Published online Mar 16, 2026. doi: 10.12998/wjcc.v14.i8.118582
Revised: January 21, 2026
Accepted: February 4, 2026
Published online: March 16, 2026
Processing time: 69 Days and 18.6 Hours
Propofol is widely considered for procedural sedation and analgesia across medical facilities. It is associated with dose-dependent hypotension, respiratory depression, and lack of analgesia. These limitations can be managed by combining propofol with N-methyl-D-aspartate (NMDA) receptor antagonists (ketami
To investigate the safety and efficacy of combining NMDA receptor antagonists (ketamine or esketamine) with propofol compared to propofol monotherapy for procedural sedation.
Randomized controlled trials comparing NMDA antagonist-propofol combinations with propofol mo
Nineteen randomized controlled trials (n = 8400+) were included, from which 13 assessed ketamine-propofol, while 6 assessed esketamine-propofol. Combination therapy significantly reduced hypotension (RR = 0.40; 95%CI: 0.26-0.60) and the need for respiratory interventions (RR = 0.73; 95%CI: 0.58-0.93), whereas hallucinations were significantly more frequent in the intervention cohort (RR = 2.24; 95%CI: 1.65-3.03). No significant differences were observed for desaturation, apnea, recovery time, systolic/diastolic blood pressure, mean arterial pressure, or heart rate. Patient satisfaction was comparable across groups. Sensitivity and subgroup analyses confirmed the strength of the key findings, and meta-regression revealed an increased risk of hypotension with higher propofol dosing, which was reduced with higher ketamine dosing.
NMDA antagonist-propofol regimen improves hemodynamic safety and reduces respiratory interventions in procedural sedation and analgesia compared to propofol, but is associated with a higher risk of hallucinations. These findings support the selective use of this approach, with dosing strategies justifying further modifications.
Core Tip: This meta-analysis of 19 randomized controlled trials evaluates the safety and efficacy of combining N-methyl-D-aspartate receptor antagonists, specifically ketamine and esketamine, with propofol for procedural sedation. Our pooled analysis demonstrates that this combination significantly reduces the risk of hypotension and the need for respiratory interventions compared to propofol monotherapy. However, clinicians must remain vigilant regarding the increased risk of transient hallucinations. These findings support the use of Ketofol and esketamine-propofol as valuable, hemodynamically stable alternatives for sedation in diverse clinical settings.
- Citation: Faheem MSB, Hassan ST, Feroze F, Khan A, Munir SU, Khaliq A, Fatima ST, Mendonca R, Surani S. Combination of N-methyl-D-aspartate receptor antagonists and propofol for procedural sedation. World J Clin Cases 2026; 14(8): 118582
- URL: https://www.wjgnet.com/2307-8960/full/v14/i8/118582.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i8.118582
Procedural sedation and analgesia (PSA) is a process of administering sedative and analgesic medications to reduce anxiety and provide comfort to patients undergoing various medical procedures. It is extensively used in emergency departments, endoscopic units, and minor surgical settings, contributing to successful procedural outcomes. More than 20 million invasive procedures are performed annually across the pediatric and adult populations in the United States, requiring moderate or deep sedation[1]. The primary aim of this process is to achieve optimal sedation, anxiolysis, amnesia, and analgesia without compromising hemodynamic or respiratory stability[2-5]. Propofol is a gamma-ami
Ketamine and esketamine are N-methyl-d-aspartate (NMDA) receptor antagonists that belong to the class of dis
Due to inconsistent PSA practices across settings and a lack of guidelines conclusively recommending one sedation strategy over another, it is necessary to to assess the efficacy and safety of NMDA antagonists combined with propofol. Therefore, we conducted a steroid-responsive meningitis-arteritis (SRMA) of randomized controlled trials (RCTs) comparing NMDA antagonist (ketamine or esketamine)-propofol combinations with propofol monotherapy. We also evaluated key outcomes such as sedation quality, respiratory and cardiovascular adverse events, recovery characteristics, and patient/provider satisfaction across diverse procedural settings.
This SRMA has been conducted in accordance with the Preferred Reporting Items for SRMA (PRISMA) guidelines, which is an international database designed for the prospective registration of systematic review protocols to ensure trans
In patients undergoing PSA (P), does the combination of an NMDA receptor antagonist (ketamine or esketamine) with propofol (I), compared with propofol monotherapy (C), improve outcomes such as hemodynamic stability, respiratory safety, procedural success, and recovery quality (O)?
Our systematic review included RCTs published in English that enrolled patients undergoing PSA and compared the safety and efficacy of the combination therapy of an NMDA receptor antagonist (ketamine or esketamine) and propofol with propofol monotherapy in PSA. The review encompassed studies involving both adult and pediatric patient populations. We excluded studies classified as non-original (review articles, editorials, case reports, abstracts, conference proceedings, grey literature), preclinical studies including animal models or in vitro research, those that were directly comparing ketamine or esketamine or any two NMDA antagonists, and lack a direct comparison of NMDA antagonist-propofol with propofol monotherapy, or studies with incomplete or missing outcome data and single arm RCTs.
Databases such as PubMed (MEDLINE), EMBASE, and clinicaltrials.gov were screened thoroughly by the author Hassan ST according to the inclusion and exclusion criteria to search for studies comparing the safety and efficacy of NMDA antagonist-propofol combined therapy with propofol monotherapy in PSA. A literature search was conducted using keywords and Medical Subject Heading terms related to procedural sedation, analgesia, ketamine, esketamine, NMDA receptor antagonists, and propofol, covering articles published from inception until June 2025. Further, a detailed PubMed search strategy is provided in the supplementary information (Supplementary Table 1).
The author (Faheem MSB) reviewed the abstract and title of all references that met the inclusion and exclusion criteria and assessed the full text to determine the eligibility of the included studies; discrepancies were resolved by a second author (Surani S).
Two reviewers (Hassan ST and Fatima ST) independently extracted and incorporated the data from the included RCTs into an Excel sheet covering information such as study ID, first author, year of publication, design, sample size, baseline characteristics, patient demographics, type of procedure, sedation regimen (ketamine/esketamine-propofol vs propofol alone), safety and efficacy outcomes while any disagreements were addressed by a third reviewer (Faheem MSB). Furthermore, the authors Hassan ST and Fatima ST utilized the Cochrane Risk of Bias (RoB) tool (RoB 2.0), which assesses five domains such as the randomization process, deviation from intended intervention, missing data, outcome mea
The primary endpoints of this study were respiratory safety events, the need for rescue interventions, procedural conditions, anesthetic consumption, and recovery profile. Hemodynamic changes, patient-centered measures, recovery parameters, and adverse effects were pooled as secondary outcomes. These outcomes were assessed in both the NMDA antagonist-propofol and propofol monotherapy groups by pooling risk ratios (RRs) with corresponding 95% confidence intervals (CIs). The Mantel-Haenszel method was applied using a random-effects model to account for interstudy heterogeneity, which was assessed using I2 statistics indicating low heterogeneity (< 50%), moderate heterogeneity (50%-75%), and substantial heterogeneity (> 75%). Sensitivity analyses (leave-one-out [LOO] method), predefined subgroup analyses, and meta-regression were performed to identify the influence of potential sources, such as drug dosage and patient age, on heterogeneity. Funnel plots were designed for the visual inspection of publication bias. All statistical computations of this study were performed and reviewed by the author using the RStudio tool[16], and forest plots were employed for the visualization of the data.
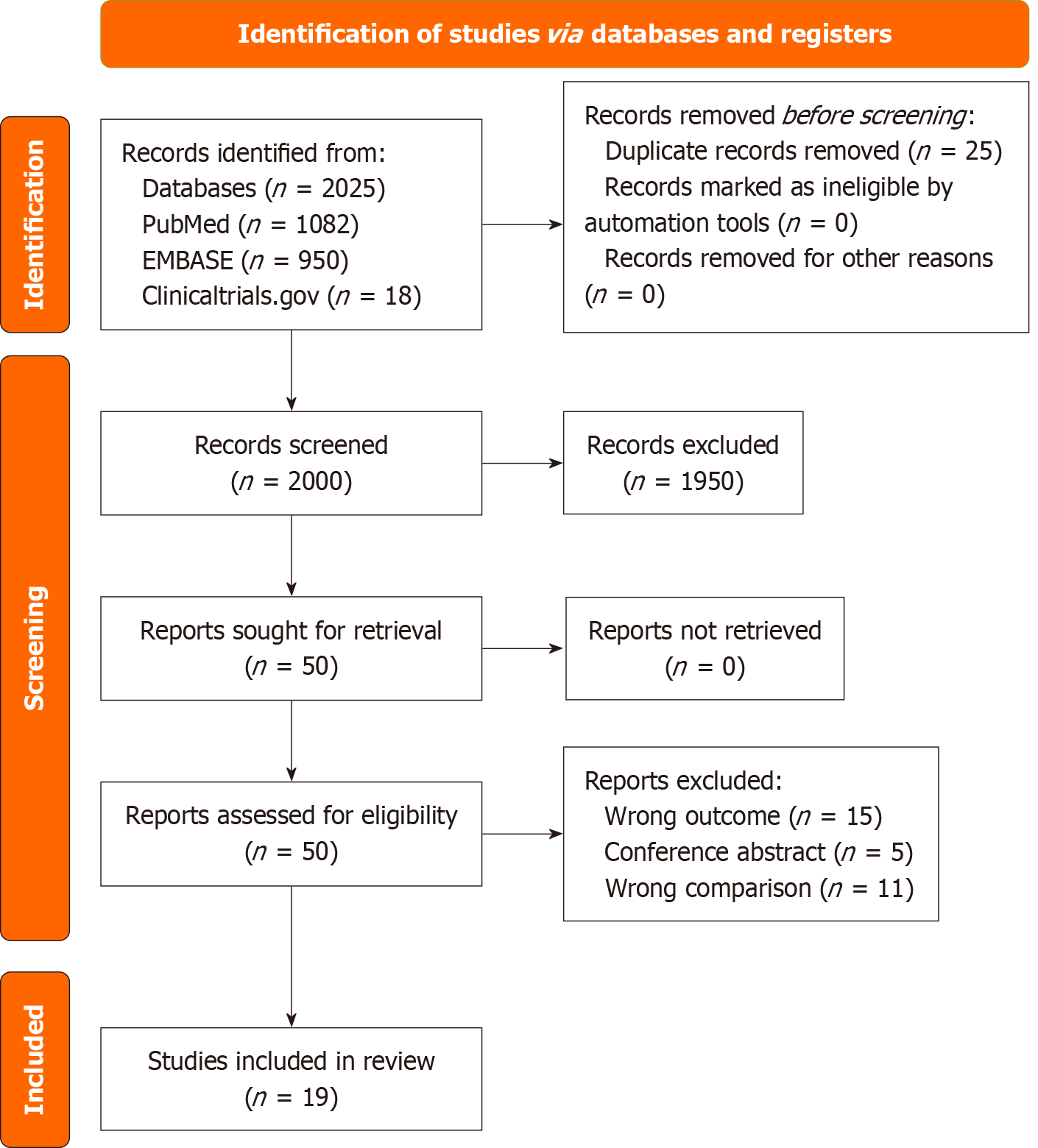
Of the 19 RCTs included, as shown in Figure 1, the sample sizes ranged from 40 to 573. Among the included studies, 13 trials utilized a ketamine-propofol combination, whereas 6 trials assessed esketamine-propofol regimens. The inclusion trials assessed the intervention vs control on respiratory, hemodynamic, recovery, and patient-centered outcomes (Figure 2A and B, Tables 1 and 2).
| Ref. | Study design | Sample size (n) | Age in years | Dose (mg/kg) | Number of males, n (%) | SBP, mmHg | DBP, mmHg | Oxygen saturation (%) | Heart rate, bpm | MAP, mmHg | |||||||||
| Control | Interv | Control | Interv | Control | Interv | Control | Interv | Control | Interv | Control | Interv | Control | Interv | Control | Interv | Control | Interv | ||
| Ferguson et al[17], 2016 | RCT | 292 | 281 | 46 ± 23.84 | 50 ± 25.3 | 1 mg/kg propofol | 1:1 ketofol = 0.25 | 145 ± 49 | 138 ± 49 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Miner et al[22], 2014 | RCT | 90 | 1:1 ketofol = 85; 4:1 ketofol = 96 | 40 ± 17.05 | 1:1 = 39 (17.8); 4:1 = 36 (16) | 1 mg/kg propofol | 1:1 ketofol = propofol - 0.5 mg/kg, ketamine - 0.5 mg/kg, 4:1 ketofol = propofol - 0.8 mg/kg, ketamine - 0.2 mg/kg | 47 ± 52 | 1:1= 45 ± 53; 4:1= 51 ± 53 | 127 ± 51.15 | 1:1 = 127 ± 17.79; 4:1 = 129 ± 14.83 | NR | NR | 100 | 1:1 = 100; 4:1 = 100 | NR | NR | NR | NR |
| Ulutas et al[32], 2023 | RCT | 34 | 30 | 53.65 ± 14.57 | 46.27 ± 18.72 | 1 mg/kg propofol | 1:1 ketofol = 0.5 mg/kg ketamine + 0.5 mg/kg propofol | 25 ± 73.5 | 18 ± 60 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Soliman et al[25], 2017 | RCT | 30 | 30 | 14.48 ± 1.26 | 14.09 ± 1.32 | A bolus dose of propofol (1-2 mg/kg) and then a contin | 1:1 ketofol = 1 mg/kg as a bolus dose and continu | 16 ± 53.53 | 13 ± 43.33 | NR | NR | NR | NR | NR | NR | 79.9 ± 7.5 | 79.0 ± 6.9 | 85.3 ± 6.9 | 85.1 ± 6.6 |
| Kumar et al[26], 2020 | RCT | NR | NR | NR | NR | 1.5-2.5 mg/kg propofol | IV ketamine 1 mg/kg followed by propofol 1-2 mg/kg | NR | NR | NR | NR | NR | NR | NR | NR | 84.7 ± 11.9 | 81.1 ± 9.7 | 95.6 ± 11.8 | 94.6 ± 9.1 |
| Andol | RCT | 142 | 142 | 54 ± 24.47 | 48 ± 30.39 | 0.75 mg/kg of propofol | 1:1 ketofol = 0.188 mg/ kg each of ketamine and propofol | 69 ± 49 | 71 ± 50 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Tandon et al[24], 2014 | RCT | 135 | 135 | 47 ± 13 | 49 ± 12 | Propofol | Ketamine and propofol | 104 ± 77.03 | 100 ± 74.04 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Khalil et al[35], 2021 | RCT | 53 | 53 | 26.49 ± 3.06 | 27.68 ± 3.67 | Propofol (2 mg/kg) | Propofol (1 mg/kg) plus ketamine (0.5 mg/kg) | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Frey et al[23], 1999 | RCT | 33 | 33 | NR | NR | Propofol | 3.3:1 ketofol | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Cui et al[27], 2023 | RCT | 67 | 68 | 65.6 ± 10.2 | 62.9 ± 9.6 | Propofol | Esketam | 42 ± 62.7 | 34 ± 50 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Liu et al[28], 2025 | RCT | 126 | 126 | 65 ± 10.38 | 64.5 ± 11.12 | 1 mg/kg propofol | 1 mg/kg propofol and 0.2 | 61 ± 48.4 | 69 ± 54.8 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Zheng et al[29], 2023 | RCT | 52 | 52 | 41.1 ± 7.9 | 42.2 ± 9.3 | 2 mg/kg propofol | 2 mg/kg propofol and 0.25 | 34 ± 65.38 | 32 ± 61.53 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Fu et al[19], 2024 | RCT | 25 | 25 for all ratios of esketofol | 54.4 ± 2.7 | 5:1 esketofol = 49.88 ± 2.6; 3.3:1 esketofol = 48.9 ± 2.4; esketofol = 1.5:1 = 48.5 ± 3.0 | 2 mg/kg propofol | 5:1 = propofol 1 mg/kg with esketam | 10 ± 40 | 5:1 = 11 ± 27.5; 3.3:1 = 12 ± 48; 1.5:1 = 16 ± 64 | 129.12 ± 21.58 | 5:1 = 127.04 ± 23.43; 3.3:1 = 126.0 ± 19.36; 1.5:1 = 120.84 ± 20.11 | 78.52 ± 16.35 | 5:1 = 78.76 ± 13.58; 3.3:1 = 78.12 ± 13.41; 1.5:1 = 75.56 ± 17.64 | 99.76 ± 0.52 | 5:1 = 99.56 ± 1.04; 3.3:1 = 99.44 ± 1.08; 1.5:1 = 99.56 ± 1.50 | NR | NR | 95.00 ± 17.06 | 5:1 = 92.16 ± 14.14; 3.3:1 = 92.96 ± 14.12; 1.5:1 = 89.12 ± 17.28 |
| Beyoğlu et al[33], 2020 | RCT | 41 | 34 | 3.23 ± 1.62 | 3.33 ± 1.77 | 1 mg/kg propofol | Ketofol | 19 ± 46.34 | 21 ± 61.76 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Frizelle et al[21], 1997 | RCT | 20 | 20 | 59 ± 13 | 55 ± 17 | Bolus of 0.5 | Initial bolus of 0.4 mg/kg propofol and 0.1 mg/kg ketamine followed by continu | 16 ± 80 | 16 ± 80 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Bhardwaj et al[30], 2024 | RCT | 29 | 29 | 24 ± 13.34 | 30 ± 22.24 | Propofol 0.5 mg/kg bolus | 1 mg/kg of ketamine and 0.5 mg/kg propofol | 19 ± 65.51 | 17 ± 58.6 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Schmitz et al[31], 2018 | RCT | 167 | 169 | 3.67 ± 3.26 | 3.91 ± 3.11 | 1 mg/kg propofol | Propofol 0.5 mg/kg and ketamine 1 mg/kg | 89 ± 53.3 | 86 ± 52.4 | NR | NR | NR | NR | 98 ± 0.74 | 98 ± 1.48 | NR | NR | NR | NR |
| Chiaretti et al[20], 2011 | RCT | 62 | 59 | 7.3 ± 5.2 | 6.9 ± 5.4 | 2 mg/kg propofol | 2 mg/kg propofol and 0.5 mg/kg ketamine | 32 ± 51 | 27 ± 44 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Xu et al[43], 2024 | RCT | 42 | 42 | 4.1 ± 1.4 | 4.2 ± 1.1 | 10 mg/mL propofol | 10:1 esketofol (propofol and esketam | 17 ± 40.47 | 11 ± 26.19 | 108 ± 13 | 105 ± 11 | NR | NR | NR | NR | 105 ± 16 | 102 ± 16 | 66 ± 12 | 63 ± 12 |
| Ref. | Desaturation | Apnea | Occurrence of any respiratory intervention | Hypotension | Patient satisfaction with sedation | Recovery time (minutes) | Vomiting | ||||||||
| Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | ||
| Ferguson et al[17], 2016 | 23 ± 8 | 17 ± 6 | 16 ± 5 | 11 ± 4 | 47 ± 16 | 38 ± 14 | 24 ± 8 | 3 ± 1 | 10 ± 0 | 10 ± 0 | NR | NR | 8 ± 3 | 12 ± 4 | |
| Miner et al[22], 2014 | NR | NR | 14 ± 16 | 1:1 = 10 ± 12; 4:1 = 15 ± 16 | 41 ± 45 | 1:1 = 33 ± 39; 4:1 = 48 ± 50 | NR | NR | 70 ± 78 | 1:1 = 61 ± 72, 4:1 = 78 ± 81 | 6 ± 4.45 | 1:1 = 10 ± 8.89; 4:1 = 8 ± 8.15 | NR | NR | |
| Ulutas et al[32], 2023 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Soliman et al[25], 2017 | NR | NR | NR | NR | NR | NR | 3 ± 10 | 2 ± 6.6 | NR | NR | 26.3 ± 4.1 | 21.4 ± 3.7 | NR | NR | |
| Kumar et al[26], 2020 | NR | NR | NR | NR | NR | NR | 18 ± 60 | 3 ± 10 | NR | NR | NR | NR | NR | NR | |
| Andolfatto et al[18], 2012 | 36 ± 25 | 38 ± 27 | 13 ± 9 | 15 ± 11 | NR | NR | 1 ± 0.7 | 0 | NR | NR | 6 ± 4.45 | 8 ± 2.22 | NR | NR | |
| Tandon et al[24], 2014 | NR | NR | NR | NR | 19 ± 14.6 | 4 ± 3 | 16 ± 12 | 6 ± 4 | NR | NR | 5 ± 2.22 | 4 ± 12.22 | NR | NR | |
| Khalil et al[35], 2021 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
| Frey et al[23], 1999 | NR | NR | NR | NR | 18 ± 54.5 | 9 ± 27 | NR | NR | NR | NR | NR | NR | NR | NR | |
| Cui et al[27], 2023 | NR | NR | NR | NR | NR | NR | 14 ± 20.9 | 11 ± 16.2 | 10 | 10 | 6 ± 1.48 | 6 ± 0.74 | NR | NR | |
| Liu et al[28], 2025 | NR | NR | NR | NR | NR | NR | 46 ± 36.5 | 18 ± 14.3 | NR | NR | NR | NR | NR | NR | |
| Zheng et al[29], 2023 | NR | NR | NR | NR | NR | NR | 12 ± 23.1 | 4 ± 7.7 | NR | NR | NR | NR | 0 | 0 | |
| Fu et al[19], 2024 | 1 ± 4.0 | For all mixtures of esketofol = 0 | NR | NR | NR | NR | 15 ± 60.0 | Propofol + esketamine 0.2 = 4 ± 16.0; propofol + esketamine 0.3 = 4 ± 16.0; popofol + esketamine 0.4 = 3 ± 12.0 | 5.0 | For all mixtures of esketofol = 5.0 | NR | NR | NR | NR | |
| Beyoğlu et al[33], 2020 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 26.9 ± 6.19 | 27.1 ± 5.41 | NR | NR | |
| Frizelle et al[21], 1997 | NR | NR | 0 | 1 | NR | NR | 4 ± 20 | 4 ± 20 | NR | NR | NR | NR | NR | NR | |
| Bhardwaj et al[30], 2024 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 26.9 ± 6.19 | 27.1 ± 5.41 | NR | NR | |
| Schmitz et al[31], 2018 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 54 ± 29.65 | 38 ± 31.87 | 4 ± 2.5 | 3 ± 1.9 | |
| Chiaretti et al[20], 2011 | 3/62 ± 4.8 | 1/59 ± 1.7 | NR | NR | NR | NR | NR | NR | NR | NR | 20 ± 2 | 8 ± 2 | NR | NR | |
| Xu et al[43], 2024 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | |
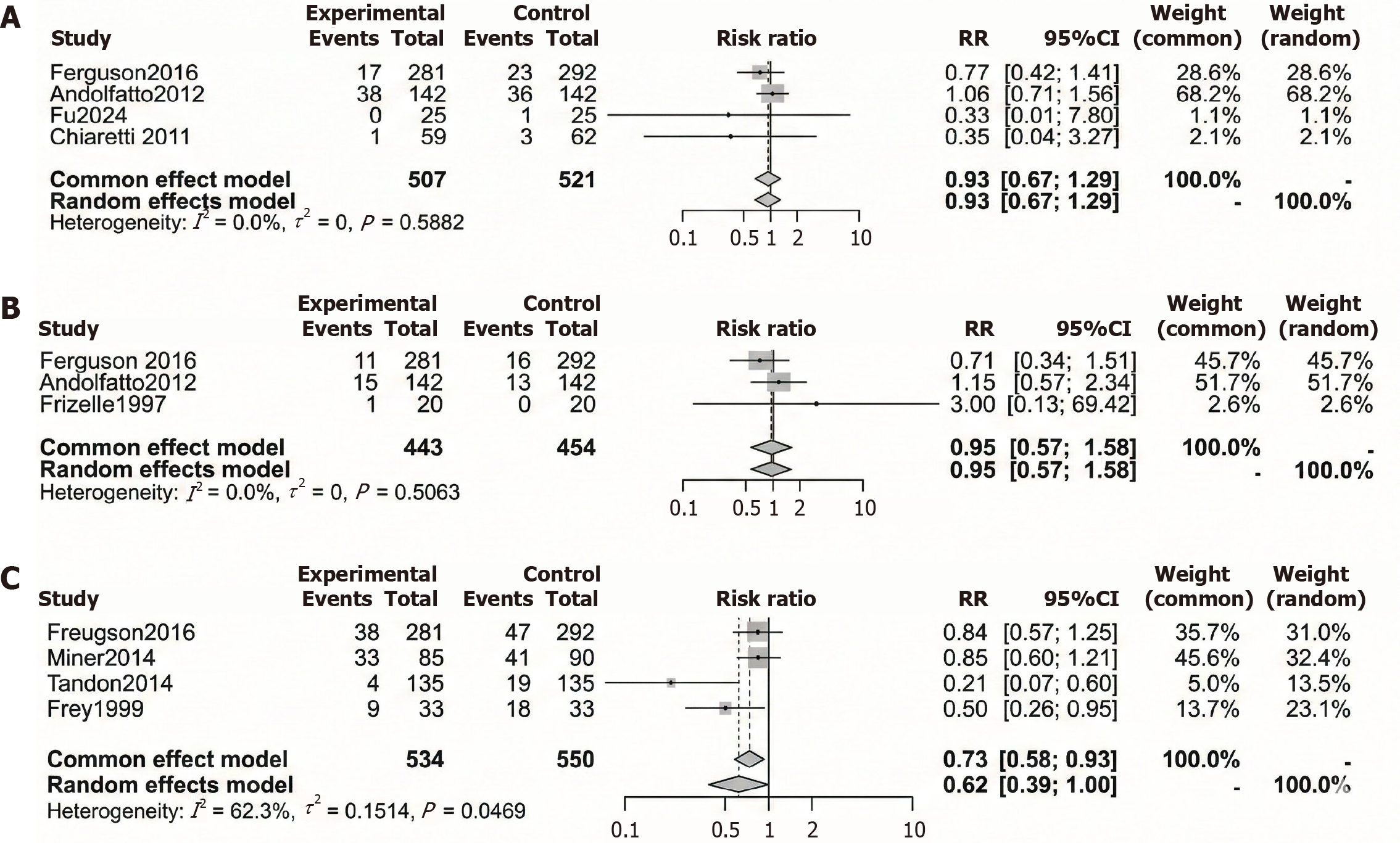
Desaturation:Figure 2A presents a forest plot of the meta-analysis of the RR for desaturation. This analysis involved four studies with 507 experimental participants and 521 control participants[17-20]. The RR s corresponding to each individual study were 0.77 (95%CI: 0.42-1.41), 1.06 (95%CI: 0.71-1.56), 0.33 (95%CI: 0.01-7.80), and 0.35 (95%CI: 0.04-3.27), respectively[17-20]. All individual studies failed to demonstrate a statistically significant effect because their confidence intervals crossed the no-effect line. The combined RR from all studies reached 0.93 (95%CI: 0.67-1.29). The meta-analysis demonstrated no statistically significant difference in desaturation risk between the experimental and control groups, as the 95%CI of the pooled estimate included 1. The included studies showed no significant heterogeneity, as indicated by I2 = 0.0% and P = 0.5882.
Apnea: As shown in Figure 2B, an analysis was performed to evaluate the RR for apnea. Three studies were included, comprising 443 participants in the experimental group and 454 in the control group. The reported RRs from the three studies were 0.71, 1.15, and 3.00, none of which were statistically significant[17,18,21]. This analysis resulted in a RR value of 0.95 with a 95%CI: 0.57-1.58. The confidence interval included the line of no effect, indicating no statistically significant difference in apnea risk between the control and experimental groups.
Figure 2C shows the forest plot for the respiratory intervention. Four studies, including 534 participants in the experimental group and 550 in the control group, were analyzed[17,22-24]. Two studies reported significant reductions in risk[23,24]. The combined RR under a fixed-effects model reached 0.73 (95%CI: 0.58-0.93), showing notable advantages. The analysis revealed moderate heterogeneity, with I2 = 52.3% and P = 0.0469. The pooled RR was borderline significant at 0.62 (95%CI: 0.39-1.00) under a random-effects model, indicating study heterogeneity.
Subgroup analysis of respiratory interventions, stratified by publication year, was conducted. An integrated evaluation of the data indicates a near-statistically significant decrease in respiratory intervention risk (RR = 0.62, 95%CI: 0.39-1.00). The two subgroups (“Year Group 2013.5” and “Year Group < 2013.5”) failed to demonstrate a statistically meaningful effect independently. The subgroup results from the analysis showed strong heterogeneity, indicating substantial variation across the different study outcomes. A test for subgroup differences was not statistically significant (P = 0.5667), indicating no strong evidence that the intervention's effect differs between studies published before or after mid-2013 (Supplementary Figure 2). Similarly, another subgroup analysis stratified studies by sample size. The complete analysis demonstrates a borderline statistically significant decrease in respiratory intervention risk (RR = 0.62, 95%CI: 0.39-1.00) because the upper boundary of the confidence interval aligns with the no-effect line. Neither of the two subgroups demonstrated statistical significance in their effects when analyzed independently. The study results showed substantial variability, reflecting the high heterogeneity within the subgroups. The P-value from the test of subgroup differences was 0.5667, indicating that the intervention effect did not differ significantly between studies with higher and lower sample sizes (Supplementary Figure 3).
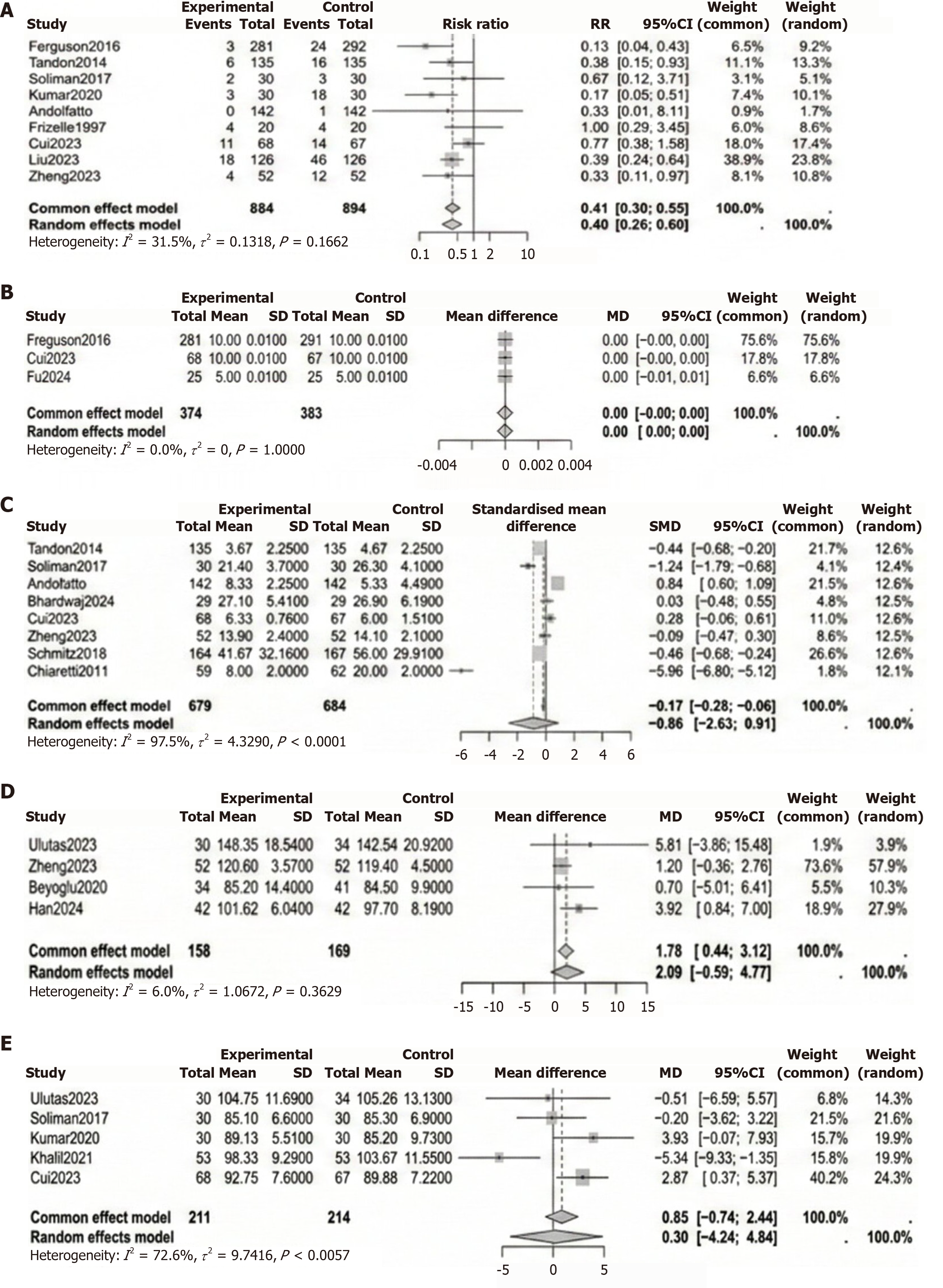
Hypotension:Figure 3A presents the forest plot for hypotension. Nine studies were included, with 884 participants in the experimental group and 894 in the control group[17,18,21,24-28]. Several individual studies demonstrated a significant risk reduction. The combined RR was 0.40 (95%CI: 0.26-0.60), indicating that the experimental group had 40% fewer hypotension cases. The results showed 31.5% heterogeneity (P-value = 0.1662), confirming the stability of the findings.
Patient satisfaction: Figure 3B displays the forest plot evaluating patient satisfaction. Three studies were included, involving 374 participants in the experimental group and 383 in the control group[17,19,27]. Each study reported a mean difference of 0.00, with narrow confidence intervals that did not cross the line of no effect. The pooled analysis also showed a mean difference of 0.00 (95%CI: -0.00 to 0.00), confirming that there was no significant difference in patient satisfaction between groups. Heterogeneity was absent (I2 = 0.0%, P = 1.0000).
Recovery time:Figure 3C displays the forest plot for patient recovery time. The research evaluation comprised eight studies, including 679 experimental participants and 684 control group members[18,20,24,25,27,29-31]. Different research findings emerged from individual studies: Some found significant reductions in recovery time, while others detected no significant change. The fixed-effects model showed a significant average difference of -0.17 (95%CI: -0.28 to -0.06). The analysis of studies revealed substantial inconsistency, as heterogeneity was high (I2 = 97.5%, P < 0.0001). A random-effects model was used to analyze the data, yielding a combined standardized mean difference of -0.86 with CIs of -2.63 to 0.91. The statistical analysis failed to show significant results because the confidence interval crossed zero, providing no definitive evidence of recovery time effects after accounting for study variation.
Systolic blood pressure: Figure 3D illustrates the results of the meta-analysis of systolic blood pressure (SBP). The analysis incorporated four studies, which included 158 participants from the experimental group and 169 from the control group[17,29,32-34]. Every individual study showed no significant findings. The combined mean difference was 2.09 (95%CI: -0.59 to 4.77), but the confidence interval included zero, yielding a statistically insignificant result. The findings showed minimal variation across studies, with an I2 value of 6.0% and P = 0.3629 indicating consistency.
Subgroup analysis based on age: Subgroup analysis was conducted to evaluate SBP effects, stratified by age (≥ 50) and (< 50). Statistical analysis of both subgroups failed to show significant treatment effects because the 95%CIs for each group included zero. The subgroup differences test produced a P-value of 0.0765, which lacked statistical significance. The results showed that the intervention impacted SBP similarly across age groups, even though there was substantial variability in the “age < 50” population (Supplementary Figure 4).
Mean arterial pressure: The results of the meta-analysis on mean arterial pressure (MAP) are presented in Figure 3E. Five studies were included, with 211 participants in the experimental group and 214 in the control group[25-27,32,35]. The 2023 research by Cui et al[27] found that MAP levels increased significantly, although most studies did not report substantial changes. A statistical evaluation using a fixed-effects approach found no significant difference between the means at 0.85, with CIs ranging from -0.74 to 2.44. Multiple studies yielded mixed results, with the analysis revealing significant heterogeneity (I2 = 72.6%, P = 0.0057). The random-effects analysis of MAP data yielded a mean difference of 0.30 (95%CI: -4.24 to 4.84), which did not reach statistical significance.
In the subgroup analysis of MAP by sample size, the overall combined analysis shows a mean difference of 0.30 with a 95%CI: -4.24 to 4.84. Since this interval crosses zero, the overall effect is not statistically significant. Neither subgroup shows a significant effect either. The analysis reveals substantial heterogeneity: The first subgroup has an I2 of 83%, and the overall I2 is 57.7%. The subgroup difference analysis showed no statistical significance (P = 0.4264), indicating that the intervention effect on MAP does not differ substantially across studies with different sample sizes (Supplementary Figure 5).
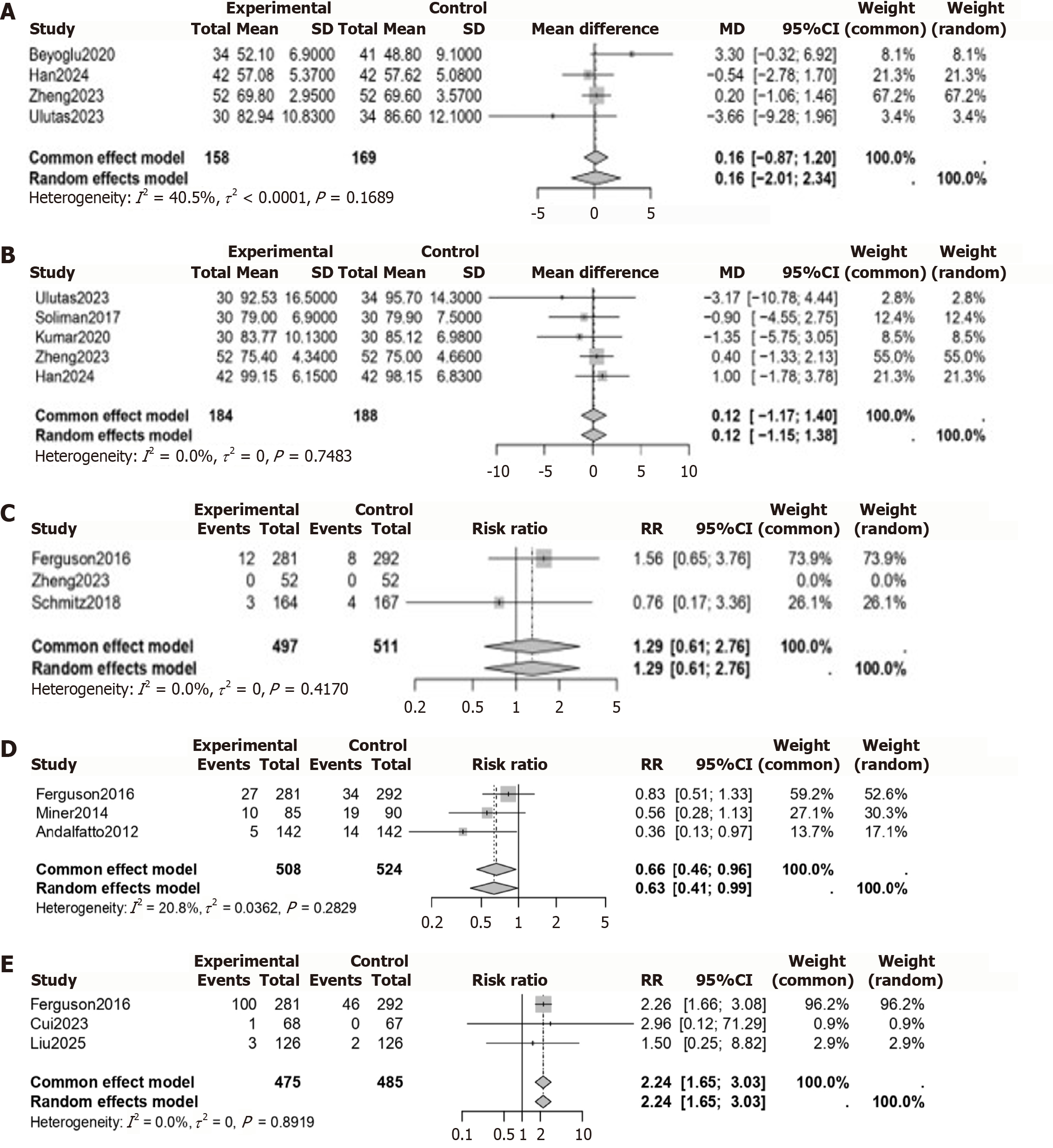
Diastolic blood pressure:Figure 4A depicts the forest plot from the meta-analysis of diastolic blood pressure (DBP). Four studies were included, involving 158 participants in the experimental group and 169 in the control group[29,32-34]. None of the individual studies had a statistically significant effect. The pooled mean difference was 0.16 (95%CI: -2.01 to 2.34), and because the CI crossed zero, the overall result was not statistically significant and did not provide clear evidence of a difference between groups. There was moderate heterogeneity (I2 = 40.5%, P = 0.1689).
The subgroup analysis of the effect on DBP, stratified by age (< 50 and ≥ 50), was conducted. For both age groups, the pooled results show no statistically significant effect, as the 95%CIs for both subgroups include zero. While there is moderate heterogeneity within each subgroup, the test for subgroup differences shows no statistically significant difference in the treatment effect between the two age groups (P = 0.1791). This means that the effect of the intervention on DBP does not significantly differ based on whether a person is under 50 or 50 and older (Supplementary Figure 6).
Heart rate: Figure 4B shows the forest plot for heart rate. Five studies were included, with 184 participants in the experimental group and 188 in the control group[25,26,29,32,34]. No individual study showed a statistically significant effect. The pooled mean difference was 0.12 (95%CI: -1.15 to 1.38); since the confidence interval crossed zero, the overall result was also not statistically significant, and there was no strong evidence of a difference in heart rate between groups.
Vomiting: Figure 4C shows a forest plot of the meta-analysis of the RR for vomiting. Three studies involving a total of 497 participants in the intervention group and 511 in the control group were included in the analysis[17,29,31]. No single study showed a significant effect. The combined RR was 1.29 (95%CI: 0.61-2.76). Although the 95%CI encloses 1, meaning that the meta-analysis overall result is not statistically significant, it did not present any specific evidence of difference in the risk for vomiting between control and experimental groups.
Airway events: The meta-analysis of airway events is summarized in the forest plot shown in Figure 4D. Three studies were included, comprising 508 participants in the experimental group and 524 in the control group[17,18,22]. Although two individual studies did not show a significant effect on their own[17,22], the combined analysis, which gives more weight to the larger studies, reveals a clear benefit (pooled mean difference was 0.63 (95%CI: 0.41-0.99). The low heterogeneity (I2 = 20.8%) and non-significant P-value (P = 0.2829) confirm that the results across the studies are consistent, strengthening the conclusion.
Hallucinations: The forest plot shown in Figure 4E demonstrates a meta-analysis of three studies concerning the risk of hallucinations[17,27,28]. The experimental group comprised 475 participants, and the control group comprised 485 participants. The overall result of this analysis was a RR of 2.24 and a 95%CI of 1.65-3.03, and since in this CI the value of 1 was excluded, there is statistical significance that the intervention or exposure was associated with more than a two-fold increased risk of hallucinations. The plot showed no heterogeneity (I2 = 0.0%), indicating very consistent results with variability likely due to chance.
Sensitivity analyses were performed across all outcomes to investigate the robustness of the pooled estimates. For DBP, the LOO analysis displayed that removing individual studies did not substantially affect the overall mean difference or the associated 95%CIs, establishing that no single study systematically influenced the pooled estimates. For MAP, both the inclusion/exclusion of the trial by Cui et al[27] and the LOO analysis exhibited only small shifts in pooled effect sizes and heterogeneity statistics, supporting the finding of stability. In the same way, for the respiratory intervention, the LOO analysis with a random-effects model showed that the recalculated RRs, derived by sequentially removing one study, were closely aligned with the overall effect and, at times, provided overlapping CIs, demonstrating that our findings were robust. Regarding apnea incidence, a sensitivity analysis limited to the two available trials indicated consistent RRs with stable precision, despite a small evidence base[17,18]. In conclusion, all analyses demonstrated that no single study drove the overall conclusions, thus demonstrating the robustness and dependability of the meta-analytic findings (Supplementary Figures 7-11).
Meta-regression was conducted to examine potential causes of heterogeneity in the notation hypotension outcome. A positive association was found between propofol dose difference (mg) and the log (RR) for hypotension, with the largest studies having the greatest effect on the overall pattern of association[28,29]. In contrast, a very slight negative correlation between mean age and log (RR), where studies with greater weight shaped the trend[21,28]. Finally, a negative cor
| Ref. | SBP (mmHg) | DBP (mmHg) | MAP at different time points, T1, T2, and T3, (mmHg) | Heart rate at time points H1, H2, H3 (beats/minutes) | Airway events | Hallucinations | ||||||
| Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | Control | Intervention | |
| Ferguson et al[17], 2016 | NR | NR | NR | NR | NR | NR | NR | NR | 34 ± 12 | 27 ± 9 | 46 ± 15 | 100 ± 35 |
| Miner et al[22], 2014 | NR | NR | NR | NR | NR | NR | NR | NR | 19 ± 21 | 1:1 = 10 ± 12; 4:1 = 25 ± 26 | NR | NR |
| Ulutas et al[32], 2023 | t1 = 140.23 ± 2.75, t2 = 143.61 ± 26.40, t3 = 143.79 ± 27.65 | t1 = 131.06 ± 1.71; t2 = 157.53 ± 2.83 t3 = 156.46 ± 24.37 | t1 = 83.64 ± 10.86 t2 = 89.47 ± 16.69 t3 = 86.70 ± 16.71 | t1 = 72.36 ± 9.07; t2 = 92.50 ± 17.67; t3 = 83.96 ± 12.69 | t1 = 102.51 ± 13.24; t2 = 107.52 ± 16.88; t3 = 105.74 ± 18.07 | t1 = 91.93 ± 9.52; t2 = 114.18 ± 18.32; t3 = 108.13 ± 14.79 | h1 = 94.85 ± 15.45; h2 = 97.35 ± 19.66; h3 = 94.91 ± 17.36 | h1 = 92.53 ± 16.53; h2 = 102.87 ± 19.51; h3 = 105.47 ± 16.60 | NR | NR | NR | NR |
| Soliman et al[25], 2017 | NR | NR | NR | NR | 85.3 ± 6.9 | 85.1 ± 6.6 | No different time points. 79.9 ± 7.5 | 79.0 ± 6.9 | NR | NR | NR | NR |
| Kumar et al[26], 2020 | NR | NR | NR | NR | Immediate postinduction = 83.6 ± 11.7, 3 minutes = 70.1 ± 6.8, 1 minute postintubation = 101.4 ± 13.1, 3 minutes = 90.4 ± 14.2, 5 minutes = 85.3 ± 14.7, 10 minutes = 83.5 ± 14.5 | Immediate postinduction = 90.4 ± 8.8, 3 minutes = 85.2 ± 8.0, 1 minute postintubation = 92.7 ± 7.7, 3 minutes = 89.2 ± 6.5, 5 minutes = 89.2 ± 6.2, 10 minutes = 88.1 ± 6.0 | Immediate postinduction = 79.2 ± 8.6, 3 minutes = 65.7 ± 5.4, 1 minute postintubation = 103.5 ± 12.4, 3 minutes = 94.1 ± 8.9, 5 minutes = 86.9 ± 9.4, 10 minutes = 81.3 ± 9.9 | Immediate postinduction = 82.2 ± 11.3, 3 minutes = 80.8 ± 12.4, 1 minute postintubation = 84.8 ± 9.5, 3 minutes = 88.0 ± 15.8, 5 minutes = 84.6 ± 15.4, 10 minutes = 82.2 ± 15.0 | NR | NR | NR | NR |
| Andolfatto et al[18], 2012 | NR | NR | NR | NR | NR | NR | NR | NR | 14 ± 10 | 5 ± 4 | NR | NR |
| Tandon et al[24], 2014 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Khalil et al[35], 2021 | NR | NR | NR | NR | T1 = 115, T2 = 110, T3 = 105, T4 = 100, T5 = 95, T6 = 90 | T1 = 110; T2 = 105; T3 = 100; T4 = 95; T5 = 90; T6 = 85 | T1 = 105; T2 = 100; T3 = 95; T4 = 90; T5 = 85; T6 = 80 | T1 = 85; T2 = 90; T3 = 85; T4 = 80; T5 = 75; T6 = 70 | NR | NR | NR | NR |
| Frey et al[23], 1999 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Cui et al[27], 2023 | NR | NR | NR | NR | t1 = 100; t2 = 100; t3 = 85; t4 = 83; t5 = 80; t6 = 83; t7 = 84; t8 = 83 | t1 = 100; t2 = 99; t3 = 89; t4 = 85; t5 = 84; t6 = 86; t7 = 89; t8 = 89 | NR | NR | NR | NR | 0 | 1 ± 1.5 |
| Liu et al[28], 2025 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | 2 ± 1.6 | 3 ± 2.4 |
| Zheng et al[29], 2023 | t1 = 124; t2 = 110; t3 = 103; t4 = 140; t5 = 120 | t1 = 123; t2 = 120; t3 = 120; t4 = 124; t5 = 125 | t1 = 71; t2 = 65; t3 = 60; t4 = 76; t5 = 69 | t1 = 70; t2 = 69; t3 = 68; t4 = 70; t5 = 69 | NR | NR | t1 = 75; t2 = 70; t3 = 68; t4 = 83; t5 = 75 | t1 = 75; t2 = 74; t3 = 73; t4 = 77; t5 = 75 | NR | NR | NR | NR |
| Fu et al[19], 2024 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Beyoğlu et al[33], 2020 | 84.5 ± 9.9 | 85.2 ± 14.4 | 48.8 ± 9.1 | 52.1 ± 6.9 | NR | NR | NR | NR | NR | NR | NR | NR |
| Frizelle et al[21], 1997 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Bhardwaj et al[30], 2024 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Schmitz et al[31], 2018 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Chiaretti et al[20], 2011 | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR | NR |
| Xu et al[43], 2024 | 0 minute = 109; 5 minutes = 99; 10 minutes = 100; 15 minutes = 102; 20 minutes = 101; 25 minutes = 104; 30 minutes = 107; 35 minutes = 104; 40 minutes = 102; 45 minutes = 98; 50 minutes = 97; 55 minutes = 97; 60 minutes = 97 | 0 minute = 42; 5 minutes = 42; 10 minutes = 42; 15 minutes = 42; 20 minutes = 40; 25 minutes = 40; 30 minutes = 33; 35 minutes = 25; 40 minutes = 19; 45 minutes = 15; 50 minutes = 12; 55 minutes = 11; 60 minutes = 11 | 0 minute = 66; 5 minutes = 55; 10 minutes = 58; 15 minutes= 62; 20 minutes = 63; 25 minutes = 64; 30 minutes = 62; 35 minutes = 62; 40 minutes = 60; 45 minutes = 58; 50 minutes = 55; 55 minutes = 52; 60 minutes = 43 | 0 minute = 64; 5 minutes = 56; 10 minutes = 67; 15 minutes = 54; 20 minutes = 66; 25 minutes = 51; 30 minutes = 61; 35 minutes = 61; 40 minutes = 59; 45 minutes = 54; 50 minutes = 50; 55 minutes = 51; 60 minutes = 50 | NR | NR | NR | NR | NR | NR | NR | NR |
| Covariate (moderator) | Direction of association | Approximate trend |
| Propofol dosage difference (msg) | Positive | Each 50 mg increase in propofol appeared to increase LogRR by approximately 0.05-0.10 |
| Mean age (years) | Slighty negative | Each approximately 10 years increase in mean age appeared to decrease LogRR by approximately 0.01 (very weak) |
| Ketamine dosage difference | Negative | Each 50 mg increase in ketamine appeared to decrease LogRR by approximately 0.05-0.08 |
This SRMA-analysis of 19 RCTs compared two related techniques of NMDA modulation with propofol: The combination of racemic ketamine with propofol (Ketofol) and the S-enantiomer esketamine with propofol, with propofol monotherapy as procedural sedation. Pooled estimates generally indicate that combining an NMDA antagonist with propofol decreases the incidence of hypotension and is associated with a tendency toward reduced respiratory interventions, but does not consistently affect desaturation, vomiting, apnea, or patient satisfaction. The recovery time results were inconsistent across trials, and neuropsychiatric adverse effects (perceptual disturbances or hallucinations) were more common with the combination of ketamine.
There was no notable difference in desaturation risk between propofol and Ketofol. Large-scale emergency department studies by Ferguson et al[17] and Andolfatto et al[18] reported similar desaturation rates with the two regimens. By contrast, Fu et al[19], assessing esketamine dosing in the context of colonoscopy, and Chiaretti et al[20], with children undergoing oncological procedures, saw reduced numbers of desaturation events using Ketofol, although the studies were underpowered and failed to achieve statistical significance. More recent studies have yielded procedure-specific findings, such as those of Foo et al[36], in an SRMA of pediatric sedation, further demonstrating that Ketofol did not significantly reduce desaturation compared with propofol alone, highlighting that any respiratory protective effect is inconsistent when analyzed across larger aggregated data. In comparison, Song et al[37] presented stronger evidence in the form of a RCTs, showing that esketamine-propofol effectively reduced desaturation during bidirectional endoscopy compared with propofol monotherapy. The findings in general suggest that while ketamine would, in principle, suppress propofol-related respiratory depression to some extent, consistent and clinically significant benefit more readily emerges with esketamine-propofol combinations in special procedural applications.
Similarly, there was no notable difference in apnea risk. This is similar to the findings of Ferguson et al[17] and Andolfatto et al[18], who reported equal apnea incidence during sedation in the emergency department, and of Frizelle et al[21], who found no advantage of Ketofol in spinal anesthesia. The risk of apnea was not reduced appreciably in the prior meta-analysis by Foo et al[36] in children. Theoretically, even though airway tone and respiratory drive are apparently maintained by ketamine alone, this has not routinely translated to clinical protection against apnea, and therefore cannot be the sole indication for Ketofol. Large-scale studies consistently using standardized monitoring protocols may explain the potential advantage in selective populations. Also note that our apnea risk analysis was based solely on Ketofol trials, with no esketamine-propofol study data included in this endpoint. Large-scale trials with similar monitoring protocols will, however, be needed to ascertain whether there is any subset of patients who benefit.
Our meta-analysis also indicated a decrease in the need for respiratory interventions with Ketofol, as assessed using fixed-effects modeling. However, significance was borderline in the random-effects analysis, suggesting moderate heterogeneity. Tandon et al[24] and Frey et al[23] both reported significant decreases in intervention rate, whereas subsequent trials failed to consistently replicate this advantage. Subgroup analysis did not unearth years of publication or sample size as effect modifiers, highlighting the effect of contextual procedural rather than methodological factors. Notably, external evidence corroborates this finding. The large meta-analysis by Jalili et al[13] showed that Ketofol significantly decreased respiratory complications requiring intervention, as indicated by pooled data analysis, and demonstrated a consistent effect size across different trial settings. Although our included trials did not evaluate esketamine, independent evidence from Song et al[37] confirmed that esketamine-propofol decreased the need for airway maneuvers, such as jaw thrust, during endoscopy. So, while the results of this study are generalizable only to Ketofol, independent evidence indicates that esketamine-propofol will yield the same respiratory advantage under certain procedural conditions.
The accompanying pharmacological rationale bolsters the observation. Propofol’s central respiratory depression and loss of upper airway reflexes are countered by ketamine’s preservation of spontaneous ventilation and airway tone. Both ketamine and its S-enantiomer, esketamine, are NMDA receptor antagonists. Esketamine is particularly striking in having 2 times to 4 times more NMDA receptor affinity than the R-enantiomer[38,39]. Greater NMDA antagonism underlies the maintenance of airway reflexes and respiratory drive. It may underlie the protective effects on respiration observed with both Ketofol and the esketamine-propofol combination, albeit with variable magnitude depending on dosing, procedure, and patient variables. Collectively, these findings highlight the imperative need for large-scale randomized trials to differentiate agent-specific efficacy and determine the population best suited for the benefit of NMDA-based sedative administration. It is important to differentiate between the two NMDA antagonists that we assessed. Esketamine, which is an S-enantiomer of ketamine, has nearly two times the analgesic potency of racemic ketamine and a higher elimination rate[40]. These variations in pharmacokinetics suggest that esketamine-propofol combinations might offer faster recovery rates and have different side effects compared to traditional Ketofol. However, our analysis found comparable safety profiles for both.
Our meta-analysis also revealed that the use of Ketofol or the esketamine-propofol combination was associated with a reduced incidence of hypotension compared with propofol alone. In our included trials, Cui et al[27], Liu et al[28], and Zheng et al[29] separately examined esketamine-propofol schedules, whereas the other trials assessed Ketofol. The difference suggests that both NMDA-centric and ketamine-centric combinations contributed to the net protection effect observed in our meta-analysis. There is supportive evidence from previous meta-analyses as well. Foo et al[36] indicated that Ketofol decreased the risk of hypotension compared with propofol monotherapy, and the same inference was drawn in the meta-analysis conducted by Jalili et al[13]. Additionally, the external randomized study conducted by Song et al[37] strengthened the inferences by providing evidence that esketamine, when paired with propofol, significantly reduced the incidence of hypotension during endoscopy procedures. Overall, the findings indicate that administering propofol with the addition of either esketamine or ketamine is associated with hemodynamic stability and a decrease in hypotension.
There were no significant differences in SBP, MAP, DBP, or heart rate between the adult and child subgroups in our study. Age- and sample size-matched subgroup analyses similarly indicated no significant variation, indicating similar effects across population subgroups. Notably, numerous included trials, such as those by Cui et al[27], Zheng et al[29], Ulutas et al[32], and Xu et al[29], assessed esketamine-propofol combinations and consistently reported non-significant hemodynamic changes, as reflected in the Ketofol trials. Individual external reports from Hwang et al[41] and Tomatir et al[42] similarly indicated little cardiovascular perturbation when ketamine and propofol were co-administered. Further external evidence from Xu et al[43] similarly confirms this finding, demonstrating that esketamine added to propofol decreased injection pain but also sustained systolic, diastolic, and MAP and heart rate within normal ranges, highlighting the hemodynamic stability of NMDA-based anesthetics. These findings are consistent with prior meta-analyses[36,13], all of which support cardiovascular stability with both Ketofol compared with propofol and esketamine-propofol compared with propofol.
The fundamental principles of drug pharmacology account for the findings. Propofol causes vasodilation as well as negative inotropy, putting patients at risk of hypotension. In contrast, the combination of ketamine and esketamine is an NMDA receptor antagonist with enhanced sympathomimetic action that elevates vascular resistance and heart rate. Esketamine has a more favorable NMDA receptor affinity than the R-enantiomer and a cardiovascular stabilizing drug profile similar to that of ketamine[38,40]. These dual drug effects balance the depressor effect of propofol as well as yield stable systolic, diastolic, and MAP in addition to heart rate in the clinical setting.
Patient outcomes, such as satisfaction and recovery duration, were generally comparable between propofol and NMDA-propofol combination therapy, in keeping with previous meta-analyses[36,13]. No recovery difference was reported from Andolfatto et al[18] with Ketofol, but shorter recovery with greater satisfaction was reported from Makkey et al[44]. Esketamine trials included in our review showed broadly similar satisfaction, with some evidence of rapid recovery, and a mild advantage in selective use[36,27,29]. Overall, results remain even, with incremental advantages depending on agent and setting.
In the context of adverse events, Ketofol and esketamine-propofol combinations were generally well tolerated regarding nausea and vomiting[38,45,46]. But the major issue at hand is hallucinations since the psychomimetic effect of ketamine and esketamine produces distorted perception, mood changes, and auditory or visual hallucinations[38,46-48]. These events are generally transitory and self-limiting in occurrence, but clinicians should be vigilant and reduce dosing, especially if there is a psychiatric predisposition. Overall, Ketofol and esketamine-propofol provide effective sedation and hemodynamic and respiratory stability, and the most common adverse event requiring consideration is hallucinations.
In short, NMDA-propofol combinations, including Ketofol and esketamine-propofol, achieve successful sedation with preserved hemodynamic and respiratory stability, comparable patient satisfaction, and negligible impact on recovery time, while carrying the added burden of transitory hallucinations. These findings highlight the hemodynamic and procedural utility of both Ketofol and esketamine-propofol, tempered by neuropsychiatric considerations. Readers should interpret these pooled results in the context of clinical heterogeneity as the trials that were included spanned across diverse settings, which range from emergency departments to elective endoscopy suites, and included both adult and pediatric populations. Though we found no significant differences in terms of age or publication year, the varying depth of sedation required for different procedures may influence the absolute rates of respiratory and hemodynamic events. Although this SRMA was comprehensive, it had limitations that should be noted. Heterogeneity was detected in some outcomes, specifically respiratory intervention and MAP, indicating variation within the study population, dosing, and procedural techniques. The finding was reproduced in the sensitivity analysis as strong and not due to any single study; however, the sparsity of trials in some outcomes, such as apnea, leads to imprecision.
Meta-regression identified differences in propofol and ketamine/esketamine dosing, as well as patient age, as partial contributors to inter-study heterogeneity in the risk of hypotension. However, these findings do highlight potential effect modifiers and the importance of individualized dosing in clinical practice. These associations were not statistically significant, and the limited number of studies and their statistical power highlight that the findings should be interpreted with caution, as they might serve as exploratory insights rather than definitive causal evidence. Additionally, including studies with heterogeneous age groups, procedural sites, and sedation regimens may reduce the generalizability of some findings. Lastly, reporting bias cannot be excluded, and outcomes assessed in a limited number of studies may affect the reliability of pooled estimates.
In summary, this SRMA of 19 RCTs establishes that NMDA-propofol combinations, such as Ketofol and esketamine-propofol, are associated with hemodynamic benefits relative to propofol monotherapy, primarily by reducing hypotension while maintaining similar respiratory stability and patient-oriented outcomes. Transient hallucinations are the most significant adverse effect, prompting clinicians’ vigilance. Recovery duration and patient satisfaction effects are typically mild and acceptable clinically. Procedural environment, patient type, and dosage regimen are most likely effectors of effectiveness and safety. Larger, standardized randomized trials are justified to determine optimal dosage strategies, identify the population most suited to benefit, and assess procedure-related outcomes, especially in the new field of mechanical ventilation therapy and neuropsychiatric adverse effects.
| 1. | Leffler DA, Bukoye B, Sawhney M, Berzin T, Sands K, Chowdary S, Shah A, Barnett S. Development and validation of the PROcedural Sedation Assessment Survey (PROSAS) for assessment of procedural sedation quality. Gastrointest Endosc. 2015;81:194-203.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 2. | Krauss B, Green SM. Sedation and analgesia for procedures in children. N Engl J Med. 2000;342:938-945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 252] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 3. | Meyer S, Grundmann U, Gottschling S, Kleinschmidt S, Gortner L. Sedation and analgesia for brief diagnostic and therapeutic procedures in children. Eur J Pediatr. 2007;166:291-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (1)] |
| 4. | Sahyoun C, Cantais A, Gervaix A, Bressan S, Löllgen R, Krauss B; Pediatric Emergency Medicine Comfort and Analgesia Research in Europe (PemCARE) group of the Research in European Pediatric Emergency Medicine. Pediatric procedural sedation and analgesia in the emergency department: surveying the current European practice. Eur J Pediatr. 2021;180:1799-1813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 5. | Hinkelbein J, Lamperti M, Akeson J, Santos J, Costa J, De Robertis E, Longrois D, Novak-Jankovic V, Petrini F, Struys MMRF, Veyckemans F, Fuchs-Buder T, Fitzgerald R. European Society of Anaesthesiology and European Board of Anaesthesiology guidelines for procedural sedation and analgesia in adults. Eur J Anaesthesiol. 2018;35:6-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 245] [Cited by in RCA: 190] [Article Influence: 23.8] [Reference Citation Analysis (4)] |
| 6. | Bryson HM, Fulton BR, Faulds D. Propofol. An update of its use in anaesthesia and conscious sedation. Drugs. 1995;50:513-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 253] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 7. | McKeage K, Perry CM. Propofol: a review of its use in intensive care sedation of adults. CNS Drugs. 2003;17:235-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 142] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 8. | Marik PE. Propofol: therapeutic indications and side-effects. Curr Pharm Des. 2004;10:3639-3649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 302] [Cited by in RCA: 340] [Article Influence: 15.5] [Reference Citation Analysis (3)] |
| 9. | Short CE, Bufalari A. Propofol anesthesia. Vet Clin North Am Small Anim Pract. 1999;29:747-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 93] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 10. | Miao Y, Zheng M, Li Q, Xiong L, Feng J, Liu X, Fan G, Chaturvedi R, Zhang F, Yin N. Comparison of propofol-esketamine versus propofol-sufentanil for deep sedation and analgesia in children with autism: A randomized double-blind clinical trial. Autism Res. 2024;17:1356-1364. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 11. | Huang X, Lin F, Chen Q, Hu X. Safety and efficacy of the combination of esketamine and propofol in procedural sedation/analgesia: a systematic review and meta-analysis. Minerva Anestesiol. 2023;89:680-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 12. | Yan JW, McLeod SL, Iansavitchene A. Ketamine-Propofol Versus Propofol Alone for Procedural Sedation in the Emergency Department: A Systematic Review and Meta-analysis. Acad Emerg Med. 2015;22:1003-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 13. | Jalili M, Bahreini M, Doosti-Irani A, Masoomi R, Arbab M, Mirfazaelian H. Ketamine-propofol combination (ketofol) vs propofol for procedural sedation and analgesia: systematic review and meta-analysis. Am J Emerg Med. 2016;34:558-569. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 67] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 14. | Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9803] [Reference Citation Analysis (0)] |
| 15. | Hassan ST, Faheem MSB, Munir SU. Combination of N-Methyl-D-Aspartate Receptor Antagonists and Propofol for Procedural Sedation: A Systematic Review and Meta-Analysis. PROSPERO 2025; CRD420251123120 [cited 30 August 2025]. Available from: https://www.crd.york.ac.uk/PROSPERO/view/CRD420251123120. |
| 16. | Posit team. RStudio: Integrated Development Environment for R. Boston, MA: Posit Software, PBC; 2025 [cited 30 August 2025]. Available from: http://www.posit.co/. |
| 17. | Ferguson I, Bell A, Treston G, New L, Ding M, Holdgate A. Propofol or Ketofol for Procedural Sedation and Analgesia in Emergency Medicine-The POKER Study: A Randomized Double-Blind Clinical Trial. Ann Emerg Med. 2016;68:574-582.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 18. | Andolfatto G, Abu-Laban RB, Zed PJ, Staniforth SM, Stackhouse S, Moadebi S, Willman E. Ketamine-propofol combination (ketofol) versus propofol alone for emergency department procedural sedation and analgesia: a randomized double-blind trial. Ann Emerg Med. 2012;59:504-12.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 19. | Fu M, Sheng B, Liu R, Li Y, Chen G, Chen H, Chen X, Duan G, Huang H, Chen J, Chen Y. Impact of different doses of esketamine on the incidence of hypotension in propofol-based sedation for colonoscopy: a randomized controlled trial. Ther Adv Drug Saf. 2024;15:20420986241278499. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 20. | Chiaretti A, Ruggiero A, Barbi E, Pierri F, Maurizi P, Fantacci C, Bersani G, Riccardi R. Comparison of propofol versus propofol-ketamine combination in pediatric oncologic procedures performed by non-anesthesiologists. Pediatr Blood Cancer. 2011;57:1163-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Frizelle HP, Duranteau J, Samii K. A comparison of propofol with a propofol-ketamine combination for sedation during spinal anesthesia. Anesth Analg. 1997;84:1318-1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 22. | Miner JR, Moore JC, Austad EJ, Plummer D, Hubbard L, Gray RO. Randomized, double-blinded, clinical trial of propofol, 1:1 propofol/ketamine, and 4:1 propofol/ketamine for deep procedural sedation in the emergency department. Ann Emerg Med. 2015;65:479-488.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 23. | Frey K, Sukhani R, Pawlowski J, Pappas AL, Mikat-Stevens M, Slogoff S. Propofol versus propofol-ketamine sedation for retrobulbar nerve block: comparison of sedation quality, intraocular pressure changes, and recovery profiles. Anesth Analg. 1999;89:317-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 26] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Tandon M, Pandey VK, Dubey GK, Pandey CK, Wadhwa N. Addition of sub-anaesthetic dose of ketamine reduces gag reflex during propofol based sedation for upper gastrointestinal endoscopy: A prospective randomised double-blind study. Indian J Anaesth. 2014;58:436-441. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 25. | Soliman R, Mofeed M, Momenah T. Propofol versus Ketofol for Sedation of Pediatric Patients Undergoing Transcatheter Pulmonary Valve Implantation: A Double-blind Randomized Study. Ann Card Anaesth. 2017;20:313-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 26. | Kumar N, Rajan S, Kumar L. Efficacy of Ketofol in Blunting Hypotensive Effects of Propofol during Induction and Its Effect on Intraoperative Anesthetic Requirements and Recovery Profile. Anesth Essays Res. 2020;14:605-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 27. | Cui S, Huang P, Wei Z, Guo T, Zhang A, Huang L. Esketamine Combined with Propofol TCI versus Propofol TCI for Deep Sedation during Endobronchial Ultrasound-Guided Transbronchial Needle Aspiration: A Prospective, Randomized, and Controlled Trial. Int J Clin Pract. 2023;2023:1155126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 28. | Liu D, Gao X, Zhuo Y, Cheng W, Yang Y, Wu X, Yang H, Yao Y. Effect of Esketamine on Cognitive Recovery After Propofol Sedation for Outpatient Colonoscopy: A Randomized Clinical Trial. Drug Des Devel Ther. 2025;19:425-437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 29. | Zheng L, Wang Y, Ma Q, Liang W, Zhang X, Ren Z, Qin W, Meng F, Li Y, Fan G, Yin N. Efficacy and Safety of a Subanesthetic Dose of Esketamine Combined with Propofol in Patients with Obesity Undergoing Painless Gastroscopy: A Prospective, Double-Blind, Randomized Controlled Trial. Drug Des Devel Ther. 2023;17:1347-1356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 30. | Bhardwaj P, Panneerselvam S, Rudingwa P, Govindaraj K, Satya Prakash MVS, Badhe AS, Nagarajan K. Comparison of airway collapsibility following single induction dose ketamine with propofol versus propofol sedation in children undergoing magnetic resonance imaging: A randomised controlled study. Indian J Anaesth. 2024;68:189-195. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 31. | Schmitz A, Weiss M, Kellenberger C, O'Gorman Tuura R, Klaghofer R, Scheer I, Makki M, Sabandal C, Buehler PK. Sedation for magnetic resonance imaging using propofol with or without ketamine at induction in pediatrics-A prospective randomized double-blinded study. Paediatr Anaesth. 2018;28:264-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 32. | Ulutas H, Ucar M, Celik MR, Agar M, Gulcek I. Sedation with Propofol and Propofol-Ketamine (Ketofol) in Flexible Bronchoscopy: A Randomized, Double-Blind, Prospective Study. Niger J Clin Pract. 2023;26:1817-1823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 4] [Article Influence: 1.3] [Reference Citation Analysis (1)] |
| 33. | Beyoğlu ÇA, Kendigelen P, Beyoğlu İ, Altındaş F, Kuruoğlu S, Kaya G. Does magnetic resonance imaging increase core body temperature in children? Results of the administration of propofol and ketofol: a randomized clinical study. Turk J Pediatr. 2020;62:224-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 34. | Xu L, Dong Q, Jin A, Zeng S, Wang K, Yang X, Zhu X. Experience of financial toxicity and coping strategies in young and middle-aged patients with stroke: a qualitative study. BMC Health Serv Res. 2024;24:94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 35. | Mohammad Khalil A, Makram Botros J, Boules ML, Gaber Ragab S. Reliable and Rapid Smooth Extubation After "Ketamine-Propfol Mixture" for Induction of General Anesthesia in Laparoscopic Drilling of Polycystic Ovary: A Randomized, Double-blind, Comparative Study. Anesth Pain Med. 2021;11:e113919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 36. | Foo TY, Mohd Noor N, Yazid MB, Fauzi MH, Abdull Wahab SF, Ahmad MZ. Ketamine-propofol (Ketofol) for procedural sedation and analgesia in children: a systematic review and meta-analysis. BMC Emerg Med. 2020;20:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (4)] |
| 37. | Song N, Yang Y, Zheng Z, Shi WC, Tan AP, Shan XS, Liu H, Meng L, Peng K, Ji FH. Effect of Esketamine Added to Propofol Sedation on Desaturation and Hypotension in Bidirectional Endoscopy: A Randomized Clinical Trial. JAMA Netw Open. 2023;6:e2347886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 60] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 38. | Choi EJ, Kim CH, Yoon JY, Kim EJ. Ketamine-propofol (ketofol) in procedural sedation: a narrative review. J Dent Anesth Pain Med. 2023;23:123-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 10] [Reference Citation Analysis (0)] |
| 39. | Kawczak P, Feszak I, Bączek T. Ketamine, Esketamine, and Arketamine: Their Mechanisms of Action and Applications in the Treatment of Depression and Alleviation of Depressive Symptoms. Biomedicines. 2024;12:2283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 34] [Reference Citation Analysis (0)] |
| 40. | Feeney A, Papakostas GI. Pharmacotherapy: Ketamine and Esketamine. Psychiatr Clin North Am. 2023;46:277-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 31] [Reference Citation Analysis (0)] |
| 41. | Hwang J, Jeon Y, Park HP, Lim YJ, Oh YS. Comparison of alfetanil and ketamine in combination with propofol for patient-controlled sedation during fiberoptic bronchoscopy. Acta Anaesthesiol Scand. 2005;49:1334-1338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 42. | Tomatir E, Atalay H, Gurses E, Erbay H, Bozkurt P. Effects of low dose ketamine before induction on propofol anesthesia for pediatric magnetic resonance imaging. Paediatr Anaesth. 2004;14:845-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 49] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 43. | Xu C, Wei X, Zhang C, Huang X, Lan H, Xu Y, Wu X, Li F, Guan X. Esketamine prevents propofol-induced injection pain: Randomized controlled trial. Front Pharmacol. 2022;13:991559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 44. | Makkey M, Baker M, Ibrahim N. Hemodynamic stability of ketamine/propofol admixture ketofol in patients undergoing endoscopic retrograde cholangiopancreatography. J Curr Med Res Pract. 2018;3:43. [DOI] [Full Text] |
| 45. | Elsaeidy AS, Ahmad AHM, Kohaf NA, Aboutaleb A, Kumar D, Elsaeidy KS, Mohamed OS, Kaye AD, Shehata IM. Efficacy and Safety of Ketamine-Dexmedetomidine Versus Ketamine-Propofol Combination for Periprocedural Sedation: A Systematic Review and Meta-analysis. Curr Pain Headache Rep. 2024;28:211-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 14] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 46. | Yang S, Wang J, Li X, Wang T, Xu Z, Xu X, Zhou X, Chen G. Adverse Effects of Esketamine for the Treatment of Major Depression Disorder: Findings from Randomized Controlled Trials. Psychiatr Q. 2022;93:81-95. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 47. | Powers AR 3rd, Gancsos MG, Finn ES, Morgan PT, Corlett PR. Ketamine-Induced Hallucinations. Psychopathology. 2015;48:376-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 88] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 48. | Mortero RF, Clark LD, Tolan MM, Metz RJ, Tsueda K, Sheppard RA. The effects of small-dose ketamine on propofol sedation: respiration, postoperative mood, perception, cognition, and pain. Anesth Analg. 2001;92:1465-1469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 109] [Article Influence: 4.4] [Reference Citation Analysis (0)] |