Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.117259
Revised: December 23, 2025
Accepted: January 29, 2026
Published online: April 28, 2026
Processing time: 135 Days and 19.8 Hours
Monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL) is a rare, agg
To improve the understanding and diagnostic accuracy of MEITL, this study ana
Patients who were diagnosed with MEITL or EATL II at Peking Union Medical College Hospital between August 2012 and June 2025 were retrospectively en
Among the 26 MEITL patients in Peking Union Medical College Hospital, the most common symptoms were weight loss (73%) and abdominal pain (65%), with 96% showing small intestinal involvement. Endoscopic findings in 10 patients showed mucosal edema (100%), roughness (100%), and villous blunting (90%). A review of 130 pu
Recognizing the characteristic symptoms and endoscopic features of MEITL is essential for improving the dia
Core Tip: This study analyzed 26 cases from Peking Union Medical College Hospital and 130 literature-reported cases of monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL). It found heterogeneous endoscopic manifestations. The Peking Union Medical College Hospital cohort mainly presented with subtle mucosal changes (edema, roughness, villous blunting), which may be early endoscopic signs of MEITL. Ulceration was the main endoscopic feature in the literature cohort, likely indicating disease progression. Multi-site deep biopsy combined with imaging is crucial for early accurate diagnosis of MEITL. Two clinical subtypes (acute aggressive and chronic consumptive) were identified, with distinct symptoms and pathological features, providing value for personalized clinical management.
- Citation: Jiang SJ, Ou CZ, Tang H, Li MH, Jia CW, Zhou WX, He ZYT, Li J, Zhang Y, Li JN. Monomorphic epitheliotropic intestinal T-cell lymphoma: Clinical, endoscopic and pathological features. World J Gastroenterol 2026; 32(16): 117259
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/117259.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.117259
Monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL) is a rare and aggressive form of intestinal non-Hodgkin lymphoma that originates from intraepithelial lymphocytes (IEL). It was initially classified as enteropathy-associated T-cell lymphoma (EATL) type II and was formally designated as MEITL in the 2016 World Health Organization classification of tumors of hematopoietic and lymphoid tissues[1]. EATL type I is more common in Western countries and is strongly associated with celiac disease[2]. In contrast, MEITL has no clear association with celiac disease and predominantly occurs in Asian and Hispanic populations.
MEITL predominantly affects older adults, with a median age at onset of 58-62 years and a male predominance[3]. Clinically, MEITL lacks specific manifestations. Patients often present with abdominal pain, diarrhea, weight loss, or abdominal distension, and some patients may develop acute abdominal symptoms due to intestinal perforation or obstruction. Because of its rapid disease progression, most patients are diagnosed at advanced stages, with a very poor prognosis. The reported median survival time for MEITL is approximately 7 months, and the 1-year overall survival rate is less than 40%[4]. Therefore, early recognition and accurate diagnosis are crucial for improving patient outcomes, with endoscopy and imaging playing key roles in the early detection of the disease[5].
However, owing to the extreme rarity of MEITL, most published reports are limited to case studies or small cohorts, and systematic summaries are lacking, resulting in frequent misdiagnoses and delayed diagnoses. In this study, the characteristics of 26 MEITL patients diagnosed at Peking Union Medical College Hospital (PUMCH) between 2012 and 2025 were systematically analyzed and integrated with data from 130 reported cases worldwide to comprehensively summarize the clinical manifestations, endoscopic features, and pathological characteristics of MEITL. Additionally, differences among clinical subtypes were explored, aiming to provide evidence-based guidance for diagnosis and management.
Retrospective cohort from PUMCH: Patients who were diagnosed with MEITL, as confirmed by pathology, at the Departments of Gastroenterology and Hematology, PUMCH, between August 2012 and June 2025, were retrospectively included. The inclusion criteria were as follows: (1) Fulfillment of the 2016 World Health Organization diagnostic criteria for MEITL, defined by diffuse infiltration of monomorphic T lymphocytes with marked epitheliotropism in pathological specimens; immunohistochemical positivity for CD3, CD8, and CD56; and negative results for Epstein-Barr virus-encoded RNA (EBER) in situ hybridization; and (2) Complete clinical data, including demographic information, clinical symptoms, laboratory tests, endoscopic or imaging findings. The exclusion criteria were: (1) Unclear diagnosis or confirmation of other lymphoma subtypes (e.g., EATL type I or natural killer/T-cell lymphoma); and (2) Incomplete clinical data with missing key information. Ultimately, 26 patients were included.
Literature cohort: A systematic search was conducted in PubMed and Web of Science from database inception to June 2025 using the following terms: “monomorphic epitheliotropic intestinal T-cell lymphoma”, “MEITL”, “type II enteropathy-associated T-cell lymphoma”, “EATL II”, “type II intestinal enteropathy-associated T-cell lymphoma”, and “enteropathy-associated T-cell lymphoma type II”. The inclusion criteria were: (1) Case reports or small case series; (2) Cases pathologically confirmed as MEITL or EATL II, using diagnostic criteria consistent with this study; and (3) Availability of complete demographic, clinical, laboratory, endoscopic, and pathological data. The exclusion criteria were: (1) Duplicate cases; (2) Incomplete data; and (3) Review articles or basic research studies. Ultimately, 109 publications reporting 130 cases were included.
Demographic data, clinical manifestations, laboratory results, imaging, endoscopic findings and pathological features were recorded using standardized forms. Endoscopic data for patients in the retrospective cohort from PUMCH were retrieved from the Endoscopy Center database of the Department of Gastroenterology and reviewed by senior endoscopists to document characteristic endoscopic features.
(1) Demographic characteristics: Sex, age at diagnosis, and geographic region of residence; (2) Clinical symptoms: Weight loss, abdominal pain, diarrhea, fever (temperature ≥ 37.3 °C), intestinal obstruction, intestinal perforation, ascites, and gastrointestinal bleeding; (3) Laboratory parameters: Anemia (hemoglobin < 120 g/L for men, < 110 g/L for women), hypoalbuminemia (serum albumin < 35 g/L), elevated C-reactive protein (CRP > 10 mg/L), elevated high-sensitivity CRP (> 8 mg/L), β2-microglobulin (β2-MG; normal range 0.7-1.8 mg/L) and baseline lactate dehydrogenase (LDH; normal range 0-250 U/L); (4) Sites of involvement: Stomach, small intestine (duodenum/jejunum/ileum), and colorectum, confirmed by endoscopy or imaging; (5) Imaging features: Abdominal computed tomography (CT) findings, including bowel wall thickening, bowel dilation, and multiple enlarged lymph nodes; (6) Endoscopic features: Mucosal edema, mucosal roughness (granular surface), villous blunting, lymphangiectasia-like changes, mosaic patterns, mucosal hyperemia, erosion, ulceration, and nodular elevation; and (7) Pathological features: Immunohistochemical markers [CD2, CD3, CD4, CD5, CD7, CD8, CD56, CD20, T-cell intracellular antigen 1 (TIA-1), granzyme B, and Ki-67] and EBER in situ hybridization results.
Statistical analyses were performed using R version 4.3.3. For categorical variables (e.g., symptom incidence and immunophenotype positivity), Pearson’s χ2 test was used to compare groups when the sample size was ≥ 5 and Fisher’s exact test was applied when the sample size was < 5. A significance level of α = 0.05 was used, and P < 0.05 was con
Among the 26 MEITL patients, 15 (57.7%) were male and 11 (42.3%) were female, with a median age at diagnosis of 61 years (range, 37-82 years). The most common symptoms were abdominal pain and/or diarrhea, including weight loss (19/26, 73.1%), abdominal pain (17/26, 65.4%), and diarrhea (9/26, 34.6%). The incidences of intestinal obstruction and perforation prior to diagnosis were both 26.9% (7/26), and one additional patient developed intestinal perforation during chemotherapy. The detailed clinical characteristics of the cohort are summarized in Table 1. Laboratory findings revealed elevated β2-MG (8/8, 100%), elevated high-sensitivity CRP (9/10, 90%), hypoalbuminemia (15/18, 83.3%), anemia (13/20, 65%), and an increased erythrocyte sedimentation rate (3/9, 33.3%), while no patients had elevated LDH levels.
| Characteristic | PUMCH cohort (n = 26) | Literature cohort (n = 130) | P value |
| Clinical characteristics | |||
| Sex (male:female) | 15:11 | 87:42 | 0.370 |
| Median age, years (range) | 61 (37-82) | 61 (35-83) | |
| Weight loss | 73 (19/26) | 46 (60/130) | 0.012a |
| Abdominal pain | 65 (17/26) | 59 (77/130) | 0.558 |
| Diarrhea | 35 (9/26) | 39 (51/130) | 0.659 |
| Fever | 31 (8/26) | 48 (21/44) | 0.164 |
| Intestinal obstruction | 27 (7/26) | 18 (23/130) | 0.276 |
| Intestinal perforation | 31 (8/26) | 35 (45/130) | 0.705 |
| Laboratory findings | |||
| Anemia | 65 (13/20) | 60 (31/52) | 0.675 |
| Hypoalbuminemia | 83 (15/18) | 92 (36/39) | 0.368 |
| Elevated CRP | 90 (9/10) | 86 (25/29) | 1.000 |
| Elevated LDH | 0 (0/16) | 31 (11/35) | 0.011a |
| Elevated β2-MG | 100 (8/8) | ||
| Sites of involvement | |||
| Stomach | 0 | 10 (13/126) | 0.126 |
| Small intestine | 96 (25/26) | 84 (106/126) | 0.128 |
| Colon and rectum | 15 (4/26) | 25 (32/126) | 0.323 |
| Imaging features | |||
| Bowel wall thickening | 83 (15/18) | 56 (54/96) | 0.037a |
| Bowel dilation | 39 (7/18) | 20 (19/96) | 0.076 |
| Lymph node enlargement | 61 (11/18) | 23 (22/96) | 0.001a |
| Endoscopic features | |||
| Diffuse involvement | 60 (6/10) | ||
| Mucosal edema | 100 (10/10) | 27 (17/64) | < 0.001a |
| Rough/granular surface | 100 (10/10) | 19 (12/64) | < 0.001a |
| Villous blunting | 90 (9/10) | 23 (15/64) | < 0.001a |
| Lymphangiectasia-like change | 80 (8/10) | ||
| Mosaic pattern | 40 (4/10) | ||
| Mucosal hyperemia | 60 (6/10) | 16 (10/64) | 0.005a |
| Nodular elevations | 40 (4/10) | 6 (4/64) | 0.010a |
| Erosion | 20 (2/10) | 17 (11/64) | 1.000 |
| Ulceration | 30 (3/10) | 66 (42/64) | 0.042a |
| Mass-like lesion | 0 | 16 (10/64) | 0.339 |
With respect to the sites of involvement, the small intestine was the primary affected organ (25/26, 96.2%), with only 4 patients (15.4%) showing colorectal involvement and no cases involving the stomach. Bone marrow aspiration was performed in 10 patients, with suspected involvement in 2. Among the 18 patients who underwent abdominal CT, the imaging features included bowel wall thickening (15/18, 83.3%), multiple enlarged lymph nodes (11/18, 61.1%), and bowel dilation (7/18, 38.9%).
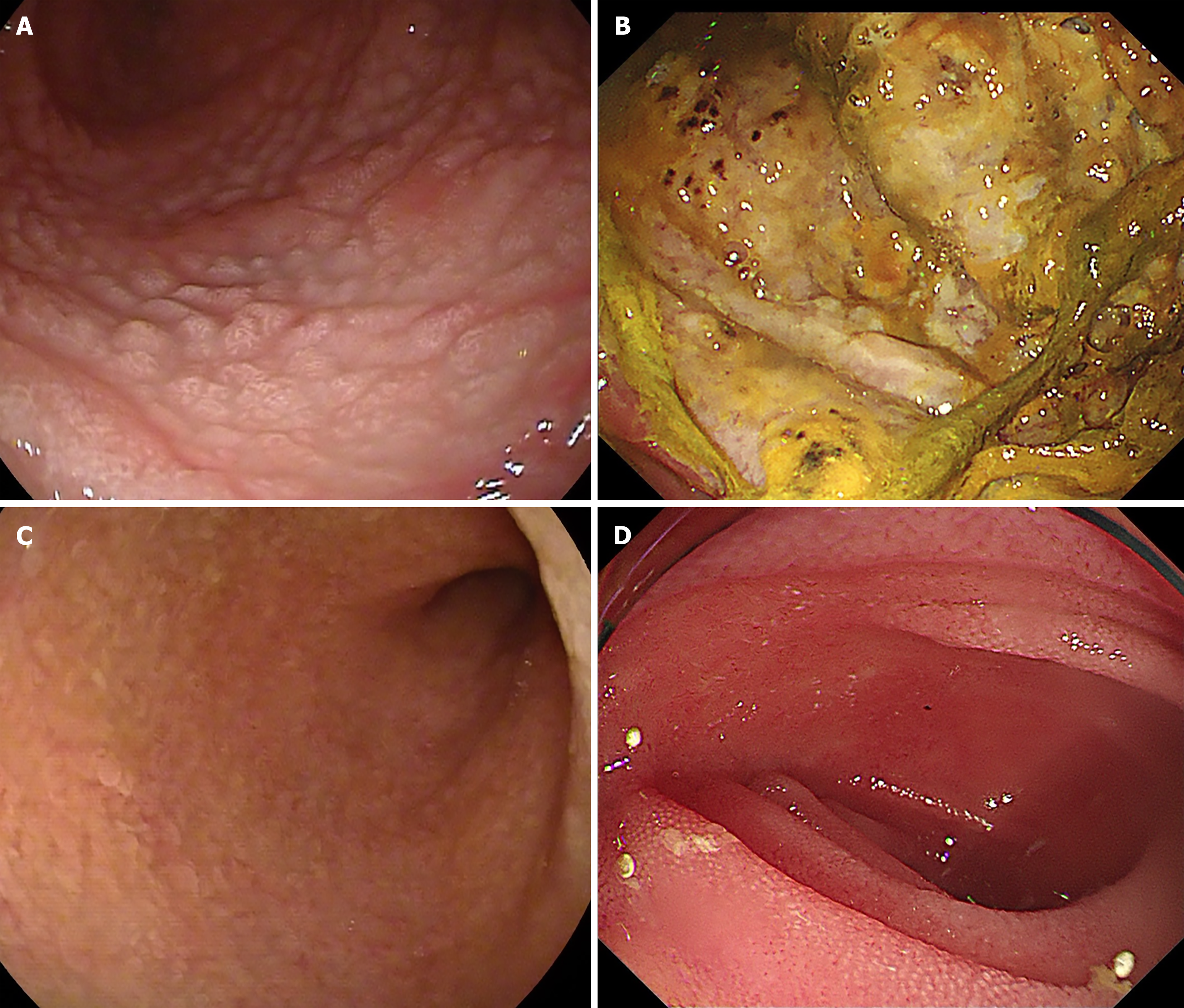
Ten of the 26 patients underwent endoscopic examination at PUMCH. Sites of gastrointestinal involvement included the duodenum (2/10), jejunum (3/10), proximal ileum (3/10), distal ileum (4/10), and colorectum (1/10). Endoscopic findings were generally subtle, including mucosal edema (10/10, 100%), mucosal roughness or granular surface (10/10, 100%), villous blunting (9/10, 90.0%), lymphangiectasia-like changes (8/10, 80.0%), blurred vascular patterns (6/10, 60.0%), mucosal hyperemia (6/10, 60.0%), mosaic patterns (4/10, 40.0%), nodular elevations (4/10, 40.0%), ulceration (3/10, 30.0%), erosion (2/10, 20.0%), and intestinal stricture (3/10, 30.0%). The endoscopic images from some typical patients are shown in Figure 1. The diagnostic yield of endoscopic biopsy was 50.0% (13/26), with the remaining 13 patients requiring surgical or core needle specimens for diagnosis.
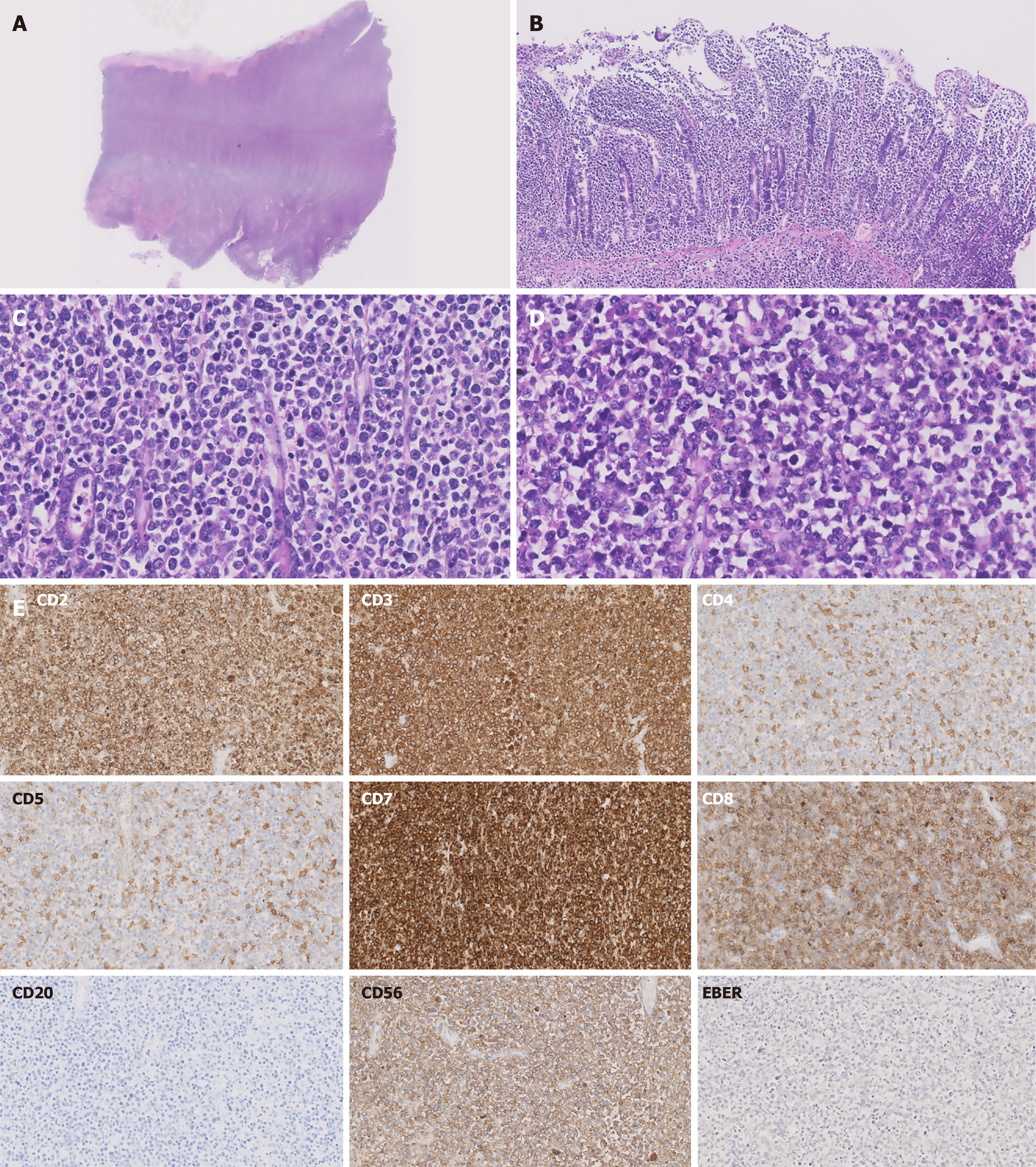
Pathologically, 23 patients provided analyzable reports. The typical histological features of MEITL included diffuse infiltration of monomorphic medium-sized lymphocytes with round nuclei and pale cytoplasmic borders and prominent epitheliotropism, usually without necrosis or a significant inflammatory response[6]. The tumor cells exhibited high proliferative activity, with Ki-67 overexpression (> 50%) observed in 19/23 patients (82.6%). Representative histopathological and immunophenotypic findings are shown in Figure 2. The immunohistochemical findings were as follows: EBER- (17/17, 100%), CD7+ (14/14, 100%), CD3+ (21/22, 95.5%), CD5- (16/17, 94.1%), CD10- (13/14, 92.9%), CD4- (15/17, 88.2%), CD2+ (9/11, 81.8%), CD56+ (16/20, 80.0%), TIA-1+ (12/15, 80.0%), Bcl2+ (8/10, 80.0%), and CD8+ (16/21, 76.2%). A subset of patients were CD20+ (6/22, 27.2%).
As shown in Table 1, among the 130 literature-reported MEITL patients, the male-to-female ratio was approximately 2:1. In terms of clinical features, only the incidence of weight loss was significantly lower in the PUMCH cohort (P = 0.012), while the incidence of other symptoms was not significantly different. Laboratory findings revealed that LDH was significantly more frequently elevated (11/35, 31.4%) in the literature cohort than in the PUMCH cohort (P = 0.011).
In terms of sites of involvement, the literature cohort included the stomach (13/126, 10.3%), central nervous system (13/130, 10.0%), lungs and thoracic cavity (19/130, 14.6%), and bone marrow (10/130, 7.7%), with occasional reports of skin, liver, gallbladder, bladder, pancreas, and ocular involvement[7-12]. Imaging features such as bowel wall thickening (P = 0.037) and multiple lymph node enlargement (P = 0.001) were significantly less frequent in the literature cohort than in the PUMCH cohort.
Endoscopic findings were described for 64 cases in the literature, with notable differences from those in the PUMCH cohort. Unlike the PUMCH cohort, ulceration was the most common feature in the literature cohort (42/64, 65.6%), and the difference was statistically significant (P = 0.042). In contrast, compared with those in the PUMCH cohort, subtle mucosal changes, including mucosal edema (17/64, 26.6%), mucosal roughness or granular surface (12/64, 18.8%), villous blunting (15/64, 23.4%), and mucosal hyperemia (10/64, 15.6%), were significantly less frequent in the literature cohort. Additionally, mass-like lesions were observed in 15.6% of the literature cases (10/64), while no mass-like lesions were identified in the PUMCH cohort. Statistical analysis indicated that patients without endoscopic ulcers were more likely to present with diarrhea (P = 0.002), weight loss (P = 0.003), and elevated CD4+ expression (P = 0.02), suggesting that these patients may exhibit a chronic, cachectic clinical phenotype.
The pathological characteristics of patients in the literature cohort were consistent with those in the PUMCH cohort. Additionally, some patients in the literature cohort exhibited the following immunophenotypic features: CD79α- (87.5%, 14/16), CD30- (90.9%, 50/55), CD103+ (90.9%, 10/11), Pax5- (100%, 8/8), cyclinD1- (100%, 12/12), and Bcl6- (92.3%, 12/13).
By combining the PUMCH cohort (26 cases) with the literature cohort (130 cases), a total of 156 patients were included and stratified into a surgical group (78 patients) and a nonsurgical group (76 patients) on the basis of whether surgery was performed prior to diagnosis. The nonsurgical group exhibited a longer disease course than the surgical group. And the nonsurgical group predominantly exhibited chronic symptoms, with higher incidences of diarrhea (54.0% vs 24.4%, P < 0.001) and weight loss (65.8% vs 37.2%, P < 0.001). In contrast, the surgical group mainly presented with acute manifestations, with a significantly higher rate of intestinal perforation (53.8% vs 14.5%, P < 0.001). The nonsurgical group also had significantly greater involvement of the stomach (13.5% vs 2.6%, P = 0.016) and colorectum (35.1% vs 13.2%, P = 0.002), whereas small intestinal involvement was comparable between the groups (P = 0.322).
Immunohistochemical analysis revealed that the surgical group had significantly higher positivity rates for TIA-1 (100% vs 80.0%, P = 0.005) and granzyme B (80.6% vs 50.0%, P = 0.015), indicating stronger cytotoxicity and more aggressive tumor behavior. Previous studies have reported that the expression of granzyme B, TIA-1, and perforin is associated with activated cytotoxic phenotypes and that the positive expression of these genes is positively correlated with the depth of tumor infiltration[13]. The detailed data are presented in Table 2.
| Characteristic | Total (n = 156) | Non-surgical group (n = 76) | Surgical group (n = 78) | P value |
| Male | 66 (102/155) | 70 (53/76) | 62 (48/77) | 0.334 |
| Female | 34 (53/155) | 30 (23/76) | 38 (29/77) | 0.334 |
| Clinical symptoms | ||||
| Abdominal pain | 60 (94/156) | 55 (42/76) | 65 (51/78) | 0.199 |
| Diarrhea | 38 (60/156) | 54 (41/76) | 24 (19/78) | < 0.001a |
| Constipation | 3 (5/156) | 3 (2/76) | 4 (3/78) | 0.671 |
| Vomiting | 9 (14/156) | 7 (5/76) | 12 (9/78) | 0.284 |
| Abdominal distension | 21 (32/156) | 22 (17/76) | 19 (15/78) | 0.631 |
| Hematochezia | 7 (11/156) | 8 (6/76) | 6 (5/78) | 0.721 |
| Fever | 41 (29/70) | 43 (16/37) | 41 (13/32) | 0.826 |
| Weight loss | 51 (79/156) | 66 (50/76) | 37 (29/78) | < 0.001a |
| Intestinal obstruction | 19 (30/156) | 20 (15/76) | 19 (15/78) | 0.937 |
| Intestinal perforation | 34 (53/156) | 14 (11/76) | 54 (42/78) | < 0.001a |
| Ascites | 6 (10/156) | 7 (5/76) | 6 (5/78) | 0.966 |
| Bleeding | 3 (4/156) | 5 (4/76) | 0 | 0.057 |
| Sites of involvement | ||||
| Stomach | 9 (13/152) | 14 (10/74) | 3 (2/76) | 0.016a |
| Small intestine | 86 (130/152) | 82 (61/74) | 88 (67/76) | 0.322 |
| Colon and rectum | 24 (36/152) | 35 (26/74) | 13 (10/76) | 0.002a |
| Laboratory findings | ||||
| Anemia | 61 (44/72) | 68 (27/40) | 53 (17/32) | 0.213 |
| Hypoalbuminemia | 89 (51/57) | 89 (32/36) | 90 (19/21) | 1.000 |
| Elevated CRP | 87 (34/39) | 87 (20/23) | 88 (14/16) | 1.000 |
| Pathological features | ||||
| CD2+ | 74 (20/27) | 86 (12/14) | 58 (7/12) | 0.190 |
| CD3+ | 99 (136/138) | 98 (61/62) | 99 (73/74) | 1.000 |
| CD4- | 81 (88/109) | 84 (41/49) | 78 (46/59) | 0.456 |
| CD5- | 86 (83/96) | 92 (35/38) | 82 (47/57) | 0.232 |
| CD7+ | 100 (58/58) | 100 (32/32) | 100 (25/25) | 1.000 |
| CD8+ | 90 (121/135) | 88 (57/65) | 91 (62/68) | 0.513 |
| CD56+ | 95 (129/136) | 94 (60/64) | 97 (68/70) | 0.425 |
| CD20+ | 13 (12/93) | 20 (7/35) | 9 (5/56) | 0.129 |
| TIA-1+ | 93 (65/70) | 80 (20/25) | 100 (43/43) | 0.005a |
| Granzyme B+ | 68 (40/59) | 50 (11/22) | 81 (29/36) | 0.015a |
In this study, the data from 26 MEITL patients diagnosed at PUMCH were integrated with the data from 130 globally reported cases to systematically analyze and summarize the clinical, pathological, and endoscopic features of the disease. MEITL patients commonly present with weight loss, abdominal pain, and diarrhea, although some cases present acutely with perforation or obstruction, indicating heterogeneity in clinical phenotypes. Endoscopic features in the PUMCH cohort were dominated by mucosal edema, roughness, and villous blunting, whereas ulcers were more frequently observed in the literature cohort, suggesting differences across populations or timing of diagnosis. The pathological diagnosis relied on the histological hallmark of diffuse infiltration by monomorphic small-to-medium lymphocytes with epitheliotropism and a typical immunophenotype (CD3+, CD8+, CD56+, and EBER-), although some patients exhibited histological and immunological heterogeneity.
Notably, all 8 MEITL patients tested for β2-MG in this study had elevated serum levels. However, owing to the limited number of tested cases and the absence of published literature on the association between β2-MG and MEITL, the clinical significance of this finding requires further validation. Previous studies have confirmed that the serum β2-MG level can serve as an independent prognostic factor in peripheral T-cell lymphoma, not otherwise specified, with elevated levels indicating poorer outcomes[14]. Moreover, simultaneously increased serum levels of β2-MG, LDH, and CRP have been identified as an independent risk factor for disease progression and survival in patients with diffuse large B-cell lymphoma[15]. These findings suggest that β2-MG, in combination with other clinical or molecular markers (such as LDH, CRP, tumor protein 53 mutations and MYC abnormalities), could be incorporated into a diagnostic or prognostic evaluation model applicable to MEITL. However, this hypothesis remains to be validated through large-sample studies.
Endoscopic manifestations of MEITL exhibit notable population heterogeneity and disease-stage associations. In the PUMCH cohort, patients predominantly showed mucosal edema, roughness, a granular surface, and villous blunting, while the incidence of ulceration was significantly lower than that reported in the literature (30% vs 66%). These findings are consistent with those of Ishibashi et al[16], who reported that diffuse mucosal edema and granular changes, with or without villous atrophy, serve as important early endoscopic clues for MEITL[16], whereas ulcers typically appear during disease progression. Further analysis of the literature cohort revealed that non-Asian patients had a higher incidence of intestinal obstruction prior to diagnosis (P = 0.047). In line with the study by Veloza et al[17], these findings suggest that Western patients may develop acute and severe symptoms because of rapid disease progression.
In terms of diagnostic efficiency, the endoscopic diagnostic rate of MEITL in the PUMCH cohort was only 50.0%, primarily because of the lack of specific endoscopic features. In addition to common findings such as mucosal edema and villous blunting, some patients exhibit a “mosaic pattern”, which can easily be confused with inflammatory bowel disease or malabsorption syndromes[5,18]. This underscores the need for clinicians to maintain a high index of suspicion for these nonspecific endoscopic changes. Early recognition may allow for a definitive diagnosis and treatment before perforation or obstruction occurs, thereby improving therapeutic opportunities and the prognosis. Further analysis of the literature cohort revealed that patients with small intestinal involvement had a significantly lower endoscopic diagnostic rate than those with upper or lower gastrointestinal involvement and were more likely to have deeper mucosal infiltration. Considering the complex anatomy of the small intestine and the hidden nature of deep lesions, for suspected cases, clinicians should adopt a pathological strategy of "multiple-site and deep-tissue sampling", combined with imaging modalities such as double-balloon enteroscopy and contrast-enhanced abdominal CT, to improve diagnostic sensitivity and avoid delays caused by relying on a single examination[19].
In summary, in clinical practice, the presence of nonspecific endoscopic findings such as mucosal edema, rough or granular mucosa, and villous blunting in the small intestine should raise suspicion for MEITL. Multiple-site, multisample biopsy, supplemented when necessary with enteroscopy and imaging studies (such as CT, positron emission tomography-CT, etc.), is recommended to improve the diagnostic yield[20]. Failure to identify and diagnose the disease early often allows progression to deep ulceration, ultimately resulting in severe complications such as perforation or bleeding, which significantly increase the treatment difficulty and mortality risk.
Histopathological examination is the gold standard for diagnosing MEITL. Tumor tissue is generally divided into three regions: The central tumor area, the peripheral zone, and the IEL proliferation area. The central tumor area is characterized by diffuse infiltration of monomorphic small-to-medium lymphocytes; the peripheral zone shows tumor cells extending laterally along the mucosa, and the IEL proliferation area contains small lymphocytes or morphologically near-normal lymphocytes, resembling an inflammatory response, which can easily lead to misdiagnosis[21,22]. Therefore, in clinical pathology practice, multiple biopsies from different sites should be obtained to improve diagnostic accuracy. Notably, some MEITL cases may exhibit atypical pathological features, including nonmonomorphic cells, necrosis, angiocentric infiltration, or a “starry-sky” pattern. Previous studies have suggested that MYC overexpression and tumor protein 53 mutation may be closely associated with these atypical morphological characteristics[23].
The typical immunophenotype of MEITL is CD3+ CD8+ CD56+ EBER-, although approximately 13% (12/93) of patients in this cohort exhibited CD20 positivity. Previous studies have suggested that aberrant expression of B-cell–related markers may be associated with better survival outcomes[17]; however, owing to incomplete prognostic follow-up in this cohort, this observation could not be further validated. CD4 positivity was significantly associated with the absence of endoscopic ulcers, but its clinical significance in MEITL remains unclear. Case reports have indicated that small intestinal CD4+ T-cell lymphomas may represent a subtype with indolent clinical behavior, although this has not been confirmed in large-scale, multicenter studies[24,25]. In MEITL research, CD4+ cases are rare and have not been sufficiently studied. Therefore, recognizing this uncommon variant and excluding other CD4+ T-cell lymphoma subtypes is critical for accurate MEITL diagnosis[26]. These pathological features not only expand the understanding of the biological behavior of MEITL but also provide potential reference markers for precise clinical diagnosis, differential diagnosis, and prognostic stratification.
This study proposed a clinical classification of MEITL patients into two groups on the basis of whether surgical intervention occurred prior to diagnosis: The “acute aggressive type” (surgical group) and the “chronic consumptive type” (nonsurgical group). Although no statistically significant difference in median overall survival was observed between the two subgroups, this classification has important clinical and biological relevance because it accurately delineates two distinct modes of clinical presentation in MEITL, which may reflect inherent heterogeneity in tumor biology.
The acute aggressive type typically manifests as acute abdominal emergencies such as intestinal perforation or obstruction. The expression of cytotoxic proteins such as TIA-1 and granzyme B is high in tumor cells of this subtype, suggesting stronger local tissue invasiveness and destructive potential. In contrast, the chronic consumptive type is characterized by prolonged nonspecific diarrhea and weight loss. Given its indolent and protracted course, this chronic form is particularly susceptible to diagnostic delay. This classification system provides clear warning signs and differential diagnostic clues for clinicians across different specialties (e.g., emergency medicine and gastroenterology), thereby potentially improving early recognition.
Thus, clinical subtyping based on a prediagnostic surgical history is valuable primarily for guiding differentiated clinical management pathways rather than for prognostic stratification. For patients with the chronic consumptive subtype, if unexplained chronic diarrhea is present, MEITL should be considered in the differential diagnosis, and more proactive endoscopic evaluation with deep tissue biopsy should be advocated to reduce diagnostic delay, thereby improving survival outcomes. For those with the acute aggressive subtype, a multidisciplinary approach should be established to ensure seamless integration of emergency surgery and postoperative systemic therapy. Future studies should further integrate molecular and genetic profiles within this clinical framework to elucidate the core mechanisms driving the distinct clinical phenotypes, ultimately paving the way for more targeted and individualized treatment strategies.
The main strengths of this study are as follows. First, it serves as a large-scale retrospective cohort of MEITL patients, providing a comprehensive overview of patients’ clinical, imaging, endoscopic, and pathological features. By integrating data from 130 cases reported in the literature, this study offers a broad perspective on the heterogeneity of this disease. Second, by combining clinical, endoscopic, and pathological characteristics, this study highlights nonspecific endoscopic features such as mucosal edema and villous shortening, which, when interpreted alongside typical immunophenotypes, may improve early recognition and diagnosis of MEITL, potentially enhancing patient outcomes. Finally, on the basis of stratification according to whether surgery was performed prior to diagnosis, this study identified two distinct clinical subtypes of MEITL: The acute highly aggressive form and the chronic cachectic form. This classification has potential utility for early diagnosis and the development of personalized clinical management pathways in MEITL.
However, several limitations should be noted. First, the study is a single-center, retrospective analysis with a relatively small sample size, and incomplete follow-up and laboratory data may limit the representativeness of the findings. Second, the literature cohort may be affected by publication bias and incomplete data, restricting comparability and homogeneity. Third, this study lacks molecular-level analyses, making it difficult to fully elucidate disease mechanisms. Future studies should aim for multicenter collaboration to increase the sample size, enhance case registration and follow-up, and integrate molecular pathology with prospective research to reduce biases and strengthen the reliability of the findings.
This study focuses on enhancing the early diagnosis rate of MEITL, a disease characterized by highly heterogeneous clinical phenotypes ranging from highly aggressive to chronic cachectic presentations. Imaging and endoscopic evaluations play crucial roles in early detection, while histopathological features are closely associated with the underlying biological behavior of the disease. Early recognition and accurate diagnosis are essential for improving patient outcomes, and further exploration of the underlying molecular mechanisms may provide novel avenues for targeted therapies and individualized management strategies.
We sincerely thank the medical staff from the Departments of Gastroenterology, Hematology, and Pathology at Peking Union Medical College Hospital for their valuable support in patient data collection, specimen processing, and endoscopic imaging analysis. We also appreciate the contributions of all study participants and their families for their trust and cooperation throughout the research. Additionally, we would like to acknowledge the colleagues who provided critical comments and suggestions during the manuscript preparation, which greatly improved the quality of this work.
| 1. | Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, Advani R, Ghielmini M, Salles GA, Zelenetz AD, Jaffe ES. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 2016;127:2375-2390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6394] [Cited by in RCA: 5661] [Article Influence: 566.1] [Reference Citation Analysis (6)] |
| 2. | Al Somali Z, Hamadani M, Kharfan-Dabaja M, Sureda A, El Fakih R, Aljurf M. Enteropathy-Associated T cell Lymphoma. Curr Hematol Malig Rep. 2021;16:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 3. | Nishimura MF, Nishimura Y, Nishikori A, Yoshino T, Sato Y. Primary Gastrointestinal T-Cell Lymphoma and Indolent Lymphoproliferative Disorders: Practical Diagnostic and Treatment Approaches. Cancers (Basel). 2021;13:5774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 4. | Lungu M, Oprea VD, Niculeț E, Apostol LL, Păduraru MI, Ionescu AM, Zaharia AL. Leptomeningitis as Rare Secondary Dissemination in MEITL (Monomorphic Intestinal Epitheliotropic T-Cell Lymphoma). Life (Basel). 2025;15:1243. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Ozaka S, Takahashi H, Hamano T, Fukuda M, Mizukami K. Monomorphic Epitheliotropic Intestinal T-cell Lymphoma Presenting With Significant Villous Atrophy in the Small Intestine. Cureus. 2025;17:e79496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 6. | Ondrejka S, Jagadeesh D. Enteropathy-Associated T-Cell Lymphoma. Curr Hematol Malig Rep. 2016;11:504-513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 38] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 7. | Onieva R, Aguirre F, Blázquez C, Carrera R, Piernas S, Ramila ME, Soler A, Ferreres JC, Papaleo N. A case report and a literature review about central nervous system involvement in monomorphic epitheliotropic intestinal T cell lymphoma. J Hematop. 2025;18:7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Olmos-Alpiste F, Vázquez I, Gallardo F, Sánchez-Gonzalez B, Colomo L, Pujol RM. Monomorphic Epitheliotropic Intestinal T-Cell Lymphoma With Secondary Cutaneous Involvement: A Diagnostic Challenge. Am J Dermatopathol. 2021;43:300-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 9. | Okuda T, Shirase T, Nishikubo M, Konishi Y, Takeoka T, Takeshima J, Ito T, Tsuji M. Monomorphic epitheliotropic intestinal T-cell lymphoma with gallbladder involvement: A case report. Mol Clin Oncol. 2025;23:67. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Dong B, Chen Y, Ding B, Li N, Li Z. A case report of monomorphic epitheliotropic intestinal T-cell lymphoma with pulmonary manifestations as the first presentation. BMC Pulm Med. 2025;25:132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 11. | Zhang X, Liu XS, Zhao C, Lai YM, Zhang MF. Ocular manifestations as first signs of systemic T cell lymphoma in two cases. BMC Ophthalmol. 2017;17:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Rand AJ, Cardona DM, Proia AD, Lagoo AS. Clinically undiagnosed enteropathy associated T-cell lymphoma type II presenting with prolonged lower gastrointestinal tract symptoms: report of an autopsy case and review of diagnostic challenges and clinicopathological correlation. J Gastrointest Oncol. 2013;4:103-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 13. | Zhou B, Guo M, Li X, Duan T, Peng L, Hao H. Clinicopathological and molecular genetic alterations in monomorphic-epitheliotropic intestinal T-cell lymphoma of the small intestine. Eur J Med Res. 2024;29:191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 14. | Kim HD, Cho H, Sohn BS, Park CS, Huh J, Ryu JS, Lee SW, Yoon SE, Kim SJ, Ko YH, Kim WS, Suh C. Prognostic significance of serum β2-microglobulin levels in patients with peripheral T-cell lymphoma not otherwise specified. Leuk Lymphoma. 2022;63:124-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 15. | Zhong J, Weng X, Chen L. Prognostic Value of Serum β2-Microglobulin in Predicting Survival of Patients with Diffuse Large B-Cell Lymphoma. Clin Lab. 2025;71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 16. | Ishibashi H, Nimura S, Kayashima Y, Takamatsu Y, Aoyagi K, Harada N, Kadowaki M, Kamio T, Sakisaka S, Takeshita M. Multiple lesions of gastrointestinal tract invasion by monomorphic epitheliotropic intestinal T-cell lymphoma, accompanied by duodenal and intestinal enteropathy-like lesions and microscopic lymphocytic proctocolitis: a case series. Diagn Pathol. 2016;11:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 17. | Veloza L, Cavalieri D, Missiaglia E, Ledoux-Pilon A, Bisig B, Pereira B, Bonnet C, Poullot E, Quintanilla-Martinez L, Dubois R, Llamas-Gutierrez F, Bossard C, De Wind R, Drieux F, Fontaine J, Parrens M, Sandrini J, Fataccioli V, Delfau-Larue MH, Daniel A, Lhomme F, Clément-Filliatre L, Lemonnier F, Cairoli A, Morel P, Glaisner S, Joly B, El Yamani A, Laribi K, Bachy E, Siebert R, Vallois D, Gaulard P, Tournilhac O, De Leval L. Monomorphic epitheliotropic intestinal T-cell lymphoma comprises morphologic and genomic heterogeneity impacting outcome. Haematologica. 2023;108:181-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 62] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 18. | Nozari N. Type II Enteropathy-Associated T-cell Lymphoma: A Rare Report from Iran. Middle East J Dig Dis. 2017;9:55-57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 19. | Naito T, Nosaka T, Takahashi K, Ofuji K, Matsuda H, Ohtani M, Hiramatsu K, Imamura Y, Yamauchi T, Nakamoto Y. Successful early diagnosis of monomorphic epitheliotropic intestinal T-cell lymphoma manifesting as chronic diarrhea and hypokalemia using video capsule endoscopy and double-balloon enteroscopy. Clin J Gastroenterol. 2022;15:913-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 20. | Zhang X, Liu S, Liu Y, Lv Z, Yang J. 18F-Fluorodeoxyglucose PET/CT and MRI Imaging Characteristics of Monomorphic Epitheliotropic Intestinal T-cell Lymphoma: A case report. Curr Med Imaging. 2023;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 21. | Ding X, Zhang M, Zhan Q, Zhang H, Zhang R, Yan X, Zhang L, Wang X. Monomorphic epitheliotropic intestinal T-cell lymphoma: report of four cases and literature review. J Int Med Res. 2024;52:3000605241271756. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 22. | Zhao L, Yang Y, Zhang Y. Type II enteropathy-associated t-cell lymphoma: A case report and literature review. Niger J Clin Pract. 2018;21:812-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 23. | Xiong DT, Cheng F, Xu JZ, Wang JH, Zhang YF, Cai YY, Gan WJ, Li XQ, Wang ZM, Yu F. [Atypical clinicopathological features of monomorphic epitheliotropic intestinal T-cell lymphoma]. Zhonghua Xue Ye Xue Za Zhi. 2025;46:642-646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 24. | Malamut G, Meresse B, Kaltenbach S, Derrieux C, Verkarre V, Macintyre E, Ruskone-Fourmestraux A, Fabiani B, Radford-Weiss I, Brousse N, Hermine O, Cerf-Bensussan N, Cellier C. Small intestinal CD4+ T-cell lymphoma is a heterogenous entity with common pathology features. Clin Gastroenterol Hepatol. 2014;12:599-608.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 51] [Article Influence: 4.3] [Reference Citation Analysis (1)] |
| 25. | Matuchansky C. Small intestinal CD4+ T-cell lymphoma: a subset with possible indolent clinical behavior. Clin Gastroenterol Hepatol. 2015;13:619-620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 26. | El Hussein S. Monomorphic epitheliotropic T-cell lymphoma with strong CD4 expression. Blood. 2024;143:1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |