INTRODUCTION
Surgical reconstruction of the gastrointestinal (GI) wall is essential to manage complications, such as anastomotic leakage and tracheoesophageal fistulas, which can be life-threatening[1,2]. Muscle flaps, including sternocleidomastoid or intercostal flaps, reinforce tissue defects and separate adjacent organs. Use of the muscle flap technique has been described in case series and clinical reports as a salvage procedure in patients with persistent leaks after esophagectomy or in patients experiencing intractable airway-GI tract fistulas[3-5]. However, muscle flap surgery requires extensive dissection, is highly invasive, and can lead to donor site morbidity and impaired cosmetic outcomes[6,7].
Recent advances in regenerative medicine have introduced myoblast cell sheet transplantation as a less invasive alternative to muscle flaps. In a rat model of gastric perforation, transplantation of skeletal muscle-derived myoblast cell sheets prevented leakage and promoted connective tissue repair[8]. In a porcine model, autologous myoblast cell sheets transplanted laparoscopically to the duodenal serosa after endoscopic submucosal dissection (ESD) prevented delayed perforation[9]. Recently, a first-in-human study demonstrated the safety and feasibility of myoblast cell sheet transplantation in patients who underwent duodenal ESD; the authors observed no delayed perforations[10]. These findings suggest that myoblast cell sheet transplantation offers structural support to weakened GI tissues.
Although these results are promising, the behavior of transplanted myoblasts and the histological changes in the esophageal wall after transplantation have not been established. To address this issue, we investigated the histological behavior of myoblast cell sheets transplanted into an esophageal muscle defect in a rat model for 4 weeks. This study evaluated the effect of myoblast cell sheets on promoting tissue repair and reinforcement and determined how their structure evolved at each time point.
MATERIALS AND METHODS
Animals
All rats received humane care according to the criteria outlined in the Guide for the Care and Use of Laboratory Animals prepared by the National Institutes of Health (NIH publication 86e23, revised 1985). Animal experimental procedures were performed in accordance with the Guidelines for Animal Experimentation of Nagasaki University, with the approval of the Institutional Animal Care and Use Committee (Approval No. 220324-1). Sprague-Dawley rats were purchased from Charles River Laboratories (Tokyo, Japan). The rats were housed at the Laboratory Animal Center for Biochemical Research at Nagasaki University Graduate School of Biomedical Sciences with a temperature of 22 ± 2 °C and a humidity of 40%-70%. Chow (Oriental Yeast, Tokyo, Japan) and autoclaved water were available ad libitum.
Culture of myoblast and cell sheet fabrication
Allogeneic neonatal-derived cell products are necessary when autologous tissue is unavailable for several weeks[11,12]. Because L8 myoblasts are derived from neonatal rats, they exhibit relatively low immunogenicity, making them suitable for allogeneic rat models[13]. Therefore, rat myoblast L8 cells (Cat. No. 95102434; American Type Culture Collection, Manassas, VA, United States), established from Wistar rats, were seeded onto temperature-responsive 6-well plates (Cat. No. CS3004; UpCell 6 Multi-well plates; Cellseed, Tokyo, Japan) at a density of 2 × 105 cells per well. Cells were cultured in Dulbecco’s modified Eagle’s medium supplemented with high glucose (Cat. No. 11995e065, Waltham, MA, United States), 10% fetal bovine serum (Cat. No. 1606731; Thermo Fisher Scientific, Waltham, MA, United States) and 10 mg/mL gentamicin (Fujiseiyaku, Tokyo, Japan) at 37 °C and 5% CO2. The medium was replaced every 48-72 hours. After 7-10 days of culture, the dishes with confluent myoblasts were transferred to 20 °C for 30 minutes before transplantation. The myoblast cell sheets were easily detached using forceps and washed three times with ice-cold Hank’s balanced salt solution containing calcium chloride and magnesium sulfate (Cat. No. 14025e092; Thermo Fisher Scientific, Waltham, MA, United States) (Figure 1A).
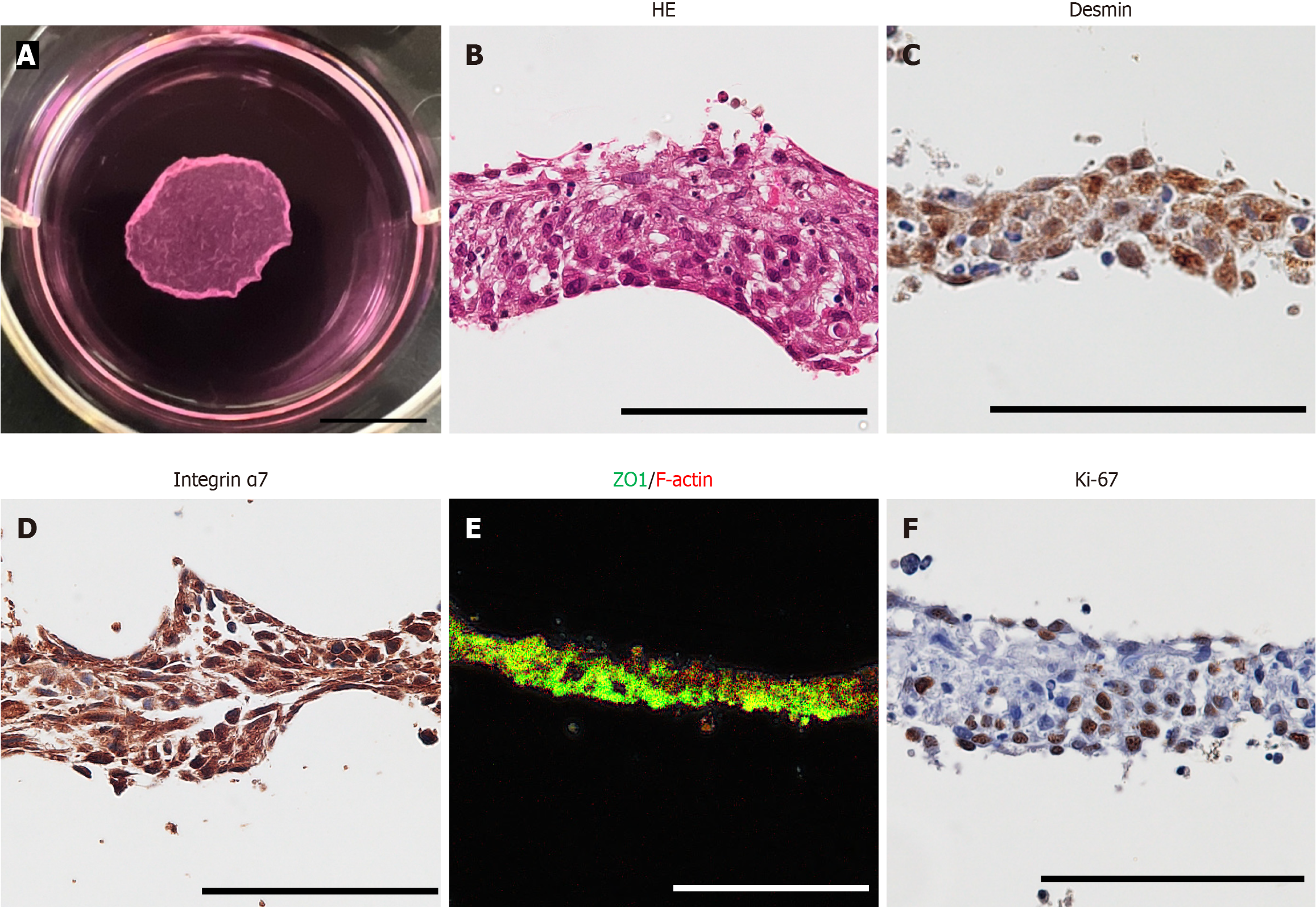
Figure 1 Characteristics of myoblast cell sheets.
A: Fabrication of myoblast cell sheets. The diameter of the myoblast cell sheet was approximately 10 mm (scale bar: 10 mm); B: Hematoxylin and eosin staining of the myoblast cell sheets (× 400, scale bar: 100 μm); C-F: Immunostaining of the myoblast cell sheets for desmin (C), Ki-67 (F), integrin α7 (D) (× 400, scale bar: 100 μm) and zonula occludens-1 (E) in myoblast cell sheets (× 200, scale bar: 100 μm). HE: Hematoxylin and eosin; ZO1: Zonula occludens-1.
Myoblast cell sheet transplantation
We used 7-9-week-old Sprague-Dawley rats to analyze the effects of myoblast cell sheet transplantation. Rats were anesthetized via isoflurane inhalation (Isoflu; DS Pharma Animal Health, Osaka, Japan). After disinfecting the abdominal surface with alcohol, a midline upper abdominal incision was made to open the abdominal cavity for the procedure. The abdominal esophagus was first identified and isolated, then a 10-mm segment encompassing 67% of the circumference of the abdominal esophageal muscle layer was resected to establish an experimental model (Figure 2A).
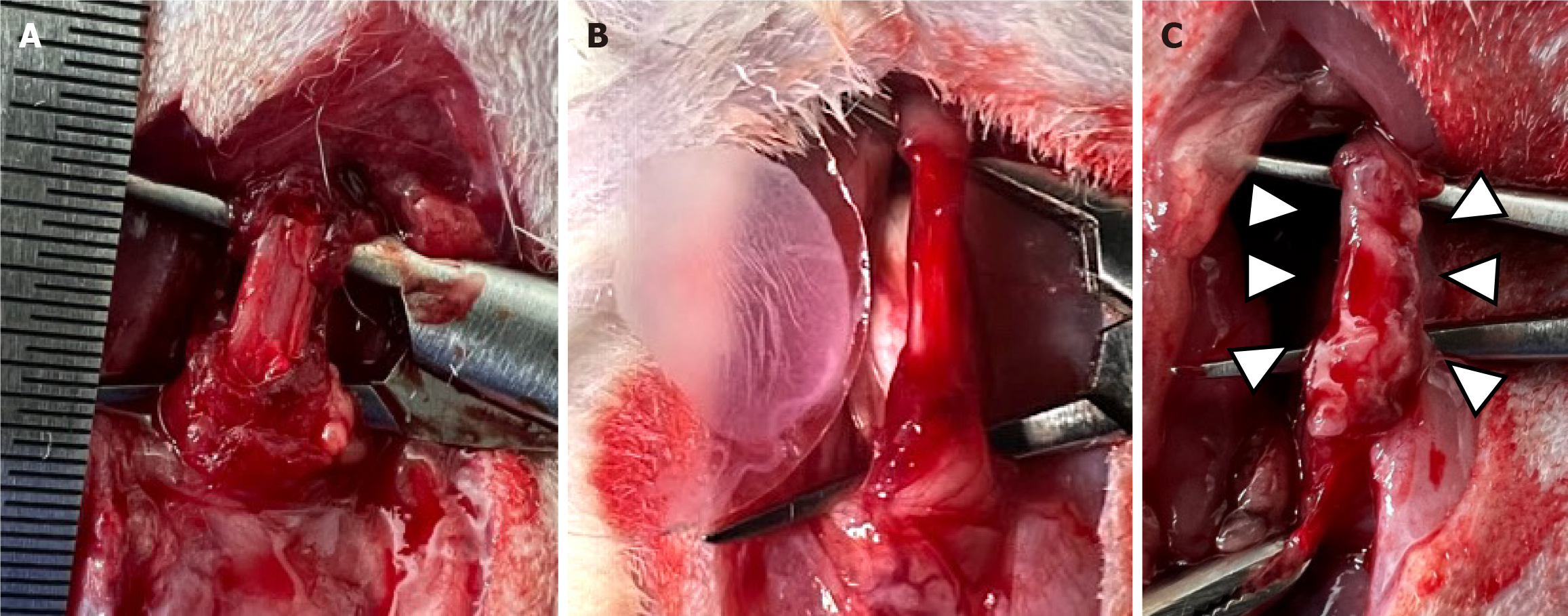
Figure 2 Animal experiments.
A: Resection of 67% of the abdominal esophageal muscularis propria. The major axis of the resected tissue measured 1 cm; B: We transplanted a myoblast cell sheet onto the resected esophageal muscularis propria using a circular glass sheet; C: Gross findings after myoblast cell sheet transplantation (△).
An allogenic myoblast cell sheet was transplanted onto the resected esophageal muscular layer using a circular coverslip as a carrier (Figure 2A and B). The circular coverslip was immediately removed and not left inside the body. Animals that received myoblast cell sheet transplantation were placed in the transplantation group. Animals treated similarly but that did not undergo myoblast cell sheet transplantation comprised the control group. In this group, the abdomen was immediately closed after resection of the esophageal muscular layers was performed. All rats were fed normally after the surgery. Euthanasia was performed at 1 week, 2 weeks, and 4 weeks after the surgical resection. Five rats in the control group and five rats in the transplantation group were examined at each time point of the study. Body weights were recorded at the time of surgery and euthanasia (4 weeks postoperatively). To evaluate postoperative growth, the differential body weight gain was calculated from the time of surgery to euthanasia. All esophageal tissue samples were fixed with 4% paraformaldehyde in phosphate buffered saline (Cat. No. 163e20145; FUJIFILM Wako Pure Chemical, Osaka, Japan) for paraffin-embedding.
Histopathological analyses
Serial sections (5 μm) were cut from the paraffin blocks. Hematoxylin and eosin staining and immunohistochemistry were performed on the tissue sections. Immunohistochemistry was performed using Dako Target Retrieval Solution, Citrate pH 6 (10 ×) (Cat. No. S236784; Dako-Agilent Technologies, Santa Clara, CA, United States), Tris/EDTA solution, pH 9 (Cat. No. S2367; Dako-Agilent Technologies, Santa Clara, CA, United States), Dako REAL Peroxidase-Blocking Solution (Cat. No. S2023; Dako-Agilent Technologies, Santa Clara, CA, United States), Protein Block Serum-Free Ready-to-Use (Cat. No. X0909; Dako-Agilent Technologies, Santa Clara, CA, United States), Dako REAL Antibody Diluent (Cat. No. S2022; Dako-Agilent Technologies, Santa Clara, CA, United States), and Dako REAL EnVision Detection System, Peroxidase/3,3’-diaminobenzidine þ, Rabbit/Mouse (Cat. No. K5007; Dako-Agilent Technologies, Santa Clara, CA, United States). The procedures were performed according to the protocol described by Dako. After visualization with 3,3’-diaminobenzidine, the sections were counterstained with hematoxylin. The stained specimens were observed under an Olympus BX53 microscope (Olympus, Tokyo). The primary antibodies used were rabbit anti-desmin (1:100, Cat. No. ab15200; Abcam, Cambridge, United Kingdom), rabbit anti-Ki67 monoclonal antibody (1:100, Cat. No. ab16667; Abcam, Cambridge, United Kingdom), rabbit anti-CD8α monoclonal antibody (1:2000, Cat. No. ab237709; Abcam, Cambridge, United Kingdom), rabbit anti-CD4 monoclonal antibody (1:4000, Cat. No. ab237722; Abcam, Cambridge, United Kingdom), rabbit anti-CD68 monoclonal antibody (1:200, Cat. No. ab283654; Abcam, Cambridge, United Kingdom), and rabbit anti-integrin alpha (α) 7 monoclonal antibody (1:200, Cat. No. ab203254; Abcam, Cambridge, United Kingdom).
To assess the proliferative capacity of the myoblast cell sheets, the percentage of Ki67-positive nuclei (Ki67-positive cells/total cells) was quantitatively determined using ImageJ software (NIH, Bethesda, MD, United States). For this quantification, three random 40 × high-power fields were selected per cell sheet (n = 5).
The distance from the muscularis mucosa to the outer margin of the esophagus at the site of muscle layer resection was measured and compared between the transplantation and control groups at each time point. Additionally, Ki67-positive nuclei and CD8-, CD4-, and CD68+ cells at the myoblast sheet graft site were quantitatively measured using ImageJ software (NIH). For quantification, three random 40 × high-power fields within the transplanted cell layer were selected per rat.
Immunofluorescence staining
Serial sections (5 μm) cut from the paraffin blocks were heated in Tris/EDTA solution, pH 9 (Cat. No. S2367; Dako-Agilent Technologies, Santa Clara, CA, United States) using a microwave for the antigen retrieval. Blocking was carried out with a Biotin-Blocking System (Dako, Kyoto, Japan) for 20 minutes and in Tris-buffered saline containing 5% bovine serum albumin and 0.1% Tween 20 for 1 hour at 20 °C. The blocked sections were then incubated with Tris-buffered saline + 5% bovine serum albumin, 0.1% Tween 20, and rabbit anti-zonula occludens-1 (ZO1) (1:100, Cat. No. ab221547; Abcam, Cambridge, United Kingdom) for 24 hours at 4 °C, followed by incubation with secondary antibody (goat anti-rabbit biotin at 1:100, Cat. No. B8895; Sigma-Aldrich, St. Louis, MO, United States) for 30 minutes at room temperature, and the high-sensitivity fluorescent reagent ExtrAvidin-TRITC (Cat. No. 1: 180; Sigma-Aldrich, St. Louis, MO, United States) for 1 hour at room temperature. Actin filaments were stained with Alexa Fluor 568-conjugated phalloidin (A12380; Invitrogen, Carlsbad, CA, United States). Fluorescence images were captured using a Ti-U inverted microscope paired with the C-HGFI fiber optic illuminator (Nikon, Tokyo, Japan).
Statistical analysis
Statistical analyses were carried out using JMP Pro software (SAS Institute Inc., Cary, NC, United States). The association between myoblast cell sheets and adhesions was analyzed using the χ2 test. Changes in body weight were analyzed using the two-tailed Student’s t-test and in esophageal thickness using a two-way analysis of variance (commonly known as ANOVA) followed by Tukey’s multiple-comparison test. Cell counts were compared across time points using one-way ANOVA followed by Tukey’s multiple-comparison test. Statistical significance was set at P < 0.05.
RESULTS
Evaluation of myoblast cell sheets
Myoblasts seeded on temperature-responsive culture dishes adhered and spread within a few hours. The cells became confluent and formed a cell sheet after 7-10 days (Figure 1A). The fabricated myoblast cell sheets were approximately 50 μm thick (Figure 1B) and comprised of desmin- and integrin α7-positive cells (Figure 1C and D, respectively) as well as high-expression ZO1-positive cells (Figure 1E). The rate of Ki67-positive cells in the myoblast cell sheets was 39.6% ± 6.8% (Figure 1F).
Transplantation experiment
We evaluated the effects of myoblast cell sheet transplantation on defects in the esophageal muscle layer. Myoblast cell sheets were transplanted after two-thirds resection of the abdominal esophageal muscle layer (Figure 2). At week 4 after transplantation, the transplant group showed a significantly higher body weight gain compared with the control group (67.8% ± 17.3% vs 44.7% ± 9.6%, P < 0.05). Macroscopic findings revealed that the incidence of multi-organ adhesion (specifically, the liver) to the operative site was significantly lower in the transplant group than in the control group (13.3% vs 60.0%, P = 0.02). Notably, no cases of peritoneal inflammation due to perforation or death were observed in either group. Furthermore, in the transplantation group, the transplanted myoblast sheets were observed as nodular formations at 1 week after transplantation, but these formations were not present at 2 weeks and 4 weeks after transplantation (Figure 3). Histological evaluation demonstrated fibrous adhesion of the liver to the resected esophageal muscle layer in the control group (Figure 4A). However, in the transplantation group, the defective esophageal muscle layer was replaced by proliferating desmin-positive cells (Figure 4B-D).
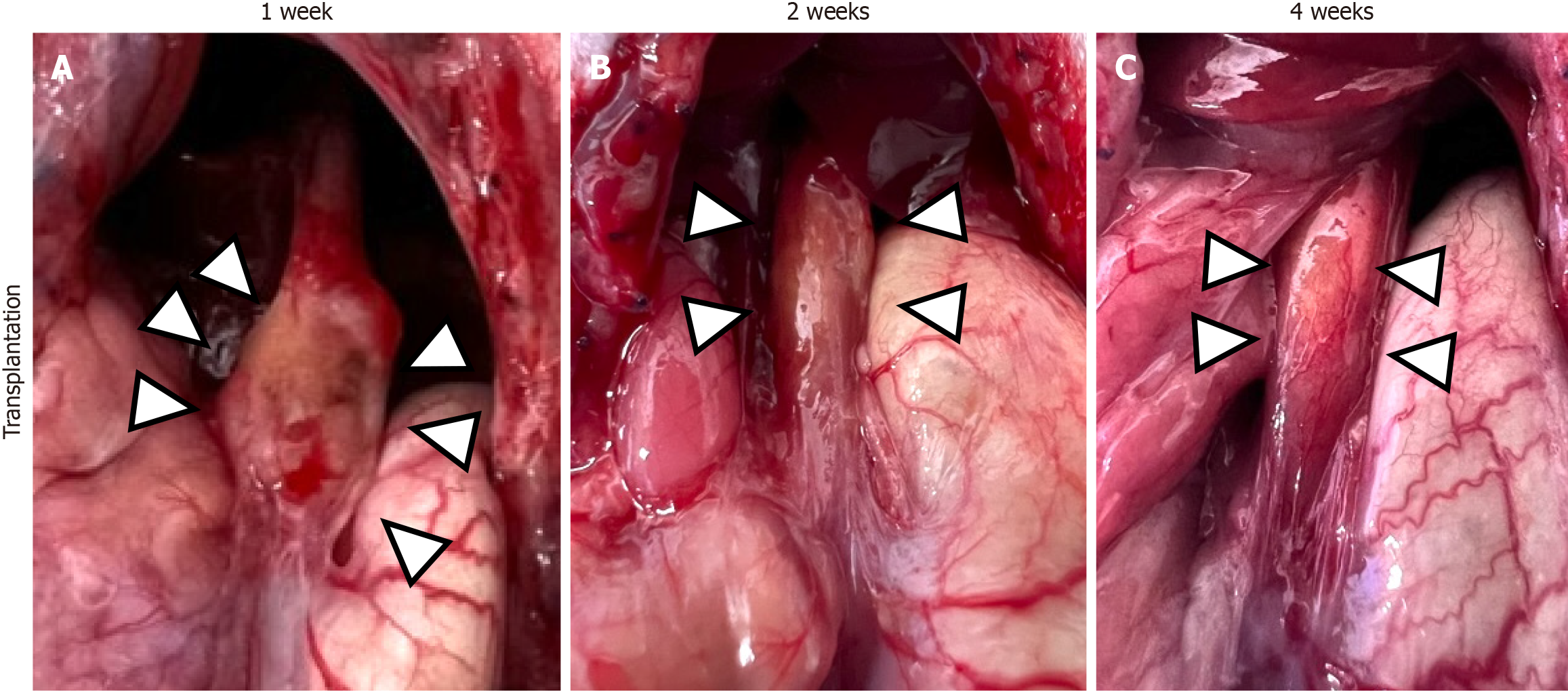
Figure 3 Macroscopic findings of grafted tissue in the transplant group.
A: At 1 week postoperatively, nodular thickening of the esophageal wall was observed at the site of myoblast cell sheet transplantation (△); B: At 2 weeks postoperatively, the nodules observed at week 1 had disappeared, and the transplantation site showed a smooth esophageal wall surface (△); C: At 4 weeks postoperatively, the macroscopic findings at the transplantation site remained consistent with those observed at week 2 (△).
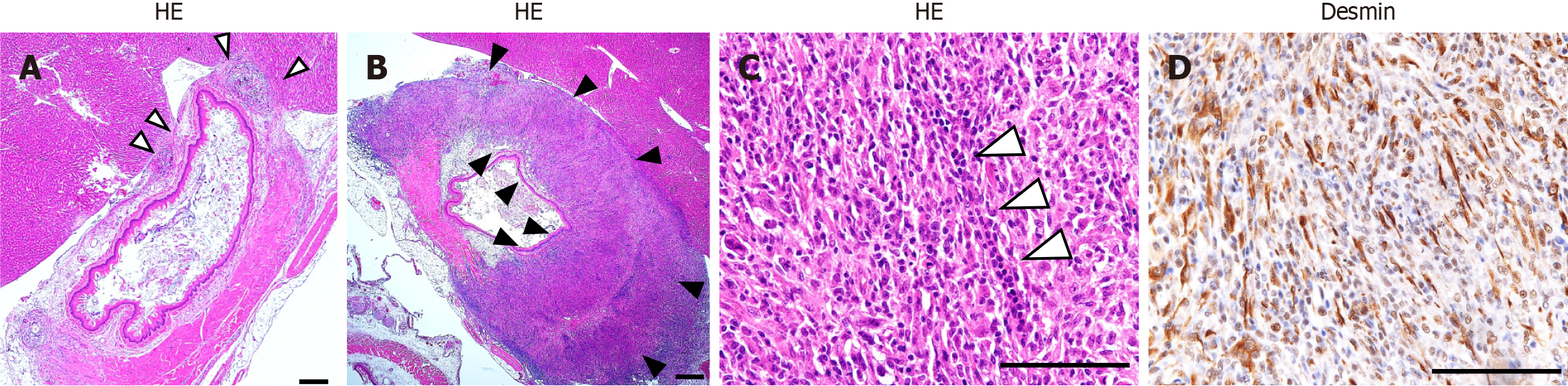
Figure 4 Histological evaluation at 1 week postoperatively.
A: Histological examination of the control group [hematoxylin and eosin (HE) staining, × 40] revealed no tissue replacement at the site of the esophageal muscularis propria resection. Fibrous adhesion was noted between the resected area and the liver (△); B: HE staining (× 40) of the transplantation group showed the myoblast cell sheet proliferating and replacing the resected esophageal muscularis propria (▲); C: HE staining (× 400, scale bar: 100 μm) of the transplantation group showed the cells in the myoblast sheet had partially formed a multinucleated myotube (△); D: Desmin staining (× 400, scale bar: 100 μm) of the transplantation group showed that the myoblast cell sheet contained fibrous desmin-positive cells, indicating proliferation of the myoblast cell sheet.
Evaluation of the behavior of myoblast cell sheets after transplantation
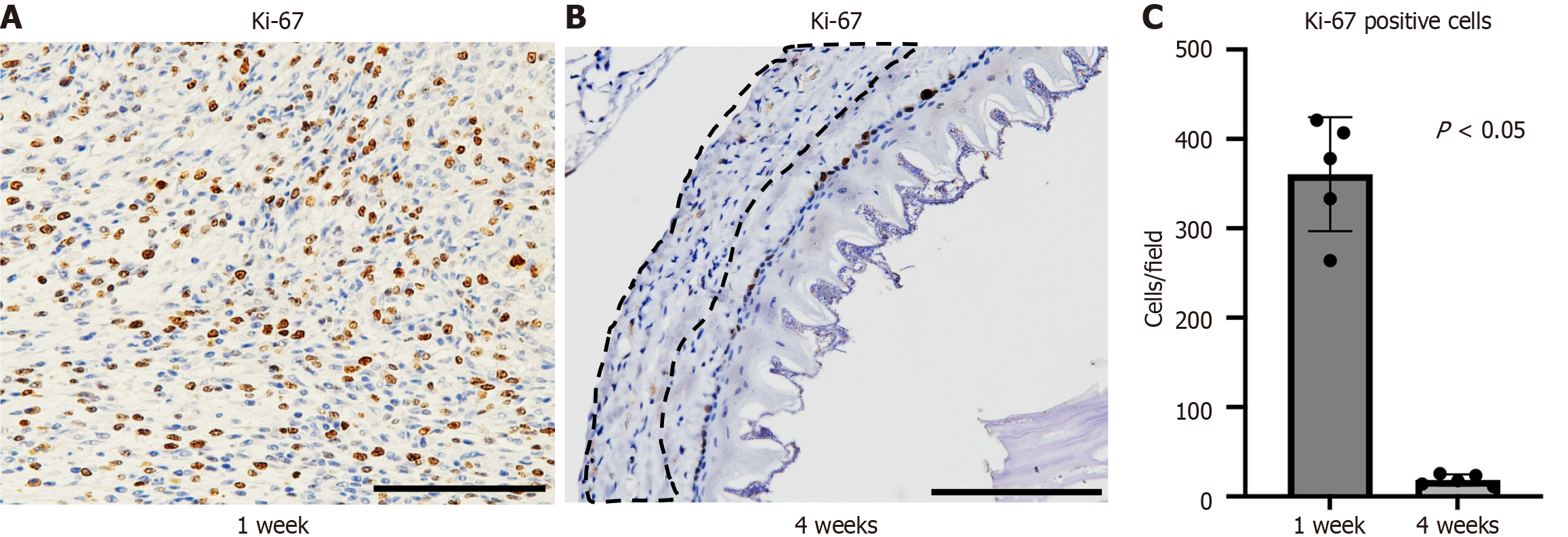
We evaluated the behavior of myoblast cells after transplantation. A gradual decrease in the number of transplanted cells was observed from 2 weeks post-transplantation, with the cell layer transforming into a monolayer by 4 weeks post-transplantation (Figure 5). Cells at the transplantation site initially contained a high number of Ki67-positive cells, which decreased by 4 weeks post-transplantation (Figure 6A and B). The rate of Ki67-positive cells decreased significantly from week 1 (360.6 ± 63.6 cells/field) to week 4 (18.4 ± 6.2 cells/field, P < 0.05) (Figure 6C).
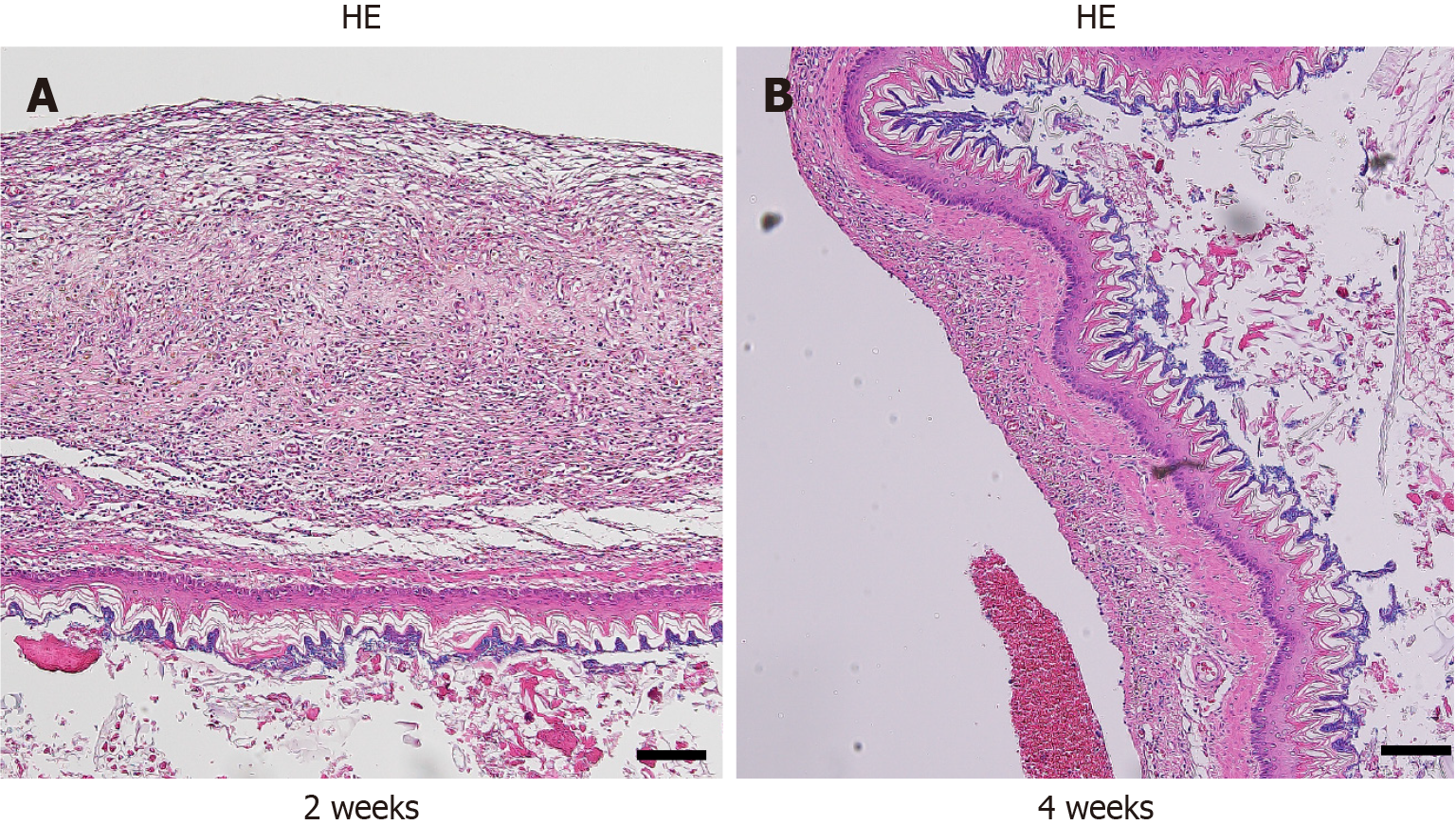
Figure 5 Histological evaluation of transplanted myoblast cell sheets at each time point.
A and B: Hematoxylin and eosin (HE) staining (× 100, scale bar: 100 μm) of the transplantation group at 2 weeks postoperatively revealed a decrease in the thickness of the cell sheet compared with the 1-week time point (A), and 4 weeks postoperatively revealed a further decrease in the thickness of the myoblast cell sheets (B).
Figure 6 Ki-67-positive cells in the transplanted myoblast cell sheets at each time point.
A and B: Ki-67 immunostaining (× 400, scale bar: 100 μm) of the transplantation group at 1 week postoperatively showed that the layer formed by the myoblast cell sheet contained numerous Ki-67-positive cells (A), and 4 weeks postoperatively showed very few Ki-67 positive cells (proliferating cells) in the transplanted myoblast sheet layer (enclosed by a dotted line) (B); C: The number of Ki-67-positive cells per high-power field (× 400) detected via Ki-67 immunostaining showed that Ki-67-positive cells in the myoblast sheet-transplanted area at 4 weeks post-transplantation were significantly reduced compared to that at 1 week post-transplantation.
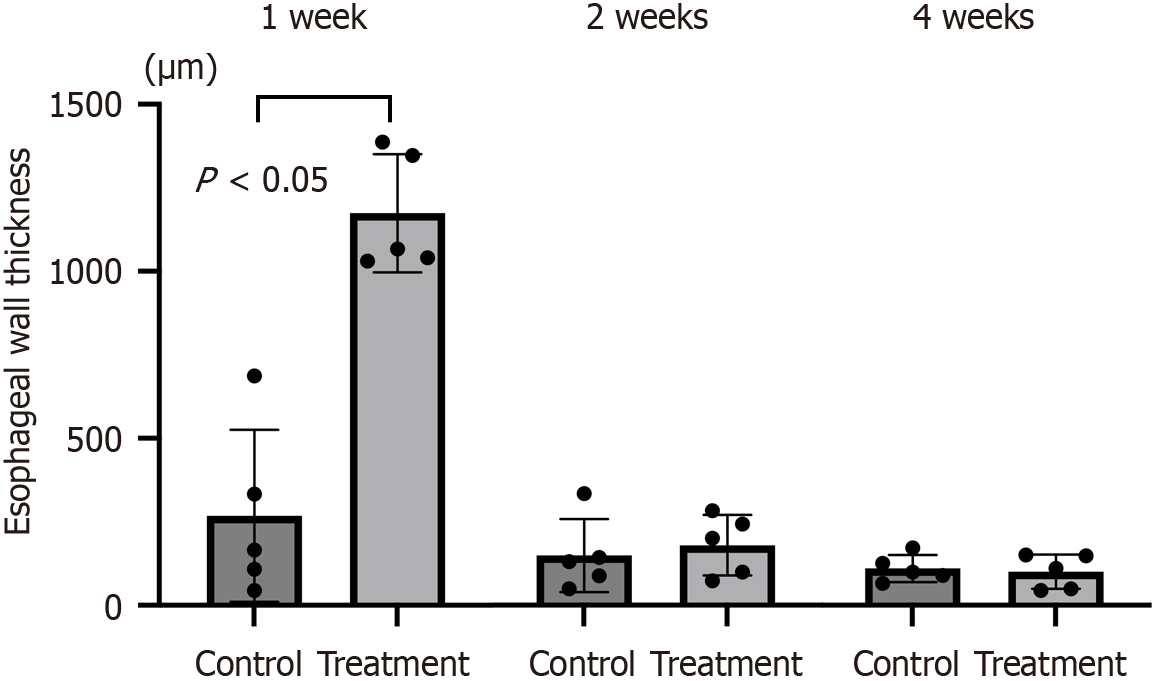
Quantitative image analysis of the cell layer thickness revealed that the transplantation group formed a cell layer approximately 1200 μm thick, 1 week after transplantation. At this time point, the esophageal wall in the transplantation group was significantly thicker than that in the control group (1174.0 ± 177.0 μm vs 267.6 ± 257.6 μm, P < 0.05). However, the thickness in the transplantation group decreased to approximately 180 μm after 2 weeks, and both groups had similar thicknesses at this point (Figure 7).
Figure 7 Esophageal wall thickness at the site of muscularis propria resection at each time point.
In the control group, the esophageal wall thickness remained constant at each time point. At 1 week postoperatively, the esophageal wall was significantly thicker in the transplantation group than in the control group (P < 0.05). The thickness of the esophageal wall decreased to a level comparable to that of the control group at 2 weeks postoperatively.
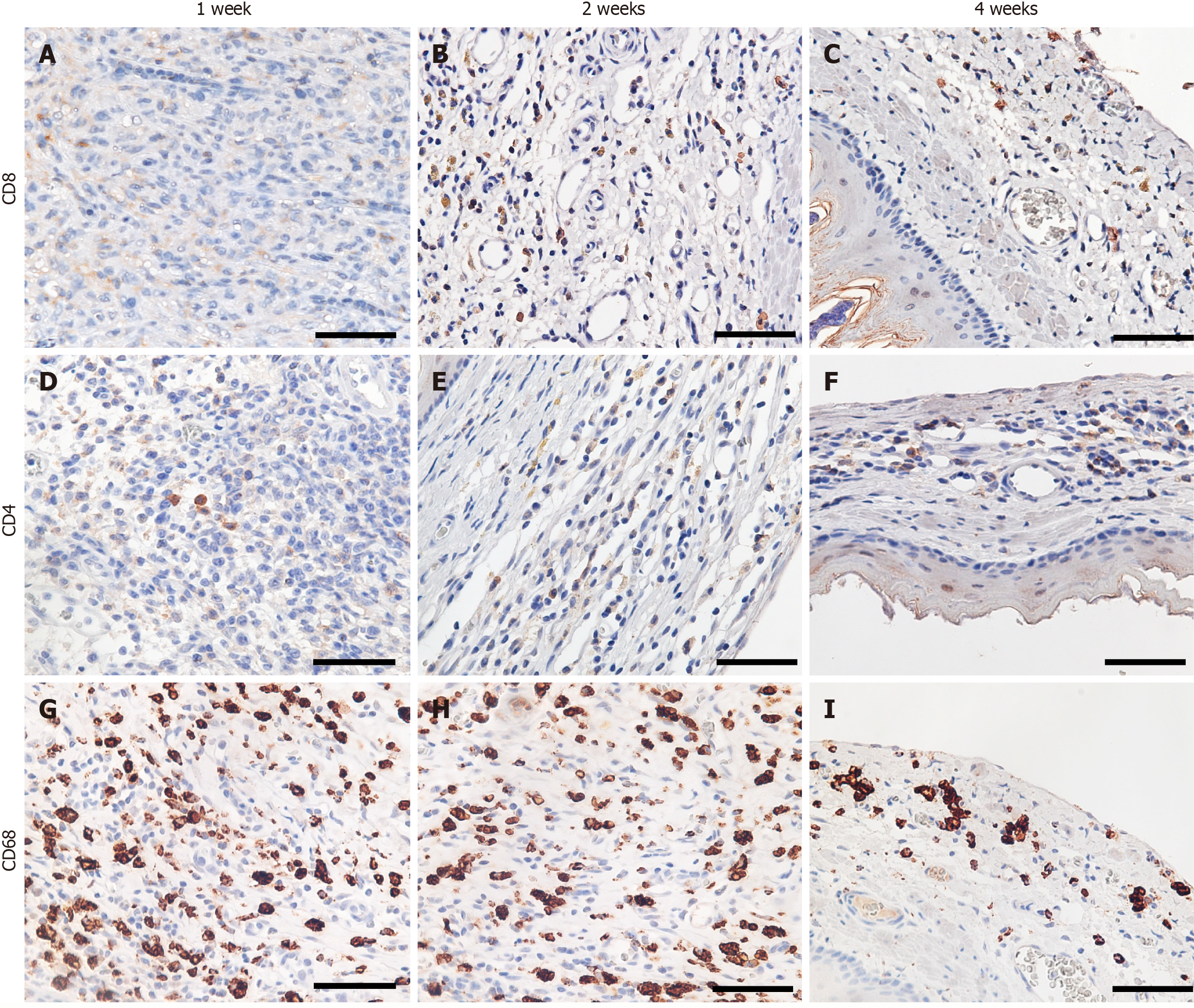
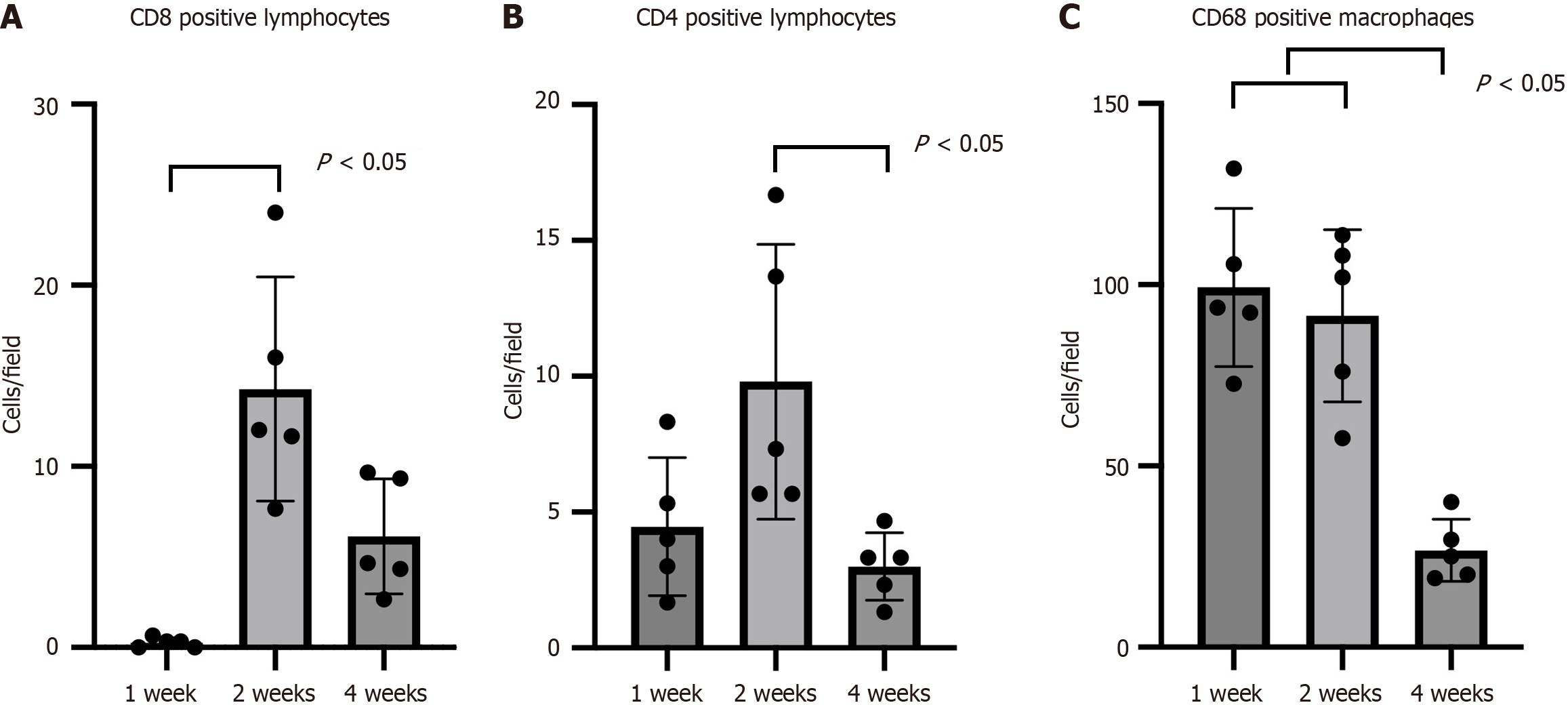
In the evaluation of CD8+ lymphocyte infiltration, very few CD8+ lymphocytes were observed at 1 week post-transplantation; however, at 2 weeks, significantly greater infiltration was observed compared to that at 1 week (0.3 ± 0.3 cells/field vs 14.3 ± 6.2 cells/field vs 6.1 ± 3.2 cells/field, respectively; P < 0.01) (Figures 8A-C and 9A). Regarding CD4+ lymphocyte infiltration, the number of infiltrating cells at 4 weeks was significantly lower than that at 2 weeks (4.5 ± 2.5 cells/field vs 9.8 ± 5.1 cells/field vs 3.0 ± 1.2 cells/field, P < 0.05). However, no robust CD4+ lymphocyte infiltration was observed at any time point (Figures 8D-F and 9B). Furthermore, regarding CD68+ macrophage infiltration, the count at 4 weeks was significantly lower than that at both 1 week and 2 weeks (99.2 ± 21.8 cells/field vs 91.5 ± 23.8 cells/field vs 26.7 ± 8.6 cells/field, P < 0.01) (Figures 8G-I and 9C).
Figure 8 Immunostaining of immune cells in transplanted myoblast cell sheets at each time point.
A-C: CD8 immunostaining in the transplantation group showed infiltration of CD8+ lymphocytes in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm); D-F: CD4 immunostaining in the transplantation group showed infiltration of CD4+ lymphocytes in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm); G-I: CD68 immunostaining in the transplantation group showed infiltration of CD68+ macrophages in the layers of the transplanted myoblast sheet (× 400, scale bar: 50 μm).
Figure 9 Quantitative analysis of immune cells.
A: The number of CD8+ cells per high-power field (× 400) detected via CD8 immunostaining showed mild infiltration of CD8+ cells at 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation; B: The number of CD4+ cells per high-power field (× 400) detected via CD4 immunostaining showed mild infiltration of CD4+ cells at 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation; C: The number of CD68+ cells per high-power field (× 400) detected via CD68 immunostaining showed mild infiltration of CD68+ cells at 1 week and 2 weeks post-transplantation, which subsided by 4 weeks post-transplantation.
DISCUSSION
We investigated the effects of myoblast cell sheet transplantation on esophageal muscle layer defects in a rat model. Our findings indicated that myoblast cell sheets effectively engrafted and proliferated at the injury site, contributing to early tissue replacement and reducing severe fibrous adhesions to the adjacent organs. A notable decrease in the thickness of the myoblast cell sheets suggested complete differentiation into mature muscle tissue 4 weeks after transplantation. The transient reinforcement is likely due to the immediate physical barrier provided by transplanted multilayered sheets. Specifically, the transplanted myoblast cell sheet can serve as a reinforcer in the absence of the esophageal muscular layer and without adhesion within 2 weeks.
Myoblasts highly express muscle-specific adhesion molecules essential for myotube formation and cell fusion, including integrin α3β1, M-cadherin, and intercellular adhesion molecule 1[14-17]. In particular, integrin α7 is known to promote the adhesion and migration of myoblast cells[18]. Furthermore, myoblasts secrete cytokines and growth factors, such as platelet-derived growth factor subunit B, fibroblast growth factor 2, and transforming growth factor β1[14-17]. These factors promote the proliferation, differentiation, and maturation of transplanted myoblasts and surrounding tissues[19,20].
The advantages of cell sheet transplantation over cell suspension injection are the preservation of intercellular junctions and the extracellular matrix. This leads to higher cell engraftment and maintenance of cell function after transplantation[21]. Furthermore, transplanted cell sheets can locally supply growth factors and promote tissue repair[22]. We previously reported that the transplantation of myoblast cell sheets promoted cytokines and growth factors, including hypoxia-inducible factor 1α, stromal cell-derived factor 1, and vascular endothelial growth factor, in the transplanted lesion[23].
Our in vitro evaluation of myoblast cell sheets revealed that the myoblasts formed a robust desmin-positive layer approximately 50 μm thick. The high proportion of Ki67-positive cells in myoblast cell sheets is important for post-transplantation engraftment and proliferation. In addition, the robust expression of integrin α7 and ZO1 suggests enhanced adhesion to the extracellular matrix and strengthened cell-to-cell junctions[18]. This enhanced effect is expected to be a physical barrier to strong intercellular adhesion and to improve the paracrine effects of the secretion of growth factors and cytokines[24,25]. Thus, we determined that the myoblast cell sheets had successfully constructed intercellular junctions and were ready for transplantation into the recipient mice. Based on the results of this study, myoblast cell sheets possess strong cell-cell adhesiveness and high proliferative capacity. Mesenchymal stem cells have both a high secretory capacity for cytokines, such as vascular growth factors[26], and a high plasticity in cell-cell junctions, allowing for adaptation to various functions, such as tissue repair and immune modulation[27,28]. Importantly, for reinforcement of the digestive tract in the pathologic acute phase, the graft must be able to withstand physical stimuli, such as peristalsis. These features collectively underlie the choice of myoblast cell sheets as suitable for this treatment.
Although myoblast cell sheets have been clinically applied to treat patients with myocardial failure[22,29], their application in the GI field has also been reported[8-10,23]. Transplantation of myoblast cell sheets reduced intra-abdominal adhesion and inflammation in a rat model of gastric perforation[8] and was effective in preventing pancreatic fistula in a pancreatic fistula model[30]. Moreover, myoblast cell sheet transplantation to the serosal site after duodenal ESD prevented delayed perforation in a porcine model[9]. A recent human clinical trial investigated the feasibility of laparoscopic autologous myoblast cell sheet transplantation to prevent delayed perforation after duodenal ESD. The transplanted myoblast cell sheets reinforced the tissue and successfully prevented peritonitis in all 3 cases[10].
In this study, myoblast cell sheets transplanted into the rat esophageal muscle layer defect showed remarkable short-term efficacy. One week after transplantation, the myoblasts proliferated and supplemented the defective esophageal muscle layer, confirming the usefulness of myoblast cell sheets for wall reinforcement. We postulated that myoblast cell sheets may be effective in preventing delayed GI perforation. Furthermore, postoperative adhesions were prevented in the transplantation group. Thus, myoblast cell sheets transplanted into the esophageal muscle layer defect suppressed fibrosis through cytokine secretion, thereby reducing intra-abdominal adhesion. The significantly higher weight gain in the transplant group at week 4 suggests that the suppression of adhesions further served to prevent reduced food intake.
The transplanted myoblast cell sheets initially formed a thick layer and significantly increased esophageal wall thickness 1 week after allogeneic transplantation. However, this thickness substantially decreased 2 weeks after transplantation and was comparable to that in the control group. In prior studies, autologously transplanted myoblast cell sheets promoted mucosal regeneration in an animal model, but the sheet structure could not be confirmed after postoperative day 14[9,31,32]. Thus, long-term engraftment has not yet been achieved even with autologous myoblast transplantation. The regenerative capacity of stem cells declines with age due to DNA damage, metabolic abnormalities, and signaling changes[33-35]. Factors such as reduced cellular proliferation and regeneration and the deterioration of cell function associated with aging contribute to lower transplant success rates. Therefore, treatment using cells from younger donors is desirable from the perspective of long-term engraftment and immune response.
The rate of Ki67-positive cells, which indicates proliferation, also decreased at each time point in the present study. After 4 weeks, the transplanted site was composed of a single layer of cells, suggesting that while the sustained presence and myodifferentiation of transplanted myoblasts were limited, the initial cell replacement and physical barrier effects were robust.
The allogeneic transplantation model used in this study served as a valuable platform for developing “off-the-shelf” regenerative medicine products to streamline future clinical applications by eliminating the need for patient-specific cell harvest and expansion. However, several factors hinder the long-term survival of transplanted allogeneic myoblast cell sheets, including hypoxia and malnutrition immediately after transplantation and immune responses[31,36]. It has been reported that myoblasts possess immunomodulatory properties similar to those of mesenchymal stem cells, exhibiting low expression of immune-related markers, such as HLA-DR, CD45, CD34, and CD31. Furthermore, they suppress the proliferation of allogeneic T cells through the indoleamine 2,3-dioxygenase pathway when stimulated with interferon-γ[37]. The number of CD68+ macrophages begins to increase from postoperative day 2, and their infiltration and activation within the tissue become prominent between 3 days and 5 days after lung transplantation[38]. Subsequently, the infiltration of CD4+ lymphocytes and activation of CD8+ cells occur after postoperative day 3, with an increase in the tissue and the resulting cytotoxic response peaking around one week post-transplantation[39,40]. In allogeneic cell transplantation, infiltration of macrophages and neutrophils plays the role of innate immune cells in the early elimination of transplanted cells, which is followed by T cell-mediated rejection in the second week[41].
In this study, CD8+ lymphocytes were observed to have infiltrated the transplanted myoblast cell sheet starting from 2 weeks after transplantation. CD4+ lymphocytes and CD68+ macrophages were mildly elevated at 1 week and 2 weeks after transplantation compared to 4 weeks after transplantation. This suggests the possibility of immune sensitization following transplantation. However, prominent lymphocytic infiltration, suggestive of acute rejection, was not detected.
Allogeneic neonatal or fetal dermal fibroblasts exhibit low immunogenicity, high proliferative capacity, and robust extracellular matrix production[42,43]. Therefore, further research aimed at achieving long-term engraftment using neonatal and fetal cells is necessary to establish a clinically viable strategy for the sustained repair of the full-circumferential and long esophageal muscular layers.
Many animal studies have focused on short-term histological changes. However, research on the in vivo behavior of myoblast cell sheets is limited. Thus, we designed a rat model to verify the engraftment and behavior of myoblast cell sheets after transplantation by selectively resecting the esophageal muscle layer propria. We primarily evaluated the engraftment and post-transplantation behavior of allogeneic myoblast cell sheets to validate the usefulness of the model in regenerative medicine as an alternative to skeletal muscle flap covering. In the future, we plan to verify its therapeutic efficacy for clinical anastomotic leakage and fistulas of the esophagus in large animal experiments.
Despite these promising findings, we must acknowledge the limitations of our study. First, the rat model may not fully replicate the physiological and anatomical conditions of the human esophagus. Notably, the muscularis externa of the rat esophagus is predominantly composed of striated muscles[44], whereas the human esophagus consists of both striated and smooth muscles. This difference may affect the long-term behavior of transplanted myoblast cell sheets and functional integration of the repaired tissue. Second, the observation period was limited to 4 weeks. This duration was insufficient to assess the long-term fate of the transplanted cells, their complete differentiation into mature muscle tissue, or the long-term functional recovery of the esophageal wall. In order to achieve clinical application, it is essential that functional evaluations, including manometry, are performed in future large-animal experiments. Furthermore, the absence of corresponding functional assessments, such as tensile strength or elasticity measurements, limits the clinical interpretation of the observed transient thickness changes. Finally, the small sample size (n = 5 in each group) used in this preliminary study limited the statistical power and, in turn, necessitates cautious interpretation. Of note however, because this was a pilot study with a small sample size, the absence of statistical significance does not rule out clinically or biologically meaningful effects in the study. These data will inform the design and sample size calculations of adequately powered confirmatory studies incorporating functional endpoints.