Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.111612
Revised: July 28, 2025
Accepted: January 12, 2026
Published online: March 25, 2026
Processing time: 253 Days and 10.1 Hours
The outcome of renal transplantation is influenced by the occurrence of various complications, including urological. One of the most frequently occurring com
To evaluate the incidence, identify potential risk factors, and assess the clinical outcomes related to post-transplant lymphocele formation.
We analyzed 86 consecutive renal transplant recipients from 2010 to 2019. All recipients had undergone protocol screening ultrasound scan at first week after surgery and 3 months post-transplant. We analyzed risk factors for lymphocele fo
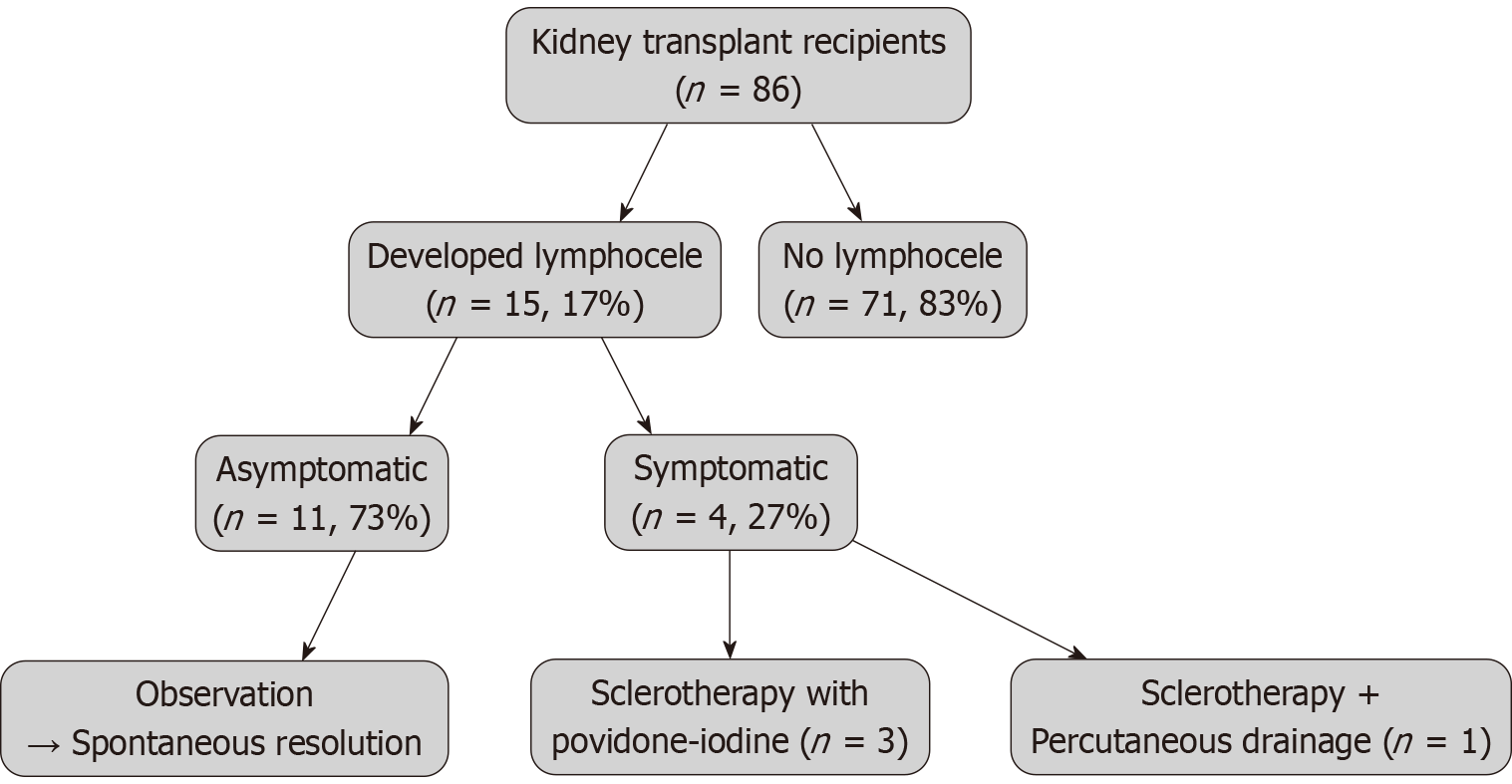
Among the 86 kidney transplant recipients, 15 patients (17.4%) developed a lymphocele, with a median onset of 30 days post-transplantation (range: 30-330 days). The condition was asymptomatic in the majority of cases (14 patients, 93%), while one patient (7%) presented with ipsilateral lower limb lymphedema, which led to the diagnosis. Therapeutic intervention was necessary in four cases. Sclerotherapy using a povidone-iodine solution was performed in three patients, while one patient underwent combined sclerotherapy and percutaneous drainage.
Multivariate analysis identified two independent risk factors: Warm ischemia time exceeding 30 minutes [P = 0.011; odds ratio (OR) = 1.103; 95%CI: 1.023-1.109] and donor age over 50 years (P = 0.041; OR = 1.072; 95%CI: 1.023-1.146). Renal function at one year was comparable between patients who developed a lymphocele and those who did not.
Our findings suggest that improved surgical techniques and optimized immunosuppressive management could contribute to reducing the incidence of lymphocele and enhancing post-transplant renal outcomes.
Core Tip: Lymphocele is a frequent complication after kidney transplantation, but its risk factors remain poorly studied in North African populations. This retrospective study of Tunisian kidney transplant recipients identified prolonged warm ischemia time and older donor age as independent predictors of lymphocele formation. The findings emphasize the importance of targeted preventive strategies in this specific regional context to improve graft outcomes and patient care.
- Citation: Tlili S, Ghabi H, Mami I, Kaaroud H, Rais L, Mohamed Karim ZR. Lymphocele after kidney transplantation: Incidence, associated factors and outcomes. World J Nephrol 2026; 15(1): 111612
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/111612.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.111612
Lymphocele is one of the surgical complication after renal transplantation. Its incidence has been reported to be up to 40% in the first year after transplantation[1-3], particularly in the first six months[3]. It is defined as a lymphatic disorder with a liquid accumulation around the graft. The lymphatic leak may originate from disruption of both recipient vessel and graft ones. Most frequently, it is described as an asymptomatic disorder progressing favorably. Nevertheless, in some other cases, it was associated with severe complication and impaired kidney transplantation (KT) outcomes.
Recent data show that lymphocele incidence varies globally, ranging from 0.6% to 26% in North America and Europe, and up to 40% in some Asian and African cohorts[4]. This highlights the importance of regional studies to identify specific risk profiles.
Since few studies had been interested to post transplantation evaluation no clear consensus on the risk factors in the literature, The aim of our study is to determine the incidence, risk factors, and clinical outcomes of post-transplant lymphocele in a North African cohort in order to an early screening and better management of high risk population. We hypothesized that specific surgical (e.g., warm ischemia time) and donor-related factors (e.g., age) would be independently associated with increased lymphocele risk.
We conducted a retrospective cohort study over a nine-year period at the Department of Nephrology, Dialysis, and Transplantation of La Rabta Hospital in Tunis, Tunisia.
This was a retrospective study, which inherently limits causal inference and is susceptible to potential selection and information biases. Nevertheless, data were consistently recorded in a prospective database, which mitigates recall bias.
The study included all adult patients (aged 18 years and older) who underwent KT between 2010 and 2019, from both living and deceased donors. All included recipients had a minimum post-transplant follow-up of 12 months. Patients were excluded if they received their transplant at another center, had incomplete medical records, or died within the first year following transplantation.
The sample size (n = 86) reflects all eligible kidney transplant recipients at our center over a 9-year period. Although modest, it was sufficient to identify statistically significant associations in multivariate models. A formal power calculation was not performed.
Data were obtained from consecutive adult kidney transplant recipients who underwent transplantation between November 2010 and November 2019. Information was retrieved from the institutional database of kidney transplant patients maintained by the Department of Nephrology, Dialysis, and Transplantation at La Rabta Hospital in Tunis, Tunisia.
The main study outcome was lymphocele onset up to one-year post discharge of KT. Concerning the surgical technic, an extraperitoneal approach was performed in all patients with a pararectal incision. Vascular anastomosis was performed between Grafts’ vessels and the iliac ones. All visible lymphatic vessels dissected were ligated. In addition, an uretero-ureteral or a vesicoureteral anastomosis with Lich-Gregoir technique has been realized. A surgical drain was placed in the extraperitoneal and was left until drainage is less than 50 mL/24 hours.
A routinely screening by regular ultrasound was performed in all recipients with a schedule: One time/week until patient’s discharge, one time/month to the end of six months and on indication after that.
Lymphocele was defined as a perirenal fluid collection detected on ultrasound with a typical anechoic or hypoechoic aspect, located near the iliac vessels or transplant ureter, without internal septations. A size threshold of > 2 cm in diameter was used to define lymphocele. Asymptomatic cases were identified through routine protocol-based ultrasound screening and were monitored unless symptoms or size progression warranted intervention.
The average post-transplant hospitalization duration was 10 ± 3 days, during which screening was performed once per week. Graft loss was defined a return to permanent dialysis within the first year after transplantation.
A renal graft dysfunction was defined as a serum creatinine ≥ 132 μmol by the end of the first renal transplantation according to the literature[5]. The diagnosis of delayed graft function (DGF) was established if dialysis was required in the first week post KT.
The following factors were statistically analyzed: Recipient age, gender, recipient’s comorbidities, BMI, primary renal disease, type and duration of dialysis, type of donor, donor age, cold and warm ischemia time, induction and maintenance immunosuppression, DGF, acute rejection episodes and the different medical or surgical complications during the first year post-operative.
All statistical analyses were conducted using SPSS software, version 25.0. Descriptive statistics were used to summarize the baseline characteristics of the study population. Comparisons between patients with and without lymphocele after KT were made using Student’s t-test, Mann-Whitney U test, and χ2 test, as appropriate. To identify potential risk factors, a multivariate analysis was carried out using binary logistic regression. This model was adjusted for variables that showed a significant association in the univariate analysis, as well as for potential confounders reported in the literature.
Variables with a P > 0.2 in the univariate analysis were included in the multivariate logistic regression model. No additional variables were forced into the model. This approach allowed for identification of independent predictors among those with the strongest univariate association with lymphocele development. Variables included in the multivariate model were: Donor age, warm ischemia time, dialysis modality (hemodialysis vs peritoneal dialysis), and DGF. A P-value of ≤ 0.05 was considered statistically significant, with a 95%CI used throughout the analysis.
A total of 86 consecutive KT were performed at our center during the study period. The mean age was 35.3 ± 9.7 (17-58 years) and 57 (66%) patients were male. Smoking was noted in 36 (41%) of patients. The mean body mass index was 24.8 ± 4.44 kg/m2 (17.4-35). Hypertension was the principal comorbidity in our sample present in 60 (70%) of patients in pre KT. Primary renal disease was to glomerulonephritis in 44% of cases. Overall, 60 patients (70%) had been on hemodialysis with a median time on dialysis before KT of 19 months. The majority of our patients had received a kidney from living donor in 95% of cases. A summary of baseline characteristics of patients are presented in Table 1.
| Variables | Lymphocele group (n = 71) | Non lymphocele group (n = 15) | P value |
| Mean age (years) | 36.2 ± 10.9 | 35.9 ± 9.4 | |
| Male gender | 9 | 48 | |
| Body mass index (kg/m2) | 23.2 ± 3.5 | 25.1 ± 4.6 | |
| Dialysis method | |||
| Hemodialysis | 7 | 53 | 0.033 |
| intra peritoneal catheter | 8 | 16 | 0.028 |
| Mean donor age (year) | 50.8 ± 11.3 | 43 ± 10.8 | 0.015 |
| Mean warm ischemia time (minute) | 42.6 ± 12.1 | 34.7 ± 7.9 | 0.002 |
| Acute rejection | 2 | 3 | |
| Infection complication | 11 | 46 | |
| Graft loss at one year | 2 | 1 |
Induction therapy consisted on anti-lymphocyte thymoglobulin in 93% of cases. Concerning maintenance therapy, all our patients were under steroids and mycophenolate mofetil associated with tacrolimus in 70 (81%) of recipients.
A total of 15 patients (17%) developed a lymphocele, with a mean onset of 30 days following KT (range: 30-330 days). The condition was diagnosed within the first 3 months in 10 cases (67%), and between 3 months and 1 year in 5 cases (33%). Most patients (14 out of 15, 93%) remained asymptomatic, whereas one patient (7%) presented with ipsilateral lower limb lymphedema, which led to the diagnosis. Intervention was necessary in four cases: Three patients underwent sclerotherapy with povidone-iodine, and one patient required sclerotherapy combined with percutaneous drainage. The median time to resolution for treated lymphoceles was 45 days (range: 30-90 days), with no treatment-related complications observed (Figure 1).
Unifactorial analysis identified four predictive factors related to the incidence of lymphocele. They were: Presence of peritoneal dialysis catheter at the time of transplantation P = 0.028; odds ratio (OR) = 3.786; 95%CI: 1.189-12.055, hemodialysis P = 0.033; OR = 3.563; 95%CI: 1.126-11.277, warm ischemia > 30 minutes P = 0.002; OR = 7.9; 95%CI: 2.947-12.933 and donor age > 50 years P = 0.015; OR = 7.6; 95%CI: 1.504-13.839.
The independent risk factors significant in multifactorial analysis were only warm ischemia > 30 minutes P = 0.011; OR = 1.103; 95%CI: 1.023-1.109 and donor age > 50 years P = 0.041; OR = 1.072; 95%CI: 1.023-1.146.
Renal function was comparable between no-lymphocele and lymphocele group at one year. At one year, graft dysfunction occurred in 3 of 15 patients (20%) in the lymphocele group vs 10 of 71 patients (14%) in the non-lymphocele group. Graft loss occurred in 2 patients with lymphocele and in 1 patient without. These differences were not statistically significant. There were not a higher risk for graft loss among patients with lymphoceles. Infectious, surgical complication and hospital readmission rate were similar between the two groups.
Lymphocele is a common surgical complication after KT. In our study, 17% of patients had experienced this complication in the first year after transplantation, which is in line with previous reports. The incidence ranges between 0.6% and 51% among literature[4-6]. The variability of these rates could be explained by the diagnosis strategy: The highest rates were reported in studies in which there was a systematic ultrasound search for lymphocele compared to others in which lymphocele screening was based only on warning signs[7,8].
Most of our renal recipients presented an asymptomatic lymphocele with incidental finding on ultrasound exam. This result is consistent with the data previously publihed[6]. However, lymphocele can be responsible for impairment graft function by compressing ureter, transplant vasculatureor the graft. Also, lymphocele can causes deep vein thrombosis, lower limb or genital edema, compressive syndrome of the vena cava or the portal vein or fever[9,10].
Thus, lymphocele management depend on clinical presentation. Authors in the most of series recommend starting with minimally invasive measures[9]. Most often, asymptomatic lymph disorder do not require treatment but should be followed closely with clinical, laboratory and radiological monitoring unless the volume exceeds 140 mL, which in line with our attitude, 93% of lymphocele had been spontaneously resolved.
The rate of patient requiring treatment ranges between 0.04% and 14.6% among literature. Therapeutic options consist on: Percutaneous drainage (simple aspiration or image-guided percutaneous catheter drainage) associated or not to sclerotherapy, which has been proven effectiveness in previous reports[11,12]. This was the strategy opted by our center in all cases.
In our center, povidone-iodine was chosen due to its wide availability, ease of use, and reported success in prior small studies. Although some concerns exist regarding potential nephrotoxicity, the treated patients maintained stable renal function throughout follow-up. No adverse effects related to sclerotherapy were observed in our series. Nonetheless, this approach should be carefully considered and individualized.
In the other side, surgical treatment with marsupialization of the lymph collection into the peritoneal cavity is considered the treatment of choice by many authors[2,13,14].
In a recent systematic review regarding the management of lymphocele, it had been reported that laparoscopic surgery is the less associated with recurrent lymphocele with a rate of 8%. This rate is reported to be around 50% after percutaneous drain placement, 31% after sclerotherapy and 16% after open surgery[4].
Given that lymphocele is a frequent surgical complication that may impaired KT results, clinician should not only mange it but also prevent its onset. Thus, identifying patients at high risk of lymphocele after KT would had benefic effect on graft recipient’s outcomes. Our analysis had showed that donor age superior to 50 years and prolonged warm ischemia are independent risk factors of lymphocele in the first year after transplantation. In fact, among literature few authors had been interested to risk factors, which remain unclearly established.
Based on causes and pathophysiology of post-operative lymphocele, statistical association showed in our study would be explained. In fact, it has been clearly demonstrated that lymphocele originates frequently from lymphatic chain coursing around iliac vessels[12]; hence, a prolonged warm ischemia may involve technical difficulties, particular vascular anastomosis with extensive dissection and lymphatic disruption[15]. Prolonged warm ischemia time reflects extended handling and vascular anastomosis duration, which can increase the risk of lymphatic vessel injury or extensive dissection[15]. Furthermore, prolonged ischemia may promote local inflammation, impair healing, and contribute to increased lymphatic leakage postoperatively[15].
In addition, prolonged warm ischemia is associated with acute rejection because of prolonged anoxia, which is well known as risk factor of lymphocele. Indeed, it has been reported that acute rejection is associated with increasing lymphatic flow and 50-fold higher lymphangiogenisis[12,16-18].
The threshold of 30 minutes for warm ischemia time and 50 years for donor age were based on prior studies identifying these cut-offs as significant predictors of surgical complications post-transplantation[14,19].
In the other side, post-operative lymphocele may also originates from donor’s lymphatic vessels dissected either during the time of organ procurement surgery or transplantation time[12,19]. A more advanced age involve poorer organ’s quality, impaired tissue healing and prolonged lymphorrhea[20,21]. Bentas et al[19], first authors who established this association, had reported that “higher fragility of renal lymphatic vessels and more abundant perihilar fatty tissue of older donor kidneys” are the probable explanation for this association[19].
Other authors had commonly reported other medical risk factors, particularly acute rejection, DGF and mammalian target of rapamycin inhibitors[3,22], our study failed to find such association, may be because of the small sample size which may weaken our statistical analysis.
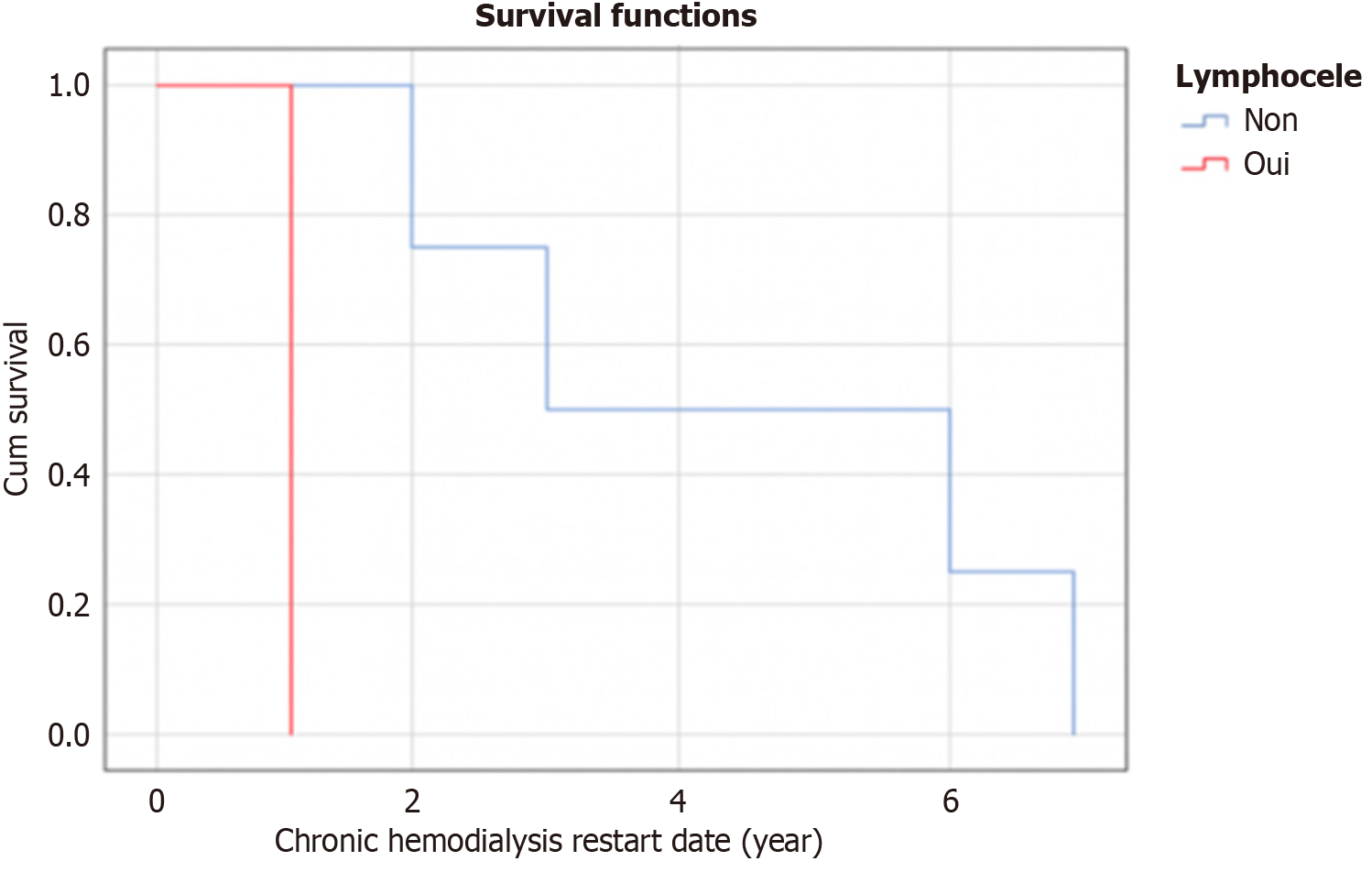
Our analysis did not highlight any association of lymphocele with neither graft dysfunction or survival, nor patient survival or other comorbidities within the first year after transplantation. This is in concordance with results of other authors[15,22] (Figure 2).
Some other studies had reported that lymphocele is associated with high risk of graft lost particular in the first year following KT[23-25].
Further larger studies are required in order to determine exact risk factors and the association between lymphocele and graft survival.
This study has several limitations. First, its retrospective and single-center design may limit generalizability. Second, the relatively small sample size may reduce the statistical power to detect associations, particularly for rare outcomes. Third, no long-term outcomes beyond one year were assessed. Prospective multicenter studies with larger cohorts are needed to validate these findings.
In conclusion, despite surgical technic development, lymphocele remain a frequent vascular complication with possible impaired KT results. It is mainly induced by surgical causes such as dissection and non-ligature of lymphatic vessel. However, many other medical factors related to either donor or recipient are involved. Our analysis had demonstrated that lymphocele development risk is correlated to warm ischemia time and donor age. Thus, patients at high risk should be closely followed up, quickly and well managed.
| 1. | Derweesh IH, Ismail HR, Goldfarb DA, Araki M, Zhou L, Modlin C, Krishnamurthi V, Flechner SM, Novick AC. Intraoperative placing of drains decreases the incidence of lymphocele and deep vein thrombosis after renal transplantation. BJU Int. 2008;101:1415-1419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 2. | Atray NK, Moore F, Zaman F, Caldito G, Abreo K, Maley W, Zibari GB. Post transplant lymphocele: a single centre experience. Clin Transplant. 2004;18 Suppl 12:46-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 70] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 3. | Heer MK, Clark D, Trevillian PR, Sprott P, Palazzi K, Hibberd AD. Functional significance and risk factors for lymphocele formation after renal transplantation. ANZ J Surg. 2018;88:597-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 4. | Lucewicz A, Wong G, Lam VW, Hawthorne WJ, Allen R, Craig JC, Pleass HC. Management of primary symptomatic lymphocele after kidney transplantation: a systematic review. Transplantation. 2011;92:663-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 91] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 5. | Adani GL, Baccarani U, Bresadola V, Lorenzin D, Montanaro D, Risaliti A, Terrosu G, Sponza M, Bresadola F. Graft loss due to percutaneous sclerotherapy of a lymphocele using acetic acid after renal transplantation. Cardiovasc Intervent Radiol. 2005;28:836-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 6. | Mihaljevic AL, Heger P, Abbasi Dezfouli S, Golriz M, Mehrabi A. Prophylaxis of lymphocele formation after kidney transplantation via peritoneal fenestration: a systematic review. Transpl Int. 2017;30:543-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Doehn C, Fornara P, Fricke L, Jocham D. Laparoscopic fenestration of posttransplant lymphoceles. Surg Endosc. 2002;16:690-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Derouiche A, Mechri M, Ktari MM, Helal I, Ben Abdallah T, Chebil M. Lymphoceles after renal transplantation: Study of risk factors. Prog Urol. 2010;20:301-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 9. | Berardinelli L, Raiteri M, Pasciucco A, Carini M. The use of a polymeric sealant for prevention of posttransplantation lymphocele. Transplant Proc. 2011;43:1072-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 10. | Syversveen T, Midtvedt K, Brabrand K, Øyen O, Foss A, Scholz T. Prophylactic peritoneal fenestration to prevent morbidity after kidney transplantation: a randomized study. Transplantation. 2011;92:196-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 11. | Iwan-Zietek I, Zietek Z, Sulikowski T, Nowacki M, Zair L, Romanowski M, Zukowski M, Rość D, Ostrowski M. Minimally invasive methods for the treatment of lymphocele after kidney transplantation. Transplant Proc. 2009;41:3073-3076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Ranghino A, Segoloni GP, Lasaponara F, Biancone L. Lymphatic disorders after renal transplantation: new insights for an old complication. Clin Kidney J. 2015;8:615-622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 83] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 13. | Lima ML, Cotrim CA, Moro JC, Miyaoka R, D'Ancona CA. Laparoscopic treatment of lymphoceles after renal transplantation. Int Braz J Urol. 2012;38:215-221; discussion 221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 14. | Ulrich F, Niedzwiecki S, Fikatas P, Nebrig M, Schmidt SC, Kohler S, Weiss S, Schumacher G, Pascher A, Reinke P, Tullius SG, Pratschke J. Symptomatic lymphoceles after kidney transplantation - multivariate analysis of risk factors and outcome after laparoscopic fenestration. Clin Transplant. 2010;24:273-280. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 15. | Zagdoun E, Ficheux M, Lobbedez T, Chatelet V, Thuillier-Lecouf A, Bensadoun H, Ryckelynck JP, Hurault de Ligny B. Complicated lymphoceles after kidney transplantation. Transplant Proc. 2010;42:4322-4325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 16. | Khauli RB, Stoff JS, Lovewell T, Ghavamian R, Baker S. Post-transplant lymphoceles: a critical look into the risk factors, pathophysiology and management. J Urol. 1993;150:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 110] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 17. | Bzoma B, Kostro J, Dębska-Ślizień A, Hellmann AR, Zadrożny D, Śledziński Z, Rutkowski B. Treatment of the Lymphocele After Kidney Transplantation: A Single-center Experience. Transplant Proc. 2016;48:1637-1640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 18. | Favi E, Puliatti C, Iesari S, Monaco A, Ferraresso M, Cacciola R. Impact of Donor Age on Clinical Outcomes of Primary Single Kidney Transplantation From Maastricht Category-III Donors After Circulatory Death. Transplant Direct. 2018;4:e396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 19. | Bentas W, Jones J, Karaoguz A, Tilp U, Probst M, Scheuermann E, Hauser IA, Jonas D, Gossmann J. Renal transplantation in the elderly: surgical complications and outcome with special emphasis on the Eurotransplant Senior Programme. Nephrol Dial Transplant. 2008;23:2043-2051. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 20. | Joosten M, d'Ancona FC, van der Meijden WA, Poyck PP. Predictors of symptomatic lymphocele after kidney transplantation. Int Urol Nephrol. 2019;51:2161-2167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 21. | Thomas C, Ziewers S, Thomas A, Dotzauer R, Bartsch G, Haferkamp A, Tsaur I. Development of symptomatic lymphoceles after radical prostatectomy and pelvic lymph node dissection is independent of surgical approach: a single-center analysis. Int Urol Nephrol. 2019;51:633-640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 22. | Chatzikyrkou C, Menne J, Gwinner W, Schmidt BM, Lehner F, Blume C, Schwarz A, Haller H, Schiffer M. Pathogenesis and management of hypertension after kidney transplantation. J Hypertens. 2011;29:2283-2294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 23. | De Coppi P, Giuliani S, Fusaro F, Zanatta C, Zacchello G, Gamba P, Zanon GF. Cadaver kidney transplantation and vascular anomalies: a pediatric experience. Transplantation. 2006;82:1042-1045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 24. | Giuliani S, Gamba P, Kiblawi R, Midrio P, Ghirardo G, Zanon GF. Lymphocele after pediatric kidney transplantation: incidence and risk factors. Pediatr Transplant. 2014;18:720-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Dubeaux VT, Oliveira RM, Moura VJ, Pereira JM, Henriques FP. Assessment of lymphocele incidence following 450 renal transplantations. Int Braz J Urol. 2004;30:18-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |