Published online May 7, 2026. doi: 10.3748/wjg.v32.i17.116867
Revised: January 7, 2026
Accepted: February 4, 2026
Published online: May 7, 2026
Processing time: 152 Days and 17.9 Hours
The effect of post-transjugular intrahepatic portosystemic shunt (TIPS) rebleeding on liver-related mortality remains uninvestigated.
To investigate the relationship between post-TIPS rebleeding and liver-related mortality in patients with cirrhosis and to conduct subgroup analyses based on liver function.
This study included 1782 patients who underwent covered TIPS at seven medical centers to prevent rebleeding. The primary endpoints were liver-related death and all-cause rebleeding. Propensity score matching, adjusted survival curves, and competing risk analyses based on liver transplantation and non-liver death were performed to ensure the robustness of the results.
During a median follow-up period of 32.25 months, 346 patients (19.4%) developed post-TIPS rebleeding, and 429 (24.1%) died from liver-related causes. Chronic HBV infection was the predominant cirrhosis etiology. Multiva
Post-TIPS rebleeding was significantly associated with higher mortality in patients with cirrhosis and variceal bleeding irrespective of liver function category.
Core Tip: Transjugular intrahepatic portosystemic shunt (TIPS) is considered a treatment option to prevent variceal rebleeding. However, the impact of post-TIPS rebleeding on mortality has not yet been investigated. Therefore, this study aimed to evaluate whether post-TIPS rebleeding increases liver-related mortality based on liver function categories in patients with cirrhosis. Our results suggested that post-TIPS rebleeding independently correlated with liver-related mortality, and it was significantly associated with higher liver-related mortality in patients with cirrhosis with variceal bleeding irrespective of liver function category. Therefore, clinical management following TIPS should include a rigorous follow-up strategy regardless of baseline Child-Pugh classification.
- Citation: Zhu JY, Tie J, Xia YF, Wang GC, Zhuge YZ, Wu H, Zhu XL, Xue H, Xu J, Zhang F, Zhao LH, Huang GJ, Zhang MY, Li Z, Wei B, Li PJ, Wang Z, Wu W, Chen C, Tang CW, Zhang CQ. Impact of rebleeding after transjugular intrahepatic portosystemic shunt on liver-related death based on liver function categories. World J Gastroenterol 2026; 32(17): 116867
- URL: https://www.wjgnet.com/1007-9327/full/v32/i17/116867.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i17.116867
The transjugular intrahepatic portosystemic shunt (TIPS) procedure is a widely used radiological intervention for treating portal hypertension and its complications[1]. According to previous practice guidelines and Baveno VII recommendations, TIPS is considered a treatment option for patients in whom standard endoscopic therapy combined with β-blockers fails to prevent variceal rebleeding[2-4].
Rebleeding following initial TIPS treatment remains a known complication with an incidence rate of approximately 6%-40% within 1-2 years[5]. Specifically, the absence of adjunctive embolization of variceal collaterals during the TIPS procedure, high model for end-stage liver disease (MELD)-Na scores, and portal hypertensive gastropathy on pre-TIPS endoscopy may contribute to an increased risk of developing post-TIPS rebleeding[5-7]. Rebleeding is associated with high incidence and mortality rates in patients with cirrhosis who have recovered from acute variceal bleeding without secondary prevention[8]. However, the impact of post-TIPS rebleeding on mortality has not yet been investigated, and whether post-TIPS rebleeding increases liver-related deaths in patients post-TIPS remains uncertain.
A recent individual patient meta-analysis found that patients treated with TIPS for acute variceal bleeding or ascites had improved survival compared with those treated with standard of care, whereas no significant survival benefit was observed in the context of rebleeding prevention although TIPS reduced rebleeding rates[9]. Several tools, including the MELD score, Mayo TIPS model, Freiburg index of post-TIPS survival score, and elderly patient calculator TIPS score, have been proposed to predict survival following TIPS implantation[10-13]. However, these scores do not consider post-TIPS rebleeding as a potential factor affecting survival; they primarily reflect the impact of liver function.
This nationwide, multicenter cohort study aimed to assess whether post-TIPS rebleeding increases liver-related mor
This study included patients with cirrhosis who underwent TIPS to prevent rebleeding at seven academic hospitals in China between June 2010 and June 2020. All hospitals had gastroenterology or liver units where experienced clinicians regularly perform TIPS. All patients received specific treatment for the underlying cause of liver cirrhosis, including ongoing antiviral therapy, alcohol abstinence, or ursodeoxycholic acid, depending on the etiology.
The inclusion criteria were as follows: (1) Diagnosis of cirrhosis (based on clinical signs, laboratory and imaging tests, or liver biopsy); and (2) TIPS performed for secondary prophylaxis of variceal bleeding. The exclusion criteria were as follows: (1) Age < 14 years or > 75 years; (2) Child-Pugh score > 13; (3) TIPS with uncovered stents; (4) Unrelieved bile duct obstruction, hepatocellular carcinoma, or other malignancies; (5) Prior TIPS procedure, portocaval surgery, or splenectomy; (6) Heart failure (New York Heart Association class > III), coronary heart disease, or arrhythmia requiring treatment; (7) Partially or completely occlusive portal vein; (8) Cavernous transformation; and (9) Incomplete clinical data.
This study was conducted according to the principles of the Declaration of Helsinki and was approved by the Biomedical Research Ethics Committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (No. SWYX 2022-1005). The requirement for informed consent was waived due to the retrospective nature.
All laboratory indicators were collected from the most recent pre-TIPS measurements, reflecting the patient’s stable baseline status, typically within 24-48 h before the procedure. The mean or median of the indicator was used as a substi
TIPS was performed under local anesthesia using a standard technique[7]. The procedure was completed under continuous X-ray guidance. A polytetrafluoroethylene-covered stent (Viatorr stent, Gore Medical, Flagstaff, AZ, United States, or Fluency stent, BD, Franklin Lakes, NJ, United States) of an appropriate diameter (6 mm, 8 mm, or 10 mm) was implanted via the transjugular approach to establish a connection between the portal vein and hepatocaval junction. The stent diameter was primarily selected based on the operator’s experience and patient characteristics, including pre-TIPS portal pressure gradient (PPG), pre-TIPS hepatic encephalopathy, liver function, and stent availability. For patients with poor preoperative liver function or a history of severe hepatic encephalopathy, smaller-diameter stents were preferred to balance portal venous decompression with the risks of liver function deterioration and worsening hepatic encephalopathy. PPG was measured as the difference between the pressure in the portal vein and the inferior vena cava (rather than in the right atrium) using a 5.5F Fogarty Thru-Lumen Embolectomy Catheter (Edwards Lifesciences LLC, Irvine, CA, United States) before and after TIPS implantation. For gastroesophageal varices with spontaneous portal shunts (such as gastrorenal shunts) discovered during intraoperative angiography, interlock coils (Boston Scientific, Marlborough, MA, United States) and/or Glubran 2 cyanoacrylate glue (GEM S.R.L., Italy) were used to embolize the lateral varicose veins before TIPS creation.
The patency of the stent was assessed by Doppler ultrasound or CT after the procedure. Stent dysfunction was defined as the presence of filling defects, thrombosis, flow obstruction, or absence of flow signals within the stent observed on abdominal ultrasound or CT.
The primary endpoints of this study were liver-related death and all-cause rebleeding. The secondary endpoint was overt hepatic encephalopathy (OHE). Liver-related deaths included liver failure, variceal bleeding, or other complications of cirrhosis. All-cause rebleeding was defined according to the Baveno VI consensus[14] and referred to any first rebleeding event originating from the gastrointestinal tract and meeting clinical significance criteria, regardless of whether it was esophagogastric variceal bleeding, occurring between 24 h after the TIPS procedure (to avoid interference from perioperative procedural bleeding) and the last follow-up. Diagnosis required clinical manifestations of hematemesis, melena, or hematochezia along with objective evidence of hemodynamic instability or active hemorrhage: A decrease in hemo
Normality of continuous variables was assessed using the Shapiro-Wilk test. Quantitative variables that followed a normal distribution were expressed as mean ± SD and compared using the Student’s t test; those that did not follow a normal distribution were expressed as median (quartiles) and compared using the Mann-Whitney U test. Qualitative variables were expressed as n (%) and compared using the χ² test. A Cox proportional hazards model was used for uni
To control for baseline confounding factors and make the rebleeding group more comparable with the non-rebleeding group on key prognostic variables, we applied propensity score matching (PSM) to construct the analysis cohort using the nearest-neighbor matching procedure with a caliper width of 0.2. Notably, a 1:2 (post-TIPS rebleeding/no post-TIPS rebleeding) PSM matching was performed to retain as many cases as possible in the post-TIPS rebleeding group to maintain its representativeness and matching the best controls from a larger control group pool to enhance statistical power and reduce estimation bias. Matching covariables included age, sex, hemoglobin level, platelet count, PT, creatinine level, Child-Pugh score, MELD-Na score, and post-TIPS PPG levels, representing baseline imbalances between the two groups. Standard mean difference (SMD) and love plot (Supplementary Figure 1) were used to evaluate group comparability before and after PSM. SMD < 0.2 indicated a small difference between the groups[16].
Subgroup analysis was performed based on different liver function categories to evaluate the consistency and robustness of the impact of rebleeding after TIPS on liver-related mortality.
A two-sided P < 0.05 was considered statistically significant. All analyses were performed using SPSS version 25 (IBM Corporation, Armonk, NY, United States) and R software version 4.4.1 (http://www.R-project.org/).
This study included 1782 patients and a median follow-up period of 32.25 months. Supplementary Figure 2 presents the patient enrollment flowchart. The median age of the patients was 51 years, and 1150 (64.5%) were male. Chronic HBV infection was the predominant etiology of cirrhosis (61.3%). Furthermore, 738 (41.4%), 887 (49.8%), and 157 (8.8%) patients were classified as Child-Pugh A, B, and C, respectively, with a median MELD score of 10 (9-12). Stent diameters of 6-7 mm, 8 mm, and 10 mm were used in 87 (4.9%), 1603 (90.0%), and 92 (5.2%) patients, respectively. PPG decreased from 24.0 (20-27) mmHg before TIPS to 8.8 (6-11) mmHg after TIPS in all patients.
Before PSM post-TIPS rebleeding occurred in 346 patients (19.4%), who were generally younger (median 50 vs 52, P = 0.020), more likely male (74.6% vs 62.1%, P < 0.001), had lower hemoglobin (median 79.5 vs 83.0, P = 0.003), had lower platelet count (median 54 vs 61, P = 0.009), had higher creatinine levels (median 75.2 vs 71.0, P < 0.001), and had pro
| Characteristics | Before PSM | After PSM | ||||||||
| Total | No post-TIPS rebleeding | Post-TIPS rebleeding | P value | SMD | Total | No post-TIPS rebleeding | Post-TIPS rebleeding | P value | SMD | |
| Age (years) | 51 (44, 60) | 52 (44, 60) | 50 (43, 59) | 0.020 | 0.140 | 50 (43, 58) | 50 (43, 58) | 50 (43, 59) | 0.803 | 0.023 |
| Sex | < 0.001 | 0.270 | 0.547 | 0.045 | ||||||
| Male | 1150 (64.5) | 892 (62.1) | 258 (74.6) | 750 (73.1) | 494 (72.4) | 256 (74.4) | ||||
| Female | 632 (35.5) | 544 (37.9) | 88 (25.4) | 276 (26.9) | 188 (27.6) | 88 (25.6) | ||||
| Cirrhosis etiology | 0.291 | 0.152 | 0.770 | 0.107 | ||||||
| Chronic HBV infection | 1092 (61.3) | 861 (60.0) | 231 (66.8) | 662 (64.5) | 432 (63.3) | 230 (66.9) | ||||
| Other viral infection | 108 (6.1) | 87 (6.1) | 21 (6.1) | 61 (5.9) | 40 (5.9) | 21 (6.1) | ||||
| Alcoholic liver disease | 106 (5.9) | 88 (6.1) | 18 (5.2) | 63 (6.1) | 46 (6.7) | 17 (4.9) | ||||
| Autoimmune hepatitis | 170 (9.5) | 143 (10.0) | 27 (7.8) | 93 (9.1) | 66 (9.7) | 27 (7.8) | ||||
| Mixed etiology | 41 (2.3) | 34 (2.4) | 7 (2.0) | 20 (1.9) | 13 (1.9) | 7 (2.0) | ||||
| Others | 265 (14.9) | 223 (15.5) | 42 (12.1) | 127 (12.4) | 85 (12.5) | 42 (12.2) | ||||
| Hepatic encephalopathy before procedure | 17 (1.0) | 15 (1.0) | 2 (0.6) | 0.622 | 0.052 | 5 (0.5) | 3 (0.4) | 2 (0.6) | 1.000 | 0.020 |
| Ascites before procedure | 0.400 | 0.069 | 0.763 | 0.049 | ||||||
| No | 589 (33.1) | 464 (32.3) | 125 (36.1) | 356 (34.7) | 232 (34.0) | 124 (36.0) | ||||
| Mild | 648 (36.4) | 528 (36.8) | 120 (34.7) | 372 (36.3) | 252 (37.0) | 120 (34.9) | ||||
| Moderate and severe | 545 (30.6) | 444 (30.9) | 101 (29.2) | 298 (29.0) | 198 (29.0) | 100 (29.1) | ||||
| Hemoglobin (g/L) | 82 (71, 96) | 83 (72, 97) | 80 (68, 94) | 0.003 | 0.183 | 80 (69, 93) | 80 (69, 92) | 80 (68, 94) | 0.881 | 0.013 |
| Platelet (109/L) | 60 (41, 88) | 61 (42, 89) | 54 (40, 82) | 0.009 | 0.082 | 58 (41, 84) | 60 (43, 87) | 54 (40, 82) | 0.062 | 0.057 |
| Albumin (g/L) | 34.3 (30.8, 37.7) | 34.3 (30.8, 37.7) | 34.4 (30.7, 37.3) | 0.744 | 0.007 | 34.4 (30.9, 37.5) | 34.3 (31.0, 37.5) | 34.4 (30.7, 37.3) | 0.786 | 0.001 |
| Total bilirubin (µmol/L) | 20.3 (14.0, 29.3) | 20.0 (13.9, 29.6) | 20.6 (14.3, 28.3) | 0.848 | 0.031 | 20.1 (13.9, 28.7) | 19.9 (13.8, 28.7) | 20.6 (14.3, 28.4) | 0.699 | 0.010 |
| Creatinine (µmol/L) | 72 (58, 88) | 71 (57, 87) | 75 (63, 89) | < 0.001 | 0.201 | 76 (61, 91) | 77 (60, 92) | 75 (63, 89) | 0.968 | 0.030 |
| Prothrombin time lengthen time (seconds) | 1.4 (0, 3.1) | 1.4 (0, 3.0) | 1.8 (0.1, 3.3) | 0.028 | 0.093 | 1.6 (0.1, 3.2) | 1.5 (0.1, 3.1) | 1.8 (0.2, 3.32) | 0.268 | 0.034 |
| International normalized ratio | 1.30 (1.18, 1.45) | 1.29 (1.18, 1.44) | 1.31 (1.20, 1.48) | 0.173 | 0.124 | 1.31 (1.20, 1.46) | 1.31 (1.20, 1.46) | 1.31 (1.20, 1.47) | 0.912 | 0.063 |
| Sodium (mmol/L) | 139.9 (137.4, 142.0) | 139.9 (137.5, 142.0) | 140.0 (137.4, 142.0) | 0.999 | 0.004 | 139.8 (137.4, 141.9) | 139.6 (137.3, 141.8) | 140.0 (137.4, 141.9) | 0.340 | 0.071 |
| Child-Pugh score | 7 (6, 8) | 7 (6, 8) | 7 (6, 8) | 0.813 | 0.012 | 7 (6, 8) | 7 (6, 8) | 7 (6, 8) | 0.917 | 0.007 |
| Child-Pugh class | 0.867 | 0.032 | 0.861 | 0.036 | ||||||
| A | 738 (41.4) | 592 (41.2) | 146 (42.2) | 431 (42.0) | 286 (41.9) | 145 (42.2) | ||||
| B | 887 (49.8) | 719 (50.1) | 168 (48.6) | 506 (49.3) | 339 (49.7) | 167 (48.5) | ||||
| C | 157 (8.8) | 125 (8.7) | 32 (9.2) | 89 (8.7) | 57 (8.4) | 32 (9.3) | ||||
| MELD score | 10 (9, 12) | 10 (9, 12) | 10 (9, 12) | 0.074 | 0.124 | 11 (9, 13) | 11 (9, 13) | 11 (9, 13) | 0.739 | 0.035 |
| MELD-Na score | 11 (9, 13) | 11 (9, 13) | 11 (9, 13) | 0.073 | 0.101 | 11 (9, 13) | 11 (9, 13) | 11 (9, 13) | 0.761 | < 0.001 |
| Combine venous embolization | 1231 (69.1) | 993 (69.2) | 238 (68.8) | 0.947 | 0.008 | 718 (70.0) | 481 (70.5) | 237 (68.9) | 0.641 | 0.036 |
| TIPS stent diameter | 0.179 | 0.107 | 0.535 | 0.073 | ||||||
| < 8 mm | 87 (4.9) | 65 (4.5) | 22 (6.4) | 55 (5.4) | 33 (4.8) | 22 (6.4) | ||||
| 8 mm | 1603 (90.0) | 1301 (90.6) | 302 (87.3) | 909 (88.6) | 609 (89.3) | 300 (87.2) | ||||
| 10 mm | 92 (5.2) | 70 (4.9) | 22 (6.4) | 62 (6.0) | 40 (5.9) | 22 (6.4) | ||||
| Pre-TIPS PPG (mmHg) | 24.00 (20.00, 27.21) | 24.00 (20.00, 27.21) | 23.56 (20.00, 27.21) | 0.664 | 0.043 | 24.00 (20.37, 27.93) | 24.00 (20.59, 27.94) | 23.53 (20.00, 27.21) | 0.347 | 0.049 |
| Post-TIPS PPG (mmHg) | 8.80 (6.00, 11.00) | 8.80 (6.00, 11.00) | 8.82 (6.60, 11.03) | 0.050 | 0.131 | 9.00 (6.60, 11.30) | 9.00 (6.60, 11.62) | 8.82 (6.60, 11.03) | 0.832 | 0.014 |
| PPG reduction (mmHg) | 14.70 (11.00, 18.38) | 14.71 (11.00, 18.40) | 14.00 (11.00, 17.66) | 0.380 | 0.051 | 14.70 (11.00, 18.36) | 14.71 (11.00, 18.44) | 14.00 (11.00, 17.68) | 0.300 | 0.057 |
Overall, 530 (29.7%) patients died with 429 (80.9%) deaths attributed to liver-related causes. Supplementary Table 1 presents the cumulative incidence of post-TIPS rebleeding at different time points. All-cause and liver-related deaths occurred more frequently in patients with post-TIPS rebleeding than in those without rebleeding (before PSM: 40.5% vs 27.2%, P < 0.001 and 35.5% vs 21.3%, P < 0.001, respectively; after PSM: 40.7% vs 27.0%, P < 0.001 and 35.8% vs 21.6%, P < 0.001, respectively; Table 2). During the follow-up period, OHE occurred more frequently in patients with post-TIPS rebleeding than in those without it (before PSM: 38.4% vs 32.5%, P = 0.040; after PSM: 38.1% vs 32.0%, P = 0.060; Table 2). Before PSM the OHE incidence rate in patients with post-TIPS rebleeding was 14.30 per 100 patient-years, significantly higher than that in patients without rebleeding (12.08 per 100 patient-years). After PSM the OHE incidence rate remained higher in the rebleeding group (13.37 vs 11.23 per 100 patient-years). Due to the retrospective nature of the study, it was difficult to precisely determine the temporal sequence between OHE and rebleeding events. The relationship between these two factors is complex and bidirectional. Future prospective studies are needed to further clarify the temporal relationship and interaction between the two.
| Characteristics | Before PSM | After PSM | ||||||||
| Total | No post-TIPS rebleeding | Post-TIPS rebleeding | P value | SMD | Total | No post-TIPS rebleeding | Post-TIPS rebleeding | P value | SMD | |
| Overt hepatic encephalopathy | 599 (33.6) | 466 (32.5) | 133 (38.4) | 0.040 | 0.125 | 349 (34.0) | 218 (32.0) | 131 (38.1) | 0.060 | 0.128 |
| All-cause death | 530 (29.7) | 390 (27.2) | 140 (40.5) | < 0.001 | 0.284 | 324 (31.6) | 184 (27.0) | 140 (40.7) | < 0.001 | 0.293 |
| Liver-related death | 429 (24.1) | 306 (21.3) | 123 (35.5) | < 0.001 | 0.320 | 270 (26.3) | 147 (21.6) | 123 (35.8) | < 0.001 | 0.318 |
Compared with survivors, patients who died from liver-related causes were older, exhibited more severe ascites, had higher MELD and MELD-Na scores, and a greater proportion were classified as Child-Pugh B and C classes (all P < 0.05; Supplementary Table 2). Furthermore, non-survivors exhibited higher total bilirubin, creatinine, and international normalized ratios, prolonged PT, and lower albumin and sodium levels (all P < 0.05; Supplementary Table 2). Additiona
The univariable analysis and multivariable Cox proportional hazard model revealed that older age, higher Child-Pugh and MELD-Na scores, and post-TIPS rebleeding as independent predictive factors for liver-related death (Table 3).
| Variable | Univariate analysis | Multivariate analysis | ||
| HR (95%CI) | P value | HR (95%CI) | P value | |
| Age | 1.030 (1.020-1.040) | < 0.001 | 1.031 (1.022-1.040) | < 0.001 |
| Sex (female vs male) | 1.080 (0.889-1.320) | 0.424 | ||
| Cirrhosis etiology | ||||
| Alcoholic liver disease vs chronic HBV infection | 0.832 (0.509-1.360) | 0.107 | ||
| Other viral infection vs chronic HBV infection | 1.330 (0.938-1.900) | 0.109 | ||
| Autoimmune hepatitis vs chronic HBV infection | 1.100 (0.768-1.570) | 0.603 | ||
| Mixed etiology vs chronic HBV infection | 0.960 (0.475-1.940) | 0.107 | ||
| Others vs chronic HBV infection | 1.210 (0.929-1.580) | 0.156 | ||
| Previous ascites | ||||
| Mild vs no | 1.260 (0.996-1.600) | 0.054 | ||
| Moderate and severe vs no | 2.070 (1.630-2.630) | < 0.001 | ||
| Child-Pugh score | 1.260 (1.200-1.330) | < 0.001 | 1.207 (1.121-1.300) | < 0.001 |
| Model for end-stage liver disease-Na score | 1.060 (1.040-1.070) | < 0.001 | 1.035 (1.016-1.055) | < 0.001 |
| Transjugular intrahepatic portosystemic shunt stent diameter | ||||
| 8 mm vs < 8 mm | 0.912 (0.484-1.720) | 0.775 | ||
| 10 mm vs < 8 mm | 1.240 (0.615-2.490) | 0.549 | ||
| Portal pressure gradient reduction | 1.010 (0.997-1.030) | 0.119 | ||
| Rebleeding | 1.490 (1.210-1.840) | < 0.001 | 1.609 (1.304-1.986) | < 0.001 |
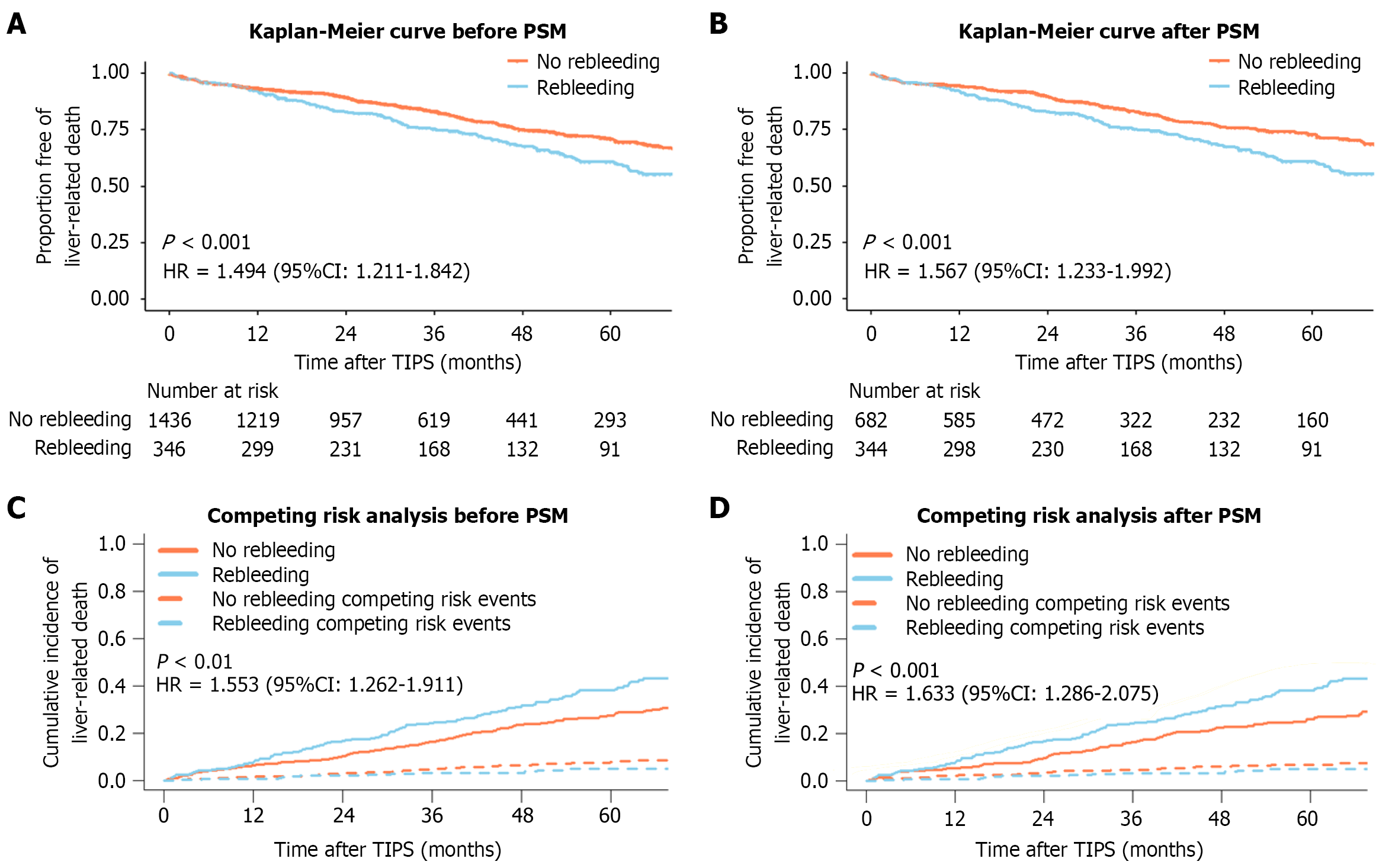
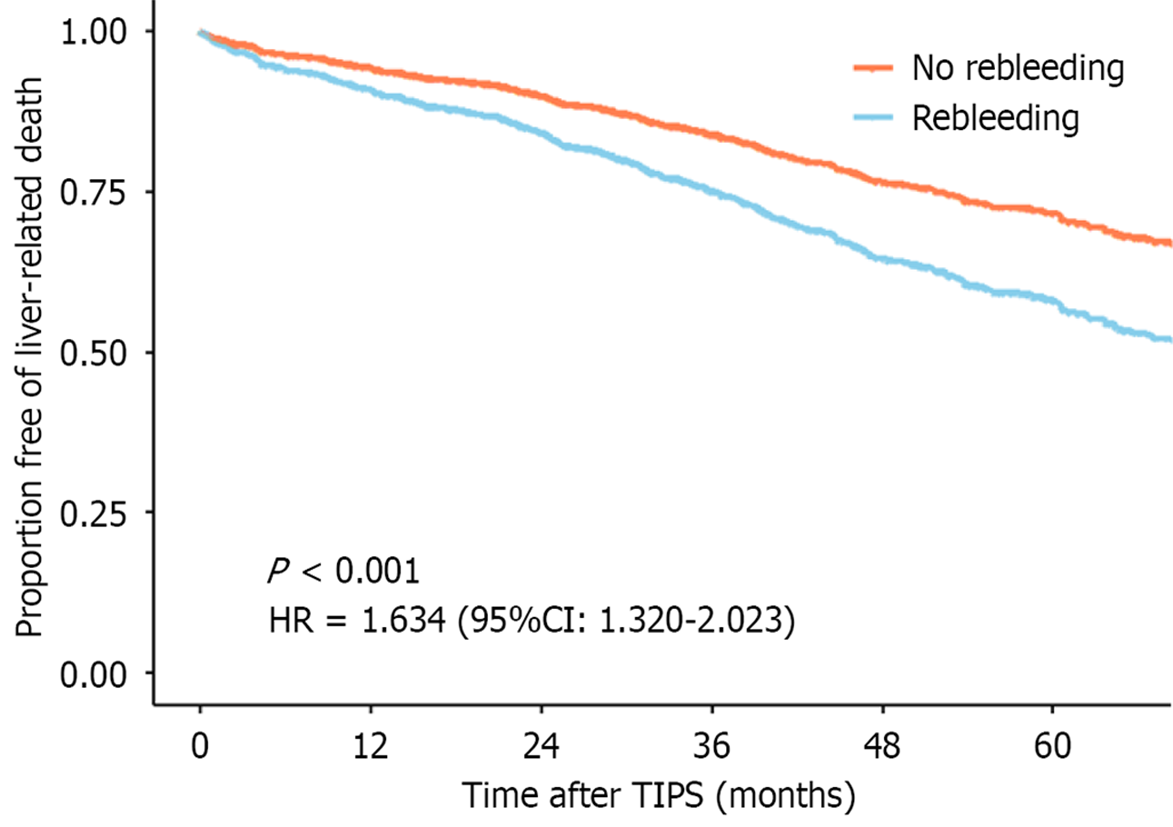
Patients with post-TIPS rebleeding had a higher cumulative liver-related mortality than those without rebleeding [before PSM (1 year: 8.3% vs 6.7%, 3 years: 25.2% vs 17.2%, 5 years: 40.6% vs 30.2%; HR = 1.494, 95%CI: 1.211-1.842, P < 0.001; Figure 1A), after PSM (1 year: 8.4% vs 5.6%, 3 years: 25.2% vs 17.4%, 5 years: 40.6% vs 28.3%; HR = 1.567, 95%CI: 1.233-1.992, P < 0.001; Figure 1B)]. This difference persisted even after accounting for competing events (before PSM: HR = 1.553, 95%CI: 1.262-1.911, P < 0.001; Figure 1C; after PSM: HR = 1.633, 95%CI: 1.286-2.075, P < 0.001; Figure 1D). The adjusted survival curve remained consistent (HR = 1.634, 95%CI: 1.320-2.023, P < 0.001; Figure 2).
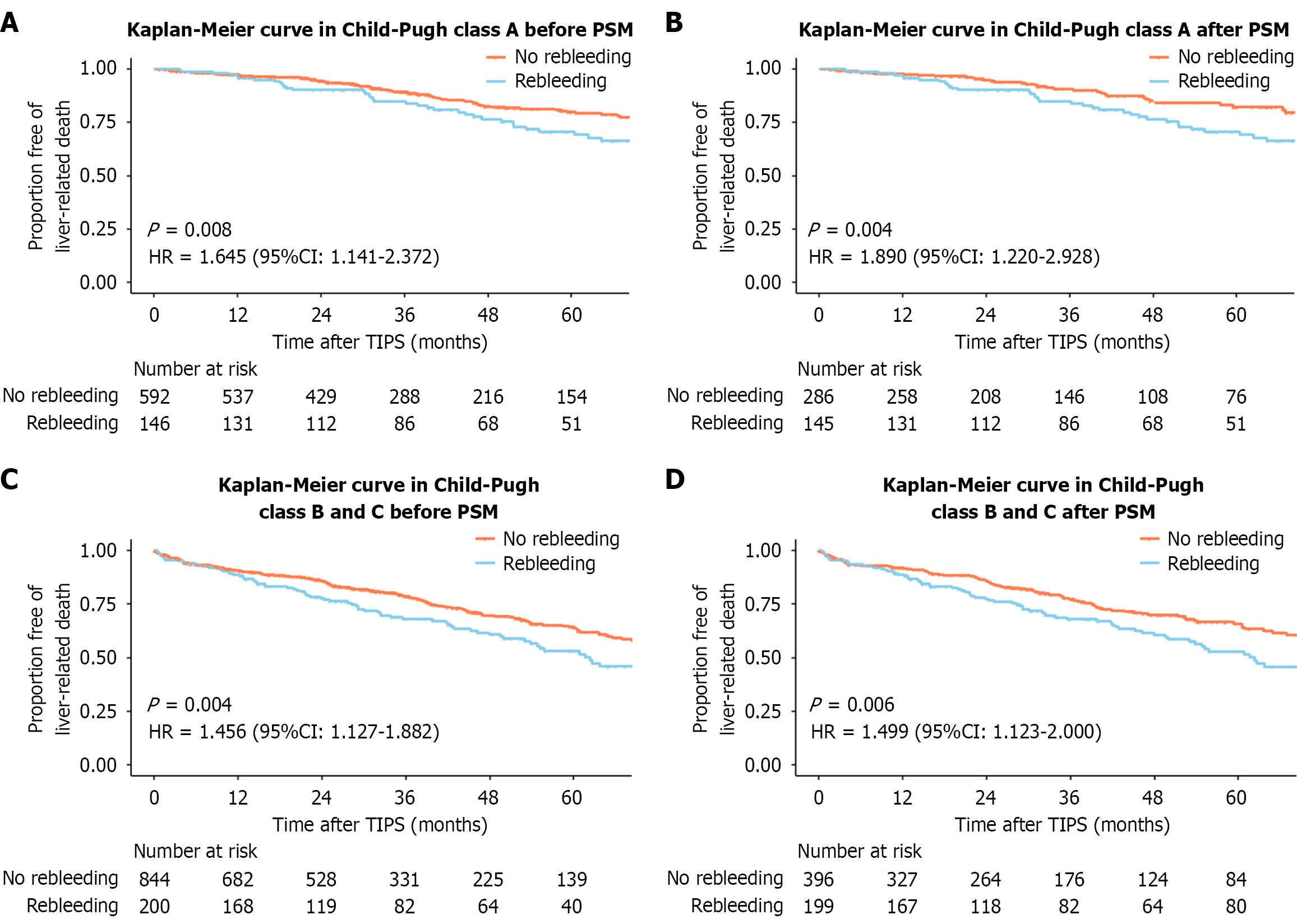
Similar results were observed when analyzing patients who underwent TIPS for variceal rebleeding prevention across different liver functions categories (Child-Pugh class A, before PSM: HR = 1.645, 95%CI: 1.141-2.372, P = 0.008; after PSM: HR = 1.890, 95%CI: 1.220-2.928, P = 0.004; Child-Pugh class B and C, before PSM: HR = 1.456, 95%CI: 1.127-1.882, P = 0.004; after PSM: HR = 1.499, 95%CI: 1.123-2.000, P = 0.006; Figure 3).
This multicenter, large-scale cohort study revealed that post-TIPS rebleeding was significantly associated with higher mortality. Multivariable analysis identified post-TIPS rebleeding as an independent predictive factor of liver-related death. The same direction of the effect was consistently observed in a series of sensitivity analyses. Similar results were obtained by separately analyzing patients with different liver functions who underwent TIPS to prevent variceal re
However, several randomized controlled trials have reported that although TIPS reduces rebleeding in patients with cirrhosis compared with standard treatment, it does not improve survival[17-21]. The discrepancy may be attributed to several factors. First, these studies compared TIPS with standard treatments that significantly reduced rebleeding rates with rates ranging from 29% to 66% over 1-3 years. This may dilute the advantage of TIPS in reducing rebleeding, tran
In the real-world clinical setting, the effects of post-TIPS rebleeding on liver-related mortality can be attributed to several interrelated mechanisms. First, massive acute blood loss can lead to hypovolemic shock. Second, post-TIPS rebleeding usually indicates ineffective control of portal hypertension or the presence of other serious complications such as stent dysfunction and liver function deterioration. Shunt dysfunction is a known risk factor for rebleeding after TIPS[24], but its incidence is usually low within the first year[25]. This may be attributed to rigorous post-procedural monitoring and high short-term patient adherence following the TIPS procedure[26]. However, disease progression often leads to a gradual decline in treatment compliance, financial resources, and access to care opportunities, resulting in an increasing rate of shunt dysfunction[27,28]. However, since the progression of the disease and the decline in patient compliance were not directly verified by our study data, the causal relationship between the two still needs to be evaluated through prospective studies. Moreover, in the late stage of decompensated liver cirrhosis, liver function, systemic inflammation response, and overall health of the patient also deteriorates[29]. Bleeding may trigger a systemic inflammatory response, exacerbate liver damage, and cause acute-on-chronic liver failure. After bleeding, the patient’s general condition worsens, and immunity is impaired, thereby significantly increasing the risk of fatal infection.
Interestingly, post-TIPS rebleeding was associated with increased liver-related mortality regardless of whether the patients were classified as Child-Pugh class A or B/C without differences due to different liver functions. The findings appear contradictory as patients with lower Child-Pugh grades are generally believed to have better liver reserve function and higher tolerance to rebleeding; therefore, rebleeding would be expected to have a less severe impact on liver-related mortality in these individuals. Although TIPS reduces the risk of bleeding by lowering portal pressure, the associated reduction in intrahepatic blood flow may exacerbate liver function damage[30]. Even among patients classified as Child-Pugh class A, TIPS postoperative liver function may also decrease, making them close to the fragile state of Child-Pugh class B/C patients during rebleeding. Furthermore, the Child-Pugh class does not account for portal hemodynamic parameters such as PPG and portal velocity, potentially leading to underestimation of the actual risk in some patients with Child-Pugh class A. Regardless of baseline liver function, acute variceal bleeding as an acute and severe clinical event may push patients with different Child-Pugh classes to a similar risk of death through mechanisms such as direct hemodynamic disorders, systemic inflammatory responses, and multiple organ failure[31].
Consequently, clinical management following TIPS should include a rigorous follow-up strategy regardless of baseline Child-Pugh classification. This may involve regular endoscopy, imaging evaluation, and hemodynamic monitoring to minimize the risk of rebleeding and improve overall prognosis. Specifically, the patency of the stent should be assessed using Doppler ultrasound or CT at 1 month, 3 months, 6 months, and annually. If an abnormality is detected during the examination, early reintervention should be performed. Meanwhile, the follow-up intensity should be stratified based on individual risks with closer monitoring for patients who experience rebleeding events early after the procedure.
This study had several advantages. First, as a large-sample, multicenter, retrospective cohort study, it included 1782 patients with cirrhosis and variceal bleeding who underwent TIPS treatment, providing sufficient statistical power to detect associations between rebleeding and liver-related mortality and to enable critical subgroup analyses. More importantly, this study was the first to systematically stratify patients based on baseline liver function (the Child-Pugh classification) in a large real-world cohort to evaluate rebleeding after TIPS as a prognostic indicator of liver-related mortality.
However, this study had some limitations. First, some biases may have persisted due to the observational nature of this study. However, enrolling consecutive patients helped minimize the bias. Second, the study population, predominantly Asian patients with chronic HBV infection, may limit the generalizability of our findings to other liver disease etiologies and populations. Specifically, compared with Western cohorts primarily characterized by alcoholic or metabolic fatty liver disease, patients with HBV-related cirrhosis may exhibit differences in the progression of portal hypertension and the risk of hepatocellular carcinoma. Additionally, differences may exist between Asia and the West regarding the intensity of monitoring after TIPS, accessibility of secondary interventions, and criteria for liver transplantation. These differences may have subtly influenced the strength of the association between rebleeding events and mortality. Furthermore, this study analyzed rebleeding as a fixed covariable and failed to fully account for its nature as a time-varying exposure. Future prospective studies should employ time-dependent Cox models for validation to more accu
Moreover, postoperative variables such as liver and renal function were not captured, particularly the inability to fully monitor dynamic changes in liver function due to the potential residual confounding issues in observational designs. Although we used rigorous statistical models and adjusted baseline liver function indicators, including the Child-Pugh score, these indicators are static. Clinically, before a rebleeding event occurs, patients may have already experienced unrecorded acute liver function deterioration or accelerated progression of chronic liver disease. This unmeasured liver function deterioration is likely a driving factor: It both triggers rebleeding and directly increases the risk of liver-related mortality. To overcome this limitation and validate the inferences of this study, future research should focus on continuous dynamic monitoring of patients’ liver function, whose indicators including Child-Pugh and MELD-Na scores at fixed intervals after the TIPS procedure, such as every 3 months, using time-varying covariable Cox models and causal mediation analysis to quantify the extent to which dynamic worsening of liver function mediates the association between rebleeding and liver-related death. Lastly, patients who underwent preemptive TIPS were not included although it is associated with reduced mortality and rebleeding rates[32].
Our study revealed that post-TIPS rebleeding was significantly associated with higher mortality in patients with cirrhosis and variceal bleeding, regardless of liver function category. Given the retrospective nature of this study, our results need to be validated in future prospective studies with larger cohorts.
| 1. | Lv Y, Fan D, Han G. Transjugular intrahepatic portosystemic shunt for portal hypertension: 30 years experience from China. Liver Int. 2023;43:18-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 2. | Tripathi D, Stanley AJ, Hayes PC, Travis S, Armstrong MJ, Tsochatzis EA, Rowe IA, Roslund N, Ireland H, Lomax M, Leithead JA, Mehrzad H, Aspinall RJ, McDonagh J, Patch D. Transjugular intrahepatic portosystemic stent-shunt in the management of portal hypertension. Gut. 2020;69:1173-1192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 272] [Cited by in RCA: 247] [Article Influence: 41.2] [Reference Citation Analysis (0)] |
| 3. | Kaplan DE, Ripoll C, Thiele M, Fortune BE, Simonetto DA, Garcia-Tsao G, Bosch J. AASLD Practice Guidance on risk stratification and management of portal hypertension and varices in cirrhosis. Hepatology. 2024;79:1180-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 288] [Article Influence: 144.0] [Reference Citation Analysis (20)] |
| 4. | de Franchis R, Bosch J, Garcia-Tsao G, Reiberger T, Ripoll C; Baveno VII Faculty. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76:959-974. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2244] [Cited by in RCA: 2065] [Article Influence: 516.3] [Reference Citation Analysis (15)] |
| 5. | Hu K, Sedki M, Kwong A, Kesselman A, Kolli KP, Morelli G, Spengler E, Said A, Lai J, Desai A, Paul S, Frenette C, Fallon M, German M, Verna E, Boike J, Gregory D, Thornburg B, VanWagner L, Goel A; Advancing Liver Therapeutic Approaches (ALTA) Study Group. Portal Hypertensive Gastropathy and MELD-Na Score Predict Recurrent Gastrointestinal Bleeding After TIPSS: An ALTA Group Study. Aliment Pharmacol Ther. 2025;61:346-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 6. | Gaba RC, Bui JT, Cotler SJ, Kallwitz ER, Mengin OT, Martinez BK, Berkes JL, Carrillo TC, Knuttinen MG, Owens CA. Rebleeding rates following TIPS for variceal hemorrhage in the Viatorr era: TIPS alone versus TIPS with variceal embolization. Hepatol Int. 2010;4:749-756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 49] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | Xia Y, Tie J, Wang G, Zhuge Y, Wu H, Zhu X, Xue H, Xu J, Zhang F, Zhao L, Huang G, Zhang M, Wei B, Li P, Wang Z, Wu W, Chen C, Yang S, Han Y, Tang C, Zhang C. Small Transjugular Intrahepatic Portosystemic Shunt Plus Variceal Embolization for Gastric Varices: A Multicenter Cohort Study. Clin Gastroenterol Hepatol. 2025;23:965-977.e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 8. | Seo YS. Prevention and management of gastroesophageal varices. Clin Mol Hepatol. 2018;24:20-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 9. | Larrue H, D'Amico G, Olivas P, Lv Y, Bucsics T, Rudler M, Sauerbruch T, Hernandez-Gea V, Han G, Reiberger T, Thabut D, Vinel JP, Péron JM, García-Pagán JC, Bureau C. TIPS prevents further decompensation and improves survival in patients with cirrhosis and portal hypertension in an individual patient data meta-analysis. J Hepatol. 2023;79:692-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 83] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 10. | Kamath PS, Kim WR; Advanced Liver Disease Study Group. The model for end-stage liver disease (MELD). Hepatology. 2007;45:797-805. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1400] [Cited by in RCA: 1240] [Article Influence: 65.3] [Reference Citation Analysis (5)] |
| 11. | Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, ter Borg PC. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology. 2000;31:864-871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2385] [Cited by in RCA: 2092] [Article Influence: 80.5] [Reference Citation Analysis (5)] |
| 12. | Bettinger D, Sturm L, Pfaff L, Hahn F, Kloeckner R, Volkwein L, Praktiknjo M, Lv Y, Han G, Huber JP, Boettler T, Reincke M, Klinger C, Caca K, Heinzow H, Seifert LL, Weiss KH, Rupp C, Piecha F, Kluwe J, Zipprich A, Luxenburger H, Neumann-Haefelin C, Schmidt A, Jansen C, Meyer C, Uschner FE, Brol MJ, Trebicka J, Rössle M, Thimme R, Schultheiss M. Refining prediction of survival after TIPS with the novel Freiburg index of post-TIPS survival. J Hepatol. 2021;74:1362-1372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 133] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 13. | Vizzutti F, Celsa C, Calvaruso V, Enea M, Battaglia S, Turco L, Senzolo M, Nardelli S, Miraglia R, Roccarina D, Campani C, Saltini D, Caporali C, Indulti F, Gitto S, Zanetto A, Di Maria G, Bianchini M, Pecchini M, Aspite S, Di Bonaventura C, Citone M, Guasconi T, Di Benedetto F, Arena U, Fanelli F, Maruzzelli L, Riggio O, Burra P, Colecchia A, Villa E, Marra F, Cammà C, Schepis F. Mortality after transjugular intrahepatic portosystemic shunt in older adult patients with cirrhosis: A validated prediction model. Hepatology. 2023;77:476-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 14. | de Franchis R; Baveno VI Faculty. Expanding consensus in portal hypertension: Report of the Baveno VI Consensus Workshop: Stratifying risk and individualizing care for portal hypertension. J Hepatol. 2015;63:743-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2652] [Cited by in RCA: 2374] [Article Influence: 215.8] [Reference Citation Analysis (2)] |
| 15. | Patidar KR, Bajaj JS. Covert and Overt Hepatic Encephalopathy: Diagnosis and Management. Clin Gastroenterol Hepatol. 2015;13:2048-2061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 195] [Cited by in RCA: 166] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 16. | Matthay EC, Hagan E, Gottlieb LM, Tan ML, Vlahov D, Adler N, Glymour MM. Powering population health research: Considerations for plausible and actionable effect sizes. SSM Popul Health. 2021;14:100789. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 17. | Pomier-Layrargues G, Villeneuve JP, Deschênes M, Bui B, Perreault P, Fenyves D, Willems B, Marleau D, Bilodeau M, Lafortune M, Dufresne MP. Transjugular intrahepatic portosystemic shunt (TIPS) versus endoscopic variceal ligation in the prevention of variceal rebleeding in patients with cirrhosis: a randomised trial. Gut. 2001;48:390-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 99] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Holster IL, Tjwa ET, Moelker A, Wils A, Hansen BE, Vermeijden JR, Scholten P, van Hoek B, Nicolai JJ, Kuipers EJ, Pattynama PM, van Buuren HR. Covered transjugular intrahepatic portosystemic shunt versus endoscopic therapy + β-blocker for prevention of variceal rebleeding. Hepatology. 2016;63:581-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 199] [Cited by in RCA: 176] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 19. | Sauerbruch T, Mengel M, Dollinger M, Zipprich A, Rössle M, Panther E, Wiest R, Caca K, Hoffmeister A, Lutz H, Schoo R, Lorenzen H, Trebicka J, Appenrodt B, Schepke M, Fimmers R; German Study Group for Prophylaxis of Variceal Rebleeding. Prevention of Rebleeding From Esophageal Varices in Patients With Cirrhosis Receiving Small-Diameter Stents Versus Hemodynamically Controlled Medical Therapy. Gastroenterology. 2015;149:660-8.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 194] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 20. | Luo X, Wang Z, Tsauo J, Zhou B, Zhang H, Li X. Advanced Cirrhosis Combined with Portal Vein Thrombosis: A Randomized Trial of TIPS versus Endoscopic Band Ligation Plus Propranolol for the Prevention of Recurrent Esophageal Variceal Bleeding. Radiology. 2015;276:286-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 90] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 21. | Lv Y, Qi X, He C, Wang Z, Yin Z, Niu J, Guo W, Bai W, Zhang H, Xie H, Yao L, Wang J, Li T, Wang Q, Chen H, Liu H, Wang E, Xia D, Luo B, Li X, Yuan J, Han N, Zhu Y, Xia J, Cai H, Yang Z, Wu K, Fan D, Han G; PVT-TIPS Study Group. Covered TIPS versus endoscopic band ligation plus propranolol for the prevention of variceal rebleeding in cirrhotic patients with portal vein thrombosis: a randomised controlled trial. Gut. 2018;67:2156-2168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 153] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 22. | Xiang Y, Tie J, Wang G, Zhuge Y, Wu H, Zhu X, Xue H, Liu S, Yang L, Xu J, Zhang F, Zhang M, Wei B, Li P, Wang Z, Wu W, Chen C, Yang S, Han Y, Tang C, Qi X, Zhang C. Post-TIPS Overt Hepatic Encephalopathy Increases Long-Term but Not Short-Term Mortality in Cirrhotic Patients With Variceal Bleeding: A Large-Scale, Multicenter Real-World Study. Aliment Pharmacol Ther. 2025;61:1183-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 23. | Kabelitz MA, Sandmann L, Praktiknjo M, Mauz JB, Auer TA, Bruns T, Chang J, Engelmann C, Fehrenbach U, Hinrichs J, Jansen C, Kloeckner R, Kluwe J, Köhler M, Meyer C, Piecha F, Pollmanns MR, Ripoll C, Schultheiss M, Seifert LL, Stöhr F, Sturm L, Trebicka J, Zipprich A, Labenz C, Bettinger D, Maasoumy B; German Cirrhosis Study Group. Early Occurrence of Hepatic Encephalopathy Following Transjugular Intrahepatic Portosystemic Shunt Insertion is Linked to Impaired Survival: A Multicenter Cohort Study. Clin Gastroenterol Hepatol. 2025;23:2214-2223.e27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 24. | Kim HK, Kim YJ, Chung WJ, Kim SS, Shim JJ, Choi MS, Kim DY, Jun DW, Um SH, Park SJ, Woo HY, Jung YK, Baik SK, Kim MY, Park SY, Lee JM, Kim YS. Clinical outcomes of transjugular intrahepatic portosystemic shunt for portal hypertension: Korean multicenter real-practice data. Clin Mol Hepatol. 2014;20:18-27. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Luo X, Zhao M, Wang X, Jiang M, Yu J, Li X, Yang L. Long-term patency and clinical outcome of the transjugular intrahepatic portosystemic shunt using the expanded polytetrafluoroethylene stent-graft. PLoS One. 2019;14:e0212658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 26. | DiMatteo MR. Variations in patients' adherence to medical recommendations: a quantitative review of 50 years of research. Med Care. 2004;42:200-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1662] [Cited by in RCA: 1437] [Article Influence: 65.3] [Reference Citation Analysis (0)] |
| 27. | Bernardi M, Caraceni P. Novel perspectives in the management of decompensated cirrhosis. Nat Rev Gastroenterol Hepatol. 2018;15:753-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 64] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 28. | Moscrop A, Ziebland S, Roberts N, Papanikitas A. A systematic review of reasons for and against asking patients about their socioeconomic contexts. Int J Equity Health. 2019;18:112. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 38] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 29. | Engelmann C, Clària J, Szabo G, Bosch J, Bernardi M. Pathophysiology of decompensated cirrhosis: Portal hypertension, circulatory dysfunction, inflammation, metabolism and mitochondrial dysfunction. J Hepatol. 2021;75 Suppl 1:S49-S66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 293] [Article Influence: 58.6] [Reference Citation Analysis (2)] |
| 30. | Casadaban LC, Parvinian A, Couture PM, Minocha J, Knuttinen MG, Bui JT, Gaba RC. Characterization of liver function parameter alterations after transjugular intrahepatic portosystemic shunt creation and association with early mortality. AJR Am J Roentgenol. 2014;203:1363-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 31. | Ibrahim M, Mostafa I, Devière J. New Developments in Managing Variceal Bleeding. Gastroenterology. 2018;154:1964-1969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 32. | Lv Y, Zuo L, Zhu X, Zhao J, Xue H, Jiang Z, Zhuge Y, Zhang C, Sun J, Ding P, Ren W, Li Y, Zhang K, Zhang W, He C, Zhong J, Peng Q, Ma F, Luo J, Zhang M, Wang G, Sun M, Dong J, Bai W, Guo W, Wang Q, Yuan X, Wang Z, Yu T, Luo B, Li X, Yuan J, Han N, Zhu Y, Niu J, Li K, Yin Z, Nie Y, Fan D, Han G. Identifying optimal candidates for early TIPS among patients with cirrhosis and acute variceal bleeding: a multicentre observational study. Gut. 2019;68:1297-1310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 111] [Article Influence: 15.9] [Reference Citation Analysis (0)] |