Published online Apr 6, 2026. doi: 10.12998/wjcc.v14.i10.119088
Revised: February 9, 2026
Accepted: March 4, 2026
Published online: April 6, 2026
Processing time: 73 Days and 11.1 Hours
The simultaneous occurrence of pathogenic variants in both neurofibromatosis type 1 (NF1) and BRCA2 genes is extremely rare and remains poorly characterized. Each gene independently confers a substantial risk of malignancy, NF1 through dysregulation of the RAS/PI3K/AKT/mTOR pathway, while BRCA2 through homologous recombination deficiency. A potential synergistic impact of dual germline defects on tumor susceptibility, onset, and phenotype has been rep
In the article, we present the case of a 40-year-old woman with clinically con
This case underscores the clinical significance of dual pathogenic variants in NF1 and BRCA2, suggesting potential synergistic effects on early tumorigenesis and tumor biology. It highlights the importance of comprehensive ge
Core Tip: This case underscores the clinical significance of dual pathogenic variants in neurofibromatosis type 1 and BRCA2, suggesting potential synergistic effects on early tumorigenesis and tumor biology. It highlights the importance of comprehensive genetic testing in complex hereditary cancer presentations, as well as multidisciplinary surveillance, and individualized risk-reduction strategies.
- Citation: Czukiewska E, Narowska D, Czukiewski T, Pasieka I, Kuźniar A. Clinical consequences of the co-occurence of BRCA2 and NF1 mutation in ovarian cancer: A case report. World J Clin Cases 2026; 14(10): 119088
- URL: https://www.wjgnet.com/2307-8960/full/v14/i10/119088.htm
- DOI: https://dx.doi.org/10.12998/wjcc.v14.i10.119088
The BRCA2 (*600185 MIM) gene is a key suppressor gene involved in the repair of double-strand DNA breaks, and its damage leads to genome instability and tumor development[1]. The repair of double-strand DNA breaks involving the BRCA2 gene occurs through homologous recombination involving the RAD51C gene[2]. Pathogenic somatic variants in the BRCA2 gene are found in ovarian, prostate, and pancreatic cancers, as well as in other solid tumors[2]. In Poland, BRCA2 mutations are detected in approximately 0.7%-4% of patients with ovarian cancer, while the total frequency of BRCA1 and BRCA2 mutations in this population is estimated at 12%-18%[3]. Germline variants lead to hereditary breast and ovarian cancer syndrome, in which cancer risk in women increases to 20%-30% by the age of 70 for ovarian cancer, and to 60%-70% over a lifetime for breast cancer. Breast cancer is carrier of a pathogenic mutation clinical tumors are often highly differentiated, estrogen receptor -positive (ER+), which contrasts with the more aggressive, triple -negative breast cancer commonly associated with BRCA1. Whereas ovarian cancer tumors are typically high-grade serous car
The neurofibromatosis type 1 (NF1) (* 613113 MIM) gene is also a suppressor gene[7], and its mutations occur not only in hereditary cancers but also in sporadic cancers, including melanoma, leukemia, and lung cancer, among others[8-10]. Germline pathogenic variants in the NF1 gene, leading to loss of neurofibromin function, result in the phakomatosis, NF1 (#162200 MIM), which is a rare disorder characterized by café-au-lait spots, lisch nodules, and fibromatous tumors of the skin. Individuals with this condition have an increased susceptibility to the development of both benign and malignant tumors. NF1 is sometimes referred to as peripheral neurofibromatosis. Malignant peripheral nerve sheat tumors is the main malignant tumor associated with NF1, optic pathway gliomas - especially in children, pheochromocytomas and abnormalities, attention-deficit hyperactivity disorder and a other neuroendocrine tumors, elevated risk of other nerous system and cranial tumors. Additionally neurological and skeletal features appear as scoliosis, pseudoarthrosis, chest wall autism spectrum traits. NF1 is sometimes referred to as peripheral neurofibromatosis. The worldwide incidence of NF1 is estimated at 1 in 2500 to 1 in 3000 individuals[11,12].
Molecular diagnosis of NF1 is challenging due to the large size of the gene, the lack of mutational hotspots, and the wide spectrum of pathogenic variants[13,14]. NF1 (neurofibromin 1) is a gene that regulates the RAS/MAPK signaling pathway. It acts as a GTPase-activating protein by inhibiting Ras protein activity and its mutations cause Ras overactivity, which activates the Raf/MEK/ERK (MAPK) pathway, promoting cell growth and division. Another signaling pathway regulated by the loss of neurofibromin is PI3K/AKT/mTOR, which supports cell survival and growth. Carrying a mutation in the BRCA2 gene is associated with a significantly increased initial risk of ovarian cancer, whereas mutations in the NF1 gene are not considered to be a factor significantly increasing the risk of this cancer, and their clinical phen
Co-occurrence of germline pathogenic variants in both BRCA2 and NF1 is exceedingly rare and has been documented only in isolated case reports[4,9,11] . In our work, we refer to three described cases of people suffering from various neo
So far, two described cases of patients comprised a 41-year-old male[15] and a 40-year-old female[16]. While specific molecular details of the variants were not reported in either case, germline status was confirmed in one case through next-generation sequencing of both tumor tissue and saliva samples. The clinical presentations differed markedly between cases: The male patient presented with neurofibromas and extensive lipomatosis but notably lacked typical NF1 features such as café-au-lait spots or multiple neurofibromas, while the female patient presented with bilateral breast cancers and an aggressive pheochromocytoma phenotype. Both studies attributed these atypical presentations to potential gene - gene interactions between the two tumor suppressor genes[15,16]. Management strategies were tailored to individual presentations, including extensive surgical resections with long-term surveillance protocols in one case and perioperative alpha-blockade followed by adrenalectomy in the other. Family history of NF1 was documented in one case with maternal transmission, but neither study reported family history of BRCA2-associated cancer. Long-term follow-up data were not available for either patient. Another case of extremely rare mutations is presented by Cohen[17]. The authors described the potential development of the rare chronic lymphocytic leukemia in patients with NF1. In their article, they present a case of a 67-year-old woman with NF1, a BRCA2 mutation, and chronic lymphocytic leukemia (CLL). The patient exhibited typical NF1 manifestations, such as multiple neurofibromas, axillary freckling, and Lisch nodules. CLL was diagnosed at age 60, and treatment with rituximab began 41 months later, resulting in a favorable and sustained response. The review of prior cases indicates more aggressive disease courses. As BRCA1/BRCA2 mutations may increase CLL risk, the patient’s leukemia may be partly related to her BRCA2 status, suggesting that the NF1-CLL association may be coincidental yet influenced by additional genetic factors.
Von Recklinghausen disease is a genetic disorder with autosomal dominant inheritance, belonging to the group of phakomatoses. It is estimated that the incidence of the disease is approximately 1:3500 live births in the Polish population[16,18,19]. Clinically, von Recklinghausen disease is diagnosed based on the National Institutes of Health criteria, according to which the diagnosis can be made when at least 2 of the 7 conditions are met: 6 cafe-au-lait spots with a diameter > 15 mm, two or more neurofibromas of any type or one plexiform neurofibroma, freckled speckles on exposed parts of the body - armpits and groin, optic nerve glioma, two or more lisch nodules, sphenoid bone dysplasia, first-degree relative with NF1.
A 40-year-old Caucasian female patient of Polish origin presented to the Genetic Counseling Clinic in Lublin, Poland, due to a family history of breast cancer and a prior clinical diagnosis of von Recklinghausen disease (NF1), established on the basis of clinical criteria.
She was of European ethnicity and had no previously diagnosed malignancies at the time of genetic evaluation. The patient sought genetic consultation to initiate molecular diagnostics and to implement preventive surveillance strategies targeting cancers occurring in her family. The patient is currently undergoing treatment for serous ovarian cancer and requires ongoing support and monitoring.
The patient has a history of scoliosis and osteoarthritis.
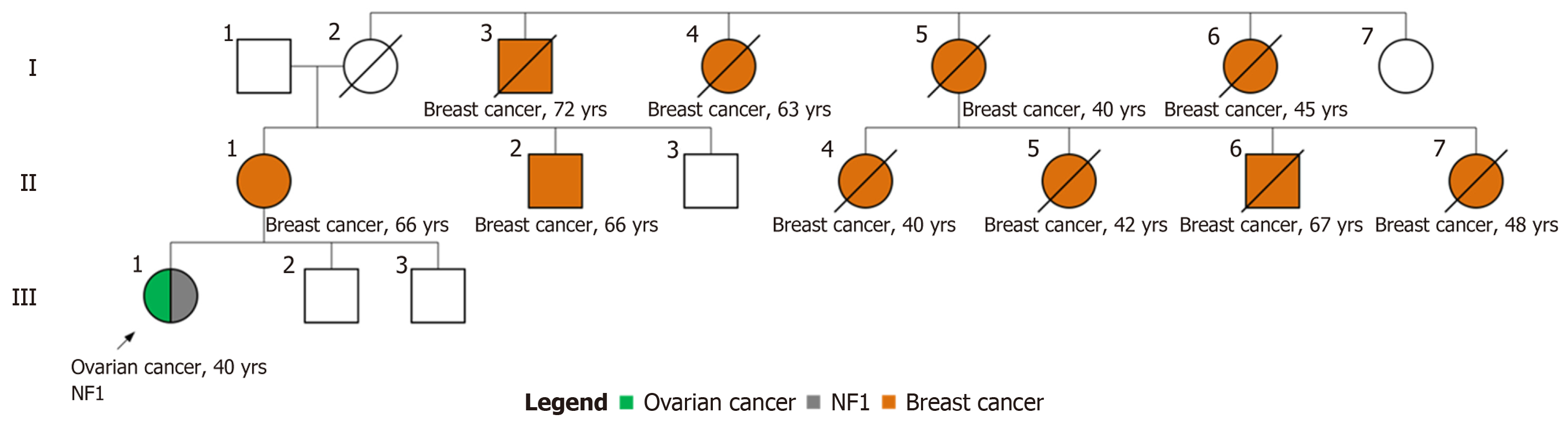
The proband’s mother as well as her maternal uncle were both diagnosed with breast cancer at the age of 66 years old. Her maternal great uncle was affected by breast cancer (72 years old at the time of the diagnosis) along with his three sisters (proband’s maternal great aunt’s) who were found to have breast cancer at the age of 63 years old, 40 years old, and 45 years old.
Figure 1 the description of the patient’s pedigree. The patient’s maternal great aunt’s descendants were also affected by breast cancer; three of her daughters at the age of 40, 42 and 48 as well as her son who was diagnosed at the age of 67.
A physical examination of the skin on the chest and back revealed several nerve-related nodules approximately 13 mm in size. Numerous thickened areas measuring 1-1.5 cm could be felt in the subcutaneous tissue of the chest and abdominal cavity.
Molecular diagnostic analysis: Genomic DNA was isolated from peripheral blood samples using the QIAsymphony system (QIAGEN GmbH, Hilden, Germany) according to the manufacturer’s instructions. To identify pathogenic variants within the coding sequences of the selected genes, targeted next-generation sequencing (NGS) was performed using the Ion AmpliSeq On-Demand DNA Panel Chef-Ready Library Preparation Kit (Thermo Fisher Scientific, Waltham, MA, United States).
Sequencing was carried out on the Ion S5 platform (Thermo Fisher Scientific) following standard protocols. Raw sequencing data were processed and analyzed using Ion Reporter software, version 5.20 (Thermo Fisher Scientific). The minimum mean coverage across targeted coding regions was ≥ 100 ×.
Variant analysis included single nucleotide variants and small insertions and deletions (indels) located within coding regions and flanking splice-site regions of the analyzed genes. The assay was not designed to detect large genomic rearrangements involving one or more exons. In addition, variant detection within homopolymer regions may be limited due to the technical characteristics of Ion Torrent sequencing technology.
Identified variants were interpreted and classified according to the recommendations of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology (ACMG)/(AMP guidelines). Variant classification was based on multiple criteria, including population frequency data (e.g., gnomAD), previously reported clinical significance (ClinVar, HGMD where available), in silico pathogenicity predictions, conservation analysis, and available literature. Variants classified as pathogenic or likely pathogenic were considered clinically relevant. Variants classified as benign or likely benign were not included in the final clinical report. As part of the molecular diagnostic work-up, a comprehensive panel of 97 genes was analyzed using NGS. The analyzed genes included: MEN1, MC1R, MAX, MAP2K1, KRAS, KIT, KIF1B, KCNK12, IDH1, IDH2, HRAS, HNF1B, HNF1A, H3F3A, GREM1, GNAQ, GNA11, GDNF, GALNT12, NFLCN, FH, FBXO11, FANCC, EXO1, ERBB3, EPCAM, EGFR, DIRC3, DICER1, CYP21A2, CTTNNA1, CHEK2, CDKN2A, CDH1, BRIP1, BRCA1, BRCA2, BRAF, BMPR1A, BARD1, BAP1, AXIN2, ATP9B, ATM, ARAF, APC, ALK, AKT1, NF1, NF2, MET, MITF, MLH1, MLH3, MRE11A, MSH2, MSH6, MUTYH, N4BP2 L1, NBN, NRAS, PALB2, PDGFRA, PIK3CA, PINLYP, PMS1, PMS2, POLD1, POLE, POT1, PRKAR1A, PRSS1, PTCH1, PTCH2, PTEN, RAD51C, RAD51D, RB1, RET, ROS1, SDHA, SDHAF2, SDHB, SDHC, SDHD, SMAD4, STK11, SUFU, TGFBR2, TMEM127, TP53, TSC1, TSC2, VHL, WT1, XRCC1, XRCC2.
BRCA2: C.8909G>A (p.Trp2970Ter) classified as pathogenic (PVS1, PM2, PP5) according to ACMG criteria associated with hereditary breast and ovarian cancer syndrome.
NF1: C.1527+1G > C classified as pathogenic (PVS1, PM2, PS4) is a splice site variant in the NF1 gene that disrupts normal RNA splicing, leading to an altered, shortened protein, and is classified as Pathogenic (P) for NF1 according to ACMG guidelines, causing disease by loss of function Abbreviation: PVS1 - pathogenic very strong; PM2 - pathogenic moderate; PP5 - pathogenic supporting; PS4 - pathogenic strong
No other pathogenic or likely pathogenic variants were detected in the remaining analyzed genes.
This comprehensive NGS panel enables the detection of germline variants associated with hereditary cancer syndr
The variant in the NF1 gene is absent from population database gnomAD; however it has been reported with a fre
The variant in the BRCA2 gene is absent from population database gnomAD; and its population frequency is unknown.
Cancer antigen 125 monitoring: As part of the diagnostic and follow-up evaluation, the patient underwent serial mea
Baseline staging computed tomography (CT) (December 2020) demonstrated peritoneal/liver capsular implants. Follow-up imaging after systemic therapy showed marked regression of lesions, and subsequent serial magnetic resonance imaging (MRI)/CT examinations through 2025 revealed no radiologic progression, with complete regression of pre
Based on clinical, laboratory, radiological, and histopathological findings, the patient was diagnosed with breast and ovarian cancer, along with von Recklinghausen disease, associated with the co-occurrence of pathogenic variants in the BRCA2 and NF1 genes.
The patient was clinically diagnosed with numerous neurofibroma-like nodules, approximately 13 mm in diameter, on the skin of her chest and back. According to her medical history, in 2017 she underwent surgery for a neurofibroma in the thoracic spine (Th). In the same year, on July 11, 2017, a neurofibroma-like lesion was removed from the left knee joint area, which was confirmed by histopathological examination. A similar lesion was excised from the skin of the right half of the chest, with a histopathological result also corresponding to neurofibroma. A CT scan of the chest revealed widening of the intervertebral foramina at the Th/L level, interpreted as the result of fibrous-neurofibromatous lesions. The main complaints reported by the patient included pain in the thoracic spine and both hip joints, associated with osteoarthritis. The risk of developing malignant breast and ovarian cancer was discussed with the patient, and re
The patient was referred for prophylactic bilateral adnexectomy. During the procedure, due to suspected ovarian cancer, an intraoperative histopathological examination was performed, which confirmed the clinical diagnosis of malignant neoplasm. A hysterectomy with adnexectomy, removal of the greater omentum and lymph nodes were performed (November 2020). The final histopathological examination revealed cancer of the left ovary – high-grade serous adenocarcinoma of the left ovary, FIGO III (T3cN0M0, according to the TNM classification). After the gynecological surgery, the patient received adjuvant postoperative chemotherapy with avastin + paclitaxel + carboplatin, three courses between January and February 2021. (January 2021-February 2021). On February 9, 2021, an implantable central venous access device (vascular port) was inserted. A follow-up chest X-ray on that day showed no abnormalities, including signs of pneumothorax. However, an X-ray performed on February 11, 2021, due to sudden dyspnea at rest, revealed iatrogenic pneumothorax on the right side. On the same day, drainage of the right pleural cavity was performed. After completion of the treatment of pneumothorax, chemotherapy was continued - on February 10, 2021, another, third course of treatment was administered in the A+S+K regimen, while the next cycle was performed on March 16, 2021, which the patient tolerated well. On March 22, 2021, the patient developed gastrointestinal obstruction due to peritoneal adhesions, requiring surgical intervention. During the procedure, the adhesions were released using an open method. The patient was discharged home in good general condition on March 28, 2021. After her general condition stabilized, adjuvant treatment was continued with three consecutive courses of chemotherapy in the tyrosine kinase inhibitor + paclitaxel regimen, administered from March 2021 to April 2021. Following a consultation, it was decided to discontinue chemotherapy after this cycle. On August 5, 2021, maintenance treatment with the PARP inhibitor Olaparib was started at a dose of 2 × 2 tablets of 150 mg for 28 days (first course). The patient continues treatment with Olaparib; the disease remains currently in remission. During oncological treatment, the patient was also diagnosed with peripheral poly
At the 24-month follow-up, the patient remained in good general condition with no evidence of disease recurrence. She tolerated olaparib-therapy well, with only mild adverse events. Regular oncological follow-up was scheduled.
The literature contains descriptions of individual patients or families in whom simultaneous variants were identified in the NF1 gene and in genes predisposing to breast cancer (more frequently mutations in BRCA1 and less often in BRCA2)[20,21]. These reports illustrate possible phenotypic combinations, such as early age at onset or atypical histological features, and suggest the need for broader genetic testing panels in selected families[22]. To better contextualize the present case, it is important to compare baseline cancer risks associated with each gene. Female carriers of pathogenic variants in BRCA1 or BRCA2 have well-established increased risks of both breast and ovarian cancer, warranting enhanced surveillance and risk-reducing strategies. Large prospective cohort studies have estimated cumulative ovarian cancer risks to age 70 of approximately 41% for BRCA1 and 15% for BRCA2 carriers, along with markedly elevated risks of breast cancer[23]. In contrast, although individuals with NF1 have an increased overall cancer risk, particularly for tumors of neural origin and early-onset breast cancer, current evidence does not indicate a clearly increased baseline risk of ovarian cancer attributable to NF1 alone. In this context, the occurrence of high-grade serous ovarian carcinoma in the present patient is most plausibly attributable to the BRCA2 pathogenic variant, consistent with the known association between homologous recombination deficiency and this tumor type. Conversely, the dermatologic manifestations and other clinical features characteristic of neurofibromatosis are consistent with the NF1 phenotype. The coexistence of pathogenic variants in both genes therefore appears to reflect overlapping but largely independent risk profiles, although a potential biological interaction cannot be excluded and requires further investigation in larger cohorts and tumor-based molecular studies.
The presented case illustrates the complex clinical and molecular background of a 40-year-old woman with NF1 and a significant family history of breast and ovarian cancer. The patient met the National Institutes of Health diagnostic criteria for NF1, presenting with multiple neurofibroma-like lesions on her chest and back, confirmed histopathologically in several locations, including the thoracic spine and extremities. Imaging studies revealed neurofibromatous lesions in the thoracolumbar spine, consistent with the systemic nature of the disease. The main symptom reported by the patient was chronic pain associated with neurofibromatous lesions and osteoarthritis. Given her family history of disease, multiple cases of breast and ovarian cancer in close relatives on her mother’s side, molecular testing was initiated to assess for the co-occurrence of pathogenic variants in NF1 and BRCA2. This dual predisposition is clinically significant because both genes are tumor suppressors involved in genomic stability and DNA repair[24]. Loss of NF1 leads to activation of the RAS/PI3K/AKT pathway and metabolic reprogramming, while BRCA2 mutations impair homologous recombination. The co-occurrence of both factors may synergistically enhance tumorigenesis, earlier disease onset, and resistance to treatment, as suggested by recent studies[20].
The patient underwent comprehensive oncological management following the diagnosis of high-grade serous ovarian carcinoma (FIGO III). Treatment included cytoreductive surgery, adjuvant chemotherapy (avastin + paclitaxel + carboplatin), and subsequent maintenance therapy with the PARP inhibitor olaparib, which remains effective with dis
Di Giosaffatte et al[26] showed that breast cancer associated with NF1 is characterized by earlier onset and a different molecular profile (Table 1). In a multicenter analysis of 86 patients with NF1 and breast cancer (and 111 cases from the literature), the average age of onset was 49 years, with carriers of missense variants developing the disease earlier than those with truncating mutations (43.9 years vs 49.5 years). The tumors were more often of high malignancy, with a HER2-enriched or luminal B phenotype, with luminal A being less common. Missense variants were concentrated in the N-HEAT domain of neurofibromin, mainly p.Leu847Pro and p.Tyr489*, and more than 70% of them were predicted to des
| Ref. | Patient (sex, age) | Clinical presentation/tumor spectrum | Germline variants (NF1; BRCA2) | Key management | Outcome/follow-up |
| Current case | Female, 40 | Clinically confirmed NF1; strong maternal family history of breast cancer; incidentally detected high-grade serous ovarian carcinoma (FIGO III; pT3cN0M0) | NF1: c.1527+1G>C (splice-site) BRCA2: c.8909G>A (p.Trp2970Ter) | Cytoreductive surgery (hysterectomy + BSO + omentectomy ± lymph nodes); platinum-taxane chemotherapy + bevacizumab; olaparib maintenance; intensified breast surveillance (MRI/MMG/US); prophylactic mastectomy declined | Durable remission; long-term CA125 normalization; no radiologic progression on serial imaging |
| Chaidarun et al[16], 2020 | Female, 40 | Bilateral breast cancer and pheochromocytoma in the context of NF1 | NF1: NR BRCA2: NR (reported as BRCA2-positive) | Preoperative alpha-blockade; adrenal surgery; oncologic management of breast cancer (details NR) | Favorable postoperative course; long-term follow-up NR |
| Choi[15], 2024 | Male, 41 | Intrathoracic neurofibroma and extensive lipomatosis; no malignancy reported | NF1: NR BRCA2: NR (combined germline NF1 and BRCA2 reported) | Surgical resection of intrathoracic neurofibroma; surveillance | Outcome NR (conference abstract; limited follow-up) |
| Cohen[17], 2021 | Female, 67 | Classic NF1 manifestations; chronic lymphocytic leukemia | NF1: NR BRCA2: NR (BRCA2-positive reported) | Rituximab therapy | Favorable and sustained response; no significant progression during reported follow-up |
The Polish case presented and previously reported cases highlight the growing recognition of complex cancer predisposition in individuals carrying pathogenic variants in both NF1 and BRCA2[27]. In the present patient, this combination was associated with high-grade serous ovarian cancer (FIGO III), whereas in a comparative report a patient with the same genetic background developed bilateral breast cancer and pheochromocytoma[16]. These observations illustrate that carriers of variants in both genes may present with diverse clinical phenotypes affecting different organs.
Pheochromocytoma, although a recognized manifestation of NF1, occurs relatively infrequently, and its coexistence with a BRCA2 pathogenic variant has rarely been described. In contrast, the Polish patient did not develop endocrine tumors but presented with extensive neurofibromatous lesions and ovarian cancer, reflecting variability in clinical ex
Taken together, these cases emphasize the heterogeneity of clinical presentation in patients with complex hereditary cancer predisposition and underscore the importance of individualized surveillance and multidisciplinary management.
In the literature, there are case reports involving male patients. Choi et al[15] described a 40-year-old man who underwent resection of a large intrathoracic neurofibroma. Both this case and the present report illustrate the rare coexistence of germline variants in NF1 and BRCA2, but with different clinical manifestations. The male patient presented mainly with multiple lipomas and intrathoracic neurofibromas and did not develop malignant tumors, whereas the 40-year-old female patient described in this study developed high-grade serous ovarian cancer in addition to multiple neurofibromas and a strong family history of breast and ovarian cancer. Management also differed, with surgical res
Another rare scenario was reported by Cohen[17], who described a 67-year-old woman with NF1, a BRCA2 pathogenic variant, and CLL. The patient presented with typical features of NF1 and achieved a favorable response to rituximab therapy. Compared with the present case, these reports demonstrate that individuals carrying pathogenic variants in both genes may develop diverse malignancies and clinical phenotypes.
Taken together, the available case reports suggest considerable variability in clinical presentation and underscore the importance of individualized surveillance strategies, genetic counseling, and long-term monitoring in patients with complex hereditary cancer predisposition[28,29].
A report of pathogenic NF1 germline variants in individuals with NF1 and breast cancer (NF1 + BC) provides valuable information on genotype-phenotype correlations and cancer predisposition[26]. Of 71 individuals with NF1 + BC, causal NF1 variants were identified in 69 (97%), with 65 classified as pathogenic/Likely pathogenic (P/LP) and four as variants of uncertain significance according to ACMG/AMP guidelines. A total of 63 unique variants were detected, including 25 novel and 38 previously reported in LOVD. The majority were truncating variants, with two multiexon deletions identified by MLPA; no whole-gene deletions were observed. Combined with data from the literature (n = 111), 180 NF1 + BC cases were analyzed, revealing pathogenic variants scattered throughout the NF1 gene. Eighteen individuals carried variants affecting splicing (SS category). Compared with the overall NF1 cohort (LOVD, n = 5444), NF1 + BC cases had fewer large deletions (0/180 vs 142/5444; P = 0.0148). Importantly, missense variants in the N-terminal HEAT domain were significantly more numerous [16/23 (70%) vs 336/805 (42%); P = 0.007775], as were variants in the CSRD domain [12/23 (52%) vs 192/805 (24%); P = 0.029311], and several individual variants were significantly overrepresented (Table 1). Consistent with previous reports[20-28], missense variants were associated with earlier breast cancer onset, with a mean age of 43.9 years vs 49.5 years for truncating variants (P = 0.014; Cohen’s d = 0.587), suggesting the functional significance of domain-specific mutations. Compared to the Polish patient, several similarities and differences emerge. The patient carries the BRCA2 gene: C.8909G>A (p.Trp2970Ter), classified as pathogenic, which likely increases breast cancer risk independently of NF1. Finally, the patient’s NF1 splice site variant c.1527+1G>C (ID 431589) was classified as pathogenic and responsible for the classic symptoms of NF1. Comparison of her genetic profile with the NF1 + BC cohort highlights several points: The patient’s NF1 variant is a splice site mutation that falls within the SS category identified in this cohort. Unlike the cohort, which shows an enrichment of missense variants in the HEAT and CSRD domains, the Polish patient carries a splice site variant, reflecting the diversity of NF1 mutations associated with breast cancer. Fur
A limitation of the present study is the lack of tumor genomic profiling. Specifically, analyses assessing loss of heterozygosity, homologous recombination deficiency scores, and somatic alterations in NF1 were not performed, which precludes definitive conclusions regarding biallelic inactivation and potential biological interactions at the tumor level. Consequently, the interpretation of the role of NF1 alterations in tumorigenesis remains limited. Future studies incor
Despite these limitations, this case highlights important clinical considerations in the context of NF1-associated breast and ovarian cancer, including the need for multidisciplinary management combining genetic counseling, targeted surveillance, and individualized systemic therapy. Emerging evidence suggests that NF1 deficiency may contribute not only to tumorigenesis through RAS pathway dysregulation, but also to specific metabolic vulnerabilities, such as inc
Early identification of patients harboring pathogenic variants in both NF1 and BRCA2 may be clinically relevant for optimizing preventive strategies and improving outcomes. The coexistence of pathogenic variants in these genes suggests a potential interaction that may contribute to increased susceptibility to malignancies affecting multiple organs and may influence tumor biology, metabolic features, and therapeutic responses. However, current evidence remains limited and largely observational, and mechanistic relationships have not yet been established. Further clinical and translational studies are therefore needed to better understand the biological and clinical implications of this rare genetic combination and to refine prevention and treatment strategies in such genetically complex patients.
| 1. | Carbone FP, Ancona P, Volinia S, Terrazzan A, Bianchi N. Druggable Molecular Networks in BRCA1/BRCA2-Mutated Breast Cancer. Biology (Basel). 2025;14:253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 2. | Quaas M, Kohler R, Nöltner L, Schmidbauer LF, Uxa S, Müller GA, Engeland K. BRCA1 and BRCA2 gene expression: p53- and cell cycle-dependent repression requires RB and DREAM. Cell Death Differ. 2026;33:51-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 3. | Feszak S, Feszak IJ, Kluźniak W, Wokołorczyk D, Stempa K, Gliniewicz K, Uciński J, Huzarski T, Dębniak T, Gronwald J, Lubiński J, Narod SA, Cybulski C. BRCA1 and BRCA2 Mutations in Polish Women with Ductal Carcinoma In Situ. Cancers (Basel). 2025;17:613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 4. | Armstrong N, Ryder S, Forbes C, Ross J, Quek RG. A systematic review of the international prevalence of BRCA mutation in breast cancer. Clin Epidemiol. 2019;11:543-561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 185] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 5. | Jasiewicz A, Rudnicka H, Kluźniak W, Gronwald W, Kluz T, Cybulski C, Jakubowska A, Lubiński J, Gronwald J. Frequency of BRCA1 and BRCA2 mutations in ovarian cancer patients in South-East Poland. Hered Cancer Clin Pract. 2022;20:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | Peng Y, Liao J, He X, Zhou Y, Zhang L, Jia Y, Yang H. Prevalence of BRCA1 and BRCA2 mutations in ovarian cancer patients from Yunnan Province in southwest China. Eur J Cancer Prev. 2025;34:231-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 7. | Wang W, Wei CJ, Cui XW, Li YH, Gu YH, Gu B, Li QF, Wang ZC. Impacts of NF1 Gene Mutations and Genetic Modifiers in Neurofibromatosis Type 1. Front Neurol. 2021;12:704639. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 37] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 8. | Lin LL, Choucair K, Patel R, Albalawy RS, Morand SM, Stanbery L, Albrethsen M, Nemunaitis M, Walter A, Charles Brunicardi F, Dworkin L, Nemunaitis J. NF1 in Solid Tumors: The Unknown Soldier of Tumor Suppressor Genes? Genet Mol Med. 2019;1:1-13. [DOI] [Full Text] |
| 9. | Kuhn E, Natacci F, Corbo M, Pisani L, Ferrero S, Bulfamante G, Gambini D. The Contribution of Oxidative Stress to NF1-Altered Tumors. Antioxidants (Basel). 2023;12:1557. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 10. | Báez-Flores J, Rodríguez-Martín M, Lacal J. The therapeutic potential of neurofibromin signaling pathways and binding partners. Commun Biol. 2023;6:436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 31] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 11. | Williams VC, Lucas J, Babcock MA, Gutmann DH, Korf B, Maria BL. Neurofibromatosis type 1 revisited. Pediatrics. 2009;123:124-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 554] [Cited by in RCA: 430] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 12. | Grech VS, Lotsaris K, Touma TE, Kefala V, Rallis E. The Role of Artificial Intelligence in Identifying NF1 Gene Variants and Improving Diagnosis. Genes (Basel). 2025;16:560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 13. | Kiraz A. Detection of Novel NF1 Variants with Next Generation-based DNA Sequencing Technology, and Genotype-Phenotype Characteristics of Neurofibromatosis. Erciyes Med J. 2023;45:152-158. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Huang J, Yang K, Wang Y, Ma X, Yang W, Huo X, Bai J, Zhu H, Wang J, Lv Y, Liao S. Unraveling novel variants in the NF1 gene and investigating potential therapeutic strategies. Sci Rep. 2025;15:24008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 15. | Choi Y. EPCO-59. First case report of combined germline NF1 and BRCA2 mutations presented with neurofibromas and lipomatosis. Neuro-Oncology. 2024;26:viii15. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 16. | Chaidarun T, Macleod SR, Chaidarun S. SAT-222 Aggressive Phenotype Pheochromocytoma Associated with NF-1 and BRCA Mutation. J Endocr Soc. 2020;4 (Suppl 1):SAT-222. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Cohen PR. Chronic Lymphocytic Leukemia in Neurofibromatosis Type 1 Patients: Case Report and Literature Review of a Rare Occurrence. Cureus. 2021;13:e14258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | Wolder D, Swat A. A family case report of Neurofibromatosis. J Educ Health Sport. 2022;12:108-115. [DOI] [Full Text] |
| 19. | Fijałkowska M, Antoszewski B. Satisfaction from surgical treatment and its influence on the wellbeing of patients with Recklinghausen disease - preliminary report. Pol Przegl Chir. 2020;92:31-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 20. | Gensini F, Sestini R, De Luca A, Pinna V, Daniele P, Orzalesi L, Petrella MC, Porfirio B, Papi L. Early-onset malignant phyllodes breast tumor in a patient with germline pathogenic variants in NF1 and BRCA1 genes. Fam Cancer. 2021;20:195-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 21. | Sukumar J, Kassem M, Agnese D, Pilarski R, Ramaswamy B, Sweet K, Sardesai S. Concurrent germline BRCA1, BRCA2, and CHEK2 pathogenic variants in hereditary breast cancer: a case series. Breast Cancer Res Treat. 2021;186:569-575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 22. | Ferrer-Avargues R, Castillejo MI, Dámaso E, Díez-Obrero V, Garrigos N, Molina T, Codoñer-Alejos A, Segura Á, Sánchez-Heras AB, Castillejo A, Soto JL. Co-occurrence of germline pathogenic variants for different hereditary cancer syndromes in patients with Lynch syndrome. Cancer Commun (Lond). 2021;41:218-228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 23. | Masgras I, Cannino G, Ciscato F, Sanchez-Martin C, Darvishi FB, Scantamburlo F, Pizzi M, Menga A, Fregona D, Castegna A, Rasola A. Tumor growth of neurofibromin-deficient cells is driven by decreased respiration and hampered by NAD(+) and SIRT3. Cell Death Differ. 2022;29:1996-2008. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 24. | Ho WK, Hassan NT, Yoon SY, Yang X, Lim JMC, Binte Ishak ND, Ho PJ, Wijaya EA, Ng PP, Luccarini C, Allen J, Tai MC, Chiang J, Zhang Z, See MH, Thong MK, Woo YL, Dunning AM, Hartman M, Yip CH, Mohd Taib NA, Easton DF, Li J, Ngeow J, Antoniou AC, Teo SH. Age-specific breast and ovarian cancer risks associated with germline BRCA1 or BRCA2 pathogenic variants - an Asian study of 572 families. Lancet Reg Health West Pac. 2024;44:101017. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 25. | Dibble KE, Donorfio LKM, Britner PA, Bellizzi KM. Stress, anxiety, and health-related quality of life in BRCA1/2-positive women with and without cancer: A comparison of four US female samples. Gynecol Oncol Rep. 2022;42:101033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 26. | Di Giosaffatte N, Daniele P, Petrizzelli F, Iacovino C, Canciani C, Garau ML, Santoro C, Trevisan V, Panfili A, Cavone S, Guida V, D'Asdia MC, Bernardini L, Majore S, Ferraris A, Valiante M, Gensini F, Radio FC, Tortora G, Cassina M, Miele G, Priolo M, Sirchia F, Piccinno L, Flex E, Zampino G, Genuardi M, Nigro V, Salviati L, Papi L, Grammatico P, Leoni C, Piluso G, Giustini S, Mazza T, Upadhyaya M, Tartaglia M, Trevisson E, De Luca A. Subtype distribution, clinical presentation, and molecular spectrum of neurofibromatosis type 1-associated breast cancer. Breast. 2025;84:104618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 27. | Mo J, Moye SL, McKay RM, Le LQ. Neurofibromin and suppression of tumorigenesis: beyond the GAP. Oncogene. 2022;41:1235-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 28. | Bogusławska A, Rzepka E, Opalińska M, Sowa-Staszczak A, Gilis-Januszewska A, Hubalewska-Dydejczyk A. A rare case of metastatic pheochromocytoma in the course of neurofibromatosis type 1. Pol Arch Intern Med. 2022;132:16291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 29. | Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E, Voelkerding K, Rehm HL; ACMG Laboratory Quality Assurance Committee. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405-424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27361] [Cited by in RCA: 25316] [Article Influence: 2301.5] [Reference Citation Analysis (8)] |