Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.113303
Revised: September 11, 2025
Accepted: January 6, 2026
Published online: March 25, 2026
Processing time: 205 Days and 11.8 Hours
Primary membranous nephropathy (MN) is a major cause of nephrotic syndrome in adults, characterized by immune complex formation on the outer side of the glomerular basement membrane. Primary MN accounts for 80% of the cases, and 20% cases are associated with a secondary etiology. While immunosuppressive therapy has improved patient outcomes in MN, the clinical profile and treatment outcomes of MN in our patient population have not been previously studied.
To investigate the clinicopathological profile and outcomes of primary MN patients. We included consenting patients aged ≥ 18 years with primary MN and excluded those with end-stage kidney disease, pregnancy, or secondary MN.
This study approved by the institutional ethics committee, was conducted at the Department of Nephrology, Sher-I-Kashmir Institute of Medical Sciences, Srinagar, India. Detailed history, physical examination, laboratory investigations [including 24-hour urinary protein, kidney and liver function tests, and anti-phospholipase A2 receptor (PLA2R) antibody levels], and screening for secondary etiologies were performed. Renal biopsy tissues were examined using light microscopy, immunofluorescence, immunohistochemistry for PLA2R, throm
Of 46 patients, 60.9% were female, with a mean age of 43.8 ± 13.7 years. The mean proteinuria was 5.8 ± 3.2 g/day, mean serum albumin was 2.5 ± 0.39 g/dL, and mean estimated glomerular filtration rate was 99.8 ± 26.6 mL/minute/1.73 m2. Edema was the most common symptom (100%), and hypertension was the most frequent comorbidity (37%). Serum anti-PLA2R antibodies were positive in 39.1% of patients. On immunohistochemistry, 58.7% were tissue PLA2R positive and 17.4% were NELL-1 positive. At baseline, 71.1% of patients were in the high-risk category. Initial non-immunosuppressive supportive care was given to 84.8% of patients, with 23.08% achieving remission. Among those receiving immunosuppressive therapy, 72.2% on modified Ponticelli regimen and 85.7% on other regimens achieved remission at 6 months (P = 0.432). At 12 months, 82.9% of patients on immunosuppressive therapy achieved remission. Both modified Ponticelli and other immunosuppressive regimens significantly improved proteinuria and serum albumin at 12 months (P < 0.0001).
PLA2R-associated MN was the most common form of MN, followed by NELL-1. Primary MN prevalence was highest in the fifth and sixth decades of life, with a slight female preponderance observed in this study. Most patients belonged to the high-risk group. Immunosuppression led to complete remission in over 80% of patients, with no significant difference in remission rates between different immunosuppressive agents. This study provides initial insights into MN in the local patient population, despite its limitations of being a single-center observational study with a relatively small sample size and short follow-up.
Core Tip: In this single-center study from Northern India, phospholipase A2 receptor-associated membranous nephropathy was identified as the most common form, with most patients presenting in a high-risk category and a slight female predominance observed. While initial supportive care had limited success, immunosuppressive therapy was highly effective, leading to remission in over 80% of patients. Importantly, there was no significant difference in remission rates between the modified Ponticelli and other immunosuppressive regimens. The study highlights the effectiveness of immunosuppression in this patient population, despite its limitations as a small-scale, single-center observational study.
- Citation: Wani MA, Khan I, Rather JI, Bhat MA, Wani MM, Wani IA, Sheikh RY, Parry MA. Clinical profile and outcome of primary membranous nephropathy: A tertiary care center experience from North India. World J Nephrol 2026; 15(1): 113303
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/113303.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.113303
The term membranous nephropathy (MN) indicates a pathological condition characterized by diffuse thickening of the glomerular basement membrane and involves the whole glomerulus[1]. It occurs due to immune complex formation on the outer side of the glomerular basement membrane. MN is a pathological diagnosis on the renal biopsy. MN can be primary MN, where the disease is limited to the kidneys without systemic involvement, or secondary MN which is associated with other diseases like autoimmune diseases, infections, malignancy, and drugs. Primary MN accounts for 80% of cases and 20% are associated with other systemic diseases or exposures[2]. Primary MN represents the commonest cause of idiopathic nephrotic syndrome in adult non-diabetics worldwide, representing between 20% and 37% in many series and rising to as high as 40% in adults over 60[3]. Antibodies to phospholipase A2 receptor (PLA2R) are found in 70% of patients and are specific for MN[4]. Other podocyte antigens have been found implicated like thrombospondin type 1 domain-containing 7A in < 5% of primary MN[5], exostosin 1 (EXT1) and EXT2, semaphorin 3B, protein kinase C-binding protein neural epidermal growth factor-like 1 (NELL1) (also known as NELL-1 protein), protocadherin 7, neural cell adhesion molecule 1 and serine protease high temperature requirement factor A1[6]. Progression of MN is variable; one-third of the patients have spontaneous remission, one-third have variable levels of proteinuria, and the remaining third progress to advanced kidney failure. Immunosuppressive therapy has substantially reduced the rate of kidney replacement therapy[2]. In untreated MN, complete remission rates of 20%-30% with 60%-80% 10-year renal survival rates are reported in many studies[7]. In patients who have persistent nephrotic syndrome, renal failure develops over a period of 10 years in 40%-50%[8].
Therapy of MN included supportive care and immunomodulatory therapies. Patients with MN need intensive monitoring to identify the estimated risk of progression of renal function deterioration. Patients at high risk of progression of kidney disease are considered for immunosuppressive therapies[9,10]. Despite being the commonest cause of nephrotic syndrome in non-diabetic adults, the clinical profile and treatment outcomes of MN are unknown in our patient population. Understanding these characteristics is essential to guide management and improve patient care.
This study conducted in the Department of Nephrology, Sher-I-Kashmir Institute of Medical Sciences, Srinagar, India. The duration of the study was 2 years. The study was approved by the Institutional Ethics Committee of Sher-I-Kashmir Institute of Medical Sciences (319/2022). The primary objective of this study was to evaluate the clinicopathological profile and outcome of the patients with primary MN. Inclusion and exclusion criteria: All the consenting patients aged more than or equal to 18 years with biopsy-proven primary MN were included in the study. Patients with secondary MN, pregnancy, and end-stage kidney disease were excluded from the study.
Patients who were diagnosed with primary MN during the study period, and those patients who were already diagnosed and were on follow-up and had required data available, were included in this study. A detailed history of current illness, past history, or comorbidity, as well as personal and drug history, was taken. A detailed general physical and systemic examination was done. History of any potential clue for secondary MN was sought, including history of skin whitening cream use, herbal medicine, or supplement intake.
Laboratory investigations included a complete hemogram, kidney, and liver function tests, 24-hour urinary protein excretion or urine protein-creatinine ratio, and a basic metabolic panel including blood glucose, lipid profile, and electrolytes. Patients were screened for any secondary etiology of MN by viral serology for hepatitis B virus, hepatitis C virus, and Human immunodeficiency virus. Appropriate patients with suspicion of autoimmune disease were screened for anti-nuclear antibody, anti-double-stranded DNA, and complement levels. Age-appropriate screening for malignancy with chest X-ray, stool for occult blood, gastrointestinal endoscopy, imaging like computed tomography and/or magnetic resonance imaging, mammography, Papanicolaou smear, tumor markers like carbohydrate antigen 125, carcinoembryonic antigen, carbohydrate antigen 19-9, alpha-fetoprotein, prostate-specific antigen was done. Serum anti-PLA2R antibody levels were measured at baseline and during monitoring of disease activity. Patients were classified based on the risk of progression of renal disease, which helped in management decisions. Treatment medications, including immunosuppressive agents, received by patients were recorded. All patients were followed periodically, and disease status was noted.
Renal biopsy tissue was examined in detail using light microscopy, immunofluorescence microscopy for immunoglobulin A, immunoglobulin M, immunoglobulin G and its subtypes, complement 3, complement 1q, kappa, and lambda light chains, immunohistochemistry for PLA2R, thrombospondin type-1 domain-containing 7A, NELL-1, EXT1, EXT2, and electron microscopy, and detailed histopathological findings were noted. Patients were risk-stratified on the basis of proteinuria, kidney function, and serum anti-PLA2R antibody levels as per Table 1.
| Very high risk | High risk | Moderate risk | Low risk |
| Presence of any 2 of the following is considered to be at very high risk of disease progression: Serum creatinine > 1.5 mg/dL, considered to be due to MN; progressive decline in kidney function, > 25% decline in eGFR from the baseline over the prior 2 years; severe, disabling, or life-threatening nephrotic syndrome is defined by serum albumin < 2.5 g/dL and a refractory edema, or a thromboembolic event | Presence of ≥ 2 of the following is considered to be high risk: Decrease in eGFR > 25% not explained by other causes at any time during the observation period; proteinuria > 8 g/day at the end of the observation period or persistent nephrotic syndrome; patient is anti-PLA2R antibody positive, serial anti-PLA2R antibody titres are high (> 150 RU/mL by ELISA) and not decreasing or are increasing to > 150 RU/mL | Presence of ≥ 2 of the following: Normal or stable eGFR (< 25% decrease) over a 3-month to 6-month period; persistent proteinuria between 4 g/day and 8 g/day at the end of the observation period; patient is anti-PLA2R antibody positive, serial anti-PLA2R antibody titres are < 150 RU/mL by ELISA and stable or increasing by < 25% over 6-month period | Normal or stable eGFR (< 25% decrease) over a 3-month to 6-month period; proteinuria < 4 g/day at the end of the observation period; patient is anti-PLA2R antibody positive, serial anti-PLA2R antibody titres are persistently low < 50 RU/mL by ELISA or are decreasing by ≥ 25% at 3-month to 6-month period |
Complete remission was defined as a reduction in proteinuria to < 300 mg/day with normal serum albumin and renal function. Partial remission was defined as a reduction of proteinuria > 50% from the baseline to > 300 mg/day and < 3.5 g/day accompanied by normalization of serum albumin and stable renal function. Relapse was defined as the return of proteinuria to > 3.5 g/day after an initial complete or partial response. Patients with a relapse after partial remission should have a > 50% increase in proteinuria from the nadir in addition to proteinuria > 3.5 g/day. Resistant disease was defined as the persistent or worsening of proteinuria and hypoalbuminemia which prompted the addition or change in immunosuppressive therapy in the absence of intolerance or adverse effects of the immunosuppressive regimen.
The data obtained was compiled in Microsoft Excel and subjected to analysis using SPSS software. Values were expressed as mean ± SD. Between-group comparisons for continuous variables were done using the independent t-test and Mann-Whitney test for normal and non-normal (distribution) data, respectively. P < 0.05 was considered to indicate statistical significance. The χ2 test was utilized for categorical variables.
A total of 46 patients were included in the study, out of which 28 (60.9%) were females and 18 (39.1%) were males. The average age of our patients was 43.8 ± 13.7 years. Of 19 (41.3%) patients fell in the age group of 45-60 years. This was followed by the age group of 30-45 years, which accounted for 14 (30.4%) patients. Only 4 (8.7%) of patients were above 60 years of age. The mean proteinuria of our patients was 5.8 ± 3.2 g per day. The mean serum albumin and creatinine were respectively 2.5 ± 0.39 g/dL and 0.8 ± 0.3 mg/dL. The mean estimated glomerular filtration rate was 99.8 ± 26.6 mL/minute/1.73 m2. The mean anti-PLA2R titer was 81.6 ± 148.5 RU/mL. The baseline characteristics are shown in Table 2. Edema was the most common symptom seen in all patients. Of 73.9% patients had nephrotic syndrome. The clinical features are shown in Table 3. The most common comorbidity was hypertension, which was present in 17 (37%) patients. The comorbidity status is shown in Table 4.
| Parameter | Mean | SD |
| BMI (kg/m2) | 24.8 | 3.4 |
| Systolic blood pressure (mmHg) | 126 | 12.7 |
| Diastolic blood pressure (mmHg) | 75.7 | 8.7 |
| Cholesterol (mg/dL) | 253.1 | 84.4 |
| Triglycerides (mg/dL) | 297 | 89.5 |
| LDL (mg/dL) | 148 | 54.1 |
| HDL (mg/dL) | 52.3 | 14.2 |
| Hemoglobin (g/dL) | 12.3 | 1.7 |
| Total serum protein (g/dL) | 6.1 | 0.85 |
| Proteinuria (g/dL) | 5.8 | 3.2 |
| Serum albumin (g/dL) | 2.5 | 0.39 |
| Urea (mg/dL) | 30.8 | 22.4 |
| Creatinine (mg/dL) | 0.8 | 0.3 |
| eGFR (ml/minute/1.73 m2) | 99.83 | 26.6 |
| Anti-PLA2R titre (RU/mL) | 81.6 | 148.5 |
| Clinical features | n (%) |
| Nephrotic syndrome | 34 (73.9) |
| Edema | 46 (100) |
| Foamy urine | 38 (82.6) |
| Microscopic hematuria | 12 (26.1) |
| Comorbidity | n (%) |
| Hypertension | 17 (37) |
| Hypothyroidism | 6 (13) |
| Obesity | 6 (13) |
| Diabetes mellitus | 3 (6.5) |
Of 18 (39.1%) patients had positive serum anti-PLA2R antibodies, and 17 (37%) of patients had negative serum anti-PLA2R antibodies. However, in 11 (23.9%) patients, serum anti-PLA2R antibodies were not available. On immunohistochemistry of the kidney biopsies, 27 (58.7%) of patients were tissue PLA2R positive, and 8 (17.4%) of patients were tissue NELL-1 positive. 11 (23.9%) of patients had no identifiable antigen on immunohistochemistry. Of 33 (71.1%) patients were in the high-risk category at the baseline, remaining 13 (28.3%) were in the low to moderate-risk category. Of 39 (84.8%) patients received initial non-immunosuppressive supportive care, while 7 (15.2%) received upfront immunosuppression. Among patients who received initial supportive care, 9 (23.08%) patients achieved remission, while 30 (76.92%) had no remission. In terms of risk category, there was no difference in the outcome with supportive care, as shown in Table 5.
| Risk | Remission | No remission | Total | P value |
| Low or moderate | 5 (38.5) | 8 (61.5) | 13 (100) | 0.129 |
| High | 4 (15.4) | 22 (84.6) | 26 (100) | |
| Total | 9 (23.08) | 30 (76.92) | 39 (100) |
Among patients who received immunosuppressive therapy, 18 (46.2%) received a modified Ponticelli regimen and 21 (53.8%) received other immunosuppressive regimens. Of 15 (83.3%) patients who received a modified Ponticelli regimen had baseline proteinuria greater than 6 g per day compared to 12 (57.1%) patients who received another immunosuppressive regimen; however, the difference was not statistically significant (P = 0.096, Fischer’s exact test). In our study baseline mean 24-hour urinary protein was higher in the modified Ponticelli group compared to other immunosuppressive groups; however, it was not statistically significant (8.77 ± 3.12 g/day vs 7.22 ± 2.69 g/day, P = 0.109). Patients who received modified Ponticelli had significantly higher serum creatinine at baseline compared with the other immunosuppressive group (0.99 ± 0.44 mg/dL vs 0.74 ± 0.14 mg/dL, P = 0.02), Table 6.
| Parameter | Modified Ponticelli regimen | Others | P value |
| Proteinuria (g/day) | 8.77 ± 3.12 | 7.22 ± 2.69 | 0.109 |
| Serum albumin (g/dL) | 2.07 ± 0.52 | 2.16 ± 0.54 | 0.6 |
| Serum creatinine (mg/dL) | 0.99 ± 0.44 | 0.74 ± 0.14 | 0.02 |
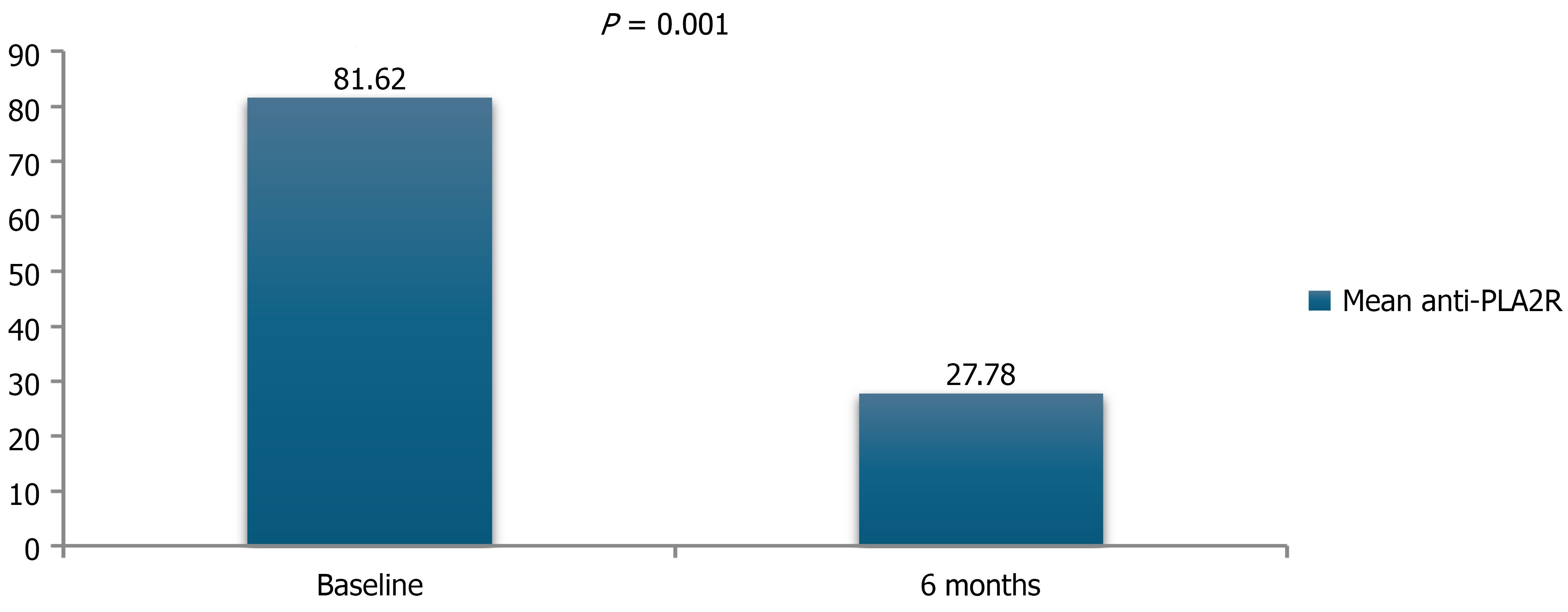
At 6 months, 13 (72.2%) patients who received a modified Ponticelli regimen had remission and 18 (85.7%) who received other immunosuppressive therapies achieved remission (P = 0.432). In patients with positive serum anti-PLA2R, there was a significant decline at 6 months with immunosuppression, Figure 1. At 12 months, 29 (82.9%) patients had remission on immunosuppressive therapy, while 6 (17.1%) patients had no remission. Our patients who received either modified Ponticelli or other immunosuppression had a significant improvement in 24-hour urinary proteinuria and serum albumin concentration at 12 months of therapy, Table 7.
| Therapy | Parameter | Baseline | At 12 months | P value |
| Modified Ponticelli regimen | Proteinuria (g/day) | 8.77 ± 3.12 | 1.75 ± 2.02 | < 0.0001 |
| Serum albumin (g/dL) | 2.07 ± 0.52 | 3.71 ± 0.57 | < 0.0001 | |
| Serum creatinine (mg/dL) | 0.99 ± 0.43 | 1.02 ± 0.52 | 0.845 | |
| Other | Proteinuria (g/day) | 7.22 ± 2.69 | 0.89 ± 1.24 | < 0.0001 |
| Serum albumin (g/dL) | 2.15 ± 0.55 | 3.94 ± 0.48 | < 0.0001 | |
| Serum creatinine (mg/dL) | 0.74 ± 0.14 | 0.78 ± 0.22 | 0.492 |
This study conducted in the Department of Nephrology at Sher-I-Kashmir Institute of Medical Sciences, Srinagar, over a period of 2 years. Primary MN is a specific pathological disorder of the glomerulus characterized by thickening of the capillary glomerular wall due to subepithelial immune complex deposition, where a secondary cause of glomerular lesion is excluded[2]. It is one of the most common causes of nephrotic syndrome in adult non-diabetic patients. Around 30% of cases of adult nephrotic syndrome are due to MN only surpassed by non-diabetic podocytopathy with focal segmental glomerulosclerosis lesions in African and Hispanic Americans[9]. There is a paucity of data on primary MN in our population, so this study was conducted.
We studied 46 patients with primary MN during the study period. The mean age at diagnosis of primary MN was 43.8 years. Our result was comparable to a study by Hemanth Kumar et al[11], who studied the profile and outcomes of primary MN in a tertiary hospital in North India; in their study mean age of participants was 43 years. In our study, 28 (60.9%) patients were females, and 18 (39.1%) were males. For unknown reasons, there is a slight male preponderance in primary MN[12]. However, our study did not demonstrate the same probability because of a small number of participants. This needs to be confirmed with larger studies of our population.
The most common clinical presentation was edema seen in all patients. Of 82.6% patients had frothuria (as reported by patients). Microscopic haematuria was observed in 26.1% of patients. Nephrotic syndrome was seen in 73.9% of patients. Ronco et al[9] reported microscopic haematuria in < 25% of patients and nephrotic syndrome in two-thirds of patients; and Couser[2] reported edema in 60%-75%, 80% of patients with primary MN present with nephrotic syndrome and haematuria in 50%-60% of patients during the course of primary MN.
In our study, serum anti-PLA2R antibody was available in 35 patients. Of the 11 patients in whom anti-PLA2R antibody was not available, 8 patients had NELL-1-related MN, and in 3 patients, anti-PLA2R antibody was not performed. Of the remaining 35 patients, 3 had qualitative anti-PLA2R antibodies done by immunofluorescence assay, which were positive, and 32 had quantitative anti-PLA2R antibodies performed by enzyme-linked immunosorbent assay. On immunohistochemistry, 27 (58.7%) patients were positive for PLA2R, and 8 (17.4%) were positive for NELL-1, 11 (23.9%) had no identifiable antigens on immunohistochemistry. McDonnell et al[13] reported 70% of primary MN associated with PLA2R, and Sethi et al[14] reported NELL-1 to be associated with approximately 10% of primary MN.
Of 33 (71.7%) patients were in the high/very high-risk category at baseline. Of 7 (15.2%) patients had received upfront immunosuppression along with supportive care. Of the remaining 39 patients, 7 (15.2%) received non-immunosuppressive antiproteinuric therapy only, while 32 (69.6%) patients received immunosuppressive therapy after a variable duration of non-immunosuppressive antiproteinuric therapy. Among 39 patients who received non-immunosuppressive antiproteinuric therapy, 9 (23.08%) patients had remission [3 (7.69%) had complete remission and 6 (15.39%) had partial remission] with supportive care only without immunosuppression, and 30 (76.92%) patients had no remission with supportive care. Dahan et al[15] reported 21.1% remission with non-immunosuppressive anti-proteinuric therapy at 1 year. There was no significant difference between the baseline risk category and the rate of remission with supportive care. The mean duration of supportive care in patients who didn’t receive any immunosuppressive therapy (n = 7) was 24.7 ± 8.17 months, while the mean duration of supportive care in patients who received immunosuppressive therapy after supportive care (n = 32) was 5.3 ± 4.02 months. Among patients who achieved remission with non-immunosuppressive antiproteinuric therapy 3 (7.69%) patients had relapse after 11.3 ± 2.08 months of non-immunosuppressive antiproteinuric therapy. All three patients had partial remission with non-immunosuppressive antiproteinuric therapy, which is a known risk factor for relapse[16].
Of the 46 patients, 39 (84.8%) received immunosuppressive therapy out of which 7 (17.9%) patients received upfront immunosuppression. At 6 months of therapy, compared from baseline there was significant improvement in proteinuria (7.93 ± 2.96 g/day to 1.96 ± 2.66 g/day, P < 0.001) and serum albumin (2.12 ± 0.53 mg/dL to 3.48 ± 0.83 mg/dL, P < 0.001). Of 31 (79.5%) had remission, 14 (35.9%) had complete remission and 17 (43.6%) had partial remission and 8 (20.5%) patients no remission. Our results were comparable with the study of Hemanth Kumar et al[11] who found an 87.5% remission rate with immunosuppressive therapy. Rao et al[16] found at the end of 6 months complete remission was seen in 25.9% and 62.9% had partial remission. The mean anti-PLA2R antibody level at baseline was 81.6 ± 148.5 RU/mL. After 6 months of immunosuppressive therapy serum anti-PLA2R antibody titres were significantly decreased to a mean 27.78 ± 82.81 RU/mL (P = 0.001), 4 of 8 patients who did not undergo remission at 6 months were resistant to initial immunosuppressive therapy and were excluded from analysis at 12 months.
At 12 months, 29 (82.9%) patients had remission, 18 (51.4%) had complete remission and 11 (31.5%) had partial remission, and 6 (17.1%) patients had no remission. Our results are comparable with the study of Hemanth Kumar et al[11], who found an 87.5% remission rate with immunosuppressive therapy. Fernández-Juárez et al[17] reported 79% overall remission at 12 months and 33% and 60% complete remission at 1 year and 2 years, respectively. There was a significant improvement in proteinuria and serum albumin concentration at 12 months with a decrease in mean 24-hour urinary protein from 7.93 ± 2.96 g/day to 1.29 ± 1.67 g/day and an increase in serum albumin from 2.12 ± 0.53 mg/dL to 3.84 ± 0.53 mg/dL.
Out of 39 patients who received immunosuppressive therapy, 18 (46.2%) received modified Ponticelli, and 21 (53.8%) received other immunosuppressive agents. In the other immunosuppressive group, 15 patients received prednisolone and tacrolimus, 3 patients received prednisolone, tacrolimus, and rituximab, 2 patients received rituximab alone, and 1 patient received tacrolimus alone. The rate of remission with the modified Ponticelli regimen was not significantly different compared to other immunosuppressive therapies at 6 months (72% vs 85%; P = 0.432) or at 12 months (68.8% vs 94.7%; P = 0.073). This non-significant higher remission rates with other immunosuppressive groups compared to the modified Ponticelli regimen could be because of higher baseline proteinuria and serum creatinine in the modified Ponticelli group (8.77 ± 3.12 g/day vs 7.22 ± 2.69 g/day, P = 0.109; 0.99 ± 0.44 mg/dL vs 0.74 ± 0.14 mg/dL, P = 0.02). We found that there is a significant decrease in proteinuria and an increase in serum albumin with both immunosuppressive groups at 6 months and 12 months. The limitations of our study are that it was a single-center study with a relatively small sample size, and the follow-up period was short. However, this study provides useful insight into the previously unstudied realm of MN in our patient population.
In conclusion, PLA2R-associated MN was the most common form of MN, followed by NELL-1 in our study population. The prevalence of primary MN was highest in the 5th and 6th decades of life with a slight female preponderance. Most of the patients were in the high-risk group. With immunosuppression, more than 80% of patients achieved complete remission. There was no significant difference in remission among different immunosuppressive agents.
| 1. | Moroni G, Ponticelli C. Secondary Membranous Nephropathy. A Narrative Review. Front Med (Lausanne). 2020;7:611317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 2. | Couser WG. Primary Membranous Nephropathy. Clin J Am Soc Nephrol. 2017;12:983-997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 548] [Cited by in RCA: 484] [Article Influence: 53.8] [Reference Citation Analysis (6)] |
| 3. | Johnson RJ, Feehally J, Floege J. Membranous nephropathy. Chapter 20. In: Comprehensive Clinical Nephrology. 5th ed. Philadelphia: Saunders, 2015: 239-251. |
| 4. | Beck LH Jr, Bonegio RG, Lambeau G, Beck DM, Powell DW, Cummins TD, Klein JB, Salant DJ. M-type phospholipase A2 receptor as target antigen in idiopathic membranous nephropathy. N Engl J Med. 2009;361:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2031] [Cited by in RCA: 1727] [Article Influence: 101.6] [Reference Citation Analysis (0)] |
| 5. | Tomas NM, Beck LH Jr, Meyer-Schwesinger C, Seitz-Polski B, Ma H, Zahner G, Dolla G, Hoxha E, Helmchen U, Dabert-Gay AS, Debayle D, Merchant M, Klein J, Salant DJ, Stahl RAK, Lambeau G. Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. N Engl J Med. 2014;371:2277-2287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 823] [Cited by in RCA: 694] [Article Influence: 57.8] [Reference Citation Analysis (0)] |
| 6. | Sethi S. New 'Antigens' in Membranous Nephropathy. J Am Soc Nephrol. 2021;32:268-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 184] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 7. | Maisonneuve P, Agodoa L, Gellert R, Stewart JH, Buccianti G, Lowenfels AB, Wolfe RA, Jones E, Disney AP, Briggs D, McCredie M, Boyle P. Distribution of primary renal diseases leading to end-stage renal failure in the United States, Europe, and Australia/New Zealand: results from an international comparative study. Am J Kidney Dis. 2000;35:157-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 202] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 8. | De Vriese AS, Glassock RJ, Nath KA, Sethi S, Fervenza FC. A Proposal for a Serology-Based Approach to Membranous Nephropathy. J Am Soc Nephrol. 2017;28:421-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 311] [Cited by in RCA: 259] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 9. | Ronco P, Beck L, Debiec H, Fervenza FC, Hou FF, Jha V, Sethi S, Tong A, Vivarelli M, Wetzels J. Membranous nephropathy. Nat Rev Dis Primers. 2021;7:69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 332] [Article Influence: 66.4] [Reference Citation Analysis (0)] |
| 10. | Turner NN, Lameire N, Goldsmith DJ, Winearls CG, Himmelfarb J, Remuzzi G. Oxford Textbook of Clinical Nephrology: Three-Volume Pack. 4th ed. Oxford: University Oxford Press, 2015. [DOI] [Full Text] |
| 11. | Hemanth Kumar MK, Sandhu J, Sandhu JS. Profile and primary treatment outcomes in membranous nephropathy. Saudi Med J. 2022;43:1051-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 12. | Hogan SL, Muller KE, Jennette JC, Falk RJ. A review of therapeutic studies of idiopathic membranous glomerulopathy. Am J Kidney Dis. 1995;25:862-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 120] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 13. | McDonnell T, Wu HHL, Sinha S, Chinnadurai R. The Role of PLA2R in Primary Membranous Nephropathy: Do We Still Need a Kidney Biopsy? Genes (Basel). 2023;14:1343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 14. | Sethi S, Beck LH Jr, Glassock RJ, Haas M, De Vriese AS, Caza TN, Hoxha E, Lambeau G, Tomas NM, Madden B, Debiec H, D'Agati VD, Alexander MP, Amer H, Appel GB, Barbour SJ, Caravaca-Fontan F, Cattran DC, Casal Moura M, D'Avila DO, Eick RG, Garovic VD, Greene EL, Herrera Hernandez LP, Jennette JC, Lieske JC, Markowitz GS, Nath KA, Nasr SH, Nast CC, Pani A, Praga M, Remuzzi G, Rennke HG, Ruggenenti P, Roccatello D, Soler MJ, Specks U, Stahl RAK, Singh RD, Theis JD, Velosa JA, Wetzels JFM, Winearls CG, Yandian F, Zand L, Ronco P, Fervenza FC. Mayo Clinic consensus report on membranous nephropathy: proposal for a novel classification. Kidney Int. 2023;104:1092-1102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 81] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 15. | Dahan K, Debiec H, Plaisier E, Cachanado M, Rousseau A, Wakselman L, Michel PA, Mihout F, Dussol B, Matignon M, Mousson C, Simon T, Ronco P; GEMRITUX Study Group. Rituximab for Severe Membranous Nephropathy: A 6-Month Trial with Extended Follow-Up. J Am Soc Nephrol. 2017;28:348-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 307] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 16. | Rao IR, Attur RP, Rangaswamy D, Shenoy S, Laxminarayana SLK, Nagaraju SP. Efficacy and safety of a modified- ‘modified Ponticelli’ regimen for treatment of primary membranous nephropathy. J Nephropathol. 2019;8:e25. [DOI] [Full Text] |
| 17. | Fernández-Juárez G, Rojas-Rivera J, Logt AV, Justino J, Sevillano A, Caravaca-Fontán F, Ávila A, Rabasco C, Cabello V, Varela A, Díez M, Martín-Reyes G, Diezhandino MG, Quintana LF, Agraz I, Gómez-Martino JR, Cao M, Rodríguez-Moreno A, Rivas B, Galeano C, Bonet J, Romera A, Shabaka A, Plaisier E, Espinosa M, Egido J, Segarra A, Lambeau G, Ronco P, Wetzels J, Praga M; STARMEN Investigators. The STARMEN trial indicates that alternating treatment with corticosteroids and cyclophosphamide is superior to sequential treatment with tacrolimus and rituximab in primary membranous nephropathy. Kidney Int. 2021;99:986-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 165] [Article Influence: 33.0] [Reference Citation Analysis (0)] |