INTRODUCTION
Diabetic retinopathy (DR) remains a leading cause of preventable vision impairment worldwide, despite considerable progress in systemic glucose control and ocular treatments. Historically, the pathophysiology and progression of DR have been understood primarily through chronic hyperglycemia, with glycated hemoglobin (HbA1c) serving as the principal biomarker for clinical decision-making and risk assessment. Landmark studies, such as the diabetes control and complications trial and its extended epidemiology of diabetes interventions and complications follow-up, conclusively demonstrated that sustained glycemic control reduces the incidence and progression of retinopathy[1]. Emerging evidence indicates that metrics beyond mean HbA1c, including long-term glycemic variability and other risk determinants, contribute to DR risk; higher visit-to-visit HbA1c variability has been associated with incident DR independent of mean HbA1c in type-2 diabetes, and glycemic profile variability has been identified as an independent predictor of DR progression in long-term cohorts[2-5]. In addition to glycemic factors, DR risk is influenced by established systemic modifiers such as hypertension, dyslipidemia, and renal dysfunction, which interact with metabolic stress to shape retinal vulnerability.
In recent years, there has been a growing focus on metabolic instability, including glycemic fluctuations, intermittent hyperglycemia, and hypoglycemic episodes. Inter-visit variability in fasting plasma glucose and HbA1c has been identified as an independent predictor of microvascular problems, such as DR, in both type 1 and type 2 diabetes populations[6,7]. These findings question the sufficiency of mean glucose indices alone and suggest that retinal tissues may be particularly susceptible to rapid metabolic fluctuations, which are consistently associated with increased risk and progression of DR. Fluctuating glucose levels induce recurrent metabolic stress on retinal endothelial cells, pericytes, and Müller glia, resulting in cumulative cellular damage that may not be indicated by average glucose exposure[5]. This instability may exacerbate oxidative stress, inflammatory signaling, and mitochondrial dysfunction, thereby accelerating microvascular damage even in the absence of prolonged hyperglycemia[8].
The introduction of continuous glucose monitoring (CGM) has provided unparalleled insight into daily glucose fluctuations, enabling the measurement of time within range, time exceeding range, time below range, and aggregate indicators of glucose variability. Numerous observational and longitudinal studies have established strong correlations between reduced time in range and the incidence or progression of DR, irrespective of HbA1c levels[9,10]. These findings have been validated across multiple groups, including individuals with type 1 diabetes, type 2 diabetes, and those using intensive insulin or automated insulin delivery devices[11,12]. The data corroborate the concept that retinal damage is influenced by both cumulative glucose exposure and the magnitude and frequency of glycemic fluctuations. Thus, dependence on HbA1c alone may underappreciate the actual retinal risk in patients undergoing significant metabolic fluctuations.
The notion of metabolic memory establishes a vital molecular foundation linking early or intermittent dysglycemia to enduring retinal injury, beyond mere clinical associations. Research involving both experimental and human studies has shown that previous episodes of inadequate glycemic control can lead to enduring epigenetic, inflammatory, and mitochondrial changes in retinal cells, which continue to facilitate disease progression despite later improvements in glycemic levels[13,14]. This event provides a molecular rationale for the initial exacerbation of retinopathy observed after rapid glucose normalization and highlights the retina's limited capacity to recover from recurrent metabolic insults[15]. Metabolic memory indicates that temporary metabolic instability may have enduring effects that extend beyond its duration, thereby complicating static risk assessment approaches.
The retinal neurovascular unit, comprising neurons, glia, and microvasculature, functions as an integrated metabolic and signaling system; intact interactions across these components are critical for retinal homeostasis. The retina has exceptionally high metabolic demands, particularly in neurons such as photoreceptors, which require continuous adenosine triphosphate (ATP) generation. Metabolic instability - including glycemic variability as reflected by HbA1c variability and glucose fluctuations - has been associated with increased DR progression, suggesting that fluctuations in glucose homeostasis may detrimentally influence both microvascular and neural elements of the retina beyond mean glycemic exposure[16].
Disruption of neuroglial homeostasis may precede the development of microvascular lesions, leading to initial functional impairment and establishing conditions for subsequent structural damage[17]. DR should increasingly be seen as a neurovascular and metabolic disorder rather than solely a microangiopathic problem. This paradigm shift corresponds with increasing evidence that neurodegenerative alterations and vascular dysfunction develop concurrently and are collectively influenced by metabolic stress.
Collectively, these data underscore a significant disparity between conventional glycemic measurements and the intricate metabolic conditions encountered by retinal tissues. A dynamic paradigm that incorporates glycemic fluctuations, CGM data, and biological indicators of metabolic stress may provide a more precise assessment of retinopathy risk and progression. Recognizing DR as a result of metabolic instability rather than merely persistent hyperglycemia significantly influences screening methods, treatment timing, and personalized patient care. This narrative review consolidates existing clinical, translational, and mechanistic evidence to position metabolic instability as a fundamental contributor to DR, while rigorously evaluating the limitations of current data and highlighting priorities for future research. While glycemic variability and metabolic memory have been individually investigated, they are often discussed as discrete phenomena. In this review, we propose the broader construct of metabolic instability as an integrative pathophysiological framework that links dynamic glycemic exposure, stress signaling activation, epigenetic embedding, and neurovascular unit dysfunction.
NARRATIVE APPROACH
This narrative review was conducted to synthesize and critically assess existing research about the influence of metabolic instability on the onset and advancement of DR. A narrative approach was selected to integrate heterogeneous study designs, metabolic metrics, and retinal outcome measures.
A thorough literature search was conducted in January 2026 utilizing major biomedical databases, with PubMed/MEDLINE as the primary source of peer-reviewed articles. Search algorithms integrated controlled-vocabulary terms and free-text keywords pertinent to DR and metabolic dynamics, including glycemic variability, metabolic instability, glucose fluctuations, hypoglycemia, CGM, time-in-range measurements, and metabolic memory. Supplementary words concerning downstream pathways, including oxidative stress, mitochondrial failure, epigenetic regulation, inflammation, and neurovascular unit impairment, were integrated to encompass mechanistic and translational investigations. Searches were confined to English-language articles to guarantee uniformity in data interpretation and reporting standards.
The selection of studies focused on original research papers and high-quality reviews that specifically investigated the correlation between dynamic metabolic exposure and retinal morphological or functional outcomes. Both type 1 and type 2 diabetes populations were included, along with studies featuring mixed cohorts, contingent upon the reporting of retinal endpoints or validated surrogate markers of DR. Experimental studies using in vitro systems or animal models were incorporated when they elucidated biological pathways that might link metabolic variations to retinal damage. Research concentrating solely on static glycemic indices, without consideration of variability or temporal glucose patterns, was typically eliminated unless it provided essential insights into long-term glycemic consequences or legacy phenomena pertinent to metabolic instability.
The literature was screened iteratively to identify studies most relevant to dynamic glycemic exposure and retinal structural or functional outcomes. Priority was given to clinical investigations reporting measures of glycemic variability or continuous CGM metrics in relation to DR, as well as experimental and translational studies supporting biological plausibility. This approach allowed integration of complementary clinical and mechanistic evidence while maintaining a focused and coherent narrative synthesis.
RETINAL INJURY DUE TO GLYCEMIC VARIABILITY
Glycemic fluctuations have become a significant factor influencing the incidence of DR, independent of average glycemic exposure. Clinical cohort studies have consistently shown that variations in fasting plasma glucose and HbA1c from visit to visit are linked to the onset and progression of retinopathy in individuals with type 2 diabetes, even after controlling for average HbA1c levels[6,7]. Collectively, these findings indicate that visit-to-visit glycemic variability may contribute to DR risk beyond mean glycemic exposure. While the underlying mechanisms remain incompletely defined, repeated glucose excursions are hypothesized to exacerbate metabolic and oxidative stress within retinal tissues[18]. The retina, known for its high metabolic demands and limited regenerative capacity, appears particularly susceptible to fluctuating metabolic stress. Consequently, glycemic fluctuations are increasingly regarded not merely as a secondary epiphenomenon but as a direct factor in retinal microvascular injury.
At the cellular level, intermittent hyperglycemia induces recurrent activation of oxidative and inflammatory pathways, resulting in endothelial dysfunction and pericyte depletion. Experimental findings indicate that variable glucose conditions elicit greater oxidative stress and mitochondrial damage than prolonged hyperglycemia with the same average glucose exposure[19,20]. This phenomenon is particularly pertinent to the retinal microvasculature, where endothelial cells exhibit limited antioxidant defenses and heightened sensitivity to redox imbalance. Repeated metabolic disturbances compromise tight junction integrity, enhance vascular permeability, and induce capillary dropout, hence hastening the microangiopathic progression typical of DR. These mechanisms provide scientific justification for the robust epidemiological correlations observed between glycemic variability and the progression of retinal disease[21-23].
Additional work suggests that glucose oscillations can enhance stress signaling programs (e.g., p53 activation) and sustain injury pathways consistent with a cellular memory response[24]. In retinal pericytes, intermittent high glucose preferentially activates ER stress-linked inflammatory pathways compared with constant high glucose, supporting a direct susceptibility of pericytes to glucose fluctuation patterns[25]. Beyond acute oxidative and inflammatory signaling, glycemic variability may also accelerate retinal injury through enhanced formation and signaling of advanced glycation end products (AGEs). Rapid glucose excursions favor non-enzymatic glycation reactions, increasing AGE generation even when average glucose levels are comparable, thereby amplifying carbonyl and oxidative stress within retinal tissues[26,27]. AGEs exert additional pathogenic effects through activation of their receptor (RAGE), which is expressed on retinal endothelial cells, pericytes, Müller glia, and inflammatory cells. AGE-RAGE engagement activates nuclear factor kappa B (NF-κB)-dependent inflammatory signaling, enhances reactive oxygen species production, and promotes endothelial dysfunction and leukostasis. AGE-RAGE signaling has been implicated in the persistence of vascular and inflammatory injury even after glycemic improvement, providing a mechanistic link between glycemic variability, metabolic memory, and progressive retinal damage[28]. It should be acknowledged that not all studies of glycemic variability have demonstrated a consistent or independent association with DR. A number of reviews and cohort analyses have pointed to heterogeneity in findings, with some longitudinal studies showing stronger links for long-term HbA1c variability than for short-term measures, and others reporting weaker or non-specific associations with microvascular outcomes. In the RIACE Italian multicenter cohort, HbA1c variability was associated with nephropathy but not retinopathy, underscoring that the strength of association may vary according to population characteristics, variability metrics, and study design[29-31].
RETINAL RISK ASSESSMENT AND CONTINUOUS GLUCOSE MONITORING
The advent of CGM has fundamentally altered the evaluation of glycemic exposure by facilitating high-resolution, temporal analysis of glucose dynamics. Metrics, including time in range, time above range, and time below range, provide a more detailed depiction of metabolic stability than HbA1c alone. It is important to distinguish between the different constructs captured by CGM-derived metrics. Measures such as standard deviation and coefficient of variation primarily quantify glucose dispersion and short-term instability around a mean value, whereas time-in-range, time above range, and time below range reflect cumulative exposure to predefined glycemic thresholds. These metrics provide complementary but non-overlapping information, and their associations with DR may reflect distinct biological pathways. Observational studies using CGM data have shown that reduced time in range is strongly associated with the incidence and severity of DR in both type 1 and type 2 diabetes populations[9]. These relationships remain significant even after controlling for conventional risk factors, underscoring that retinal damage reflects dynamic rather than averaged metabolic exposure. Significantly, measurements developed from CGM identify clinically inconspicuous glucose fluctuations that would remain unnoticed in standard therapy.
Longitudinal evaluations further substantiate the predictive significance of CGM measures for retinal outcomes. Individuals with greater time in range exhibit a lower incidence of retinopathy and a slower progression of pre-existing disease over time, despite similar mean HbA1c values[32]. Increased exposure to hyperglycemic and hypoglycemic levels seems to pose cumulative retinal danger, underscoring the reciprocal damage caused by metabolic extremes. Empirical evidence indicates that the use of CGM may be associated with a reduced incidence of sight-threatening retinopathy complications, presumably due to improved identification and management of glucose fluctuations[10,33,34]. Collectively, this research establishes CGM as a potentially significant instrument for both glucose regulation and personalized retinal risk evaluation. Relying exclusively on HbA1c as a measure of glycemic control may obscure significant interindividual variation in retinal risk, particularly among individuals with comparable average glycemic exposure but differing glucose dynamics. Integrating measurements of glycemic fluctuations and time-in-range into standard evaluations may facilitate more precise identification of patients at increased risk of retinopathy onset or progression. CGM provides an effective method for recording these dynamic metabolic markers in actual clinical environments. The integration of CGM data with ocular observations may facilitate customized screening schedules, emphasizing high-risk individuals for more frequent retinal assessments. Furthermore, the use of CGM may enable prompt intervention by detecting harmful glucose patterns before irreversible structural damage occurs[9-12]. This strategy aligns with the broader trend toward precision medicine in diabetes management, prioritizing personalized risk profiles over population-based thresholds.
RETINAL DAMAGE AND METABOLIC MEMORY
The notion of metabolic memory provides a cohesive framework for elucidating why transient phases of metabolic instability may have enduring effects on retinal health. Experimental and clinical investigations have shown that early exposure to dysglycemia induces enduring molecular changes that perpetuate retinopathy progression, even after subsequent glycemic recovery[35]. Evidence supporting the concept of metabolic memory derives from both retinal-specific experimental models and systemic vascular studies, which together provide complementary, but not interchangeable, insights into the persistence of glucose-induced cellular injury. These abnormalities encompass persistent activation of inflammatory signaling, mitochondrial dysfunction, and epigenetic alterations that influence gene expression in retinal cells. Metabolic memory indicates that retinal damage results not only from cumulative glucose exposure but also from the timing and intensity of metabolic shocks.
Epigenetic processes are crucial in facilitating metabolic memory in the retina. Retinal-specific studies have demonstrated persistent alterations in DNA methylation and histone modifications in retinal endothelial cells and pericytes following variable or transient hyperglycemic exposure, leading to sustained activation of pro-oxidant and pro-inflammatory genes. In parallel, systemic vascular and endothelial models have provided converging evidence that intermittent hyperglycemia induces long-lasting epigenetic reprogramming, supporting the biological plausibility of similar mechanisms in retinal tissues. Enduring alterations in DNA methylation and histone modification have been seen in retinal endothelial cells and pericytes after variable glucose exposure, resulting in sustained activation of pro-oxidant and pro-inflammatory genes[13,14,36]. The link between persistent epigenetic alterations and stress-activated signaling pathways provides an important mechanistic bridge between dynamic glycemic exposure and long-term cellular dysfunction. Experimental evidence shows that glucose oscillations induce activation of the p53 pathway, contributing to metabolic memory in endothelial cells exposed to fluctuating glucose levels, and implicating p53 as a mediator of stress-associated transcriptional regulation in diabetic vascular cells[37]. Moreover, epigenomic profiling in type 1 diabetes cohort revealed persistent differential DNA methylation at specific loci correlated with prior glycemic exposure, supporting a role for stable epigenetic marks in the long-term regulation of genes relevant to metabolic memory and microvascular outcomes[13]. These alterations persist despite the reestablishment of normoglycemia. This effect is clinically evident in the early exacerbation of retinopathy after quick glycemic correction, highlighting the retina's susceptibility to sudden metabolic changes[15,38]. The recognition of metabolic memory underscores the importance of early and consistent glycemic regulation, as well as the prevention of excessive glucose fluctuations throughout disease progression. While systemic vascular studies offer important mechanistic insight, extrapolation to DR must be interpreted within the context of retina-specific cellular metabolism and neurovascular architecture.
METABOLIC INSTABILITY AND NEUROVASCULAR UNIT DYSFUNCTION
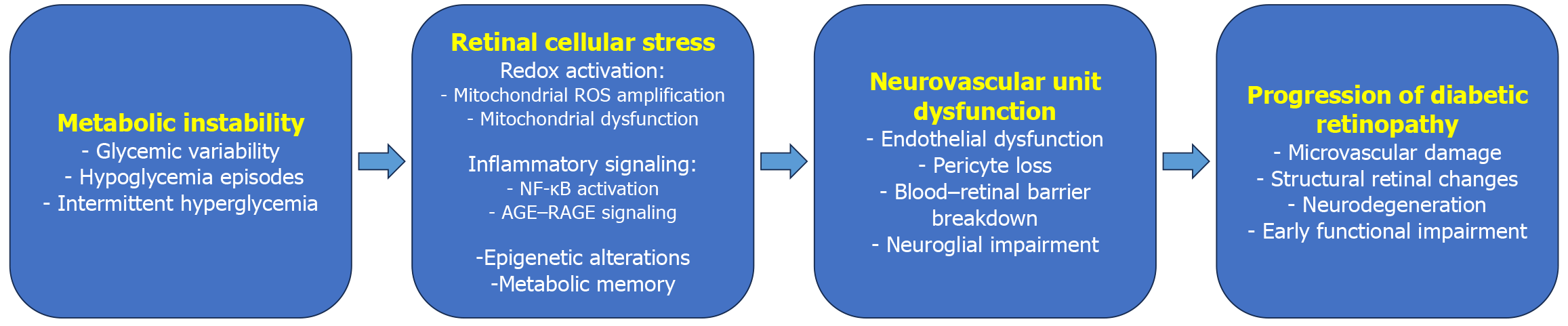
DR is increasingly acknowledged as a problem of the neurovascular unit rather than a mere microvascular disease[39]. Neurons, glial cells, and vascular components are metabolically interconnected, and disturbances in this integrated system lead to early functional impairment. Glycemic instability adversely affects Müller glia and retinal neurons, leading to disrupted neurotransmitter recycling, compromised metabolic support, and heightened susceptibility to oxidative damage[40]. Neurodegenerative changes have been increasingly noted early in DR and may occur before the appearance of visible vascular lesions in certain contexts, although the exact temporal sequence is not fully established. Variable glucose levels and persistent hypoglycemia intensify neurovascular uncoupling by disrupting autoregulatory mechanisms and enhancing inflammatory interactions between the brain and vascular systems[41,42]. Experimental models indicate that oscillatory glucose exposure induces greater neuroglial activation and vascular leakage than continuous hyperglycemia, underscoring the distinct toxicity of metabolic instability. This paradigm is clinically consistent with observed functional deficits, including diminished contrast sensitivity and electrical irregularities, in individuals with modest vascular signs of retinopathy[17,43-45]. In contrast to hyperglycemia-induced oxidative and inflammatory stress, hypoglycemia may exert injury through partially distinct mechanisms. The retina has exceptionally high metabolic demands and limited glycogen reserves, rendering retinal neurons and Müller glia particularly vulnerable to acute glucose deprivation. Hypoglycemia leads to rapid cellular energy failure, impaired ATP-dependent ion homeostasis, and excitotoxic stress mediated by dysregulated glutamate handling. Recurrent hypoglycemic episodes may further compromise neurovascular coupling and autoregulatory responses. Importantly, restoration of normoglycemia following hypoglycemia can generate a reperfusion-like phenomenon, characterized by abrupt mitochondrial reactivation and reactive oxygen species overproduction, thereby amplifying oxidative injury. These oscillatory metabolic transitions-particularly when repeated-may potentiate cumulative neuronal and microvascular damage independent of sustained hyperglycemia[46,47]. Examining DR from a neurovascular perspective underscores the need for earlier detection methods and therapies that prioritize metabolic stability over vascular damage alone. Figure 1 illustrates a conceptual overview of the pathogenic pathways connecting metabolic instability to neurovascular damage in DR.
Figure 1 Influence of metabolic instability on the etiology of diabetic retinopathy.
Variable glucose exposure, encompassing glycemic variability, intermittent hyperglycemia, hypoglycemia, and reduced time in range, induces retinal cellular stress characterized by oxidative damage, mitochondrial dysfunction, inflammatory activation, and epigenetic alterations associated with metabolic memory. These processes compromise the retinal neurovascular unit, resulting in endothelial dysfunction, pericyte depletion, disruption of the blood-retinal barrier, and neuroglial damage. The combined effects of these pathways lead to progressive microvascular and neuronal damage in the retina. NF-κB: Nuclear factor kappa B; AGE: Advanced glycation end product; ROS: Reactive oxygen species; RAGE: Receptor for advanced glycation end-products.
OXIDATIVE STRESS, INFLAMMATION, AND MITOCHONDRIAL DYSFUNCTION
Inflammation and oxidative stress are fundamental mechanisms linking metabolic instability to progressive retinal damage. Repeated fluctuations in glucose levels consistently stimulate redox-sensitive signaling pathways, thereby prolonging reactive oxygen species generation in retinal endothelial cells, pericytes, and Müller glia. In contrast to chronic hyperglycemia, which elicits a relatively steady adaptive response, variable glucose exposure exacerbates oxidative stress by consistently surpassing antioxidant defenses, resulting in cumulative cellular damage[48,49]. Mitochondrial dysfunction is a significant downstream effect, characterized by compromised oxidative phosphorylation, elevated mitochondrial DNA damage, and altered fission and fusion dynamics. The mitochondrial modifications sustain a self-perpetuating loop of oxidative stress that persists after normalization of glucose levels, consistent with the notion of metabolic memory[50]. Beyond mitochondrial reactive oxygen species (ROS) generation, increasing evidence supports nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-particularly the NADPH oxidase 2 (NOX2) isoform-as a key upstream driver of retinal oxidative stress in DR. NOX2 activation promotes an early rise in cytosolic superoxide, amplifies inflammatory signaling (e.g., leukostasis, barrier dysfunction, vascular endothelial growth factor -linked vascular activation), and contributes directly to microvascular injury. Importantly, NOX2-derived ROS can initiate and perpetuate mitochondrial dysfunction, establishing a feed-forward ‘ROS-induced ROS release’ loop that sustains oxidative damage and inflammatory amplification even when glycemic exposure is intermittent. Mechanistically, diabetes-associated activation of Rac1-dependent NOX2 signaling has been linked to mitochondrial damage and capillary cell apoptosis, providing a plausible bridge between dynamic glucose excursions and redox-driven neurovascular injury. In vivo studies further indicate that NOX2 activity in both resident retinal cells and bone marrow-derived inflammatory cells contribute to diabetes-induced retinal vascular pathology, underscoring the role of NOX2 in coupling metabolic stress to inflammatory vascular damage. Collectively, these findings position NOX2 as a mechanistic nexus through which glycemic variability and metabolic instability may translate into persistent mitochondrial injury, inflammatory escalation, and progressive retinal microangiopathy[51-55].
Inflammatory signaling pathways are closely linked to oxidative mechanisms in glycemic variability. Fluctuating glucose levels increase activation of NF-κB and other transcriptional regulators, thereby promoting prolonged production of pro-inflammatory cytokines, adhesion molecules, and chemokines in the retinal microenvironment. This inflammatory environment leads to leukostasis, endothelial dysfunction, and disruption of the blood-retinal barrier, therefore expediting microvascular disease. Significantly, recurrent inflammatory activation appears more harmful than sustained stimulation of comparable intensity, reinforcing the idea that metabolic instability has disproportionate biological effects[56,57]. Pericytes are likely critical amplifiers of this process. Pericyte dysfunction and loss undermine capillary stability and blood-retinal barrier integrity, while also promoting inflammatory crosstalk within the neurovascular unit, mechanisms increasingly recognized as central across retinal vascular diseases, including DR[58]. Importantly, retinal pericytes themselves show maladaptive stress responses to glucose oscillations intermittent (but not constant) high glucose increases monocyte chemoattractant protein-1 and engages ER-stress-associated inflammatory signaling in human retinal pericytes[20]. Importantly, these stress-activated pathways are not merely biochemical phenomena but directly relate to the structural lesions observed in human DR. Oxidative stress and NF-κB-mediated inflammatory activation promotes pericyte apoptosis and endothelial dysfunction, destabilizing capillary walls and contributing to microaneurysm formation. Mitochondrial dysfunction and sustained reactive oxygen species amplification impair endothelial tight junction integrity and increase vascular permeability, facilitating retinal hemorrhages and edema. Concurrent leukostasis and inflammatory signaling promote capillary occlusion and non-perfusion, leading to localized ischemia and subsequent neovascular responses. Thus, dynamic glucose-induced cellular stress provides a mechanistic substrate linking metabolic instability to the characteristic microvascular lesions of DR. These findings elucidate why patients with relatively moderate average glycemic exposure may still develop retinopathy rapidly when glucose variability is significant.
LIMITATIONS
Numerous constraints must be recognized when analyzing the existing evidence connecting metabolic instability to DR. The inconsistency in the definition and measurement of glycemic variability hinders cross-study comparisons and restricts the formulation of clinically relevant thresholds. Metrics like standard deviation, coefficient of variation, time in range, and visit-to-visit HbA1c variability are not synonymous and may reflect different dimensions of metabolic instability. Secondly, much of the research is observational, which precludes causal inference and introduces the potential for residual confounding. Despite the persistence of associations after multivariable adjustment, unmeasured variables, such as treatment adherence, comorbidities, and socioeconomic characteristics, may influence both glycemic trends and retinal outcomes.
A further limitation is the limited longitudinal integration of metabolic and ocular data. Limited research has integrated CGM data with comprehensive retinal imaging or functional evaluations during prolonged follow-up durations. Consequently, the temporal association between metabolic instability and particular retinal alterations remains poorly defined. Moreover, laboratory models may inadequately replicate the complexities of human DR, particularly with respect to the long-term progression of the disease and the effects of treatment. Another important limitation relates to the multidimensional nature of metabolic instability. Glycemic variability encompasses distinct constructs, including excursion amplitude, frequency of fluctuations, cumulative exposure to hyperglycemic and hypoglycemic extremes, and time within target range. These dimensions are not interchangeable, and current evidence does not allow definitive weighting of which specific instability pattern is most detrimental to retinal tissue. Differences in metric definitions, study design, and population characteristics further complicate interpretation. Clarifying the relative pathogenic contribution of these components will require prospective studies specifically designed to compare variability dimensions against standardized retinal outcomes. Furthermore, interpretation of associations between glycemic variability and DR must account for several potential confounding factors. Insulin treatment intensity, use of complex insulin regimens, and frequent therapy adjustments are often associated with increased glucose fluctuations, while simultaneously reflecting more advanced disease or higher baseline retinopathy risk. Similarly, comorbid conditions such as renal impairment, hypertension, dyslipidemia, and cardiovascular disease may independently influence both metabolic stability and retinal microvascular vulnerability. Socioeconomic determinants, including access to diabetes technologies, healthcare continuity, education level, and treatment adherence, can further modulate glycemic patterns and screening practices, thereby affecting observed retinal outcomes. Although many observational studies adjust for mean HbA1c and selected clinical variables, residual confounding remains likely, particularly in real-world CGM datasets. These limitations highlight the necessity of cautious causal interpretation and reinforce the importance of longitudinal designs and interventional studies specifically targeting glycemic stability rather than glycemic averages alone. Finally, the preponderance of research from high-income healthcare environments may limit applicability to settings with varying access to diabetes technologies and ophthalmology services; access to continuous CGM technology is generally inconsistent. Costs, infrastructure requirements, and limited healthcare coverage may hinder adoption in low-resource settings, potentially exacerbating disparities in metabolic monitoring and retinal risk classification.
FUTURE OUTLOOKS
Future research must focus on standardizing glycemic variability measurements and determining clinically significant thresholds linked to retinal risk. Standardization of data from CGM would enhance comparability across studies and facilitate their integration into routine clinical practice. Longitudinal studies that integrate CGM data with multimodal retinal imaging, functional assessments, and genetic biomarkers are essential for elucidating the sequence of neurovascular alterations induced by metabolic instability. Such methodologies may facilitate the prompt identification of retinal damage and enhance personalized screening protocols. Among CGM-derived metrics, time in range currently appears particularly promising for clinical implementation, given its reproducibility, interpretability, and consistent associations with retinal outcomes across multiple cohorts. The coefficient of variation may offer additional value as a standardized measure of relative glycemic dispersion, particularly when exceeding proposed stability thresholds. Furthermore, cumulative exposure to glycemic extremes-reflected by time above range and time below range-may capture biologically relevant metabolic stress linked to oxidative injury and neurovascular dysfunction. Although definitive interventional evidence remains limited, these parameters represent pragmatic and mechanistically grounded candidates for future variability-targeted retinal outcome trials.
From a therapeutic perspective, there remains a critical need for interventional trials specifically targeting glycemic stability rather than mean glycemia alone. While intensive glucose lowering reduces long-term retinopathy risk, it may paradoxically exacerbate retinal injury when achieved rapidly or with excessive variability. Glucose-stabilizing technologies-including automated insulin delivery systems, hybrid closed-loop platforms, and advanced CGM-guided treatment algorithms-may reduce both excursion amplitude and exposure to glycemic extremes, thereby attenuating recurrent metabolic stress at its source. Prospective studies evaluating retinal imaging biomarkers in patients using these systems could clarify whether improved glucose stability translates into structural retinal protection. However, it is important to recognize that direct interventional evidence demonstrating that targeted reduction of glycemic variability independently decreases the incidence or progression of DR remains limited. Most available data derive from observational analyses or secondary evaluations of glucose metrics rather than prospective trials specifically designed to test variability as an independent therapeutic target. Carefully controlled longitudinal studies will therefore be necessary to determine whether improving metabolic stability translates into measurable retinal protection beyond mean glycemic control.
Beyond glucose modulation, adjunctive therapies targeting oxidative stress, inflammation, mitochondrial dysfunction, and epigenetic reprogramming represent a compelling frontier. Antioxidant strategies, anti-inflammatory agents, and interventions aimed at restoring mitochondrial homeostasis or reversing metabolic memory may help mitigate the persistent retinal damage induced by prior dysglycemia[59-61].
Mitochondrial injury is repeatedly implicated as a self-perpetuating source of oxidative stress in DR, and reviews emphasize that mitochondrial dysfunction/mitochondrial DNA damage and impaired mitochondrial homeostasis represent rational therapeutic targets, while also noting delivery challenges imposed by the blood-retinal barrier[62]. Inflammatory amplification-including NF-κB-linked signaling-is positioned as a key pathway connecting oxidative stress to leukostasis, barrier dysfunction, and neuroglial activation; preclinical work supports that inhibiting NF-κB signaling can attenuate glial reactivity in diabetic models, although clinical translation requires further validation[63]. Targeted inhibition of NADPH oxidase isoforms, particularly NOX2 and NOX4, has shown protective effects against diabetes-induced retinal oxidative damage and neurovascular injury in experimental systems, suggesting a feasible upstream intervention point[64]. Overall, these adjunct approaches should be evaluated cautiously and ideally in combination with improved glycemic stability, using standardized retinal structural and functional endpoints. Although most approaches remain preclinical interventions on oxidative and inflammatory stress pathways may offer translational benefit within the metabolic instability framework. Innovations in diabetes technology, such as automated insulin delivery devices, offer promising approaches to reduce metabolic fluctuations and should be evaluated explicitly for their effects on microvascular outcomes. Incorporating metabolic dynamics into research and clinical frameworks may transform preventive treatments for DR.
CONCLUSION
DR is increasingly recognized as a clinical manifestation closely associated with metabolic instability, rather than solely a consequence of chronic hyperglycemia. Glycemic variability, intermittent hyperglycemia, and hypoglycemia have distinct and cumulative effects on the retinal neurovascular unit, accelerating microvascular and neurodegenerative damage despite seemingly acceptable average glycemic indices. A mechanistic understanding of oxidative stress, mitochondrial dysfunction, inflammation, and metabolic memory provides molecular justification for these clinical findings. CGM is an effective method for assessing dynamic metabolic exposure and may improve retinal risk categorization beyond HbA1c alone. Implementing a dynamic metabolic framework may enhance screening, inform therapy timing, and ultimately alleviate the burden of vision loss linked to DR.
Peer review: Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Ophthalmology
Country of origin: Italy
Peer-review report’s classification
Scientific quality: Grade A, Grade B, Grade B
Novelty: Grade A, Grade B, Grade B
Creativity or innovation: Grade B, Grade B, Grade B
Scientific significance: Grade A, Grade B, Grade B
P-Reviewer: Singh PK, PhD, Post Doctoral Researcher, United States; Yang J, PhD, China S-Editor: Liu JH L-Editor: A P-Editor: Xu J