Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.117544
Revised: January 1, 2026
Accepted: February 12, 2026
Published online: May 7, 2026
Processing time: 135 Days and 19.5 Hours
Long-term low-dose aspirin (LDA) use is associated with an increased risk of peptic ulcer bleeding (PUB). Helicobacter pylori (H. pylori) infection is a well-known risk factor for peptic ulcer disease (PUD).
To evaluate the prevalence of H. pylori infection in long-term LDA users, assess the impact of eradication on PUB risk, and explore its association with cardiova
A prospective observational study was conducted at Benha Teaching Hospital from January 2023 to January 2024. Four hundred adults (≥ 18 years) on daily LDA for at least one year were enrolled. All participants underwent a comprehensive evaluation, including assessment of CV comorbidities. H. pylori was diagnosed via a stool antigen test, urea breath test, and histopathology. Infected patients received levofloxacin-based triple therapy for 14 days. Eradication was assessed at six weeks, and participants were followed for six months to evaluate PUB incidence and CV outcomes, including disease progression.
The prevalence of H. pylori infection was 65.5% (262/400). H. pylori-positive patients exhibited significantly lower albumin levels (3.5 ± 0.4 g/dL vs 3.9 ± 0.6 g/dL, P < 0.001) and higher levels of urea (29.3 ± 6.4 mg/dL vs 23.5 ± 4.6 mg/dL, P < 0.001), sodium (137.8 ± 2.9 mmol/L vs 135.3 ± 3.5 mmol/L, P < 0.001), potassium (4.1 ± 0.9 mmol/L vs 3.8 ± 0.6 mmol/L, P = 0.014), and cholesterol (191.0 ± 29.7 mg/dL vs 177.0 ± 35.9 mg/dL, P = 0.004). PUB was significantly more prevalent in H. pylori-positive patients with gastric erosions (12.2% vs 0.0%, P < 0.001), peptic ulcers (23.7% vs 0.0%, P < 0.001), and gastric ulcers (10.7% vs 0.0%, P < 0.001). After eradication therapy, 51.9% (136/262) of patients tested negative for H. pylori, while 48.1% (126/262) remained positive. PUB was significantly more frequent in patients with persistent H. pylori infection (8.7% at 6 weeks, 11.9% at 6 months) compared to those with successful eradication (1.5% at 6 weeks, 3.7% at 6 months, P < 0.001), highlighting the importance of successful eradication in reducing PUB risk. Additionally, H. pylori-positive patients had a higher prevalence of ischemic heart disease (54.3% vs 33.3%, P < 0.001) and a higher burden of CV disease progression during follow-up (hazard ratio = 3.5, P < 0.001), suggesting a potential interaction between H. pylori infection and CV risk.
Successful screening and eradicating H. pylori in high-risk LDA users was associated with significantly reduced PUB risk and was linked to less CV disease progression in patients with persistent infection.
Core Tip: This prospective observational study shows that persistent Helicobacter pylori infection markedly increases the risk of peptic ulcer bleeding and cardiovascular (CV) disease progression in long-term low-dose aspirin users. Successful eradication lowered ulcer bleeding rates from 11.9% to 3.7% - an approximately 69% relative risk reduction and was associated with significantly fewer CV events. These findings support routine Helicobacter pylori screening and eradication as a practical strategy to improve both gastrointestinal safety and CV outcomes in high-risk aspirin users.
- Citation: Semeya AA, Makled WA, Elnagdy MA, Elgamal R, Othman AAA. Helicobacter pylori eradication and the prevention of peptic ulcer bleeding and cardiovascular disease progression in chronic aspirin users. World J Gastroenterol 2026; 32(17): 117544
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/117544.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.117544
Helicobacter pylori (H. pylori) is a Gram-negative bacterium that colonizes the gastric mucosa and is pivotal in various gastrointestinal pathologies. Approximately 50% of the population globally harbors H. pylori, making it a significant public health concern[1]. Studies have shown that H. pylori infection is responsible for 80%-90% of peptic and duodenal ulcers, with infected individuals having a 3-10 times higher lifetime risk of developing peptic ulcer disease (PUD) than non-infected individuals[2,3]. Beyond gastrointestinal complications, H. pylori infection is associated with systemic effects, including chronic inflammatory responses, altered lipid and glucose metabolism, and deficiencies in vitamins such as B12, C, and D[4]. These systemic effects may contribute to comorbidities, such as coronary heart disease, under
In Egypt, the standard H. pylori eradication regimen involves a triple therapy consisting of a proton pump inhibitor (PPI) and two antibiotics-typically clarithromycin and either amoxicillin or metronidazole. However, due to rising antibiotic resistance, quadruple therapy, which includes bismuth compounds along with a PPI and two antibiotics, is gaining popularity for higher eradication success rates in many countries[6]. Adapting treatment to local resistance patterns is crucial for effectiveness, as resistance to clarithromycin and other antibiotics is common in the region. Treat
Aspirin, a widely used antiplatelet agent, is essential in preventing CVD[8]. LDA therapy offers substantial benefits in primary and secondary CVD prevention, notably decreasing the risk of CV events by 21% and reducing all-cause mortality by 13% in individuals with preexisting CVD[9,10]. Furthermore, emerging evidence suggests potential benefits of LDA in reducing the risk of colorectal cancer[11,12], cognitive impairment, dementia, and Alzheimer’s disease patients, which may lead to expanded usage[13-15]. However, these benefits are tempered by a 2-4-fold increase in the risk of severe bleeding in this population, including PUD and gastrointestinal bleeding[16-18]. This dual impact necessitates careful evaluation of individual risk factors when considering aspirin therapy for long-term use. H. pylori eradication typically involves a combination of antibiotics and PPIs.
The interplay between LDA use and H. pylori infection presents a significant clinical challenge[19]. Aspirin use exacerbates the risk of peptic ulcers and bleeding, particularly in the presence of H. pylori infection[16-18]. However, eradicating H. pylori has been shown to reduce the recurrence of peptic ulcers, particularly in high-risk individuals[20,21]. New study findings suggest that long-term aspirin use increases the likelihood of acquiring H. pylori infection and raises the recurrence rate of H. pylori-related conditions after eradication therapy[22]. Despite this, the global prevalence of H. pylori infection and the widespread use of LDA raise critical questions regarding the benefits of systematic H. pylori eradication in all aspirin users.
Given the clinical significance of this interplay, this study investigates the prevalence of H. pylori infection among long-term LDA users, examines the impact of H. pylori eradication on the incidence of peptic ulcer bleeding (PUB), and explores the potential association between H. pylori infection and CV outcomes. We hypothesize that persistent H. pylori infection exacerbates PUB risk and negatively influences CV health in long-term LDA users.
This prospective observational study was conducted at Benha Teaching Hospital from January 2023 to January 2024. The study aimed to evaluate the prevalence of H. pylori infection in long-term LDA users and assess the impact of H. pylori eradication on the risk of PUB. The hospital was selected due to its high prevalence of H. pylori infections and access to advanced diagnostic tools.
The study enrolled 400 adult patients (≥ 18 years) who had been on daily LDA therapy (81-325 mg) for at least one year. The sample size was calculated based on a 95% confidence level (Z-score = 1.96) and the latest H. pylori prevalence in Egypt of 48.9%[23]. The following formula for estimating the sample size in proportion studies was used: “n = [Z2 × P × (1 - P)]/E2”, Where: n = required sample size, Z = Z-score corresponding to the desired confidence level (1.96 for a 95%CI), P = estimated prevalence of H. pylori infection (0.489), and E = margin of error (0.05 for a 5% margin of error).
Calculation: n = [1.962 × 0.489 × (1 - 0.489)]/0.052 = (3.8416 × 0.489 × 0.511)/0.0025 = 0.960/0.0025 = 384.
To account for potential dropouts and ensure adequate power, the sample size was increased to 400 participants. A power analysis confirmed that this sample size provided 80% power to detect a 15% difference in the incidence of PUB between H. pylori-positive and H. pylori-negative groups, assuming a two-sided alpha level of 0.05. Wilson's method calculated confidence intervals (CIs) for prevalence estimates. The subjects were divided into G I (H. pylori-positive patients) and G II (H. pylori-negative patients; control group). Following gastroduodenoscopy, patients were monitored for six months. Eradication was assessed at six weeks, and follow-up evaluations were conducted to assess PUB incidence and CV outcomes.
This study was reported according to the STROBE guidelines for observational studies.
PUB: Was defined as overt gastrointestinal bleeding (hematemesis, melena, or hematochezia) accompanied by endosco
CVD progression: Was a pre-specified composite endpoint defined as the first occurrence during follow-up of: (1) New coronary revascularization procedure (PCI or CABG); (2) Hospitalization for unstable angina; or (3) Documented worsening of Canadian Cardiovascular Society (CCS) angina class by at least one grade. CV events were adjudicated by a board-certified cardiologist blinded to H. pylori status.
The current study was implemented in coordination with the guidelines of the Declaration of Helsinki. Ethical approval was gained according to the recommendations of the Ethics Committee of Benha Teaching Hospitals, Egypt, Approval No. HB-000131. Written informed consent was obtained from the patients, which addressed all the steps of the study and their right to withdraw at any time.
All consecutively enrolled participants in this study were adults (≥ 18 years) on daily LDA (81-325 mg) for at least one year as part of secondary prevention for established CVD [e.g., ischemic heart disease (IHD), prior myocardial infarction (MI), or stroke]. Aspirin therapy was continued throughout the study period, including during and after H. pylori eradication for those diagnosed, as per standard clinical guidelines for secondary prevention of CVD. No participants were advised to discontinue aspirin, and its use was monitored at each follow-up visit. In addition to aspirin, participants were on up to two other antithrombotic or anticoagulant medications [e.g., clopidogrel, warfarin, or direct oral anticoagulants (DOACs)] as indicated by their clinical condition. This inclusion criterion ensured a homogeneous cohort of patients at high risk for CV events, providing a stable population in which to study the added impact of H. pylori infection. Following enrollment, all participants underwent systematic diagnostic testing for H. pylori infection via a combination of stool antigen test, urea breath test (UBT), and histopathological examination during protocol-mandated upper gastrointestinal endoscopy. Participants who tested positive for H. pylori were required to be willing to undergo eradication therapy and adhere to the associated follow-up protocol. The use of all medications was recorded, and their potential confounding effects were adjusted for using multivariate regression analysis. Written informed consent was obtained from all participants before enrollment, ensuring their understanding of the study objectives and procedures and their right to withdraw at any time.
Participants were excluded from the study if they had a known allergic reaction or contraindication to any of the medications used in H. pylori eradication therapy, including levofloxacin, amoxicillin, or PPIs. Additionally, patients with severe comorbidities that could independently cause significant gastropathy or drastically alter the natural history of gastrointestinal or CV outcomes were excluded, including advanced liver disease (Child-Pugh B/C), end-stage renal disease (on dialysis), or active malignancies. Patients with portal hypertensive gastropathy, uremic gastropathy, or any cause of bleeding in the upper gastrointestinal tract (e.g., Mallory-Weiss tears, Dieulafoy’s lesion, and Aortoenteric fistula) were also excluded. Pregnant or lactating women were excluded due to potential risks to the fetus or infant. Patients with a documented history of prior H. pylori eradication therapy were excluded to avoid confounding by antibiotic resistance or treatment-related mucosal changes. Other exclusion criteria included recent use (within the past two months) of PPIs, antibiotics, or non-steroidal anti-inflammatory drugs (NSAIDs), as these could interfere with H. pylori detection and treatment efficacy. Chronic use of gastroprotective medications (PPIs or H2-receptor antagonists) outside of the study protocol was prohibited during the follow-up period to standardize the assessment of ulcer bleeding risk. Patients with prior gastric surgery (e.g., gastrectomy or vagotomy) or conditions associated with gastric acid hypersecretion, such as Zollinger-Ellison syndrome, were also excluded. Patients on excessively complex antithrombotic regimens (typically involving four or more agents) were excluded to minimize extreme heterogeneity in bleeding risk. Finally, individuals with a history of alcohol or drug abuse were not eligible for participation.
Baseline assessment: Before treatment, baseline data were collected, including demographic information (age, sex, body mass index), medical history (comorbidities, duration of LDA use), and gastrointestinal symptoms (abdominal pain, bloating, nausea, vomiting). Symptoms were assessed using the Gastrointestinal Symptom Rating Scale (GSRS).
Laboratory investigations: Fasting blood samples were collected from all participants to assess baseline biochemical parameters and monitor treatment outcomes. Approximately 5 mL of venous blood was drawn from each participant using sterile vacutainers. The blood samples were divided into two aliquots: One collected in ethylenediaminetetraacetic acid tubes for complete blood count (CBC) analysis, and the other in plain tubes for serum separation. The serum samples were centrifuged at 3000 rpm for 15 minutes, aliquoted, and stored at -20 °C until further analysis.
All samples were promptly sent to the laboratory, where serum tests for lipid profile (total cholesterol, triglycerides, low-density lipoprotein, high-density lipoprotein), liver function (alanine transaminase, aspartate transaminase, and albumin), kidney function (creatinine, urea), electrolytes [sodium (Na) and potassium (K)], coagulation profile (proth
Diagnosis of CVDs: CVDs, including IHD, percutaneous coronary intervention (PCI), coronary artery bypass grafting (CABG), chronic stable angina (CSA), MI, and valvular diseases, were diagnosed under the supervision of a board-certified cardiologist, ensuring the highest standard of clinical accuracy. Participants with suspected or known CV con
These diagnostic methods are widely accepted and recommended in clinical guidelines for the accurate identification of CVDs. The involvement of a cardiologist in the diagnostic process ensured that all cases were thoroughly evaluated and confirmed, minimizing the risk of misdiagnosis or oversight.
H. pylori infection was confirmed using a combination of three diagnostic modalities: Stool antigen testing, UBT, and upper gastrointestinal endoscopy with histopathological examination of gastric biopsies. This multimodal approach was employed to maximize diagnostic sensitivity and specificity, in line with recommendations for research settings where accurate baseline classification is critical. The non-invasive tests (stool antigen and UBT) provided functional confirma
Stool antigen testing: Stool samples were examined for H. pylori antigens using the rapid OnSite® one-step test (Cat. # R0192C, CTK Biotech, Inc., United States). This qualitative immunochromatographic test detects H. pylori antigens in stool samples. Following the manufacturer’s guidelines, the stool samples were assessed using the card test. A single red band at the control line indicated a negative result, while the presence of red bands at both the control and result lines indicated a positive result. The test was considered invalid if the control band was absent, regardless of the result band’s appearance.
UBT: The UBT was used to confirm H. pylori infection and assess eradication post-treatment. Participants were instructed to fast for at least 6 hours before the test. They were then given a labeled urea solution (containing 13C) to drink. If H. pylori was present in the stomach, the bacteria would break down the urea, releasing labeled carbon dioxide (CO2), which is absorbed into the bloodstream and exhaled in the breath. Breath samples were collected at baseline and 30 minutes after ingestion of the urea solution. The samples were analyzed using an infrared spectrometer (for 13C-UBT) or a scintillation counter (for 14C-UBT). A positive result was defined as a change in labeled CO2 concentration ≥ 4.0‰ for 13C-UBT or > 200 dpm for 14C-UBT, based on the manufacturer's guidelines (HeliFan Plus, Fischer Analysen Instrumente GmbH, Germany). The UBT was performed according to standardized protocols recommended by recent consensus guidelines[24].
Endoscopic procedures: All patients underwent upper gastrointestinal endoscopy using a standard video endoscope (Fujifilm EG-760Z). Biopsy samples were obtained according to the Updated Sydney System to optimize the detection of H. pylori and assess gastric mucosal pathology. A total of five biopsies were taken: Two from the antrum, two from the corpus, and one from the incisura angularis. Endoscopic findings, including gastric erosion, peptic ulcers, and other mucosal abnormalities, were documented. In this study, peptic ulcers refer to ulcers occurring in the stomach (gastric ulcers) or the duodenum (duodenal ulcers). Both types are caused by an imbalance between mucosal defense mechanisms and aggressive factors such as gastric acid, pepsin, and H. pylori infection. Gastric ulcers are located in the stomach lining, while duodenal ulcers occur in the duodenum. The distinction between these two types is clinically relevant, as their underlying causes, clinical presentations, and management strategies may differ. In this study, we analyzed gastric ulcers and duodenal ulcers separately to better understand their association with H. pylori infection and PUB.
Histopathological examination: Formalin fixation of biopsy samples obtained during endoscopy was performed, followed by paraffin embedding and staining with hematoxylin & eosin for routine histopathological examination. Additional staining with Giemsa or immunohistochemistry was performed to detect H. pylori infection. The degree of gastritis was determined using the Updated Sydney System, in which the severity of inflammation, atrophy, intestinal metaplasia, and H. pylori density were assessed[25]. All histopathological evaluations were performed by investigators blinded to the participants' H. pylori status to minimize potential bias.
Rationale for treatment regimen selection: Based on local Egyptian antibiotic resistance patterns showing high rates of clarithromycin and metronidazole resistance, and emerging evidence supporting levofloxacin-containing regimens in this context[26], we opted for a levofloxacin-based triple therapy.
Treatment regimen: Patients with confirmed H. pylori infection received levofloxacin-based triple therapy, consisting of levofloxacin 500 mg once daily, amoxicillin 1 g twice daily, and omeprazole 20 mg twice daily for 14 days. Omeprazole was selected as the PPI based on its inclusion in local Egyptian treatment guidelines, widespread availability in our hospital formulary, proven efficacy at this dose, and cost-effectiveness for our patient population[26]. Adherence was monitored using pill counts at follow-up visits, self-reported diaries, and pharmacy refill records to ensure accuracy and minimize recall bias. Participants with < 80% adherence were excluded from the per-protocol analysis. The primary analyses for all clinical outcomes (PUB and CVD progression) were performed on the intention-to-treat population, which included all enrolled patients according to their original H. pylori status group assignment, regardless of adherence or treatment completion. Sensitivity analyses using the per-protocol population yielded consistent results.
Follow-up and outcome assessment: Follow-up testing was performed six weeks post-treatment using stool antigen testing to confirm eradication success. The stool antigen test was selected as the primary follow-up tool due to its non-invasive nature, high concordance with the UBT in post-treatment settings, and superior practicality in our clinical setting. Performing repeat endoscopy in all asymptomatic patients was not ethically or practically justified. Endoscopy was reserved only for cases with suspected PUB or persistent symptoms requiring direct visualization. Similarly, the UBT was not routinely repeated to minimize patient burden and cost. The stool antigen test provided a reliable and validated alternative for assessing the success of eradication. Patients were instructed to discontinue PPIs for at least two weeks before testing. Eradication was defined as a negative stool antigen test at this time point. Patients were followed for six months to monitor PUB incidence and CV events. Symptom resolution was evaluated using the GSRS at baseline, 6 weeks, and 6 months. Upper gastrointestinal endoscopy was repeated in cases of suspected PUB or persistent symptoms. Adverse events were recorded and graded according to the Common Terminology Criteria for Adverse Events (CTCAE).
Data were analyzed using SPSS version 28 (IBM Corp., United States). Continuous variables were expressed as mean ± SD, and categorical variables as n (%). Differences between groups were assessed using Student’s t-test for normally distributed continuous variables and Pearson’s χ2 test for categorical variables. Kaplan-Meier survival analysis was employed to evaluate time-to-event data for PUB and CV outcomes, with group comparisons made using the log-rank test. Normality was assessed using the Shapiro-Wilk test. Based on these results, normally distributed variables are presented as mean ± SD and compared using Student’s t-test; non-normally distributed variables are presented as median (interquartile range) and compared using the Mann-Whitney U test. Categorical variables are presented as n (%) and compared using Pearson’s χ2 test.
Primary outcomes and multiple comparisons: The pre-specified primary outcomes of this study were PUB and CVD progression (as defined in the “Analysis of CV status” subsection below). All other comparisons, including biochemical parameters, symptom scores, and subgroup analyses, were considered exploratory. To mitigate the risk of type I error inflation due to multiple testing, we applied the Benjamini-Hochberg false discovery rate (FDR) correction to all exploratory univariate and subgroup comparisons. Adjusted P values (q values) are reported alongside nominal P values in relevant tables. For regression models (Cox and logistic), which focused on the primary outcomes and their pre-specified covariates, no further multiplicity adjustment was performed, as recommended for confirmatory analyses of primary endpoints.
Cox proportional hazards regression was used to identify independent predictors of PUB and CV outcomes. All Cox models were adjusted for age, sex, diabetes mellitus, baseline albumin levels, and adherence to treatment. For models predicting CVD progression, baseline multivessel coronary disease was also included as a covariate. The proportional hazards assumption was verified using Schoenfeld residuals; no significant violations were detected (P > 0.05 for all covariates). Models were fit using complete-case analysis (n = 400). Adjusted hazard ratios (aHRs) with 95%CIs are reported.
Multivariate logistic regression was conducted to analyze predictors of PUB, treatment failure, and adverse events, adjusting for potential confounders, including diabetes status, and including the impact of combination antithrombotic and anticoagulant therapy (e.g., aspirin + clopidogrel, aspirin + warfarin, aspirin + DOACs). All logistic regression models were adjusted for the same covariates as the Cox models, where applicable. Adjusted odds ratios (aORs) with 95%CIs are reported.
Subgroup analyses were performed to evaluate the impact of demographic and clinical factors, including age, sex, and comorbidities, on outcomes of interest. For age, patients were stratified into two groups: ≥ 65 years and < 65 years. For sex, comparisons were made between males and females. Comorbidities were assessed by comparing patients with IHD to those without. Outcomes for patients with persistent H. pylori infection were compared to those with successful eradication. Patients with high adherence (≥ 80% of prescribed medication) were compared to those with low adherence (< 80%). Participants with hypoalbuminemia (albumin < 3.5 g/dL) were compared to those with normal levels, and those with elevated urea levels (> 25 mg/dL) were analyzed as a subgroup. Hazard ratios (HRs) with 95%CIs and corresponding P values were reported for each subgroup. A P value < 0.05 was considered statistically significant. To further investigate potential differences among patients with IHD, PCI, and CABG, we conducted an additional subgroup analysis. This allowed us to assess whether CV outcomes differed significantly among these subgroups.
Analysis of CV status: Given the relatively short (6-month) follow-up period and the chronic nature of atherosclerotic disease, the analysis of CV 'outcomes' focused on the prevalence and progression of CVD status during follow-up, rather than solely on incident acute events. For patients with baseline CVD, progression was defined as: A new coronary revascularization procedure (PCI or CABG), hospitalization for unstable angina, or a documented change in CCS angina class. For Kaplan-Meier and regression analyses, the time-to-event was defined as the time to the first documentation of this progressive disease status during follow-up. Patients without baseline CVD who developed new-onset angina with objective evidence of ischemia were also considered to have reached this endpoint.
Handling of missing data: Missing baseline laboratory values (e.g., albumin, lipids) were handled using multiple imputation with predictive mean matching, generating 20 imputed datasets. The imputation model included all analysis variables and auxiliary predictors of missingness. For longitudinal outcomes (e.g., GSRS scores at follow-up), missing data in the per-protocol analysis were addressed using the last observation carried forward (LOCF) approach only when a participant had at least one post-baseline measurement and dropout was unrelated to the outcome. Complete-case analysis was used for sensitivity testing, and results were consistent across methods. Analyses were conducted on the imputed datasets for baseline comparisons and on the LOCF-modified dataset for longitudinal per-protocol analyses, ensuring no inappropriate mixing of imputation strategies.
The study population enrolled 400 participants, comprising 216 males (54.0%) and 184 females (46.0%), with a mean age of 60.2 years. As shown in Table 1, there was no significant difference in age or sex distribution between H. pylori-positive and H. pylori-negative groups (P = 0.938 and P = 0.604, respectively), indicating homogeneity in these baseline characte
| Category | Variable | H. pylori-positive | H. pylori-negative | Nominal P value | Adjusted q value |
| Sociodemographic | Age (years) | 60.2 ± 9.2 | 60.3 ± 7.3 | 0.938 | 0.938 |
| Male | 138 (52.7) | 78 (56.5) | 0.604 | 0.672 | |
| Female | 124 (47.3) | 60 (43.5) | 0.604 | 0.672 | |
| Diabetes mellitus | 72 (27.5) | 43 (31.2) | 0.452 | 0.603 | |
| Biochemical | ALT (IU/L), median (interquartile range) | 40.4 (22.6) | 36.3 (28.8) | 0.269 | 0.403 |
| AST (IU/L), median (interquartile range) | 42.7 (23.2) | 37.9 (20.6) | 0.150 | 0.300 | |
| Albumin (g/dL) | 3.5 ± 0.4b | 3.9 ± 0.6 | < 0.001 | < 0.001 | |
| Urea (mg/dL) | 29.3 ± 6.4b | 23.5 ± 4.6 | < 0.001 | < 0.001 | |
| Sodium (mmol/L) | 137.8 ± 2.9b | 135.3 ± 3.5 | < 0.001 | < 0.001 | |
| Potassium (mmol/L) | 4.1 ± 0.9a | 3.8 ± 0.6a | 0.014 | 0.028 | |
| Cholesterol (mg/dL) | 191.0 ± 29.7a | 177.0 ± 35.9a | 0.004 | 0.012 | |
| Triglycerides (mg/dL), median (interquartile range) | 94.6 (58.6) | 87.6 (55.2) | 0.414 | 0.518 | |
| HDL (mg/dL), median (interquartile range) | 43.1 (41.2) | 53.5 (50.2) | 0.118 | 0.236 | |
| LDL (mg/dL), median (interquartile range) | 156.3 (88.9) | 140.6 (81.5) | 0.224 | 0.374 | |
| Random blood sugar (mg/dL), median (interquartile range) | 128.5 (89.2) | 130.1 (92.3) | 0.850 | 0.850 | |
| HbA1c (%) | 6.8 ± 1.2 | 6.9 ± 1.3 | 0.587 | 0.671 | |
| Digestive symptoms (GSRS) | Total GSRS score | 2.9 ± 0.8b | 1.7 ± 0.6 | < 0.001 | < 0.001 |
| Abdominal pain | 90 (34.4)a | 30 (21.7) | 0.012 | 0.024 | |
| Bloating | 60 (22.9) | 20 (14.5) | 0.045 | 0.068 | |
| Nausea | 50 (19.1)a | 10 (7.2) | 0.003 | 0.009 |
The prevalence of diabetes mellitus in the overall cohort was 28.8% (115/400), reflecting the high CV risk profile of the study population. No significant difference in diabetes prevalence was observed between H. pylori-positive (27.5%, 72/262) and H. pylori-negative (31.2%, 43/138) groups (P = 0.452). Similarly, mean HbA1c levels were comparable between groups (6.8% ± 1.2% vs 6.9% ± 1.3%, P = 0.587), indicating balanced glycemic status at baseline. These findings confirm that diabetes was not a confounding factor in the initial group allocation and was subsequently adjusted for in all multivariate analyses.
Gastrointestinal symptoms were evaluated using the GSRS at baseline. H. pylori-positive patients had significantly higher total GSRS scores (2.9 ± 0.8 vs 1.7 ± 0.6, P < 0.001) compared to H. pylori-negative patients, indicating greater symptom severity. Additionally, individual symptoms were more prevalent in H. pylori-positive patients, with abdominal pain reported in 34.4% vs 21.7% (P = 0.012), bloating in 22.9% vs 14.5% (P = 0.045), and nausea in 19.1% vs 7.2% (P = 0.003) (Table 1). These findings highlight the increased burden of gastrointestinal symptoms among infected patients, emphasizing the importance of H. pylori eradication to improve symptom outcomes.
However, significant differences were observed in biochemical and endoscopic parameters. H. pylori-positive patients had significantly lower albumin levels (3.5 ± 0.4 g/dL vs 3.9 ± 0.6 g/dL, P < 0.001) and significantly higher levels of urea (29.3 ± 6.4 mg/dL vs 23.5 ± 4.6 mg/dL, P < 0.001), Na (137.8 ± 2.9 mmol/L vs 135.3 ± 3.5 mmol/L, P < 0.001), K (4.1 ± 0.9 mmol/L vs 3.8 ± 0.6 mmol/L, P = 0.014), and cholesterol (191.0 ± 29.7 mg/dL vs 177.0 ± 35.9 mg/dL, P = 0.004) compared to H. pylori-negative patients (Table 1). The significantly lower albumin levels and higher urea levels in H. pylori-positive patients may reflect chronic inflammation and systemic effects associated with persistent infection.
H. pylori infection was confirmed in 65.5% (262/400) of participants using a combination of stool antigen testing, UBT, upper gastrointestinal endoscopy, and histopathological examination.
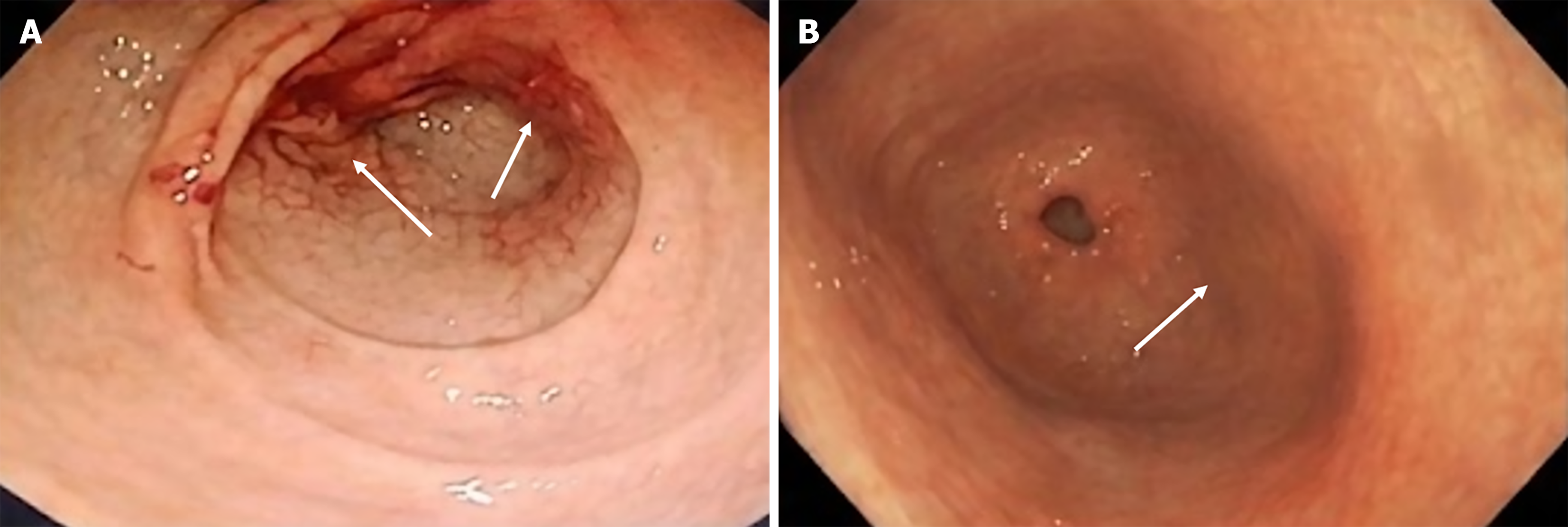
Endoscopic findings are critical for diagnosing H. pylori-related gastric pathologies and assessing the severity of mucosal damage. The endoscopic examination revealed significant differences between H. pylori-positive and H. pylori-negative patients. Among H. pylori-positive patients, 23.7% (62/262) had atrophic gastritis, 12.2% (34/262) had gastric erosions, and 10.7% (28/262) had gastric ulcers. Duodenal ulcers were less common, identified in 2.3% (6/262) of H. pylori-positive patients. The higher prevalence of gastric vs duodenal ulcerative lesions in our cohort may be explained by several factors: (1) The predominant gastric mucosal injury associated with long-term aspirin use, which is more strongly associated with gastric rather than duodenal ulceration; (2) The advanced age and CV risk profile of our patients on concomitant antithrombotic therapy; and (3) Potential regional differences in H. pylori strain virulence. In contrast, none of the H. pylori-negative patients exhibited these lesions (P < 0.001 for all comparisons) (Figure 1, Table 2).
| Category | Variable | H. pylori-positive (n = 262) | H. pylori-negative (n = 138) | P value |
| Endoscopic | Normal | 122 (46.6) | 136 (98.6) | < 0.001 |
| Atrophic gastritis | 62 (23.7) | 0 (0.0) | < 0.001 | |
| Gastric erosion | 34 (12.2) | 0 (0.0) | < 0.001 | |
| Gastric ulcer | 28 (10.7) | 0 (0.0) | < 0.001 | |
| Antral erosion | 6 (2.3) | 0 (0.0) | 0.378 | |
| Duodenal ulcer | 6 (2.3) | 0 (0.0) | 0.378 | |
| Duodenitis | 6 (2.3) | 0 (0.0) | 0.378 | |
| Histopathological | H. pylori colonization | 262 (100) | 0 (0.0) | < 0.001 |
| Inflammation severity | ||||
| Mild | 80 (30.5) | 40 (29.0) | 0.75 | |
| Moderate | 120 (45.8) | 30 (21.7) | < 0.001 | |
| Severe | 50 (19.1) | 0 (0.0) | < 0.001 | |
| Atrophy | 40 (15.3) | 0 (0.0) | < 0.001 | |
| Intestinal metaplasia | 20 (7.6) | 0 (0.0) | 0.002 |
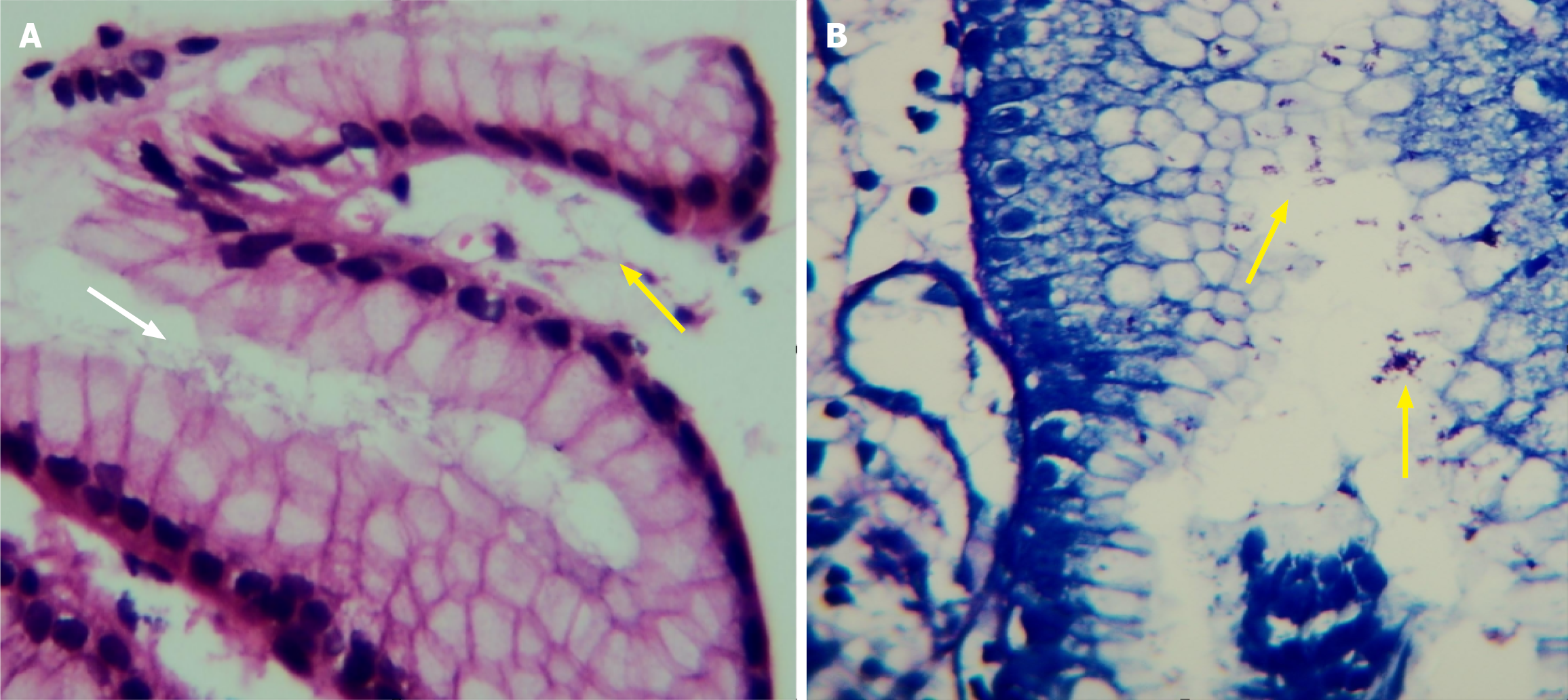
Histopathological examination provides definitive evidence of H. pylori colonization and the degree of gastric inflammation, atrophy, and intestinal metaplasia. Histopathological examination of biopsy samples confirmed H. pylori colonization in 65.5% (262/400) of patients. Histopathological examination using the Updated Sydney System revealed that H. pylori-positive patients had significantly higher rates of moderate to severe inflammation (78.3% vs 12.3%, P < 0.001), atrophy (15.3% vs 0.0%, P < 0.001), and intestinal metaplasia (7.6% vs 0.0%, P = 0.002) compared to H. pylori-negative patients. These findings underscore the role of H. pylori in inducing chronic gastric inflammation and precancerous changes (Figure 2, Table 2).
The distribution of diseases associated with LDA use is presented in Table 3. IHD was the most prevalent condition, accounting for 47.1% of the study population, with a significantly higher prevalence in H. pylori-positive patients (54.3%) compared to H. pylori-negative patients (33.3%; P < 0.001). PCI and IHD-PCI were more common in H. pylori-negative patients (12.5% each) than in H. pylori-positive patients (8.7% and 6.5%, respectively; P = 0.012 for PCI, P = 0.018 for IHD-PCI). Conversely, CABG was more prevalent in H. pylori-positive patients (17.4%) compared to H. pylori-negative patients (12.5%; P = 0.045).
| Disease | H. pylori positive | H. pylori negative | P value |
| IHD | 142 (54.3) | 46 (33.3) | < 0.001 |
| PCI | 23 (8.7) | 17 (12.5) | 0.012 |
| IHD-PCI | 17 (6.5) | 17 (12.5) | 0.018 |
| CABG | 46 (17.4) | 17 (12.5) | 0.045 |
| CSA | 6 (2.1) | 0 (0.0) | 0.078 |
| Post CABG | 11 (4.2) | 17 (12.5) | 0.064 |
| Post PCI | 6 (2.1) | 0 (0.0) | 0.210 |
| Inferior MI | 6 (2.1) | 6 (4.2) | 0.320 |
| Prosthetic MV | 6 (2.1) | 6 (4.2) | 0.320 |
| Post PCS | 6 (2.1) | 0 (0.0) | 0.210 |
These findings suggest a potential interaction between H. pylori infection and CV risk, which may influence the management of long-term LDA users. The significantly higher prevalence of IHD in H. pylori-positive patients under
To further explore the relationship between PUB and CVDs, we compared the prevalence of specific CV conditions in patients with and without PUB. As shown in Table 4, patients with PUB had a significantly higher prevalence of IHD (54.3% vs 33.3%, P < 0.001) and were more likely to undergo CABG (17.4% vs 12.5%, P = 0.045). These findings suggest that PUB is associated with an increased burden of CVD in long-term aspirin users.
| Cardiovascular disease | PUB incidence | No PUB incidence | Adjusted odds ratio (95%CI) | P value |
| Category | 54.3% | 33.3% | 2.5 (1.3-4.8) | 0.006 |
| Endoscopic | 17.4% | 12.5% | 1.8 (1.2-2.7) | 0.045 |
| Histopathological | 8.7% | 6.0% | 1.2 (0.8-1.8) | 0.120 |
| Category | 2.1% | 1.5% | 1.1 (0.9-1.3) | 0.320 |
All participants in this study were on daily LDA in combination with one or more additional antithrombotic or anticoagulant medications. When analyzed using the total study cohort as the denominator, stratification by the type of additional medication revealed no significant differences in the incidence of PUB or CV events between groups (e.g., aspirin plus clopidogrel vs aspirin plus warfarin; P > 0.05). In contrast, participants receiving triple therapy (aspirin plus two additional agents) exhibited a significantly higher incidence of PUB compared with those on dual therapy (aspirin plus one additional agent) (15.2% vs 8.7%, P = 0.032). Multivariate regression analysis confirmed that the number of concomitant antithrombotic or anticoagulant medications was an independent predictor of PUB, with triple therapy associated with a significantly increased risk compared with dual therapy (aOR = 1.8, 95%CI: 1.2-2.7, P = 0.004) (Table 5).
| Medication group | PUB incidence n/N (%) | Cardiovascular events n/N (%) | Adjusted odds ratio for PUB | 95%CI for PUB | P value for PUB |
| By type of additional medication | |||||
| Aspirin + clopidogrel | 42/400 (10.5) | 101/400 (25.3) | 1.2 | 0.8-1.8 | 0.120 |
| Aspirin + warfarin | 39/400 (9.8) | 99/400 (24.7) | 1.1 | 0.7-1.6 | 0.150 |
| Aspirin + DOACs | 45/400 (11.2) | 104/400 (26.1) | 1.3 | 0.9-1.9 | 0.200 |
| By number of medications | |||||
| Dual therapy (aspirin + 1 drug) | 35/400 (8.7) | 94/400 (23.5) | 1.0 (reference) | - | - |
| Triple therapy (aspirin + 2 drugs) | 61/400 (15.2) | 116/400 (28.9) | 1.8 | 1.2-2.7 | 0.004 |
Following the 6-week post-treatment assessment, H. pylori-positive patients were categorized into two groups: (1) The eradicated group (n = 136), defined by a negative stool antigen test at 6 weeks; and (2) The persistent infection group (n = 126), defined by a positive stool antigen test at 6 weeks. The same 126 patients in the persistent infection group remained positive at the 6-month follow-up assessment. Therefore, the ‘persistent 6-week post-treatment’ and ‘persistent 6-month follow-up’ columns in Table 6 represent the same cohort assessed at two different time points, allowing comparison of outcomes between eradicated and persistently infected patients alongside the H. pylori-negative group (n = 138). Overall, H. pylori eradication was achieved in 51.9% of infected patients (136/262), reflecting the challenges of achieving optimal treatment outcomes in this setting.
| Category | Variable | H. pylori-positive group | H. pylori-negative group | P value (eradicated vs persistent 6-week post-treatment) | P value (persistent 6-month vs negative) | ||
| Eradicated | Persistent 6-week post-treatment | Persistent 6-month follow-up (n = 126) | |||||
| Gastric | PUB | 5 (3.7) | 26 (20.6) | 15 (11.9) | 0 (0.0) | < 0.001 | < 0.001 |
| Outcomes | Gastric erosions | 8 (5.9) | 26 (20.6) | 12 (9.5) | 0 (0.0) | < 0.001 | < 0.001 |
| Gastric ulcer | 6 (4.4) | 22 (17.5) | 10 (7.9) | 0 (0.0) | < 0.001 | < 0.001 | |
| Antral erosion | 2 (1.5) | 6 (4.8) | 2 (1.6) | 0 (0.0) | 0.02 | 0.06 | |
| Duodenal ulcer | 3 (2.2) | 8 (6.3) | 4 (3.2) | 0 (0.0) | 0.03 | 0.08 | |
| CV disease progression status1 | CV disease progression | 40 (29.4) | 96 (76.2) | 92 (73.0) | 44 (32.0) | < 0.001 | < 0.001 |
| PCI | 10 (7.4) | 11 (8.7) | 10 (7.9) | 15 (11.0) | 0.05 | 0.06 | |
| IHD-PCI | 16 (11.8) | 22 (17.5) | 20 (15.9) | 6 (4.3) | 0.01 | 0.02 | |
| CABG | 12 (8.8) | 30 (23.8) | 28 (22.2) | 17 (12.0) | 0.002 | 0.005 | |
| CSA | 4 (2.9) | 6 (4.8) | 3 (2.4) | 0 (0.0) | 0.03 | 0.07 | |
| Post CABG | 5 (3.7) | 12 (9.5) | 10 (7.9) | 4 (2.9) | 0.07 | 0.08 | |
| Post PCI | 5 (3.7) | 6 (4.8) | 5 (4.0) | 0 (0.0) | 0.22 | 0.33 | |
| Inferior MI | 4 (2.9) | 6 (4.8) | 4 (3.2) | 0 (0.0) | 0.33 | 0.34 | |
| Prosthetic MV | 4 (2.9) | 6 (4.8) | 4 (3.2) | 0 (0.0) | 0.33 | 0.34 | |
| Post PCS | 4 (2.9) | 6 (4.8) | 4 (3.2) | 0 (0.0) | 0.22 | 0.33 | |
| Adverse events | Fatigue | 4 (2.9) | 14 (11.1) | 10 (7.9) | 8 (5.8) | 0.01 | 0.02 |
| Dizziness | 3 (2.2) | 10 (7.9) | 6 (4.8) | 5 (3.6) | 0.02 | 0.03 | |
| Skin Rash | 2 (1.5) | 6 (4.8) | 4 (3.2) | 2 (1.4) | 0.02 | 0.04 | |
Gastric outcomes demonstrated significant differences among these subgroups. PUB was overwhelmingly more frequent in patients with persistent H. pylori infection, with the highest incidence in the persistent 6-week post-treatment group (20.6%), but decreased slightly by the 6-month follow-up in the persistent follow-up group (11.9%). In contrast, the eradicated group maintained a significantly lower but not absent rate of PUB (3.7%, P < 0.001 compared to both persistent groups). The few PUB events occurring despite successful eradication highlight the persistent, independent ulcerogenic risk of long-term aspirin use.
CV outcomes were also markedly different between the subgroups. The burden of CVD progression (as defined by new revascularization, hospitalization for angina, or worsening angina class) was highest in the persistent 6-week post-treatment group (76.2%), decreased slightly in the persistent 6-month follow-up group (73.0%), but remained significantly higher than in the eradicated group (29.4%, P < 0.001). CABG was performed most frequently in the persistent 6-week post-treatment group (23.8%), compared to 8.8% in the eradicated group (P = 0.002).
Adverse events were predominantly observed in the persistent 6-week post-treatment group, including fatigue (11.1%) and dizziness (7.9%), which decreased only slightly by the 6-month follow-up period (7.9% and 4.8%, respectively). These rates were significantly higher than in the eradicated group (2.9% and 2.2%, respectively; P = 0.010 for fatigue and P = 0.020 for dizziness). The higher burden of adverse events in the Persistent groups highlights the lingering systemic effects of H. pylori infection.
This stratification underscores the critical importance of achieving eradication and maintaining adherence to treatment protocols. Despite the moderate eradication rate of 51.9%, the data demonstrate significant reductions in both gastric and CV risks among successfully treated patients. The inclusion of both 6-week and 6-month follow-ups in the study metho
Kaplan-Meier survival analysis was performed to evaluate the time-to-event data for PUB and CV outcomes in 6 weeks and 6 months. The cumulative incidence of PUB was significantly higher in H. pylori-positive patients with persistent infection compared to the eradicated and H. pylori-negative groups (log-rank test, P < 0.001). At 6 weeks, 8.7% of patients with persistent infection experienced PUB, compared to 1.5% in the eradicated group and 0% in the H. pylori-negative group. By 6 months, the cumulative incidence of PUB increased to 11.9% in the persistent infection group, while reaching 3.7% in the eradicated group and 0% in the H. pylori-negative group. These findings emphasize the critical role of eradication in preventing long-term complications.
For incident CVD progression, defined as new revascularization, hospitalization for angina, or worsening angina class occurring during follow-up, the persistent infection group had a significantly higher burden compared to the eradicated and H. pylori-negative groups. At 6 weeks, the cumulative incidence of new CV disease progression was 8.0% in the persistent infection group, compared to 3.7% in the eradicated group and 4.3% in the H. pylori-negative group. By 6 months, this increased to 14.3% in the persistent group, while reaching 5.9% in the eradicated group and 7.2% in the negative group. The approximately 2.4-fold higher risk of CV disease progression in the persistent group compared to the eradicated group underscores the potential CV implications of ongoing H. pylori infection. Similarly, new procedures related to disease progression (e.g., CABG, PCI) were more frequent in the persistent infection group at both time points, though these differences did not reach statistical significance (Table 7).
| Outcome | Time point | H. pylori-positive (persistent), incidence (95%CI) | H. pylori-positive (eradicated), incidence (95%CI) | H. pylori-negative, incidence (95%CI) | P value |
| PUB | 6 weeks | 8.7% (5%-13%) | 1.5% (0.5%-4%) | 0% (0%-2%) | < 0.001 |
| 6 months | 11.9% (8%-17%) | 3.7% (2%-7%) | 0% (0%-2%) | < 0.001 | |
| Incident CV disease progression | 6 weeks | 8.0% (5%-12%) | 3.7% (2%-7%) | 4.3% (2%-8%) | 0.045 |
| 6 months | 14.3% (10%-20%) | 5.9% (3%-10%) | 7.2% (4%-12%) | 0.002 | |
| New CABG procedure | 6 weeks | 2.4% (1%-6%) | 0.7% (0.2%-3%) | 1.4% (0.5%-4%) | 0.210 |
| 6 months | 4.0% (2%-8%) | 1.5% (0.5%-4%) | 2.9% (1%-6%) | 0.120 | |
| New PCI procedure | 6 weeks | 3.2% (1%-7%) | 1.5% (0.5%-4%) | 2.2% (1%-5%) | 0.320 |
| 6 months | 5.6% (3%-10%) | 2.9% (1%-6%) | 4.3% (2%-8%) | 0.180 |
Adjusted Cox regression analysis identified H. pylori infection (aHR = 4.2, 95%CI: 2.1-8.3, P < 0.001) and low albumin levels (aHR = 2.8, 95%CI: 1.5-5.2, P = 0.002) as independent predictors of PUB. Age (aHR = 1.1, 95%CI: 0.9-1.3, P = 0.320) and sex (aHR = 1.2, 95%CI: 0.8-1.8, P = 0.450) were not significantly associated with PUB.
For CVD progression, H. pylori infection (aHR = 3.5, 95%CI: 1.8-6.7, P < 0.001) and multivessel coronary disease at baseline (aHR = 2.8, 95%CI: 1.5-5.2, P = 0.001) were significant predictors, while age and sex were not significant (Table 8).
| Variable | Adjusted hazard ratio | 95%CI | P value |
| Predictors of PUB | |||
| H. pylori infection | 4.2 | 2.1-8.3 | < 0.001 |
| Low albumin levels | 2.8 | 1.5-5.2 | 0.002 |
| Diabetes mellitus | 1.3 | 0.8-2.0 | 0.28 |
| Age | 1.1 | 0.9-1.3 | 0.32 |
| Sex (male) | 1.2 | 0.8-1.8 | 0.45 |
| Predictors of CV disease progression | |||
| H. pylori infection | 3.5 | 1.8-6.7 | < 0.001 |
| Multivessel coronary disease at baseline | 2.8 | 1.5-5.2 | 0.001 |
| Diabetes mellitus | 1.6 | 1.0-2.5 | 0.045 |
| Age | 1 | 0.9-1.2 | 0.55 |
| Sex (male) | 1.1 | 0.8-1.5 | 0.6 |
| Variable | Adjusted odds ratio | 95%CI | P value |
| Predictors of PUB | |||
| H. pylori infection | 4.2 | 2.1-8.3 | < 0.001 |
| Low albumin levels | 2.8 | 1.5-5.2 | 0.002 |
| Diabetes mellitus | 1.2 | 0.8-1.9 | 0.35 |
| Poor adherence | 3.1 | 1.8-5.4 | < 0.001 |
| Predictors of CV disease progression | |||
| H. pylori infection | 3.2 | 1.7-6.0 | < 0.001 |
| Multivessel coronary disease at baseline | 2.8 | 1.5-5.2 | 0.001 |
| Diabetes mellitus | 1.5 | 1.0-2.4 | 0.04 |
| Poor adherence | 2.1 | 1.1-3.9 | 0.012 |
Adjusted multivariate logistic regression identified several independent predictors of adverse outcomes. H. pylori infec
Similarly, H. pylori infection (aOR = 3.2, 95%CI: 1.7-6.0, P < 0.001), multivessel coronary disease at baseline (aOR = 2.8, 95%CI: 1.5-5.2, P = 0.001), and poor adherence (aOR = 2.1, 95%CI: 1.1-3.9, P = 0.012) were significant contributors to CVD progression (Table 9).
Adherence to the treatment regimen, defined as taking ≥ 80% of prescribed medications, was observed in 85.5% of participants (342/400). Adherence rates were notably higher in the eradicated group (92.6%, 126/136) compared to the persistent infection group (78.6%, 99/126). These findings highlight the critical role of adherence in reducing the risk of both PUB and CV complications. Poor adherence (< 80%) significantly increased the likelihood of adverse outcomes, emphasizing the importance of patient education and support to improve adherence and achieve better clinical outcomes.
Histopathology demonstrated the highest sensitivity (95%) and specificity (98%) for H. pylori detection. The stool antigen test and UBT showed good concordance with histopathology (kappa = 0.85 and 0.82, respectively) (Table 10).
| Diagnostic method | Sensitivity (%) | 95%CI | Specificity (%) | 95%CI | Kappa statistic |
| Stool antigen test | 90 | 85-94 | 92 | 88-95 | 0.85 |
| Urea breath test | 88 | 83-92 | 90 | 86-93 | 0.82 |
| Histopathology | 95 | 91-98 | 98 | 95-99 | 1.00 |
Adverse events were recorded and graded using the CTCAE. The most common adverse events included fatigue, dizziness, skin rash, nausea, and diarrhea.
The findings related to adverse events at the 6-week follow-up, presented in Table 11, reveal a higher incidence of fatigue, dizziness, skin rash, nausea, and diarrhea in patients with persistent H. pylori infection compared to those whose infection was eradicated or those who were H. pylori-negative. Fatigue was the most prevalent, affecting 11.1% of patients, and it was notably more common in the persistent infection group (11.1%) compared to the eradicated (2.9%) and negative groups (5.8%). Dizziness also showed a similar trend, with 7.9% of patients in the persistent infection group reporting it, compared to 2.2% in the eradicated group and 3.6% in the H. pylori-negative group. Additionally, nausea, skin rash, and diarrhea were more frequently reported in the persistent infection group. These findings suggest that adverse events may be linked to the persistence of H. pylori infection or possibly to the side effects of treatment. Logistic regression analysis indicated that these adverse events were significant predictors of lower treatment adherence, with fatigue, dizziness, and nausea showing strong associations (OR = 2.5, 2.2, and 2.0 respectively).
| Adverse event | Frequency (%) | OR (95%CI) | P value |
| Fatigue | 11.1 | 2.5 (1.3-4.8) | 0.006 |
| Dizziness | 7.9 | 2.2 (1.1-4.2) | 0.020 |
| Skin rash | 4.8 | 1.8 (1.0-3.2) | 0.040 |
| Nausea | 5.6 | 2.0 (1.1-3.6) | 0.030 |
| Diarrhea | 3.2 | 1.5 (0.8-2.8) | 0.200 |
In comparison to Table 6, which shows the outcomes in H. pylori-positive and negative patients across various follow-up periods, adverse events in Table 10 highlight a different set of concerns. While Table 6 focuses on gastric and CV outcomes, Table 10 specifically addresses the side effects related to treatment adherence. The adverse events reported in Table 6 (fatigue, dizziness, and skin rash) appear in a broader context of gastric issues such as gastric erosions, ulcers, and other conditions, with significant differences observed between the eradicated and persistent infection groups. The higher frequency of adverse events in patients with persistent infection from both tables underscores the ongoing challenges in managing H. pylori-related conditions, with implications for treatment adherence and overall patient outcomes.
The subgroup analysis revealed several significant findings regarding the impact of age, sex, comorbidities, and other factors on the risk of PUB and CV events. Older age (≥ 65 years) was associated with an increased risk of both PUB (HR = 2.0) and CV events (HR = 1.8), indicating that older patients are more vulnerable to these adverse outcomes. Additionally, males exhibited a higher risk of PUB (HR = 1.7) and CV events (HR = 1.5) compared to females, highlighting the need for gender-specific prevention strategies. The presence of IHD was a strong risk factor for PUB (HR = 2.8) and treatment failure (HR = 2.4), suggesting that patients with IHD may require more intensive management to mitigate these risks. Eradication of H. pylori significantly reduced both PUB (HR = 3.5) and CV events (HR = 3.2), emphasizing the importance of successful H. pylori treatment in reducing long-term complications.
Subgroup analysis by diabetes status revealed that while diabetes was associated with increased CV events (HR = 1.7, 95%CI: 1.1-2.5, P = 0.015), it was not a significant independent predictor of PUB (HR = 1.4, 95%CI: 0.9-2.1, P = 0.120) after adjustment for H. pylori infection status and other covariates. This suggests that the effect of H. pylori on PUB risk ope
Moreover, low adherence to treatment was strongly associated with higher risks of PUB (HR = 2.6) and treatment failure (HR = 3.1), further underlining the critical role of patient adherence in improving clinical outcomes. Biochemical markers, such as hypoalbuminemia (HR = 2.5 for PUB) and high urea levels (HR = 1.9 for CV events), also identified high-risk subgroups. These findings suggest that targeted interventions addressing these factors could reduce the incidence of PUB and CV events in high-risk populations (Table 12).
| Subgroup | Outcome | HR | 95%CI | Nominal P value | Adjusted q value |
| Age | Risk of PUB (≥ 65 years vs < 65 years) | 2 | 1.4-2.9 | 0.002 | 0.003 |
| CV events (≥ 65 years vs < 65 years) | 1.8 | 1.2-2.5 | 0.008 | 0.009 | |
| Sex | Risk of PUB (males vs females) | 1.7 | 1.2-2.3 | 0.011 | 0.013 |
| CV events (males vs females) | 1.5 | 1.1-2.1 | 0.042 | 0.042 | |
| Comorbidities | Risk of PUB (IHD vs no IHD) | 2.8 | 1.6-5.1 | 0.004 | 0.005 |
| Treatment failure (IHD vs no IHD) | 2.4 | 1.3-4.3 | 0.006 | 0.007 | |
| Diabetes status | Risk of PUB (DM vs no DM) | 1.4 | 0.9-2.1 | 0.12 | 0.12 |
| CV event (DM vs no DM) | 1.7 | 1.1-2.5 | 0.015 | 0.018 | |
| Eradication status | PUB risk (persistent vs eradicated H. pylori) | 3.5 | 2.1-5.9 | < 0.001 | < 0.001 |
| CV events (persistent vs eradicated) | 3.2 | 2.0-5.0 | < 0.001 | < 0.001 | |
| Adherence levels | PUB risk (low vs high adherence) | 2.6 | 1.5-4.5 | 0.003 | 0.004 |
| Treatment failure (low vs high adherence) | 3.1 | 1.8-5.4 | < 0.001 | < 0.001 | |
| Biochemical markers | PUB risk (hypoalbuminemia vs normal albumin) | 2.5 | 1.4-4.2 | 0.005 | 0.006 |
| CV events (high urea vs normal) | 1.9 | 1.2-3.1 | 0.02 | 0.022 |
To address the potential limitations of combining heterogeneous CV conditions in the same groups, and to ensure robust estimates from adequately powered subgroups, we performed a focused subgroup analysis on the three most prevalent and clinically relevant conditions: IHD, PCI, and CABG. Analyses for less common CV subtypes (e.g., CSA, post-PCI) were underpowered due to small sample sizes and are therefore not presented in this focused analysis. The results demonstrate a significant association between H. pylori infection and increased risks of both PUB and CV events in these subgroups. For example, patients with IHD had a higher risk of PUB (HR = 2.8, 95%CI: 1.6-5.1, P = 0.004) and CV events (HR = 2.5, 95%CI: 1.3-4.8, P = 0.006). Similarly, patients undergoing PCI or CABG also showed elevated risks of PUB and CV events, underscoring the systemic impact of H. pylori infection on both gastrointestinal and CV health (Table 13).
| CV disease | Outcome | HR | 95%CI | Nominal P value | Adjusted q value |
| IHD | Risk of PUB | 2.8 | 1.6-5.1 | 0.004 | 0.012 |
| CV events | 2.5 | 1.3-4.8 | 0.006 | 0.012 | |
| PCI | Risk of PUB | 1.8 | 1.2-2.7 | 0.045 | 0.068 |
| CV events | 1.5 | 1.1-2.1 | 0.042 | 0.068 | |
| CABG | Risk of PUB | 2 | 1.4-2.9 | 0.002 | 0.012 |
| CV events | 1.8 | 1.2-2.5 | 0.008 | 0.024 |
This focused analysis confirms that H. pylori infection is a significant modifier of risk in patients with major CVDs. The consistent elevation in HRs across these core subgroups highlights the importance of targeted screening and eradication of H. pylori in high-risk populations, particularly those with IHD, PCI, or CABG. Findings for less common conditions (e.g., CSA, post-PCI) were not statistically reliable due to small sample sizes and should be interpreted with caution. Future studies with larger sample sizes are needed to further explore associations in those specific subgroups.
To quantitatively assess the potential impact of unmeasured lifestyle confounders (e.g., smoking, alcohol use, dietary patterns) on our primary results, we performed an E-value sensitivity analysis. The E-value estimates the minimum strength of association an unmeasured confounder would need to have with both the exposure (H. pylori infection) and the outcome to fully explain away the observed HR, conditional on the measured covariates.
For PUB, the aHR for H. pylori infection was 4.2. The corresponding E-value was 5.0 (95%CI lower bound: 3.1). This indicates that an unmeasured confounder would need to be associated with both H. pylori infection and PUB by HRs of at least 5.0 each to nullify the observed association. For CVD progression (aHR = 3.5), the E-value was 4.2 (95%CI lower bound: 2.6). These substantial E-values suggest that the observed associations are robust to plausible unmeasured confounding (Table 14).
| Outcome | Adjusted HR for H. pylori | 95%CI | E-value (point estimate) | E-value for CI lower bound | Interpretation |
| PUB | 4.2 | 2.1-8.3 | 5.0 | 3.1 | To fully explain the observed HR of 4.2, an unmeasured confounder would need to be associated with both H. pylori and PUB by hazard ratios of ≥ 5.0 each, beyond the measured covariates |
| CV disease progression | 3.5 | 1.8-6.7 | 4.2 | 2.6 | To fully explain the observed HR of 3.5, an unmeasured confounder would need to be associated with both H. pylori and CV progression by hazard ratios of ≥ 4.2 each |
H. pylori infection is a significant global health concern with profound implications for gastrointestinal and systemic health[27]. In long-term LDA users, H. pylori infection presents a unique clinical challenge, as it exacerbates the risk of PUD and gastrointestinal bleeding[19] while also potentially influencing CV outcomes[28]. Aspirin, widely used for CV prevention, inhibits cyclooxygenase-1 (COX-1), reducing prostaglandin synthesis and compromising gastric mucosal defense mechanisms[29]. Combined with H. pylori infection, which independently induces chronic inflammation, mucosal injury, and systemic metabolic disturbances, the risk of PUB is significantly amplified[30]. Furthermore, emerging evidence suggests that H. pylori may contribute to CVD through chronic inflammation, endothelial dysfunction, and pro-atherogenic effects[31]. This study aimed to evaluate the prevalence of H. pylori infection in long-term LDA users, assess the impact of eradication on PUB risk, and explore its association with CV outcomes. By addressing these interconnected aspects, our findings provide critical insights into optimizing gastroprotective and CV management strategies in this high-risk population.
Our study revealed a high prevalence of H. pylori infection (65.5%) among long-term LDA users, significantly higher than the general population. This finding underscores the importance of screening for H. Pylori in this high-risk group, particularly in regions with high endemicity. Scientifically, long-term LDA use may create a favorable environment for H. pylori colonization by reducing gastric mucosal resistance and altering the gastric microenvironment[32]. Successful eradication was achieved in 51.9% of patients, leading to a drastic reduction in PUB incidence (3.7% in the eradicated group vs 11.9% in the persistent infection group, P < 0.001). This demonstrates a strong association between successful eradication and a lower incidence of aspirin-related gastrointestinal complications. Although PUB occurred predominantly in patients with persistent infection, the small residual risk in the eradicated group underscores the multifaceted etiology of aspirin-induced mucosal injury, where H. pylori is a major modifiable cofactor. The persistence of H. pylori infection exacerbates aspirin-induced mucosal injury by increasing gastric acid secretion and impairing mucosal repair mechanisms[33]. Furthermore, H. pylori-positive patients had a significantly higher prevalence of IHD (54.3% vs 33.3%, P < 0.001) and higher rates of CABG procedures (17.4% vs 12.5%, P = 0.045). Successful eradication was associated with a significantly lower burden of CVD progression (29.4% in the eradicated group vs 73.0% in the persistent infection group, P < 0.001). H. pylori infection is proposed to induce systemic inflammation, which may contribute to endothelial dysfunc
The eradication success rate in our study was 51.9% (136/262), which is moderate and reflects the real-world challen
Our findings indicated that compared with H. pylori-negative individuals, H. pylori-positive patients had significantly lower albumin levels and higher levels of urea, Na, K, and cholesterol (Table 1). These changes may indicate that H. pylori infection could lead to systemic metabolic changes, possibly due to chronic low-grade inflammation and oxidative stress. Besides, digestive symptoms such as abdominal pain, bloating, and nausea were more frequent in H. pylori-positive patients. In this regard, such findings are compatible with the accepted role of H. pylori in inducing damage to the gastric mucosa and impairing its mobility[33]. Interestingly, despite such biochemical and symptomatic differences, no significant discrepancies in age and sex distribution could be found among the groups. This again shows that H. pylori performs its pathogenic role mainly in physiological and metabolic dysregulation rather than via demographic predisposition[21]. Importantly, glycemic parameters (HbA1c: 6.8% vs 6.9%, P = 0.587) showed no significant difference, suggesting that H. pylori's metabolic effects are selective rather than global. This pattern aligns with emerging evidence that H. pylori infection may specifically dysregulate lipid metabolism and protein homeostasis through chronic inflammation-mediated pathways, while having less direct impact on glucose regulation in non-diabetic populations. The dissociation between H. pylori-associated dyslipidemia and glycemic control in our cohort supports the hypothesis that different infectious and inflammatory mechanisms underlie these distinct metabolic disturbances.
The association between H. pylori infection and hypoalbuminemia is well-documented, with evidence indicating that chronic inflammation induced by the bacterium leads to decreased albumin synthesis and increased catabolism, con
Notably, while our cohort reflected the high prevalence of diabetes mellitus (28.8%) expected in a population with established CVD on secondary prevention, diabetes status was evenly distributed between H. pylori-positive and negative groups (27.5% vs 31.2%, P = 0.452). This balanced distribution, coupled with multivariate adjustment, strengthens the specificity of our findings regarding H. pylori's independent effects. The lack of significant association between diabetes and PUB risk (HR = 1.3, P = 0.280) in our models, contrasted with its expected association with CV events (HR = 1.6, P = 0.045), suggests distinct pathogenic mechanisms: H. pylori exerts its primary ulcerogenic effect through local gastric mucosal disruption and inflammation, whereas diabetes contributes to CV risk through systemic metabolic and vascular pathways. This distinction is clinically important as it supports targeted H. pylori screening and eradication as a specific intervention to reduce aspirin-associated PUB, separate from general CV risk management strategies.
Our findings indicate that digestive symptoms, including abdominal pain, bloating, and nausea, were significantly more prevalent among H. pylori-positive patients compared to H. pylori-negative individuals, which aligns with the bacterium’s well-established role in disrupting gastric mucosal integrity and impairing gastric motility[43]. Chronic H. pylori infection triggers an inflammatory response that compromises the protective mucus barrier, exposing the gastric epithelium to acid-related injury, while also contributing to delayed gastric emptying and altered gut hormone regula
Our study's endoscopic and histopathological findings revealed striking differences between H. pylori-positive and H. pylori-negative participants (Table 2). Among H. pylori-positive individuals, nearly half (46.6%) exhibited normal endo
Such findings are consistent with previous studies that have established a strong link between H. pylori infection and gastric mucosal damage. The high prevalence of peptic ulcers and gastric erosions in H. pylori-positive patients aligns with evidence that H. pylori is a major cause of these conditions, as demonstrated in large-scale consensus reports[47]. The significant proportion of H. pylori-positive patients with moderate to severe inflammation further supports the role of H. pylori in driving chronic gastritis, which can progress to atrophy and intestinal metaplasia, as highlighted in prior research[50]. Notably, the absence of atrophy and intestinal metaplasia in H. pylori-negative participants reinforces the pathogen’s role in promoting precancerous changes, a finding consistent with the Kyoto global consensus on H. pylori gastritis[46]. Mechanistically, H. pylori disrupts gastric epithelial cells by inducing inflammation, altering cell signaling, and impairing mucosal integrity. The bacterium translocates its CagA protein into epithelial cells, activating nuclear factor kappa B (NF-κB) and promoting the secretion of proinflammatory cytokines such as IL-8, which recruits immune cells and sustains chronic inflammation. Additionally, H. pylori urease stimulates the release of IL-6 and TNF-α, further exacerbating tissue damage. These inflammatory responses weaken the epithelial barrier, increasing susceptibility to gastric ulcers and carcinogenesis[51]. The histopathological findings are consistent with the Updated Sydney System, as described by Dixon et al[25], which classifies H. pylori-associated gastritis based on inflammation severity, atrophy, and metaplasia. Chronic H. pylori–induced inflammation is clinically significant, as persistent infection is associated with progressive mucosal damage and an increased risk of neoplastic transformation, underscoring the importance of app
Our findings highlighted significant differences in the prevalence of CVDs between H. pylori-positive and H. pylori-negative participants before treatment (Table 3). Among H. pylori-positive individuals, IHD at baseline was notably more common (54.3%) compared to H. pylori-negative participants (33.3%), suggesting a potential association between H. pylori infection and CV risk. Additionally, H. pylori-positive patients had a higher prevalence of CABG (17.4% vs 12.5%) and a lower prevalence of PCI (8.7% vs 12.5%), indicating possible differences in disease severity or management between the two groups. Interestingly, CSA and post-PCI cases were exclusively observed in H. pylori-positive patients, though these differences did not reach statistical significance. These findings suggest that H. pylori infection may be linked to a higher burden of CVD, particularly IHD, and could influence the progression or management of these conditions.
The association between H. pylori infection and a higher prevalence of IHD aligns with previous studies suggesting that chronic illnesses, including H. pylori, may contribute to systemic inflammation, oxidative stress, and endothelial dysfunction, thereby increasing CV risk and the formation of atherosclerotic plaques. The bacterium's ability to increase levels of C-reactive protein and other inflammatory markers further accelerates atherosclerosis[54]. The higher prevalence of CABG in H. pylori-positive patients is consistent with findings that H. pylori infection may accelerate atherosclerosis, leading to more severe coronary artery disease requiring surgical intervention[55,56]. However, the lower prevalence of PCI in H. pylori-positive patients contrasts with some studies, which have reported no significant difference in PCI rates between H. pylori-positive and -negative individuals[57]. This discrepancy may reflect differences in study populations or the influence of confounding factors such as comorbidities or treatment practices. The exclusive presence of CSA and post-PCI cases in H. pylori-positive patients, though not statistically significant, warrants further investigation, as it may indicate a unique interaction between H. pylori and CV pathophysiology. H. pylori infection may contribute to CVDs through multiple mechanisms, including chronic inflammation, molecular mimicry, lipid metabolism alterations, and endothelial dysfunction. The persistent inflammatory response induced by H. pylori leads to elevated cytokine levels and acute-phase proteins, which are implicated in atherosclerosis development. Additionally, the bacterium’s antigens may mimic host tissues, triggering autoimmune responses that damage vascular structures. H. pylori infection is also asso
Our analysis revealed a significant association between PUB and specific CVDs, particularly IHD and CABG. As shown in Table 4, patients with PUB had a higher prevalence of IHD (54.3% vs 33.3%, P < 0.001) and were more likely to undergo CABG (17.4% vs 12.5%, P = 0.045). These findings suggest that PUB may not only be a gastrointestinal com
Several mechanisms may explain this association. First, H. pylori infection induces chronic systemic inflammation, which is a known risk factor for atherosclerosis and endothelial dysfunction[31,56]. H. pylori infection may contribute to plaque formation and vascular damage through the proposed release of pro-inflammatory cytokines such as IL-6 and TNF-α, as suggested by previous research[31,51]. Second, the use of LDA, while beneficial for CV prevention, exacerbates gastric mucosal injury in the presence of H. pylori, leading to PUB[19,30]. This dual burden of infection and medication may amplify both gastrointestinal and CV risks[56]. Furthermore, the higher prevalence of CABG in H. pylori-positive patients suggests that persistent infection may accelerate atherosclerosis, necessitating surgical intervention[55,56]. The lower prevalence of PCI in this group may reflect differences in disease severity or management strategies[57]. These findings underscore the association between H. pylori infection, PUB, and higher CV risk in long-term aspirin users.
The higher baseline prevalence of IHD in H. pylori-positive patients compared to H. pylori-negative patients (54.3% vs 33.3%, P < 0.001) suggests a potential interaction between H. pylori infection and CV risk. Furthermore, persistent H. pylori infection was a strong independent predictor of CVD progression during follow-up (adjusted HR = 3.5, P < 0.001), suggesting that H. pylori may contribute to worsening CV status through pathways independent of conventional meta
Our study outcomes revealed significant differences between H. pylori-positive patients with eradicated vs persistent infection and H. pylori-negative individuals (Tables 6 and 7). In the eradicated group, the incidence of PUB was markedly lower (1.5% at 6 weeks and 3.7% at 6 months) compared to the persistent infection group (8.7% at 6 weeks and 11.9% at 6 months), highlighting the association between successful eradication and reduced PUB risk. Similarly, the burden of incident CVD progression, defined as new revascularization, hospitalization for angina, or worsening angina class occurring during follow-up, was significantly lower in the eradicated group. The time-to-event analysis (Table 7) showed that incident CV disease progression occurred in 3.7% of the eradicated group vs 8.0% of the persistent group at 6 weeks, and 5.9% vs 14.3% at 6 months, representing an approximately 2.4-fold higher risk in the persistent group. The time-to-event analysis for CVD progression further supports these findings, showing a clear temporal relationship between persistent infection and worsening CV status, while the eradicated group resembled the H. pylori-negative baseline. Adverse events, such as fatigue and dizziness, were also more common in the persistent group, suggesting that ongoing H. pylori infection exacerbates both gastrointestinal and systemic complications. These results underscore the importance of successful eradication in reducing the burden of both gastric and CV pathology.
Beyond gastrointestinal outcomes, persistent H. pylori infection was associated with a significantly higher risk of incident CVD progression (as defined by new revascularization, hospitalization for angina, or worsening angina class occurring during follow-up) over the six-month follow-up period (aHR = 3.5). However, it is important to note that these CV findings are based on a relatively short six-month observation period and reflect symptomatic progression and healthcare utilization patterns rather than long-term atherosclerotic endpoints.
Our findings are consistent with previous research highlighting the benefits of H. pylori eradication in reducing both gastrointestinal and CV risks. For example, our results align with the randomized, double-blind, placebo-controlled trial (Helicobacter Eradication Aspirin Trial) trial by Hawkey et al[21], which demonstrated that H. pylori eradication signifi
The six-month follow-up period provided valuable insights into the short-term effects of H. pylori eradication on PUB and CV outcomes. However, atherosclerosis-related CV events develop over years, and a longer follow-up may be necessary to fully capture the long-term impact of H. pylori eradication on CV risk. Future studies with extended follow-up periods will help determine whether successful eradication leads to sustained reductions in major CV events (e.g., MI, stroke, CV mortality).
The Cox proportional hazards regression (Table 8) and multivariate logistic regression (Table 9) analyses identify key predictors of PUB and CVD progression in patients with H. pylori infection. For PUB, H. pylori infection emerged as the strongest predictor, with an aHR of 4.2 and an aOR of 4.2, highlighting its central role in ulcer-related complications. Low albumin levels were also a significant predictor, with aHR and aOR values of 2.8, suggesting that nutritional status and systemic inflammation may exacerbate ulcer risk. However, this association should be interpreted cautiously, as hypoalbuminemia likely serves as a marker of overall illness severity and chronic inflammation rather than a direct causal mediator of ulcer bleeding. Reverse causation is also plausible, where patients with more severe underlying conditions have both lower albumin levels and greater susceptibility to aspirin-induced mucosal injury.
For CVD progression, H. pylori infection remained a significant predictor (aHR = 3.5; aOR = 3.2), alongside multivessel coronary disease at baseline (aHR = 2.8; aOR = 2.8) (Tables 8 and 9), emphasizing the interplay between infection and underlying coronary severity. Poor adherence to treatment was another critical factor, significantly increasing the odds of both PUB (aOR = 3.1) and CVD progression (aOR = 2.1) (Table 9), underscoring the importance of patient compliance in managing these conditions. These findings collectively highlight the multifactorial nature of PUB and CV disease progression, with H. pylori infection serving as a common and modifiable risk factor. A sensitivity analysis for unmea
The findings in Tables 6 and 7 build on existing evidence but also offer new perspectives on the predictors of PUB and CVD progression, particularly in long-term LDA users. This suggests that the chronic systemic inflammatory burden induced by H. pylori may accelerate underlying atherosclerosis, promote endothelial dysfunction, and potentially destabilize coronary plaques in high-risk patients on long-term aspirin therapy. This is further supported by our regression analyses, which identified H. pylori infection and multivessel coronary disease as strong predictors of CV progression. This finding aligns with emerging evidence linking chronic infections to accelerated atherogenesis and adverse CV trajectories.
While the strong association between H. pylori infection and PUB is well-documented[62,63], our study highlights the additional role of low albumin levels, which may reflect underlying malnutrition or chronic inflammation, further compounding ulcer risk. It is important to note that hypoalbuminemia in this context is more likely a biomarker of the systemic inflammatory burden and nutritional compromise associated with persistent H. pylori infection, rather than an independent causal factor in ulcer pathogenesis. Studies suggested that H. pylori infection may adversely affect nutri
The diagnostic accuracy of H. pylori detection methods plays a critical role in guiding treatment decisions and pre
Our study findings shed light on the adverse events associated with H. pylori treatment and their impact on adherence, a critical factor in achieving successful eradication. Fatigue (11.1%), dizziness (7.9%), and nausea (5.6%) were the most frequently reported side effects, with fatigue showing the strongest association with poor adherence (OR = 2.5) (Table 11). These findings corroborate previous studies that have documented increased gastrointestinal and systemic side effects with H. pylori eradication therapy, particularly with levofloxacin-based regimens[72]. Scientifically, levofloxacin’s ability to cross the blood-brain barrier is thought to contribute to central nervous system side effects like dizziness and fatigue, which are further exacerbated by its impact on mitochondrial function and neurotransmitter activity[73,74]. These adverse events, while generally mild, can significantly influence patient compliance, particularly in older adults or those with comorbidities. The high odds ratios for treatment failure in patients with low adherence further emphasize the importance of managing side effects to improve outcomes. Given the strong association between adverse events and adherence, strategies such as patient counseling, medication reminders, and symptom management protocols should be incorporated into eradication programs to optimize adherence and treatment success[72]. Addressing these challenges through tailored interventions could enhance adherence and reduce the risk of complications like PUB and CV events.
The subgroup analysis provided valuable insights into the risk factors for PUB and CV events in LDA users, highlighting the interplay between H. pylori infection, patient characteristics, and treatment outcomes (Table 12). Older age (≥ 65 years) and male sex were associated with significantly higher risks of both PUB and CV events. Comorbidities like IHD further exacerbated these risks, with HRs of 2.8 for PUB and 2.4 for treatment failure. Older patients (≥ 65 years) had a 2.0-fold increased risk of PUB and a 1.8-fold increased risk of CV events. Men had a 1.7-fold increased risk of PUB and 1.5-fold increased CV risk. Baseline IHD had a 2.8-fold higher risk of PUB and a 2.4-fold greater treatment failure rate. Increased gastric damage in older adults is primarily due to weakened mucosal defense mechanisms, chronic inflammation, reduced gastric blood flow, and higher susceptibility to oxidative stress. Additionally, H. pylori infection and prolonged NSAID use further exacerbate mucosal injury, increasing the risk of ulcers and complications[75]. The male sex is associated with a higher prevalence of atherosclerosis and CVD, particularly at younger ages. Men tend to develop CVD earlier than women due to differences in sex hormones, lipid metabolism, and vascular inflammation. However, after menopause, women experience a rapid increase in CV risk, often surpassing men in later life. These differences highlight the importance of sex-specific risk assessment and treatment strategies[76,77]. Persistent H. pylori infection was the strongest predictor of adverse outcomes, with HRs of 3.5 for PUB and 3.2 for CV events, reinforcing the importance of successful eradication. Low adherence to treatment also emerged as a critical risk factor, with HRs of 2.6 for PUB and 3.1 for treatment failure, highlighting the need for strategies to improve patient compliance. These findings collectively emphasize the importance of personalized risk stratification and tailored interventions to optimize outcomes in LDA users with H. pylori infection[72]. This comprehensive analysis strongly supports routine H. pylori screening and eradication in long-term LDA users to reduce the risk of both gastric and CV complications. Our study reinforces prior evidence that persistent H. pylori infection amplifies the risks of PUB and IHD through chronic inflammation, metabolic alterations, and mucosal injury.
The subgroup analysis of CV conditions in Table 13 reveals a clear association between H. pylori infection and adverse outcomes in patients with IHD, PCI, and CABG. Notably, patients with IHD had a 2.8-fold increased risk of PUB and a 2.5-fold increased risk of CV events. Similarly, those undergoing PCI or CABG also showed elevated risks, with HRs of 1.8 and 2.0 for PUB, respectively. These findings underscore the systemic impact of H. pylori infection, which extends beyond gastrointestinal complications to influence CV health. Importantly, this analysis was performed to address the concern regarding the potential limitations of combining heterogeneous CV conditions in the same groups, ensuring a more precise evaluation of the association between H. pylori infection and specific CV outcomes.
The elevated risks of PUB and CV events in patients with IHD, PCI, or CABG may be explained by the potential interplay between H. pylori-associated systemic inflammation and the pro-thrombotic effects of LDA. H. pylori infection triggers a chronic inflammatory response characterized by the release of pro-inflammatory cytokines such as IL-6 and TNF-α, which contribute to endothelial dysfunction and atherosclerosis. This inflammatory milieu not only exacerbates gastric mucosal injury but also accelerates the progression of CVD, particularly in patients with preexisting conditions like IHD[31,56]. In patients undergoing PCI or CABG, the persistent inflammatory state induced by H. pylori may further compromise vascular repair mechanisms, increasing the risk of adverse CV events. Additionally, the use of LDA, while beneficial for CV prevention, may amplify PUB risk in these patients by impairing gastric mucosal defenses[19,30]. The dual burden of H. pylori infection and aspirin use creates a vicious cycle, where systemic inflammation and mucosal injury reinforce each other, leading to worse outcomes. The strong association between persistent H. pylori infection and adverse outcomes (HR = 3.5 for PUB and HR = 3.2 for CV events) highlights the importance of successful eradication. By eliminating H. pylori, clinicians may not only reduce the risk of PUB but also mitigate systemic inflammation, potentially improving CV outcomes[21,61]. However, the small number of cases for less common conditions (e.g., CSA, post-PCI) limited our ability to detect significant differences, underscoring the need for larger studies with longer follow-up periods to validate these findings.
Regarding the impact of concomitant antithrombotic/anticoagulant therapy, our study found that all participants were on aspirin plus at least one other antithrombotic or anticoagulant agent, reflecting contemporary secondary prevention practice. While this combination increases overall bleeding risk, our multivariate analyses explicitly adjusted for antithrombotic regimen intensity (dual vs triple therapy). As shown in Table 5, the number of additional agents was an independent predictor of PUB (aOR = 1.8 for triple therapy). This statistical adjustment strengthens the conclusion that the observed associations between H. pylori status and both gastrointestinal and CV outcomes are independent of the background antithrombotic burden. The study title appropriately focuses on 'chronic aspirin users' as aspirin represents the constant, ulcerogenic agent common to all participants and is the primary therapeutic intervention under investigation.
This study has several notable strengths. First, the prospective observational design with systematic follow-up allowed for the assessment of temporal relationships between H. pylori status and clinical outcomes. Second, the use of a multi-modal diagnostic approach (histopathology, UBT, stool antigen) ensured accurate identification of H. pylori infection. A key strength is the inclusion of a substantial proportion of patients with diabetes mellitus (28.8% of the cohort), which reflects the real-world clinical population where CVD, diabetes, and aspirin use frequently coexist. The balanced distribution of diabetes between H. pylori-positive and negative groups at baseline, and its subsequent adjustment in multivariate analyses, strengthens the internal validity of our findings regarding H. pylori's independent effects. This inclusion enhances the external validity and clinical relevance of our conclusions for managing complex patients with multimorbidity. Finally, the comprehensive evaluation of both gastrointestinal and CV endpoints provides a holistic view of the systemic impact of H. pylori infection in high-risk aspirin users.
While our findings suggest an association between H. pylori eradication and less documented CVD progression during short-term follow-up, the six-month observation period is insufficient to evaluate the impact on hard atherosclerotic endpoints such as MI or stroke. The observed differences may reflect changes in angina symptoms, medication adjust
This study has several limitations. First, its single-center design may limit the generalizability of findings to broader populations. Second, the six-month follow-up period may not fully capture long-term CV risks or ulcer recurrence post-eradication. Third, adherence monitoring relied on self-reporting, pill counts, and pharmacy records, which may introduce reporting bias. Fourth, the specific reasons for eradication failure (e.g., primary antibiotic resistance vs poor metabolizer status) could not be definitively ascertained, as pretreatment culture and susceptibility testing were not routinely performed. Fifth, while we adjusted for major clinical and biochemical confounders, unmeasured lifestyle factors (e.g., detailed dietary patterns, smoking, alcohol consumption) could introduce residual confounding. We addressed this possibility quantitatively via an E-value sensitivity analysis, which suggests the observed association between H. pylori and PUB is robust to such confounding; however, its potential influence cannot be entirely ruled out. Sixth, all participants were on daily LDA in combination with one or more other antithrombotic or anticoagulant medications, which may limit the generalizability of our findings to patients on aspirin monotherapy; however, this design reflects real-world clinical practice where combination therapy is common in secondary prevention. Seventh, while our study suggests a link between persistent H. pylori infection and CVD, the observational design precludes causal inference. Finally, residual confounding and potential selection bias cannot be ruled out. Future randomized controlled trials with longer follow-up are needed to confirm these findings.
Future studies should address these limitations by incorporating longer follow-up periods to investigate the long-term effects of H. pylori eradication on CV and gastrointestinal outcomes. Multicentric studies with larger, more diverse populations would enhance the external validity of findings. Comparative trials of various eradication regimens could identify optimal therapeutic approaches for aspirin users. The incorporation of newer CV biomarkers and genetic predisposing factors may further elucidate the role of H. pylori infection in atherosclerosis. Additionally, strategies to enhance treatment adherence, such as patient education programs and alternative medication delivery systems, should be explored to maximize eradication success. Finally, future research should investigate the impact of H. pylori eradication in patients on different combinations of antithrombotic and anticoagulant medications (e.g., aspirin + clopidogrel or anticoagulants) to better understand the interplay between infection, medication use, and bleeding risk. The role of PPI co-therapy in reducing gastrointestinal bleeding in high-risk patients should also be explored.
Persistent H. pylori infection was strongly associated with significantly higher risks of PUB and CVD progression in long-term LDA users, with effects that are independent of diabetes mellitus and other traditional CV risk factors. Successful eradication was associated with fewer of these complications, supporting the need for routine screening and treatment in high-risk populations, including those with complex multimorbidity such as concurrent CVD and diabetes. This study highlights the systemic impact of H. pylori beyond the gastrointestinal tract, emphasizing its potential relevance in CV health. Future research should focus on optimizing eradication strategies, improving adherence, and assessing long-term outcomes to enhance the clinical management of aspirin users with H. pylori infection.
| 1. | Ali A, AlHussaini KI. Helicobacter pylori: A Contemporary Perspective on Pathogenesis, Diagnosis and Treatment Strategies. Microorganisms. 2024;12:222. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 89] [Article Influence: 44.5] [Reference Citation Analysis (3)] |
| 2. | Bagheri N, Razavi A, Pourgheysari B, Azadegan-Dehkordi F, Rahimian G, Pirayesh A, Shafigh M, Rafieian-Kopaei M, Fereidani R, Tahmasbi K, Shirzad H. Up-regulated Th17 cell function is associated with increased peptic ulcer disease in Helicobacter pylori-infection. Infect Genet Evol. 2018;60:117-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 3. | Li R, Wang W, Ma Y, Chen H. Analysis of risk factors for ulcer recurrence and upper gastrointestinal bleeding in children with peptic ulcer treated with Helicobacter pylori eradication therapy. Transl Pediatr. 2023;12:618-630. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (33)] |
| 4. | Kountouras J, Papaefthymiou A, Polyzos SA, Deretzi G, Vardaka E, Soteriades ES, Tzitiridou-Chatzopoulou M, Gkolfakis P, Karafyllidou K, Doulberis M. Impact of Helicobacter pylori-Related Metabolic Syndrome Parameters on Arterial Hypertension. Microorganisms. 2021;9:2351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 5. | Li B, Zhang Y, Zheng Y, Cai H. The causal effect of Helicobacter pylori infection on coronary heart disease is mediated by the body mass index: a Mendelian randomization study. Sci Rep. 2024;14:1688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 6. | Afifi S, Elantouny N, Elsokkary R, Abdelbaser E. Helicobacter Pylori Treatment Eradication in Egypt: Standard Clarithromycin-based Triple versus Quadruple Regimen Therapy. Afro-Egypt J Infect Enem Dis. 2020;10:100-107. [DOI] [Full Text] |
| 7. | Barakat SH, Hanafy HM, Guimei M, Elsawy EH, Khalil AFM. Different regimens for eradication of Helicobacter pylori infection in children: a randomized controlled trial. Eur J Pediatr. 2024;184:13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Cofer LB, Barrett TJ, Berger JS. Aspirin for the Primary Prevention of Cardiovascular Disease: Time for a Platelet-Guided Approach. Arterioscler Thromb Vasc Biol. 2022;42:1207-1216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 9. | Kim SP, Ryu J, Kim SH, Yoon HJ. Low-dose aspirin in the primary prevention of cardiovascular diseases: A retrospective, propensity score matched study. Atherosclerosis. 2023;371:54-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Patrono C. Low-dose aspirin for the prevention of atherosclerotic cardiovascular disease. Eur Heart J. 2024;45:2362-2376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 63] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 11. | Biccler J, Bollaerts K, Vora P, Sole E, Rodriguez LAG, Lanas A, Langley RE, Gabarró MS. Public health impact of low-dose aspirin on colorectal cancer, cardiovascular disease and safety in the UK - Results from micro-simulation model. Int J Cardiol Heart Vasc. 2021;36:100851. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Nafisi S, Støer NC, Veierød MB, Randel KR, Hoff G, Löfling L, Bosetti C, Botteri E. Low-Dose Aspirin and Prevention of Colorectal Cancer: Evidence From a Nationwide Registry-Based Cohort in Norway. Am J Gastroenterol. 2024;119:1402-1411. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 13. | Nguyen TNM, Chen LJ, Trares K, Stocker H, Holleczek B, Beyreuther K, Brenner H, Schöttker B. Long-term low-dose acetylsalicylic use shows protective potential for the development of both vascular dementia and Alzheimer's disease in patients with coronary heart disease but not in other individuals from the general population: results from two large cohort studies. Alzheimers Res Ther. 2022;14:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 22] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 14. | Thong EH, Lee ECY, Yun C, Li TYW, Sia C. Aspirin Therapy, Cognitive Impairment, and Dementia-A Review. Future Pharmacol. 2023;3:144-161. [DOI] [Full Text] |
| 15. | Skriver C, Maltesen T, Dehlendorff C, Skovlund CW, Schmidt M, Sørensen HT, Friis S. Long-term aspirin use and cancer risk: a 20-year cohort study. J Natl Cancer Inst. 2024;116:530-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 35] [Article Influence: 17.5] [Reference Citation Analysis (1)] |
| 16. | Slomski A. Daily Aspirin Increases Gastrointestinal Bleeding Among Older Adults. JAMA. 2020;324:1599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Mahady SE, Margolis KL, Chan A, Polekhina G, Woods RL, Wolfe R, Nelson MR, Lockery JE, Wood EM, Reid C, Ernst ME, Murray A, Thao L, McNeil JJ. Major GI bleeding in older persons using aspirin: incidence and risk factors in the ASPREE randomised controlled trial. Gut. 2021;70:717-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 96] [Article Influence: 19.2] [Reference Citation Analysis (4)] |
| 18. | Bittl JA, Laine L. Gastrointestinal Injury Caused by Aspirin or Clopidogrel Monotherapy Versus Dual Antiplatelet Therapy. J Am Coll Cardiol. 2022;79:129-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 19. | Sarri GL, Grigg SE, Yeomans ND. Helicobacter pylori and low-dose aspirin ulcer risk: A meta-analysis. J Gastroenterol Hepatol. 2019;34:517-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 20. | Ng EK, Lam YH, Sung JJ, Yung MY, To KF, Chan AC, Lee DW, Law BK, Lau JY, Ling TK, Lau WY, Chung SC. Eradication of Helicobacter pylori prevents recurrence of ulcer after simple closure of duodenal ulcer perforation: randomized controlled trial. Ann Surg. 2000;231:153-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 137] [Cited by in RCA: 103] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Hawkey C, Avery A, Coupland CAC, Crooks C, Dumbleton J, Hobbs FDR, Kendrick D, Moore M, Morris C, Rubin G, Smith M, Stevenson D; HEAT Trialists. Helicobacter pylori eradication for primary prevention of peptic ulcer bleeding in older patients prescribed aspirin in primary care (HEAT): a randomised, double-blind, placebo-controlled trial. Lancet. 2022;400:1597-1606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 44] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 22. | Zhou Y, Qiao Y, Zhao L, Zhang X, Yang S, Bai L, Liu S. Association of long-term use of low-dose aspirin with Helicobacter pylori infection and effect on recurrence rate. Sci Rep. 2024;14:22084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 23. | Chen YC, Malfertheiner P, Yu HT, Kuo CL, Chang YY, Meng FT, Wu YX, Hsiao JL, Chen MJ, Lin KP, Wu CY, Lin JT, O'Morain C, Megraud F, Lee WC, El-Omar EM, Wu MS, Liou JM. Global Prevalence of Helicobacter pylori Infection and Incidence of Gastric Cancer Between 1980 and 2022. Gastroenterology. 2024;166:605-619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 325] [Article Influence: 162.5] [Reference Citation Analysis (0)] |
| 24. | Said ZNA, El-Nasser AM. Evaluation of urea breath test as a diagnostic tool for Helicobacter pylori infection in adult dyspeptic patients. World J Gastroenterol. 2024;30:2302-2307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (3)] |
| 25. | Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161-1181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3861] [Cited by in RCA: 3609] [Article Influence: 120.3] [Reference Citation Analysis (6)] |
| 26. | Zhang X, Li X, Li J, Deng Y, Xu W, Chen D, Wei L. Comparison of tegoprazan-based and proton pump inhibitor-based regimens for Helicobacter pylori eradication: a meta-analysis and systematic review. Front Med (Lausanne). 2025;12:1580203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 27. | Malfertheiner P, Camargo MC, El-Omar E, Liou JM, Peek R, Schulz C, Smith SI, Suerbaum S. Helicobacter pylori infection. Nat Rev Dis Primers. 2023;9:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 773] [Cited by in RCA: 642] [Article Influence: 214.0] [Reference Citation Analysis (18)] |
| 28. | Abi Khalil C, Omar OM, Al Suwaidi J, Taheri S. Aspirin Use and Cardiovascular Outcome in Patients With Type 2 Diabetes Mellitus and Heart Failure: A Population-Based Cohort Study. J Am Heart Assoc. 2018;7:e010033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Warner TD, Nylander S, Whatling C. Anti-platelet therapy: cyclo-oxygenase inhibition and the use of aspirin with particular regard to dual anti-platelet therapy. Br J Clin Pharmacol. 2011;72:619-633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 179] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 30. | Sostres C, Gargallo CJ, Lanas A. Interaction between Helicobacter pylori infection, nonsteroidal anti-inflammatory drugs and/or low-dose aspirin use: old question new insights. World J Gastroenterol. 2014;20:9439-9450. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 31. | Aramouni K, Assaf RK, Azar M, Jabbour K, Shaito A, Sahebkar A, Eid AA, Rizzo M, Eid AH. Infection with Helicobacter pylori may predispose to atherosclerosis: role of inflammation and thickening of intima-media of carotid arteries. Front Pharmacol. 2023;14:1285754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (2)] |
| 32. | Ong L, Lin C. Adhesion, infection, and therapeutic treatment of Helicobacter pylori: a review on current aspects and future promise. Discov Appl Sci. 2024;6:323. [DOI] [Full Text] |
| 33. | Yoshida N, Sugimoto N, Hirayama F, Nakamura Y, Ichikawa H, Naito Y, Yoshikawa T. Helicobacter pylori infection potentiates aspirin induced gastric mucosal injury in Mongolian gerbils. Gut. 2002;50:594-598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 34. | Metwally M, Ragab R, Abdel Hamid HS, Emara N, Elkholy H. Helicobacter pylori Antibiotic Resistance in Egypt: A Single-Center Study. Infect Drug Resist. 2022;15:5905-5913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 19] [Reference Citation Analysis (4)] |
| 35. | Elazazi NADA, Eltabbakh M, Hussein HM, Mahmood YM, Elwakil R. Efficacy of Potassium Competitive Acid Blockers (P-CABs) versus Proton Pump Inhibitors (PPIs) in the First and Second Line Eradication Regimens for Helicobacter pylori in Egyptian Patients. 2023 Preprint. Available from: Qeios. [DOI] [Full Text] |
| 36. | Liu H, Qin Y, Yang J, Huang G, Wei X, Wang L, Li W. Helicobacter pylori Infection as a Risk Factor for Abnormal Serum Protein Levels in General Population of China. J Inflamm Res. 2022;15:2009-2017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 37. | Xu Y, Wang Q, Liu Y, Cui R, Zhao Y. Is Helicobacter pylori infection a critical risk factor for vascular dementia? Int J Neurosci. 2016;126:899-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 38. | Aljahdli E, Almaghrabi SJ, Alhejaili TL, Alghamdi W. Association Between Helicobacter pylori Eradication and Kidney Function in Patients With Chronic Gastritis: A Retrospective Single-Center Study. Cureus. 2022;14:e21621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 39. | Hashim M, Mohammed O, G/Egzeabeher T, Wolde M. The association of Helicobacter Pylori infection with dyslipidaemia and other atherogenic factors in dyspeptic patients at St. Paul's Hospital Millennium Medical College. Heliyon. 2022;8:e09430. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 40. | Abdu A, Cheneke W, Adem M, Belete R, Getachew A. Dyslipidemia and Associated Factors Among Patients Suspected to Have Helicobacter pylori Infection at Jimma University Medical Center, Jimma, Ethiopia. Int J Gen Med. 2020;13:311-321. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 41. | Izhari MA, Al Mutawa OA, Mahzari A, Alotaibi EA, Almashary MA, Alshahrani JA, Gosady ARA, Almutairi AM, Dardari DMM, AlGarni AKA. Helicobacter pylori (H. pylori) Infection-Associated Dyslipidemia in the Asir Region of Saudi Arabia. Life (Basel). 2023;13:2206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 42. | Yang C, You N, Chen Y, Zhang J. Helicobacter pylori infection increases the risk of dyslipidemia in Chinese diabetic Population: a retrospective cross-sectional study. BMC Infect Dis. 2024;24:730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 43. | Xu W, Xu L, Xu C. Relationship between Helicobacter pylori infection and gastrointestinal microecology. Front Cell Infect Microbiol. 2022;12:938608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 44. | Dharan M, Wozny D. Helicobacter pylori infection and small intestinal bacterial overgrowth-more than what meets the eye. World J Clin Cases. 2022;10:7209-7214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 45. | Wei YF, Li X, Zhao MR, Liu S, Min L, Zhu ST, Zhang ST, Xie SA. Helicobacter pylori disrupts gastric mucosal homeostasis by stimulating macrophages to secrete CCL3. Cell Commun Signal. 2024;22:263. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 46. | Sugano K, Tack J, Kuipers EJ, Graham DY, El-Omar EM, Miura S, Haruma K, Asaka M, Uemura N, Malfertheiner P; faculty members of Kyoto Global Consensus Conference. Kyoto global consensus report on Helicobacter pylori gastritis. Gut. 2015;64:1353-1367. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1515] [Cited by in RCA: 1293] [Article Influence: 117.5] [Reference Citation Analysis (21)] |
| 47. | Malfertheiner P, Megraud F, O'Morain CA, Gisbert JP, Kuipers EJ, Axon AT, Bazzoli F, Gasbarrini A, Atherton J, Graham DY, Hunt R, Moayyedi P, Rokkas T, Rugge M, Selgrad M, Suerbaum S, Sugano K, El-Omar EM; European Helicobacter and Microbiota Study Group and Consensus panel. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut. 2017;66:6-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2431] [Cited by in RCA: 2095] [Article Influence: 232.8] [Reference Citation Analysis (4)] |
| 48. | Wauters L, Burns G, Ceulemans M, Walker MM, Vanuytsel T, Keely S, Talley NJ. Duodenal inflammation: an emerging target for functional dyspepsia? Expert Opin Ther Targets. 2020;24:511-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 49] [Article Influence: 8.2] [Reference Citation Analysis (2)] |
| 49. | Kabil S, Mohamed Ali A, Ghazy A, El-Shewi M. Gastric Bacterial Flora in Patients with Helicobacter Pylori Infection with and without Dyspepsia. Benha Med J. 2022;39:319-333. [DOI] [Full Text] |
| 50. | Schulz C, Schütte K, Koch N, Vilchez-Vargas R, Wos-Oxley ML, Oxley APA, Vital M, Malfertheiner P, Pieper DH. The active bacterial assemblages of the upper GI tract in individuals with and without Helicobacter infection. Gut. 2018;67:216-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 157] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 51. | Alzahrani S, Lina TT, Gonzalez J, Pinchuk IV, Beswick EJ, Reyes VE. Effect of Helicobacter pylori on gastric epithelial cells. World J Gastroenterol. 2014;20:12767-12780. [PubMed] [DOI] [Full Text] |
| 52. | Shah SC, Iyer PG, Moss SF. AGA Clinical Practice Update on the Management of Refractory Helicobacter pylori Infection: Expert Review. Gastroenterology. 2021;160:1831-1841. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 197] [Cited by in RCA: 174] [Article Influence: 34.8] [Reference Citation Analysis (4)] |
| 53. | Fu H, Ma Y, Yang M, Zhang C, Huang H, Xia Y, Lu L, Jin W, Cui D. Persisting and Increasing Neutrophil Infiltration Associates with Gastric Carcinogenesis and E-cadherin Downregulation. Sci Rep. 2016;6:29762. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 54. | Watanabe J, Kotani K. The effect of Helicobacter pylori eradication on C-reactive protein: results from a meta-analysis. Arch Med Sci. 2022;18:958-964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 55. | Sharma V, Aggarwal A. Helicobacter pylori: Does it add to risk of coronary artery disease. World J Cardiol. 2015;7:19-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 56. | Fang Y, Fan C, Xie H. Effect of Helicobacter pylori infection on the risk of acute coronary syndrome: A systematic review and meta-analysis. Medicine (Baltimore). 2019;98:e18348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 57. | Wang B, Yu M, Zhang R, Chen S, Xi Y, Duan G. A meta-analysis of the association between Helicobacter pylori infection and risk of atherosclerotic cardiovascular disease. Helicobacter. 2020;25:e12761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (3)] |
| 58. | Jamkhande PG, Gattani SG, Farhat SA. Helicobacter pylori and cardiovascular complications: a mechanism based review on role of Helicobacter pylori in cardiovascular diseases. Integr Med Res. 2016;5:244-249. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 59. | Tali LDN, Faujo GFN, Konang JLN, Dzoyem JP, Kouitcheu LBM. Relationship between active Helicobacter pylori infection and risk factors of cardiovascular diseases, a cross-sectional hospital-based study in a Sub-Saharan setting. BMC Infect Dis. 2022;22:731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 60. | Sun L, Zheng H, Qiu M, Hao S, Liu X, Zhu X, Cai X, Huang Y. Helicobacter pylori infection and risk of cardiovascular disease. Helicobacter. 2023;28:e12967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 61. | Yaslianifard S, Sameni F, Kazemi K, Atefpour Y, Hajikhani B, Baradaran Bagheri A, Yazdani S, Dadashi M. Beyond the gut: a comprehensive meta-analysis on Helicobacter pylori infection and cardiovascular complications. Ann Clin Microbiol Antimicrob. 2025;24:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 62. | Narayanan M, Reddy KM, Marsicano E. Peptic Ulcer Disease and Helicobacter pylori infection. Mo Med. 2018;115:219-224. [PubMed] |
| 63. | McConaghy JR, Decker A, Nair S. Peptic Ulcer Disease and H. pylori Infection: Common Questions and Answers. Am Fam Physician. 2023;107:165-172. [PubMed] |
| 64. | Wiedermann CJ. Hypoalbuminemia as Surrogate and Culprit of Infections. Int J Mol Sci. 2021;22:4496. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 263] [Article Influence: 52.6] [Reference Citation Analysis (8)] |
| 65. | Lee CT, Wu CT, Chang WL, Yang EH, Hsieh MT, Chen WY, Sheu BS, Cheng HC. The nonadherence and risk factors of eradication failure by sequential therapy as first-line anti-Helicobacter pylori treatment in real-world clinical practice. Helicobacter. 2024;29:e13033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 66. | Genta RM, Graham DY. Comparison of biopsy sites for the histopathologic diagnosis of Helicobacter pylori: a topographic study of H. pylori density and distribution. Gastrointest Endosc. 1994;40:342-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 191] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 67. | Misra V, Misra S, Dwivedi M, Singh UP, Bhargava V, Gupta SC. A topographic study of Helicobacter pylori density, distribution and associated gastritis. J Gastroenterol Hepatol. 2000;15:737-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 68. | Patel SK, Pratap CB, Jain AK, Gulati AK, Nath G. Diagnosis of Helicobacter pylori: what should be the gold standard? World J Gastroenterol. 2014;20:12847-12859. [PubMed] [DOI] [Full Text] |
| 69. | Miftahussurur M, Yamaoka Y. Diagnostic Methods of Helicobacter pylori Infection for Epidemiological Studies: Critical Importance of Indirect Test Validation. Biomed Res Int. 2016;2016:4819423. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 95] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 70. | Best LM, Takwoingi Y, Siddique S, Selladurai A, Gandhi A, Low B, Yaghoobi M, Gurusamy KS. Non-invasive diagnostic tests for Helicobacter pylori infection. Cochrane Database Syst Rev. 2018;3:CD012080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (3)] |
| 71. | Fashner J, Gitu AC. Diagnosis and Treatment of Peptic Ulcer Disease and H. pylori Infection. Am Fam Physician. 2015;91:236-242. [PubMed] |
| 72. | Tai WC, Chiu CH, Liang CM, Chang KC, Kuo CM, Chiu YC, Wu KL, Hu ML, Chou YP, Chiou SS, Chiu KW, Kuo CH, Hu TH, Lin MT, Chuah SK. Ten-Day versus 14-Day Levofloxacin-Containing Triple Therapy for Second-Line Anti-Helicobacter pylori Eradication in Taiwan. Gastroenterol Res Pract. 2013;2013:932478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 73. | Nishikubo M, Kanamori M, Nishioka H. Levofloxacin-Associated Neurotoxicity in a Patient with a High Concentration of Levofloxacin in the Blood and Cerebrospinal Fluid. Antibiotics (Basel). 2019;8:78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 74. | Reddy VASK, Mittal GK, Sekhar S, Singhdev J, Mishra R. Levofloxacin-Induced Myoclonus and Encephalopathy. Ann Indian Acad Neurol. 2020;23:405-407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 75. | Tarnawski AS, Ahluwalia A. Increased susceptibility of aging gastric mucosa to injury and delayed healing: Clinical implications. World J Gastroenterol. 2018;24:4721-4727. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (3)] |
| 76. | Man JJ, Beckman JA, Jaffe IZ. Sex as a Biological Variable in Atherosclerosis. Circ Res. 2020;126:1297-1319. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 209] [Cited by in RCA: 304] [Article Influence: 50.7] [Reference Citation Analysis (0)] |
| 77. | Kim HL. Differences in Risk Factors for Coronary Atherosclerosis According to Sex. J Lipid Atheroscler. 2024;13:97-110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 19] [Article Influence: 9.5] [Reference Citation Analysis (0)] |