Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.113212
Revised: November 10, 2025
Accepted: January 19, 2026
Published online: March 25, 2026
Processing time: 207 Days and 15 Hours
Veno-arterial extracorporeal membrane oxygenation (VA-ECMO) improves survival in patients with cardiogenic shock (CS), but data on chronic kidney disease (CKD) as a predictor of mortality in these patients is limited.
To assess the association between CKD and in-hospital mortality in CS patients undergoing VA-ECMO through a systematic review and meta-analysis.
We reviewed studies up to February 2024 that evaluated CKD’s impact on in-hospital mortality in CS patients treated with extracorporeal membrane oxygenation. Databases included PubMed, Google Scholar, EMBASE, and Scopus. A random-effects model was used to calculate the pooled unadjusted odds ratio, I2 statistics to determine heterogeneity, and leave-one-out sensitivity analysis to assess robustness, with P < 0.05 considered statistically significant.
Thirteen studies, involving 12185 CS patients treated with VA-ECMO (mean age 63, 68% males), were included. CKD was significantly associated with an increased in-hospital mortality (odds ratio = 1.89, 95% confidence interval: 1.4-2.55, P < 0.01). Leave-one-out analysis confirms robustness of the association between CKD and increased in-hospital mortality post-extracorporeal membrane oxygenation (I2 = 75%). Despite substantial heterogeneity, the association remains significant, indicating that CKD is a critical risk factor in these patients.
CKD is strongly associated with an increased in-hospital mortality in CS patients treated with VA-ECMO. Despite study heterogeneity, this association remains robust, highlighting CKD as a critical prognostic factor.
Core Tip: The impact of chronic kidney disease on in-hospital mortality of patients receiving veno-arterial extracorporeal membrane oxygenation for cardiogenic shock is assessed through our study. It emphasizes how chronic kidney disease, which is frequently present in critically ill patients as a pre-existing comorbidity, may be a significant prognostic factor influencing clinical outcomes. The study offers new insights by examining this relationship across diverse populations and clinical settings. The study provides new insights into patient selection, risk assessment, and future directions for veno-arterial extracorporeal membrane oxygenation care in this patient population.
- Citation: Vadiyala MR, Varghese J, Chilingarashvili G, Ahluwalia V, Sharma S, Raza SMM, Sura H, Pingili A, Desai R. Association of chronic kidney disease with in-hospital mortality of cardiogenic shock patients undergoing veno-arterial extracorporeal membrane oxygenation. World J Nephrol 2026; 15(1): 113212
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/113212.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.113212
Cardiogenic shock (CS) represents a complicated condition of severe clinical severity that develops when cardiac output becomes severely insufficient to maintain tissue perfusion. The condition exists as a range of circulatory disturbances that need progressively more advanced circulatory support measures[1,2]. The primary cause of CS stems from myocardial ischemia, which leads to mechanical complications. The development of severe right or left ventricular dysfunction, which leads to CS, can result from valvular disease and myocarditis as well as pulmonary embolism and stress-induced cardiomyopathy. The number of hospitalizations related to CS has increased, but nationwide mortality rates from this condition have decreased. The combination of early detection, along with invasive monitoring and prompt revascularization procedures and temporary mechanical circulatory support has led to this decline[3,4]. Veno-arterial extracorporeal membrane oxygenation (VA-ECMO) functions as a temporary mechanical circulatory support method that medical professionals increasingly use to treat refractory CS[5]. VA-ECMO functions as a vital tool for delivering cardiac and respiratory assistance to patients who need recovery support or additional medical interventions[6]. The selection of appropriate patients for VA-ECMO remains vital because this therapy requires substantial resources while carrying major risks[7]. Prior research has demonstrated that chronic kidney disease (CKD) along with older age, elevated lactate levels, high sequential organ failure assessment scores, and prolonged cardiopulmonary resuscitation predict poor for patients undergoing VA-ECMO treatment for CS[5,8-10]. Although CKD is generally recognized as a risk factor for unfavourable outcomes in critically ill patients, little is known about the outcomes of CKD patients with CS receiving VA-ECMO, particularly with regards to in-hospital mortality outcomes. Through this systematic review and meta-analysis, we aim to address this knowledge gap by assessing the prognostic significance of CKD in this high-risk population and its effect on in-hospital mortality.
Our meta-analysis was contacted utilizing Preferred Reporting Items for Systematic Reviews and Meta-Analyses. A comprehensive literature search was performed through PubMed and Scopus databases until February 2024 to identify studies exploring outcomes in patients with CS undergoing VA-ECMO. We used the following keywords, “cardiogenic shock”, “refractory cardiogenic shock”, “ECMO”, “veno-arterial ECMO”, “VA-ECMO”, “ECLS”, “extracorporeal life support”, “extracorporeal membrane oxygenation”, “chronic kidney disease”, “CKD”, “renal insufficiency”, “renal dysfunction”, and “kidney failure”. Boolean operators (AND, OR) were used for search strategy. Reference lists of included studies were screened to identify any eligible study.
Included studies fulfilling following criterias: (1) Studies involving only patients aged ≥ 18 years; (2) Studies reporting pre-existing CKD at hospital admission, defined as per kidney disease: Improving Global Outcomes (KDIGO) guidelines; and (3) Studies reporting in-hospital mortality. Excluding criteria were following: (1) Case reports or case series; (2) Reviews, editorials, or conference abstracts without full-text data; and (3) Non-English language publications without a full English translation.
Initial search results were screened by titles and abstracts by two independent reviewers to mark potentially eligible studies. Followed by, full-text articles assessment for eligibility. A second reviewer cross-verified the screening process ensuring adherence to protocol. Any discrepancies were resolved through discussion with a third reviewer.
Primary data from eligible studies were extracted into a standardized Excel spreadsheet by two independent investigators. Extracted data included baseline characteristics and primary outcomes. Discrepancies during extraction were resolved through consensus, and corresponding authors were contacted for clarification or missing data when necessary.
Pre-existing CKD associated odds ratio (OR) of in-hospital mortality among VA-ECMO populations was defined as a primary outcome. A random-effects model (DerSimonian-Laird method) was used to estimate pool effect. Forest plots were generated to visualize pooled estimates. All statistical analysis was conducted through R (version 4.2.2) with the “meta” and “metafor” packages. A two-sided P < 0.05 is accepted as statistically significant.
Using Newcastle-Ottawa Scale for the cohort studies approach used to evaluate methodological quality and risk of bias of included studies. Two reviewers independently assessed study across multiple variables: (1) Selection; (2) Comparability; and (3) Outcome. Scoring system classified studies as high quality, 4-6 as moderate quality, and ≤ 3 as low quality. Disagreements were resolved through discussion or consultation with a third reviewer. A leave-one-out sensitivity analysis was conducted to assess pooled results robustness.
Publication bias was assessed using a Doi plot, which graphs the log-odds ratio against the absolute Z-score (an inverse-precision metric), and the Luis Furuya-Kanamori (LFK) index was calculated to quantify asymmetry. An LFK index between -1 and +1 indicates symmetry (no publication bias), 1 to 2 suggests minor asymmetry, and ≥ 2 indicates major asymmetry, potentially reflecting small-study effects or selective reporting. The LFK index was interpreted cautiously, considering the limited number of studies.
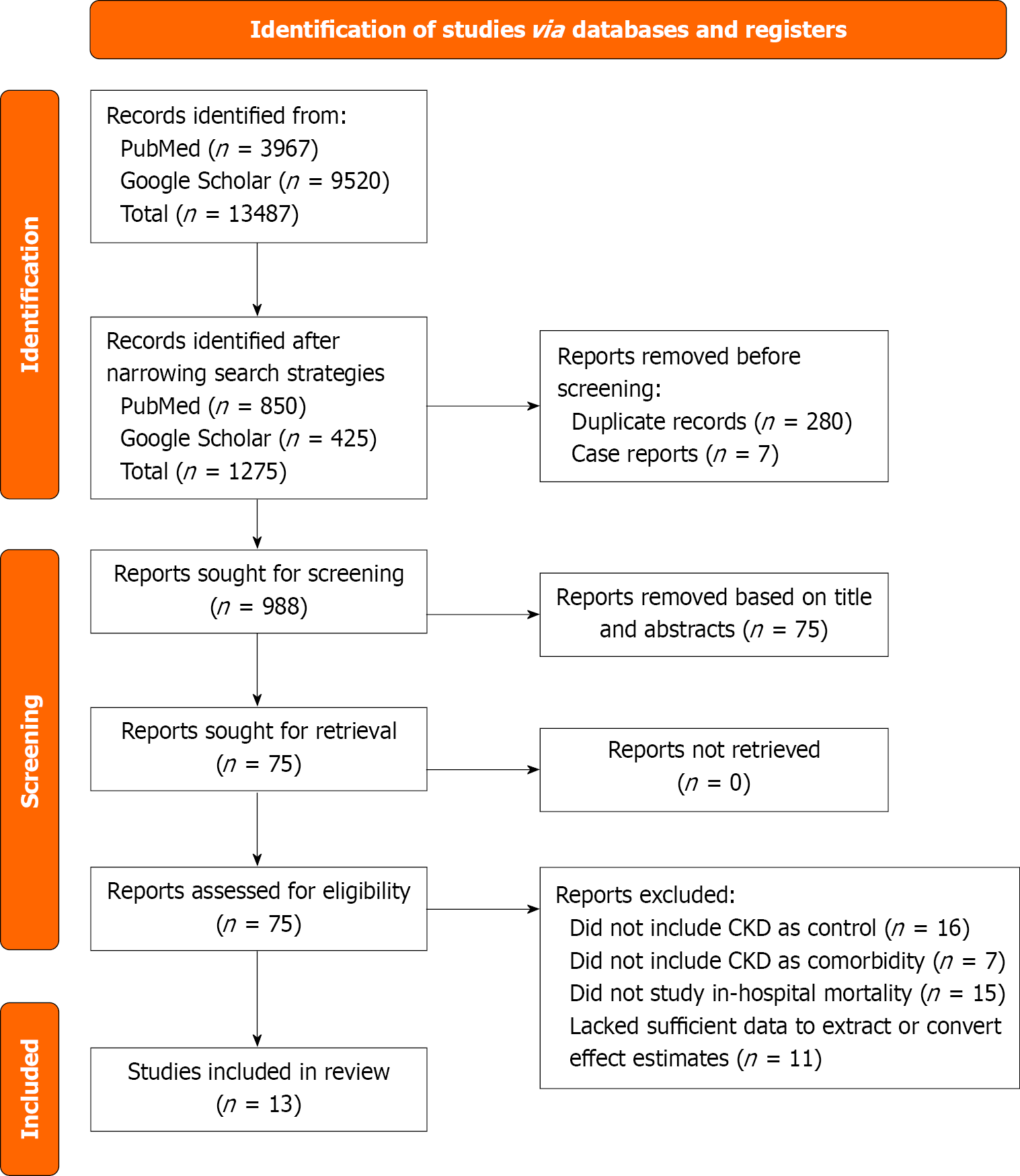
Our literature search across the chosen databases yielded an initial pool of 13487 articles. Following the removal of 280 duplicates, we proceeded to screen the abstracts of the remaining 988 records. This screening process identified 75 articles for full-text retrieval. Ultimately, we excluded 62 of these due to their meeting our inclusion criteria, primarily stemming from insufficient data for deriving effect estimates related to CKD, an inappropriate study population, or the absence of in-hospital mortality as an assessed outcome. Consequently, 13 cohort studies were deemed eligible for inclusion in our systematic review and meta-analysis (Figure 1).
All the 13 studies we included were published by February 2024 and together involved 12185 patients with CS who were treated with VA-ECMO. Average age of the patients was about 63 years, and 68% of them were male. Detailed breakdown of the patient demographics and their baseline health characteristics are shown in Table 1[11-23].
| Ref. | Setting | Sample size | Age (years, mean ± SD) | BMI (mean ± SD) | Male (n) | Chronic kidney disease (non-survivor/ | Hypertension (non-survivor/ | Diabetes mellitus (non-survivor/ | Mortality (%) |
| Jeong et al[11], 2024 | Multicenter | 322 | 65.2 ± 12.1 | 23.5 ± 3.2 | 241 | 21/5 | 109/64 | 137/60 | 52.1 |
| Zhang et al[12], 2022 | Single-center | 510 | 66.0 | - | 337 | 72/79 | 112/137 | 86/83 | 44.5 |
| Laimoud and Alanazi[13], 2020 | Single-center | 106 | 64.7 ± 7.0 | 26.5 ± 7.0 | 74 | 19/2 | 20/14 | 11/9 | 56.6 |
| Schmidt et al[14], 2015 | Single-center | 3846 | 40.2 ± 14.4 | - | 2548 | 87/25 | - | - | 58.4 |
| Peigh et al[15], 2015 | Single-center | 73 | 48.0 ± 14.0 | - | 47 | 14/6 | - | 9/12 | 35.6 |
| Aso et al[16], 2016 | Multicenter | 4658 | 64.8 ± 13.7 | - | 3399 | 389/130 | - | - | 73.6 |
| Wang et al[17], 2022 | Single-center | 574 | - | - | 394 | 107/71 | 205/183 | 110/78 | - |
| Rubino et al[18], 2018 | Single-center | 101 | 57.1 ± 15.8 | - | 64 | 5/0 | 25/9 | 12/4 | 66.3 |
| Rastan et al[19], 2010 | Single-center | 517 | 63.5 ± 11.2 | - | 370 | 69/12 | 274/89 | 143/24 | 75.2 |
| Papadopoulos et al[20], 2015 | Single-center | 360 | 62.0 ± 17.0 | - | 274 | 32/8 | 173/54 | 122/29 | 70.0 |
| Vigneshwar et al[21], 2020 | Multicenter | 789 | 54.8 ± 9.8 | 29.8 | 535 | 117/72 | 145/83 | 190/111 | 57.9 |
| Salna et al[22], 2020 | Single-center | 112 | 69.0 | 30.1 | 66 | 33/6 | 52/18 | 31/8 | 72.3 |
| Kuroki et al[23], 2016 | Single-center | 217 | 58.9 ± 15.0 | 24.0 ± 4.9 | 180 | 116/28 | - | - | 71.4 |
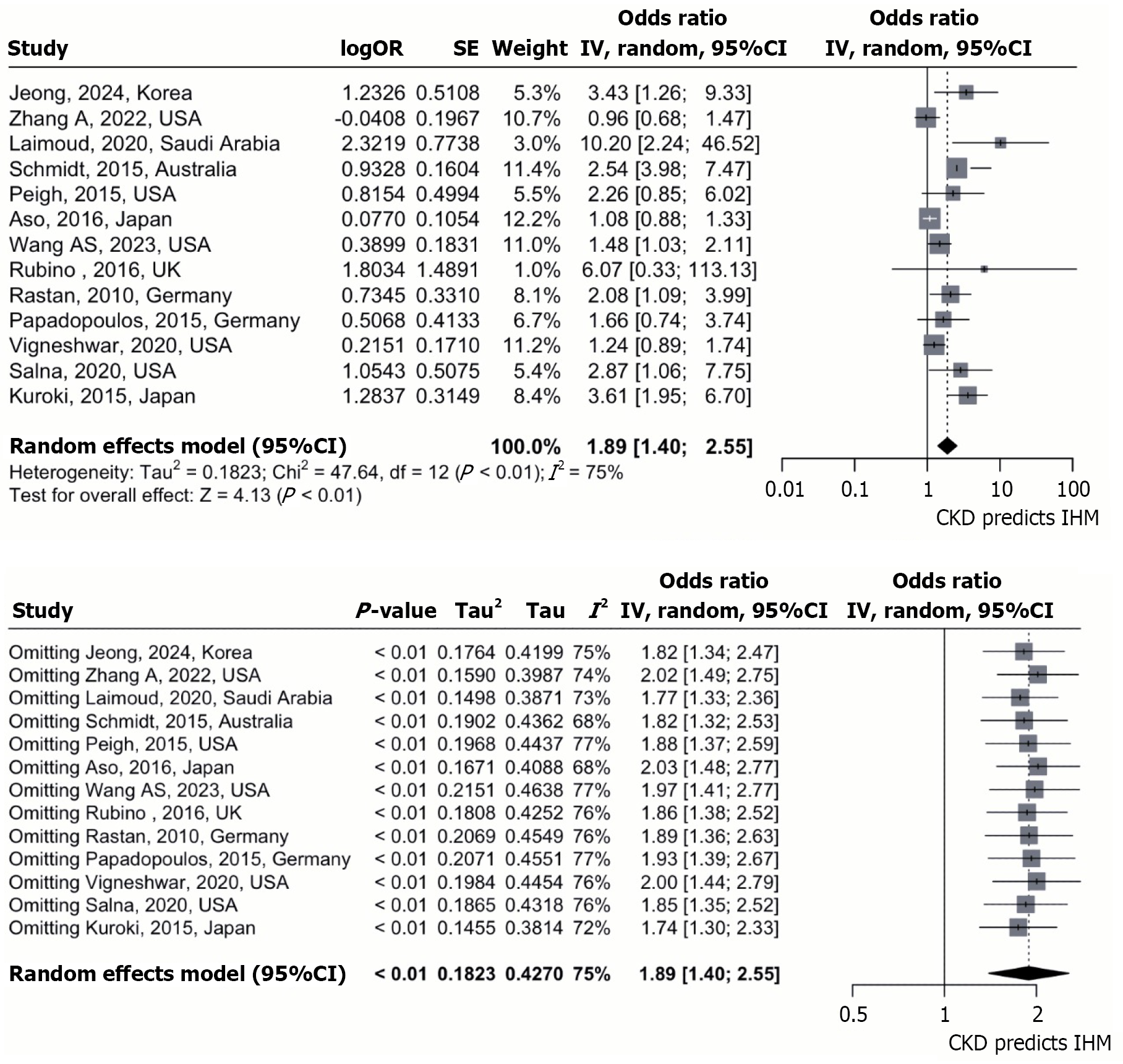
Our main analysis utilized a random-effects model, we observed a strong and statistically significant link between prior CKD and increased risk of death during hospitalization. The forest plot depicts this clearly (Figure 2, upper panel), with CKD patients facing 89% greater odds of in-hospital mortality than those without CKD [pooled OR = 1.89, 95% confidence interval (CI): 1.40-2.55; P < 0.01].
We observed considerable statistical heterogeneity across the included studies (I2 = 75%; Cochran’s Q P < 0.01), indicating notable variation in effect sizes. To evaluate the stability of this primary result, we conducted a leave-one-out sensitivity analysis (Figure 2, lower panel). The overall estimate did not significantly change when each study was excluded one after the other; the pooled unadjusted OR consistently retained statistical significance and validated CKD as a risk factor (range: 1.74-2.03). This result highlights the strength of our conclusions and shows minimal dependence on any individual study.
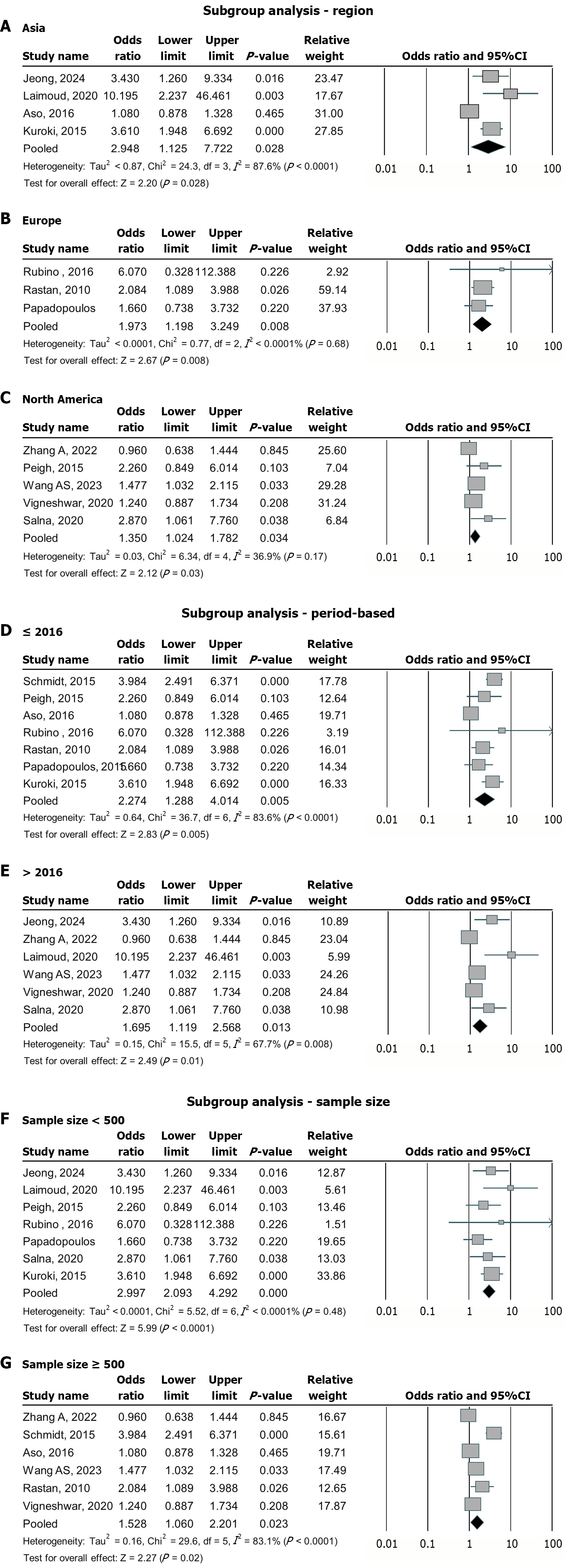
Across all subgroups, CKD was associated with higher in-hospital mortality among patients with CS supported with VA-ECMO. (Figure 3). For region, the association was significant in Asia (4 studies; pooled OR = 2.95, 95%CI: 1.13-7.72; I2 = 87.6%; Z = 2.20, P = 0.028), Europe (3 studies; OR = 1.97, 95%CI: 1.20-3.25; I2 approximately 0%; Z = 2.67, P = 0.008), and North America (5 studies; OR = 1.35, 95%CI: 1.02-1.78; I2 = 36.9%; Z = 2.12, P = 0.03) (Figure 3). Effect sizes were largest in Asian cohorts, with considerable heterogeneity, and smallest in North American cohorts with low-moderate heterogeneity.
Based on the study period, studies published ≤ 2016 showed a stronger association (7 studies; OR = 2.27, 95%CI: 1.29-4.01; I2 = 83.6%; Z = 2.83, P = 0.005) than studies published after 2016 (6 studies; OR = 1.69, 95%CI: 1.12-2.57; I2 = 67.7%; Z = 2.49, P = 0.01), suggesting attenuation in more recent literature, though between-study heterogeneity remained substantial in both strata. In terms of sample size, smaller studies (< 500 patients) reported the largest effect (7 studies; OR = 3.00, 95%CI: 2.09-4.29; I2 approximately 0%; Z = 5.99, P < 0.0001), whereas larger studies (≥ 500 patients) showed a more modest but still significant association (6 studies; OR = 1.53, 95%CI: 1.06-2.20; I2 = 83.1%; Z = 2.27, P = 0.02).
Assessment of methodological quality via the Newcastle-Ottawa Scale revealed that the 13 incorporated studies were of high caliber, with a correspondingly low risk of bias. Every study achieved a score of at least 7 out of 9 possible points, and ten attained the maximum of 9/9. These quality ratings are outlined in Table 2.
| Ref. | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Overall |
| Jeong et al[11], 2024 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Laimoud and Alanazi[13], 2020 | √ | √ | √ | √ | - | √ | √ | √ | 7/9 |
| Kuroki et al[23], 2016 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Aso et al[16], 2016 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Rastan et al[19], 2010 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Wang et al[17], 2022 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Salna et al[22], 2020 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Zhang et al[12], 2022 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Peigh et al[15], 2015 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Schmidt et al[14], 2015 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Vigneshwar et al[21], 2020 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Papadopoulos et al[20], 2015 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
| Rubino et al[18], 2018 | √ | √ | √ | √ | √ | √ | √ | √ | 9/9 |
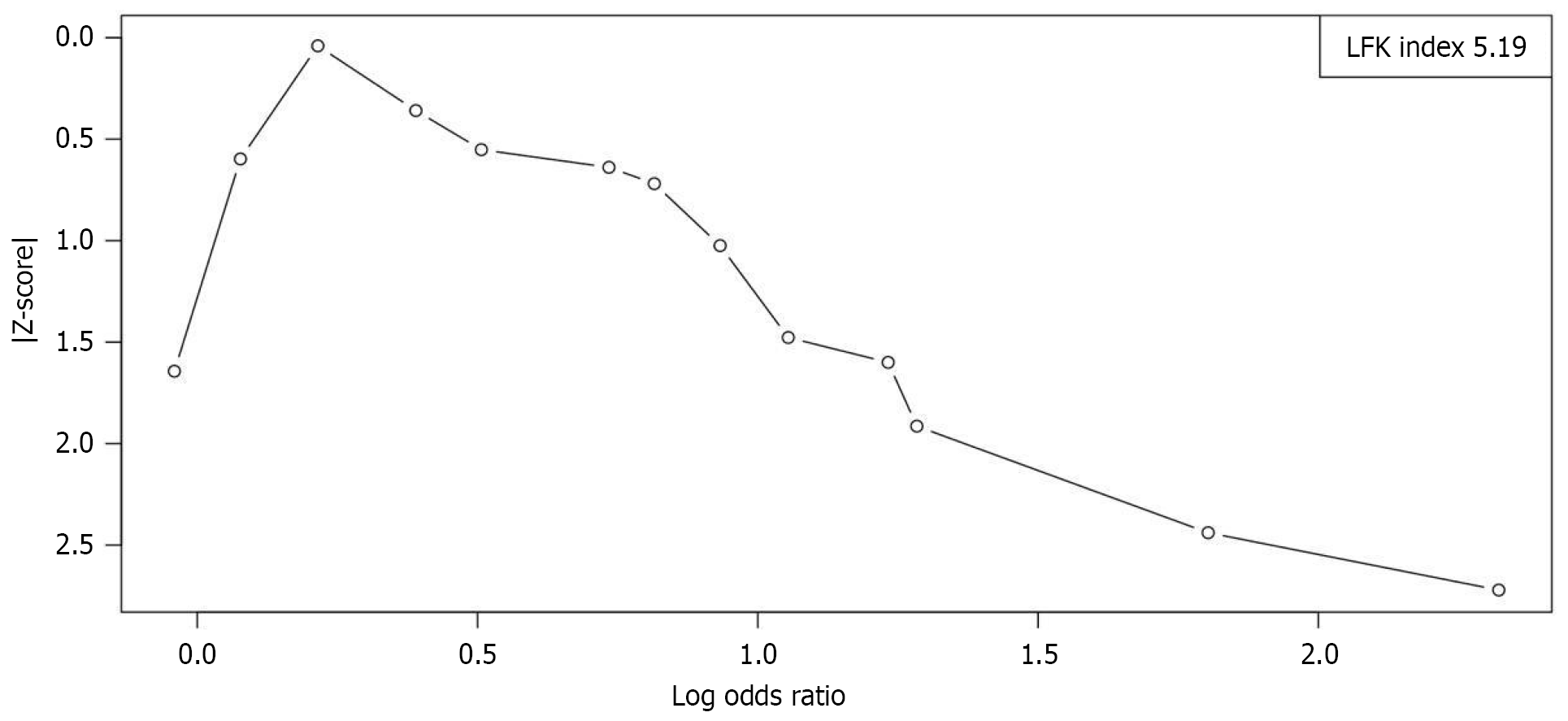
We further examined the potential for publication bias through the LFK index (Figure 3) and Doi plot (Figure 4). The plot displayed pronounced right-sided asymmetry, corroborated by an LFK index value of 5.19. Given that this exceeds 2, it signals substantial asymmetry and raises concerns about the underrepresentation of smaller studies reporting null or nonsignificant results in our synthesis.
Our systematic review and meta-analysis, consisting of thirteen observational cohort studies involving 12185 patients, provides compelling evidence on the impact of CKD on in-hospital mortality in patients with CS undergoing VA-ECMO. In summary, the pooled analysis showed that CKD is significantly associated with in-hospital mortality when compared to patients without CKD (pooled OR = 1.89; 95%CI: 1.40-2.55; P < 0.001). After leave-one-out sensitivity analyses, the result remained robust. Hence, CKD should be considered an important prognostic factor in this high-risk population. To the best of our knowledge, our study is the most extensive meta-analysis conducted to date assessing the association of CKD with outcomes in CS patients supported by VA-ECMO.
Previous studies have shown that there was increased mortality in critically ill patients with pre-existing comorbid CKD compared to patients without CKD[24]. FRENSHOCK study is a prospective, observational, multicenter survey involving 772 patients in France admitted for CS. According to the study, patients with CKD had a higher all-cause mortality rate at one month (36.6% vs 23.2%; hazard ratio: 1.39, 95%CI: 1.01-1.9; P = 0.04) and at one year (62.8% vs 40.5%, hazard ratio: 1.39, 95%CI: 1.09-1.77; P < 0.01)[25]. In a different study that included a smaller cohort (n = 248) of patients in CS, specifically in acute myocardial infarction, they highlighted that CKD as a comorbidity is a strong, independent predictor of in-hospital and long-term mortality[26]. Kwon et al[27] in their study examined the association between preexisting severity of CKD and VA-ECMO outcomes. They demonstrated that advanced stages of CKD are independently associated with a greater risk of in-hospital mortality and perioperative complications. They further stratified patients undergoing VA-ECMO based on operative indications, and it was found that CKD 3-5 was associated with the highest risk-adjusted rate of mortality, particularly in patients with CS[27]. In a retrospective study by Lu et al[7] which involved 1008 patients with CKD and 2016 patients without CKD, after propensity score matching, found that CKD was associated with higher in-hospital mortality rates (69.5% vs 62.2%; adjusted OR: 1.41; 95%CI: 1.15-1.72) in patients who received extracorporeal membrane oxygenation (ECMO) treatment.
Our findings support and expand upon existing literature indicating that renal dysfunction worsens the outcomes in critically ill populations and particularly those undergoing VA-ECMO. An effort has been made to extend these observations to the CS and ECMO population, a group characterized by extreme hemodynamic instability and high baseline mortality. Another study by Laimoud and Alanazi[13] which is included in our meta-analysis, found chronic renal failure as a strong independent predictor of in-hospital mortality (OR: 10.2, P = 0.001). They further noted that the presence of CKD and the development of acute kidney injury, along with the need for renal replacement therapy, were significantly prevalent in non-survivors. It should also be highlighted that renal dysfunction has consistently emerged as a key component in ECMO mortality risk prediction tools such as the survival after VA-ECMO score which includes renal failure, and the risk estimation model for ECMO after coronary artery bypass grafting (CABG) score which includes serum creatinine as one of the variables to predict mortality post-CABG and VA-ECMO[14,17]. Jeong et al[11] in their PRECISE risk model for ischemic CS patients receiving VA-ECMO, identified CKD as one of 15 independent predictors of in-hospital mortality, reinforcing the prognostic significance of CKD in this patient population. Aso et al[16] found a strong correlation between the use of renal replacement therapy and in-hospital mortality, which further supports the prognostic weight of renal burden in these patients, although they did not find a significant correlation with renal impairment.
The degree of heterogeneity in our meta-analysis (I2 = 75%) reflects clinical and methodological diversity among the included studies. However, the strength of the association between CKD and in-hospital mortality varied across all strata, with the strongest correlation in Asian cohorts, intermediate in European cohorts, and least in North American cohorts. This suggests that regional practice differences and baseline population characteristics may influence outcomes. The attenuation of effect observed in studies published after 2016 is likely due to advancements in patient selection, peri-ECMO management, and ECMO technology over time. Comparably, larger studies provided more modest but still significant associations, while smaller studies reported larger effect sizes, which is consistent with small-study effects and possible publication bias. Heterogeneity was also caused by variations in the definition and staging of CKD. While nine studies used broadly used terms such as “pre-existing CKD” or “chronic renal insufficiency”, others used specific criteria. For example, Schmidt et al[14] defined CKD as kidney damage or glomerular filtration rate < 60 mL/minute/1.73 m2 for ≥ 3 months, Peigh et al[15] used a risk, injury, failure, loss, and end-stage kidney disease score of injury or worse, Rastan et al[19] applied creatinine > 1.8 mg/dL, and Kuroki et al[23] classified patients with estimated glomerular filtration rate < 60 mL/minute/1.73 m2 as having CKD. These discrepancies likely affected patient selection and risk assessment, signifying the need for future ECMO research to use standardized, KDIGO-based renal definitions[28]. In addition to patient demographics and differences in CKD severity and definitions, differences in ECMO protocols across institutions are also the probable contributing factors to this heterogeneity. The Doi plot’s right-sided asymmetry (LFK index = 5.19), which suggests that smaller studies with larger effect sizes might be overrepresented, could inflate the pooled odds ratio. Nevertheless, the consistent direction and statistical significance across sensitivity and subgroup analyses, especially the leave-one-out approach, mitigate this concern and reinforce the robustness and clinical applicability of our findings.
CKD seems to have a consistent prognostic impact across the various CS etiologies treated with VA-ECMO. However, Alba et al[10] large meta-analysis assessing the mortality following VA-ECMO across CS etiologies revealed markedly disparate mortality estimates for each etiology (P < 0.001). Mortality rate vary significantly depending on the underlying cause, with approximately 35% in post-transplant patients, 40% for myocarditis, 53% for heart failure to over 75% in patients with out-of-hospital cardiac arrest. This variability underscores the importance of considering shock etiology when interpreting mortality risk and suggests that the additive impact of CKD may differ by primary diagnosis. To further refine patient selection and management to improve outcomes, additional studies are needed to evaluate the impact on outcomes by specific CS etiology.
Our study has several limitations. Firstly, the only studies we included were retrospective observational studies, which are vulnerable to confounding and biases in measurement or selection. Residual confounding by factors such as age, comorbidities and illness severity cannot be ruled out since we relied on the different methods and adjustments of individual studies rather than conducting a unified multivariate analysis. Therefore, our pooled unadjusted odds ratio shows an overall association between CKD and in-hospital mortality, rather than a definitive causal relationship. Secondly, there is significant heterogeneity noted. This is likely due to studies varying in their definitions and staging of CKD, CS etiologies, and ECMO protocols, including weaning strategies, anticoagulation protocols. This leads to limited direct comparability among studies and limits the precision of the pooled estimates. Thirdly, the potential for publication bias suggests that smaller studies and those with neutral or negative results might be underrepresented. Finally, the generalizability of our findings may be limited by institutional and geographic variations in ECMO protocols, patient populations, and clinical practice. Thus, more research is required to validate CKD as an independent risk factor in this patient population.
Given that CKD is associated with inflammation, metabolic derangement, fluid overload, poor tolerance to critical illness, and a diminished ability to respond to physiological stressors - all of which exacerbate ECMO outcomes - these findings are clinically plausible. Several pre-existing chronic conditions, including diabetes mellitus, hypertension, and anemia, are also frequently present in patients with CKD, further complicating management and worsening prognosis. When CKD coexists with CS, impaired natriuresis promotes fluid overload, making hemodynamic management more difficult and increasing mortality risk[29]. VA-ECMO itself can predispose the kidneys to further insult. Exposure of blood to artificial surfaces may trigger hemolysis and hemoglobinuria-induced tubular damage, creating a self-perpetuating cycle of renal and systemic dysfunction. Moreover, VA-ECMO is frequently complicated by bleeding and thromboembolic events[30]. CKD may also amplify these risks: Uremic platelet dysfunction and vascular fragility increase bleeding risk, whereas endothelial activation and a prothrombotic milieu increase the risk of thromboembolic events.
Our study findings have significant implications for clinical practice and future research. CKD should be viewed as a prognostic indicator in patients undergoing VA-ECMO for CS rather than merely a comorbidity. These patients require early risk assessment, optimization of kidney function, and individualized decision-making. From a clinical standpoint, CKD should not automatically preclude VA-ECMO candidacy but should inform pre-implant risk counseling, anticipated duration of support, and timing of renal replacement therapy. Since these patients with pre-existing CKD undergoing VA-ECMO require close monitoring and have a high risk of in-hospital mortality, a nephrology consultation prior to starting VA-ECMO treatment may be beneficial to guide individualized management. Fluid balance should be closely monitored and titrated, as both overload and aggressive depletion can worsen outcomes by exacerbating pulmonary edema or reducing circuit flow. In this setting, unfractionated heparin remains the preferred anticoagulant, but altered clearance and platelet dysfunction necessitate cautious dosing and frequent anti-Xa or assertive community treatment monitoring to balance bleeding and thrombosis[31]. Renal replacement therapy should be considered early for progressive azotemia, metabolic acidosis, or refractory volume overload, with modality selection based on hemodynamic stability. Proactive identification and mitigation of these interrelated risks are crucial for enhancing survival in this high-risk population.
Beyond its recognition as a prognostic marker, CKD could be pragmatically incorporated into established and emerging ECMO risk-stratification tools. This could help physicians assess who is a good fit for VA-ECMO and further discuss goals and prognosis with patients and their families. Existing models, such as the survival after VA-ECMO and risk estimation model for ECMO after CABG scores, already include renal dysfunction indicators (renal failure or serum creatinine levels), whereas newer models, like the PRECISE score, explicitly list CKD as an independent variable. Our findings reinforce the value of formalizing CKD within these models, either as a categorical variable (presence vs absence or stratified by KDIGO stage) or as a continuous variable that interacts with other renal parameters (e.g., baseline creatinine or estimated glomerular filtration rate). Future studies should investigate the mechanisms by which CKD influences the results of VA-ECMO in patients with CS. It also helps evaluate individualized ECMO strategies and assess the function of adjunctive renal therapies in this high-risk subgroup. It is also essential to compare the effects of CKD severity (e.g., stages 3-5) on outcomes, given the heterogeneity among the included studies.
In conclusion, our study adds to the evidence that pre-existing CKD is significantly associated with increased in-hospital mortality in patients with CS undergoing VA-ECMO support. By demonstrating CKD as a prognostic factor for in-hospital mortality, our findings have implications for risk assessment, patient selection, and developing ECMO strategies and decision-making. Further studies, particularly large multicenter prospective studies or randomized controlled trials are needed to validate our findings across diverse patient populations and to establish clinical thresholds. Understanding the mechanisms of associations will potentially enhance outcomes. Thus, identifying CKD as a prognostic factor for patients with CS undergoing VA-ECMO could help in risk assessment and optimizing care.
| 1. | Reynolds HR, Hochman JS. Cardiogenic shock: current concepts and improving outcomes. Circulation. 2008;117:686-697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 529] [Cited by in RCA: 557] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 2. | Angelini F, Bocchino PP, Frea S, De Ferrari GM. Does end-organ dysfunction precede or follow cardiogenic shock in acute decompensated heart failure? The two-faced Janus. Letter regarding the article 'Epidemiology, pathophysiology and contemporary management of cardiogenic shock - a position statement from the Heart Failure Association (HFA) of the European Society of Cardiology (ESC)'. Eur J Heart Fail. 2021;23:197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Osman M, Syed M, Patibandla S, Sulaiman S, Kheiri B, Shah MK, Bianco C, Balla S, Patel B. Fifteen-Year Trends in Incidence of Cardiogenic Shock Hospitalization and In-Hospital Mortality in the United States. J Am Heart Assoc. 2021;10:e021061. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 154] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 4. | Vahdatpour C, Collins D, Goldberg S. Cardiogenic Shock. J Am Heart Assoc. 2019;8:e011991. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 351] [Cited by in RCA: 297] [Article Influence: 42.4] [Reference Citation Analysis (0)] |
| 5. | Syed M, Khan MZ, Osman M, Sulaiman S, Agrawal P, Raina S, Patel B, Bianco C, Balla S, Daggubati R. Sixteen-Year National Trends in Use and Outcomes of VA-ECMO in Cardiogenic Shock. Cardiovasc Revasc Med. 2022;44:1-7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 6. | Eckman PM, Katz JN, El Banayosy A, Bohula EA, Sun B, van Diepen S. Veno-Arterial Extracorporeal Membrane Oxygenation for Cardiogenic Shock: An Introduction for the Busy Clinician. Circulation. 2019;140:2019-2037. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 103] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 7. | Lu YA, Chen SW, Lee CC, Wu VC, Fan PC, Kuo G, Chen JJ, Chu PH, Chang SH, Chang CH. Mid-term survival of patients with chronic kidney disease after extracorporeal membrane oxygenation. Interact Cardiovasc Thorac Surg. 2020;31:595-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Muller G, Flecher E, Lebreton G, Luyt CE, Trouillet JL, Bréchot N, Schmidt M, Mastroianni C, Chastre J, Leprince P, Anselmi A, Combes A. The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock. Intensive Care Med. 2016;42:370-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 337] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 9. | Xie A, Phan K, Tsai YC, Yan TD, Forrest P. Venoarterial extracorporeal membrane oxygenation for cardiogenic shock and cardiac arrest: a meta-analysis. J Cardiothorac Vasc Anesth. 2015;29:637-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 10. | Alba AC, Foroutan F, Buchan TA, Alvarez J, Kinsella A, Clark K, Zhu A, Lau K, McGuinty C, Aleksova N, Francis T, Stanimirovic A, Vishram-Nielsen J, Malik A, Ross HJ, Fan E, Rac VE, Rao V, Billia F. Mortality in patients with cardiogenic shock supported with VA ECMO: A systematic review and meta-analysis evaluating the impact of etiology on 29,289 patients. J Heart Lung Transplant. 2021;40:260-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 11. | Jeong JH, Kook H, Lee SH, Joo HJ, Park JH, Hong SJ, Kim MN, Park SM, Jung JS, Yang JH, Gwon HC, Ahn CM, Jang WJ, Kim HJ, Bae JW, Kwon SU, Lee WS, Jeong JO, Park SD, Lim SH, Lee J, Lee J, Yu CW. Prediction of In-Hospital Mortality for Ischemic Cardiogenic Shock Requiring Venoarterial Extracorporeal Membrane Oxygenation. J Am Heart Assoc. 2024;13:e032701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 12. | Zhang A, Kurlansky P, Ning Y, Wang A, Kaku Y, Fried J, Takeda K. Outcomes following successful decannulation from extracorporeal life support for cardiogenic shock. J Thorac Cardiovasc Surg. 2024;167:1033-1046.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 13. | Laimoud M, Alanazi M. The Validity of SOFA Score to Predict Mortality in Adult Patients with Cardiogenic Shock on Venoarterial Extracorporeal Membrane Oxygenation. Crit Care Res Pract. 2020;2020:3129864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 14. | Schmidt M, Burrell A, Roberts L, Bailey M, Sheldrake J, Rycus PT, Hodgson C, Scheinkestel C, Cooper DJ, Thiagarajan RR, Brodie D, Pellegrino V, Pilcher D. Predicting survival after ECMO for refractory cardiogenic shock: the survival after veno-arterial-ECMO (SAVE)-score. Eur Heart J. 2015;36:2246-2256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 749] [Cited by in RCA: 672] [Article Influence: 61.1] [Reference Citation Analysis (0)] |
| 15. | Peigh G, Cavarocchi N, Keith SW, Hirose H. Simple new risk score model for adult cardiac extracorporeal membrane oxygenation: simple cardiac ECMO score. J Surg Res. 2015;198:273-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 16. | Aso S, Matsui H, Fushimi K, Yasunaga H. In-hospital mortality and successful weaning from venoarterial extracorporeal membrane oxygenation: analysis of 5,263 patients using a national inpatient database in Japan. Crit Care. 2016;20:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 90] [Cited by in RCA: 124] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 17. | Wang AS, Nemeth S, Kurlansky P, Brodie D, Takayama H, Naka Y, Kaku Y, Fried J, Nir U, Takeda K. Sex differences in patients with cardiogenic shock requiring extracorporeal membrane oxygenation. J Thorac Cardiovasc Surg. 2022;164:960-969.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 18. | Rubino A, Costanzo D, Stanszus D, Valchanov K, Jenkins D, Sertic F, Fowles JA, Vuylsteke A. Central Veno-Arterial Extracorporeal Membrane Oxygenation (C-VA-ECMO) After Cardiothoracic Surgery: A Single-Center Experience. J Cardiothorac Vasc Anesth. 2018;32:1169-1174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 19. | Rastan AJ, Dege A, Mohr M, Doll N, Falk V, Walther T, Mohr FW. Early and late outcomes of 517 consecutive adult patients treated with extracorporeal membrane oxygenation for refractory postcardiotomy cardiogenic shock. J Thorac Cardiovasc Surg. 2010;139:302-311, 311.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 386] [Cited by in RCA: 452] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 20. | Papadopoulos N, Marinos S, El-Sayed Ahmad A, Keller H, Meybohm P, Zacharowski K, Moritz A, Zierer A. Risk factors associated with adverse outcome following extracorporeal life support: analysis from 360 consecutive patients. Perfusion. 2015;30:284-290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 21. | Vigneshwar NG, Kohtz PD, Lucas MT, Bronsert M, J Weyant M, F Masood M, Itoh A, Rove JY, Reece TB, Cleveland JC, Pal JD, Fullerton DA, Aftab M. Clinical predictors of in-hospital mortality in venoarterial extracorporeal membrane oxygenation. J Card Surg. 2020;35:2512-2521. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Salna M, Sanchez J, Fried J, Masoumi A, Witer L, Kurlansky P, Agerstrand CL, Brodie D, Garan AR, Takeda K. Ten-year outcomes of extracorporeal life support for in-hospital cardiac arrest at a tertiary center. J Artif Organs. 2020;23:321-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 23. | Kuroki N, Abe D, Iwama T, Sugiyama K, Akashi A, Hamabe Y, Aonuma K, Sato A. Prognostic effect of estimated glomerular filtration rate in patients with cardiogenic shock or cardiac arrest undergoing percutaneous veno-arterial extracorporeal membrane oxygenation. J Cardiol. 2016;68:439-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Abdalrahim MS, Khalil AA, Alramly M, Alshlool KN, Abed MA, Moser DK. Pre-existing chronic kidney disease and acute kidney injury among critically ill patients. Heart Lung. 2020;49:626-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 25. | Delmas C, Roubille F, Lamblin N, Bonello L, Leurent G, Levy B, Elbaz M, Danchin N, Champion S, Lim P, Schneider F, Cariou A, Khachab H, Bourenne J, Seronde MF, Schurtz G, Harbaoui B, Vanzetto G, Quentin C, Delabranche X, Aissaoui N, Combaret N, Manzo-Silberman S, Tomasevic D, Marchandot B, Lattuca B, Henry P, Gerbaud E, Bonnefoy E, Puymirat E. Baseline characteristics, management, and predictors of early mortality in cardiogenic shock: insights from the FRENSHOCK registry. ESC Heart Fail. 2022;9:408-419. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 26. | Peters AE, Smith LA, Corotto P, Bilchick KC, Mazimba S. Baseline Chronic Kidney Disease Predicts In-Hospital and Long-Term Mortality in Cardiogenic Shock. J Card Fail. 2017;23:S52-S53. |
| 27. | Kwon OJ, Aguayo E, Tabibian K, Balian J, Chaturvedi A, Yalzadeh D, Hadaya J, Sanaiha Y, Benharash P. National Outcomes of Venoarterial Extracorporeal Life Support in Patients with Chronic Kidney Disease. Surg Open Sci. 2025;26:87-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 28. | Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int. 2024;105:S117-S314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3017] [Cited by in RCA: 2532] [Article Influence: 1266.0] [Reference Citation Analysis (2)] |
| 29. | Arbel Y, Mass R, Ziv-Baran T, Khoury S, Margolis G, Sadeh B, Flint N, Ben-Shoshan J, Finn T, Keren G, Shacham Y. Prognostic implications of fluid balance in ST elevation myocardial infarction complicated by cardiogenic shock. Eur Heart J Acute Cardiovasc Care. 2017;6:462-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 30. | Askenazi DJ, Selewski DT, Paden ML, Cooper DS, Bridges BC, Zappitelli M, Fleming GM. Renal replacement therapy in critically ill patients receiving extracorporeal membrane oxygenation. Clin J Am Soc Nephrol. 2012;7:1328-1336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 168] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 31. | Vajter J, Volod O. Anticoagulation Management During ECMO: Narrative Review. JHLT Open. 2025;8:100216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |