Published online May 26, 2026. doi: 10.12998/wjcc.v14.i15.120825
Revised: April 1, 2026
Accepted: April 22, 2026
Published online: May 26, 2026
Processing time: 61 Days and 19.6 Hours
Dupilumab has shown therapeutic efficacy in bullous pemphigoid (BP) by blocking interleukin (IL)-4/IL-13 signaling; inducing a type 22 immune signature with increased AHR signaling, keratinocyte activation and IL-22 receptor upregulation; and indirectly regulating STAT3 pathway activity to reduce the need for systemic immunosuppressants and glucocorticoids. The pathogenesis of Kaposi sarcoma (KS), a rare BP complication, is closely associated with human her
An 87-year-old female was diagnosed with BP based on clinical, histopathological, and immunological findings. Good control was achieved with low-dose oral prednisone, dupilumab, and topical halometasone-triclosan cream. After 32 weeks of treatment, she developed violaceous-red papules on both feet, which were confirmed as KS by skin biopsy and immunohistochemistry. Human immunodeficiency virus, syphilis, systemic disease, and internal malignancy were excluded. The patient declined laser/cryotherapy for KS due to advanced age and poor mobility, and was treated with topical 5% imiquimod cream. At the 6-month follow-up, no new KS lesions occurred, with partial regression of existing lesions. BP remained well controlled under regular surveillance.
Further research and follow-up are needed to clarify BP-KS association and dupilumab’s safety.
Core Tip: Dupilumab was used to treat bullous pemphigoid (BP) complicated with Kaposi sarcoma (KS). The causes were analyzed, and the immune drift induction hypothesis was proposed. STAT3 plays a key role in KS pathogenesis, as it supports human herpesvirus-8 latency and viral replication. It can be activated by virally encoded interleukin-6 via gp130 receptor signaling to promote the survival and proliferation of infected cells. Dupilumab-induced immune deviation (toward Th1/Th17/Th22 pathways) and STAT3 signaling activation may contribute to KS occurrence in susceptible BP patients. Further investigation is needed.
- Citation: Wang SN, Wang JM, Xu M, Wu JJ. Kaposi sarcoma following dupilumab treatment for bullous pemphigoid: A case report and review of literature. World J Clin Cases 2026; 14(15): 120825
- URL: https://www.wjgnet.com/2307-8960/full/v14/i15/120825.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i15.120825
Bullous pemphigoid (BP) is an autoimmune subepidermal blistering disease that predominantly affects elderly in
Dupilumab, a monoclonal antibody targeting interleukin (IL)-4 receptor alpha, blocks IL-4 and IL-13 signaling and has shown promise in the treatment of BP[5-7]. By suppressing type 2 inflammation, dupilumab may reduce the need for systemic corticosteroids and other immunosuppressive agents. This could lower the risk of treatment-related adverse events in elderly patients.
Kaposi sarcoma (KS) is a rare vascular endothelial tumor associated with human herpesvirus-8 (HHV-8) infection. Its incidence increases in immunocompromised individuals, including those receiving immunosuppressive therapy for autoimmune blistering diseases[8]. Here, we report a case of KS developing several months after initiating dupilumab therapy in a patient with confirmed BP and discuss the possible clinical association.
An 87-year-old woman complained of widespread vesicles and bullae for 9 months, and multiple tender erythematous papules on both feet for 32 weeks.
After 32 weeks of combination treatment with prednisone and dupilumab, the patient developed multiple tender erythematous papules on both feet.
She denied any previous history of infectious diseases or malignancy.
No relevant personal or family history was documented.
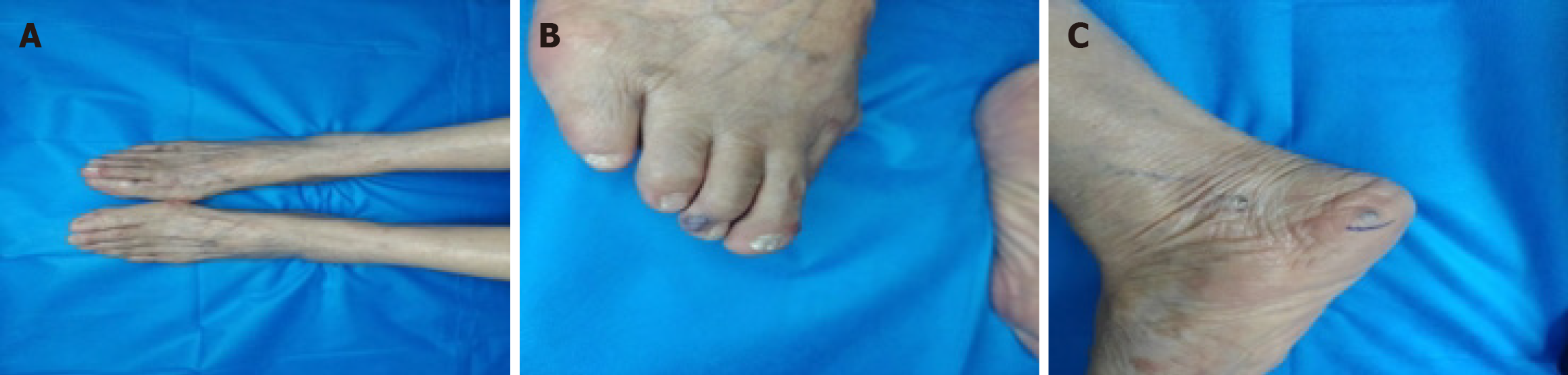
Physical examination revealed multiple tense vesicles and bullae on the trunk and extremities, with thickened walls and a negative Nikolsky sign. There were > 10 discrete, hemispherical, violaceous-red papules on both feet (Figure 1). There were no signs of stasis dermatitis or varicose changes in the lower extremities.
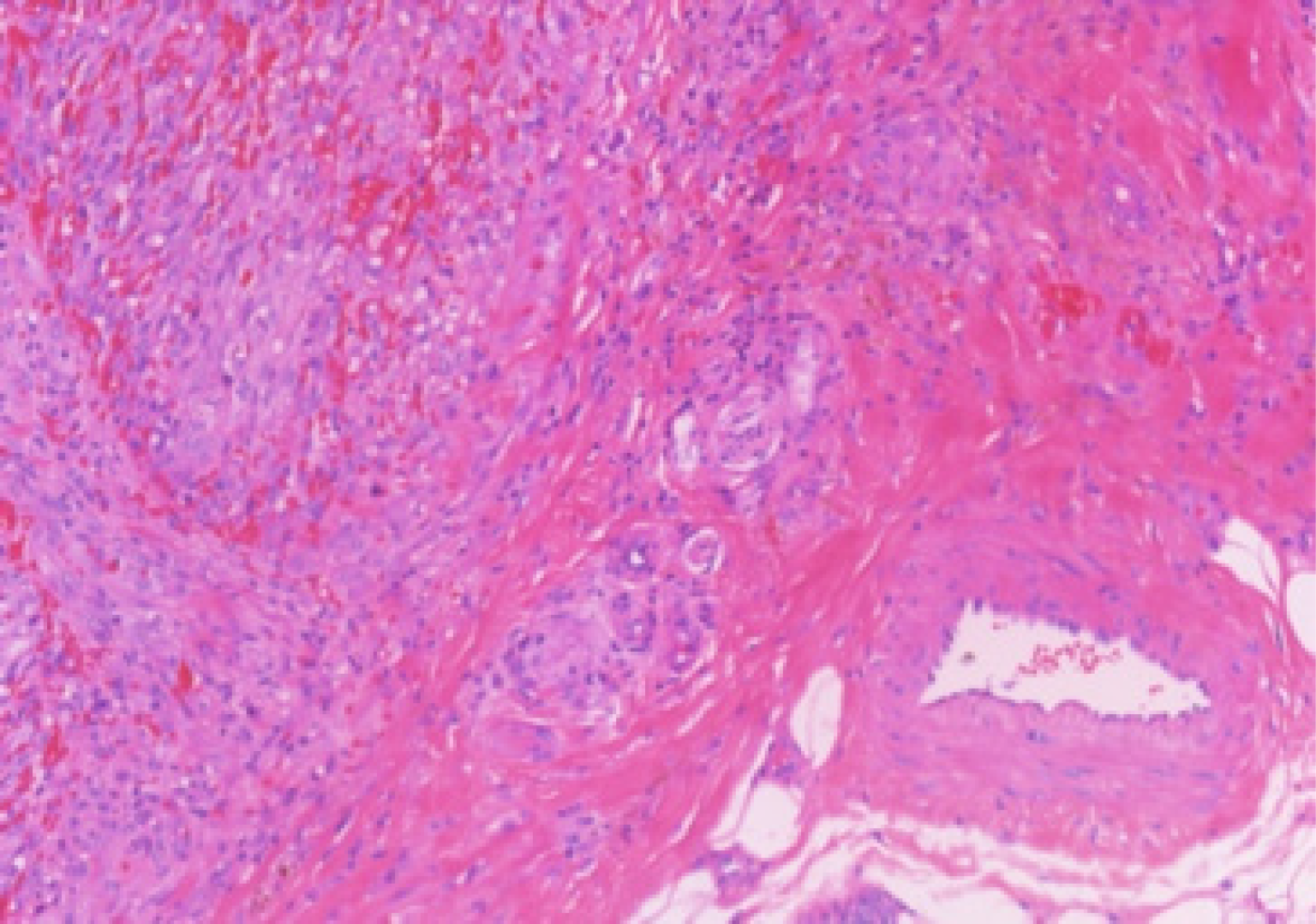
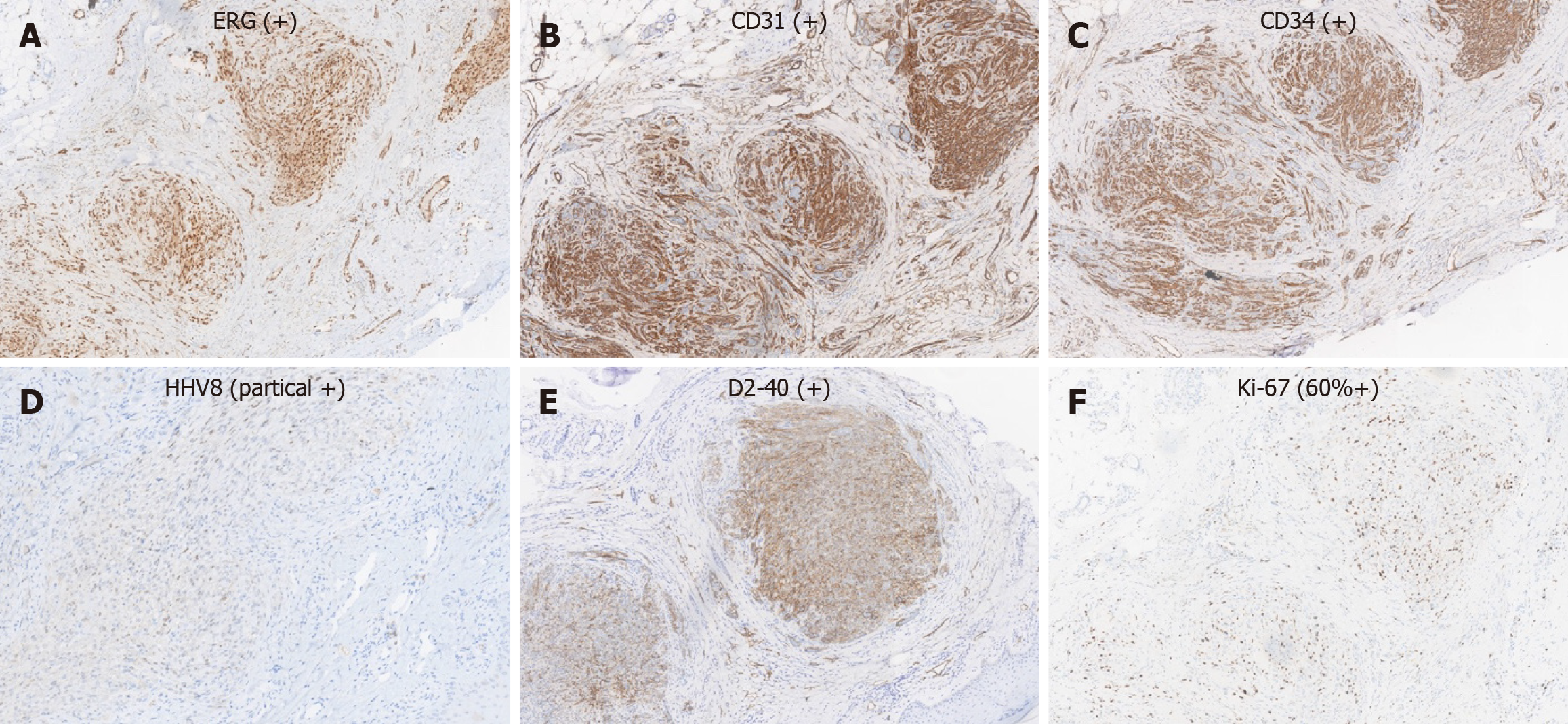
Laboratory testing was negative for human immunodeficiency virus (HIV) and syphilis (Treponema pallidum particle agglutination assay/toluidine red unheated serum test), and routine laboratory parameters were within normal limits. Skin biopsy obtained from a fresh blister on the trunk demonstrated subepidermal blister formation with eosinophilic infiltration, consistent with BP. Punch biopsy obtained from a papule on the right heel revealed vascular proliferation suggestive of KS (Figure 2). Immunohistochemical analysis showed tumor cells positive for erythroblast transformation specific related gene, CD31, CD34, HHV-8 (partial), D2-40, and Ki-67 (60%+) (Figure 3).
Whole-body computed tomography and gastrointestinal endoscopy showed no evidence of systemic abnormalities or malignancy. Dermoscopic examination of the lesions on both feet demonstrated purplish–red lacunar structures.
A final diagnosis of BP with KS was established.
Initial treatment for BP was oral prednisone (initial dose 15 mg/day for 2 weeks, followed by gradual tapering to 5 mg/day and then reduced to 2.5 mg every other day after 24 weeks as maintenance therapy) in combination with dupilumab (loading dose 600 mg administered subcutaneously, followed by 300 mg every 2 weeks). Halometasone-triclosan cream was applied topically to affected areas.
The patient developed multiple tender erythematous papules on both feet. After 32 weeks of treatment, BP remains well controlled with low-dose prednisone (2.5 mg every other day) and dupilumab (300 mg every 2 weeks). For the KS lesions on the feet, carbon dioxide laser therapy or cryotherapy was recommended; however, these options were declined by the patient’s family due to the patient’s advanced age and limited mobility. Instead, topical 5% imiquimod cream was initiated.
At the 6-month follow-up visit, BP was stable, with almost no new blisters. Regarding KS, the patient experienced local erythema and pruritus during imiquimod use, which was tolerable. At the latest follow-up, no new foot lesions were observed. The smaller rashes had basically resolved, and the larger ones had decreased in size. The patient remains under regular clinical observation.
BP is an autoimmune subepidermal blistering disorder that primarily affects elderly individuals and is characterized clinically by tense blisters on normal or erythematous skin[1]. BP is associated with a range of complications[9,10] and may become life-threatening in severe cases, highlighting the need for careful and individualized management. A review of previously reported BP-associated complications identified several cases of KS occurring in patients with BP, providing a relevant clinical context for this case report.
KS is a rare multifocal vascular tumor caused by KS-associated herpesvirus (KSHV/HHV-8) and is traditionally classified into classic, African endemic, iatrogenic, and acquired immunodeficiency syndrome-associated forms[8,11,12]. Reported cases of BP with KS frequently involve patients with underlying immunosuppression, including HIV infection, chronic comorbidities (e.g., diabetes, hypertension, and chronic kidney disease)[13], or long-term exposure to immuno
Dupilumab was initiated in combination with low-dose oral prednisone to minimize prolonged systemic corticosteroid exposure in our elderly patient. Corticosteroids were rapidly tapered to a minimal maintenance dose. The patient had no history of HIV infection, major chronic comorbidities, or previous immunosuppressive therapy. However, approximately 8 months after treatment initiation, progressive violaceous papules developed on the feet and were then diagnosed as KS. HHV-8 serological indicators were not tested either at baseline or after the diagnosis of KS, and the potential for latent HHV-8 infection or reactivation cannot be ruled out. This constitutes a limitation of the present study. Although concomitant STS use cannot be completely excluded as a contributing factor, current evidence linking STS, alone or in combination with low-dose systemic corticosteroids, to KS development remains limited. To our knowledge, no reports have described KS occurring after dupilumab treatment in patients with BP, suggesting a potential association.
Dupilumab is a fully human monoclonal antibody that targets the IL-4 receptor alpha, blocking IL-4 and IL-13 signaling. This mechanism downregulates eosinophil recruitment and suppresses B-cell activation[16,17]. The Th2 immune response plays a key role in BP pathogenesis; elevated levels of IL-4 and IL-13 derived from CD4+ and CD8+ T cells have been detected in both blister fluid and serum, accompanied by increased serum immunoglobulin E levels and eosinophilia[18-20]. These findings support the therapeutic rationale for dupilumab in BP. In clinical practice, dupilumab is often combined with conventional therapies such as corticosteroids or immunosuppressants. This may shorten treatment duration and reduce cumulative steroid exposure, lowering the risk of adverse events. Serious adverse events associated with dupilumab are relatively uncommon and mainly include soft tissue infections, conjunctivitis, psoria
Following reports of dupilumab-associated head and neck dermatitis, Bangert et al[23] investigated the underlying immunological mechanisms and found that while dupilumab normalized downstream IL-4/IL-13 markers (CCL13, CCL17, CCL18, and CCL26), it also induced a pronounced type 22 immune signature characterized by increased IL-22 and aryl hydrocarbon receptor signaling, keratinocyte activation, and upregulation of IL-22 receptors. These findings suggest that inhibition of the Th2/Tc2 pathway by dupilumab may trigger compensatory immune shifts toward the Th1, Th17, and Th22 pathways. The occurrence of psoriasiform eruptions, conjunctivitis, and arthritis during dupilumab therapy further supports the hypothesis of immune deviation[24].
From a systems biology perspective, immune regulation involves complex signaling pathways and gene regulatory networks rather than isolated pathways. Whether through IL-4/IL-13 blockade or compensatory activation of IL-17 and IL-22 pathways, dupilumab-modulated signals are transmitted via modulation of the STAT3 pathway, leading to diverse biological effects[25]. One downstream outcome of STAT3 activation is increased IL-6 signaling, which can promote pro-inflammatory and pro-tumorigenic microenvironments. In KS, KSHV infection induces endothelial cells to produce cytokines such as IL-6, IL-10, and IL-13, which drive monocyte differentiation and polarization into tumor-associated macrophages that shape the tumor microenvironment. Tumor-associated macrophages can be polarized into M1-like (pro-inflammatory) and M2-like macrophages. The latter inhibits tumor immunity via IL-10 secretion and promotes tumorigenesis by activating the STAT3 signaling pathway[26]. STAT3, a positive regulator of viral productive replication, is activated in endothelial and primary effusion lymphoma cells during de novo and latent KSHV infection. This is critical for maintaining latently infected cell viability and efficient viral replication. The KSHV-encoded IL-6 homologue, frequently detected in latently infected primary effusion lymphoma cells, is key to KSHV-associated disease pathogenesis and immune escape[27,28]. It’s signaling through the gp130 receptor is the main mechanism of STAT3 activation[29]. Additionally, the KSHV-encoded IL-6 homologue promotes tumor angiogenesis by upregulating VEGF expression[30]. In summary, STAT3-related pathways play an important role in KS development.
Based on these observations, dupilumab-induced immune modulation could alter the local immune microenvironment via STAT3 signaling pathways, potentially facilitating KS development in susceptible individuals. However, given the complexity of immune regulatory networks, such effects are likely affected by multiple genetic and environmental factors rather than a single pathway. Furthermore, the uniqueness of this case does not exclude the possibility of individual genetic susceptibility. Due to the lack of verification of previous latent infection or viral reactivation of KSHV/HHV-8 in this case, the possibility that viral activation induces tumors cannot be ruled out. Therefore, the proposed mechanism remains speculative and requires further mechanistic investigation.
The association between BP and KS in this clinical context remains incompletely understood. It should be emphasized that the immune drift hypothesis proposed in this case is speculative and requires further exploration and verification. In this single case, we cannot rule out potential effects of low-dose systemic glucocorticoids, topical glucocorticoids, advanced age, and pre-existing latent HHV-8 infection. The evaluation criteria and impact of these factors require further verification. The potential contributions of immunosuppressive therapies (e.g., systemic and topical corticosteroids) according to their cumulative doses and treatment durations also warrant further investigation. Continued clinical observation and additional reports will be important for elucidating the long-term safety profile of dupilumab and optimizing therapeutic strategies for BP.
| 1. | Karakioulaki M, Eyerich K, Patsatsi A. Advancements in Bullous Pemphigoid Treatment: A Comprehensive Pipeline Update. Am J Clin Dermatol. 2024;25:195-212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 36] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 2. | Seyed Jafari SM, Feldmeyer L, Bossart S, Simon D, Schlapbach C, Borradori L. Case Report: Combination of Omalizumab and Dupilumab for Recalcitrant Bullous Pemphigoid. Front Immunol. 2020;11:611549. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 3. | James T, Salman S, Stevenson B, Bundell C, Kelly G, Nolan D, John M. IgE blockade in autoimmunity: Omalizumab induced remission of bullous pemphigoid. Clin Immunol. 2019;198:54-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 4. | Vico-Alonso C, Calleja-Algarra A, Aragón-Miguel R, Sánchez-Velázquez A, Velasco-Tamariz V, Ortiz-Romero PL, Monsálvez-Honrubia V. Omalizumab as an alternative therapeutic tool in the treatment of bullous pemphigoid: A case report. Dermatol Ther. 2019;32:e12829. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 5. | Kaye A, Gordon SC, Deverapalli SC, Her MJ, Rosmarin D. Dupilumab for the Treatment of Recalcitrant Bullous Pemphigoid. JAMA Dermatol. 2018;154:1225-1226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 78] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 6. | Saleh M, Reedy M, Torok H, Weaver J. Successful treatment of bullous pemphigoid with dupilumab: a case and brief review of the literature. Dermatol Online J. 2021;27:13030/qt0dv3f9h6. [PubMed] [DOI] [Full Text] |
| 7. | Seidman JS, Eichenfield DZ, Orme CM. Dupilumab for bullous pemphigoid with intractable pruritus. Dermatol Online J. 2019;25:13030/qt25q9w6r9. [PubMed] [DOI] [Full Text] |
| 8. | Tourlaki A, Genovese G, Guanziroli E, Scoppio BM, Berti E, Brambilla L. Autoimmune bullous diseases in non-HIV Kaposi’s sarcoma: a retrospective study in a large cohort of patients. J Eur Acad Dermatol Venereol. 2018;32:1777-1783. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Huttelmaier J, Benoit S, Goebeler M. Comorbidity in bullous pemphigoid: up-date and clinical implications. Front Immunol. 2023;14:1196999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 10. | Pratasava V, Sahni VN, Suresh A, Huang S, Are A, Hsu S, Motaparthi K. Bullous Pemphigoid and Other Pemphigoid Dermatoses. Medicina (Kaunas). 2021;57:1061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 11. | Chen SA, Wang LT, Chen ZH, Yang FP, Zhu QY, Chen HY, Luo XQ. Extensive cutaneous iatrogenic Kaposi’s sarcoma after bullous pemphigoid treatment with oral methylprednisolone: a rare Chinese case report. J Int Med Res. 2021;49:300060520982829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Lopes AO, Marinho PDN, Medeiros LDS, de Paula VS. Human Gammaherpesvirus 8 Oncogenes Associated with Kaposi’s Sarcoma. Int J Mol Sci. 2022;23:7203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 13. | Kooner A, Anthony MR, Gandhi D, Dutt A, Dolehide C. Balancing the Treatment of Kaposi Sarcoma and Bullous Pemphigoid: A Therapeutic Challenge in a 63-Year-Old Male. Cureus. 2025;17:e77708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Wang T, Li C, Yang C, Cui H, Fu X, Wen L, Cui Y, Yang S, Zhang X. Identification of human herpesvirus-8 in Kaposi’s sarcoma with bullous pemphigoid. Chin Med J (Engl). 2014;127:786-788. [PubMed] [DOI] [Full Text] |
| 15. | Binois R, Nadal M, Esteve E, De Muret A, Kerdraon R, Gheit T, Tommasino M, Gaboriaud P, Touze A, Samimi M. Cutaneous Kaposi sarcoma during treatment with superpotent topical steroids and methotrexate for bullous pemphigoid: three cases. Eur J Dermatol. 2017;27:369-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 16. | Klepper EM, Robinson HN. Dupilumab for the treatment of nivolumab-induced bullous pemphigoid: a case report and review of the literature. Dermatol Online J. 2021;27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 36] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 17. | Oren-Shabtai M, Mimouni D, Nosrati A, Atzmony L, Kaplan B, Barzilai A, Baum S. Biological treatment for bullous pemphigoid. Front Immunol. 2023;14:1157250. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 12] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Liang G, Qian H, Sun C, Zhang H, Li Z, Li S, Jing K, Zhao C, Wang Y, Xiang R, Li X, Feng S. Dupilumab, corticosteroids and their combination for the treatment of bullous pemphigoid. An Bras Dermatol. 2025;100:243-252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | da Silva JON, E Silva RR, Zattar Ribeiro PV, Farah PS, Steglich RB. Efficacy and safety of dupilumab in patients with moderate-to-severe bullous pemphigoid: a systematic review and meta-analysis. An Bras Dermatol. 2025;100:429-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 20. | Yan T, Xie Y, Liu Y, Shan Y, Wu X, Wang J, Zuo YG, Zhang Z. Dupilumab effectively and rapidly treats bullous pemphigoid by inhibiting the activities of multiple cell types. Front Immunol. 2023;14:1194088. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 21. | Bridgewood C, Wittmann M, Macleod T, Watad A, Newton D, Bhan K, Amital H, Damiani G, Giryes S, Bragazzi NL, McGonagle D. T Helper 2 IL-4/IL-13 Dual Blockade with Dupilumab Is Linked to Some Emergent T Helper 17‒Type Diseases, Including Seronegative Arthritis and Enthesitis/Enthesopathy, but Not to Humoral Autoimmune Diseases. J Invest Dermatol. 2022;142:2660-2667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 93] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 22. | Narla S, Silverberg JI, Simpson EL. Management of inadequate response and adverse effects to dupilumab in atopic dermatitis. J Am Acad Dermatol. 2022;86:628-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 66] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 23. | Bangert C, Alkon N, Chennareddy S, Arnoldner T, Levine JP, Pilz M, Medjimorec MA, Ruggiero J, Cohenour ER, Jonak C, Damsky W, Griss J, Brunner PM. Dupilumab-associated head and neck dermatitis shows a pronounced type 22 immune signature mediated by oligoclonally expanded T cells. Nat Commun. 2024;15:2839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 43] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 24. | Matucci-Cerinic C, Viglizzo G, Pastorino C, Corcione A, Prigione I, Bocca P, Bustaffa M, Cecconi M, Gattorno M, Volpi S. Remission of eczema and recovery of Th1 polarization following treatment with Dupilumab in STAT3 hyper IgE syndrome. Pediatr Allergy Immunol. 2022;33:e13770. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (1)] |
| 25. | Yang Z, Xiang Q, Nicholas J. Direct and biologically significant interactions of human herpesvirus 8 interferon regulatory factor 1 with STAT3 and Janus kinase TYK2. PLoS Pathog. 2023;19:e1011806. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 26. | Neumeyer S, Tagawa T. The Kaposi sarcoma herpesvirus control of monocytes, macrophages, and the tumour microenvironment. Virology. 2025;601:110286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 27. | Liang C, Lee JS, Jung JU. Immune evasion in Kaposi’s sarcoma-associated herpes virus associated oncogenesis. Semin Cancer Biol. 2008;18:423-436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 58] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 28. | Lee HR, Brulois K, Wong L, Jung JU. Modulation of Immune System by Kaposi’s Sarcoma-Associated Herpesvirus: Lessons from Viral Evasion Strategies. Front Microbiol. 2012;3:44. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 56] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 29. | Chatterjee M, Osborne J, Bestetti G, Chang Y, Moore PS. Viral IL-6-induced cell proliferation and immune evasion of interferon activity. Science. 2002;298:1432-1435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 166] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 30. | Bhaskaran N, Ghosh SK, Yu X, Qin S, Weinberg A, Pandiyan P, Ye F. Kaposi’s sarcoma-associated herpesvirus infection promotes differentiation and polarization of monocytes into tumor-associated macrophages. Cell Cycle. 2017;16:1611-1621. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 2.3] [Reference Citation Analysis (0)] |