Published online Apr 26, 2026. doi: 10.12998/wjcc.v14.i12.120145
Revised: March 8, 2026
Accepted: March 19, 2026
Published online: April 26, 2026
Processing time: 57 Days and 16.4 Hours
Ampullary adenocarcinoma is often associated with favourable outcomes due to early presentation and high resectability. Small ampullary lesions are more commonly well-differentiated neuroendocrine tumours, which typically demon
A 69-year-old woman presented with painless obstructive jaundice. Imaging revealed a small ampullary lesion without evidence of metastatic disease. Endo
This case highlights the size-biology paradox in ampullary tumours, where small lesions may harbour highly aggressive histology. LCNEC of the ampulla of Vater remains rare and carries a potentially poor prognosis, warranting accurate pathological diagnosis and multidisciplinary management.
Core Tip: Large-cell neuroendocrine carcinoma (LCNEC) of the ampulla of Vater is a rare and aggressive malignancy that may present as a small lesion but requires oncologically intensive management. This striking discordance between tumour size, anatomical location, and biological behaviour underpins the size-biology paradox observed in ampullary LCNEC and highlights the importance of accurate histopathological classification and multidisciplinary oncological management.
- Citation: Florou E, Velayutham A, Zen Y, Waters J, Srinivasan P. Size-biology paradox in ampullary tumours - large-cell neuroendocrine carcinoma of the ampulla of Vater: A case report. World J Clin Cases 2026; 14(12): 120145
- URL: https://www.wjgnet.com/2307-8960/full/v14/i12/120145.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i12.120145
Adenocarcinoma of the ampulla of Vater is the most common malignant tumour arising from the ampullary region and generally carries a more favourable prognosis than other periampullary malignancies, largely owing to earlier pre
Neuroendocrine neoplasms (NENs) of the ampulla of Vater are rare, accounting for approximately 1%-2% of am
In contrast, poorly differentiated neuroendocrine carcinomas (NECs) of the ampulla of Vater, including large-cell neuroendocrine carcinoma (LCNEC), represent an exceptionally rare but highly aggressive subset[6]. LCNEC is characterised histologically by large polygonal cells, extensive necrosis, high mitotic activity, and a markedly elevated Ki-67 proliferation index[7,8]. Published case reports and small series describe early recurrence, frequent lymph node and liver metastases, and limited survival despite radical surgery, with reported median survival generally under two years[6]. Although pancreaticoduodenectomy remains the primary treatment modality, long-term outcomes are often limited by early systemic relapse[6].
This striking discordance between tumour size, anatomical location, and biological behaviour underpins the size-biology paradox observed in ampullary LCNEC and highlights the importance of accurate histopathological classification and multidisciplinary oncological management.
A 69-year-old woman presented with painless obstructive jaundice.
The patient initially presented in Jordan with progressive jaundice. Cross-sectional imaging demonstrated a double duct sign secondary to an ampullary lesion. She underwent endoscopic retrograde cholangiopancreatography (ERCP) with biliary stent placement prior to referral for further management.
Her medical history included hypertension, deep vein thrombosis treated with rivaroxaban, bilateral knee osteoarthritis, and gastroesophageal reflux disease. Surgical history was notable for three prior lower segment caesarean sections.
She denied tobacco or alcohol use and reported no family history of upper gastrointestinal malignancy.
Physical examination was unremarkable apart from clinical jaundice. No abdominal masses were palpable.
Laboratory investigations were consistent with obstructive jaundice. Tumour markers were not elevated.
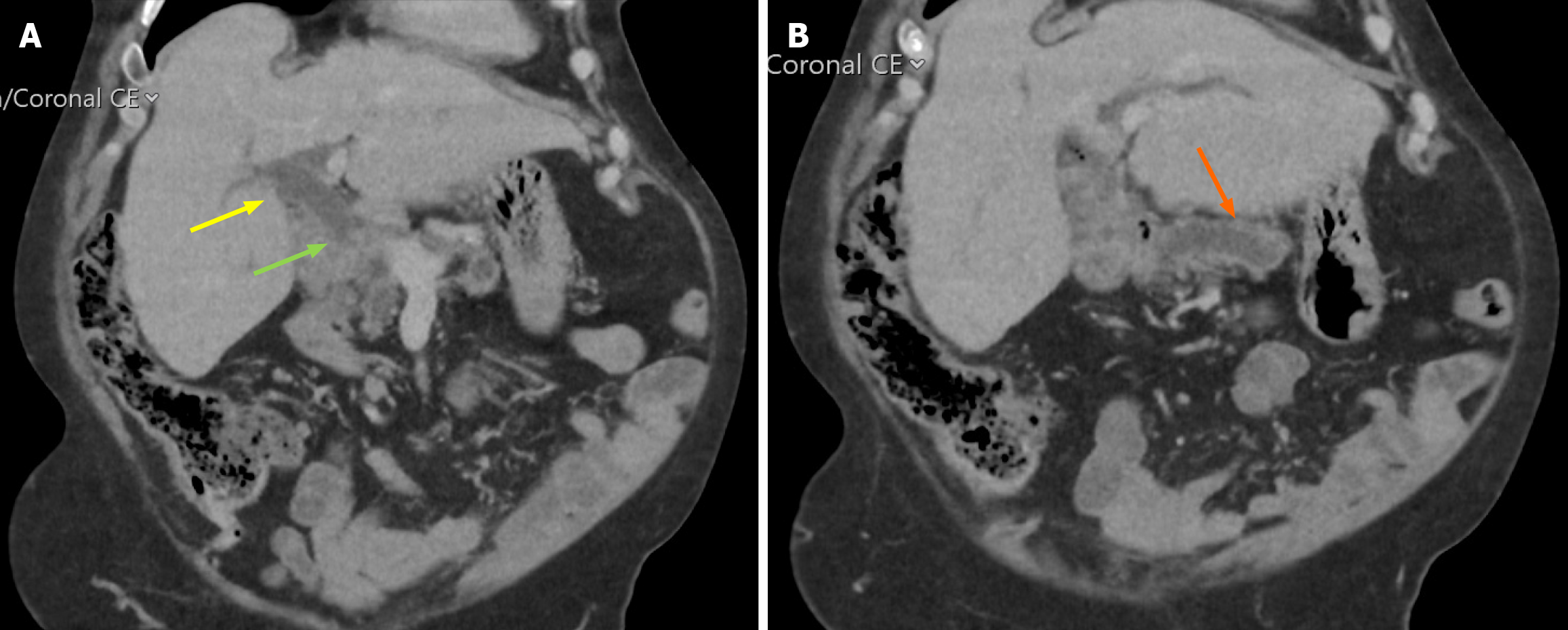
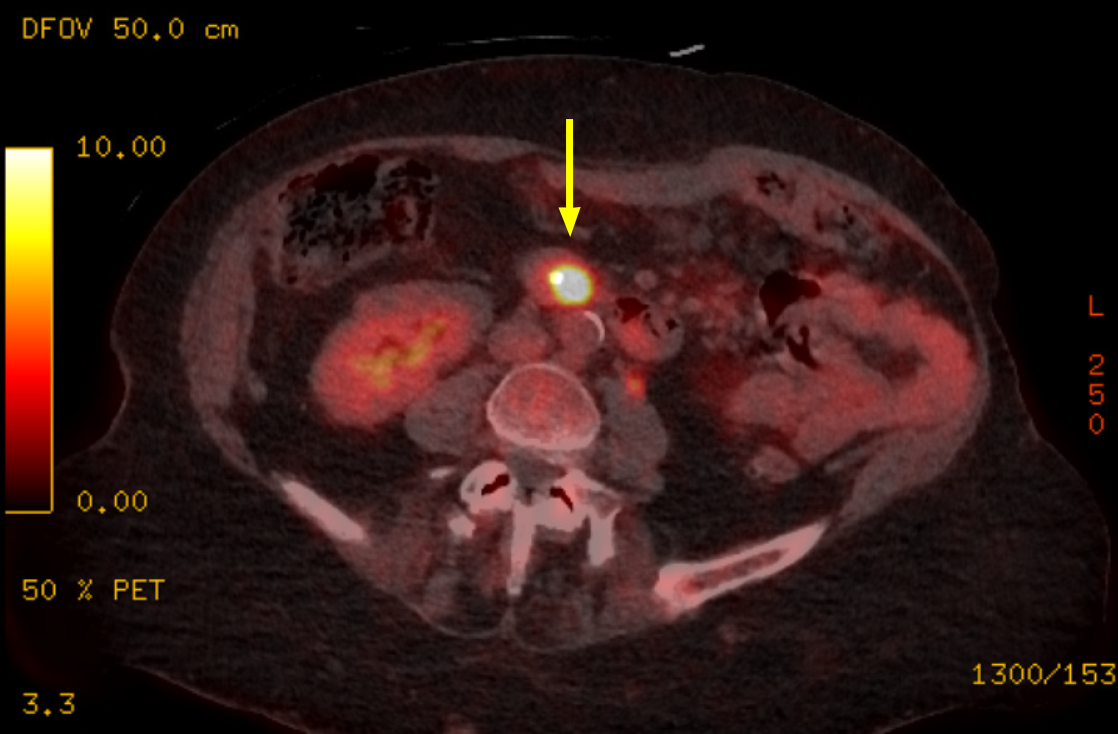
Contrast-enhanced computed tomography demonstrated dilated pancreatic duct measuring 9 mm with associated pancreatic parenchymal atrophy (Figure 1). Endoscopic visualisation of the ampullary lesion during ERCP was limited due to luminal collapse; a biliary stent was inserted. Positron emission tomography revealed no evidence of distant metastatic disease (Figure 2).
Endoscopic biopsy demonstrated moderately differentiated adenocarcinoma. Based on these findings, the working diagnosis was ampullary adenocarcinoma (cT2N0M0), and the case was discussed at the institutional hepatopancreatobiliary multidisciplinary team meeting, where pancreatoduodenectomy was recommended.
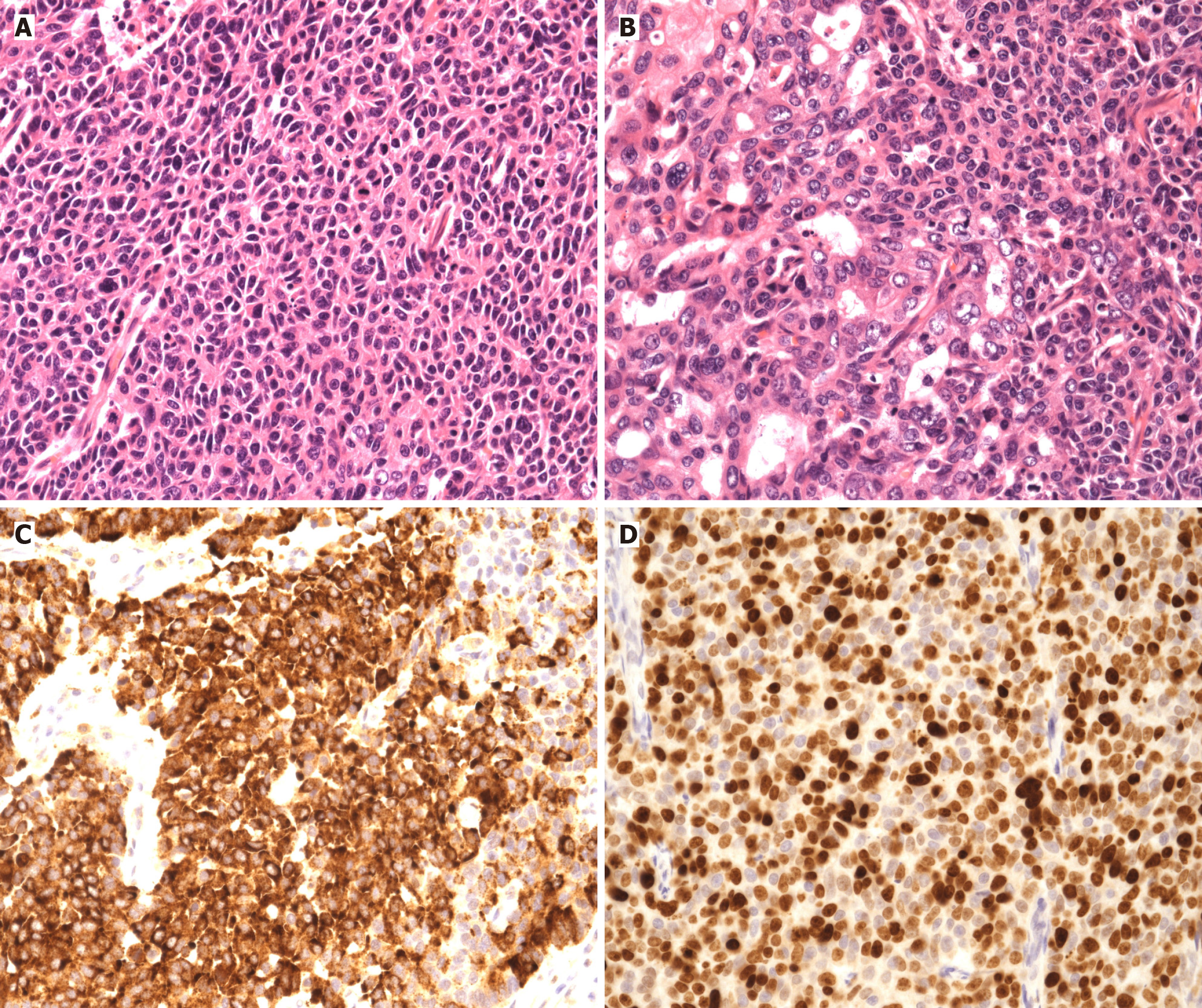
Histological examination of the resection specimen demonstrated a 16-mm poorly differentiated LCNEC of the ampulla of Vater, focally associated (< 10%) with adenocarcinoma and adenomatous elements (Figure 3). The tumour invaded the duodenal mucosa and submucosa and demonstrated metastatic involvement in 3 of 19 regional lymph nodes. Lymphovascular invasion was present and perineural invasion was focal. All resection margins were negative. Final pathological staging was pT1bN1 (3/19), LV1, PN1, R0. Immunohistochemistry showed diffuse chromogranin positivity and focal synaptophysin expression, with a Ki-67 proliferation index up to 70%.
The case was reviewed at a dedicated neuroendocrine tumour multidisciplinary meeting. In view of the high-grade histology and nodal involvement, adjuvant chemotherapy was recommended.
The patient underwent pylorus-preserving pancreaticoduodenectomy in July 2025.
The patient received six cycles of carboplatin (area under the curve 5) and etoposide (100 mg/m2 intravenously on day 1, followed by 200 mg/m2 orally on days 2 and 3) administered every 21 days.
Postoperative recovery was largely uncomplicated but prolonged due to a mild wound infection and multi-resistant Escherichia coli cultured from the biliary stent, requiring extended intravenous antibiotic therapy. The patient remained clinically stable throughout, with no episodes of sepsis.
At 8 months following surgery and completion of adjuvant chemotherapy, she remains clinically well with no radiological evidence of disease recurrence.
NENs of the ampulla of Vater are rare, accounting for a very small proportion of gastroenteropancreatic neuroendocrine neoplasms and less than 2%-6% of all ampullary malignancies[9,10]. Within this already uncommon group, poorly differentiated NECs represent a distinct and more aggressive biological subset[9]. Both small-cell NEC and large-cell NEC have been described in the ampullary region, with small-cell variants reported more frequently in early literature[10,11].
LCNEC of the ampulla of Vater is exceptionally rare, with fewer than several dozen well-documented cases reported to date[9,12]. The largest pooled analyses suggest that LCNEC demonstrates highly aggressive behaviour, characterised by frequent lymphovascular invasion, nodal metastasis, and early distant spread, most commonly to the liver[9]. Reported median overall survival in published series generally ranges between 8 months and 18 months despite complete surgical resection[9,12]. Mortality within the first 1-2 years after diagnosis remains common, reflecting the tumour’s high meta
Mixed histology, including adenomatous or adenocarcinoma components admixed with neuroendocrine carcinoma, has also been described and may support a convergent or shared carcinogenic pathway[11,12]. In the present case, focal glandular elements (< 10%) were identified, but the tumour did not meet criteria for mixed neuroendocrine-non-neuroendocrine neoplasm, consistent with prior reports of predominantly neuroendocrine-driven biology[9].
Surgical resection via pancreaticoduodenectomy remains the cornerstone of treatment when feasible[9]. However, surgery alone is rarely sufficient for high-grade NEC biology, as recurrence is common even after R0 resection[9,12]. Given the biological similarities between ampullary LCNEC and other extrapulmonary high-grade NECs, platinum-based chemotherapy regimens, typically combining cisplatin or carboplatin with etoposide, are widely adopted in the adjuvant setting despite the absence of prospective trials[9].
The present case reinforces the size-biology paradox in ampullary malignancy. Despite a small primary tumour measuring only 16 mm, nodal metastases were present at resection, supporting the concept that tumour biology rather than anatomical size is the dominant determinant of prognosis in high-grade ampullary NEC[9,10]. While long-term survival has been reported in rare instances, these cases remain exceptional and are often associated with early mul
This report has limitations inherent to single-case observations. Follow-up in our patient is currently limited to 8 months after completion of adjuvant chemotherapy, and longer surveillance is required to determine whether durable remission can be achieved. Given the aggressive natural history described in most published series, continued close radiological and clinical follow-up is essential. Close postoperative surveillance is recommended, including cross-sectional imaging with computed tomography or positron emission tomography/computed tomography every 3-6 months during the first two years, together with regular clinical review and biochemical monitoring where appropriate.
Large-cell neuroendocrine carcinoma of the ampulla of Vater is a rare but highly aggressive malignancy associated with early metastatic potential and limited survival in most reported cases. Accurate histopathological diagnosis, awareness of biopsy limitations, and multidisciplinary management incorporating systemic therapy are critical. Although our patient remains disease-free at short-term follow-up, longer observation is necessary before definitive prognostic conclusions can be drawn. Further collaborative studies are required to better define optimal management strategies for this challenging disease.
| 1. | Albores-Saavedra J, Schwartz AM, Batich K, Henson DE. Cancers of the ampulla of vater: demographics, morphology, and survival based on 5,625 cases from the SEER program. J Surg Oncol. 2009;100:598-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 225] [Article Influence: 13.2] [Reference Citation Analysis (3)] |
| 2. | Carter JT, Grenert JP, Rubenstein L, Stewart L, Way LW. Tumors of the ampulla of vater: histopathologic classification and predictors of survival. J Am Coll Surg. 2008;207:210-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 118] [Article Influence: 6.6] [Reference Citation Analysis (3)] |
| 3. | Kimura W, Futakawa N, Yamagata S, Wada Y, Kuroda A, Muto T, Esaki Y. Different clinicopathologic findings in two histologic types of carcinoma of papilla of Vater. Jpn J Cancer Res. 1994;85:161-166. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 206] [Cited by in RCA: 162] [Article Influence: 5.1] [Reference Citation Analysis (1)] |
| 4. | de Castro SM, van Heek NT, Kuhlmann KF, Busch OR, Offerhaus GJ, van Gulik TM, Obertop H, Gouma DJ. Surgical management of neoplasms of the ampulla of Vater: local resection or pancreatoduodenectomy and prognostic factors for survival. Surgery. 2004;136:994-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 5. | Randle RW, Ahmed S, Newman NA, Clark CJ. Clinical outcomes for neuroendocrine tumors of the duodenum and ampulla of Vater: a population-based study. J Gastrointest Surg. 2014;18:354-362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 80] [Article Influence: 6.7] [Reference Citation Analysis (1)] |
| 6. | Stojsic Z, Brasanac D, Bilanovic D, Mitrovic O, Stevanovic R, Boricic I. Large-cell neuroendocrine carcinoma of the ampulla of Vater. Med Oncol. 2010;27:1144-1148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 7. | Cavazza A, Gallo M, Valcavi R, De Marco L, Gardini G. Large cell neuroendocrine carcinoma of the ampulla of vater. Arch Pathol Lab Med. 2003;127:221-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 8. | Kagaya T, Miki A, Mito K, Fukushima N, Lefor AK, Sata N. Large Cell Neuroendocrine Carcinoma of the Ampulla of Vater With Long-Term Survival. Am Surg. 2026;92:1334-1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Karlafti E, Charalampidou M, Fotiadou G, Abba Deka I, Raptou G, Kyriakidis F, Panidis S, Ioannidis A, Protopapas AA, Netta S, Paramythiotis D. Ampullary Large-Cell Neuroendocrine Carcinoma, a Diagnostic Challenge of a Rare Aggressive Neoplasm: A Case Report and Literature Review. Diagnostics (Basel). 2022;12:1797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 1.0] [Reference Citation Analysis (1)] |
| 10. | Nassar H, Albores-Saavedra J, Klimstra DS. High-grade neuroendocrine carcinoma of the ampulla of vater: a clinicopathologic and immunohistochemical analysis of 14 cases. Am J Surg Pathol. 2005;29:588-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 77] [Article Influence: 3.7] [Reference Citation Analysis (4)] |
| 11. | Zhang L, DeMay RM. Cytological features of mixed adenoneuroendocrine carcinoma of the ampulla: two case reports with review of literature. Diagn Cytopathol. 2014;42:1075-1084. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (1)] |
| 12. | Imamura N, Nanashima A, Hiyoshi M, Fujii Y. Report of two cases of large cell neuroendocrine carcinoma of duodenal ampulla with contrasting outcomes following pancreaticoduodenectomy according to the use of adjuvant chemotherapy. Int J Surg Case Rep. 2017;31:132-138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (1)] |