Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.110867
Revised: July 28, 2025
Accepted: January 6, 2026
Published online: March 25, 2026
Processing time: 269 Days and 14.3 Hours
The Wilms tumor 1 (WT1) mutation is significantly associated with steroid-resistant nephrotic syndrome (SRNS) in children. It causes a spectrum of di
We report a rare familial occurrence of shared mutation of WT1 (at intron 9 splice site: C.1432+5G>A) and COL4A5 genes in two siblings. The elder sibling (46, XY) presented with infantile steroid-resistant nephrotic syndrome and rapidly deteriorating and progressing to end-stage kidney disease with XY karyotype. The genetic testing confirmed the diagnosis of Frasier syndrome associated with a WT1 mutation in the elder sibling. She is currently on maintenance dialysis, awaiting renal transplantation. The younger sibling presented at age four with facial edema and SRNS with slower disease progression and no apparent gonadal anomalies. Her renal biopsy revealed minimal change disease, and she maintains stable renal function under antiproteinuric therapy. Genetic testing confirmed the diagnosis of WT1 mutation with 46, XX karyotype in the younger sibling. Both siblings also carried a COL4A5 gene variant of uncertain significance. Family history reveals consanguineous marriage and a similar presentation in a sibling.
Despite identical mutations, phenotypic differences highlight complex genotype-phenotype relations, stressing the need for research, genetic counseling, and family member screening.
Core Tip: This report describes two siblings with identical pathogenic Wilms tumor 1 but distinct karyotypes (46, XY and 46, XX) leading to divergent clinical presentations. The elder sibling had classical features with early-onset steroid-resistant nephrotic syndrome with rapidly deteriorating renal function, while the younger sibling presented with steroid-resistant nephrotic syndrome only without gonadal abnormalities. This rare familial occurrence emphasizes sex-dependent phenotypic variability despite identical mutations, expanding understanding of Wilms tumor 1-related disorders. The findings underscore the importance of genetic testing, detailed family evaluation, and long-term monitoring to guide management in such rare syndromic presentations.
- Citation: Khandelwal MH, Piparva KG, Parchwani D. Familial WT1-associated nephropathy - 46, XY Frasier syndrome and 46, XX steroid-resistant nephrotic syndrome in female siblings: A case report and review of literature. World J Nephrol 2026; 15(1): 110867
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/110867.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.110867
Steroid-Resistant Nephrotic Syndrome (SRNS) is a challenging clinical entity in pediatric nephrology, representing a significant subset of cases that fail to respond to standard corticosteroid therapy. Approximately up to 25% of SRNS cases are attributed to mutations in genes predominantly expressed in podocytes, specialized epithelial cells essential for the integrity of the glomerular filtration barrier[1-3]. Among these genes, Wilms tumor 1 (WT1) was the first identified to harbor pathogenic variants associated with SRNS[4]. Initially recognized as a tumor suppressor gene linked to Wilms tumor. WT1 disorder is characterized by congenital or infantile onset of SNRS, mainly with glomerular lesions[5].
Frasier syndrome is a rare autosomal dominant genetic disorder caused by mutations in the WT1 gene, characterized by SRNS with onset in early childhood, progressive kidney failure, and disorders of sex development. The prevalence rate of Frasier syndrome is less than 1 in 1000000, with approximately 150 cases reported worldwide to date[6]. WT1 disorder is caused by a heterozygous pathogenic variant in the WT1 gene. The WT1 gene primarily functions in the regulation of genitourinary system development. WT1 mutations cause a wide spectrum of renal and extrarenal manifestations[5]. WT1 encodes a transcription factor containing an N-terminal transactivation domain (exon 1) and four zinc fingers at the C-terminus (exons 7-10). It serves as a regulatory transcription factor for both renal and gonadal development and plays multiple essential roles in the embryonic development of various systems, including the central nervous system and mesothelial organs[7].
Depending on the exon involved, pathogenic variants of the WT1 gene can lead to Denys-Drash syndrome (DDS) or Frasier syndrome. Both conditions are characterized by progressive glomerulopathy and a predisposition to Wilms’ tumor[8]. Frasier syndrome is caused by heterozygous mutations in the donor splice site in intron 9 of the Wilms tumor gene, resulting in insertion of the amino acids lysine (K), threonine (T) and serine (S), altering the ratio of +KTS/-KTS isoforms of WT1. A splice site at exon 9 inserts three additional amino acids between the third and the fourth zinc finger (usually referred to as the KTS splice insert). Mutations in the KTS site result in Frasier syndrome[9]. A group of mostly missense mutations in exons 8 and 9 affect the zinc-finger domains, which impair the DNA-binding capacity of WT1 and cause either DDS or Meacham syndrome[10].
Patients with Frasier syndrome have delayed or poor pubertal development. It is also associated with a high risk of developing End Stage Kidney Disease, gonadal tumors, and male pseudo hermaphroditism (female external genitalia with XY sex chromosomes) in the second or third decade of life[6,11]. The rarity and complex inheritance of WT1-related nephropathies make familial occurrences extremely uncommon. Here, we report the clinical and genetic findings in two female siblings with identical WT1 mutations who presented with differing phenotypes: One consistent with Frasier syndrome (XY karyotype) and the other with SRNS with XX karyotype. This case provides valuable insight into the spectrum of WT1-associated disorders and highlights the variable expressivity that can occur even within the same family.
Elder sibling: A female child presented with intermittent periorbital puffiness in the last 2 months at the age of one year (December 2015). Her parents consulted a pediatric nephrologist. After clinical examination, he advised urine examination, which revealed proteinuria, confirming the case of infantile nephrotic syndrome with right-sided hydronephrosis. She was treated with oral methylprednisolone 2 mg/kg/day for a six-week duration; however, proteinuria persisted, and she was resistant to steroids and diagnosed with SRNS. Then she was treated with calcineurin inhibitor cyclosporine (4-5 mg/kg in two divided doses) with continuous monitoring of kidney function and serum electrolytes. After an initial response, over time, the response diminished, with intermittent periods of clinical improvement. Eventually, she developed resistance to calcineurin inhibitors, and cyclosporine was discontinued.
Younger sibling: Her younger sister presented with similar symptoms, i.e. facial edema at the age of 4 years (June 2019) and was evaluated by a pediatric nephrologist. Urinalysis confirmed nephrotic syndrome. She was prescribed a full course of corticosteroids at 2 mg/kg/day for six weeks. However, the proteinuria persisted, and she was resistant to it. The pediatric nephrologist confirmed a clinical diagnosis of primary SRNS.
Elder girl: Her renal function progressively declined, eventually leading to chronic kidney disease.
Younger girl: She is under antiproteinuric therapy with stable renal function.
Elder sibling: There is no history of any past medical illness.
Younger sibling: Elder sister was diagnosed with Frasier syndrome 2 years ago (2018).
No known genetic disorders have been reported in either parent’s family or the maternal lineage. The mother, of Nepali origin residing in India, has a history of consanguineous marriage and two miscarriages prior to the birth of these daughters. A similar clinical presentation was reported in a female child from her previous marriage; however, no medical records are available to verify this.
Elder sibling: On examination, the patient appeared alert and well-oriented. Periorbital and facial edema were noted, without generalized edema or ascites. Vital signs were stable with a normal heart rate, blood pressure, and respiratory rate. Cardiovascular, respiratory, abdominal, and neurological examinations were unremarkable. No lymphadenopathy or organomegaly was detected. Growth parameters were appropriate for age.
Younger sibling: On examination, the patient with periorbital and facial edema was noted, without generalized edema or ascites, with stable vital parameters. Cardiovascular, respiratory, abdominal, and neurological examinations were unremarkable. No lymphadenopathy or organomegaly was detected. Growth parameters were appropriate for age.
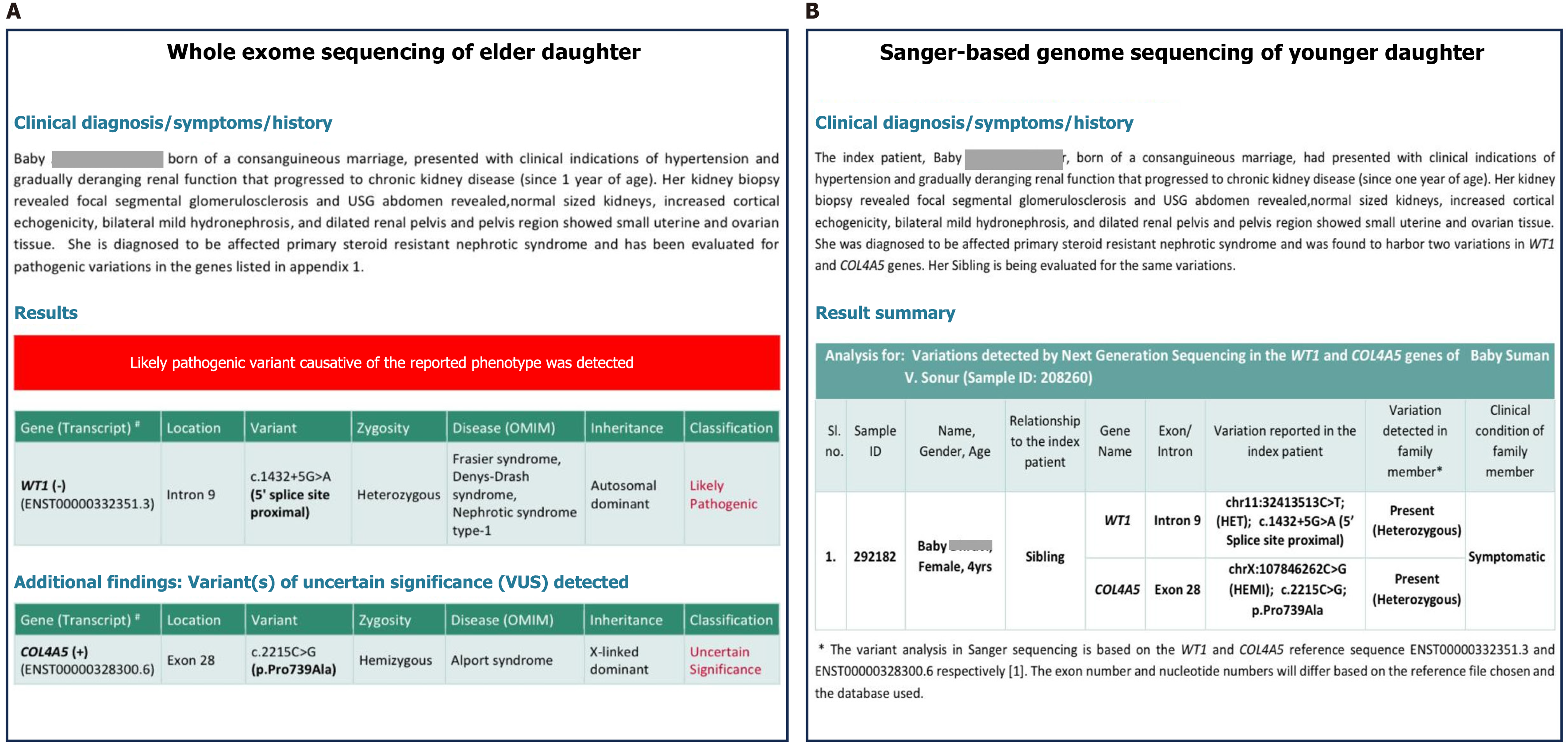
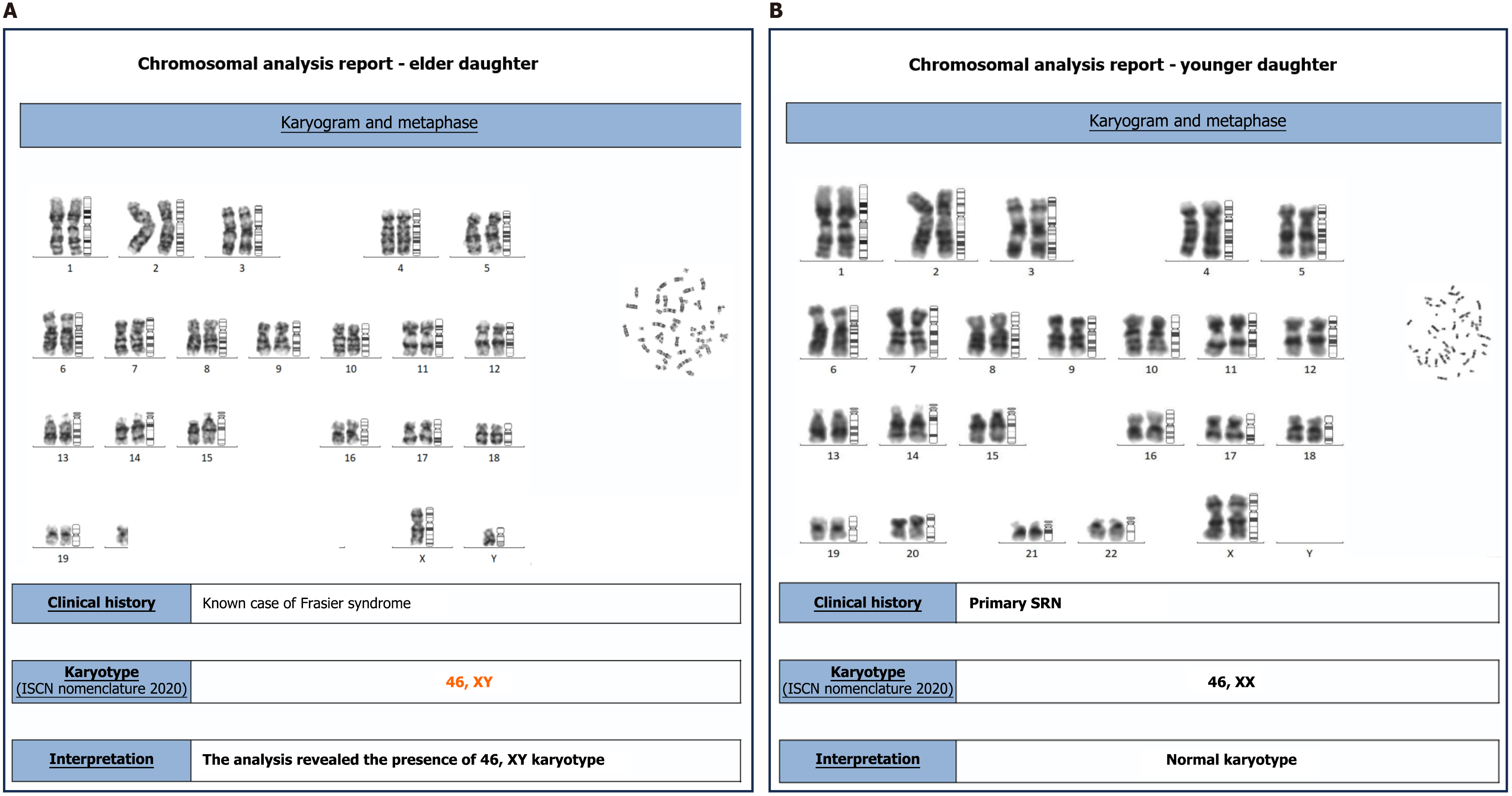
Elder sibling: (1) Renal biopsy: In view of clinically diagnosed SRNS, the pediatric nephrologist advised her renal biopsy (January 2016), which revealed focal mesangial-proliferative glomerulonephritis; (2) Genetic analysis: As she was a case of SRNS and rapid progressive renal dysfunction, she was advised to undergo genetic testing (August 2018). Next generation sequencing for the WT1 and COL4A5 genes revealed presence of two pathogenic variants: A heterozygous variant in the WT1 gene (chr11: 32413513C>T; c.1432+5G>A, located in intron 9 at the 5’ splice site), and a hemizygous variant in the COL4A5 gene (chrX: 107846262C>G; c.2215C>G; p.Pro739Ala in exon 28). These findings are consistent with a genetic etiology of SRNS (Figure 1); and (3) Karyotype analysis: Following the genetic testing results, the nephrologist recommended karyotyping (August 2018) for further evaluation. It showed a 46, XY chromosomal complement in a phenotypic female (Figure 2).
Younger sibling: (1) Renal biopsy: Due to the family history of SRNS in the elder sibling, the pediatric nephrologist advised a renal biopsy, which revealed the finding of “minimal change disease”; (2) Genetic analysis: She was advised to undergo Sanger sequence for the target gene analysis (September 2019). The test result revealed that the younger one also carries two heterozygous genetic variants: One in the WT1 gene (chr11: 32413513C>T affecting intron 9) and one in the COL4A5 gene (chrX: 107846262C>G affecting exon 28, resulting in p.Pro739Ala). These are identical to the variants identified in her elder sibling (Figure 1); and (3) Karyotype analysis: She also underwent karyotype analysis (September 2019). Unlike her elder sister, her karyotype analysis showed a normal female chromosome pattern (46, XX) (Figure 2).
Both siblings were advised to undergo abdominal ultrasonography, which revealed normal findings, with no structural abnormalities observed in the kidneys or other abdominal organs.
Based on the clinical presentation of SRNS, a 46, XY karyotype with a female phenotype, and the identification of a WT1 mutation at the 5’ splice site of intron 9 on genetic testing, the pediatric nephrologist confirmed the diagnosis of Frasier syndrome.
Based on the clinical presentation of SRNS, a 46, XX karyotype with a female phenotype, and the identification of a WT1 mutation at the 5’ splice site of intron 9 on genetic testing, the pediatric nephrologist confirmed the diagnosis of WT1-associated nephropathy.
Currently, she is on maintenance hemodialysis twice a week as well as an antiproteinuric protocol.
She is treated with an antiproteinuric protocol with continuous monitoring of renal function.
She is progressing to End Stage Renal Disease (ESRD). Her family was advised and counseled for renal transplantation.
She remains asymptomatic with preserved renal function and is under ongoing antiproteinuric therapy.
Both siblings had pathogenic WT1 mutation at the 5’ splice site of intron 9, along with a COL4A5 variant; however, their clinical and phenotypic presentations differed markedly. The elder sibling, with a 46, XY karyotype, presented with classical features of Frasier syndrome, and had rapid progressive nephropathy leading to chronic kidney disease[12]. In contrast, the younger sibling, who had a 46, XX karyotype, exhibited isolated SRNS with a later onset and a more indolent progression of renal dysfunction, without any extrarenal manifestations. A similar pattern was observed in the cases reported by Klamt et al[13] describing two sisters - ws131 (46, XX) and ws131B (46, XY) - who carried the same +5 G→A splice-site mutation in intron 9 of the WT1 gene but demonstrated differing clinical outcomes, highlighting the variable expressivity and phenotypic spectrum associated with WT1 mutations. We have included published case reports of WT1-associated nephropathy, as shown in Table 1[9,13-24], which provide comprehensive details on both genotypic and phenotypic characteristics.
| Ref. | WT1-mutation type | Phenotype | Karyotype | Renal pathology | Syndrome |
| Barbaux et al[9], 1997 | IVS9+1G>A and IVS9+5G>A splice-site | Female | 46, XY | FSGS, SRNS, gonadal dysgenesis | Frasier syndrome |
| Klamt et al[13], 1998 | IVS9+5G>A splice-site | Females | 46, XY and 46, XX | FSGS, SRNS | Frasier syndrome |
| Guaragna et al[14], 2012 | WT1 IVS9+5G>A (cases 1-3), IVS9+1G>A | Female | 46, XY | FSGS, SRNS, ESRF, puberty, dysgerminoma, streak gonads | Frasier syndrome |
| Dattolo et al[15], 2013 | Exon 6 truncating (c.1012A>T, p.R338X) | Female | 46, XX | FSGS, Gonadoblastoma | Atypical WT1-related syndrome |
| Ahn et al[16], 2017 | Exon 7-9 missense and Intron 9 splice | Female and male | 46, XY (Predominantly, 46, XX | FSGS, SRNS, DSD and ESRD | Denys-Drash syndrome |
| Akramov et al[17], 2021 | Exon 7 (chr11:32417947G>A), | Male | 46, XY | Nephroblastoma, ambiguous genitalia | Variant of Denys-Drash syndrome |
| Wang et al[18], 2021 | Exon 7 novel missense (c.1420C>T, p.His474 Tyr) | Female | 46, XY | Gonadoblastoma, dysgerminoma | Denys-Drash syndrome |
| Zhang et al[19], 2022 | intron 9 of, c.1432+5G>A splice-site | Female | 46, XY | FSGS, SRNS and pulmonary dysplasia | Frasier syndrome |
| Arroyo-Parejo et al[20], 2022 | WT1 | Female | 46, XY | ESKD, abnormalities of the male external genitalia | Frasier syndrome |
| Shao Q, et al[21], 2023 | WT1 intron 9 mutation (c.1447+4C>T) | Female | 46, XY | SNRS with progressive nephropathy | Frasier syndrome |
| Al Zabali et al[22], 2024 | WT1 mutations | Female and male | 46, XY and 46, XX | ESRF, thrombotic microangiopathy | WT1-associated atypical hemolytic uremic syndrome |
| Tran Thuy et al[23], 2024 | p.Arg467Gln exon 9 missense | Female and male (Neonate) | 46, XY and 46, XX | ESRD within the first month of their life | Aggressive Denys-Drash syndrome variant |
| McTaggart SJ et al[28], 2001 | T1 c.1447+4C>T (intron 9) | Female | 46, XY | FSGS | Frasier syndrome |
| Chan et al[24], 2006 | WT1 intron 9 mutation (c.1447+4C>T) | Female and male | 46, XY | FSGS, SRNS, progressed to ESRD | Frasier syndrome |
Table 1 compiles reported cases of WT1 gene mutation-associated nephropathies. It summarizes key features, including mutation type, phenotype, karyotype, and renal pathology, helping to elucidate genotype–phenotype correlations. So far, WT1-related nephropathies have almost exclusively been associated with changes in intron 9 or exon 8 or 9, and they also include emerging atypical presentations (e.g., a novel sequence variant (c.1012A>T) of the WT1 gene in exon 6, atypical hemolytic uremic syndrome-associated WT1 mutations, offering a comprehensive view of the expanding WT1-related disease spectrum. Classic Frasier syndrome is consistently associated with intron 9 splice-site mutations (e.g.+4C>T, +5G>A) in 46, XY individuals, presenting with focal segmental glomerulosclerosis and gonadal dysgenesis[9,14-16,19,24]. DDS variants often involve exon 7-9 missense mutations, manifesting as early-onset diffuse mesangial sclerosis, having a higher risk of Wilms tumor[16-18,23]. Some incomplete or atypical presentations, including exon 6 or truncating mutations, may present later and with milder or overlapping features. Recently, novel WT1 missense variants (e.g. p.Arg467Gln) have been identified in neonatal DDS patients with extremely severe ESRD[21]. Published case series linking WT1 mutations with atypical hemolytic uremic syndrome expand the phenotypic spectrum beyond classic nephropathy[22] (Table 1).
A variety of specialized genetic databases and software tools are available to explore and interpret WT1 mutations. They are “Online Mendelian Inheritance in Man”(OMIM) (Available from: https://www.omim.org), “ClinVar” (Available from: https://www.ncbi.nlm.nih.gov/clinvar/), “Human Gene Mutation Database “(HGMD) (Available from: https://www.hgmd.cf.ac.uk), “Leiden Open Variation Database” (LOVD) (Available from: https://www.lovd.nl), “DECIPHER” (Available from: https://www.deciphergenomics.org), “Orphanet” (Available from: https://www.orpha.net), “Gene Curation Coalition” (GenCC) (Available from: https://thegencc.org) and “GeneReviews “ (Available from: https://www.ncbi.nlm.nih.gov/books/NBK1249/) provides evidence-based clinical validity of gene-disease relationships.
The COL4A5 p.Pro739Ala variant identified in these cases is currently classified as a variant of uncertain significance. The COL4A5 gene, located on the X chromosome, encodes the α5 chain of type IV collagen and is implicated in X-linked Alport syndrome when pathogenic variants are present. In hemizygous males (46, XY), pathogenic COL4A5 variants typically manifest as hematuria, progressive renal failure, sensorineural hearing loss, and ocular abnormalities, characteristic of classical X-linked Alport syndrome[25]. This variant is extremely rare and largely unreported in major population databases such as the Genome Aggregation Database, Exome Variant Server, and TOPMed, highlighting its limited recognition and unclear clinical relevance[26]. Although both siblings had a COL4A5 variant in addition to WT1 mutations, the absence of hematuria and extrarenal manifestations (such as hearing loss or ocular abnormalities) at present strongly supports a diagnosis of WT1-associated nephropathy. However, we cannot entirely exclude the potential role of X-linked inheritance or the COL4A5 variant as a phenotypic modifier, particularly in light of the evolving understanding of digenic inheritance in renal disorders; the overall phenotype aligns more closely with WT1-related glomerulopathy[25]. Frasier syndrome follows a slower course, typically progressing during adolescence or early adulthood during the second decade of life[27]. But in this case, the elder sibling’s rapidly progressive, deteriorating kidney function might be influenced or contributed to by the presence of the COL4A5 variant.
Frasier syndrome is typically suspected during adolescence when phenotypic females with a history of SRNS are evaluated for delayed puberty or absence of menstruation, since most cases have normal-looking female genitalia and are raised as girls[14]. However, diagnosis may be delayed as modest breast development can occur despite estrogen deficiency, and pubertal delay may be mistakenly attributed to chronic illness or immunosuppressive therapy. The diagnosis is confirmed by detecting WT1 mutations[28]. The elder sibling in our case represents a rare presentation of Frasier syndrome, with a 46, XY karyotype, normal-appearing external female genitalia, and no detectable gonadal abnormalities at present. A similar rare presentation of Frasier syndrome was reported by Huang et al[29] in a female sibling who had early-onset SRNS and a WT1 mutation, with no gonadal abnormalities initially, but who later developed gonadoblastoma during adolescence[3]. In our case, early diagnosis in the elder sibling was possible through genetic and karyotyping analysis before she entered adolescence. The relative rarity of 46, XX presentations may also reflect diagnostic bias, as these individuals lack the gonadal abnormalities that trigger early evaluation in 46, XY cases, leading to underdiagnosis[9,30]. Interestingly, the younger sibling had a 46, XX karyotype. However, in our case, the younger sibling with a 46, XX karyotype was diagnosed early due to a known history of Frasier syndrome in the elder sibling with similar manifestations.
The clinical management of WT1-associated SRNS is challenging, as it typically does not respond to corticosteroids or immunosuppressive therapies. Progression to ESRD is inevitable. Once ESRD develops, dialysis and, ultimately, renal transplantation remain the only definite treatment options. Notably, there have been no reported cases of nephrotic syndrome recurrence following kidney transplantation[31]. In addition to renal complications, patients with a 46, XY karyotype and gonadal dysgenesis face a significantly elevated risk of gonadal tumors, particularly in Frasier syndrome, where the risk is estimated at around 60%, compared to approximately 40% in DDS. As a result, early diagnosis and timely prophylactic bilateral gonadectomy are strongly recommended for individuals with Frasier syndrome and DDS to mitigate the risk of malignancy[14]. Until surgical removal is performed, regular monitoring with imaging studies such as pelvic ultrasound or MRI is crucial to detect any early signs of gonadal tumors. After gonadectomy, the child may require long-term hormone replacement therapy to support normal pubertal development and maintain bone health, neces
Genetic counseling is also recommended for the parents to assess the inheritance pattern. The patients have been informed about the potential risk of tumor development and advised to continue regular follow-ups with ongoing monitoring and periodic reassessment of gonadal status. Genetic analysis and long-term follow-up of such exceptional cases will advance the understanding of genotype-phenotype correlations in WT1-related disorders and improve clinical management strategies for affected families. The parents of these siblings have been advised to undergo whole exome sequencing to evaluate potential carrier status or mosaicism, which may help assess recurrence risk and guide future genetic counseling.
Two siblings within a single family shared an identical WT1 mutation, but differing clinical courses, disease progression, and karyotypic presentations highlight the complexity of genotype-phenotype correlations in WT1-associated nephropathies. This case illustrates the intricate interplay between genetics and sex determination in WT1 disorders. Early diagnosis remains challenging. Genetic counseling, long-term follow-up, and proactive monitoring are essential. With confirmed familial inheritance, parental genetic testing and counseling are strongly recommended to help anticipate future reproductive risks. Limitation: Parental genetic testing has not yet been performed, which restricts the ability to determine inheritance patterns.
We sincerely thank the hospital administration for their support, and we are also grateful to the patient’s parents for providing consent to share the clinical information for publication.
| 1. | Benoit G, Machuca E, Antignac C. Hereditary nephrotic syndrome: a systematic approach for genetic testing and a review of associated podocyte gene mutations. Pediatr Nephrol. 2010;25:1621-1632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 116] [Article Influence: 7.3] [Reference Citation Analysis (1)] |
| 2. | Hinkes BG, Mucha B, Vlangos CN, Gbadegesin R, Liu J, Hasselbacher K, Hangan D, Ozaltin F, Zenker M, Hildebrandt F; Arbeitsgemeinschaft für Paediatrische Nephrologie Study Group. Nephrotic syndrome in the first year of life: two thirds of cases are caused by mutations in 4 genes (NPHS1, NPHS2, WT1, and LAMB2). Pediatrics. 2007;119:e907-e919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 303] [Article Influence: 15.9] [Reference Citation Analysis (1)] |
| 3. | Lipska BS, Iatropoulos P, Maranta R, Caridi G, Ozaltin F, Anarat A, Balat A, Gellermann J, Trautmann A, Erdogan O, Saeed B, Emre S, Bogdanovic R, Azocar M, Balasz-Chmielewska I, Benetti E, Caliskan S, Mir S, Melk A, Ertan P, Baskin E, Jardim H, Davitaia T, Wasilewska A, Drozdz D, Szczepanska M, Jankauskiene A, Higuita LM, Ardissino G, Ozkaya O, Kuzma-Mroczkowska E, Soylemezoglu O, Ranchin B, Medynska A, Tkaczyk M, Peco-Antic A, Akil I, Jarmolinski T, Firszt-Adamczyk A, Dusek J, Simonetti GD, Gok F, Gheissari A, Emma F, Krmar RT, Fischbach M, Printza N, Simkova E, Mele C, Ghiggeri GM, Schaefer F; PodoNet Consortium. Genetic screening in adolescents with steroid-resistant nephrotic syndrome. Kidney Int. 2013;84:206-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 67] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 4. | Coppes MJ, Huff V, Pelletier J. Denys-Drash syndrome: relating a clinical disorder to genetic alterations in the tumor suppressor gene WT1. J Pediatr. 1993;123:673-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 63] [Article Influence: 1.9] [Reference Citation Analysis (1)] |
| 5. | Lipska BS, Ranchin B, Iatropoulos P, Gellermann J, Melk A, Ozaltin F, Caridi G, Seeman T, Tory K, Jankauskiene A, Zurowska A, Szczepanska M, Wasilewska A, Harambat J, Trautmann A, Peco-Antic A, Borzecka H, Moczulska A, Saeed B, Bogdanovic R, Kalyoncu M, Simkova E, Erdogan O, Vrljicak K, Teixeira A, Azocar M, Schaefer F; PodoNet Consortium. Genotype-phenotype associations in WT1 glomerulopathy. Kidney Int. 2014;85:1169-1178. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 113] [Article Influence: 9.4] [Reference Citation Analysis (1)] |
| 6. | Pelletier J, Bruening W, Kashtan CE, Mauer SM, Manivel JC, Striegel JE, Houghton DC, Junien C, Habib R, Fouser L. Germline mutations in the Wilms' tumor suppressor gene are associated with abnormal urogenital development in Denys-Drash syndrome. Cell. 1991;67:437-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 681] [Cited by in RCA: 590] [Article Influence: 16.9] [Reference Citation Analysis (1)] |
| 7. | Wagner KD, Wagner N, Schedl A. The complex life of WT1. J Cell Sci. 2003;116:1653-1658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 97] [Article Influence: 4.2] [Reference Citation Analysis (1)] |
| 8. | Nagano C, Nozu K. A review of the genetic background in complicated WT1-related disorders. Clin Exp Nephrol. 2025;29:1-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 10.0] [Reference Citation Analysis (1)] |
| 9. | Barbaux S, Niaudet P, Gubler MC, Grünfeld JP, Jaubert F, Kuttenn F, Fékété CN, Souleyreau-Therville N, Thibaud E, Fellous M, McElreavey K. Donor splice-site mutations in WT1 are responsible for Frasier syndrome. Nat Genet. 1997;17:467-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 511] [Cited by in RCA: 452] [Article Influence: 15.6] [Reference Citation Analysis (1)] |
| 10. | Huff V. Wilms' tumours: about tumour suppressor genes, an oncogene and a chameleon gene. Nat Rev Cancer. 2011;11:111-121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 227] [Cited by in RCA: 199] [Article Influence: 13.3] [Reference Citation Analysis (1)] |
| 11. | Tsuji Y, Yamamura T, Nagano C, Horinouchi T, Sakakibara N, Ishiko S, Aoto Y, Rossanti R, Okada E, Tanaka E, Tsugawa K, Okamoto T, Sawai T, Araki Y, Shima Y, Nakanishi K, Nagase H, Matsuo M, Iijima K, Nozu K. Systematic Review of Genotype-Phenotype Correlations in Frasier Syndrome. Kidney Int Rep. 2021;6:2585-2593. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 12. | Moorthy AV, Chesney RW, Lubinsky M. Chronic renal failure and XY gonadal dysgenesis: "Frasier" syndrome--a commentary on reported cases. Am J Med Genet Suppl. 1987;3:297-302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 44] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 13. | Klamt B, Koziell A, Poulat F, Wieacker P, Scambler P, Berta P, Gessler M. Frasier syndrome is caused by defective alternative splicing of WT1 leading to an altered ratio of WT1 +/-KTS splice isoforms. Hum Mol Genet. 1998;7:709-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 217] [Article Influence: 7.8] [Reference Citation Analysis (1)] |
| 14. | Guaragna MS, Lutaif AC, Bittencourt VB, Piveta CS, Soardi FC, Castro LC, Belangero VM, Maciel-Guerra AT, Guerra-Junior G, Mello MP. Frasier syndrome: four new cases with unusual presentations. Arq Bras Endocrinol Metabol. 2012;56:525-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 15. | Dattolo P, Allinovi M, Iatropoulos P, Michelassi S. Atypical clinical presentation of a WT1-related syndrome associated with a novel exon 6 gene mutation. BMJ Case Rep. 2013;2013:bcr2013009543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.2] [Reference Citation Analysis (1)] |
| 16. | Ahn YH, Park EJ, Kang HG, Kim SH, Cho HY, Shin JI, Lee JH, Park YS, Kim KS, Ha IS, Cheong HI. Genotype-phenotype analysis of pediatric patients with WT1 glomerulopathy. Pediatr Nephrol. 2017;32:81-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (1)] |
| 17. | Akramov NR, Shavaliev RF, Osipova IV. New mutation in WT1 gene in a boy with an incomplete form of Denys-Drash syndrome: A CARE-compliant case report. Medicine (Baltimore). 2021;100:e25864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 18. | Wang F, Cai J, Wang J, He M, Mao J, Zhu K, Zhao M, Guan Z, Li L, Jin H, Shu Q. A novel WT1 gene mutation in a chinese girl with denys-drash syndrome. J Clin Lab Anal. 2021;35:e23769. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 19. | Zhang B, Ding Y, Ren X, Song C, Zhang X, Wang F, Yang X. Congenital diaphragmatic eventration with pulmonary dysplasia in Frasier syndrome due to a WT1 mutation of c.1432+5(IVS9)G>A. Eur J Med Genet. 2022;65:104655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 20. | Arroyo-Parejo Drayer P, Seeherunvong W, Katsoufis CP, DeFreitas MJ, Seeherunvong T, Chandar J, Abitbol CL. Spectrum of Clinical Manifestations in Children With WT1 Mutation: Case Series and Literature Review. Front Pediatr. 2022;10:847295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 21. | Shao Q, Xie X, Geng J, Yang X, Li W, Zhang Y. Frasier Syndrome: A 15-Year-Old Phenotypically Female Adolescent Presenting with Delayed Puberty and Nephropathy. Children (Basel). 2023;10:577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 22. | Al Zabali S, Alseneidi S, Faqeehi H, Albatati S, Al Anazi A. Association of Atypical Hemolytic Uremic Syndrome With Wilms' Tumor 1 Gene Mutations: A Case Series and Literature Review. Cureus. 2024;16:e70016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 23. | Tran Thuy HQ, Nguyen TTT, Fukui K, Nagano C, Nozu K, Hamada R, Tsukaguchi H. Severe Kidney Dysplastic Phenotypes Caused by WT1 Exon 9 Missense Variants. J Am Soc Nephrol. 2024;35. [DOI] [Full Text] |
| 24. | Chan WK, To KF, But WM, Lee KW. Frasier syndrome: a rare cause of delayed puberty. Hong Kong Med J. 2006;12:225-227. [PubMed] |
| 25. | Voskarides K, Damianou L, Neocleous V, Zouvani I, Christodoulidou S, Hadjiconstantinou V, Ioannou K, Athanasiou Y, Patsias C, Alexopoulos E, Pierides A, Kyriacou K, Deltas C. COL4A3/COL4A4 mutations producing focal segmental glomerulosclerosis and renal failure in thin basement membrane nephropathy. J Am Soc Nephrol. 2007;18:3004-3016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 161] [Article Influence: 8.5] [Reference Citation Analysis (1)] |
| 26. | Gibson J, Fieldhouse R, Chan MMY, Sadeghi-Alavijeh O, Burnett L, Izzi V, Persikov AV, Gale DP, Storey H, Savige J; Genomics England Research Consortium. Prevalence Estimates of Predicted Pathogenic COL4A3-COL4A5 Variants in a Population Sequencing Database and Their Implications for Alport Syndrome. J Am Soc Nephrol. 2021;32:2273-2290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 158] [Article Influence: 31.6] [Reference Citation Analysis (1)] |
| 27. | Rey RA, Josso N. Diagnosis and Treatment of Disorders of Sexual Development. Endocrinology: Adult and Pediatric, 2016. [DOI] [Full Text] |
| 28. | McTaggart SJ, Algar E, Chow CW, Powell HR, Jones CL. Clinical spectrum of Denys-Drash and Frasier syndrome. Pediatr Nephrol. 2001;16:335-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 58] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 29. | Huang YC, Tsai MC, Tsai CR, Fu LS. Frasier Syndrome: A Rare Cause of Refractory Steroid-Resistant Nephrotic Syndrome. Children (Basel). 2021;8:617. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 6] [Reference Citation Analysis (1)] |
| 30. | Chernin G, Vega-Warner V, Schoeb DS, Heeringa SF, Ovunc B, Saisawat P, Cleper R, Ozaltin F, Hildebrandt F; Members of the GPN Study Group. Genotype/phenotype correlation in nephrotic syndrome caused by WT1 mutations. Clin J Am Soc Nephrol. 2010;5:1655-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 85] [Article Influence: 5.3] [Reference Citation Analysis (1)] |
| 31. | Popa L, Balgradean M, Croitoru A. Long-Term Study in Children with Steroid-Resistant Nephrotic Syndrome Progressing to End-Stage Renal Disease. Maedica (Bucur). 2022;17:271-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |