in hUVECs by delivering novel miRNA N-194. Mol Med. 2025;31:173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 19. | Chen HW, Zhang SF, He HT. Effects of different irradiators on the establishment of osteoradionecrosis model of rat mandible. Hua Xi Kou Qiang Yi Xue Za Zhi. 2021;39:524-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 20. | Yamasaki MC, Roque-Torres GD, Peroni LV, Nascimento EHL, Salmon B, Oliveira ML, Freitas DQ, Correr-Sobrinho L. Does the administration of meloxicam before head and neck radiotherapy reduce the risk of mandibular osteoradionecrosis? An animal model study. Clin Oral Investig. 2021;25:3739-3745. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 21. | Ma X, Bian Y, Yuan H, Chen N, Pan Y, Zhou W, Gao S, Du X, Hao S, Yan Z, Li X, Liu K, Xu F, Wang Y, Du Y. Human amnion-derived mesenchymal stem cells promote osteogenic differentiation of human bone marrow mesenchymal stem cells via H19/miR-675/APC axis. Aging (Albany NY). 2020;12:10527-10543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 20] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 22. | Chim SM, Qin A, Tickner J, Pavlos N, Davey T, Wang H, Guo Y, Zheng MH, Xu J. EGFL6 promotes endothelial cell migration and angiogenesis through the activation of extracellular signal-regulated kinase. J Biol Chem. 2011;286:22035-22046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 106] [Article Influence: 7.1] [Reference Citation Analysis (1)] |
| 23. | Jin Y, Kang Y, Wang M, Wu B, Su B, Yin H, Tang Y, Li Q, Wei W, Mei Q, Hu G, Lukacs-Kornek V, Li J, Wu K, Yuan X, Wang W. Targeting polarized phenotype of microglia via IL6/JAK2/STAT3 signaling to reduce NSCLC brain metastasis. Signal Transduct Target Ther. 2022;7:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 126] [Article Influence: 31.5] [Reference Citation Analysis (1)] |
| 24. | Liu QW, Huang QM, Wu HY, Zuo GS, Gu HC, Deng KY, Xin HB. Characteristics and Therapeutic Potential of Human Amnion-Derived Stem Cells. Int J Mol Sci. 2021;22:970. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (1)] |
| 25. | Xiao S, Xiao C, Miao Y, Wang J, Chen R, Fan Z, Hu Z. Human acellular amniotic membrane incorporating exosomes from adipose-derived mesenchymal stem cells promotes diabetic wound healing. Stem Cell Res Ther. 2021;12:255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 110] [Article Influence: 22.0] [Reference Citation Analysis (1)] |
| 26. | Wen S, Dooner M, Cheng Y, Papa E, Del Tatto M, Pereira M, Deng Y, Goldberg L, Aliotta J, Chatterjee D, Stewart C, Carpanetto A, Collino F, Bruno S, Camussi G, Quesenberry P. Mesenchymal stromal cell-derived extracellular vesicles rescue radiation damage to murine marrow hematopoietic cells. Leukemia. 2016;30:2221-2231. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 130] [Cited by in RCA: 177] [Article Influence: 17.7] [Reference Citation Analysis (4)] |
| 27. | Wang Y, Zhu G, Wang J, Chen J. Irradiation alters the differentiation potential of bone marrow mesenchymal stem cells. Mol Med Rep. 2016;13:213-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 28. | Kumari P, Sarovar Bhavesh N. Birth and death view of DNA, RNA, and proteins. Gene. 2023;883:147672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 29. | Majchrzak A, Witkowska M, Smolewski P. Inhibition of the PI3K/Akt/mTOR signaling pathway in diffuse large B-cell lymphoma: current knowledge and clinical significance. Molecules. 2014;19:14304-14315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 72] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 30. | Lauzon MA, Drevelle O, Daviau A, Faucheux N. Effects of BMP-9 and BMP-2 on the PI3K/Akt Pathway in MC3T3-E1 Preosteoblasts. Tissue Eng Part A. 2016;22:1075-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (1)] |
| 31. | Lee MY, Lim HW, Lee SH, Han HJ. Smad, PI3K/Akt, and Wnt-dependent signaling pathways are involved in BMP-4-induced ESC self-renewal. Stem Cells. 2009;27:1858-1868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 69] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 32. | Liu X, Li Q, Niu X, Hu B, Chen S, Song W, Ding J, Zhang C, Wang Y. Exosomes Secreted from Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Prevent Osteonecrosis of the Femoral Head by Promoting Angiogenesis. Int J Biol Sci. 2017;13:232-244. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 222] [Cited by in RCA: 211] [Article Influence: 23.4] [Reference Citation Analysis (1)] |
| 33. | Liang B, Liang JM, Ding JN, Xu J, Xu JG, Chai YM. Dimethyloxaloylglycine-stimulated human bone marrow mesenchymal stem cell-derived exosomes enhance bone regeneration through angiogenesis by targeting the AKT/mTOR pathway. Stem Cell Res Ther. 2019;10:335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 188] [Cited by in RCA: 167] [Article Influence: 23.9] [Reference Citation Analysis (1)] |
| 34. | Dekker H, Bravenboer N, van Dijk D, Bloemena E, Rietveld DHF, Ten Bruggenkate CM, Schulten EAJM. The irradiated human mandible: A quantitative study on bone vascularity. Oral Oncol. 2018;87:126-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 35. | Xu J, Zheng Z, Fang D, Gao R, Liu Y, Fan ZP, Zhang CM, Wang SL. Early-stage pathogenic sequence of jaw osteoradionecrosis in vivo. J Dent Res. 2012;91:702-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 36. | Cao X, Wu X, Frassica D, Yu B, Pang L, Xian L, Wan M, Lei W, Armour M, Tryggestad E, Wong J, Wen CY, Lu WW, Frassica FJ. Irradiation induces bone injury by damaging bone marrow microenvironment for stem cells. Proc Natl Acad Sci U S A. 2011;108:1609-1614. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 204] [Article Influence: 13.6] [Reference Citation Analysis (1)] |
| 37. | Guipaud O, Jaillet C, Clément-Colmou K, François A, Supiot S, Milliat F. The importance of the vascular endothelial barrier in the immune-inflammatory response induced by radiotherapy. Br J Radiol. 2018;91:20170762. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 80] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 38. | Wang Y, Chen X, Yin Y, Li S. Human amnion-derived mesenchymal stem cells induced osteogenesis and angiogenesis in human adipose-derived stem cells via ERK1/2 MAPK signaling pathway. BMB Rep. 2018;51:194-199. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 39. | Zhou M, Pei B, Cai P, Yi C, Akanyibah FA, Lyu C, Mao F. Human umbilical cord mesenchymal stem cell-derived exosomes repair IBD by activating the SIRT1-FXR pathway in macrophages. Stem Cell Res Ther. 2025;16:233. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 40. | Kusserow A, Pang K, Sturm C, Hrouda M, Lentfer J, Schmidt HA, Technau U, von Haeseler A, Hobmayer B, Martindale MQ, Holstein TW. Unexpected complexity of the Wnt gene family in a sea anemone. Nature. 2005;433:156-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 437] [Article Influence: 20.8] [Reference Citation Analysis (1)] |
| 41. | Qian C, Zhu C, Yu W, Jiang X, Zhang F, Sun J. Bone morphogenetic protein 2 promotes osteogenesis of bone marrow stromal cells in type 2 diabetic rats via the Wnt signaling pathway. Int J Biochem Cell Biol. 2016;80:143-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 42. | Wan X, Guan S, Hou Y, Qin Y, Zeng H, Yang L, Qiao Y, Liu S, Li Q, Jin T, Qiu Y, Liu M. FOSL2 promotes VEGF-independent angiogenesis by transcriptionnally activating Wnt5a in breast cancer-associated fibroblasts. Theranostics. 2021;11:4975-4991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (1)] |
| 43. | Zhang B, Wu X, Zhang X, Sun Y, Yan Y, Shi H, Zhu Y, Wu L, Pan Z, Zhu W, Qian H, Xu W. Human umbilical cord mesenchymal stem cell exosomes enhance angiogenesis through the Wnt4/β-catenin pathway. Stem Cells Transl Med. 2015;4:513-522. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 421] [Cited by in RCA: 404] [Article Influence: 36.7] [Reference Citation Analysis (1)] |