Published online May 14, 2026. doi: 10.3748/wjg.v32.i18.116018
Revised: December 4, 2025
Accepted: February 25, 2026
Published online: May 14, 2026
Processing time: 187 Days and 0.6 Hours
Tumor enucleation via thoracoscopic surgery (TS) and submucosal tunneling endoscopic resection are both effective and widely used therapies for resecting most esophageal subepithelial lesions (SELs) originating from the muscularis propria (MP). However, when a lesion exceeds 35 mm in diameter and/or is located in the cervical esophagus, it is classified as a complex esophageal tumor. In such cases, submucosal tunneling endoscopic resection is not feasible due to insufficient tunnel space and the inability to successfully extract the lesion from the tunnel. Exposed endoscopic full-thickness resection (EFTR), which does not require tunnel creation, is a potential alternative treatment for complex esopha
To compare the feasibility and safety of esophageal exposed EFTR with TS for the resection of complex esophageal SELs.
Between November 2016 and October 2023, the clinical records of patients with esophageal SELs-MP who underwent resection at the First Affiliated Hospital of Zhengzhou University were retrospectively reviewed. Patients with lesions larger than 35 mm in diameter and/or located in the cervical esophagus were included in the study. Clinicopathological characteristics, perioperative outcomes, complications, and follow-up data were col
A total of 60 patients with complex esophageal SELs-MP were included, with 15 patients in the EFTR group and 45 in the TS group. The EFTR and TS groups demonstrated comparable technical success rates (100% vs 97.8%, P = 0.574) and en bloc resection rates (86.7% vs 75.6%, P = 0.423). Compared to the TS group, the EFTR group had a significantly longer median procedure time (240.0 minutes vs 120.0 minutes, P < 0.001) but a shorter postoperative nasogastric decompression period (5.6 ± 4.9 days vs 10.7 ± 13.2 days, P = 0.016). In the EFTR group, complete defect closure was achieved in 3 patients (20.0%), incomplete closure in 8 patients (53.3%), and non-closure in 4 patients (26.7%). Postoperative adverse events occurred in 3 patients in the EFTR group and 4 patients in the TS group. Both groups experienced cases of esophageal stricture and fistula. Notably, chylothorax was observed exclusively in the TS group.
Esophageal-exposed EFTR demonstrated clinical outcomes comparable to those of TS, suggesting that it is a safe and feasible option for the treatment of complex esophageal SEL-MP. Further studies are warranted to validate these findings.
Core Tip: Esophageal exposed endoscopic full-thickness resection (EFTR) proved to be a safe procedure for complex esophageal subepithelial lesions-muscularis propria. Postoperative adverse events were conservatively managed and occurred at an acceptable rate. Exposed EFTR achieved 100% technical success and curative resection. Even for large tumors requiring piecemeal resection for retrieval, patient outcomes were not compromised. Patients undergoing esophageal exposed EFTR demonstrated faster recovery, evidenced by earlier initiation of oral intake and shorter nasogastric intubation durations.
- Citation: Li XX, Zheng QF, Garcia AM, Shi ZY, Shi M, Islam MS, Liu D. Exposed endoscopic full-thickness resection vs thoracoscopic surgery for complex esophageal subepithelial lesions arising from the muscularis propria. World J Gastroenterol 2026; 32(18): 116018
- URL: https://www.wjgnet.com/1007-9327/full/v32/i18/116018.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i18.116018
Esophageal subepithelial lesions (SELs) originate from the muscularis mucosa, submucosa, or muscularis propria (MP) and are predominantly benign. Among them, leiomyomas, mesenchymal tumors, and lipomas are the most common pathologic types[1]. The detection rate of esophageal SELs has risen markedly in recent years, primarily due to the widespread adoption of gastrointestinal endoscopy and advances in endoscopic ultrasound. Esophageal SELs smaller than 2 cm are generally considered low risk and require only close surveillance. However, depending on the size of the lesion and the extent of its extension into the esophageal lumen, symptoms such as dysphagia and thoracic pain may occur, and resection may be recommended[2]. Treatment options include endoscopic and surgical procedures, including esophagectomy, and the choice of treatment should depend on lesion size, location, and local expertise[3].
Surgical enucleation is an effective treatment for most SELs, and Mutrie et al[4] reported that it is technically feasible in up to 97 % of cases. In the past, open transthoracic enucleation of esophageal SELs was associated with significant adverse events. However, with the advent of minimally invasive procedures, such as video-assisted thoracoscopic surgery (TS), postoperative complications and mortality have decreased significantly. In recent years, with the rapid development of endoscopic techniques, endoscopic resection has become a standard approach for removing SELs, including transmural lesions, offering a less invasive and safer alternative. Accepted endoscopic techniques include endoscopic submucosal resection, endoscopic submucosal dissection, submucosal tunneling endoscopic resection (STER), and endoscopic full-thickness resection (EFTR). Among these, STER is a highly feasible and safe treatment for lesions arising from the MP because it maintains mucosal integrity over the SEL, thereby reducing the risk of perforation[5]. However, STER is technically challenging for large SELs-MP or for lesions located in the cervical esophagus because the excised tumor may not be retrievable from the tunnel, the submucosal tunneling space is limited, and endoscopic maneuverability is restricted[6]. In such cases, tumor enucleation by TS is often the preferred approach. Nevertheless, EFTR offers an alternative to STER by enabling en bloc, curative resection without the need for a submucosal tunnel. Although EFTR is widely performed for gastric and colorectal tumors[7-11], its application to esophageal SELs-MP remains limited. Therefore, in this study, we aimed to compare the feasibility and safety of esophageal exposed EFTR with TS for the resection of complex esophageal SELs.
We retrospectively analyzed 328 patients with SEL-MP who underwent resection at the First Affiliated Hospital of Zhengzhou University between November 2016 and October 2023. The inclusion criteria were as follows: (1) Preoperative endoscopic ultrasound confirmed that the esophageal SEL originated from the MP; and (2) The lesion was > 35 mm in diameter or located in the cervical esophagus, regardless of size. The exclusion criteria: (1) Age < 18 years; and (2) Endoscopic resection not performed as exposed EFTR or surgical resection not performed as TS.
Of the 328 patients with esophageal SELs-MP, 159 had lesions located in the cervical esophagus or measuring > 35 mm; of these, 82 underwent surgical resection and 77 underwent endoscopic resection. We then screened patients who underwent TS or EFTR. Ultimately, 60 patients met the inclusion criteria and were allocated to the EFTR (n = 15) and TS (n = 45) groups. Clinicopathological characteristics, perioperative outcomes, complications, and follow-up data were collected. The flowchart of enrollment is shown in Figure 1.
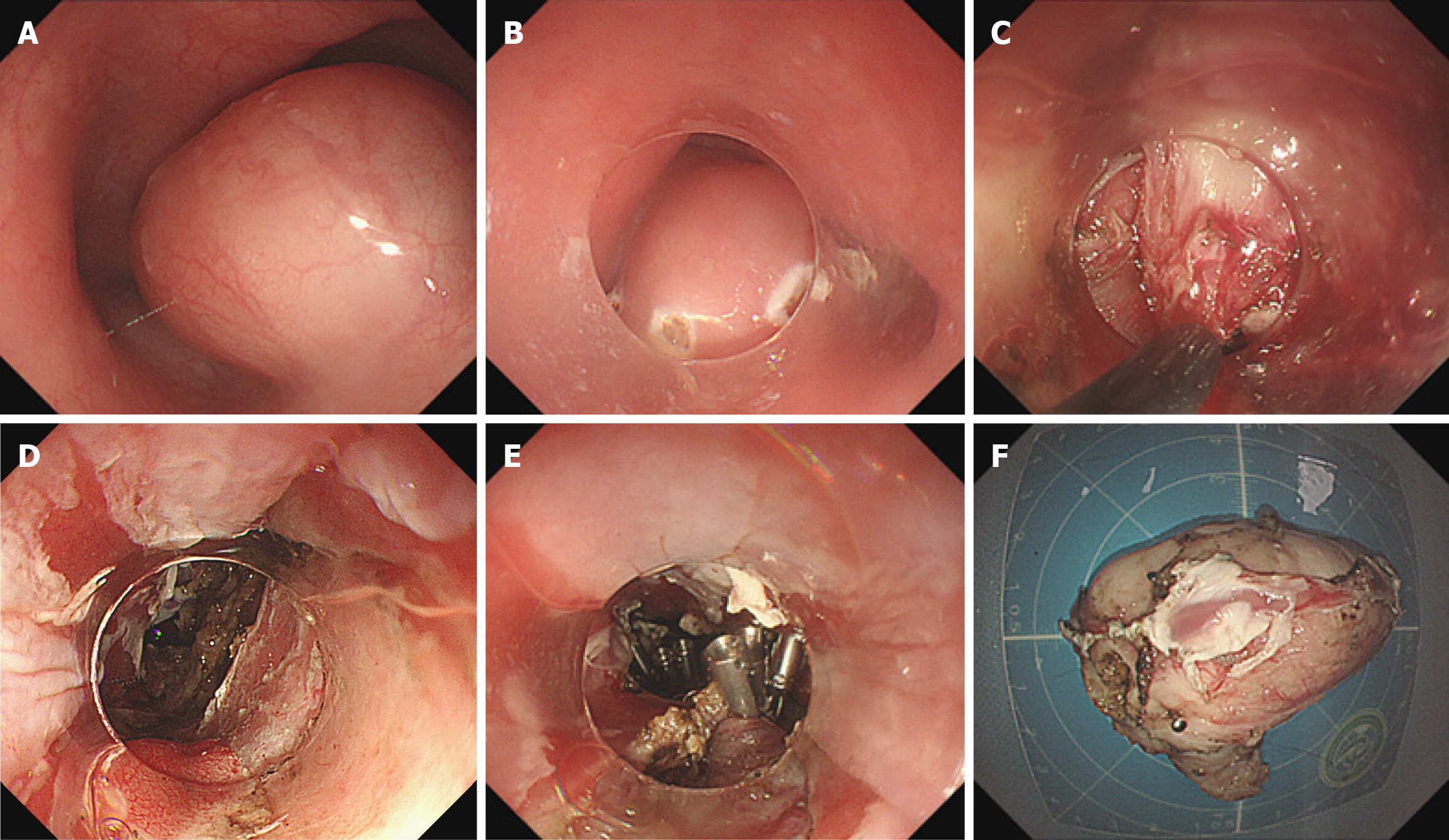
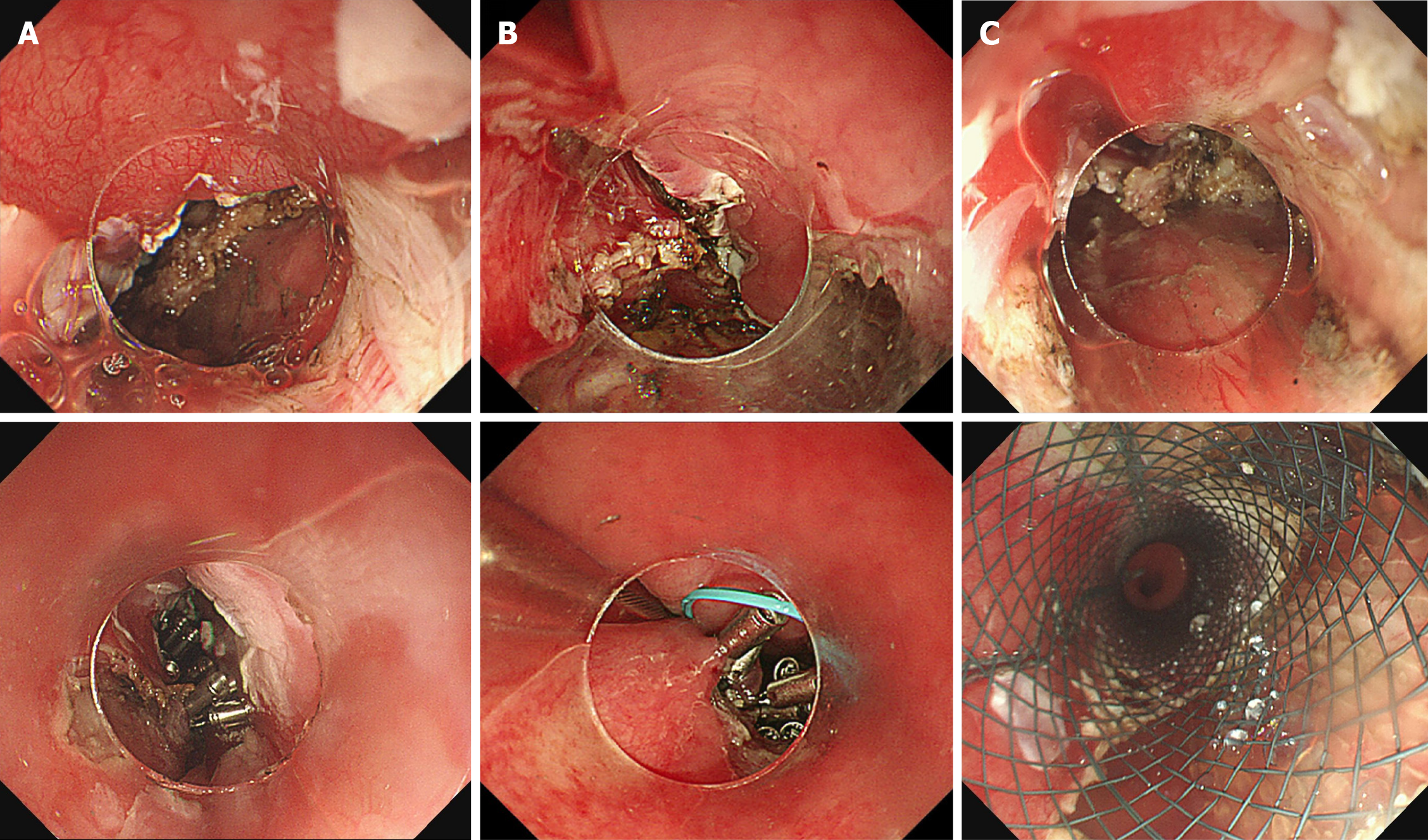
Exposed EFTR procedures were performed by four experienced endoscopists at our endoscopy center under general anesthesia with CO2 insufflation. The technique included lesion marking, submucosal dissection, full-thickness resection, and defect closure using metallic clips, nylon loops, or stents as needed. Details of the procedure are outlined in Figure 2 and in prior publications[12]. Figure 3 shows the different endoscopic defect-closure techniques and outcomes.
Procedures were performed with patients in the left lateral decubitus position under combined inhalational and intravenous anesthesia, and a double-lumen endotracheal tube was used for single-lung ventilation. The procedure was as follows: (1) A viewing port was established in the seventh intercostal space (ICS) along the right midaxillary line, and three working ports were established in the fourth ICS along the right anterior axillary line and the sixth and ninth ICSs along the right posterior axillary line, respectively; (2) Thoracoport trocars were inserted into these ports, and an artificial pneumothorax was established; (3) The mediastinal pleura above the azygos vein was opened, and the esophagus was mobilized; (4) The esophageal muscular layer over the tumor was incised longitudinally using an electric hook, and the tumor was dissected thoroughly; and (5) Mucosal integrity was assessed, the muscular layer was intermittently sutured with 3-0 Vicryl, and a mediastinal drainage tube was placed through the ninth ICS along the posterior axillary line.
Patients in both groups received standard postoperative care, including fasting, nasogastric decompression, and total parenteral nutrition. In the TS group, both a thoracic catheter and a nasogastric tube were placed during surgery. The intrathoracic tube was typically removed within 7 days, and oral intake was initiated after removal of the nasogastric decompression tube, provided there were no signs of thoracic infection.
The follow-up protocol was determined based on histopathological findings. All patients were advised to undergo upper endoscopy and computed tomography 3 to 6 months after treatment to assess for residual tumor. Follow-up consultations were scheduled every 6 to 12 months. When in-person visits were not feasible, outcomes were monitored via telephone.
The primary outcomes were compared between esophageal exposed EFTR and TS using the following criteria: (1) Curative resection, defined as a histologically negative margin with no residual tumor on postoperative pathology; (2) En bloc resection, defined as complete tumor removal without capsular disruption; (3) Technical success, defined as successful, complete resection of the lesion; and (4) Recurrence-free survival, defined as the time from the procedure to the last follow-up visit without tumor recurrence. Secondary outcomes included: (1) Procedure-related parameters, including procedure time, length of hospital stay, antibiotic prescription, intraesophageal decompression time, fasting period, and hospitalization expenses; and (2) Intraoperative and postoperative adverse events, defined as Clavien-Dindo grade III-IV complications.
Continuous variables are presented as the mean ± SD or as the median (range). Categorical variables were compared using the Pearson χ2 test or Fisher’s exact test, whereas continuous variables were analyzed using the Student’s t test or the Mann-Whitney U test. Recurrence-free survival was assessed using the log-rank test. Statistical analyses were performed using SPSS version 27.0 (SPSS, Inc., Chicago, IL, United States). Statistical significance was defined as a P value < 0.05.
A total of 60 patients met the inclusion criteria for complex esophageal SELs-MP, including 10 (16.7%) with cervical lesions, 35 with large lesions (> 35 mm in diameter), and 15 (25.0%) with both characteristics. Based on treatment modality, 15 patients underwent esophageal exposure to EFTR, and 45 underwent TS. As shown in Table 1, there were no significant differences between the EFTR and TS groups in terms of age, sex, or American Society of Anesthesiologists classification. Although lesion size did not differ significantly between groups, the mean maximum lesion diameter was slightly larger in the EFTR group than in the TS group (59.3 ± 21.7 mm vs 48.7 ± 19.3 mm, P = 0.087).
| Variables | EFTR (n = 15) | TS (n = 45) | P value |
| Age, years | 47.2 ± 15.6 | 41.6 ± 11.3 | 0.148 |
| Male sex | 8 (57.1) | 28 (62.2) | 0.734 |
| Location | 0.775 | ||
| Cervical | 7 (46.6) | 18 (40.0) | |
| Thoracic | 4 (26.7) | 17 (37.8) | |
| Abdominal | 4 (26.7) | 10 (22.2) | |
| Lesion size, mm | 59.3 ± 21.7 | 48.7 ± 19.3 | 0.087 |
| Pathology | 0.041 | ||
| Leiomyoma | 10 (66.7) | 40 (88.9) | |
| GIST | 3 (20.0) | 1 (2.2) | |
| Schwannomas | 0 | 4 (8.9) | |
| Others1 | 2 (13.3) | 0 (0.0) | |
| ASA class | 0.325 | ||
| I | 12 (80.0) | 40 (88.9) | |
| II | 3 (20.0) | 5 (11.1) | |
| III | 0 | 0 |
In the EFTR group, defect closure methods were tailored to lesion location and size. Complete closure was achieved in 3 patients (20.0%) using a nylon loop combined with metallic clips (n = 1), a fully covered retrievable metallic stent (n = 1), or metallic clips alone (n = 1). Incomplete closure was achieved in 8 patients (53.3%) using metallic clips, whereas 4 patients (26.7%) had unclosed defects. Of the four patients with unclosed defects, three had cervical lesions, and one had a 100-mm-diameter lesion. Intraesophageal decompression was mandatory in all cases with unclosed defects but was not used in some cases with complete or incomplete closure.
The technical success rate was comparable between the EFTR and TS groups (100% vs 97.8%, respectively; P = 0.574). Both groups achieved a 100% curative resection rate (Table 2). Although the overall en bloc resection rate was numerically higher in the EFTR group (86.7% vs 75.6%), the difference was not statistically significant (P = 0.423). Subgroup analysis by lesion location provided additional clinical insights. Exposed EFTR may offer a technical advantage in achieving en bloc resection for lesions in the abdominal esophagus (100% vs 50%), although this difference did not reach statistical significance (P = 0.118), likely due to the small sample size. Compared with the TS group, the EFTR group showed similarly high en bloc resection rates in the cervical and thoracic segments. For lesions < 75 mm in diameter, en bloc resection was achieved in 90.0% of the EFTR group and 73.9% of the TS group. However, in 2 EFTR cases (13.3%), lesions measuring 100 mm and 50 mm in diameter that were resected en bloc were divided into two fragments to facilitate retrieval using a snare. During follow-up, no recurrence was observed in the EFTR group, whereas 2 patients in the TS group experienced recurrence at 14 months and 64 months, respectively. Both recurrences were successfully managed with endoscopic resection. Detailed perioperative data for both groups are provided in Table 2.
| Variables | EFTR (n = 15) | TS (n = 45) | P value |
| Primary outcomes | |||
| Curative resection | 15 (100) | 45 (100) | > 0.999 |
| En bloc resection | 13 (86.7) | 34 (75.6) | 0.423 |
| Cervical | 71.4 (5/7) | 77.8 (14/18) | 0.739 |
| Thoracic | 100 (4/4) | 88.2 (15/17) | 0.471 |
| Abdominal | 100 (4/4) | 50 (5/10) | 0.118 |
| Recurrence-free survival | 15 (100) | 43 (95.6) | 0.422 |
| Technical success | 15 (100) | 44 (97.8) | 0.574 |
| Clinical success | 15 (100) | 45 (100) | > 0.999 |
| Secondary outcomes | |||
| Procedure time, minutes | 240.0 (172.3, 262.5) | 120.0 (87.50, 145.0) | < 0.001 |
| Hospital stay, days | 18.1 ± 7.4 | 18.78 ± 7.4 | 0.757 |
| Postoperative hospital stay, days | 12.2 ± 4.9 | 11.1 ± 5.2 | 0.474 |
| Duration of antibiotic use after surgery, days | 8.9 ± 5.3 | 8.9 ± 5.0 | 0.967 |
| Nasogastric decompression tube placement | 12 (80.0) | 43 (95.6) | 0.059 |
| Nasogastric decompression duration, days | 5.6 ± 4.9 | 10.7 ± 13.2 | 0.016 |
| Postoperative fasting time, days | 7.36 ± 3.8 | 10.2 ± 13.2 | 0.430 |
| Hospitalization expenses, yuan | 55608.1 (43969.7, 65099.5) | 48680.6 (43429.0, 56037.4) | 0.291 |
| Intraoperative adverse events | 0.591 | ||
| Bleeding | 0 | 2 (4.4) | |
| Perforation | 0 | 1 (2.2) | |
| Postoperative adverse events | 0.234 | ||
| Stricture | 2 (12.5) | 1 (2.2) | |
| Fistula | 1 (6.3) | 1 (2.2) | |
| Chylothorax | 0 | 2 (4.4) | |
| Follow-up, months | 36.9 ± 19.9 | 58.9 ± 25.1 | 0.004 |
The median procedure time was significantly longer for exposed EFTR than for TS (240 minutes vs 120 minutes, P < 0.001; Table 2). Nasogastric decompression tubes were placed in 12 patients (80.0%) in the EFTR group and 43 patients (95.6%) in the TS group (P = 0.059). However, the duration of postoperative nasogastric decompression was significantly shorter in the EFTR group than in the TS group (5.6 ± 4.9 days vs 10.7 ± 13.2 days, P = 0.016; Table 2). Postoperative length of hospital stay, antibiotic administration, fasting duration, and hospitalization expenses were similar between groups.
During the procedure, severe adverse events occurred in 3 patients (6.7%) in the TS group (Table 2). One patient experienced an esophageal perforation and required open surgery as definitive treatment. Two patients (4.4%) had intraoperative bleeding, with blood losses of 400 mL and 300 mL, requiring intraoperative transfusion. One of these patients ultimately underwent radical esophagectomy with digestive tract reconstruction. In the EFTR group, no severe intraoperative adverse events were reported.
Postoperative adverse events occurred in 4 patients in the TS group (Table 3). These included esophageal stricture in 1 patient (2.2%), which was successfully managed with a retrievable metallic stent for 9 days, and esophageal fistula in 1 patient (2.2%), which was treated conservatively with intraesophageal decompression and nasojejunal feeding for 35 days. Two patients (4.4%) developed chylothorax and required thoracoscopic duct ligation (Table 3).
| Case No. | Sex/age (years) | Adverse events | Method | Location | Size (mm) | Procedure time (minutes) | Hospital stay (days) | Use of antibiotic | Treatment |
| 1 | Female/51 | Stricture | EFTR | Thoracic | 85 | 333 | 21 | Yes | Balloon dilation |
| 2 | Male/55 | Stricture | EFTR | Abdominal | 50 | 250 | 18 | Yes | Stent placement |
| 3 | Male/44 | Fistula | EFTR | Abdominal | 57 | 140 | 40 | Yes | Conservative |
| 4 | Male/22 | Fistula | TS | Abdominal | 60 | 278 | 19 | Yes | Conservative |
| 5 | Male/67 | Stricture | TS | Abdominal | 75 | 170 | 20 | Yes | Stent placement |
| 6 | Male/16 | Chylothorax | TS | Cervical | 25 | 156 | 23 | Yes | Thoracic duct ligation |
| 7 | Female/72 | Chylothorax | TS | Thoracic | 130 | 226 | 40 | Yes | Thoracic duct ligation |
In the EFTR group, postoperative adverse events occurred in 3 patients (Table 3). These included esophageal stricture in 2 cases, managed with balloon dilation and a retrievable metallic stent, respectively, and esophageal fistula in 1 case, which was successfully managed conservatively with percutaneous drainage and intraesophageal decompression. The percutaneous drainage tube was removed, and oral intake was resumed within 30 days after the EFTR procedure.
STER is widely considered the optimal method for removing SELs-MP because of its feasibility and safety[13]. However, STER is unsuitable for complex esophageal lesions that are large and/or located in the cervical esophagus, because there is insufficient space to create a submucosal tunnel and to allow effective endoscopic manipulation. EFTR offers a non-tunneling approach while still achieving en bloc and curative resection. Zhang et al[14] described six cases of esophageal SELs resected using EFTR with satisfactory outcomes. Our study provides additional evidence, with the following key findings: (1) Safety: Exposed esophageal EFTR is a safe procedure for complex esophageal SELs-MP. To our knowledge, this is the largest cohort of complex esophageal EFTR procedures reported to date. Although no severe intraoperative adverse events occurred, the rate of postoperative adverse events was acceptable and was managed conservatively; (2) Feasibility: Exposed esophageal EFTR achieved 100% technical success and curative resection rates. Although large tumors required piecemeal resection, this did not compromise outcomes; and (3) Recovery: Patients undergoing exposed EFTR experienced faster recovery, as evidenced by earlier postoperative oral intake and shorter nasogastric intubation duration. However, in our study, exposed EFTR required longer procedure times than TS.
Given the limited space within the esophageal tunnel, STER is typically indicated for lesions < 35 mm. Even with this limitation, en bloc resection is not always achievable. Chai et al[15] compared STER with video-assisted thoracoscopic enucleation for esophageal SELs-MP and reported a higher en bloc resection rate with thoracoscopic enucleation. Theoretically, EFTR can achieve en bloc resection regardless of tumor size; however, the difficulty increases with larger tumors. In our study, en bloc resection rates were comparable between the EFTR and TS groups, even for large cervical SELs (71.4% vs 77.8%, P = 0.739). In addition, piecemeal resection is often necessary for tumors > 80 mm in diameter. Some SELs, such as gastrointestinal stromal tumor, have malignant potential, making it critical to avoid fragmentation during surgical intervention. However, endoscopic piecemeal resection is considered safe, with a negligible risk of tumor seeding because of the protective role of the gastrointestinal wall[16,17]. Our findings support this, as no recurrences or metastases were observed during follow-up among tumors resected piecemeal.
Secure defect closure is the most challenging and critical step in exposed esophageal EFTR, particularly for transmural post-EFTR defects[18]. Various methods are widely used, including clip closure, endoloop-assisted closure, over-the-scope clip closure, and endosuturing[12,19]. Zhou et al[20] first reported successful post-EFTR defect closure using multi
Because EFTR is still in the early stages of application for esophageal lesions, operator experience remains limited, particularly for large or cervical lesions. Rigorous intraoperative and postoperative surveillance is crucial to mitigate adverse events. No severe intraoperative adverse events occurred in the EFTR group. Major adverse events associated with EFTR include bleeding, perforation, secondary infection, pneumoperitoneum, and hydrothorax[23,24].
Proper hemostasis and defect closure are critical for preventing bleeding and perforation. Esophageal stricture was the most common adverse event in the EFTR group and was managed conservatively. In contrast to previous reports, no cases of pneumoperitoneum requiring intervention occurred in the EFTR group, likely because carbon dioxide insuffla
In contrast, the TS group had one case requiring conversion to open surgery because of active intraoperative bleeding, as well as two cases of postoperative chylothorax requiring thoracoscopic catheter ligation. These findings support the use of exposed EFTR as a reasonable treatment option for complex esophageal lesions, with manageable adverse events and satisfactory outcomes.
In this study, exposed EFTR required a longer procedure time but offered no advantages in hospital length of stay, antibiotic duration, fasting time, or total hospitalization expenses. These findings may be attributable to the larger and more challenging cervical lesions in the EFTR group, in which the median tumor size was 59 mm vs 49 mm in the TS group. Increased lesion complexity likely contributed to the prolonged procedure time. We anticipate that the advantages of EFTR will become more apparent as technical proficiency improves with experience along the learning curve.
Several limitations should be acknowledged. First, the retrospective design introduces potential selection bias. Second, because exposed esophageal EFTR is a novel procedure, the cohort size and number of events were relatively small, which restricted our comprehensive evaluation of its advantages and limited our ability to detect differences with ade
Esophageal EFTR demonstrated clinical outcomes comparable to those of TS, suggesting that it is a safe and feasible option for complex esophageal SEL-MP. Further studies are needed to validate these findings.
| 1. | Luo Z, Yu J, Zhang H, Yang S, Li J, Liu J, Wang S. A novel technique for submucosal tumors in the esophagus: mucosal zipper endoscopic resection. Endoscopy. 2025;57:1112-1117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Schneider MA, Vetter D, Gutschow CA. Management of subepithelial esophageal tumors. Innov Surg Sci. 2025;10:21-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 3. | Bhagat VH, Kim M, Kahaleh M. A Review of Endoscopic Full-thickness Resection, Submucosal Tunneling Endoscopic Resection, and Endoscopic Submucosal Dissection for Resection of Subepithelial Lesions. J Clin Gastroenterol. 2021;55:309-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (1)] |
| 4. | Mutrie CJ, Donahue DM, Wain JC, Wright CD, Gaissert HA, Grillo HC, Mathisen DJ, Allan JS. Esophageal leiomyoma: a 40-year experience. Ann Thorac Surg. 2005;79:1122-1125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 104] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 5. | Wadhwa V, Franco FX, Erim T. Submucosal Tunneling Endoscopic Resection. Surg Clin North Am. 2020;100:1201-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 6. | Feng Y, Yu L, Yang S, Li X, Ding J, Chen L, Xu Y, Shi R. Endolumenal endoscopic full-thickness resection of muscularis propria-originating gastric submucosal tumors. J Laparoendosc Adv Surg Tech A. 2014;24:171-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 7. | Al-Bawardy B, Rajan E, Wong Kee Song LM. Over-the-scope clip-assisted endoscopic full-thickness resection of epithelial and subepithelial GI lesions. Gastrointest Endosc. 2017;85:1087-1092. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 8. | Huang LY, Cui J, Lin SJ, Zhang B, Wu CR. Endoscopic full-thickness resection for gastric submucosal tumors arising from the muscularis propria layer. World J Gastroenterol. 2014;20:13981-13986. [PubMed] [DOI] [Full Text] |
| 9. | Dolan RD, Bazarbashi AN, McCarty TR, Thompson CC, Aihara H. Endoscopic full-thickness resection of colorectal lesions: a systematic review and meta-analysis. Gastrointest Endosc. 2022;95:216-224.e18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (3)] |
| 10. | Chen T, Zhang YW, Lian JJ, Zhang HB, Xu AP, Li F, Yan XH, Duan BS, Zhao ZY, Chu Y, Shen L, Cao J, Zhang L, Zheng L, Chu SG, Xu MD. No-touch endoscopic full-thickness resection technique for gastric gastrointestinal stromal tumors. Endoscopy. 2023;55:557-562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 11. | Kappelle WFW, Backes Y, Valk GD, Moons LMG, Vleggaar FP. Endoscopic full-thickness resection of gastric and duodenal subepithelial lesions using a new, flat-based over-the-scope clip. Surg Endosc. 2018;32:2839-2846. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 12. | Wang K, Gao P, Cai M, Song B, Zhou P. Endoscopic full-thickness resection, indication, methods and perspectives. Dig Endosc. 2023;35:195-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 13. | Xu MD, Cai MY, Zhou PH, Qin XY, Zhong YS, Chen WF, Hu JW, Zhang YQ, Ma LL, Qin WZ, Yao LQ. Submucosal tunneling endoscopic resection: a new technique for treating upper GI submucosal tumors originating from the muscularis propria layer (with videos). Gastrointest Endosc. 2012;75:195-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 247] [Article Influence: 17.6] [Reference Citation Analysis (4)] |
| 14. | Zhang Y, Yao L, Xu M, Berzin TM, Li Q, Chen W, Hu J, Wang Y, Cai M, Qin W, Xu J, Huang Y, Zhou P. Treatment of leakage via metallic stents placements after endoscopic full-thickness resection for esophageal and gastroesophageal junction submucosal tumors. Scand J Gastroenterol. 2017;52:76-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Chai N, Du C, Gao Y, Niu X, Zhai Y, Linghu E, Liu Y, Yang B, Lu Z, Li Z, Wang X, Tang P. Comparison between submucosal tunneling endoscopic resection and video-assisted thoracoscopic enucleation for esophageal submucosal tumors originating from the muscularis propria layer: a randomized controlled trial. Surg Endosc. 2018;32:3364-3372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 16. | Kim HH, Kim JH, Park SJ, Park MI, Moon W. Risk factors for incomplete resection and complications in endoscopic mucosal resection for lateral spreading tumors. Dig Endosc. 2012;24:259-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 17. | Luigiano C, Consolo P, Scaffidi MG, Strangio G, Giacobbe G, Alibrandi A, Pallio S, Tortora A, Melita G, Familiari L. Endoscopic mucosal resection for large and giant sessile and flat colorectal polyps: a single-center experience with long-term follow-up. Endoscopy. 2009;41:829-835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 88] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 18. | Mori H, Rahman A, Kobara H, Fujihara S, Nishiyama N, Ayaki M, Matsunaga T, Murakami M, Masaki T. Current Status of Exposed Endoscopic Full-Thickness Resection and Further Development of Non-Exposed Endoscopic Full-Thickness Resection. Digestion. 2017;95:6-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 19. | Granata A, Martino A, Ligresti D, Zito FP, Amata M, Lombardi G, Traina M. Closure techniques in exposed endoscopic full-thickness resection: Overview and future perspectives in the endoscopic suturing era. World J Gastrointest Surg. 2021;13:645-654. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 20. | Zhou PH, Yao LQ, Qin XY, Cai MY, Xu MD, Zhong YS, Chen WF, Zhang YQ, Qin WZ, Hu JW, Liu JZ. Endoscopic full-thickness resection without laparoscopic assistance for gastric submucosal tumors originated from the muscularis propria. Surg Endosc. 2011;25:2926-2931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 251] [Article Influence: 16.7] [Reference Citation Analysis (2)] |
| 21. | Shi Q, Chen T, Zhong YS, Zhou PH, Ren Z, Xu MD, Yao LQ. Complete closure of large gastric defects after endoscopic full-thickness resection, using endoloop and metallic clip interrupted suture. Endoscopy. 2013;45:329-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 113] [Article Influence: 8.7] [Reference Citation Analysis (1)] |
| 22. | Guo JT, Zhang JJ, Wu YF, Liao Y, Wang YD, Zhang BZ, Wang S, Sun SY. Endoscopic full-thickness resection using an over-the-scope device: A prospective study. World J Gastroenterol. 2021;27:725-736. [PubMed] [DOI] [Full Text] |
| 23. | Mori H, Kobara H, Nishiyama N, Masaki T. Current status and future perspectives of endoscopic full-thickness resection. Dig Endosc. 2018;30 Suppl 1:25-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Jian G, Tan L, Wang H, Lv L, Wang X, Qi X, Le M, Tan Y, Liu D. Factors that predict the technical difficulty during endoscopic full-thickness resection of a gastric submucosal tumor. Rev Esp Enferm Dig. 2021;113:35-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 25. | Li B, Chen T, Qi ZP, Yao LQ, Xu MD, Shi Q, Cai SL, Sun D, Zhou PH, Zhong YS. Efficacy and safety of endoscopic resection for small submucosal tumors originating from the muscularis propria layer in the gastric fundus. Surg Endosc. 2019;33:2553-2561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 33] [Article Influence: 4.7] [Reference Citation Analysis (0)] |