Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.115162
Revised: December 15, 2025
Accepted: January 30, 2026
Published online: April 14, 2026
Processing time: 175 Days and 22.4 Hours
Early diagnosis of upper gastrointestinal bleeding (UGIB) relies on invasive endoscopy and laboratory tests, which carry procedural risks and diagnostic de
To investigate the feasibility of BS acoustic signatures as UGIB screening bio
A prospective study enrolled 40 UGIB patients (endoscopy-confirmed within 24 hours) and 40 age-/sex-matched healthy controls. BS signals were recorded at the right lower umbilical quadrant using a G-200 device (60 seconds/subject, 4 kHz sampling). After denoising via variational mode decomposition, 78-dimensional features were extracted across four domains: Time-domain, frequency-domain, time-frequency domain, and nonlinear dynamics. Weighted feature importance was calculated using an integrated strategy and gradient-optimized feature subsets were used to train four classifiers: Support vector machine, random forest, logistic regression, and K-nearest neighbor. SHapley Additive exPlanations analysis was conducted on the features of the optimal model. Model performance was evaluated by fivefold cross-validation. Spearman’s correlation analysis was performed to assess key BS features against red blood cell count, hemoglobin, hematocrit, C-reactive protein (CRP), and high-sensitivity CRP.
The support vector machine classifier with 25-feature subsets achieved optimal performance (area under the curve > 0.89), significantly outperforming other models. Acoustic feature importance analysis identified band_Energy and Mel-frequency cepstral coefficient variance as core biomarkers (cumulative contribution > 60%). Key pathological correlations included: (1) Significant negative correlations between spectral centroid and red blood cell count/hemoglobin/hematocrit (P < 0.01); (2) Positive correlation between wavelet entropy and these hematological parameters (P < 0.05), suggesting multiscale microcirculatory flow fluctuations; and (3) Positive wavelet energy correlations with CRP/high-sensitivity-CRP (P < 0.05).
Multidimensional BS features enable noninvasive UGIB screening. Their strong correlation with anemia/inflammation indicators reveals an acoustic-hemato-physiological coupling mechanism, providing a novel paradigm for early UGIB monitoring.
Core Tip: This study develops a machine learning-based screening tool to identify upper gastrointestinal bleeding (UGIB) patients using multidimensional bowel sound (BS) features. It also explores pathological associations between UGIB-related BSs characteristics and blood biochemical indicators, leveraging feature importance evaluation and SHapley Additive exPlanations analysis to identify interpretable predictors. This study pioneers a noninvasive BS analysis tool for UGIB screening, leveraging multidimensional acoustic feature engineering and machine learning optimization.
- Citation: Zhang L, Wang MY, Wei SY, Su C, Hu SY, Ren XY, Liu YP, Liu C, Wan Y. Predicting gastrointestinal bleeding and audio biomarkers based on machine learning analysis of bowel sounds. World J Gastroenterol 2026; 32(14): 115162
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/115162.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.115162
Upper gastrointestinal bleeding (UGIB) refers to hemorrhage originating from the gastrointestinal (GI) tract proximal to the ligament of Treitz, encompassing bleeding from lesions in the esophagus, stomach, duodenum, or pancreas/biliary tract. It manifests clinically as fecal occult blood, anemia, hematemesis, melena, or hematochezia. The global annual incidence of UGIB ranges from 47 to 150 cases per 100000 individuals. If the blood loss exceeds 20% of the circulatory blood volume (approximately 1 L) and is not promptly diagnosed and managed, it can lead to hemorrhagic shock and multiple organ failure. The overall mortality rate associated with UGIB varies between 3% and 15%[1-3]. The etiology of UGIB involves peptic ulcers, esophageal or gastric variceal rupture, tumors, and other causes. Although endoscopy is considered the diagnostic gold standard for locating the bleeding site and performing therapeutic interventions, and is often supplemented by imaging and laboratory investigations, these methods generally suffer from limitations such as suboptimal timeliness, inherent invasiveness in some cases, and elevated costs. Consequently, the development of rapid, noninvasive, and accurate technologies for early screening and diagnosis of UGIB holds significant potential[4].
Bowel sounds (BSs) are noninvasive, portable, and cost-effective physiological acoustic signals, enabling continuous monitoring of intestinal motility. They provide a novel means for real-time assessment of gut health[5,6]. Traditional clinical assessment of BSs relied on subjective qualitative auscultation using stethoscopes to guide diagnosis and management. However, this approach exhibits limited sensitivity and accuracy, detecting only acoustic events exceeding 20 dB, while lacking quantitative parameterization capabilities. Modern BS monitoring devices represent a technological breakthrough, enabling machine learning-driven feature parsing to establish clinically validated correlations with diverse intestinal pathologies. These portable systems further facilitate extended dynamic monitoring in ambulatory settings[7-9]. Dimoulas et al[10] achieved quantitative characterization of BS patterns through wavelet time-frequency analysis, establishing an objective foundation for monitoring GI motility and investigating the acoustic correlates of intestinal disorders. Goto et al[6] described a two-step method for real-time BS identification under noisy conditions, intended for sepsis monitoring in the intensive care unit. Recent studies have utilized machine learning techniques combined with time-frequency domain features to explore the relationship between BS characteristics and pathological conditions[11-13]. These studies have demonstrated that integrating signal processing methods, such as variational mode decomposition (VMD), empirical mode decomposition (EMD), and wavelet-based transforms with machine learning modeling, can capture subtle feature distinctions imperceptible to the human ear. This integration enables more objective, quantitative, and efficient identification and classification of disease states, thus significantly unlocking the potential of BSs for noninvasive diagnosis and therapeutic applications[14-16]. Wang et al[17] developed an integrated predictive model combining dual-channel BS monitoring with back-propagation neural networks, achieving 82.61% accuracy in acute GI injury severity classification. Key BS parameters (wave count, power, duration) showed strong correlations with acute GI injury progression, while model performance was enhanced through integration of clinical indicators including sequential organ failure assessment score, lactate levels, and mechanical ventilation status.
While BS analysis shows significant promise and leverages technological advances empowered by machine learning for diagnosing other GI disorders, research directly investigating its correlation with UGIB, a condition characterized by high incidence, high risk, and acute severity, remains to be explored. Recent studies have demonstrated a strong correlation between sound features and blood glucose levels in patients with cystic-fibrosis-related diabetes, offering an innovative, noninvasive monitoring pathway for diabetes management[18]. A recent study led by Goto et al[6] used BS monitoring systems to gain deeper insights into the relationship between exercise and inflammation; the latter indicated by serum interleukin-6 levels. In light of these correlations between body sounds and clinically significant indicators, a compelling rationale emerges: Investigating whether BSs have a measurable relationship with UGIB could provide a novel, noninvasive monitoring pathway for this critical condition.
Addressing the aforementioned challenges, this study primarily aimed to provide a noninvasive, precise, and automated approach for the screening and early risk warning of UGIB, while simultaneously enhancing the understanding of acoustic-physiological alterations in the bowel following UGIB. The BS classification tool developed in this study is designed to serve as an adjunct screening method for emergency triage or pre-endoscopic evaluation in critically ill patients, particularly targeting populations with hemodynamic instability, advanced age, or contraindications to endoscopy (e.g., cardiopulmonary insufficiency), rather than replacing the existing gold-standard diagnostic methods. The specific objectives were as follows: (1) Developing an effective screening tool. To determine if machine learning models can accurately identify UGIB patients based on multidimensional features extracted from BSs; and (2) Delving into pathological associations to investigate whether clinically meaningful statistical associations exist between UGIB-related BS characteristics and relevant blood biochemical indicators. We propose leveraging feature importance evaluation and SHapley Additive exPlanations (SHAP) analysis to identify interpretable predictors.
The prospective study (August 2025 to September 2025) enrolled 40 UGIB patients confirmed by gastroscopy within 24 hours of onset and 40 age- and sex-matched healthy controls. Enrollment followed predefined inclusion and exclusion criteria and was conducted at The First Affiliated Hospital of Xi’an Jiaotong University and its partnered community centers.
BS signals were collected using the G-200 sound acquisition device at the standard anatomical location two-finger breadths inferior-right to the umbilicus (Figure 1). Each recording session lasted 60 seconds, with a sampling frequency set at 4 kHz. All participants were required to fast for ≥ 2 hours to minimize alimentary tract interference and standardize gastric motility. Subjects maintained a supine position with quiet, consistent breathing throughout recording to reduce respiratory artifacts and ensure stable abdominal wall tension. Concurrently, laboratory examination data from all UGIB patients were collected, including five key indicators: Red blood cell (RBC) count, hemoglobin level, hematocrit, and C-reactive protein (CRP) and high-sensitivity CRP (hs-CRP), providing a clinical basis for subsequent analysis.
Inclusion criteria were: (1) Age 18-80 years; (2) UGIB involving the esophagus, stomach, duodenum, or pancreatobiliary lesions proximal to the ligament of Treitz, confirmed by gastroscopy; (3) Enrollment within 24 hours of the bleeding event; (4) Conscious and cooperative for examinations; and (5) Having the required biochemical indicators within 3 days after gastroscopy.
Healthy control group inclusion criteria: (1) Age and sex matched (± 5 years) to the patient group; and (2) No history of digestive diseases and absence of symptoms such as abdominal pain, melena, or hematemesis within the last 3 months.
Common exclusion criteria for both groups: (1) History of intestinal obstruction, intestinal perforation, or abdominal surgery within the past 6 months; (2) Skin lesions on the abdominal wall at the auscultation site; (3) Use of prokinetic agents (e.g., mosapride) or antidiarrheal agents (e.g., loperamide) within 48 hours prior to enrollment; (4) Pregnant or lactating women; (5) New York Heart Association functional class III/IV or oxygen saturation < 90%; (6) Severe anemia requiring urgent blood transfusion; (7) Environmental noise > 40 dB during recording (measured according to ISO 362 standard); (8) Use of anticoagulants (international normalized ratio > 1.5) or antiplatelet agents (excluding aspirin) within the past week; and (9) Concurrent systemic inflammatory disease or malignancy.
Demographic characteristics, including age and sex, were statistically characterized for both UGIB patients and healthy control groups. Additionally, biochemical indicators of UGIB patients underwent formal distribution assessment using the Shapiro-Wilk normality test. All analyses were performed in Python 3.9 (NumPy 1.22.4, SciPy 1.8.1) under standard parametric assumptions unless otherwise specified.
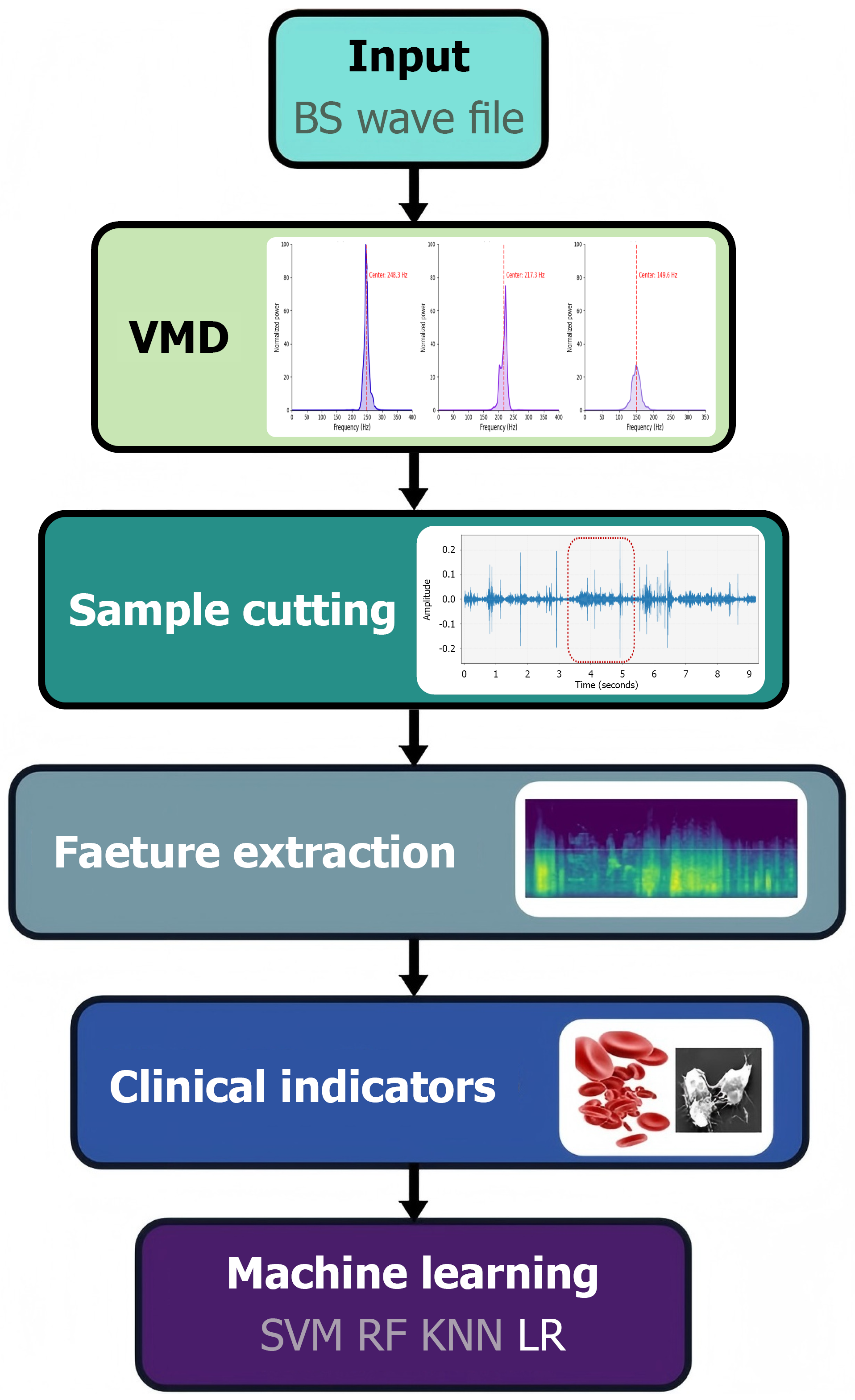
In audio analysis, signal decomposition is a commonly used denoising approach. As an adaptive, data-driven method, EMD has been widely applied across multiple domains[19]. It decomposes signals by sequentially generating intrinsic mode functions (IMFs) and subtracting them from the original signal. In contrast, VMD uses a nonrecursive strategy, generating all IMFs simultaneously and enabling flexible decomposition through parameter tuning[20]. Several studies have demonstrated the superior performance of VMD over EMD in practical applications. Maji and Pal[21] found that VMD better characterizes high-frequency components in electrocardiogram signals; Mohanty et al[20] demonstrated its advantage in capturing multitone signals with closely spaced frequencies. Considering the nonstationary nature of BS signals and interference from background noise, the VMD algorithm[22] was used for denoising in this study. Within this study, BSs were decomposed into five modes. Noise within the BS signal was discriminated against and removed through frequency spectral density analysis within distinct frequency bands[23].
Two senior gastroenterologists, operating in a dual-blind manner, segmented BS samples based on simultaneous visual inspection of audio waveforms and auditory analysis. Strict quality control was enforced by including only segments where both experts independently identified the same sound segment. To achieve sample balance across different categories and mitigate potential data bias in subsequent analyses, a randomized retention strategy was implemented: For each sample, three BS segments were randomly selected and retained (Figure 2).
Comprehensive feature extraction of BS signals was performed across four dimensions: Time domain, frequency domain, time-frequency domain, and nonlinear domain: (1) Time-domain analysis of the BS signals was conducted to extract 24 statistical parameters, including peak values, mean value, and variance. Additionally, waveform factors and zero-crossing rate-related parameters were calculated to quantify signal morphology and dynamic characteristics[24]; (2) The signal was transformed into the frequency domain using the fast Fourier transform. Key spectral parameters such as spectral centroid and bandwidth were extracted. The spectrum was partitioned into five sub-bands, and the energy within each frequency band was computed to analyze the distribution of signal frequency components; (3) Time-frequency domain features. Mel-frequency cepstral coefficients (MFCC) techniques were used to extract 13-dimensional static coefficients along with their first-order delta coefficients. These were supplemented by wavelet packet decompo
Four conventional machine learning models, support vector machine (SVM), random forest (RF), logistic regression (LR), and K-nearest neighbors (KNN), were constructed for GI hemorrhage detection. (1) Based on statistical learning theory, SVM uses the radial basis function kernel to handle linearly inseparable data, seeking an optimal hyperplane in the feature space. Hyperparameters, specifically the parameter for the radial basis function kernel and the penalty parameter, were optimized via cross-validation to balance model fitting and generalization performance; (2) As a bagging algorithm in ensemble learning, RF trains multiple decision trees using bootstrap sampling. Randomness is introduced by selecting a feature subset at each node during splitting. The final classification is determined by a majority voting mechanism among the trees, thereby mitigating overfitting risk; (3) Operating within the framework of generalized linear models, LR utilizes the logistic function to transform a linear output into the probability of a sample belonging to the positive class. Model parameters were optimized using gradient descent, offering favorable model interpretability; and (4) For each test sample, KNN identifies its K nearest neighbors in the training set based on Euclidean distance. The class label of the test sample is then determined by the majority class among these neighbors. The optimal K value was determined via grid search combined with cross-validation to adapt to complex data distributions. All feature extraction and model classification tasks in this experiment were performed using Python 3.9. Standard libraries including Pandas[26], Numpy[27], and Libros[28] were utilized for data processing and feature extraction. The machine learning models were implemented using their respective standard implementations available in the Scikit-learn library[29].
A multimethod approach was adopted to determine the importance ranking of all BS features. By integrating the SVM recursive feature elimination algorithm and the RF feature importance score, a multidimensional feature importance ranking framework was constructed. We conducted SHAP analysis on the features of the optimal model. This approach leveraged game theory to assign each feature an importance value, representing its marginal impact on model predic
Based on the established feature importance ranking, sequential feature subsets of progressively increasing size were constructed by incorporating features in descending order of importance. The four machine learning models were trained and evaluated using a fivefold cross-validation strategy. This involved randomly partitioning the dataset into five equal folds. During each iteration, four folds served as the training set and the remaining fold was used for validation. The average performance across all five validation cycles was calculated as the performance metric. Model classification performance was comprehensively evaluated using several metrics: Accuracy, F1-score, specificity, sensitivity, and the area under the curve (AUC). The performance of each model across the different feature subsets was compared to identify the optimal combination of features and model type. The selected subset of features demonstrating high contribution within the classification models was subsequently correlated with biochemical test indicators obtained during the hospitalization of patients with UGIB. Given that most biochemical indicators exhibited non-normal distributions, Spearman’s rank correlation coefficient was used to assess the relationships. Clinically significant feature-biomarker correlations were interpreted by drawing upon clinical knowledge to elucidate their intrinsic associations. This analysis explored the potential clinical utility of BS features as biomarkers and their applicability for clinical translation.
The healthy control group (n = 40) had a mean age of 57 years (range: 38-79 years), comprising 22 males and 18 females. The UGIB patients group (n = 40) had a higher mean age of 58 years (range: 42-78 years), with 22 males and 18 females. Individual matching was used to balance confounding factors (age and sex) between groups. After matching, no significant differences were found in age (P = 0.76) between the two groups, and sex distribution was completely con
| Age | Sex | ||||
| Median | Lower quartile | Upper quartile | Male | Female | |
| UGIB patients | 59.00 | 45.50 | 67.75 | 22 | 18 |
| Healthy controls | 58.50 | 46.75 | 66.75 | 22 | 18 |
| P value | > 0.05 | > 0.05 | |||
| Median | Lower quartile | Upper quartile | Medical reference range | |
| Red blood cell count, 1012/L | 3.05 | 2.77 | 3.51 | 4.3-5.8 |
| Hemoglobin, g/L | 90.5 | 83 | 105 | 130-175 |
| Hematocrit, % | 29.6 | 25.7 | 32.4 | 40-50 |
| CRP, mg/L | 6.8 | 6 | 24.65 | 0-10 |
| hs-CRP, mg/L | 6 | 2.84 | 6 | 0.3 |
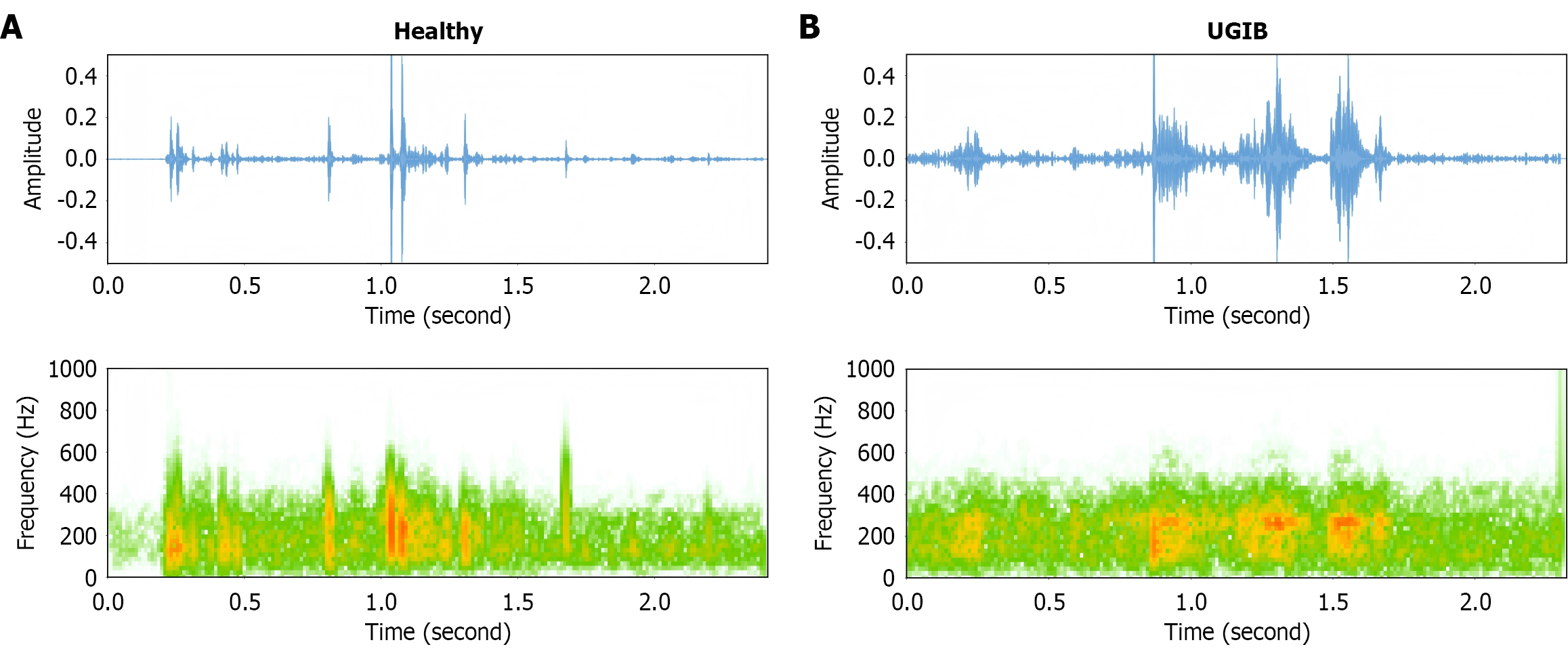
Figure 3 compares the BS signals of the healthy group and patients with UGIB, showing differences in both time-domain waveforms and spectrograms. The time-domain waveforms revealed disparities in amplitude and regularity. The healthy group showed waveforms with clear, intermittent pulse-like peaks and concentrated amplitude (y-axis) fluctuations. The UGIB group displayed more chaotic waveforms with larger amplitude fluctuations, irregular peak intervals, and prolonged duratios of high-amplitude segments (e.g., 1.2-1.8 seconds). The spectrograms demonstrated differences in frequency distribution. The healthy group showed energy concentrated primarily in lower frequency ranges (mainly 200-600 Hz), with a regular clustered distribution over time (x-axis). The UGIB group showed more dispersed energy distribution, significant energy in higher frequency ranges (600-1000 Hz), and irregular frequency variations (scattered energy distribution). Healthy BSs are characterized by temporal regularity and low-frequency, concentrated spectral features, whereas UGIB patients exhibit temporally chaotic BSs with high-frequency and dispersed spectral patterns. These acoustic differences may assist in evaluating pathological states of the digestive tract through BS analysis.
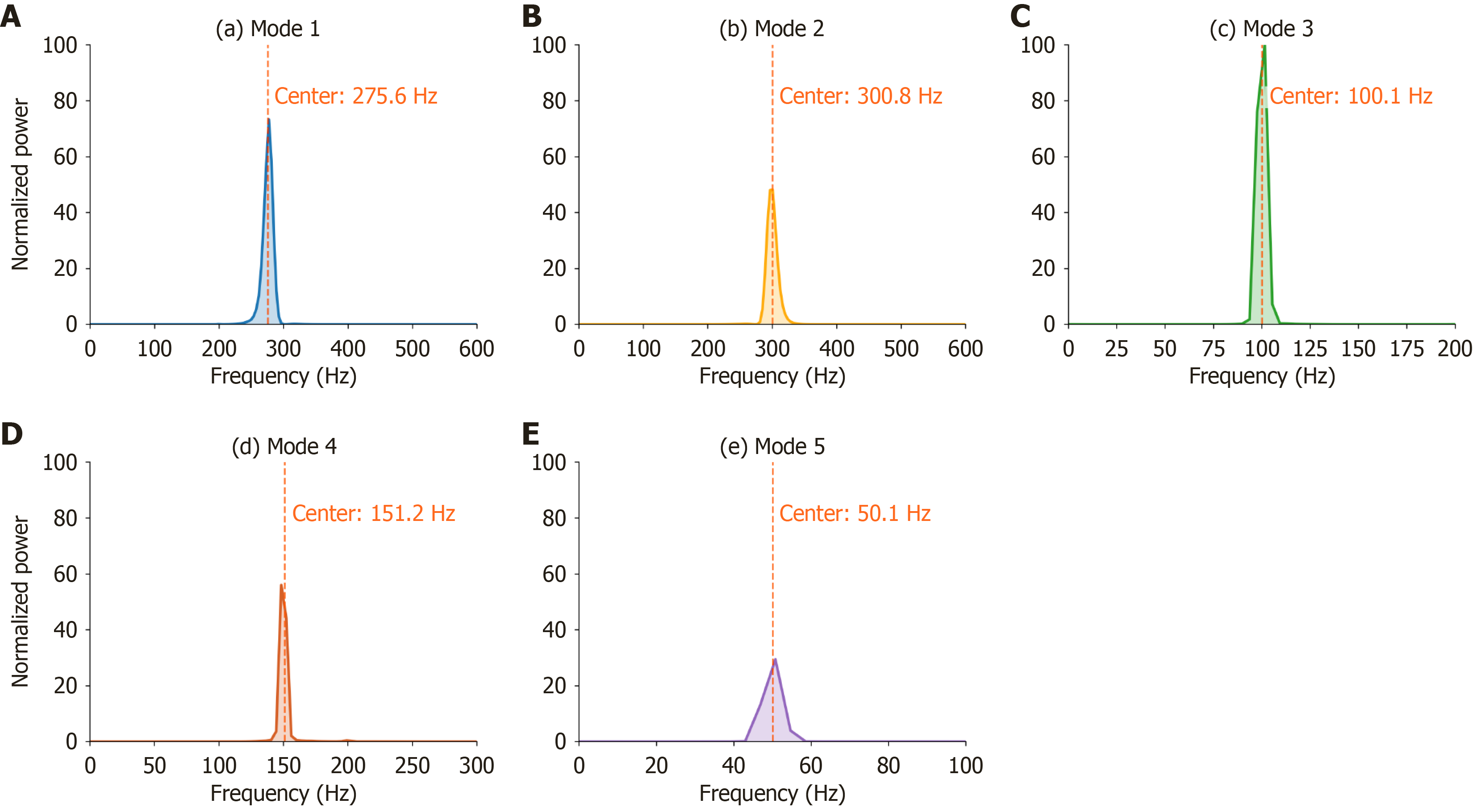
To enhance noise suppression in BS analysis, this study evaluated the modal decomposition performance of VMD. Figure 4 illustrates the power spectral characteristics of five IMFs in the frequency domain. All IMFs exhibited unimodal power distributions, with center frequencies at 300.8 Hz, 275.6 Hz, 151.2 Hz, 100.1 Hz, and 50.1 Hz, respectively. Accor
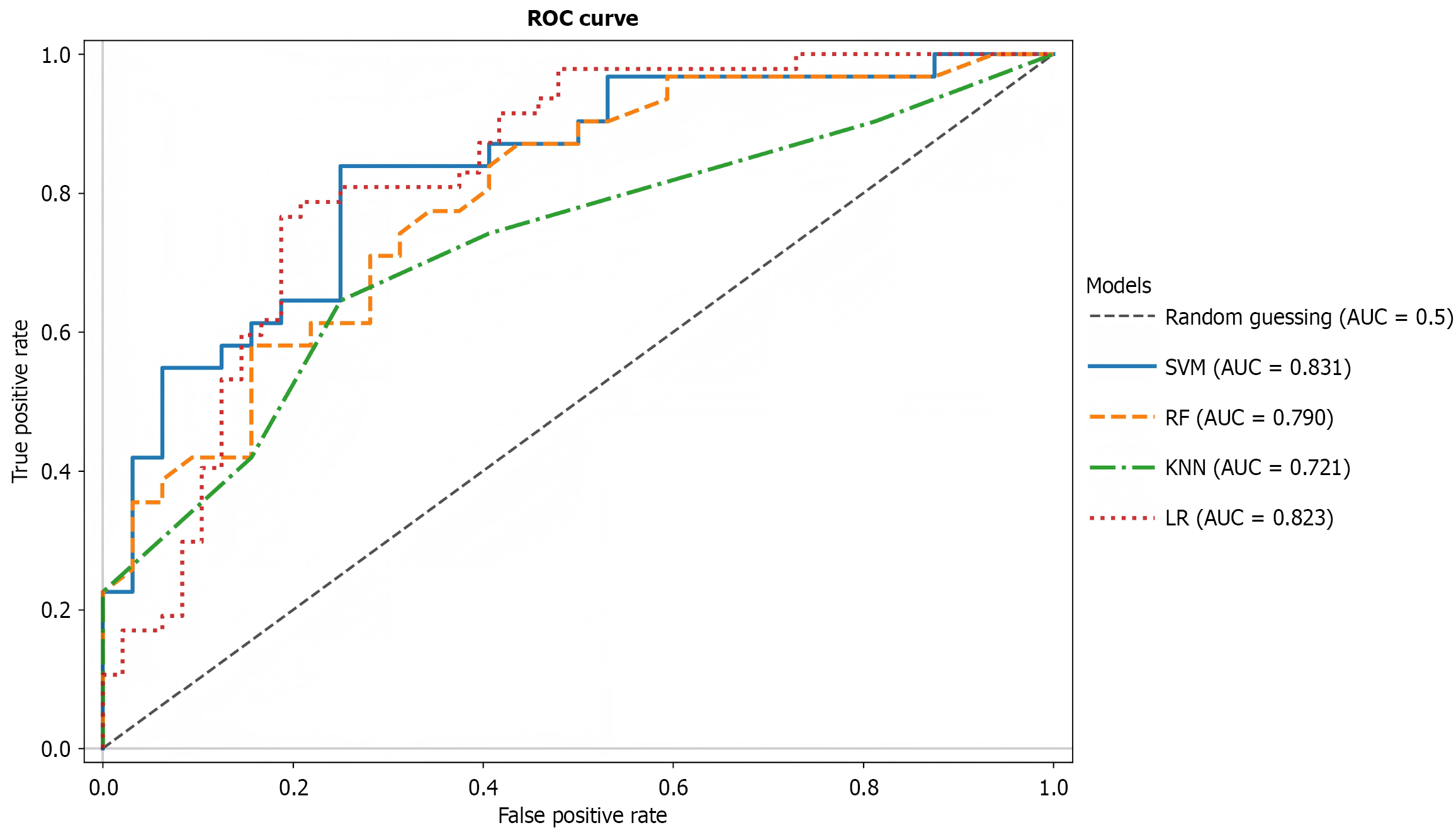
A total of 78 sound features were initially extracted from BSs. After excluding non-normally distributed features (MFCC mean_12, MFCC mean_11, and MFCC mean_4) via the Shapiro-Wilk normality test, 75 features were retained for model training. When utilizing all 75 features, the SVM demonstrated superior discriminative performance with an AUC of 0.831 (Figure 5), significantly outperforming other models (DeLong test, P < 0.01). The LR model ranked second (AUC = 0.823), while the RF and KNN models exhibited weaker performance (AUC = 0.772 and 0.721, respectively). As shown in Table 3, SVM achieved the highest comprehensive stability: Accuracy = 0.81, specificity = 0.875 (significantly higher than for other models, P < 0.01), and F1 score = 0.793; LR showed optimal sensitivity (0.766) but lower specificity (0.771)); RF displayed an imbalanced performance profile with high sensitivity (0.742) but low specificity (0.688); and KNN performed weakest across all metrics (accuracy = 0.698, AUC = 0.721).
| Feature type | Feature | Physical meaning |
| Time domain | Max, Min, Mean, Peak_to_Peak | Amplitude peak, minimum, arithmetic mean value, difference between maximum and minimum |
| Abs_Mean, Variance, Std_Dev, RMS | Amplitude absolute value average, variance, standard, root mean square | |
| Waveform_Length, Skewness | Cumulative change magnitude, signal distribution asymmetry | |
| Kurtosis, Crest_Factor | Signal peakiness, normalized fourth moment | |
| Zero_Crossing_Rate_Mean, Varian, Skewne | Signal oscillation frequency, oscillation degree, oscillation skewness characteristic | |
| Q1, Q3, interquartile range | Amplitude 25th, 75th percentile value, range between 25th and 75th | |
| Shape_Factor, Impluse_Factor | Regularity, peak component intensity of signals | |
| Margin_Factor, Hjorth_Activity, Hjorth Mobility | Signal large amplitude change index, the total power of the signal, contraction frequency fluctuation | |
| Autocoor_Peak, Fractal_Dimension, Energy, Avg_energy | Maximum signal self-similarity, signal complexity/irregularity, total squared amplitude, energy per sample | |
| Frequency domain | Spectral_Centroid, Slope, Bandwidth, Flatness | Signal frequency, variance around of centroid, tonal vs noise-like character |
| Spectral_Rolloff85, Rolloff95, Steepness | Frequency point with 85%, 95% total energy, the average energy ratio of high frequency to low frequency | |
| Band_Energy_[1-5] | 50-100 Hz, 100-500 Hz, 500-1000 Hz, 1000-2000 Hz, 2000-4000 Hz | |
| MFCC_Mean_[1-13] | Spectral envelope characteristics | |
| MFCC_Var_[1-13] | Spread of spectral envelope | |
| Spectral Steepness, PSD_Peak, Harmonic_Ratio | Average spectral slope, maximum power spectral density, harmonic content proportion | |
| Time-frequency domain | Wavelet_Energy_[0-2] | Wavelet coefficient energy |
| Wavelet_Kurtosis_[0-2] | Peakiness in wavelet coefficients | |
| STFT_Energy_Kurtosis | Peak concentration of short-time Fourier transform energy | |
| Nonlinear characteristics | Hjorth_Complexity | The complexity of signal changes |
| Wavelet_Entropy_[0-2], STFT_Energy_Entropy | Signal complexity in wavelet domain, uncertainty in time-frequency energy distribution |
Feature subsets comprising the top 20, 25, 30, and 40 features alongside the complete set of 75 features were constructed based on feature importance ranking. t-tests revealed significant sound features differences between healthy controls and UGIB patients. For MFCC, six variance parameters showed strong significance (MFCC_Var_2/3/4/5/6/7: P < 0.05), while only MFCC_Mean_3/4/7/13 (P < 0.05 exhibited significant mean differences. In Band_Energy, Band_Energy_2/3/4/5 (P < 0.055) were significantly distinct. These results highlight UGIB-induced disruptions in spectral stability (MFCC variances) and energy redistribution across mid-to-high frequency bands (Figure 4).
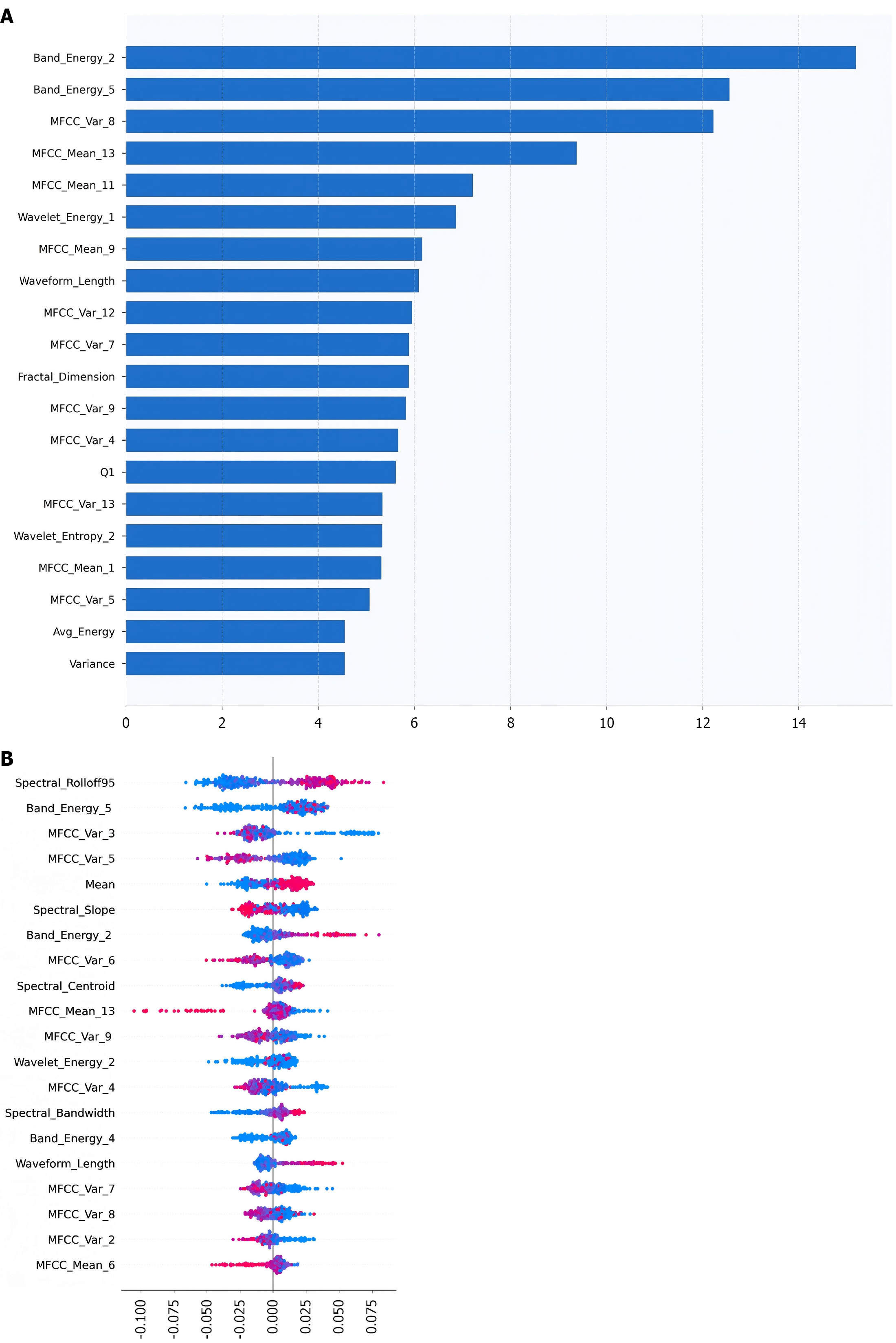
As illustrated in Figure 6A, the top 20 features identified by SVM-recursive feature elimination demonstrated key discriminative characteristics. Figure 6B visualizes SHAP values where color intensity represents feature magnitude and horizontal positioning indicates the direction and strength of influence on healthy probability prediction. Both figures consistently established Band_Energy and MFCC_Var as dominant predictive variables, with band_Energy_2 exhibiting the most pronounced effect gradient. Parallel findings emerged during feature importance analysis for SVM and RF, confirming band_energy_2 and band_energy_5 as major contributors. Collectively, features related to band_energy and MFCC_VAR accounted for > 60% of cumulative feature importance across models. Consequently, features were partitioned into graded subsets according to model-specific importance scores to further evaluate performance stability.
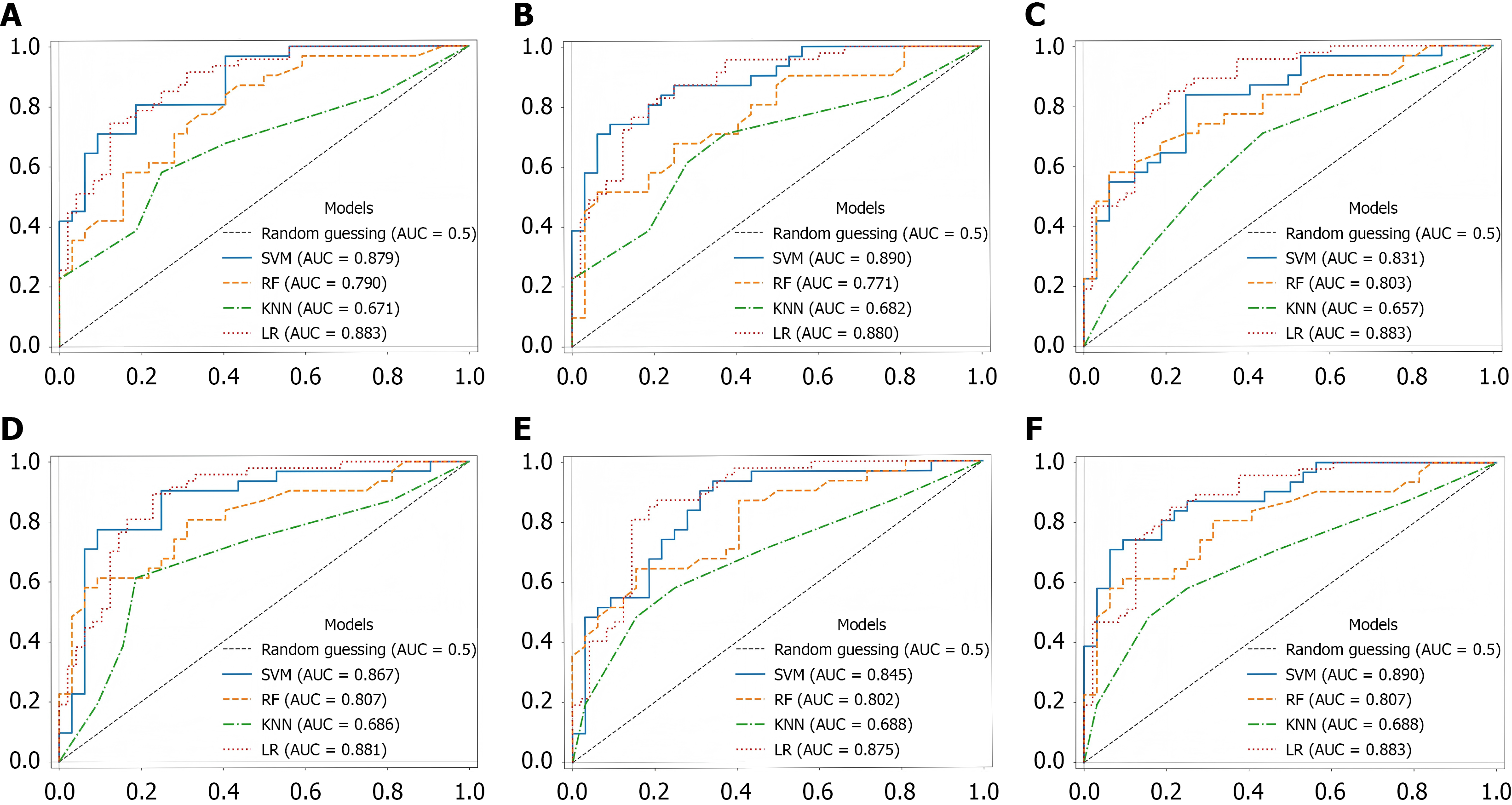
The SVM achieved peak discrimination (AUC = 0.890) at 25 features while demonstrating robust stability. LR reached optimal performance (AUC = 0.883) at 30 features, whereas RF showed maximum efficacy (AUC = 0.807) at 35 features (Figure 7). KNN consistently underperformed across all configurations. Complementary metrics in Tables 4 and 5 reveal that SVM attained 0.826 accuracy at 35 features with minimal fluctuation between subsets, contrasting with RF’s volatile accuracy (range: 0.683-0.762) and LR’s moderate stability (0.779-0.811), which remained inferior to those of SVM. Regarding sensitivity and specificity, SVM maintained balanced performance at 35 features (sensitivity = 0.774, specificity = 0.813-0.906). RF exhibited substantial sensitivity/specificity variability that may induce diagnostic errors during mass screening, necessitating supplementary clinical assessment. KNN demonstrated severe instability compromising clinical utility, while LR showed limited capability in detecting subtle pathological patterns compared to SVM despite relative stability.
| Algorithms | Accuracy | F1 score | Sensitivity | Specificity |
| SVM | 0.81 | 0.793 | 0.742 | 0.875 |
| RF | 0.714 | 0.719 | 0.742 | 0.688 |
| KNN | 0.698 | 0.678 | 0.645 | 0.75 |
| LR | 0.789 | 0.783 | 0.766 | 0.813 |
| Sensitivity | Specificity | Accuracy | F1 score | |||||||||||||||||
| Model | 40 | 35 | 30 | 25 | 20 | 40 | 35 | 30 | 25 | 20 | 40 | 35 | 30 | 25 | 20 | 40 | 35 | 30 | 25 | 20 |
| SVM | 0.581 | 0.774 | 0.71 | 0.71 | 0.71 | 0.813 | 0.875 | 0.875 | 0.906 | 0.844 | 0.698 | 0.826 | 0.794 | 0.81 | 0.778 | 0.655 | 0.814 | 0.772 | 0.786 | 0.759 |
| RF | 0.677 | 0.742 | 0.742 | 0.677 | 0.71 | 0.688 | 0.719 | 0.719 | 0.719 | 0.813 | 0.683 | 0.73 | 0.73 | 0.698 | 0.762 | 0.677 | 0.73 | 0.73 | 0.689 | 0.746 |
| KNN | 0.58 | 0.713 | 0.516 | 0.613 | 0.581 | 0.75 | 0.813 | 0.719 | 0.719 | 0.75 | 0.667 | 0.714 | 0.619 | 0.667 | 0.667 | 0.632 | 0.679 | 0.571 | 0.644 | 0.632 |
| LR | 0.702 | 0.713 | 0.723 | 0.713 | 0.744 | 0.854 | 0.854 | 0.875 | 0.865 | 0.875 | 0.789 | 0.779 | 0.8 | 0.789 | 0.811 | 0.773 | 0.759 | 0.782 | 0.767 | 0.795 |
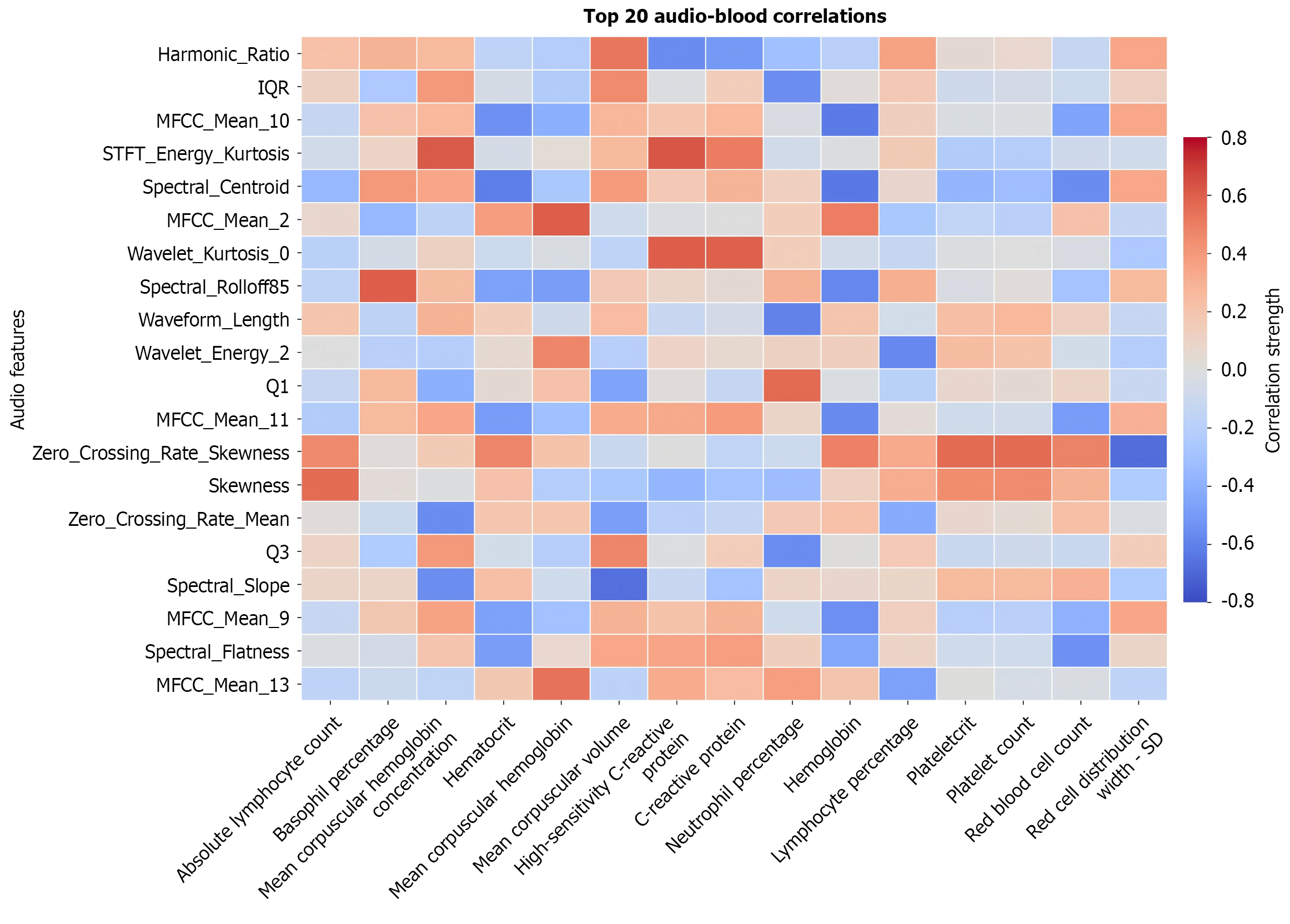
Correlations between the selected BS features and clinical biochemical indicators were investigated. Figure 8 presents the heat map of the correlation between BS features and clinical biochemical indicators in the correlation analysis. Spectral centroid showed significant negative correlations with RBC count r = -0.561, P = 0.007), hemoglobin r = -0.632, P = 0.002), and hematocrit (r = -0.611, P = 0.003). Similar negative correlations were observed for spectral bandwidth, spectral flatness, and MFCC-based metrics. The MFCC_mean_10 feature demonstrated more pronounced negative correlations with RBC count (r = -0.460, P = 0.03), hemoglobin (r = -0.631, P = 0.002), and hematocrit (r = -0.541, P = 0.01). In contrast, wavelet entropy exhibited modest positive correlations with these parameters (r = 0.362-0.528), reaching statistical significance for hemoglobin (P = 0.01) and hematocrit (P = 0.01). When analyzing inflammatory markers, morphological characteristics of BS segments, including kurtosis, kurtosis of wavelet energy, and impulse factor, showed positive correlations with both CRP and hs-CRP. Wavelet energy demonstrated the strongest correlation (CRP: r = 0.505, P = 0.02; hs-CRP: r = 0.630, P = 0.002). Conversely, harmonic ratio showed negative correlations with both CRP (r = -0.501, P = 0.02) and hs-CRP (r = -0.562, P = 0.007). The positive correlation pattern of wavelet entropy (r = 0.362-0.528) highlights the unique value of multiscale analysis. Unlike global spectral features, wavelet decomposition effectively captures the temporal complexity of BSs across different scales. The observed increase in wavelet entropy may reflect enhanced multiscale fluctuations in microcirculatory blood flow associated with elevated RBC parameters. This physiological association potentially originates from two mechanisms: Normal hematocrit levels support complex hemorheological properties that facilitate sophisticated vasomotion fluctuations, while optimal oxygen supply maintains the rhythmic electrical activity of intestinal interstitial cells of Cajal, generating BSs with distinctive fractal characteristics.
The diagnosis and detection of GIB currently rely on comprehensive assessments integrating clinical manifestations, imaging studies, and biochemical evaluations. This multifaceted approach necessitates prolonged and diverse testing procedures, often resulting in delayed clinical decision-making for gastric hemorrhage events[30]. The primary objective of this study was to investigate BSs as potential physiological biomarker for UGIB, establishing a noninvasive screening system to differentiate between UGIB patients and healthy individuals. We implemented a machine learning framework utilizing BS characteristics with four classification models: SVM, LR, KNN, and RF. This framework enabled the classification of BS segments from UGIB patients vs healthy controls. Complementary analysis examined correlations between BS features and laboratory biochemical indicators, providing substantial theoretical support for the diagnostic applicability of BS signatures. Although the audio recording equipment had limited technical specifications, it facilitated convenient and rapid acquisition of BS data from participants. This approach demonstrated potential for reducing patients’ exposure to repetitive and uncomfortable abdominal examinations while maintaining diagnostic efficacy.
We developed and evaluated machine learning models for UGIB detection using authentic BSs collected in clinical settings. Analysis of receiver operating characteristic curves across five feature subsets (20, 25, 30, 35, and 40 features) demonstrated that the SVM model exhibited minimal AUC fluctuations (0.811-0.879) within the 20- to 40-feature range, indicating robust adaptability to varying feature dimensions. This characteristic translates to strong generalization capability in clinical implementation. While the RF model showed moderate AUC variations, performance consistently remained within an acceptable range (0.771-0.790). The ensemble nature of RF enables it to provide supplementary diagnostic support across diverse data acquisition scenarios. In contrast, both KNN and LR models displayed significant AUC oscillations with changing feature numbers, suggesting high sensitivity to feature set size. Consequently, stringent quality control over feature acquisition is required to ensure diagnostic stability when implementing these models. Regarding sensitivity and specificity, SVM achieved sensitivity = 0.774 at 35 features, while maintaining specificity between 0.813 and 0.906, whereas KNN demonstrated substantial instability. This evidence indicates SVM’s consistent ability to accurately identify UGIB patients, while differentiating them from healthy individuals, demonstrating sig
Analysis of feature-biomarker correlations revealed that MFCC and spectral band energy features emerged as predominant model parameters. MFCC features primarily characterized the discrete processing stages of BSs, with our results indicating distinct acoustic signatures between UGIB and normal states. These differences manifest as amplified variance in MFCC features[31], likely attributable to increased time-varying properties of hemorrhage-associated acoustic signals. We hypothesize that intestinal bleeding alters motility patterns, generating BSs with abnormal frequency-specific characteristics. Significant differences in the 100-500 Hz frequency band between UGIB patients and healthy controls were identified, forming the primary classification basis for our SVM model. Mechanistically, when blood enters the intestinal lumen as an atypical content, it increases luminal viscosity. This elevated viscosity promotes turbulent flow during peristalsis, acoustically manifesting as abnormal signatures, a finding consistent with prior research. Concurrently, blood coagulation releases substantial gas volumes that destabilize existing intestinal bubbles, potentially increasing transient burst sounds within the 300-500 Hz frequency range[32]. These pathophysiological insights highlight the potential for developing predictive models targeting patients with cirrhosis-related portal hypertension, enabling pre-emptive interventions before overt bleeding occurs.
We used a multidimensional acoustic feature analysis approach encompassing spectral, Mel-frequency, and wavelet-domain metrics. This multimodal fusion strategy provides comprehensive information for evaluating the association between BSs and the hematological system. Different feature sets capture distinct physical properties and physiological significance of BSs from complementary perspectives. Variations in their correlations with blood parameters reflect both differential sensitivity across features and underlying physiological mechanisms operating at multiple levels.
Correlation analyses revealed that spectral centroid, as a first-order statistical measure quantifying spectral energy concentration, demonstrated a strong negative correlation with hemoglobin (r = -0.632). This indicates its capacity to effectively capture systemic hematological influences on intestinal motility. Reduced spectral centroid values typically signify energy redistribution from high to low frequencies, potentially attributable to: (1) Enhanced high-frequency absorption due to elevated hematocrit levels; and (2) Increased slow-wave peristalsis induced by heightened blood viscosity, which aligns with classification feature results. Conversely, wavelet entropy showed modest positive correlations (r = 0.362-0.528), highlighting the unique value of multiscale analysis. Unlike global spectral features, wavelet decomposition captures the temporal complexity of BSs across diverse scales. Elevated RBC parameters associate with richer multiscale fluctuations in microcirculatory blood flow, acoustically manifested as increased wavelet entropy. This relationship may originate from two mechanisms: (1) Physiological hemorheological properties at normal hematocrit levels support complex vasomotion fluctuations; and (2) Optimal oxygen delivery sustains rhythmic electrical activity in interstitial cells of Cajal, generating fractal BS patterns[33]. The robust correlations between BS features and hematological parameters establish a foundation for novel noninvasive monitoring applications, particularly in early detection of digestive disorders and hematological abnormalities. Whereas conventional hematological analysis requires invasive blood sampling, BS monitoring offers completely noninvasive and highly repeatable assessment, bridging a critical gap for clinical surveillance.
This study revealed a significant positive correlation between wavelet energy and inflammatory markers (CRP/hs-CRP), suggesting intestinal inflammation affects BSs through dual mechanisms: (1) Cytokine-mediated suppression of smooth muscle contractility (e.g., interleukin-6) altering sound frequency/amplitude; and (2) Mucosal edema/micro
Despite establishing predictive models for UGIB using BSs and identifying correlations between sound features and hematological biomarkers, several methodological limitations warrant consideration in clinical translation. These constraints simultaneously delineate critical research directions: Longitudinal studies are required to elucidate temporal relationships between acoustic feature evolution and biochemical indicators fluctuations. The modest sample size (n = 80) may overestimate model performance; external validation in larger cohorts is required. And multicenter trials are essential to validate model applicability and generalizability across diverse populations.
This study pioneers a noninvasive BS analysis tool for UGIB screening, leveraging multidimensional acoustic feature engineering and machine learning optimization. By integrating VMD denoising with time-frequency-spectral feature extraction, we developed a gradient-boosted SVM classifier that achieved robust diagnostic efficacy. Key BS biomarkers, particularly band energy and MFCC variance, were identified as sensitive indicators of hemorrhage-induced intestinal motility alterations, while correlation analyses validated their physiological linkage to hematological dynamics. This tool provides a rapid preliminary screening protocol for UGIB in prehospital/bedside settings, which will be embedded into tiered diagnostic workflow systems in future implementations to assist clinicians in prioritizing management for high-risk patients under resource-constrained clinical scenarios, with future translation dependent on multicenter validation and real-time monitoring implementation.
| 1. | Kamboj AK, Hoversten P, Leggett CL. Upper Gastrointestinal Bleeding: Etiologies and Management. Mayo Clin Proc. 2019;94:697-703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 104] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 2. | Fouad TR, Abdelsameea E, Abdel-Razek W, Attia A, Mohamed A, Metwally K, Naguib M, Waked I. Upper gastrointestinal bleeding in Egyptian patients with cirrhosis: Post-therapeutic outcome and prognostic indicators. J Gastroenterol Hepatol. 2019;34:1604-1610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 3. | Hearnshaw SA, Logan RF, Lowe D, Travis SP, Murphy MF, Palmer KR. Acute upper gastrointestinal bleeding in the UK: patient characteristics, diagnoses and outcomes in the 2007 UK audit. Gut. 2011;60:1327-1335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 510] [Cited by in RCA: 437] [Article Influence: 29.1] [Reference Citation Analysis (5)] |
| 4. | Laine L, Barkun AN, Saltzman JR, Martel M, Leontiadis GI. ACG Clinical Guideline: Upper Gastrointestinal and Ulcer Bleeding. Am J Gastroenterol. 2021;116:899-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 392] [Article Influence: 78.4] [Reference Citation Analysis (2)] |
| 5. | Du X, Allwood G, Webberley KM, Osseiran A, Marshall BJ. Bowel Sounds Identification and Migrating Motor Complex Detection with Low-Cost Piezoelectric Acoustic Sensing Device. Sensors (Basel). 2018;18:4240. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Goto J, Matsuda K, Harii N, Moriguchi T, Yanagisawa M, Sakata O. Usefulness of a real-time bowel sound analysis system in patients with severe sepsis (pilot study). J Artif Organs. 2015;18:86-91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 7. | Inderjeeth AJ, Webberley KM, Muir J, Marshall BJ. The potential of computerised analysis of bowel sounds for diagnosis of gastrointestinal conditions: a systematic review. Syst Rev. 2018;7:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 8. | Nowak JK, Nowak R, Radzikowski K, Grulkowski I, Walkowiak J. Automated Bowel Sound Analysis: An Overview. Sensors (Basel). 2021;21:5294. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 9. | Ficek J, Radzikowski K, Nowak JK, Yoshie O, Walkowiak J, Nowak R. Analysis of Gastrointestinal Acoustic Activity Using Deep Neural Networks. Sensors (Basel). 2021;21:7602. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 20] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 10. | Dimoulas C, Kalliris G, Papanikolaou G, Petridis V, Kalampakas A. Bowel-sound pattern analysis using wavelets and neural networks with application to long-term, unsupervised, gastrointestinal motility monitoring. Expert Syst Appl. 2008;34:26-41. [DOI] [Full Text] |
| 11. | Kumar TS, Soiland E, Stavdahl O, Fougner AL. Pilot Study of Early Meal Onset Detection from Abdominal Sounds. 2019 E-Health and Bioengineering Conference (EHB); 2019 Nov 21-23; Iasi, Romania. NJ, United States: IEEE, 2019: 1-4. [DOI] [Full Text] |
| 12. | Lin BS, Sheu MJ, Chuang CC, Tseng KC, Chen JY. Enhancing bowel sounds by using a higher order statistics-based radial basis function network. IEEE J Biomed Health Inform. 2013;17:675-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 13. | Ulusar UD. Recovery of gastrointestinal tract motility detection using Naive Bayesian and minimum statistics. Comput Biol Med. 2014;51:223-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Al Mamun KA, Habib MHU, Mcfarlane N, Paul N. A low power integrated bowel sound measurement system. 2015 IEEE International Instrumentation and Measurement Technology Conference (I2MTC) Proceedings; 2015 May 11-14; Pisa, Italy. NJ, United States: IEEE, 2015: 779-783. [DOI] [Full Text] |
| 15. | Mamun KAA, Mcfarlane N. Integrated real time bowel sound detector for artificial pancreas systems. Sens Biosensing Res. 2016;7:84-89. [RCA] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 7] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 16. | Yan XX, Zhang YL, Zhang YP, Yang YY, Wu D. Diagnostic Accuracy of Computerized Bowel Sound Analysis with Non-Invasive Devices for Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis. Chin Med Sci J. 2024;39:122-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 17. | Wang Y, Wang J, Liu W, Zhang G. [Establishment of comprehensive prediction model of acute gastrointestinal injury classification of critically ill patients]. Zhonghua Wei Chang Wai Ke Za Zhi. 2018;21:325-330. [PubMed] |
| 18. | Suppakitjanusant P, Kasemkosin N, Sivapiromrat AK, Weinstein S, Ongphiphadhanakul B, Hunt WR, Sueblinvong V, Tangpricha V. Predicting glycemic control status and high blood glucose levels through voice characteristic analysis in patients with cystic fibrosis-related diabetes (CFRD). Sci Rep. 2023;13:8617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 19. | Maji U, Pal S. Empirical mode decomposition vs. variational mode decomposition on ECG signal processing: A comparative study. 2016 International Conference on Advances in Computing, Communications and Informatics (ICACCI); 2016 Sep 21-24; Jaipur, India. NJ, United States: IEEE, 2016: 1129-1134. [DOI] [Full Text] |
| 20. | Mohanty S, Gupta KK, Raju KS. Comparative study between VMD and EMD in bearing fault diagnosis. 2014 9th International Conference on Industrial and Information Systems (ICIIS); 2014 Feb 15-17; Gwalior, India. NJ, United States: IEEE, 2014: 1-6. [DOI] [Full Text] |
| 21. | Martis RJ, Acharya UR, Tan JH, Petznick A, Yanti R, Chua CK, Ng EY, Tong L. Application of empirical mode decomposition (emd) for automated detection of epilepsy using EEG signals. Int J Neural Syst. 2012;22:1250027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 108] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 22. | Sujadevi VG, Mohan N, Sachin Kumar S, Akshay S, Soman KP. A hybrid method for fundamental heart sound segmentation using group-sparsity denoising and variational mode decomposition. Biomed Eng Lett. 2019;9:413-424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 23. | Kölle K, Aftab MF, Andersson LE, Fougner AL, Stavdahl Ø. Data driven filtering of bowel sounds using multivariate empirical mode decomposition. Biomed Eng Online. 2019;18:28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 11] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 24. | Tao W, Wang G, Sun Z, Xiao S, Wu Q, Zhang M. Recognition Method for Broiler Sound Signals Based on Multi-Domain Sound Features and Classification Model. Sensors (Basel). 2022;22:7935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 25. | Sato R, Emoto T, Gojima Y, Akutagawa M. Automatic Bowel Motility Evaluation Technique for Noncontact Sound Recordings. Appl Sci. 2018;8:999. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 27. | Harris CR, Millman KJ, van der Walt SJ, Gommers R, Virtanen P, Cournapeau D, Wieser E, Taylor J, Berg S, Smith NJ, Kern R, Picus M, Hoyer S, van Kerkwijk MH, Brett M, Haldane A, Del Río JF, Wiebe M, Peterson P, Gérard-Marchant P, Sheppard K, Reddy T, Weckesser W, Abbasi H, Gohlke C, Oliphant TE. Array programming with NumPy. Nature. 2020;585:357-362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21438] [Cited by in RCA: 6020] [Article Influence: 1003.3] [Reference Citation Analysis (4)] |
| 28. | Mcfee B, Raffel C, Liang D, Ellis D, Mcvicar M, Battenberg E, Nieto O. librosa: Audio and Music Signal Analysis in Python. SciPy, 2015. [DOI] [Full Text] |
| 29. | Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, Blondel M, Prettenhofer P, Weiss R, Dubourg V, Vanderplas J, Passos A, Cournapeau D, Brucher M, Perrot M, Duchesnay E. Scikit-learn: Machine Learning in Python. J Mach Learn Res. 2011;12:2825-2830. |
| 30. | Tokar JL, Higa JT. Acute Gastrointestinal Bleeding. Ann Intern Med. 2022;175:ITC17-ITC32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 31. | Hafiz NF, Mashohor S, Shazril MHSEMA, Rasid MFA, Ali A. Comparison of Mel Frequency Cepstral Coefficient (MFCC) and Mel Spectrogram Techniques to Classify Industrial Machine Sound. 2023 15th International Conference on Software, Knowledge, Information Management and Applications (SKIMA); 2023 Dec 8-10; Kuala Lumpur, Malaysia. NJ, United States: IEEE, 2023: 273-278. [DOI] [Full Text] |
| 32. | Wang G, Chen Y, Liu H, Yu X, Han Y, Wang W, Kang H. Differences in intestinal motility during different sleep stages based on long-term bowel sounds. Biomed Eng Online. 2023;22:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 33. | Felder S, Margel D, Murrell Z, Fleshner P. Usefulness of bowel sound auscultation: a prospective evaluation. J Surg Educ. 2014;71:768-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |