Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.117443
Revised: January 16, 2026
Accepted: February 13, 2026
Published online: April 28, 2026
Processing time: 131 Days and 8.3 Hours
Ulcerative colitis (UC) is a chronic non-specific inflammatory bowel disease cha
Core Tip: This review summarizes recent research advances in natural products for ulcerative colitis, systematically analyzes their core bioactive components and potential mechanisms of action, and further identifies the challenges in clinical translation. In the future, it is imperative to clarify their therapeutic targets and conduct standardized clinical studies to facilitate the development of natural products as novel therapeutic agents or adjuvants for ulcerative colitis treatment.
- Citation: Yu X, Zhao CX, Wang C. Research progress in the treatment of ulcerative colitis with natural products. World J Gastroenterol 2026; 32(16): 117443
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/117443.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.117443
Ulcerative colitis (UC) is a lifelong inflammatory bowel disease affecting the rectum and colon. In 2023, the global prevalence of UC was estimated to reach 5 million cases, with a rising incidence worldwide[1]. Historically, UC has long been regarded as a “Western disease”, with an incidence of 300-400 per 100000 individuals in Western populations[2]. However, amid the accelerated industrialization and westernization of dietary patterns, the incidence and prevalence of UC have increased significantly in other regions including Asia, accompanied by a distinct trend toward younger age at onset. Notably, while the incidence of UC in China is lower than that in Western countries, it remains higher than in other developing nations[3,4]. The ongoing acceleration of urbanization and industrialization is expected to exacerbate the burden of UC in developing countries, posing significant challenges to global healthcare systems[5]. Currently, the pathogenesis of UC remains incompletely understood, with multiple factors including genetic susceptibility, immune-inflammatory dysregulation, and alterations in the gut microbiota implicated as contributing causes[6].
No definitive cure exists for UC to date, with the core goal of current clinical management focusing on achieving mu
While 5-aminosalicylates the first-line therapy for mild-to-moderate UC play a crucial role in controlling mild inflammation, long-term administration tends to disrupt gut microbiota homeostasis and induce dysbiosis. Although corticosteroids can rapidly suppress acute inflammatory flares in severe UC, 30%-40% of patients remain non-responsive to this monotherapy[14,15]. The use of immunosuppressants significantly elevates patients’ infection risk, posing a threat to their overall health. As a second-line therapeutic option for UC, emerging biotherapies have achieved therapeutic breakthroughs for some patients unresponsive to conventional treatments; however, their clinical application is limited by high costs attributed to substantial research and development (RD) investments and complex manufacturing processes, which render long-term access unaffordable for most patients and biosafety concerns[7]. For example, a phase III randomized controlled trial has demonstrated that integrin antagonists may be associated with risks such as appendicitis, infusion reactions, and increased susceptibility to infections[16]. Thus, the development of more effective therapeutic approaches for UC has become increasingly imperative. Natural products (NPs), leveraging their advantages of wide availability, multi-target synergistic effects (MTSEs), and low toxicity with minimal side effects, have emerged as a key research direction for UC treatment[17]. Therefore, this review summarizes recent research advances in NPs-based interventions for UC, systematically analyzes their core active components and underlying mechanisms of action, provides an in-depth dissection of the core bottlenecks in basic research, clinical translation, and industrialization, clarifies future development directions, and aims to offer academic references and practical guidance for advancing the innovation of NPs-enabled UC treatment.
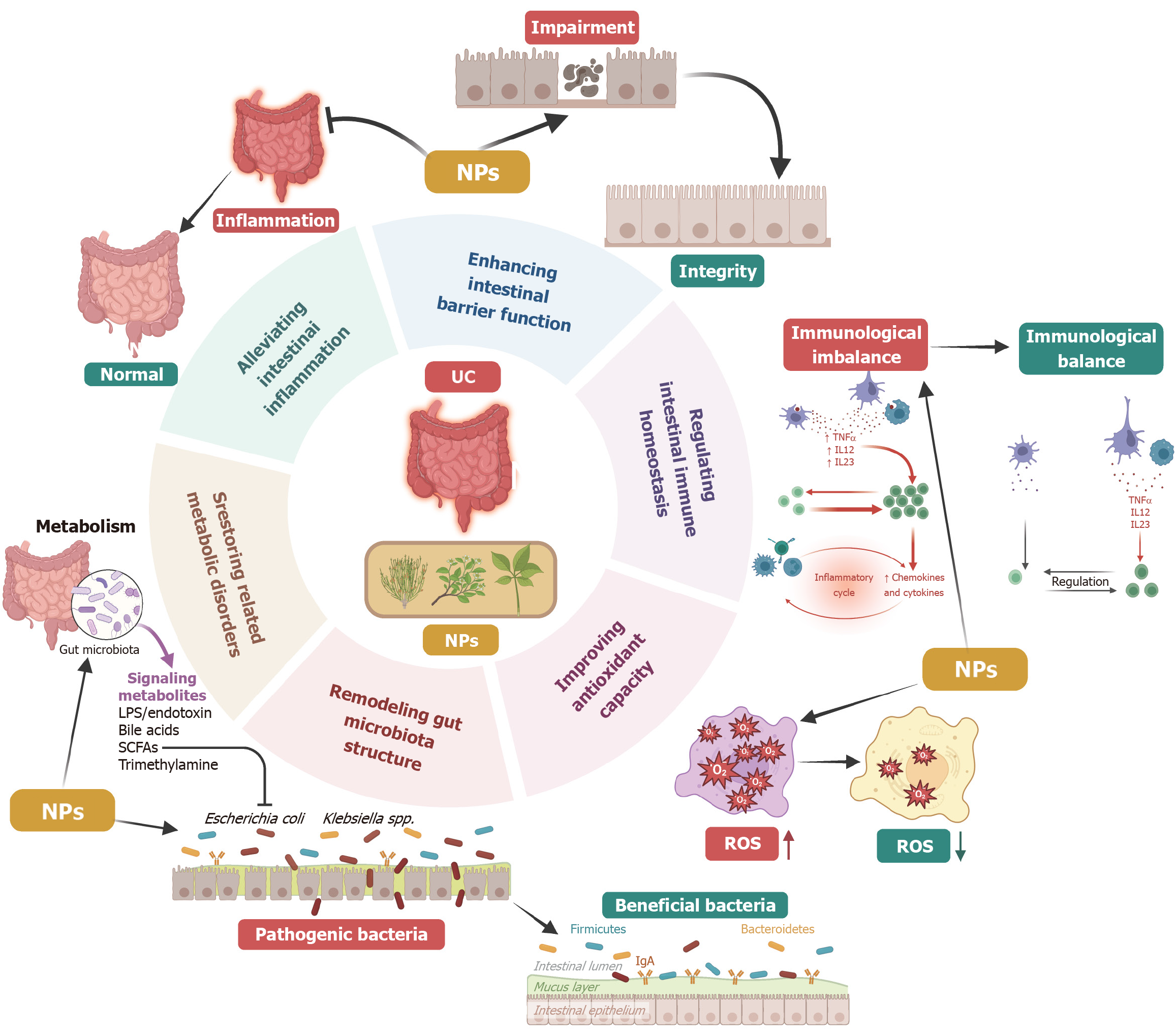
The pathogenesis of UC involves multiple links, including inflammatory activation, mucosal damage, immune imbalance, and oxidative stress[13,18,19]. NPs can act on multiple pathological nodes simultaneously, such as alleviating intestinal inflammation[20], enhancing intestinal barrier function[21], regulating intestinal immune homeostasis[22], improving antioxidant capacity[23], remodeling gut microbiota structure[24], and restoring related metabolic disorders[25], further breaking the vicious cycle of “inflammation-damage-dysbiosis”. NPs intervene in the pathological process of UC through multiple key pathways: (1) Restoring intestinal barrier function: Reversing intestinal barrier damage in the UC state and reestablishing the structural integrity of the mucosa; (2) Regulating immune homeostasis: Correcting immune imbalance and modulating the abnormal expression of immune cells and inflammatory factors (e.g., TNF-α, interleukin-12); (3) Enhancing antioxidant capacity: Reducing UC-related reactive oxygen species levels and alleviating oxidative stress damage; (4) Remodeling gut microbiota structure: Inhibiting the proliferation of pathogenic bacteria (e.g., Escherichia coli, Klebsiella spp.) and promoting the colonization of beneficial bacteria (e.g., Firmicutes, Bacteroidetes); (5) Ameliorating metabolism-related disorders: Regulating gut microbiota-derived metabolic signals [e.g., lipopolysaccharides (LPS), bile acids, short-chain fatty acids (SCFAs)] and correcting metabolic abnormalities; and (6) Alleviating intestinal inflammation: Reversing the excessive inflammatory state of UC to the homeostatic level of a normal intestine, as illustrated in Figure 1. In addition, the active components of NPs are mostly endogenous or homologous substances in organisms, which are highly tolerated by the human body with no significant hepatotoxicity or nephrotoxicity after long-term use[26], aligning with the clinical needs of long-term maintenance therapy for UC.
The World Health Organization reveals that 80% of the population in developing countries worldwide relies on herb-based traditional medicines for better treatment outcomes[27]. Scientific research on these active components and folk remedies, developed by diverse ethnic and cultural groups (i.e., ethnopharmacology), has made extensive contributions to the discovery and refinement of modern emerging therapeutic strategies. Numerous studies have demonstrated that core NPs components including flavonoids (containing flavonoid glycosides)[28], polyphenols (containing phenolic glycosides)[29], polysaccharides (containing glycosides)[30], alkaloids[31], and terpenoids[32] exert anti-UC effects by regulating multiple nodes, providing a solid theoretical basis for clinical application. Highly active monomers screened from medicinal plants and characteristic microorganisms (e.g., curcumin, quercetin derivatives) exhibit superior anti-inflammatory activity compared to conventional drugs. Moreover, clinical studies have confirmed that NPs may improve clinical remission rates and reduce relapse risks in UC patients[33,34].
Crude plant extracts are common forms of intervention for UC in traditional medicine and daily dietary therapy, and their research can directly bridge traditional application experience with modern scientific validation, holding significant translational value. For example, Rehman et al[35] found that crude extracts of Calliandra haematocephala could ameliorate acetic acid-induced UC, confirming the therapeutic potential of crude extracts. Notably, as mixtures containing multiple active components such as flavonoids, polysaccharides, and phenolic acids, crude plant extracts often exert their the
Plant-derived NPs are currently the most well-studied, thus exhibiting prominent potential for clinical application. Quercetin, a representative flavonoid, alleviates UC by regulating the signal transducer and activator of transcription (STAT) 1/peroxisome proliferator-activated receptor γ balance and determining the alternative activation of mac
| Name | Source | Related mechanisms | Experimental models | Ref. |
| Nicotiflorin | San-Ye-Qing | Inhibiting the NF-κB pathway and activating the NLRP3 inflammasome to exert anti-inflammatory effects | LPS + BMDMs | Ruan et al[39] |
| Berberine | Coptis chinensis Franch. | Inhibiting the PI3K/AKT/mTOR pathway by targeting IRGM1 | 2.0% DSS induced UC in C57BL/6 J mice | Meng et al[40] |
| Epiberberine | Coptidis Rhizoma | Activating intestinal FXR, regulating BAs metabolism/FGF15, and relieving intestinal BAs accumulation/inflammation | 3% DSS induced UC in C57BL/6 J mice | Chen et al[41] |
| Anemoside B4 | P. chinensis | Enhancing intestinal epithelial barrier via regulating MLCK-pMLC2 signaling | 3% DSS induced UC in C57BL/6 J mice | Feng et al[42] |
| Paeoniflorin | Paeonia emodi, Paeonia obovata | Targeting CDC42 inhibits the CDC42/JNK signaling pathway | 4.0% DSS induced UC in SD rats | Hu et al[43] |
| Benzoylpaeo-niflorin | Baishao | Suppressing ferroptosis by regulating Nrf2/SLC7A11/GPX4 signaling pathway | 3.5% DSS induced UC in C57BL/6 J mice | Tang et al[44] |
| Formononetin | Astragalus membranaceus | Regulating gut microbiota and M1/M2 macrophage polarization balance | 2.5% DSS induced UC in C57BL/6 J mice | Xiao et al[45] |
| Anemoside B4 | P. chinensis | Regulating Lactobacillus abundance, promoting SCFA metabolism/butyrate production, and activating the AhR pathway to inhibit ROS/NLRP3 inflammasome activation | 3% DSS induced UC in C57BL/6 J mice | Wu et al[46] |
| Gastrodin | Gastrodia elata | Promoting gut microbiota-derived tryptophan metabolite production and inhibiting inflammation via the AhR/NLRP3 pathway | 2.5% DSS induced UC in C57BL/6 J mice | Zhang et al[47] |
| Plantamajoside | Plantago | Regulating gut microbiota, enhancing intestinal barrier, increasing cystathionine β-synthase expression, and inhibiting the NF-κB pathway | 2.5% DSS induced UC in C57BL/6 J mice | Jia et al[48] |
| Morin | Moraceae family | Inhibiting MAPK/NF-κB pathways, regulating gut microbiota | 2.5% DSS induced UC in C57BL/6 J mice | Qiu et al[49] |
| Caffeic acid | Coffee, Argan | Alleviating intestinal injury by inhibiting the Toll/Imd signaling pathways and suppressing mitochondria damage-mediated apoptosis | 4% DSS induced UC in drosophila | Xiu et al[50] |
| Curcumin, tryptophan | Turmeric | Restoring intestinal barrier function, reducing oxidative stress and inflammation, and regulating gut microbiota homeostasis | 2.5% DSS induced UC in BALB/c mice | Jiang et al[51] |
| Vanillic acid | Vanilla bean | Inhibiting arachidonic acid metabolic pathway and promoting macrophage polarization from M1 to M2 phenotype | 2.5% DSS induced UC in C57BL/6 J mice | Zhao et al[52] |
| Gastrodin | Gastrodia elata Blume | Enriching the Akkermansia mucinniphila population | 2% DSS induced UC in BALB/c mice | Zhang et al[53] |
| Vanillic acid | Angelica sinensis | Regulating oxidative stress, inflammatory responses, macrophage polarization, and the AMPK signaling pathway | 2.5% DSS induced UC in BALB/c mice | Ma et al[54] |
| Theabrownin | Fu Brick Tea | Enhancing antioxidant and anti-inflammatory capacities, regulating gut microbiota, and participating in the AhR pathway | 1.5% DSS induced UC in C57BL/6 J mice | Yang et al[55] |
| Butylchloro-genate | Chaenomeles speciosa | Inhibiting the activation of the NLRP3 inflammasome | 4% DSS induced UC in C57BL/6 J mice | Huang et al[56] |
| Quercetin | Vegetables and Fruits | Regulating the gut microbiota-isovanillic acid-intestinal barrier axis | 3% DSS induced UC in BALB/c mice | Lei et al[57] |
| Proanthocyanidins | Ephedra sinica Stapf | Regulating gut microbiota and restoring impaired serum tryptophan and glycerophospholipid metabolism | 4% DSS induced UC in KM mice | Lv et al[58] |
| TCP80-1 | Tupistra chinensis Baker | Enhancing the intestinal barrier and promoting the proliferation and differentiation of intestinal stem cells into intestinal epithelial cells | 4% DSS induced UC in drosophila | Wang et al[59] |
| Kaji-ichigoside F1 | Rosa roxburghii Trat | Regulating gut microbiota and metabolism, and inhibiting the PI3K/AKT pro-inflammatory signaling pathway | 3% DSS induced UC in C57BL/6 J mice | Liu et al[60] |
| Fraxin | Cortex Fraxini | Inhibiting ROS production, reducing pro-inflammatory cytokines, and regulating the TLR4/NF-κB and MAPK signaling pathways | 2.5% DSS induced UC in C57BL/6 J mice | Sun et al[61] |
| Isosinensetin | Pericarpium Citri Reticulatae | Inhibiting the PI3K/AKT signaling pathway, alleviating oxidative stress and inflammatory responses, and enhancing barrier function | 3% DSS induced UC in C57BL/6 J mice | Li et al[62] |
| Homogalacturonan | Passiflora edulis f. flavicarpa | Enhancing MUC-2 expression and promoting epithelial barrier restoration | 5% DSS induced UC in Swiss mice | Mazeti et al[63] |
| Bergenin | Ardisia japonica | Reducing Bacteroides vulgatus abundance to regulate branched-chain amino acid metabolism, thereby inhibiting TLR4 and modulating the phosphorylation of NF-κB and mTOR | 2.5% DSS induced UC in C57BL/6 J mice | Huang et al[64] |
| Theabrownin | Pu-erh tea | Increasing Akkermansia abundance and intestinal epithelial barrier function, regulating CD4+ T cell differentiation and Treg/Th17 balance, and inhibiting TLR2/4-mediated MyD88-dependent NF-κB, MAPK, and AKT signaling pathways | 5% DSS induced UC in Swiss mice | Zhao et al[65] |
| Gypenosides | Gynostemma pentaphyllum | Regulating the tricarboxylic acid cycle and glutamine metabolism, modulating stem cell bioactivity, and promoting the repair of damaged mucosa | 3% DSS induced UC in C57BL/6 J mice | Yang et al[66] |
| Glycyrrhizic acid, patchouli alcohol | Huoxiang Zhengqi | Modulating 11β-HSD1-mediated endogenous corticosterone metabolism to exert anti-inflammatory effects | 2% DSS induced UC in C57BL/6 J mice | Wang et al[67] |
| Neohesperidin | Brassicaceae family plants | Inhibiting the MAPK/NF-κB inflammatory signaling pathway, maintaining intestinal barrier integrity, and regulating gut microbiota | 2.5% DSS induced UC in C57BL/6 J mice | Ju et al[68] |
| Linderanine C | Lindera aggregate | Inhibiting the MAPK signaling pathway and macrophage M1 polarization | 2.5% DSS induced UC in C57BL/6 J mice | Lan et al[69] |
| Loganic acid | Swertia cincta | Inhibiting TLR4/NF-κB-mediated inflammation and activating the SIRT1/Nrf2 antioxidant response | 2.5% DSS induced UC in BALB/c mice | Prakash et al[70] |
| Pulchinenoside B4 | Bai-Tou-weng-Tang | Targeting the CD1d-mediated AKT-STAT1-PRDX1-NF-κB signaling pathway and inhibiting NLRP3 inflammasome activation | 3% DSS induced UC in C57BL/6 J mice | Li et al[71] |
| Rhoifolin | Citrus grandis | Activating CEMIP/SLC7A11-mediated cystine uptake and inhibiting epithelial ferroptosis | 2.5% DSS induced UC in C57BL/6 J mice | Liu et al[72] |
| Berberine | Coptis chinensis Franch. | Modulating the bile acid/S1PR2/RhoA/ROCK pathway to restore intestinal epithelial barrier function | 2.5% DSS induced UC in C57BL/6 J mice | Yu et al[73] |
| Gypenoside LXXV | Gynostemma pentaphyllum | Targeting the glucocorticoid receptor to inhibit NF-κB-COX2 signaling and promote M1-to-M2 macrophage polarization | 2.5% DSS induced UC in C57BL/6 J mice | Wu et al[74] |
| Mangiferin | Pueraria tuberosa | Restoring colon length and body weight, and reducing inflammatory responses | Inducing colitis via rectal administration of 2 mL of 4% acetic acid in BALB/c mice | Dharmapuri et al[75] |
| Didymin | Citrus grandis | Regulating gut microbiota and altering metabolites, thereby modulating the STAT3 and NF-κB pathways | 2% DSS induced UC in ICR mice | Chu et al[76] |
| JNUTS013 | Marine fungi | Inhibiting inflammation through inducing NLRP3 protein degradation | 3% DSS induced UC in C57BL/6 J mice | Wang et al[77] |
| Isolinderalactone | Lindera aggregata | Binding to LXRα and upregulating the expression of LXRα target genes such as ABCA1, thereby activating the LXRα pathway and inhibiting macrophage M1 polarization | 2.5% DSS induced UC in C57BL/6 J mice | Huang et al[78] |
| Linderane | Lindera aggregata | Suppressing IL-6/STAT3-mediated Th17 differentiation and apoptosis resistance | 2.5% DSS induced UC in C57BL/6 J mice | Lai et al[79] |
| (-)-Fenchone | Foeniculum vulgare Mill | Enhancing intestinal barrier function, exerting anti-inflammatory and antioxidant effects, and regulating immune function | Inducing colitis via rectal administration of trinitrobenzene sulfonic acid in Wistar rats | Araruna et al[80] |
| Ganoderic acid A | Ganoderma lucidum | Regulating gut microbiota-related tryptophan metabolism (especially 3-IAld), activating the aryl hydrocarbon receptor, and inducing IL-22 production | 2.5% DSS induced UC in C57BL/6 J mice | Kou et al[81] |
| Oxymatrine | Sophora flavescens | Regulating ferroptosis and alleviating intestinal inflammation | 4% DSS induced UC in ICR mice | Gao et al[82] |
| Diosmin | Citrus grandis | Inhibiting PANoptosis, regulating gut microbiota and metabolites | 3% DSS induced UC in C57BL/6 J mice | Tan et al[83] |
| Naringenin | Citrus grandis | Activating the Nrf2 signaling pathway to exert antioxidant effects | 3.5% DSS induced UC in BALB/c mice | Li et al[84] |
| Dihydromyricetin | Ampelopsis grossedentata | Inhibiting the formation of neutrophil extracellular traps and regulating the HIF-1α/VEGFA signaling pathway | 3.5% DSS induced UC in C57BL/6 J mice | Ma et al[85] |
| Homoplantaginin | Salvia plebeia R. Brown | Regulating the MMP9-RLN2 signaling axis | 2.5% DSS induced UC in C57BL/6 J mice | Tao et al[86] |
| Apigenin | Citrus grandis | Regulating mast cell degranulation via the PAMP-MRGPRX2 feedback loop | 5% DSS induced UC in C57BL/6 J mice | Huang et al[87] |
Microbial-derived NPs represent an emerging research direction, as they are more conducive to gut microbiota modulation[88]. SCFAs are representative microbial-derived metabolites; for instance, butyrate, a metabolite of pro
Although animal-derived NPs have been less extensively studied to date, their application potential remains to be explored. As a representative animal-derived NPs, propolis is a complex mixture formed by honeybees through blending resin collected from plant buds and exudates with their own secretions. The main bioactive components of propolis include flavonoids, phenolic acids, and terpenoids[91,92], which exhibit diverse biological activities such as antitumor, antiapoptotic, anti-inflammatory, antioxidant, barrier-regulating, and antibacterial effects[93]. Therefore, the therapeutic effects of propolis in alleviating UC are pleiotropic: (1) Liquiritigenin, one of its bioactive components, may alleviate coli
To date, based on existing research on the pathogenesis of UC, the core mechanisms of action of NPs for alleviating UC include regulating inflammation-related signaling pathways, repairing the intestinal mucosal barrier, balancing immune function, and remodeling the gut microbiota. Numerous studies have demonstrated that NPs can exert alleviative and therapeutic effects on UC through multiple signaling pathways[97,98], such as inhibiting the nuclear transcriptional activity of NF-κB and blocking the mitogen-activated protein kinase (MAPK) signaling cascade; regulating the Janus kinase/STAT signaling pathway; targeted inhibition of the phosphatidylinositol 3-kinase/protein kinase B signaling pathway; participating in TLRs- and NOD-like receptor thermal protein domain associated protein 3-related signaling pathways; and activating Nrf2/HO-1-related pathways. The intestinal barrier is a crucial defense mechanism in the gut that effectively protects against internal and external harmful microorganisms, and it plays an increasingly important role in understanding disease progression and developing therapeutic agents. NPs can upregulate the expression of tight junction proteins, reduce intestinal epithelial cell permeability, decrease endotoxin and antigen leakage, while promoting intestinal epithelial cell proliferation and accelerating the healing of damaged mucosa[21,99]. In addition, NPs regulate the balance of intestinal immune function, modulate the activation and differentiation of immune cells (macrophages, T cells, B cells, dendritic cells), reduce the proportion of pro-inflammatory cell subsets, and reconstruct intestinal immune homeostasis[22,100]. Dysbiosis of the gut microbiota induces and promotes the occurrence and progression of UC. After oral administration, NPs sequentially pass through the upper gastrointestinal tract (e.g., stomach, duodenum) to reach the intestinal lumen, where they are gradually metabolized and converted into SCFAs by gut-resident microbiota (including commensal beneficial bacteria such as Bifidobacteria and Lactobacilli) through enzymatic reactions like hydrolysis and fermentation. Meanwhile, NPs promote the proliferation and colonization of beneficial bacteria, inhibit the growth of pathogenic bacteria, enhance gut microbiota diversity, remodel the gut microbiota structure, and improve the gut microecological environment[24,101]. Traditional signaling pathways such as NF-κB and MAPK have long been the focus of mechanistic research on NPs-based therapies for UC. However, current studies targeting these pathways exhibit notable limitations: First, research on the NF-κB/MAPK pathways in the field of NPs-facilitated UC treatment has become increasingly saturated, as most studies merely repeatedly validate the simplistic mechanism that “NPs inhibit NF-κB activation to alleviate inflammation”, without further exploring upstream regulatory nodes or crosstalk with other signaling pathways; second, some studies overinterpret the regulatory effects of NPs on these pathways, lacking suf
In recent years, research on the pathogenesis of UC has expanded to more refined molecular levels. Emerging mechanisms, such as autophagic dysregulation[102], abnormal programmed cell death[103], ferroptosis[104], mitochondrial dysfunction[105], metabolic reprogramming[106], and abnormal post-translational modifications[107], have all been confirmed to be involved in the pathogenesis of UC. For instance, Modified Tou Nong Powder can induce autophagy and improve mitochondrial function by activating the adenosine 5’-monophosphate-activated protein kinase/peroxisome proliferator-activated receptor γ coactivator 1-alpha signaling pathway, thereby exerting a therapeutic effect on alleviating UC in both in vitro and in vivo experiments[108]. Ferroptosis is a novel form of non-apoptotic cell death, characterized primarily by iron homeostasis imbalance and lipid peroxidation triggered by redox system disorder[109]. Studies have confirmed that inhibiting ferroptosis in intestinal epithelial cells can effectively protect the intestinal mucosal barrier and mitigate mucosal damage[110]. Therefore, targeted intervention in the ferroptosis pathway is expected to become a potential novel strategy for the treatment of UC. For example, the ameliorative effect of Scutellaria baicalensis Georgi on UC is closely associated with its regulation of lipid peroxidation and suppression of intestinal epithelial cell ferroptosis mediated by the GPX4/ACSL4 axis[111]. Gancao Xiexin Decoction may alleviate inflammatory damage in UC by regulating the TEAD4/ACSL4 signaling pathway to inhibit ferroptosis in intestinal epithelial cells[112]. However, to date, research on the anti-UC effects of NPs has mostly focused on traditional signaling pathways and gut microbiota modulation, while studies on their targeted effects against these emerging mechanisms remain scarce. Their specific regulatory targets, modes of action, and synergistic effects have not yet been fully elucidated, urgently requiring further investigation. Notably, the regulatory modes of NPs in metabolic reprogramming and their precise regulatory effects on post-translational modifications of proteins (e.g., affecting the modification levels of specific inflammatory proteins) still lack in-depth mechanistic validation and in vivo experimental support. Furthermore, the synergistic regulatory relationships between NPs and emerging mechanisms (e.g., the synergistic effects of simultaneously targeting ferroptosis and mitochondrial dysfunction) as well as the impact of structure-activity relationships on the efficiency of mechanism regulation urgently require supplementation through more basic research. These gaps provide important directions for subsequent innovative research on NPs against UC.
Basic research has clarified the therapeutic effects and underlying mechanisms of NPs from various sources in the treatment of UC, laying a solid foundation for their clinical translation. Moreover, the conduct of clinical studies on traditional Chinese medicine compound preparations and other related areas has further directly promoted the transformation of NPs from laboratory settings to clinical practice, and a clear exploration path has been formed for the verification of their clinical efficacy.
In the exploration of monotherapy, NPs have been primarily targeted at patients with mild to moderately active UC and those intolerant to conventional treatments, with several products demonstrating definitive therapeutic efficacy. For instance, Fufangkushen colon-coated capsules demonstrate non-inferior efficacy and safety compared with mesalazine enteric-coated tablets in the treatment of active UC patients presenting with the traditional Chinese medicine syndrome of damp-heat accumulation, and exert superior therapeutic effects in patients with left hemicolon involvement[113]. Achillea millefolium hydroalcoholic extract capsules can effectively improve the disease activity index and multiple inflammation-related parameters in patients with mild to moderately active UC[114]. Thymus kotschyanus extract is also capable of significantly reducing fecal calprotectin levels and simple clinical colitis activity index scores in patients with mild to moderate UC[115]. Furthermore, indigo naturalis (Qingdai) monotherapy even exhibits advantages in improving clinical response rates and facilitating mucosal healing among treatment-refractory patients, such as those with steroid dependence or a history of anti-TNF-α agent administration[116]. Polyphenon E, in turn, offers a novel therapeutic option for patients with mild to moderately active UC who are refractory to 5-aminosalicylic acid medications and/or aza
Given the limitations of monotherapy, the combined application of NPs with conventional drugs and novel therapeutic approaches has emerged as a research hotspot in clinical practice, which enhances therapeutic efficacy and expands treatment scenarios through synergistic mechanisms. Among these strategies, the combination of NPs with conventional drugs represents the mainstream model, whose core mechanism lies in enhancing anti-inflammatory effects and regulating intestinal flora balance to achieve synergistic efficacy. For example, in a 12-week treatment of moderate active UC patients refractory to 5-aminosalicylic acid, Qing-Chang-Hua-Shi granules combined with 5-aminosalicylic acid yielded significantly higher rates of clinical remission, clinical response, and relief of mucopurulent bloody stool compared with the placebo group, with a comparable incidence of adverse reactions[119]. Systematic reviews and meta-analyses have confirmed that curcumin combined with mesalazine can markedly increase the probabilities of clinical remission, clinical response, and endoscopic response in patients, exhibiting superior efficacy to placebo with mild side effects[120]. As an adjuvant therapy to standard UC treatment, pomegranate peel aqueous extract can reduce the Lichtiger colitis activity index in patients, with a higher clinical response rate than the placebo group at week 4[121]. In addition, the combination of NPs with novel therapeutic methods has shown promising potential. For instance, fecal microbiota transplantation combined with pectin therapy can lower patients’ Mayo scores and make the composition of patients’ intestinal flora more similar to that of donors, which verifies that pectin can improve fecal microbiota transplantation efficacy by maintaining the diversity of transplanted flora[122]. Although in vitro culture experiments using colonic biopsy samples from UC patients have confirmed that oleuropein can decrease Escherichia coli-LPS-induced inflammatory factor expression and alleviate inflammatory damage to colonic tissues[123], laying a foundation for its clinical application and combination therapy exploration. Additionally, a systematic review and meta-analysis sum
NPs often exhibit significant anti-inflammatory activity in vitro, exerting effects by directly inhibiting the release of inflammatory factors and regulating inflammatory signaling pathways. However, they are prone to obvious application bottlenecks after in vivo administration. Following oral administration, their active components are susceptible to degradation and destruction by gastric acid and digestive enzymes in the gastrointestinal tract. Additionally, due to characteristics such as poor water solubility and weak mucosal penetration ability, their in vivo bioavailability is generally less than 20%[125,126]. This imbalance between high in vitro activity and low in vivo bioavailability makes it difficult to effectively translate the therapeutic effects observed in animal experiments into human clinical applications, seriously hindering the clinical translation of NPs. Enterotyping (or bacterial community typing) enables the stratification of patients based on the characteristics of their gut microbiota[127]. Four enterotypes have been identified to date, namely Bacteroides 1, Bacteroides 2, Prevotella, and Ruminococcus[128]. Up to 80% of inflammatory bowel disease patients harbour the Bacteroides 2 enterotype, which is defined as a dysbiotic enterotype; while the prevalence of this enterotype in healthy individuals is less than 15%[128,129]. So significant interindividual variability exists in the therapeutic efficacy of NPs for UC, a phenomenon closely associated with patients’ individual biological characteristics: On one hand, different patients exhibit distinct gut microbiota typing. As gut microbiota serves as both metabolic carriers and therapeutic targets of NPs, differences in its composition directly influence the metabolic conversion efficiency and bioactivity exertion of NPs[130]. On the other hand, variations in patients’ genetic backgrounds (e.g., inflammation-related gene polymorphisms) also lead to differences in therapeutic sensitivity to NPs. These aforementioned factors collectively result in substantial disparities in therapeutic response rates of NPs among UC patients[13]. Currently, there is a lack of precision medication guidance systems based on individual characteristics, making it challenging to formulate personalized treatment regimens.
NPs possess complex and diverse chemical composition systems. Their core active components (e.g., flavonoids, polysaccharides, terpenoids) generally have low contents in raw materials and are significantly influenced by factors such as soil, climate, season, and processing technology of the producing area’s ecological environment, leading to substantial variations in the type and content of components among raw materials from different sources[131-133]. Currently, the separation and purification technologies for active components of NPs still lack a unified standardized process. Inconsistencies in extraction solvents, separation methods, and purification purity standards adopted in different research or production links result in significant batch-to-batch variations in the final active components. This makes it difficult to ensure pharmacodynamic uniformity in subsequent pharmacodynamic evaluations and clinical applications, becoming a key bottleneck restricting the development of NPs. At the level of quality control systems, influenced by the MTSEs of NPs, the existing systems lack exclusive quality control indicators that can fully reflect pharmacodynamic effects. Moreover, current standards only focus on the detection of a single marker component, which can neither cover all pharmacodynamically relevant substances nor reflect the balance of multi-component proportions. Ultimately, this leads to insufficient control over the stability of product quality, affecting the safety and reliability of clinical applications.
Clinical trials of NPs for UC treatment still exhibit significant scale and design limitations. Details of relevant clinical trials over the past three years are presented in Table 2[134-142]. Most conducted studies are small sample sizes, resulting in insufficient representativeness and generalizability of the research findings, which makes it difficult for existing conclusions to fully support the widespread clinical application and promotion of NPs. Some studies have inadequate reporting on key aspects such as randomization methods, allocation concealment, and the implementation of blinding procedures, thus carrying a high risk of selection bias and performance bias. In addition, most studies have a relatively short follow-up duration (usually 4-8 weeks), which makes it difficult to evaluate the long-term efficacy and safety of NPs in the treatment of UC, and also impossible to clarify their potential impact on disease recurrence rates. Moreover, differences in therapeutic efficacy across studies may stem from multiple factors: (1) The included UC patients vary in terms of disease severity, disease duration, and comorbidity status; (2) There is inconsistency in intervention strategies, including the types of NPs used, dosage administered, treatment course, and whether combined with Western medicines, all of which are not unified among different studies; and (3) There is a lack of unified standards for outcome evaluation some studies only adopt clinical symptom scores as efficacy indicators, while others combine endoscopic scores and histological scores, leading to difficulties in directly comparing the results of different studies. Existing indicators cover multiple dimensions, such as clinical symptoms, endoscopic mucosal healing, and histological repair, but significant variations exist among different studies in the selection of core indicators, detection methods, and judgment criteria, leading to difficulties in cross-study comparison of research results. Furthermore, there are obvious gaps in safety data: Safety monitoring for long-term use (> 1 year) and safety assessments in special populations (e.g., elderly, pediatric, and pregnant patients) are relatively scarce, and the risks of potential adverse reactions remain unclear, which further restricts their clinical promotion and application.
| Natural product/active component | Trial design | Sample size | Ref. |
| Anthocyanin-rich extract | Multicenter, randomized, placebo-controlled, double-blind study | 48 | Biedermann et al[134] |
| Hudi enteric-coated capsule, modified Wumei pill, and Qingchang Wenzhong decoction | Multicenter, randomized, double-blind study | 143 | Li et al[135] |
| Zataria multiflora | Multicenter, randomized, placebo-controlled, triple-blind trial | 92 | Morvaridi et al[136] |
| Curcumin, resveratrol | Prospective multicenter three-arm RCT | 48 | Erol Doğan et al[137] |
| Zuoqing San | Single-center, double-blind study | 126 | Li et al[138] |
| 1-kestose | Randomized, double-blind, placebo-controlled pilot trial | 40 | Ikegami et al[139] |
| Curcumin-QingDai | A retrospective multicenter adult cohort study | 88 | Yanai et al[140] |
| Icanbelimod | Single-center, double-blind study | 28 | Lickliter et al[141] |
| Pegmispotide | Double-blind study | 29 | Allegretti et al[142] |
Currently, NPs still face multiple practical obstacles in the industrialization process related to UC treatment: (1) Immature production technology: The extraction and purification of NPs involve complex separation steps, resulting in a long overall production cycle and high costs. This makes large-scale production difficult, directly restricting the market launch and popularization of related products; (2) Insufficient industry-university-research collaboration mechanism: Research institutions mainly focus on laboratory-scale activity verification and mechanism elucidation, while enterprises prioritize industrial feasibility and market demand; the lack of effective connection and cooperation platforms between the two leads to an obvious disconnection between laboratory achievements and industrial production technologies; and (3) Inadequate policy support system: The approval process for natural medicines (including traditional Chinese medicine compounds and NPs derivatives) is relatively complex; currently, there is a lack of exclusive evaluation standards tailored to their multi-component and multi-target characteristics, and most still follow the approval framework for chemical drugs; this results in a lengthy approval cycle and uncertain expectations of return on RD investment, sig
In summary, the application of NPs in UC treatment is hampered by multiple challenges, including inadequate depth in mechanistic research, low in vivo bioavailability, inconsistent quality control, and insufficient clinical translation evidence. These unresolved issues not only restrict the clinical transformation of NPs but also point out clear directions for subsequent research.
In recent years, the in-depth integration of multi-omics technologies and network pharmacology has established a precise and efficient research system. Through the integrative analysis of multi-omics data combined with target prediction in network pharmacology, panoramic correlation analysis of the “component-target-pathway” of NPs can be achieved, rapidly identifying core active components with synergistic regulatory effects and their corresponding key therapeutic targets[143]. Single-cell omics technology, with its unique advantage in resolving cellular heterogeneity, can accurately capture the specific responses of different cell subsets in the UC intestinal microenvironment to NPs, further refining the cellular targeting and action specificity of active components[144]. Ultimately, this will clarify the core regulatory nodes of the MTSEs of NPs and elucidate the mechanisms underlying interindividual response differences.
In addition, precision modeling technology for animal models better mimics the pathophysiological characteristics of human UC, overcoming the species differences between traditional mouse models and clinical patients. It can more authentically reflect the therapeutic effects and mechanisms of NPs in vivo, such as the humanized NSG mouse model[145] and fecal microbiota transplantation mouse model[146]. A novel three-dimensional in vitro model composed of multi-layered gastrointestinal tissue that exhibits functional immune responses under inflammatory conditions, which can accelerate drug development and reduce unnecessary animal experiments by providing reliable permeability and efficacy data for emerging therapies[147]. The combined application of these technologies enables multi-dimensional mechanistic validation from the molecular and cellular levels to the whole-animal level. It not only improves the precision of target and pathway elucidation for NPs but also enhances the reliability and clinical reference value of research findings, providing robust technical support for innovative mechanism research and clinical translation of NPs.
UC is closely associated with primary sclerosing cholangitis (PSC), with approximately 5%-10% of UC patients de
To address the core bottlenecks in the RD and application of NPs-based anti-UC therapies, joint efforts from three dimensions process optimization, quality control, and formulation innovation are required. From the perspective of separation and purification process optimization, innovative technologies such as high hydrostatic pressure, ultrasound, pulsed electric field, and supercritical fluid extraction are increasingly replacing conventional methods[155]. Furthermore, integrating real-time monitoring techniques like near-infrared online detection can enhance the extraction rate and purity of core active components. Simultaneously, traceability of raw material origins and standardized control of harvesting and pretreatment processes help reduce compositional variations among different batches of raw materials. In terms of quality control system construction, integrating multi-component quantitative analysis and fingerprinting technology enables the establishment of full-chain quality standards covering raw material access, critical control points in production, and finished product inspection. This achieves accurate quantification of core pharmacodynamic components and comprehensive characterization of product composition, ensuring product quality stability and uniformity. With regard to formulation innovation and upgrading, continuous RD of colon-targeted and long-acting sustained-release formulations is crucial to improve bioavailability and lesion targeting[156,157]. Additionally, active exploration of novel drug delivery methods such as transdermal absorption and mucosal drug delivery can overcome the limitations of traditional oral administration, expanding the application scope of NPs across different patient populations and disease scenarios[158].
To promote the clinical translation and standardized application of NPs in UC treatment, focused efforts are required in three aspects: First, conduct multi-center, double-blind, randomized controlled trials covering over 10 tertiary hospitals with a sample size of ≥ 500 cases, strictly adhering to good clinical practice guidelines to enhance the credibility and persuasiveness of research evidence; Second, develop exclusive clinical efficacy evaluation guidelines for NPs in UC treatment, unify primary endpoint indicators such as mucosal healing rate and remission maintenance duration, as well as follow-up periods, to achieve cross-study comparability of research results; Third, systematically supplement safety data by conducting long-term (≥ 1 year) safety follow-up and clinical trials in special populations (e.g., elderly, pediatric, and pregnant patients), clarify long-term medication risks and suitability for special populations, and provide solid evidence for safe clinical medication. Finally, predictive biomarkers (e.g., gut microbiota typing, inflammation-related gene polymorphisms) need to be identified to distinguish populations sensitive to NPs monotherapy or adjunct therapy, thereby enhancing precision treatment.
NPs represent a cornerstone of modern medical advancement, with their applications extending from traditional herbal therapies to the discovery of landmark drugs such as morphine and quinine. The mid-20th century was hailed as the “golden age” for the exploration of naturally derived antibiotics, after which the research scope of NPs gradually expanded into other therapeutic fields. According to statistics, a total of 58 NP-related drugs have been approved worldwide from January 2014 to June 2025, including 45 NPs and their derived new chemical entities, as well as 13 NP-derived antibody-drug conjugates[159]. Commonly used clinical immunosuppressants such as the fungus-sourced cyclosporine and mycophenolate, and the streptomycete-derived sirolimus and tacrolimus have been approved for the prevention of organ rejection in transplant recipients[160]. Tazarotene, a bacterially derived agent, has obtained approval from the United States Food and Drug Administration for the treatment of plaque psoriasis, a chronic autoimmune skin disease[161]. For UC, the fungus-sourced drug etrasimod first received approval in the United States in October 2023 for the treatment of moderate-to-severe active UC in adults; subsequently, the European Medicines Agency issued a positive opinion in December 2023, recommending the granting of its marketing authorization[162].
To accelerate the industrialization process and clinical translation of NPs in UC treatment, a full-chain support system needs to be established: First, build industry-university-research collaborative innovation platforms to promote in-depth cooperation between universities, research institutions, and pharmaceutical enterprises, bridging the key connecting links between basic research, technological breakthroughs, and industrial production; Second, optimize industrial production processes, focus on the demands of low-cost and large-scale production, and develop high-efficiency extraction and purification technologies as well as standardized production procedures; Third, improve the policy support system national and government departments should promote the establishment of green channels for natural medicine approval, formulate exclusive evaluation standards tailored to the multi-component and MTSEs of traditional Chinese medicine compounds and NPs derivatives, and provide policy guarantees for the high-quality development of the NPs-based anti-UC industry.
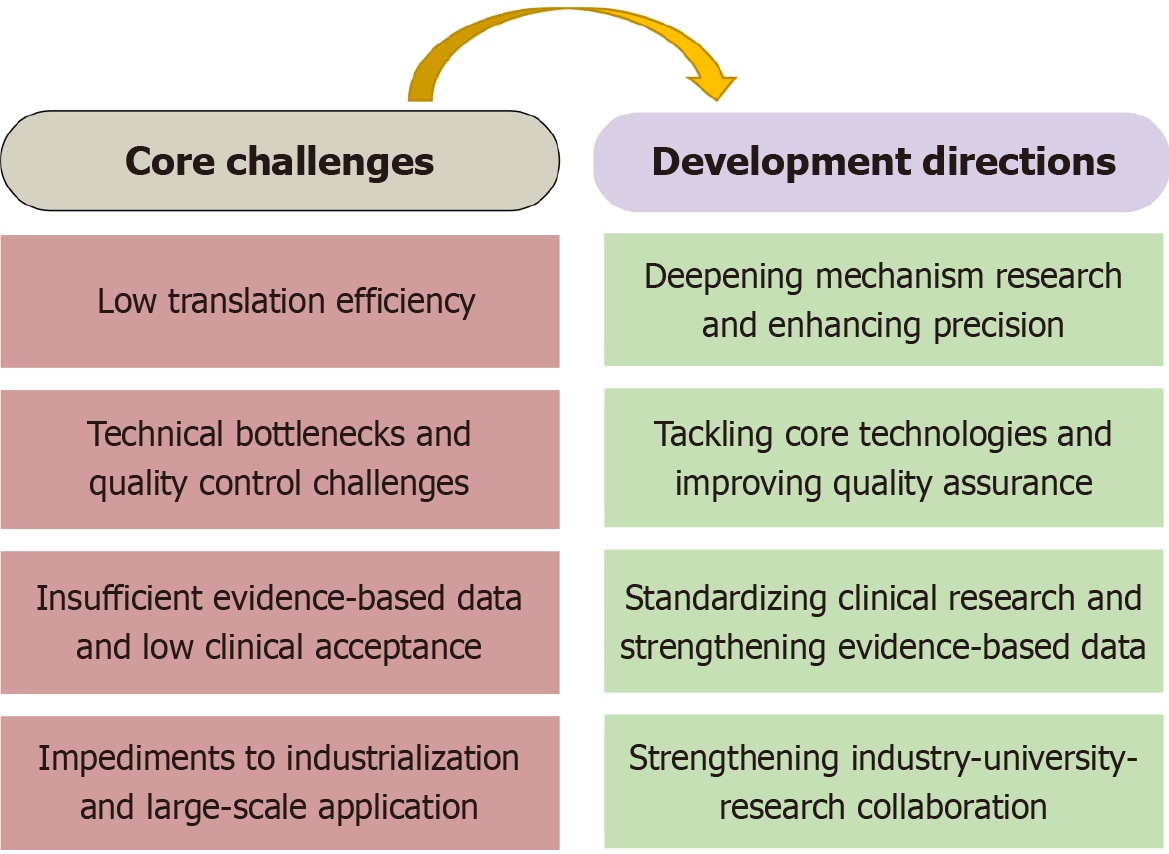
Based on the above content, this paper proposes a problem-oriented matching framework that aligns the core challenges of NPs application in UC treatment with the targeted development directions, as illustrated in Figure 2. The left panel summarizes four key challenges in the current field: Low translation efficiency, technical bottlenecks and quality control difficulties, insufficient evidence-based data coupled with low clinical acceptance, and impediments to industrialization and large-scale application. Corresponding to these challenges (linked by the directional arrow), the right panel puts forward four key development directions: Deepening mechanism research and enhancing precision, tackling core technologies and improving quality assurance, standardizing clinical research and strengthening evidence-based data, and strengthening industry-university-research collaboration. This diagram clarifies the “problem-solution” logical connection in this field, providing a clear guiding framework for subsequent research, technological breakthroughs, and the industrial translation of NPs in UC treatment.
NPs have achieved remarkable progress in UC treatment research due to their advantages of MTSEs, low toxicity, and high efficacy. They provide novel insights for clinical diagnosis and treatment, possessing both significant academic value and broad application potential. However, several challenges remain, including low translation efficiency, prominent technical bottlenecks, and insufficient evidence-based data, which require targeted breakthroughs. Through multi-disciplinary collaborative innovation such as deepening mechanism research, tackling core technologies, standardizing clinical research, and strengthening industry-university-research collaboration NPs can be transformed from laboratory achievements into clinically practical therapeutic approaches, as in Figure 2. This will facilitate the improvement of UC diagnosis and treatment quality, ultimately providing patients with safer and higher-quality treatment options.
| 1. | Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023;402:571-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1251] [Cited by in RCA: 1075] [Article Influence: 358.3] [Reference Citation Analysis (5)] |
| 2. | Gros B, Kaplan GG. Ulcerative Colitis in Adults: A Review. JAMA. 2023;330:951-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 562] [Cited by in RCA: 481] [Article Influence: 160.3] [Reference Citation Analysis (1)] |
| 3. | Wan J, Zhou J, Wang Z, Liu D, Zhang H, Xie S, Wu K. Epidemiology, pathogenesis, diagnosis, and treatment of inflammatory bowel disease: Insights from the past two years. Chin Med J (Engl). 2025;138:763-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 41] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 4. | Wei SC, Sollano J, Hui YT, Yu W, Santos Estrella PV, Llamado LJQ, Koram N. Epidemiology, burden of disease, and unmet needs in the treatment of ulcerative colitis in Asia. Expert Rev Gastroenterol Hepatol. 2021;15:275-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 5. | Buie MJ, Quan J, Windsor JW, Coward S, Hansen TM, King JA, Kotze PG, Gearry RB, Ng SC, Mak JWY, Abreu MT, Rubin DT, Bernstein CN, Banerjee R, Yamamoto-Furusho JK, Panaccione R, Seow CH, Ma C, Underwood FE, Ahuja V, Panaccione N, Shaheen AA, Holroyd-Leduc J, Kaplan GG; Global IBD Visualization of Epidemiology Studies in the 21st Century (GIVES-21) Research Group, Balderramo D, Chong VH, Juliao-Baños F, Dutta U, Simadibrata M, Kaibullayeva J, Sun Y, Hilmi I, Raja Ali RA, Paudel MS, Altuwaijri M, Hartono JL, Wei SC, Limsrivilai J, El Ouali S, Vergara BI, Dao VH, Kelly P, Hodges P, Miao Y, Li M. Global Hospitalization Trends for Crohn's Disease and Ulcerative Colitis in the 21st Century: A Systematic Review With Temporal Analyses. Clin Gastroenterol Hepatol. 2023;21:2211-2221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 175] [Article Influence: 58.3] [Reference Citation Analysis (0)] |
| 6. | Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017;389:1756-1770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3085] [Cited by in RCA: 2810] [Article Influence: 312.2] [Reference Citation Analysis (7)] |
| 7. | Wangchuk P, Yeshi K, Loukas A. Ulcerative colitis: clinical biomarkers, therapeutic targets, and emerging treatments. Trends Pharmacol Sci. 2024;45:892-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 182] [Article Influence: 91.0] [Reference Citation Analysis (0)] |
| 8. | Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. ACG Clinical Guideline: Ulcerative Colitis in Adults. Am J Gastroenterol. 2019;114:384-413. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1361] [Cited by in RCA: 1172] [Article Influence: 167.4] [Reference Citation Analysis (7)] |
| 9. | Burr NE, Gracie DJ, Black CJ, Ford AC. Efficacy of biological therapies and small molecules in moderate to severe ulcerative colitis: systematic review and network meta-analysis. Gut. 2021;gutjnl-2021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 118] [Article Influence: 23.6] [Reference Citation Analysis (1)] |
| 10. | Shehab M, Alrashed F, Alsayegh A, Aldallal U, Ma C, Narula N, Jairath V, Singh S, Bessissow T. Comparative Efficacy of Biologics and Small Molecule in Ulcerative Colitis: A Systematic Review and Network Meta-analysis. Clin Gastroenterol Hepatol. 2025;23:250-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 36] [Article Influence: 36.0] [Reference Citation Analysis (1)] |
| 11. | Khan AU, Ali M, Wahab MA. Comparative efficacy of pharmacologic interventions in ulcerative colitis: a network meta analysis. Inflammopharmacology. 2025;33:2679-2687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Lasa JS, Olivera PA, Danese S, Peyrin-Biroulet L. Efficacy and safety of biologics and small molecule drugs for patients with moderate-to-severe ulcerative colitis: a systematic review and network meta-analysis. Lancet Gastroenterol Hepatol. 2022;7:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 293] [Article Influence: 73.3] [Reference Citation Analysis (5)] |
| 13. | Liang Y, Li Y, Lee C, Yu Z, Chen C, Liang C. Ulcerative colitis: molecular insights and intervention therapy. Mol Biomed. 2024;5:42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 80] [Article Influence: 40.0] [Reference Citation Analysis (0)] |
| 14. | Calméjane L, Laharie D, Kirchgesner J, Uzzan M. Review article: Updated management of acute severe ulcerative colitis: From steroids to novel medical strategies. United European Gastroenterol J. 2023;11:722-732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 42] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 15. | Gisbert JP, García MJ, Chaparro M. Rescue Therapies for Steroid-refractory Acute Severe Ulcerative Colitis: A Review. J Crohns Colitis. 2023;17:972-994. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 16.7] [Reference Citation Analysis (1)] |
| 16. | Peyrin-Biroulet L, Hart A, Bossuyt P, Long M, Allez M, Juillerat P, Armuzzi A, Loftus EV Jr, Ostad-Saffari E, Scalori A, Oh YS, Tole S, Chai A, Pulley J, Lacey S, Sandborn WJ; HICKORY Study Group. Etrolizumab as induction and maintenance therapy for ulcerative colitis in patients previously treated with tumour necrosis factor inhibitors (HICKORY): a phase 3, randomised, controlled trial. Lancet Gastroenterol Hepatol. 2022;7:128-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 68] [Article Influence: 17.0] [Reference Citation Analysis (3)] |
| 17. | Cao F, Liu J, Sha BX, Pan HF. Natural Products: Experimental Efficient Agents for Inflammatory Bowel Disease Therapy. Curr Pharm Des. 2019;25:4893-4913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 18. | Guo M, Wang X. Pathological mechanism and targeted drugs of ulcerative colitis: A review. Medicine (Baltimore). 2023;102:e35020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 19. | Du L, Ha C. Epidemiology and Pathogenesis of Ulcerative Colitis. Gastroenterol Clin North Am. 2020;49:643-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 546] [Cited by in RCA: 458] [Article Influence: 76.3] [Reference Citation Analysis (5)] |
| 20. | Gandhi GR, Mohana T, Athesh K, Hillary VE, Vasconcelos ABS, Farias de Franca MN, Montalvão MM, Ceasar SA, Jothi G, Sridharan G, Gurgel RQ, Xu B. Anti-inflammatory natural products modulate interleukins and their related signaling markers in inflammatory bowel disease: A systematic review. J Pharm Anal. 2023;13:1408-1428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 44] [Reference Citation Analysis (0)] |
| 21. | Peng J, Li H, Olaolu OA, Ibrahim S, Ibrahim S, Wang S. Natural Products: A Dependable Source of Therapeutic Alternatives for Inflammatory Bowel Disease through Regulation of Tight Junctions. Molecules. 2023;28:6293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 22. | Zhao D, Ge A, Yan C, Liu X, Yang K, Yan Y, Hao M, Chen J, Daga P, Dai CC, Li C, Cao H. T helper cell 17/regulatory T cell balance regulates ulcerative colitis and the therapeutic role of natural plant components: a review. Front Med (Lausanne). 2024;11:1502849. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 23. | Li B, Wang Y, Jiang X, Du H, Shi Y, Xiu M, Liu Y, He J. Natural products targeting Nrf2/ARE signaling pathway in the treatment of inflammatory bowel disease. Biomed Pharmacother. 2023;164:114950. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 30] [Reference Citation Analysis (0)] |
| 24. | Zhu M, Song Y, Xu Y, Xu H. Manipulating Microbiota in Inflammatory Bowel Disease Treatment: Clinical and Natural Product Interventions Explored. Int J Mol Sci. 2023;24:11004. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 25. | Yu Z, Li D, Sun H. Herba Origani alleviated DSS-induced ulcerative colitis in mice through remolding gut microbiota to regulate bile acid and short-chain fatty acid metabolisms. Biomed Pharmacother. 2023;161:114409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 88] [Reference Citation Analysis (0)] |
| 26. | Sai Priya T, Ramalingam V, Suresh Babu K. Natural products: A potential immunomodulators against inflammatory-related diseases. Inflammopharmacology. 2024;32:2821-2859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 27. | Aware CB, Patil DN, Suryawanshi SS, Mali PR, Rane MR, Gurav RG, Jadhav JP. Natural bioactive products as promising therapeutics: A review of natural product-based drug development. Afr J Bot. 2022;151:512-528. [DOI] [Full Text] |
| 28. | Xue JC, Yuan S, Meng H, Hou XT, Li J, Zhang HM, Chen LL, Zhang CH, Zhang QG. The role and mechanism of flavonoid herbal natural products in ulcerative colitis. Biomed Pharmacother. 2023;158:114086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 78] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 29. | Jamieson PE, Carbonero F, Stevens JF. Dietary (poly)phenols mitigate inflammatory bowel disease: Therapeutic targets, mechanisms of action, and clinical observations. Curr Res Food Sci. 2023;6:100521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 24] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 30. | Guo Y, Li Y, Cao Q, Ye L, Wang J, Guo M. The Function of Natural Polysaccharides in the Treatment of Ulcerative Colitis. Front Pharmacol. 2022;13:927855. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (1)] |
| 31. | Li C, Wang J, Ma R, Li L, Wu W, Cai D, Lu Q. Natural-derived alkaloids exhibit great potential in the treatment of ulcerative colitis. Pharmacol Res. 2022;175:105972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (1)] |
| 32. | Rivera Rodríguez R, Johnson JJ. Terpenes: Modulating anti-inflammatory signaling in inflammatory bowel disease. Pharmacol Ther. 2023;248:108456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 28] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 33. | Vazirian F, Samadi S, Abbaspour M, Taleb A, Bagherhosseini H, Mozaffari HM, Mohammadpour AH, Emami SA. Evaluation of the efficacy of Thymus kotschyanus extract as an additive treatment in patients with ulcerative colitis: a randomized double-blind placebo-controlled trial. Inflammopharmacology. 2022;30:2145-2152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 34. | Nikkhah-Bodaghi M, Darabi Z, Agah S, Hekmatdoost A. The effects of Nigella sativa on quality of life, disease activity index, and some of inflammatory and oxidative stress factors in patients with ulcerative colitis. Phytother Res. 2019;33:1027-1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 39] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 35. | Rehman IU, Saleem M, Raza SA, Bashir S, Muhammad T, Asghar S, Qamar MU, Shah TA, Bin Jardan YA, Mekonnen AB, Bourhia M. Anti-ulcerative colitis effects of chemically characterized extracts from Calliandra haematocephala in acetic acid-induced ulcerative colitis. Front Chem. 2024;12:1291230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 36. | Zhou HF, Yang C, Li JY, He YY, Huang Y, Qin RJ, Zhou QL, Sun F, Hu DS, Yang J. Quercetin serves as the major component of Xiang-lian Pill to ameliorate ulcerative colitis via tipping the balance of STAT1/PPARγ and dictating the alternative activation of macrophage. J Ethnopharmacol. 2023;313:116557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 34] [Reference Citation Analysis (0)] |
| 37. | Li S, Lu S, Xiong J, Chen L, Lin Y, Chen X, Wang X, Lin L, Cai X, Yang X. Hydroxyethyl starch-curcumin nanoparticles ameliorate DSS-induced ulcerative colitis in mice via synergistic TLR4/NF-κB suppression, Nrf2 activation, intestinal barrier restoration, and gut microbiota modulation. Front Pharmacol. 2025;16:1610711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 38. | Shu L, Fu H, Pi A, Feng Y, Dong H, Si C, Li S, Zhu F, Zheng P, Zhu Q. Protective effect of andrographolide against ulcerative colitis by activating Nrf2/HO-1 mediated antioxidant response. Front Pharmacol. 2024;15:1424219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 39. | Ruan Y, Zhu X, Shen J, Chen H, Zhou G. Mechanism of Nicotiflorin in San-Ye-Qing rhizome for anti-inflammatory effect in ulcerative colitis. Phytomedicine. 2024;129:155564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 49] [Reference Citation Analysis (0)] |
| 40. | Meng G, Li P, Du X, Feng X, Qiu F. Berberine alleviates ulcerative colitis by inhibiting inflammation through targeting IRGM1. Phytomedicine. 2024;133:155909. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 39] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 41. | Chen M, Wang Y, Chen L, Chen M, Li X, Wang G. Epiberberine ameliorates ulcerative colitis by regulating bile acids hepatoenteral circulation through intestinal FXR. Phytomedicine. 2025;143:156831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 42. | Feng W, Zhang Y, Zhang Y, Dong Y, Wu J, Chen Q, Liu M, Wang D, Wu Y, Wang T. Anemoside B4 ameliorates dextran sulfate sodium (DSS)-induced colitis through inhibiting NLRP3 inflammasome and modulating gut microbiota. Eur J Pharmacol. 2024;963:176164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 43. | Hu Q, Xie J, Jiang T, Gao P, Chen Y, Zhang W, Yan J, Zeng J, Ma X, Zhao Y. Paeoniflorin alleviates DSS-induced ulcerative colitis by suppressing inflammation, oxidative stress, and apoptosis via regulating serum metabolites and inhibiting CDC42/JNK signaling pathway. Int Immunopharmacol. 2024;142:113039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 44. | Tang Y, Wang Z, Zhou F, Li L, Sun C, Li L, Tang F, Huang D, Li Z, Tan Y, Pei G. Benzoylpaeoniflorin alleviates ulcerative colitis by inhibiting ferroptosis through targeting phosphogluconic dehydrogenase. Phytomedicine. 2025;147:157111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 45. | Xiao Q, Luo L, Zhu X, Yan Y, Li S, Chen L, Wang X, Zhang J, Liu D, Liu R, Zhong Y. Formononetin alleviates ulcerative colitis via reshaping the balance of M1/M2 macrophage polarization in a gut microbiota-dependent manner. Phytomedicine. 2024;135:156153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 46. | Wu H, Li YL, Wang Y, Wang YG, Hong JH, Pang MM, Liu PM, Yang JJ. Anemoside B4 alleviates ulcerative colitis by attenuating intestinal oxidative stress and NLRP3 inflammasome via activating aryl hydrocarbon receptor through remodeling the gut microbiome and metabolites. Redox Biol. 2025;85:103746. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 26.0] [Reference Citation Analysis (3)] |
| 47. | Zhang D, Wu J, Feng H, Tang P, Zhou Y, Zhao C, Liu J, Feng W, Peng C. Gastrodin ameliorates ulcerative colitis via modulating gut microbial tryptophan metabolism and AhR/NLRP3 pathway. Phytomedicine. 2025;147:157217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 48. | Jia Y, Liu X, Gao X, Yin S, Wu K, Meng X, Ren H, Liu J, Liu Z, Li H, Jiang Y. Plantamajoside alleviates DSS-induced ulcerative colitis by modulating gut microbiota, upregulating CBS, and inhibiting NF-κB. Phytomedicine. 2025;143:156827. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 49. | Qiu L, Yan C, Yang Y, Liu K, Yin Y, Zhang Y, Lei Y, Jia X, Li G. Morin alleviates DSS-induced ulcerative colitis in mice via inhibition of inflammation and modulation of intestinal microbiota. Int Immunopharmacol. 2024;140:112846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 34] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 50. | Xiu M, Li B, He L, Shi Y, Zhang Y, Zhou S, Liu Y, Wang N, He J. Caffeic Acid Protects Against Ulcerative Colitis via Inhibiting Mitochondrial Apoptosis and Immune Overactivation in Drosophila. Drug Des Devel Ther. 2025;19:2157-2172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 51. | Jiang H, Li G, Xie L, Zhang N, Huang Y, Liang X, Guo F, Jiang Q, Liao Z. Curcumin Combined with Tryptophan Ameliorates DSS-Induced Ulcerative Colitis via Reducing Inflammation and Oxidative Stress and Regulation of Gut Microbiota. Nutrients. 2025;17:2988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 52. | Zhao H, Fu X, Wang Y, Shang Z, Li B, Zhou L, Liu Y, Liu D, Yi B. Therapeutic Potential of Vanillic Acid in Ulcerative Colitis Through Microbiota and Macrophage Modulation. Mol Nutr Food Res. 2025;69:e202400785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 53. | Zhang X, Cai J, Chen L, Tai J, Liu J, Cao Y. Gastrodin ameliorates DSS-induced ulcerative colitis via enhancing the abundance of Akkermansia muciniphila and modulating the metabolites. Phytomedicine. 2025;146:157138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 54. | Ma X, Ouyang C, Tang P, Ma H, Wang Z, Hou Y, Zhu L, Huang H. Vanillic acid protects against ulcerative colitis by modulating oxidative stress, inflammatory response, macrophage polarization and AMPK signaling pathways. Int Immunopharmacol. 2026;168:115928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 55. | Yang W, Ren D, Shao H, Zhang X, Li T, Zhang L, Liu L, Zhao Y, Niu P, Yang X. Theabrownin from Fu Brick Tea Improves Ulcerative Colitis by Shaping the Gut Microbiota and Modulating the Tryptophan Metabolism. J Agric Food Chem. 2023;71:2898-2913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 47] [Reference Citation Analysis (0)] |
| 56. | Huang J, Liu Z, Liu Y, Liu X, Tian Z, Yao M, Wang W, Liu Z. Butylchlorogenate from Chaenomeles speciosa alleviates ulcerative colitis via suppressing NLRP3 inflammasome activation and remodeling intestinal homeostasis. Phytomedicine. 2025;148:157282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 57. | Lei L, Wang J, Wang J, He W, Wu T, Li J, Bi X, Mei M, Guan X, Zhu X. Quercetin boosts gut microbiota-driven production of isovanillic acid to alleviate colitis via enhancing intestinal barrier function. Curr Res Food Sci. 2025;11:101183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 58. | Lv M, Wan X, Wang Y, Jiang H, Qin X, Wang Z, Yang C, Shuai J, Lu Q, Xu F, Liu Y. Combined gut microbiome and metabolomics to reveal the mechanism of proanthocyanidins from the roots of Ephedra sinica Stapf on the treatment of ulcerative colitis. J Pharm Biomed Anal. 2024;249:116351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 59. | Wang T, Wu Z, Li M, Cao B, Li J, Jiang J, Liu H, Zhang Q, Zhang S. TCP80-1, a new levan-neoseries fructan from Tupistra chinensis Baker rhizomes alleviates ulcerative colitis induced by dextran sulfate sodium in Drosophila melanogaster model. Food Res Int. 2025;203:115860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 60. | Liu J, Wu H, Wu J, Jiang X, Fan Y, Liu Y, Wang L, Sun X. Kaji-ichigoside F1 alleviates the effect of ulcerative colitis: An integrated strategy of gut microbiota, metabolomics, and transcriptomics. Phytomedicine. 2025;148:157320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 61. | Sun W, Hang L, Chen L, Qu Y, Zhang Y, Miao Z, Wang X. Fraxin Ameliorates Ulcerative Colitis by Modulating Oxidative Stress, Inflammation and TLR4/NF-κB and MAPK Signaling Pathways. Altern Ther Health Med. 2024;30:117-125. [PubMed] |
| 62. | Li Q, Wu SS, Chen BZ, Li MH, Wang TS, Li C. Bioactive components from Pericarpium Citri Reticulatae 'Chachi' alleviates ulcerative colitis by PI3K/AKT signaling pathway: Network pharmacology, molecular docking and experimental verification. Fitoterapia. 2025;185:106736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 63. | Mazeti SSS, Turini AA, Bueno LR, Lima CD, Bastos RS, Rocha JA, Cordeiro LMC, Maria-Ferreira D, Gois MB. Homogalacturonan From Yellow Passion Fruit Peel Ameliorates Intestinal Injury in DSS-Induced Colitis in Mice. J Cell Mol Med. 2025;29:e70910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 64. | Huang TQ, Chen YX, Zeng SL, Lin Y, Li F, Jiang ZM, Liu EH. Bergenin Alleviates Ulcerative Colitis By Decreasing Gut Commensal Bacteroides vulgatus-Mediated Elevated Branched-Chain Amino Acids. J Agric Food Chem. 2024;72:3606-3621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 65. | Zhao L, Zhao C, Miao Y, Lei S, Li Y, Gong J, Peng C. Theabrownin from Pu-erh Tea Improves DSS-Induced Colitis via Restoring Gut Homeostasis and Inhibiting TLR2&4 Signaling Pathway. Phytomedicine. 2024;132:155852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 66. | Yang Y, Li X, Huang F, Cai F, Ning X, Jiang Q, Zhou L, Zeng B, Zhou W, Hu G. Comprehensive multiomics profiling reveals the protective function of gypenosides against dextran sulfate sodium-induced colitis. Sci Rep. 2025;15:31803. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 67. | Wang Y, Sun C, Cao Y, Jiao T, Wang K, Li J, Zhang M, Jiang J, Zhong X, Yu S, Xu H, Wang J, Yi T, Tian X, Zhu H, Zhou H, Huang C, Wu T, Guo X, Xie C. Glycyrrhizic acid and patchouli alcohol in Huoxiang Zhengqi attenuate intestinal inflammation and barrier injury via regulating endogenous corticosterone metabolism mediated by 11β-HSD1. J Ethnopharmacol. 2025;338:119025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 68. | Ju T, Song Z, Qin D, Cheng J, Li T, Hu G, Fu S. Neohesperidin Attenuates DSS-Induced Ulcerative Colitis by Inhibiting Inflammation, Reducing Intestinal Barrier Damage, and Modulating Intestinal Flora Composition. J Agric Food Chem. 2024;72:20419-20431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 69. | Lan M, Lin C, Zeng L, Hu S, Shi Y, Zhao Y, Liu X, Sun J, Liang G, Huang M. Linderanine C regulates macrophage polarization by inhibiting the MAPK signaling pathway against ulcerative colitis. Biomed Pharmacother. 2024;178:117239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 70. | Prakash AN, Prasad N, Puppala ER, Panda SR, Jain S, Ravichandiran V, Singh M, Naidu VGM. Loganic acid protects against ulcerative colitis by inhibiting TLR4/NF-κB mediated inflammation and activating the SIRT1/Nrf2 anti-oxidant responses in-vitro and in-vivo. Int Immunopharmacol. 2023;122:110585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 35] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 71. | Li J, Li P, Yuan S, Xue JC, Zhang QG, Gao BH. Pulchinenoside B4 alleviates DSS-induced colitis by inhibiting CD1d-dependent NLRP3 inflammasome activation in macrophages. Int Immunopharmacol. 2025;148:114118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 72. | Liu Z, Liang L, Lu S, Chen J, Guo H, Xiong Y. Citrus rhoifolin alleviated DSS-induced acute colitis by activating CEMIP/SLC7A11-mediated cystine uptake and inhibiting epithelial ferroptosis. Eur J Nutr. 2024;64:20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 73. | Yu J, Zheng Y, Liu C, Xie Z, Liu Q, Yang S, Tian Q, Song C, Chen S. Multi-omics reveals the alleviating effect of berberine on ulcerative colitis through modulating the gut microbiome and bile acid metabolism in the gut-liver axis. Front Pharmacol. 2024;15:1494210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 74. | Wu W, Qu X, Hu C, Zhu X, Wan M, Zhou Y, Cheng H. Gypenoside LXXV Alleviates Colitis by Reprograming Macrophage Polarization via the Glucocorticoid Receptor Pathway. J Agric Food Chem. 2024;72:20444-20457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 75. | Dharmapuri G, Kotha AK, Kalangi SK, Reddanna P. Mangiferin, A Naturally Occurring Glucosylxanthone, Induces Apoptosis in Caco-2 Cells In Vitro and Exerts Protective Effects on Acetic Acid-Induced Ulcerative Colitis in Mice through the Regulation of NLRP3. ACS Pharmacol Transl Sci. 2024;7:1270-1277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 76. | Chu Z, Hu Z, Yang F, Zhou Y, Tang Y, Luo F. Didymin Ameliorates Dextran Sulfate Sodium (DSS)-Induced Ulcerative Colitis by Regulating Gut Microbiota and Amino Acid Metabolism in Mice. Metabolites. 2024;14:547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 77. | Wang F, Zhang M, Yuan M, Xia Z, Yang F, Zhang S, Lin T, Luo L, Tang J, Zhang Y. A novel sorbicillinoid compound as a potent anti-inflammation agent through inducing NLRP3 protein degradation. Br J Pharmacol. 2023;180:1930-1948. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 78. | Huang M, Lan M, Liu X, Lin C, Zeng L, Li Y, Li F, Dou X, Zhao Y, Shi Y, Xu X, Sun J, Liang G. Isolinderalactone regulates macrophage polarization and efferocytosis by activating the LXRα pathway against ulcerative colitis. Chin Med. 2025;20:152. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 79. | Lai H, Chen M, Xuan W, Xie F, Xu P, Huang M, Wang H. Linderane from Lindera aggregata attenuates ulcerative colitis by suppressing IL-6/STAT3-mediated Th17 differentiation and apoptosis resistance. J Ethnopharmacol. 2026;357:120965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 80. | Araruna MEC, Alves Júnior EB, de Lima Serafim CA, Pessoa MMB, de Souza Pessôa ML, Alves VP, Sobral MV, da Silva MS, Alves AF, de Paiva Sousa MC, Araújo AA, Batista LM. (-)-Fenchone Ameliorates TNBS-Induced Colitis in Rats via Antioxidant, Immunomodulatory, and Cytoprotective Mechanisms. Pharmaceuticals (Basel). 2024;18:18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 81. | Kou RW, Li ZQ, Wang JL, Jiang SQ, Zhang RJ, He YQ, Xia B, Gao JM. Ganoderic Acid A Mitigates Inflammatory Bowel Disease through Modulation of AhR Activity by Microbial Tryptophan Metabolism. J Agric Food Chem. 2024;72:17912-17923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 82. | Gao BB, Wang L, Li LZ, Fei ZQ, Wang YY, Zou XM, Huang MC, Lei SS, Li B. Beneficial effects of oxymatrine from Sophora flavescens on alleviating Ulcerative colitis by improving inflammation and ferroptosis. J Ethnopharmacol. 2024;332:118385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 24] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 83. | Tan C, Xiang Z, Wang S, He H, Li X, Xu M, Guo X, Pu Y, Zhen J, Dong W. Diosmin alleviates colitis by inhibiting PANoptosis of intestinal epithelial cells and regulating gut microbiota and metabolites. Phytomedicine. 2025;141:156671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 84. | Li J, Hua L, Hu M, Zhu N, Dong S, Jing X, Zhu Z, Liu Y, Zhou Y. The protective effect of naringenin on ulcerative colitis in mice through increasing Nrf2 pathway activity. Acta Biochim Biophys Sin (Shanghai). 2025;57:1068-1080. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 85. | Ma X, Li M, Wang X, Xu H, Jiang L, Wu F, Wei L, Qi G, Zhang D. Dihydromyricetin ameliorates experimental ulcerative colitis by inhibiting neutrophil extracellular traps formation via the HIF-1α/VEGFA signaling pathway. Int Immunopharmacol. 2024;138:112572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 86. | Tao Y, Shao R, Cui M, Wang H, Xiang M, Ge S, Deng M, Li X, Liu F, Fan F. Homoplantaginin inhibits the progression of ulcerative colitis in mice by regulating the MMP9-RLN2 signaling axis. Front Med (Lausanne). 2025;12:1582066. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 87. | Huang Y, Wang N, Ji X, Luo S, Gong L, Zhao C, Zheng G, Liu R, Zhang T. Apigenin ameliorates inflamed ulcerative colitis by regulating mast cell degranulation via the PAMP-MRGPRX2 feedback loop. Phytomedicine. 2025;140:156564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 88. | Tambovtseva RS, Arslan LA, Grigoryeva TA, Abdulkhakov SR, Doludin YV, Stoma IO, Rizvanov AA, Miftakhova RR, Gabdoulkhakova AG. Narrative review on bacteria-derived metabolites in the pathogenesis of ulcerative colitis. Clin Microbiol Rev. 2025;38:e0021024. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 89. | Chen H, Qian Y, Jiang C, Tang L, Yu J, Zhang L, Dai Y, Jiang G. Butyrate ameliorated ferroptosis in ulcerative colitis through modulating Nrf2/GPX4 signal pathway and improving intestinal barrier. Biochim Biophys Acta Mol Basis Dis. 2024;1870:166984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 78] [Article Influence: 39.0] [Reference Citation Analysis (0)] |
| 90. | Xie J, Liu Y, Chen B, Zhang G, Ou S, Luo J, Peng X. Ganoderma lucidum polysaccharide improves rat DSS-induced colitis by altering cecal microbiota and gene expression of colonic epithelial cells. Food Nutr Res. 2019;63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 90] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 91. | Hossain R, Quispe C, Khan RA, Saikat ASM, Ray P, Ongalbek D, Yeskaliyeva B, Jain D, Smeriglio A, Trombetta D, Kiani R, Kobarfard F, Mojgani N, Saffarian P, Ayatollahi SA, Sarkar C, Islam MT, Keriman D, Uçar A, Martorell M, Sureda A, Pintus G, Butnariu M, Sharifi-Rad J, Cho WC. Propolis: An update on its chemistry and pharmacological applications. Chin Med. 2022;17:100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 88] [Cited by in RCA: 98] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 92. | Huang S, Zhang CP, Wang K, Li GQ, Hu FL. Recent advances in the chemical composition of propolis. Molecules. 2014;19:19610-19632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 335] [Cited by in RCA: 412] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 93. | Ayad AS, Benchaabane S, Daas T, Smagghe G, Loucif-Ayad W. Propolis Stands out as a Multifaceted Natural Product: Meta-Analysis on Its Sources, Bioactivities, Applications, and Future Perspectives. Life (Basel). 2025;15:764. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 94. | Barbosa Bezerra G, de Menezes de Souza L, Dos Santos AS, de Almeida GK, Souza MT, Santos SL, Aparecido Camargo E, Dos Santos Lima B, de Souza Araújo AA, Cardoso JC, Gomes SV, Gomes MZ, de Albuquerque RL Júnior. Hydroalcoholic extract of Brazilian red propolis exerts protective effects on acetic acid-induced ulcerative colitis in a rodent model. Biomed Pharmacother. 2017;85:687-696. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 37] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 95. | Hua Z, Hui LI, Haihua W. Potential protective effects of the water-soluble Chinese propolis on experimental ulcerative colitis. J Tradit Chin Med. 2023;43:925-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 96. | Qian Z, Zhang M, Lu T, Yu J, Yin S, Wang H, Wang J. Propolis alleviates ulcerative colitis injury by inhibiting the protein kinase C - transient receptor potential cation channel subfamily V member 1 - calcitonin gene-related peptide/substance P (PKC-TRPV1-CGRP/SP) signaling axis. PLoS One. 2024;19:e0294169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 97. | Zhou Y, Wang D, Yan W. Treatment Effects of Natural Products on Inflammatory Bowel Disease In Vivo and Their Mechanisms: Based on Animal Experiments. Nutrients. 2023;15:1031. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 36] [Reference Citation Analysis (0)] |
| 98. | Long D, Mao C, Zhang W, Zhu Y, Xu Y. Natural products for the treatment of ulcerative colitis: focus on the JAK/STAT pathway. Front Immunol. 2025;16:1538302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 99. | Zhou Y, Zhang D, Cheng H, Wu J, Liu J, Feng W, Peng C. Repairing gut barrier by traditional Chinese medicine: roles of gut microbiota. Front Cell Infect Microbiol. 2024;14:1389925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 26] [Reference Citation Analysis (0)] |
| 100. | Moudgil KD, Venkatesha SH. The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation. Int J Mol Sci. 2022;24:95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 141] [Article Influence: 35.3] [Reference Citation Analysis (0)] |
| 101. | Nascimento RPD, Machado APDF, Galvez J, Cazarin CBB, Maróstica Junior MR. Ulcerative colitis: Gut microbiota, immunopathogenesis and application of natural products in animal models. Life Sci. 2020;258:118129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 108] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 102. | Zhang W, Zou M, Fu J, Xu Y, Zhu Y. Autophagy: A potential target for natural products in the treatment of ulcerative colitis. Biomed Pharmacother. 2024;176:116891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 103. | Chen B, Dong X, Zhang JL, Sun X, Zhou L, Zhao K, Deng H, Sun Z. Natural compounds target programmed cell death (PCD) signaling mechanism to treat ulcerative colitis: a review. Front Pharmacol. 2024;15:1333657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 104. | Long D, Mao C, Huang Y, Xu Y, Zhu Y. Ferroptosis in ulcerative colitis: Potential mechanisms and promising therapeutic targets. Biomed Pharmacother. 2024;175:116722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 45] [Reference Citation Analysis (0)] |
| 105. | Zhang YF, Fan MY, Bai QR, Zhao R, Song S, Wu L, Lu JH, Liu JW, Wang Q, Li Y, Chen X. Precision therapy for ulcerative colitis: insights from mitochondrial dysfunction interacting with the immune microenvironment. Front Immunol. 2024;15:1396221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 106. | Zhang YF, Shi TT, Lin YL, Zhu YT, Lin S, Fang TY. Macrophage Metabolic Reprogramming in Inflammatory Bowel Diseases: From Pathogenesis to Therapy. J Inflamm Res. 2025;18:11821-11839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 107. | Che X, Zhang Y, Chen X, Xie G, Li J, Xu C, Zhang C, Zhu Y, Yang X. The lactylation-macrophage interplay: implications for gastrointestinal disease therapeutics. Front Immunol. 2025;16:1608115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 108. | Li L, Ye Z, Qian H, Chen L, Hu Y, Liu X, Zhu J, Bao T, Ganesan K, Lu F, Wang J, Wen X, Qin K, Ye Q. Modified Tou Nong Powder obstructs ulcerative colitis by regulating autophagy and mitochondrial function. J Ethnopharmacol. 2025;340:119220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 109. | Dixon SJ, Olzmann JA. The cell biology of ferroptosis. Nat Rev Mol Cell Biol. 2024;25:424-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1019] [Cited by in RCA: 917] [Article Influence: 458.5] [Reference Citation Analysis (5)] |
| 110. | Liu Y, Zhang JT, Sun M, Song J, Sun HM, Wang MY, Wang CM, Liu W. Targeting ferroptosis in the treatment of ulcerative colitis by traditional Chinese medicine: A novel therapeutic strategies. Phytomedicine. 2025;139:156539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 111. | Zhang J, Tan B, Wu H, Han T, Fang D, Cai H, Hu B, Kang A. Scutellaria baicalensis Extracts Restrict Intestinal Epithelial Cell Ferroptosis by Regulating Lipid Peroxidation and GPX4/ACSL4 in Colitis. Phytomedicine. 2025;141:156708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 112. | Pan Z, Gan C, Zhi S, Yang Y, Zhang Y, Li L, Zhang S, Huang Q. Gancao Xiexin decoction attenuated experimental colitis through suppressing ACSL4-mediated ferroptosis. J Ethnopharmacol. 2025;344:119532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 113. | Gong Y, Zha Q, Li L, Liu Y, Yang B, Liu L, Lu A, Lin Y, Jiang M. Efficacy and safety of Fufangkushen colon-coated capsule in the treatment of ulcerative colitis compared with mesalazine: a double-blinded and randomized study. J Ethnopharmacol. 2012;141:592-598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 47] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 114. | Amiri M, Navabi J, Shokoohinia Y, Heydarpour F, Bahrami G, Behbood L, Derakhshandeh P, Momtaz S, Farzaei MH. Efficacy and safety of a standardized extract from Achillea wilhelmsii C. Koch in patients with ulcerative colitis: A randomized double blind placebo-controlled clinical trial. Complement Ther Med. 2019;45:262-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 115. | Kanauchi O, Mitsuyama K, Homma T, Takahama K, Fujiyama Y, Andoh A, Araki Y, Suga T, Hibi T, Naganuma M, Asakura H, Nakano H, Shimoyama T, Hida N, Haruma K, Koga H, Sata M, Tomiyasu N, Toyonaga A, Fukuda M, Kojima A, Bamba T. Treatment of ulcerative colitis patients by long-term administration of germinated barley foodstuff: multi-center open trial. Int J Mol Med. 2003;12:701-704. [PubMed] |
| 116. | Naganuma M, Sugimoto S, Fukuda T, Mitsuyama K, Kobayashi T, Yoshimura N, Ohi H, Tanaka S, Andoh A, Ohmiya N, Saigusa K, Yamamoto T, Morohoshi Y, Ichikawa H, Matsuoka K, Hisamatsu T, Watanabe K, Mizuno S, Abe T, Suzuki Y, Kanai T; INDIGO Study Group. Indigo naturalis is effective even in treatment-refractory patients with ulcerative colitis: a post hoc analysis from the INDIGO study. J Gastroenterol. 2020;55:169-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 32] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 117. | Dryden GW, Lam A, Beatty K, Qazzaz HH, McClain CJ. A pilot study to evaluate the safety and efficacy of an oral dose of (-)-epigallocatechin-3-gallate-rich polyphenon E in patients with mild to moderate ulcerative colitis. Inflamm Bowel Dis. 2013;19:1904-1912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 118. | Irving PM, Iqbal T, Nwokolo C, Subramanian S, Bloom S, Prasad N, Hart A, Murray C, Lindsay JO, Taylor A, Barron R, Wright S. A Randomized, Double-blind, Placebo-controlled, Parallel-group, Pilot Study of Cannabidiol-rich Botanical Extract in the Symptomatic Treatment of Ulcerative Colitis. Inflamm Bowel Dis. 2018;24:714-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 107] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 119. | Shen H, Zhang S, Zhao W, Ren S, Ke X, Gu Q, Tang Z, Xie J, Chen S, Chen Y, Zou J, Zhang L, Shen Z, Zheng K, Liu Y, Gu P, Cheng J, Hu J, Zhu L. Randomised clinical trial: Efficacy and safety of Qing-Chang-Hua-Shi granules in a multicenter, randomized, and double-blind clinical trial of patients with moderately active ulcerative colitis. Biomed Pharmacother. 2021;139:111580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 120. | Chandan S, Mohan BP, Chandan OC, Ahmad R, Challa A, Tummala H, Singh S, Dhawan P, Ponnada S, Singh AB, Adler DG. Curcumin use in ulcerative colitis: is it ready for prime time? A systematic review and meta-analysis of clinical trials. Ann Gastroenterol. 2020;33:53-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 121. | Kamali M, Tavakoli H, Khodadoost M, Daghaghzadeh H, Kamalinejad M, Gachkar L, Mansourian M, Adibi P. Efficacy of the Punica granatum peels aqueous extract for symptom management in ulcerative colitis patients. A randomized, placebo-controlled, clinical trial. Complement Ther Clin Pract. 2015;21:141-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 122. | Wei Y, Gong J, Zhu W, Tian H, Ding C, Gu L, Li N, Li J. Pectin enhances the effect of fecal microbiota transplantation in ulcerative colitis by delaying the loss of diversity of gut flora. BMC Microbiol. 2016;16:255. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 123. | Larussa T, Oliverio M, Suraci E, Greco M, Placida R, Gervasi S, Marasco R, Imeneo M, Paolino D, Tucci L, Gulletta E, Fresta M, Procopio A, Luzza F. Oleuropein Decreases Cyclooxygenase-2 and Interleukin-17 Expression and Attenuates Inflammatory Damage in Colonic Samples from Ulcerative Colitis Patients. Nutrients. 2017;9:391. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 62] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 124. | Iyengar P, Godoy-Brewer G, Maniyar I, White J, Maas L, Parian AM, Limketkai B. Herbal Medicines for the Treatment of Active Ulcerative Colitis: A Systematic Review and Meta-Analysis. Nutrients. 2024;16:934. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 125. | McClements DJ, Saliva-Trujillo L, Zhang R, Zhang Z, Zou L, Yao M, Xiao H. Boosting the bioavailability of hydrophobic nutrients, vitamins, and nutraceuticals in natural products using excipient emulsions. Food Res Int. 2016;88:140-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 76] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 126. | Tenore GC, Campiglia P, Giannetti D, Novellino E. Simulated gastrointestinal digestion, intestinal permeation and plasma protein interaction of white, green, and black tea polyphenols. Food Chem. 2015;169:320-326. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 99] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 127. | Costea PI, Hildebrand F, Arumugam M, Bäckhed F, Blaser MJ, Bushman FD, de Vos WM, Ehrlich SD, Fraser CM, Hattori M, Huttenhower C, Jeffery IB, Knights D, Lewis JD, Ley RE, Ochman H, O'Toole PW, Quince C, Relman DA, Shanahan F, Sunagawa S, Wang J, Weinstock GM, Wu GD, Zeller G, Zhao L, Raes J, Knight R, Bork P. Enterotypes in the landscape of gut microbial community composition. Nat Microbiol. 2018;3:8-16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 899] [Cited by in RCA: 751] [Article Influence: 93.9] [Reference Citation Analysis (4)] |
| 128. | Vieira-Silva S, Sabino J, Valles-Colomer M, Falony G, Kathagen G, Caenepeel C, Cleynen I, van der Merwe S, Vermeire S, Raes J. Quantitative microbiome profiling disentangles inflammation- and bile duct obstruction-associated microbiota alterations across PSC/IBD diagnoses. Nat Microbiol. 2019;4:1826-1831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 195] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 129. | Libby P. Statin drugs might boost healthy gut microbes. Nature. 2020;581:263-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 130. | Glassner KL, Abraham BP, Quigley EMM. The microbiome and inflammatory bowel disease. J Allergy Clin Immunol. 2020;145:16-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 770] [Cited by in RCA: 646] [Article Influence: 107.7] [Reference Citation Analysis (1)] |
| 131. | Afshar M, Najafian S, Radi M. The effect of harvest time on the natural product of Rosmarinus officinalis L. from South Iran (Fars province). Nat Prod Res. 2022;36:2637-2642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 132. | Dong W, Chen Q, Wei C, Hu R, Long Y, Zong Y, Chu Z. Comparison of the effect of extraction methods on the quality of green coffee oil from Arabica coffee beans: Lipid yield, fatty acid composition, bioactive components, and antioxidant activity. Ultrason Sonochem. 2021;74:105578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 44] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 133. | Maksimovic S, Tadic V, Skala D, Zizovic I. Separation of phytochemicals from Helichrysum italicum: An analysis of different isolation techniques and biological activity of prepared extracts. Phytochemistry. 2017;138:9-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 134. | Biedermann L, Doulberis M, Schreiner P, Nielsen OH, The FO, Brand S, Burk S, Hruz P, Juillerat P, Krieger-Grübel C, Leu K, Leventhal GE, Misselwitz B, Scharl S, Schoepfer A, Seibold F, Herfarth H, Rogler G. Efficacy and Safety of Anthocyanin-Rich Extract in Patients with Ulcerative Colitis: A Randomized Controlled Trial. Nutrients. 2024;16:4197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 135. | Li N, Shang X, Shi L, Li Y, Mao T, Wang Q, Li J, Peng G. Effects of three Chinese herbal therapies on gut microbiota and short-chain fatty acid metabolism in patients with mild, moderate, and severe ulcerative colitis: Multi-center, randomized, controlled trials. Int Immunopharmacol. 2025;152:114444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 136. | Morvaridi M, Aryaeian N, Alavinejad P, Seyedian SS, Ghafourian M, Bakhtiari N, Seyedtabib M. Zatariamultiflora hydroalcoholic extract: A triple-blind randomized controlled trial on immune genes, inflammation, and ulcerative colitis symptoms. J Ethnopharmacol. 2025;344:119527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 137. | Erol Doğan Ö, Karaca Çelik KE, Baş M, Alan EH, Çağın YF. Effects of Mediterranean Diet, Curcumin, and Resveratrol on Mild-to-Moderate Active Ulcerative Colitis: A Multicenter Randomized Clinical Trial. Nutrients. 2024;16:1504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 138. | Li M, Deng H, Xu S, Fang X, Tang K, Chen L, Zha A. The active ingredients and mechanism of Zuoqing San in the treatment of sigmoid ulcerative colitis by retention enema. J Complement Integr Med. 2025;22:343-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 139. | Ikegami S, Nakamura M, Honda T, Yamamura T, Maeda K, Sawada T, Ishikawa E, Yamamoto K, Furune S, Ishikawa T, Furukawa K, Ohno E, Ishigami M, Kinoshita F, Kadota Y, Tochio T, Shimomura Y, Hirooka Y, Kawashima H. Efficacy of 1-kestose supplementation in patients with mild to moderate ulcerative colitis: A randomised, double-blind, placebo-controlled pilot study. Aliment Pharmacol Ther. 2023;57:1249-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 140. | Yanai H, Salomon N, Lahat A, Ungar B, Eliakim R, Kriger-Sharabi O, Reiss-Mintz H, Koslowsky B, Shitrit AB, Tamir-Degabli N, Dotan I, Zittan E, Maharshak N, Hirsch A, Ben-Horin S, Kopylov U. Real-world experience with Curcumin-QingDai combination for patients with active ulcerative colitis: A retrospective multicentre cohort study. Aliment Pharmacol Ther. 2023;58:175-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 141. | Lickliter J, Yang X, Guo J, Pan W, Wei Z. Icanbelimod (CBP-307), a next-generation Sphingosine-1-phosphate receptor modulator, in healthy men: pharmacokinetics, pharmacodynamics, safety, and tolerability in a randomized trial in Australia. Front Immunol. 2024;15:1380975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 142. | Allegretti JR, Cheifetz AS, Dulai PS, Stevens AC, Chapas-Reed J, Chesnel L, Dixit B, Farquhar R, Ghahramani P, Miller BW, Murphy CK, Quintas M, Tanase R, Telia T, Woźniak-Stolarska B, Gupta R. Safety, Pharmacokinetics, and Clinical Efficacy of ADS051, a Neutrophil Modulator, in Ulcerative Colitis: Results of a Randomized Phase 1b Trial. Am J Gastroenterol. 2024;120:1624-1635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 143. | Chen J, Feng C, Liu Y, Cai Z. Identification of hub necroptosis-related targets and discovery of potential natural inhibitors in ulcerative colitis based on bioinformatics and computer-aided drug design. J Comput Aided Mol Des. 2025;39:96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 144. | Corridoni D, Antanaviciute A, Gupta T, Fawkner-Corbett D, Aulicino A, Jagielowicz M, Parikh K, Repapi E, Taylor S, Ishikawa D, Hatano R, Yamada T, Xin W, Slawinski H, Bowden R, Napolitani G, Brain O, Morimoto C, Koohy H, Simmons A. Single-cell atlas of colonic CD8(+) T cells in ulcerative colitis. Nat Med. 2020;26:1480-1490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 176] [Article Influence: 29.3] [Reference Citation Analysis (3)] |
| 145. | Weß V, Schuster-Winkelmann P, Karatekin YH, Malik S, Beigel F, Kühn F, Gropp R. Humanized NSG Mouse Models as a Preclinical Tool for Translational Research in Inflammatory Bowel Diseases. Int J Mol Sci. 2023;24:12348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 146. | Guggeis MA, Andreani NA, López-Agudelo VA, Tran F, Kadibalban AS, Moors KA, Marinos G, Saboukh A, Harris D, Falk-Paulsen M, Weber-Stiehl S, Järke L, Sommer F, Welz L, Bang C, Franke A, Chung CJ, Bronowski C, Schuchardt S, Künzel S, Aden K, Schreiber S, Kaleta C, Baines JF, Rosenstiel P. Cross-species engraftment biases and metabolic divergence in gnotobiotic mice humanized with ulcerative colitis microbiota. Gut Microbes. 2025;17:2581445. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 147. | Ferreira B, Ferreira C, Martins C, Nunes R, das Neves J, Leite-Pereira C, Sarmento B. Establishment of a 3D multi-layered in vitro model of inflammatory bowel disease. J Control Release. 2025;377:675-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 148. | Mertz A, Nguyen NA, Katsanos KH, Kwok RM. Primary sclerosing cholangitis and inflammatory bowel disease comorbidity: an update of the evidence. Ann Gastroenterol. 2019;32:124-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 51] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 149. | Tsaitas C, Semertzidou A, Sinakos E. Update on inflammatory bowel disease in patients with primary sclerosing cholangitis. World J Hepatol. 2014;6:178-187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 41] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 150. | Duboc H, Rajca S, Rainteau D, Benarous D, Maubert MA, Quervain E, Thomas G, Barbu V, Humbert L, Despras G, Bridonneau C, Dumetz F, Grill JP, Masliah J, Beaugerie L, Cosnes J, Chazouillères O, Poupon R, Wolf C, Mallet JM, Langella P, Trugnan G, Sokol H, Seksik P. Connecting dysbiosis, bile-acid dysmetabolism and gut inflammation in inflammatory bowel diseases. Gut. 2013;62:531-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 810] [Cited by in RCA: 721] [Article Influence: 55.5] [Reference Citation Analysis (3)] |
| 151. | Zhou Q, Gao S, Yu X, Zhang L, Zhang Z, Fu Y, Liu W, Mu Y, Zhang H, Liu P, Chen J. Total astragalus saponins attenuate primary sclerosing cholangitis in mice by upregulation of TGR5. Phytother Res. 2024;38:4502-4518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 152. | Hu RW, Carey EJ, Lindor KD, Tabibian JH. Curcumin in Hepatobiliary Disease: Pharmacotherapeutic Properties and Emerging Potential Clinical Applications. Ann Hepatol. 2017;16:835-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 153. | Cai J, Zhu Z, Li Y, Li Q, Tian T, Meng Q, Wang T, Ma Y, Wu J. Artemisia capillaris Thunb. Polysaccharide alleviates cholestatic liver injury through gut microbiota modulation and Nrf2 signaling pathway activation in mice. J Ethnopharmacol. 2024;327:118009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 154. | Gao TH, Liao W, Lin LT, Zhu ZP, Lu MG, Fu CM, Xie T. Curcumae rhizoma and its major constituents against hepatobiliary disease: Pharmacotherapeutic properties and potential clinical applications. Phytomedicine. 2022;102:154090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 86] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 155. | Jha AK, Sit N. Extraction of bioactive compounds from plant materials using combination of various novel methods: A review. Trends Food Sci Tech. 2022;119:579-591. [RCA] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 198] [Article Influence: 49.5] [Reference Citation Analysis (0)] |
| 156. | Zhang J, Sun S, Chen H, Feng Y, Li Y, Dong Z. Advances in natural compound-based nanomedicine and the interaction with gut microbiota in ulcerative colitis therapy. Front Pharmacol. 2023;14:1197144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 157. | Wu T, Bai X, Zhang Y, Dai E, Ma J, Yu C, He C, Li Q, Yang Y, Kong H, Qu H, Zhao Y. Natural medicines-derived carbon dots as novel oral antioxidant administration strategy for ulcerative colitis therapy. J Nanobiotechnology. 2024;22:511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 158. | Liu H, Liu Y, Peng S, Zhou L, McClements DJ, Fang S, Liu W. Colonic delivery and controlled release of curcumin encapsulated within plant-based extracellular vesicles loaded into hydrogel beads. Food Res Int. 2025;202:115540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 15] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 159. | Butler MS, Capon RJ, Blaskovich MAT, Henderson IR. Natural product-derived compounds in clinical trials and drug approvals. Nat Prod Rep. 2026;43:20-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 160. | Cantarovich D, Kervella D, Karam G, Dantal J, Blancho G, Giral M, Garandeau C, Houzet A, Ville S, Branchereau J, Delbos F, Guillot-Gueguen C, Volteau C, Leroy M, Renaudin K, Soulillou JP, Hourmant M. Tacrolimus- versus sirolimus-based immunosuppression after simultaneous pancreas and kidney transplantation: 5-year results of a randomized trial. Am J Transplant. 2020;20:1679-1690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 161. | Silverberg JI, Boguniewicz M, Quintana FJ, Clark RA, Gross L, Hirano I, Tallman AM, Brown PM, Fredericks D, Rubenstein DS, McHale KA. Tapinarof validates the aryl hydrocarbon receptor as a therapeutic target: A clinical review. J Allergy Clin Immunol. 2024;154:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 53] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 162. | Shirley M. Etrasimod: First Approval. Drugs. 2024;84:247-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |