Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.114331
Revised: November 14, 2025
Accepted: January 29, 2026
Published online: April 14, 2026
Processing time: 186 Days and 22.6 Hours
One of the main challenges in treating gastric cancer is chemoresistance, particularly when using 5-fluorouracil (5-FU). Traditional Chinese medicine Xiangshaliujunzi decoction (XSLJZD) has been widely used for managing chemotherapy-related side effects; however, its potential antitumor effects remain unexplored. We aimed to study the role of XSLJZD in addressing 5-FU resistance in gastric cancer cells.
To investigate whether XSLJZD overcomes 5-FU resistance in gastric cancer by regulating miR-200a-3p/ZEB1/IRF1-mediated PANoptosis.
BGC-823/5-FU cell line was constructed. ZEB1 and IRF1 knockdown cell lines were constructed by transfecting lentivirus vectors containing shRNAs targeting ZEB1 and IRF1. Cell viability and proliferation were detected by CCK8 and colony formation assays. Cell apoptosis was determined by flow cytometry. Cell necrop
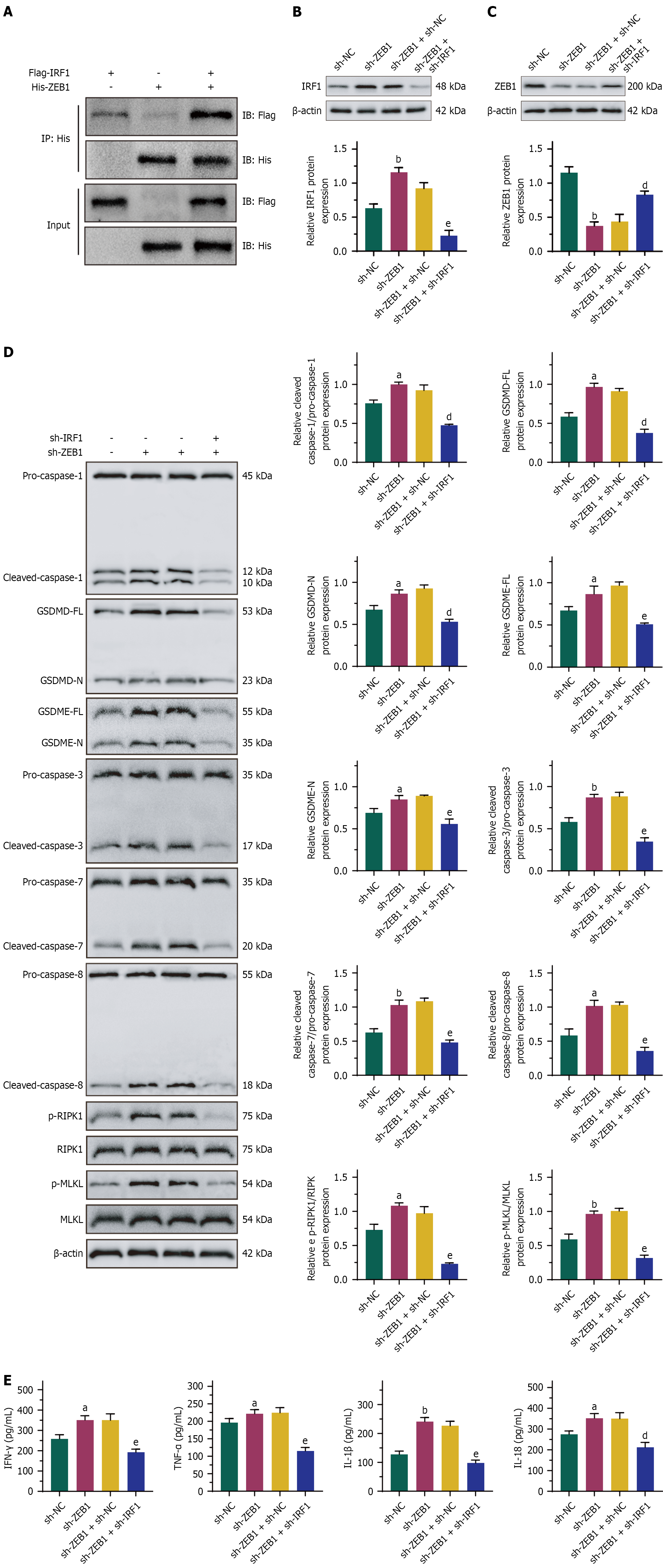
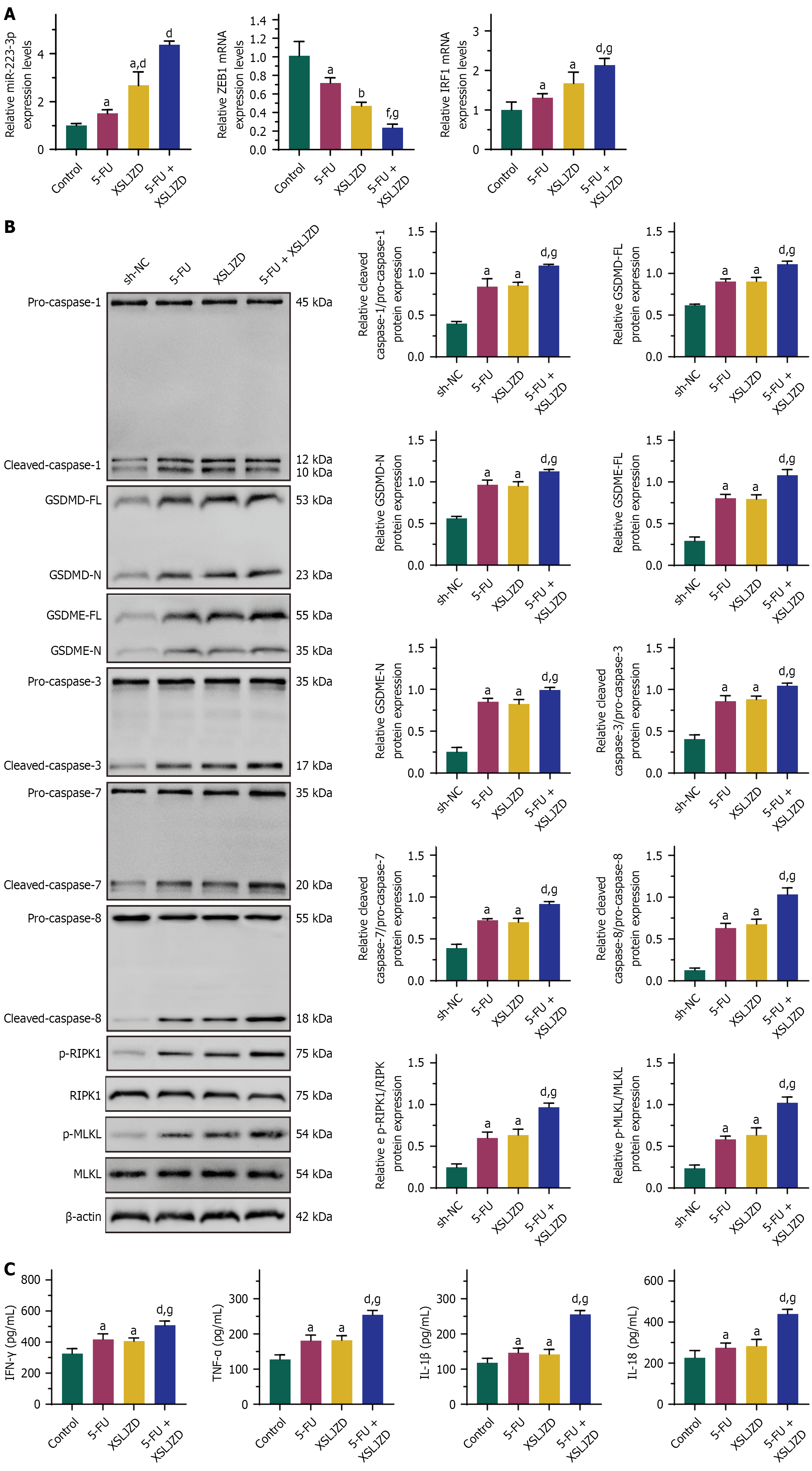
XSLJZD significantly inhibited cell viability and proliferation while promoting PANoptosis in BGC-823/5-FU cells (5-FU-resistant cells). ZEB1 knockdown upregulated pyroptosis-, apoptosis-, and other programmed cell death (PCD)-related proteins. Simultaneous knockdown of ZEB1 and IRF1 suppressed the expression of pyroptosis-, apoptosis- and PCD-related proteins. The combination of XSLJZD and 5-FU promoted miR-200a/ZEB1/IRF1-mediated PANoptosis in transplanted tumor tissues from mice.
Our findings suggest that XSLJZD sensitizes gastric cancer cells to 5-FU by modulating the miR-200a-3p/ZE
Core Tip: Xiangshaliujunzi decoction (XSLJZD) overcomes 5-fluorouracil (5-FU) resistance in gastric cancer by inducing PANoptosis via the miR-200a-3p/ZEB1/IRF1 pathway. In vitro, XSLJZD inhibits viability and promotes cell death. In vivo, combination with 5-FU enhances this effect. This provides a novel traditional Chinese medicine strategy for chemoresistance, though limitations include lack of standalone controls and generalizability validation.
- Citation: Gao H, Yin DF, Xing XR, Zhou LJ, Yu R. MiR-200a-3p/ZEB1/IRF1-mediated PANoptosis prompts Xiangshaliujunzi decoction to overcome 5-fluorouracil resistance in gastric cancer. World J Gastroenterol 2026; 32(14): 114331
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/114331.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.114331
Gastric cancer is the fifth most common malignancy worldwide with high mortality rates[1]. It has a poor prognosis, mainly because it is often diagnosed at a late stage. The global 5-year survival rate is approximately 20%, except in Japan and South Korea[2]. For resectable gastric cancer, the standard treatment involves surgical resection in combination with perioperative chemotherapy. For nonresectable or metastatic gastric cancer, systemic chemotherapy is the primary treat
PANoptosis is a type of regulated cell death characterized by the coordinated activation of apoptosis, pyroptosis, and necroptosis, potentially involving a multiprotein structure termed the PANoptosome[5]. Increasing evidence has revealed the significance of PANoptosis in gastric cancer. A PANscore model has been developed to quantify PANoptosis patterns in patients with gastric cancer, associating low scores with good immunotherapy response and prognosis[6]. Research showed that the ubiquitination-mediated degradation of YBX1 can reduce oxaliplatin resistance by promoting PANop
Traditional Chinese medicine (TCM) is widely employed in China in combination with chemotherapy and radiothe
Gastric adenocarcinoma cell line BGC-823 was purchased from Shanghai Huiying Biotechnology Co., Ltd. BGC-823, a well-established human gastric adenocarcinoma model with moderate aggressiveness, is commonly used in studies on chemoresistance and exhibits reliable 5-FU resistance. The cells were maintained in RPMI-1640 medium (Gibco, China, 11875093) with 10% fetal bovine serum (FBS) (Excell Bio, China, FSD500) in a humidified incubator with 5% CO2, at 37 °C. A cell line resistant to 5-FU (BGC-823/5-FU) was developed as follows: Cells were first cultured for 24 hours in RPMI-1640 medium containing 5 mg/L 5-FU, then washed and cultured in a complete medium without the drug for another 24 hours. Once the cells resumed growth, 5-FU was reintroduced at increasing concentrations (incrementing by 5 mg/L each time), and the cells were cultured for another 24 hours. The above operation was repeated eight times until the concentration of 5-FU reached 40 mg/L. Following the above steps, the BGC-823/5-FU cell line was cultured in a medium containing 10 mg/L 5-FU and utilized for subsequent experiments 2 weeks after drug withdrawal (maintained after 10 passages without 5-FU). Resistance was verified by IC50 (IC50 BGC-823/5-FU approximately 40 mg/L vs parental approximately 5 mg/L, approximately 8-fold).
The herbs were added to distilled water according to their respective dosages and decocted to prepare an herbal de
The miR-200a-3p sequence information was retrieved from the NCBI database (https://www.ncbi.nlm.nih.gov/gene). The sequences of the miR-200a-3p inhibitor, mimic, and the negative control (NC) were designed and synthesized by Shanghai GenePharma Co., Ltd. Detailed sequence data are available in Supplementary Table 1. The transfection of miR-200a-3p mimics, inhibitors, and their NC sequences was performed following the manufacturer’s instructions.
ShRNAs targeting ZEB1 and IRF1 were designed according to their coding sequences (CDS, assessed via the NCBI database). Plasmid pLKO.1, which contains the shRNA construct, was used as the vector. Recombinant plasmids pLKO.1-sh-ZEB1 and pLKO.1-sh-IRF1 were engineered by Shanghai GenePharma Co., Ltd. Detailed information about the vector and shRNA sequences is available in Supplementary Table 2 and Supplementary Figure 1. Lentivirus vectors containing the recombinant plasmids pLKO.1-sh-ZEB1 (Lv-sh-ZEB1) and pLKO.1-sh-IRF1 (Lv-sh-IRF1) were subsequently generated by Sangon Biotech (Shanghai) Co., Ltd. (Supplementary Table 2). The cells were incubated for 12 hours in a six-well plate. Lipofectamine 3000 (Invitrogen, United States) was used to transfect the lentiviruses into the gastric cancer cells.
The cells were maintained in a 96-well plate at a concentration of 2000 cells per well at 37 °C with 5% CO2 for 8 hours. Afterward, the cells were handled according to their specific experimental groups and then cultured for 0, 24, 48, or 72 hours. After each designated culture period, CCK-8 reagent (10 µL/well) (BA00208, Bioss, China) was added and left to incubate for 2 hours, followed by the determination of the optical density at 450 nm.
After being incubated for 8 hours in a six-well plate, the cells were treated according to their experimental groups. Following the treatment, the cells were cultured for 2 weeks. The medium was then removed, and the cells were fixed with methanol for 20 minutes. The cells were stained using 0.2% crystal violet for 5 minutes. After staining, the cells were rinsed with running water. The colonies were counted. Colony formation rate was computed as follows: Colony forma
The cells were incubated in RPMI-1640 medium without FBS for 12 hours at 37 °C with 5% CO2 and then treated according to the experimental groups. After being washed using phosphate-buffered saline (PBS), the cells were subsequently resuspended in 1 × annexin V binding solution (C1062 L, Beyotime, China) to obtain a cell solution with concentration of 1 × 106 cells/m. The cell suspension (100 μL), annexin V-FITC (5 μL), and PI solution (5 μL) were mixed in a tube and then incubated in the dark for 15 minutes. Afterward, 1 × annexin V binding solution (400 μL) was introduced into the tube, and Attune NxT flow cytometry (Thermo Fisher Scientific, United States) was used for analysis.
The cells were subjected to treatments based on the experimental groups. The RIPA buffer containing 0.5 mL PMSF was used to lyse the cells to obtain the lysate. After the lysate was centrifuged for 10 minutes at 4 °C and 12000 × g, the supernatant was collected. Protein concentration was assessed using the BCA protein assay kit. The protein was loaded into the wells of a 12% SDS-PAGE gel (BioFroxx, China) (40 μg/well) and separated at 80 V for 30 minutes, followed by 120 V until the protein was fully resolved. The proteins were transferred onto a PVDF membrane, which was then blocked for 15 minutes using a blocking solution and incubated overnight at 4 °C with the following primary antibodies: Anti-pro-caspase-1 (1:1000, ab179515, Abcam, United Kingdom), anti-GSDMD (1:1000, ab210070, Abcam, United Kingdom), anti-cleaved caspase-3 (1:500, ab32042, Abcam, United Kingdom), anti-cleaved caspase-7 (1:1000, ab256469, Abcam, United Kingdom), anti-cleaved caspase-8 (1:1000, #98134, CST, United States), anti-RIPK1 (1:1000, AB300617, Abcam, United Kingdom), anti-p-RIPK1 (1:1000, ab316923, Abcam, United Kingdom), anti-MLKL (1:2000, ab184718, Abcam, United Kingdom), anti-p-MLKL (1:1000, ab187091, Abcam, United Kingdom), anti-GSDME-N (1:1000, ab215191, Abcam, United Kingdom), anti-IRF1 (1:1000, #8478, CST, United States), anti-ZEB1 (1:1000, ab203829, Abcam, United Kingdom), and anti-β-actin (1:1000, ab8226, Abcam, United Kingdom). The membrane was washed with TBST and incubated with anti-rabbit/mouse IgG secondary antibodies (1:20000, Bioss, China) for 2 hours at room temperature. ECL solution (NCM Biotech, China) was added to the membrane to visualize the proteins using the JP-K6000 system (Shanghai Jiapeng, China). Protein expression levels, including phosphorylated and cleaved forms, were quantified using ImageJ software and normalized to β-actin as the internal reference.
The cells were treated in a six-well plate following the experimental group protocols. The FastPure® Cell/Tissue Total RNA Isolation Kit V2 (RC112, Vazyme, China) was used to extract the total RNA. The MiPure Cell/Tissue miRNA Kit (RC201, Vazyme, China) was used to extract the miRNA. RNA concentration and purity were assessed using the Nano-600 Ultra Micro Nucleic Acid and Protein Analysis System manufactured by Shanghai Jiapeng, China. cDNA was synthesized following the instructions of the HiScript III 1st Strand cDNA Synthesis Kit (R312) and the miRNA 1st Strand cDNA Synthesis Kit (MR101). Quantitative PCR (qPCR) was performed following the guidelines of the Taq Pro Universal SYBR qPCR Master Mix (Q712) and the miRNA Universal SYBR qPCR Master Mix (MQ101). The sequences of the primers are listed in Supplementary Table 3.
The cells were treated according to the experimental groups. The culture medium from different groups was collected and centrifuged at 1000 × g for 20 minutes. Thes concentration of interferon (IFN)-γ, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-18 were measured using the Human IFN-γ enzyme-linked immunosorbent assay (ELISA) Kit (D711044-0096, Sangon, China), Human TNF-α ELISA Kit (PT518, Beyotime, China), Human IL-1β ELISA Kit (PI305, Beyotime, China), and Human IL-18 ELISA Kit (PI558, Beyotime, China) following the manufacturer’s instructions. Tumor tissue from the mice was treated with liquid nitrogen and thawed at 2-8 °C. After homogenization with a homoge
Apoptosis, necroptosis, and pyroptosis were detected using the YO-PRO-1/PI Apoptosis and Necrosis Detection Kit (C1075S, Beyotime, China). YO-PRO-1/PI working solution was prepared by adding YO-PRO-1 (1000 ×, 1 µL) and PI (1000 ×, 1 µL) to 998 µL of buffer. Following treatment in a six-well plate, the cells were rinsed with PBS buffer. The working solution (1 mL/well) was added to the wells, and the plate was incubated at 37 °C for 15 minutes. A fluo
PmirGLO-ZEB1 3'UTR-WT and PmirGLO-ZEB1 3'UTR-mut reporter plasmids were constructed on the basis of the predictive binding sites of miR-200a-3p and ZEB1 (Sangon Co., Ltd., Shanghai, China). The plasmid structure information is provided in Supplementary Figure 2. The cells were cotransfected with either miR-200a-3p mimics or NC, along with PmirGLO-ZEB1 3’UTR-WT and PmirGLO-ZEB1 3’UTR-mut reporter plasmids, following the manufacturer’s instructions. A dual-luciferase reporter assay system (RG027, Beyotime, China) was applied to evaluate the Firefly and Renilla luciferase activities 48 hours after transfection.
After being treated according to the experimental protocol, 1 × 107 cells were collected and spun for 5 minutes at a speed of 1000 × g. The polysome lysis buffer (900 μL) was mixed with the protease inhibitor (9 μL) and the RNase inhibitor (4.5 μL). DNA was removed from the lysate. The mixture was used to resuspend the cell lysate. Anti-Ago2 antibodies or control IgG was immobilized on magnetic beads. Immunoprecipitation was performed at 4 °C. The recovered RNA was subsequently amplified. The HiScript III 1st Strand cDNA Synthesis Kit (R312, Vazyme, China) was used to synthesize cDNA. qPCR was performed using the Taq Pro Universal SYBR qPCR Master Mix (Q712, Vazyme, China). The primer information is shown in Supplementary Table 3.
Protein was extracted using the RIPA buffer. Co-immunoprecipitation (CO-IP) assays were carried out using the Magnetic IP/Co-IP Kit (P2179M, Beyotime, China) following the manufacturer's guidelines. Protein A agarose (5 μL) and Protein G agarose (5 μL) were mixed with 500 μL of cell lysate and left to incubate for 1 hour. The supernatant was collected. An additional 500 μL of lysate was incubated with anti-ZEB1 or anti-IRF1 antibodies for 12 hours at 4 °C. Protein A/G magnetic beads were then added and incubated overnight. After centrifugation at 12000 × g and 4 °C for 1 minute, the antigen-antibody complexes were recovered. Following washing, the antigen-antibody complexes were eluted using an elution buffer. WB was employed to detect the expression levels of ZEB1 and IRF1.
Twenty-four male BALB/c nude mice (approximately 4 weeks old, Vital River Laboratory Animal Technology Co., Ltd., China) were used. Approximately 5 × 107 BGC-823 cells were subcutaneously administered into the right flank of each mouse. When the tumor grew to 100-150 mm³, the mice received treatment based on the following groupings (six mice in each group): Control group, receiving saline gavage (once daily for 4 weeks) and intraperitoneal injection of saline (every 3 days for 4 weeks); 5-FU group, receiving saline gavage (once daily for 4 weeks) and intraperitoneal injection of a 5-FU solution (50 mg/kg, every 3 days for 3 weeks); XSLJZD group, receiving XSLJZD [6.2 g/(kg·day), once daily for 3 weeks][22] and intraperitoneal injection of saline (every 3 days for 4 weeks); and 5-FU + XSLJZD group, receiving XSLJZD [6.2 g/(kg·day), once daily for 3 weeks] and intraperitoneal injection of a 5-FU solution (20 mg/kg, every 3 days for 3 weeks). After treatment, the mice were euthanized, and tumors were excised. Tumor samples were either snap-frozen in liquid nitrogen for WB analysis or preserved in 4% formaldehyde and embedded in paraffin as a preparatory step.
ImageJ software was applied to analyze the images. GraphPad Prism 9 (Version 9.5.1) was used for data analysis. Data were presented as mean ± SD from three independent replicates (n = 3). Homogeneity of variance was confirmed using Levene’s test (P > 0.05). Differences were assessed by one-way ANOVA followed by Tukey’s post-hoc test, with signifi
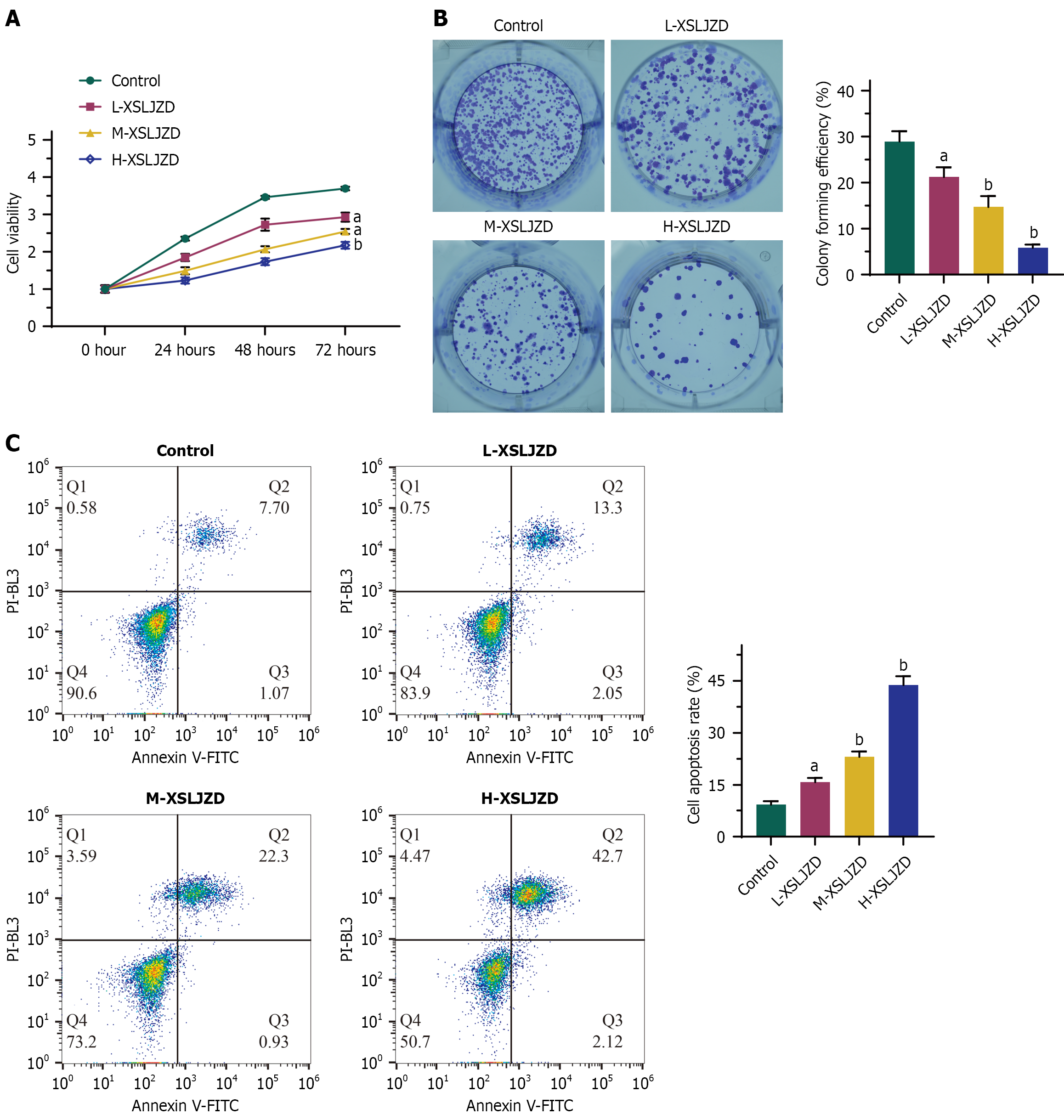
We treated BGC-823/5-FU cells with XSLJZD at low, medium, and high doses to evaluate its effects on 5-FU-resistant cells. XSLJZD significantly reduced cell viability in a dose-dependent manner, with the high-dose group exhibiting lower viability compared with the control group at 24, 48, and 72 hours (Figure 1A). Colony formation assays indicated that XSLJZD treatment dose-dependently decreased colony formation rates, with the high-dose group showing fewer colonies compared with the control (Figure 1B). Flow cytometry analysis of apoptosis revealed that XSLJZD dose-dependently increased the percentage of apoptotic cells (annexin V-FITC/PI positive), with the high-dose group showing the highest apoptosis rate (Figure 1C). These findings collectively confirm that XSLJZD inhibits cell viability and proliferation while promoting apoptosis in BGC-823/5-FU cells.
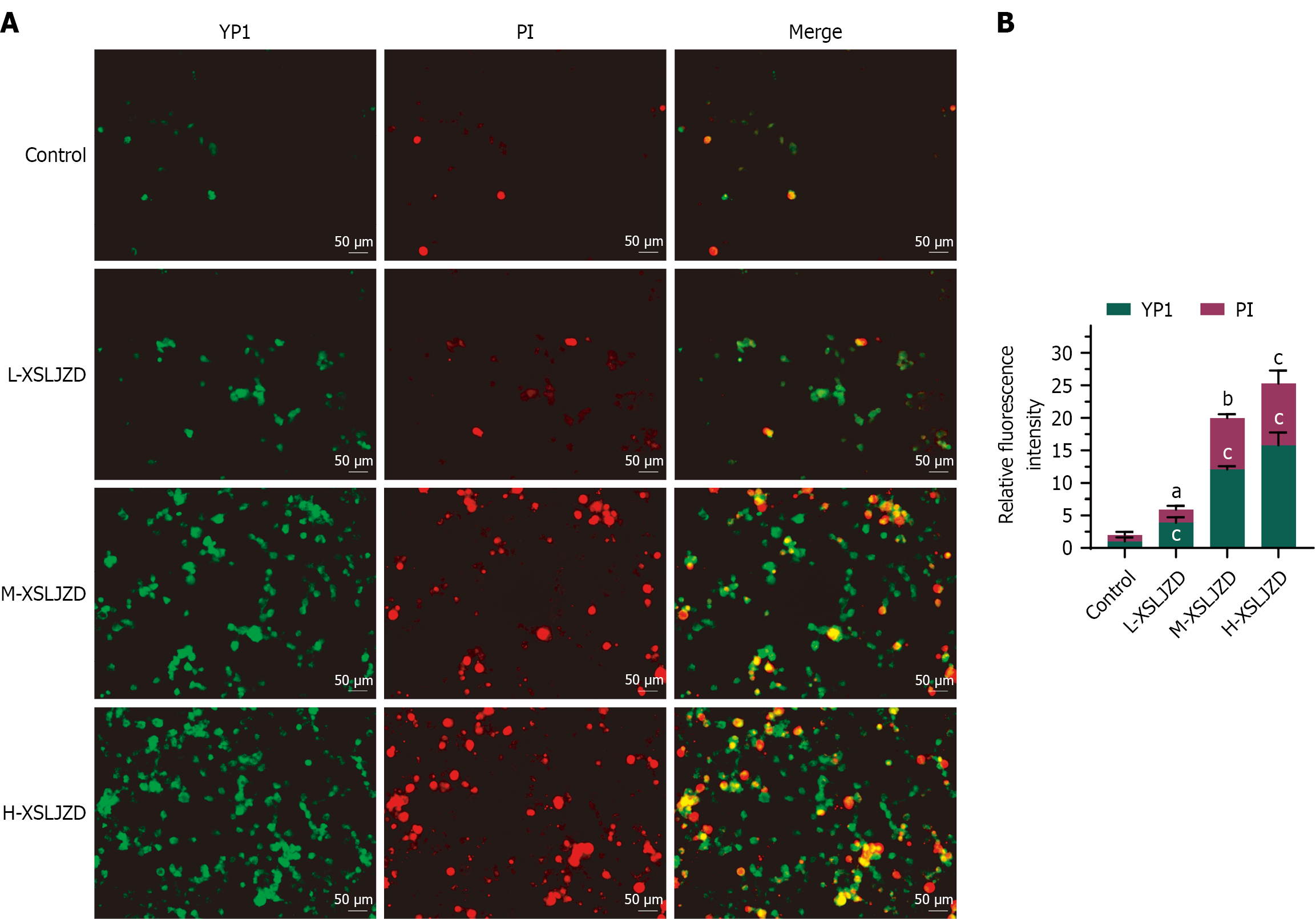
We used YO-PRO-1/PI staining to further assess XSLJZD’s impact on various forms of programmed cell death (PCD) in BGC-823/5-FU cells. Figure 2A shows representative fluorescence microscopy images, where YO-PRO-1-positive cells (green fluorescence) indicate early apoptosis or necroptosis due to initial membrane permeability, YO-PRO-1/PI double-positive cells (green/red fluorescence) suggest late apoptosis or necrosis, and PI-positive cells (red fluorescence) indicate necroptosis or pyroptosis due to rapid membrane rupture. XSLJZD treatment dose-dependently increased YO-PRO-1-positive and PI-positive cell populations. Quantification of relative fluorescence intensity revealed that the high-dose XSLJZD group showed significantly higher fluorescence for YO-PRO-1 and PI compared with the control group, confirming the enhanced induction of apoptosis, necroptosis, and pyroptosis.
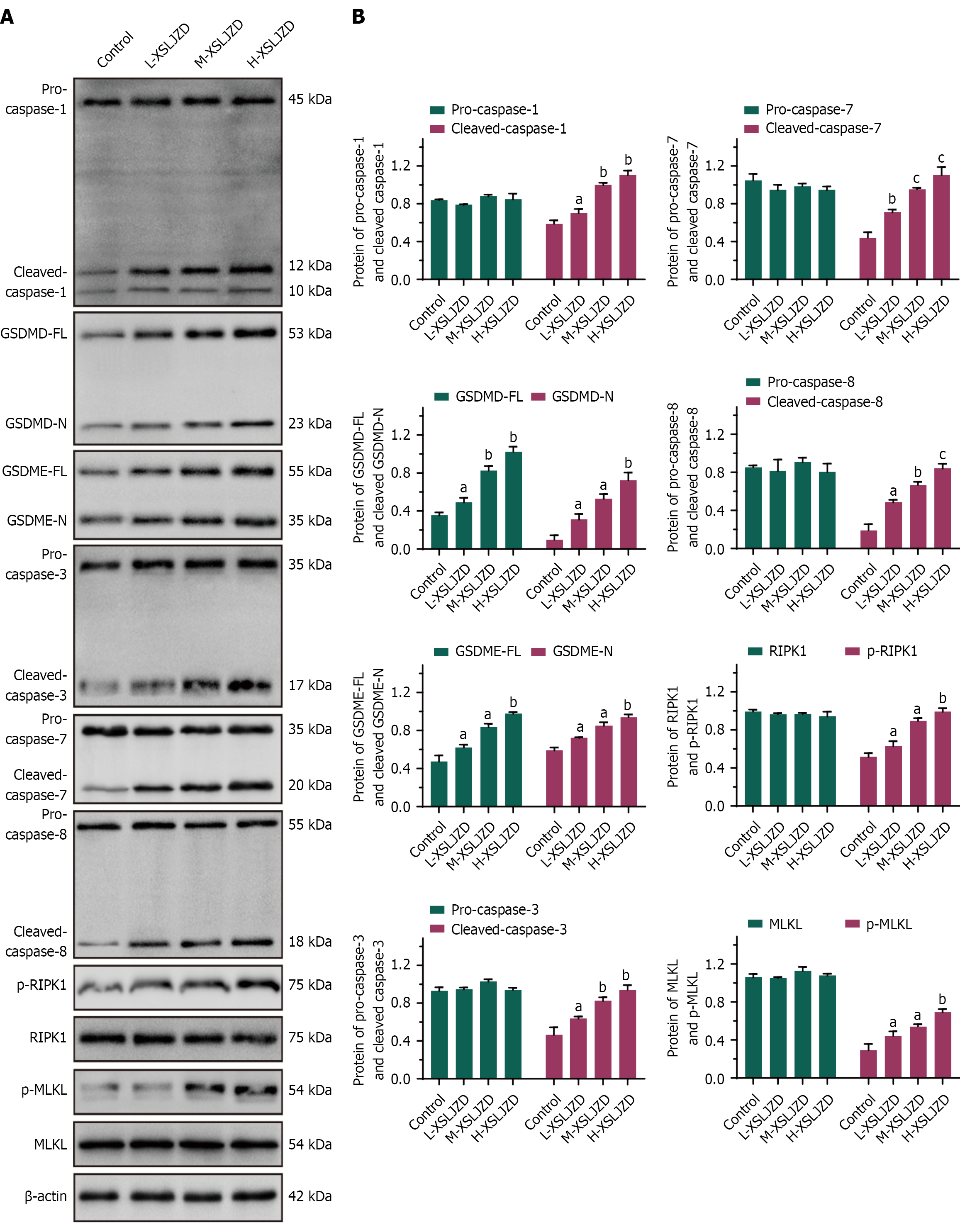
WB analysis was performed to investigate the effect of XSLJZD on PANoptosis-related protein expression in BGC-823/5-FU cells. Figure 3A shows representative WB bands for pyroptosis-related proteins (cleaved caspase-1, GSDMD-FL, GSDMD-N, GSDME-FL, and GSDME-N), apoptosis-related proteins (cleaved caspase-3, cleaved caspase-7, and cleaved caspase-8), and other PCD-related proteins (p-RIPK1 and p-MLKL). Figure 3B demonstrates the relative protein expression levels. All these proteins were up-regulated in the cells exposed to XSLJZD. Their levels increased with the dose of XSLJZD. By contrast, the levels of pro-caspase-1, pro-caspase-3, pro-caspase-7, pro-caspase-8, RIPK1, and MLKL were unaffected by XSLJZD treatment.
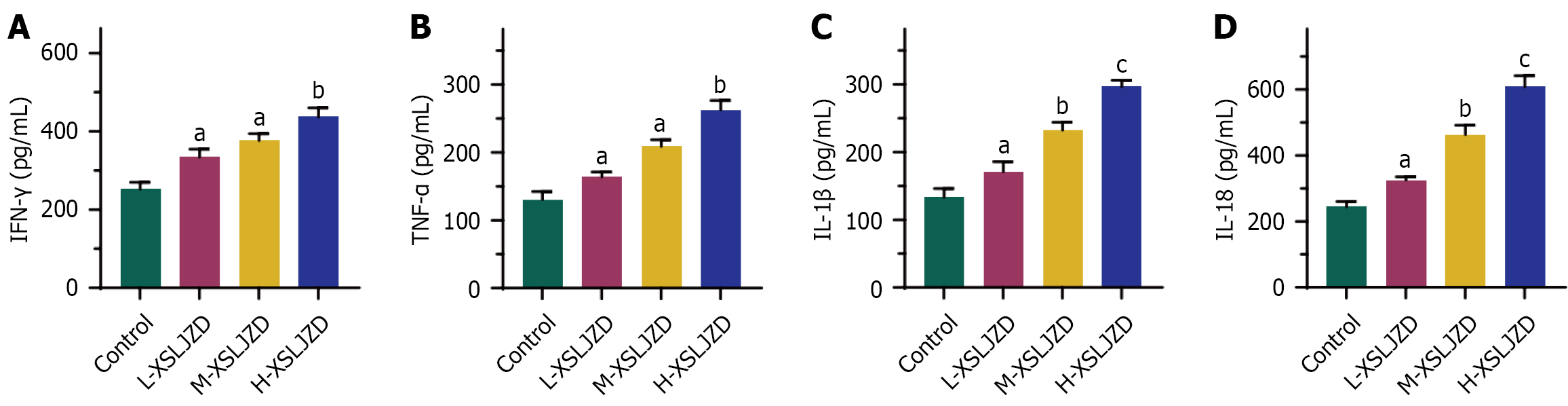
We measured the concentration of IFN-γ, TNF-α, IL-1β, and IL-18 in the cell culture medium of different XSLJZD treatment groups and control groups using ELISA. The IFN-γ levels in the culture medium dose-dependently increased with XSLJZD treatment, with the high-dose group exhibiting the highest concentration (Figure 4A). A similar dose-dependent increase was observed for TNF-α secretion (Figure 4B). Figure 4C illustrates elevated IL-1β levels, with significant increases in the medium- and high-dose groups. Figure 4D shows that IL-18 secretion was significantly enhanced in all XSLJZD-treated groups, with the high-dose group showing the greatest increase. These increases in PANoptosis-associated cytokines support the induction of apoptosis, pyroptosis, and necroptosis, as corroborated by YO-PRO-1/PI staining (Figure 2) and WB analysis of specific PCD markers (Figure 3). However, direct evidence of PANoptosome formation was not assessed in this study.
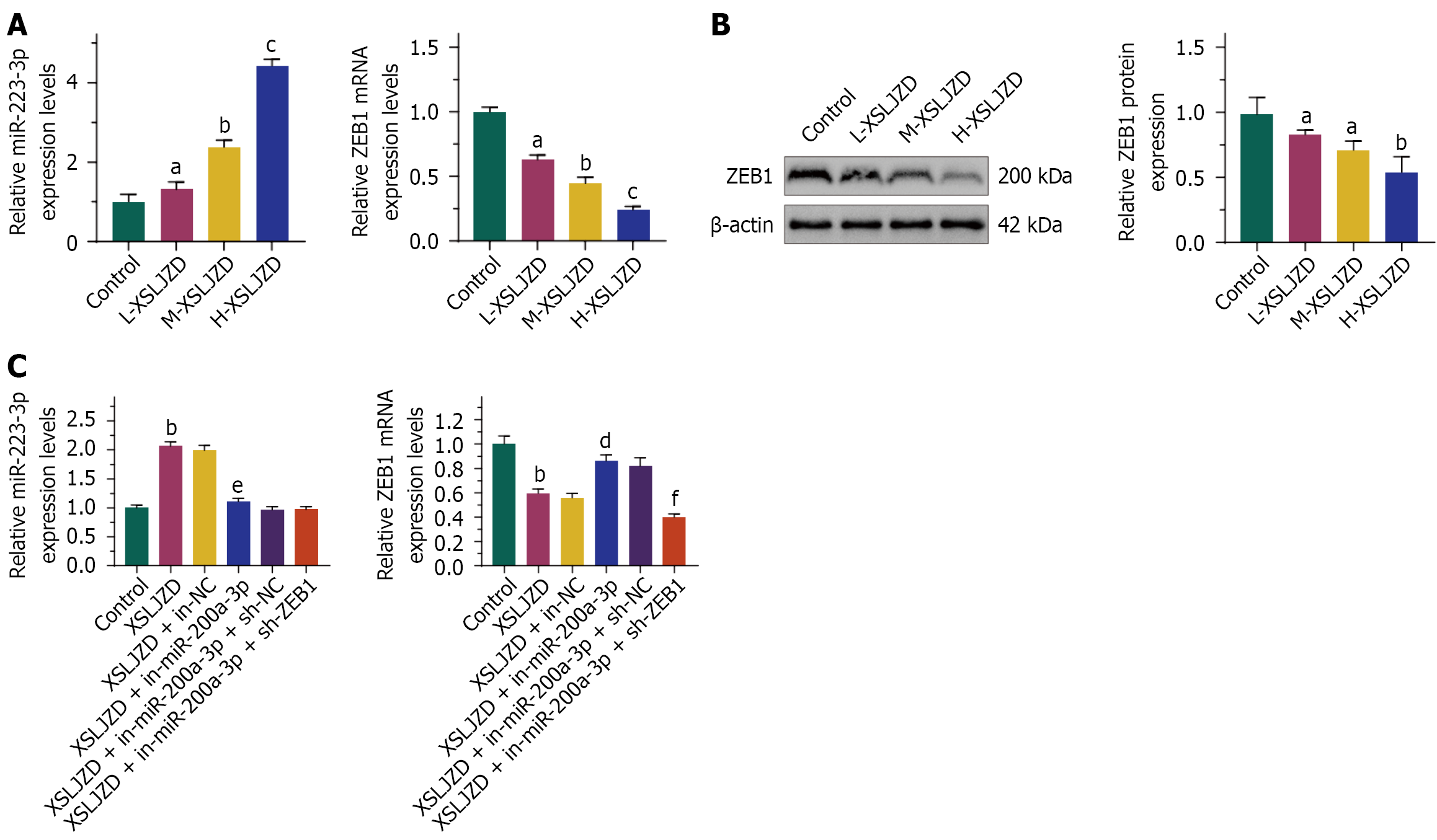
On the basis of prior reports of miR-200a-3p downregulation in gastric cancer and its role in chemoresistance via ZEB1, we investigated its regulation by XSLJZD in BGC-823/5-FU cells. We examined the expression of miR-200a-3p and ZEB1 in BGC-823/5-FU cells exposed to XSLJZD. MiR-200a-3p expression was significantly increased after treatment with XSLJZD, whereas ZEB1 expression was decreased. The influence of XSLJZD on miR-200a-3p and ZEB1 expression was dose dependent (Figure 5A and B). Inhibiting miR-200a-3p in the XSLJZD group partially restored ZEB1 expression (Figure 5C). These data imply that ZEB1 expression is influenced by miR-200a-3p.
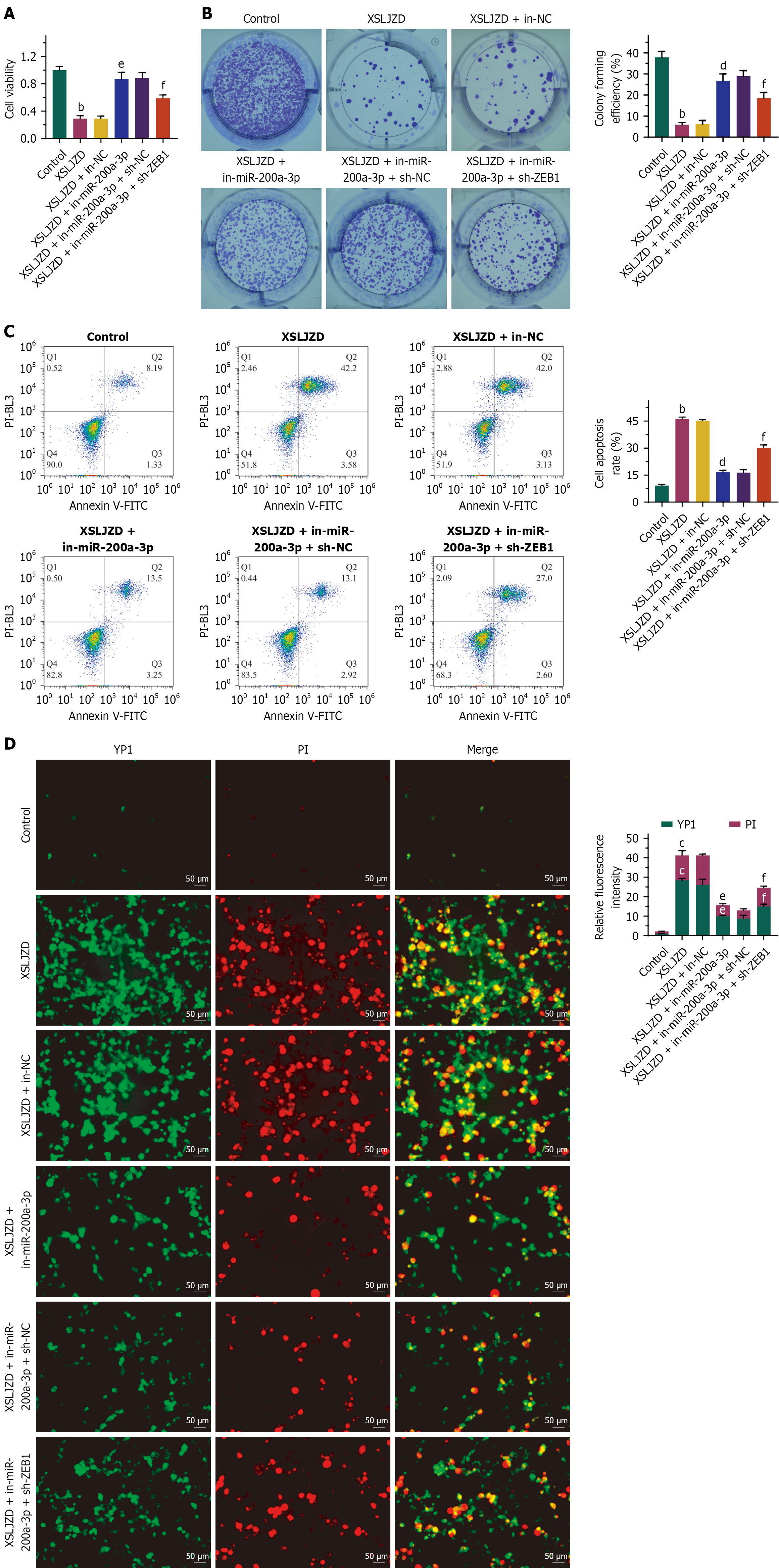
Cell viability and proliferation were significantly suppressed after treatment with XSLJZD (Figure 6A and B). By contrast, cell apoptosis was significantly increased in the XSLJZD-treated group (Figure 6C). In addition, cell apoptosis/necroptosis (YP1 positive) and necroptosis/pyroptosis (PI positive) were elevated in the XSLJZD-treated group (Figure 6D). The XSLJZD-induced effects were partially abolished after miR-200a-3p inhibitors were transfected into the 5-FU-resistant cells. Moreover, knocking down ZEB1 expression in 5-FU-resistant cells with miR-200a-3p inhibition restored the effects of XSLJZD on cell viability, proliferation, and PANoptosis (Figure 6) to some extent. These results demonstrate that the impacts of XSLJZD on cell viability, proliferation, and apoptosis are mediated partially by the miR-200a-3p/ZEB1 axis.
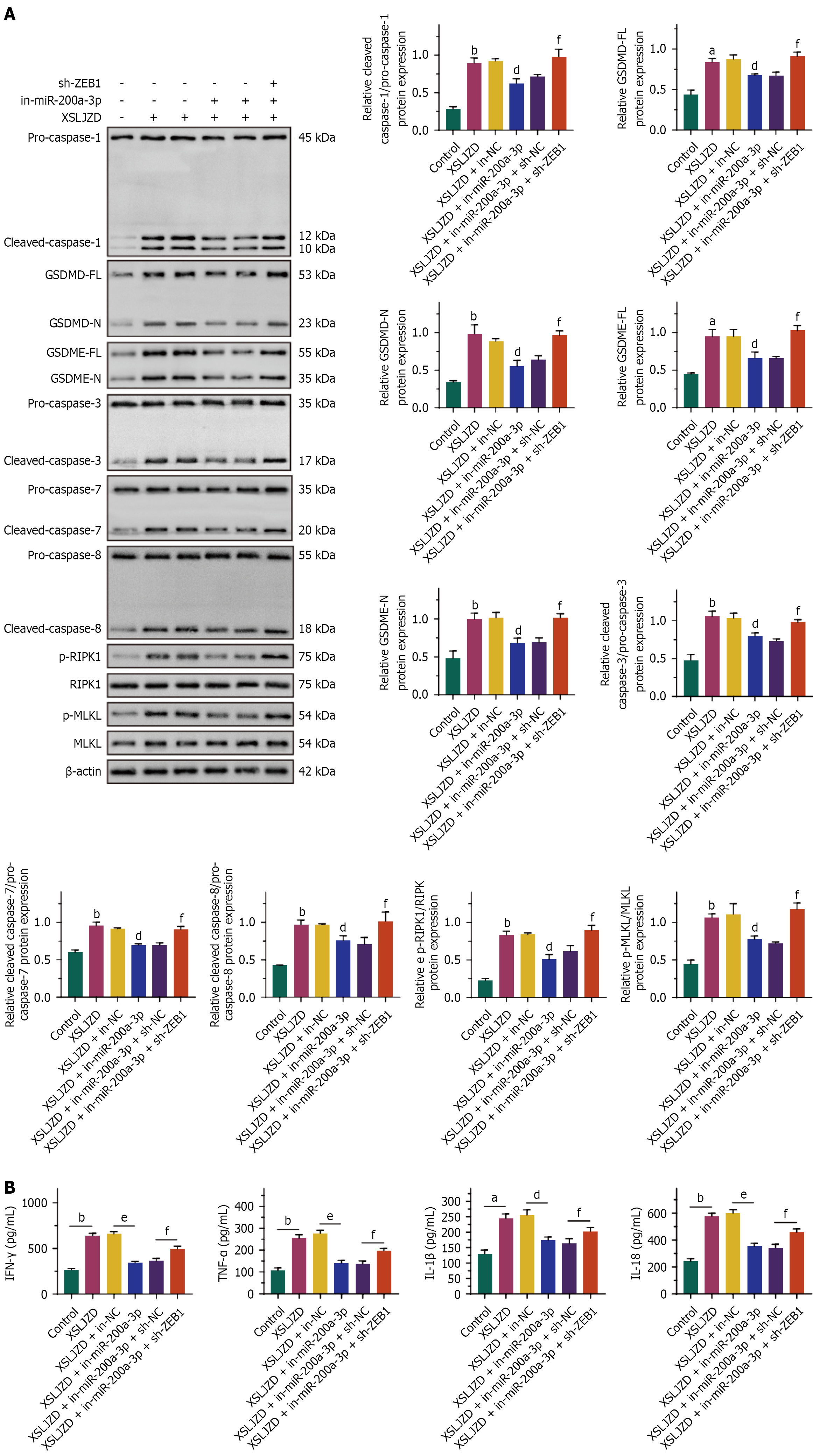
XSLJZD promoted the expression of pyroptosis-, apoptosis-, and PCD-related proteins without affecting the expression of pro-caspase-1, pro-caspase-3, pro-caspase-7, pro-caspase-8, RIPK1, and MLKL. In addition, XSLJZD upregulated the secretion of IFN-γ, TNF-α, IL-1β, and IL-18 in BGC-823/5-FU cells. The transfection of miR-200a-3p inhibitors into BGC-823/5-FU cells partially abolished the effects of XSLJZD on the expression of PANoptosis-related proteins and inflammatory factors. Furthermore, knocking down ZEB1 n in BGC-823/5-FU cells with miR-200a-3p inhibition partially restored the effects of XSLJZD on PANoptosis-related proteins and inflammatory factor expression (Figure 7).
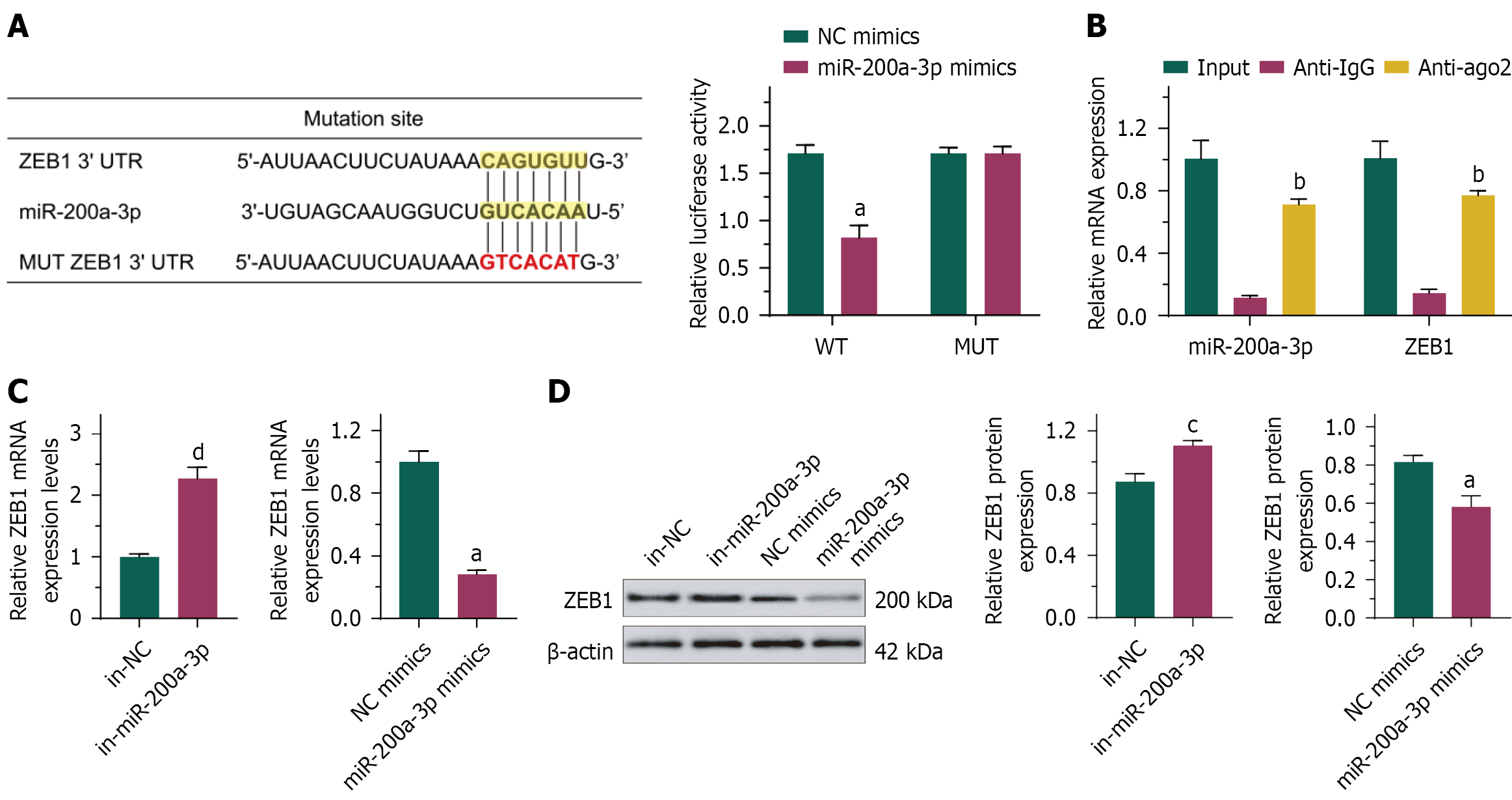
According to the dual-luciferase reporter assay results, WT ZEB1 3’UTR reduced luciferase activity, whereas MUT ZEB1 3’UTR had no impact (Figure 8A). RNA immunoprecipitation (RIP) assay was conducted to explore how miR-200a-3p interacts with ZEB1. The enrichment of miR-200a-3p and ZEB1 was observed in the RNA-induced silencing complex after immunoprecipitation using the Ago2 antibody (Figure 8B). Knocking down miR-200a-3p led to an elevation of ZEB1 expression, whereas overexpressing miR-200a-3p decreased ZEB1 expression (Figure 8).
Co-IP revealed that IRF1 was present in the ZEB1 immunoprecipitation complex, and ZEB1 was conversely detected in the IRF1 immunoprecipitation complex, indicating a direct interaction between these two proteins (Figure 9A). Conver
Tumor tissues from BALB/c nude mice (n = 6 per group) were excised and snap-frozen in liquid nitrogen for protein extraction. The levels of miR-200a-3p, ZEB1, and IRF1 mRNA in tumor samples were analyzed by real-time quantitative PCR. In contrast to the control group, the 5-FU and XSLJZD treatment groups exhibited higher miR-200a-3p and IRF1 mRNA expression and lower ZEB1 mRNA expression. In addition, the combination treatment group (5-FU + XSLJZD) showed even higher levels of miR-200a-3p and IRF1 mRNA and lower ZEB1 mRNA compared with the individual 5-FU and XSLJZD groups (Figure 10A). Further analysis of PANoptosis-related proteins and inflammatory cytokines in tumor tissues revealed that the 5-FU and XSLJZD groups had significantly increased expression of cleaved caspase-1, GSDMD-FL, GSDMD-N, GSDME-FL, GSDME-N, cleaved caspase-3, cleaved caspase-7, cleaved caspase-8, p-RIPK1, and p-MLKL. The combination treatment group (5-FU + XSLJZD) showed even further increased levels of these proteins compared with the 5-FU and XSLJZD groups. However, the expression of the precursor forms of these proteins, including pro-caspase-1, pro-caspase-3, pro-caspase-7, pro-caspase-8, RIPK1, and MLKL, did not show significant changes across the groups (Figure 10B). Moreover, the concentrations of IFN-γ, TNF-α, IL-1β, and IL-18 were significantly increased in the 5-FU and XSLJZD groups. The combination treatment group (5-FU + XSLJZD) further increased the levels of these inflammatory cytokines compared with the 5-FU and XSLJZD groups (Figure 10C).
In China, the traditional Chinese decoction XSLJZD has been widely used to alleviate treatment-related adverse events in patients with cancer, particularly chemotherapy-induced nausea and vomiting[23]. However, its antitumor effects are rarely reported. In this study, we found that XSLJZD exerts an antitumor effect on 5-FU-resistant gastric cells by sup
We further explored the regulatory mechanism of XSLJZD on PANoptosis. Previous studies showed that miRNAs contribute to the formation of 5-FU resistance in gastric cancer. Some miRNAs play an oncogenic role, promoting 5-FU resistance. For instance, miR-BART20-5p promotes 5-FU resistance by downregulating BAD in gastric cancer cells[26]. Similarly, miR-193-3p enhances 5-FU resistance by inhibiting PTEN expression[27], and miR-17 induces resistance by suppressing DEDD expression[28]. Some miRNAs function as tumor suppressors and help overcome 5-FU resistance. For example, miR-429 decreases resistance to 5-FU by silencing Bcl-2 expression[29], and miR-BART15-3p sensitizes cancer cells to 5-FU by suppressing Tax1-binding protein 1 expression[30]. We found that XSLJZD upregulated miR-200a-3p in gastric cancer cells. Inhibiting miR-200a-3p abolished the therapeutic effect of XSLJZD in XSLJZD-treated gastric cancer cells. These results suggest that XSLJZD overcomes, at least partially, 5-FU resistance in gastric cancer cells in a miR-200a-3p-dependent manner, indicating that miR-200a-3p acts as a tumor suppressor in the context of 5-FU resistance in gastric cancer. By binding to the 3’UTR of the mRNA, miRNA regulates gene expression and thereby influences the biological processes of cancer cells. Through online predictive software, ZEB1 was identified as one of the potential targets of miR-200a-3p. We further confirmed the interaction between ZEB1 and miR-200a-3p using dual luciferase and RIP assays. Previous studies pointed out that ZEB1 induces chemoresistance by promoting the clearance of chemotherapy-induced DNA damage[31]. The high ZEB1 expression in patients with breast cancer has been associated with poor response to chemotherapy[32]. Similarly, ZEB1 expression is elevated in cisplatin-resistant ovarian cancer cells, and its knockdown increases sensitivity to cisplatin[33].
We demonstrated that XSLJZD overcomes 5-FU resistance by upregulating miR-200a-3p. MiR-200a-3p inhibition or ZEB1 knockdown diminished the effect of XSLJZD, suggesting that XSLJZD reverses 5-FU resistance via the miR-200a-3p/ZEB1 axis. These results align with previous reports on the role of the miR-200a-3p/ZEB1 axis in promoting chemoresistance. How miR-200a-3p/ZEB1 regulates PANoptosis warrants further investigation. IRF1 plays a critical role in PANoptosis by regulating the formation of the NLRP12-PANoptosome, which mediates PANoptosis development in response to TNF and IFNγ[34,35]. In respiratory virus infection, ZEB1 silences IRF1 expression[36]. However, our study revealed that a ZEB1/IRF1 complex is present in 5-FU-resistant cells. Knocking down ZEB1 led to an increase in IRF1 Levels. Interestingly, silencing IRF1 also elevated ZEB1 expression, suggesting the presence of a regulatory loop between ZEB1 and IRF1. The precise mechanisms underlying this mutual regulation remain unknown and need further explo
In summary, our study demonstrated that XSLJZD helps reduce 5-FU resistance in gastric cancer. The underlying mechanism involves XSLJZD promoting miR-200a-3p expression, which in turn induces PANoptosis through the miR-200a-3p/ZEB1/IRF1 axis. However, several limitations must be acknowledged. First, the standalone effects of miR-200a-3p inhibition or ZEB1 knockdown without XSLJZD were not evaluated, potentially overlooking their independent contributions to resistance. Second, the unequal 5-FU doses in vivo (50 mg/kg monotherapy vs 20 mg/kg combination) may confound the interpretations of synergy vs additivity. Third, pharmacodynamic data, including tumor growth curves, weights, and survival rates, were not comprehensively collected because of resource constraints. Finally, PANoptosome assembly was not directly confirmed, and no specific inhibitors were used to dissect individual cell death pathways. Future studies should address these gaps by incorporating equivalent doses, additional models, pathway-specific in
Our study demonstrates that XSLJZD overcomes 5-FU resistance in gastric cancer by upregulating miR-200a-3p, which in turn suppresses ZEB1 expression, thereby relieving the ZEB1-mediated inhibition of IRF1 and inducing PANoptosis. In vitro experiments showed that XSLJZD inhibits cell viability and proliferation while promoting apoptosis, necroptosis, and pyroptosis in 5-FU-resistant BGC-823/5-FU cells. This effect is mediated through the miR-200a-3p/ZEB1/IRF1 axis. In vivo xenograft results supported these findings with molecular evidence of increased levels of PANoptosis-related proteins and cytokines. However, limitations include the lack of in vivo pharmacodynamic data, standalone effects of miR-200a-3p/ZEB1 manipulations, and direct PANoptosome confirmation due to resource constraints. Future clinical trials are essential to validate XSLJZD as a therapeutic strategy for overcoming chemoresistance in gastric cancer.
The authors express their appreciation to staff in the Affiliated Hospital of Liaoning University of Traditional Chinese Medicine, for their technical assistance.
| 1. | Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, Jemal A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16785] [Cited by in RCA: 15069] [Article Influence: 7534.5] [Reference Citation Analysis (23)] |
| 2. | Karimi P, Islami F, Anandasabapathy S, Freedman ND, Kamangar F. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol Biomarkers Prev. 2014;23:700-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1420] [Cited by in RCA: 1392] [Article Influence: 116.0] [Reference Citation Analysis (9)] |
| 3. | Lordick F, Carneiro F, Cascinu S, Fleitas T, Haustermans K, Piessen G, Vogel A, Smyth EC; ESMO Guidelines Committee. Gastric cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022;33:1005-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1159] [Cited by in RCA: 1022] [Article Influence: 255.5] [Reference Citation Analysis (13)] |
| 4. | Sethy C, Kundu CN. 5-Fluorouracil (5-FU) resistance and the new strategy to enhance the sensitivity against cancer: Implication of DNA repair inhibition. Biomed Pharmacother. 2021;137:111285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 463] [Cited by in RCA: 351] [Article Influence: 70.2] [Reference Citation Analysis (2)] |
| 5. | Ocansey DKW, Qian F, Cai P, Ocansey S, Amoah S, Qian Y, Mao F. Current evidence and therapeutic implication of PANoptosis in cancer. Theranostics. 2024;14:640-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 81] [Article Influence: 40.5] [Reference Citation Analysis (0)] |
| 6. | Pan H, Pan J, Li P, Gao J. Characterization of PANoptosis patterns predicts survival and immunotherapy response in gastric cancer. Clin Immunol. 2022;238:109019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 120] [Article Influence: 30.0] [Reference Citation Analysis (0)] |
| 7. | Lin C, Lin P, Yao H, Liu S, Lin X, He R, Teng Z, Zuo X, Li Y, Ye J, Zhu G. Modulation of YBX1-mediated PANoptosis inhibition by PPM1B and USP10 confers chemoresistance to oxaliplatin in gastric cancer. Cancer Lett. 2024;587:216712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 8. | Qi F, Zhao L, Zhou A, Zhang B, Li A, Wang Z, Han J. The advantages of using traditional Chinese medicine as an adjunctive therapy in the whole course of cancer treatment instead of only terminal stage of cancer. Biosci Trends. 2015;9:16-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 402] [Cited by in RCA: 343] [Article Influence: 31.2] [Reference Citation Analysis (1)] |
| 9. | Zhang X, Qiu H, Li C, Cai P, Qi F. The positive role of traditional Chinese medicine as an adjunctive therapy for cancer. Biosci Trends. 2021;15:283-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 283] [Article Influence: 56.6] [Reference Citation Analysis (3)] |
| 10. | Liao YH, Li CI, Lin CC, Lin JG, Chiang JH, Li TC. Traditional Chinese medicine as adjunctive therapy improves the long-term survival of lung cancer patients. J Cancer Res Clin Oncol. 2017;143:2425-2435. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 134] [Article Influence: 14.9] [Reference Citation Analysis (5)] |
| 11. | Tian JX, Li M, Liao JQ, Liu WK, Tong XL. Xiangshaliujunzi Decoction for the treatment of diabetic gastroparesis: a systematic review. World J Gastroenterol. 2014;20:561-568. [PubMed] [DOI] [Full Text] |
| 12. | Han Y, Fan X, Fan L, Wu Y, Zhou Z, Wang G, Guo L, Gao W, Chen Y, Gao Q. Liujunzi decoction exerts potent antitumor activity in oesophageal squamous cell carcinoma by inhibiting miR-34a/STAT3/IL-6R feedback loop, and modifies antitumor immunity. Phytomedicine. 2023;111:154672. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Jia C, Zhang Y, Xie Y, Ren Y, Zhang H, Zhou Y, Gao N, Ding S, Han S. miR-200a-3p plays tumor suppressor roles in gastric cancer cells by targeting KLF12. Artif Cells Nanomed Biotechnol. 2019;47:3697-3703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 14. | Yu SJ, Yang L, Hong Q, Kuang XY, Di GH, Shao ZM. MicroRNA-200a confers chemoresistance by antagonizing TP53INP1 and YAP1 in human breast cancer. BMC Cancer. 2018;18:74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 15. | Liu N, Zeng J, Zhang X, Yang Q, Liao D, Chen G, Wang Y. [Involvement of miR-200a in chemosensitivity regulation of ovarian cancer]. Zhonghua Yi Xue Za Zhi. 2014;94:2148-2151. [PubMed] |
| 16. | Liu X, Chen L, Wang T. Overcoming cisplatin resistance of human lung cancer by sinomenine through targeting the miR-200a-3p-GLS axis. J Chemother. 2023;35:357-366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 17. | Wang Z, Chen Y, Lin Y, Wang X, Cui X, Zhang Z, Xian G, Qin C. Novel crosstalk between KLF4 and ZEB1 regulates gemcitabine resistance in pancreatic ductal adenocarcinoma. Int J Oncol. 2017;51:1239-1248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 31] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 18. | Title AC, Hong SJ, Pires ND, Hasenöhrl L, Godbersen S, Stokar-Regenscheit N, Bartel DP, Stoffel M. Genetic dissection of the miR-200-Zeb1 axis reveals its importance in tumor differentiation and invasion. Nat Commun. 2018;9:4671. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 102] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 19. | Cong N, Du P, Zhang A, Shen F, Su J, Pu P, Wang T, Zjang J, Kang C, Zhang Q. Downregulated microRNA-200a promotes EMT and tumor growth through the wnt/β-catenin pathway by targeting the E-cadherin repressors ZEB1/ZEB2 in gastric adenocarcinoma. Oncol Rep. 2013;29:1579-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 132] [Article Influence: 10.2] [Reference Citation Analysis (1)] |
| 20. | Yu J, Chen J, Yang H, Chen S, Wang Z. Overexpression of miR200a3p promoted inflammation in sepsisinduced brain injury through ROSinduced NLRP3. Int J Mol Med. 2019;44:1811-1823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 21. | Zheng M, Kanneganti TD. The regulation of the ZBP1-NLRP3 inflammasome and its implications in pyroptosis, apoptosis, and necroptosis (PANoptosis). Immunol Rev. 2020;297:26-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 495] [Cited by in RCA: 453] [Article Influence: 75.5] [Reference Citation Analysis (0)] |
| 22. | Zhang J, Wang X, Wang F, Tang X. Xiangsha Liujunzi Decoction improves gastrointestinal motility in functional dyspepsia with spleen deficiency syndrome by restoring mitochondrial quality control homeostasis. Phytomedicine. 2022;105:154374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 34] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 23. | Xiao H, Liu L, Ke S, Zhang Y, Zhang W, Xiong S, Zhang W, Ouyang J. Efficacy of Xiang-Sha-Liu-Jun-Zi on chemotherapy-induced nausea and vomiting: A protocol for systematic review and meta-analysis. Medicine (Baltimore). 2021;100:e25848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 24. | Yin X, Zhang H, Wang J, Bian Y, Jia Q, Yang Z, Shan C. lncRNA FLJ20021 regulates CDK1-mediated PANoptosis in a ZBP1-dependent manner to increase the sensitivity of laryngeal cancer-resistant cells to cisplatin. Discov Oncol. 2024;15:265. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 25. | Huang J, Jiang S, Liang L, He H, Liu Y, Cong L, Jiang Y. Analysis of PANoptosis-Related LncRNA-miRNA-mRNA Network Reveals LncRNA SNHG7 Involved in Chemo-Resistance in Colon Adenocarcinoma. Front Oncol. 2022;12:888105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (4)] |
| 26. | Kim H, Choi H, Lee SK. Epstein-Barr virus miR-BART20-5p regulates cell proliferation and apoptosis by targeting BAD. Cancer Lett. 2015;356:733-742. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 82] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 27. | Jian B, Li Z, Xiao D, He G, Bai L, Yang Q. Downregulation of microRNA-193-3p inhibits tumor proliferation migration and chemoresistance in human gastric cancer by regulating PTEN gene. Tumour Biol. 2016;37:8941-8949. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 28. | Wu DM, Hong XW, Wang LL, Cui XF, Lu J, Chen GQ, Zheng YL. MicroRNA-17 inhibition overcomes chemoresistance and suppresses epithelial-mesenchymal transition through a DEDD-dependent mechanism in gastric cancer. Int J Biochem Cell Biol. 2018;102:59-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Zhu P, Zhang J, Zhu J, Shi J, Zhu Q, Gao Y. MiR-429 Induces Gastric Carcinoma Cell Apoptosis Through Bcl-2. Cell Physiol Biochem. 2015;37:1572-1580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 30. | Choi H, Lee SK. TAX1BP1 downregulation by EBV-miR-BART15-3p enhances chemosensitivity of gastric cancer cells to 5-FU. Arch Virol. 2017;162:369-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 31. | Zhang P, Wei Y, Wang L, Debeb BG, Yuan Y, Zhang J, Yuan J, Wang M, Chen D, Sun Y, Woodward WA, Liu Y, Dean DC, Liang H, Hu Y, Ang KK, Hung MC, Chen J, Ma L. ATM-mediated stabilization of ZEB1 promotes DNA damage response and radioresistance through CHK1. Nat Cell Biol. 2014;16:864-875. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 390] [Cited by in RCA: 386] [Article Influence: 32.2] [Reference Citation Analysis (4)] |
| 32. | Zhang X, Zhang Z, Zhang Q, Zhang Q, Sun P, Xiang R, Ren G, Yang S. ZEB1 confers chemotherapeutic resistance to breast cancer by activating ATM. Cell Death Dis. 2018;9:57. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 87] [Cited by in RCA: 89] [Article Influence: 11.1] [Reference Citation Analysis (4)] |
| 33. | Cui Y, Qin L, Tian D, Wang T, Fan L, Zhang P, Wang Z. ZEB1 Promotes Chemoresistance to Cisplatin in Ovarian Cancer Cells by Suppressing SLC3A2. Chemotherapy. 2018;63:262-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 34. | Karki R, Sharma BR, Tuladhar S, Williams EP, Zalduondo L, Samir P, Zheng M, Sundaram B, Banoth B, Malireddi RKS, Schreiner P, Neale G, Vogel P, Webby R, Jonsson CB, Kanneganti TD. Synergism of TNF-α and IFN-γ Triggers Inflammatory Cell Death, Tissue Damage, and Mortality in SARS-CoV-2 Infection and Cytokine Shock Syndromes. Cell. 2021;184:149-168.e17. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1530] [Cited by in RCA: 1343] [Article Influence: 268.6] [Reference Citation Analysis (2)] |
| 35. | Sundaram B, Pandian N, Mall R, Wang Y, Sarkar R, Kim HJ, Malireddi RKS, Karki R, Janke LJ, Vogel P, Kanneganti TD. NLRP12-PANoptosome activates PANoptosis and pathology in response to heme and PAMPs. Cell. 2023;186:2783-2801.e20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 349] [Cited by in RCA: 331] [Article Influence: 110.3] [Reference Citation Analysis (0)] |
| 36. | Yang J, Tian B, Sun H, Garofalo RP, Brasier AR. Epigenetic silencing of IRF1 dysregulates type III interferon responses to respiratory virus infection in epithelial to mesenchymal transition. Nat Microbiol. 2017;2:17086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |