Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.117079
Revised: January 13, 2026
Accepted: February 4, 2026
Published online: March 25, 2026
Processing time: 106 Days and 1.1 Hours
Dengue fever remains a major cause of acute febrile illness in India, particularly in rural areas where diagnostic facilities are limited. Early detection using point-of-care tests helps guide timely management and reduce unnecessary antibiotic use.
To determine the seroprevalence and clinical profile of dengue infection among acute febrile patients attending a rural tertiary care hospital in Maharashtra.
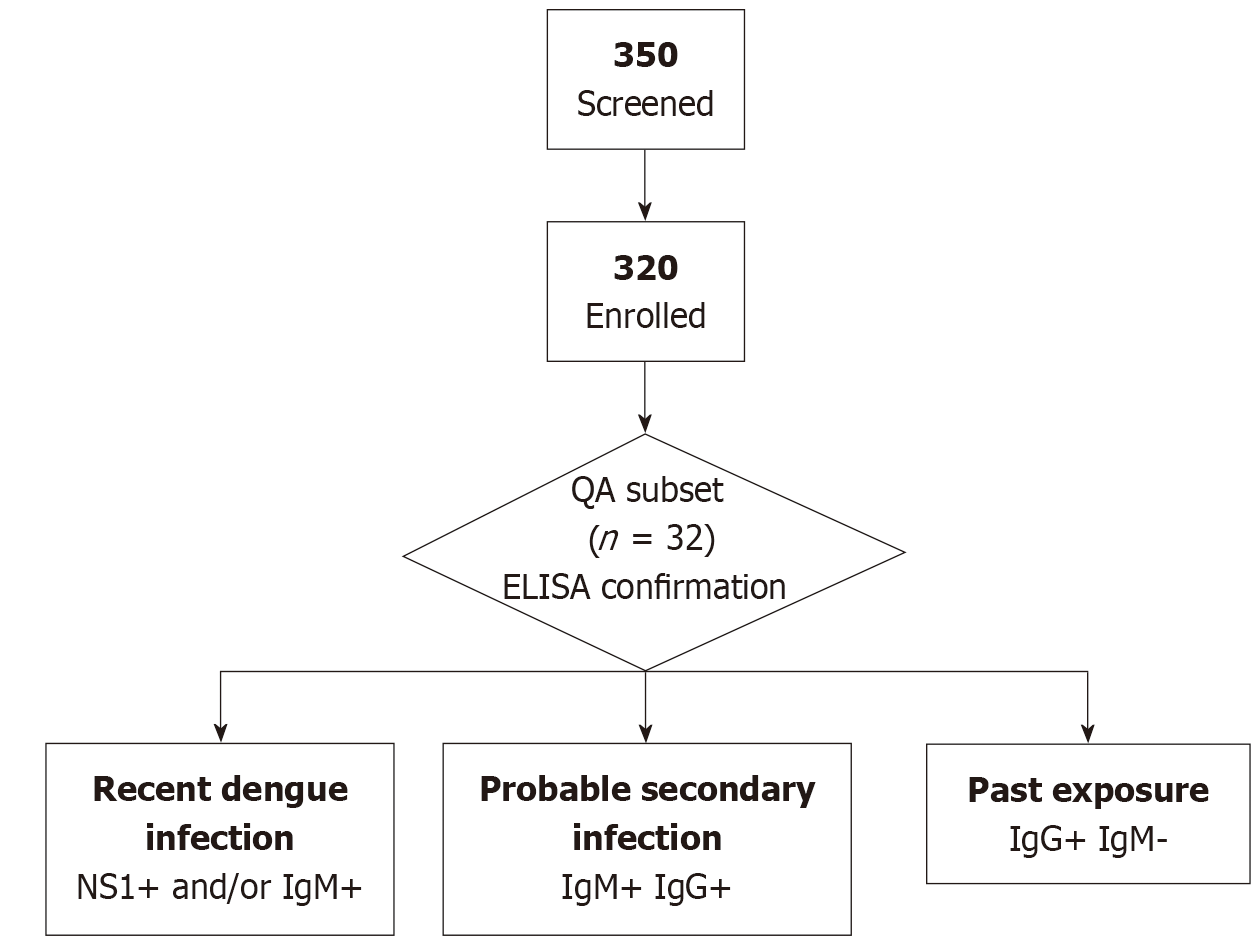
A prospective cross-sectional study was conducted at Ashwini Rural Medical College Hospital and Research Centre, Solapur from May to October 2025. A total of 320 consecutive patients presenting with acute fever of ≤ 7 days duration prior to hospital presentation were enrolled. Patients of all age groups were included. Serum samples were tested for dengue NS1 antigen and anti-dengue IgM and IgG antibodies using a commercial rapid test (Manufacturer: Meril diagnostics; Meriscreen dengue NS1 antigen and IgM + IgG antibodies). A 10% subset (n = 32) was tested by enzyme-linked immunosorbent assay (ELISA) for quality assurance. Patients were classified as recent dengue (NS1+ and/or IgM+), probable secondary infection (IgM+ IgG+), past exposure (IgG+ IgM-), or negative (NS1- IgM-). Clinical data and outcomes were recorded. Statistical analysis included χ2, Mann-Whitney U, and logistic regression (α = 0.05), adjusting for age, sex, platelet count, total leukocyte count, and presence of rash.
Overall, 28% (90/320) had evidence of recent dengue, while 35% (112/320) showed IgG positivity, and 10% (32/320) had probable secondary infection. Median platelet count was significantly lower in the recent dengue group [78000/mm³; interquartile range (IQR): 55000-110000] compared with non-dengue patients (210000/mm³; IQR: 160000-240000; P < 0.001). The most common symptoms were headache (68%), myalgia (61%), and retro-orbital pain (44%). Antibiotic use prior to testing was 60%; among those diagnosed with dengue, a relative 25% reduction in antibiotic prescriptions was observed following laboratory confirmation. Quality assurance analysis showed rapid test sensitivity of 92%, specificity of 95%, and Cohen’s kappa of 0.85 vs ELISA. Logistic regression identified thrombocytopenia (< 100000/mm³) [adjust odds ratio (aOR) 5.2; 95%CI: 2.4-10.9] and rash (aOR 3.6; 95%CI: 1.8-7.0) as independent predictors of dengue positivity.
A substantial proportion of acute febrile cases in this rural setting were due to dengue, highlighting the utility of NS1/IgM rapid tests for early diagnosis where molecular facilities are unavailable. Integrating rapid dengue testing into routine fever evaluation can improve case detection and rationalize antibiotic use.
Core Tip: This prospective study from a rural tertiary hospital in Maharashtra found that nearly one-third of acute febrile illnesses were due to dengue infection. Rapid NS1/IgM testing showed high diagnostic accuracy (sensitivity 92%, specificity 95%) and led to a clinically meaningful reduction in unnecessary antibiotic use. Incorporating such point-of-care diagnostics in rural healthcare can enable early dengue detection, improve patient management, and strengthen antimicrobial stewardship.
- Citation: Dhotre SV, Ghatole MP, Dhotre PS, Kashetty VA, Nagoba BS. Seroprevalence and clinical profile of dengue infection among acute febrile patients at a rural tertiary care hospital. World J Virol 2026; 15(1): 117079
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/117079.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.117079
Dengue fever, caused by Dengue virus (DENV) and transmitted by Aedes mosquitoes, continues to pose a significant public health burden in India[1,2]. The disease manifests as a wide clinical spectrum ranging from self-limiting fever to severe dengue hemorrhagic fever and shock[3,4]. Early and accurate diagnosis is critical for patient management and outbreak control.
In rural India, limited access to advanced molecular diagnostics such as reverse transcription polymerase chain reaction (RT-PCR) challenges early case confirmation. Point-of-care rapid tests detecting NS1 antigen and anti-dengue IgM/IgG antibodies are valuable tools in these settings[5,6]. The NS1 antigen is detectable from day 1 to 5 of fever, while IgM and IgG appear after day 4, facilitating differentiation of acute and secondary infections[7,8].
Previous studies in urban tertiary hospitals report seroprevalence ranging between 20%-40% among febrile patients[9,10]. Most of these studies relied primarily on serological assays, including NS1 antigen detection and IgM/IgG enzyme-linked immunosorbent assay (ELISA), with limited use of molecular confirmation. However, data from rural tertiary care hospitals remain scarce. Assessing local seroprevalence and correlating it with clinical and hematological parameters is crucial for improving diagnostic strategies and antibiotic stewardship in such areas.
This study aimed to estimate the seroprevalence of dengue infection using rapid diagnostic tests among acute febrile patients at a rural tertiary care hospital and to analyze associated clinical and laboratory profiles.
A prospective cross-sectional study was carried out at Ashwini Rural Medical College Hospital and Research Centre, Solapur, Maharashtra, from May 2025 to October 2025. This 800-bed rural tertiary hospital caters to semi-urban and village populations in western Maharashtra. During the study period, dengue testing was not routinely performed for all febrile patients and was generally requested at the discretion of the treating physician; the present study implemented systematic screening of all eligible acute febrile cases.
Based on an expected dengue prevalence of 25% among acute febrile patients, with 95% confidence level (z = 1.96) and 5% absolute precision, the required sample size was 289. Allowing 10% for incomplete data, the final sample size was 320 patients.
Patients of any age presenting with acute fever of ≤ 7 days duration prior to hospital presentation. Willingness to provide written informed consent.
Fever of known bacterial or non-infectious etiology. Refusal to consent.
Venous blood (3-5 mL) was collected aseptically. Serum was separated and tested immediately using a commercial dengue rapid combo test (NS1/IgM/IgG; Manufacturer Meril diagnostics, Meriscreen Dengue NS1 Antigen and IgM + IgG Antibodies) as per the manufacturer’s instructions. Ten percent of samples (n = 32) were randomly selected and retested using ELISA for NS1 and IgM/IgG antibodies at the State Public Health Laboratory for quality assurance.
Recent infection: NS1+ and/or IgM+.
Probable secondary infection: IgM+ and IgG+.
Past exposure: IgG+ IgM-.
Negative: NS1- IgM-.
Clinical and demographic data were recorded in a structured proforma, including age, sex, residence, duration of fever, symptoms, laboratory parameters (complete blood cell count, platelet count, liver function tests), and outcome. Patients were also assessed for clinical or laboratory evidence suggestive of concurrent bacterial infection during hospitalization.
Data was analyzed using SPSS version 26. Descriptive statistics summarized demographic and clinical characteristics. Continuous variables were expressed as median [interquartile range (IQR)]; categorical variables as n (%). Comparisons between dengue-positive and dengue-negative groups used χ2/Fisher’s exact test for categorical variables and Mann-Whitney U test for continuous data. Logistic regression identified independent predictors of recent dengue infection, adjusting for age, sex, platelet count, total leukocyte count, and presence of rash. Statistical significance was set at P < 0.05.
The study was conducted in accordance with the ethical principles of the Declaration of Helsinki. As this was an observational study involving investigations performed as part of routine clinical care, formal Institutional Ethics Committee approval was not required. Written informed consent was obtained from all participants or their legally authorized representatives, and patient confidentiality was strictly maintained
A total of 320 patients were enrolled. The median age was 31 years (IQR: 22-44), and 56% were male. Most patients (62%) resided in rural villages within a 30 km radius of the hospital.
Among 320 patients, 90 (28%) had evidence of recent dengue infection, 32 (10%) probable secondary infection, and 112 (35%) past exposure (Table 1). NS1 antigen was detected in 64 patients (20%), and IgM antibodies in 58 (18%). Co-positivity (NS1+IgM+) occurred in 32 patients (10%).
| Serological category | Criteria | n (%) |
| Recent dengue infection | NS1+ and/or IgM+ | 90 (28.1) |
| Probable secondary infection | IgM+ IgG+ | 32 (10.0) |
| Past exposure | IgG+ IgM- | 112 (35.0) |
| Negative | NS1- IgM- | 86 (26.9) |
The most frequent symptoms among dengue-positive patients were headache (68%), myalgia (61%), retro-orbital pain (44%), and rash (26%). Bleeding manifestations were seen in 8%. Median platelet count was significantly lower among dengue-positive cases (78000/mm³, IQR: 55000-110000) compared to negatives (210000/mm³, IQR: 160000-240000; P < 0.001) (Table 2).
| Parameter | Recent dengue (n = 90) | Non-dengue (n = 230) | P value |
| Median age (years) | 30 (20-42) | 33 (24-45) | 0.18 |
| Male sex (%) | 55.6 | 57.0 | 0.82 |
| Headache (%) | 68.0 | 42.0 | < 0.001 |
| Rash (%) | 26.0 | 8.0 | 0.002 |
| Bleeding signs (%) | 8.0 | 2.0 | 0.04 |
| Median platelet count (/mm³) | 78000 (55000-110000) | 210000 (160000-240000) | < 0.001 |
Overall, 60% of patients received empirical antibiotics before dengue testing. Among confirmed dengue cases, antibiotic usage declined by 25% following laboratory confirmation, indicating partial but clinically relevant rationalization of antibiotic prescribing.
Of the 32 samples tested by ELISA, the rapid test demonstrated a sensitivity of 92% and specificity of 95%, with Cohen’s kappa of 0.85, indicating excellent agreement.
On multivariable logistic regression, two independent predictors of recent dengue infection were identified: Thrombocytopenia (< 100000/mm³): Adjust odds ratio (aOR) = 5.2; 95%CI: 2.4-10.9; P < 0.001. Rash: AOR = 3.6; 95%CI: 1.8-7.0; P = 0.001 (Figure 1).
This study demonstrates that nearly one-third of acute febrile illnesses in a rural tertiary hospital were attributable to dengue, consistent with regional studies reporting 20%-35% prevalence[11,12]. The use of NS1 and IgM rapid tests effectively identified cases during the acute phase, supporting their value where PCR is unavailable.
The predominance of young adults and males parallels findings from other Indian cohorts[13,14]. Common clinical features-headache, myalgia, retro-orbital pain-remain characteristic[15,16]. Platelet counts were significantly reduced in dengue-positive patients, reinforcing thrombocytopenia as a strong predictor. The observed association of rash and thrombocytopenia with seropositivity was statistically significant, consistent with previous literature[17,18]. Although antibiotic therapy is inherently inappropriate in uncomplicated dengue infection, this reduction reflects the real-world impact of early diagnostic confirmation on prescribing practices in resource-limited rural settings. Similar trends have been reported in endemic regions, where point-of-care dengue diagnostics contributed to reductions in unnecessary antibiotic use without compromising patient outcomes[19,20].
No laboratory-confirmed bacterial co-infections were identified among dengue-positive patients in this study. Antibiotics were continued only in a small subset of patients with clinical features suggestive of secondary bacterial infection, pending further evaluation. This underscores the importance of diagnostic stewardship alongside antimicrobial stewardship in acute febrile illness management.
Rapid tests demonstrated high sensitivity (92%) and specificity (95%) with excellent agreement (κ = 0.85) against ELISA, validating their operational reliability. However, cross-reactivity with other flaviviruses remains a potential limi
Strengths include prospective design, consecutive sampling, and quality assurance via ELISA. Limitations are the lack of molecular confirmation and potential cross-reactivity in serology. Nevertheless, findings provide critical local epidemiological data and demonstrate how rapid testing can enhance case detection and antibiotic stewardship in resource-limited settings.
Limitations include the absence of molecular confirmation by RT-PCR and the inherent possibility of serological cross-reactivity. Although microbiological culture facilities were available, cultures were not systematically performed in all patients with suspected dengue, as clinical management was guided primarily by laboratory-confirmed dengue diagnosis and clinical judgment. Nevertheless, the findings offer important real-world evidence supporting the role of rapid diagnostic testing in dengue-endemic, resource-constrained settings.
Routine integration of NS1/IgM rapid tests into fever clinics in rural hospitals can expedite diagnosis, enable timely supportive management, and reduce antibiotic misuse. Periodic serosurveys can aid in tracking local transmission trends and guiding vector-control efforts.
A significant proportion of acute febrile illnesses in this rural tertiary hospital were due to dengue infection. NS1/IgM rapid diagnostic tests proved reliable for early diagnosis, with strong correlation to clinical and hematological findings. Implementation of point-of-care dengue testing in similar rural settings can improve diagnostic accuracy, support more rational antibiotic prescribing, and strengthen dengue surveillance and control efforts.
The authors thank the Department of Medicine, the Central Clinical Laboratory, and the technical staff of the Department of Microbiology, Ashwini Rural Medical College Hospital and Research Centre, Solapur, for their cooperation during this study.
| 1. | World Health Organization. Dengue-Global situation. [cited 28 January 2026]. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2024-DON518. |
| 2. | Ganeshkumar P, Murhekar MV, Poornima V, Saravanakumar V, Sukumaran K, Anandaselvasankar A, John D, Mehendale SM. Dengue infection in India: A systematic review and meta-analysis. PLoS Negl Trop Dis. 2018;12:e0006618. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 129] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 3. | Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wint GR, Simmons CP, Scott TW, Farrar JJ, Hay SI. The global distribution and burden of dengue. Nature. 2013;496:504-507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7940] [Cited by in RCA: 6575] [Article Influence: 505.8] [Reference Citation Analysis (2)] |
| 4. | Shastri PS, Gupta P, Kumar R. A prospective 3 year study of clinical spectrum and outcome of dengue fever in ICU from a tertiary care hospital in North India. Indian J Anaesth. 2020;64:181-186. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 5. | Hunsperger EA, Sharp TM, Lalita P, Tikomaidraubuta K, Cardoso YR, Naivalu T, Khan AS, Marfel M, Hancock WT, Tomashek KM, Margolis HS. Use of a Rapid Test for Diagnosis of Dengue during Suspected Dengue Outbreaks in Resource-Limited Regions. J Clin Microbiol. 2016;54:2090-2095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 6. | Shukla MK, Singh N, Sharma RK, Barde PV. Utility of dengue NS1 antigen rapid diagnostic test for use in difficult to reach areas and its comparison with dengue NS1 ELISA and qRT-PCR. J Med Virol. 2017;89:1146-1150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (1)] |
| 7. | World Health Organization. Laboratory testing for dengue virus: interim guidance. [cited 28 January 2026]. Available from: https://szu.gov.cz/wp-content/uploads/2025/04/Laboratory-testing-for-dengue-virus-Interim-guidance-April-2025.pdf. |
| 8. | Singh MP, Majumdar M, Singh G, Goyal K, Preet K, Sarwal A, Mishra B, Ratho RK. NS1 antigen as an early diagnostic marker in dengue: report from India. Diagn Microbiol Infect Dis. 2010;68:50-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 9. | Dinkar A, Singh J. Dengue infection in North India: An experience of a tertiary care center from 2012 to 2017. Tzu Chi Med J. 2020;32:36-40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 10. | Gupta S, Shakya A, Mishra N, Islahi S, Singh S, Nag S, Dwivedi A, Shukla M, Sen S, Dwivedi P, Agarwal M, Mukherjee A. Investigating the Clinico-Demographic Characteristics of Dengue Fever and Its Seroprevalence at a Tertiary Care Hospital in Northern India. Cureus. 2024;16:e57640. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |
| 11. | Pooja PS, Kokate SB, Shrikhande S. Seroprevalence of dengue virus infection among the patients admitted with acute febrile illness in a tertiary care hospital in Central India. Int J Community Med Public Health. 2025;12:2674-2679. |
| 12. | Mishra AC, Arankalle VA, Gadhave SA, Mahadik PH, Shrivastava S, Bhutkar M, Vaidya VM. Stratified sero-prevalence revealed overall high disease burden of dengue but suboptimal immunity in younger age groups in Pune, India. PLoS Negl Trop Dis. 2018;12:e0006657. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 13. | Kumar M, Verma RK, Nirjhar S, Singh M. Dengue in children and young adults, a cross-sectional study from the western part of Uttar Pradesh. J Family Med Prim Care. 2020;9:293-297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 14. | Agrawal VK, Prusty BSK, Reddy CS, Mohan Reddy GK, Agrawal RK, Sekher Srinivasarao Bandaru VC. Clinical profile and predictors of Severe Dengue disease: A study from South India. Caspian J Intern Med. 2018;9:334-340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 15. | Centers for Disease Control and Prevention. Clinical features of dengue. [cited 28 January 2026]. Available from: https://www.cdc.gov/dengue/hcp/clinical-signs/index.html. |
| 16. | Mohan K, Malaiyan J, Nasimuddin S, Devasir RS, Meenakshi-Sundaram P, Selvaraj S, Krishnasamy B, Gnanadesikan S, Karthikeyan M, Kandasamy M, Jayakumar N, Elumalai D, Ra GG. Clinical profile and atypical manifestation of dengue fever cases between 2011 and 2018 in Chennai, India. J Family Med Prim Care. 2020;9:1119-1123. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 17. | Das S, Abreu C, Harris M, Shrader J, Sarvepalli S. Severe Thrombocytopenia Associated with Dengue Fever: An Evidence-Based Approach to Management of Thrombocytopenia. Case Rep Hematol. 2022;2022:3358325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (1)] |
| 18. | Haq FU, Imran M, Aslam Z, Mukhtar F, Jabeen K, Chaudhry M, Rahman SU, Muhammad N. Severity of Dengue Viral Infection Based on Clinical and Hematological Parameters among Pakistani Patients. Am J Trop Med Hyg. 2023;109:1284-1289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 19. | Robert S, Diagbouga PS, Djibougou AD, Guy D, Bagnall R, Ravel F. Dengue diagnosis and impact on clinical management: A literature review. PLoS Negl Trop Dis. 2025;19:e0013196. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 7] [Reference Citation Analysis (1)] |
| 20. | Tello-Cajiao ME, Osorio L. Impact of Dengue Rapid Diagnostic Tests on the Prescription of Antibiotics and Anti-Inflammatory Drugs by Physicians in an Endemic Area in Colombia. Am J Trop Med Hyg. 2019;101:696-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | Zamil MF, Hasan A, Trina AT, Hossain MS, Afreen S, Kumkum A, Ahmed D, Alam MS. Diagnostic accuracy of the OnSite Dengue Ag rapid test in symptomatic patients from Dhaka, Bangladesh. BMC Infect Dis. 2025;25:991. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 22. | Luangaram P, Tamdet C, Saengwong C, Prommool T, Kraivong R, Nilchan N, Punyadee N, Avirutnan P, Srisawat C, Malasit P, Kasinrerk W, Puttikhunt C. Differential critical residues on the overlapped region of the non-structural protein-1 recognized by flavivirus and dengue virus cross-reactive monoclonal antibodies. Sci Rep. 2022;12:21548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (1)] |