Published online Mar 16, 2026. doi: 10.12998/wjcc.v14.i8.118789
Revised: January 30, 2026
Accepted: February 24, 2026
Published online: March 16, 2026
Processing time: 64 Days and 19.8 Hours
Polyneuropathy, organomegaly, endocrinopathy, monoclonal protein and skin changes (POEMS) syndrome is a rare plasma cell neoplasm with associated paraneoplastic syndrome. Castleman disease variant of POEMS syndrome, lacking detectable M-protein is uncommon and presents substantial diagnostic and therapeutic challenges. We illustrate a case of Castleman disease variant of POEMS syndrome and the complexity of its clinical management.
A 56-year-old man presented with limb weakness, sensory neuropathy, weight loss, and hyperpigmentation. He was diagnosed with Castleman disease variant of POEMS syndrome, having fulfilled 3 major criteria (poly
Although a monoclonal plasma cell disorder is a mandatory diagnostic criterion for POEMS syndrome, it may be minimal or undetectable in some patients with the Castleman disease variant who otherwise fulfil diagnostic criteria.
Core Tip: Castleman disease variant of polyneuropathy, organomegaly, endocrinopathy, monoclonal protein and skin changes syndrome is a rare and challenging multisystem disorder to manage, with atypical variants without detectable M-protein exceptionally rare. This case highlights the importance of recognizing key clinical features and utilizing supportive markers, such as vascular endothelial growth factor and histopathology. To improve functional outcomes, quality of life, and minimize long-term morbidity, early diagnosis enables timely systemic chemotherapy treatment and multidisciplinary supportive care.
- Citation: Paw LJ, Tan JK, Teoh CX, Megat Mohamad Aminuddin PNA, Agusalim AF, Cheong XK, Payus AO, Wan Nik Ahmad Mustafa WAK, Omar UA, Mustafa N, Chong GY, Tumian NR, Abdul Wahid SF, Chong CY, Tan HJ, Remli R. Castleman disease variant of polyneuropathy, organomegaly, endocrinopathy, monoclonal protein and skin changes syndrome: A case report. World J Clin Cases 2026; 14(8): 118789
- URL: https://www.wjgnet.com/2307-8960/full/v14/i8/118789.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i8.118789
Polyneuropathy, organomegaly, endocrinopathy, monoclonal protein and skin changes (POEMS), is a rare and de
Castleman disease (or angiofollicular lymph node hyperplasia) is a rare lymphoproliferative disorder, diagnosed based on histopathological findings. A retrospective study of 99 patients with POEMS syndrome, showed that 25 of 43 biopsied lymph nodes were diagnostic of Castleman disease, and 84% of these had a hyaline vascular type. Only patients with both peripheral neuropathy and a plasma cell clone can be classified as having classic POEMS syndrome. Those without either characteristic can be classified as having a Castleman disease variant of POEMS syndrome[4,7]. Despite its myriad of symptoms, it is commonly misdiagnosed as chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) or other paraproteinemic neuropathies, and it would not be unusual to reach a final diagnosis of POEMS only after 1-2 years from the initial presentation[5].
There have been few published reports on patients presenting with features similar to POEMS syndrome but without any detectable M-protein, a mandatory criterion for its diagnosis. We present an exceptionally rare case of Castleman disease variant of POEMS syndrome to highlight the diagnostic and management issues encountered, with a review of existing literature.
A 56-year-old Malay man presented to the emergency department of a tertiary care center with acute left upper limb weakness and numbness upon awakening.
He woke up with weakness and numbness in his left upper and lower limbs. He could still ambulate with aid as his muscle power progressively improved. There was no associated slurring of speech, facial numbness, headache, vomiting, or giddiness. He was last known to be well the night before going to sleep and presented to the emergency department 12 hours later. Upon further questioning, he reported feeling unwell for the past 4 months, with lethargy, easy fatiguability, unintentional weight loss (18 kg), poor appetite and hyperpigmented skin. Furthermore, he reported progressive leg swelling with peripheral numbness over the past month.
He was recently diagnosed with diabetes mellitus, hypertension and dyslipidemia 4 months prior to presentation. He had no significant past medical or surgical history otherwise.
He was a reformed smoker, having stopped 20 years ago with a history of 5 pack years. He did not consume alcohol and was a pensioner who previously served in the military. There was no significant medical history or malignancy among his family members.
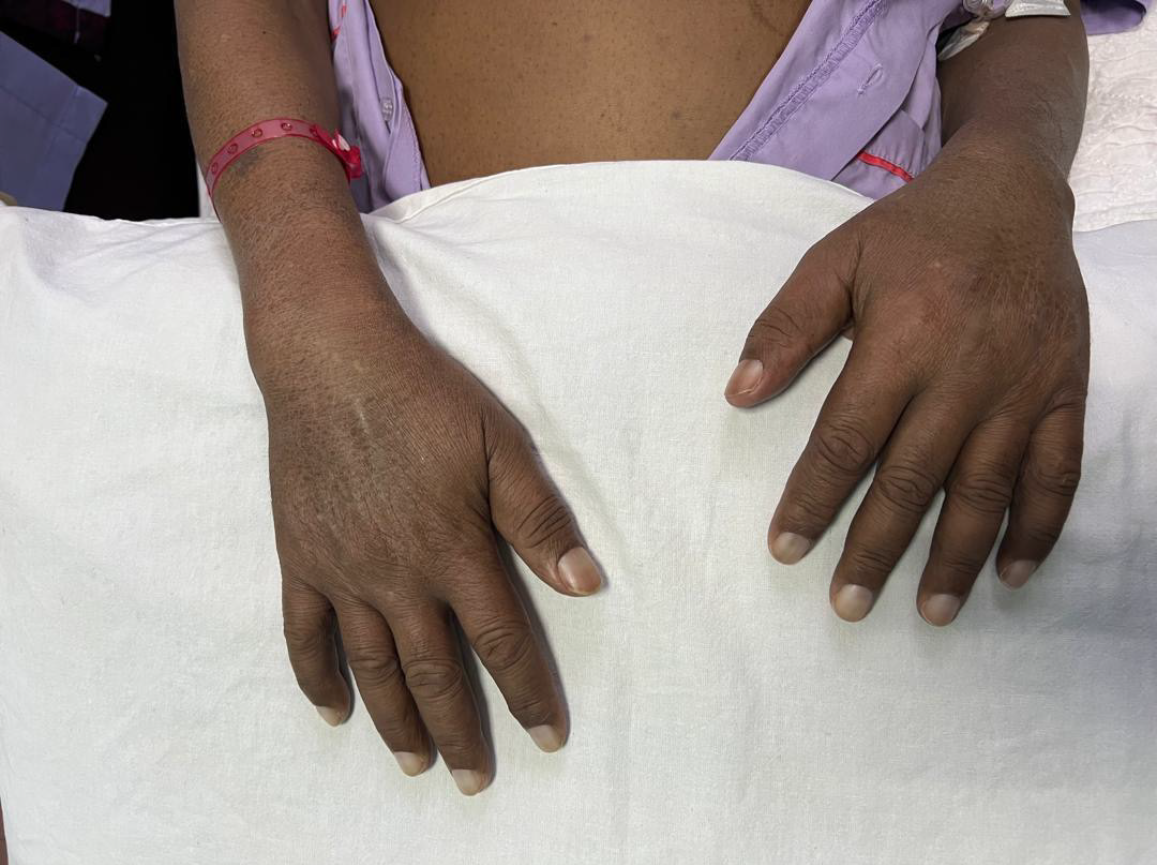
Clinically, he had anasarca, leukonychia, and finger clubbing (Figure 1). He had peripheral sensory loss of a stocking distribution until mid-shin with hyporeflexia over bilateral knees and ankles. Otherwise, muscle power and cranial nerves were grossly normal. He had a palpable 2 cm × 2 cm cervical lymph node, but no organomegaly clinically. Fundoscopy demonstrated bilateral papilledema.
Blood investigation results demonstrated persistent thrombocytosis, with platelet counts ranging from 600 × 109/L to 900 × 109/L, hypoalbuminemia (25-32 g/L), and a reversal of the albumin-globulin ratio (0.7). Serum protein electrophoresis and immunofixation revealed a polyclonal increase in immunoglobulins, with no paraprotein band present. Serum free light chain shows an elevated free Kappa/Lambda ratio of 1.97, while paired urine protein electrophoresis and im
Otherwise, his coagulation profile, liver enzymes, renal function and electrolytes were within normal limits. A long Synacthen and thyroid function test performed was suggestive of primary adrenal insufficiency and subclinical hypothyroidism. JAK-2 gene mutation, anticardiolipin, lupus anticoagulant and tumor markers demonstrated negative results. The anti-nuclear antibody (ANA) titer was elevated at 1:320 with a homogenous pattern, but complement levels were normal
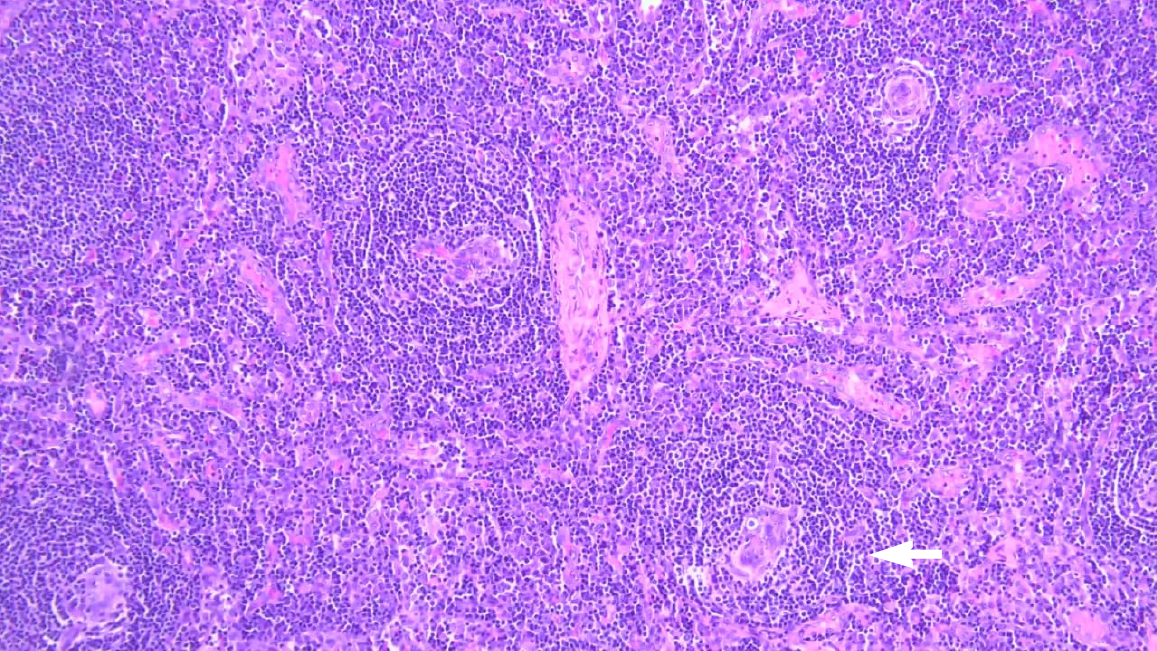
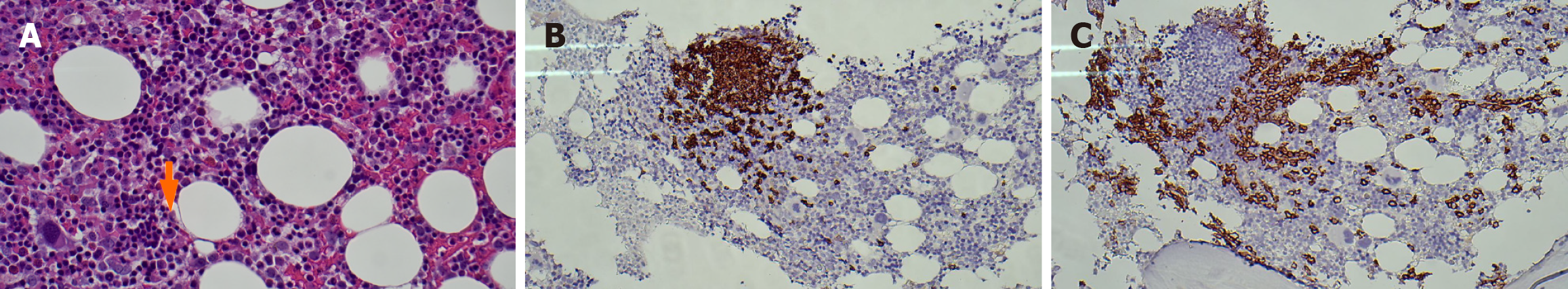
Excisional biopsy of the right cervical lymph node was suggestive of Castleman disease of mixed hyaline vascular and plasma cell variant (Figure 2). Furthermore, a bone marrow aspirate smear showed patchy hyper-normocellular marrow with 5% of plasma cells with mild plasmacytosis in the trephine sample, but no kappa and lambda light chain restriction (Figure 3A). Immunohistochemical staining for CD20 demonstrated aggregates of mature B-cells (Figure 3B), rimmed by plasma cells, which were highlighted with CD138 (Figure 3C). The presence of lymphoid nodules rimmed by clusters of plasma cells was suggestive of POEMS. To further support the diagnosis of Castleman disease variant of POEMS syndrome, the VEGF was markedly elevated (> 700 pg/mL).
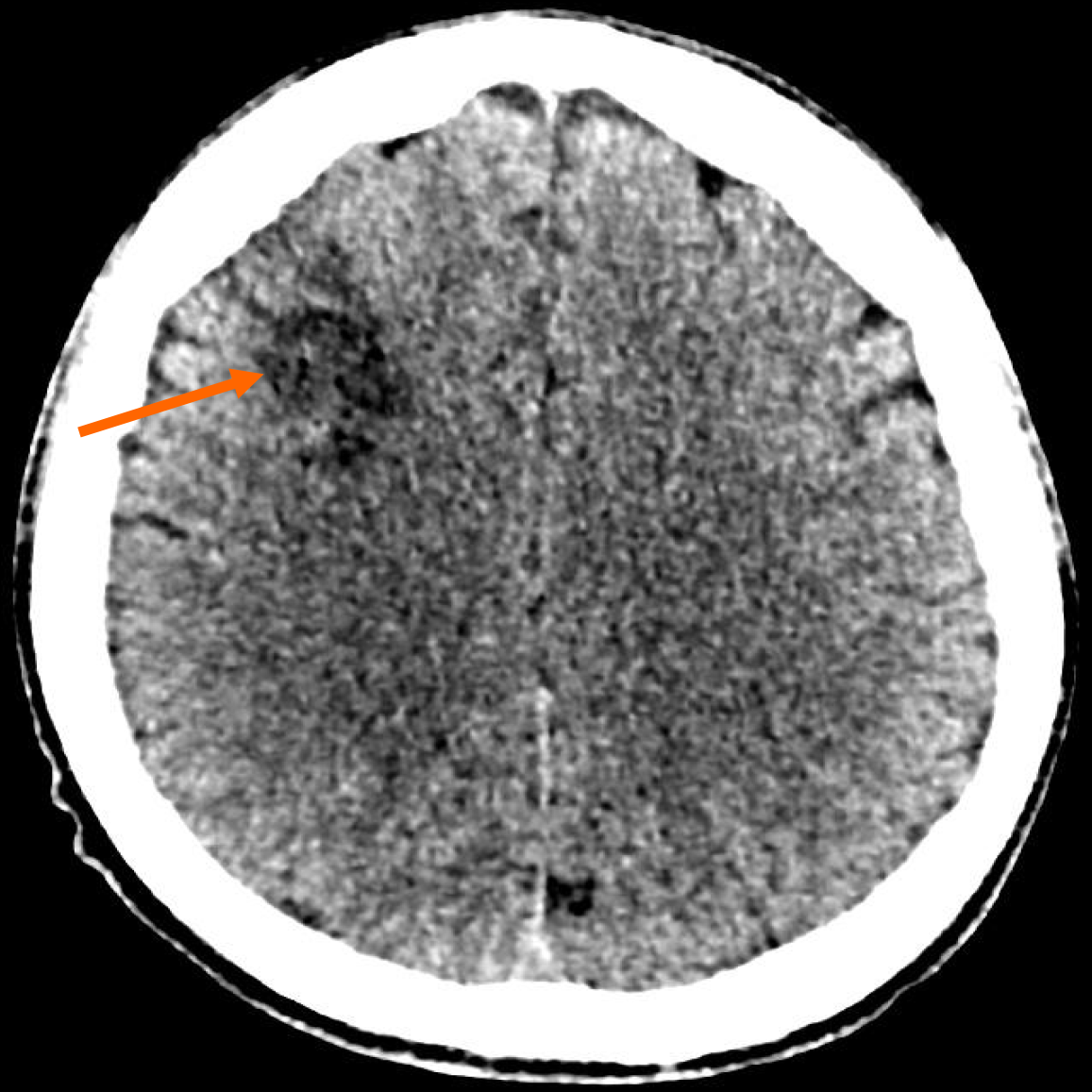
Plain computed tomography (CT) of the brain was done as a first-line radiological investigation. It revealed a well-defined hypodensity at the right frontoparietal lobe, suggestive of a chronic watershed region infarct (Figure 4).
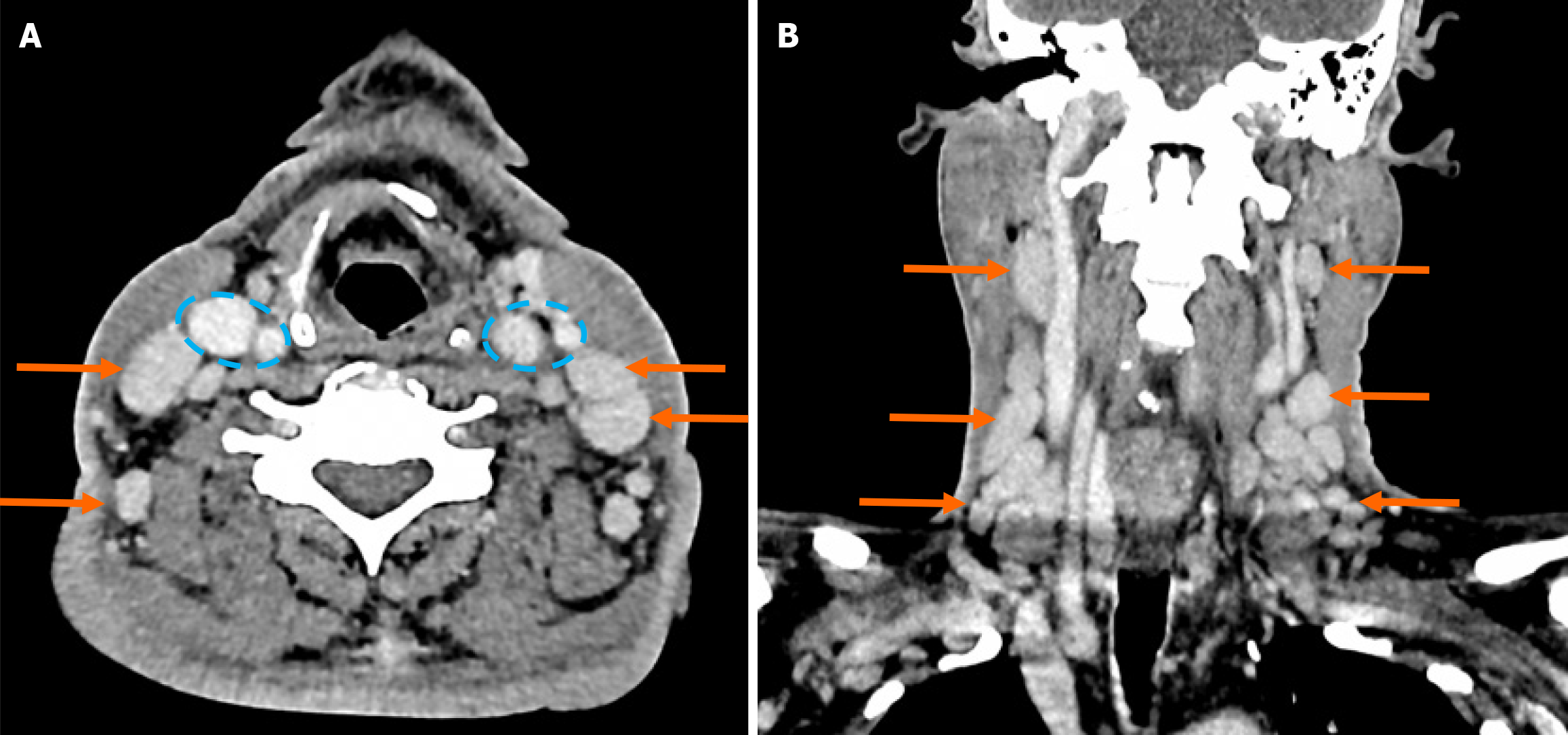
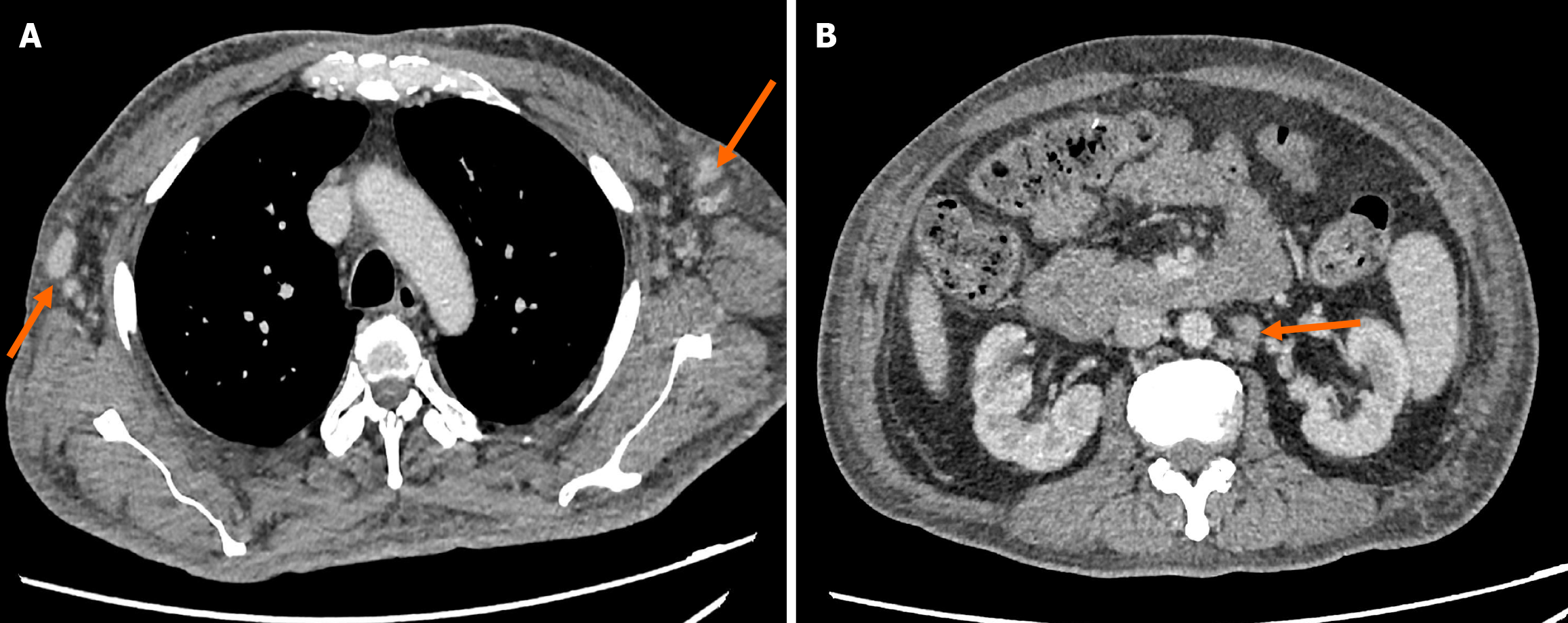
Initial ultrasound of the abdomen showed grade 2 fatty liver with hepatosplenomegaly. The patient further underwent a contrast-enhanced CT (CECT) neck, thorax, abdomen and pelvis, which demonstrated multiple enlarged lymph nodes in the cervical, supraclavicular, axillary, paraaortic and bilateral inguinal regions (Figures 5 and 6). Some of these lymph nodes appeared matted, but without a necrotic center within. An esophagogastroduodenoscopy and colonoscopy showed pangastritis and gastropathy with low-grade tubular adenoma in the colon.
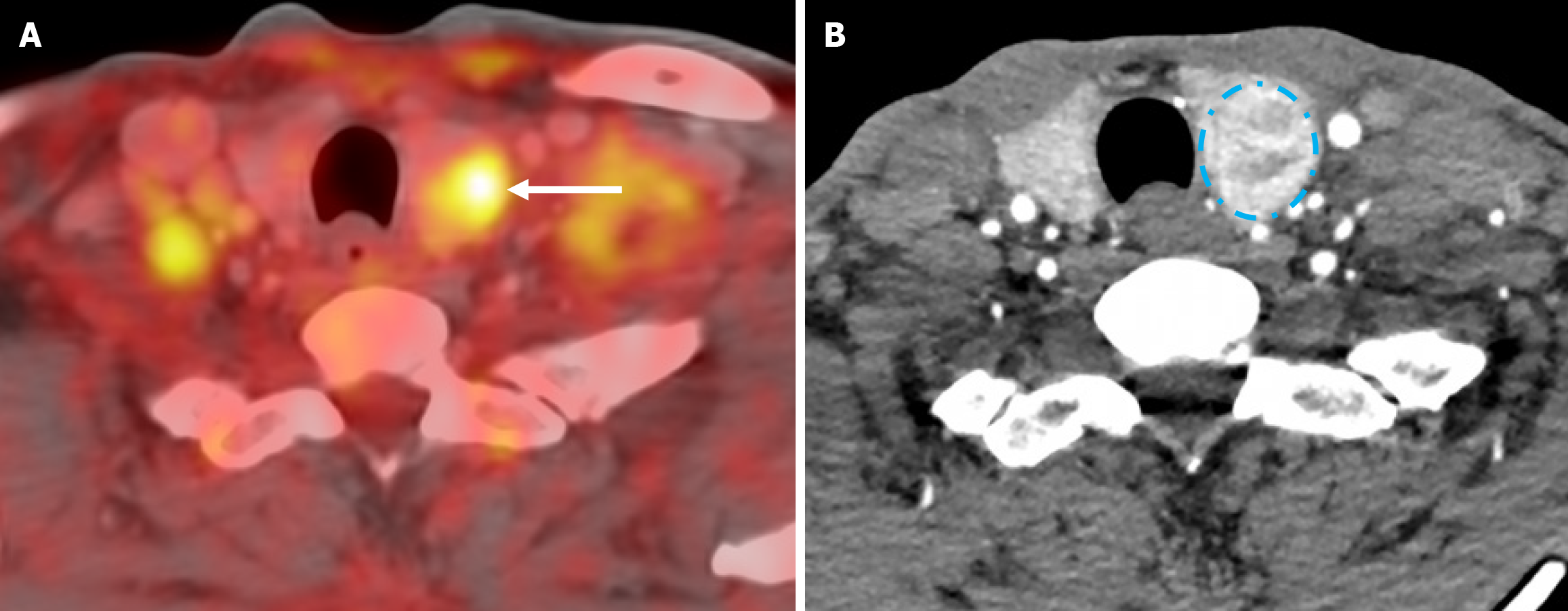
Subsequent positron emission tomography (PET) scan with CT (PET CT) done within 2 weeks from the previous CECT reveals stable multiple supra- and infra-diaphragmatic lymphadenopathies with background metabolism. However, there was a hypermetabolic left thyroid nodule seen, which corresponded to the heterogeneously enhancing left thyroid nodule in CT as depicted in Figure 7. There was also mildly hypermetabolic bone marrow in PET CT without an 18F-fluorodeoxyglucose (FDG)-avid bone lesion, which was of indeterminate clinical significance.
Serial nerve conduction studies demonstrated absent sensory response over bilateral radial and sural nerves, while other tested nerves showed prolonged distal sensory latency with reduced sensory nerve action potential, illustrated in Table 1. The motor response over the left radial and bilateral peroneal was absent. At the same time, the other tested nerves had significantly prolonged distal motor latency and reduced compound muscle action potential with conduction blocks over several nerves, as listed in Table 2, suggesting a generalized sensorimotor demyelinating polyneuropathy with conduction block.
| Sensory NCS | ||||||
| Nerve/sites | Rec. site | Latency, ms | Amplitude, µV | Segments | Distance, cm | Vel., m/s |
| Left median - digit II (antidromic) | ||||||
| Wrist | Index | 5.311 | 2.71 | Wrist - index | 14 | 34.9 |
| Right median - digit II (antidromic) | ||||||
| Wrist | Index | 5.421 | 5.61 | Wrist - index | 15 | 35.1 |
| Left ulnar - digit V (antidromic) | ||||||
| Wrist | Dig V | 4.641 | 4.41 | Wrist - dig V | 12 | 33.9 |
| Right ulnar - digit v (antidromic) | ||||||
| Wrist | Dig V | 5.101 | 4.01 | Wrist - dig V | 12 | 31.6 |
| Left radial - superficial (antidromic) | ||||||
| Forearm | Wrist | NR1 | NR1 | Forearm - wrist | 10 | NR |
| Right radial - superficial (antidromic) | ||||||
| Forearm | Wrist | NR1 | NR1 | Forearm - wrist | 10 | NR |
| Left sural - (antidromic) | ||||||
| Calf | Ankle | NR1 | NR1 | Calf - ankle | 14 | NR |
| Right sural - (antidromic) | ||||||
| Calf | Ankle | NR1 | NR1 | Calf - ankle | 14 | NR |
| Motor NCS | |||||||
| Nerve/sites | Muscle | Latency, ms | Amplitude, mV | Segments | Distance, cm | Velocity, m/s | Dur., ms |
| Left median – abductor pollicis brevis (APB) | |||||||
| Wrist | APB | 5.731 | 4.01 | Wrist - APB | 8 | 8.81 | |
| Elbow | APB | 16.06 | 1.8 | Elbow - Wrist | 24 | 23 | 10.52 |
| Right median - abductor pollicis brevis (APB) | |||||||
| Wrist | APB | 5.271 | 5.7 | Wrist - APB | 8 | 8.96 | |
| Elbow | APB | 15.19 | 2.7 | Elbow - Wrist | 22 | 22 | 10.92 |
| Left ulnar – abductor digiti minimi (ADM) | |||||||
| Wrist | ADM | 4.481 | 4.51 | Wrist - ADM | 8 | 10.52 | |
| B. elbow | ADM | 24.27 | 0.31 | B. elbow - wrist | 22 | 11 | 18.00 |
| A. elbow | ADM | 32.88 | 0.51 | A. elbow - B. elbow | 14 | 16 | 15.50 |
| Right ulnar – abductor digiti minimi (ADM) | |||||||
| Wrist | ADM | 4.501 | 3.91 | Wrist - ADM | 8 | 13.04 | |
| B. elbow | ADM | 35.27 | 0.41 | B. elbow - wrist | 20 | 6 | 21.23 |
| A. elbow | ADM | 46.10 | 0.21 | A. elbow - B. elbow | 14 | 13 | 20.42 |
| Left peroneal - extensor digitorum brevis (EDB) | |||||||
| Ankle | EDB | NR1 | NR1 | Ankle - EDB | 8 | NR | |
| Right peroneal - extensor digitorum brevis (EDB) | |||||||
| B. fib head | EDB | NR1 | NR1 | B. fib head - ankle | NR | ||
| Left tibial – abductor hallucis (AH) | |||||||
| Ankle | AH | 8.311 | 1.81 | Ankle - AH | 8 | 7.15 | |
| Knee | AH | 26.15 | 0.91 | Knee - ankle | 40 | 22 | 8.46 |
| Right tibial – abductor hallucis (AH) | |||||||
| Ankle | AH | 7.401 | 1.51 | Ankle - AH | 8 | 6.65 | |
| Knee | AH | 26.29 | 0.31 | Knee - ankle | 37 | 20 | 8.56 |
| Left peroneal - tibialis anterior (TA) | |||||||
| Fib head | Tib Ant | 4.33 | 2.41 | Fib head - tib ant | 14.77 | ||
| Pop fossa | Tib Ant | 6.58 | 2.2 | Pop fossa - fib head | 10 | 44 | |
| Right peroneal - tibialis anterior (TA) | |||||||
| Fib head | Tib Ant | 6.191 | 2.31 | Fib head - tib ant | |||
| Pop fossa | Tib ant | 8.38 | 2.0 | Pop fossa - fib head | 6 | 27 | 14.71 |
| Left radial – extensor indicis propius (EIP) | |||||||
| Forearm | EIP | NR1 | NR1 | Forearm - EIP | NR | ||
| Elbow | EIP | NR1 | NR1 | Elbow - forearm | 14 | NR | NR |
| Spiral Gr | EIP | NR1 | NR1 | Spiral Gr - elbow | 13 | NR | NR |
| Right radial – extensor indicis propius (EIP) | |||||||
| Forearm | EIP | 3.041 | 3.5 | Forearm - EIP | 9.44 | ||
| Elbow | EIP | 6.00 | 2.2 | Elbow - forearm | 10 | 34 | 9.06 |
| Spiral Gr | EIP | 8.69 | 2.7 | Spiral Gr - elbow | 10 | 37 | 9.52 |
A diagnosis of Castleman disease variant of POEMS syndrome, without detectable M-proteins, was made based on the clinical and investigation findings.
He received melphalan plus dexamethasone, followed by three cycles of cyclophosphamide with dexamethasone while awaiting the availability of lenalidomide. Bortezomib was not administered, considering the existing severe neuropathy. Given that the serum testosterone level was low, testosterone replacement was prescribed monthly. The long-term plan was to proceed with an autologous stem cell transplant (ASCT) once adequate control of the disease was attained.
His treatment was complicated with a new-onset post-infarct seizure requiring antiseizure medication, as well as a left upper limb swelling due to a long-segment basilic vein thrombosis, necessitating anticoagulation. During the ensuing 5 months post-chemotherapy, the patient generally improved, most notably a reduction in skin pigmentation, improved muscle power and resolving fluid retention. Considering his clinical improvement and the cost of tests that require outsourcing (VEGF), we did not arrange for a follow-up study.
Unfortunately, before the planning phase for ASCT, he was admitted with severe sepsis, which was further com
Our patient was admitted with neuropathy with the onset of symptoms dating several months back. Similar to reported literature, he experienced cramps and spasms in his extremities before they progressed proximally[8]. Subsequently, patients develop hyperesthesia and dysesthesia with weakness, experiencing difficulty climbing stairs and reduced grip strength[8,9]. The clinical course of the disease spans from days to weeks or weeks to months[8].
CIDP: CIDP is similar in many aspects, characterized by immune-mediated, motor and sensory, chronic and progressive neuropathy with polyradicular features (distal and proximal involvement) and may be misdiagnosed in 60% of POEMS syndrome cases[10,11]. Several clinical features in favor of Castleman disease variant of POEMS syndrome are the painful and severe neuropathy experienced, with a greater degree of sensorimotor impairment, as the unmyelinated C fibers are commonly well-preserved as opposed to those with CIDP, although this is arbitrary[10,12]. Cranial nerve involvement and dysautonomia are also far less common in patients with this variant of POEMS syndrome. Non-invasively, nerve conduction studies of patients with POEMS syndrome demonstrate conduction slowing predominantly in the in
Multiple myeloma: Castleman Disease variant of POEMS syndrome is associated with underlying plasma cell disorders. Differential diagnosis should include multiple myeloma, which may present with neuropathy as well. Distinguishing between these pathologies is essential as treatment and prognosis differ[4]. Multiple myeloma tends to present with bone pain, anemia, hypercalcemia, extensive infiltration of bone marrow by plasma cells, or renal failure, which is uncommon in the Castleman disease variant of POEMS[14]. Furthermore, imaging would demonstrate the presence of lytic bone lesions instead of sclerotic lesions and without marked elevation in VEGF levels, as seen in POEMS[4].
Paraneoplastic syndrome: Castleman disease variant of POEMS syndrome can closely mimic or coexist with paraneoplastic syndromes, making accurate differentiation essential. Paraneoplastic syndromes present as secondary manifestations of an underlying malignancy mediated by immune mechanisms or ectopic hormone or cytokine production. In contrast, Castleman disease variant of POEMS syndrome represents an underlying plasma cell dyscrasia, with disease biology driven predominantly by VEGF, although inflammatory cytokines may also be elevated[15].
In this context, a diagnosis of Castleman disease variant of POEMS syndrome is favored when imaging demonstrates diffuse, fluorodeoxyglucose-avid lymphadenopathy without evidence of a discrete occult primary tumor, which would otherwise support a paraneoplastic etiology.
Immunoglobulin G4-related disease: Immunoglobulin G4 (IgG4)-related disease (IgG4-RD) is another important diagnostic consideration, as it may present with lymphadenopathy and systemic symptoms that overlap with those of Castleman Disease variant of POEMS syndrome. Demographically, IgG4-RD tends to affect older patients, with a mean age in the sixth decade, whereas Castleman disease variant of POEMS syndrome commonly presents earlier, in the fourth to fifth decades of life[15,16]. In contrast to the systemic inflammatory manifestation in Castleman disease variant of POEMS syndrome, IgG4-RD characteristically forms localized inflammatory “pseudotumor” involving organs such as the pancreas, salivary glands, lacrimal glands, and kidneys, and is often associated with atopic or allergic conditions[16]. Castleman disease typically exhibits markedly elevated inflammatory markers, including C-reactive protein, erythrocyte sedimentation rate, and serum IL-6 levels, whereas these markers are comparatively lower in IgG4-RD[15]. Therapeutically, IgG4-RD often responds well to corticosteroid monotherapy, whereas Castleman disease variant of POEMS syndrome generally requires treatment directed at plasma cell disorders to achieve disease control[15].
Amyloid light chain or primary amyloidosis: Amyloid light chain or primary amyloidosis (AL amyloidosis) and Castleman disease variant of POEMS syndrome represent distinct but occasionally overlapping conditions. AL amy
Our patient, however, did not fulfil the mandatory criteria of monoclonal plasma cell disorder, demonstrating a polyclonal gammopathy. This is despite having polyneuropathy and fulfilling 3 major criteria (polyneuropathy, Castleman disease and VEGF elevation) and 6 minor criteria (organomegaly, extravascular volume overload, endo
Although the presence of a monoclonal plasma cell disorder is a mandatory criterion for the diagnosis of POEMS syndrome, rare cases have been reported in the literature where patients exhibit typical clinical and laboratory features (polyneuropathy, elevated VEGF, multiple major and minor criteria) but lack a demonstrable monoclonal gammopathy[18]. There were recent cases of M-protein-negative variants that demonstrated successful management with lenali
In the Castleman disease variant of POEMS, VEGF links the plasma cell disorder and its diverse manifestations. VEGF overproduction represents the dominant pathogenic mediator rather than an interleukin-6–driven cytokine storm. Pathophysiologically, VEGF acts as a potent pro-angiogenic factor that increases vascular permeability, directly leading to extravascular fluid overload, as evidenced by edema, ascites, and organomegaly. Furthermore, it causes endoneural edema, leading to polyneuropathy[4]. As this variant lacks a detectable M-protein, elevated serum VEGF remains the most reliable biomarker for confirming the diagnosis and monitoring the efficacy of treatment.
There’s currently no standard therapy for the Castleman disease variant of POEMS syndrome[20]. The principle of treatment is based on the degree of plasma cell infiltration[5,9,22]. Targeted radiotherapy is favored for isolated sclerotic bone lesions (up to 3) without marrow involvement, and is associated with a 10-year survival rate of 70%[5,9,22]. Extensive osteoporotic lesions or bone marrow involvement would require systemic chemotherapy. High-dose melphalan in combination with ASCT, and lenalidomide with dexamethasone are examples of effective systemic regimes. Two prospective studies of lenalidomide with dexamethasone achieved a remission rate of over 70% and a 3-year progression-free survival rate of 60%-75%[23,24]. Other treatment regime may include thalidomide and bortezomib, which are agents with anti-VEGF and anti-TNF effects[4,22].
Supportive therapy such as physical therapy, continuous positive airway pressure, analgesics for neuropathic pain, psychological support as well as orthotics, positively influence overall long-term outcomes and wellbeing[4,22]. Endocrinopathies may require hormone replacement therapy, with thyroxine or steroids, as in our case, to address hypothyroidism and adrenal insufficiency. Subsequent follow-up should include an echocardiogram and diffusing capacity for carbon monoxide every six months for those receiving systemic chemotherapy to monitor its side effects. PET CTs could be used to assess for relapses based on availability of facility[4,22].
Castleman disease variant of POEMS syndrome leads to a significant deterioration in quality of life through neuropathy, fluid retention, thromboembolic events and cachexia. Patients frequently succumb to the illness due to respiratory and circulatory failure, progressive malnutrition, infection and kidney failure. Good prognostic factors include albumin > 3.2 g/dL, attaining complete hematological response, and a younger onset of disease[4,22]. Poor prognostic factors include age greater than 50 years, presence of pleural effusion, severe renal impairment and pulmonary hypertension[4,22].
Castleman disease variant of POEMS syndrome remains a diagnostic and therapeutic challenge. We highlight this case to raise awareness of prompt full diagnostic work-up and a multidisciplinary approach in order to reduce long-term irreversible morbidity and mortality.
The authors would like to take this opportunity to extend our thanks to the patient and his caregivers for granting us permission to publish his case for the benefit of the medical community.
| 1. | Suichi T, Misawa S, Beppu M, Takahashi S, Sekiguchi Y, Shibuya K, Amino H, Tsuneyama A, Suzuki YI, Nakamura K, Sato Y, Kuwabara S. Prevalence, clinical profiles, and prognosis of POEMS syndrome in Japanese nationwide survey. Neurology. 2019;93:e975-e983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 52] [Article Influence: 7.4] [Reference Citation Analysis (1)] |
| 2. | Livingston J, Cobiella C, Hall-Craggs MA. POEMS syndrome - a unique presentation of a rare paraneoplastic syndrome. BMJ Case Rep. 2010;2010:bcr0920103324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 8] [Article Influence: 0.5] [Reference Citation Analysis (1)] |
| 3. | Mangalik A, Veliath AJ. Osteosclerotic myeloma and peripheral neuropathy. A case report. Cancer. 1971;28:1040-1045. [PubMed] [DOI] [Full Text] |
| 4. | Dispenzieri A. POEMS syndrome: 2021 Update on diagnosis, risk-stratification, and management. Am J Hematol. 2021;96:872-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 90] [Article Influence: 18.0] [Reference Citation Analysis (1)] |
| 5. | Zhou GX, Xiao L, Bi YM, Yang F, Yang CZ, Zhang D. Case report: POEMS syndrome masquerades as diabetic foot. Front Neurol. 2023;14:1336382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 6. | Wang Y, Huang LB, Shi YH, Fu H, Xu Z, Zheng GQ, Wang Y. Characteristics of 1946 Cases of POEMS Syndrome in Chinese Subjects: A Literature-Based Study. Front Immunol. 2019;10:1428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 7. | Yu H, Yao F, Li Y, Li J, Cui QC. Castleman disease variant of POEMS syndrome complicated with multiple cerebral infarction: a rare case report and review of literature. Int J Clin Exp Pathol. 2015;8:13578-13583. [PubMed] |
| 8. | D'Sa S, Khwaja J, Keddie S, Keh RY, Smyth D, Ronneberger R, Dubash S, Sivabalasingham S, Wan S, Hoskote C, Baldeweg S, Sive J, Lunn MP. Comprehensive Diagnosis and Management of POEMS Syndrome. Hemasphere. 2022;6:e796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 20] [Reference Citation Analysis (1)] |
| 9. | Wang W, Yang G. POEMS syndrome presents with a distended abdomen: A case report. J Int Med Res. 2023;51:3000605221148410. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 10. | Piccione EA, Engelstad J, Dyck PJ, Mauermann ML, Dispenzieri A, Dyck PJ. Nerve pathologic features differentiate POEMS syndrome from CIDP. Acta Neuropathol Commun. 2016;4:116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 39] [Article Influence: 3.9] [Reference Citation Analysis (1)] |
| 11. | Nasu S, Misawa S, Sekiguchi Y, Shibuya K, Kanai K, Fujimaki Y, Ohmori S, Mitsuma S, Koga S, Kuwabara S. Different neurological and physiological profiles in POEMS syndrome and chronic inflammatory demyelinating polyneuropathy. J Neurol Neurosurg Psychiatry. 2012;83:476-479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 128] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 12. | Koike H, Iijima M, Mori K, Yamamoto M, Hattori N, Watanabe H, Tanaka F, Doyu M, Sobue G. Neuropathic pain correlates with myelinated fibre loss and cytokine profile in POEMS syndrome. J Neurol Neurosurg Psychiatry. 2008;79:1171-1179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 72] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 13. | Sung JY, Kuwabara S, Ogawara K, Kanai K, Hattori T. Patterns of nerve conduction abnormalities in POEMS syndrome. Muscle Nerve. 2002;26:189-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 80] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 14. | Nyunt WWT, Remli R, Abdul Muttlib FA, Leong CF, Masir N, Tumian NR, Abdul Wahid SFS. POEMS syndrome. Malays J Pathol. 2017;39:297-303. [PubMed] |
| 15. | Fajgenbaum DC, Uldrick TS, Bagg A, Frank D, Wu D, Srkalovic G, Simpson D, Liu AY, Menke D, Chandrakasan S, Lechowicz MJ, Wong RS, Pierson S, Paessler M, Rossi JF, Ide M, Ruth J, Croglio M, Suarez A, Krymskaya V, Chadburn A, Colleoni G, Nasta S, Jayanthan R, Nabel CS, Casper C, Dispenzieri A, Fosså A, Kelleher D, Kurzrock R, Voorhees P, Dogan A, Yoshizaki K, van Rhee F, Oksenhendler E, Jaffe ES, Elenitoba-Johnson KS, Lim MS. International, evidence-based consensus diagnostic criteria for HHV-8-negative/idiopathic multicentric Castleman disease. Blood. 2017;129:1646-1657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 527] [Cited by in RCA: 474] [Article Influence: 52.7] [Reference Citation Analysis (1)] |
| 16. | Sasaki T, Akiyama M, Kaneko Y, Mori T, Yasuoka H, Suzuki K, Yamaoka K, Okamoto S, Takeuchi T. Distinct features distinguishing IgG4-related disease from multicentric Castleman's disease. RMD Open. 2017;3:e000432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 41] [Article Influence: 4.6] [Reference Citation Analysis (1)] |
| 17. | Merlini G, Dispenzieri A, Sanchorawala V, Schönland SO, Palladini G, Hawkins PN, Gertz MA. Systemic immunoglobulin light chain amyloidosis. Nat Rev Dis Primers. 2018;4:38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 407] [Article Influence: 50.9] [Reference Citation Analysis (2)] |
| 18. | Ding C, Li Y. A case of paraprotein-negative POEMS syndrome: Case report and literature review. Medicine (Baltimore). 2024;103:e39267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 19. | Du HP, Zhang Y, Sun LM, Luo L. Two cases of special POEMS syndrome without monoclonal protein expression: a case report and literature review. J Int Med Res. 2021;49:300060521990967. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 20. | Ji M, Jin S, Zang S, Li P, Lu F, Zhao C, Zhang C, Ji C, Ye J. Castleman disease variant of POEMS syndrome without M protein: a case report. Front Oncol. 2024;14:1449945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 21. | Nomura T, Iwata I, Eguchi K, Fujii S, Inoue T, Tarisawa M, Ishio T, Toyama Y, Uwatoko H, Shirai S, Matsushima M, Yaguchi H, Yabe I. Requirement of Repeated Serum VEGF Measurements in POEMS Syndrome. Intern Med. 2025;64:769-773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 22. | Kanuganti D, Nagarjunakonda VS, Bandarupalli P, Gorijala VK, Konagalla VLSR, Kowtha P. POEMS Syndrome: A Case Report and Review of the Literature. Cureus. 2022;14:e27001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 23. | Nozza A, Terenghi F, Gallia F, Adami F, Briani C, Merlini G, Giordano L, Santoro A, Nobile-Orazio E. Lenalidomide and dexamethasone in patients with POEMS syndrome: results of a prospective, open-label trial. Br J Haematol. 2017;179:748-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (1)] |
| 24. | Li J, Huang XF, Cai QQ, Wang C, Cai H, Zhao H, Zhang L, Cao XX, Gale RP, Zhou DB. A prospective phase II study of low dose lenalidomide plus dexamethasone in patients with newly diagnosed polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, and skin changes (POEMS) syndrome. Am J Hematol. 2018;93:803-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 56] [Article Influence: 7.0] [Reference Citation Analysis (1)] |