Published online Mar 26, 2026. doi: 10.12998/wjcc.v14.i9.119273
Revised: February 14, 2026
Accepted: March 5, 2026
Published online: March 26, 2026
Processing time: 55 Days and 16.1 Hours
Venous air embolism (VAE) is a recognized complication of neurosurgical pro
We report the case of a 22-year-old male undergoing awake craniotomy in supine position for resection of a right fronto-temporal low-grade glioma, identified on preoperative magnetic resonance imaging as a non-enhancing cortical lesion in close proximity to eloquent motor cortex, with functional magnetic resonance imaging demonstrating perilesional motor and supplementary motor area acti
VAE can occur during awake craniotomy even in the supine position, potentially precipitated by seizure-related deep inspiratory efforts. Given the nonspecific presentation and diagnostic limitations in awake neurosurgery, a high index of suspicion and prompt supportive management are essential to ensure favourable outcomes.
Core Tip: Venous air embolism (VAE), though classically associated with neurosurgical procedures in the sitting position, can also occur during awake craniotomy in the supine position and may present with subtle, nonspecific symptoms. In awake patients, seizure-related deep inspiratory efforts and transient negative intrathoracic pressure can facilitate air entrainment through exposed cerebral veins, particularly when the surgical field is positioned above the level of the heart. Overlap between seizure activity and the clinical manifestations of VAE can delay recognition in the absence of advanced mo
- Citation: Shiva Priya K, Kaushal A, Lokho P, Balasubramanian N, Agrawal A, Kant S, Badetiya N. Seizure-associated venous air embolism during awake craniotomy in the supine position: A case report. World J Clin Cases 2026; 14(9): 119273
- URL: https://www.wjgnet.com/2307-8960/full/v14/i9/119273.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i9.119273
Venous air embolism (VAE) refers to the entrainment of air into the venous circulation from exposed surgical sites, potentially leading to significant cardiopulmonary and neurological consequences[1]. While many episodes remain subclinical, clinically significant VAE has been most frequently reported during neurosurgical procedures performed in the sitting position, with an incidence ranging from 10% to 80%[1]. However, VAE may occur in any surgical position in which the operative field is above the level of the heart.
Awake craniotomy has become increasingly common for lesions involving eloquent cortical regions, allowing real-time neurological assessment. Despite its advantages, detection of VAE during awake craniotomy is particularly challenging due to limited monitoring modalities and the absence of invasive airway control[2,3]. Reports of VAE during awake craniotomy, especially in the supine position, are scarce, with only isolated cases described in the literature[2].
We describe a rare case of VAE occurring toward the end of tumor resection in a patient undergoing awake craniotomy in the supine position, likely precipitated by seizure-associated deep inspiratory efforts.
A 22-year-old male undergoing awake craniotomy in supine position for resection of a right fronto-temporal low-grade glioma. Intraoperatively, near the completion of tumor excision during awake craniotomy, the patient developed sudden dyspnea associated with persistent coughing, chest tightness, tachypnea, tachycardia, and a drop in oxygen saturation.
The patient was undergoing awake craniotomy in the supine position for resection of a right fronto-temporal glioma. Standard monitoring was in place, including electrocardiography, pulse oximetry, invasive arterial blood pressure monitoring, and capnography via nasal cannula. Supplemental oxygen was administered at 3 L/minute through nasal prongs. Sedation was provided using dexmedetomidine (loading dose of 1 μg/kg over 15 minutes followed by infusion at 0.5 μg/kg/hour) and fentanyl infusion at 1 μg/kg/hour. A scalp block was performed using 0.5% bupivacaine.
The patient’s head was fixed in a Mayfield clamp with approximately 10° of neck flexion, and the head end of the operating table was elevated by about 20°. Sedation was discontinued during tumor resection to allow continuous neu
Toward the end of tumor excision, the patient complained of facial paresthesia and dyspnea, raising concern for non-convulsive seizure activity. The surgical field was irrigated with cold saline, and intravenous midazolam 1 mg was administered. Over the next 5-10 minutes, the patient developed persistent coughing, increasing respiratory discomfort, and chest tightness. Oxygen saturation decreased from 98%-100% to 89%, accompanied by tachypnea and tachycardia. End-tidal carbon dioxide values decreased, although interpretation was limited due to displacement of the sampling cannula during coughing episodes. On auscultation, the lung fields were clear; however, a new, continuous, mild pre
The patient had a six-month history of progressive left-sided upper and lower limb weakness accompanied by recurrent focal seizures. He was diagnosed with a right fronto-temporal lobe glioma and was receiving antiepileptic therapy with phenytoin, levetiracetam, and lacosamide. There was no history of cardiovascular, respiratory, or metabolic disease. He had no prior surgeries and no known drug allergies.
The patient was a non-smoker and did not consume alcohol or illicit substances. There was no history of recent infection or trauma. Family history was non-contributory, with no known hereditary neurological, cardiovascular, or pulmonary disorders.
Preoperatively, the patient was conscious, oriented, and cooperative. Vital signs were stable. Neurological examination revealed mild left-sided hemiparesis (Medical Research Council grade 4/5) with no cranial nerve deficits. Cardiovascular and respiratory examinations were unremarkable, and airway assessment was normal.
Preoperative laboratory investigations, including complete blood count, serum electrolytes, renal and liver function tests, coagulation profile, electrocardiographic (ECG), and chest radiography, were within normal limits.
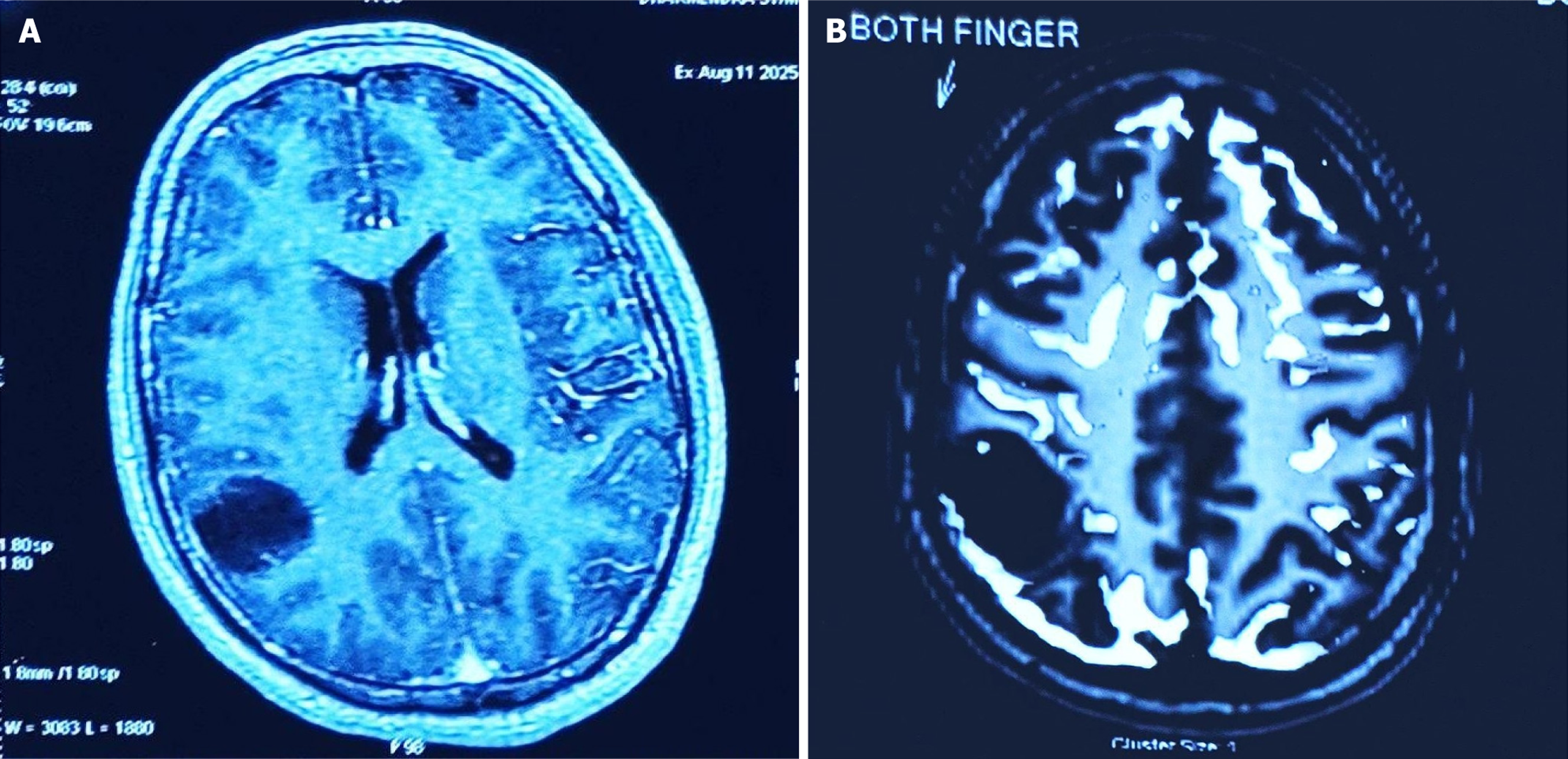
Preoperative magnetic resonance imaging (MRI) brain revealed a well-defined, irregular intra-axial lesion measuring approximately 3.8 cm × 3.2 cm × 3.7 cm involving the right fronto-temporal cortical region (Figure 2A). The lesion was hypointense on T1-weighted images and hyperintense on T2-weighted and fluid-attenuated inversion recovery sequences, with no diffusion restriction, hemorrhage, calcification, or significant post-contrast enhancement. There was no surrounding edema or mass effect. Imaging features were suggestive of a low-grade cortical neoplasm, such as diffuse neuroepithelial tumor or low-grade glioma.
Functional MRI of the brain was subsequently performed for preoperative motor cortex mapping using blood-oxygen-level-dependent sequences. Motor paradigms included bilateral finger tapping and bilateral foot tapping. During right and left finger tapping, few activation stripes were observed predominantly in the periphery of the lesion, with some activation extending within the lesion. Supplementary motor area activation was noted bilaterally (Figure 2B). During bilateral foot tapping, few activation stripes were again noted in the perilesional region, with predominant activation involving the right supplementary motor area. These findings indicated close proximity of eloquent motor cortical areas to the lesion, emphasizing the need for awake craniotomy with continuous neurological monitoring.
Intraoperative VAE during awake craniotomy in the supine position, likely precipitated by seizure-associated deep inspiratory efforts.
Immediate management included flooding of the surgical field with saline, positioning the patient in the Trendelenburg position, administration of 100% oxygen via a tightly sealed face mask, and application of 5 cm H2O positive end-ex
Post-event transthoracic echocardiography demonstrated no evidence of cardiac dysfunction. Lung ultrasound revealed bilateral B-lines, consistent with pulmonary interstitial involvement secondary to VAE. The patient remained hemodynamically stable postoperatively, with no neurological deterioration or respiratory sequelae. He was monitored in the neurosurgical intensive care unit and discharged with no residual cardiopulmonary complications attributable to the in
VAE is a well-recognized complication of neurosurgical procedures, particularly when performed in the sitting position, where a favorable pressure gradient between the surgical field and the right atrium facilitates air entrainment[1]. Nevertheless, VAE can occur in any surgical position in which the operative site is elevated above the level of the heart, including the supine position, especially in the presence of additional predisposing factors. Reports of VAE during awake craniotomy remain rare. In awake patients, spontaneous deep inspiratory efforts can generate negative intrathoracic pressure that is transmitted to central and cerebral venous structures, thereby increasing the likelihood of air entrainment through exposed venous channels[3].
VAE can present with highly variable and nonspecific clinical signs, especially when advanced monitoring is limited during awake procedures. The clinical presentation may include sudden dyspnea, coughing, desaturation, chest dis
The diagnosis of VAE is based on clinical suspicion supported by physiological and imaging modalities. Capnography is widely available and a sudden decrease in end-tidal carbon dioxide (EtCO2), particularly > 5 mmHg and when ass
Despite the availability of multiple monitoring modalities, no single technique offers uniformly high sensitivity and specificity across all clinical scenarios. In practice, a multimodal approach, integrating vigilant clinical observation with trends in end-tidal CO2, and adjunctive use of precordial Doppler or transesophageal echocardiography when feasible, provides the highest diagnostic yield.
In the present case, seizure-associated dyspnea and deep inspiratory efforts likely led to transient reductions in cerebral venous pressure, promoting venous air entrainment. This risk may have been further exacerbated by head elevation relative to the heart. Recognition of VAE was relatively delayed due to clinical overlap between seizure-related symptoms and the nonspecific signs of air embolism, compounded by the limited sensitivity of conventional intraoperative monitoring techniques typically employed during awake craniotomy.
Pulmonary air embolism can produce a spectrum of physiological effects, including sympathetic activation, pulmonary vasoconstriction, ventilation-perfusion mismatch, and hypoxemia[7,8]. Beyond these immediate effects, endothelial activation and inflammatory responses may increase pulmonary capillary permeability, leading to interstitial pulmonary edema, as documented in previous reports[8-10]. Collectively, these mechanisms provide a plausible explanation for the hypoxemia and ultrasound findings observed in the present patient.
The clinical severity of VAE is primarily determined by both the volume and rate of air entrainment. While small volumes of air may remain clinically insignificant, rapid introduction of larger volumes can markedly reduce cardiac output, resulting in hemodynamic instability and characteristic ECG changes[3]. In the current case, the absence of hy
This case underscores the importance of maintaining high clinical vigilance for atypical presentations of VAE during awake neurosurgical procedures. Symptoms such as persistent coughing, unexplained dyspnea, chest discomfort, or sudden hypoxemia in an awake patient should prompt consideration of VAE, even when overt hemodynamic deterioration is not evident.
VAE can occur during awake craniotomy even in the supine position and may be precipitated by seizure-associated alterations in respiratory mechanics, including deep inspiratory efforts. Because clinical manifestations are often subtle and nonspecific in awake patients, diagnosis may be delayed. A high index of suspicion and prompt institution of sup
| 1. | Orebaugh SL. Venous air embolism: clinical and experimental considerations. Crit Care Med. 1992;20:1169-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 152] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 2. | Balki M, Manninen PH, McGuire GP, El-Beheiry H, Bernstein M. Venous air embolism during awake craniotomy in a supine patient. Can J Anaesth. 2003;50:835-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 3. | Scuplak SM, Smith M, Harkness WF. Air embolism during awake craniotomy. Anaesthesia. 1995;50:338-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | White-Dzuro GA, Smith MR, Guo A, West T, Mueller AL, Houle T, Akeju O, Nahed B, Rhee J. Detection of Venous Air Embolism in Nonsitting Craniotomy for Tumor Patients: A Retrospective Case Series. J Neurosurg Anesthesiol. 2026;38:76-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Moitra V, Permut TA, Penn RM, Roth S. Venous air embolism in an awake patient undergoing placement of deep brain stimulators. J Neurosurg Anesthesiol. 2004;16:321-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 6. | Rau TR, Plaschke K, Weigand MA, Maier C, Schramm C. Automatic detection of venous air embolism using transesophageal echocardiography in patients undergoing neurological surgery in the semi-sitting position: a pilot study. J Clin Monit Comput. 2021;35:1103-1109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Lam KK, Hutchinson RC, Gin T. Severe pulmonary oedema after venous air embolism. Can J Anaesth. 1993;40:964-967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 8. | Frim DM, Wollman L, Evans AB, Ojemann RG. Acute pulmonary edema after low-level air embolism during craniotomy. Case report. J Neurosurg. 1996;85:937-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 9. | Wood S, Fuzaylov G. Acute pulmonary edema due to occult air embolism detected on an automated anesthesia record: illustrative case. J Neurosurg Case Lessons. 2021;1:CASE2075. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 10. | Perschau RA, Munson ES, Chapin JC. Pulmonary interstitial edema after multiple venous air emboli. Anesthesiology. 1976;45:364-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 21] [Article Influence: 0.4] [Reference Citation Analysis (0)] |