Published online May 14, 2026. doi: 10.3748/wjg.v32.i18.116902
Revised: January 7, 2026
Accepted: February 24, 2026
Published online: May 14, 2026
Processing time: 161 Days and 16.4 Hours
Pediatric liver transplantation (pLT) has evolved from a purely life-saving inter
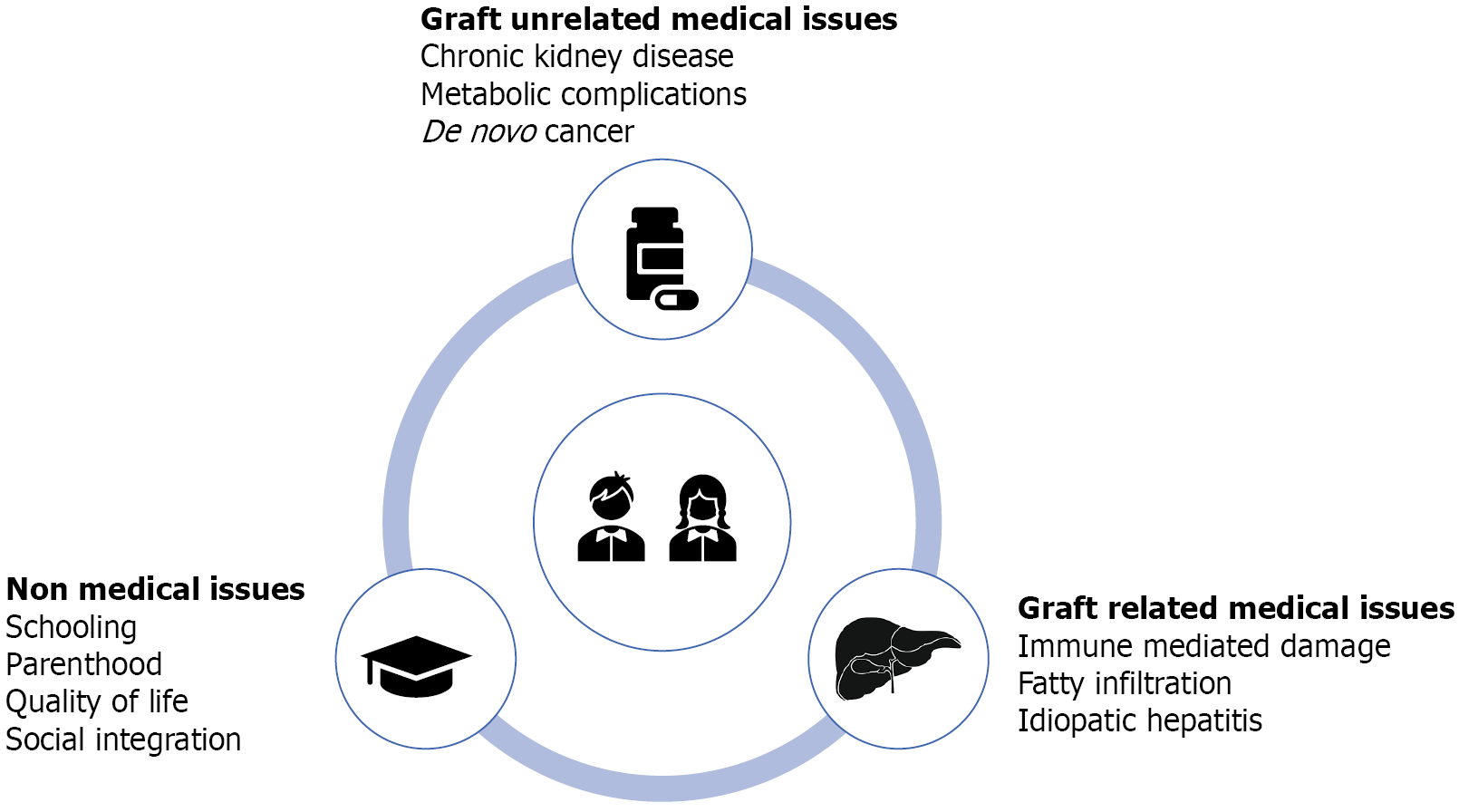
Core Tip: Pediatric liver transplantation has progressed from a life-saving procedure to one offering excellent long-term survival, with most recipients reaching adulthood. The focus has shifted from survival to global health, encompassing medical outcomes, psychological well-being, social integration, quality of life, and long-term functional and developmental success. However, only a minority achieve the ideal outcome, largely due to medical complications. Graft-related issues (such as persistent liver function test abnormalities) and extrahepatic complications, including chronic kidney disease, and metabolic disorders, and de novo cancers, often result from long-term immunosuppression. This minireview highlights determinants of long-term outcomes and proposes refinements to better assess the ideal outcome.
- Citation: Ferrarese A, Cananzi M, Bosa L, Senzolo M, Germani G, Vivian LM, Mescoli C, Dolcet A, Gringeri E, D’Antiga L, Spada M, Dias JA, Cillo U, Burra P. Outcomes of pediatric liver transplant recipients who reach adulthood: Current perspectives and unmet needs. World J Gastroenterol 2026; 32(18): 116902
- URL: https://www.wjgnet.com/1007-9327/full/v32/i18/116902.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i18.116902
Liver transplantation (LT) remains a life-saving therapy for a wide range of inherited and acquired pediatric diseases. Over recent decades, pediatric LT (pLT) programs have expanded worldwide, alongside the harmonization of organ allocation policies[1,2]. Two factors underpin the transplant benefit at pediatric age. First, pharmacological therapies for many pediatric conditions, including neoplastic, metabolic and cholestatic disorders, remain limited[3,4]. Second, out
In light of these considerations, there has been a growing need to develop composite scores capable of adequately measuring overall health during long-term follow-up, and enabling comparisons between cohorts. Analyzing a retro
| Variables associated with ideal outcome at 10 years follow-up[9] | Prevalence1 (%) | Variables associated with AYA-ideal outcome at long term follow-up[8] | Prevalence (%) |
| Graft related issues | |||
| No re-transplantation | 88 | No late re-transplantation2 | 76.7 |
| No chronic rejection | 91 | No chronic rejection | 93.3 |
| Normal liver enzymes (alanine aminotransferase) | 89 | Normal liver enzymes (alanine aminotransferase) | 66.7 |
| Normal liver enzymes (gamma-glutamyl transpeptidase) | 85 | Normal liver enzymes (gamma glutamyl transpeptidase) | 74.2 |
| Normal liver function (albumin) | 98 | Normal liver function (albumin) | 87.5 |
| Normal liver function (total bilirubin) | 99 | ||
| Immunosuppression-induced issues | |||
| No post-transplant lymphoproliferative disease | 94 | No malignancy2 | 95 |
| No renal dysfunction (i.e., GFR < 90 mL/minute/ | 91 | No renal dysfunction (i.e., GFR < 90 mL/minute/1.73 m2) | 61.7 |
| Acceptable linear growth | 93 | No arterial hypertension2 | 68.3 |
| No diabetes | 99 | No diabetes | 99.2 |
| Additional medications | |||
| No ongoing use of prednisone | 81 | No antiseizure medication | 97.5 |
| No use of antihypertensive agents | 87 | No treatment with steroids | 80.8 |
| No use of antiseizure medication | 100 | ||
Reaching long post-transplant follow-up often coincides with adolescence or young adulthood (AYA), a life phase characterized by increased vulnerability to non-adherence and significant changes in patients’ perceptions, behaviors, and priorities[10]. This period frequently overlaps with the transfer of care from pediatric to adult healthcare services, a critical phase marked by the loss of established relationships with pediatric healthcare professionals and the need to develop new therapeutic alliances within adult care settings[11]. A structured transition process may provide a safe framework to address both medical and non-medical challenges, thereby facilitating the achievement of overall health.
The primary aim of this narrative minireview is to describe the prevalence and risk factors of medical complications, both graft-related and extrahepatic, that may hinder the attainment of an ideal outcome after pLT, with particular attention to issues relevant to adult transplant hepatologists. A secondary aim is to highlight current challenges and future perspectives for each of these complications. To this end, all relevant manuscripts indexed in PubMed were reviewed, prioritizing multicenter, prospective studies published within the last 15 years, in order to provide an up-to-date overview of the available evidence.
In the long-term follow-up after pLT, the trajectories of patient and graft survival may diverge, indicating that a subset of recipients, particularly in AYA, may require late re-transplantation (re-LT). The leading indications of re-LT differ from those observed in adult LT recipients and are predominantly related to immune-mediated mechanisms. Consequently, the overarching goal of long-term follow-up is the preservation of graft function through the early identification of subclinical (immune-mediated) injury, and the prevention of its progression. Studies have shown that elevations of transaminases and γ-glutamyl transferase tests occur in approximately 15% of pLT recipients at 10 years post-transplant, doubling at 20 years[8,9,12,13]. This chronic increase also predicts graft fibrosis, which may in turn determine graft loss[14-17]. Persistent elevations of transaminases and γ-glutamyl transferase have therefore been incorporated among the items defining ideal outcome (Table 1).
Although elevations in liver function tests may reflect a variety of conditions, including fatty infiltration, de novo idiopathic or viral hepatitis, immune-mediated complications remain the most prevalent and clinically concerning. Among modifiable risk factors associated with the development of immune-mediated graft injury, non-adherence to immunosuppressive therapy plays a particularly critical role and is more prevalent among AYA recipients than in older LT populations. Fluctuations in immunosuppressive drug trough levels have been associated with both acute and chronic immune-mediated liver injury, further underscoring the importance of adherence monitoring[18].
A major priority is the development of internationally shared protocols for the assessment of graft function in the long-term follow-up, with the aim of enabling earlier detection of graft injury[19-21]. Although serum transaminases currently represent the main indicators used in routine clinical practice to monitor graft function, growing evidence indicates that a substantial proportion of liver allografts may harbor immune-mediated injury, such as subclinical acute cellular rejection, even in the presence of normal transaminase levels. In this setting, the application of biomarkers already used in adult transplantation, including donor-specific antibodies, may improve the diagnostic accuracy for silent graft injury and facilitate earlier diagnosis[22,23]. Moreover, the adoption of innovative approaches, such as genome-wide analyses and artificial intelligence-based platforms, may allow the identification of new patient-tailored biomarkers, both serological and histological, which could complement or potentially replace transaminases as indicators of allograft health in future composite scores of overall health[24,25].
Another aim for the near future will be to identify the true prevalence of fibrosis during fixed follow-up endpoints, and its prognostic impact. Although liver biopsy remains the gold standard for fibrosis assessment, serum biomarkers, transient elastography, and/or the combination thereof, could improve the longitudinal evaluation of fibrosis progression[26-29]. Furthermore, elucidating the relationship between the phenotype of the inflammatory tissue infiltrate and the concomitant presence of fibrosis could enable the identification of specific immunological therapeutic targets[30].
Another key question will be to assess the influence of immunosuppression minimization (up to its complete withdrawal) on graft injury, using dedicated tools that extend beyond conventional transaminase measurements. Pending the identification and validation of such biomarkers, close surveillance of allograft function remains essential, including the use of protocol liver biopsies in dedicated settings, which may guide timely and appropriate modulation of immunosuppressive therapy[31-33].
Incidence of post-pLT CKD is expected to rise with extended follow-up, affecting the long-term outcome, especially at deeper stages. Stage III CKD has been reported in 1%-3% of patients after pLT in recent studies. Liver disease etiology (e.g., Alagille syndrome, Wilson’s disease), pre-transplant renal impairment, older age at pLT and cyclosporine-based immunosuppression have been identified as associated risk factors[34-36]. Two studies specifically addressed the incidence of end-stage renal disease after pLT. In the study by Ruebner et al[37] on pLTs performed between 1990 and 2010, 2% of recipients developed this condition, after a median follow-up of 7.8 years. Notably, male gender, older age, viral hepatitis, late re-LT were the independent predictors of this condition, which significantly correlated with mortality. In another retrospective observational study, Umemura et al[38] showed a progressive decrease of renal function over time, highlighting a prevalence of end-stage renal disease up to 5.9% in subjects surviving more than 20 years after pLT.
The risk of CKD development during the long-term follow-up is largely due to calcineurin inhibitors (CNI) toxicity. To slow CKD progression while preserving graft function, protocols that minimize CNI exposure have been considered, including the off-label use of everolimus and minimization strategies with mycophenolic acid[39-41]. In a prospective study, 56 pLT recipients who started everolimus within 6 months post-transplant demonstrated a modest improvement of renal function (glomerular filtration rate increase of 6.2 mL/minute/1.73 m2 from baseline to month 12). However, 44% discontinued treatment due to adverse events[42]. A multicenter retrospective study evaluated everolimus or sirolimus use in various settings (e.g., nephrotoxicity, neoplasms, rejection) at a longer interval after pLT (median 3 years); in that study, the overall discontinuation rate was 23.3%. Although no significant renal benefit was observed in the whole cohort, creatinine levels remained stable in patients with pre-existing renal impairment[43]. Additional long-term strategies for preserving renal function include the early management of metabolic complications, such as hypertension, diabetes and obesity, which can themselves adversely impact kidney health. These complications may be overlooked more often in the pediatric population compared with adults, underscoring the need for vigilant long-term follow-up. Furthermore, the implementation of biomarkers such as cystatin C may be of help in diagnosis of CKD, alongside indirect formulas for estimating glomerular filtration rate that appropriately account for patient age. Once CKD develops, a nephrological assessment becomes essential to evaluate the degree of renal dysfunction and proteinuria, and to initiate targeted pharmacological therapies (e.g., angiotensin-converting enzyme inhibitors, or newer drugs such as sodium-glucose cotransporter-2 inhibitors) aimed at slowing disease progression[44]. Kidney transplantation in adolescence, young adulthood, or adulthood should be pursued for patients who develop ESRD following a previous pLT, while promoting organ-allocation strategies that do not disadvantage these patients compared with those without a history of transplan
The onset of DNC and its consequences have a significant impact on overall health, affecting long-term ideal outcomes after pLT[46]. Considering available epidemiological data, and comparing pLT recipients with age-matched non-transplanted controls, these patients exhibit a markedly higher prevalence of DNC, which justifies the implementation of dedicated surveillance policies for this at-risk population. Moreover, the relationship between DNC and time is crucial, as prevalence increases from 2%-5% to 15%-22% at 10 and 25 years after LT, respectively[47-49]. Therefore, it is likely that the development of DNC will be identified once patients have transitioned to the adult healthcare setting. When comparing pediatric and adult LT recipients, cancer types and outcomes differ substantially: Pediatric/AYA recipients exhibit a higher prevalence of lymphoproliferative disorders, whereas other cancers that are more typical of adults, such as gastrointestinal and lung cancers, are rare. Risk factors are less well characterized in pediatric recipients; however, long-term immunosuppression, Epstein-Barr virus serostatus, and age at transplant have been identified as key contri
Future challenges require larger datasets to better stratify oncological risk, particularly according to types of immunosuppression, and to personalize surveillance, especially in patients with comorbidities (e.g., primary sclerosing cholangitis, inflammatory bowel disease) or modifiable risk factors (such as smoking, alcohol intake and sexually transmitted infections)[51]. Such surveillance should target cancer types different from those seen in adult LT recipients and should be implemented especially during transition to adult healthcare, given that the risk of DNC increases over time. Regarding composite surveillance strategies, the field of biomarkers, including liquid biopsy and circulating tumor cells, holds significant potential in terms of diagnosis and prognosis[52]. Appropriate long-term immunosuppression management aimed at reducing excessive toxic exposures may help lower the risk of DNC. The antiproliferative effects of mammalian target of rapamycin inhibitors remain poorly characterized in the pLT/AYA setting and warrant further investigation. Moreover, the impact of immunosuppression withdrawal on the reduction of DNC cumulative incidence remains to be fully elucidated[33].
Metabolic complications are increasingly encountered in AYA pLT recipients. A systematic review reported a prevalence of diabetes, hypertriglyceridemia, hypertension and obesity in approximately 10%, 16%-50%, 4%-100%, 10%-60% of pLT recipients, respectively[53]. Overall, the wide heterogeneity in prevalence can be attributed to several factors, including study design, variability in diagnostic criteria, approaches to case identification (direct diagnosis vs inference from chronic medication use), and differences in the timing of assessment according to age at transplantation.
That said, growing evidence underscores that the prevalence of many of these complications is higher in pLT recipients compared with age- and sex-matched non-transplanted controls[54,55]. Risk factors for post-pLT metabolic complications include long-term immunosuppressive therapy, in particular CNI and corticosteroids. Notably, the prevalence of im
The significant clinical implications of these metabolic complications underscore the need for regular monitoring, early lifestyle interventions, and careful immunosuppressive management. Routine screening of metabolic complications to enable early diagnosis, even in the absence of risk factors commonly seen in adult LT recipients, is a crucial action, parti
Preventive strategies could include those already implemented in the general adolescent population to reduce modifiable risk factors, such as dietary patterns, physical activity levels, and limiting sedentary behaviors related to technology use[63]. In the whole pediatric population, obesity prevalence tends to be higher in pre-school and school-age children than in adolescents, likely due to differences in hormonal changes during puberty and greater psychological attention to weight control during adolescence[64]. More data on AYA pLT recipients are needed to understand whether long-term immunosuppressive therapy may alter this pattern, maintaining a rising trend also during adolescence.
Considering therapeutic approaches, a major challenge for the future will be to apply novel therapies for metabolic conditions, particularly diabetes and obesity, to AYA pLT recipients. Studies in both transplanted patients and adolescent populations have demonstrated the safety and effectiveness of novel molecules, paving the way for their potential large-scale use in patients who share both conditions[65-68].
A final aspect concerns adherence to chronic therapy, which often declines during adolescence. This can affect both immunosuppressive regimens and other long-term treatments, such as for diabetes. Fostering adherence through a multidimensional approach, including reducing the number of daily tablets, simplifying therapeutic algorithms, and providing psychological support, may represent an additional effective therapeutic strategy.
Neurological complications are a significant cause of morbidity and mortality early after pLT, and are reported in 8%-46% of patients. Seizures and encephalopathy are the most common, typically occurring in the early post-transplant period, whereas tremor, headaches, ataxia, and neuropathy usually develop later[69-72]. Common causes of seizures include metabolic derangements (electrolyte, hepatic, or renal abnormalities) and the use of immunosuppressive agents (such as cyclosporine and tacrolimus)[73]. Although often transient, these conditions may leave sequelae that require long-term chronic medications, for example anticonvulsants. Moreover, certain types of drug-resistant epilepsy may lead, even years later, to complications related to polypharmacy, interactions with immunosuppressive medications, reduced neurocognitive development, and a significant impairment in quality of life[74]. In the previously proposed scores of ideal outcome, the need for antiseizure medications was an important but uncommon factor. Indeed, in the study by Lund et al[8], only 2.5% of patients were still receiving such chronic therapy at the last follow-up, whereas in the study by Ng et al[9], none of the patients continued anticonvulsants. An important goal for the future will be to complement, or even replace, antiseizure medications with other indicators of chronic polypharmacy (hypertension, CKD, diabetes, dyslipidemia, insomnia, or depression), better suited to capturing the achievement of an ideal outcome in AYA pLT recipients. In this context, the number of agents used for each class, and the total number of daily medications could serve as a useful indicator of the patient’s overall health, potentially guiding future therapy simplification to improve adhe
Good short- and long-term survival after pLT is well established; accordingly, the focus of long-term follow-up has progressively shifted from survival to optimal long-term functioning[75]. Increased awareness of the specific needs of AYA pLT recipients among parents, healthcare professionals, and policymakers has contributed to improved outcomes; nevertheless, several unmet needs and challenges persist.
This minireview underscores that currently available parameters for outcome assessment remain suboptimal, both because they fail to comprehensively capture the concept of overall health and because they require updating in light of advances in medical knowledge. Proposed actions for implementation of overall health assessment within three domains (graft-related issues, medical complications, and non-medical parameters such as social integration, parenthood, and patient-reported outcomes) are summarized in Table 2. Adaptation to different socio-economic contexts and their periodic updates could be subsequent steps to ensure broad applicability of these scores and long-term relevance.
| Topic | Progress on the management of paediatric LT recipients reaching adulthood | Future actions |
| pLT overall health during and beyond transition | Recognition of overall health as the ideal outcome to be achieved in the long-term follow-up | Change of current scores or development of novel composite scores, preferably derived from multicenter studies, capable of capturing overall health from both medical and psychosocial perspectives |
| Long-term allograft health | Increased awareness of importance of monitoring allograft health, especially beyond transition; lack of universal association between normal transaminases and absence of graft inflammation/immune-mediated injury; variable degree of histologically proven fibrosis after pLT without significant rates of progression over time, according to studies in paired biopsies | Identification of novel biomarkers capable of enabling early detection of immune-mediated graft injury and fibrosis, as well as predicting long-term clinical outcomes; integration of graft function-related biomarkers into algorithms for the assessment of overall health during long-term follow-up, either alongside or as a replacement for conventional transaminase measurements |
| Extrahepatic complications affecting long-term overall health | Implementation of CNI sparing/minimization strategies to reduce the risk of long-term CKD development; increased awareness of metabolic syndrome as a potentially dangerous complication after pediatric LT; increased awareness about reproductive health (including sexual dysfunction, infertility, contraception) as a key topic to address in the long-term follow-up | Prevention and aggressive correction of modifiable risk factors for CKD development; development of tailored (universal) surveillance algorithms for de novo cancer; evaluation of safety and effectiveness of available drugs for the general population of young adult LT recipients with metabolic syndrome; evaluation of effectiveness and safety of artificial reproductive technologies even in the setting of young adult LT recipients |
| Transition | Increased awareness of the importance of transition among many pediatric and adult transplant hepatologists; development and validation of tools to assess readiness for transition; development of structured transition pathways in many transplant centers, managed by qualified healthcare professionals | Development of a universal transition protocol applicable in all LT centers, to be tailored according to local expertise and needs; greater integration of psychosocial support into transition programs; systematic application of reliable endpoints to evaluate the quality and effectiveness of the transition process; long-term, multicenter data on the outcomes of structured transition programs to guide best practices |
Finally, particular emphasis should be placed on the transition process from pediatric to adult care, which has been demonstrated to be effective in supporting AYA during this vulnerable life phase. Favorable long-term outcomes asso
| 1. | Spada M, Angelico R, Trapani S, Masiero L, Puoti F, Colledan M, Cintorino D, Romagnoli R, Cillo U, Cardillo M; Italian College of Liver Transplant Program. Tailoring allocation policies and improving access to paediatric liver transplantation over a 16-year period. J Hepatol. 2024;80:505-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 2. | Rodriguez-Davalos MI, Lopez-Verdugo F, Kasahara M, Muiesan P, Reddy MS, Flores-Huidobro Martinez A, Xia Q, Hong JC, Niemann CU, Seda-Neto J, Miloh TA, Yi NJ, Mazariegos GV, Ng VL, Esquivel CO, Lerut J, Rela M; Pediatric Liver Transplantation Global Census Group. International Liver Transplantation Society Global Census: First Look at Pediatric Liver Transplantation Activity Around the World. Transplantation. 2023;107:2087-2097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 3. | Vandriel SM, Li LT, She H, Wang JS, Gilbert MA, Jankowska I, Czubkowski P, Gliwicz-Miedzińska D, Gonzales EM, Jacquemin E, Bouligand J, Spinner NB, Loomes KM, Piccoli DA, D'Antiga L, Nicastro E, Sokal É, Demaret T, Ebel NH, Feinstein JA, Fawaz R, Nastasio S, Lacaille F, Debray D, Arnell H, Fischler B, Siew S, Stormon M, Karpen SJ, Romero R, Kim KM, Baek WY, Hardikar W, Shankar S, Roberts AJ, Evans HM, Jensen MK, Kavan M, Sundaram SS, Chaidez A, Karthikeyan P, Sanchez MC, Cavalieri ML, Verkade HJ, Lee WS, Squires JE, Hajinicolaou C, Lertudomphonwanit C, Fischer RT, Larson-Nath C, Mozer-Glassberg Y, Arikan C, Lin HC, Bernabeu JQ, Alam S, Kelly DA, Carvalho E, Ferreira CT, Indolfi G, Quiros-Tejeira RE, Bulut P, Calvo PL, Önal Z, Valentino PL, Desai DM, Eshun J, Rogalidou M, Dezsőfi A, Wiecek S, Nebbia G, Pinto RB, Wolters VM, Tamara ML, Zizzo AN, Garcia J, Schwarz K, Beretta M, Sandahl TD, Jimenez-Rivera C, Kerkar N, Brecelj J, Mujawar Q, Rock N, Busoms CM, Karnsakul W, Lurz E, Santos-Silva E, Blondet N, Bujanda L, Shah U, Thompson RJ, Hansen BE, Kamath BM; Global ALagille Alliance (GALA) Study Group. Natural history of liver disease in a large international cohort of children with Alagille syndrome: Results from the GALA study. Hepatology. 2023;77:512-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 65] [Article Influence: 21.7] [Reference Citation Analysis (1)] |
| 4. | Antala S, Taylor SA. Biliary Atresia in Children: Update on Disease Mechanism, Therapies, and Patient Outcomes. Clin Liver Dis. 2022;26:341-354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 5. | Elisofon SA, Magee JC, Ng VL, Horslen SP, Fioravanti V, Economides J, Erinjeri J, Anand R, Mazariegos GV; Society of Pediatric Liver Transplantation Research Group. Society of pediatric liver transplantation: Current registry status 2011-2018. Pediatr Transplant. 2020;24:e13605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 92] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 6. | Baumann U, Karam V, Adam R, Fondevila C, Dhawan A, Sokal E, Jacquemin E, Kelly DA, Grabhorn E, Pawlowska J, D'Antiga L, Jara Vega P, Debray D, Polak WG, de Ville de Goyet J, Verkade HJ; European Liver and Intestine Transplant Association (ELITA) and all ELTR contributing centers. Prognosis of Children Undergoing Liver Transplantation: A 30-Year European Study. Pediatrics. 2022;150:e2022057424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 7. | Martinelli J, Habes D, Majed L, Guettier C, Gonzalès E, Linglart A, Larue C, Furlan V, Pariente D, Baujard C, Branchereau S, Gauthier F, Jacquemin E, Bernard O. Long-term outcome of liver transplantation in childhood: A study of 20-year survivors. Am J Transplant. 2018;18:1680-1689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 69] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 8. | Lund LK, Grabhorn EF, Rüther D, Buchholz A, Lang M, Herden U, Fischer L, Sterneck M. Long-term Outcome of Pediatric Liver Transplant Recipients Who Have Reached Adulthood: A Single-center Experience. Transplantation. 2023;107:1756-1763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 9. | Ng VL, Alonso EM, Bucuvalas JC, Cohen G, Limbers CA, Varni JW, Mazariegos G, Magee J, McDiarmid SV, Anand R; Studies of Pediatric Liver Transplantation (SPLIT) Research Group. Health status of children alive 10 years after pediatric liver transplantation performed in the US and Canada: report of the studies of pediatric liver transplantation experience. J Pediatr. 2012;160:820-6.e3. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 210] [Cited by in RCA: 194] [Article Influence: 13.9] [Reference Citation Analysis (1)] |
| 10. | Burra P. The adolescent and liver transplantation. J Hepatol. 2012;56:714-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 11. | Lawrence ZE, Martinez M, Lobritto S, Chen J, Breslin N, Fox A, Vittorio J. Adherence, Medical Outcomes, and Health Care Costs in Adolescents/Young Adults Following Pediatric Liver Transplantation. J Pediatr Gastroenterol Nutr. 2020;70:183-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 33] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 12. | Kelly D, Verkade HJ, Rajanayagam J, McKiernan P, Mazariegos G, Hübscher S. Late graft hepatitis and fibrosis in pediatric liver allograft recipients: Current concepts and future developments. Liver Transpl. 2016;22:1593-1602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 85] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 13. | Montano-Loza AJ, Rodríguez-Perálvarez ML, Pageaux GP, Sanchez-Fueyo A, Feng S. Liver transplantation immunology: Immunosuppression, rejection, and immunomodulation. J Hepatol. 2023;78:1199-1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 137] [Article Influence: 45.7] [Reference Citation Analysis (0)] |
| 14. | Evans HM, Kelly DA, McKiernan PJ, Hübscher S. Progressive histological damage in liver allografts following pediatric liver transplantation. Hepatology. 2006;43:1109-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 222] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 15. | Ekong UD, Melin-Aldana H, Seshadri R, Lokar J, Harris D, Whitington PF, Alonso EM. Graft histology characteristics in long-term survivors of pediatric liver transplantation. Liver Transpl. 2008;14:1582-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 96] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 16. | Venturi C, Sempoux C, Bueno J, Ferreres Pinas JC, Bourdeaux C, Abarca-Quinones J, Rahier J, Reding R. Novel histologic scoring system for long-term allograft fibrosis after liver transplantation in children. Am J Transplant. 2012;12:2986-2996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 102] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 17. | Rhu J, Ha SY, Lee S, Kim JM, Choi GS, Joh JW, Lee SK. Risk factors of silent allograft fibrosis 10 years post-pediatric liver transplantation. Sci Rep. 2020;10:1833. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 18. | Ferrarese A, Germani G, Lazzaro S, Cananzi M, Russo FP, Senzolo M, Gambato M, Zanetto A, Cillo U, Gringeri E, Perilongo G, Burra P. Short-term outcomes of paediatric liver transplant recipients after transition to Adult Healthcare Service. Liver Int. 2018;38:1316-1321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Feng S, Bucuvalas JC, Demetris AJ, Burrell BE, Spain KM, Kanaparthi S, Magee JC, Ikle D, Lesniak A, Lozano JJ, Alonso EM, Bray RA, Bridges NE, Doo E, Gebel HM, Gupta NA, Himes RW, Jackson AM, Lobritto SJ, Mazariegos GV, Ng VL, Rand EB, Sherker AH, Sundaram S, Turmelle YP, Sanchez-Fueyo A. Evidence of Chronic Allograft Injury in Liver Biopsies From Long-term Pediatric Recipients of Liver Transplants. Gastroenterology. 2018;155:1838-1851.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 20. | Pinon M, Pizzol A, Chiadò C, David E, Chiusa L, Dell'Olio D, Isolato G, Amoroso A, Deaglio S, Catalano S, Tandoi F, Romagnoli R, Calvo PL. Evaluation of Graft Fibrosis, Inflammation, and Donor-specific Antibodies at Protocol Liver Biopsies in Pediatric Liver Transplant Patients: A Single-center Experience. Transplantation. 2022;106:85-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 21. | Ohlsson S, Kathemann S, Pilic D, Prusinskas B, Baba HA, Theurer S, Dechene A, Paul A, Heinold A, Hoyer PF, Lainka E. Protocol liver biopsies in stable long-term pediatric liver transplant recipients: risk or benefit? Eur J Gastroenterol Hepatol. 2021;33:e223-e232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 22. | Quintero-Bernabeu J, Salcedo-Allende MT, Juamperez-Goñi J, Mercadal-Hally M, Padrós-Fornieles C, Larrarte-King M, Mameli S, Molino-Gahete JA, Coma-Muñoz A, Moreno-Galdo A. Histological findings in follow-up liver biopsies up to 15 years after pediatric liver transplantation: Associations with subclinical rejection, fibrosis, and immunological markers. Liver Transpl. 2026;32:529-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Kanaan E, Ohlsson S, Kathemann S, Prusinskas B, Tsaka S, Heinemann FM, Heinold A, Schulze M, Pape L, Lainka E. Assessment of Donor-Specific Human Leukocyte Antigen Antibodies Following Pediatric Liver Transplantation: Predictors, Protectors, and Clinical Relevance. Clin Transplant. 2025;39:e70390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 24. | Vinciguerra T, Brunati A, David E, Longo F, Pinon M, Ricceri F, Castellino L, Piga A, Giraudo MT, Tandoi F, Cisarò F, Dell Olio D, Isolato G, Romagnoli R, Salizzoni M, Calvo PL. Transient elastography for non-invasive evaluation of post-transplant liver graft fibrosis in children. Pediatr Transplant. 2018;22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 25. | Chanpong A, Angkathunyakul N, Sornmayura P, Tanpowpong P, Lertudomphonwanit C, Panpikoon T, Treepongkaruna S. Late allograft fibrosis in pediatric liver transplant recipients: Assessed by histology and transient elastography. Pediatr Transplant. 2019;23:e13541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 26. | Lv Z, Yong JK, Liu Y, Zhou Y, Pan Y, Xiang X, Li L, Wang Y, Zhao Y, Liu Z, Zhang Z, Xia Q, Feng H. A blood-based PT-LIFE (Pediatric Liver Transplantation-LIver Fibrosis Evaluation) biomarker panel for noninvasive evaluation of pediatric liver fibrosis after liver transplantation: A prospective derivation and validation study. Am J Transplant. 2025;25:501-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 27. | Lee CK, Nastasio S, Mitchell PD, Fawaz R, Elisofon SA, Vakili K, Kim HB, Nguyen D, Jonas MM. Transient elastography assessment of liver allograft fibrosis in pediatric transplant recipients. Pediatr Transplant. 2020;24:e13736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 28. | Perito ER, Persyn E, Bucuvalas J, Martinez M, Mohammad S, Squires JE, Demetris AJ, Feng S. Graft Fibrosis Over 10 to 15 Years in Pediatric Liver Transplant Recipients: Multicenter Study of Paired, Longitudinal Surveillance Biopsies. Liver Transpl. 2022;28:1051-1062. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 29. | Cheng K, Feng S, Bucuvalas JC, Levitsky J, Perito ER. Not everything that counts can be counted: Tracking long-term outcomes in pediatric liver transplant recipients. Am J Transplant. 2022;22:1182-1190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 30. | Cousin VL, Rougemont AL, Rubbia-Brandt L, Wildhaber BE, Villard J, Ferrari-Lacraz S, McLin VA. Peripheral Donor-specific Antibodies Are Associated With Histology and Cellular Subtypes in Protocol Liver Biopsies of Pediatric Recipients. Transplantation. 2020;104:1633-1643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 31. | Uebayashi EY, Okajima H, Uno S, Kadohisa M, Yamamoto M, Ogawa E, Okamoto T, Okumura S, Ogiso S, Ito T, Takeuchi Y, Hirata M, Yamada Y, Minamiguchi S, Hatano E, Haga H. Thirty-year follow-up of immunosuppression modulation: Impact on graft fibrosis and anti-HLA antibodies after pediatric liver transplantation. Liver Transpl. 2026;32:243-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 32. | Feng S, Bucuvalas JC, Mazariegos GV, Magee JC, Sanchez-Fueyo A, Spain KM, Lesniak A, Kanaparthi S, Perito E, Venkat VL, Burrell BE, Alonso EM, Bridges ND, Doo E, Gupta NA, Himes RW, Ikle D, Jackson AM, Lobritto SJ, Jose Lozano J, Martinez M, Ng VL, Rand EB, Sherker AH, Sundaram SS, Turmelle YP, Wood-Trageser M, Demetris AJ. Efficacy and Safety of Immunosuppression Withdrawal in Pediatric Liver Transplant Recipients: Moving Toward Personalized Management. Hepatology. 2021;73:1985-2004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 33. | Boillot O, Wischlen E, Dubois V, Chopinet S, Laverdure N, Hervieu V, Collardeau-Frachon S, Rivet C, Lachaux A, Dumortier J. Dynamics and risk factors for long term pediatric liver graft fibrosis progression according to protocol liver biopsies over a 30-years follow-up. Dig Liver Dis. 2025;57:2419-2427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 34. | Sato M, Kaneko T, Ogura M, Kamei K, Ito S, Fukuda A, Sakamoto S, Kasahara M, Ishikura K. Favorable Kidney Function in Pediatric Liver Transplant Recipients: Results of a Single-center Cohort Study. Transplantation. 2019;103:1655-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 35. | Campbell K, Ng V, Martin S, Magee J, Goebel J, Anand R, Martz K, Bucuvalas J; SPLIT Renal Function Working Group. Glomerular filtration rate following pediatric liver transplantation--the SPLIT experience. Am J Transplant. 2010;10:2673-2682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 36. | Mention K, Lahoche-Manucci A, Bonnevalle M, Pruvot FR, Declerck N, Foulard M, Gottrand F. Renal function outcome in pediatric liver transplant recipients. Pediatr Transplant. 2005;9:201-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 37. | Ruebner RL, Reese PP, Denburg MR, Rand EB, Abt PL, Furth SL. Risk factors for end-stage kidney disease after pediatric liver transplantation. Am J Transplant. 2012;12:3398-3405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 38. | Umemura K, Mita A, Ohno Y, Masuda Y, Yoshizawa K, Kubota K, Notake T, Hosoda K, Kamachi A, Goto T, Tomida H, Yamazaki S, Shimizu A, Soejima Y. Late-onset Chronic Kidney Disease Over 2 Decades After Pediatric Liver Transplantation: A Single-center, Retrospective Study. Transplantation. 2023;107:1535-1544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 39. | Matloff RG, Arnon R, Saland JM. The kidney in pediatric liver transplantation: an updated perspective. Pediatr Transplant. 2012;16:818-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (3)] |
| 40. | Tannuri U, Gibelli NE, Maksoud-Filho JG, Santos MM, Pinho-Apezzato ML, Velhote MC, Ayoub AA, Silva MM, Maksoud JG. Mycophenolate mofetil promotes prolonged improvement of renal dysfunction after pediatric liver transplantation: experience of a single center. Pediatr Transplant. 2007;11:82-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 41. | Evans HM, McKiernan PJ, Kelly DA. Mycophenolate mofetil for renal dysfunction after pediatric liver transplantation. Transplantation. 2005;79:1575-1580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 42. | Ganschow R, Ericzon BG, Dhawan A, Sharif K, Martzloff ED, Rauer B, Ng J, Lopez P. Everolimus and reduced calcineurin inhibitor therapy in pediatric liver transplant recipients: Results from a multicenter, prospective study. Pediatr Transplant. 2017;21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 2.4] [Reference Citation Analysis (2)] |
| 43. | Dumortier J, Couchonnal E, Lacaille F, Rivet C, Debray D, Boillot O, Lachaux A, Ackermann O, Gonzales E, Wildhaber BE, Jacquemin E, McLin V. mTOR inhibitors in pediatric liver transplant recipients. Clin Res Hepatol Gastroenterol. 2019;43:403-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 44. | Smoyer WE, Gillespie BS, Oni L, Nester C, Nelson RG, Bjornstad P, Heerspink HJL, Denburg M, Gross O, Marquard J, Steubl D, Lim MD, Creighton C, Balogun S, Eva L, Helm K, Tarnoff JM, Tuchman S, Khurana M, Mulugeta L, Yao L, Mistry K, Thompson A, Trachtman H. Strategies for the development of sodium-glucose cotransporter-2 inhibitors for kidney protection in pediatric chronic kidney disease: proceedings of a workshop meeting in July 2023. Kidney Int. 2026;109:57-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 45. | Husain SA, King KL, Owen-Simon NL, Fernandez HE, Ratner LE, Mohan S. Access to kidney transplantation among pediatric candidates with prior solid organ transplants in the United States. Pediatr Transplant. 2022;26:e14303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 46. | Nathan PC, Lau C, Ng VL, Miserachs M, Teoh CW, Solomon M, Dipchand AI, Locke M, Gupta S. Outcomes after solid organ transplantation in survivors of childhood, adolescent, and young adult cancer: a population-based study. J Natl Cancer Inst. 2025;117:1809-1816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 47. | Åberg F, Isoniemi H, Pukkala E, Jalanko H, Rasmussen A, Storm HH, Schultz N, Bennet W, Ekvall N, Ericzon BG, Malenicka S, Tretli S, Line PD, Boberg KM, Østensen A, Karlsen TH, Nordin A. Cancer After Liver Transplantation in Children and Young Adults: A Population-Based Study From 4 Nordic Countries. Liver Transpl. 2018;24:1252-1259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 48. | Kitchlu A, Dixon S, Dirk JS, Chanchlani R, Vasilevska-Ristovska J, Borges K, Dipchand AI, Ng VL, Hebert D, Solomon M, Michael Paterson J, Gupta S, Joseph Kim S, Nathan PC, Parekh RS. Elevated Risk of Cancer After Solid Organ Transplant in Childhood: A Population-based Cohort Study. Transplantation. 2019;103:588-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 49. | Robinson C, Chanchlani R, Kitchlu A. Malignancies after pediatric solid organ transplantation. Pediatr Nephrol. 2021;36:2279-2291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 50. | Quintero Bernabeu J, Juamperez J, Mercadal-Hally M, Larrarte King M, Gallego Melcon S, Gros Subias L, Sábado Álvarez C, Soler-Palacin P, Melendo Pérez S, Esperalba J, Navarro Jiménez A, Garrido Pontnou M, Camacho Soriano J, Hidalgo Llompart E, Bilbao Aguirre I, Charco Torra R. Epstein-Barr virus-associated risk factors for post-transplant lymphoproliferative disease in pediatric liver transplant recipients. Pediatr Transplant. 2022;26:e14292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 51. | Rela M, Quintero J, Kasahara M, Muiesan P, Hernández-Oliveros F, Rajalingam R, Shankar S, Sayed BA, di Sabato D, Rammohan A, Fung J, Bilbao I. Nonhepatic Cancer in the Pediatric Liver Transplant Population: Guidelines From the ILTS-SETH Consensus Conference. Transplantation. 2022;106:e46-e51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 52. | Sundby RT, Pan A, Shern JF. Liquid biopsies in pediatric oncology: opportunities and obstacles. Curr Opin Pediatr. 2022;34:39-47. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 53. | Rothbaum Perito E, Lau A, Rhee S, Roberts JP, Rosenthal P. Posttransplant metabolic syndrome in children and adolescents after liver transplantation: a systematic review. Liver Transpl. 2012;18:1009-1028. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 54. | Trier C, Schaffalitzky de Muckadell V, Borgwardt L, Rasmussen A, Hørby Jørgensen M. Markers of obesity in Danish pediatric liver transplantation recipients. Pediatr Transplant. 2022;26:e14320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 55. | Dagher M, Ng VL, Carpenter A, Rankin S, De Angelis M, Avitzur Y, Mouzaki M. Overweight, central obesity, and cardiometabolic risk factors in pediatric liver transplantation. Pediatr Transplant. 2015;19:175-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 56. | Perito ER, Lustig RH, Rosenthal P. Metabolic Syndrome Components After Pediatric Liver Transplantation: Prevalence and the Impact of Obesity and Immunosuppression. Am J Transplant. 2016;16:1909-1916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 57. | Perito ER, Mohammad S, Rosenthal P, Alonso EM, Ekong UD, Lobritto SJ, Feng S. Posttransplant metabolic syndrome in the withdrawal of immunosuppression in Pediatric Liver Transplant Recipients (WISP-R) pilot trial. Am J Transplant. 2015;15:779-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 58. | Regelmann MO, Goldis M, Arnon R. New-onset diabetes mellitus after pediatric liver transplantation. Pediatr Transplant. 2015;19:452-459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 59. | Vafaei F, Dehghani SM, Malekhoseini SA, Karamifar H, Nikeghbalian S. Prevalence of Postoperation Metabolic Syndrome in Pediatric Liver Transplant Patients: A Single Center Experience. Exp Clin Transplant. 2018;16:582-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 60. | Hathout E, Alonso E, Anand R, Martz K, Imseis E, Johnston J, Lopez J, Chinnock R, McDiarmid S; SPLIT study group. Post-transplant diabetes mellitus in pediatric liver transplantation. Pediatr Transplant. 2009;13:599-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 37] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 61. | Bellini MI, Lauro A, D'Andrea V, Marino IR. Pediatric Liver Transplantation: Long-Term Follow-Up Issues. Exp Clin Transplant. 2022;20:27-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 62. | Black DS, Vidmar AP, Barnett B. Characterising the design and methods of continuous glucose monitoring used in behavioural interventions to inform future research in prediabetes. Lancet Digit Health. 2025;7:100904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 63. | Lister NB, Baur LA, Felix JF, Hill AJ, Marcus C, Reinehr T, Summerbell C, Wabitsch M. Child and adolescent obesity. Nat Rev Dis Primers. 2023;9:24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 406] [Cited by in RCA: 324] [Article Influence: 108.0] [Reference Citation Analysis (0)] |
| 64. | Zhang X, Liu J, Ni Y, Yi C, Fang Y, Ning Q, Shen B, Zhang K, Liu Y, Yang L, Li K, Liu Y, Huang R, Li Z. Global Prevalence of Overweight and Obesity in Children and Adolescents: A Systematic Review and Meta-Analysis. JAMA Pediatr. 2024;178:800-813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 393] [Cited by in RCA: 310] [Article Influence: 155.0] [Reference Citation Analysis (0)] |
| 65. | Weghuber D, Barrett T, Barrientos-Pérez M, Gies I, Hesse D, Jeppesen OK, Kelly AS, Mastrandrea LD, Sørrig R, Arslanian S; STEP TEENS Investigators. Once-Weekly Semaglutide in Adolescents with Obesity. N Engl J Med. 2022;387:2245-2257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 560] [Cited by in RCA: 451] [Article Influence: 112.8] [Reference Citation Analysis (6)] |
| 66. | Hannon TS, Chao LC, Barrientos-Pérez M, Pamidipati KC, Landó LF, Lee CJ, Patel H, Bergman BK. Efficacy and safety of tirzepatide in children and adolescents with type 2 diabetes (SURPASS-PEDS): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2025;406:1484-1496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 67. | Chow KW, Ibrahim B, Rahal K, Hsu RH, Tan T, Meneses K, Saab S. Semaglutide is effective in achieving weight loss in liver transplant recipients. Liver Transpl. 2024;30:223-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 68. | Yakubu I, Spengler J, Taylor P, LaPorte M, Brown A, Sterling S, Agegnehu B, Iaria A, Marks R, Sprague T, Pontinha V, Patel V, Patidar KR, Siddiqui MS. Impact of Glucagon-like Peptide-1 Receptor Agonists on Metabolic Health in Liver Transplant Recipients. Transplantation. 2025;109:e501-e507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 69. | Lee YJ, Yum MS, Kim EH, Choi HW, Oh SH, Kim DY, Kim KM, Ko TS. Risk factors for neurological complications and their correlation with survival following pediatric liver transplantation. Pediatr Transplant. 2014;18:177-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 70. | Gungor S, Kilic B, Arslan M, Selimoglu MA, Karabiber H, Yilmaz S. Early and late neurological complications of liver transplantation in pediatric patients. Pediatr Transplant. 2017;21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 71. | Menon J, Shanmugam N, Rammohan A, Hakeem A, Reddy MS, Rela M. Neurological complications in pediatric liver transplant recipients. Pediatr Transplant. 2022;26:e14376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 72. | Piñero F, Cheang Y, Mendizabal M, Cagliani J, Gonzalez Campaña A, Pages J, Colaci C, Barreiro M, Alonso C, Malla I, Fauda M, Bueri J, Podesta LG, Silva M. Incidence, risk factors, and outcomes related with neurological events after liver transplantation in adult and pediatric recipients. Pediatr Transplant. 2018;22:e13159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 73. | Ueno T, Toyama C, Deguchi K, Masahata K, Nomura M, Watanabe M, Kamiyama M, Tazuke Y, Bessho K, Okuyama H. Long-Term Outcome After Tacrolimus-Related Neurotoxicity in Pediatric Living Donor Liver Transplantation. Transplant Proc. 2022;54:468-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 74. | Strzelczyk A, Zuberi SM, Striano P, Rosenow F, Schubert-Bast S. The burden of illness in Lennox-Gastaut syndrome: a systematic literature review. Orphanet J Rare Dis. 2023;18:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 49] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 75. | Ng VL, Mazariegos GV, Kelly B, Horslen S, McDiarmid SV, Magee JC, Loomes KM, Fischer RT, Sundaram SS, Lai JC, Te HS, Bucuvalas JC. Barriers to ideal outcomes after pediatric liver transplantation. Pediatr Transplant. 2019;23:e13537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 34] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 76. | Kosmach-Park B, Coyne B, Gupta N, Mazariegos G. Bridging the Gap: A Review of Pediatric to Adult Transition of Care in Liver Transplantation. Pediatr Transplant. 2025;29:e14900. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 77. | Culnane E, Loftus H, Peters R, Haydar M, Hodgson A, Herd L, Hardikar W. Enabling successful transition-Evaluation of a transition to adult care program for pediatric liver transplant recipients. Pediatr Transplant. 2022;26:e14213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 78. | Mayer K, Junge N, Goldschmidt I, Leiskau C, Becker T, Lehner F, Richter N, van Dick R, Baumann U, Pfister ED. Psychosocial outcome and resilience after paediatric liver transplantation in young adults. Clin Res Hepatol Gastroenterol. 2019;43:155-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 79. | Vittorio J, Kosmach-Park B, King LY, Fischer R, Fredericks EM, Ng VL, Narang A, Rasmussen S, Bucuvalas J. Health Care Transition for Adolescents and Young Adults With Pediatric-Onset Liver Disease and Transplantation: A Position Paper by the North American Society of Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr. 2023;76:84-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |