Published online Mar 16, 2026. doi: 10.12998/wjcc.v14.i8.118134
Revised: January 17, 2026
Accepted: February 12, 2026
Published online: March 16, 2026
Processing time: 81 Days and 22.5 Hours
Musculoskeletal manifestations are the most common extraintestinal manifestations of pediatric inflammatory bowel disease (IBD). However, the coexistence of Crohn’s disease (CD) with chronic non-bacterial osteomyelitis (CNO) and axial spondyloarthropathy creates a complex autoinflammatory phenotype that is challenging to manage and prone to severe structural damage.
We report the longitudinal course of a male patient with CD and progressive skeletal involvement. Despite sequential trials of adalimumab and infliximab (both discontinued due to anaphylaxis) and vedolizumab, the patient developed extensive cervical-occipital fusion and C6-C7 pseudoarthrosis. At age 15, whole-body magnetic resonance imaging (WB-MRI) was pivotal in identifying multifocal periphyseal bone marrow edema (BME) characteristic of concomitant CNO, which had been missed by targeted spinal imaging. Genetic testing identified a heterozygous nucleotide-binding oligomerization domain-containing protein 2 p.Leu762Phe variant. Following the diagnosis of this phenotype, treatment was intensified to tofacitinib and intravenous zoledronic acid. A follow-up WB-MRI at age 16 demonstrated a marked reduction in multifocal BME and resolution of active inflammatory lesions, though structural spinal fusion remained permanent.
This case suggests that in pediatric patients with IBD with refractory skeletal pain, clinicians might consider the possibility of overlapping autoinflammatory syndromes. While based on a single observation, the use of WB-MRI was instrumental in identifying subclinical lesions, assisting in the differentiation between active inflammation and permanent structural damage.
Core Tip: Pediatric Crohn’s disease can coexist with chronic non-bacterial osteomyelitis and spondyloarthropathy, creating a severe autoinflammatory phenotype linked to nucleotide-binding oligomerization domain-containing protein 2 variants. This case demonstrates that persistent skeletal pain in inflammatory bowel disease patients requires a high index of suspicion for multifocal bone involvement. Whole-body magnetic resonance imaging should be considered for mapping total inflammatory load and preventing irreversible structural damage, such as cervical fusion. Management often necessitates a multi-target approach, utilizing Janus kinase inhibitors and bisphosphonates to address refractory systemic inflammation and preserve skeletal integrity.
- Citation: Triantafyllou G, Prountzos S, Bolou K, Arkoudis NA, Liontou K, Piagkou M, Fotis L, Papakonstantinou O. Value of whole-body magnetic resonance imaging on progressive axial fusion and chronic non-bacterial osteomyelitis in pediatric Crohn’s disease: A case report. World J Clin Cases 2026; 14(8): 118134
- URL: https://www.wjgnet.com/2307-8960/full/v14/i8/118134.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i8.118134
The association between inflammatory bowel disease (IBD) and musculoskeletal manifestations is well-documented in pediatric populations, typically presenting as peripheral arthritis or axial spondyloarthropathy[1,2]. However, the clinical landscape becomes significantly more complex when overlapping autoinflammatory conditions coexist with Crohn’s disease (CD). When these conditions overlap, patients often face a severe disease course characterized by progressive structural damage, such as spinal fusion, and profound systemic complications including severe growth failure and osteoporosis[3,4].
Herein, this case illustrates the longitudinal diagnostic and unusual cervical-occipital fusion of a young patient carrying a nucleotide-binding oligomerization domain-containing protein 2 (NOD2) variant, highlighting the critical role of advanced imaging in managing complex pediatric systemic inflammation.
A 13-year-old male patient was referred to our Pediatric Rheumatology Unit for persistent back and neck pain, with multiple imaging examinations. He had a confirmed diagnosis of CD since age 7. The interesting imaging characteristics by chronological order are further presented.
Growth failure was a persistent feature of the clinical course. At age 7, the patient’s height was 122 cm (Z-score -0.42). By age 13, despite treatments, his height Z-score had progressively declined to -2.13 (140 cm), and his weight Z-score dropped from -1.85 to -2.31, confirming severe persistent growth impairment.
At age 15, whole-body magnetic resonance imaging (WB-MRI) was pivotal in identifying multifocal periphyseal bone marrow edema (BME) characteristic of concomitant CNO, which had been missed by targeted spinal imaging. Genetic testing identified a heterozygous NOD2 p.Leu762Phe variant.
Not applicable.
Not applicable.
Not applicable.
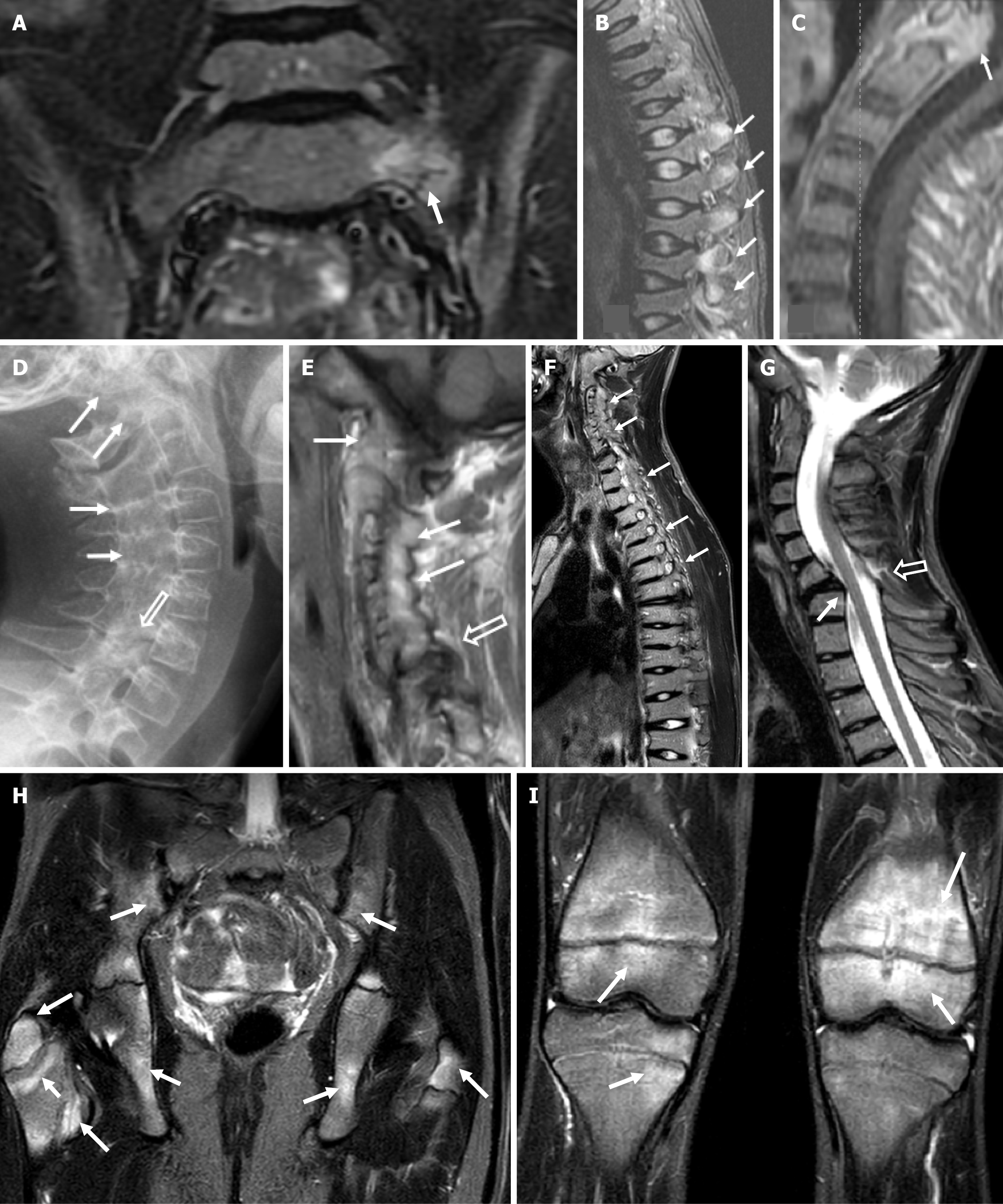
At age 7, an initial magnetic resonance imaging (MRI) performed for low-back pain revealed BME on the iliac side of the sacroiliac joint and a small stress fracture of the left sacral wing, that was initially undetected (Figure 1A).
At age 10, spine MRI demonstrated reduced vertebral height with normal signal intensity of vertebral bodies, multifocal BME of the posterior elements (Figure 1B) and pannus formation around the dens, suggesting inflammatory spondyloarthropathy (Figure 1C).
Sacral stress fracture and reduced vertebral height were attributed to osteoporosis due to malabsorption of CD.
At age 14, X-ray and spinal MRI showed progressive ossification and fusion of the cervical spine and occiput with pseudoarthrosis at the C6-C7 level (Figure 1D and E). Bisphosphonate and anti-inflammatory therapy were administered.
WB-MRI, performed at age 15, for diffuse skeletal pain demonstrated further progression of fusion of posterior elements of the spine (Figure 1F), prolapse of the C6-C7 intervertebral disk, at the level of pseudoarthrosis (Figure 1G) and levoscoliosis of the lumbar spine; however BME of the posterior elements had subsided whereas the height of thoracic and lumbar vertebral bodies was significantly restored. In addition, multiple areas of BME in the peripheral skeleton were seen, most of them in periphyseal distribution, suggestive of concomitant chronic non-bacterial osteomyelitis (CNO) (Figure 1H and I).
The final diagnosis is CD with concomitant CNO and IBD-associated spondyloarthropathy. Genetic testing via whole-exome sequencing further identified a heterozygous NOD2 p.Leu762Phe variant.
The WB-MRI was a pivotal diagnostic turning point. While previous targeted spinal MRIs suggested that the disease was predominantly structural, the WB-MRI revealed widespread, subclinical active inflammation in the peripheral metaphyses.
The treatment course for this patient was complex and required multiple adjustments due to disease progression and medication intolerance. Initially, at age 7, the patient received a combination of adalimumab (anti-tumor necrosis factor [TNF]), azathioprine (AZA), and a 2-month course of corticosteroids, but adalimumab was discontinued following an anaphylactic reaction. A subsequent attempt with infliximab (anti-TNF) plus AZA also ended due to an anaphylactic response, leading to a period of AZA monotherapy followed by 1 year of clinical remission without treatment. Upon the recurrence of symptoms at age 12, the patient was treated with vedolizumab and AZA, then switched to vedolizumab and methotrexate (MTX) due to a lack of clinical response. Following the identification of a NOD2 p.Leu762Phe variant and the diagnosis of CNO, therapy was intensified in June 2023 to include tofacitinib (a Janus kinase [JAK] inhibitor), MTX, and intravenous bisphosphonates (zoledronic acid) to manage the multifocal skeletal lesions and severe low bone mass.
Repeat WB-MRI at age 16, revealed a global reduction of the inflammatory load with no change of the structural changes after stable structural deformities of the spine.
The longitudinal comparison between WB-MRI at age 15 and age 16 depicted a clear distinction. Active inflammatory lesions, characterized by T2-weighted short tau inversion recovery hyperintensity, demonstrated significant response to therapy. Conversely, structural lesions involving ossification and remodeling showed no regression.
This imaging case illustrates the early onset and progressive course of IBD-associated spondyloarthropathy, with striking cervical-occipital fusion, a pattern rarely reported in childhood. The association between IBD and musculoskeletal manifestations is a well-recognized challenge in pediatric rheumatology and gastroenterology. This case underscores the complexity of managing a patient with an overlapping phenotype of CD, IBD-associated spondyloarthritis (SpA), and CNO[1].
Musculoskeletal disorders are the most frequent extraintestinal manifestations in children with IBD, affecting approximately 16% to 33% of patients[1]. Recent longitudinal cohort data suggests that children and adolescents diagnosed with IBD face a significantly increased risk of developing SpA and psoriatic arthritis, with these conditions often manifesting within 10 years of the initial IBD onset[2]. While peripheral arthritis in IBD often correlates with intestinal disease activity, axial involvement, such as sacroiliitis and spinal inflammation, frequently follows a course independent of gut flares[1].
The coexistence of CNO and IBD, as seen in this patient, is a documented clinical entity that suggests shared autoinflammatory pathways. CNO is a sterile, non-infectious inflammatory bone disease that primarily affects the metaphyses of long bones but can involve any skeletal site[5]. While IBD primarily involves mucosal immune dysregulation, both conditions involve cytokine imbalances, genetic predispositions, and potential microbiome influences. In pediatric populations, CD appears more frequently associated with CNO than ulcerative colitis[6].
Advanced imaging was pivotal in this patient’s longitudinal care, particularly the transition to WB-MRI. Traditional “targeted” MRI may miss subclinical inflammation foci, leading to an underestimation of the total disease burden. In contrast, WB-MRI provides comprehensive coverage of both the axial and peripheral skeleton in a single examination[3]. WB-MRI excels at identifying asymptomatic inflammatory lesions, which is crucial for conditions like CNO and SpA that often involve multifocal sites[4]. The lack of ionizing radiation makes WB-MRI an ideal modality for repeated monitoring of systemic inflammation and treatment response in sensitive pediatric cohorts[4]. It also allows for the objective assessment of both skeletal and extra-skeletal inflammation, facilitating a more accurate evaluation of “inflammatory load” vs permanent structural damage[4].
The identification of a NOD2 variant in this patient is highly relevant to the clinical presentation. NOD2 (previously CARD15) is an established susceptibility gene for CD, encoding a protein that functions as an intracellular receptor for bacterial products[5]. Research has hypothesized that NOD2 variants may contribute to an “extended phenotype” of CD that includes non-bacterial bone inflammation. While NOD2 variants are common in CD, their specific role in predisposing patients to co-occurring CNO suggests a shared autoinflammatory process involving the innate immune system[5].
Management of overlapping IBD and CNO requires a multi-target approach, often complicated by treatment in
There are several limitations to this report. Primarily, as a single-patient case study, the clinical outcomes and the observed association between the NOD2 variant and the ‘triple overlap’ of CD, SpA, and CNO cannot be generalized to all patients. Furthermore, while the radiological resolution of bone marrow edema was clear following the initiation of tofacitinib and bisphosphonates, the specific contribution of each agent to the systemic improvement is difficult to isolate in a single subject. These observations should be considered hypothesis-generating, highlighting the need for larger cohort studies to investigate the prevalence and optimal management of this extended autoinflammatory phenotype.
In conclusion, this case highlights that when pediatric IBD presents with severe or persistent skeletal pain, a high index of suspicion for concomitant SpA or CNO is required. Utilizing WB-MRI early in the diagnostic workup allows for a comprehensive assessment of systemic inflammation, while genetic testing can provide insights into the underlying autoinflammatory drivers, ultimately guiding personalized and intensified therapeutic strategies. Nevertheless, it is important to highlight the collaborative work of several specialties (rheumatologists, pediatricians, gastroenterologists, radiologists) to ensure the maximum outcomes of such complex cases.
| 1. | Cardile S, Romano C. Current issues in pediatric inflammatory bowel disease-associated arthropathies. World J Gastroenterol. 2014;20:45-52. [PubMed] [DOI] [Full Text] |
| 2. | Jølving LR, Zegers FD, Lund K, Wod M, Nielsen J, Qvist N, Nielsen RG, Nørgård BM. Children and Adolescents Diagnosed With Inflammatory Bowel Disease Are at Increased Risk of Developing Diseases With a Possible Autoimmune Pathogenesis. Inflamm Bowel Dis. 2025;31:87-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 3. | Choida V, Madenidou AV, Sen D, Hall-Craggs MA, Ciurtin C. The role of whole-body MRI in musculoskeletal inflammation detection and treatment response evaluation in inflammatory arthritis across age: A systematic review. Semin Arthritis Rheum. 2022;52:151953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 4. | Zecca F, Dejua D, Rennie WJ, Saba L. Whole-body MRI in arthritis. Skeletal Radiol. 2025;54:2517-2539. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 5. | Morbach H, Dick A, Beck C, Stenzel M, Müller-Hermelink HK, Raab P, Girschick HJ. Association of chronic non-bacterial osteomyelitis with Crohn's disease but not with CARD15 gene variants. Rheumatol Int. 2010;30:617-621. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 6. | Tzaneti A, Athanasopoulou E, Fessatou S, Fotis L. Chronic Nonbacterial Osteomyelitis in Inflammatory Bowel Disease. Life (Basel). 2023;13:2347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 7. | Fujisaki T, Matsuishi T, Kamizono J, Amamoto M, Mizuochi T. Crohn's disease and chronic recurrent multifocal osteomyelitis in a Japanese boy. Pediatr Int. 2021;63:115-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 8. | Song K, Satsangi J, Coates LC. Arthritis complicating inflammatory bowel disease- the future is now. Lancet Rheumatol. 2024;6:e805-e810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (1)] |