Published online Mar 25, 2026. doi: 10.5527/wjn.v15.i1.113474
Revised: September 23, 2025
Accepted: January 7, 2026
Published online: March 25, 2026
Processing time: 199 Days and 21.8 Hours
Kidney transplantation is a moderate to high-risk surgery, and postoperative outcome is closely related to good intraoperative fluid management. Suboptimal fluid management is adversely associated with poor graft and recipient outcomes. Hence, meticulous intraoperative fluid management is essential and requires in-depth knowledge of newly added fluids and hemodynamic tools for fluid monitoring. Over the years, fluid management during kidney transplant has significantly changed with the inclusion of newer fluids and dynamic indices of volume responsiveness. In this review, we will discuss the different types of fluids and hemodynamic tools available for perioperative fluid management in kidney transplantation and their pros and cons on postoperative outcomes.
Core Tip: Fluid therapy is one of the core domains of intraoperative anaesthesia management in kidney transplantation. Newer advancements in recent years have changed the outlook of intraoperative hemodynamic monitoring, resulting in better transplant outcomes. In this review, we focused on discussing newer indices of fluid responsiveness and their utility in kidney transplant surgery, as well as newer crystalloids and colloids and their impact on metabolic profile and post-transplant graft outcomes.
- Citation: Goyal VK, Shekhrajka P, Bhargava SK, Mittal S. Perioperative fluid management in kidney transplantation: What’s new and future directives? World J Nephrol 2026; 15(1): 113474
- URL: https://www.wjgnet.com/2220-6124/full/v15/i1/113474.htm
- DOI: https://dx.doi.org/10.5527/wjn.v15.i1.113474
Fluid therapy is an essential part of perioperative anaesthetic management for intermediate to high-risk surgeries, including cardiac, transplant, polytrauma, major abdominal, and vascular. Optimal fluid management in the perioperative period is required for enhanced patient recovery, as it is closely associated with postoperative surgical outcomes. Euvolemia is highly desired, as improper fluid therapy by means of the wrong fluid type or poor monitoring guidance may lead to adverse consequences related to both hypovolemia and hypervolemia. Hypovolemia increases the risk of organ hypoperfusion, hypotension, lactic acidosis, increased need for vasopressors, infections, and organ failure, while hypervolemia enhances the risk of cardiopulmonary complications, tissue oedema, poor wound healing, postoperative ileus, mechanical ventilation, and coagulopathy[1,2].
In kidney transplantation, postoperative graft function and patient recovery depend on perioperative fluid management; hence, meticulous fluid therapy is highly recommended. In this review, we highlighted the different aspects of fluid therapy, including types of fluids (crystalloids vs colloids), monitoring tools, and hemodynamic targets during kidney transplantation based on recent literature.
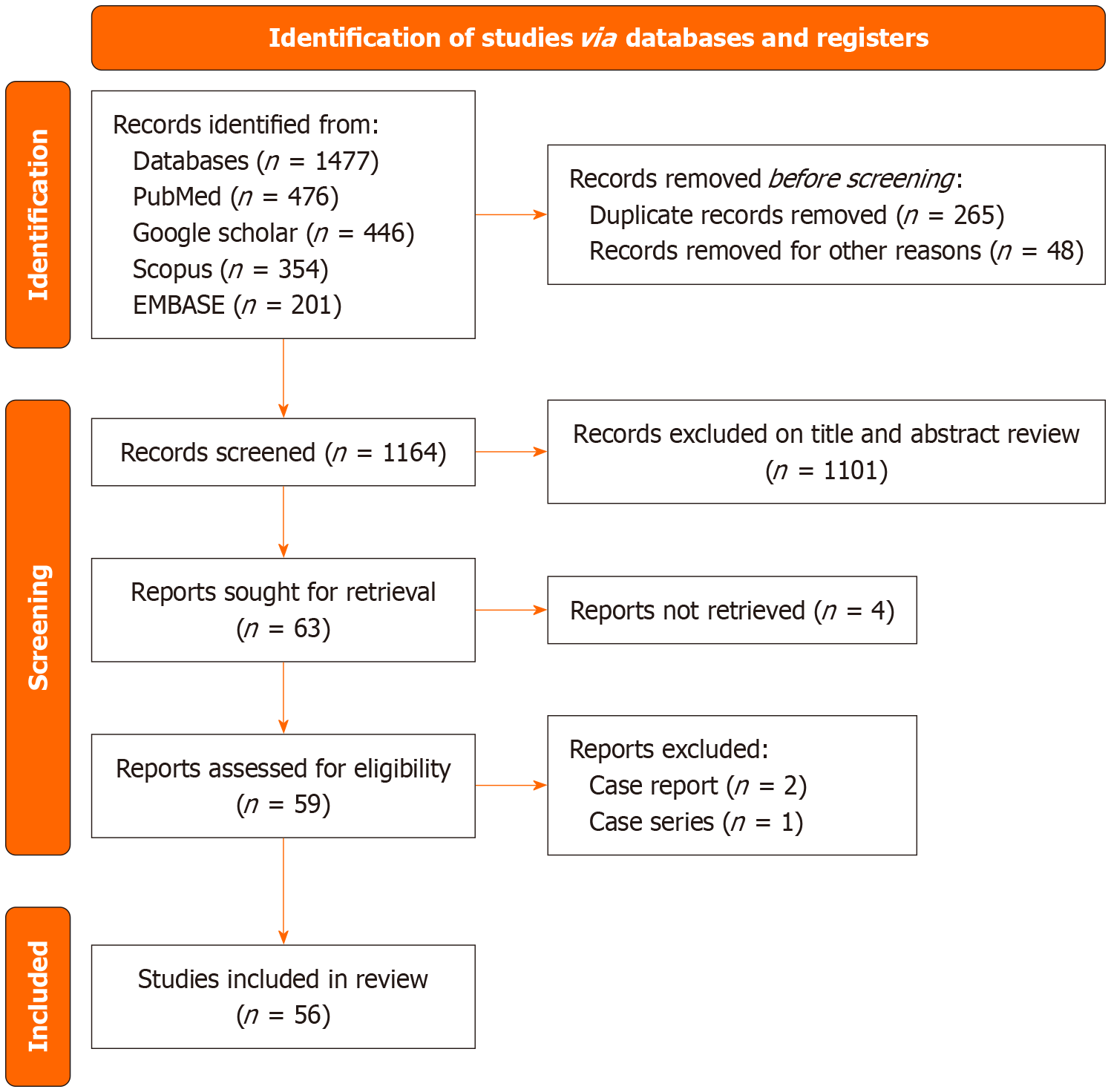
The medical literatures were searched for data using search engines (PubMed, Scopus, Google Scholar, and Cochrane Library) for recent practices in perioperative fluid management during kidney transplantation. The key terms used for search included (kidney transplant OR kidney transplantation OR renal transplant OR renal transplantation) AND (normal saline OR lactated ringer OR plasmaLyte A OR hydroxyethyl starch OR albumin OR central venous pressure OR mean arterial pressure OR pulse pressure variation OR stroke volume variation OR transoesophageal doppler OR perioperative goal directed fluid therapy OR floTrac OR hypotension prediction index OR delayed graft function). We included data from randomized controlled trials and retrospective studies in this review and excluded the case reports, case studies, review articles, meta-analysis, and protocols observing simultaneous multiple interventions and outcomes in perioperative period as shown in Figure 1.
Primary outcome included was delayed graft function (DGF) while urine output, change in postoperative creatinine level, requirements of intraoperative fluids, metabolic changes (serum electrolytes and acid/base), and postoperative complications were as secondary outcomes.
The levels of evidence and strength of recommendations were determined using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework. Evidence quality was classified as high, moderate, low, or very low, based on the available literature. Generally, multiple randomized controlled trials with consistent outcomes or systematic reviews were rated as high quality, whereas observational studies, particularly retrospective ones, were considered low or very low quality. The strength of recommendations was graded as strong or weak, depending on whether they supported or opposed an intervention. Strong recommendations were given when benefits clearly outweighed risks (or vice versa), while weak recommendations were made when evidence was limited and decisions needed to be individualized.
Postoperative graft and recipient outcomes are affected by the type of fluid used and should be considered in anaesthesia planning beforehand. However, the selection of fluids (crystalloids or colloids) during kidney transplantation is highly controversial, and practices vary among anaesthesiologists.
Normal saline, Ringer lactate, and low chloride solutions are commonly used crystalloids in kidney transplantation and have differences in solute contents. Normal saline is a potassium-free crystalloid and has been considered an ideal fluid during kidney transplantation by anaesthesiologists since decades, but recent evidence discourages intraoperative use of normal saline in large volumes, as it is associated with increased risk of hyperchloremic metabolic acidosis due to supraphysiologic levels of chloride (154 mmol/L) compared to plasma (96-108 mmol/L)[3-7]. Hyperchloremic metabolic acidosis eventually causes hyperkalaemia by outward movement of K+ ions from the cells, afferent renal vasoconstriction mediated through tubulo-glomerular feedback mechanism related to delivery of high chloride to distal tubules, reduced glomerular filtration rate (GFR), urine production, acute kidney injury (AKI), DGF, requirement of renal replacement therapy[8-12]. Balanced low chloride crystalloids (e.g., Kabilyte, Plasma-Lyte A, and Physiomax) are isotonic to plasma and contain chloride in physiological concentration to plasma (98 mmol/L vs 96-108 mmol/L) and acetate as a buffer[13-17]. In kidney transplantation, numerous studies have concluded that balanced low chloride solutions reduce the risk of hyperchloremia, hyperkalaemia, metabolic acidosis, AKI, and DGF as compared to normal saline. However, balanced low chloride solutions have not shown any benefit in terms of vasopressor requirement, length of intensive care unit stay, and postoperative creatinine level[18-22].
Ringer lactate is a slightly hypotonic crystalloid containing lactate as a buffer and potassium (4 mmol/L) with an inherent theoretical risk of lactic acidosis and hyperkalaemia. But recent studies have shown favourable metabolic outcomes as compared to 0.9% saline with less metabolic acidosis and hyperkalaemia in kidney transplantation[23-25].
Recommendations: Based on recent evidence, balanced low chloride solutions are strongly recommended for kidney transplantation and must be considered as crystalloids of choice in perioperative periods (GRADE high-quality of evidence, strong recommendation).
The use of intraoperative colloids (starches, gelatin, and albumin) in kidney transplantation has many controversies among perioperative physicians and area of debate. There is very scarce evidence in support of perioperative use of colloids.
Colloids containing starches have shown increased risk of kidney injury, need for renal replacement therapy, and coagulation abnormalities in critically ill patients[26,27]. The proposed mechanisms of kidney injury include osmotic nephrosis-like lesions, cytoplasmic vacuolisation, and tubular swelling by tissue uptake of starches[28,29]. Administration of hydroxyethyl starches (HES) in brain-dead donor management is associated with increased incidence of DGF in kidney transplant recipients due to effects on graft quality[30-32]. However, studies on recipients during kidney transplant did not reveal any significant negative impact on postoperative kidney function as compared to crystalloids and gelatin[33,34].
Intravenous administration of human albumin plays potential roles, including increasing plasma oncotic pressure, antioxidant effects, immunomodulation, protection from ischaemia-reperfusion injury, and decreasing the requirement of crystalloids and vasopressors[35,36]. However, studies have shown either no benefit or an increased risk of postoperative complications, pulmonary oedema, and mortality. Also, human albumin is associated with increased risk of disease transmission, immunogenicity, and high cost[37-40]. In kidney transplantation, 20% albumin does not have any advantage on early graft functions as compared to crystalloids[41,42]. Table 1 outlines different types of intravenous fluids used during kidney transplantation.
| Type of fluid | Pros | Cons |
| Normal saline | Less expensive, easily available, potassium free | Potentially hypertonic, Risk of hyperchloremic metabolic acidosis, hyperkalemia, decreased GFR and urine output, AKI, and DGF large volume infusion should be avoided |
| Low chloride solutions (e.g., Kabilyte, Plasm-Lyte A) | Physiologically like plasma, no risk of hyperkalemia, metabolic acidosis, and DGF. Fluid of choice during kidney transplant | More expensive than other crystalloids |
| Ringer lactate | Balanced crystalloid, isotonic, has buffering capacity, is cost-effective, and safe | Theoretical risk of lactic acidosis and hyperkalemia |
| Hydroxyethyl starch | Volume expander, easily available | Risk of AKI, renal replacement therapy, and coagulopathy |
| Albumin | Increases plasma oncotic pressure, antioxidant properties, immunomodulation, protection from ischemia-reperfusion injury, less requirement of crystalloids, and vasopressors | Increased cost, risk of allergic reactions, and disease transmission. No benefit or increased risk of volume-related complications in recipients with cardio-pulmonary compromise. Routine use is not recommended |
Recommendations: Recent evidence suggests no benefit of routine use of albumin in kidney transplantation and use should be individualized based on serum albumin level (GRADE moderate-quality evidence, weak recommendation). Studies on recipients did not reveal any significant negative impact of HES on postoperative kidney function. Still, routine use of HES is not recommended for kidney transplantation and should be avoided in the perioperative period (GRADE moderate-quality of evidence, weak recommendation).
Haemodynamic monitoring is the cornerstone of fluid therapy in kidney transplantation to maintain optimal volume status and for excellent postoperative graft function. Suboptimal fluid management (hypovolemia or hypervolemia) has detrimental effects on postoperative graft and recipient outcomes. Excessive volume administration may cause tissue oedema, pulmonary oedema, the requirement of mechanical ventilation, dilutional coagulopathy, poor wound healing, postoperative ileus, and/or multi-organ dysfunction, while inadequate fluid volume results in hypotension, increased need for inotropes or vasopressors, metabolic acidosis, poor perfusion to the grafted kidney, AKI, and DGF. In the initial days of kidney transplantation, fluid administration was considered till no further response, but supranormal volume loading is not warranted in recent studies[43,44]. Over the decades, a lot has changed in perioperative fluid monitoring, and it is discussed here one by one.
Older studies have recommended monitoring of central venous pressure (CVP) to assess volume responsiveness during kidney transplantation to guide intraoperative fluids, but there were no uniformities among authors for upper and lower CVP targets, and it ranged between 1-2 kPa[44-46]. CVP is a static parameter and poorly predicts volume status. It has many limitations, including complications associated with central venous catheters (injury to major vessels, haematoma, haemothorax, infection, difficult cannulation in kidney recipients, etc.) and erroneous results (valvular regurgitation, pulmonary hypertension, right heart disease, etc.). Recent studies have not shown any advantages of CVP monitoring on post-transplant renal functions; hence, routine use of CVP for fluid management during kidney transplantation is highly discouraged due to the availability of newer and better haemodynamic tools to assess volume responsiveness[47-50].
Recommendations: Recent evidence suggested that there are no advantages of CVP monitoring on post-transplant renal functions; hence, routine use of CVP for fluid management during kidney transplantation is not recommended (GRADE moderate-quality evidence, strong recommendation).
Pulse pressure variation (PPV) is a dynamic index of volume responsiveness derived from cardiorespiratory interaction. It is a minimally invasive and easily measured parameter of volume responsiveness. Recent evidence has shown that postoperative renal function is better in recipients who received fluid by PPV guidance, with a lower incidence of DGF in comparison to CVP. Also, the intraoperative need for crystalloids is significantly less[51-53]. Suggested targets for PPV were between 6% to 15%, but upper and lower limits varied among authors. Similarly, stroke volume variation (SVV) predicts better preload status in comparison to CVP during kidney transplantation and has a good correlation to change in cardiac index[54-56]. High risk kidney transplant recipients like patients with dilated cardiomyopathy (ejection fraction < 40%), coronary artery disease, severe pulmonary hypertension etc. have low safety of margin and prone to higher fluid related complications. Goyal et al[57] in their retrospective study in patients with dilated cardiomyopathy observed decreased incidence of immediate post-transplant complication when fluids were guided using SVV as compared to CVP[56,57]. These dynamic indices are easy to interpret and guide fluids, as fluid is stopped once the value is below the lower limit, and a fluid bolus (250-500 mL of crystalloid or colloid) is infused when the upper limit is crossed. A few limitations, including low tidal volume ventilation (< 8 mL/kg), spontaneous ventilation, and arrhythmias, must be considered during PPV or SVV-guided fluid therapy. Currently, lung protective ventilation (< 8 mL/kg) is no more of a limitation with the use of SVV or PPV if the tidal volume challenge strategy is considered during kidney transplantation[58].
Less studied dynamic indices during kidney transplantation are stroke volume and Transoesophageal Doppler (TED), and have also shown promising results. Perioperative goal-directed fluid therapy with stroke volume optimization protocol effectively reduces the postoperative complications and DGF[59]. TED-guided fluid therapy during kidney transplantation has also shown a reduced requirement of intraoperative fluids along with decreased fluid-related postoperative complications, including postoperative dyspnoea and tissue oedema, compared to CVP[60].
Recommendations: Recent evidence has shown that postoperative renal function is better in recipients who received fluid by PPV/SVV guidance, with a lower incidence of DGF in comparison to CVP (GRADE high-quality evidence, strong recommendation).
Stroke volume optimization and TED have effectively reduced the DGF and fluid related postoperative complications (GRADE low-quality evidence, weak recommendation).
Intraoperative hypotension (IOH) during kidney transplant is closely associated with poor postoperative graft outcomes, including AKI, DGF, increased incidence of rejection, and poor long-term graft survival. Monitoring systemic arterial pressure is vital, as perfusion of the renal graft largely depends on pressure due to loss of autoregulation by ischaemia-reperfusion injury and denervation of the explanted kidney. Post-ischaemic tissue oedema can further deteriorate the perfusion of the graft[61]. Hence, higher mean arterial pressure (MAP) targets are necessary for adequate perfusion of the graft, and these targets are widely affected by donor (age, live/brain dead/donation after cardiac death, hypertensive, number of vessels) and recipient characteristics (preoperative blood pressure, dialysis, comorbidities, etc.). Exposure to MAP below the threshold (magnitude as well as the duration) is directly related to the increased incidence of DGF. Intraoperative target MAP varies among institutions; however, the MAP ≥ 11 kPa after reperfusion is generally recommended. Perioperative use of vasopressors (preferred agents and dosing) to increase MAP to the target level is not well studied, and further work is needed to find optimal strategies. However, large doses of vasopressors should be avoided due to the inherent risk of intrarenal vasoconstriction and impaired graft perfusion[62,63].
MAP at reperfusion is advocated to be related to early recovery of graft function, but Oh et al[64], in a retrospective study, concluded that high baseline MAP is directly associated with early recovery of eGFR and urine output[64-66]. Also, the practice of arterial pressure monitoring, either invasive or non-invasive, depends on the anaesthesiologist's discretion and is a subject of controversy. Table 2 outlines the intraoperative haemodynamic monitoring to assess fluid responsiveness.
| Hemodynamic indices | Advantages and targets | Limitations |
| Central venous pressure | Relatively cheap, measures right atrial pressure to predict volume status 1-2 kPa, upper and lower limits vary among institutions | Static parameter, complications associated with central venous cannulation (injury to major vessels, haematoma, and infection, etc.), erroneous results in PAH, valvular heart disease, and pulmonary disease, etc., poorly predict volume status. No benefit in post-transplant renal function recovery |
| Pulse pressure variation/stroke volume variation | Dynamic indices, minimally invasive, easy to interpret, and high accuracy. Decreased incidence of DGF in postoperative periods. 6%-15% varies among institutions | Confounding factors: Low tidal volume (< 8 mL/kg), spontaneous ventilation, high PEEP, and arrhythmias |
| Stroke volume | Dynamic index, minimally invasive, easy to interpret, and more accurate. Reduced incidence of DGF in postoperative periods% change in stroke volume (</> 10%) | Confounding factors: Low tidal volume (< 8 mL/kg), spontaneous ventilation, high PEEP, and arrhythmias |
| Transoesophageal Doppler | Dynamic index, non-invasive, continuous, real-time, and more accurate reduces requirements of intraoperative fluids along with fluid-related complications (tissue edema, dyspnoea) | Equipment cost, operator dependent, requires training, risk of oesophageal injury |
| Invasive blood pressure | Beat-to-beat monitoring of arterial pressure, post reperfusion MAP ≥ 11 kPa, varies among institutions | Difficult arterial line placement due to AV fistula or objected by treating physicians, risk of infection, hematoma, etc. No specified targets, depends on donor and recipient’s characteristics (age, co-morbidities, and type of donation) |
Recommendations: Recent evidence have shown that higher MAP (≥ 11 kPa) is necessary for adequate perfusion of the graft (GRADE high-quality evidence, strong recommendation).
As discussed, intraoperative fluid therapy is a key to good postoperative transplant outcomes. Over the decades, continuous evolutions in haemodynamic monitoring and incorporation of newer fluid compositions have drastically changed the graft and recipient survival. Continuous beat-to-beat arterial pressure monitoring is pivotal to detecting hypotension and taking reactive measures, but placement of an arterial cannula is challenging due to the presence of an AV fistula and is sometimes objected to by nephrologists. Kakuta et al[67] reported successful use of non-invasive continuous arterial pressure monitoring using ClearSight in kidney transplant recipients. Further studies with large numbers of patients are required to support its use in routine practice. Recently, the role of artificial intelligence in pe
Early recovery of graft function largely depends on close adherence to intraoperative blood pressure targets. IOH requires urgent measures to hasten its impact on graft functioning, but the ideal strategy (fluid or vasopressors) to combat hypotension is not well defined. Also, the use of vasopressors during transplant is debatable due to their effect on renal perfusion and necessitates further studies to choose the ideal agent and dosing regimen.
In conclusion, perioperative fluid management during kidney transplantation is an important concern for the transplant team. Meticulous fluid therapy based on recent evidence and with the inclusion of newer fluids and haemodynamic tools will be helpful to improve postoperative outcomes further.
| 1. | Ostermann M, Auzinger G, Grocott M, Morton-Bailey V, Raphael J, Shaw AD, Zarbock A; POQI XI Investigators. Perioperative fluid management: evidence-based consensus recommendations from the international multidisciplinary PeriOperative Quality Initiative. Br J Anaesth. 2024;133:1263-1275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 2. | Makaryus R, Miller TE, Gan TJ. Current concepts of fluid management in enhanced recovery pathways. Br J Anaesth. 2018;120:376-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 111] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 3. | Chang Y, Qin Y, Zou Y, Zeng H, Li C, Qin M, Wu J, Ban J. Plasma-lyte solution versus saline in kidney transplantation: A systematic review and meta-analysis of randomized controlled trials. PLoS One. 2025;20:e0320082. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 4. | Chang T, Shih MC, Wu YL, Wu TT, Yang JT, Wu CY. Comparative efficacy of balanced crystalloids versus 0.9% saline on delayed graft function and perioperative outcomes in kidney transplantation: a meta-analysis of randomised controlled trials. Br J Anaesth. 2024;133:1173-1182. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Carvalho Pereira L, Carvalho Pereira I, Dias Delfino Cabral T, Viana P, Mendonça Ribeiro A, Amaral S. Balanced Crystalloids Versus Normal Saline in Kidney Transplant Patients: An Updated Systematic Review, Meta-analysis, and Trial Sequential Analysis. Anesth Analg. 2024;139:58-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 6. | Collins MG, Fahim MA, Pascoe EM, Hawley CM, Johnson DW, Varghese J, Hickey LE, Clayton PA, Dansie KB, McConnochie RC, Vergara LA, Kiriwandeniya C, Reidlinger D, Mount PF, Weinberg L, McArthur CJ, Coates PT, Endre ZH, Goodman D, Howard K, Howell M, Jamboti JS, Kanellis J, Laurence JM, Lim WH, McTaggart SJ, O'Connell PJ, Pilmore HL, Wong G, Chadban SJ; BEST-Fluids Investigators; Australasian Kidney Trials Network. Balanced crystalloid solution versus saline in deceased donor kidney transplantation (BEST-Fluids): a pragmatic, double-blind, randomised, controlled trial. Lancet. 2023;402:105-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 64] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 7. | Medeiros H, Lima PH, Junior VS, Souza DA, Pinheiro AM, Martins RR, A H Costa KM, Junior JHD, Medeiros PJ, Da Silva WA Sr. A Comparison Between Saline and Balanced Solutions in Kidney Transplants: A Randomized Clinical Trial. Cureus. 2023;15:e49813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 8. | Kolodzie K, Cakmakkaya OS, Boparai ES, Tavakol M, Feiner JR, Kim MO, Newman TB, Niemann CU. Perioperative Normal Saline Administration and Delayed Graft Function in Patients Undergoing Kidney Transplantation: A Retrospective Cohort Study. Anesthesiology. 2021;135:621-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 9. | do Nascimento Junior P, Dohler LE, Ogawa CMU, de Andrade LÍGM, Braz LG, M Dolo NSP. Effects of Plasma-Lyte. ½ and 0.9% saline in renal function after deceased-donor kidney transplant: a randomized controlled trial. Braz J Anesthesiol. 2022;72:711-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Wagener G, Bezinover D, Wang C, Kroepfl E, Diaz G, Giordano C, West J, Kindscher JD, Moguilevitch M, Nicolau-Raducu R, Planinsic RM, Rosenfeld DM, Lindberg S, Schumann R, Pivalizza EG. Fluid Management During Kidney Transplantation: A Consensus Statement of the Committee on Transplant Anesthesia of the American Society of Anesthesiologists. Transplantation. 2021;105:1677-1684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 11. | Nesseler N, Rached A, Ross JT, Launey Y, Vigneau C, Bensalah K, Beloeil H, Mallédant Y, Garlantezec R, Seguin P. Association between perioperative normal saline and delayed graft function in deceased-donor kidney transplantation: a retrospective observational study. Can J Anaesth. 2020;67:421-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Rein JL, Coca SG. "I don't get no respect": the role of chloride in acute kidney injury. Am J Physiol Renal Physiol. 2019;316:F587-F605. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 63] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 13. | González-Castro A, Ortiz-Lasa M, Rodriguez-Borregan JC, Rodrigo Calabia E, Ruiz San Millan JC, Dierssen Soto T. Influence of Proportion of Normal Saline Administered in the Perioperative Period of Renal Transplantation on Kalemia Levels. Transplant Proc. 2018;50:569-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Calixto Fernandes MH, Schricker T, Magder S, Hatzakorzian R. Perioperative fluid management in kidney transplantation: a black box. Crit Care. 2018;22:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 15. | Pfortmueller C, Funk GC, Potura E, Reiterer C, Luf F, Kabon B, Druml W, Fleischmann E, Lindner G. Acetate-buffered crystalloid infusate versus infusion of 0.9% saline and hemodynamic stability in patients undergoing renal transplantation: Prospective, randomized, controlled trial. Wien Klin Wochenschr. 2017;129:598-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 13] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 16. | Gonzalez-Castro A, Ortiz-Lasa M, Peñasco Y, González C, Blanco C, Rodriguez-Borregan JC. Choice of fluids in the perioperative period of kidney transplantation. Nefrología. 2017;37:572-578. [DOI] [Full Text] |
| 17. | Weinberg L, Harris L, Bellomo R, Ierino FL, Story D, Eastwood G, Collins M, Churilov L, Mount PF. Effects of intraoperative and early postoperative normal saline or Plasma-Lyte 148® on hyperkalaemia in deceased donor renal transplantation: a double-blind randomized trial. Br J Anaesth. 2017;119:606-615. [PubMed] [DOI] [Full Text] |
| 18. | Adwaney A, Randall DW, Blunden MJ, Prowle JR, Kirwan CJ. Perioperative Plasma-Lyte use reduces the incidence of renal replacement therapy and hyperkalaemia following renal transplantation when compared with 0.9% saline: a retrospective cohort study. Clin Kidney J. 2017;10:838-844. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 36] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Wan S, Roberts MA, Mount P. Normal saline versus lower-chloride solutions for kidney transplantation. Cochrane Database Syst Rev. 2016;2016:CD010741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Potura E, Lindner G, Biesenbach P, Funk GC, Reiterer C, Kabon B, Schwarz C, Druml W, Fleischmann E. An acetate-buffered balanced crystalloid versus 0.9% saline in patients with end-stage renal disease undergoing cadaveric renal transplantation: a prospective randomized controlled trial. Anesth Analg. 2015;120:123-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 21. | Kim SY, Huh KH, Lee JR, Kim SH, Jeong SH, Choi YS. Comparison of the effects of normal saline versus Plasmalyte on acid-base balance during living donor kidney transplantation using the Stewart and base excess methods. Transplant Proc. 2013;45:2191-2196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 22. | Hadimioglu N, Saadawy I, Saglam T, Ertug Z, Dinckan A. The effect of different crystalloid solutions on acid-base balance and early kidney function after kidney transplantation. Anesth Analg. 2008;107:264-269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 156] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 23. | O'Malley CMN, Frumento RJ, Hardy MA, Benvenisty AI, Brentjens TE, Mercer JS, Bennett-Guerrero E. A randomized, double-blind comparison of lactated Ringer's solution and 0.9% NaCl during renal transplantation. Anesth Analg. 2005;100:1518-1524. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 238] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 24. | Khajavi MR, Etezadi F, Moharari RS, Imani F, Meysamie AP, Khashayar P, Najafi A. Effects of normal saline vs. lactated ringer's during renal transplantation. Ren Fail. 2008;30:535-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 25. | Tontu F. Fluid Selection in Renal Transplant Patients: Considerations for Hyperkalemia Management. Turk J Anaesthesiol Reanim. 2025;53:138-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 26. | Westphal M, James MF, Kozek-Langenecker S, Stocker R, Guidet B, Van Aken H. Hydroxyethyl starches: different products--different effects. Anesthesiology. 2009;111:187-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 307] [Cited by in RCA: 231] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 27. | Mutter TC, Ruth CA, Dart AB. Hydroxyethyl starch (HES) versus other fluid therapies: effects on kidney function. Cochrane Database Syst Rev. 2013;2013:CD007594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 89] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 28. | Vuiblet V, Nguyen TT, Wynckel A, Fere M, Van-Gulick L, Untereiner V, Birembaut P, Rieu P, Piot O. Contribution of Raman spectroscopy in nephrology: a candidate technique to detect hydroxyethyl starch of third generation in osmotic renal lesions. Analyst. 2015;140:7382-7390. [PubMed] [DOI] [Full Text] |
| 29. | Dickenmann M, Oettl T, Mihatsch MJ. Osmotic nephrosis: acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis. 2008;51:491-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 199] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 30. | Cittanova ML, Leblanc I, Legendre C, Mouquet C, Riou B, Coriat P. Effect of hydroxyethylstarch in brain-dead kidney donors on renal function in kidney-transplant recipients. Lancet. 1996;348:1620-1622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 370] [Cited by in RCA: 277] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 31. | Patel MS, Niemann CU, Sally MB, De La Cruz S, Zatarain J, Ewing T, Crutchfield M, Enestvedt CK, Malinoski DJ. The Impact of Hydroxyethyl Starch Use in Deceased Organ Donors on the Development of Delayed Graft Function in Kidney Transplant Recipients: A Propensity-Adjusted Analysis. Am J Transplant. 2015;15:2152-2158. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 32. | Vuiblet V, Fere M, Bankole E, Wynckel A, Gobinet C, Birembaut P, Piot O, Rieu P. Raman-based detection of hydroxyethyl starch in kidney allograft biopsies as a potential marker of allograft quality in kidney transplant recipients. Sci Rep. 2016;6:33045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 33. | Hokema F, Ziganshyna S, Bartels M, Pietsch UC, Busch T, Jonas S, Kaisers U. Is perioperative low molecular weight hydroxyethyl starch infusion a risk factor for delayed graft function in renal transplant recipients? Nephrol Dial Transplant. 2011;26:3373-3378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 34. | Wu Y, Wu AS, Wang J, Tian M, Jia XY, Rui Y, Yue Y. Effects of the novel 6% hydroxyethyl starch 130/0.4 on renal function of recipients in living-related kidney transplantation. Chin Med J (Engl). 2010;123:3079-3083. [PubMed] |
| 35. | Severs D, Hoorn EJ, Rookmaaker MB. A critical appraisal of intravenous fluids: from the physiological basis to clinical evidence. Nephrol Dial Transplant. 2015;30:178-187. [PubMed] [DOI] [Full Text] |
| 36. | Bernardi M, Angeli P, Claria J, Moreau R, Gines P, Jalan R, Caraceni P, Fernandez J, Gerbes AL, O'Brien AJ, Trebicka J, Thevenot T, Arroyo V. Albumin in decompensated cirrhosis: new concepts and perspectives. Gut. 2020;69:1127-1138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 255] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 37. | Wiedermann CJ. Human Albumin Infusion in Critically Ill and Perioperative Patients: Narrative Rapid Review of Meta-Analyses from the Last Five Years. J Clin Med. 2023;12:5919. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 38. | Lewis SR, Pritchard MW, Evans DJ, Butler AR, Alderson P, Smith AF, Roberts I. Colloids versus crystalloids for fluid resuscitation in critically ill people. Cochrane Database Syst Rev. 2018;8:CD000567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 112] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 39. | Zou Y, Ma K, Xiong JB, Xi CH, Deng XJ. Comparison of the effects of albumin and crystalloid on mortality among patients with septic shock: systematic review with meta-analysis and trial sequential analysis. Sao Paulo Med J. 2018;136:421-432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 40. | Lazzareschi DV, Fong N, Mavrothalassitis O, Whitlock EL, Chen CL, Chiu C, Adelmann D, Bokoch MP, Chen LL, Liu KD, Pirracchio R, Mathis MR, Legrand M; MPOG Collaborators. Intraoperative Use of Albumin in Major Noncardiac Surgery: Incidence, Variability, and Association With Outcomes. Ann Surg. 2023;278:e745-e753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 41. | Abdallah E, El-Shishtawy S, Mosbah O, Zeidan M. Comparison between the effects of intraoperative human albumin and normal saline on early graft function in renal transplantation. Int Urol Nephrol. 2014;46:2221-2226. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 42. | Shah RB, Shah VR, Butala BP, Parikh GP. Effect of intraoperative human albumin on early graft function in renal transplantation. Saudi J Kidney Dis Transpl. 2014;25:1148-1153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 43. | Carlier M, Squifflet JP, Pirson Y, Decocq L, Gribomont B, Alexandre GP. Confirmation of the crucial role of the recipient's maximal hydration on early diuresis of the human cadaver renal allograft. Transplantation. 1983;36:455-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 44. | De Gasperi A, Narcisi S, Mazza E, Bettinelli L, Pavani M, Perrone L, Grugni C, Corti A. Perioperative fluid management in kidney transplantation: is volume overload still mandatory for graft function? Transplant Proc. 2006;38:807-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 45. | Bacchi G, Buscaroli A, Fusari M, Neri L, Cappuccilli ML, Carretta E, Stefoni S. The influence of intraoperative central venous pressure on delayed graft function in renal transplantation: a single-center experience. Transplant Proc. 2010;42:3387-3391. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 46. | Goyal VK, Mittal S, Shekhrajka P. Unlocking Insights: A Survey on Perioperative Hemodynamic Monitoring in Kidney Transplantation Across India. Indian J Transplant. 2025;19:214-217. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 47. | Collange O, Tacquard C, Oulehri W, Biehler J, Moulin B, Mertes PM, Lejay A, Caillard S. Hemodynamic Management During Kidney Transplantation: A French Survey. Transplant Proc. 2021;53:1450-1453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | Adelmann D, Bicknell L, Niemann CU, Feiner J, Roll GR, Burdine L, Whitlock EL. Central venous pressure monitoring in living donor kidney recipients does not affect immediate graft function: A propensity score analysis. Clin Transplant. 2018;32:e13238. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 49. | Gingell-Littlejohn M, Koh H, Aitken E, Shiels PG, Geddes C, Kingsmore D, Clancy MJ. Below-target postoperative arterial blood pressure but not central venous pressure is associated with delayed graft function. Transplant Proc. 2013;45:46-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 50. | Aref A, Zayan T, Sharma A, Halawa A. Utility of central venous pressure measurement in renal transplantation: Is it evidence based? World J Transplant. 2018;8:61-67. [PubMed] [DOI] [Full Text] |
| 51. | De Cassai A, Bond O, Marini S, Panciera G, Furian L, Neri F, Andreatta G, Rigotti P, Feltracco P. [Pulse pressure variation guided fluid therapy during kidney transplantation: a randomized controlled trial]. Braz J Anesthesiol. 2020;70:194-201. [PubMed] [DOI] [Full Text] |
| 52. | Goyal VK, Gupta P, Baj B, Garg VK. A randomized comparison between pulse pressure variation and central venous pressure in patients undergoing renal transplantation. J Anaesthesiol Clin Pharmacol. 2021;37:628-632. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 53. | Kannan G, Loganathan S, Kajal K, Hazarika A, Sethi S, Sen IM, Subramanyam R, Singh S. The effect of pulse pressure variation compared with central venous pressure on intraoperative fluid management during kidney transplant surgery: a randomized controlled trial. Can J Anaesth. 2022;69:62-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 54. | Kim KM, Kim GS, Han M. A comparative study of pulse pressure variation, stroke volume variation and central venous pressure in patients undergoing kidney transplantation. Singapore Med J. 2022;63:731-739. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 55. | Toyoda D, Fukuda M, Iwasaki R, Terada T, Sato N, Ochiai R, Kotake Y. The comparison between stroke volume variation and filling pressure as an estimate of right ventricular preload in patients undergoing renal transplantation. J Anesth. 2015;29:40-46. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 56. | Chin JH, Jun IG, Lee J, Seo H, Hwang GS, Kim YK. Can stroke volume variation be an alternative to central venous pressure in patients undergoing kidney transplantation? Transpl Proc. 2014;46:3363-3366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 57. | Goyal VK, Gupta P, Baj B. Anesthesia for renal transplantation in patients with dilated cardiomyopathy: a retrospective study of 31 cases. Braz J Anesthesiol. 2019;69:477-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 58. | Nimje GR, Goyal VK, Singh P, Shekhrajka P, Mishra A, Mittal S. Assessment of fluid responsiveness after tidal volume challenge in renal transplant recipients: a nonrandomized prospective interventional study. Clin Transplant Res. 2024;38:188-196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 59. | Cavaleri M, Veroux M, Palermo F, Vasile F, Mineri M, Palumbo J, Salemi L, Astuto M, Murabito P. Perioperative Goal-Directed Therapy during Kidney Transplantation: An Impact Evaluation on the Major Postoperative Complications. J Clin Med. 2019;8:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 60. | Srivastava D, Sahu S, Chandra A, Tiwari T, Kumar S, Singh PK. Effect of intraoperative transesophageal Doppler-guided fluid therapy versus central venous pressure-guided fluid therapy on renal allograft outcome in patients undergoing living donor renal transplant surgery: a comparative study. J Anesth. 2015;29:842-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 61. | Adelmann D, Legrand M. Intraoperative blood pressure management during kidney transplantation: Grafts under pressure. Am J Transplant. 2024;24:1925-1927. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Pongpruksa C, Khampitak N, Chang D, Bunnapradist S, Gritsch H, Xia VW. Intraoperative Mean Arterial Pressure and Postoperative Delayed Graft Function in Kidney Transplantation: Evaluating Three Commonly Used Thresholds. Clin Transplant. 2024;38:e15458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 63. | Sicova M, McGinn R, Emerson S, Perez P, Gonzalez R, Li Y, Famure O, Randall I, Mina DS, Santema M, Wijeysundera DN, Van Klei W, Kim SJ, McCluskey SA. Association of Intraoperative Hypotension With Delayed Graft Function Following Kidney Transplant: A Single Centre Retrospective Cohort Study. Clin Transplant. 2024;38:e70000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 11] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 64. | Oh C, Bang JB. #621 The effect of intra-operative mean arterial pressure on early graft function in deceased donor kidney transplantation. Nephrol Dial Transpl. 2024;39:gfae069-1671. [DOI] [Full Text] |
| 65. | Kinoshita K, Yamanaga S, Kaba A, Tanaka K, Ogata M, Fujii M, Hidaka Y, Kawabata C, Toyoda M, Uekihara S, Kashima M, Miyata A, Inadome A, Kobayashi T, Yokomizo H. Optimizing Intraoperative Blood Pressure to Improve Outcomes in Living Donor Renal Transplantation. Transplant Proc. 2020;52:1687-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 66. | Campos L, Parada B, Furriel F, Castelo D, Moreira P, Mota A. Do intraoperative hemodynamic factors of the recipient influence renal graft function? Transplant Proc. 2012;44:1800-1803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 68] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 67. | Kakuta N, M Tsutsumi Y, Murakami C, Sakai Y, Oyama T, Kasai A, Kume K, Tanaka K. Effectiveness of using non-invasive continuous arterial pressure monitoring with ClearSight in hemodynamic monitoring during living renal transplantation in a recipient:a case report. J Med Invest. 2018;65:139-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 68. | Goyal VK, Shekhrajka P, Mittal S, Bhardwaj M. Impact of FloTrac versus hypotension prediction index (HPI)-guided haemodynamic management on intraoperative hypotension in kidney transplantation: A retrospective observational study. Indian J Anaesth. 2025;69:496-501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |