Published online Apr 28, 2026. doi: 10.3748/wjg.v32.i16.117799
Revised: January 18, 2026
Accepted: February 12, 2026
Published online: April 28, 2026
Processing time: 121 Days and 23.6 Hours
Gastric cancer remains one of the leading causes of cancer-related mortality worldwide, and surgery remains the cornerstone of curative treatment. However, the extent of surgical resection may significantly influence postoperative immune and inflammatory responses, which are closely related to recovery and compli
To compare the effects of super minimally invasive surgery (SMIS) and organ-resection surgery (ORS) for gastric cancer on postoperative immune and inflammatory responses.
This prospective study included patients with gastric cancer who underwent SMIS or conventional ORS between May 2023 and April 2024. Peripheral blood samples were collected one day preoperatively and on the third postoperative day. Plasma levels of 92 trauma- and immunity-related proteins were quantified using the Olink proteomic platform. Differentially expressed proteins (DEPs) before and after surgery were identified within each group, and comparative analyses were performed to assess immune-inflammatory alterations between SMIS and ORS.
A total of 38 patients were included: 20 in the SMIS group and 18 in the ORS group. Baseline demographic and clinical characteristics were comparable between the groups (all P > 0.05). Proteomic analysis identified 13 DEPs after SMIS and 37 DEPs after ORS. Nine DEPs were shared between the surgical models (TMPRSS15, CNTN2, HPGDS, PLXDC1, NPPC, CA14, calcitonin, platelet-derived growth factor C, and nitric oxide synthase). Among these, calcitonin, platelet-derived growth factor C, and nitric oxide synthase showed the most prominent postoperative upregulation, particularly after ORS, indicating a stronger trauma-related immune response than SMIS.
SMIS for gastric cancer was associated with a reduced postoperative immune-inflammatory response and lower trauma-related protein expression than ORS, suggesting better immune preservation and lower surgical stress.
Core Tip: The optimal extent of surgical intervention for gastric cancer remains controversial, particularly with respect to its impact on postoperative immune homeostasis. This study integrates super minimally invasive surgery concepts with proteomic profiling to investigate immune-inflammatory alterations following different surgical strategies, providing mechanistic insights that may support more individualized, trauma-conscious surgical decision-making in early gastric cancer.
- Citation: Yuan YQ, Chen QQ, Xiao P, Li HK, Lv KM, Tian S, Shi H, Hao J, Yan Y, Linghu EQ. Plasma proteomic profiling of gastric cancer: Super minimally invasive surgery vs organ-resection on trauma immunity. World J Gastroenterol 2026; 32(16): 117799
- URL: https://www.wjgnet.com/1007-9327/full/v32/i16/117799.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i16.117799
Gastric cancer is one of the most prevalent malignant neoplasms globally, with high incidence and mortality rates that pose a significant public health threat[1]. In China, it ranks as the second most common cancer, substantially affecting public health and life expectancy[2]. The high burden of gastric cancer highlights the critical need for early diagnosis and treatment. Advances in medical technology have led to diversified treatment approaches, with surgery, chemotherapy, radiotherapy, targeted therapy, and immunotherapy as the primary modalities[3]. Among these, surgery remains the mainstay of treatment for gastric cancer and the only curative option.
Surgical intervention inevitably causes direct mechanical tissue injury, local hemorrhage, and ischemia, followed by hypoxic cellular stress and ischemia-reperfusion injury, which activate innate and adaptive immune responses at both local and systemic levels[4,5]. Dysregulation of the host response increases the incidence of infectious complications, impairs wound healing, and adversely affects other clinical outcomes, including length of hospital stay, mortality, and long-term cognitive function[6,7]. However, the clinical manifestations of postoperative inflammation can be insidious[8].
Among the various surgical approaches, super minimally invasive surgery (SMIS) and conventional organ-resection surgery (ORS) are the two predominant treatment modalities in current clinical practice. SMIS offers several advantages over ORS, including reduced trauma, preservation of organ function, lower postoperative pain, and a reduced risk of infection and contamination[9]. SMIS has become a standard of care for early-stage gastric cancer. However, SMIS has limitations, including the need for surgeons to have advanced endoscopic skills to perform the procedure successfully. Accordingly, this exploratory pilot study aims to compare plasma levels of organ damage-related molecules following SMIS and ORS to determine which surgical method is more deleterious to the human body, an area that has not been previously explored. The findings will provide a scientific foundation for the targeted development of postoperative intervention strategies.
This study was designed as a prospective, exploratory pilot study. In accordance with current clinical guidelines and ethical standards, the surgical strategy (SMIS or ORS) was determined by clinicians based on patients’ clinicopathologic characteristics (e.g., tumor stage and differentiation). Because the extent of surgery is strictly dictated by disease severity, random allocation was not feasible in this real-world clinical setting.
The study enrolled 38 consecutive patients with gastric cancer (20 in the SMIS group and 18 in the ORS group) who were hospitalized at the First Medical Center of Chinese PLA General Hospital between May 2023 and April 2024 and scheduled for surgical treatment. Written informed consent was obtained from all participants. Ethical approval was granted by the Ethics Committee of the Chinese PLA General Hospital, No. S2023-188-01.
The surgical approaches for the two groups were as follows: (1) The SMIS group underwent endoscopic high-frequency electrodissection, endoscopic mucosal resection, pre-incision endoscopic mucosal resection, or endoscopic submucosal dissection; and (2) The ORS group underwent laparoscopic-assisted radical gastrectomy for gastric cancer, robot-assisted radical total gastrectomy, laparoscopic-assisted radical resection of distal gastric cancer, laparoscopic total gastrectomy, or total gastrectomy.
In this study, the decision to perform SMIS or ORS was made by clinicians based on patients’ clinicopathologic characteristics. The distinction between the groups was based on the extent of surgical intervention, specifically whether the stomach was partially or completely removed.
Comparative analyses were performed across the following subgroups: Preoperative vs postoperative comparisons within the SMIS group and within the ORS group. The subgroups were defined as follows: GEP = SMIS preoperative, GEA = SMIS postoperative, GSP = ORS preoperative, and GSA = ORS postoperative.
Data collected included: (1) General data. Sex, age, body mass index (BMI), smoking history, and alcohol consumption history; and (2) Clinical characteristics and treatment data. Past medical history, prior medication use, preoperative medication use, Helicobacter pylori infection history, tumor pathology type, preoperative blood counts, surgical procedure, postoperative temperature changes, antibiotic use during hospitalization, antipyretic and analgesic drug use during hospitalization, time to discharge after surgery, and hospitalization costs.
Inclusion criteria: (1) Pathologically confirmed diagnosis of gastric cancer or high-grade intraepithelial neoplasia; (2) Elective surgery with signed informed consent for surgical treatment; (3) Age 18-80 years, male or female; (4) Willingness to cooperate with the study; and (5) No contraindications to endoscopic or surgical procedures.
Exclusion criteria: (1) Documented history of gastrointestinal surgery; (2) History of palliative resection surgery; (3) Presence of malignant tumors at other sites; (4) Severe organ dysfunction; (5) Administration of antibiotics or anti-inflammatory medications within 1 week before surgery; (6) Intellectual disability; (7) Patients who did not meet surgical indications or refused the procedure; and (8) Patients requiring other study-related treatments due to concurrent illnesses during the study period.
Normalized protein expression: The blood sample collection protocol was as follows. Samples were collected from two patient groups (SMIS and ORS) at two time points. The first set of samples was collected 1 day before the surgical procedure, and the second set was collected on postoperative day (POD) 3. Specifically, preoperative sampling was chosen to capture patients’ baseline immune status, independent of the acute stress, anesthesia, or blood transfusions associated with surgery. This 24-hour window also minimizes potential confounding from longer-term variables such as tumor progression or nutritional changes. The selection of POD 3 is based on the biological kinetics of the systemic inflammatory response, as inflammatory mediators, including cytokines, chemokines, and damage-associated molecular patterns, typically reach significant or peak levels 48-72 hours after surgical trauma. Furthermore, POD 3 sampling avoids immediate, transient fluctuations related to residual anesthesia, fluid resuscitation, and the acute hormonal stress response that are most pronounced within the first 24 postoperative hours[10,11]. Samples were then centrifuged at 3500 rpm (2500 × g) for 10 minutes at 4 °C. After centrifugation, the samples were stored at -80 °C.
EDTA plasma samples from 20 patients in the SMIS group and 18 in the ORS group (40 plasma samples from the SMIS group and 36 plasma samples from the ORS group) were analyzed using the Olink Proseek Organ Injury Panel (PEA technology; Shanghai Biotech, Shanghai, China) to assess organ damage markers. The panel includes 92 proteins and uses protein-binding plates in which targets are recognized by pairs of oligonucleotide-labeled antibody probes. The resulting DNA sequences were quantified by microfluidic real-time polymerase chain reaction amplification of the oligonucleotide sequences. Internal and external controls were used to generate threshold cycle (Ct) data for quality control and normalization. Normalized protein expression (NPX) values were calculated by normalizing Ct values using the extension control, inter-plate control, and calibration factor. NPX values were used as the final assay readout; NPX represents relative protein abundance after log2 transformation of the protein values by Olink. Additional details are available on the Olink website (https://www.olink.com).
Principal component analysis: Principal component analysis aggregates signal values from protein samples to generate a new set of composite variables. Typically, two to three composite variables are selected to capture as much information as possible from the original variables, thereby reducing dimensionality based on the specific problem being addressed.
Cluster analysis: Cluster analysis is a widely used exploratory data analysis technique that groups and classifies data based on similarity. Patterns within clusters are highly similar, whereas similarities between different clusters are comparatively low.
Differential protein analysis: The method for screening differentially expressed proteins (DEPs) involved using the PEA technique, the core technology of the Olink proteomics assay, to analyze plasma samples. Differential protein analysis was performed using normalized NPX values. NPX is a quantitative representation of protein levels that aims to minimize variation between internal test components and external standards. Olink standard difference analysis utilizes Student’s t-test (or ANOVA when comparing more than two groups) to calculate protein expression differences from normalized NPX data under various experimental conditions. Differential protein information is identified by screening for conditions with P values < 0.05 (Student’s t-test). Subsequent differential protein expression analysis is then performed, leading to the creation of heat maps and box plots.
Gene Ontology and Kyoto Encyclopedia of Genes and Genomes enrichment analysis: Functional enrichment analysis was performed only for statistically significant DEPs (P < 0.05). These DEPs were compared with the full panel of 92 proteins quantified in the Olink assay, which served as the background reference set. Enrichment significance was evaluated using Fisher’s exact test (P < 0.05) to identify overrepresented Gene Ontology (GO) terms and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways. The analysis was conducted at the GO term or KEGG pathway level, providing insight into overall functional enrichment patterns among the DEPs.
Statistical analyses were performed using SPSS version 27.0. Continuous variables are presented as the mean ± SD or median (interquartile range), as appropriate. Comparisons between two independent groups were performed using the independent-samples t test or the Mann-Whitney U test, depending on the data distribution. Paired comparisons before and after surgery within the same group were conducted using paired t tests or Wilcoxon signed-rank tests. Categorical variables were compared using the χ2 test or Fisher’s exact test, as appropriate. A generalized estimating equation was used to analyze repeated measurements over time. All statistical tests were two-sided, and a P value < 0.05 was considered statistically significant. Owing to the exploratory nature of this study, no adjustment for multiple comparisons was performed.
A total of 38 patients were included in the study, with 20 in the SMIS group and 18 in the ORS group. The SMIS group comprised 18 males and 2 females, with a mean age of 61 years (range, 55.5-72 years), whereas the ORS group comprised 13 males and 5 females, with a mean age of 60.5 years (range, 46.5-65 years). As shown in Table 1, no statistically significant differences were observed between the groups in sex, age, BMI, history of Helicobacter pylori infection, history of chronic diseases, preoperative blood counts, prior medication use, smoking, or alcohol consumption (all P > 0.05).
| Variables | SMIS (n = 20) | ORS (n = 18) | P value |
| Gender | 0.164 | ||
| Male | 18 (90) | 13 (72) | |
| Female | 2 (10) | 5 (28) | |
| Age (years) | 61 (55.5, 72) | 60.5 (46.5 65) | 0.219 |
| BMI (kg/m2) | 24.95 (23.6, 27.25) | 22.95 (21.15, 26.05) | 0.106 |
| Smoking history | 7 (35) | 7 (39) | 1.000 |
| History of alcohol use | 10 (50) | 9 (50) | 1.000 |
| History of Helicobacter pylori infection | 3 (15) | 3 (17) | 1.000 |
| Hypertension | 10 (50) | 6 (33) | 0.342 |
| Diabetes | 5 (25) | 4 (22) | 1.000 |
| Coronary heart disease, pre-operative blood test | 4 (20) | 1 (6) | 0.344 |
| White blood cells (× 109/L) | 6.085 (5.515, 7.57) | 5.57 (4.385, 7.185) | 0.148 |
| Neutrophils (× 109/L) | 0.558 ± 0.721 | 0.608 ± 0.949 | 0.076 |
| Lymphocytes (× 109/L) | 0.335 ± 0.729 | 0.293 ± 0.853 | 0.12 |
| Monocytes (× 109/L) | 0.0775 (0.065, 0.088) | 0.074 (0.057, 0.082) | 0.342 |
| Previous medication history | 1.000 | ||
| Anti-inflammatory drugs (anticoagulants, anti-platelet drugs, non-steroidal anti-inflammatory drugs, hormonal drugs) | 2 (10) | 1 (5.6) | |
| No anti-inflammatory medication | 18 (90) | 17 (94.4) |
A comparison of clinical characteristics and treatment plans showed statistically significant differences between the two groups in pathological tumor type (P < 0.001), type of postoperative antibiotics (P < 0.05), duration of antibiotic therapy (P < 0.001), use of analgesia (P < 0.001), length of postoperative hospital stay (P < 0.05), postoperative body temperature, and postoperative hospitalization costs (P < 0.001) (Table 2).
| SMIS (n = 20) | ORS (n = 18) | P value | |
| Tumor pathology type | < 0.001 | ||
| Severe atypical hyperplasia (high-grade intraepithelial neoplasia) | 4 (20) | 0 | |
| High-grade intraepithelial neoplasia with carcinoma | 9 (45) | 0 | |
| Moderately differentiated adenocarcinoma | 3 (15) | 0 | |
| Poorly differentiated adenocarcinoma | 0 | 6 (33.3) | |
| Low and intermediate grade glandular cancer | 2 (10) | 3 (16.7) | |
| Signet ring cell carcinoma | 1 (5) | 0 | |
| Low-to-moderately differentiated adenocarcinoma combined with signet ring cell carcinoma | 0 | 4 (20) | |
| Undifferentiated adenocarcinoma combined with signet ring cell carcinoma | 1 (5) | 5 (25) | |
| Number of types of antibiotics used | 0.03829 | ||
| Use of 1 antibiotic | 9 (45) | 2 (11) | |
| Use of 2 antibiotics | 4 (20) | 7 (39) | |
| Use of 3 antibiotics | 5 (25) | 9 (50) | |
| Use of 4 antibiotics | 0 | 0 | |
| No antibiotics used | 2 (10) | 0 | |
| Duration of antibiotic use | 2 (2, 3) | 6 (5.5, 7) | < 0.001 |
| Postoperative analgesia | < 0.001 | ||
| Yes | 2 (10) | 17 (94.4) | |
| No | 18 (90) | 1 (5.6) | |
| Length of stay after surgery | 5 (5, 7) | 7 (6, 7.5) | 0.002849 |
| Hospitalization costs | 24710.9 (20873.79, 31738.355) | 93442.45 (84969.0, 101357.365) | < 0.001 |
| Postoperative temperature | 0.01544 | ||
| Low-grade fever (37.3-38.0 °C) | 1 (5) | 7 (38.9) | |
| Medium fever (38.1-39.0 °C) | 2 (10) | 1 (5.6) | |
| High fever (> 39.1 °C) | 0 | 1 (5.6) | |
| Normal temperature | 17 (85) | 9 (50) |
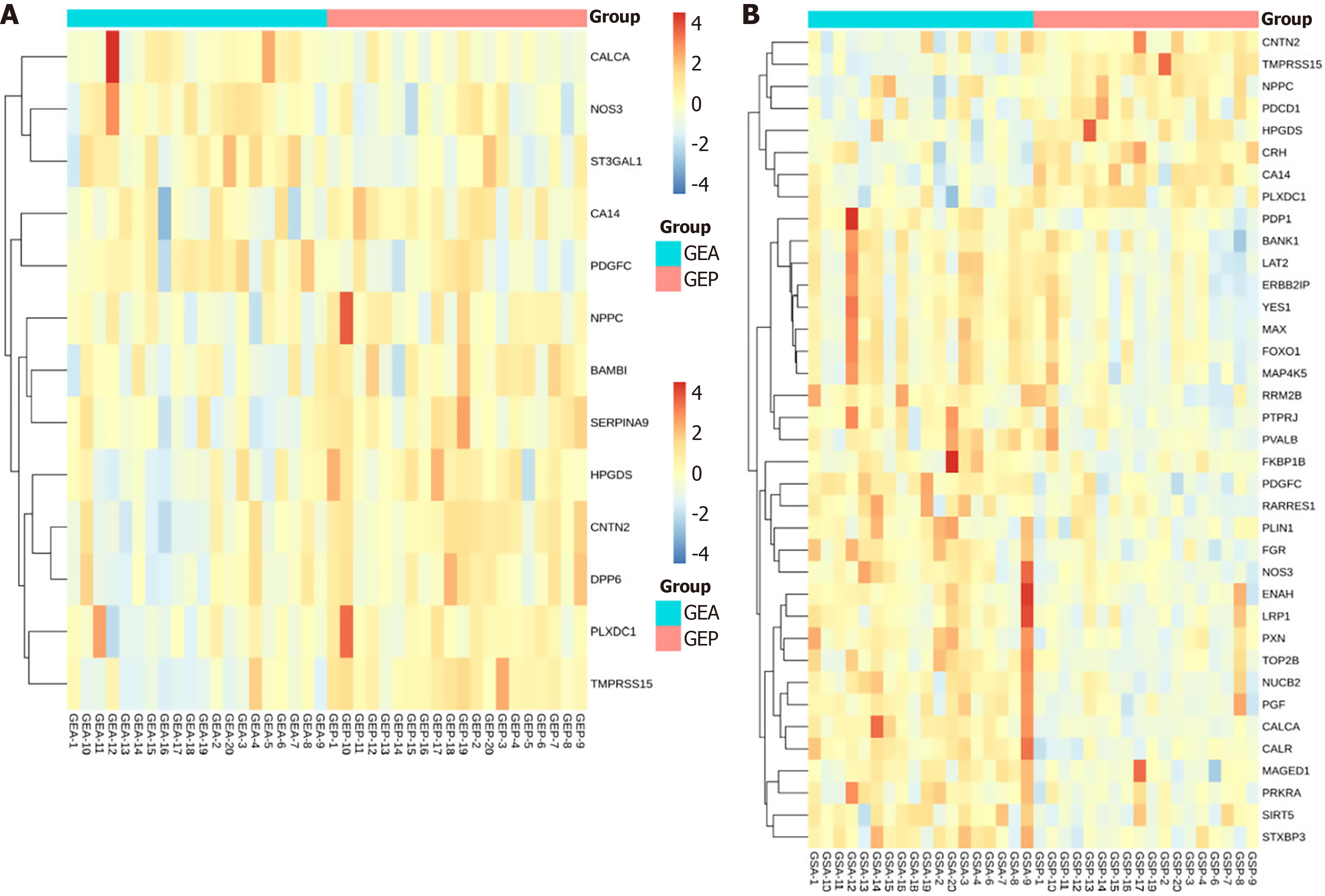
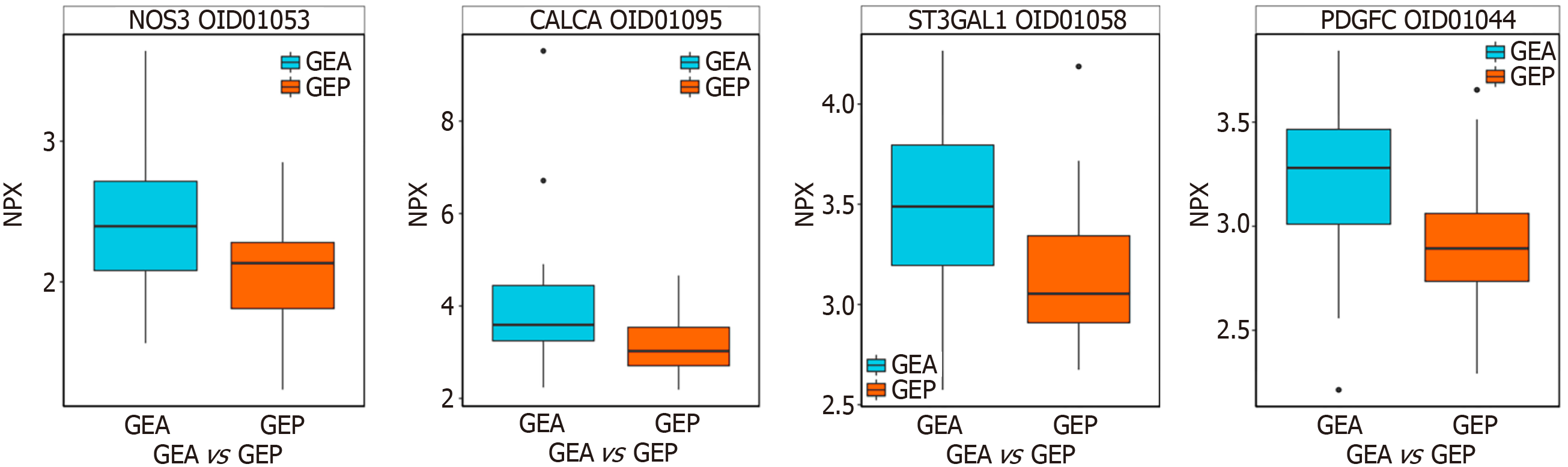
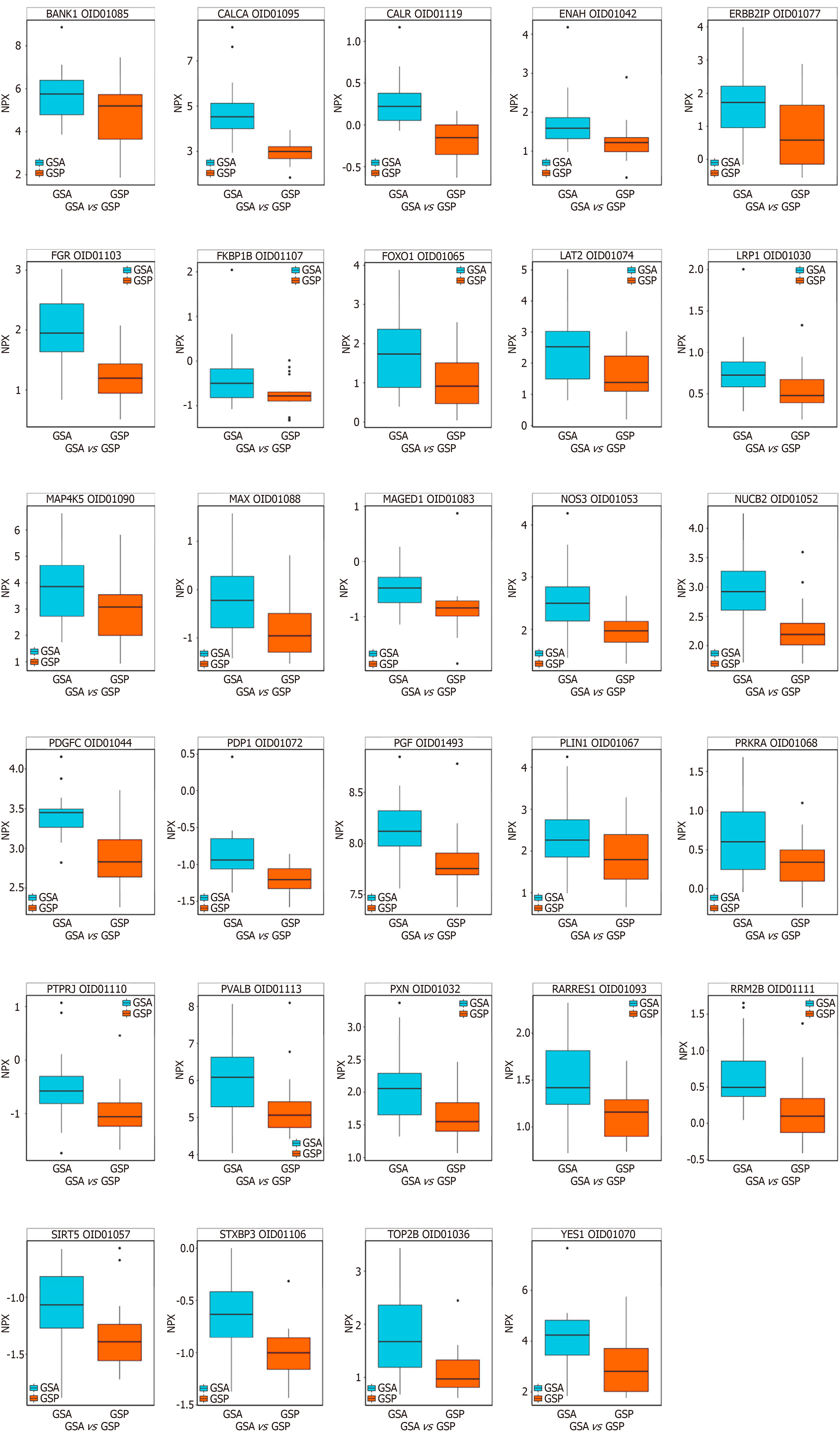
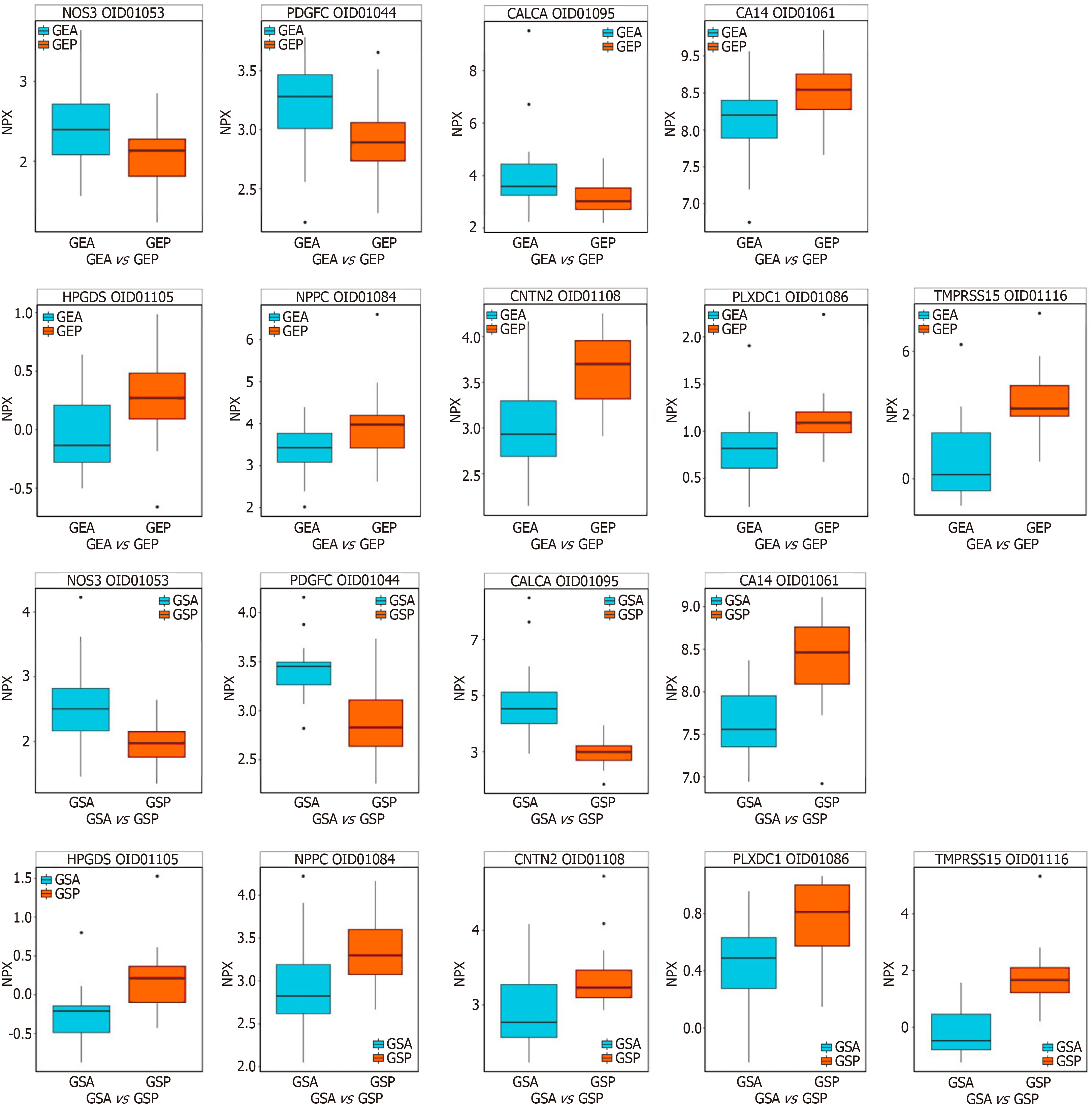
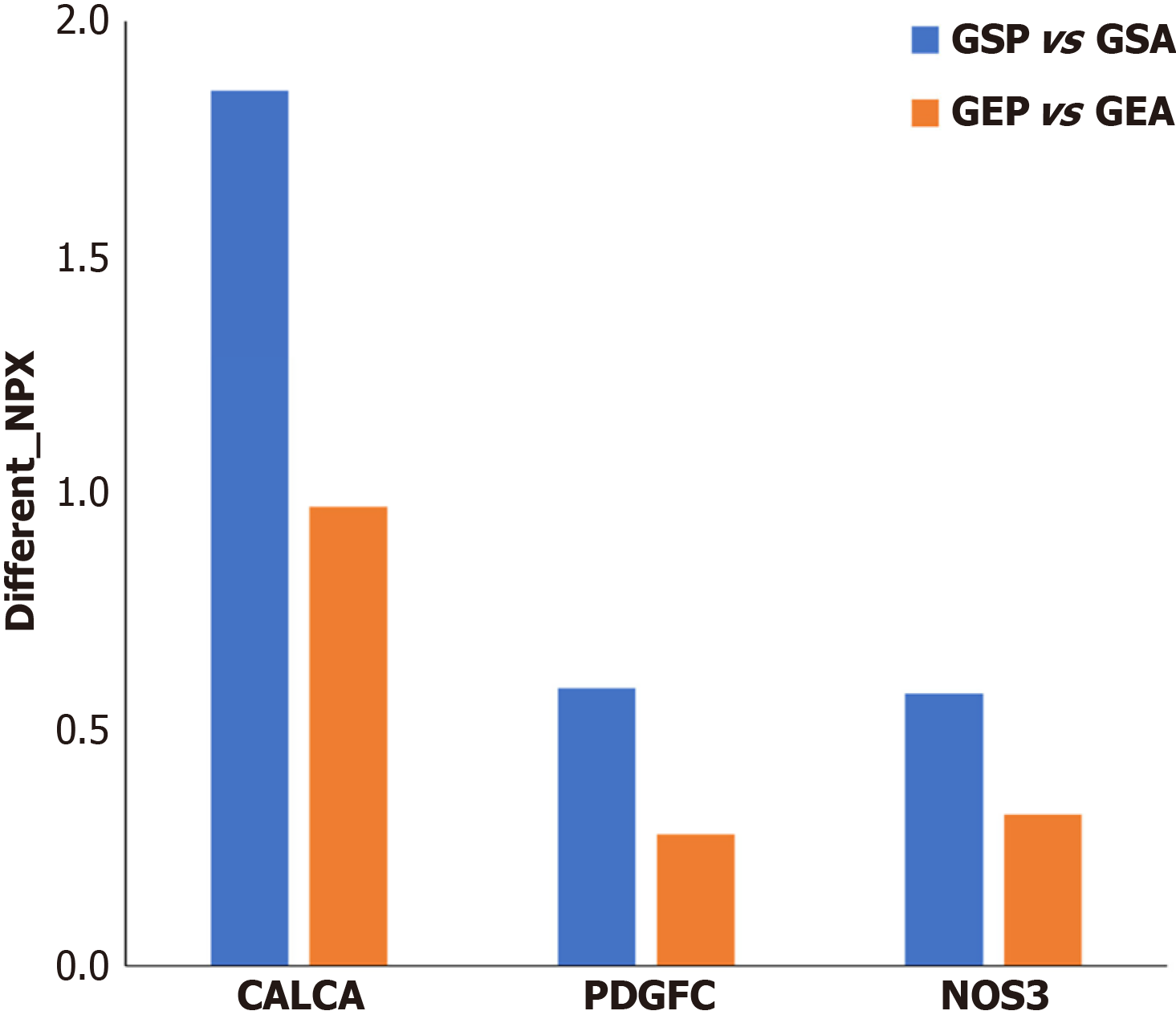
Differential protein analysis: We performed preoperative and postoperative differential protein expression analysis on the two groups (Figure 1). Comparative analysis of the SMIS group before and after surgery identified 13 DEPs, of which 4 were upregulated post-SMIS (Figure 2). A similar analysis of the ORS group revealed 37 DEPs, with 29 upregulated after ORS (Figure 3). A total of nine DEPs were identified in both the SMIS group and ORS group. These included TMPRSS15, CNTN2, HPGDS, PLXDC1, NPPC, CA14, calcitonin (CALCA), platelet-derived growth factor C (PDGFC), and nitric oxide synthase (NOS3) (Figure 4). Among these nine common DEPs, CALCA, PDGFC, and NOS3 were found to be upregulated post-surgically in comparison to the pre-surgical state. Notably, CALCA, PDGFC, and NOS3 showed the greatest increase following ORS (Figure 5).
The 9 DEPs were functionally classified as follows. (1) Proteins involved in the repair and regeneration of cells: CNTN2[12], NOS3[13]; (2) Protein involved in immune and inflammatory processes: HPGDS[14,15], PDGFC[16-18], NOS3[19]; (3) Protein involved in biological metabolism: NPPC[20,21], CA14[22], CALCA[23]; (4) Cancer-related protein: PLXDC1[24,25], PDGFC[26-28], NOS3[29]; and (5) Trauma-related: TMPRSS15[30].
The four DEPs that were upregulated after SMIS surgery were functionally classified as follows. (1) Proteins involved in the repair and regeneration of cells: NOS3[13], PDGFC[31]; (2) Protein involved in immune and inflammatory processes: PDGFC[16]; NOS3[19,32]; CMP-N-acetylneuraminate-beta-galactosamide-alpha-2,3-sialyltransferase1[33]; (3) Protein involved in biological metabolism: CALCA[23]; and (4) Cancer-related protein: CMP-N-acetylneuraminate-beta-galactosamide-alpha-2,3-sialyltransferase1[34,35].
The 29 DEPs that were found to be upregulated after ORS were functionally classified as follows: (1) Proteins involved in the repair and regeneration of cells: CALR[36,37], TOP2B[38], RRM2B[39,40], NUCB2[41], MAX[42], FKBP1B, PDGFC[43], YES1[44], PXN[45,46], ENAH[47,48]; (2) Protein involved in immune and inflammatory processes: SIRT5[49], PDGFC[18,27], FGR[50-52], STXBP3[53,54], PLIN1[55,56], PVALB[57], MAP4K5[58], LAT2[59], BANK1[60,61], RARRES1[62,63], NOS3[32], ERBB2IP[64]; (3) Protein involved in biological metabolism: NUCB2[65,66], PDGFC[28], PLIN1[67], PDP1[68], NOS3[69], FOXO1[70], SIRT5[71,72], CALCA[73], PTPRJ[74]; and (4) Cancer-related protein: SIRT5[75], NUCB2[76], CALR[77], PDGFC[26], YES1[44,78], PXN[79], PLIN1[80], ERBB2IP[81], LRP1[82], MAGED1[83], PGF[84], ENAH[85], PRKRA[86].
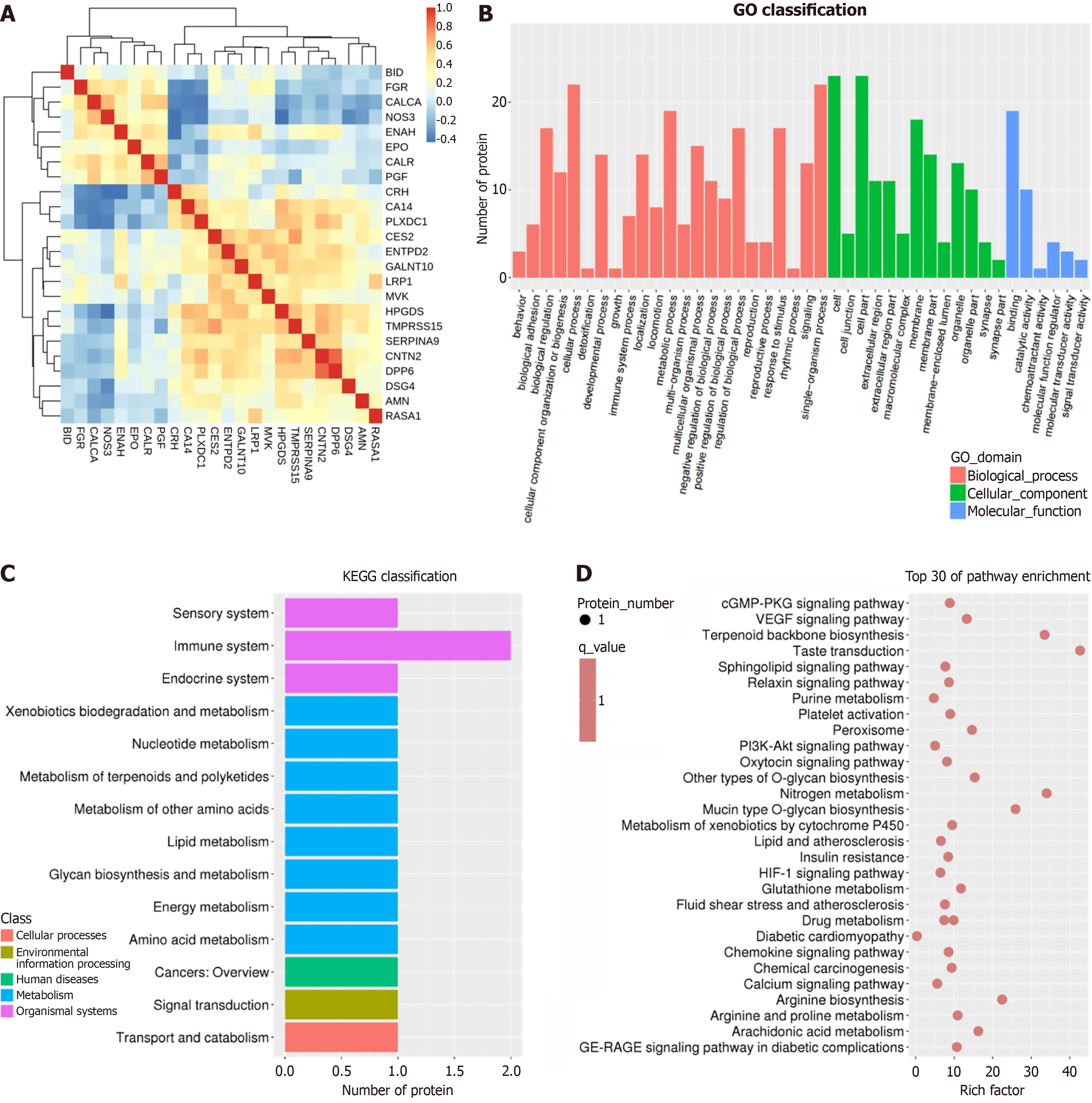
Correlation and biological function analysis of DEPs in the SMIS and ORS group: We analyzed the correlations among DEPs and performed functional enrichment analyses in the SMIS and ORS groups. Figure 6A shows a correlation heatmap of proteins in the endoscopic super minimally invasive surgery and laparoscopic surgery groups. The correlation heatmap includes only DEPs (|log2fold change| > 1, adjusted P < 0.05). The full protein list is provided in Supplementary Table 1.
GO analysis indicated enrichment in immune system processes, cellular processes, metabolic processes, and single-organism processes. In terms of molecular function, binding and catalytic activity were enriched (Figure 6B). KEGG analysis showed significant enrichment in immune system-related pathways (Figure 6C). Pathway enrichment analysis further indicated that differentially expressed postoperative immune-related proteins were mainly enriched in terpenoid backbone biosynthesis, taste transduction, and nitrogen metabolism pathways. Nitrogen metabolism includes the production of nitric oxide (NO), an important immune effector molecule generated by activated macrophages and other leukocytes during inflammation and wound repair, reflecting alterations in immune status (Figure 6D)[87].
This study systematically evaluated the comparative changes in trauma-induced immune inflammation in gastric cancer patients undergoing SMIS vs those undergoing ORS. Specifically, it analyzed alterations in trauma immunity before and after surgery, as well as the impact of different surgical methods on organ damage in patients. The results indicated that the SMIS group, before and after surgery, yielded 13 DEPs, while the ORS group revealed 37 DEPs. Notably, a significant increase in the number of DEPs related to organ damage was observed in the ORS group, both preoperatively and postoperatively, compared to the SMIS group. This suggests that ORS has a more pronounced effect on the patients, contributing to greater organ damage.
Clinical data from the 38 enrolled patients, including sex, age, BMI, chronic diseases, history of Helicobacter pylori infection, and preoperative use of anti-inflammatory drugs, were analyzed. No significant differences were observed between the SMIS and ORS groups, indicating comparable baseline inflammatory and immune status between groups, which is important for interpreting the study findings.
We then compared postoperative outcomes between the SMIS and ORS groups and found significant differences in the type and duration of postoperative antibiotic use, postoperative body temperature, and postoperative analgesia. The ORS group had higher postoperative body temperature and a longer duration of antibiotic therapy than the SMIS group. This finding supports the hypothesis that ORS induces disparities in inflammatory and immune-related proteins in the proteome, which are comparatively overexpressed. Statistical analysis also revealed significant differences in hospital stay between the SMIS and ORS groups, further substantiating the notion that ORS results in differential expression of proteins related to cell repair and regeneration. These proteins exhibited a general upward trend, with substantial discrepancies observed across pathological tumor types, influenced by individual conditions, the chosen surgical method, and indications for surgery.
This study also investigated the expression levels and functional characteristics of three proteins. NOS3 is predominantly expressed in endothelial cells and has been identified as a key contributor to NO synthesis, a critical signaling process. NO plays an important role in cardiovascular function, including regulation of vascular tone, endothelial function, cardiac performance, blood coagulation, and thrombosis[32,88]. The ORS preoperative and postoperative groups showed greater upregulation of NOS3, suggesting that ORS generally causes greater damage to the vascular endothelium, with a more significant impact on the circulatory system and vascular endothelial function.
PDGFC is expressed in most organs and cell types[31] and plays a pivotal role in multiple biological, physiological, and pathological processes, including development, angiogenesis, tumor growth, tissue remodeling, wound healing, atherosclerosis, fibrosis, and metabolism. Aberrant PDGFC expression has been linked to several pathological conditions, including choroidal neoplasms, chronic myocarditis, glomerulosclerosis, tissue fibrosis, atherosclerosis, and various cancers. Cancer-associated fibroblasts have been shown to secrete PDGFC, activating PDGFC/PDGFRA signaling in gastric stromal tumor cells and promoting tumor growth and metastasis[26]. Elevated PDGFC levels in gastrointestinal stromal tumors have been associated with increased tumor growth, migration, and invasion. Therefore, avoiding marked postoperative upregulation of PDGFC-related proteins may help mitigate tumor-related risks. In addition, PDGFC overexpression has been shown to enhance the proliferation, migration, and invasion of pancreatic ductal adenocarcinoma cells, suggesting a potential association with tumor recurrence. In the present study, PDGFC was more strongly upregulated in the preoperative and postoperative ORS groups, suggesting that ORS may be associated with greater physiological risk than SMIS; however, further validation is required.
CALCA is a polypeptide hormone that regulates metabolism and plays a role in maintaining bone density and reducing fracture rates. CALCA is also involved in pain regulation and exhibits analgesic effects[23]. In this study, CALCA was significantly upregulated in the preoperative and postoperative ORS groups, suggesting that patients undergoing ORS may experience more pronounced postoperative pain, greater metabolic disturbance, and a longer recovery period. The increase was particularly evident alongside routine use of pain pumps and antipyretic analgesics after ORS, consistent with a higher postoperative analgesic requirement in these patients.
In summary, the increases in CALCA, NOS3, and PDGFC observed with both surgical approaches suggest postoperative inflammation and immune-related responses to these procedures. Implementing preventive measures in clinical settings may help reduce complications related to metabolic disturbance, inflammation, and vascular endothelial injury, thereby facilitating faster recovery and earlier return to normal activities. Although both surgical methods result in the production of DEPs, ORS appears to have a more significant impact on the human body, affecting a broader range of processes, whereas SMIS has a less pronounced effect, with fewer significant changes in the number and function of DEPs.
In comparison with the SMIS group, the ORS group demonstrated a greater capacity to detect proteins associated with cancer cell metastasis. This finding is likely attributable, at least in part, to the more advanced clinicopathological staging of tumors in the ORS group, rather than solely to the surgical approach itself. This underscores the importance of early diagnosis and treatment of digestive tract cancers, as early-stage disease preserves the option for less invasive interventions. Regarding potential confounders, although anesthetics may transiently affect immune responses, they are typically metabolized within 24 hours postoperatively. Given that blood samples were collected on postoperative day 3, residual anesthetic effects are unlikely to have substantially influenced the proteomic results; nonetheless, this possibility cannot be entirely excluded.
However, this study also has limitations. First, patient grouping was determined by clinicians based on tumor pathological characteristics and was not randomized. The SMIS group consisted predominantly of early-stage lesions, whereas the ORS group comprised exclusively invasive adenocarcinomas. We acknowledge that these fundamental differences in disease biology introduce significant selection bias.
Because the extent of surgery is intrinsically linked to tumor progression, the identified DEPs likely represent a composite molecular signature reflecting both the systemic response to surgical trauma and the underlying tumor-burden. Although this limits isolation of the surgical impact alone, it provides a “real-world” snapshot of the molecular alterations associated with different standard-of-care interventions. Future studies employing propensity score matching are needed to further disentangle these factors. Second, although Olink technology offers high sensitivity and microliter-scale detection capability, protein expression levels are determined through relative comparisons. Therefore, new technologies will be needed to enable quantitative analysis of key proteins in the future. Third, this prospective exploratory study is limited by its small sample size (n = 38) and single-center design, which reduces statistical power and precludes subgroup analyses. Thus, these findings represent a preliminary molecular “snapshot” and require validation in large-scale, multicenter prospective trials. Furthermore, our study primarily focused on the acute-phase response and did not investigate long-term proteomic adaptations. Future research should explore how early molecular markers evolve into sustained physiological outcomes.
Despite these limitations, this study provides a preliminary comparison of trauma-associated immune proteins in patients treated with SMIS and ORS. It also highlights the potential adverse effects of organ resection on immune and inflammatory responses, other biological processes, and overall quality of life. Early detection and treatment of gastric cancer are crucial for maintaining viable options for SMIS, thereby minimizing the impact of surgical interventions on patients. However, for locally advanced gastric cancer, ORS remains the prevalent option, as current technological limitations restrict minimally invasive surgery to treating cancers up to stage T1. Consequently, early detection and diagnosis are paramount to ensuring SMIS remains a viable treatment option for patients.
Organ-preserving SMIS has been shown to result in fewer DEP species related to trauma and inflammation, as well as fewer upregulated proteins. Conversely, ORS has been observed to yield a greater number of DEP species associated with trauma and inflammation, along with higher expression of these proteins. These findings suggest that ORS exerts a more substantial influence on the body’s responses to trauma and inflammation, as well as on immune function. In light of these observations, implementing targeted preventive measures may be a viable approach to mitigate these effects.
| 1. | Wong MCS, Huang J, Chan PSF, Choi P, Lao XQ, Chan SM, Teoh A, Liang P. Global Incidence and Mortality of Gastric Cancer, 1980-2018. JAMA Netw Open. 2021;4:e2118457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 328] [Cited by in RCA: 291] [Article Influence: 58.2] [Reference Citation Analysis (6)] |
| 2. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 14950] [Article Influence: 7475.0] [Reference Citation Analysis (21)] |
| 3. | Guan WL, He Y, Xu RH. Gastric cancer treatment: recent progress and future perspectives. J Hematol Oncol. 2023;16:57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 763] [Cited by in RCA: 664] [Article Influence: 221.3] [Reference Citation Analysis (8)] |
| 4. | Marik PE, Flemmer M. The immune response to surgery and trauma: Implications for treatment. J Trauma Acute Care Surg. 2012;73:801-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 225] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 5. | Weigand K, Brost S, Steinebrunner N, Büchler M, Schemmer P, Müller M. Ischemia/Reperfusion injury in liver surgery and transplantation: pathophysiology. HPB Surg. 2012;2012:176723. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 108] [Article Influence: 7.7] [Reference Citation Analysis (4)] |
| 6. | Cai Z, Yuan X, Li H, Feng X, Du C, Han K, Chen Q, Linghu E. Bowel function, quality of life, and mental health of patients with high-grade intraepithelial neoplasia or T1 colorectal cancer after organ-preserving versus organ-resection surgeries: a cross-sectional study at a Chinese tertiary care center. Surg Endosc. 2024;38:5756-5768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Terrando N, Brzezinski M, Degos V, Eriksson LI, Kramer JH, Leung JM, Miller BL, Seeley WW, Vacas S, Weiner MW, Yaffe K, Young WL, Xie Z, Maze M. Perioperative cognitive decline in the aging population. Mayo Clin Proc. 2011;86:885-893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 139] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 8. | Máca J, Peteja M, Reimer P, Jor O, Šeděnková V, Panáčková L, Ihnát P, Burda M, Ševčík P. Surgical injury: comparing open surgery and laparoscopy by markers of tissue damage. Ther Clin Risk Manag. 2018;14:999-1006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 9. | Chen Q, Yuan Y, Li H, Sun Y, Zhu X, Zhai Y, Liu M, Lyu K, Ning B, Shao Q, Xu J, Zhang Z, Li Y, Tian S, Zuo X, Han K, Wang J, Du C, Zhang L, Du J, Liu L, Chen X, Linghu E; Chinese Society of Digestive Endoscopy, SMIS Committee of World Endoscopy Organization. Chinese clinical practice guidelines for super minimally invasive surgery of digestive tract tumors. J Transl Int Med. 2025;13:487-507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Desborough JP. The stress response to trauma and surgery. Br J Anaesth. 2000;85:109-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1598] [Cited by in RCA: 1324] [Article Influence: 50.9] [Reference Citation Analysis (1)] |
| 11. | Sereda AP, Rukina AN, Trusova YV, Dzhavadov AA, Cherny AA, Bozhkova SA, Shubnyakov II, Tikhilov RM. Dynamics of C-reactive protein level after orthopedic surgeries. J Orthop. 2024;47:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 12. | Savvaki M, Kafetzis G, Kaplanis SI, Ktena N, Theodorakis K, Karagogeos D. Neuronal, but not glial, Contactin 2 negatively regulates axon regeneration in the injured adult optic nerve. Eur J Neurosci. 2021;53:1705-1721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Ma L, Wallace JL. Endothelial nitric oxide synthase modulates gastric ulcer healing in rats. Am J Physiol Gastrointest Liver Physiol. 2000;279:G341-G346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 62] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 14. | Mohri I, Aritake K, Taniguchi H, Sato Y, Kamauchi S, Nagata N, Maruyama T, Taniike M, Urade Y. Inhibition of prostaglandin D synthase suppresses muscular necrosis. Am J Pathol. 2009;174:1735-1744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | Rittchen S, Jandl K, Lanz I, Reiter B, Ferreirós N, Kratz D, Lindenmann J, Brcic L, Bärnthaler T, Atallah R, Olschewski H, Sturm EM, Heinemann A. Monocytes and Macrophages Serve as Potent Prostaglandin D(2) Sources during Acute, Non-Allergic Pulmonary Inflammation. Int J Mol Sci. 2021;22:11697. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 16. | Jiang Z, Kang Q, Qian H, Xu Z, Tong H, Yang J, Li L, Li R, Li G, Chen F, Lin N, Zhao Y, Shi H, Huang J, Ma X. Revealing the crucial roles of suppressive immune microenvironment in cardiac myxoma progression. Signal Transduct Target Ther. 2024;9:193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 17. | Klinkhammer BM, Floege J, Boor P. PDGF in organ fibrosis. Mol Aspects Med. 2018;62:44-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 182] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 18. | Lu W, Xie Y, Huang B, Ma T, Wang H, Deng B, Zou S, Wang W, Tang Q, Yang Z, Li X, Wang L, Fang L. Platelet-derived growth factor C signaling is a potential therapeutic target for radiation proctopathy. Sci Transl Med. 2021;13:eabc2344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Cirino G, Fiorucci S, Sessa WC. Endothelial nitric oxide synthase: the Cinderella of inflammation? Trends Pharmacol Sci. 2003;24:91-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 139] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 20. | Bao Q, Zhang B, Zhou L, Yang Q, Mu X, Liu X, Zhang S, Yuan M, Zhang Y, Che J, Wei W, Liu T, Li G, He J. CNP Ameliorates Macrophage Inflammatory Response and Atherosclerosis. Circ Res. 2024;134:e72-e91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 34] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 21. | Perez-Ternero C, Aubdool AA, Makwana R, Sanger GJ, Stimson RH, Chan LF, Moyes AJ, Hobbs AJ. C-type natriuretic peptide is a pivotal regulator of metabolic homeostasis. Proc Natl Acad Sci U S A. 2022;119:e2116470119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 22. | Qian J, Shen Q, Zhang T, Chen J, Chen L, Dong Y, Yan R, Chen Z. Carbonic anhydrase 14 protects the liver against the cytotoxicity of bile acids in a biliary bicarbonate umbrella-related manner. Life Sci. 2022;310:121117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 23. | Yazdani J, Khiavi RK, Ghavimi MA, Mortazavi A, Hagh EJ, Ahmadpour F. [Calcitonin as an analgesic agent: review of mechanisms of action and clinical applications]. Braz J Anesthesiol. 2019;69:594-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 24. | Li X, Fan Y, Tang M, Li H, Zhang Y, Mi J, Wang Y, Zhao M, Wang Z, Su F. PLXDC1 Can Be a Biomarker for Poor Prognosis and Immune Evasion in Gastric Cancer. J Inflamm Res. 2022;15:5439-5455. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 25. | Fu Y, Zhang X, Qiao Q. PLXDC1 serves as a potential prognostic marker and involves in malignant progression and macrophage polarization in colon cancer. J Biochem Mol Toxicol. 2024;38:e23832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 26. | Yoon H, Tang CM, Banerjee S, Yebra M, Noh S, Burgoyne AM, Torre J, Siena M, Liu M, Klug LR, Choi YY, Hosseini M, Delgado AL, Wang Z, French RP, Lowy A, DeMatteo RP, Heinrich MC, Molinolo AA, Gutkind JS, Harismendy O, Sicklick JK. Cancer-associated fibroblast secretion of PDGFC promotes gastrointestinal stromal tumor growth and metastasis. Oncogene. 2021;40:1957-1973. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 27. | Turrell FK, Orha R, Guppy NJ, Gillespie A, Guelbert M, Starling C, Haider S, Isacke CM. Age-associated microenvironmental changes highlight the role of PDGF-C in ER(+) breast cancer metastatic relapse. Nat Cancer. 2023;4:468-484. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 65] [Article Influence: 21.7] [Reference Citation Analysis (0)] |
| 28. | Shi YH, Liu ZD, Ma MJ, Zhao GY, Zhu YQ, Wang JQ, Yu YY, Huang XT, Ye JY, Li FX, Wang XY, Xu QC, Yin XY. Platelet-Derived Growth Factor C Facilitates Malignant Behavior of Pancreatic Ductal Adenocarcinoma by Regulating SREBP1 Mediated Lipid Metabolism. Adv Sci (Weinh). 2024;11:e2407069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 29. | Zou D, Li Z, Lv F, Yang Y, Yang C, Song J, Chen Y, Jin Z, Zhou J, Jiang Y, Ma Y, Jing Z, Tang Y, Zhang Y. Pan-Cancer Analysis of NOS3 Identifies Its Expression and Clinical Relevance in Gastric Cancer. Front Oncol. 2021;11:592761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 30. | Yang X, Yin H, Peng L, Zhang D, Li K, Cui F, Xia C, Huang H, Li Z. The Global Status and Trends of Enteropeptidase: A Bibliometric Study. Front Med (Lausanne). 2022;9:779722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 31. | Jung SC, Kang D, Ko EA. Roles of PDGF/PDGFR signaling in various organs. Korean J Physiol Pharmacol. 2025;29:139-155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 17] [Reference Citation Analysis (5)] |
| 32. | Ying L, Hofseth LJ. An emerging role for endothelial nitric oxide synthase in chronic inflammation and cancer. Cancer Res. 2007;67:1407-1410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 190] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 33. | Zou Y, Guo S, Liao Y, Chen W, Chen Z, Chen J, Wen L, Xie X. Ceramide metabolism-related prognostic signature and immunosuppressive function of ST3GAL1 in osteosarcoma. Transl Oncol. 2024;40:101840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 34. | Lin WD, Fan TC, Hung JT, Yeo HL, Wang SH, Kuo CW, Khoo KH, Pai LM, Yu J, Yu AL. Sialylation of CD55 by ST3GAL1 Facilitates Immune Evasion in Cancer. Cancer Immunol Res. 2021;9:113-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 47] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 35. | Pietrobono S, Anichini G, Sala C, Manetti F, Almada LL, Pepe S, Carr RM, Paradise BD, Sarkaria JN, Davila JI, Tofani L, Battisti I, Arrigoni G, Ying L, Zhang C, Li H, Meves A, Fernandez-Zapico ME, Stecca B. ST3GAL1 is a target of the SOX2-GLI1 transcriptional complex and promotes melanoma metastasis through AXL. Nat Commun. 2020;11:5865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 75] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 36. | Sawaya AP, Vecin NM, Burgess JL, Ojeh N, DiBartolomeo G, Stone RC, Pastar I, Tomic-Canic M. Calreticulin: a multifunctional protein with potential therapeutic applications for chronic wounds. Front Med (Lausanne). 2023;10:1207538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 37. | Michalak M, Corbett EF, Mesaeli N, Nakamura K, Opas M. Calreticulin: one protein, one gene, many functions. Biochem J. 1999;344 Pt 2:281-292. [PubMed] |
| 38. | Neha S, Dholaniya PS. The Prevailing Role of Topoisomerase 2 Beta and its Associated Genes in Neurons. Mol Neurobiol. 2021;58:6443-6459. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 39. | Wu L, Yin L, Ma L, Yang J, Yang F, Sun B, Nianzeng X. Comprehensive bioinformatics analysis of ribonucleoside diphosphate reductase subunit M2(RRM2) gene correlates with prognosis and tumor immunotherapy in pan-cancer. Aging (Albany NY). 2022;14:7890-7905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 40. | Takaya K, Kishi K. Identification of a new human senescent skin cell marker ribonucleoside-diphosphate reductase subunit M2 B. Biogerontology. 2024;25:1239-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 41. | Zhou Q, Liu Y, Feng R, Zhang W. NUCB2: roles in physiology and pathology. J Physiol Biochem. 2022;78:603-617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 42. | Tucker DW, Pangal DJ, Du R, Gogia AS, Tafreshi A, Ruzevick J, Hurth KT, Triche T Jr, Micko A, Carpten JD, Shiroishi MS, Carmichael JD, Rhie SK, Zada G. Validation of Myc-Associated Protein X (MAX) regulation in growth hormone secreting and nonfunctional pituitary adenoma. PLoS One. 2023;18:e0284949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 43. | Tian Y, Zhan Y, Jiang Q, Lu W, Li X. Expression and function of PDGF-C in development and stem cells. Open Biol. 2021;11:210268. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 44. | Kook E, Chun KS, Kim DH. Emerging Roles of YES1 in Cancer: The Putative Target in Drug Resistance. Int J Mol Sci. 2024;25:1450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 45. | Zaidel-Bar R, Milo R, Kam Z, Geiger B. A paxillin tyrosine phosphorylation switch regulates the assembly and form of cell-matrix adhesions. J Cell Sci. 2007;120:137-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 376] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 46. | Liang P, Wu Y, Zheng S, Zhang J, Yang S, Wang J, Ma S, Zhang M, Gu Z, Liu Q, Jiang W, Xing Q, Wang B. Paxillin phase separation promotes focal adhesion assembly and integrin signaling. J Cell Biol. 2024;223:e202209027. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 26] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 47. | Deng G, Luo Y, Zhang Y, Zhang J, He Z. Enabled homolog (ENAH) regulated by RNA binding protein splicing factor 3b subunit 4 (SF3B4) exacerbates the proliferation, invasion and migration of hepatocellular carcinoma cells via Notch signaling pathway. Bioengineered. 2022;13:2194-2206. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 48. | Li Y, Zhang Y, Wang M, Su J, Dong X, Yang Y, Wang H, Li Q. The mammalian actin elongation factor ENAH/MENA contributes to autophagosome formation via its actin regulatory function. Autophagy. 2024;20:1798-1814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 49. | Cha XD, Zou QY, Li FZ, Wang TY, Wang SL, Cai BY, Cao ZW, Ji ZH, Liu HB, Wang WW, Li TF, Liang CQ, Ren WW, Liu HH. SIRT5 exacerbates eosinophilic chronic rhinosinusitis by promoting polarization of M2 macrophage. J Allergy Clin Immunol. 2024;154:644-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 50. | Acín-Pérez R, Iborra S, Martí-Mateos Y, Cook ECL, Conde-Garrosa R, Petcherski A, Muñoz MDM, Martínez de Mena R, Krishnan KC, Jiménez C, Bolaños JP, Laakso M, Lusis AJ, Shirihai OS, Sancho D, Enríquez JA. Fgr kinase is required for proinflammatory macrophage activation during diet-induced obesity. Nat Metab. 2020;2:974-988. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 56] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 51. | Kao TI, Chen PJ, Wang YH, Tseng HH, Chang SH, Wu TS, Yang SH, Lee YT, Hwang TL. Bletinib ameliorates neutrophilic inflammation and lung injury by inhibiting Src family kinase phosphorylation and activity. Br J Pharmacol. 2021;178:4069-4084. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 52. | Crainiciuc G, Palomino-Segura M, Molina-Moreno M, Sicilia J, Aragones DG, Li JLY, Madurga R, Adrover JM, Aroca-Crevillén A, Martin-Salamanca S, Del Valle AS, Castillo SD, Welch HCE, Soehnlein O, Graupera M, Sánchez-Cabo F, Zarbock A, Smithgall TE, Di Pilato M, Mempel TR, Tharaux PL, González SF, Ayuso-Sacido A, Ng LG, Calvo GF, González-Díaz I, Díaz-de-María F, Hidalgo A. Behavioural immune landscapes of inflammation. Nature. 2022;601:415-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 81] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 53. | Ouahed J, Kelsen JR, Spessott WA, Kooshesh K, Sanmillan ML, Dawany N, Sullivan KE, Hamilton KE, Slowik V, Nejentsev S, Neves JF, Flores H, Chung WK, Wilson A, Anyane-Yeboa K, Wou K, Jain P, Field M, Tollefson S, Dent MH, Li D, Naito T, McGovern DPB, Kwong AC, Taliaferro F, Ordovas-Montanes J, Horwitz BH, Kotlarz D, Klein C, Evans J, Dorsey J, Warner N, Elkadri A, Muise AM, Goldsmith J, Thompson B, Engelhardt KR, Cant AJ, Hambleton S, Barclay A, Toth-Petroczy A, Vuzman D, Carmichael N, Bodea C, Cassa CA, Devoto M, Maas RL, Behrens EM, Giraudo CG, Snapper SB. Variants in STXBP3 are Associated with Very Early Onset Inflammatory Bowel Disease, Bilateral Sensorineural Hearing Loss and Immune Dysregulation. J Crohns Colitis. 2021;15:1908-1919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 54. | Yao Q, Wang C, Wang Y, Xiang W, Chen Y, Zhou Q, Chen J, Jiang H, Chen D. STXBP3 and GOT2 predict immunological activity in acute allograft rejection. Front Immunol. 2022;13:1025681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 55. | Wang M, Cao X, Shang Y, Jiang Y, Chen P, Duan C, Zhang D, Wang P, Ji J, Gong Z. Correlational analysis of PLIN1 with inflammation in diabetic foot ulcer wounds. Diabetes Res Clin Pract. 2024;209:111605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (2)] |
| 56. | Cho KY, Miyoshi H, Nakamura A, Greenberg AS, Atsumi T. Lipid Droplet Protein PLIN1 Regulates Inflammatory Polarity in Human Macrophages and is Involved in Atherosclerotic Plaque Development by Promoting Stable Lipid Storage. J Atheroscler Thromb. 2023;30:170-181. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 57. | Gong Y, Zhou M, Zhu Y, Pan J, Zhou X, Jiang Y, Zeng H, Zheng H, Geng X, Huang D. PVALB Was Identified as an Independent Prognostic Factor for HCC Closely Related to Immunity, and Its Absence Accelerates Tumor Progression by Regulating NK Cell Infiltration. J Hepatocell Carcinoma. 2024;11:813-838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 58. | Yu R, Zhang J, Zhuo Y, Hong X, Ye J, Tang S, Liu N, Zhang Y. ARG2, MAP4K5 and TSTA3 as Diagnostic Markers of Steroid-Induced Osteonecrosis of the Femoral Head and Their Correlation With Immune Infiltration. Front Genet. 2021;12:691465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 59. | Wang Z, Li B, Li S, Lin W, Wang Z, Wang S, Chen W, Shi W, Chen T, Zhou H, Yinwang E, Zhang W, Mou H, Chai X, Zhang J, Lu Z, Ye Z. Metabolic control of CD47 expression through LAT2-mediated amino acid uptake promotes tumor immune evasion. Nat Commun. 2022;13:6308. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 88] [Reference Citation Analysis (0)] |
| 60. | Yang J, Ren J, Yang Y, Sun J, Zhou X, Zheng S, Xuan D, Xue Y, Fan H, Zhang J, Zou H, Wan W, Kong N. BANK1 alters B cell responses and influences the interactions between B cells and induced T regulatory cells in mice with collagen-induced arthritis. Arthritis Res Ther. 2018;20:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 61. | Gómez Hernández G, Morell M, Alarcón-Riquelme ME. The Role of BANK1 in B Cell Signaling and Disease. Cells. 2021;10:1184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 46] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 62. | Patel J, Xun D, Creswell K, Kim DK, Wu M, Hwang JW, Kim TS, Bansal S, Hong SH, Galli S, Kim HJ, Deng C, Byers SW, Lee MH. Loss of RARRES1 function Promotes Follicular Lymphomagenesis and Inhibits B cell Differentiation in Mice. Int J Biol Sci. 2022;18:2670-2682. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 63. | Möller-Hackbarth K, Dabaghie D, Charrin E, Zambrano S, Genové G, Li X, Wernerson A, Lal M, Patrakka J. Retinoic acid receptor responder1 promotes development of glomerular diseases via the Nuclear Factor-κB signaling pathway. Kidney Int. 2021;100:809-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 64. | Fang Q, Jing G, Zhang Y, Wang H, Luo H, Xia Y, Jin Q, Liu Y, Zuo J, Yang C, Zhang X, Liu S, Wu X, Song X. Erbin accelerates TFEB-mediated lysosome biogenesis and autophagy and alleviates sepsis-induced inflammatory responses and organ injuries. J Transl Med. 2023;21:916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 65. | Schalla MA, Unniappan S, Lambrecht NWG, Mori M, Taché Y, Stengel A. NUCB2/nesfatin-1 - Inhibitory effects on food intake, body weight and metabolism. Peptides. 2020;128:170308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 66. | Zheng J, Han J, Wang Y, Tian Z. Role of brain NUCB2/nesfatin-1 in stress and stress-related gastrointestinal disorders. Peptides. 2023;167:171043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 67. | Wang Y, Peng Y, Yang Y, Hu Y, Wang H, Xu L, Ju H. The effect of the PLIN1 gene on the metabolism and mitochondria of porcine skeletal muscle satellite cells. Genes Genomics. 2022;44:1555-1563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 68. | Karagiota A, Kanoura A, Paraskeva E, Simos G, Chachami G. Pyruvate dehydrogenase phosphatase 1 (PDP1) stimulates HIF activity by supporting histone acetylation under hypoxia. FEBS J. 2023;290:2165-2179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 69. | Tenopoulou M, Doulias PT. Endothelial nitric oxide synthase-derived nitric oxide in the regulation of metabolism. F1000Res. 2020;9:F1000 Faculty Rev-F1000 Faculty1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 40] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 70. | Teaney NA, Cyr NE. FoxO1 as a tissue-specific therapeutic target for type 2 diabetes. Front Endocrinol (Lausanne). 2023;14:1286838. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 55] [Reference Citation Analysis (0)] |
| 71. | Wu M, Tan J, Cao Z, Cai Y, Huang Z, Chen Z, He W, Liu X, Jiang Y, Gao Q, Deng B, Wang J, Yuan W, Zhang H, Chen Y. Sirt5 improves cardiomyocytes fatty acid metabolism and ameliorates cardiac lipotoxicity in diabetic cardiomyopathy via CPT2 de-succinylation. Redox Biol. 2024;73:103184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 72] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 72. | Fabbrizi E, Fiorentino F, Carafa V, Altucci L, Mai A, Rotili D. Emerging Roles of SIRT5 in Metabolism, Cancer, and SARS-CoV-2 Infection. Cells. 2023;12:852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 55] [Reference Citation Analysis (0)] |
| 73. | Srinivasan A, Wong FK, Karponis D. Calcitonin: A useful old friend. J Musculoskelet Neuronal Interact. 2020;20:600-609. [PubMed] |
| 74. | Ulke J, Chopra S, Kadiri OL, Geserick P, Stein V, Cheshmeh S, Kleinridders A, Kappert K. PTPRJ is a negative regulator of insulin signaling in neuronal cells, impacting protein biosynthesis, and neurite outgrowth. J Neuroendocrinol. 2024;36:e13446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 75. | Teng P, Cui K, Yao S, Fei B, Ling F, Li C, Huang Z. SIRT5-mediated ME2 desuccinylation promotes cancer growth by enhancing mitochondrial respiration. Cell Death Differ. 2024;31:65-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 52] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 76. | Ji R, Wang Y, Pan D, Han J, Wang Y, Zheng S, Zhao W, Li X, Han C, Zhang L. NUCB2 inhibition antagonizes osteosarcoma progression and promotes anti-tumor immunity through inactivating NUCKS1/CXCL8 axis. Cancer Lett. 2024;591:216893. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 77. | Fucikova J, Spisek R, Kroemer G, Galluzzi L. Calreticulin and cancer. Cell Res. 2021;31:5-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 322] [Article Influence: 53.7] [Reference Citation Analysis (0)] |
| 78. | Zhou H, Sun D, Tao J, Xu M, Zhang X, Hou H. Role of YES1 signaling in tumor therapy resistance. Cancer Innov. 2023;2:210-218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 79. | Chen Y, Zhao H, Xiao Y, Shen P, Tan L, Zhang S, Liu Q, Gao Z, Zhao J, Zhao Y, Guo Y, Feng Y. Pan-cancer analysis reveals an immunological role and prognostic potential of PXN in human cancer. Aging (Albany NY). 2021;13:16248-16266. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 80. | Huang JH, Wei Y, Fang Z, Yu C, Zhang R, Feng ZB, Zeng LP. Clinical pathological significance and biological function of PLIN1 in hepatocellular carcinoma: bioinformatics analysis and in vitro experiments. BMC Cancer. 2024;24:1073. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 81. | Qiu T, Tan L, Yan J, Luo Q. Erbin: an important therapeutic target for blocking tumor metastasis. Front Pharmacol. 2024;15:1474798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 82. | Lin H, Fu L, Zhou X, Yu A, Chen Y, Liao W, Shu G, Zhang L, Tan L, Liang H, Wang Z, Deng Q, Wang J, Jin M, Chen Z, Wei J, Cao J, Chen W, Li X, Li P, Lu J, Luo J. LRP1 induces anti-PD-1 resistance by modulating the DLL4-NOTCH2-CCL2 axis and redirecting M2-like macrophage polarisation in bladder cancer. Cancer Lett. 2024;593:216807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 83. | Zhang B, Li D, Jin X, Zhang K. The CDK4/6 inhibitor PD0332991 stabilizes FBP1 by repressing MAGED1 expression in pancreatic ductal adenocarcinoma. Int J Biochem Cell Biol. 2020;128:105859. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 84. | Kim N, Ko Y, Shin Y, Park J, Lee AJ, Kim KW, Pyo J. Comprehensive Analysis for Anti-Cancer Target-Indication Prioritization of Placental Growth Factor Inhibitor (PGF) by Use of Omics and Patient Survival Data. Biology (Basel). 2023;12:970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 85. | Chen D, Xu L, Li X, Chu Y, Jiang M, Xu B, Zhao M, Wang W, Wang H, Kang H, Wang K, Wu K, Liang J, Ren G. Enah overexpression is correlated with poor survival and aggressive phenotype in gastric cancer. Cell Death Dis. 2018;9:998. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 86. | Qiu J, Feng M, Yang G, Su D, Zhao F, Liu Y, Tao J, Luo W, Zhang T. PRKRA promotes pancreatic cancer progression by upregulating MMP1 transcription via the NF-κB pathway. Heliyon. 2023;9:e17194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 87. | Coleman JW. Nitric oxide in immunity and inflammation. Int Immunopharmacol. 2001;1:1397-1406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 602] [Cited by in RCA: 647] [Article Influence: 25.9] [Reference Citation Analysis (3)] |
| 88. | Harrison DG, Widder J, Grumbach I, Chen W, Weber M, Searles C. Endothelial mechanotransduction, nitric oxide and vascular inflammation. J Intern Med. 2006;259:351-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 228] [Article Influence: 11.4] [Reference Citation Analysis (0)] |