Copyright: ©Author(s) 2026.
World J Gastroenterol. Mar 28, 2026; 32(12): 115853
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.115853
Published online Mar 28, 2026. doi: 10.3748/wjg.v32.i12.115853
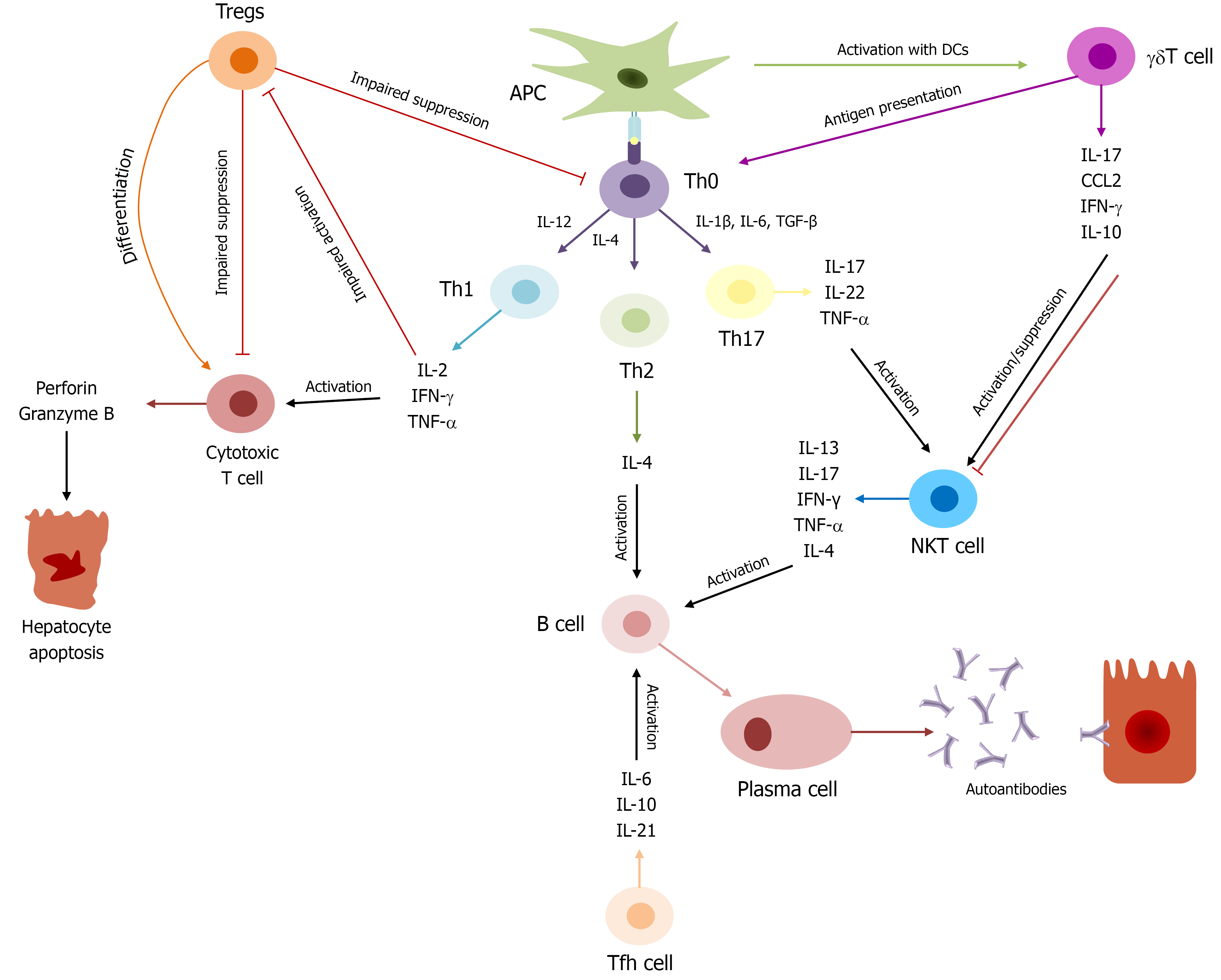
Figure 1 Immunopathogenesis of autoimmune hepatitis.
Hepatic autoantigens interact with antigen-presenting cells, which lead to the activation of cluster of differentiation (CD) 4+ T helper (Th) 0 with differentiation into Th1, Th2, Th17 cells, secreting different cytokines [various interleukin (IL), tumor necrosis factor-α, and interferon-γ]. Activation of cytotoxic CD8+ T cells with Th1 cytokines inducing hepatocyte apoptosis. CD4+ Th2, natural killer T cells and T follicular helper activate B cells with different cytokines (IL-4, IL-6, IL-21 and etc.) and promote their differentiation into plasma cells, producing autoantibodies. Regulatory T cells are defective in autoimmune hepatitis patients and cannot suppress CD8+ and CD4+ T cells, but can transform into cytotoxic T cells with further hepatocyte apoptosis. Gamma delta T cells can act as pro- and anti-inflammatory cells. Proinflammatory cytokines induce inflammation, cellular infiltration and liver damage. Tregs: Regulatory T cells; Th: T helper; IL: Interleukin; IFN: Interferon; TNF: Tumor necrosis factor; APC: Antigen-presenting cells; TGF: Transforming growth factor; DC: Dendritic cell; NKT: Natural killer T cell; Tfh: T follicular helper cell; γδT: Gamma delta T cell.
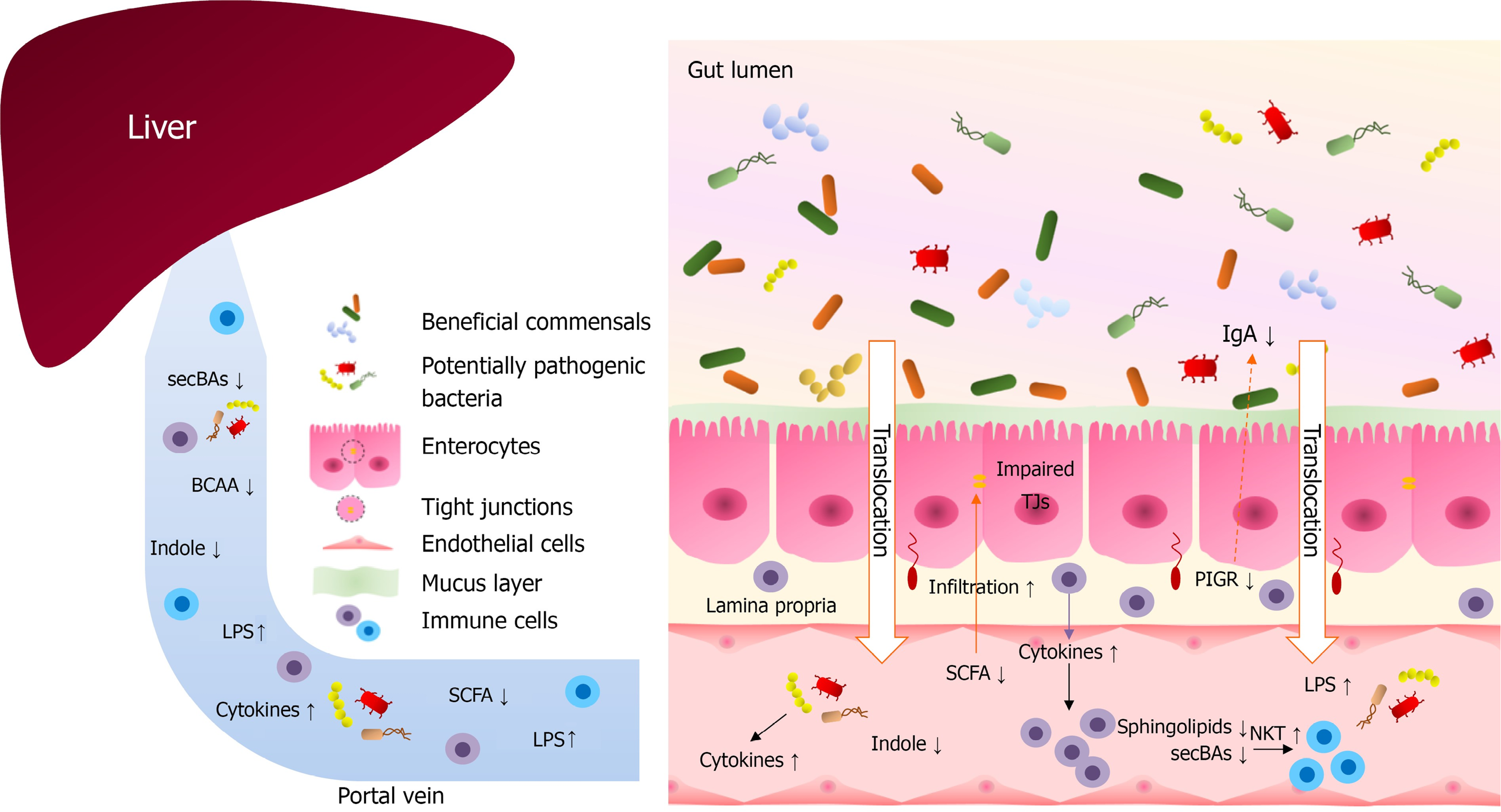
Figure 2 Microbiota-associated pathogenesis of autoimmune hepatitis.
Dysbiosis induce decreased tight junction (TJ) proteins expression and increased intestinal permeability, which leads to elevated serum lipopolysaccharide (LPS), infiltration of lamina propria and enhanced cytokines production. Increased LPS, bacteria and inflammatory cells migrate to the liver and initiate inflammation. Dysbiosis also leads to decreased production of short-chain fatty acids, branched-chain amino acids, secondary bile acids, indole, sphingolipids and etc., which play a protective role in the gut and liver. Decreased level of polymeric immunoglobulin receptor leads to reduced immunoglobulin A secretion and dysbiosis, inhibited TJ proteins expression and increased intestinal permeability. secBAs: Secondary bile acids; BCAA: Branched-chain amino acid; LPS: Lipopolysaccharide; SCFA: Short-chain fatty acid; IgA: Immunoglobulin A; TJ: Tight junction; PIGR: Polymeric immunoglobulin receptor; NKT: Natural killer T cell.
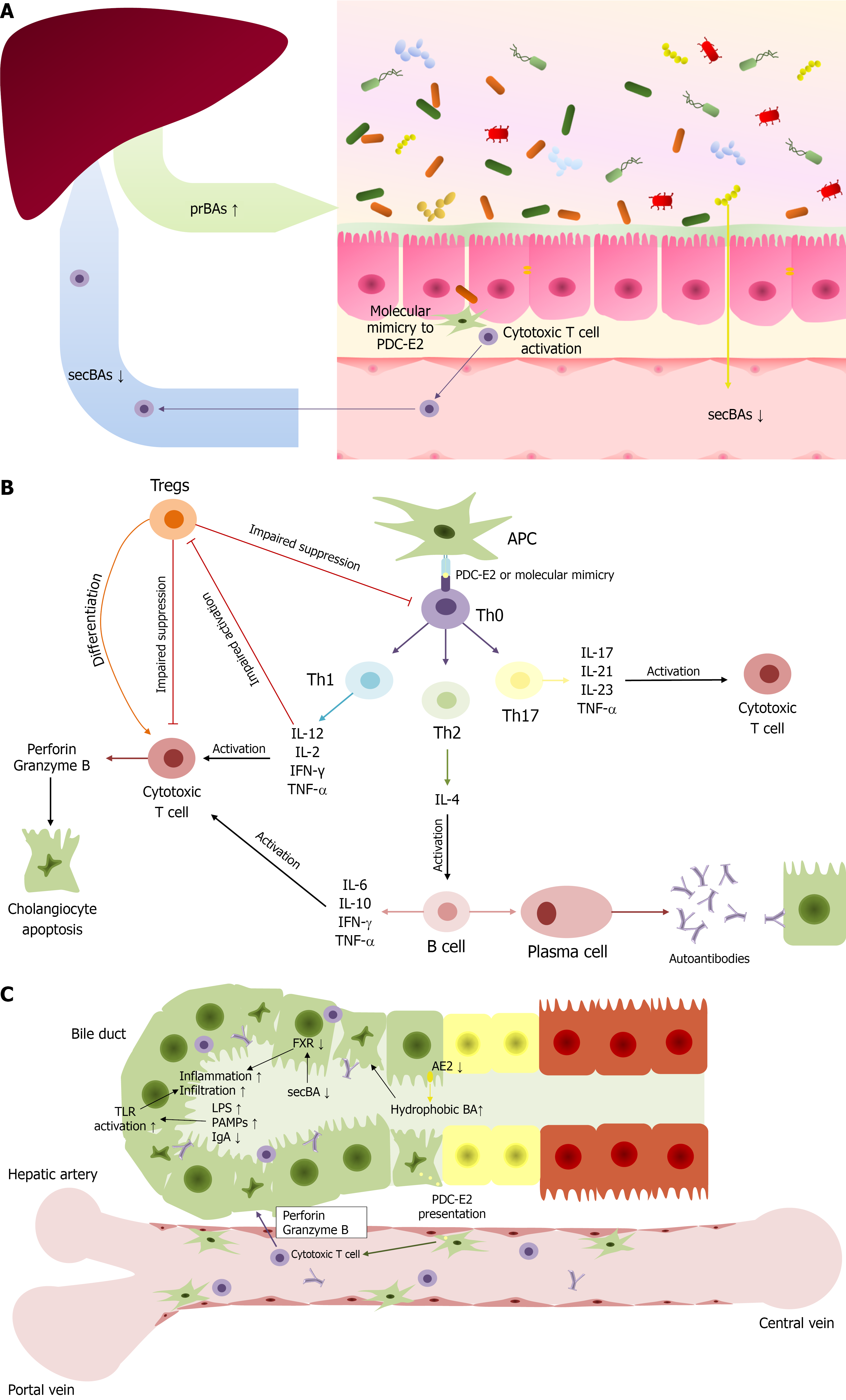
Figure 3 Gut microbiota in primary biliary cholangitis pathogenesis.
A: Bacterial molecular mimicry for E2 subunit of the pyruvate dehydrogenase complex (PDC-E2) can activate cytotoxic T cells, which transfer to the liver and attack cholangiocytes. Dysbiosis leads to the increased intestinal permeability and endotoxinemia, and decreased secondary bile acids (secBAs) production despite the increased intake of primary bile acids into the intestine; B: Antigen-presenting cells present PDC-E2 or molecules similar to it to cluster of differentiation 4+ T cells, that transform into T helper (Th) 1, Th2, Th17 cells with production of different cytokines (various interleukin, tumor necrosis factor-α, and interferon-γ) and activate both cytotoxic T cells and B cells. This process leads to increased autoantibodies secretion and cholangiocyte damage; C: Activated cytotoxic T cells induce cholangiocytes apoptosis. Suppressed anion exchanger 2 reduces the protection of cholangiocytes from hydrophobic bile acids in bile. Decreased secBAs lead to insufficient farnesoid X receptor activation and enhanced inflammation, as well as excessive levels of lipopolysaccharide and pathogen-associated molecular patterns activate Toll-like receptors. secBAs: Secondary bile acids; prBAs: Primary bile acids; PDC-E2: E2 subunit of the pyruvate dehydrogenase complex; Tregs: Regulatory T cells; Th: T helper; IL: Interleukin; IFN: Interferon; TNF: Tumor necrosis factor; APC: Antigen-presenting cells; TLR: Toll like receptor; LPS: Lipopolysaccharide; PAMPs: Pathogen-associated molecular patterns; IgA: Immunoglobulin A; FXR: Farnesoid X receptor; AE2: Anion exchanger 2; BA: Bile acid.
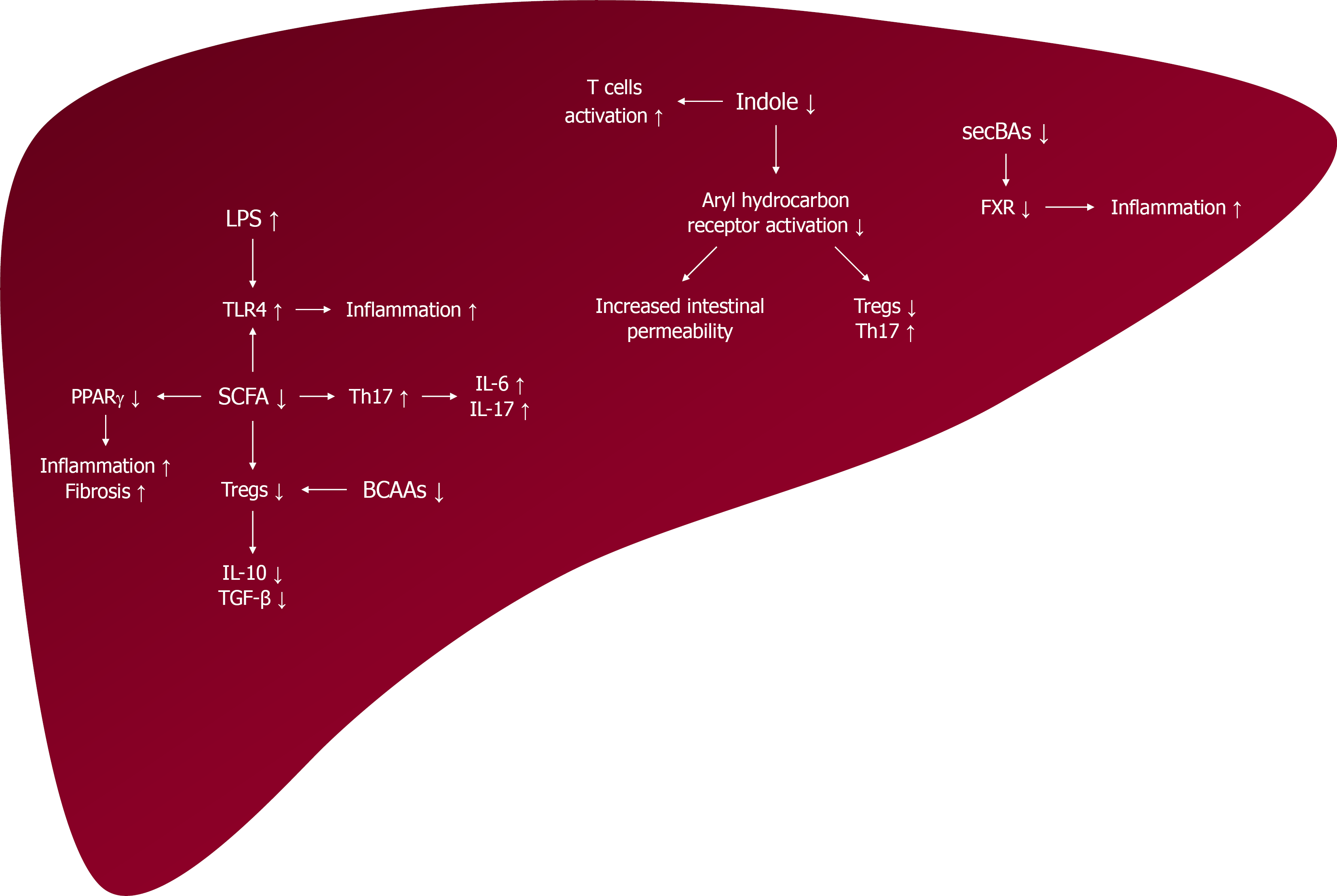
Figure 4 Microbiota-derived metabolites and immune system.
Gut dysbiosis leads to a reduced production of short-chain fatty acids (SCFA), branched-chain amino acids, secondary bile acids, and indole metabolites. These changes result in enhanced activation of pro-inflammatory cells (T helper 17 and others) and in the production of pro-inflammatory cytokines. In immune liver diseases in both patients and animal models, there is a deficiency and dysfunction of regulatory T cells (Tregs). Dysbiosis-associated decrease in the production of SCFAs and indole metabolites leads to more significant decrease in anti-inflammatory Treg cells and the cytokines they secrete (interleukin-10, transforming growth factor-β). Additionally, dysbiosis increases intestinal permeability, leading to high concentrations of lipopolysaccharide in blood and activation of Toll like receptor 4, which also amplifies inflammation. SCFA can induce the hepatic gamma-type peroxisome proliferator-activated receptor; therefore, a decrease in the production of the former leads to reduced activation of the latter, which results in increased inflammation and fibrosis processes. TLR: Toll like receptor; LPS: Lipopolysaccharide; Tregs: Regulatory T cells; SCFAs: Short-chain fatty acids; IL: Interleukin; TGF: Transforming growth factor; PPAR: Peroxisome proliferator-activated receptor; Th: T helper; BCAA: Branched-chain amino acid; FXR: Farnesoid X receptor; secBAs: Secondary bile acids.
- Citation: Kiseleva Y, Maslennikov R, Poluektova E, Zolnikova O, Sigidaev A, Zharikov Y, Shirokova E, Ivashkin V. Microbiome-immune interactions in autoimmune liver diseases. World J Gastroenterol 2026; 32(12): 115853
- URL: https://www.wjgnet.com/1007-9327/full/v32/i12/115853.htm
- DOI: https://dx.doi.org/10.3748/wjg.v32.i12.115853