Published online Apr 7, 2026. doi: 10.3748/wjg.v32.i13.115758
Revised: November 19, 2025
Accepted: January 21, 2026
Published online: April 7, 2026
Processing time: 154 Days and 3.4 Hours
Traditional hemorrhoidectomy is often associated with problems such as long operation time, excessive bleeding, and slow recovery. As an emerging surgical method, biological welding technology integrates tissue fusion, cutting, and he
To evaluate the safety, efficacy, and underlying mechanism of biological welding technology for hemorrhoidectomy in an animal mod.
Twenty adult healthy New Zealand rabbits were randomly allocated to two groups: The biological welding group (hemorrhoidectomy performed with bio
The bio-welding group demonstrated a significantly shorter operation time (44.9 ± 17.2 seconds vs 222.2 ± 43.9 seconds, P < 0.001) and achieved zero intraoperative blood loss (0 g vs 18.75 ± 5.83 g, P < 0.001) compared to the control group. Despite a relatively high local temperature (59.85 ± 5.48 °C), only mild thermal damage occurred. Wound healing was better on post-operative days 7 and 14, with a significantly shorter average recovery time (12.83 ± 1.35 days vs 16.0 ± 1.91 days, P = 0.029). Mechanistic studies revealed that the technology promotes spatial reconstruction of collagen fibers and reduces postoperative inflammation, thereby accelerating orderly tissue repair.
Biological welding technology can safely and efficiently perform hemorrhoidectomy, significantly shorten the ope
Core Tip: This study demonstrates that biological welding technology, which integrates tissue fusion, cutting, and hemostasis into a single procedure, offers a rapid and bloodless approach to hemorrhoidectomy. In a rabbit model, it significantly reduced operative time by approximately 80%, achieved zero intraoperative blood loss, and accelerated postoperative recovery compared to traditional excision and suturing. Mechanistically, it promotes organized tissue repair by remodeling collagen fibers and reducing inflammation. These findings highlight its potential as a safe, efficient, and innovative surgical technique for clinical application in the treatment of hemorrhoids.
- Citation: Chen Y, Zhang QR, Guo MG, Sun ZY, Zeng FC. Biological welding: A rapid and bloodless approach to hemorrhoidectomy. World J Gastroenterol 2026; 32(13): 115758
- URL: https://www.wjgnet.com/1007-9327/full/v32/i13/115758.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i13.115758
Hemorrhoids, swollen and inflamed blood vessels in the lower rectum and anus, are among the most common and widespread conditions globally[1,2]. It is estimated that up to 44% of the general population may suffer from hemorr
The treatment for hemorrhoids varies and is tailored to the severity of the condition. According to clinical guidelines, treatment options include pharmacological therapy, non-pharmacological interventions, outpatient surgery, and surgical intervention[6]. Medications, particularly topical treatments, help relieve symptoms, reduce inflammation, and alleviate discomfort. Non-pharmacological interventions such as dietary changes, increased fluid intake, and sitz baths can improve symptoms and reduce the frequency of flare-ups[7]. Grade I and II hemorrhoids are usually managed with medication, lifestyle modifications, and dietary adjustments, whereas grade III and IV hemorrhoids require surgical intervention[1,2]. Hemorrhoidectomy is the most common and safest procedure for hemorrhoid removal, typically performed using scissors, electrocautery devices, vascular sealing instruments, or harmonic scalpels[1,2]. However, traditional hemorrhoidectomy is associated with significant postoperative pain, prolonged recovery periods, and high complication rates, especially in elderly or pregnant patients, where surgical risks are heightened. Therefore, there is a pressing need to develop minimally invasive, fast-recovery techniques.
Biological welding technology, also known as high-frequency welding, high-frequency electro-field welding, electro-surgical welding, and bipolar electro-surgical surgery, is an emerging surgical technique that has gained widespread attention in recent years. Unlike high-frequency electrosurgery, biological welding combines tissue electro-sectioning, electrocoagulation, and welding (tissue fusion) into one integrated procedure. By precisely adjusting high-frequency current, electrical energy is converted into thermal energy, increasing tissue temperature in an orderly and controlled manner. The application of pressure with welding forceps results in strong, seamless tissue anastomosis[8,9]. Compared to traditional surgical techniques, the multifunctional nature of biological welding simplifies the surgical process, enhances efficiency, and minimizes tissue damage while achieving effective hemostasis and tissue fusion[10,11]. This technique eliminates the need for implants, vascular clamps, or sutures, significantly reducing surgical time and bleeding, with welding temperatures kept below 60 °C[12]. Additionally, the biological welding device is easy to operate, and the use of bipolar forceps or tweezers to hold tissues allows for rapid cutting, hemostasis, or welding without changing instruments.
Clinically, biological welding has shown notable success in fields such as ophthalmology, gastrointestinal surgery, and oncology. In ophthalmology, high-frequency electro-field welding has significantly improved retinal adhesion strength, reduced postoperative inflammation, and demonstrated no significant complications in follow-up studies[13]. In gastrointestinal surgery, biological welding has been applied in treating esophageal atresia, with patients recovering faster compared to those undergoing laser welding[14]. In skull base tumor surgery, biological welding has effectively sealed dural defects, reducing postoperative cerebrospinal fluid leaks and improving surgical safety[15]. These studies collectively demonstrate the substantial benefits of biological welding in various clinical settings, enhancing surgical outcomes and patient recovery.
Given its significant advantages in various surgical procedures, including precise hemostasis, accelerated healing, and reduced infection risk, biological welding technology has attracted considerable attention in the medical community. However, its application in hemorrhoidectomy remains unexplored. This study aims to evaluate the safety, efficacy, and advantages of biological welding technology in hemorrhoidectomy using an animal model (New Zealand rabbit anal tissue). We hope to establish a solid scientific foundation for the clinical application of biological welding in hemorr
The animals used in this study (adult healthy New Zealand rabbits) were sourced from the Guangdong Medical Laboratory Animal Center (Shenzhen, Guangdong Province, China). The study adhered to laboratory animal care and usage guidelines and was approved by the Institutional Animal Care and Use Committee of Shenzhen University Medical School (No. IACUC-202400075). A total of 20 adult healthy New Zealand rabbits were randomly divided into two groups: The biological welding group (undergoing hemorrhoidectomy with biological welding technology) and the control group (undergoing traditional hemorrhoidectomy with excision and suturing). All rabbits were housed under identical breeding standards and environmental conditions.
The equipment used in this study was a high-frequency biological welding device developed in-house by our team[11].
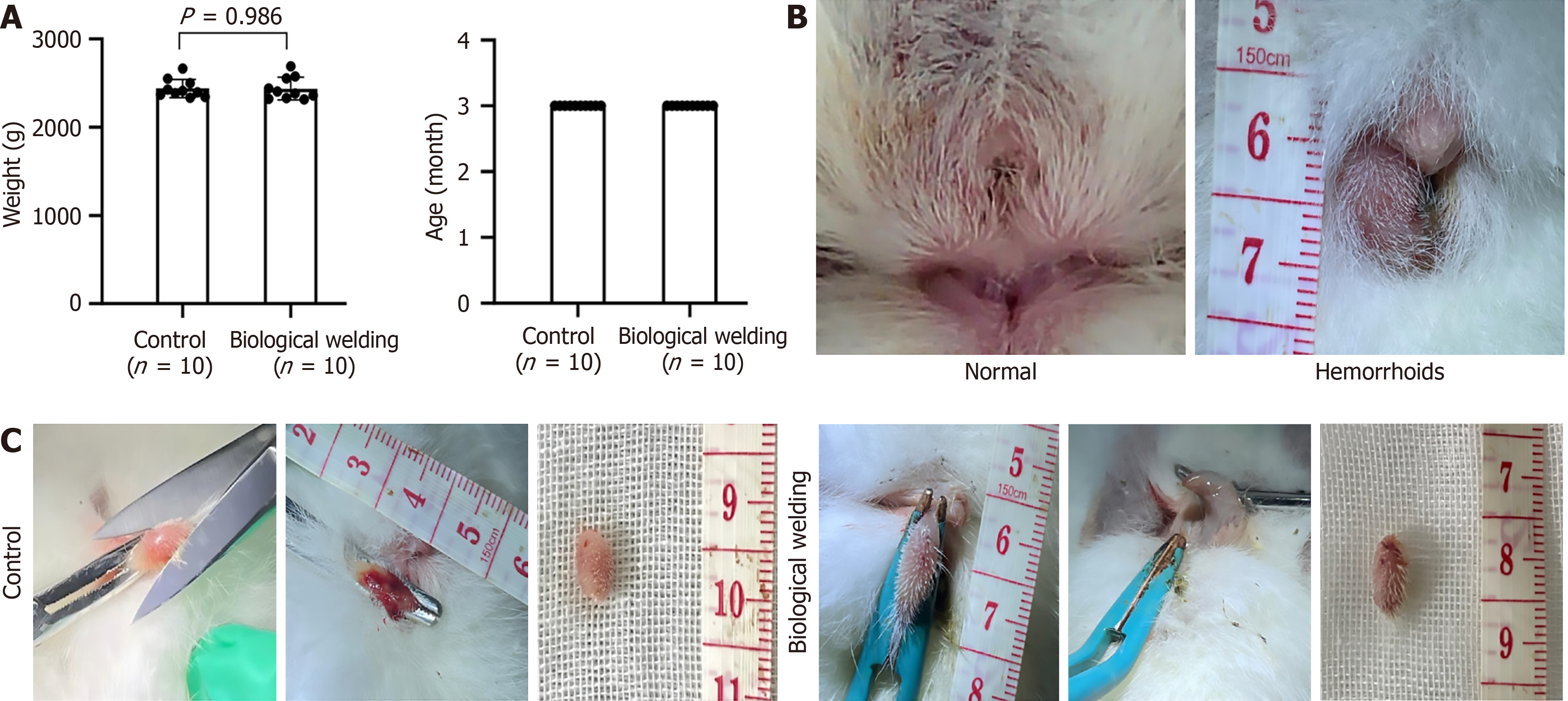
We adopted an improved standardized protocol to establish a hemorrhoid model in order to simulate acute external hemorrhoids combined with perianal inflammation[16-18]. Specifically, three days before the surgery, a mixture of castor oil and acetic acid [with the optimized ratio of 2% castor oil + 1% acetic acid (solvent: 95% ethanol, volume ratio 1:1 mixture)] was applied topically to the perianal skin of New Zealand rabbits. This chemical stimulation causes local acute inflammation, manifested as a soft, red, swollen mass in the anal region (Figure 1), which is similar to the early ap
Surgical time was defined as the total time required for complete hemorrhoidal excision and wound closure. Intraoperative blood loss was recorded using the dry gauze absorption method. The temperature of the surgical site was continuously monitored with an infrared thermometer, and the highest recorded temperature was considered the surgical temperature.
Tissue specimens were fixed in formalin buffer, embedded, and sectioned, followed by hematoxylin and eosin (HE) staining and Sirius red staining. Tissue degradation and pathological changes were assessed by analyzing the slides. For electron microscopy, tissue specimens were fixed in glutaraldehyde, dehydrated using graded ethanol organic solvents, embedded, sectioned, and stained with lead citrate. Ultrastructural degradation and pathological changes were evaluated by analyzing the slides.
The fused region of hemorrhoids was analyzed using a Raman spectrometer (NTEGRA Spectra-II). The central area of the hemorrhoidal tissue sample was excised and rinsed with deionized water to remove adherent damaged adipose tissue (appearing as reddish-brown residue at this stage). Raman spectra were collected from the central region, with multiple frequency points acquired per sample and averaged to reduce signal noise. The acquired Raman spectral data were first edited and smoothed using Origin 2024 software. Subsequently, the baseline was removed via polynomial fitting, and the Raman spectral range was confined to 900-2000 cm-1. Finally, Gaussian-Lorentzian functions were employed to calibrate characteristic peaks, from which information such as peak intensity was extracted.
Statistical data are expressed as mean ± SD. Data analysis was performed using GraphPad Prism 9.0 software. Differences between two independent samples were assessed using a two-tailed t-test. For normally distributed data with homo
This study included 20 adult male healthy New Zealand rabbits, which were randomly divided into the biological welding group and the control group. To ensure the accuracy of the experiment, we compared the age and weight of the two groups of animals. Statistical analysis revealed no significant differences between the groups (Figure 1), indicating that the rabbits were comparable at baseline.
Hemorrhoid models were established using croton oil and glacial acetic acid, resulting in a soft, red, swollen mass in the anal region (Figure 1). After anesthesia and disinfection, the control group underwent traditional excision and suturing for hemorrhoidectomy, while the biological welding group underwent hemorrhoidectomy using a biological welding technique, which integrates electrocautery, electrocoagulation, and welding.
Postoperative observations showed that the control group had more prominent surgical incisions, with visible suture marks in a linear arrangement and some tissue distortion around the incision due to suture tension. The edges of the incision were swollen and hemorrhagic. In contrast, the biological welding group’s surgical site appeared dry with no abnormal leakage or bleeding. The incision edges were well-aligned, and the surrounding skin had a normal color, with an overall appearance superior to that of the control group (Figure 2A). Both groups had a 100% success rate for hemo
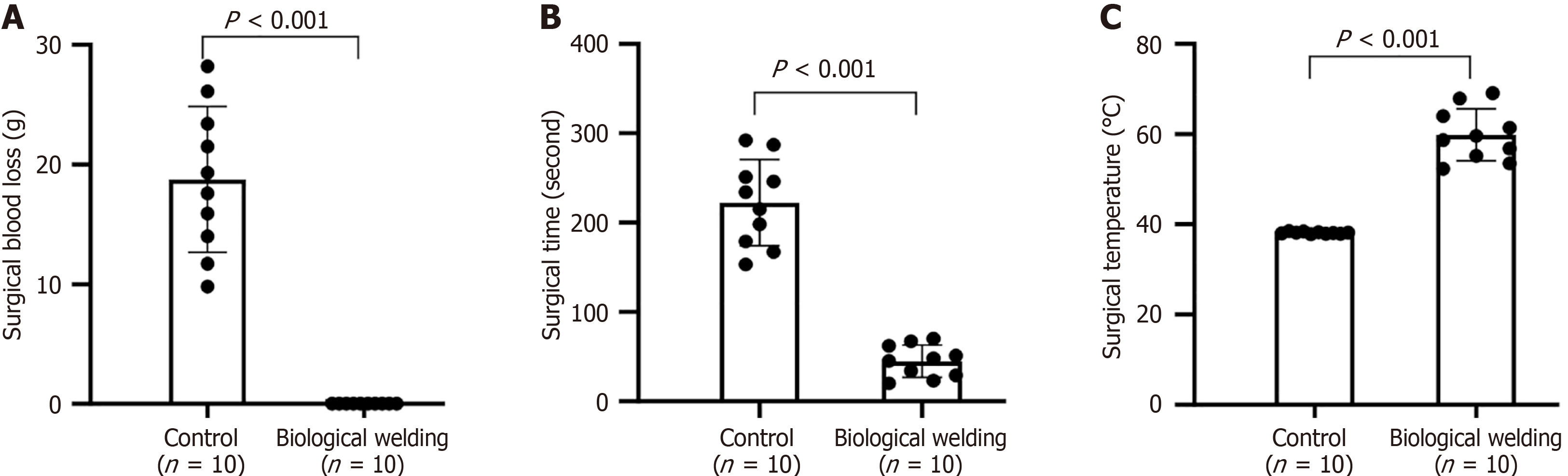
Surgical safety is a critical indicator of surgical efficacy, particularly in terms of controlling surgical time and managing intraoperative blood loss. Regarding surgical time, the biological welding group had a significantly shorter procedure time of 44.9 ± 17.2 seconds, compared to the control group’s 222.2 ± 43.9 seconds (P < 0.001) (Figure 2A). In terms of blood loss, the biological welding group exhibited no blood loss (0 g) during hemorrhoidectomy, significantly lower than the control group’s 18.75 ± 5.83 g (P < 0.001) (Figure 2B). In intraoperative temperature monitoring, the control group’s temperature was 38.23 ± 0.24 °C, while the biological welding group’s temperature was 59.85 ± 5.48 °C, significantly higher than the control group (P < 0.001) (Figure 2C). Further examination using HE staining revealed that the control group exhibited mild mechanical damage, while the primary pathological changes in the biological welding group were mild thermal and pressure injuries, including protein denaturation, coagulative necrosis, and hyaline degeneration (Figure 3A). These findings demonstrate that biological welding technology significantly outperforms traditional methods in reducing both surgical time and blood loss during hemorrhoidectomy.
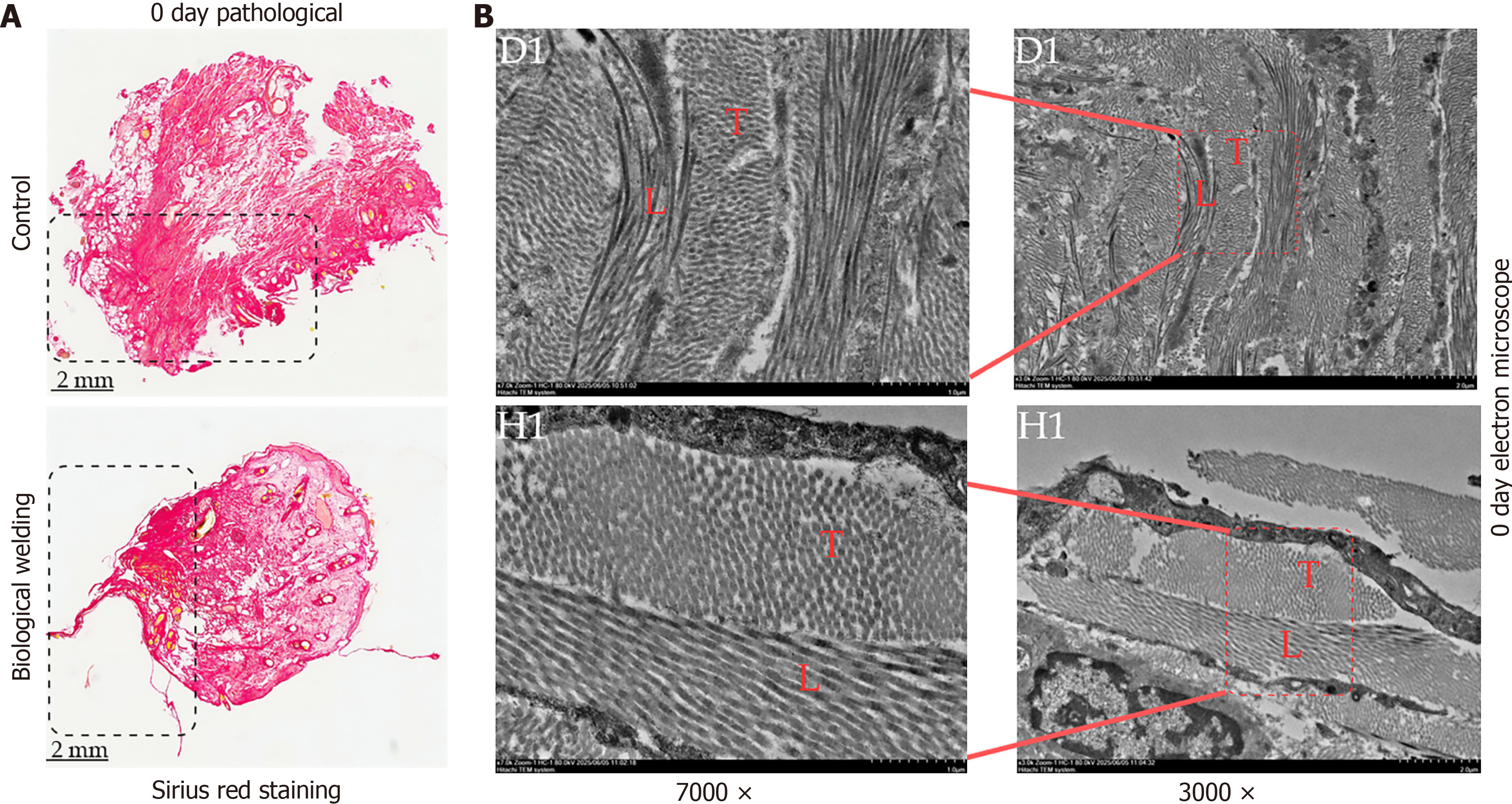
Biological welding technology achieves tissue welding through the synergistic effects of high-frequency current and mechanical pressure. Since collagen is the primary component of the tissue matrix, we employed Sirius red staining to observe the changes in the microstructure of hemorrhoidal tissue following welding.
The results showed that the control group retained its natural collagen fiber structure, which appeared as regularly arranged, well-defined collagen fibers (Figure 3B). In contrast, in the biological welding group, the collagen fibers at the welding site fused together, and the spatial structure was remodeled, showing cross-linking and tight fusion with slight thermal and mechanical damage. This observation indicates that biological welding is not simply a process of tissue cutting but involves inducing structural remodeling of the collagen fibers to achieve tissue closure.
Furthermore, this study analyzed the ultrastructural changes of the hemorrhoidal tissue after excision. Transmission electron microscopy revealed that in the control group, both transverse and longitudinal sections of the collagen fibers showed no significant changes, with collagen fibers arranged in an orderly fashion within the tissue matrix. In contrast, the biological welding group exhibited an increase in the gaps between collagen fibers in the transverse section, with larger diameters and a more uniform, round or near-round shape. The boundaries of collagen fibers in both transverse and longitudinal sections were blurred, and their arrangement was disordered. These results suggest that biological welding technology mediates the remodeling of collagen fiber structures at the ultrastructural level, facilitating hemorrhoid excision through collagen fiber reorganization.
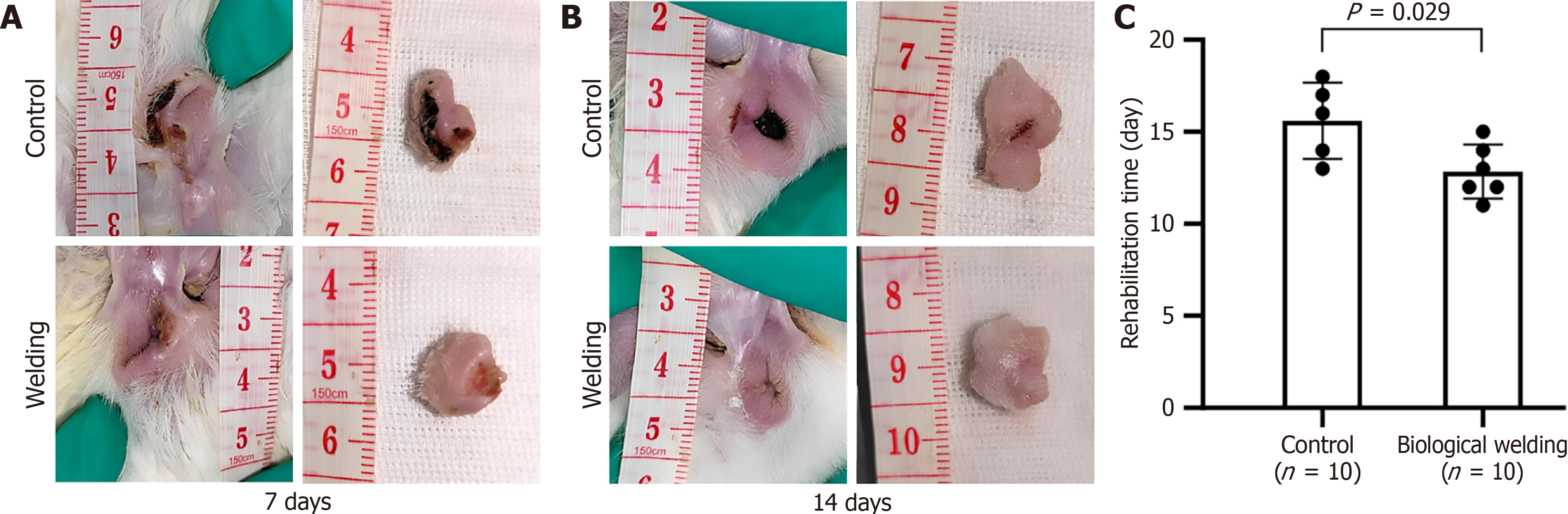
Biological welding technology can significantly enhance postoperative recovery. In this study, observations of the two groups of rabbits revealed normal daily activities and excretory functions in both groups. At postoperative days 7 and 14, we assessed the recovery status of the two groups, which underwent different surgical methods. On postoperative day 7, a critical period for inflammation and tissue proliferation, the two groups exhibited distinct differences in wound healing (Figure 4A). In the control group, the wound exhibited sustained and pronounced inflammation, characterized by significant erythema, edema, and intact scabbing. In contrast, the biological welding group demonstrated a marked reduction in inflammatory response, with a faster healing initiation process, characterized by reduced redness and swelling, scab detachment, and visible formation of new epithelial tissue (Figure 4A).
This healing advantage became more pronounced by postoperative day 14 (Figure 4B). At this time, the control group’s wounds were still in the later stages of healing, with some scab still adhered (Figure 4B), indicating ongoing healing. In contrast, the biological welding group’s wounds were nearly fully healed, with scabs almost completely detached, and the wound surface was smooth and restored to a near-normal tissue appearance (Figure 4B). To quantify the difference in healing rates, we defined clinical healing as complete epithelialization of the wound, with no significant erythema or scabbing. Based on this criterion, statistical analysis revealed that the control group’s average recovery time was 16.0 ± 1.91 days, while the biological welding group’s average full recovery time was 12.83 ± 1.35 days, with this difference being statistically significant (P = 0.029) (Figure 4C). These results indicate that biological welding technology sig
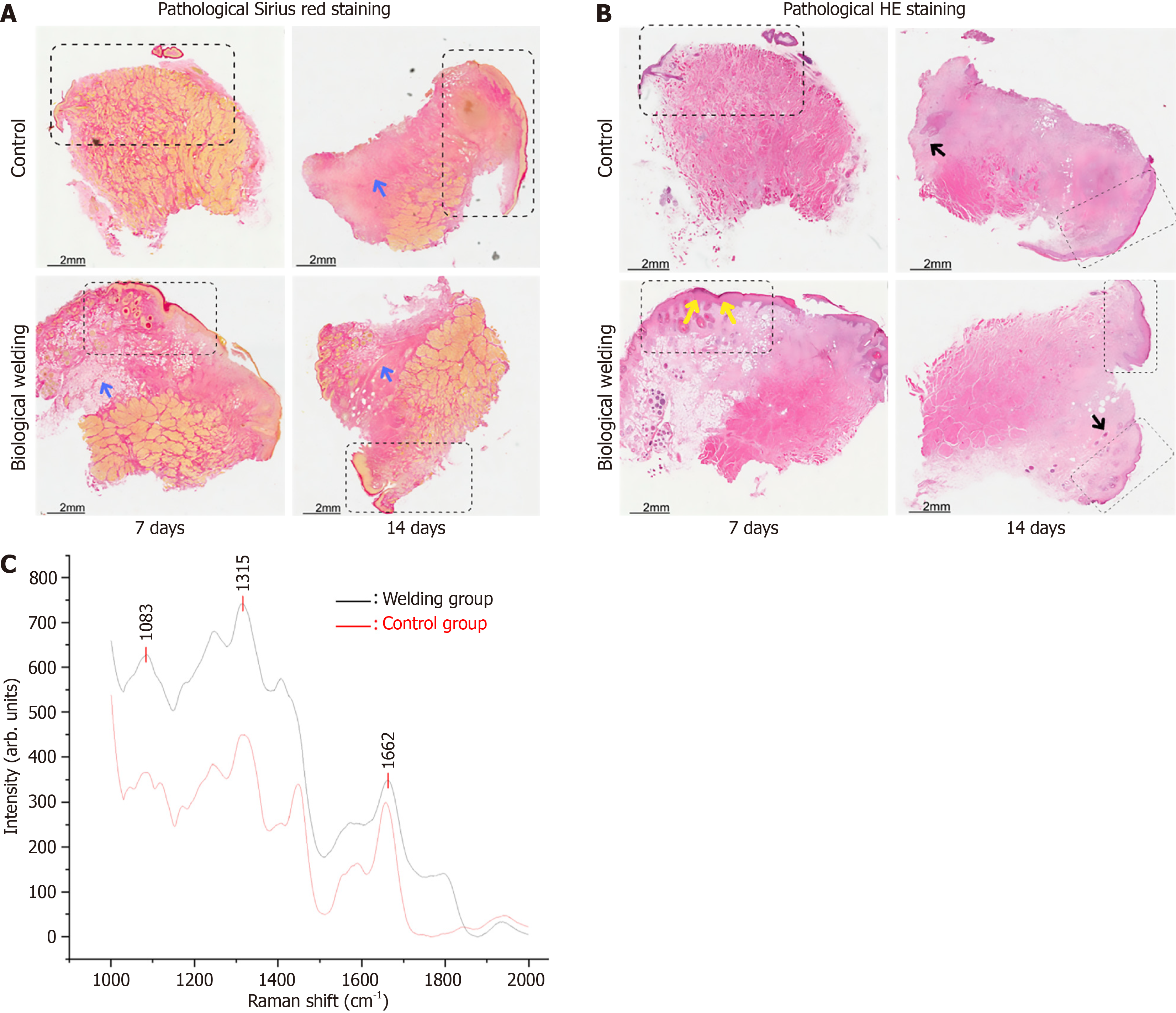
This study employed HE staining and Sirius red staining to perform histological evaluations of hemorrhoids in both the control and biological welding groups at postoperative days 7 and 14.
At postoperative day 7, HE staining analysis revealed significant inflammatory reactions in the control group, characterized by abundant infiltration of inflammatory cells and tissue edema (Figure 5), indicating severe chronic inflammation induced by the scissor incision. Additionally, fibrous tissue formation was observed. In contrast, the hemorrhoidal tissue in the biological welding group exhibited partial recovery at the welding site, with a relatively mild inflammatory response (Figure 5). Inflammatory cell infiltration was less pronounced, tissue edema was minimal, and fibrous tissue formation was more prominent, suggesting that the welding procedure had a less detrimental long-term effect on the tissue, with faster recovery of the inflammatory response. By postoperative day 14, the inflammatory response in the control group had mostly subsided, and fibrous tissue had matured further; however, some areas remained incompletely repaired. In contrast, the biological welding group showed near-complete resolution of the inflammatory response, with more mature fibrous tissue and a tissue structure approaching normal, demonstrating superior repair outcomes. These findings indicate that biological welding technology effectively alleviates the postoperative inflammatory response of hemorrhoidectomy and promotes tissue repair.
Sirius red staining analysis revealed significant differences in the dynamic changes of collagen fibers and tissue structure between the two groups (control and biological welding) at different time points (7 days and 14 days). From 7 days to 14 days post-operation, the collagen fibers in the control group were relatively disordered and not closely connected. In contrast, the biological welding group presented a collagen structure similar to the normal one, with the collagen fibers in the tissue matrix arranged regularly, in a dense and orderly state without any breakage (Figure 5). These results indicate that the biological welding process mediates the remodeling of the collagen structure in the tissue matrix, and the postoperative recovery restores the matrix structure to an ordered state.
Raman imaging of fused hemorrhoidal tissue was performed on selected regions within the welded area at 7 days post-welding (Figure 5, 7- welding). Collagen terminal end-group spectra in each map included all features previously reported in Raman studies of collagen and collagen-rich tissues. Specifically, the Raman spectra corresponding to the C-O stretching amide I band (1600-1690 cm-1) and the C-N stretching amide III band (1200-1360 cm-1) are noteworthy, as the vibrations of amide I and amide III are most sensitive to conformational changes in collagen secondary structure[8,19]. The amide I band at 1662 cm-1 in the hemorrhoidal tissue after bio-welding treatment indicates that bio-welding may promote the formation of β-sheet structures during the repair process by affecting the secondary structure of proteins. The clear amide III band at 1246 cm-1 further confirms the integrity of the structure and the abundant presence of α-helices. These structural changes were clearly demonstrated by the significant enhancement of the Raman signal intensities of the amide I band (approximately 1662 cm-1) and the amide III band (approximately 1315 cm-1). These results indicate that after undergoing hemorrhoidectomy using the biological welding technique, the spatial structure of collagen molecules in the hemorrhoidal tissue has changed.
An ideal operation for hemorrhoids should remove internal and external component of hemorrhoids completely, have minimal postoperative pain and complication, demonstrate less recurrence, and are easy to learn and perform. The procedure could be cheap and cost-effective too. Unfortunately, none of the currently available operation achieves all the ideal conditions. So far, excisional hemorrhoidectomy is the mainstay operation for grade III-IV hemorrhoids and complicated hemorrhoids. Of note, closed (Ferguson) hemorrhoidectomy and open (Milligan-Morgan) hemorrhoi
Biological welding technology, as an innovative approach in the field of surgery, has demonstrated clear advantages in reducing surgical time, minimizing intraoperative bleeding, and accelerating postoperative recovery in multiple surgical procedures[10,14,24]. This study marks the first application of biological welding technology in an animal model for the treatment of hemorrhoidal disease. The study results indicate that this surgical method demonstrates good safety and efficacy for the treatment of hemorrhoids in New Zealand rabbits. The study data validate international experience, showing comparable surgical completion rates, significantly reduced surgical time, decreased intraoperative bleeding, and earlier return to normal daily activities and bowel function postoperatively.
However, the study has certain limitations that warrant acknowledgment. Firstly, regarding the animal model: While it replicates key acute features of hemorrhoidal disease, it may not fully capture chronic vascular changes. Thus, the findings should be interpreted primarily as proof-of-concept for the technology’s efficacy, and future validation using chronic hemorrhoid models is recommended. Secondly, the relatively small sample size (10 rabbits per group) may restrict the statistical power and generalizability of the results. Future animal studies should employ larger cohorts and adopt a randomized controlled trial design to further confirm the safety and efficacy of biological welding technology in hemorrhoidectomy.
Looking ahead, our team plans to launch a pilot clinical study enrolling 30 patients with grade III-IV hemorrhoids, aiming to compare outcomes between biological welding and traditional excision surgery. Furthermore, the interdisciplinary nature of this research ensures technical rigor, we will explore its application in urological procedures, including orchiectomy, vasectomy, and partial nephrectomy, to evaluate its versatility in minimizing bleeding and optimizing tissue repair across surgical specialties.
The duration of surgery directly affects the amount of anesthetic used during the procedure, which in turn significantly impacts patient safety[25]. Among various hemorrhoidectomy procedures, surgery duration varies, primarily depending on their respective surgical techniques and technical principles. Traditional closed (Ferguson) hemorrhoidectomy and open (Milligan-Morgan) hemorrhoidectomy have a long history in hemorrhoidectomy, both being equally effective and safe. However, the Ferguson method outperforms the Milligan-Morgan method in terms of long-term patient satisfaction and incontinence[20,22]. However, the Ferguson procedure involves suturing, which prolongs the surgical time. The average surgical duration for the traditional Milligan-Morgan procedure is 24 minutes, while the average surgical duration for the Ferguson procedure is 30 minutes[20,22,26,27]. The novel laser hemorrhoidoplasty technique further reduces the average surgical time to 16 minutes[28]. Bio-welding technology is an advanced surgical technique that integrates electrocautery, electrocoagulation, and welding functions, simplifying surgical procedures while significantly reducing surgical time. In this study, the average time for hemorrhoidectomy using bio-welding technology was only 20.22% of the traditional cutting and suturing method, a reduction of approximately 79.78%, totaling only 44.9 ± 17.2 seconds. This demonstrates that bio-welding technology has a significant advantage in reducing surgical time, providing a new option for improving surgical efficiency and patient safety.
Intraoperative bleeding is a common challenge in hemorrhoidectomy and must be strictly controlled. Bleeding is a critical factor that affects the surgical field and patient safety, and excessive bleeding can lead to severe complications or even death[2,29,30]. Therefore, blood loss has become an important indicator in evaluating the safety of surgical techniques. Traditional open surgery techniques, such as the Milligan-Morgan method, typically result in relatively high blood loss, with one clinical study reporting an average blood loss of 70.34 mL[31]. In contrast, advanced energy-based devices have significantly reduced intraoperative bleeding. For example, the LigaSure bipolar coagulation technique has been reported to reduce the average blood loss to 11.5 mL[32].
In the current study, using traditional excision and suturing techniques for hemorrhoidectomy in New Zealand rabbits resulted in an average blood loss of approximately 18.75 ± 5.83 mL. However, the application of biological welding technology reduced the blood loss to 0 mL. This is due to the fact that, in addition to cutting the tissue, the technology utilizes the thermal effect for electrocoagulation and hemostasis, significantly reducing intraoperative bleeding. These results demonstrate that biological welding technology has a clear advantage in minimizing blood loss during surgery, providing a safer and more effective option to enhance surgical efficiency and patient safety.
Thermal injury is a common issue in electrosurgical procedures[33]. In this study, biological welding technology significantly reduced the extent of thermal damage, precisely controlling it within 2 millimeters. During the procedure, the average temperature remained stable at 59.85 °C, which is notably lower than the operating temperature of traditional high-frequency electrosurgical knives. This reduced temperature cutting technique minimizes the range and severity of thermal damage to tissues, promoting faster wound healing and potentially shortening the postoperative recovery period.
Histological analysis of the excised tissue revealed significant structural changes, which may be attributed to the synergistic effects of electrical current and mechanical force applied by the bipolar forceps during hemorrhoidectomy. The voltage between the two poles of the bipolar forceps induces current flow through soft tissue, causing spatial structural remodeling of tissue proteins under specific pressure and temperature conditions. This process enables rapid tissue cutting, hemostasis, and anastomosis.
Recovery time is a key indicator for evaluating surgical outcomes[34]. Postoperative recovery time is an essential metric in assessing hemorrhoidectomy efficacy, including wound healing time, duration of pain, and time to resume normal activities. In comparison, traditional hemorrhoidectomy, due to its larger wound size, has the longest recovery time. Patients typically require 15.3 ± 5.6 days to resume normal activities, and the average hospital stay is 3.2 days[35]. In contrast, studies comparing the recovery time of procedure for prolapse and hemorrhoids (PPH) technology have shown that recovery time is significantly reduced, with patients returning to normal activities 7.6 ± 3.1 days earlier[35]. A 2007 meta-analysis also demonstrated that while LigaSure hemorrhoidectomy significantly reduced surgical time and blood loss, it did not show advantages over traditional surgery in terms of postoperative pain, hospital stay, or time to return to work or normal activities, highlighting the need to consider the additional equipment cost[36].
In this study, the use of biological welding technology in hemorrhoidectomy in New Zealand rabbits resulted in a 19.82% reduction in recovery time compared to traditional excision and suturing techniques. This improvement may stem from the bloodless nature of the procedure and the absence of sutures, which prevents potential rejection reactions. Additionally, histological and electron microscopy analysis revealed that biological welding technology, through the synergistic effects of thermal energy and pressure, remodeled the naturally loose collagen fiber network into a dense, fused structure with stronger mechanical properties. This process promotes a more ordered arrangement and healing of collagen fibers, alleviates postoperative inflammation, and ultimately leads to faster tissue repair. This technology demonstrates a clear advantage in recovery time, offering patients a quicker recovery option.
The occurrence of complications is a key indicator of surgical safety. Comprehensive analyses of different hemo
In this study, biological welding technology demonstrated significant safety advantages, with no common complications such as intraoperative bleeding or fecal incontinence. These results indicate that biological welding technology offers a clear advantage in reducing complications associated with hemorrhoidectomy, providing a safer surgical option for patients.
This investigation validates bio-welding as a safe and effective modality for hemorrhoidectomy. Our findings, based on a comprehensive evaluation including animal experiments, histology, and optical analysis, demonstrate that bio-welding significantly outperforms traditional techniques. Key advantages include shortened operative duration, decreased intraoperative blood loss, and accelerated postoperative recovery. The procedural simplicity and the elimination of foreign body implantation further underscore its clinical superiority. Consequently, bio-welding technology emerges as a promising advancement in hemorrhoidal surgery, offering substantial benefits for both patient safety and healthcare system efficiency.
The authors gratefully acknowledge the Guangdong Medical Laboratory Animal Center for providing the experimental animals and for their professional support in animal husbandry. We are also indebted to the Animal Ethics Committee of Shenzhen University for their rigorous review and approval of this study protocol. The technical assistance provided by colleagues from the Guangdong Key Laboratory for Biomedical Measurements and Ultrasound Imaging, and the National-Regional Key Technology Engineering Laboratory for Medical Ultrasound was instrumental in the development of the high-frequency biological welding device used in this research. Special thanks are due to the clinical teams from Shenzhen University General Hospital for their valuable insights regarding clinical applications and surgical procedures. We acknowledge all contributors who participated in data collection, statistical analysis, and manuscript preparation, particularly recognizing the joint efforts of the first authors Chen Y, Zhang QR. Finally, we thank the reviewers and editors for their constructive comments that helped improve the quality of this manuscript.
| 1. | van Tol RR, Kleijnen J, Watson AJM, Jongen J, Altomare DF, Qvist N, Higuero T, Muris JWM, Breukink SO. European Society of ColoProctology: guideline for haemorrhoidal disease. Colorectal Dis. 2020;22:650-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 116] [Article Influence: 19.3] [Reference Citation Analysis (1)] |
| 2. | Hawkins AT, Davis BR, Bhama AR, Fang SH, Dawes AJ, Feingold DL, Lightner AL, Paquette IM; Clinical Practice Guidelines Committee of the American Society of Colon and Rectal Surgeons. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Hemorrhoids. Dis Colon Rectum. 2024;67:614-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 48] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 3. | Riss S, Weiser FA, Schwameis K, Riss T, Mittlböck M, Steiner G, Stift A. The prevalence of hemorrhoids in adults. Int J Colorectal Dis. 2012;27:215-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 353] [Cited by in RCA: 260] [Article Influence: 18.6] [Reference Citation Analysis (2)] |
| 4. | Grossi U. Expert Commentary on Diagnosis and Management of Anal Stenosis. Dis Colon Rectum. 2025;68:12-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 5. | Wald A, Bharucha AE, Limketkai B, Malcolm A, Remes-Troche JM, Whitehead WE, Zutshi M. ACG Clinical Guidelines: Management of Benign Anorectal Disorders. Am J Gastroenterol. 2021;116:1987-2008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 102] [Article Influence: 20.4] [Reference Citation Analysis (1)] |
| 6. | Davis BR, Lee-Kong SA, Migaly J, Feingold DL, Steele SR. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Hemorrhoids. Dis Colon Rectum. 2018;61:284-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 276] [Cited by in RCA: 186] [Article Influence: 23.3] [Reference Citation Analysis (1)] |
| 7. | Gallo G, Martellucci J, Sturiale A, Clerico G, Milito G, Marino F, Cocorullo G, Giordano P, Mistrangelo M, Trompetto M. Consensus statement of the Italian society of colorectal surgery (SICCR): management and treatment of hemorrhoidal disease. Tech Coloproctol. 2020;24:145-164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 211] [Cited by in RCA: 171] [Article Influence: 28.5] [Reference Citation Analysis (2)] |
| 8. | Zhu C, Yin L, Xu J, Yang X, Wang H, Xiang X, Liu H, Liu K. Characteristics of Collagen Changes in Small Intestine Anastomoses Induced by High-Frequency Electric Field Welding. Biomolecules. 2022;12:1683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (1)] |
| 9. | Zhu C, Na Y, Yan Z, Cheng X, Xie P, Tao X, Chen L, Zhao H, Qiu J, Gu X, Xiang J, Liu K. In-vivo tissue healing mechanism at the intestinal anastomosis site following high-frequency electric welding. Int J Surg. 2025;111:1614-1618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 10. | Tryliskyy Y, Kebkalo A, Wong CS, Rublenko S, Rublenko M, Ilnytskiy M, Negria I, Hordovskiy V. Safety analysis of endoscopic haemostasis using a high-frequency live tissue electric welding device - EKVZ300 PATONMED. Prz Gastroenterol. 2018;13:234-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 11. | Zeng F, Cao Q, Fu F, Wang B, Sun Z. Biological welding: a rapid and bloodless approach to circumcision. World J Urol. 2024;43:25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 12. | Wang H, Yang X, Madeniyeti N, Qiu J, Zhu C, Yin L, Liu K. Temperature Distribution of Vessel Tissue by High Frequency Electric Welding with Combination Optical Measure and Simulation. Biosensors (Basel). 2022;12:209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 13. | Umanets N, Pasyechnikova NV, Naumenko VA, Henrich PB. High-frequency electric welding: a novel method for improved immediate chorioretinal adhesion in vitreoretinal surgery. Graefes Arch Clin Exp Ophthalmol. 2014;252:1697-1703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 14. | Tyselskyi V, Poylin V, Kebkalo A. Biological welding - novel technique in the treatment of esophageal metaplasia. Pol Przegl Chir. 2020;92:1-5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 15. | Zabolotnyi D, Kvasha O. Bipolar high-frequency electric welding in radical resection of frontal sinus tumors. Carcinogenesis. 2023;44:175-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 16. | Ke M, Huang S, Lin H, Xu Z, Li X, Li Z, Chen F, Wu H. Establishment and study of a rat internal haemorrhoid model. Sci Rep. 2023;13:21385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (3)] |
| 17. | Koohi-Hosseinabadi O, Koohpeyma F, Safarpour AR, Nematollahy P, Kazemi M, Shahriarirad R, Tanideh R, Mojahedtaghi M, Ghaemmaghami P, Iraji A, Goudarzi K, Tanideh N. Antioxidant and anti-inflammatory effects of Equisetum arvense L. on acid-induced ulcerative colitis in rats. Sci Rep. 2025;15:13727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 18. | Pekacar S, Özüpek B, Akkol EK, Taştan H, Ersan H, Orhan DD. Identification of bioactive components on antihemorrhoidal activity of Cistus laurifolius L. using RP-HPLC and LC-QTOF-MS. J Ethnopharmacol. 2024;319:117122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 19. | Becker L, Lu CE, Montes-Mojarro IA, Layland SL, Khalil S, Nsair A, Duffy GP, Fend F, Marzi J, Schenke-Layland K. Raman microspectroscopy identifies fibrotic tissues in collagen-related disorders via deconvoluted collagen type I spectra. Acta Biomater. 2023;162:278-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (1)] |
| 20. | Ho YH, Buettner PG. Open compared with closed haemorrhoidectomy: meta-analysis of randomized controlled trials. Tech Coloproctol. 2007;11:135-143. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (5)] |
| 21. | Arbman G, Krook H, Haapaniemi S. Closed vs. open hemorrhoidectomy--is there any difference? Dis Colon Rectum. 2000;43:31-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 84] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 22. | Jóhannsson HO, Påhlman L, Graf W. Randomized clinical trial of the effects on anal function of Milligan-Morgan versus Ferguson haemorrhoidectomy. Br J Surg. 2006;93:1208-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (5)] |
| 23. | Yeo D, Tan KY. Hemorrhoidectomy - making sense of the surgical options. World J Gastroenterol. 2014;20:16976-16983. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 60] [Cited by in RCA: 39] [Article Influence: 3.3] [Reference Citation Analysis (4)] |
| 24. | Zhu C, Na Y, Cheng X, Tao X, Xie P, Chen L, Zhao H, Qiu J, Gu X, Xiang J, Liu K. Assessing the influence of parameters on tissue welding in small bowel end-to-end anastomosis in vitro and in vivo. Surg Endosc. 2024;38:3126-3137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 25. | Cheng H, Clymer JW, Po-Han Chen B, Sadeghirad B, Ferko NC, Cameron CG, Hinoul P. Prolonged operative duration is associated with complications: a systematic review and meta-analysis. J Surg Res. 2018;229:134-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 747] [Cited by in RCA: 651] [Article Influence: 81.4] [Reference Citation Analysis (2)] |
| 26. | Bhatti MI, Sajid MS, Baig MK. Milligan-Morgan (Open) Versus Ferguson Haemorrhoidectomy (Closed): A Systematic Review and Meta-Analysis of Published Randomized, Controlled Trials. World J Surg. 2016;40:1509-1519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (2)] |
| 27. | Cheng PL, Chen CC, Chen JS, Wei PL, Huang YJ. Diode laser hemorrhoidoplasty versus conventional Milligan-Morgan and Ferguson hemorrhoidectomy for symptomatic hemorrhoids: Meta-analysis. Asian J Surg. 2024;47:4681-4690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 9] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 28. | Durgun C, Yiğit E. Laser Hemorrhoidoplasty Versus Ligasure Hemorrhoidectomy: A Comparative Analysis. Cureus. 2023;15:e43119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 29. | Lavikainen LI, Guyatt GH, Luomaranta AL, Cartwright R, Kalliala IEJ, Couban RJ, Aaltonen RL, Aro KM, Cárdenas JL, Devereaux PJ, Galambosi PJ, Ge FZ, Halme ALE, Haukka J, Izett-Kay ML, Joronen KM, Karjalainen PK, Khamani N, Oksjoki SM, Pourjamal N; ROTBIGGS Investigators, Singh T, Tähtinen RM, Vernooij RWM, Tikkinen KAO. Risk of thrombosis and bleeding in gynecologic cancer surgery: systematic review and meta-analysis. Am J Obstet Gynecol. 2024;230:403-416. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 30. | Parks AL, Fang MC. Periprocedural Anticoagulation. Ann Intern Med. 2023;176:ITC49-ITC64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 31. | Bakhtiar N, Moosa FA, Jaleel F, Qureshi NA, Jawaid M. Comparison of hemorrhoidectomy by LigaSure with conventional Milligan Morgan's hemorrhoidectomy. Pak J Med Sci. 2016;32:657-661. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 32. | Khanna R, Khanna S, Bhadani S, Singh S, Khanna AK. Comparison of Ligasure Hemorrhoidectomy with Conventional Ferguson's Hemorrhoidectomy. Indian J Surg. 2010;72:294-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 33. | Sano MB, Fesmire CC, Petrella RA. Electro-Thermal Therapy Algorithms and Active Internal Electrode Cooling Reduce Thermal Injury in High Frequency Pulsed Electric Field Cancer Therapies. Ann Biomed Eng. 2021;49:191-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (1)] |
| 34. | Paavola M, Kanto K, Ranstam J, Malmivaara A, Inkinen J, Kalske J, Savolainen V, Sinisaari I, Taimela S, Järvinen TL; Finnish Shoulder Impingement Arthroscopy Controlled Trial (FIMPACT) Investigators. Subacromial decompression versus diagnostic arthroscopy for shoulder impingement: a 5-year follow-up of a randomised, placebo surgery controlled clinical trial. Br J Sports Med. 2021;55:99-107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (1)] |
| 35. | Zhang C, Zhang W, Xu J. Comparison of the outcomes of hemorrhoidectomy and PPH in the treatment of grades III and IV hemorrhoids. Medicine (Baltimore). 2022;101:e29100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 36. | Tan EK, Cornish J, Darzi AW, Papagrigoriadis S, Tekkis PP. Meta-analysis of short-term outcomes of randomized controlled trials of LigaSure vs conventional hemorrhoidectomy. Arch Surg. 2007;142:1209-18; discussion 1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (2)] |
| 37. | Tavani ME, Partovi Y, Poursaki T, Gharibi F. The Complications of Hemorrhoidectomy From Patients' Perspective: A Qualitative Study. Health Sci Rep. 2025;8:e70724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 38. | Eberspacher C, Mascagni P, Di Nardo D, Pironi D, Pontone S, Martellucci J, Naldini G, Mascagni D. Caiman Versus LigaSure Hemorrhoidectomy: Postoperative Pain, Early Complications, Long-Term Follow-up, and Costs. Surg Innov. 2020;27:272-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 39. | Sayfan J, Becker A, Koltun L. Sutureless closed hemorrhoidectomy: a new technique. Ann Surg. 2001;234:21-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (2)] |