Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.117479
Revised: January 7, 2026
Accepted: February 6, 2026
Published online: May 7, 2026
Processing time: 137 Days and 2.6 Hours
Complete portal vein thrombosis (PVT) is a severe form of PVT that carries a high risk of portal hypertension-related complications and poses major therapeutic challenges. Although contrast-enhanced computed tomography (CT) is widely used for diagnosis and treatment planning, current classifications in both international and domestic practice are largely time-based (acute vs chronic). However, clinical onset frequently fails to align with true histopathological maturity, parti
To analyze the radiological signs on contrast-enhanced CT images of the livers of patients with complete PVT, propose a new classification scheme, and establish the clinical significance of the scheme.
We retrospectively analyzed 171 patients who were diagnosed with complete PVT treated at one of three Beijing centers from January 2018 to December 2023. Among patients without contraindications to anticoagulation who underwent interventional or surgical treatment combined with or followed by anticoagulation therapy, thrombus samples were obtained from 102 cases for pathological examination. Treatment outcomes were compared among groups based on the newly proposed imaging feature-based thrombus classifications.
Acute PVT (26.9%) was characterized by very low-density thrombus shadows in the blood vessel with uniform density, absent or minimal collateral blood vessel formation and no vascular wall thickening. Chronic PVT accounted for 34.5% of the patients, characterized by somewhat low-density thrombus shadows in the blood vessel, potentially with uneven density, increased collateral blood vessel formation, and thickening of the blood vessel wall. Cavernous transformation, which was observed in 12.9% of patients, involved the complete replacement of normal vascular anatomy with collateral vessels, whereas mixed PVT (25.7%) displayed heterogeneous features. Pathological examination revealed that different compositions correlated with distinct thrombus types. The posttreatment portal pressure gradient significantly decreased across all groups (P < 0.001), indicating favorable therapeutic efficacy.
Complete PVT has distinct properties and imaging manifestations. The proposed new thrombosis classification includes four types with distinct properties. Thrombosis with acute or chronic properties can be treated locally or with thrombolysis with good results.
Core Tip: This study aimed to analyze contrast-enhanced computed tomography imaging features of complete portal vein thrombosis and establish a novel imaging-based classification system with clinical relevance. In a retrospective multicenter analysis of 171 patients, thrombus samples from 102 cases were examined pathologically. Four distinct thrombus types were identified: Acute (26.9%), chronic (34.5%), cavernous transformation (12.9%), and mixed (25.7%), each characterized by specific radiological and pathological profiles. Treatment resulted in a significant reduction in portal pressure gradient across all groups (P < 0.001), supporting the clinical utility of the proposed classification in guiding therapeutic strategy.
- Citation: Lv YF, Liu BW, Han Y, Meng MM, Li DZ, Tian H, Wang FC, Dong JH, Yang YP, Zhou GD, Ding HG, Zhang YN, Liu FQ, Zhu B. Proposal of a new classification scheme for complete portal vein thrombosis and its clinical significance: A retrospective study. World J Gastroenterol 2026; 32(17): 117479
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/117479.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.117479
Portal vein thrombosis (PVT) is characterized by the formation of a thrombus within the main portal vein, intrahepatic portal vein branches, mesenteric vein, or splenic vein. It represents one of the most common vascular disorders of the liver[1,2]. PVT is traditionally classified as acute or chronic PVT on the basis of its duration. Thrombosis with a duration less than six months is defined as acute PVT, whereas thrombosis that persists beyond six months is considered chronic PVT[3]. The most typical symptoms of acute PVT are sudden onset of severe abdominal pain, nausea, fever and diarrhea. Severe cases manifest as acute portal hypertension syndrome. The onset time of chronic PVT is difficult to determine, and clinical manifestations can range from a completely asymptomatic presentation to symptoms revealing obvious portal hypertension[4-6]. Acute PVT is characterized by a high-density shadow within the portal vein on plain computed tomography (CT) scans. In contrast, chronic PVT manifests as a lower density shadow or as cavernous transformation of the portal vein on CT imaging[7,8]. There are notable distinctions in the management of acute and chronic PVT. For acute PVT, the primary treatment objective is to alleviate the occlusion of the portal vein and prevent both transition to chronic thrombosis, and thrombus dissemination. Conversely, for chronic PVT, the treatment is aimed at preventing thrombus progression and mitigating the complications of portal hypertension[9-12].
However, in clinical practice, typical symptoms rarely coincide with the onset of PVT formation[13,14]. Typical ima
This study initially enrolled a cohort of 171 patients diagnosed with complete PVT, defined as complete or near-complete (> 90%) occlusion of the portal vein trunk or associated superior mesenteric vein trunk and/or splenic vein trunk[17]. These patients received treatment at Beijing Shijitan Hospital Affiliated with Capital Medical University, the Fifth Medical Center of Chinese People’s Liberation Army General Hospital (302 Hospital), and Beijing You’an Hospital Affiliated with Capital Medical University between January 2018 and December 2023. The exclusion criteria for this study included the following: (1) Age greater than 75 years or less than 18 years; (2) Severe cardiopulmonary dysfunction or hepatic encephalopathy; (3) Portal system tumor thrombus; and (4) Missing clinical data or an inability to obtain clear contrast-enhanced CT images. The study was approved by the institutional review boards of the participating hospitals. All patients provided written informed consent to participate in this study. This study was registered in the Chinese Clinical Trials Registry (registration number: No. ChiCTR1800015268).
Prior to CT examination, patients fasted for 4-6 hours to ensure gastric emptying. All scans were performed in the supine position using a 64-slice or higher-resolution spiral CT scanner (e.g., GE Discovery CT 750 HD, GE Optima 660, or United Imaging uCT 960+). The scanning range extended from the diaphragm to the anterior superior iliac spine. Scanning parameters were individually adapted, including a tube voltage of 100-120 kVp, an automatic tube current modulation set within a range of 100-300 mA, a slice thickness of 5 mm, and a slice interval of 5 mm. Following the non-contrast scan, a nonionic contrast agent was administered intravenously via an antecubital vein at a rate of 3-4 mL/second and a dose of 1.5-2.0 mL/kg body weight. Contrast-enhanced acquisitions were performed in the arterial, venous, and delayed phases. Thin-section images with a thickness of 1.25 mm were reconstructed for each phase. All images were reviewed indepen
To minimize observation bias, all radiological evaluations were performed by two senior radiologists with over 10 years of experience in abdominal imaging. The readers were masked (blinded) to the patients’ clinical data and specific treatment groups. Any inter-observer discrepancies were resolved through consensus or by a third senior consultant.
Thrombus specimens were obtained intraoperatively. The acquisition of pathological samples was contingent upon the clinical feasibility and the type of surgical intervention. Specimens were strictly collected from patients undergoing surgical thrombectomy or endovascular procedures where thrombus aspiration/retrieval was part of the standard thera
Transjugular intrahepatic portosystemic shunts: Routine disinfection and draping were performed on the area of skin over the right internal jugular vein and right femoral artery. Under local anesthesia, a catheter was inserted into the femoral artery up to the hepatic artery for portal vein positioning. If this approach was unsuccessful, percutaneous hepatic puncture and intraportal vein cannulation were used for positioning. The right internal jugular vein was punctured and cannulated. Then, the RUPS-100 set [specialized for transjugular intrahepatic portosystemic shunts (TIPS) by COOK Medical, United States] was placed into the right atrium and inferior vena cava, and the pressure was mea
Percutaneous transhepatic portal vein puncture: Routine local disinfection and draping were performed under local anesthesia. Then, puncture through the right hypochondriac region (or under the xiphoid process) or jugular vein was conducted. Upon successful portal vein access, a 5F end-hole catheter (Cordis, United States) or a pigtail catheter was inserted into the distal vessel beyond the thrombus for angiography and portal pressure measurement. The catheter was then replaced with a vascular sheath and an 8F guiding catheter (Cordis, United States) for thrombectomy. After the varicose veins were embolized, a 5F balloon catheter (smaller than the diameter of the vessel containing the thrombus, from Covidien, United States) was used for thrombus fragmentation. Using angiography, it was confirmed that blood flow could bypass the thrombus margin into the liver. The catheter was left in place for thrombolysis. Thrombolysis was conducted using a 5F end-hole catheter or a 5F thrombolytic catheter (Edwards Lifesciences, United States) and administration of 200000 IU/day to 600000 IU/day of urokinase. If the outcomes were not ideal, a transition to TIPS was made. Patients were instructed to remain in bed under electrocardiogram monitoring. The puncture site was regularly checked for bleeding, portal vein pressure was measured at least three times daily, and blood pressure was maintained below 130/90 mmHg. There were 19 patients who received this treatment, 11 of whom obtained thrombus samples.
Surgical procedures: Thrombus specimens were obtained from 4 patients who underwent bowel resection due to intestinal ischemia and necrosis, and from 5 patients during liver transplantation. Among the 4 cases with intestinal ischemia and necrosis, 2 underwent surgical resection following TIPS, 1 underwent surgery after percutaneous transhepatic portal vein intervention, and 1 did not receive any specific intervention for portal venous system thrombosis prior to surgery.
Thrombus tissue sections were incubated in xylene and alcohol at different concentrations. These sections were stained with hematoxylin and eosin (HE). The stained sections were examined using an Olympus microscope system (Tokyo, Japan).
Categorized data are presented as absolute n (%), and continuous variables are expressed as the means and SD. SPSS Version 25.0 (SPSS, Inc., Chicago, IL, United States) and R software (version 4.5.2) was used for the statistical analysis. Differences before and after treatment were examined via paired t tests or, if the criteria for a normal distribution were not met, Wilcoxon signed-rank tests. Categorical data were analyzed by the χ2 test or Fisher’s exact test, if appropriate. Continuous variables were compared using the Student’s t-test, while categorical variables were analyzed using the χ2 test to assess statistical differences between the pathology group and the non-pathology group. Two-tailed P values < 0.05 were considered statistically significant. Inter-method consistency was evaluated using Kappa statistics. Pairwise agreement between the indicators (density, wall thickening, and collateral vessels) was assessed using Cohen’s kappa, while the overall agreement among all three indicators was measured using Fleiss’ kappa. The results were presented as kappa values (κ) with corresponding 95% confidence intervals (CIs). Statistical significance was defined as P < 0.05.
Complete revascularization was characterized by the absence of intraluminal thrombus or a completely patent portal venous system/TIPS shunt. Partial recanalization was defined as the presence of residual thrombus occupying less than 30% of the vessel diameter.
From January 2018 to December 2023, a total of 171 patients with complete PVT with complete clinical data were enrolled from three hospitals, including 124 males and 47 females aged 20-71 years, with an average age of 46.9 ± 4.8 years (Figure 1). The underlying comorbidities included hepatitis B-related cirrhosis in 80 patients, hepatitis C-related cirrhosis in 2 patients, alcoholic cirrhosis in 35 patients, primary biliary cirrhosis in 7 patients, autoimmune cirrhosis in 10 patients, Budd-Chiari syndrome in 12 patients, hepatic veno-occlusive disease in 6 patients, and unknown cause in 19 patients. The participants’ Child-Pugh classifications were as follows: Class A in 37.4% (64/171), class B in 44.4% (76/171), and class C in 18.1% (31/171) of the cohort. The primary clinical symptoms in these 171 patients included abdominal pain, gas
| Acute PVT (n = 46) | Chronic PVT (n = 59) | Cavernous transformation | Mixed PVT (n = 44) | |
| Age (years) | 45.2 ± 5.9 | 47.8 ± 4.2 | 45.4 ± 4.3 | 48.4 ± 3.6 |
| Gender | ||||
| Male | 33 (71.7) | 42 (71.2) | 17 (77.3) | 32 (72.7) |
| Female | 13 (28.3) | 17 (28.8) | 5 (22.7) | 12 (27.3) |
| Background disease | ||||
| HBV cirrhosis | 24 (52.2) | 29 (49.2) | 7 (31.8) | 20 (45.5) |
| HCV cirrhosis | 0 (0) | 1 (1.7) | 1 (4.5) | 0 (0) |
| ALC | 10 (21.7) | 13 (22.0) | 5 (22.7) | 7 (15.9) |
| PBC | 2 (4.3) | 3 (5.1) | 0 (0) | 2 (4.5) |
| AIC | 2 (4.3) | 6 (10.2) | 1 (4.5) | 1 (2.3) |
| BCS | 3 (6.5) | 5 (8.5) | 1 (4.5) | 3 (6.8) |
| HVOD | 1 (2.2) | 2 (3.4) | 1 (4.5) | 2 (4.5) |
| Unknown cause | 4 (8.7) | 0 (0) | 6 (27.3) | 9 (20.5) |
| Clinical symptoms | ||||
| Abdominal pain | 10 (21.7) | 10 (17.0) | 0 (0) | 6 (13.6) |
| Variceal bleeding | 12 (26.1) | 21 (35.6) | 8 (36.4) | 15 (34.1) |
| Refractory ascites | 15 (32.6) | 11 (18.6) | 5 (22.7) | 14 (31.8) |
| Both above | 5 (10.9) | 13 (22.0) | 8 (36.4) | 7 (15.9) |
| Intestinal necrosis | 0 (0.0) | 2 (3.4) | 1 (4.5) | 1 (2.3) |
| Severe jaundice | 4 (8.7) | 2 (3.4) | 0 (0) | 1 (2.3) |
| Child-Pugh grade | ||||
| Child A | 17 (37.0) | 21 (35.6) | 10 (45.5) | 16 (36.4) |
| Child B | 21 (45.6) | 26 (44.1) | 10 (45.5) | 19 (43.2) |
| Child C | 8 (17.4) | 12 (20.3) | 2 (9.1) | 9 (20.4) |
| Pre-PPG (mmHg) | 24.76 ± 5.69 | 22.73 ± 6.70 | 21.05 ± 5.65 | 24.63 ± 6.23 |
| Post-PPG (mmHg) | 12.88 ± 3.24 | 18.26 ± 5.57 | 14.07 ± 4.04 | 17.72 ± 4.48 |
The agreement among the three key radiological signs thrombus density, vascular wall thickening, and collateral vessel formation was evaluated to assess their consistency. Pairwise comparisons using Cohen’s kappa revealed substantial to almost perfect agreement between indicators, with κ values ranging from 0.713 to 0.806. Furthermore, the overall agreement across all three morphological indicators, assessed by Fleiss’ kappa, was substantial (κ = 0.755; 95%CI: 0.642-0.869; P < 0.001). These findings indicate a statistically significant concordance among the imaging features used for PVT classification (Table 2).
| Comparison | Kappa value | 95%CI | P value |
| Density vs wall thickening | 0.713 | 0.578-0.847 | < 0.001 |
| Density vs collateral vessels | 0.749 | 0.623-0.876 | < 0.001 |
| Wall thickening vs collateral vessels | 0.806 | 0.693-0.920 | < 0.001 |
| Density vs wall thickening vs collateral vessels | 0.755 | 0.642-0.869 | < 0.001 |
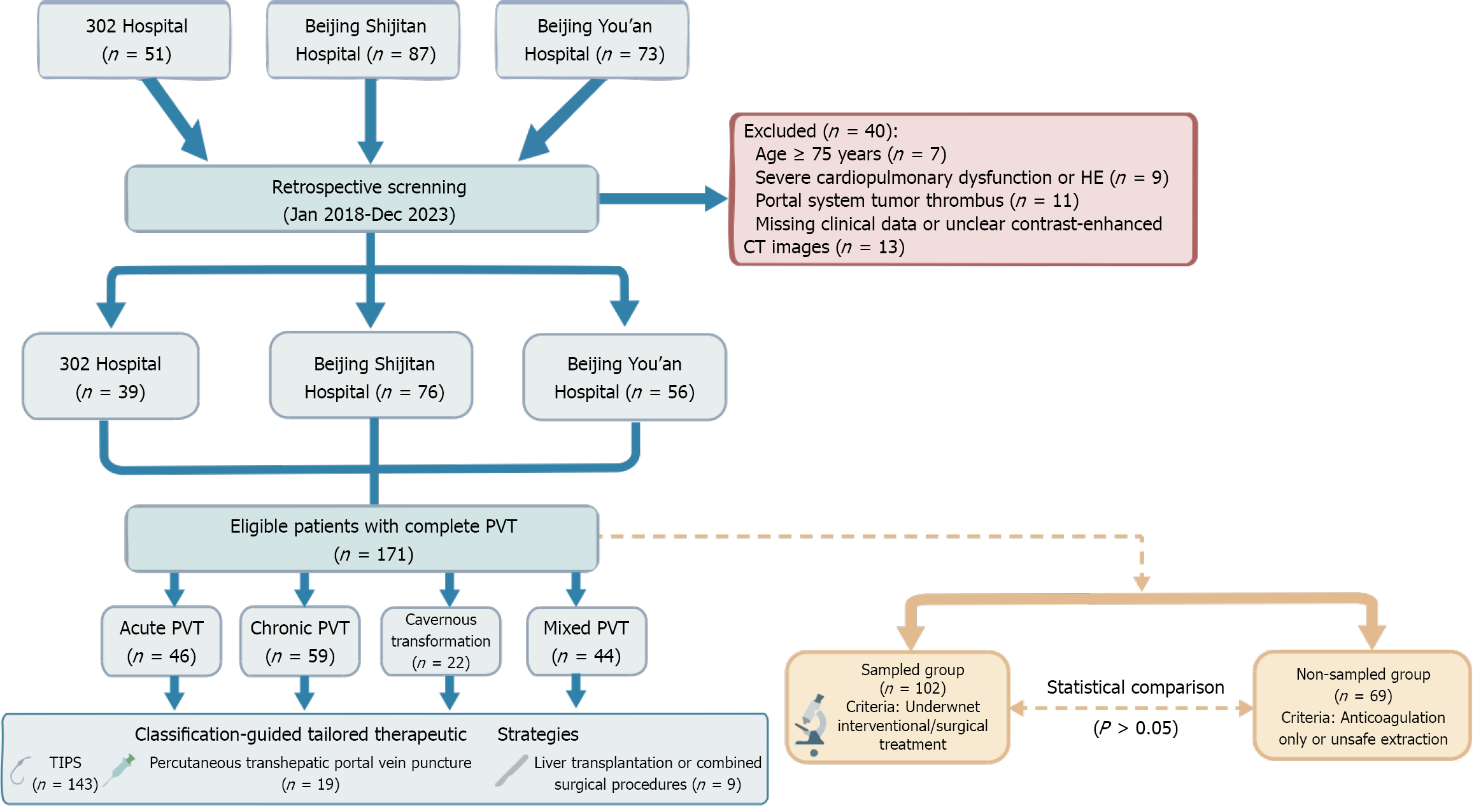
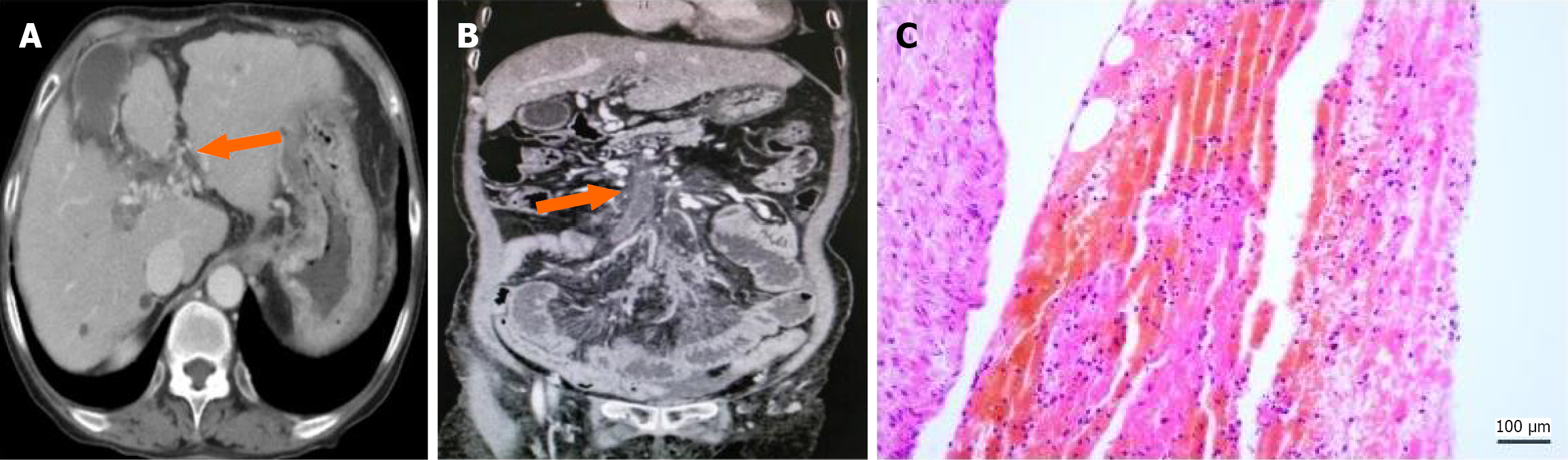
Acute PVT: In 46 patients (26.9%), the primary enhanced CT features included PVT that matched the vascular anatomy with very low-density shadows, uniform density (CT value: 39.98 ± 5.92 HU), no or minimal collateral vessel formation, and no thickening of the vessel wall (Figure 2A). In 4 patients, plain CT scans revealed that a small number of intrava
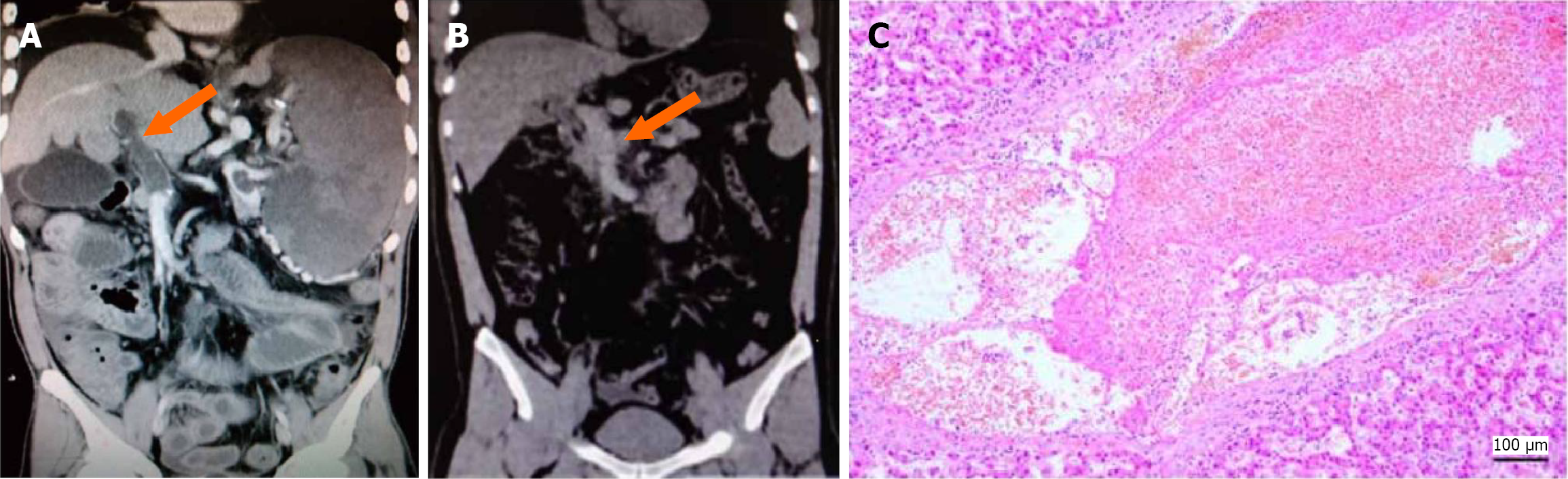
Chronic PVT: In 59 patients (34.5%), enhanced imaging revealed somewhat low-density thrombus shadows consistent with vascular anatomy, with either uniform or uneven density (CT value: 52.83 ± 9.84 HU), numerous collateral vessels forming around the thrombus, and thickened vessel walls (1.99 ± 0.46 mm) (Figure 3A).
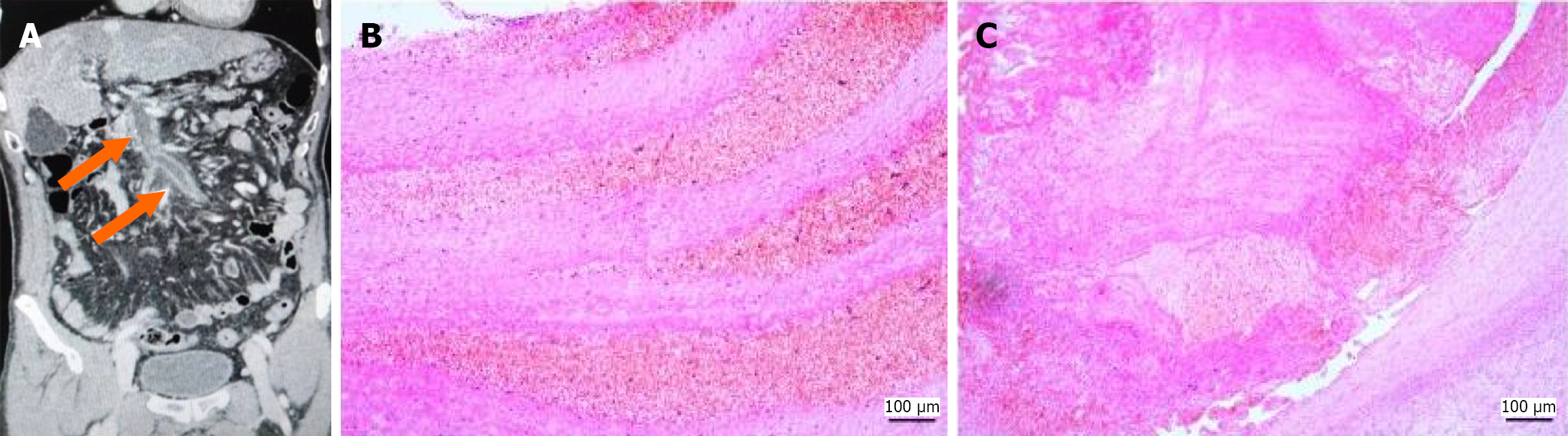
Cavernous transformation: In 22 patients (12.9%), the normal anatomical structure of the vessels at the site of the PVT was fully replaced by the formation of collateral vessels (Figure 4A).
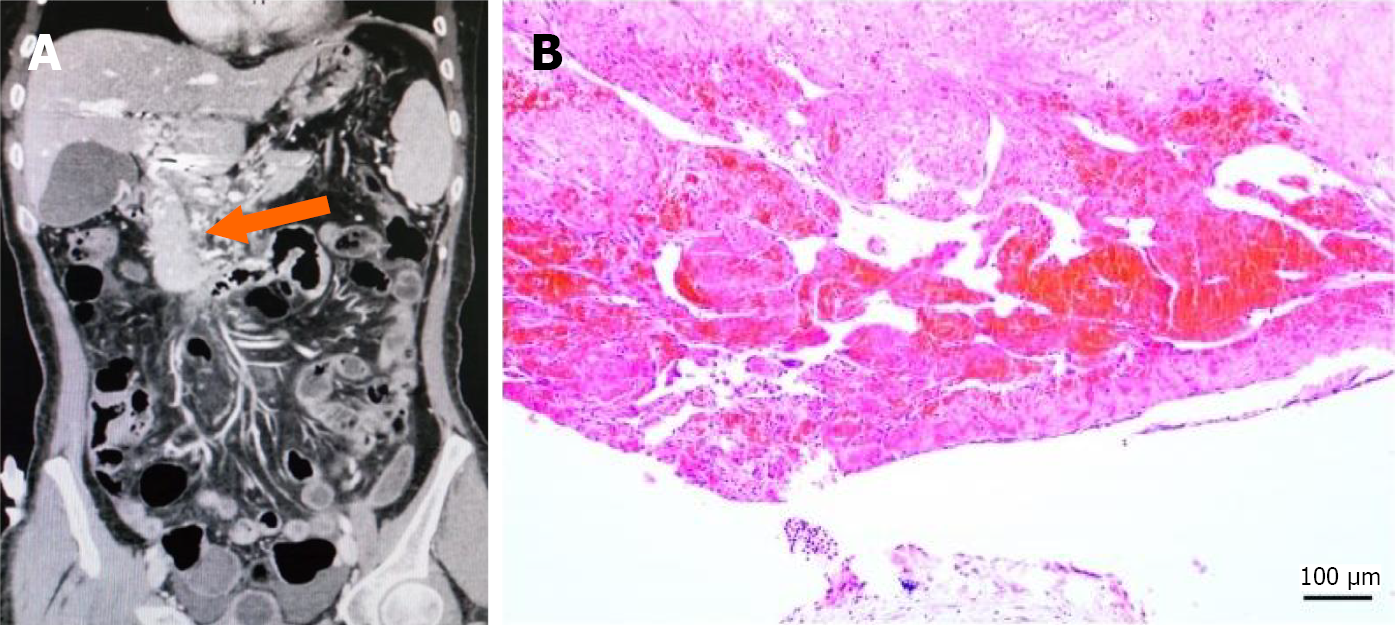
Mixed PVT: In 44 patients (25.7%), enhanced imaging demonstrated changes indicative of at least two or more types of thrombosis acute, chronic, and cavernous transformation within the PVT (Figure 5A and B, Supplementary Figures 1 and 2).
We compared the baseline characteristics of the sampled group (n = 102) vs the non-sampled group (n = 69). As shown in Table 3, there were no statistically significant differences between the two groups regarding age, gender distribution, background liver disease, clinical symptoms, or liver function (Child-Pugh grade). Importantly, the distribution of PVT classification (acute, chronic, cavernous transformation, mixed) was also balanced between the groups. These results suggest that the sampled cohort is representative of the overall study population and the pathological findings are not subject to significant spectrum bias.
| Sampled group (n = 102) | Non-sampled group (n = 69) | t/χ2 value | P value | |
| Age (years) | 47.0 ± 4.4 | 46.9 ± 5.0 | 0.116 | 0.908 |
| Gender | 0.471 | 0.493 | ||
| Male | 72 (70.6) | 52 (75.4) | ||
| Female | 30 (29.4) | 17 (24.6) | ||
| Background disease | 2.013 | 0.959 | ||
| HBV cirrhosis | 46 (45.1) | 34 (49.3) | ||
| HCV cirrhosis | 2 (2.0) | 0 (0.0) | ||
| ALC | 22 (21.5) | 13 (18.8) | ||
| PBC | 4 (3.9) | 3 (4.4) | ||
| AIC | 6 (5.9) | 4 (5.8) | ||
| BCS | 7 (6.9) | 5 (7.2) | ||
| HVOD | 3 (2.9) | 3 (4.4) | ||
| Unknown cause | 12 (11.8) | 7 (10.1) | ||
| Clinical symptoms | 7.179 | 0.208 | ||
| Abdominal pain | 20 (19.6) | 6 (8.7) | ||
| Variceal bleeding | 30 (29.4) | 26 (37.7) | ||
| Refractory ascites | 25 (24.5) | 20 (29.0) | ||
| Both above | 19 (18.7) | 14 (20.3) | ||
| Intestinal necrosis | 4 (3.9) | 0 (0.0) | ||
| Severe jaundice | 4 (3.9) | 3 (4.3) | ||
| Child-Pugh grade | 0.059 | 0.971 | ||
| Grade A | 38 (37.3) | 26 (37.7) | ||
| Grade B | 46 (45.1) | 30 (43.5) | ||
| Grade C | 18 (17.6) | 13 (18.8) | ||
| PVT classification | 0.789 | 0.852 | ||
| Acute PVT | 27 (26.5) | 19 (27.5) | ||
| Chronic PVT | 34 (33.3) | 25 (36.2) | ||
| Cavernous transformation | 15 (14.7) | 7 (10.2) | ||
| Mixed PVT | 26 (25.5) | 18 (26.1) |
Acute PVT was present in 27 patients (19 underwent TIPS; 7 underwent percutaneous transhepatic portal vein access; 1 underwent liver transplantation). Pathologically, these thrombus samples were primarily composed of platelets and red blood cells, with normal vascular walls. Figure 2C shows a HE stained section demonstrating aggregates of red blood cells and a vascular wall of normal thickness.
Chronic PVT was found in 34 patients (26 underwent TIPS; 4 underwent percutaneous transhepatic portal vein access; 2 underwent surgical procedures; 2 underwent liver transplantation). The pathological composition mainly included platelets, red blood cells, and fibrous tissue, with thickened vascular walls and proliferation of surrounding small vessels. Figure 3B and C shows HE stained sections demonstrating alternating aggregation of red blood cells and fibrin, along with a thickened vascular wall.
Cavernous transformation was observed in 15 patients (13 underwent TIPS; 1 underwent surgical procedures; 1 underwent liver transplantation). The pathology was characterized primarily by fibrous tissue and vascular proliferation, with no intact vascular walls visible. Figure 4B demonstrates a HE stained section showing aggregates of red blood cells, fibrous tissue, and prominent proliferation of small vessels.
Mixed PVT occurred in 26 patients (24 underwent TIPS; 1 underwent surgical procedures; 1 underwent liver trans
Acute PVT (n = 46): 34 patients underwent TIPS, 11 underwent percutaneous transhepatic portal vein puncture and 1 underwent liver transplantation. Complete disappearance of the primary vascular thrombus was achieved in 42 patients (91.3%), with residual thrombi smaller than 20% of the vessel diameter occurring in 3 patients (6.5%); 1 patient (2.2%) presented smooth portal blood flow post-transplantation. The mean preoperative portal pressure gradient (PPG) was 24.76 ± 5.69 mmHg, and the postoperative mean PPG was 12.88 ± 3.24 mmHg; the change in PPG was statistically different (P < 0.001).
Chronic PVT (n = 59): 48 patients underwent TIPS, 7 patients underwent percutaneous transhepatic portal vein puncture, 2 patients underwent surgical bowel resection following TIPS, and 2 patients underwent liver transplantation. Complete disappearance of the primary vascular thrombus occurred in 46 patients (78.0%), with residual thrombi smaller than 30% of the vessel diameter occurring in 11 patients (18.6%); 2 patients (3.4%) presented smooth portal blood flow post-transplantation. The mean preoperative PPG was 22.73 ± 6.70 mmHg, and the postoperative mean PPG was 18.26 ± 5.57 mmHg; the change in PPG was statistically different (P < 0.001).
Cavernous transformation (n = 22): 20 patients underwent TIPS, 8 of whom established shunts within the collateral branches and 12 of whom established shunt channels through potential original blood vessels and their distal vessels. Surgical bowel resection and liver transplantation were performed in 1 patient each. Complete patency was achieved in 21 cases (95.5%). 1 patient (4.5%) underwent solely bowel resection for necrosis, with no intervention for the portal thrombus. The mean preoperative PPG was 21.05 ± 5.65 mmHg, and the postoperative mean PPG was 14.07 ± 4.04 mmHg; the change in PPG was statistically different (P < 0.001).
Mixed PVT (n = 44): 41 patients underwent TIPS, 1 patient underwent surgical bowel resection following percutaneous transhepatic portal vein puncture, and 1 patient underwent liver transplantation. Complete disappearance of the main vascular thrombus was observed in 31 patients (70.5%) (Supplementary Figures 3 and 4), with residual thrombi smaller than 30% of the vessel diameter occurring in 12 patients (27.3%); 1 patient (2.2%) presented smooth portal blood flow post-transplantation. The mean preoperative PPG was 24.63 ± 6.23 mmHg, and the postoperative mean PPG was 17.72 ± 4.48 mmHg; the change in PPG was statistically different (P < 0.001).
The treatment outcomes in each group were mainly complete recanalization of blood vessels or complete patency of blood flow after reconstitution (Table 4). Fisher’s exact test indicated a significant overall difference in treatment outcomes among the groups (P = 0.005), particularly between the mixed PVT and acute PVT or cavernous transformation. Comparisons between acute PVT, chronic PVT, and cavernous transformation revealed no significant differences, indicating effective treatment outcomes. For mixed thrombosis, treatments should be tailored according to the predomi
Historically, PVT has been limited to classification as acute and chronic types, primarily based on the duration of abdominal pain. The duration of acute PVT is highly variable; however, it can extend up to six months. Thrombosis persisting beyond this period is classified as chronic[18]. A distinctive feature between acute and chronic PVT on unenhanced CT scans is the high-density appearance at the thrombus site[19,20]. However, in clinical practice, the actual duration of abdominal pain seldom coincides with the formation time of the thrombus, and high-density manifestations on CT are rare. Traditionally, chronic thrombosis is clinically characterized by abdominal pain persisting beyond six months, with imaging findings similar to those of acute thrombosis, with a low-density appearance within the vessels[21]. It is only at the stage of cavernous transformation, marked by significant formation of collateral vessels, that a clear distinction can be made[22].
In this study, a new classification system for thrombosis was developed on the basis of clinical observations and radiological findings. Clinically, it is exceedingly rare for the timing of abdominal pain to align with thrombus formation; here, only four cases showed such an alignment, all of which had a clear history of abdominal surgery during which thrombi formed perioperatively. Given the clinical challenges in differentiating between acute and chronic PVT, this study utilized imaging characteristics to delineate the nature of thrombosis, especially in cases of complete PVT. In cases of partial PVT, the characterization of thrombus based on imaging remains challenging due to persistent blood flow through the vessel. In contrast, complete thrombosis displays unequivocal imaging features. Therefore, this study exclusively enrolled patients with complete thrombosis. Pathological examination of the thrombus was performed to further validate the novel imaging-based classification. PVT was rigorously classified on the basis of imaging as acute, chronic, cavernous transformation, or mixed thrombosis. The principal elements considered by this classification are low-density vascular shadows consistent with vascular anatomy, whether and how many collateral vessels form at the margins, whether the vessel wall is thickened, and whether the vascular anatomy is absent; the nature of the thrombus can be clearly diagnosed through these imaging findings. Additionally, imaging from the same patient can reveal features characteristic of multiple types of thrombosis, suggesting that thrombi that form at different times during the overall process of thrombus formation exhibit distinct radiological characteristics. For example, acute thrombosis can develop into a chronic thrombosis or cavernous transformation background, here identified as mixed thrombosis, which accoun
The new classification holds significant clinical relevance. Relying solely on clinical symptoms or the duration of abdominal pain to determine the nature of thrombosis is imprecise unless the exact timing of thrombus formation is known[23,24]. Imaging manifestations can reveal various types of thrombi, each with distinct characteristic features, and correspond to different pathological compositions, thus guiding tailored therapeutic approaches. For acute thrombosis, the primary treatment includes anticoagulation and complete thrombus removal, which can be achieved via percutaneous transhepatic or jugular vein access to the portal vein for local thrombus management (including thrombectomy, thro
Meanwhile, our study has several limitations. First, the retrospective design and relatively small sample size in specific subgroups (e.g., cavernous transformation) may introduce inherent biases. Second, pathological analysis was limited to qualitative histological assessment in 102 patients due to surgical feasibility; however, a statistical audit confirmed that these sampled patients were representative of the entire cohort. Third, regarding statistical rigor, the study employed a classification-guided treatment strategy, which created structural confounding and severe multicollinearity between thrombus type and treatment modality. This precluded the use of multivariable regression models to calculate adjusted odds ratios. Nevertheless, the uniformly high recanalization rates across all groups validate the clinical utility of this tailored management approach.
In summary, the classification of thrombus is challenging in clinical settings, primarily because most patients lack definitive information on the timing of thrombus formation, and abdominal pain does not correlate with initial thrombus formation. This ambiguity complicates treatment decisions, leading to delays in appropriate treatment or inappropriate management and ultimately resulting in adverse outcomes. This study leveraged radiological signs to identify the nature of thrombus and analyzed the pathological compositions of different thrombus types. Then a novel classification system was proposed, which facilitated tailored therapeutic strategies accordingly, leading to favorable clinical outcomes.
Complete PVT has distinct properties and imaging manifestations. The proposed new thrombosis classification includes four types with distinct properties. Thrombosis with acute or chronic properties can be treated locally or with throm
| 1. | Intagliata NM, Caldwell SH, Tripodi A. Diagnosis, Development, and Treatment of Portal Vein Thrombosis in Patients With and Without Cirrhosis. Gastroenterology. 2019;156:1582-1599.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 269] [Article Influence: 38.4] [Reference Citation Analysis (2)] |
| 2. | Kodali S, Singal AK. Portal and Mesenteric Venous Thrombosis. Clin Liver Dis (Hoboken). 2020;16:142-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 3. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2065] [Article Influence: 516.3] [Reference Citation Analysis (15)] |
| 4. | Xu XY, Ding HG, Li WG, Xu JH, Han Y, Jia JD, Wei L, Duan ZP, Ling-Hu EQ, Zhuang H. Chinese guidelines on the management of liver cirrhosis (abbreviated version). World J Gastroenterol. 2020;26:7088-7103. [PubMed] [DOI] [Full Text] |
| 5. | Kallini JR, Gabr A, Kulik L, Ganger D, Lewandowski R, Thornburg B, Salem R. Noncirrhotic complete obliterative portal vein thrombosis: Novel management using trans-splenic transjugular intrahepatic portosystemic shunt with portal vein recanalization. Hepatology. 2016;63:1387-1390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 6. | Alzubaidi S, Patel I, Saini A, Knuttinen G, Naidu S, Kriegshuaser S, Albadawi H, Oklu R. Current concepts in portal vein thrombosis: etiology, clinical presentation and management. Abdom Radiol (NY). 2019;44:3453-3462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 7. | Loss M, Lang SA, Uller W, Wohlgemuth WA, Schlitt HJ. Combined surgical and interventional therapy of acute portal vein thrombosis without cirrhosis: a new effective hybrid approach for recanalization of the portal venous system. J Am Coll Surg. 2014;218:e79-e86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Marra P, Dulcetta L, Carbone FS, Muglia R, Muscogiuri G, Cheli M, D'Antiga L, Colledan M, Fagiuoli S, Sironi S. The Role of Imaging in Portal Vein Thrombosis: From the Diagnosis to the Interventional Radiological Management. Diagnostics (Basel). 2022;12:2628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 9. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines on vascular diseases of the liver. J Hepatol. 2026;84:399-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (4)] |
| 10. | Ma J, Yan Z, Luo J, Liu Q, Wang J, Qiu S. Rational classification of portal vein thrombosis and its clinical significance. PLoS One. 2014;9:e112501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 11. | Salei A, El Khudari H, McCafferty BJ, Varma RK. Portal Interventions in the Setting of Venous Thrombosis or Occlusion. Radiographics. 2022;42:1690-1704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 12. | Wang JT, Zhao HY, Liu YL. Portal vein thrombosis. Hepatobiliary Pancreat Dis Int. 2005;4:515-518. [PubMed] |
| 13. | Haris M, Thachil J. The problem with incidental and chronic portal vein thrombosis. Eur J Intern Med. 2017;39:e29-e30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 14. | Basit SA, Stone CD, Gish R. Portal vein thrombosis. Clin Liver Dis. 2015;19:199-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 15. | Hayat M, Aramvith S. Transformer’s Role in Brain MRI: A Scoping Review. IEEE Access. 2024;12:108876-108896. [DOI] [Full Text] |
| 16. | Hayat M, Aramvith S. Superpixel-Guided Graph-Attention Boundary GAN for Adaptive Feature Refinement in Scribble-Supervised Medical Image Segmentation. IEEE Access. 2025;13:196654-196668. [DOI] [Full Text] |
| 17. | Senzolo M, Garcia-Tsao G, García-Pagán JC. Current knowledge and management of portal vein thrombosis in cirrhosis. J Hepatol. 2021;75:442-453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 162] [Article Influence: 32.4] [Reference Citation Analysis (0)] |
| 18. | Bhangui P, Lim C, Levesque E, Salloum C, Lahat E, Feray C, Azoulay D. Novel classification of non-malignant portal vein thrombosis: A guide to surgical decision-making during liver transplantation. J Hepatol. 2019;71:1038-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 89] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 19. | Mathieu D, Vasile N, Grenier P. Portal thrombosis: dynamic CT features and course. Radiology. 1985;154:737-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 73] [Article Influence: 1.8] [Reference Citation Analysis (3)] |
| 20. | Marn CS, Francis IR. CT of portal venous occlusion. AJR Am J Roentgenol. 1992;159:717-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 35] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 21. | Sofia C, Cattafi A, Silipigni S, Pitrone P, Carerj ML, Marino MA, Pitrone A, Ascenti G. Portal vein thrombosis in patients with chronic liver diseases: From conventional to quantitative imaging. Eur J Radiol. 2021;142:109859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Sarin SK, Philips CA, Kamath PS, Choudhury A, Maruyama H, Nery FG, Valla DC. Toward a Comprehensive New Classification of Portal Vein Thrombosis in Patients With Cirrhosis. Gastroenterology. 2016;151:574-577.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 124] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 23. | Riva N, Ageno W. Clinical manifestations and imaging tools in the diagnosis of splanchnic and cerebral vein thromboses. Thromb Res. 2018;163:252-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Russell CE, Wadhera RK, Piazza G. Mesenteric venous thrombosis. Circulation. 2015;131:1599-1603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 25. | Naymagon L. Venous thrombosis of the liver: current and emerging concepts in management. Transl Res. 2020;225:54-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Qi X, Han G, Fan D. Management of portal vein thrombosis in liver cirrhosis. Nat Rev Gastroenterol Hepatol. 2014;11:435-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 123] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 27. | Parikh S, Shah R, Kapoor P. Portal vein thrombosis. Am J Med. 2010;123:111-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 134] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 28. | Meissner MH, Gloviczki P, Comerota AJ, Dalsing MC, Eklof BG, Gillespie DL, Lohr JM, McLafferty RB, Murad MH, Padberg F, Pappas P, Raffetto JD, Wakefield TW; Society for Vascular Surgery; American Venous Forum. Early thrombus removal strategies for acute deep venous thrombosis: clinical practice guidelines of the Society for Vascular Surgery and the American Venous Forum. J Vasc Surg. 2012;55:1449-1462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 276] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 29. | Winter MP, Schernthaner GH, Lang IM. Chronic complications of venous thromboembolism. J Thromb Haemost. 2017;15:1531-1540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 66] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 30. | Bartholomew JR. Update on the management of venous thromboembolism. Cleve Clin J Med. 2017;84:39-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 65] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 31. | Knott E, Levitin A, Obuchowski N, Karuppasamy K, Gadani S. Abstract No. 215 TIPS in Patients with Portal and Splanchnic Vein Thrombosis: A Single Center Retrospective Analysis. J Vasc Interv Radiol. 2023;34:S97-S98. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 32. | Wei B, Huang Z, Tang C. Optimal Treatment for Patients With Cavernous Transformation of the Portal Vein. Front Med (Lausanne). 2022;9:853138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 33. | Li ZP, Wang SS, Wang GC, Huang GJ, Cao JQ, Zhang CQ. Transjugular intrahepatic portosystemic shunt for the prevention of recurrent esophageal variceal bleeding in patients with cavernous transformation of portal vein. Hepatobiliary Pancreat Dis Int. 2018;17:517-523. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 34. | Gadani S, Partovi S, Levitin A, Zerona N, Sengupta S, D'Amico G, Diago Uso T, Menon KVN, Quintini C. Narrative review of portal vein thrombosis in cirrhosis: pathophysiology, diagnosis, and management from an interventional radiology perspective. Cardiovasc Diagn Ther. 2022;12:135-146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 35. | Guo FF, Wu ZY, Zhou PL, Han XW. [Transjugular intrahepatic portosystemic shunt for the treatment cavernous transformation of the portal vein with vareceal bleeding]. Zhonghua Yi Xue Za Zhi. 2020;100:387-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |