Published online May 14, 2026. doi: 10.3748/wjg.v32.i18.116229
Revised: January 5, 2026
Accepted: February 26, 2026
Published online: May 14, 2026
Processing time: 182 Days and 0.1 Hours
Colon ischemia (CI) is a common disorder with heterogeneous etiology, whereas CI secondary to inferior mesenteric vein (IMV) pathology is rare and presents distinct diagnostic challenges. Delayed diagnosis and inappropriate management may worsen prognosis.
To explore the clinical characteristics of IMV-related CI and increase awareness of this rare condition.
This retrospective case series described 11 IMV-related CI inpatients at Peking Union Medical College Hospital and summarized their clinical characteristics, imaging and endoscopic features, and prognosis. An additional cohort of 29 arterial-related CI patients was included, and univariate analyses were conducted to compare features between arterial- and IMV-related CI patients.
IMV-related CI patients were all male, with a median age of 44.5 (41.5-63.3) years. Tenesmus is a common symp
IMV-related CI patients have a chronic course, thrombosis comorbidities, and predominant rectal involvement with marked stenosis. Anticoagulation constitutes a cornerstone of therapy.
Core Tip: As a rare subtype of colon ischemia (CI), inferior mesenteric vein (IMV)-related CI often presents with nonspecific symptoms that delay diagnosis. In this study, we reviewed 11 cases from our seven-year experience, highlighting the predominance of middle-aged males, chronic disease, rectal irritation, thrombotic comorbidities, and continuous involvement of the sigmoid and rectum. Identifying both direct and indirect signs of IMV lesions on computed tomography is crucial for accurate diagnosis. Comparative analysis with arterial-related CI patients revealed substantial differences, further highlighting the necessity for prompt recognition and early intervention in IMV-related CI patients.
- Citation: Jin X, Xu TM, Ruan GC, Zhang XF, Chen XY, Li XQ, Tang H, Xu H, Wang QP, Liu W, Li J. Clinical manifestations and imaging features of inferior mesenteric vein-related colon ischemia. World J Gastroenterol 2026; 32(18): 116229
- URL: https://www.wjgnet.com/1007-9327/full/v32/i18/116229.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i18.116229
Colon ischemia (CI) represents the most common form of intestinal ischemia. When hypoperfusion occurs, it deprives the bowel of oxygen and nutrients, leading to hypoxia and direct cell death. As the ischemic period progresses, damage spreads from the mucosa to the serosa[1]. CI can present with a wide spectrum of clinical manifestations, ranging from transient lesions to severe forms with high mortality, depending on the underlying etiology and severity. Several classifications of CI have been proposed[2,3]. For example, CI may be classified as spontaneous or iatrogenic. CI is usually categorized into occlusive ischemic diseases, including embolic and thrombotic arterial occlusion as well as mesenteric venous thrombosis (MVT), and nonocclusive diseases due to alterations in systemic circulation and hypoperfusion of the mesenteric microvasculature, based on the underlying pathophysiological mechanisms[4].
Epidemiological data concerning CI remain limited and quite variable. The estimated annual incidence of acute CI is 15.6/100000 patient-years[5]. Both nonocclusive and arterial ischemia are recognized as the most common mechanisms, characterized by the involvement of watershed zones and typically sparing the rectum[6]. In contrast, venous ischemia, particularly related to the large bowel, is less frequently reported. MVT most commonly affects the superior mesenteric vein (SMV), with approximately 11% of cases involving the inferior mesenteric vein (IMV). Although both conditions may result in CI, lesions of the IMV specifically impact the distal gastrointestinal tract[7]. Only a very few case reports have described CI caused by IMV thrombosis and mesenteric arteriovenous fistula[8,9]. Some studies in the surgical field have shown that IMV ligation during colorectal surgery, even if the inferior mesenteric artery is preserved, can lead to CI, potentially affecting the anal-side colon of the anastomosis and extending to the rectum[10,11]. The diagnosis and differential diagnosis of IMV-related CI are always challenging due to its rarity. The delay in diagnosis might lead to a missed optimal timing of medications and cause poor prognosis of colectomy. Based on the experience of one medical center, this study systematically describes the clinical features of IMV-related CI and analyzes the difference in manifestations between IMV-related CI and arterial-related CI.
We searched the admission database at Peking Union Medical College Hospital from January 2016 to July 2023 and used the diagnostic code for CI or portal/MVT. The diagnosis of CI was established through a comprehensive assessment integrating clinical manifestations (abdominal pain, diarrhea, hematochezia), radiographic imaging, endoscopic findings, and histopathology[12]. Patients were included in the IMV-related CI group if they met the following inclusion criteria: (1) The diagnosis of CI was confirmed by two gastroenterologists according to the criteria (Li J and Ruan GC) on the basis of endoscopic and radiological findings; and (2) The demonstration of IMV lesions on contrast-enhanced computed tomography (CT) scans was confirmed by a senior radiologist (Liu W). Patients were excluded based on the following criteria: CI due to large bowel obstruction, idiopathic myointimal hyperplasia of the mesenteric vein, and idiopathic mesenteric phlebosclerosis. Patients lacking in-hospital imaging and endoscopic data were also excluded from the study. CI patients without predisposing shock, with radiological evidence of mesenteric arterial lesions, were enrolled as arterial-related CI (Figure 1).
The data included sex, age at diagnosis, and cardiovascular risk factors including history of smoking or alcohol consumption, diabetes, obesity, a history of myocardial infarction or stroke, and hypertension. Comorbidities such as malignancy, medication use, hematological disease, autoimmune disease, previous venous thrombosis events, and liver cirrhosis were also recorded. Clinical manifestations included abdominal signs and symptoms and abnormalities on physical examination. Laboratory, radiologic, and endoscopic examination results were recorded. All radiologic images of IMV-related CI patients were retrospectively reviewed, and the radiologic features, including the location of the involved bowel segment; the appearance and degree of wall thickness and enhancement; the presence of intramural, mesenteric or portal venous gas; mesenteric vessel patency; peritoneal fluid; mesenteric exudation; and the development of collateral vessels, were recorded.
The final diagnosis and the initial medications prescribed were recorded. Follow-up outcomes were determined by reviewing the chart and telephone calls until March 2025. Poor prognosis was defined as clinical deterioration (aggravated hematochezia, perforation, worsening obstruction, or hemodynamic instability) or the need for surgical intervention.
Statistical analyses were performed using SPSS version 26.0 software (SPSS, Inc., Chicago, IL, United States). Non
Eleven patients with IMV-related CI were enrolled (Table 1). All were male, with a median age at diagnosis of 44.5 (41.5-63.3) years. All patients had a chronic disease course with a median disease duration of 7 (5-8.5) months. The most common symptoms were weight loss (100%), abdominal pain (81.8%), diarrhea (81.8%), hematochezia (72.7%) and tenesmus (72.7%). Two patients (18.2%) had mild to moderate fever, and 3 (27.3%) had bowel obstruction. Concurrent venous thrombosis, including deep vein thrombosis, pulmonary embolism and internal jugular vein thrombosis, was identified in 5 (45.5%) patients. Abdominal tenderness was noted in 6 patients (54.5%), and abdominal masses were palpable in 3 patients (27.3%). Laboratory investigations revealed elevated D-dimer levels in 81.8% of patients and elevated C-reactive protein levels in 55.6% of patients. Abnormal protein S and antithrombin III levels were observed in 54.5% and 50.0% of the patients, respectively. Tests for antiphospholipid antibodies, lupus anticoagulant, protein C deficiency, and activated protein C resistance yielded no positive results.
| No. | Sex | Age | Disease duration | Chief complaint | Complication | Comorbidity | Endoscopic appearance | Abnormal vessels | Etiology | Treatment | Prognosis |
| 1 | Male | 62 | 5 months | Defecation difficulty, abdominal pain | Incomplete intestinal obstruction | PE, DVT; HBP; DM | Involvement of left colon and proximal rectum; multifocal lesions; congestion, edema, fragileness, ecchymosis, patchy erythema, erosion, ulceration, and stricture | IMV | Thrombosis, thrombophilia | Liquid diet, anticoagulation | Symptoms initially improved, but then worsened again, leading to surgery |
| 2 | Male | 38 | 9 months | Abdominal pain with mucous bloody stools | Sigmoid colon perforation | PE; portal vein cavernous transformation | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, patchy erythema, erosion, ulceration and stricture | SMV, IMV, PV | Thrombosis, thrombophilia | Liquid diet, prednisone, antibiotics | At the onset, inflammatory bowel disease with infection was considered, followed by surgery due to gastrointestinal perforation |
| 3 | Male | 65 | 7 months | Diarrhea | None | CHD; DM | Not reach the cecum; involvement of both left and right colon, entire rectum; multifocal lesions; congestion, edema, patchy erythema and stricture | SMV, IMV, PV | Thrombosis | Anticoagulation | Surgery due to poor therapeutic effect |
| 4 | Male | 63 | 5 years | Diarrhea | None | Portal vein cavernous transformation with parietal thrombus formation; dilatation of the small intestinal lymphangion | Involvement of entire colon and rectum; multifocal lesions; congestion, edema, and patchy erythema | SMV, IMV, PV | Abdominal tuberculosis | MCT diet; anticoagulation | Partial relief of symptoms |
| 5 | Male | 60 | 2 years | Diarrhea | Protein-losing enteropathy | HBP | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, patchy erythema and erosion | IMV | Thrombosis | Liquid diet, anticoagulation, vasodilators | Surgery due to poor therapeutic effect |
| 6 | Male | 36 | 5 months | Abdominal pain, mucous bloody stools | Incomplete intestinal obstruction | HBP; acute lymphocytic leukemia treated by allogeneic hematopoietic stem cell transplantation | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, patchy erythema, erosion, ulceration and stricture | IMV | Thrombotic microangiopathy | Fasting, anticoagulation, vasodilators | Relief of symptoms |
| 7 | Male | 69 | 8 months | Abdominal pain, diarrhea | None | Portal vein cavernous transformation; HBP | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, patchy erythema, erosion, and stricture | IMV, PV | Thrombotic microangiopathy | Liquid diet, anticoagulation | Surgery due to poor therapeutic effect |
| 8 | Male | 72 | 8 months | Abdominal pain, flatulence and reduced defecation | Incomplete intestinal obstruction | None | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, patchy erythema, erosion, and stricture | IMV | Thrombosis | Liquid diet, anticoagulation | Relief of symptoms |
| 9 | Male | 43 | 4 months | Abdominal pain and diarrhea | None | PE | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, ecchymosis, hemorrhage, patchy erythema, erosion, ulceration, and stricture | IMV | Thrombosis, thrombophilia | Liquid diet, anticoagulation | Relief of symptoms |
| 10 | Male | 46 | 7 months | Diarrhea | Hemorrhagic shock | DVT; membranous nephropathy | Not reach the cecum; involvement of both left and right colon, entire rectum; multifocal lesions; congestion, edema, fragileness, ecchymosis, erosion, ulceration, and stricture | SMV, IMV | Thrombosis | Liquid diet | Surgery due to hemorrhagic shock |
| 11 | Male | 41 | 3 months | Diarrhea | None | DVT | Not reach the cecum; involvement of left colon and entire rectum; multifocal lesions; congestion, edema, fragileness, patchy erythema, erosion, ulceration, and stricture | SMV, IMV, PV | Thrombosis, thrombophilia | Liquid diet, anticoagulation | Relief of symptoms |
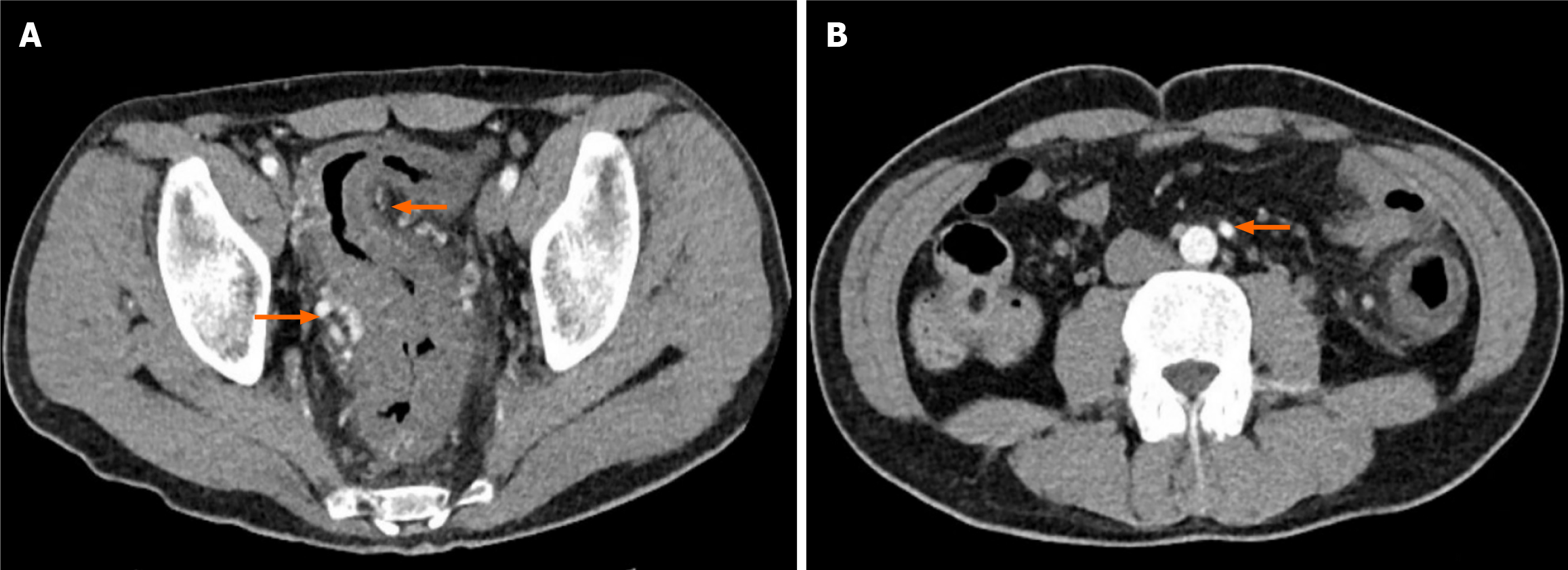
All patients underwent contrast-enhanced CT for evaluation, with involvement extending from the cecum to the rectum. Vascular lesions primarily involved the IMV in all patients, with additional involvement of the SMV (36.4%) and portal vein (45.5%). The most frequently affected segments were the sigmoid colon (100%) and rectum (81.8%). All patients had continuous lesions with bowel wall thickening, ranging from 5.6 mm to 18.2 mm. Decreased enhancement in the arterial phase was noted in 10 patients (90.9%) and in the portal venous phase in 4 patients (36.4%). Intestinal luminal stenosis (81.8%) and the absence of colonic haustration (90.9%) were also common findings. Marked mesenteric exudation was observed in 7 patients (63.6%), and one patient presented with massive ascites. No patient had bowel wall pneumatosis. In terms of the vasculature, thrombus was not directly visualized in any of the patients. Defects in vessel opacification were identified in 10 patients (90.9%), accompanied by compensatory arterial enlargement (9, 81.8%) and collateral circulation formation (8, 72.7%) (Figure 2; Table 2).
| Characteristics | Patients (n = 11) |
| Involved bowel segments | |
| Terminal ileum | 2 |
| Ascending colon | 3 |
| Transverse colon | 6 |
| Descending colon | 8 |
| Sigmoid colon | 11 |
| Rectum | 9 |
| Continuous lesions | 11 (100) |
| Bowel wall thickening | 11 (100) |
| Maximum thickness (mm), median (interquartile range) | 13.5 (11.6-15.6) |
| Abnormal enhancement | 10 (90.9) |
| Arterial phase | |
| Attenuated | 10 (90.9) |
| Enhanced | 0 |
| Unenhanced | 1 (9.1) |
| Portal vein phase | |
| Attenuated | 4 (36.4) |
| Enhanced | 1 (9.1) |
| Unenhanced | 6 (54.5) |
| Luminal stenosis | 9 (81.8) |
| Disappearance of the colonic haustration | 10 (90.9) |
| Mesenteric exudation | 10 (90.9) |
| Pneumatosis | 0 |
| Affected vessel | |
| PV | 5 (45.5) |
| SMV | 4 (36.4) |
| IMV | 11 (100) |
| Thrombus/embolus of IMV | 0 |
| Vascular opacification defect | 10 (90.9) |
| Collateral circulation | 8 (72.7) |
| Compensatory thickening of the arteries | 9 (81.8) |
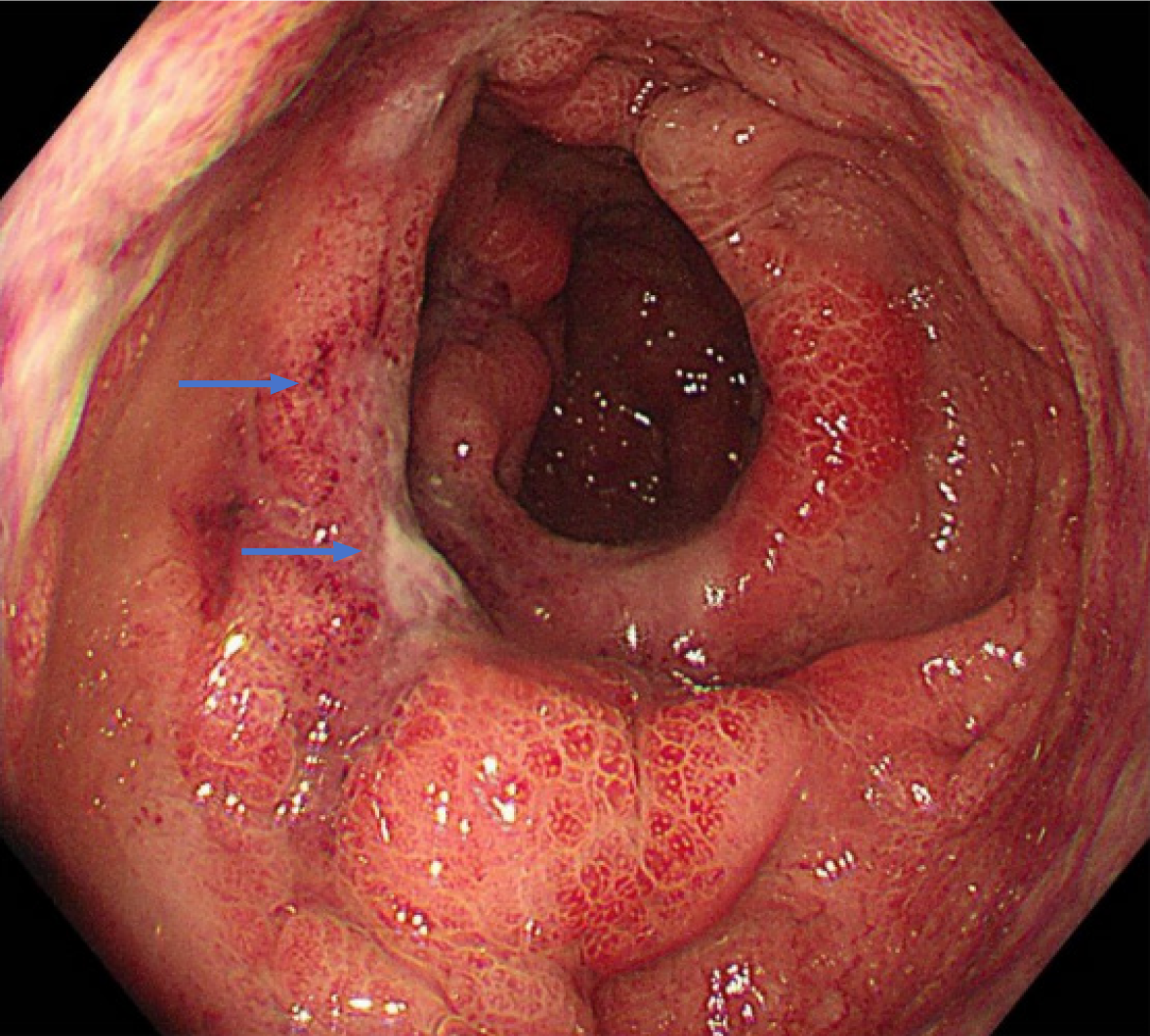
All patients underwent colonoscopic examinations. Nevertheless, due to factors such as intolerance to standard bowel preparation and complete colonic evaluation or the severity of bowel lesions, only 2 patients (18.2%) achieved cecal intubation. Both proximal rectum and sigmoid involvement were observed in all patients, and distal rectal involvement was present in 91.7% of the patients. Continuous lesions were documented in 91.7% of the patients. A crucial aspect of IMV-related CI was stenosis, which was found in 9 patients (81.8%). Ulcers were identified in 6 patients (54.5%), and were predominantly characterized as multiple, irregular, and deep. Additionally, several nonspecific findings included edema (100%), patchy erythema (100%), erosion (81.8%), and enhanced friability (72.7%). Four patients (36.4%) presented with dark purple mucosa (Figure 3). Complete loss of the vascular pattern was observed in all affected areas, and capillary dilation was noted in 3 patients. However, no patient had tortuous veins.
The etiologies of IMV lesions were identified as follows: Thrombophilia in 4 patients, portal vein cavernous trans
Compared with 29 patients with arterial-related CI, males had a more prominent predominance of IMV-related CI (100% vs 62.1%, P = 0.019), while there were no significant differences in age at diagnosis among those with IMV-related CI. Patients with IMV-related CI often had a history of drinking (54.5% vs 17.2%, P = 0.042). IMV-related CI tended to follow a chronic course (100% vs 55.2%, P = 0.007), which was often accompanied by symptoms of tenesmus (72.7% vs 24.1%, P = 0.009) and a higher incidence of concurrent thrombotic events (45.5% vs 10.3%, P = 0.025).
Laboratory examination revealed that the D-dimer level was slightly greater in patients with IMV-related CI, although this difference did not reach statistical significance. Elevated myoglobin levels were observed in 3 arterial-related CI patients, whereas all IMV-related CI patients demonstrated normal muscle enzyme levels. Imaging revealed intestinal wall thickening occurred in both groups. IMV-related CI was associated with significantly increased odds of opacification defects of affected veins and the establishment of collateral circulation (P < 0.001).
Endoscopically, patients with arterial- and venous-related disease both presented with continuous lesions, whereas clear margins were more frequently observed in arterial-related CI patients (P = 0.037). Compared with arterial-related CI patients, IMV-related CI patients had a significantly lower success rate of cecal intubation (18.2% vs 62.1%, P = 0.031). Compared with arterial-related CI patients, IMV-related CI patients demonstrated a greater prevalence of rectal involve
| Parameter | IMV-related CI (n = 11) | Arterial-related CI (n = 29) | P value |
| Sex (male) | 11 (100) | 18 (62.1) | 0.019 |
| Age (years) | 44.5 (41.5-63.3) | 66 (59.5-74) | 0.064 |
| Course (chronic) | 11 (100) | 16 (55.2) | 0.007 |
| HBP | 4 (36.4) | 12 (41.4) | 1.000 |
| CHD | 1 (9.1) | 4 (13.8) | 1.000 |
| DM | 2 (18.2) | 5 (17.2) | 1.000 |
| Smoking | 5 (45.5) | 9 (31.0) | 0.469 |
| Drinking | 6 (54.5) | 5 (17.2) | 0.042 |
| Abdominal pain | 9 (81.8) | 25 (86.2) | 1.000 |
| Diarrhea | 9 (81.8) | 22 (75.9) | 1.000 |
| Hematochezia | 8 (72.7) | 24 (82.8) | 0.660 |
| Tenesmus | 8 (72.7) | 7 (24.1) | 0.009 |
| Fever | 1 (9.1) | 12 (41.4) | 0.068 |
| Concurrent thrombosis | 5 (45.5) | 3 (10.3) | 0.025 |
| Emergency surgery | 0 | 3 (10.3) | 0.543 |
| Poor prognosis | 6 (54.5) | 6 (20.7) | 0.056 |
| Laboratory test | |||
| WBC (× 109/L) | 5.23 (4.07-6.37) | 6.17 (4.62-8.36) | 0.196 |
| Hb (g/L) | 130 (125-140) | 111 (100-142) | 0.175 |
| Alb | 36.0 (33.0-37.5) | 35.0 (31.0-31.0) | 0.473 |
| hsCRP (mg/L) | 9.96 (1.33-25.6) | 15.9 (1.97-52.3) | 0.664 |
| ESR (mm/hour) | 20.0 (9.00-36.8) | 20.0 (18.0-44.0) | 0.332 |
| D-dimer (mg/L) | 1.51 (0.89-3.15) | 0.81 (0.49-2.50) | 0.701 |
| CT | |||
| Thickening | 11 (100) | 21 (72.4) | 0.080 |
| Stenosis | 5 (45.5) | 5 (17.2) | 0.103 |
| Dilation | 1 (9.1) | 3 (10.3) | 1.000 |
| Thrombus | 0 | 1 (3.4) | 1.000 |
| Vascular opacification defect | 8 (72.7) | 0 | < 0.001 |
| Collateral circulation | 9 (81.8) | 2 (6.9) | < 0.001 |
| Endoscopy | |||
| Cecum intubation | 2 (18.2) | 18 (62.1) | 0.031 |
| Right colon | 4 | 16 | 1.000 |
| Descending colon | 4 | 20 | 0.596 |
| Sigmoid colon | 11 | 21 | 0.159 |
| Proximal rectum (or upper rectum) | 11 | 15 | 0.004 |
| Distal rectum (or lower rectum) | 10 | 7 | < 0.001 |
| Continuous lesions | 10 (90.9) | 21 (72.4) | 0.399 |
| Clear margins | 0 | 12 (41.4) | 0.017 |
| Congestion and edema | 11 (100) | 29 (100) | |
| Fragile | 8 (72.7) | 14 (48.3) | 0.286 |
| Ecchymosis, hematoma | 2 (18.2) | 2 (6.9) | 0.300 |
| Hemorrhage | 1 (9.1) | 1 (3.4) | 0.479 |
| Erosion | 9 (81.8) | 22 (75.9) | 1.000 |
| Stenosis | 9 (81.8) | 8 (27.6) | 0.005 |
| Endoscope unable to pass | 3 (27.3) | 4 (13.8) | 0.369 |
| Ulceration | 6 (54.5) | 18 (62.1) | 0.727 |
| Multifocal | 5 | 16 | 1.000 |
IMV-related CI is quite rare and easily misdiagnosed. This study systematically described the clinical characteristics of IMV-related CI patients and revealed a predominance of middle-aged males, a chronic disease duration, rectal irritation symptoms, a high prevalence of thrombotic comorbidities, and continuous involvement of the sigmoid and rectum. Recognizing both the direct and indirect findings of IMV lesions on CT was pivotal for the diagnosis of IMV-related CI. The most common etiology was thrombophilia, and anticoagulation was the primary medication choice. Delayed diagnosis and anticoagulation resulted in poor prognosis. Furthermore, compared with arterial-related CI, IMV-related CI was associated with a greater likelihood of distal rectum involvement, a greater prevalence of colon stenosis, and less pronounced clear margins.
The epidemiology and demographic data concerning venous-related CI, especially IMV-related CI, are limited. In contrast to previous epidemiological patterns in CI, which revealed a female predominance and an advanced mean age of 65 years[5,13], our findings demonstrated significant demographic divergence, with a male predominance and a mean age of 56 years, which is consistent with the features of MVT[7]. As a vascular disorder, CI has previously been found to be associated with risk factors commonly linked to vascular lesions, such as advanced age, diabetes mellitus, and hypertension[4]. However, these features were not prominently observed in IMV-related CI patients. The patients always exhibit local intra-abdominal factors, as well as inherited or acquired thrombophilia, which aligns with the significant proportion of concomitant venous thromboembolic events observed in our cohort[14-16].
Most of the patients in this study exhibited a chronic course. However, previous studies have indicated that chronic MVT comprises merely 20%-40% of MVT cases. This discrepancy may be attributed to the fact that MVT, predominantly involving the SMV, prefers acute small bowel pathology, which was not included in our study. The colonic circulation primarily drains into the IMV, and IMV involvement accounts for only 0% to 11% of cases of MVT[7]. CI can manifest nonspecifically as various degrees of abdominal pain, diarrhea, and hematochezia. Notably, we identified significant pronounced rectal irritative symptoms, which was consistent with radiological evidence of a prominent rectal lesion. A chronic course may lead to weight loss, and a limited subset of patients may progress to a critical state due to hemorrhage or perforation. Additionally, a subset of patients with MVT present features of portal hypertension or remain asympto
Laboratory evaluation is integral to the process of diagnosis and etiological screening. As in other thrombosis diseases, D-dimer is regarded as a routine biomarker. Several studies have reported that D-dimer levels are helpful for diagnosing or indicating transmural injury in acute MVT patients[17]. However, D-dimer is a fibrin degradation product, and its level depends on the timing of the test and the severity and duration of ischemia. The diagnostic utility of D-dimer for acute mesenteric ischemia exclusion demonstrates limited generalizability to chronic intestinal ischemia populations[15]. Similarly, elevated D-dimer levels were not significantly relevant in our cohort, which may be attributed to the predominantly chronic disease course rather than the acute phase. No biomarker has yet been shown to be sensitive or specific enough to establish or exclude a chronic CI diagnosis. Testing for genetics can be conducted to exclude hereditary etiology.
Splanchnic vein thrombosis primarily affects the portal vein and mesenteric vein[18]. Venous drainage largely parallels the arterial supply. Veins from the right colon converge into the SMV, and the left colon drains into the IMV. The superior rectal vein drains the upper two-thirds of the rectum into the IMV, and the lower third is drained by the middle and inferior rectal veins into the internal iliac veins[19]. The IMV usually appears slender and has great individual variation. The diagnostic hallmark of CI venous occlusion is intravascular thrombosis, with a finding of luminal filling defects[20], albeit a rare observation. Indirect signs, such as suboptimal opacification of the mesenteric vein and the formation of collateral circulation for vein drainage, suggesting a chronic course, are more common. Bowel wall thickening is often prominent. A meta-analysis revealed that in the venous-related acute mesenteric ischemia subgroup, decreased or absent bowel wall enhancement was significantly related to transmural intestinal necrosis, reflecting impaired bowel wall perfusion and irreversible damage[21].
Endoscopy serves as a pivotal diagnostic tool in CI patients. Endoscopic lesions of arterial-related CI typically include continuous changes with descending colon or sigmoid colon involvement, with nonspecific presentations, including congestion, edema, ulceration, and erosion[22]. In addition to the similarities in endoscopic findings, two major features of venous-related CI were identified in this study and compared with those of arterial-related CI. First, in contrast to the conventionally recognized pattern in which arterial-related CI typically spares the rectum, IMV-related CI has a propensity to affect the rectum. Despite the dural blood supply of the rectum, the middle and distal rectal vein might not be able to compensate for thrombosis or obliteration of the IMV. The exact pathogenesis underlying this distinctive involvement remains elusive. Second, stenosis emerged as a significant clinical manifestation. Moreover, only a limited number of patients with IMV-related CI were able to undergo a complete colonoscopy. Relying solely on colonoscopic evaluation poses an impediment to gauging the full extent of ischemic injury. The specific pathohistological manifestations are ghost cells and mucosal ischemia; however, the positivity rate in endoscopic and surgical specimens is low[23].
For management, therapeutic decisions hinge on the detection of peritoneal signs, which may necessitate emergent surgical intervention. After the exclusion of acute abdomen, anticoagulation therapy is considered the first-line treatment for acute MVT or the exacerbation of chronic MVT[24]. Consequently, pharmacotherapy was the initial treatment modality in our cohort. The widely accepted approach involves initial anticoagulation with unfractionated heparin or low-molecular-weight heparin[25]. Despite the advantages of oral administration and absence of necessity of strict laboratory monitoring, the use of NOACs remains controversial. Low-molecular-weight heparin appeared more effective in certain patients, likely due to enhanced antithrombin-mediated inhibition of activated factor Xa, providing a broad-spectrum anticoagulant effect[26]. Clinical responses are heterogeneous, necessitating prompt formulation adjustments when required. The optimal anticoagulation duration lacks consensus, although a minimum anticoagulation duration of 3-6 months is recommended. Lifelong anticoagulation may be warranted in individuals with an irreversible hypercoagulable state[27]. Two patients did not receive anticoagulation therapy in time, and both experienced poor clinical outcomes. We also observed that compared with the more rapid alleviation observed in arterial-related cases, IMV-related CI tended to result in a delayed response to therapy. Missed cases can yield irreversible and even fulminant disease that may necessitate surgical resection.
CI is generally associated with a milder course than small bowel ischemia because of several protective mechanisms[28]. Previously, CI was considered a relatively benign and self-limiting condition, with symptoms resolving within 2-3 days and mucosal healing occurring within 1-2 weeks. However, this perspective is contingent upon the assumption that CI results from a transient disruption in arterial blood flow. One patient was diagnosed with SMV thrombosis at another hospital. Upon admission to our hospital, imaging revealed resolution of the thrombus, but signs of ischemia, such as intestinal wall thickening, still presented. The prognosis of IMV-related CI patients is influenced by the rapidity of thrombus formation, as well as the extent and location of vascular involvement[29]. With respect to acute events, the lack of well-developed collateral circulation results in severe clinical presentations. Conversely, in subacute or chronic MVT patients, while venous obstruction may precipitate ischemia, the development of robust collateral circulation may mitigate the onset of ischemic injury. Nevertheless, a significant proportion of these patients ultimately receive surgical intervention for exacerbation or persistence of symptoms throughout the long-term disease course. This may correlate with the presence of irreversible tissue lesions.
Owing to its rarity and limited knowledge, this study represents the first comprehensive attempt to summarize the clinical characteristics of CI lesions caused by IMV-related diseases. Moreover, we excluded congenital vascular malformations and idiopathic mesenteric phlebosclerosis[30,31], whose disease behavior differed from that of our IMV-related CI patients. This study has several limitations. As a comprehensive tertiary hospital, the inpatients enrolled potentially had more complex comorbidities and severe conditions than those from another hospital. With an increase in clinical awareness of this condition, multicenter cohorts should be included to validate these findings.
As a rare subtype of CI, IMV-related CI shows different disease behaviors. Tenesmus, a high prevalence of thrombosis comorbidities, rectum involvement and stenosis under endoscopy, and indirect signs of IMV lesions contributed to the early diagnosis of IMV-related CI. Revealing the etiology of IMV lesions and appropriate anticoagulation is fundamental for protecting against poor prognosis.
| 1. | Theodoropoulou A, Koutroubakis IE. Ischemic colitis: clinical practice in diagnosis and treatment. World J Gastroenterol. 2008;14:7302-7308. [PubMed] [DOI] [Full Text] |
| 2. | Maimone A, De Ceglie A, Siersema PD, Baron TH, Conio M. Colon ischemia: A comprehensive review. Clin Res Hepatol Gastroenterol. 2021;45:101592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 3. | Uberti G, Goldblum JR, Allende DS. Ischemic enterocolitis and its differential diagnosis. Semin Diagn Pathol. 2014;31:152-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 4. | Walker AM, Bohn RL, Cali C, Cook SF, Ajene AN, Sands BE. Risk factors for colon ischemia. Am J Gastroenterol. 2004;99:1333-1337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 68] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Longstreth GF, Yao JF. Epidemiology, clinical features, high-risk factors, and outcome of acute large bowel ischemia. Clin Gastroenterol Hepatol. 2009;7:1075-80.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 79] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 6. | Brandt LJ, Feuerstadt P, Longstreth GF, Boley SJ; American College of Gastroenterology. ACG clinical guideline: epidemiology, risk factors, patterns of presentation, diagnosis, and management of colon ischemia (CI). Am J Gastroenterol. 2015;110:18-44; quiz 45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 211] [Article Influence: 19.2] [Reference Citation Analysis (4)] |
| 7. | Singal AK, Kamath PS, Tefferi A. Mesenteric venous thrombosis. Mayo Clin Proc. 2013;88:285-294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 129] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 8. | Zheng K, Brandt LJ, LeFrancois D. Homozygous Factor V Leiden presenting as irreversible chronic colon ischemia resulting from inferior mesenteric vein thrombosis. Clin J Gastroenterol. 2021;14:1142-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 9. | Metcalf DR, Nivatvongs S, Andrews JC. Ischemic colitis: an unusual case of inferior mesenteric arteriovenous fistula causing venous hypertension. Report of a case. Dis Colon Rectum. 2008;51:1422-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 24] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 10. | Suh JW, Park J, Lee J, Yang IJ, Ahn HM, Oh HK, Kim DW, Kang SB. Clinical impact of inferior mesenteric vein preservation during left hemicolectomy with low ligation of the inferior mesenteric artery for distal transverse and descending colon cancers: A comparative study based on computed tomography. Front Oncol. 2022;12:986516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 11. | Fujii T, Toda S, Nishihara Y, Maeda Y, Hiramatsu K, Hanaoka Y, Sato R, Matoba S, Ueno M, Kuroyanagi H. Congestive ischemic colitis occurring after resection of left colon cancer: 4 case series. Surg Case Rep. 2020;6:175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 12. | Cotter TG, Bledsoe AC, Sweetser S. Colon Ischemia: An Update for Clinicians. Mayo Clin Proc. 2016;91:671-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 13. | Yadav S, Dave M, Edakkanambeth Varayil J, Harmsen WS, Tremaine WJ, Zinsmeister AR, Sweetser SR, Melton LJ 3rd, Sandborn WJ, Loftus EV Jr. A population-based study of incidence, risk factors, clinical spectrum, and outcomes of ischemic colitis. Clin Gastroenterol Hepatol. 2015;13:731-8.e1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 126] [Cited by in RCA: 103] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 14. | Reintam Blaser A, Mändul M, Björck M, Acosta S, Bala M, Bodnar Z, Casian D, Demetrashvili Z, D'Oria M, Durán Muñoz-Cruzado V, Forbes A, Fuglseth H, Hellerman Itzhaki M, Hess B, Kase K, Kirov M, Lein K, Lindner M, Loudet CI, Mole DJ, Murruste M, Nuzzo A, Saar S, Scheiterle M, Starkopf J, Talving P, Voomets AL, Voon KKT, Yunus MA, Tamme K; AMESI Investigators (Collaborators). Incidence, diagnosis, management and outcome of acute mesenteric ischaemia: a prospective, multicentre observational study (AMESI Study). Crit Care. 2024;28:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 48] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 15. | Zafirovski A, Zafirovska M, Kuhelj D, Pintar T. The Impact of Biomarkers on the Early Detection of Acute Mesenteric Ischemia. Biomedicines. 2023;12:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 16. | Harnik IG, Brandt LJ. Mesenteric venous thrombosis. Vasc Med. 2010;15:407-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 118] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 17. | Reintam Blaser A, Starkopf J, Björck M, Forbes A, Kase K, Kiisk E, Laisaar KT, Mihnovits V, Murruste M, Mändul M, Voomets AL, Tamme K. Diagnostic accuracy of biomarkers to detect acute mesenteric ischaemia in adult patients: a systematic review and meta-analysis. World J Emerg Surg. 2023;18:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 18. | Ageno W, Riva N, Schulman S, Beyer-Westendorf J, Bang SM, Senzolo M, Grandone E, Pasca S, Di Minno MN, Duce R, Malato A, Santoro R, Poli D, Verhamme P, Martinelli I, Kamphuisen P, Oh D, D'Amico E, Becattini C, De Stefano V, Vidili G, Vaccarino A, Nardo B, Di Nisio M, Dentali F. Long-term Clinical Outcomes of Splanchnic Vein Thrombosis: Results of an International Registry. JAMA Intern Med. 2015;175:1474-1480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 182] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 19. | Sakorafas GH, Zouros E, Peros G. Applied vascular anatomy of the colon and rectum: clinical implications for the surgical oncologist. Surg Oncol. 2006;15:243-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 20. | Kanasaki S, Furukawa A, Fumoto K, Hamanaka Y, Ota S, Hirose T, Inoue A, Shirakawa T, Hung Nguyen LD, Tulyeubai S. Acute Mesenteric Ischemia: Multidetector CT Findings and Endovascular Management. Radiographics. 2018;38:945-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 21. | Zeng Y, Yang F, Hu X, Zhu F, Chen W, Lin W. Radiological predictive factors of transmural intestinal necrosis in acute mesenteric ischemia: systematic review and meta-analysis. Eur Radiol. 2023;33:2792-2799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 22. | Ahmed M. Ischemic bowel disease in 2021. World J Gastroenterol. 2021;27:4746-4762. [PubMed] [DOI] [Full Text] |
| 23. | Ten Heggeler LB, van Dam LJ, Bijlsma A, Visschedijk MC, Geelkerken RH, Meijssen MA, Kolkman JJ. Colon ischemia: Right-sided colon involvement has a different presentation, etiology and worse outcome. A large retrospective cohort study in histology proven patients. Best Pract Res Clin Gastroenterol. 2017;31:111-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 24. | Clair DG, Beach JM. Mesenteric Ischemia. N Engl J Med. 2016;374:959-968. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 481] [Cited by in RCA: 360] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 25. | Sterne JA, Bodalia PN, Bryden PA, Davies PA, López-López JA, Okoli GN, Thom HH, Caldwell DM, Dias S, Eaton D, Higgins JP, Hollingworth W, Salisbury C, Savović J, Sofat R, Stephens-Boal A, Welton NJ, Hingorani AD. Oral anticoagulants for primary prevention, treatment and secondary prevention of venous thromboembolic disease, and for prevention of stroke in atrial fibrillation: systematic review, network meta-analysis and cost-effectiveness analysis. Health Technol Assess. 2017;21:1-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 121] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 26. | Davis JPE, Lim JK, Francis FF, Ahn J. AGA Clinical Practice Update on Management of Portal Vein Thrombosis in Patients With Cirrhosis: Expert Review. Gastroenterology. 2025;168:396-404.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 33] [Article Influence: 33.0] [Reference Citation Analysis (1)] |
| 27. | Stancu B, Chira A, Coman HF, Mihaileanu FV, Ciocan R, Gherman CD, Andercou OA. Intestinal Obstruction as Initial Presentation of Idiopathic Portal and Mesenteric Venous Thrombosis: Diagnosis, Management, and Literature Review. Diagnostics (Basel). 2024;14:304. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 28. | Grootjans J, Hundscheid IH, Lenaerts K, Boonen B, Renes IB, Verheyen FK, Dejong CH, von Meyenfeldt MF, Beets GL, Buurman WA. Ischaemia-induced mucus barrier loss and bacterial penetration are rapidly counteracted by increased goblet cell secretory activity in human and rat colon. Gut. 2013;62:250-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 86] [Article Influence: 6.6] [Reference Citation Analysis (3)] |
| 29. | Russell CE, Wadhera RK, Piazza G. Mesenteric venous thrombosis. Circulation. 2015;131:1599-1603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 30. | Rozner R, Gisriel S, Damianos J, Grimshaw AA, Rizwan R, Nawaz A, Chan K, Wan D, Pantel H, Bhutta AQ, Fenster M, Brandt LJ, Barbieri A, Robert ME, Feuerstadt P, Li DK. Idiopathic myointimal hyperplasia of the mesenteric veins: A systematic review and individual patient data regression analysis. J Gastroenterol Hepatol. 2023;38:1040-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 31. | Iwashita A, Yao T, Schlemper RJ, Kuwano Y, Yao T, Iida M, Matsumoto T, Kikuchi M. Mesenteric phlebosclerosis: a new disease entity causing ischemic colitis. Dis Colon Rectum. 2003;46:209-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 25] [Reference Citation Analysis (1)] |