Copyright: ©Author(s) 2026.
World J Gastroenterol. Apr 14, 2026; 32(14): 114331
Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.114331
Published online Apr 14, 2026. doi: 10.3748/wjg.v32.i14.114331
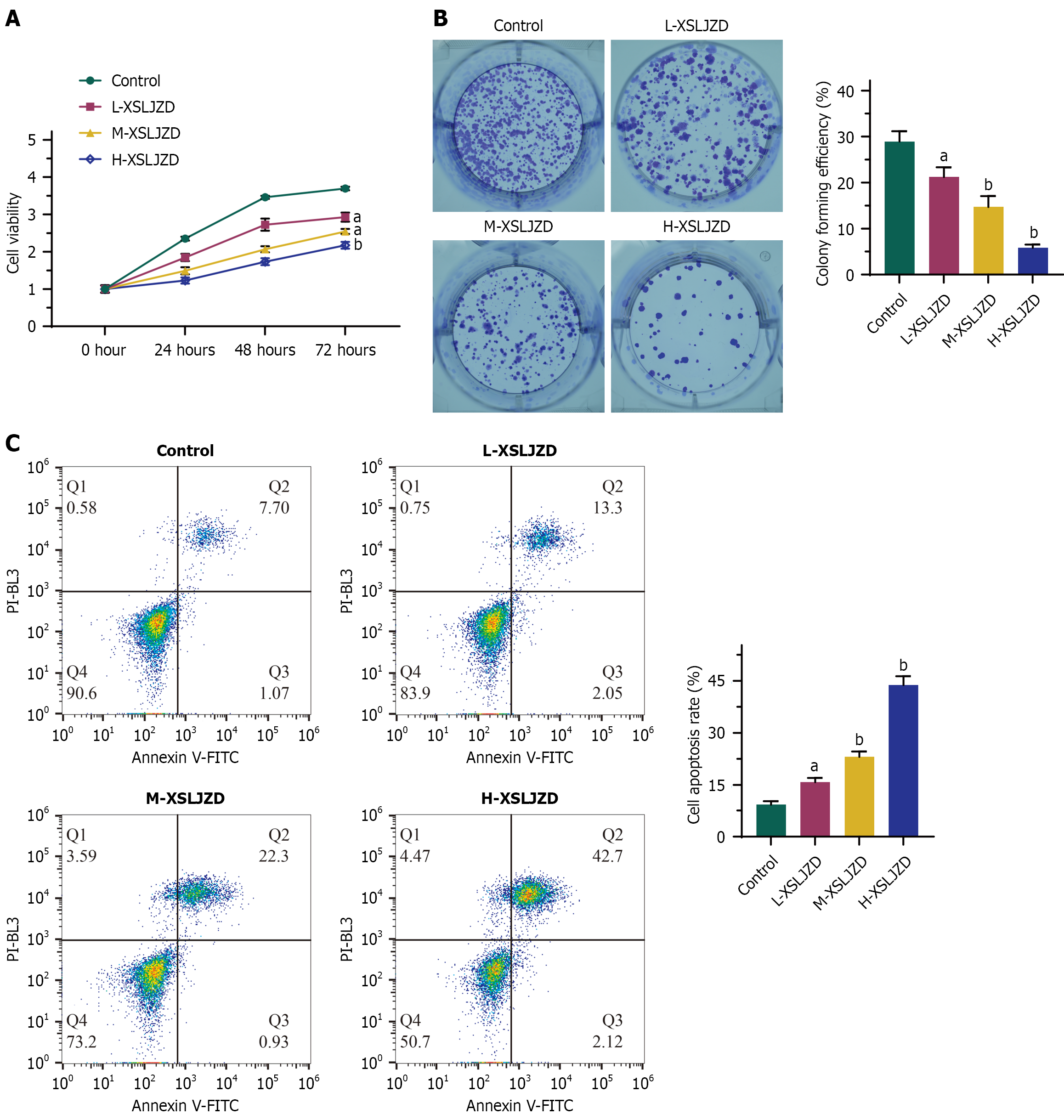
Figure 1 Xiangshaliujunzi decoction suppressed cell viability, cell proliferation and promoted apoptosis in a dose-dependent manner.
A: Cell viability detected by CCK8 assays; B: Cell proliferation detected by colony formation assays; C: Cell apoptosis detected by flow cytometry. L-Xiangshaliujunzi decoction (XSLJZD): 1.55 g/(kg·day); M-XSLJZD: 3.1 g/(kg·day); H-XSLJZD: 6.2 g/(kg·day). The vertical axis represents optical density at 450 nm from CCK-8 assays (A), normalized to the control at 0 hour (set as 1). aP < 0.05, bP < 0.01 vs control. One-way ANOVA with Tukey's post-hoc test was used. XSLJZD: Xiangshaliujunzi decoction.
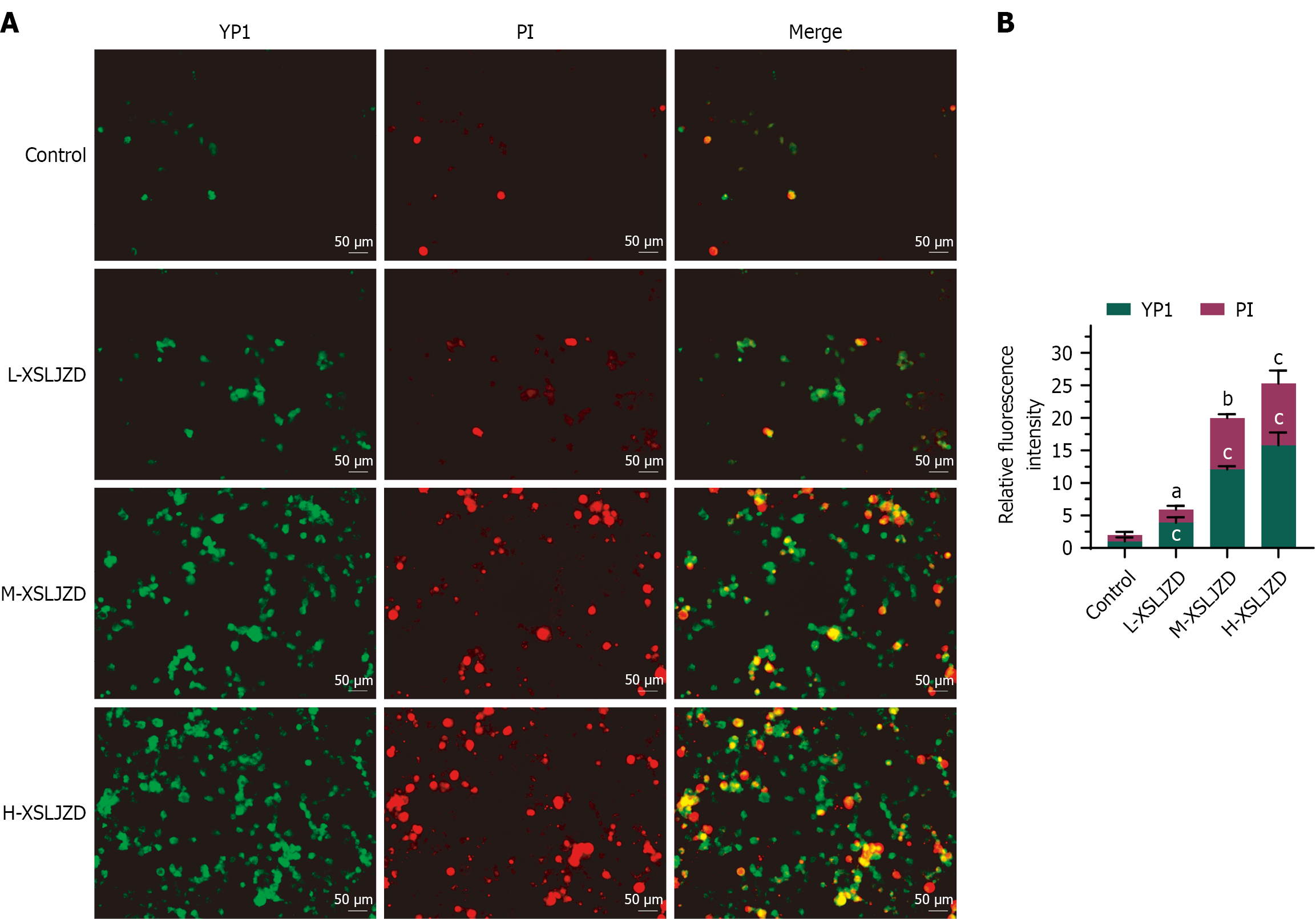
Figure 2 Xiangshaliujunzi decoction induced apoptosis, necroptosis and pyroptosis in a dose-dependent manner.
A: Apoptotic and necrotic cells were identified by green fluorescence (YO-PRO-1 positive), while necrotic and pyroptotic cells were identified by red fluorescence (PI positive); B: Bar chart of relative fluorescence intensity. The fluorescence intensity of the control group in the first replicate was set as the reference (value = 1), and intensities of the second and third replicates, as well as all Xiangshaliujunzi decoction (XSLJZD)-treated groups, were normalized to this reference. L-XSLJZD: 1.55 g/(kg·day); M-XSLJZD: 3.1 g/(kg·day); H-XSLJZD: 6.2 g/(kg·day). aP < 0.05, bP < 0.01, cP < 0.001 vs control, determined by one-way ANOVA with Tukey's post-hoc test. XSLJZD: Xiangshaliujunzi decoction.
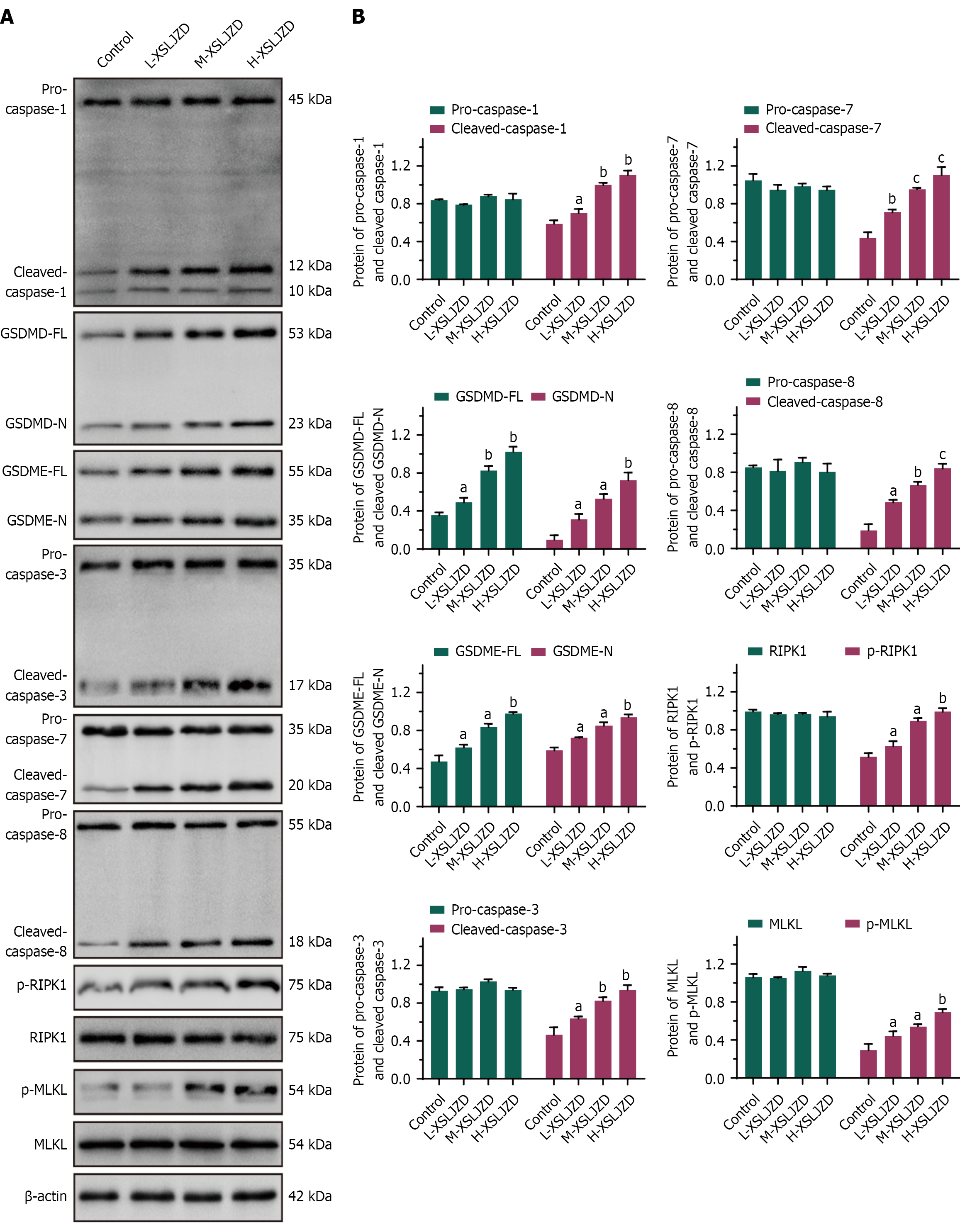
Figure 3 Xiangshaliujunzi decoction promoted the expression of PANoptosis-related proteins.
In BGC-823/5-fluorouracil cells treated with Xiangshaliujunzi decoction (XSLJZD), the expression levels of pyroptosis-related proteins (cleaved caspase-1, GSDMD-FL, GSDMD-N, GSDME-FL, GSDME-N), apoptosis-related proteins (cleaved caspase-3, cleaved caspase-7, and cleaved caspase-8), and programmed cell death-related proteins (p-RIPK1, p-MLKL) increased in a dose-dependent manner. A: Western blot bands of PANoptosis proteins; B: Bar chart of relative protein expression levels. L-XSLJZD: 1.55 g/(kg·day); M-XSLJZD: 3.1 g/(kg·day); H-XSLJZD: 6.2 g/(kg·day). aP < 0.05, bP < 0.01, cP < 0.001 vs control. XSLJZD: Xiangshaliujunzi decoction.
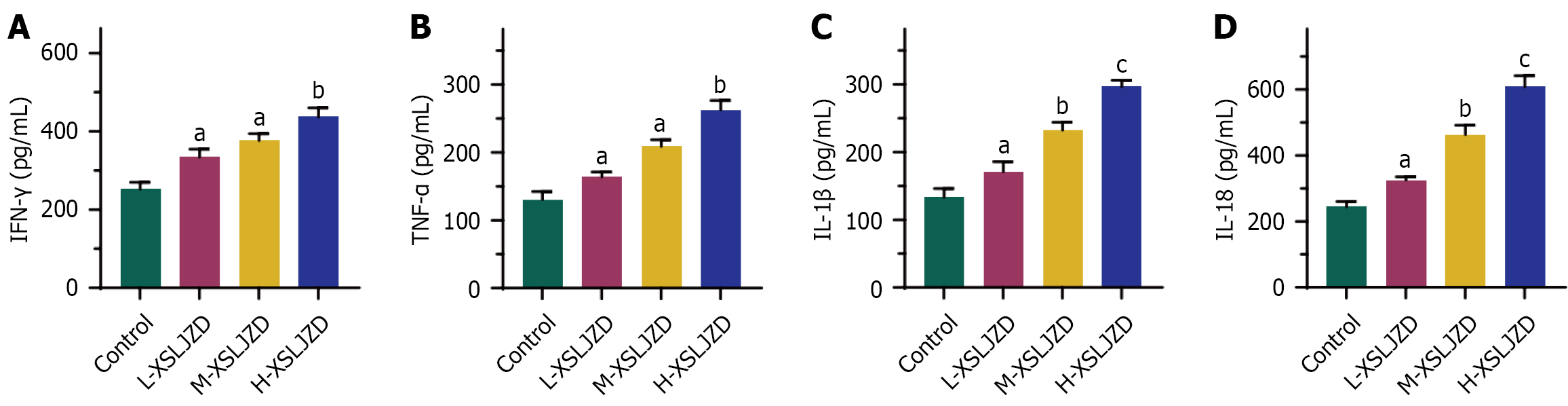
Figure 4 Interferon-γ, tumor necrosis factor-α, interleukin-1β, and interleukin-18 secreted were increased in correlation with the concentration of Xiangshaliujunzi decoction in BGC-823/5-fluorouracil cells treated with Xiangshaliujunzi decoction.
A: Interferon-γ; B: Tumor necrosis factor-α; C: Interleukin (IL)-1β; D: IL-18. L-Xiangshaliujunzi decoction (XSLJZD): 1.55 g/(kg·day); M-XSLJZD: 3.1 g/(kg·day); H- XSLJZD: 6.2 g/(kg·day). aP < 0.05, bP < 0.01, cP < 0.001 vs control. XSLJZD: Xiangshaliujunzi decoction; IFN: Interferon; TNF: Tumor necrosis factor; IL: Interleukin.
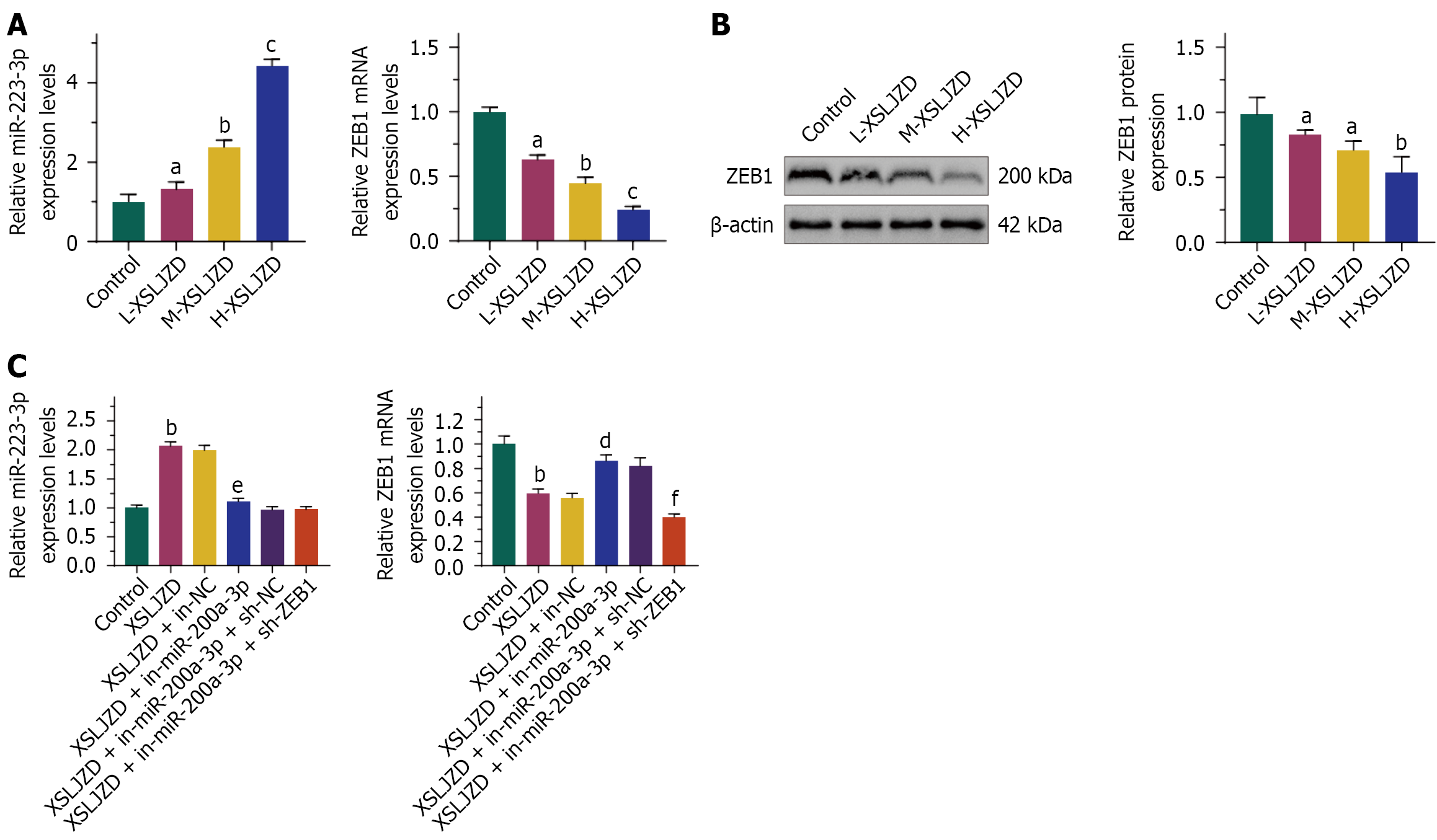
Figure 5 Xiangshaliujunzi decoction inhibited ZEB1 expression by up-regulation of miR-200a-3p.
MiR-200a-3p expression was increased in the Xiangshaliujunzi decoction (XSLJZD) group, while ZEB1 expression was significantly decreased. The effect of XSLJZD on miR-200a-3p and ZEB1 expression was dose-dependent. Inhibiting miR-200a-3p expression in the XSLJZD group partially restored ZEB1 expression. A: MiR-200a-3p and ZEB1 mRNA expression; B: ZEB1 protein expression in different treatment groups; C: MiR-200a-3p and ZEB1 mRNA expression in XSLJZD-treated BGC-823/5-fluorouracil cells transfected with miR-200a-3p or ZEB1 knock down. L-XSLJZD: 1.55 g/(kg·day); M-XSLJZD: 3.1 g/(kg·day); H- XSLJZD: 6.2 g/(kg·day). aP < 0.05, bP < 0.01, cP < 0.001 vs control. dP < 0.05, eP < 0.01 vs XSLJZD + in-negative control (NC). fP < 0.01 vs XSLJZD + sh-NC. XSLJZD: Xiangshaliujunzi decoction; NC: Negative control.
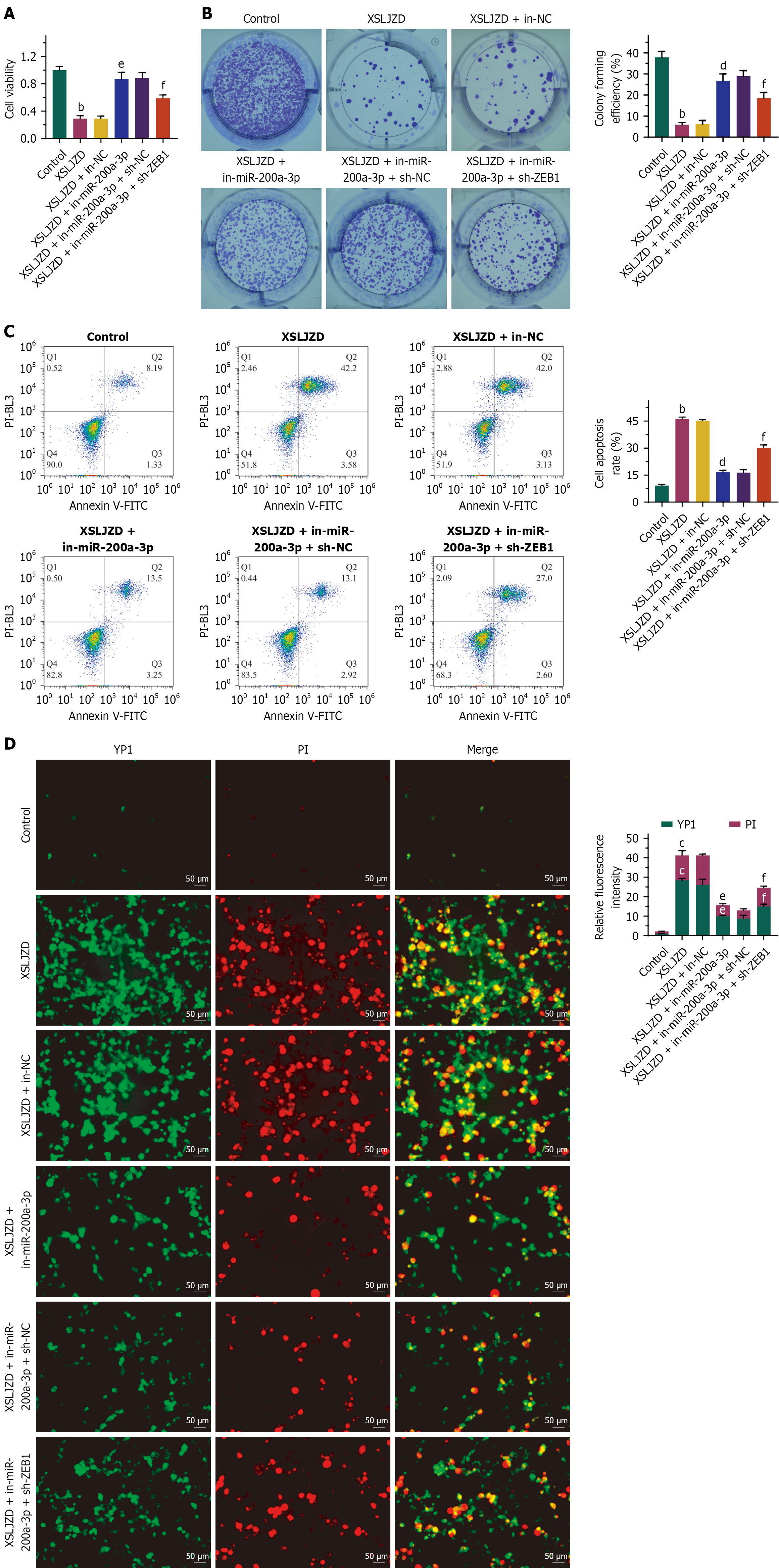
Figure 6 The effect of Xiangshaliujunzi decoction on cell functions was mediated by the miR-200a-3p/ZEB1 axis.
Cell viability and proliferation were significantly decreased, while apoptosis was significantly increased in the Xiangshaliujunzi decoction (XSLJZD)-treated group. Cell apoptosis/necroptosis (YP1 positive) and necroptosis/pyroptosis (PI positive) were also elevated in the XSLJZD-treated group. Transfection of miR-200a-3p inhibitors into BGC-823/5-fluorouracil (5-FU) cells partially reversed the effects of XSLJZD. Knocking down ZEB1 expression in BGC-823/5-FU cells transfected with miR-200a-3p inhibitors partially restored the effects of XSLJZD. A: Cell viability detected by CCK8 assays; B: Cell proliferation detected by colony formation assays; C: Cell apoptosis detected by flow cytometry; D: Cell PANoptosis detected by YO-PRO-1/PI staining. bP < 0.01, cP < 0.001 vs control. dP < 0.05, eP < 0.01 vs XSLJZD + in-negative control (NC). fP < 0.05 vs XSLJZD + sh-NC. XSLJZD: Xiangshaliujunzi decoction; NC: Negative control.
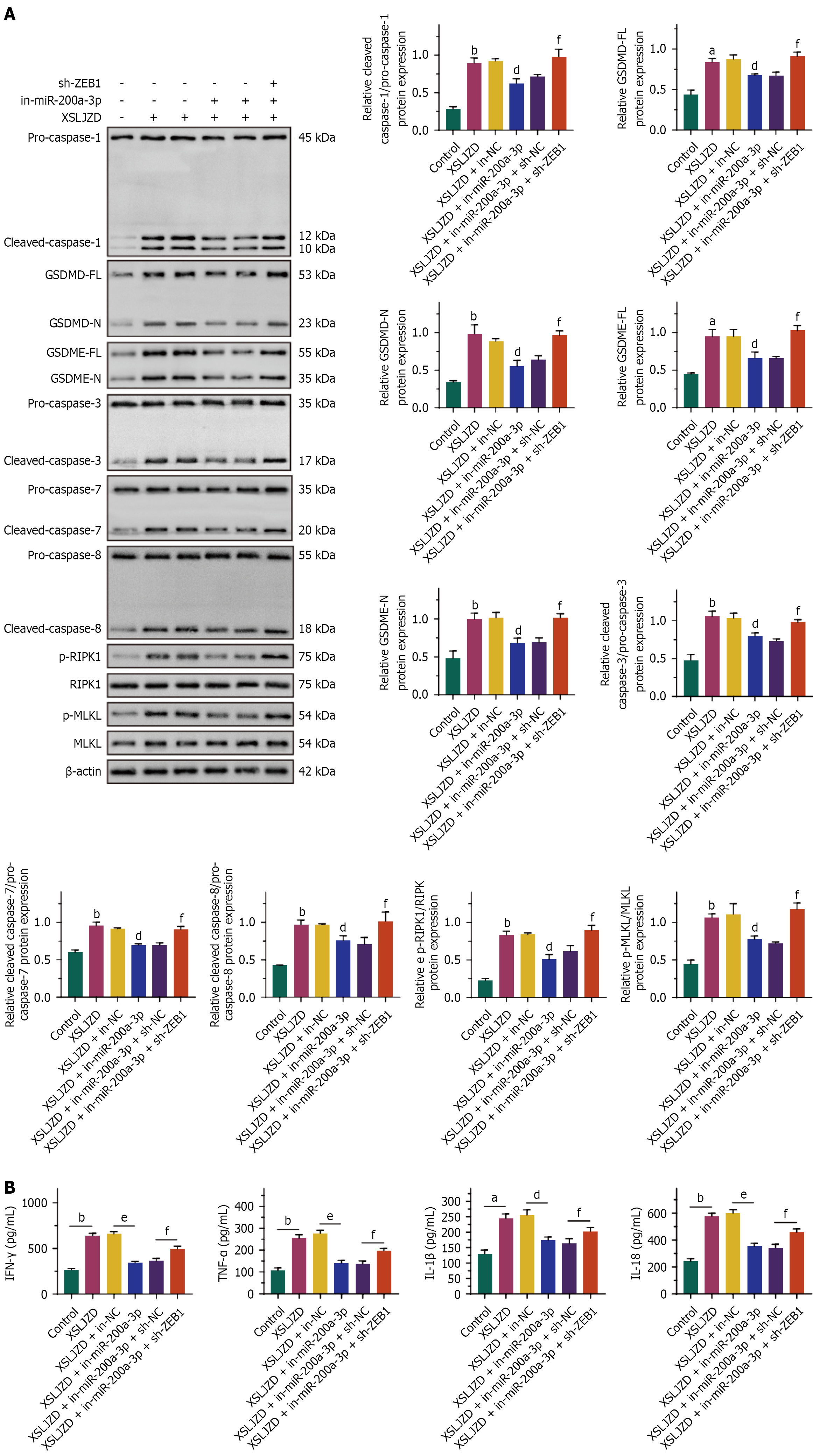
Figure 7 The effect of Xiangshaliujunzi decoction on the expression of PANoptosis-related proteins and inflammatory factors was mediated by the miR-200a-3p/ZEB1 axis.
Xiangshaliujunzi decoction (XSLJZD) enhanced the expression levels of pyroptosis-related proteins, apoptosis-related proteins, and programmed cell death (PCD)-related proteins. XSLJZD upregulated the secretion of interferon (IFN)-γ, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-18 in BGC-823/5-fluorouracil (5-FU) cells. Transfection of miR-200a-3p inhibitors into BGC-823/5-FU cells partially abolished the effects of XSLJZD on the expression of PANoptosis-related proteins and inflammatory factors. Knocking down ZEB1 expression in BGC-823/5-FU cells transfected with miR-200a-3p inhibitors partially restored the effects of XSLJZD on PANoptosis-related proteins and inflammatory factor expression. A: Expression levels of pyroptosis-related proteins, apoptosis-related proteins, and PCD-related proteins; B: IFN-γ, TNF-α, IL-1β, and IL-18 in cell culture medium detected by ELISA. aP < 0.05, bP < 0.01, cP < 0.001 vs Control. dP < 0.05, eP < 0.01 vs XSLJZD + in-negative control (NC). fP < 0.05 vs XSLJZD + sh-NC. XSLJZD: Xiangshaliujunzi decoction; IFN: Interferon; TNF: Tumor necrosis factor; IL: Interleukin.
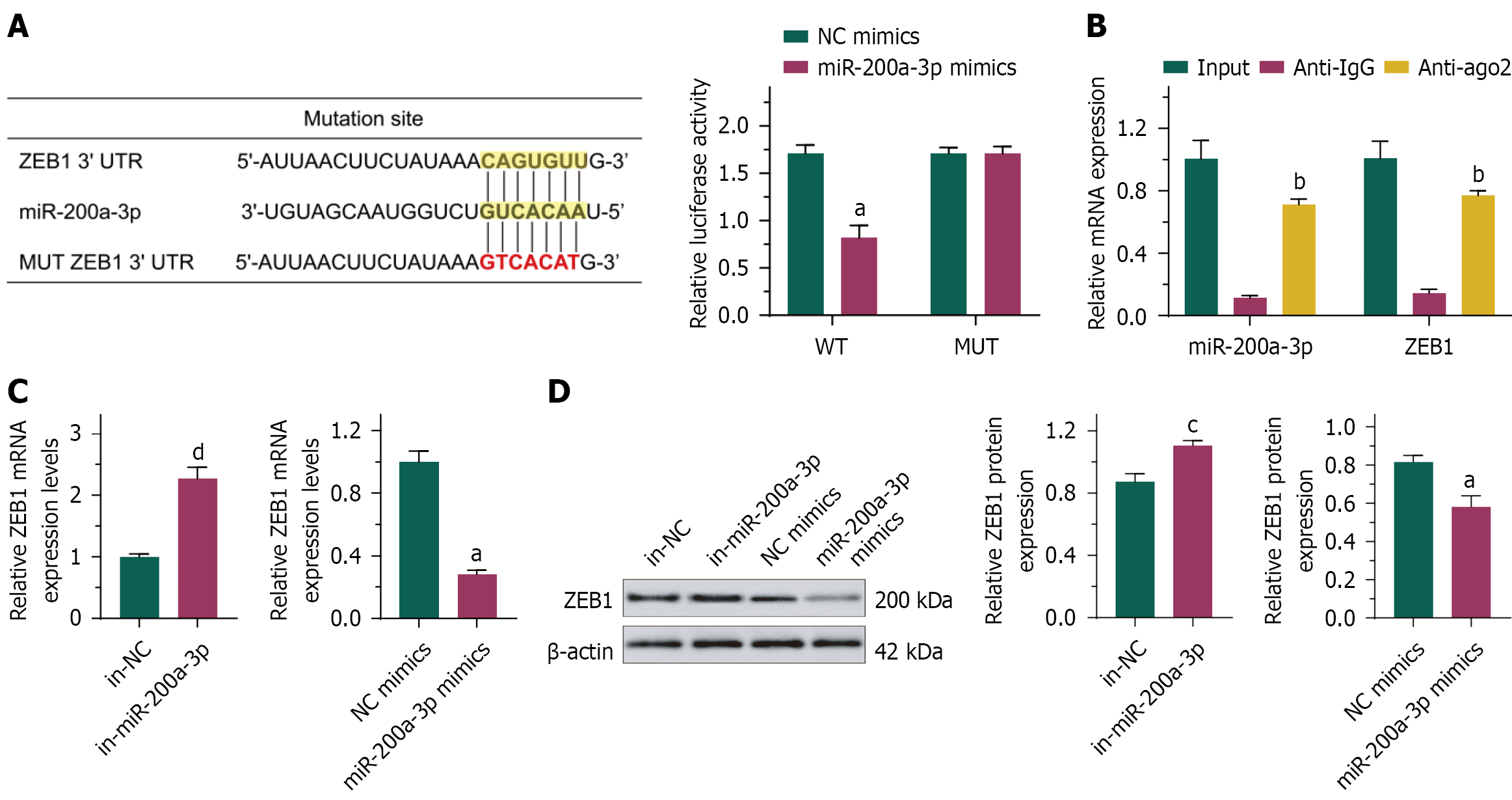
Figure 8 MiR-200a-3p inhibited ZEB1 expression by binding to the ZEB1 3’UTR.
A: Interaction between ZEB1 3’UTR and ZEB1 detected by dual-luciferase reporter assays; B: The interaction between miR-200a-3p and ZEB1 detected by RIP assays; C: ZEB1 mRNA expression; D: ZEB1 protein expression. aP < 0.01 vs negative control (NC) mimics. bP < 0.05 vs anti-IgG. cP < 0.05, dP < 0.01 vs in-NC. XSLJZD: Xiangshaliujunzi decoction; NC: Negative control.
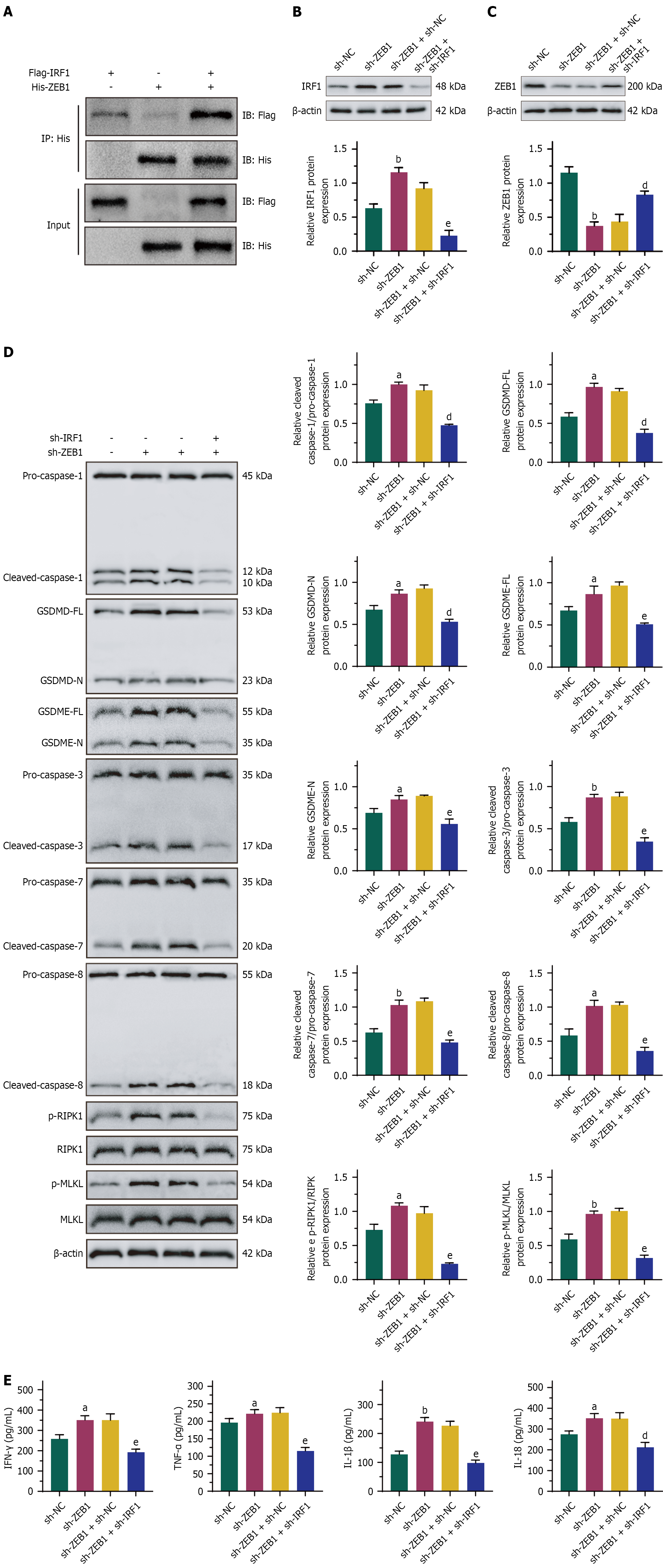
Figure 9 Regulation of ZEB1 to PANoptosis was mediated by IRF1 was present in the ZEB1 immunoprecipitation complex, and con
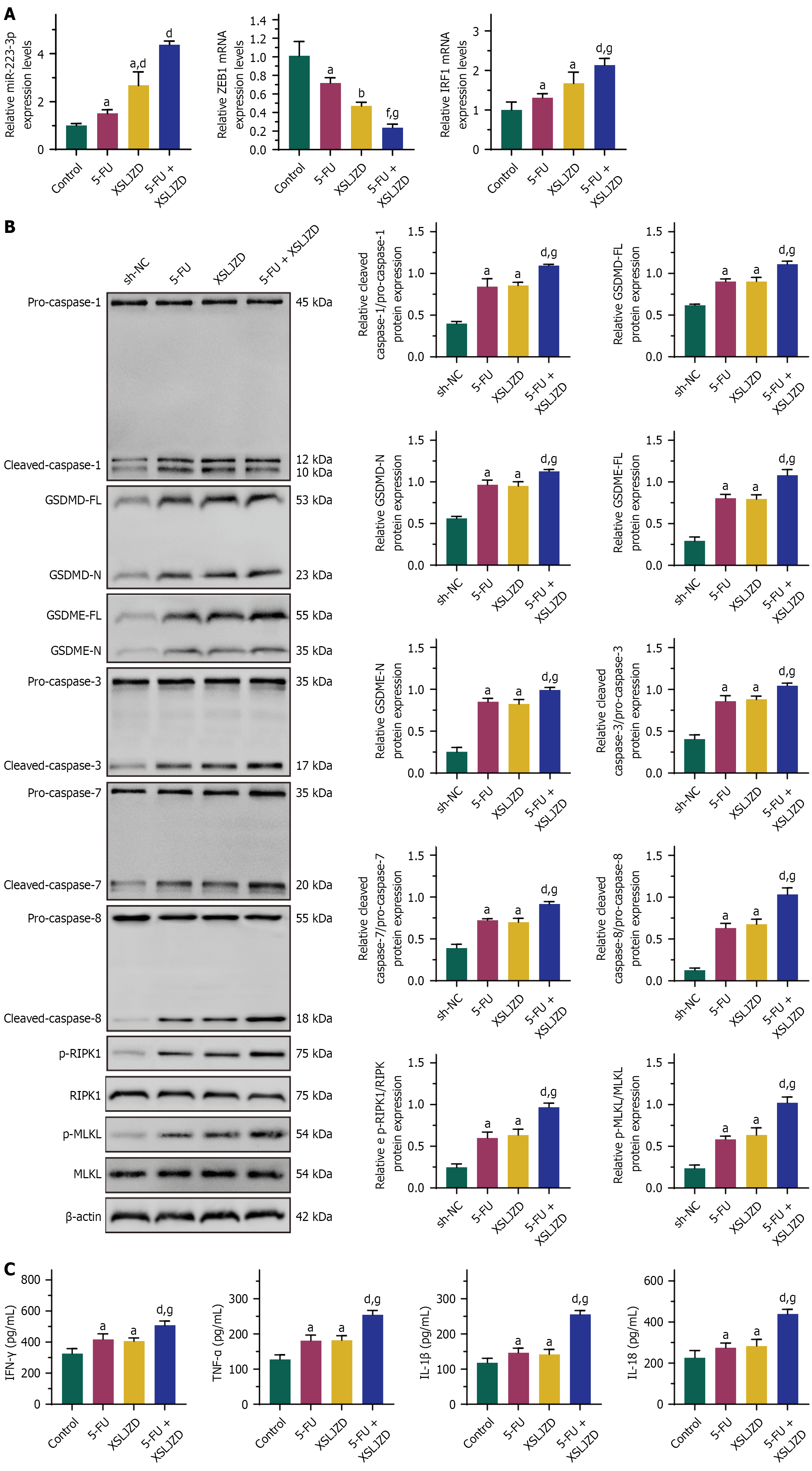
Figure 10 The combination of Xiangshaliujunzi decoction and 5-fluorouracil promoted miR-200a/ZEB1/IRF1-mediated PANoptosis in transplanted tumor tissues in mice.
The combination treatment group [5-fluorouracil (5-FU) + Xiangshaliujunzi decoction (XSLJZD)] showed higher levels of miR-200a-3p and IRF1 mRNA and lower ZEB1 mRNA compared to the individual 5-FU and XSLJZD groups. The combination treatment group (5-FU + XSLJZD) showed further increased levels of PANoptosis-related proteins and inflammatory cytokines [interferon (IFN)-γ, tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-18] compared to the 5-FU and XSLJZD groups individually. A: MiR-200a-3p, IRF1 mRNA and lower ZEB1 mRNA expression; B: PANoptosis-related proteins expression; C: IFN-γ, TNF-α, IL-1β, and IL-18 expression. aP < 0.05, bP < 0.01 vs control/sh-negative control. dP < 0.05, fP < 0.001 vs 5-FU. gP < 0.05 vs XSLJZD. XSLJZD: Xiangshaliujunzi decoction; NC: Negative control; 5-FU: 5-fluorouracil.
- Citation: Gao H, Yin DF, Xing XR, Zhou LJ, Yu R. MiR-200a-3p/ZEB1/IRF1-mediated PANoptosis prompts Xiangshaliujunzi decoction to overcome 5-fluorouracil resistance in gastric cancer. World J Gastroenterol 2026; 32(14): 114331
- URL: https://www.wjgnet.com/1007-9327/full/v32/i14/114331.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i14.114331