Published online May 26, 2026. doi: 10.12998/wjcc.v14.i15.120061
Revised: March 12, 2026
Accepted: April 22, 2026
Published online: May 26, 2026
Processing time: 88 Days and 6 Hours
Neuromuscular choristoma (NMC) is a rare developmental anomaly characterised by the aberrant presence of mature skeletal muscle fibres within peripheral nerve fascicles. Involvement of the sciatic nerve is uncommon, with most reported ins
We describe a paediatric patient who was diagnosed in infancy with a sciatic nerve NMC, confirmed by open biopsy, and has been followed for over ten years. Neurologically, the patient remained stable, exhibiting only mild weakness in the left posterior compartment of the leg. Serial imaging over the follow-up period showed no significant progression or malignant transformation of the lesion. However, over time a progressive limb length discrepancy and lower limb muscle imbalance developed, eventually becoming the dominant clinical issues. A multi
Our case adds to the limited body of long-term follow-up data and supports a conservative management approach for NMC, with an emphasis on secondary musculoskeletal consequences.
Core Tip: Neuromuscular choristoma (NMC) is a rare benign developmental lesion of peripheral nerves in which mature muscle fibres are intermingled with nerve fascicles. Although NMC is histologically benign, unnecessary surgical intervention can provoke an aggressive desmoid-type fibromatosis in the affected nerve. Paediatric sciatic nerve NMC cases are extremely uncommon, with only one published report providing long-term follow-up. We present an 11-year follow-up of a child with a sciatic NMC, underscoring the significant orthopaedic sequelae that can arise and necessitate complex multidisciplinary management. This under-recognised aspect of NMC care is a key clinical consideration highlighted by our case.
- Citation: Saravanan K, Nagappan PG, Simpson AI. Benign but not inconsequential, paediatric sciatic nerve neuromuscular choristoma and its longitudinal impact: A case report. World J Clin Cases 2026; 14(15): 120061
- URL: https://www.wjgnet.com/2307-8960/full/v14/i15/120061.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i15.120061
Neuromuscular choristoma (NMC) is a rare developmental lesion characterised by the aberrant presence of mature skeletal muscle fibres within peripheral nerve fascicles[1]. NMC typically presents in the sciatic nerve or brachial plexus[2,3], but cases have also been reported in other sites such as the cranial nerves[4-6]. Common differential diagnoses may include intraneural lipoma, benign peripheral nerve sheath tumours. A PubMed literature search for “choristoma and neuromuscular and sciatic” identified 12 relevant articles on sciatic nerve NMC. The majority of these are case reports[2,3,7-9], with the exception of two retrospective cohort studies[10-15] and one prospective cohort study[16]. Among the 10 case reports, equal numbers of adult and paediatric sciatic nerve NMC cases have been documented.
From the existing literature, it is well established that unnecessary operative interventions (e.g., surgical resection or biopsy) are associated with a significantly increased risk of NMC transforming into a desmoid-type fibromatosis (DTF) tumour[15,17-19]. In view of this risk, current management of sciatic nerve NMC in children is generally conservative, avoiding surgery unless absolutely indicated[3,8,9,12,20]. Nevertheless, longitudinal data on the natural history, long-term neurological stability, and optimal management of paediatric NMC are scarce; notably, only one published case report has described the long-term course of a sciatic nerve NMC[9]. Even in this report, their longitudinal follow-up was predominantly focused around monitoring the radiological progression of the tumour itself and the neurological symptoms of weakness caused by it directly.
Existing literature has described orthopaedic sequelae of sciatic nerve NMC, including limb length discrepancy, limb hip dysplasia and limb undergrowth in up to 85% of cases[16]. However, published reports have not, to our knowledge, detailed the longitudinal orthopaedic surveillance, multidisciplinary decision-making, and timing of guided growth intervention required as an affected child approaches skeletal maturity. Here, we present a paediatric patient with a sciatic nerve NMC diagnosed in infancy and followed for 11 years. Over the ensuing decade, orthopaedic sequelae, rather than neurological deterioration, emerged as the dominant clinical issue. The principal contribution of this case is therefore not the mere presence of musculoskeletal sequelae, but the demonstration that neurological and radiological quiescence of the nerve lesion may coexist with progressive limb length discrepancy, pelvic obliquity, compensatory scoliosis, Achilles contracture and foot deformity, requiring active longitudinal orthopaedic management and carefully timed guided growth surgery. This case emphasizes that even a histologically benign NMC can have significant long-term orthopaedic consequences, such as limb undergrowth and scoliosis, which may ultimately require intervention.
The patient had an 11-year history of a progressively worsening left lower limb length discrepancy and muscle weakness and imbalance predominantly affecting the posterior compartment of the left leg.
The patient’s limb length discrepancy and muscle imbalance were first noted in infancy and gradually progressed over 11 years. The left leg became noticeably shorter than the right, and weakness in the left posterior leg musculature led to gait abnormalities. At the clinic visit in March 2025, the patient’s magnetic resonance imaging (MRI) and radiographic evaluations were reviewed. These showed no significant interval change in the nerve lesion compared to the previous MRI from 2019, indicating stable disease.
The patient was diagnosed with a NMC involving the left sciatic nerve (extending into the lumbosacral plexus) in the first month of life, confirmed by open surgical biopsy at two weeks of age. The biopsy was conducted for diagnostic purposes, which we used to guide our long-term monitoring and management strategies once the mass was confirmed to be sciatic nerve NMC. Throughout childhood, she developed multiple orthopaedic sequelae secondary to the NMC. These sequelae included: A limb length discrepancy (with the left leg 2.5 cm shorter than the right, primarily due to inhibited growth of the left tibia); chronic left Achilles tendinopathy with an equinus contracture; a fixed calcaneal deformity of the left foot; and muscular imbalance predominantly affecting the left calf and hamstring. She underwent an Achilles tendon lengthening procedure in 2020 to address the contracture and improve ankle dorsiflexion. Aside from interventions related to the choristoma, she has been managed conservatively with routine follow-up and serial MRI scans over the past ten years to monitor the lesion’s progression.
Aside from the NMC, the patient’s medical history is unremarkable. Her family history is non-contributory; there are no known familial predispositions to benign tumours or malignancies.
At the latest evaluation, the patient was well-developed for her age and remained highly active. She participated in sports, including Irish dancing, without major limitations. On examination, a pelvic obliquity due to the limb length discrepancy was evident, along with a compensatory right-sided thoracolumbar scoliosis in the standing posture. This is consistent with reports in existing literature in which leg length discrepancy (LLD) of only a few cm causes pelvic obliquity and compensatory scoliosis[21]. The left lower limb was approximately 2.5 cm shorter than the right, with the discrepancy arising primarily from the tibia. She ambulated with a mild limp. The left foot had a calcaneal alignment deformity and was smaller than the right by approximately three shoe sizes. Despite a slight increase in the size of the nerve lesion on imaging over the years, her neurological examination remained unchanged. Muscle power in the left lower limb was near normal, with only mild weakness in the hamstring and calf muscles (medical research council grade 4+ out of 5). Sensation and reflexes in the affected limb were normal.
Laboratory investigations were unremarkable. Routine blood tests, including inflammatory markers and metabolic panels, have consistently remained within normal limits.
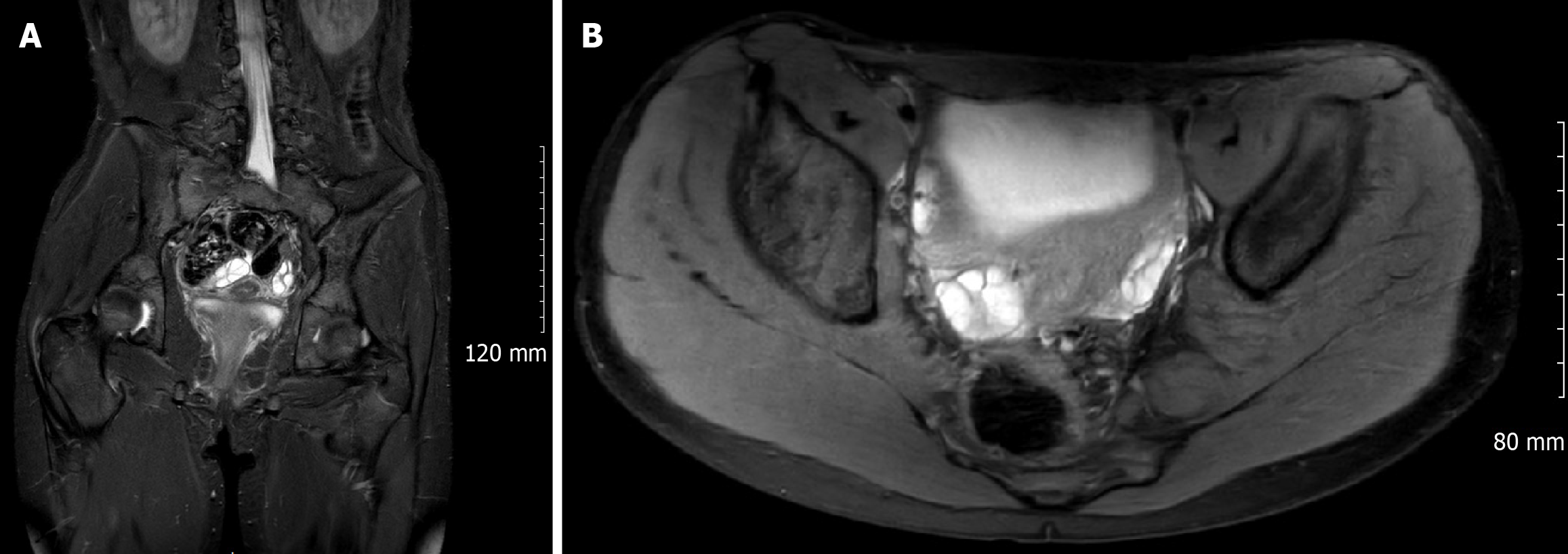
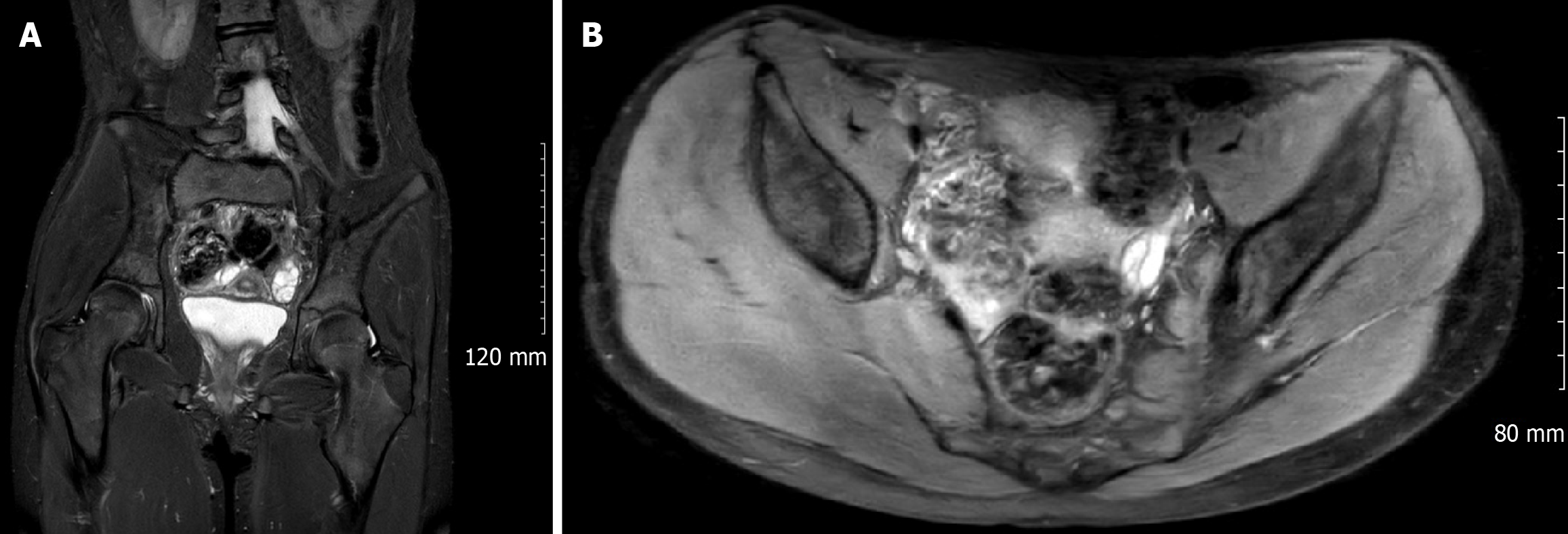
MRI: T2-weighted MRI of the lumbosacral pelvis in 2019 revealed an expanded, fusiform enlargement of the left L4-S2 nerve roots consistent with the known NMC in both axial and coronal views (Figure 1). The left sciatic nerve measured 2.7 cm in diameter on 2017 MRI and increased to 3.1 cm on the 2019 scan (measured at the maximal cross-section on axial T2 sequences). There was also associated enlargement of the adjacent neural exit foramina, with thickening of the left lumbosacral nerve roots up to the level of L5 (Figure 2). No destructive bone lesions or abnormal soft-tissue masses (such as a DTF) were identified. The remainder of the 2019 MRI was unremarkable: The hip joints, lower limb muscle signal, sacroiliac joints, and pubic symphysis were normal. A mild right thoracolumbar scoliotic curvature was incidentally noted on the scan.
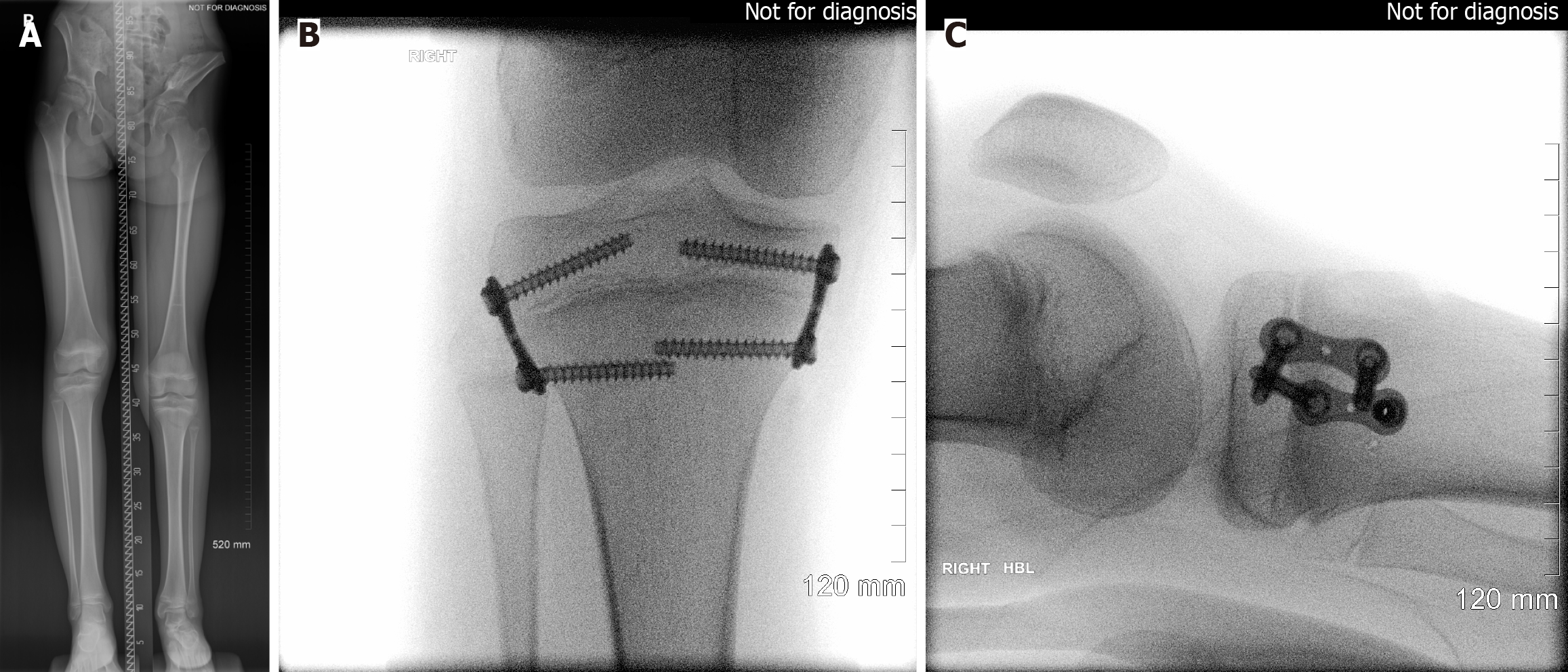
In August 2024 the anteroposterior diameter measured 2.6 cm, essentially unchanged within measurement error. The left sciatic nerve remained markedly enlarged but morphologically similar, with no new lesions and no signal characteristics suggestive of malignant transformation. In light of the patient’s continued growth and the progressive limb length discrepancy, these recent imaging findings reinforced a conservative approach for the tumour itself. However, it was determined that the patient would benefit from a guided growth procedure to address the limb length discrepancy. After multidisciplinary discussion, the patient underwent epiphysiodesis using an 8-plate on the right proximal tibial growth plate to correct the limb length discrepancy.
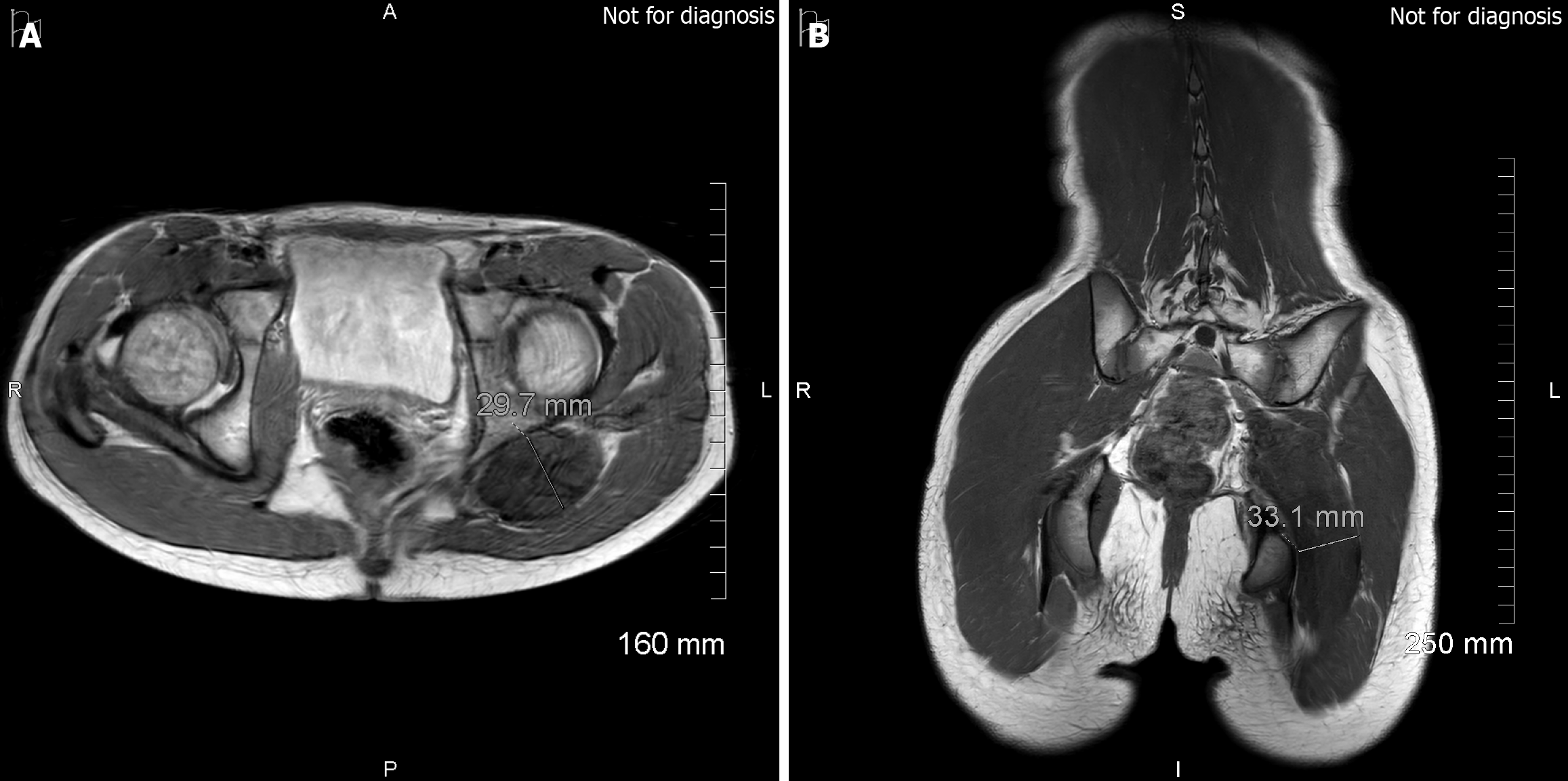
Following guided growth surgery in August 2025, interval MRI was repeated in November 2025 (Figure 3). As on previous studies, there was diffuse hypointense thickening of the left sciatic nerve involving the L4 to S2 nerve roots, with associated thickening of the L5 roots at the greater sciatic foramen. On the November 2025 examination, the sciatic nerve measured approximately 3.0 cm in anteroposterior diameter posterior to the ischium and 3.3 cm transversely at the level of the ischial tuberosity. On the preceding study, corresponding measurements at similar anatomical levels were approximately 2.6 cm and 3.2 cm respectively. Direct comparison was limited by lesion morphology and by small differences in imaging plane and anatomical landmark. However, there was no convincing interval progression in overall morphology or signal characteristics, and no accompanying clinical deterioration, supporting the interpretation of radiological stability within expected inter-scan measurement variability.
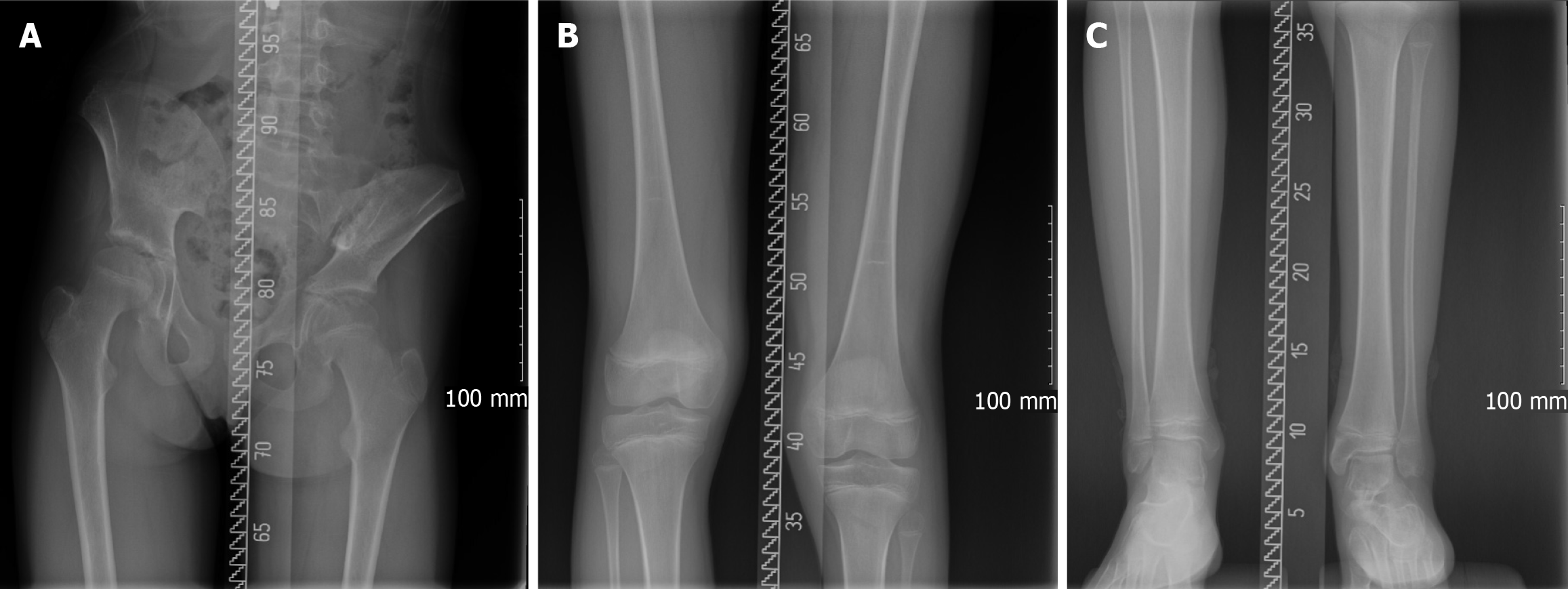
X-ray photograph: Full-length standing radiographs obtained in December 2024 (Figure 4) showed a limb length discrepancy of approximately 2.5 cm (shorter on the left), consistent with prior measurements. The discrepancy stemmed predominantly from shortening of the left tibia. A pelvic tilt due to the limb length discrepancy was evident, along with a compensatory mild right thoracolumbar scoliosis, consistent with the clinical exam findings. An ankle and foot ra
| Timepoint and modality | Key findings and measurements | Clinical impression action | Patient age |
| 2017 (MRI) | Nerve diameter: 2.7 cm | Baseline measurement | 3 years, 5 months |
| 2019 (MRI pelvis) | Nerve diameter: Increased to about 3.1 cm. Anatomy: Fusiform enlargement (L4-S2); isointense to muscle. Negatives: No bone lesions or soft tissue masses | NMC. Conservative management | 5 years, 5 months |
| August 2024 (follow-up MRI) | AP diameter: 2.6 cm (post to ischium). Transverse: 3.2 cm (ischial tuberosity). Morphology: Unchanged; no malignant signal | Stable disease. Continued conservative approach for tumor | 10 years, 5 months |
| December 2024 (standing X-Ray) | LLD: 2.5 cm (left side shorter; tibial origin). Spine: Pelvic tilt with compensatory right scoliosis. Foot: Left calcaneal deformity (elevated heel) | Confirmed indication for guided growth surgery to address LLD | 10 years, 7 months |
| August 2025 (surgery) | Right proximal tibial epiphysiodesis (8-plate) performed to slow growth on contralateral limb | 11 years, 5 months | |
| November 2025 (interval MRI) | AP diameter: 3.0 cm. Transverse: 3.3 cm. Anatomy: Gross hypointense thickening (L4-S2) and L5 root thickening | Fairly stable. Slight measurement increased attributed to difficult morphology rather than progression | 11 years, 7 months |
The patient’s case was reviewed at the local multidisciplinary sarcoma tumour board. The panel, which included paediatric oncologists, orthopaedic surgeons, radiologists, and sarcoma specialists, examined the most recent MRI scans. The panel found no suspicious features suggestive of malignant transformation or aggressive progression of the choristoma. The consensus recommendation was to continue conservative management of the NMC, with interventions focused on the orthopaedic issues[22]. The plan for guided growth surgery to address the limb length discrepancy was fully endorsed, and periodic surveillance imaging of the sciatic nerve NMC was recommended for ongoing monitoring of the lesion.
Congenital NMC of the left sciatic nerve (diagnosed in infancy via open biopsy), with secondary orthopaedic sequelae including limb length discrepancy, pelvic obliquity with compensatory scoliosis, left Achilles contracture, and left calcaneal foot deformity.
Definitive management of the sciatic nerve NMC has been conservative. After the initial diagnostic biopsy, no resection or other invasive procedure has been performed on the nerve lesion in order to minimize the risk of inducing a DTF. Instead, management has focused on addressing the orthopaedic complications. The patient underwent left Achilles tendon lengthening in 2020 to relieve the equinus contracture and improve foot position.
As the patient approached puberty, recent imaging and growth monitoring guided the decision to perform a right proximal tibial epiphysiodesis in August 2025 to gradually correct the limb length discrepancy by skeletal maturity. The procedure involved insertion of two 16-mm 8-plates deep to the fascia on the medial and lateral aspects of the right pro
At the 2-week post-operative review, the surgical incision was well healed and the patient was recovering well. She was ambulating independently without crutches and had ceased analgesics, although a slight antalgic gait persisted. Lower limb function was improving. The patient was already using rowing and ski ergometer machines at the gym, and she demonstrated the ability to perform a half-squat (albeit with unequal knee flexion). She was advised to proceed with a gentle return to sports, specifically swimming and light cycling, but to strictly avoid contact sports for six weeks. By the latest clinical review in January 2026, the patient presented with an excellent range of movement in the knee, with no evidence of stiffness or joint effusion.
For the NMC itself, adjunctive measures have included physiotherapy to maintain muscle strength and flexibility, shoe lifts to compensate for the limb length difference, and regular clinical evaluations to monitor spinal alignment and lower limb development.
The patient remains neurologically stable and continues to participate in age-appropriate physical activities. The NMC has shown no convincing radiological or clinical progression over serial follow-up. Ongoing management now focuses primarily on the orthopaedic sequelae. Key priorities include post-epiphysiodesis monitoring of limb length correction, continued surveillance of pelvic tilt and spinal alignment, and management of residual foot deformity with orthotic support or further surgery if required. Interval MRI surveillance of the pelvis and affected nerve will continue to ensure ongoing stability of the NMC. The patient remains under multidisciplinary follow-up involving paediatric orthopaedic, oncology and radiology teams.
Sciatic nerve NMC is an exceptionally rare entity; a review of the literature reveals only approximately ten reported cases (across adults and children) and two small cohort studies focusing on this condition. This scarcity of cases underscores the limited information available, particularly regarding long-term outcomes in paediatric patients[9]. Historically, surgical intervention was often undertaken for diagnostic confirmation or attempted definitive treatment of NMCs[2,13,16]. However, accumulating evidence suggests that invasive procedures such as biopsy or resection can precipitate an aggressive DTF within the affected nerve territory[15,17,18]. NMC-associated DTF can harbour CTNNB1 p.S45 mutations and may demonstrate more aggressive clinical behaviour than sporadic DTF, sometimes requiring adjuvant systemic or radiotherapy strategies for disease control[19,22,23]. Recognising this risk, contemporary management of sciatic nerve NMC, especially in children, has shifted toward conservative, non-operative strategies[3,9,12,16]. The rationale is that avoiding unnecessary surgery prevents iatrogenic fibroproliferative changes while preserving neurological function.
The initial open biopsy reflects historical diagnostic practice in infancy, when exclusion of a malignant peripheral nerve tumour was prioritised; in contemporary practice, where imaging is characteristic, a more conservative diagnostic approach may be preferred. The extent to which the infancy biopsy influenced the later clinical course in this patient cannot be determined.
In light of the recognised association between NMC and post-intervention DTF, we adopted a conservative approach towards the nerve lesion in this case. Aside from the initial diagnostic open biopsy in infancy, no direct surgical intervention was performed on the sciatic NMC, and the lesion was monitored with serial MRI. The patient’s neurological status remained essentially unchanged throughout childhood, with only mild, non-progressive motor weakness and no sensory loss. This broadly mirrors the long-term clinical course reported by Park[9], in which conservative management was associated with prolonged neurological and radiological stability. Our case supports the view that, in the absence of concerning clinical or imaging features, a watchful waiting strategy is appropriate for the nerve lesion itself.
A distinctive aspect of the present case is the prominence and progression of the orthopaedic sequelae over time (Table 2). Although the nerve lesion itself remained neurologically and radiologically quiescent, the patient developed progressive musculoskeletal consequences including limb length discrepancy, pelvic obliquity with compensatory scoliosis, Achilles contracture and fixed calcaneal deformity. Kumar et al[14] reported that sciatic nerve NMC extending into the lumbosacral plexus may be associated with nerve-territory undergrowth involving soft tissue and bone, in
| Ref. | Main focus | Orthopaedic sequelae described | Follow-up emphasis | Why our case adds value |
| Kumar et al[14], 2014 | Nerve-territory undergrowth in sciatic/lumbosacral NMC | Yes: Limb shortening and hip dysplasia | Mechanistic association and phenotype description | Our case extends this by detailing longitudinal surveillance, orthopaedic decision-making and timing of guided growth intervention |
| Park[9], 2019 | Long-term natural history of sciatic NMC | Orthopaedic issues not the primary focus | Neurological and radiological stability over long follow-up | Our case complements this by showing that prolonged lesion stability may coexist with progressive orthopaedic burden requiring treatment |
| Present case | Longitudinal paediatric multidisciplinary management | Yes: Limb length discrepancy, pelvic obliquity, scoliosis, achilles contracture, calcaneal deformity | Orthopaedic surveillance and post-biopsy conservative nerve management | Distinguishing contribution is the detailed orthopaedic pathway from observation to guided growth planning and intervention near skeletal maturity |
At a mechanistic level, the available literature supports a developmental and nerve-territory-based model rather than a purely local mass effect explanation. Animal models show that loss of nerve supply impairs bone growth. In rats, early sciatic neurectomy reduced foot bone length by about 3%-5%. Similarly, vertebrate experiments demonstrated 15% tibial shortening with early denervation. These findings support that chronic NMC involvement (denervation/underuse) can retard growth of the affected limb[15,26]. In practical terms, this means that the clinical burden of sciatic NMC choristoma may extend beyond neuropathy to include progressive asymmetry of bone and soft-tissue development within the affected distribution. Further molecular and developmental work is needed to clarify the relative contributions of altered innervation, trophic signalling and regional growth disturbance.
By age 11, the left leg was considerably shorter, leading to gait asymmetry, pelvic tilt, and secondary spinal curvature. Additionally, muscle imbalance around the ankle and hindfoot led to an Achilles tendon contracture and a calcaneal foot deformity. These sequelae are only sparingly mentioned in prior reports and have not been a central focus of the NMC literature. Our case thus highlights that NMC, despite being a benign neural tumour, can have far-reaching effects on a child’s musculoskeletal development.
Effective management of the orthopaedic sequelae of sciatic nerve NMC requires multidisciplinary care. In our patient, paediatric orthopaedic input, physiotherapy and rehabilitation were central to management. Achilles tendon lengthening was required to address the equinus contracture and improve foot positioning during gait. As the patient approached puberty, serial growth monitoring and predictive modelling of limb length discrepancy informed the timing of right proximal tibial epiphysiodesis. The aim was not treatment of the nerve lesion, but mitigation of its secondary musculoskeletal consequences. This distinction is important: Even when the NMC itself remains stable, its indirect impact on the musculoskeletal system can be clinically significant and may require active orthopaedic intervention.
This case illustrates the complexity of caring for a child with sciatic nerve NMC over the long term. Early diagnosis enabled appropriate surveillance and helped avoid unnecessary re-intervention on the nerve. Longitudinal follow-up, however, also demonstrated that neurological and radiological stability of the lesion did not equate to overall clinical stability. Instead, progressive orthopaedic sequelae emerged gradually and required planned intervention. On the basis of this case and the limited existing literature, regular orthopaedic review should be considered in children with sciatic nerve NMC, particularly where limb asymmetry, pelvic obliquity, hip dysplasia or foot deformity is present or evolving. Future reports should document musculoskeletal outcomes systematically, as this is the area in which current evidence remains particularly sparse.
This report has several limitations. First, as a single case report, it is inherently limited in generalisability. Second, while the temporal association and anatomical concordance strongly suggest that the orthopaedic sequelae were related to the NMC, causality cannot be established from a single observational case. Third, no genetic or molecular analysis was available to explore the biological basis of the observed musculoskeletal phenotype. Fourth, formal functional outcome measures were not collected longitudinally. Finally, post-epiphysiodesis follow-up remains relatively short at the time of writing, and the final degree of correction at skeletal maturity is therefore not yet known.
Paediatric sciatic nerve NMC is histologically benign but not clinically inconsequential. This case demonstrates that prolonged neurological and radiological stability of the lesion may coexist with progressive musculoskeletal con
| 1. | Bonneau R, Brochu P. Neuromuscular choristoma. A clinicopathologic study of two cases. Am J Surg Pathol. 1983;7:521-528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Lam S, Grandhi R, Wong R, Hamilton R, Greene S. Neuromuscular hamartoma of the sciatic nerve: Case report and review of the literature. Surg Neurol Int. 2013;4:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 3. | O'Brien TG, Spinner RJ, Boon AJ. Neuromuscular choristoma presenting with unilateral limb hypoplasia in a 3-year-old boy. Muscle Nerve. 2016;54:797-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Boyaci S, Moray M, Aksoy K, Sav A. Intraocular neuromuscular choristoma: a case report and literature review. Neurosurgery. 2011;68:E551-5; discussion E555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 5. | Castro DE, Raghuram K, Phillips CD. Benign triton tumor of the trigeminal nerve. AJNR Am J Neuroradiol. 2005;26:967-969. [PubMed] |
| 6. | Nikolaou G, Röösli C, Huber A, Probst R. Neuromuscular choristoma of the internal auditory meatus. ORL J Otorhinolaryngol Relat Spec. 2012;74:246-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Maher CO, Spinner RJ, Giannini C, Scheithauer BW, Crum BA. Neuromuscular choristoma of the sciatic nerve. Case report. J Neurosurg. 2002;96:1123-1126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 8. | Stone JJ, Prasad NK, Laumonerie P, Howe BM, Amrami KK, Carter JM, Jentoft ME, Spinner RJ. Recurrent desmoid-type fibromatosis associated with underlying neuromuscular choristoma. J Neurosurg. 2019;131:175-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 9. | Park JE. Long-term natural history of a neuromuscular choristoma of the sciatic nerve: a case report and literature review. Clin Imaging. 2019;55:18-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 10. | Maldonado AA, Broski SM, Carter JM, Spinner RJ. Unrecognized neuromuscular choristoma with recurrent desmoid-type fibromatosis and Marjolin ulcer: expanding the spectrum of neuromuscular choristoma sequelae within the nerve territory? Illustrative case. J Neurosurg Case Lessons. 2023;5:CASE22554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Ricciardelli A, Flores A, Lu HC, Guney E, Perry A, Grochmal J. Adult neuromuscular choristoma, a rare peripheral nerve pathology: illustrative case. J Neurosurg Case Lessons. 2024;7:CASE23517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Passi GR, Maheshwari S, Udani V. Progressive Unilateral Foot Drop in a Child due to Neuromuscular Choristoma. Ann Indian Acad Neurol. 2024;27:580-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Stone JJ, Amrami KK, Carter JM, Spinner RJ. Stimulation Contractility Gradient of a Neuromuscular Choristoma Within the Sciatic Nerve: 2-Dimensional Operative Video. Oper Neurosurg. 2019;16:E17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Kumar R, Howe BM, Amrami KK, Spinner RJ. Neuromuscular choristoma of the sciatic nerve and lumbosacral plexus: an association with nerve-territory undergrowth in the pelvis affecting soft tissue and bone. Acta Neurochir (Wien). 2014;156:1041-1046. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Maldonado AA, Broski SM, Carter JM, Marek T, Howe BM, Spinner RJ. Neuromuscular choristoma and circumferential nerve territory desmoid-type fibromatosis: imaging findings supporting a nerve-driven mechanism. J Neurosurg. 2024;140:1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Guo W, Wang H, Chen T, Yang W, Wang SF, Chen SL. Clinical features and ultrasound findings of a rare musculoskeletal system disease-neuromuscular choristoma. BMC Musculoskelet Disord. 2022;23:464. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 17. | Hébert-Blouin MN, Scheithauer BW, Amrami KK, Durham SR, Spinner RJ. Fibromatosis: a potential sequela of neuromuscular choristoma. J Neurosurg. 2012;116:399-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Hébert-Blouin MN, Amrami KK, Spinner RJ. Addendum: Evidence supports a "no-touch" approach to neuromuscular choristoma. J Neurosurg. 2013;119:252-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 19. | Knijn N, Mogk SC, Teerenstra S, Simmer F, Nagtegaal ID. Perineural Invasion is a Strong Prognostic Factor in Colorectal Cancer: A Systematic Review. Am J Surg Pathol. 2016;40:103-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 188] [Article Influence: 18.8] [Reference Citation Analysis (3)] |
| 20. | Niederhauser BD, Spinner RJ, Jentoft ME, Everist BM, Matsumoto JM, Amrami KK. Neuromuscular choristoma: characteristic magnetic resonance imaging findings and association with post-biopsy fibromatosis. Skeletal Radiol. 2013;42:567-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 21. | Raczkowski JW, Daniszewska B, Zolynski K. Functional scoliosis caused by leg length discrepancy. Arch Med Sci. 2010;6:393-398. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 92] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 22. | Marek T, Howe BM, Spinner RJ. Expanding on MRI characteristics of neuromuscular choristoma: a single center retrospective review. Skeletal Radiol. 2025;54:2107-2115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 23. | Carter JM, Maldonado AA, Howe BM, Okuno S, Spinner RJ. Frequent CTNNB1 p.S45 Mutations and Aggressive Clinical Behavior in Neuromuscular Choristoma-Associated Fibromatosis. Neurosurgery. 2021;88:804-811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 24. | Dietz FR. Effect of peripheral nerve on limb development. J Orthop Res. 1987;5:576-585. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 25. | Tomlinson RE, Christiansen BA, Giannone AA, Genetos DC. The Role of Nerves in Skeletal Development, Adaptation, and Aging. Front Endocrinol (Lausanne). 2020;11:646. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 62] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 26. | Maldonado AA, Spinner RJ, Broski SM, Stone JJ, Howe BM, Carter JM. Neuromuscular choristoma-associated desmoid-type fibromatosis: Establishing a nerve territory concept. Acta Neurochir (Wien). 2020;162:1137-1146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |