Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.117558
Revised: January 12, 2026
Accepted: February 11, 2026
Published online: May 7, 2026
Processing time: 132 Days and 21.7 Hours
The 9th edition American Joint Committee on Cancer (AJCC) staging system has been proposed by the AJCC Cancer Expert Panel (AJCCCCEP). The prognostic discriminatory validity of the new staging system in actual patient cohorts has not been fully validated.
To investigate and validate the proposed changes to the pathologic staging for colon cancer (CC) and rectal cancer (RC) by the AJCCCCEP.
The study included 1251 eligible patients with stage I-III CC and 1162 patients with RC between July 2015 and July 2021. Clinicopathological features of the pa
One thousand, two hundred and fifty-one patients with CC and 1162 patients with RC were analyzed, with a median follow-up of 5.45 and 5.30 years for DFS and OS in the CC cohort and 5.36 and 5.35 years in the RC cohort. All patients were re-classified according to the updated staging system proposed by AJCCCCEP. The survival analysis revealed a hierarchical separation across all sub-levels in the CC cohort and the RC cohort in terms of DFS and OS.
This study indicates that the 9th edition AJCC staging system has better prognostic validity than the 8th edition AJCC staging system for patients with CC and RC.
Core Tip: This validation study of 2413 stage I-III colon and rectal cancer patients demonstrates that the proposed 9th edition American Joint Committee on Cancer staging system shows clearer hierarchical separation in overall and disease-free survival, offering better prognostic discrimination compared to the 8th edition.
- Citation: Sun ZG, Wu WX, Chen SX, Zhang YJ, Jie JZ. Improved prognostic stratification of the revised ninth edition American Joint Committee on Cancer staging system for colorectal cancer. World J Gastroenterol 2026; 32(17): 117558
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/117558.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.117558
Colorectal cancer (CRC) is the third most common cancer and the second leading cause of mortality among all cancer types worldwide[1]. The American Joint Committee on Cancer (AJCC) staging manual, which classifies tumor from the perspective of pathological anatomy remains the cornerstone for treatment and prognosis of patients with CRC, for example, stage III patients are routinely given adjuvant chemotherapy, while in stage II patients, a decision is often made on a case-by-case basis. However, the discrimination and accuracy of prognostic competency using AJCC staging criteria remains controversial. The latest 8th edition AJCC staging system was released in 2016, in which the regional lymph nodes involvement (N component) is considered to be a more important prognostic factor than the depth of tumor invasion into the colorectal wall (T component), whereas numerous previous studies demonstrated that T4N0 colon cancer (CC) had significantly poorer outcomes than T1-2N1-2a CCs, regardless of the number of lymph nodes examined[2-4]. Moreover, the survival curves by the 8th edition summary stage are not hierarchical, with stage I and stage III patients exhibiting similar overall survival (OS) and stage IIIB patients exhibiting superior OS compared to stage IIB and IIC patients. Recently, the 9th edition AJCC staging system has been proposed by the AJCC Cancer Expert Panel (AJCCCCEP) and reported in American Society of Clinical Oncology 2025, wherein the weight of the pathological T component for pre
Consecutive patients with pathologically confirmed primary CC and rectal cancer (RC) who underwent curative-intent surgical resection from July 2015 to July 2021 were enrolled. The inclusion criteria were: (1) Histologically verified malignancy following radical resection performed either laparoscopically or via open laparotomy; and (2) Availability of comprehensive clinicopathological and follow-up records. The exclusion criteria were: (1) Postoperative metastasis or death within 3 months; (2) Distant metastases upon initial diagnosis; (3) Follow-up duration < 2 years; and (4) Pathologi
Clinical and pathological data were extracted retrospectively from electronic medical records. Demographic and surgical details (age, sex, operative date, tumor location, and metastatic status) were recorded. Pathological assessments included nodal lymph nodes (positive/total lymph nodes), TDs, and pT stage. Cases with incomplete pathology data were excluded.
Recurrence and mortality data were obtained via telephonic interviews with patients/families or electronic record review. Disease-free survival (DFS) was estimated from the day of primary surgery to the date of diagnosis of recurrent disease or the last date of follow-up without recurrence or the date of death from other causes. OS was estimated from the day of primary surgery to death from any causes or the last day of follow-up for those who were alive.
All patients underwent radical surgery via complete mesocolic excision for CC and total mesorectal excision for RC. Neoadjuvant chemotherapy or adjuvant chemotherapy with the CAPOX (capecitabine + oxaliplatin) or FOLFOX (fluorouracil + oxaliplatin + leucovorin) regimen was applied over six cycles, or neoadjuvant chemoradiotherapy or adjuvant chemoradiotherapy was recommended if necessary. Postoperative surveillance followed National Comprehensive Cancer Network guidelines: Quarterly visits for 2 years, semiannually for 3 subsequent years, and annually thereafter until 10 years. At every visit, interim clinical history and laboratory blood tests including carcinoembryonic antigen levels were checked.
Intergroup survival disparities (DFS/OS) were analyzed using Kaplan-Meier curves and log-rank tests. Significance was set at a two-sided P value < 0.05 with 95% confidence intervals. Analyses were conducted in R v4.0.5 (http://www.R-project.org).
During the study period, a total of 1251 patients with stage I-III CC and 1162 patients with stage I-III RC underwent curative surgery. As detailed in Table 1, the CC cohort comprised 533 female (42.6%) and 718 male (57.4%) patients, with a median age of 65 years. Pathological staging revealed the following T-stage distribution: PT1 in 40 cases (3.2%), pT2 in 132 cases (10.6%), pT3 in 991 cases (79.2%), pT4a in 32 cases (2.5%), and pT4b in 56 cases (4.5%). Lymph node involvement was distributed as follows: 774 patients (61.8%) had no positive lymph nodes (PLN), while 142 (11.4%), 117 (9.4%), 60 (4.8%), 44 (3.5%), and 114 (9.1%) had 1, 2, 3, 4, or > 4 PLNs, respectively. TDs were present in 186 cases (14.9%) and absent in 1065 (85.1%). For the RC cohort, 716 male (61.6%) and 446 female (38.4%) patients with RC were enrolled. Pathological staging revealed the following T-stage distribution: PT1 in 70 cases (6.0%), pT2 in 260 cases (22.4%), pT3 in 794 cases (68.3%) and pT4 in 38 cases (3.3%). Lymph node involvement was distributed as follows: 693 patients (59.6%) had no PLN, while 154 (13.3%), 89 (7.7%), 52 (4.5%), 36 (3.1%), and 138 (11.8%) had 1, 2, 3, 4, or > 4 PLNs, respectively.
| Overall (n = 2413) | Colon cancer (n = 1251) | Rectal cancer (n = 1162) | |
| Sex | |||
| Male | 533 (42.6) | 716 (61.6) | |
| Female | 718 (57.4) | 446 (38.4) | |
| pT stage | |||
| T1 | 40 (3.2) | 70 (6.0) | |
| T2 | 132 (10.6) | 260 (22.4) | |
| T3 | 991 (79.2) | 794 (68.3) | |
| T4 | 88 (7.0) | 38 (3.3) | |
| Number of metastatic lymph nodes | |||
| 0 | 774 (61.8) | 693 (59.6) | |
| 1 | 142 (11.4) | 154 (13.3) | |
| 2 | 117 (9.4) | 89 (7.7) | |
| 3 | 60 (4.8) | 52 (4.5) | |
| 4 | 44 (3.5) | 36 (3.1) | |
| > 4 | 114 (9.1) | 138 (11.8) |
In the CC cohort, the median follow-up time for DFS and OS was 65.43 (48.90-82.47) months and 63.63 (46.53-81.77) months, respectively. In the RC cohort, the median follow-up time was 64.37 (48.07-83.93) months and 64.27 (46.87-83.17) months for DFS and OS, respectively.
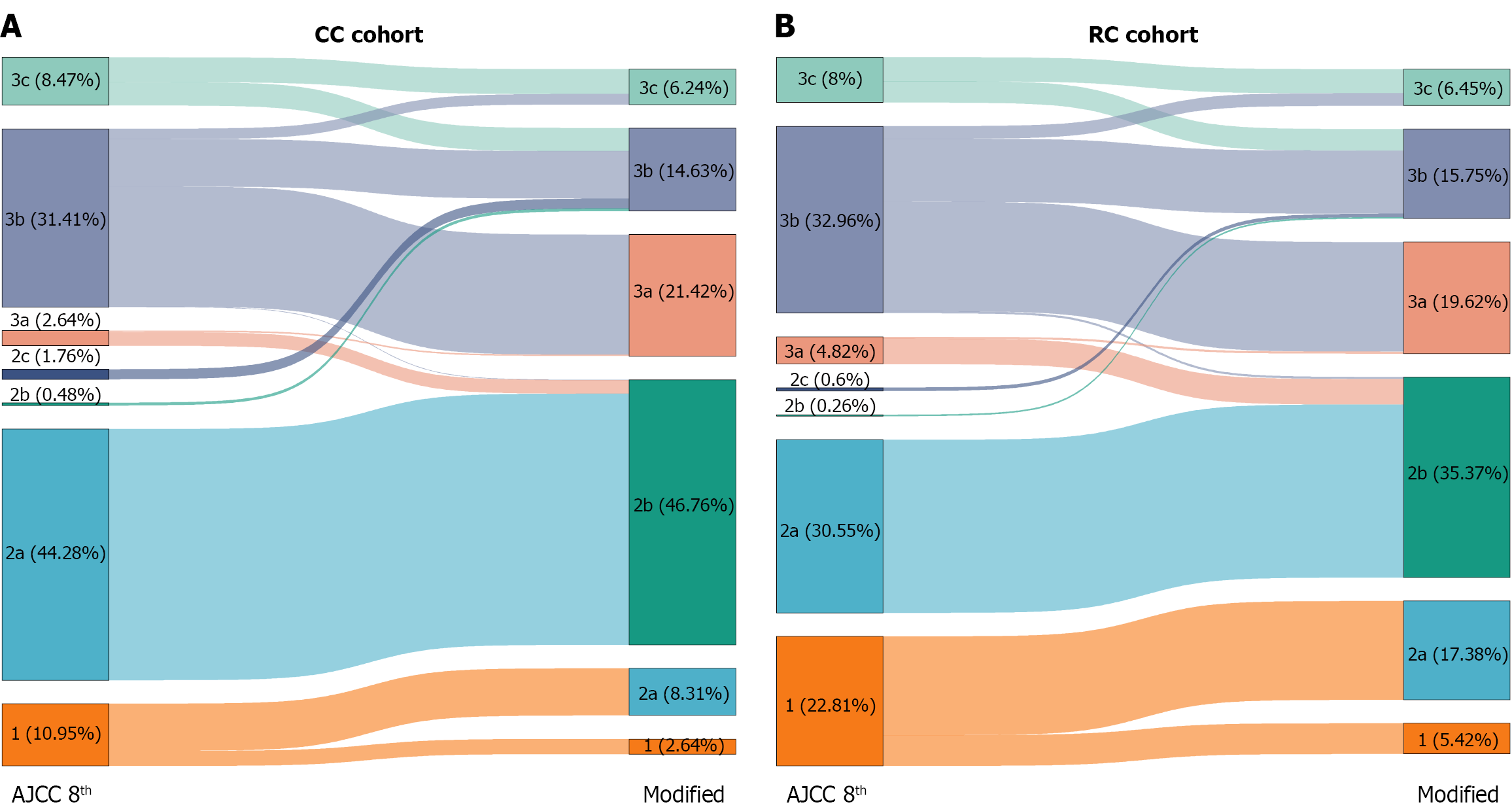
According to proposed changes to the pathologic staging for CC by the AJCCCCEP, CC with M0 disease was classified as three groups with only two subgroups within stage II, which is divided into three subgroups in the 8th staging system. The distribution of pathological staging in the CC and RC cohort according to the 8th and 9th criteria is shown in Figure 1.
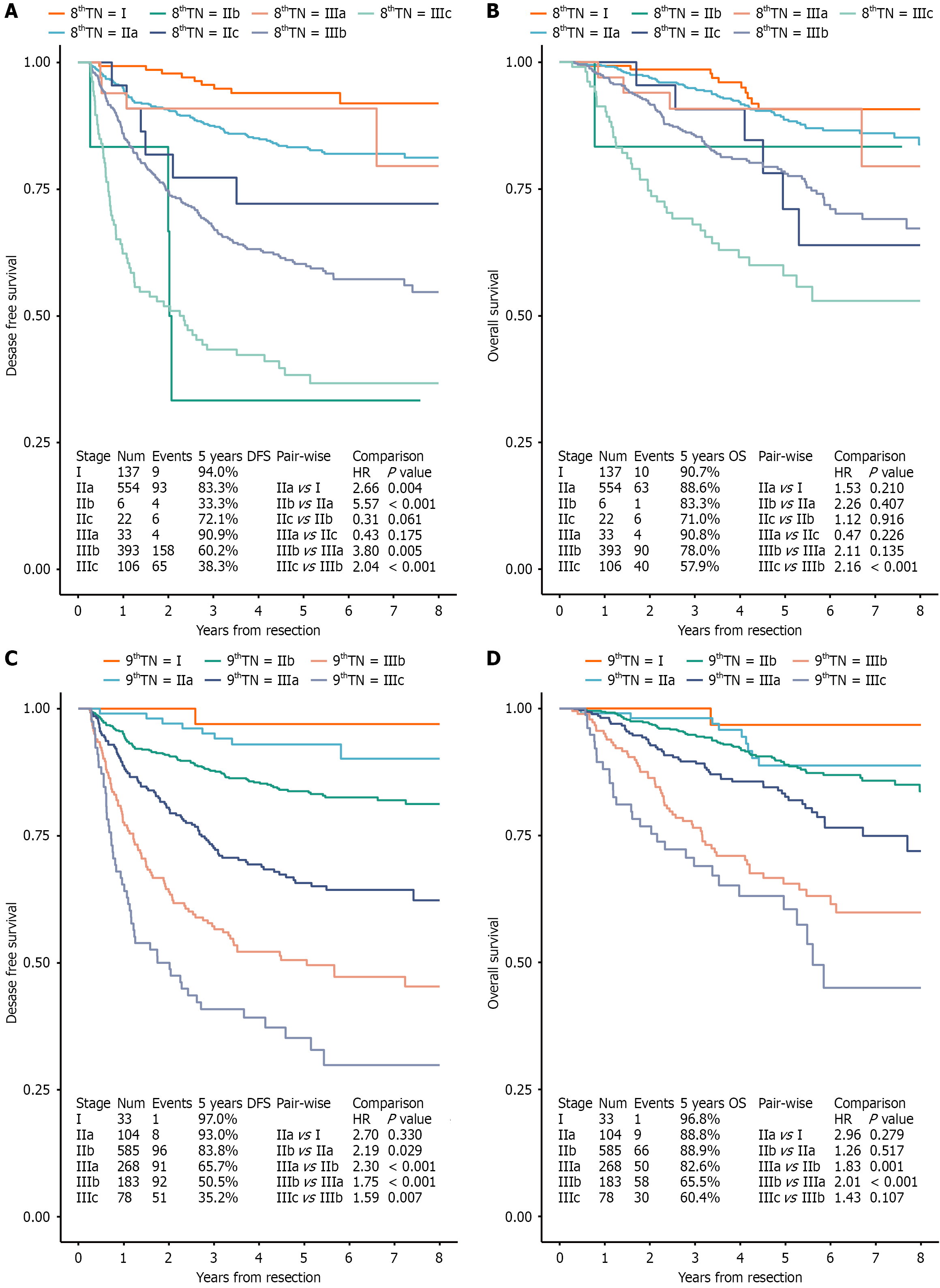
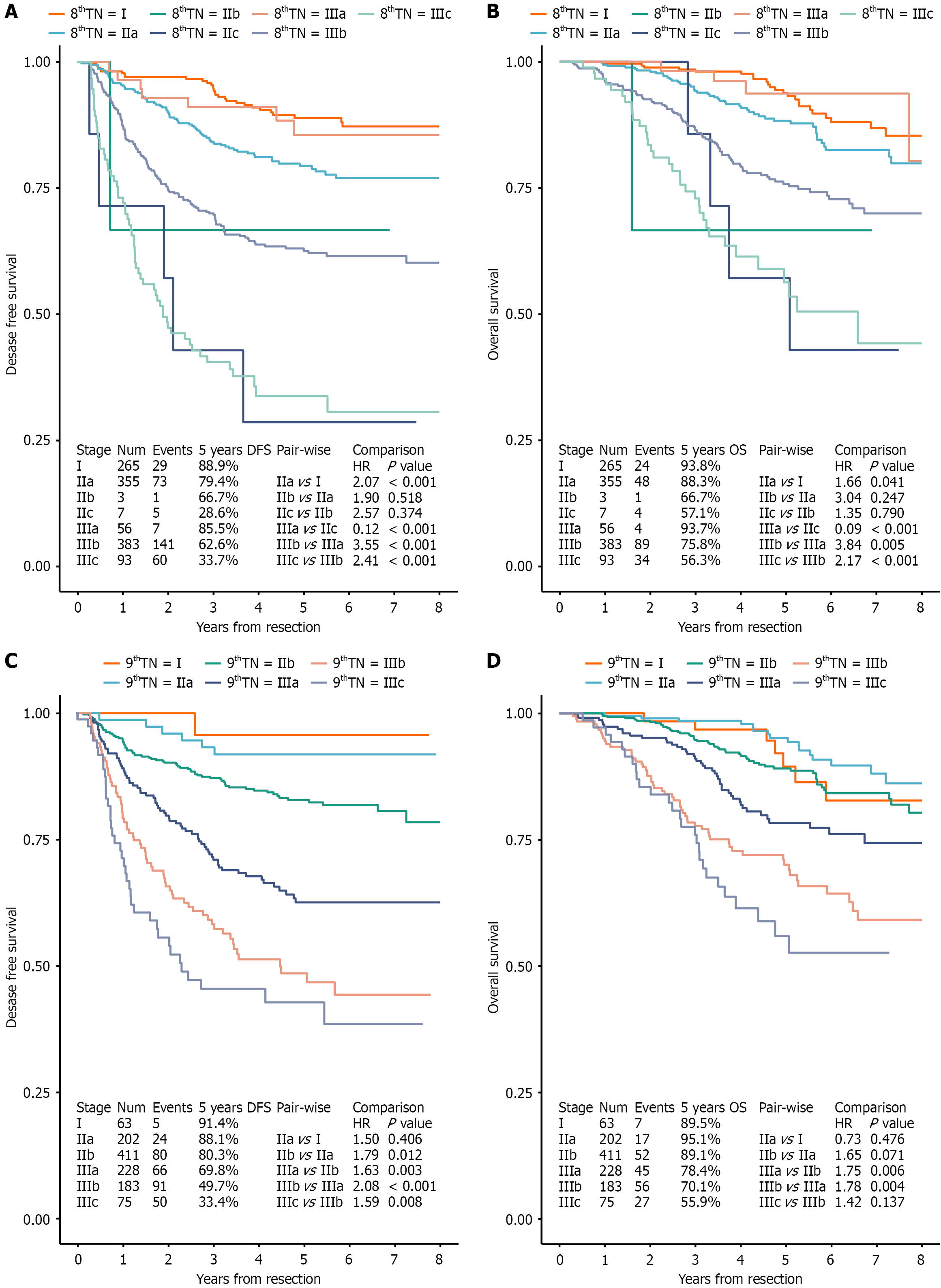
The survival analysis of the CC cohort including stage description and the corresponding 5 years survival, as well as consecutive pair-wise comparison results are shown in Figure 2. The 5-year DFS based on the 9th edition steadily de
Furthermore, the C-indices of the 8th edition and 9th edition were calculated in terms of DFS (0.689 and 0.674, P = 0.036) and OS (0.668 and 0.656, P = 0.180) in the CC cohort. Likewise, the predictive performance was also detected in cohort RC with C-indices of 0.685 vs 0.659 (P = 0.014) and 0.679 vs 0.662 (P = 0.168) in terms of DFS and OS.
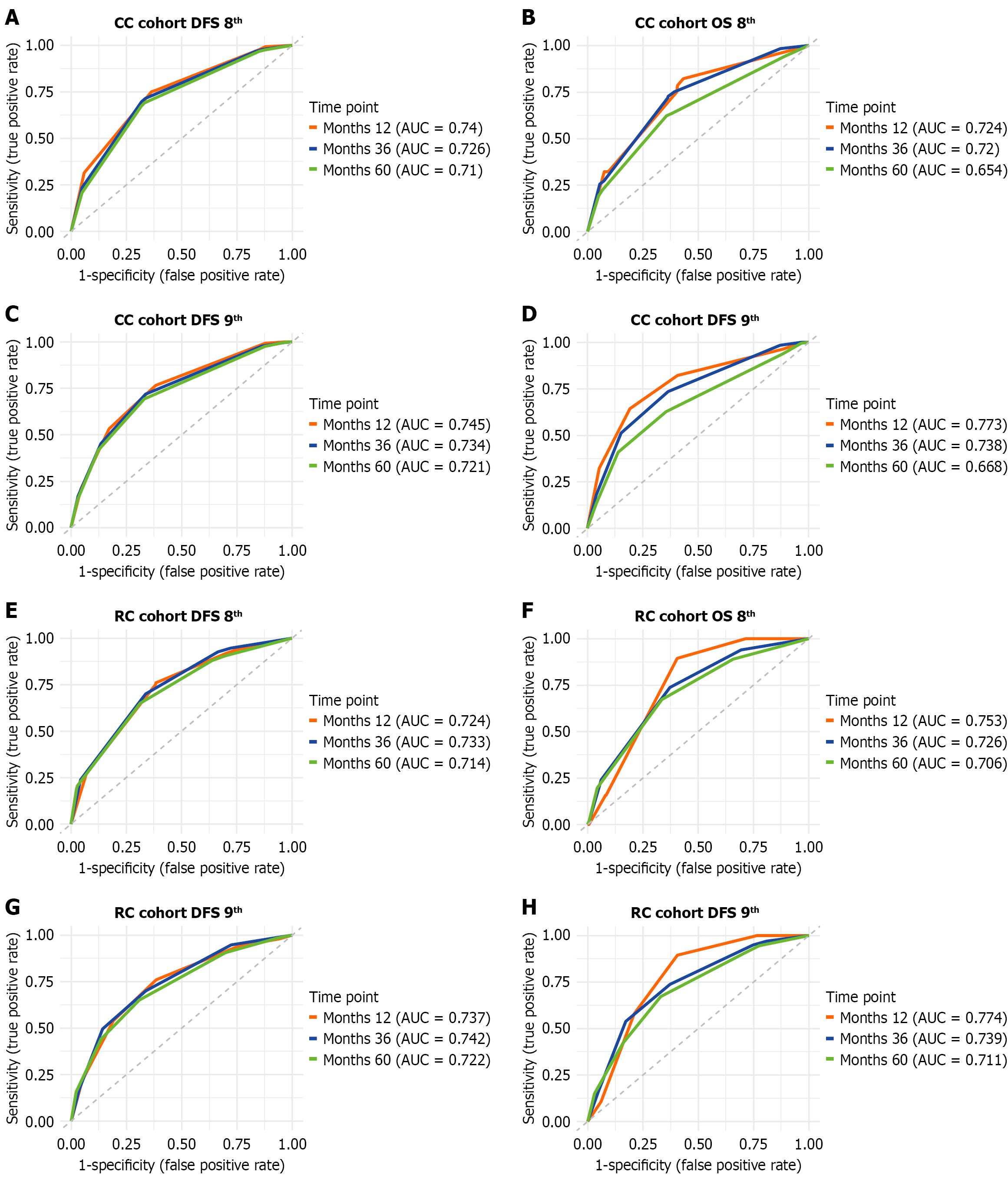
In the CC cohort, the area under the curves (AUC) at 1 year, 3 years and 5 years were 0.740, 0.726, 0.710 and 0.745, 0.734, 0.721 for the 8th edition and 9th edition system for DFS, respectively. For OS, the AUCs were 0.724, 0.720, 0.654 and 0.773, 0.738, 0.668 (Figure 4A-D). The integrated AUCs (iAUC) were 0.72 vs 0.731 (P = 0.06) and 0.704 vs 0.723 (P = 0.016) for the 8th edition and 9th edition in terms of DFS and OS, respectively. In the RC cohort, the AUCs at 1 year, 3 years and 5 years were 0.724, 0.733, 0.714 vs 0.737, 0.742, 0.722 in terms of DFS for the 8th edition and 9th edition, respectively, and 0.753, 0.726, 0.706 vs 0.774, 0.739, 0.711 for OS (Figure 4E-H). The iAUCs were 0.735 vs 0.744 (P = 0.096) and 0.741 vs 0.752 (P = 0.178) for the 8th edition and 9th edition in terms of DFS and OS, respectively. Time dependent receiver operating characteristic analysis revealed better performance of the 9th edition in discriminatory power with a statistically significant difference for CC.
In accordance with previous studies which indicated the nonhierarchical nature of current summary staging with respect to OS in patients with CC, in the present study, we demonstrated the prognostic value of the 9th edition AJCC staging system, which exhibited superior discriminative performance than the 8th edition AJCC staging system in terms of patient outcomes[6]. Different indicators for outcome including OS and DFS were assessed, respectively, and the survival curves were hierarchically separated among patients with different stages. The paradoxical phenomenon that patients with better survival but advanced staging observed in the current 8th edition is attributed to the complicated interplay between T category and N category, while the current 8th edition prioritized the N category[6,7].
According to the 9th edition, patients were divided into six subgroups ranging from subgroup I featuring best survival to subgroup IIIc displaying poorest survival. Patients with stage I CC according to the 8th edition show heterogeneous outcomes, which is reasonable as advanced T component is associated with poor outcome. Patients with IIIb CC exhibited the most various outcomes ranging from IIb to IIIc after restaging. Patients with IIa and most patients with IIIa, as well as a small portion of patients with IIIb shared similar outcome categorizing as IIb. A fraction of patients with IIIa and some with IIIb were classified as IIIa. Patients with IIb, IIc, a portion of IIIb and IIIc CC were grouped into IIIb according to the 9th edition. Patients with the worst prognosis from stages IIIb and IIIc were aggregated into a single high-risk group designated as IIIc. The aforementioned findings are in agreement with previous studies, with Rottoli et al[8] showing that stage IIc cancers had poorer outcomes than IIIa and were comparable to IIIb cancers[8-11]. In addition, the finding in the current study that patients with IIIb CC had heterogeneous survival was also indicated in a previous study[12]. This stratification brings about novel insight into the tumor biological behaviors that the depth of tumor invasion into the colorectal wall calls for enhanced clinical vigilance. Moreover, the 9th edition with homogeneous prognosis in specific groups may facilitate real effectiveness of therapy being evaluated in clinical trials.
Multiple studies have demonstrated that TDs are a factor for poor prognosis, in addition, in our previous study, we indicated that the presence of TDs warrants further investigation[13-15]. Another population-based study demonstrated that the concomitant presence of TDs and PLNs in CC was associated with worse OS and that TDs had similar prognostic implications to lymph node metastasis[16]. As an independent prognostic factor in CC, TDs are not well represented in the 8th edition, wherein TDs are taken into consideration only when the lymph node is not involved[17]. Furthermore, the presence of TDs was associated with a worse prognosis across all pN categories, indicating that valuable prognostic information was lost by incorporating TDs into the N category[18]. In these new staging criteria, TDs upgrade stage across pT and pN categories, which provides more accuracy in the prediction of prognosis.
In this study, we included patients with CC as well as RC and the results suggest that the 9th edition could also be useful for RC.
The current study has several limitations. First, the 9th edition was established based on survival outcomes. Impor
The 9th edition AJCC staging system can hierarchically separate survival outcome among patients with different disease stages, therefore provides better prognostic discrimination for stage I-III CC and RC than the 8th edition. In addition, the 9th edition AJCC staging system intuitively confers prognosis to patients and optimizes communication regarding prog
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15080] [Article Influence: 7540.0] [Reference Citation Analysis (23)] |
| 2. | Weiser MR. AJCC 8th Edition: Colorectal Cancer. Ann Surg Oncol. 2018;25:1454-1455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 834] [Cited by in RCA: 745] [Article Influence: 93.1] [Reference Citation Analysis (5)] |
| 3. | Chu QD, Zhou M, Medeiros KL, Peddi P, Kavanaugh M, Wu XC. Poor survival in stage IIB/C (T4N0) compared to stage IIIA (T1-2 N1, T1N2a) colon cancer persists even after adjusting for adequate lymph nodes retrieved and receipt of adjuvant chemotherapy. BMC Cancer. 2016;16:460. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 4. | Chu QD, Zhou M, Medeiros K, Peddi P. Positive surgical margins contribute to the survival paradox between patients with stage IIB/C (T4N0) and stage IIIA (T1-2N1, T1N2a) colon cancer. Surgery. 2016;160:1333-1343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 36] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 5. | Shi Q, Chang GJ, Pederson LD, Asare EA, Jin Z, Cohen R, Meyerhardt JA, Andre T, Tie J, Dixon JG, Palis BE, Ballman KV, Yothers G, Ma Z, Alberts SR, Shi C, Washington MK, Taieb J, Steele S, Goldberg RM. Proposed changes to the pathologic staging for colon cancer (CC): AJCC Colon Cancer Expert Panel (AJCCCCEP). J Clin Oncol. 2025;43:3520. [DOI] [Full Text] |
| 6. | Bhutiani N, Hu CY, Palis B, Cotler J, Shi Q, Washington MK, Goldberg RM, Steele SR, Chang GJ. Lack of Hierarchical Survival Prognosis in AJCC Staging for Colon and Rectal Cancer-Implications for Future Summary Stage Classification. Clin Colorectal Cancer. 2025;24:159-165.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Gunderson LL, Jessup JM, Sargent DJ, Greene FL, Stewart A. Revised tumor and node categorization for rectal cancer based on surveillance, epidemiology, and end results and rectal pooled analysis outcomes. J Clin Oncol. 2010;28:256-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 185] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 8. | Rottoli M, Stocchi L, Dietz DW. T4N0 colon cancer has oncologic outcomes comparable to stage III in a specialized center. Ann Surg Oncol. 2012;19:2500-2505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Kim MJ, Jeong SY, Choi SJ, Ryoo SB, Park JW, Park KJ, Oh JH, Kang SB, Park HC, Heo SC, Park JG. Survival paradox between stage IIB/C (T4N0) and stage IIIA (T1-2N1) colon cancer. Ann Surg Oncol. 2015;22:505-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 81] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 10. | Chan DKH, Lim TZ, Tan KK. T4N0 colon cancers should be treated like T3N1 disease. J Gastrointest Oncol. 2019;10:6-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 11. | Zhang C, Mei Z, Pei J, Abe M, Zeng X, Huang Q, Nishiyama K, Akimoto N, Haruki K, Nan H, Meyerhardt JA, Zhang R, Li X, Ogino S, Ugai T. A Modified Tumor-Node-Metastasis Classification for Primary Operable Colorectal Cancer. JNCI Cancer Spectr. 2021;5:pkaa093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 12. | Ueno H, Sekine S, Oshiro T, Kanemitsu Y, Hamaguchi T, Shida D, Takashima A, Ishiguro M, Ito E, Hashiguchi Y, Kondo F, Shimazaki H, Mochizuki S, Kajiwara Y, Shinto E, Yamamoto J, Shimada Y. Disentangling the prognostic heterogeneity of stage III colorectal cancer through histologic stromal categorization. Surgery. 2018;163:777-783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 13. | Sassun R, Sileo A, Ng JC, Violante T, Gomaa I, Mandrekar J, Rumer KK, McKenna NP, Larson DW. Validated Integration of Tumor Deposits in N Staging for Prognostication in Colon Cancer. JAMA Surg. 2025;160:408-414. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 14. | Chang Z, Fu H, Song J, Kong C, Xie R, Pi M, Sun X, Zhang W, Liu Y, Huang R, Yang T, Han D. The past, present, and future of tumour deposits in colorectal cancer: Advancing staging for improved prognosis and treatment decision-making. J Cell Mol Med. 2024;28:e18562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Jörgren F, Agger E, Lydrup ML, Buchwald P. Tumour deposits in colon cancer predict recurrence and reduced survival in a nationwide population-based study. BJS Open. 2023;7:zrad122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Mirkin KA, Kulaylat AS, Hollenbeak CS, Messaris E. Prognostic Significance of Tumor Deposits in Stage III Colon Cancer. Ann Surg Oncol. 2018;25:3179-3184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Wong-Chong N, Motl J, Hwang G, Nassif GJ Jr, Albert MR, Monson JRT, Lee L. Impact of Tumor Deposits on Oncologic Outcomes in Stage III Colon Cancer. Dis Colon Rectum. 2018;61:1043-1052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 41] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 18. | Delattre JF, Cohen R, Henriques J, Falcoz A, Emile JF, Fratte S, Chibaudel B, Dauba J, Dupuis O, Bécouarn Y, Bibeau F, Taieb J, Louvet C, Vernerey D, André T, Svrcek M. Prognostic Value of Tumor Deposits for Disease-Free Survival in Patients With Stage III Colon Cancer: A Post Hoc Analysis of the IDEA France Phase III Trial (PRODIGE-GERCOR). J Clin Oncol. 2020;38:1702-1710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 64] [Article Influence: 10.7] [Reference Citation Analysis (2)] |
| 19. | Leijssen LGJ, Dinaux AM, Taylor MS, Deshpande V, Kunitake H, Bordeianou LG, Berger DL. Perineural Invasion Is a Prognostic but not a Predictive Factor in Nonmetastatic Colon Cancer. Dis Colon Rectum. 2019;62:1212-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 26] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 20. | van Wyk HC, Going J, Horgan P, McMillan DC. The role of perineural invasion in predicting survival in patients with primary operable colorectal cancer: A systematic review. Crit Rev Oncol Hematol. 2017;112:11-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 21. | Zaanan A, Shi Q, Taieb J, Alberts SR, Meyers JP, Smyrk TC, Julie C, Zawadi A, Tabernero J, Mini E, Goldberg RM, Folprecht G, Van Laethem JL, Le Malicot K, Sargent DJ, Laurent-Puig P, Sinicrope FA. Role of Deficient DNA Mismatch Repair Status in Patients With Stage III Colon Cancer Treated With FOLFOX Adjuvant Chemotherapy: A Pooled Analysis From 2 Randomized Clinical Trials. JAMA Oncol. 2018;4:379-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 106] [Article Influence: 13.3] [Reference Citation Analysis (4)] |
| 22. | Formica V, Sera F, Cremolini C, Riondino S, Morelli C, Arkenau HT, Roselli M. KRAS and BRAF Mutations in Stage II and III Colon Cancer: A Systematic Review and Meta-Analysis. J Natl Cancer Inst. 2022;114:517-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (0)] |