Published online Mar 25, 2026. doi: 10.5501/wjv.v15.i1.117008
Revised: December 29, 2025
Accepted: February 3, 2026
Published online: March 25, 2026
Processing time: 107 Days and 16.1 Hours
Dengue virus infection is one of the most prevalent arboviral diseases worldwide, particularly affecting tropical and subtropical regions, and continues to pose a significant public health burden. Although hepatic involvement is a well-re
Core Tip: Dengue-associated acute liver failure is a severe but potentially reversible condition. With early recognition and prompt supportive management, many patients can achieve transplant-free survival. Understanding the key prognostic indicators and pathophysiological mechanisms is critical for optimizing outcomes, especially in resource-limited, dengue-endemic regions where access to liver transplantation may be restricted.
- Citation: Madhwani A, Chaudhry MQ, Rabbani JA, Kakar WG, Ayesha S, Abid S. Transplant-free survival in dengue-associated acute liver failure: Current evidence and future directions. World J Virol 2026; 15(1): 117008
- URL: https://www.wjgnet.com/2220-3249/full/v15/i1/117008.htm
- DOI: https://dx.doi.org/10.5501/wjv.v15.i1.117008
Dengue virus (DENV) infection remains a major public health challenge in tropical and subtropical regions, with an estimated 400 million infections occurring annually and billions of people at risk[1]. While the classic clinical spectrum includes febrile illness, hemorrhagic complications, and shock, hepatic involvement is a well-recognized but underappreciated facet of dengue pathophysiology[2]. Most patients with dengue exhibit transient elevations of serum aminotransferases, but a small subset develop severe hepatic injury or, more rarely, acute liver failure (ALF)[3,4].
ALF due to dengue is exceedingly uncommon, but when it occurs, it carries extremely high morbidity and mortality due to superimposed multi organ dysfunction, hypo perfusion, and systemic inflammatory injury[5]. The standard of care in non-infectious ALF often includes urgent liver transplantation; however, in dengue-associated ALF, transpla
Despite dispersed case reports and small case series, there is no comprehensive synthesis of transplant free survival outcomes, prognostic indicators, or management strategies in dengue-associated ALF. Existing literature is hampered by heterogeneity, publication bias, and lack of systematic follow up. Moreover, therapies such as N-acetylcysteine (NAC), plasma exchange, or extracorporeal liver support have been used but without clear evidence-based guidance[8,9].
This review aims to: (1) Summarize the available literature on dengue-associated ALF with a focus on transplant-free survival rates; (2) Examine clinical, biochemical, and therapeutic predictors of survival; (3) Critically assess the role of emerging adjunctive therapies; and (4) Propose directions for future research particularly prospective registries or multicenter collaborations for better clinicians’ guidance working in resource constrained settings.
This narrative review aimed to synthesize current evidence on dengue-associated ALF, with a particular focus on transplant-free survival. A comprehensive literature search was conducted using the PubMed database to identify peer reviewed articles published in English. The search strategy employed multiple keywords and Medical Subject Headings terms, including, but not limited to “dengue”, “dengue complications”, “dengue acute liver failure”, “dengue survival”, “dengue transplant”, “dengue mortality”, and “plasma exchange”, among others. Reference lists of relevant articles were also screened to identify additional studies. Studies were selected based on their relevance to the pathophysiology, clinical outcomes, management strategies, and survival outcomes especially transplant-free survival of dengue-associated ALF.
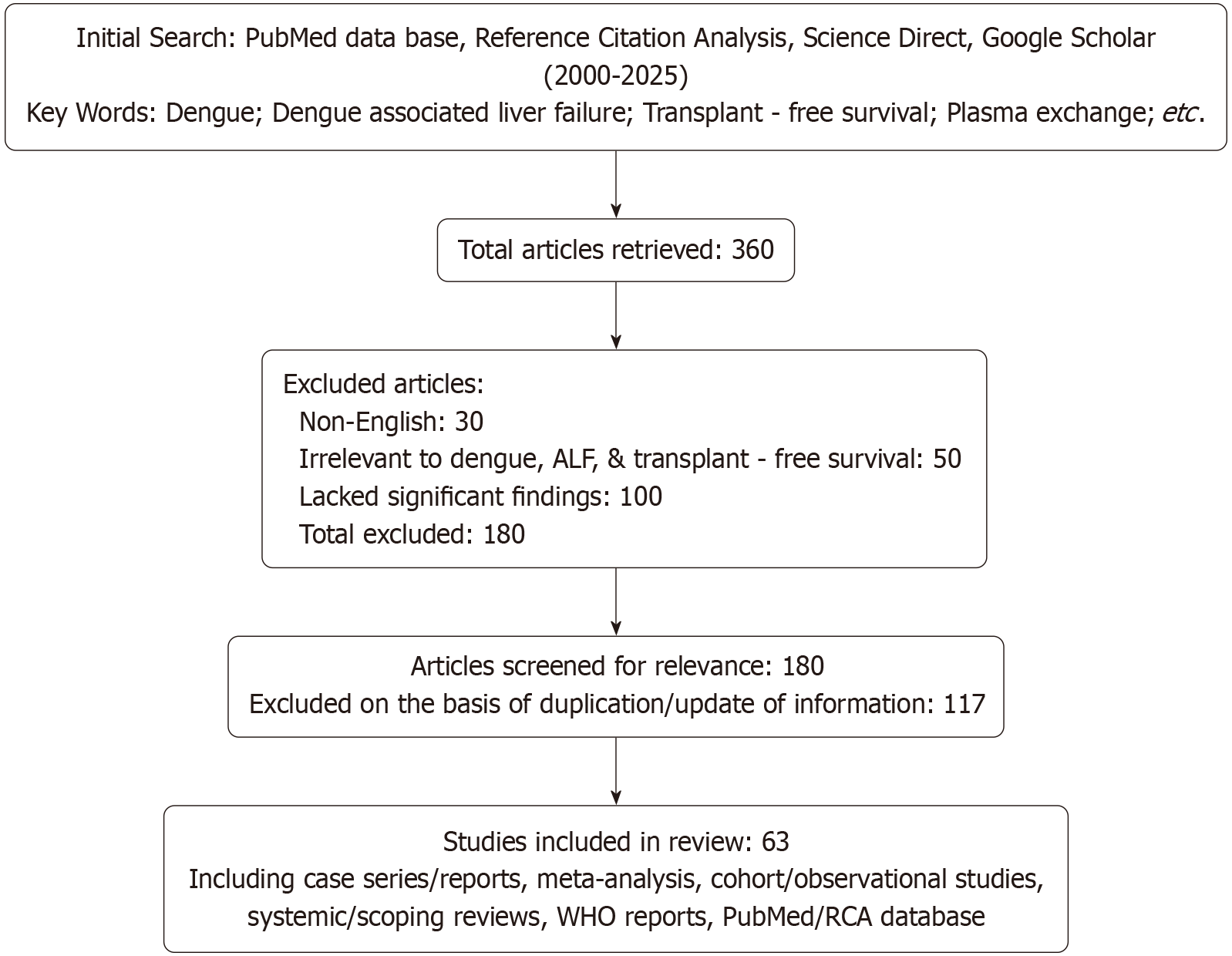
Initially, a total 360 articles were retrieved. After screening, a total 180 articles were excluded based on following exclusion criteria: (1) Non-English language publications; (2) Irrelevant focus (i.e., not addressing ALF and transplant free survival); and (3) Articles lacking significant pathophysiological or clinical relevance. This resulted in 180 articles that were screened in detail, from which 63 studies were included in the final review, after eliminating duplicated and outdated information (Figure 1).
This review included original research articles (prospective and retrospective cohort studies), meta-analyses, systematic reviews, clinically relevant case reports, and basic or translational studies focusing on dengue-associated ALF and its clinical outcomes, particularly transplant-free survival, published from 2000 onwards. Studies reporting the incidence, pathophysiology, management strategies, and liver transplantation outcomes in dengue-related ALF were selected for inclusion. Reference lists of eligible studies were also manually screened for additional relevant articles. Due to the narrative nature of this review, no formal quality assessment or risk-of-bias analysis was performed. Despite thorough efforts to capture all pertinent literature, some eligible studies may have been inadvertently excluded.
Rapidly evolving into a major global health concern, dengue has been ranked amongst the top ten infections by the World Health Organization (WHO), as of 2019. The substantial threat to public health is backed by the fact that approximately 40% of the world’s population is at risk, estimating a rate of 390 million infections/year[10,11]. And it isn’t confined to tropical and subtropical regions any more, due to urbanization, globalization, and climate change[12].
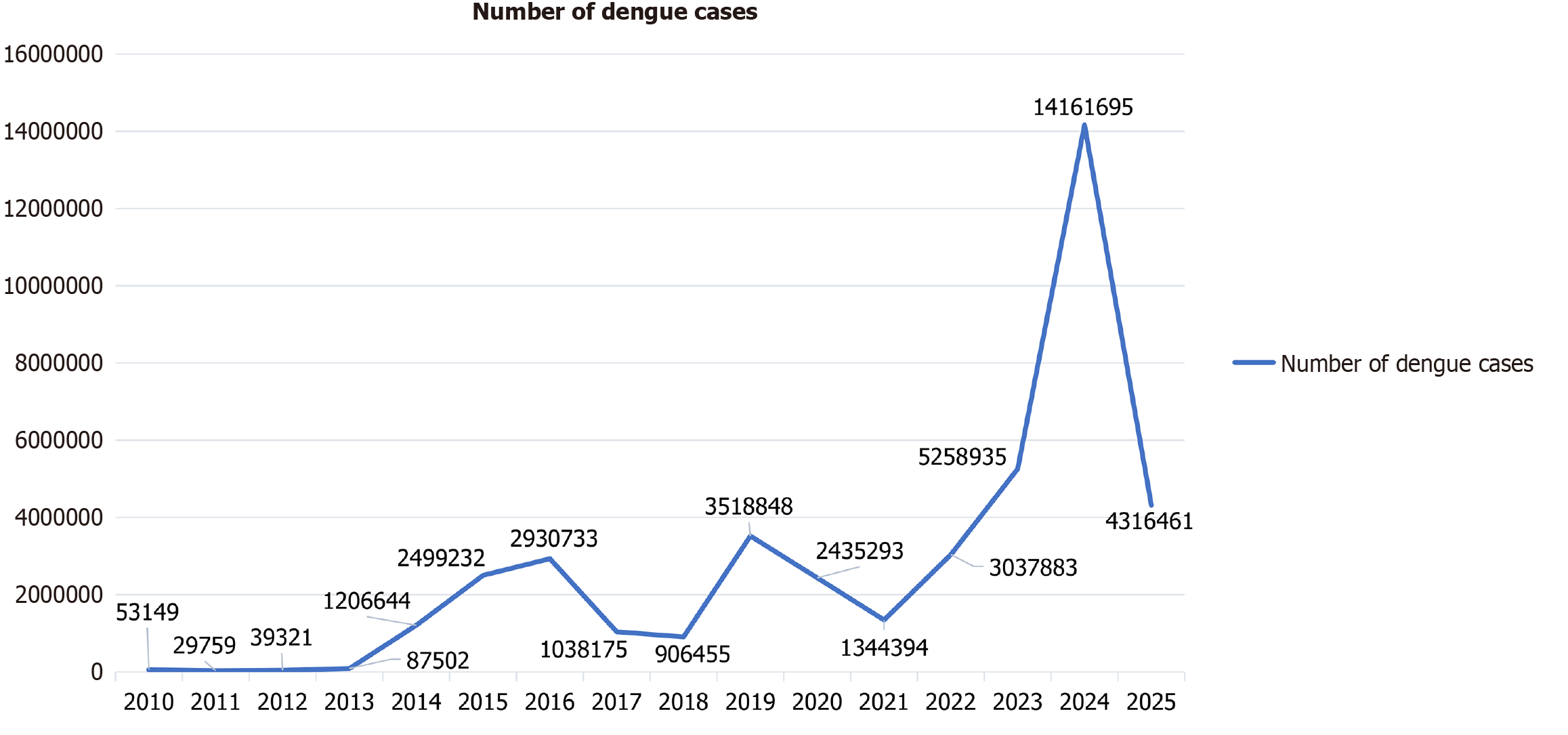
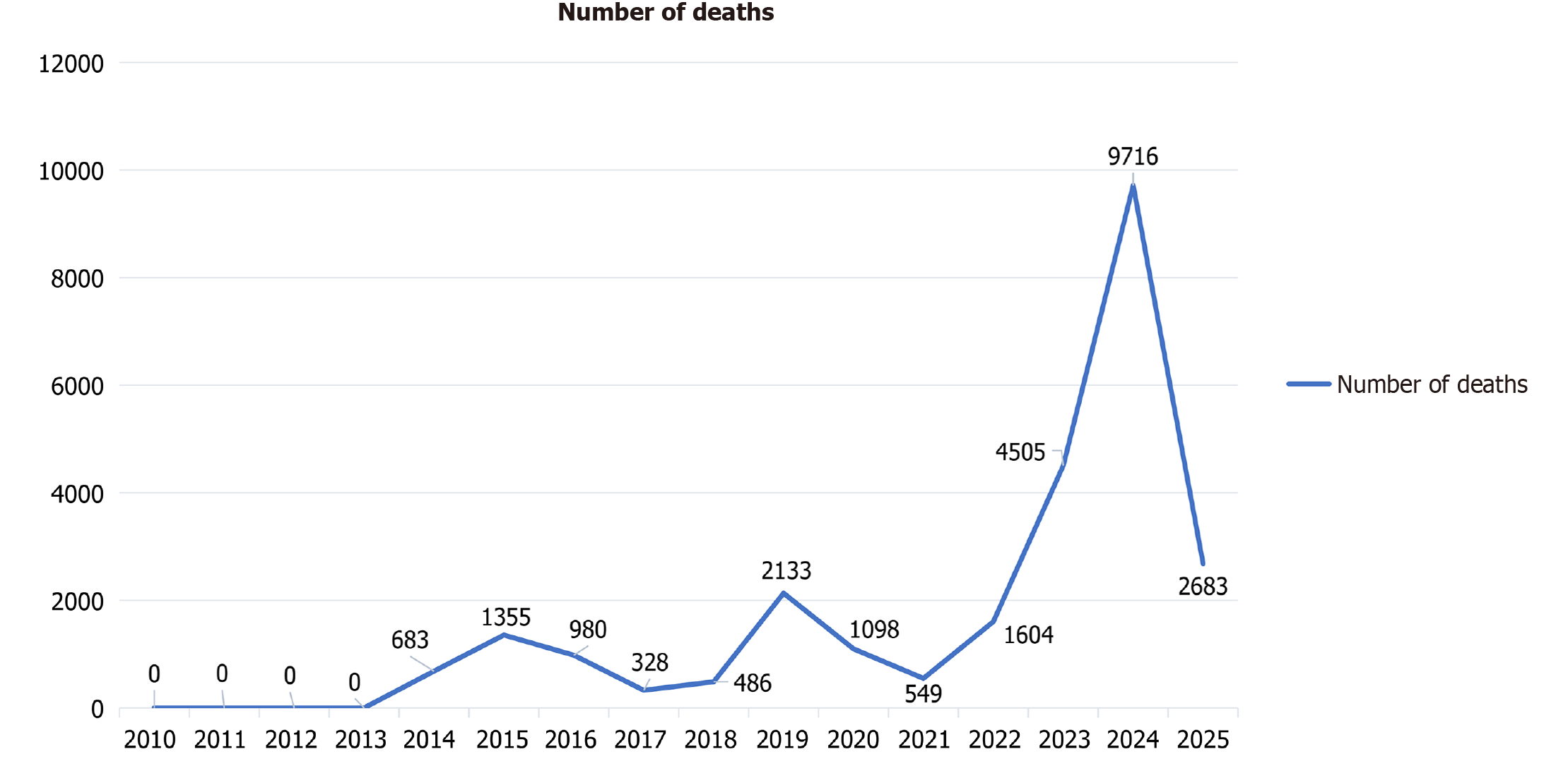
Dengue has had an unprecedented scale of global expansion. Considering the time period from 1990 to 2021, global dengue incidence has more than doubled, rising from 26.45 million cases to 58.96 million. Mortality figures have increased in a similar pattern, starting at 14315 to 29075 deaths[13]. Figure 2 shows the number of dengue cases from 2010 to 2025, while Figure 3 reveals the mortality over the same period of time. 2019, alone showed outbreaks in 129 docu
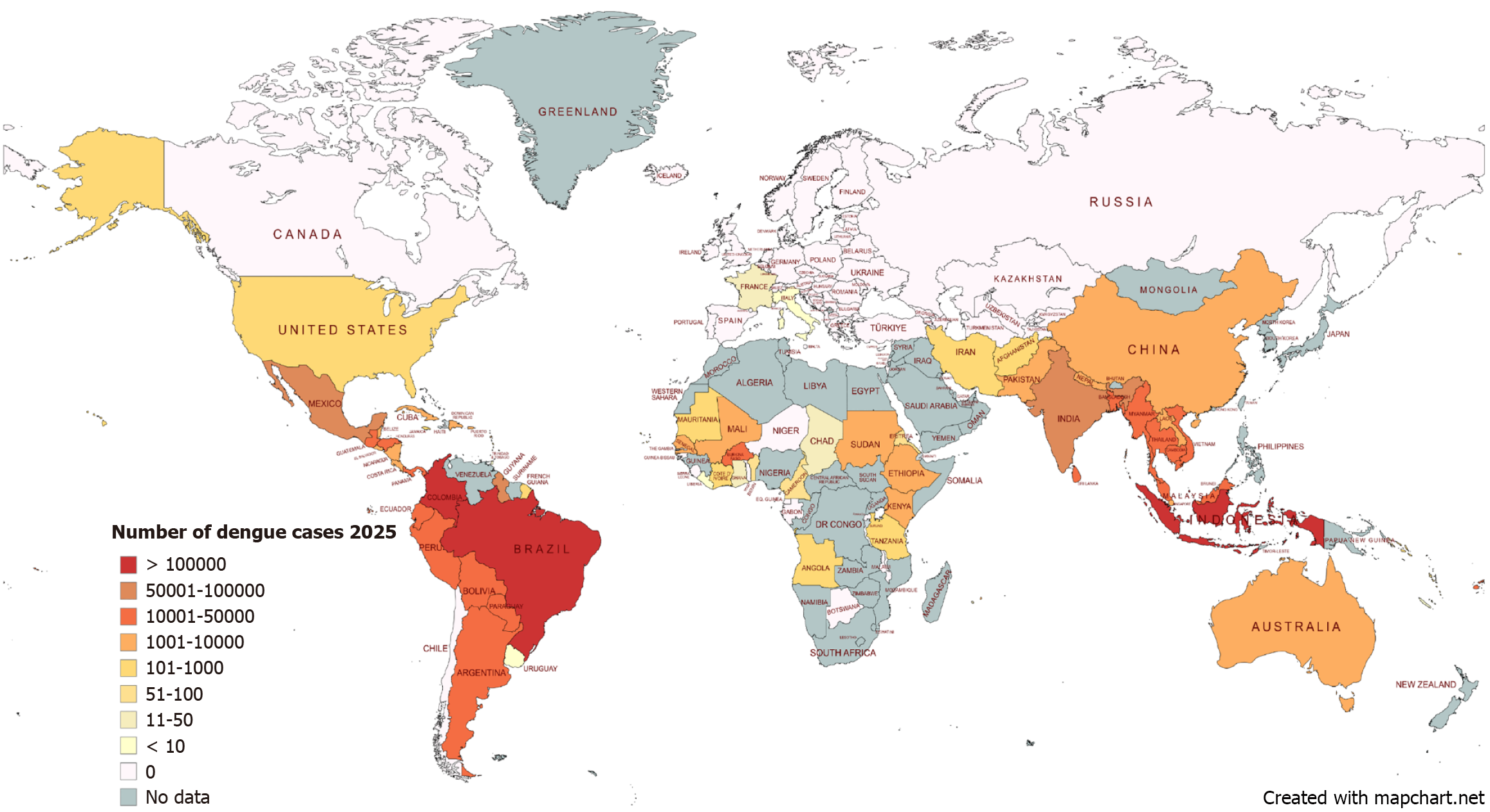
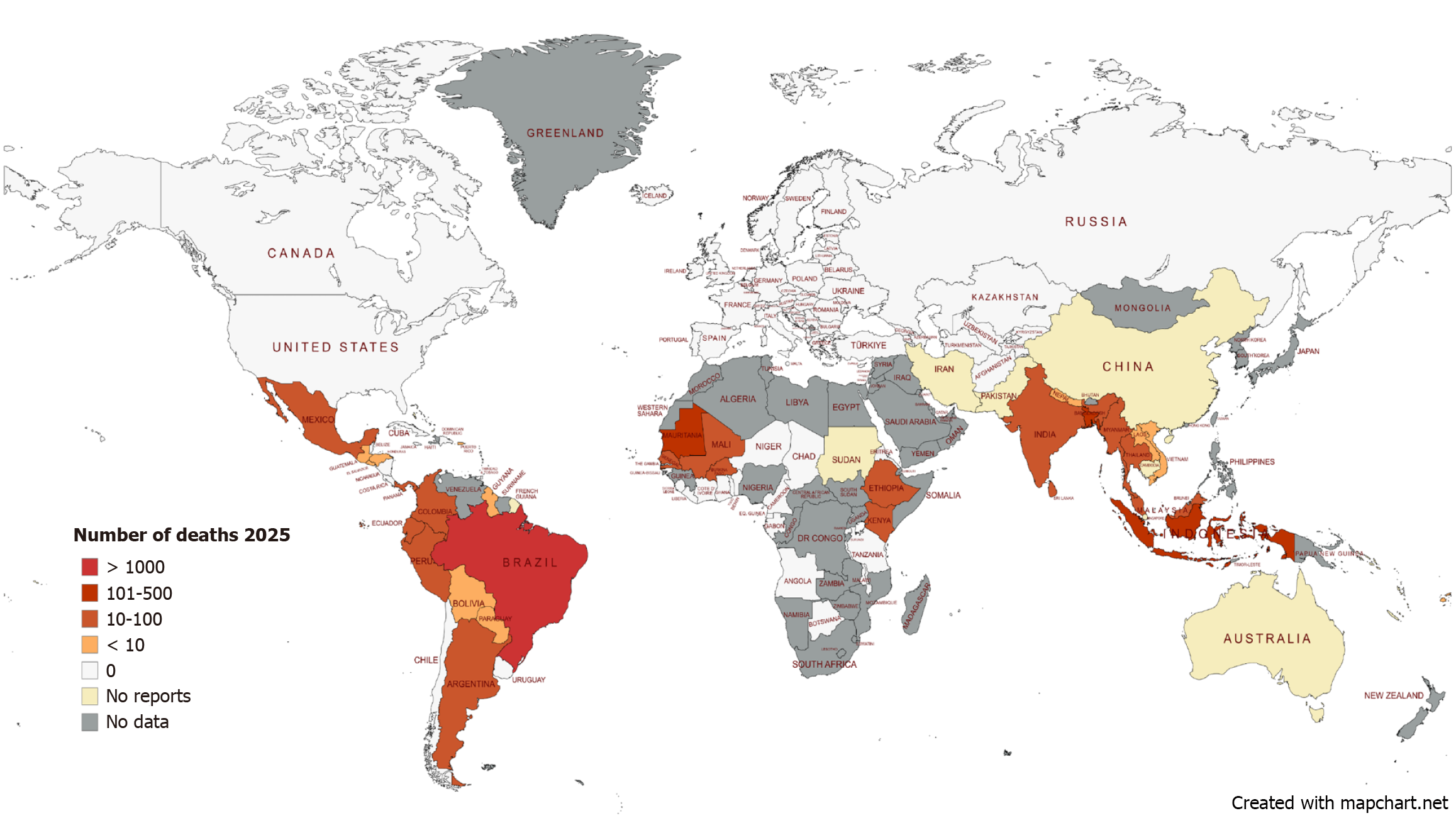
The heaviest burden of dengue continues to target the regions of South and Southeast Asia, as well as the Americas, in particular tropical Latin America. WHO reports, of December 2023, revealed the highest cases and mortality burden in America and Southeast Asia, with approximately 2049 deaths over 4.1 million cases (at an incidence rate of 419 cases per 100000 population), and 1800 deaths over 500000 cases respectively[14]. Comparing this to 2024, America and Southeast Asia have alone reported over 7000 deaths in 12.67 million cases and over 2000 deaths in approximately 700000 cases respectively[16], showing the rapid increase in dengue cases. Western Pacific showed a similar number of cases as Southeast Asia in 2023, with mortality only being 750 deaths. This mortality is mirrored in the African region, which had 171991 cases. The Eastern Mediterranean region showed smaller burdens, with Pakistan alone accounting for more than 20000 cases; however, the death toll was suspected to be lower, but isn’t documented. Europe has had the lowest incidence, with only 128 confirmed cases, and only 1 reported death in Italy[14]. Distribution of dengue cases and related mortality in 2025 is outlined in the maps provided in Figures 4 and 5.
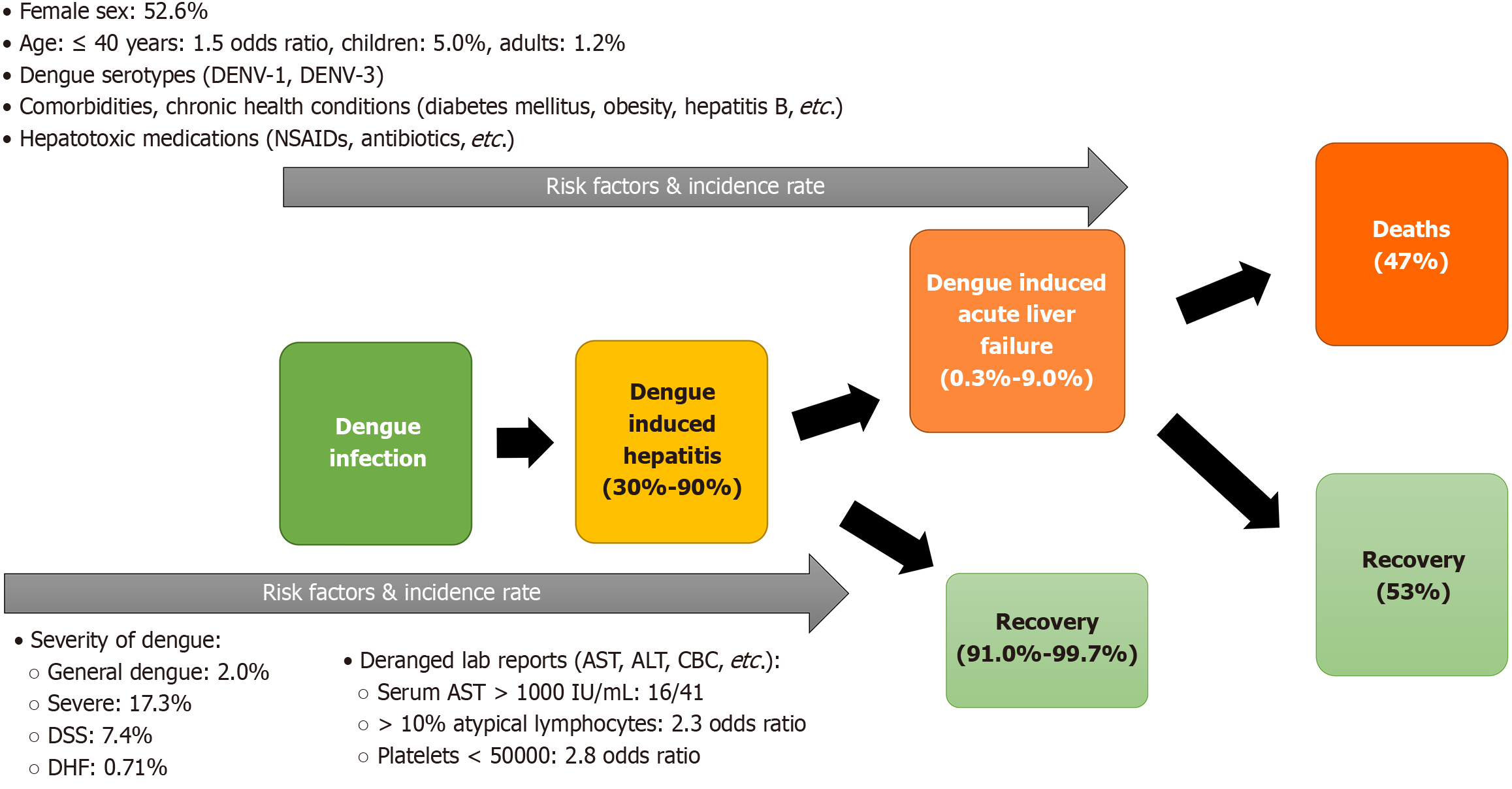
ALF is one of the most severe complications of dengue and is life threatening though rare. Reported incidence rates vary from 0.3% to 4% depending on the population and study. In Thailand, multiple studies have reported the incidence of ALF among hospitalized dengue patients. A 2016 study reported 0.31% in 1926 patients hospitalized between 2011-2015[17]. Another study in 2020, revealed the incidence to be 0.64% among 1484 hospitalized patients[6,18]. A smaller Thailand study observed 1 case of ALF among 95 hospitalized dengue patients, resulting in an incidence of approximately 1.05%[19]. Recently, a 2024 meta-analysis found the incidence to range from 0.31% to 3.68%, with a 50%-70% mortality rate[18]. In India, a study of 93 hospitalized adults reported 1 case of ALF, revealing an incidence of 1.1%[20]. In Sri Lanka, 16 out of 240 dengue patients developed ALF, resulting in a 6.7% incidence[21]. Globally, of 21 studies of 26839 dengue patients found an ALF incidence of 2.0% [95% confidence interval (CI): 1.2%-3.0%], with 1.2% in adults and 5.0% in children. The incidence was notably higher in severe dengue (17.3%) and dengue shock syndrome (DSS; 7.4%), and the combined mortality rate reached 47% (95%CI: 32.9%-61.2%)[20]. Although ALF in dengue is rare, the resulting consequences are severe. Case series have reported. ALF rates as high as 5%-9%, while larger retrospective studies reported a 0.3%-1.1% incidence. Data from Thailand and India reported 0.6%-0.8%. Once ALF occurs, the prognosis is grim, with a 47% pooled mortality and individual studies reporting 50%-68% mortality compared to 0.8% in severe hepatitis without ALF. These findings show that although rare, ALF is a critical turning point in dengue infection, so early detection and close monitoring of liver function is important[6,20].
Although ALF is a rare complication of dengue infection, the risk is notable especially among certain high-risk populations. A prospective Sri Lankan study involving 240 dengue patients demonstrated that 16 patients of 41 patients with serum aspartate aminotransferase levels exceeding 1000 IU/L progressed to ALF, underscoring the strong correlation between markedly elevated transaminases and liver failure[21]. Similarly, a 2016 Thailand study found that severe dengue significantly increased the risk of ALF particularly in young adults aged ≤ 40 years. Additional clinical predictors identified included a > 10% proportion of atypical lymphocytes and platelet counts < 5,000/mm3[17].
A 2024 meta-analysis further revealed that female patients have a higher risk of dengue-associated ALF, with an incidence of 52.6%. The presence of comorbidities such as diabetes mellitus, hypertension, coronary artery disease, dyslipidemia, alcoholic liver disease, obesity, and chronic hepatitis B also heightened susceptibility[1,22,23]. Moreover, hepatotoxic medications such as paracetamol, nonsteroidal anti-inflammatory drug, and specific antibiotics may amplify the risk by compounding liver injury[1].
Disease severity plays a critical role: The incidence of ALF is approximately 2.0% in uncomplicated dengue, rising sharply to 17.3% in severe dengue and 7.4% in DSS[18,24]. Children are particularly vulnerable, with an ALF incidence of 5.0% in pediatric dengue cases compared to 1.2% in adults[18,25,26]. While all dengue serotypes can precipitate ALF, DENV-1 and DENV-3 are most frequently implicated; notably, DENV-3 has a stronger association with liver injury in children[22,26]. A summary of risk factors and their reported incidence, and incidence of complications are shown in Figure 6.
Temporal trends (outbreak years vs endemic periods): Dengue incidence continues to rise globally, now endemic in over 100 countries, with outbreaks becoming more frequent and geographically widespread. Although epidemics typically follow 3-5-year cycles, overall case numbers have increased steadily in recent decades, often involving co-circulation of multiple serotypes that heighten the risk of severe disease and complications. WHO data from 2023-2025 highlight regional disparities: In the Americas, Eastern Mediterranean, Africa, and South-East Asia remain high-burden zones, while Europe reports only sporadic, mostly travel related cases. In Africa, Burkina Faso accounted for most of the 2023 cases across 11 affected nations. In the Americas, Brazil contributed approximately of 83% of regional cases, with 6.3 million suspected infections by mid-2024, surpassing the 2023 total; Colombia recorded the most severe presentations. Pakistan, Saudi Arabia, and Oman experienced major outbreaks in 2023, and Pakistan saw a resurgence in 2025, though below 2022-2024 peaks. In South-East Asia, dengue spiked in Bangladesh, Thailand, Nepal, and India in 2023, while Sri Lanka peaked in 2017 and 2019 before declining. In the Western Pacific, Fiji, the Philippines, and Viet Nam reported prolonged epidemics during 2023[13,27,28]. During non-epidemic years, the incidence of dengue associated ALF remains low, typically approximately 0.2%-0.5%. Accurate estimation of ALF prevalence in dengue remains challenging due to substantial reporting and methodological limitations. Although dengue is widespread, ALF is rare, and existing data are constrained by under-reporting, heterogeneous case definitions, and inconsistent surveillance. Many studies include only hospitalized or severe cases, limiting generalizability. Inadequate laboratory capacity, overlap with other febrile illnesses (e.g., malaria, chikungunya), and delays in disease confirmation further obscure true incidence. Systemic barriers including insufficiently trained personnel, inconsistent reporting systems, and vector resistance compound these chal
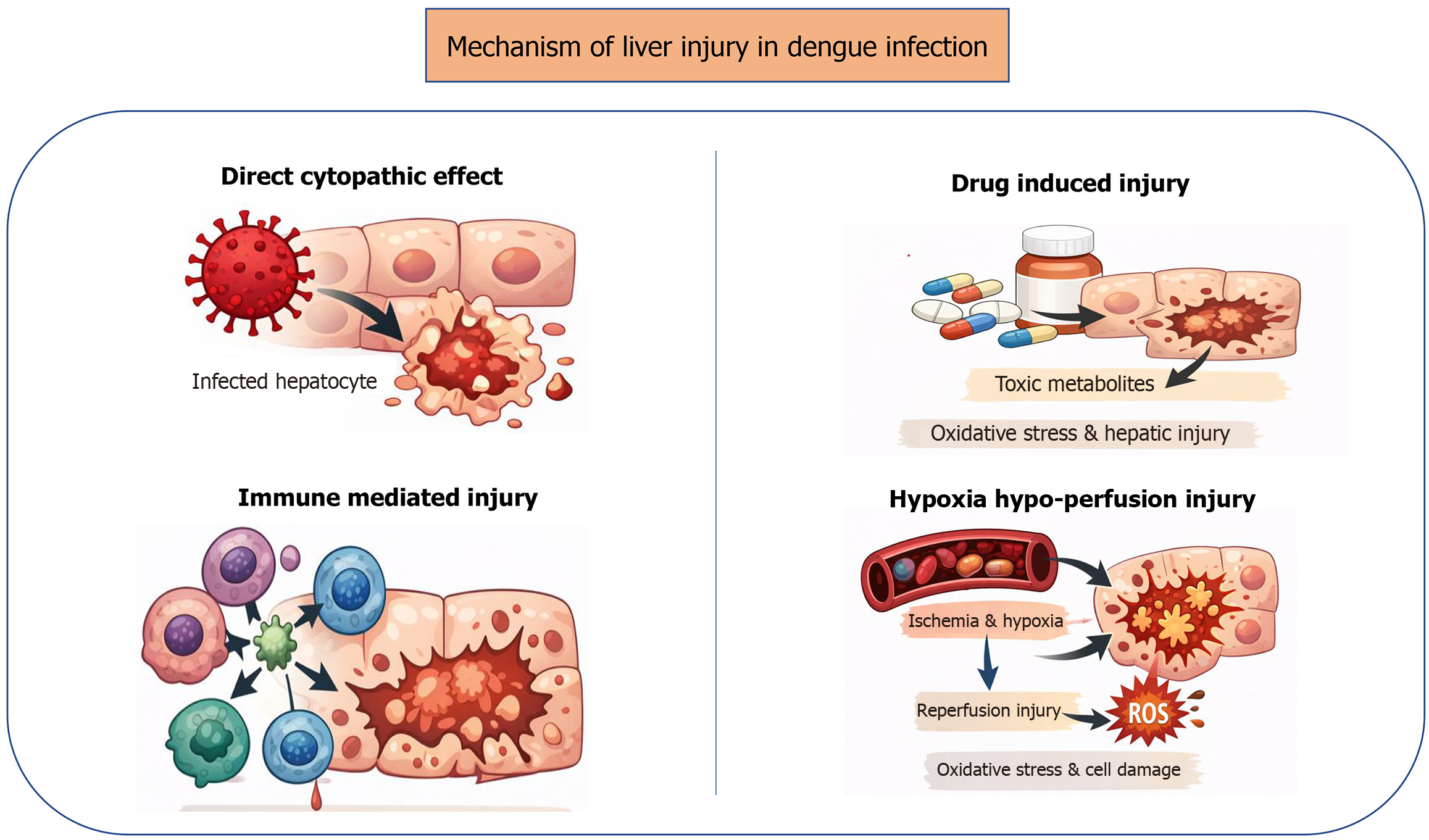
Pathophysiology: The primary mechanism of action of the single-stranded positive-sense RNA flavivirus causing dengue[30], is by invading different cells of the human body, especially the immune system. However, the damage isn’t limited to this, other cells and response systems of the human body are also affected, causing damage to other organs and organ systems, which can result in organ failure. One such organ, is the liver, damage to which results in liver injury, which may escalate to the point of causing ALF. This damage may be direct, due to the virus invading the hepatocytes itself, or indirectly, as a result of the immune response sent by the body[31]. Other methods resulting in dengue-induced ALF include ischemia, hypo perfusion, and drug induced injuries.
Direct cytopathic effect: Direct invasion of the virus into the hepatocytes and Kupffer cells[32], causes major damage by binding to the receptors present on the cell membrane of the host cell, using the E protein[32], and replicates inside the cells, which results in apoptosis (cell death)[33], mitochondrial dysfunction, and endoplasmic reticular stress. This results in the presence of apoptotic Councilman bodies[27], something also seen in yellow fever[1]. Postmortem studies have confirmed that the primary sites for replication are the hepatocytes and Kupffer cells[1,22,34]. NAC therapy is a possible way to repair said damage (as explained below).
Immune mediated injury: The immune response of the body, as a reaction to the foreign organism, can damage the normal cells within the body, especially that of the liver, which, if left untreated, may result in ALF. This may happen due to dysregulation of the immune response. Elevated cytokines such as tumor necrosis factor-α, interleukin (IL)-6, IL-10, IL-17, IL-22, and IP-10, can trigger hepatocyte apoptosis and amplify liver destruction, while increased viral loads can exacerbate the damage. Immature CD4+ T cell and CD8+ T cell, along with natural killer cells, can end up infiltrating the liver, worsening the trauma. Antibody-dependent enhancement may worsen the situation, resulting in permitting more viral entry, further fueling the inflammatory response, and causing plasma leakage[18]. This leakage also occurs due to microvascular dysfunction, which occurs as a result of endothelial cell activation and glycocalyx degradation[35]. Experimental studies show that blocking IL-17 reduces hepatic injury, supporting the importance of immune-driven pathways, and the role they play in causing liver injury (Figure 1)[1,22,23,33,36]. Extracorporeal liver support, such as therapeutic plasma exchange (TPE), is another way of removing said cytokines (explained in more detail below), while NAC therapy can have anti-inflammatory effects.
Ischemia and hypo perfusion: Hemodynamic factors also play a role in causing ALF, by causing myocardial dysfunction and collapse of circulatory pathways. Hypotension and reduced hepatic perfusion can lead to ischemic liver damage, especially in patients DSS[1]. Hypoxia is another cause of liver damage[28], which is a result of swelling of the endothelial cells, as well as microvascular destruction, as well as compromise of the circulation, or shock[31]. Multiorgan failure in dengue, especially in its severe forms, occurs due to increased vascular permeability, myocardial dysfunction, and dehydration[37]. Autopsies have revealed portal vein congestion, reduced portal blood flow, and reversal of the blood flow direction (hepato-fugal flow) in some cases[31,38]. However, it isn’t necessary for shock to cause patients with dengue to develop severe liver destruction[33]. Microvascular and immune mechanisms can cause injury independently as well[22]. Systemic complications, such as disseminated intravascular coagulation, coagulopathy, and electrolyte imbalance, may also result in aggravated liver damage in patients with severe dengue, while hypoglycemia may also result in complications[31]. Biopsies and autopsies of livers in ALF have revealed supportive findings to all of the above causes of severe liver destruction. Multi-lobular massive, focal, or patchy necrosis has been seen[33,38,39], with destroyed, or hyperplasic Kupffer cells[32,40]. Apoptotic hepatocytes have been noted to be present in clusters[32]. Portal vein congestion, decreased flow, and hepato-fugal flow have also been noted in some specimens. Midzonal and centrilobular necrosis has been seen in histopathological studies[34]. Micro-vesicular steatosis, sinusoidal congestion, and Councilman bodies have also been seen[32]. Cholestasis has also been seen in some cases[39]. However, unlike most liver diseases, fibrosis is not a common finding in livers affected by ALF due to dengue[4]. TPE is a possible method to im
Drug induced injury: Usage of hepato-toxic medicines, such as paracetamol (acetaminophen), certain antibiotics[13,33], and herbal preparations for symptomatic treatment of fever and body aches[4], are known to cause hepatocellular injury and worsen hypoxia. When combined with other pathological mechanisms, such drug-induced liver injury can progress to ALF. Various mechanisms by which DENV causes liver injury is shown in Figure 7.
Management of dengue-associated ALF remains primarily supportive, as no antiviral agent directly targeting the DENV exists. Early detection, vigilant monitoring, and prompt critical care interventions are essential to prevent rapid progression to multiorgan failure. Hemodynamic stabilization should be carefully balanced to avoid fluid overload and cerebral edema. Correction of metabolic derangements including glucose, electrolytes, and acid base imbalances is vital. Hyponatremia should be avoided to mitigate cerebral edema, and hypoglycemia prevented through continuous glucose infusion. Coagulopathy is corrected only when bleeding is clinically significant. Invasive intracranial pressure monitoring may be considered for advanced encephalopathy. Multiorgan support often requires mechanical ventilation, vasopre
NAC has gained attention as a potential adjunctive therapy in non-acetaminophen ALF, and by extension, in dengue related cases. Its proposed mechanisms include glutathione replenishment, improved hepatic microcirculation, and antioxidant effects. However, while randomized trials in non-acetaminophen ALF demonstrate improved transplant free survival with early NAC the evidence in dengue ALF is limited to small, non-randomized case series.
Sriphongphankul et al[42] observed higher survival when NAC was initiated early in Thai pediatric ALF, and Ro
Extracorporeal liver support techniques most notably TPE have been evaluated as a bridge to hepatic recovery or transplantation. TPE removes cytokines, bilirubin, and ammonia, and may transiently improve hemodynamics. However, the evidence base is exceedingly weak. A 2025 systematic review identified only nine dengue ALF cases treated with TPE, most showing biochemical improvement but without standardized endpoints or long term follow up[36].
Existing studies are largely descriptive, with no comparative analyses against standard care. While the high-volume plasma exchange trial by Larsen et al[44] demonstrated improved survival in non-dengue ALF through modulation of systemic inflammation, extrapolating these findings to dengue ALF remains speculative due to differing pathophysiology and disease kinetics.
Consequently, TPE should be viewed as an experimental adjunct, best reserved for early-stage patients in centers with experience in extracorporeal support. Data supporting an effect on transplant-free survival remain anecdotal and hy
Liver transplantation remains the definitive therapy for irreversible hepatic failure, but its role in dengue ALF is highly restricted and poorly characterized. Only isolated successful cases have been reported[44,45], with the majority of published data being anecdotal and potentially biased toward favorable outcomes. Severe coagulopathy, shock, and ongoing viremia significantly increase perioperative risk, and donor scarcity in endemic settings further limits feasibility. No prospective data exist comparing survival with vs without transplantation.
Therefore, transplantation should be considered only in rare, highly selected cases where viremia has cleared and multiorgan failure is absent. For the majority, aggressive supportive therapy, early NAC use, and selective TPE remain the most practical strategies to enhance transplant free survival in resource limited regions.
Risk stratification in dengue-associated ALF remains challenging, as existing models were not developed in this context. Traditional prognostic frameworks such as the King’s College criteria (KCC), Acute Physiology and Chronic Health Evaluation II (APACHE II), and the Model for End-Stage Liver Disease (MELD) have been applied with variable success.
While KCC developed originally for acetaminophen and non-acetaminophen ALF, remains widely used to identify poor outcomes and the need for transplantation, incorporating parameters such as advanced encephalopathy, international normalized ratio > 6.5, renal dysfunction, and severe acidosis[46]. APACHE II, although shown to be useful in acetaminophen-related ALF (with scores > 15 identifying high-risk patients)[47] has limited applicability in viral or dengue-associated ALF. This is because the score primarily reflects the burden of multiorgan failure rather than liver-specific dysfunction. As highlighted in a recent review, while APACHE II (and Sequential Organ Failure Assessment) can predict overall mortality in critical illness, they are not optimal for guiding transplant decisions in ALF[48].
By contrast, MELD appears more robust, in recent dengue cohorts a comparative analysis of prognostic models was done, where MELD score performed best for predicting both the development of ALF and patient survival. A cutoff of 16 predicted progression to ALF with high accuracy (sensitivity approximately of 88%, specificity approximately of 89%), while a cutoff of 18 predicted mortality with similarly strong performance (78.6% sensitivity, 87.7% specificity)[49].
Beyond scoring systems, laboratory parameters provide important prognostic signals. Coagulopathy, reflected by elevated international normalized ratio and reduced factor V levels, is a hallmark of ALF and correlates with adverse outcomes[50,51]. Elevated arterial ammonia levels strongly correlate with the development of cerebral edema and poor prognosis[52]. Similarly, admission pH and lactate strongly predicted mortality[39].
Recent work has highlighted the role of emerging biomarkers in severe dengue. Elevated cytokine levels, including IL-6, IL-10, and tumor necrosis factor-α, have been associated with disease severity and immune dysregulation[53,54]. More recently, alterations in circulating microRNA profiles, particularly microRNA-122, have been linked to hepatic invo
Conventional prognostic systems such as KCC and the MELD offer predictive value in ALF. However, they insufficiently capture the multifactorial pathophysiology of dengue-associated ALF, which is driven by viral replication, im
Host genetic variation contributes to differences in dengue severity. Polymorphisms in major histocompatibility com
Dengue-associated ALF varies globally due to viral, host, and healthcare differences. Co-endemicity with chikungunya and Zika > 80% overlap within 50 kilometers of dengue transmission affects serotype behavior and disease severity[58]. A meta-analysis found approximately 2% ALF incidence and 47% mortality, higher in children (approximately 5%) and DSS (approximately 17%)[18]. Access to critical care is the main survival determinant; in a French West Indies intensive care unit, 27.8% developed ALF but mortality fell to approximately 22%[59]. Transplant experience remains anecdotal, limited to a few case reports with variable outcomes[60]. Management remains supportive; no dengue specific protocols or transplant criteria exist, despite WHO and regional guidance emphasizing early recognition and hemodynamic control[14,15,45,61]. Global survival gaps mirror disparities in intensive care access, not viral virulence. Climatic factors (rainfall, temperature, humidity), rapid urbanization, and vector density drive dengue transmission, while nutrition and host vulnerability further shape severity. These interacting forces influence both disease incidence and the risk of ALF, underscoring that outcomes reflect environmental exposure, host resilience, and healthcare capacity rather than viral virulence alone[62].
ALF secondary to dengue infection remains rare but carries a disproportionately high mortality compared with other viral or drug induced etiologies. A recent meta-analysis reported a pooled mortality of 47% (95%CI: 32.9%-61.2%) and an incidence of around 2% among hospitalized dengue cases[18]. Most deaths occur rapidly, within the first week of hepatic decompensation, due to cerebral edema, refractory shock, and multiorgan failure[39].
Transplant free survival is generally limited and largely confined to patients with early encephalopathy, preserved hemodynamics, and milder coagulopathy. Available data are derived predominantly from small retrospective cohorts or single center series, with significant heterogeneity in patient selection and definitions of ALF. In an Indian cohort, nearly all patients presenting with grade III-IV encephalopathy or circulatory collapse died despite maximal care[42]. Pediatric data are more favorable: A Thai study noted higher but not statistically significant transplant-free survival (75% vs 53%) among children treated with NAC, though the study was underpowered to detect a meaningful difference[45].
Evidence supporting NAC or plasma exchange remains low certainty, primarily based on small case series and lacking randomized comparison. While both interventions are biologically plausible and associated with biochemical impro
Liver transplantation has been attempted in only a handful of well-documented cases, typically involving carefully selected patients without ongoing viremia or hemodynamic instability[63]. Although isolated reports describe good short term graft function, outcomes are subject to strong publication bias, and overall feasibility in dengue endemic, resource limited regions remain extremely low. Active infection, coagulopathy, and organ failure are frequent contraindications, compounded by donor scarcity and limited infrastructure[41].
In summary, current evidence on survival in dengue associated ALF is limited, heterogeneous, and largely descriptive. Most patients achieve recovery if at all through early recognition, intensive supportive management, and empiric NAC therapy rather than transplantation. Well designed, multicenter prospective studies are essential to identify reliable prognostic indicators, standardize management, and clarify which patients may truly benefit from advanced inter
There is an imperative need for large-scale, prospective data collection to better understand the incidence, clinical course, and transplant free survival rates in dengue associated ALF. Multicenter collaborations across endemic regions can help provide robust, generalizable evidence.
Development of validated diagnostic criteria and prognostic scoring systems specific to dengue induced ALF would help clinicians in early recognition, risk stratification, and decision-making regarding advanced supportive or transplantation-based interventions.
Emerging data suggest that immunological pathways, oxidative stress, and direct viral cytopathic effects contribute to dengue-associated liver injury. Further molecular and translational research is needed to identify reliable biomarkers for early hepatic injury and potential therapeutic targets.
Currently, management of dengue-related ALF is largely supportive. Clinical trials evaluating liver support systems, immune modulating agents, antiviral therapies, and hepatoprotective drugs could significantly improve transplant-free outcomes.
Clinical settings with scarcity of resources often do not have access to liver transplantation and intensive care facilities. Investment in early detection systems, point of care diagnostics, telemedicine support, and critical care infrastructure is crucial for improving survival in dengue-associated ALF without transplantation.
Vector control, community education, and vaccination efforts must be prioritized to reduce the burden of severe dengue disease. Preventing progression to ALF starts with effective control of dengue transmission and timely management of early complications.
Dengue associated ALF is rare but often fatal, demanding rapid recognition and intensive supportive care. Yet, timely diagnosis and meticulous management of encephalopathy, coagulopathy, and fluid balance can enable transplant free recovery in many patients. Because dengue-related hepatic injury is often potentially reversible, continuous clinical reassessment is crucial to determine when transplantation is truly necessary. Evidence remains scarce and largely descriptive, underscoring the need for prospective registries, validated prognostic tools, and dengue specific manage
| 1. | Juneja D, Jain R, Nasa P. Dengue induced acute liver failure: A meta summary of case reports. World J Virol. 2024;13:91457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 2] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (6)] |
| 2. | Campana V, Inizan C, Pommier JD, Menudier LY, Vincent M, Lecuit M, Lamballerie X, Dupont-Rouzeyrol M, Murgue B, Cabié A. Liver involvement in dengue: A systematic review. Rev Med Virol. 2024;34:e2564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (1)] |
| 3. | Arora S, Nathaniel SD, Paul JC, Hansdak SG. Acute liver failure in dengue haemorrhagic fever. BMJ Case Rep. 2015;2015:bcr2015209443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (1)] |
| 4. | Leite G. Research on tuberculosis: 75 years of IBIT contributions. Braz J Infect Dis. 2013;17:205-210. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (1)] |
| 5. | Dissanayake DMDIB, Gunaratne WMSN, Kumarihamy KWMPP, Kularatne SAM, Kumarasiri PVR. Use of intravenous N-acetylcysteine in acute severe hepatitis due to severe dengue infection: a case series. BMC Infect Dis. 2021;21:978. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (1)] |
| 6. | Teerasarntipan T, Chaiteerakij R, Komolmit P, Tangkijvanich P, Treeprasertsuk S. Acute liver failure and death predictors in patients with dengue-induced severe hepatitis. World J Gastroenterol. 2020;26:4983-4995. [PubMed] [DOI] [Full Text] |
| 7. | Polpichai N, Saowapa S, Wattanachayakul P, Danpanichkul P, Trongtorsak A, Chan SY, Choudhury A, Kaewdech A. Role of Plasma Exchange and Combining Therapies in Dengue-Associated Acute Liver Failure: A Systematic Review of Individual Cases. J Clin Exp Hepatol. 2025;15:102407. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 8. | Nakaviroj S. First case report of high volume therapeutic plasma exchange as a rescue therapy in dengue hemorrhagic fever with acute liver failure. Chulalongkorn Med J. 2021;65:335-338. [DOI] [Full Text] |
| 9. | Galante A, Adeyi O, Lau L, Humar A, Galvin Z, Selzner N, Lilly L, Sapisochin G, Bhat M. Liver Transplantation for Acute Liver Failure Due to Dengue Fever. Hepatology. 2019;70:1863-1865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (2)] |
| 10. | World Health Organization. Ten threats to global health in 2019. 10 Jan 2019. Available from: https://www.who.int/news-room/spotlight/ten-threats-to-global-health-in-2019. |
| 11. | Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wint GR, Simmons CP, Scott TW, Farrar JJ, Hay SI. The global distribution and burden of dengue. Nature. 2013;496:504-507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7940] [Cited by in RCA: 6575] [Article Influence: 505.8] [Reference Citation Analysis (2)] |
| 12. | Li L, Guo X, Zhang X, Zhao L, Li L, Wang Y, Xie T, Yin Q, Jing Q, Hu T, Li Z, Wu R, Zhao W, Xin SX, Shi B, Liu J, Xia S, Peng Z, Yang Z, Zhang F, Chen XG, Zhou X. A unified global genotyping framework of dengue virus serotype-1 for a stratified coordinated surveillance strategy of dengue epidemics. Infect Dis Poverty. 2022;11:107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 13. | Zhang WX, Zhao TY, Wang CC, He Y, Lu HZ, Zhang HT, Wang LM, Zhang M, Li CX, Deng SQ. Assessing the global dengue burden: Incidence, mortality, and disability trends over three decades. PLoS Negl Trop Dis. 2025;19:e0012932. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 37] [Article Influence: 37.0] [Reference Citation Analysis (2)] |
| 14. | World Health Organization. Dengue - Global Situation. Dec 21 2023. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2023-DON498. |
| 15. | Venkatesan P. Global upsurge in dengue in 2024. Lancet Infect Dis. 2024;24:e620. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 16. | World Health Organization. Global dengue surveillance. [cited 15 October 2025]. Available from: https://worldhealthorg.shinyapps.io/dengue_global/. |
| 17. | Kye Mon K, Nontprasert A, Kittitrakul C, Tangkijvanich P, Leowattana W, Poovorawan K. Incidence and Clinical Outcome of Acute Liver Failure Caused by Dengue in a Hospital for Tropical Diseases, Thailand. Am J Trop Med Hyg. 2016;95:1338-1344. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (2)] |
| 18. | Wongtrakul W, Charatcharoenwitthaya K, Karaketklang K, Charatcharoenwitthaya P. Incidence of acute liver failure and its associated mortality in patients with dengue infection: A systematic review and meta-analysis. J Infect Public Health. 2024;17:102497. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (35)] |
| 19. | Laoprasopwattana K, Jundee P, Pruekprasert P, Geater A. Outcome of Severe Dengue Viral Infection-caused Acute Liver Failure in Thai Children. J Trop Pediatr. 2016;62:200-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 20. | Kulkarni AV, Choudhury AK, Premkumar M, Jain P, Gupta E, Sarin SK. Spectrum, Manifestations and Outcomes of Dengue Infection in Individuals with and without Liver Disease. J Clin Transl Hepatol. 2019;7:106-111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 21. | Kumarasena RS, Niriella MA, Ranawaka CK, Miththinda JK, de Silva AP, Dassanayaka AS, de Silva HJ. Predicting acute liver failure in dengue infection. Ceylon Med J. 2016;61:35-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.9] [Reference Citation Analysis (3)] |
| 22. | Leowattana W, Leowattana T. Dengue hemorrhagic fever and the liver. World J Hepatol. 2021;13:1968-1976. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 49] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 23. | Nazeer A, Rai AA, Luck NH, Tasneem AAY. Dengue Hemorrhagic Fever: ARare Cause of Acute Liver Failure. J Coll Physicians Surg Pak. 2017;27:511-513. [PubMed] |
| 24. | Ling LM, Wilder-Smith A, Leo YS. Fulminant hepatitis in dengue haemorrhagic fever. J Clin Virol. 2007;38:265-268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 2.9] [Reference Citation Analysis (2)] |
| 25. | Poovorawan Y, Hutagalung Y, Chongsrisawat V, Boudville I, Bock HL. Dengue virus infection: a major cause of acute hepatic failure in Thai children. Ann Trop Paediatr. 2006;26:17-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (1)] |
| 26. | Osorio J, Carvajal C, Sussman O, Buitrago R, Franco-Paredes C. Acute liver failure due to dengue virus infection. Int J Infect Dis. 2008;12:444-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (1)] |
| 27. | World Health Organization. Dengue - Global situation. May 30 2024. Available from: https://www.who.int/emergencies/disease-outbreak-news/item/2024-DON518. |
| 28. | Fernando L, Wickramasinghe VP, Kalra P, Kastner R, Gallagher E. Epidemiological burden of dengue in Sri Lanka: A systematic review of literature from 2000-2020. IJID Reg. 2024;13:100436. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (1)] |
| 29. | Thanh NT, Dat NT, Thinh TN, Phuong NTM, Thanh MTH, Bao NT, Son PT, Viet DC, Tung TH, Thien V, Luan VT. Therapeutic plasma exchange and continuous renal replacement therapy in pediatric dengue-associated acute liver failure: A case series from Vietnam. Transfus Apher Sci. 2023;62:103617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 21] [Article Influence: 7.0] [Reference Citation Analysis (2)] |
| 30. | Khanam A, Gutiérrez-Barbosa H, Lyke KE, Chua JV. Immune-Mediated Pathogenesis in Dengue Virus Infection. Viruses. 2022;14:2575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 78] [Article Influence: 19.5] [Reference Citation Analysis (1)] |
| 31. | Gunasekera AM, Eranthaka U, Priyankara D, Kalupahana R. A rare case of acute liver failure with intrahepatic cholestasis due to dengue hemorrhagic fever: CytoSorb® and plasma exchange aided in the recovery: case report. BMC Infect Dis. 2022;22:938. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (1)] |
| 32. | Samanta J, Sharma V. Dengue and its effects on liver. World J Clin Cases. 2015;3:125-131. [PubMed] [DOI] [Full Text] |
| 33. | Fernando S, Wijewickrama A, Gomes L, Punchihewa CT, Madusanka SD, Dissanayake H, Jeewandara C, Peiris H, Ogg GS, Malavige GN. Patterns and causes of liver involvement in acute dengue infection. BMC Infect Dis. 2016;16:319. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 125] [Article Influence: 12.5] [Reference Citation Analysis (2)] |
| 34. | Huerre MR, Lan NT, Marianneau P, Hue NB, Khun H, Hung NT, Khen NT, Drouet MT, Huong VT, Ha DQ, Buisson Y, Deubel V. Liver histopathology and biological correlates in five cases of fatal dengue fever in Vietnamese children. Virchows Arch. 2001;438:107-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 95] [Article Influence: 3.8] [Reference Citation Analysis (1)] |
| 35. | McBride A, Chanh HQ, Fraser JF, Yacoub S, Obonyo NG. Microvascular dysfunction in septic and dengue shock: Pathophysiology and implications for clinical management. Glob Cardiol Sci Pract. 2020;2020:e202029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (1)] |
| 36. | Srikiatkhachorn A, Mathew A, Rothman AL. Immune-mediated cytokine storm and its role in severe dengue. Semin Immunopathol. 2017;39:563-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 200] [Article Influence: 22.2] [Reference Citation Analysis (1)] |
| 37. | Rajapakse S. Dengue shock. J Emerg Trauma Shock. 2011;4:120-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 50] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 38. | Khongphatthanayothin A, Mahayosnond A, Poovorawan Y. Possible cause of liver failure in patient with dengue shock syndrome. Emerg Infect Dis. 2013;19:1161-1163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (2)] |
| 39. | Giri S, Anirvan P, Vaidya A, Praharaj DL. Dengue-related acute liver failure-A scoping review. Indian J Gastroenterol. 2024;43:407-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 40. | Aye KS, Charngkaew K, Win N, Wai KZ, Moe K, Punyadee N, Thiemmeca S, Suttitheptumrong A, Sukpanichnant S, Prida M, Halstead SB. Pathologic highlights of dengue hemorrhagic fever in 13 autopsy cases from Myanmar. Hum Pathol. 2014;45:1221-1233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 138] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 41. | Bernal W, Wendon J. Acute liver failure. N Engl J Med. 2013;369:2525-2534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1040] [Cited by in RCA: 884] [Article Influence: 68.0] [Reference Citation Analysis (0)] |
| 42. | Sriphongphankul H, Liabsuetrakul T, Osatakul S. Clinical Outcomes of Children Diagnosed Dengue-Associated Acute Liver Failure with or without N-Acetylcysteine Treatment: A Retrospective Cohort Study. J Trop Pediatr. 2021;67:fmab039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (1)] |
| 43. | Rosenbaum MW, Gonzalez RS. Immunohistochemistry as predictive and prognostic markers for gastrointestinal malignancies. Semin Diagn Pathol. 2022;39:48-57. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 44. | Larsen FS, Schmidt LE, Bernsmeier C, Rasmussen A, Isoniemi H, Patel VC, Triantafyllou E, Bernal W, Auzinger G, Shawcross D, Eefsen M, Bjerring PN, Clemmesen JO, Hockerstedt K, Frederiksen HJ, Hansen BA, Antoniades CG, Wendon J. High-volume plasma exchange in patients with acute liver failure: An open randomised controlled trial. J Hepatol. 2016;64:69-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 604] [Cited by in RCA: 479] [Article Influence: 47.9] [Reference Citation Analysis (1)] |
| 45. | Rajakumar A, Gupta P, Rammohan A, Devarajan V, Jothimani D, Shanmugam N, Kaliamoorthy I, Rela M. Liver Transplantation for Dengue-induced Acute Liver Failure. J Clin Exp Hepatol. 2024;14:101405. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 46. | McPhail MJ, Wendon JA, Bernal W. Meta-analysis of performance of Kings's College Hospital Criteria in prediction of outcome in non-paracetamol-induced acute liver failure. J Hepatol. 2010;53:492-499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 99] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 47. | Saluja V, Sharma A, Pasupuleti SS, Mitra LG, Kumar G, Agarwal PM. Comparison of Prognostic Models in Acute Liver Failure: Decision is to be Dynamic. Indian J Crit Care Med. 2019;23:574-581. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (1)] |
| 48. | Panackel C, Raja K, Fawas M, Jacob M. Prognostic models in acute liver failure-historic evolution and newer updates "prognostic models in acute liver failure". Best Pract Res Clin Gastroenterol. 2024;73:101957. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 49. | Teerasarntipan T, Thanapirom K, Chaiteerakij R, Komolmit P, Treeprasertsuk S. Validation of prognostic scores for predicting acute liver failure and in-hospital death in patients with dengue-induced severe hepatitis. World J Gastroenterol. 2024;30:4781-4790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 1] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (5)] |
| 50. | Agarwal B, Wright G, Gatt A, Riddell A, Vemala V, Mallett S, Chowdary P, Davenport A, Jalan R, Burroughs A. Evaluation of coagulation abnormalities in acute liver failure. J Hepatol. 2012;57:780-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 127] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 51. | Stravitz RT, Lee WM. Acute liver failure. Lancet. 2019;394:869-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 656] [Article Influence: 93.7] [Reference Citation Analysis (3)] |
| 52. | Bhatia V, Singh R, Acharya SK. Predictive value of arterial ammonia for complications and outcome in acute liver failure. Gut. 2006;55:98-104. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 235] [Cited by in RCA: 182] [Article Influence: 9.1] [Reference Citation Analysis (1)] |
| 53. | de-Oliveira-Pinto LM, Marinho CF, Povoa TF, de Azeredo EL, de Souza LA, Barbosa LD, Motta-Castro AR, Alves AM, Ávila CA, de Souza LJ, da Cunha RV, Damasco PV, Paes MV, Kubelka CF. Regulation of inflammatory chemokine receptors on blood T cells associated to the circulating versus liver chemokines in dengue fever. PLoS One. 2012;7:e38527. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (1)] |
| 54. | Malavige GN, Huang LC, Salimi M, Gomes L, Jayaratne SD, Ogg GS. Cellular and cytokine correlates of severe dengue infection. PLoS One. 2012;7:e50387. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 93] [Article Influence: 6.6] [Reference Citation Analysis (1)] |
| 55. | Limothai U, Jantarangsi N, Suphavejkornkij N, Tachaboon S, Dinhuzen J, Chaisuriyong W, Trongkamolchai S, Wanpaisitkul M, Chulapornsiri C, Tiawilai A, Tiawilai T, Tantawichien T, Thisyakorn U, Srisawat N. Discovery and validation of circulating miRNAs for the clinical prognosis of severe dengue. PLoS Negl Trop Dis. 2022;16:e0010836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 56. | Faridah IN, Dania H, Maliza R, Chou WH, Wang WH, Chen YH, Perwitasari DA, Chang WC. Genetic Association Studies of MICB and PLCE1 with Severity of Dengue in Indonesian and Taiwanese Populations. Diagnostics (Basel). 2023;13:3365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 57. | Vejbaesya S, Luangtrakool P, Luangtrakool K, Kalayanarooj S, Vaughn DW, Endy TP, Mammen MP, Green S, Libraty DH, Ennis FA, Rothman AL, Stephens HA. TNF and LTA gene, allele, and extended HLA haplotype associations with severe dengue virus infection in ethnic Thais. J Infect Dis. 2009;199:1442-1448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 61] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 58. | Lim A, Shearer FM, Sewalk K, Pigott DM, Clarke J, Ghouse A, Judge C, Kang H, Messina JP, Kraemer MUG, Gaythorpe KAM, de Souza WM, Nsoesie EO, Celone M, Faria N, Ryan SJ, Rabe IB, Rojas DP, Hay SI, Brownstein JS, Golding N, Brady OJ. The overlapping global distribution of dengue, chikungunya, Zika and yellow fever. Nat Commun. 2025;16:3418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 69] [Reference Citation Analysis (1)] |
| 59. | Camous L, Markowicz S, Loraux C, Jabot J, Pommier JD. High frequency of severe liver dysfunction in critically ill Dengue patients in the French West Indies. J Intensive Med. 2024;4:403-404. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (1)] |
| 60. | Chikkala BR, Pandey Y, Acharya R, Sreekumar S, Dey R, Agarwal S, Gupta S. Living Donor Liver Transplant for Dengue-Related Acute Liver Failure: A Case Report. Exp Clin Transplant. 2021;19:163-166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 61. | World Health Organization; Special Programme for Research and Training in Tropical Diseases. Dengue guidelines, for diagnosis, treatment, prevention and control. Geneva: WHO, 2009. |
| 62. | Banu S, Hu W, Hurst C, Tong S. Dengue transmission in the Asia-Pacific region: impact of climate change and socio-environmental factors. Trop Med Int Health. 2011;16:598-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 92] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 63. | Kurinami N, Sugiyama S, Yoshida A, Hieshima K, Miyamoto F, Kajiwara K, Jinnouch K, Jinnouchi T, Jinnouchi H. Body muscle-to-fat ratio gender-specific cut-off values for impaired insulin sensitivity in patients with treatment-naïve type 2 diabetes mellitus. Endocrine. 2019;66:503-508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |