INTRODUCTION
Epidemiology and clinical context
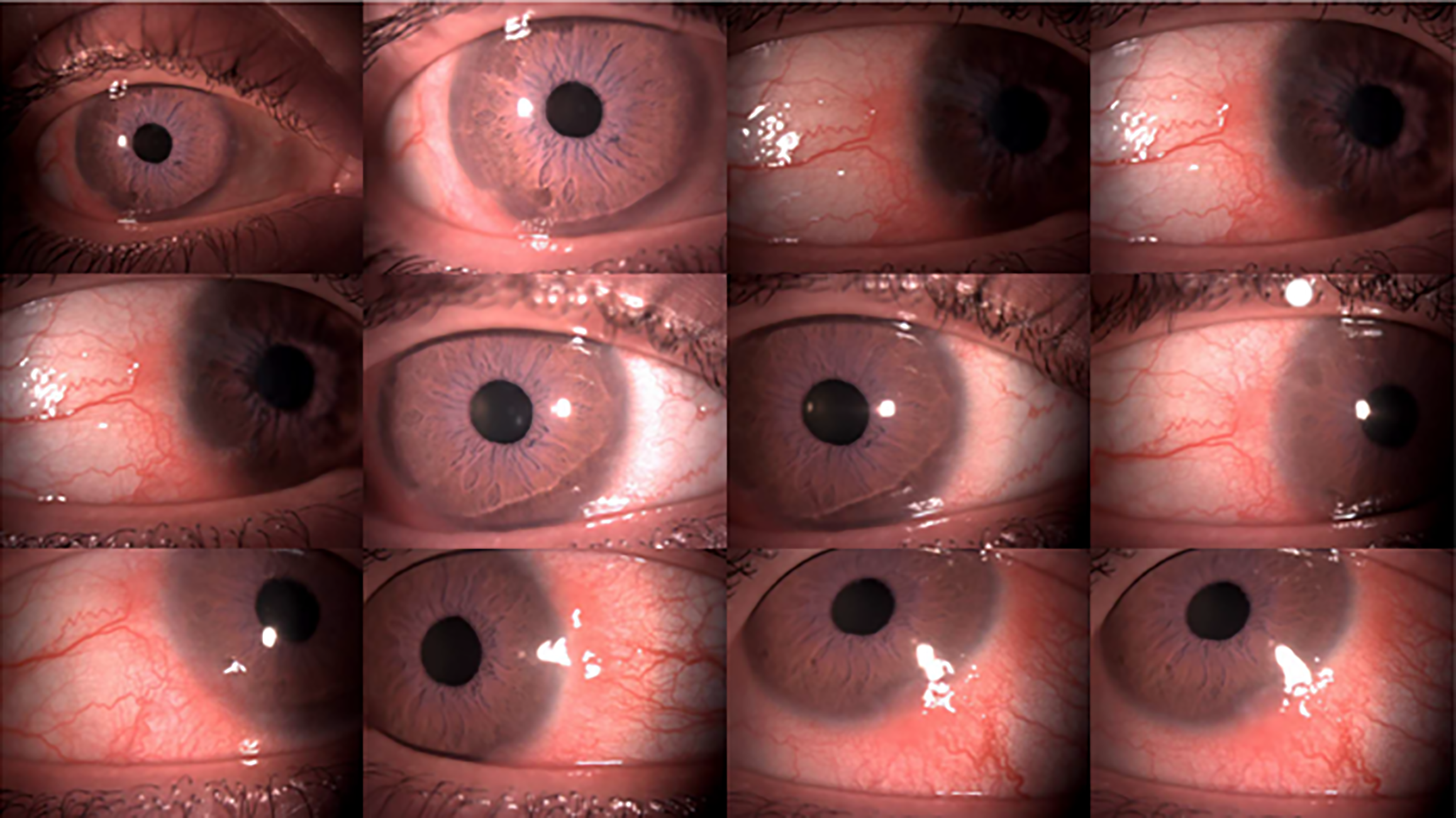
Phlyctenular keratoconjunctivitis (PKC) is characterized by small nodular inflammatory lesions at the corneal or conjunctival surface (Figure 1), caused by a hypersensitivity response to foreign antigens. It is a recurrent nodular inflammatory disease of the ocular surface, classically framed as a Gell-Coombs type IV (delayed) hypersensitivity to microbial antigens rather than a primary infection[1-4]. Before the 1950s, it was frequently a manifestation of delayed hypersensitivity to tuberculin in settings where tuberculosis was highly prevalent, classically affecting undernourished children with positive tuberculin skin tests. With improved public health measures and the decline of tuberculosis, the condition became less common, and many subsequent patients showed negative tuberculin tests[5-7] (Figure 1).
Figure 1
Patient with follicular keratoconjunctivitis, characterized by typical gelatinous-nodular lesions and marked conjunctival injection.
In contemporary practice in the United States, the most frequently implicated antigens are microbial proteins from Staphylococcus aureus, particularly in individuals with chronic blepharitis or suppurative keratitis[3,6,8]. Etiology is context-dependent: In regions where tuberculosis is endemic, mycobacterial antigens remain prominent triggers and are associated with more severe or recurrent disease, whereas in high-income settings, PKC often occurs within the broader spectrum of pediatric blepharokeratoconjunctivitis, with Staphylococcus aureus lid disease, meibomian gland dysfunction, and ocular rosacea as common antigen sources[3,6,9-11].
PKC is a frequent reason for pediatric ophthalmology referrals and predominantly affects children aged 6 months to 16 years, with higher rates in females and a seasonal peak in spring[3,12,13]. Although PKC can occur at any age, it is predominantly pediatric and often recurrent, and contemporary series highlight its potential for sight-threatening corneal involvement, including neovascularization, scarring, amblyopia, and, rarely, perforation. In a recent United States tertiary-center cohort, corneal neovascularization was reported in approximately 40% of affected eyes, scarring in about 27%, amblyopia in 17%, and perforation in around 3%, with earlier case reports describing perforation even in nontuberculous (Staphylococcal) disease[4,12,14,15].
Pathophysiology
The disorder is thought to represent a delayed hypersensitivity reaction at the cornea or conjunctiva that occurs upon re-exposure to an infectious antigen to which the host has already been sensitized[8,16,17]. Antigens from Staphylococcus aureus and Mycobacterium tuberculosis are most commonly involved. Still, other pathogens, such as herpes simplex virus (HSV), Chlamydia, Streptococcus viridans, Dolosigranulum pigrum, and various intestinal parasites (including Hymenolepis nana), have also been implicated[5,12,18-22].
Cytological examination of lesional scrapings typically reveals a predominance of helper T cells, along with suppressor/cytotoxic T lymphocytes, monocytes, and Langerhans cells, with most cells expressing human leukocyte antigen (HLA)-DR. Histologic and cytologic studies thus demonstrate a T-cell–predominant infiltrate with HLA-DR–positive inflammatory cells in phlyctenules, supporting a cell-mediated response to exogenous antigen. The presence of these antigen-presenting cells and T lymphocytes endorses the concept of a type IV, cell-mediated immune reaction. A possible link with ocular rosacea-another condition suspected to have a similar delayed hypersensitivity background-has been reported, and associations with asthma and other allergic diseases further suggest that an altered immune response plays a central role in the pathogenesis[1,8,13,23-25].
Viral antigens can also trigger a type IV hypersensitivity response with a PKC phenotype[1,6]. Evidence includes PKC/phlyctenular phenotypes in the setting of HSV infection (with documented response to antiviral therapy)[20,22,25-27], a pediatric case occurring during severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection[21], and an antigen-driven form associated with molluscum contagiosum that resolved after removal of the lesions[14,28,29]. These observations converge into a model in which persistence and/or load of bacterial or viral antigens at the lid margin drives a localized Th1 response. Clinical implication: In addition to short courses of topical steroids, it is necessary to eliminate the antigenic source and, when viral, to add targeted therapy (antivirals for HSV; eradication of molluscum), together with a steroid-sparing strategy (e.g. cyclosporine) to reduce recurrences, limit the cumulative steroid burden, and achieve more sustained control of ocular surface inflammation, thereby lowering the risk of chronic conjunctival remodeling, corneal involvement, and long-term treatment-related adverse effects[13,22,23,30-32].
This article is a narrative minireview that synthesizes epidemiologic, clinical, immunopathologic, and therapeutic data on PKC, with a specific focus on cases associated with viral triggers, including HSV, SARS-CoV-2, and molluscum contagiosum. The primary aim was to contextualize virus-associated PKC within the broader spectrum of antigen-driven hypersensitivity disease and to derive practical management recommendations for pediatric and adult patients.
A comprehensive literature search was performed in the main biomedical databases, including PubMed/MEDLINE, EMBASE, Web of Science, and the Cochrane Library, from database inception through the most recent available update at the time of writing. Additional targeted searches were conducted in ClinicalTrials.gov and Google Scholar to identify any ongoing studies, difficult-to-retrieve case reports, or gray literature. Search terms combined controlled vocabulary and free-text synonyms for “phlyctenular keratoconjunctivitis”/“phlyctenulosis”, bacterial and mycobacterial triggers (e.g., “Staphylococcus aureus”, “blepharitis”, “tuberculosis”, “Mycobacterium tuberculosis”), and viral triggers (“herpes simplex virus”, “HSV”, “SARS-CoV-2”, “COVID-19”, “molluscum contagiosum”). Only articles in English were considered; titles and abstracts were screened for relevance, and full texts of potentially eligible reports were reviewed.
Clinical evidence from case reports, case series, observational studies, and key reviews describing PKC or closely related phlyctenular disease with an identifiable or strongly suspected antigenic trigger was qualitatively synthesized; no formal meta-analysis was attempted because of heterogeneity and small sample sizes. In addition to published data, a small number of illustrative anonymized cases from the authors’ institutions were retrospectively selected to exemplify typical presentations associated with HSV, SARS-CoV-2, and molluscum. No new patient recruitment or prospective data collection was performed. As this is a narrative review that includes only anonymized retrospective clinical information, formal ethics committee approval was not required, and written informed consent for the scientific use of anonymized data and images was obtained from patients or their legal guardians in accordance with institutional policies and the Declaration of Helsinki.
DISCUSSION AND FUTURE DIRECTIONS
Viral triggers
Classically labeled as a bacterial- or mycobacterial-driven disorder, PKC can also be precipitated by viral antigen exposure in a minority of cases. Three clinical lines of evidence recur across the literature.
HSV: During an outbreak of herpes gladiatorum among wrestlers, a subset with ocular involvement developed phlyctenular disease, and HSV-1 was cultured from conjunctiva and eyelid vesicles in most of those with eye findings-strongly supporting a virus-linked, antigen-driven mechanism. More recently, a patient with conjunctival phlyctenule and nodular scleritis improved after systemic valacyclovir, suggesting that targeted antiviral therapy can be decisive when a herpetic trigger is plausible[6,14,20,22].
SARS-CoV-2: A pediatric case described unilateral PKC during acute coronavirus disease 2019 (COVID-19), interpreted as an immune-mediated manifestation rather than direct cytopathic keratitis-evidence that systemic viral infection can modulate ocular surface immunity toward a phlyctenular phenotype. Similarly, Barbara et al[33] reported a 16-year-old girl with multisystem inflammatory syndrome in children secondary to SARS-CoV-2 who developed bilateral nongranulomatous keratouveitis with subtle peripheral anterior stromal corneal infiltrates and nonpurulent conjunctivitis, in the context of a negative uveitis work-up and complete resolution after topical corticosteroids. This case further supports the hypothesis that, beyond direct viral invasion, the post-infectious hyperinflammatory state and cytokine storm induced by SARS-CoV-2 can trigger immune-mediated corneal inflammation along a spectrum that includes PKC[21,33,34].
Molluscum contagiosum: Eyelid lesions from this benign cutaneous poxvirus can chronically seed the ocular surface with viral antigen, driving a picture of unilateral follicular keratoconjunctivitis with superior pannus and phlyctenule-like nodules. The periocular lesions are classically small dome-shaped papules (2-6 mm) with a central umbilication. Still, they may be non-umbilicated or masquerade as chalazion, papilloma, sebaceous cyst, milium, or fibroma, and in both immunocompromised and immunocompetent children, may reach “giant” dimensions at the lid margin. Case reports describe otherwise healthy pediatric and young adult patients with weeks to months of chronic, refractory conjunctivitis and marginal keratitis, often mismanaged with repeated courses of topical antibiotics, antiallergic drops, and steroids until a careful inspection of the upper lid margin reveals a subtle or atypical molluscum nodule; histopathology typically shows epidermal hyperplasia with intracytoplasmic molluscum bodies. In such cases, the ocular surface disease is thought to represent a delayed-type hypersensitivity response to viral proteins shed into the tear film, producing follicular conjunctivitis, subepithelial infiltrates, marginal keratitis, pannus, and phlyctenule-like lesions that are clinically indistinguishable from classic PKC. This fits well with the modern series of pediatric PKC from developed settings, in which adjacent lid/external disease is the dominant driver and viral infections-including periocular molluscum contagiosum-are recognized among the associated antigenic triggers alongside staphylococcal blepharokeratoconjunctivitis.
Importantly, across published cases of molluscum-related keratoconjunctivitis, simple excision, curettage, or eradication of the eyelid focus (with or without adjunctive cryotherapy) leads to rapid and often complete resolution of the follicular response, keratitis, and pannus within weeks, without the need for prolonged topical steroids or systemic therapy. Molluscum thus provides one of the clearest “proof-of-mechanism” models for antigen-driven phlyctenular disease: Once the cutaneous antigen reservoir is removed, ocular surface inflammation typically switches off, highlighting the importance of meticulous lid-margin examination in any child with chronic, unilateral, “treatment-resistant” follicular or PKC[21,28,29,35,36].
Immunopathology model
Cytology from phlyctenules consistently shows T-cell–predominant infiltrates with abundant HLA-DR-positive inflammatory cells-features of a type IV (delayed) hypersensitivity response. Classic immunocytological series have demonstrated that helper T cells markedly outnumber cytotoxic T cells, that B-cell markers are confined to the phlyctenular lesion rather than the surrounding conjunctiva, and that S-100-positive accessory cells are present, with the vast majority of cells expressing HLA-DR, all of which reinforces a cell-mediated, antigen-driven mechanism. In this framework, antigen persistence or high antigen load at the lid margin/ocular surface-whether bacterial (e.g., Staphylococcus aureus or, less commonly, Streptococcus viridans) or viral (HSV, molluscum)-biases toward a localized, Th1-skewed reaction at the limbus and superficial cornea. Clinically, this is mirrored by the course of nontuberculous PKC, in which failure to control the underlying lid-margin disease can lead to progressive corneal vascularization, scarring and even permanent visual loss, whereas strategies that reduce the antigenic burden or its downstream inflammatory cascade-vigorous lid hygiene, targeted topical antibiotics and corticosteroids, and, in recurrent or steroid-limited cases, systemic tetracyclines-can rapidly quieten symptoms and arrest progression. This model integrates geographic variation (tuberculous and helminth-related disease in endemic settings; staphylococcal blepharitis/rosacea and other bacterial lid disease elsewhere) with the less frequent but well-documented virus-linked presentations, which appear to exploit the same antigen-driven, T-cell–mediated pathway[8,13,16,18].
Diagnostic clinical implications
For most patients, PKC remains non-viral; however, several bedside cues should prompt a virus-aware work-up: A history of herpetic skin or mucosal lesions, recent COVID-19, or umbilicated eyelid nodules suggestive of molluscum. In such contexts, confirmatory laboratory testing is often not required before acting: Initiating antiviral therapy when HSV is clinically plausible and proceeding to mechanical removal of molluscum lesions are both aligned with available case-based evidence and carry a favorable risk-benefit profile. At the same time, clinicians should not neglect the “core business” of PKC care: Rapid control of the inflammatory response with short courses of topical corticosteroids (with appropriate intraocular pressure monitoring) and timely introduction of steroid-sparing agents such as topical cyclosporine A in recurrent or steroid-dependent cases, in order to limit cataract, ocular hypertension, and other steroid-related sequelae.
In parallel, the source of antigenic stimulation at the lid margin must be addressed through lid hygiene, management of blepharitis or ocular rosacea (including topical azithromycin or systemic macrolides/tetracyclines in age-appropriate patients), and targeted systemic therapy when a communicable trigger such as tuberculosis or chlamydia is identified. Virus-aware practice also means recognizing when the presentation is “too atypical” to be managed as routine non-viral PKC: Disproportionately severe pain, evolution from conjunctival phlyctenule to nodular scleritis, or poor response to intensive topical steroids should lower the threshold for a broader infectious work-up (including HSV-1/2 serology and swabs) and for adding systemic antivirals.
Case reports of HSV-2–associated phlyctenular conjunctivitis with nodular scleritis describe limited benefit from corticosteroid monotherapy and subsequent, marked improvement only after the introduction of oral valacyclovir, underscoring that in herpes-suspect disease, anti-inflammatory therapy should be coupled with antiviral cover rather than escalated in isolation. Taken together, these practical points translate the immunopathogenic model into bedside practice: Reduce inflammation, remove or suppress the antigen source, and maintain a low threshold for antiviral treatment or extended infectious work-up whenever the clinical pattern deviates from the usual, staphylococcus-driven PKC phenotype[12,22,23,37].
Therapeutic considerations
Topical corticosteroids remain appropriate for rapid control of inflammation, but source control is what changes the trajectory. When bacterial drivers are suspected (blepharitis/ocular rosacea), reducing antigen load through lid hygiene and antimicrobials (systemic tetracycline-class or erythromycin in younger children) lowers the risk of recurrence. Historical pediatric series of nontuberculous PKC show that a limited course of oral tetracycline or erythromycin can induce long-lasting remission and halt progressive corneal neovascularization and scarring, offering an alternative to prolonged steroid exposure in severe or recurrent disease. More recently, macrolide-based regimens-including azithromycin 1.5% eye drops, oral macrolides, and low-dose systemic clarithromycin in meibomitis-related keratoconjunctivitis-have been shown to improve lid disease, ocular surface inflammation, and keratoepitheliopathy, further supporting a “treat-the-source” strategy[16,23,26,37,38].
In steroid-dependent or recurrent pediatric PKC, topical cyclosporine A (2%) provides effective steroid-sparing control in prospective series, with sustained quiescence after tapering in many children; in more refractory cases, additional immunomodulators such as topical tacrolimus or, in extreme situations, systemic biologic agents have been successfully used as escalation therapy. In virus-associated scenarios, add pathogen-directed treatment (e.g., oral antivirals for HSV, full anti-tuberculosis regimens when ocular TB is documented, or physical eradication for molluscum) rather than escalating steroids alone, and consider screening for less-obvious infectious sources, such as intestinal parasitosis, in high-risk settings. Overall, therapeutic priorities thus shift from simply “cooling down” the phlyctenule to a layered approach that couples a brief steroid course with aggressive management of the inciting antigen and, when needed, targeted immunomodulation[23,37-42].
Strengths, limitations and future directions
The viral-PKC link currently rests on outbreak reports, case reports, and small series. These designs cannot quantify incidence or prove causality, but they repeatedly show temporal association and therapeutic reversibility (resolution with antivirals or lesion removal). Herpes gladiatorum clusters with culture-proven HSV-1 and concomitant phlyctenular disease, single HSV-2 cases with phlyctenule plus nodular scleritis responding only after valacyclovir, and peri-varicella/zoster limbal nodules all point in the same direction, yet they remain anecdotal and small-number signals against a background of very high viral seroprevalence. Moreover, several “viral” reports rely on indirect markers (past IgG positivity, syndromic diagnoses) without biopsy or PCR confirmation, and PKC-like lesions may sit on the border between classic phlyctenulosis and broader virus-induced hypersensitivity phenotypes (e.g., Epstein Barr virus/hydroa vacciniforme).
Modern pediatric PKC cohorts and prospective series continue to show that viral triggers constitute, at most, a small minority of cases compared with staphylococcal, tuberculous, or parasitic antigens, underscoring the risk of over-attributing causality to coincidental viral exposure. Until controlled, adequately powered studies with standardized case definitions and virologic work-up are available, a trigger-focused approach remains the most defensible: Aggressively search for and treat plausible antigen sources (including viruses when clinical cues are strong), but frame decisions as pragmatic, low-risk interventions aligned with the immunopathology rather than as proof that a given virus “causes” PKC, and always with the parallel goal of minimizing cumulative steroid exposure[20,22,43-45].
Measles provides a classic example that systemic viral exanthems can be accompanied by keratitis/keratoconjunctivitis with clinically significant corneal involvement, including potentially blinding sequelae in susceptible children[45].
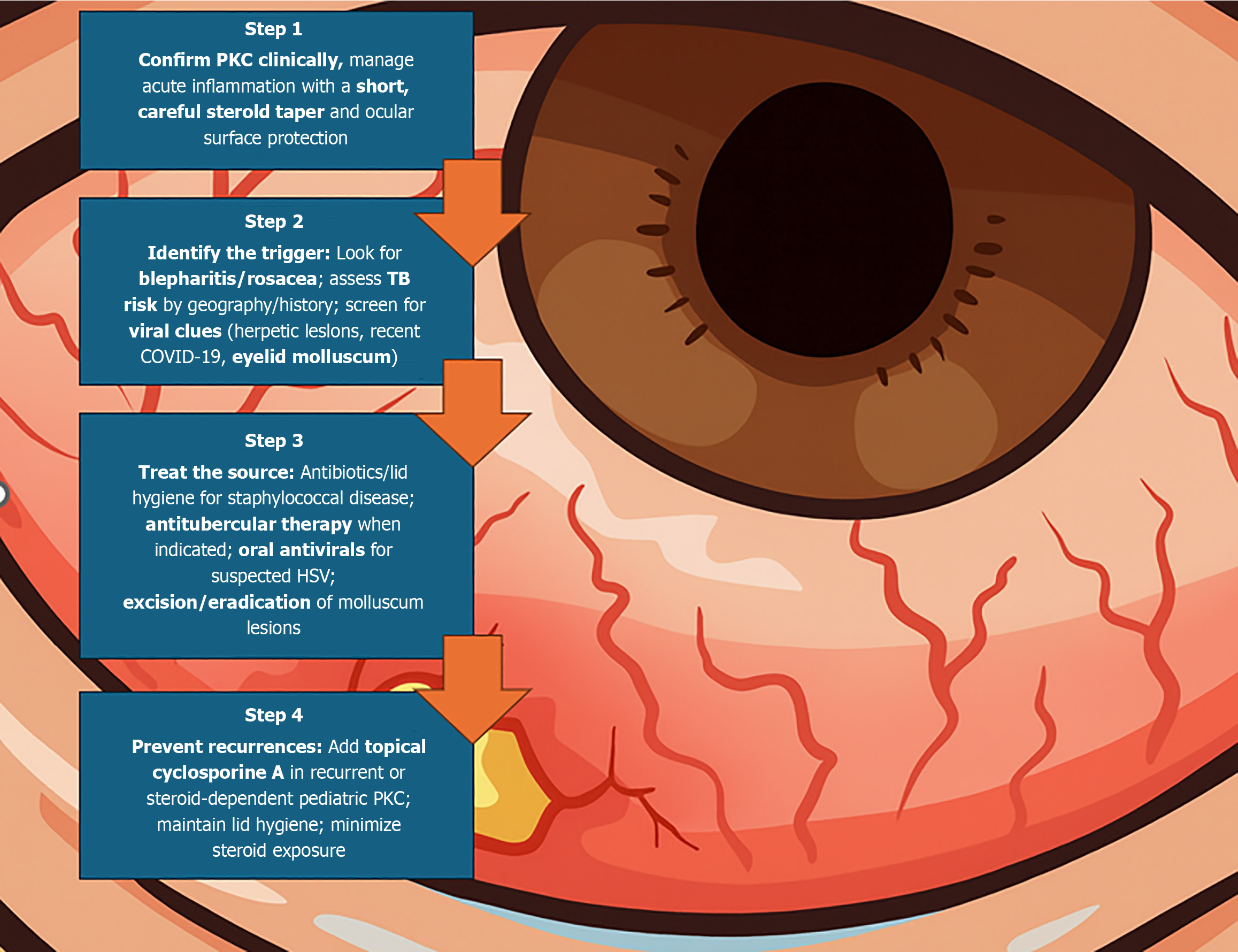
To clarify the role of viruses in PKC, several pragmatic steps are needed. First, prospective multicenter registries should estimate the true frequency of virus-associated presentations and collect standardized outcomes-time to resolution, recurrence, need for procedures or surgery, and cumulative steroid exposure-allowing reliable comparisons across centers. Second, tear and conjunctival immune profiling during active disease, using minimally invasive methods (impression cytology and targeted assays on tear samples), can test the expected type-IV/Th1 pattern in virus-linked cases vs classic staphylococcal PKC. Third, interventional studies are warranted: In HSV-suspect PKC, compare a short steroid taper alone with steroid plus systemic antivirals using predefined ocular outcomes and steroid-sparing endpoints; in eyelid molluscum contagiosum, perform randomized evaluations of lesion-eradication strategies (curettage, cryotherapy, topical immunomodulators). Finally, on the public-health side, during HSV outbreaks in contact sports or surges of respiratory viruses, simple ocular screening checklists in first-line settings (sports medicine, pediatrics, primary care) may support earlier identification and targeted management of antigen sources. These are realistic research and organizational priorities; well-designed studies should confirm their clinical impact. Figure 2 summarizes a trigger-focused, virus-aware management algorithm for PKC.
Figure 2 Practical, trigger-focused, and virus-aware algorithm for the management of phlyctenular keratoconjunctivitis.
PKC: Phlyctenular keratoconjunctivitis; TB: Tuberculosis; COVID-19: Coronavirus disease 2019; HSV: Herpes simplex virus.
CONCLUSION
PKC is best understood as an antigen-driven, type IV (delayed) hypersensitivity disease of the ocular surface. While Staphylococcus aureus lid disease and, in specific settings, mycobacterial antigens remain the dominant triggers, credible reports show that viral exposures-notably HSV, SARS-CoV-2, and eyelid molluscum contagiosum-can precipitate the same phlyctenular phenotype in a minority of patients. A unifying feature across these scenarios is antigen persistence or high antigen load at the lid margin, which biases a localized T-cell–mediated response at the limbus and superficial cornea.
For clinical practice, the priority is to pair a brief topical steroid course with source control. In staphylococcal/rosacea phenotypes, that means lid hygiene and targeted antimicrobials; in tuberculosis-risk contexts, appropriate testing and therapy; in virus-linked presentations, add pathogen-directed treatment (oral antivirals when HSV is plausible; excision/eradication of molluscum lesions). In recurrent or steroid-dependent pediatric PKC, topical cyclosporine offers a steroid-sparing path to durable control. Escalating steroids alone in suspected herpetic disease is ill-advised. The evidence base for virus-associated PKC is still limited (outbreaks, case reports, small series). To move beyond plausibility, the field needs multicenter registries, standardized case definitions with virologic work-ups, and interventional studies testing “steroid-only” vs “steroid + pathogen-directed” strategies, alongside tear/conjunctival immune profiling to verify the predicted type IV/Th1 signature. Until such data are available, a trigger-focused, virus-aware approach aligns with immunopathology, supports antimicrobial stewardship, and-most importantly-reduces recurrences and vision-threatening complications while minimizing cumulative steroid exposure.