Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.116452
Revised: December 6, 2025
Accepted: February 3, 2026
Published online: May 19, 2026
Processing time: 169 Days and 0.4 Hours
Schizophrenia is a chronic and disabling psychiatric disorder affecting appro
Core Tip: Schizophrenia remains one of the most complex psychiatric disorders, characterized by profound neurobiological, cognitive, and social dysfunctions. Despite decades of clinical and neuroimaging research, diagnosis still depends largely on subjective observation. This review integrates classical perspectives on epidemiology, etiology, and treatment with recent developments in artificial intelligence (AI). It highlights how AI-driven models-particularly those analyzing neuroimaging and electroencephalography data-offer objective insights into brain alterations and symptom mechanisms. By bridging traditional psychiatry with emerging computational tools, the paper outlines a roadmap for early detection, personalized intervention, and transparent decision support in schizophrenia care.
- Citation: Ozsoy F, Tasci G, Tasci B, Dogan S, Tuncer T. Schizophrenia in the age of artificial intelligence: A review of advances in diagnosis, prediction, and digital psychiatry. World J Psychiatry 2026; 16(5): 116452
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/116452.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.116452
Schizophrenia is a complex neurobiological disorder that affects millions of people worldwide and has profound consequences for lifelong functioning. The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) defines schizophrenia through its criteria which produce a broad range of mood and cognitive and behavioral symptoms that show significant clinical diversity. The field of neuropsychiatric research has made progress but doctors continue to depend on clinical observations and patient self-reports for diagnosis because there are no established biological or computational biomarkers for automated decision support. The process of psychiatric diagnosis through subjective evaluation results in delayed diagnosis times which span from one year to several years before patients receive their official diagnosis thus causing their treatment outcomes to worsen and their life quality to decline[1-3]. Researchers use machine learning (ML) and deep learning (DL) techniques to build automated systems which perform psychiatric disorder assessments and predict symptom patterns. Researchers have used ML to analyze neuroimaging data from structural magnetic resonance imaging (sMRI) and functional MRI (fMRI) and electroencephalography (EEG) during the last ten years to develop schizophrenia identification tools. The diagnostic accuracy of these multimodal approaches reaches between 74% and 99% through the use of discriminative features which include cortical thickness and gray-matter volume and functional connectivity patterns and cross-modal data integration[4-8]. Such research shows that artificial intelligence (AI)-based analysis methods identify concealed brain patterns which distinguish patients from normal people thus enabling the creation of objective psychiatric diagnosis methods[1-3,5]. Most ML and DL models show promising accuracy but they function as unexplainable black-box systems which generate highly accurate results without showing their decision-making processes. Medical staff develop distrust toward AI tools because they cannot understand the operational mechanisms of these systems. The classification process in psychiatric disorders requires equal importance to both model understanding and the actual classification system. The model needs to reveal its decision limits and evidence basis and confidence level to help clinicians make responsible and accountable decisions[9-12]. The fundamental knowledge gap needs explainable AI (XAI) to operate as a multidisciplinary field which reveals model operations while making them understandable to humans. The Shapley Additive Explanations (SHAP) and Local Interpretable Model-Agnostic Explanations (LIME) techniques provide feature influence explanations through basic visualizations which display the main factors that determine results. XAI methods in schizophrenia research have shown that the caudate nuclei and frontal operculum cortex and temporal fusiform cortex play essential roles in diagnosis while demonstrating ability to merge behavioral data with neuroimaging information[5,13-16]. The research demonstrates XAI serves two essential purposes by making scientific processes more transparent and by improving doctors' ability to understand AI-based medical systems. The current state shows major obstacles which prevent successful algorithm implementation in medical practice. The raw XAI outputs which include ranked feature lists and attribution heatmaps do not provide clinical value and cannot be easily used by psychiatrists during their decision-making process. The complex nature of schizophrenia which combines various neurobiological factors with multiple symptom presentations under one diagnosis makes it difficult to apply models between different groups and environments thus needing multiple tests for validation[1-3,5,9-16]. The fast development of AI technology and explainable modeling systems has become vital for schizophrenia diagnosis and clinical treatment. The research performs a systematic review to achieve three primary goals which involve: (1) Assessing ML and XAI systems for neuroimaging-based medical diagnosis; (2) Analyzing recent models that use behavioral and psychiatric data; and (3) Identifying critical methodological and ethical and translational barriers that must be addressed for medical practice adoption. The review aims to establish the development route from AI algorithm advancement to digital psychiatry systems which provide both trustworthy and clinically useful results and interpretable outcomes. The review investigates schizophrenia through an analysis of current AI system operations. The review extends past traditional epidemiological and etiological and clinical frameworks to demonstrate how modern computational tools using ML and multimodal neuroimaging analysis and explainable AI systems break through traditional diagnostic challenges to establish new methods for personalized psychiatric care. The review combines different perspectives to confirm its recently discovered research findings.
A general definition presents an extensive explanation which describes the core elements of a particular concept. The definition establishes basic characteristics of the concept through its explanation. The definition lacks precision because it provides a basic understanding of the concept through its main elements. The definition serves as an entry point to understand the concept better while providing a basis to study it in greater depth. Schizophrenia exists as a complicated mental health condition which produces recurring cycles of worsening symptoms and improvement[17]. The condition shows positive symptoms through delusions and hallucinations and disorganized speech and behavior together with negative symptoms that include social withdrawal and anhedonia and lack of motivation. The condition produces additional symptoms which include attention problems and memory issues and executive function deficits that indicate broad cognitive and perceptual and behavioral disturbances. Schizophrenia creates a severe impact on both people who have it and their families and the entire community[18,19]. The traditional clinical descriptions of schizophrenia together with its epidemiological patterns and etiological mechanisms and diverse clinical expressions serve as the essential base for understanding this disorder. The same features which define schizophrenia lead to ongoing diagnostic and therapeutic obstacles for clinicians. AI-driven analytical methods now use epidemiological data and neurobiological indicators and symptom patterns to create predictive models which enhance both early diagnosis and risk assessment and personalized treatment strategies. The conventional knowledge base serves as both historical background and structural base which enables AI systems to generate more precise data-based understanding of schizophrenia.
Research studies in the literature show that schizophrenia exists in 0.75% of worldwide population according to epidemiological data[20]. The condition affects 21 million people worldwide according to estimates which place it within the schizophrenia spectrum disorders category[21,22]. Research studies have demonstrated that schizophrenia prevalence rates differ between different geographic areas with rates between 8 cases and 43 cases per 100000 population[22]. The worldwide number of schizophrenia patients amounts to 21 million people according to recent meta-analytic research which predicts that population aging will lead to increased numbers of affected individuals. Most patients live in developing nations which continue to experience major gaps in their treatment options[23]. The exact measurement of schizophrenia prevalence continues to be difficult because doctors struggle to make accurate diagnoses and because the condition shares symptoms with other mental health conditions and research methods vary between studies. Given these challenges, schizophrenia and related psychotic disorders are often grouped together in prevalence research. The World Health Organization (WHO) states that schizophrenia and related psychotic disorders occur in 0.25% to 0.64% of the United States population according to research from household surveys and medical records and structured diagnostic interviews[24-27]. Research studies have shown that this condition affects 0.33% to 0.75% of people worldwide[22,28-30]. In the literature, studies using AI models have been conducted to determine the prevalence of schizophrenia[8,29]. In one conducted study, the prevalence in the general population was reported as 1.6%. This rate was calculated to be somewhat higher than what is found in the literature. This situation has been attributed to various factors such as AI systems estimating lifetime prevalence rather than point prevalence and potentially using a more comprehensive disorder classification methodology[8].
The disorder typically starts affecting people between ages 15 and 25 before it leads to early death and suicide attempts become more common[30]. The WHO reports that schizophrenia first appears in people between their late teenage years and their early thirty's. The first signs of schizophrenia appear during late adolescence through early twenties in males but females show symptoms between their early twenties and early thirties[29,31,32]. Gender-related differences in schizophrenia have been recognized since the earliest descriptions of the disorder. Women develop their symptoms at an older age and their symptoms become more severe after they reach menopause. Although the incidence is higher in males, overall prevalence does not differ significantly between sexes. These disparities have been attributed to genetic and neurodevelopmental factors, as well as the potential neuroprotective effects of estrogen in women[33]. In the results obtained using AI systems in this field, it has also been shown that the disorder is more common among individuals who are single or separated from their partners, and that patients have difficulty working[8].
Research findings indicate that schizophrenia affects unmarried people at higher rates and people with schizophrenia experience difficulties when trying to build or sustain marriages[34-36]. Research findings show that people with schizophrenia tend to marry less often and face difficulties in maintaining their romantic partnerships. The condition leads to negative impacts on work performance because patients face challenges when looking for work and keeping their existing jobs[37-39]. The societal and economic burden of schizophrenia is substantial[20]. Employment rates among individuals with schizophrenia are remarkably low; studies in the United States and Europe report that only about 20% of patients hold regular paid employment[20,23,40]. The socioeconomic burden of chronic disorders continues to rise because patients experience high unemployment rates and their caregivers also face increased joblessness[41]. The United States spends 155.7 billion USD annually on schizophrenia treatment which includes 38% from unemployment costs and 34% from caregiver productivity losses and 24% from direct healthcare expenses[41].
Schizophrenia creates economic problems because it reduces worker productivity during their most productive years while also causing family members to lose productivity and forcing patients to pay for their own treatment and rehabilitation costs[23]. The total economic burden increases because emergency visits happen frequently and patients need hospital care and outpatient follow-ups and pharmacological treatment and extended rehabilitation periods[42,43]. Consequently, schizophrenia remains one of the most costly psychiatric disorders for both individuals and society[20,23].
The exact causes of schizophrenia have not been fully determined because the condition develops from multiple genetic and neurobiological and environmental elements. Research shows that psychosis emerges through the combination of genetic factors with environmental elements which affect neurotransmitters and inflammation and birth complications and advanced paternal age and traumatic events and poverty and social isolation and migration and substance abuse[43-48]. In recent years, a considerable number of studies have been conducted using AI systems to elucidate the etiology of the disorder. In these studies, AI models have been developed based on various biomarkers such as gene signals, brain imaging, and inflammatory markers[49-53]. Data from the 2014 Psychiatric Genomics Consortium Schizophrenia Working Group identified 128 independent genetic variants across 108 chromosomal loci associated with schizophrenia[43]. Family and twin research studies demonstrate that genetic elements dominate development while environmental elements have little influence[44], The research findings do not support the expected pattern that was predicted. The research by Torrey and Yolken[44] demonstrated that monozygotic twins developed schizophrenia together at a 28% rate but dizygotic twins shared the condition at 6% which suggests environmental elements contribute to schizophrenia development[43]. The disorder results from multiple genetic variants which collectively contribute to susceptibility rather than being caused by a single gene or allele[45,46]. Polygenic risk scores constructed from schizophrenia-related alleles explain only about 7.7% of the variance in disorder liability[45]. In a study using AI modeling in this field, it was con
Research shows prenatal and perinatal complications serve as established environmental risk factors yet scientists have not identified their exact mechanisms of action. Research has established that low birth weight and springtime births and maternal substance or alcohol exposure during pregnancy and paternal age above 40 and childhood experiences of neglect and abuse and peer bullying are major risk factors[18]. Research findings show that obstetric complications together with fetal development abnormalities increase schizophrenia risk at a moderate level[18,47,48]. Research shows that people born during late winter and early spring face higher health risks because of seasonal factors[54,55].
Schizophrenia develops through neurochemical disruptions which affect both dopaminergic and glutamatergic signaling pathways. Studies indicate that too much activity in the mesolimbic dopaminergic system results in the formation of positive psychotic symptoms[56]. Abnormalities in GABAergic and glutamatergic neurotransmission and an imbalance between these systems are increasingly recognized as contributors to both positive and negative symptom domains[57]. In addition, serotonergic, muscarinic, and glycinergic systems have been explored in recent research as potential modulators of the disorder[58,59]. Research now focuses on neuroinflammatory processes because scientists believe pro-inflammatory cytokines and oxidative stress markers play a role in causing disorder symptoms and disorder advancement[18].
Neuroimaging data shows that neuroanatomical abnormalities exist throughout the brain because of gray matter reduction and global brain volume loss and ventricular enlargement[60]. The lateral ventricles show enlargement while the cortex becomes thinner and the brain hemispheres lose their normal symmetry according to multiple studies[60-63]. Pronounced volumetric reductions are observed in the hippocampus, frontal, and temporal regions[64,65]. The presence of structural changes becomes evident through two lines of evidence which show decreased gray matter density and disrupted neural connections between neurons[66]. Functional imaging research supports these results by showing prefrontal cortical activity decreases which scientists refer to as hypofrontality[67-71]. In studies conducted with ML models, it has been shown that AI systems developed using different brain imaging techniques may play an important role in the diagnosis of schizophrenia[8]. In a meta-analysis, when various intracranial imaging and signal/signal-processing methods were used together with AI techniques to diagnose the disorder, a sensitivity and specificity of 80% were identified[69].
Schizophrenia exists as a persistent mental health condition which produces multiple clinical symptoms including psychotic episodes and reality distortion and social isolation and motivational loss and reduced thinking capacity and neurocognitive deficits in attention and memory and executive abilities. The condition presents with positive symptoms which include hallucinations and delusions and disorganized speech and behavioral disturbances that result from abnormal function amplification and negative symptoms which cause decreased emotional and behavioral abilities through flat affect and reduced speech and motivation and social isolation and general functional decline[18].
Research findings indicate that psychosis patients exhibited unusual conduct during their pre-illness period through their academic and social interaction difficulties[70-72]. The initial symptoms typically emerge during late adolescence or early adulthood[23]. Research data shows that people with schizophrenia have homicidal behavior and suicide attempt rates at 0.48% to 0.75% which exceeds general population statistics[73-76].
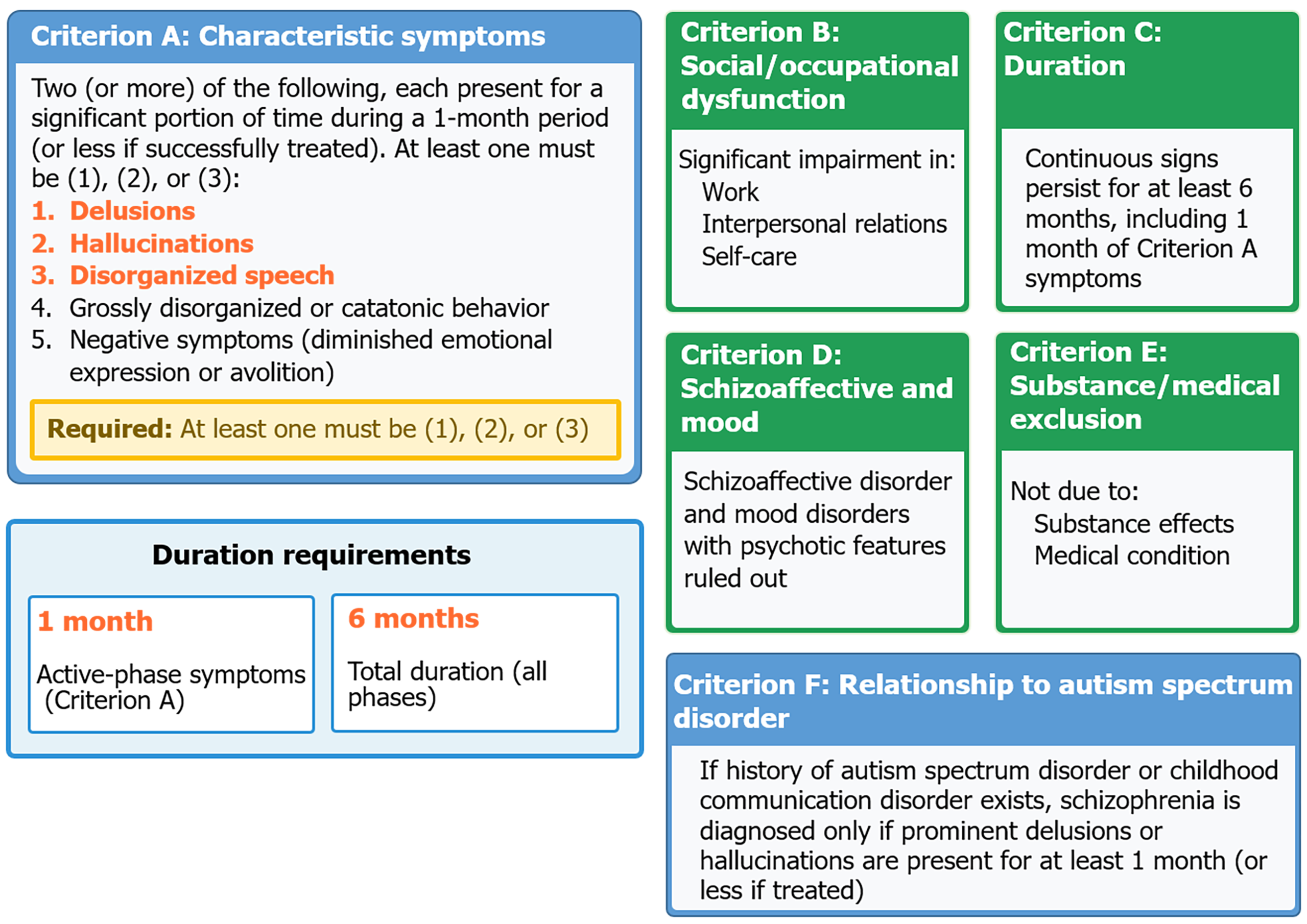
In clinical practice, the diagnosis of schizophrenia is established based on the DSM-5 criteria (Figure 1), The diagnosis requires three elements which include specific symptoms and time duration and functional impairment.
Schizophrenia has been linked to relapse and remission and chronic disorder patterns throughout its historical development[74]. Research studies published in the literature showed that 16.4% of patients recovered from their illness without any sex-related differences and found that recovery rates were higher in developing nations[75]. Research conducted in a meta-analysis study showed that 24.2% of patients reached recovery status while 35.5% achieved good or improved results and 59.7% received moderate outcomes[74]. The variability in these results is thought to be influenced by methodological differences across studies, variations in the stage and severity of illness among participants, and geographical differences in where the studies were conducted[74]. With the increase in AI studies in the field and the achievement of highly specific results, it has been shown that ML models may be effective in diagnosing schizophrenia, identifying clinical symptoms, and developing personalized treatment models in the coming years[2,76].
The main goal of schizophrenia treatment involves symptom reduction to help patients regain their functional abilities and reintegrate into their community. Therapy success depends on physicians building strong collaborative bonds with patients through respectful communication and trust based relationships and open dialogue. The main treatment approach involves pharmacological interventions which use antipsychotic medications as their primary therapeutic option[73].
The main action of typical antipsychotics occurs through D2 receptor blockade in the mesolimbic pathway which reduces excessive dopamine activity that causes positive psychotic symptoms[77] (Table 1). The first-generation agent chlorpromazine and haloperidol and fluphenazine effectively treat hallucinations and delusions yet they cause extrapyramidal side effects and tardive dyskinesia in many patients.
| Typical antipsychotics (first-generation) | Atypical antipsychotics (second-generation) |
| Phenothiazines: Chlorpromazine, fluphenazine, mesoridazine, perphenazine, thioridazine, trifluoperazine | Atypical agents: Aripiprazole, clozapine, olanzapine, quetiapine, risperidone, ziprasidone |
| Butyrophenones: Haloperidol | |
| Others: Loxapine, molindone, thiothixene |
The second-generation antipsychotics known as atypical agents form a diverse group which shows different receptor binding patterns and side effects and chemical properties compared to first-generation antipsychotics. These medications exhibit a higher affinity for serotonin (5-HT2A) receptors relative to dopamine D2 receptors, which contributes to their lower incidence of motor side effects and their efficacy in addressing both positive and negative symptoms[77] (Table 1). The treatment of schizophrenia includes aripiprazole and clozapine and olanzapine and quetiapine and risperidone and ziprasidone as standard medications.
The therapeutic response to antipsychotic medication may begin within days but often requires four to six weeks for full effect. Research studies through meta-analyses show that patients achieve their best treatment results during the initial seven to fourteen days of medication[77]. The treatment approach for patients who do not respond to standard antipsychotic doses and durations includes either increasing the medication dose or selecting a different antipsychotic medication[78]. The evaluation of medication adherence represents a vital process because depot formulations enable patients to maintain their treatment schedule when doctors identify non-adherence problems[79]. The recommended use of depot antipsychotics should be reserved for patients who fail to follow their medication schedule and for doctors who want to verify patient compliance with their medication regimen. The literature is increasingly highlighting that ML-based personalized treatment models may be used for many psychiatric disorders, including schizophrenia[80-82].
Electroconvulsive therapy (ECT) serves as an additional treatment for schizophrenia patients who do not respond to standard treatments or experience severe psychotic episodes. ECT produces its best results for patients who have recently developed their illness and show both sudden symptoms and strong emotional changes. Research studies show that ECT treatment leads to major short-term improvements for patients who experience their first schizophrenia episode[83,84].
The prognosis of schizophrenia shows different outcomes for different people because of multiple biological and psychological and social elements that affect the condition. The outcome of disorder depends on four essential factors which include premorbid personality traits and family structure and social support and overall functional status. The prognosis for patients becomes worse when they show schizoid or schizotypal personality features and poor premorbid functioning and limited social support. The table shows that better long-term results occur when patients receive early and continuous treatment and when they have strong family connections and social networks and normal cognitive abilities (Table 2)[85].
| Favorable prognostic indicators | Unfavorable prognostic indicators |
| Female sex | Male sex |
| Being married | Being single, divorced, or living alone |
| Acute onset | Insidious onset |
| Later age of onset | Early age of onset |
| Family history of mood disorder | Family history of schizophrenia |
| Good premorbid social adjustment | Premorbid schizoid or schizotypal personality |
| Living in a rural area | Living in an urban environment |
| Higher intelligence level | Lower intelligence level |
| Few and brief exacerbations | Frequent or prolonged hospitalizations |
| Early initiation and regular adherence to treatment | Irregular or absent treatment adherence |
| Absence of comorbid psychiatric disorders | Presence of comorbid psychiatric conditions |
| Strong family collaboration with the treatment team | Dysfunctional family environment or high expressed emotion |
The illness of schizophrenia progresses through a long-term pattern of recurring episodes which alternate between times of improvement and times of worsening symptoms. The treatment and support of patients lead to satisfactory functional recovery for one-third of patients but two-thirds experience partial improvement with ongoing symptoms and the last third develops enduring disability and social integration issues. Research indicates that patients who get early diagnosis and continuous medication and psychological support and build strong doctor-patient relationships will achieve improved long-term outcomes with reduced relapse rates. The prognosis of schizophrenia worsens when patients fail to follow their treatment plan and when their condition starts before age 20 and when they are male and when their family environment shows high levels of stress and expressed emotion.
The implementation of AI technologies in psychiatry during the last few years brought about a major shift in schizophrenia biomarker detection and clinical decision support systems. AI processes complex neurobiological and cognitive and behavioral data to achieve better diagnostic results and predict treatment outcomes and start interventions at an early stage. AI-driven models analyze neuroimaging data together with EEG recordings and speech patterns and social behavior measurements to create a complete understanding of schizophrenia's neurophysiological and phenotypic diversity. The AI-assisted methods enable biomarker-based diagnostic system development and improve clinical decision-making through XAI principles which provide explainable AI.
The diagnosis of complex psychiatric disorders such as schizophrenia has long relied on subjective clinical observations based on DSM-5 criteria. The practice of relying on clinical judgment leads to different treatment approaches between healthcare providers while the lack of biological markers makes it difficult to diagnose patients correctly and create proper treatment plans[86,87]. The diverse symptoms of schizophrenia along with its unknown brain mechanisms require scientists to create new diagnostic methods which can measure symptoms objectively. The new digital psychiatry paradigm uses AI to analyze complex clinical and neuroimaging and neurophysiological data for early disorder detection and individualized treatment development[88,89]. AI systems process EEG and MRI data together with genomic information and behavioral metrics and electronic health records to create biomarker-based data-driven decision support systems for schizophrenia and psychiatric disorders[90,91]. The fast progress of digital psychiatry models has not solved the essential problem of making their operations transparent and easy to understand. Establishing trust in clinical decision-making and ensuring the ethical adoption of AI systems require that model reasoning processes be explainable. The development of XAI methodologies works to enhance model output understanding which leads to better clinical decision-making confidence[92,93]. Specifically, SHAP quantifies the contribution of individual input features to model decisions; Gradient-weighted Class Activation Mapping visualizes salient regions in DL networks; and LIME generates localized explanations for individual predictions. The combination of these methods enables AI models to show their causal reasoning for schizophrenia detection and other neuropsychiatric classifications which enhances clinicians' ability to interpret results[94-96]. Digital psychiatry brought a fundamental change to medical practice because doctors now use data analysis to make diagnoses instead of depending on their personal opinions. The combination of AI with explainable modeling systems produces better diagnostic results while creating an AI-assisted mental health system which operates ethically and provides individualized care through human-AI teamwork[97-99].
The research of schizophrenia depends on neuroimaging techniques which use MRI and fMRI and diffusion tensor imaging (DTI) as their core methods. Research using structural MRI has shown that schizophrenia patients develop smaller brain volumes in their hippocampus and prefrontal cortex starting from the beginning of their illness. The disorder produces significant brain structure abnormalities which scientists have identified through these studies[100,101]. Functional MRI investigations have further shown that disruptions in large-scale neural networks particularly the default mode and salience networks are closely linked to cognitive dysfunctions and impaired integration of neural information[100,102]. Complementing the research findings show that DTI analyses detected white matter integrity breakdowns in brain connections between frontal and temporal areas which indicates neural communication problems and decreased brain information processing speed[100,103].
The combination of AI with EEG technology now serves as a revolutionary method for schizophrenia diagnosis and assessment and ongoing monitoring. The system enables researchers to detect complex neurophysiological patterns in EEG data through an automated non-invasive method which replaces traditional behavioral observation and subjective clinical evaluation. AI technology helps researchers analyze complex EEG data to study schizophrenia brain abnormalities which leads to better detection methods and individualized treatment plans. EEG recordings from schizophrenia patients show continuous disruptions across different frequency bands which extend throughout their entire recording duration. Research has shown that theta (4-7 Hz) and alpha (8-11 Hz) and beta (12-30 Hz) and gamma (> 30 Hz) power bands show abnormal increases which mainly affect the temporal and posterior cingulate regions[104,105]. Research shows that middle temporal gyrus theta activity at elevated levels creates a negative relationship with verbal memory abilities which proves EEG signals directly affect cognitive performance[105,106]. Research indicates that brain region synchronization problems result in phase discontinuity which represents a basic neurophysiological brain function defect. The brain shows two main patterns of disruption through its neural communication system because alpha-phase discontinuity increases and theta-phase continuity decreases. The alpha peak frequency in temporal and cingulate regions shows a substantial decrease which scientists identify as a distinct marker of the disorder[105,106]. The brain responses known as event related potentials (ERPs) show two types of abnormalities in schizophrenia: P50 gating and mismatch negativity which link to social and cognitive impairments[106]. Research has demonstrated that AI achieves successful EEG-based schizophrenia classification through its application of ML and DL frameworks. The classical algorithms support vector machines (SVM) and decision trees and random forests and logistic regression show effective interpretable results in small datasets where decision trees reach 89.12% specificity and random forest models achieve 91% specificity[107,108]. DL systems use their operation to find both time-based and space-based patterns which exist in EEG data. The system employs convolutional neural networks (CNNs) to detect particular patterns between channels and long short-term memory (LSTM) networks to detect time-dependent patterns. The hybrid CNN-LSTM models achieve 99.25% classification accuracy according to[109,110]. The ten-layer CNN models which process multiple EEG channels achieve 99.18% accuracy but GRU networks that focus on alpha rhythms reach 88.88%[110]. The method needs to undergo multiple site testing for clinical use because it requires both strong validation results and established measurement protocols. The study used 81 participants from various educational institutions and 80 schizophrenia patients to obtain reliable ERP and quantitative EEG results which showed connections to cognitive performance and functional assessment results[111]. EEG-based AI systems require sophisticated preprocessing and feature extraction methods to generate reliable analytical results. The signals undergo delta theta alpha beta and gamma band decomposition after band-pass filtering and noise reduction. The mathematical feature extraction methods of fast Fourier transform, approximate entropy, log-energy entropy, Shannon entropy, and kurtosis are commonly used, with entropy measures quantifying signal complexity and irregularity, and kurtosis reflecting the shape of the amplitude distribution[112]. The model achieves better performance through normalization techniques which use z-score and L2 normalization to distribute data values evenly[109]. The combination of Bayesian optimization with recursive feature elimination helps prevent overfitting in high-dimensional EEG data which results in better model performance[109]. AI-assisted EEG analyses demonstrate the ability to identify pre-conditions which appear before the development of the condition. People in this stage show cognitive problems which they cannot measure but these symptoms do not reach the level needed for psychotic disorder diagnosis. Research conducted with the Bonn Scale for the Assessment of Basic Symptoms demonstrates that psychosis detection at an early stage prevents its development in people who experience it[113,114]. AI models gain improved disorder onset prediction during the prodromal stage through the combination of EEG biomarkers with natural language cues from clinical notes that include symptoms like “hearing voices” and “social withdrawal”[115]. Healthcare professionals require model interpretability as an essential factor to accept these models for clinical use. The DL system operates as a black box which makes it difficult for clinicians to understand diagnostic results so they need XAI methods including SHAP and LIME for explanation[15]. The methods enable researchers to visualize EEG features together with brain regions and connectivity patterns which drive classification results for better model understanding. XAI methods in research studies identified age and gender and spectral EEG patterns as the essential diagnostic factors for schizophrenia diagnosis. The SHAP analysis showed that schizophrenia classification depends on three brain regions which include the bilateral pallidum and inferior frontal gyrus (pars triangularis) and temporal regions[1,5,15]. Schizophrenia neurobiological origins exist beyond EEG because EEG combined with fMRI and DTI and MR spectroscopy and MEG allows researchers to study structural and functional and electrophysiological aspects at the same time[1,5,15]. AI-based multimodal frameworks use gray matter volume loss and functional connectivity reductions and N-acetylaspartate depletion and EEG rhythmic disturbances to create personalized biomarker profiles which improve treatment prediction accuracy[116,117]. The new field of precision psychiatry requires these intricate evaluation methods for its operation. AI systems use EEG biomarkers together with genetic information and molecular data and environmental factors to develop individualized neurobiological subtypes which direct personalized treatment approaches[118,119]. Nonetheless, many existing studies are limited by small sample sizes, leading to overfitting and reduced external validity[118,120]. The use of pharmacological treatments in treatment allows disorders to produce specific EEG patterns which remain undetectable to algorithms because Western researchers collect most of the available data[108,121,122]. The application of transfer learning and domain adaptation methods has proven successful for improving generalization performance when working with restricted EEG datasets[123,124]. The combination of CNN, LSTM and transformer architectures in hybrid models produces better results in terms of accuracy and interpretability and attention mechanisms help detect important EEG segments for diagnosis[125]. AI-assisted EEG applications provide schizophrenia researchers with objective neurophysiological indicators which help them detect the condition at its initial stages and throughout its progression. The CNN-LSTM-based models reached 99% accuracy which demonstrates the potential of EEG-based methods for diagnosis through their ability to detect theta, alpha and gamma band irregularities and phase disruptions and ERP changes and theta-band coherence variations[106,109,110,126]. The clinical validity and reliability of these technologies need to be proven through extensive multicenter research that includes diverse participants and follows uniform EEG testing methods and implements clinical decision support systems with XAI. AI-driven EEG analysis methods have the ability to create an early diagnostic system for schizophrenia which provides personalized and objective psychiatric assessments.
XAI represents a new method which has established itself as an essential tool for schizophrenia diagnosis and treatment in medical settings. The psychiatric disorder schizophrenia depends on clinical observation and patient self-reports for diagnosis because its neurobiological mechanisms are complex. AI technologies show promise to improve diagnostic precision and increase early detection chances through biomarker-based decision support systems[1,127]. XAI methodologies enhance model decision interpretation which results in higher clinical trust. The techniques of LIME and SHAP and ELI5 help doctors understand DL model decisions by showing which input features affect the output results[15]. The combination of fMRI with AI technology enables researchers to detect prefrontal cortex and hippocampus abnormalities which helps them understand how default mode network connectivity problems lead to cognitive impairments[12]. The BiLSTM-Attention DL models which use EEG data enable researchers to visualize temporal neurophysiological features in an interpretable way which enhances medical professionals' ability to understand model results[15]. Natural language processing-based systems help detect symptoms through their analysis of speech content and fluency patterns. The UCL Institute of Neurology conducts research which demonstrates that language model analysis helps scientists detect semantic in-coherence and reduced verbal predictability in schizophrenia patients relative to healthy participants[128]. Research studies have shown that AI-based diagnostic systems reach 99.25% classification accuracy for schizophrenia detection according to recent systematic reviews[127,129]. The new method produces better results than conventional clinical tests because it achieves results that traditional methods cannot match since they produce 55%-76% incorrect diagnoses. The diagnostic and treatment planning capabilities of SVM and Random Forests and Vision Transformers have shown significant promise in medical applications[12,127]. Multiple major obstacles exist during XAI im
In the AI era, understanding schizophrenia requires integrating established clinical knowledge with emerging computational tools that increasingly redefine how the disorder is detected, characterized, and managed. The current diagnostic methods depend on doctor observations and patient feedback which hinders both early disorder detection and personalized medical treatment development. The development of psychiatric research now depends on AI methods which serve as its primary driving force. AI performs objective schizophrenia analysis through multiple biological indicators which combine EEG and MRI and fMRI and DTI and genomic and behavioral and linguistic data to enhance diagnostic accuracy and develop reliable clinical prediction systems. The development of XAI methodologies has accelerated this transformation by enabling medical professionals to understand model decisions at a clinical level which leads to the development of human-centered decision-support systems that maintain trustworthiness. Research data indicates AI systems using EEG data can precisely detect neurophysiological biomarkers and neuroimaging methods generate brain structure and function models of abnormalities and AI systems evaluate social and speech data to assess schizophrenia-related language and behavioral patterns. Multimodal AI frameworks merge various data modalities into a single analytical domain which enables researchers to develop customized disorder profiles. The current foundation enables AI predictive technology to develop precise psychiatry through personalized treatments which analyze neurobiological and behavioral patterns of each patient. Nevertheless, current literature reveals several important limitations. AI models have been created through research using restricted data sets which mostly stem from Western populations thus restricting their ability to work effectively in diverse cultural settings. The DL models operate as black boxes because they prevent medical staff from understanding their decision-making processes. XAI techniques solve part of this problem but researchers need to develop methods which convert algorithmic explanations into medical terms and create simple clinical interfaces for users. The ethical framework lacks solutions for data privacy protection and informed consent management and bias control and legal responsibility enforcement at a standardized global level. The research needs to progress through three fundamental directions for future studies. The development of extensive datasets which span multiple centers and include diverse cultural groups will improve model performance and clinical trustworthiness. The analysis will track disorder progression through a single analytical system which combines EEG data with fMRI results and linguistic and behavioral indicators. The medical field needs standardized ethical and explainable AI frameworks as its most critical requirement to achieve responsible AI system deployment in the future. The European Union Artificial Intelligence Act along with other international regulations require clinical AI systems to follow principles which include transparency and accountability and fairness. AI technology implementation in schizophrenia research brings new tools but also transforms the entire method of psychiatric scientific research. AI models that explain their decisions while following ethical standards and working as complete systems will enable clinicians to receive strong assistance for both disorder detection at the beginning and treatment outcome forecasting and symptom tracking and rehabilitation planning. The success of this transformation depends on accurate algorithms and human-centered design approaches and ethical practices and teamwork between different fields of expertise. The review shows that AI systems which provide explainable functions and follow ethical guidelines will enhance diagnostic precision and develop personalized treatment approaches which result in superior long-term outcomes for schizophrenia research and clinical practice.
| 1. | Shivaprasad S, Chadaga K, Dias CC, Sampathila N, Prabhu S. An interpretable schizophrenia diagnosis framework using machine learning and explainable artificial intelligence. Syst Sci Control Eng. 2024;12:2364033. [DOI] [Full Text] |
| 2. | Jiang S, Jia Q, Peng Z, Zhou Q, An Z, Chen J, Yi Q. Can artificial intelligence be the future solution to the enormous challenges and suffering caused by Schizophrenia? Schizophrenia (Heidelb). 2025;11:32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 3. | Hansen L, Bernstorff M, Enevoldsen K, Kolding S, Damgaard JG, Perfalk E, Nielbo KL, Danielsen AA, Østergaard SD. Predicting Diagnostic Progression to Schizophrenia or Bipolar Disorder via Machine Learning. JAMA Psychiatry. 2025;82:459-469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 4. | Jimenez-Mesa C, Ramirez J, Yi Z, Yan C, Chan R, Murray GK, Gorriz JM, Suckling J. Machine learning in small sample neuroimaging studies: Novel measures for schizophrenia analysis. Hum Brain Mapp. 2024;45:e26555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 5. | Chavez C, Schurger-Foy A, Oh J, Oh J, Linstead E, Yun K, Maoz U. Toward the Trusted Medical Imaging Ai: An Explainable Machine Learning Model for Schizophrenia Brain Mris. 2023 Preprint. Available from: SSRN: 5185071. [DOI] [Full Text] |
| 6. | Lei D, Pinaya WHL, Young J, van Amelsvoort T, Marcelis M, Donohoe G, Mothersill DO, Corvin A, Vieira S, Huang X, Lui S, Scarpazza C, Arango C, Bullmore E, Gong Q, McGuire P, Mechelli A. Integrating machining learning and multimodal neuroimaging to detect schizophrenia at the level of the individual. Hum Brain Mapp. 2020;41:1119-1135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 74] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 7. | Qureshi MNI, Oh J, Cho D, Jo HJ, Lee B. Multimodal Discrimination of Schizophrenia Using Hybrid Weighted Feature Concatenation of Brain Functional Connectivity and Anatomical Features with an Extreme Learning Machine. Front Neuroinform. 2017;11:59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 54] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Lai JW, Ang CKE, Acharya UR, Cheong KH. Schizophrenia: A Survey of Artificial Intelligence Techniques Applied to Detection and Classification. Int J Environ Res Public Health. 2021;18:6099. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 9. | Omiyefa S. Artificial Intelligence and Machine Learning in Precision Mental Health Diagnostics and Predictive Treatment Models. Int J Res Publ Rev. 2025;6:85-99. [DOI] [Full Text] |
| 10. | Andrew J, Rudra M, Eunice J, Belfin RV. Artificial intelligence in adolescents mental health disorder diagnosis, prognosis, and treatment. Front Public Health. 2023;11:1110088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 11. | Gao W, Lu L, Yin X. Editorial: AI approach to the psychiatric diagnosis and prediction. Front Psychiatry. 2024;15:1387370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 12. | Di Stefano V, D'Angelo M, Monaco F, Vignapiano A, Martiadis V, Barone E, Fornaro M, Steardo L, Solmi M, Manchia M, Steardo L Jr. Decoding Schizophrenia: How AI-Enhanced fMRI Unlocks New Pathways for Precision Psychiatry. Brain Sci. 2024;14:1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 13. | Kandala R, Moharir AK, Nayak DA. From Explainability to Action: A Generative Operational Framework for Integrating XAI in Clinical Mental Health Screening. 2025 Preprint. Available from: arXiv: 2510.13828. [DOI] [Full Text] |
| 14. | Egbuna I. Explainable Artificial Intelligence (XAI) in Diagnosing Neurodevelopmental Disorders: From Black Boxes to Clinical Transparency. Int J Biol Pharm Sci Arch. 2025;10:031-057. [DOI] [Full Text] |
| 15. | Almadhor A, Ojo S, Nathaniel TI, Alsubai S, Alharthi A, Hejaili AA, Sampedro GA. An interpretable XAI deep EEG model for schizophrenia diagnosis using feature selection and attention mechanisms. Front Oncol. 2025;15:1630291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 16. | Vimbi V, Shaffi N, Mahmud M. Interpreting artificial intelligence models: a systematic review on the application of LIME and SHAP in Alzheimer's disease detection. Brain Inform. 2024;11:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 105] [Reference Citation Analysis (0)] |
| 17. | Leucht S, Barnes TR, Kissling W, Engel RR, Correll C, Kane JM. Relapse prevention in schizophrenia with new-generation antipsychotics: a systematic review and exploratory meta-analysis of randomized, controlled trials. Am J Psychiatry. 2003;160:1209-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 269] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 18. | Tandon R, Nasrallah H, Akbarian S, Carpenter WT Jr, DeLisi LE, Gaebel W, Green MF, Gur RE, Heckers S, Kane JM, Malaspina D, Meyer-Lindenberg A, Murray R, Owen M, Smoller JW, Yassin W, Keshavan M. The schizophrenia syndrome, circa 2024: What we know and how that informs its nature. Schizophr Res. 2024;264:1-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 119] [Article Influence: 59.5] [Reference Citation Analysis (0)] |
| 19. | Bowie CR, Harvey PD. Cognition in schizophrenia: impairments, determinants, and functional importance. Psychiatr Clin North Am. 2005;28:613-633, 626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 171] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 20. | Moreno-Küstner B, Martín C, Pastor L. Prevalence of psychotic disorders and its association with methodological issues. A systematic review and meta-analyses. PLoS One. 2018;13:e0195687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 438] [Cited by in RCA: 353] [Article Influence: 44.1] [Reference Citation Analysis (0)] |
| 21. | Keskinen E, Marttila A, Marttila R, Jones PB, Murray GK, Moilanen K, Koivumaa-Honkanen H, Mäki P, Isohanni M, Jääskeläinen E, Miettunen J. Interaction between parental psychosis and early motor development and the risk of schizophrenia in a general population birth cohort. Eur Psychiatry. 2015;30:719-727. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 27] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 22. | Orrico-Sánchez A, López-Lacort M, Muñoz-Quiles C, Sanfélix-Gimeno G, Díez-Domingo J. Epidemiology of schizophrenia and its management over 8-years period using real-world data in Spain. BMC Psychiatry. 2020;20:149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 23. | Charlson FJ, Ferrari AJ, Santomauro DF, Diminic S, Stockings E, Scott JG, McGrath JJ, Whiteford HA. Global Epidemiology and Burden of Schizophrenia: Findings From the Global Burden of Disease Study 2016. Schizophr Bull. 2018;44:1195-1203. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1314] [Cited by in RCA: 1087] [Article Influence: 135.9] [Reference Citation Analysis (0)] |
| 24. | Kessler RC, Birnbaum H, Demler O, Falloon IR, Gagnon E, Guyer M, Howes MJ, Kendler KS, Shi L, Walters E, Wu EQ. The prevalence and correlates of nonaffective psychosis in the National Comorbidity Survey Replication (NCS-R). Biol Psychiatry. 2005;58:668-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 244] [Cited by in RCA: 281] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 25. | Wu EQ, Shi L, Birnbaum H, Hudson T, Kessler R. Annual prevalence of diagnosed schizophrenia in the USA: a claims data analysis approach. Psychol Med. 2006;36:1535-1540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 129] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 26. | Desai PR, Lawson KA, Barner JC, Rascati KL. Estimating the direct and indirect costs for community-dwelling patients with schizophrenia. J Pharm Health Serv Res. 2013;4:187-194. [RCA] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 72] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 27. | National Institute of Mental Health. Schizophrenia. [cited 16 January 2026]. Available from: https://www.nimh.nih.gov/health/publications/schizophrenia. |
| 28. | Saha S, Chant D, Welham J, McGrath J. A systematic review of the prevalence of schizophrenia. PLoS Med. 2005;2:e141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1323] [Cited by in RCA: 1371] [Article Influence: 65.3] [Reference Citation Analysis (0)] |
| 29. | Choomung P, He Y, Matsunaga M, Sakuma K, Kishi T, Li Y, Tanihara S, Iwata N, Ota A. Estimating the Prevalence of Schizophrenia in the General Population of Japan Using an Artificial Neural Network-Based Schizophrenia Classifier: Web-Based Cross-Sectional Survey. JMIR Form Res. 2025;9:e66330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 30. | Bai W, Liu ZH, Jiang YY, Zhang QE, Rao WW, Cheung T, Hall BJ, Xiang YT. Worldwide prevalence of suicidal ideation and suicide plan among people with schizophrenia: a meta-analysis and systematic review of epidemiological surveys. Transl Psychiatry. 2021;11:552. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 92] [Article Influence: 18.4] [Reference Citation Analysis (1)] |
| 31. | McGrath J, Saha S, Chant D, Welham J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev. 2008;30:67-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1782] [Cited by in RCA: 1506] [Article Influence: 83.7] [Reference Citation Analysis (0)] |
| 32. | Atkinson M, Hollis C. NICE guideline: attention deficit hyperactivity disorder. Arch Dis Child Educ Pract Ed. 2010;95:24-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 33. | Falkenburg J, Tracy DK. Sex and schizophrenia: a review of gender differences. Psychosis. 2014;6:61-69. [RCA] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 34. | Barnett P, Oshinowo I, Cooper C, Taylor C, Smith S, Pilling S. The association between social class and the impact of treatment for mental health problems: a systematic review and narrative synthesis. Soc Psychiatry Psychiatr Epidemiol. 2023;58:581-603. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 16] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 35. | Lyngdoh LAM, Antony S, Basavarajappa C, Kalyanasundaram JR, Ammapattian T. Marriage in persons with severe mental illness: A narrative review-based framework for a supported relationship. J Family Med Prim Care. 2023;12:3033-3041. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 36. | Mendrek A, Mancini-Marïe A. Sex/gender differences in the brain and cognition in schizophrenia. Neurosci Biobehav Rev. 2016;67:57-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 218] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 37. | Li X, Wu J, Liu J, Li K, Wang F, Sun X, Ma S. The influence of marital status on the social dysfunction of schizophrenia patients in community. Int J Nursi Sci. 2015;2:149-152. [RCA] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 38. | Agerbo E, Byrne M, Eaton WW, Mortensen PB. Marital and labor market status in the long run in schizophrenia. Arch Gen Psychiatry. 2004;61:28-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 115] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 39. | Thara R, Kamath S, Kumar S. Women with schizophrenia and broken marriages--doubly disadvantaged? Part I: patient perspective. Int J Soc Psychiatry. 2003;49:225-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 51] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 40. | Chong HY, Teoh SL, Wu DB, Kotirum S, Chiou CF, Chaiyakunapruk N. Global economic burden of schizophrenia: a systematic review. Neuropsychiatr Dis Treat. 2016;12:357-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 269] [Article Influence: 26.9] [Reference Citation Analysis (1)] |
| 41. | Zaprutko T, Kus K, Bilobryvka R, Rakhman L, Göder R, Michalak M, Pogłodziński A, Nowakowska E. Schizophrenia and Employment: Evaluation From Professionals Point of View. Psychiatr Q. 2015;86:569-579. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 42. | Cloutier M, Aigbogun MS, Guerin A, Nitulescu R, Ramanakumar AV, Kamat SA, DeLucia M, Duffy R, Legacy SN, Henderson C, Francois C, Wu E. The Economic Burden of Schizophrenia in the United States in 2013. J Clin Psychiatry. 2016;77:764-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 350] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 43. | Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421-427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7241] [Cited by in RCA: 5859] [Article Influence: 488.3] [Reference Citation Analysis (0)] |
| 44. | Torrey EF, Yolken RH. Schizophrenia as a pseudogenetic disease: A call for more gene-environmental studies. Psychiatry Res. 2019;278:146-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 45. | Hindley G, Bahrami S, Steen NE, O'Connell KS, Frei O, Shadrin A, Bettella F, Rødevand L, Fan CC, Dale AM, Djurovic S, Smeland OB, Andreassen OA. Characterising the shared genetic determinants of bipolar disorder, schizophrenia and risk-taking. Transl Psychiatry. 2021;11:466. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 46. | Brainstorm Consortium; Anttila V, Bulik-Sullivan B, Finucane HK, Walters RK, Bras J, Duncan L, Escott-Price V, Falcone GJ, Gormley P, Malik R, Patsopoulos NA, Ripke S, Wei Z, Yu D, Lee PH, Turley P, Grenier-Boley B, Chouraki V, Kamatani Y, Berr C, Letenneur L, Hannequin D, Amouyel P, Boland A, Deleuze JF, Duron E, Vardarajan BN, Reitz C, Goate AM, Huentelman MJ, Kamboh MI, Larson EB, Rogaeva E, St George-Hyslop P, Hakonarson H, Kukull WA, Farrer LA, Barnes LL, Beach TG, Demirci FY, Head E, Hulette CM, Jicha GA, Kauwe JSK, Kaye JA, Leverenz JB, Levey AI, Lieberman AP, Pankratz VS, Poon WW, Quinn JF, Saykin AJ, Schneider LS, Smith AG, Sonnen JA, Stern RA, Van Deerlin VM, Van Eldik LJ, Harold D, Russo G, Rubinsztein DC, Bayer A, Tsolaki M, Proitsi P, Fox NC, Hampel H, Owen MJ, Mead S, Passmore P, Morgan K, Nöthen MM, Rossor M, Lupton MK, Hoffmann P, Kornhuber J, Lawlor B, McQuillin A, Al-Chalabi A, Bis JC, Ruiz A, Boada M, Seshadri S, Beiser A, Rice K, van der Lee SJ, De Jager PL, Geschwind DH, Riemenschneider M, Riedel-Heller S, Rotter JI, Ransmayr G, Hyman BT, Cruchaga C, Alegret M, Winsvold B, Palta P, Farh KH, Cuenca-Leon E, Furlotte N, Kurth T, Ligthart L, Terwindt GM, Freilinger T, Ran C, Gordon SD, Borck G, Adams HHH, Lehtimäki T, Wedenoja J, Buring JE, Schürks M, Hrafnsdottir M, Hottenga JJ, Penninx B, Artto V, Kaunisto M, Vepsäläinen S, Martin NG, Montgomery GW, Kurki MI, Hämäläinen E, Huang H, Huang J, Sandor C, Webber C, Muller-Myhsok B, Schreiber S, Salomaa V, Loehrer E, Göbel H, Macaya A, Pozo-Rosich P, Hansen T, Werge T, Kaprio J, Metspalu A, Kubisch C, Ferrari MD, Belin AC, van den Maagdenberg AMJM, Zwart JA, Boomsma D, Eriksson N, Olesen J, Chasman DI, Nyholt DR, Avbersek A, Baum L, Berkovic S, Bradfield J, Buono RJ, Catarino CB, Cossette P, De Jonghe P, Depondt C, Dlugos D, Ferraro TN, French J, Hjalgrim H, Jamnadas-Khoda J, Kälviäinen R, Kunz WS, Lerche H, Leu C, Lindhout D, Lo W, Lowenstein D, McCormack M, Møller RS, Molloy A, Ng PW, Oliver K, Privitera M, Radtke R, Ruppert AK, Sander T, Schachter S, Schankin C, Scheffer I, Schoch S, Sisodiya SM, Smith P, Sperling M, Striano P, Surges R, Thomas GN, Visscher F, Whelan CD, Zara F, Heinzen EL, Marson A, Becker F, Stroink H, Zimprich F, Gasser T, Gibbs R, Heutink P, Martinez M, Morris HR, Sharma M, Ryten M, Mok KY, Pulit S, Bevan S, Holliday E, Attia J, Battey T, Boncoraglio G, Thijs V, Chen WM, Mitchell B, Rothwell P, Sharma P, Sudlow C, Vicente A, Markus H, Kourkoulis C, Pera J, Raffeld M, Silliman S, Boraska Perica V, Thornton LM, Huckins LM, William Rayner N, Lewis CM, Gratacos M, Rybakowski F, Keski-Rahkonen A, Raevuori A, Hudson JI, Reichborn-Kjennerud T, Monteleone P, Karwautz A, Mannik K, Baker JH, O'Toole JK, Trace SE, Davis OSP, Helder SG, Ehrlich S, Herpertz-Dahlmann B, Danner UN, van Elburg AA, Clementi M, Forzan M, Docampo E, Lissowska J, Hauser J, Tortorella A, Maj M, Gonidakis F, Tziouvas K, Papezova H, Yilmaz Z, Wagner G, Cohen-Woods S, Herms S, Julià A, Rabionet R, Dick DM, Ripatti S, Andreassen OA, Espeseth T, Lundervold AJ, Steen VM, Pinto D, Scherer SW, Aschauer H, Schosser A, Alfredsson L, Padyukov L, Halmi KA, Mitchell J, Strober M, Bergen AW, Kaye W, Szatkiewicz JP, Cormand B, Ramos-Quiroga JA, Sánchez-Mora C, Ribasés M, Casas M, Hervas A, Arranz MJ, Haavik J, Zayats T, Johansson S, Williams N, Dempfle A, Rothenberger A, Kuntsi J, Oades RD, Banaschewski T, Franke B, Buitelaar JK, Arias Vasquez A, Doyle AE, Reif A, Lesch KP, Freitag C, Rivero O, Palmason H, Romanos M, Langley K, Rietschel M, Witt SH, Dalsgaard S, Børglum AD, Waldman I, Wilmot B, Molly N, Bau CHD, Crosbie J, Schachar R, Loo SK, McGough JJ, Grevet EH, Medland SE, Robinson E, Weiss LA, Bacchelli E, Bailey A, Bal V, Battaglia A, Betancur C, Bolton P, Cantor R, Celestino-Soper P, Dawson G, De Rubeis S, Duque F, Green A, Klauck SM, Leboyer M, Levitt P, Maestrini E, Mane S, De-Luca DM, Parr J, Regan R, Reichenberg A, Sandin S, Vorstman J, Wassink T, Wijsman E, Cook E, Santangelo S, Delorme R, Rogé B, Magalhaes T, Arking D, Schulze TG, Thompson RC, Strohmaier J, Matthews K, Melle I, Morris D, Blackwood D, McIntosh A, Bergen SE, Schalling M, Jamain S, Maaser A, Fischer SB, Reinbold CS, Fullerton JM, Guzman-Parra J, Mayoral F, Schofield PR, Cichon S, Mühleisen TW, Degenhardt F, Schumacher J, Bauer M, Mitchell PB, Gershon ES, Rice J, Potash JB, Zandi PP, Craddock N, Ferrier IN, Alda M, Rouleau GA, Turecki G, Ophoff R, Pato C, Anjorin A, Stahl E, Leber M, Czerski PM, Cruceanu C, Jones IR, Posthuma D, Andlauer TFM, Forstner AJ, Streit F, Baune BT, Air T, Sinnamon G, Wray NR, MacIntyre DJ, Porteous D, Homuth G, Rivera M, Grove J, Middeldorp CM, Hickie I, Pergadia M, Mehta D, Smit JH, Jansen R, de Geus E, Dunn E, Li QS, Nauck M, Schoevers RA, Beekman AT, Knowles JA, Viktorin A, Arnold P, Barr CL, Bedoya-Berrio G, Bienvenu OJ, Brentani H, Burton C, Camarena B, Cappi C, Cath D, Cavallini M, Cusi D, Darrow S, Denys D, Derks EM, Dietrich A, Fernandez T, Figee M, Freimer N, Gerber G, Grados M, Greenberg E, Hanna GL, Hartmann A, Hirschtritt ME, Hoekstra PJ, Huang A, Huyser C, Illmann C, Jenike M, Kuperman S, Leventhal B, Lochner C, Lyon GJ, Macciardi F, Madruga-Garrido M, Malaty IA, Maras A, McGrath L, Miguel EC, Mir P, Nestadt G, Nicolini H, Okun MS, Pakstis A, Paschou P, Piacentini J, Pittenger C, Plessen K, Ramensky V, Ramos EM, Reus V, Richter MA, Riddle MA, Robertson MM, Roessner V, Rosário M, Samuels JF, Sandor P, Stein DJ, Tsetsos F, Van Nieuwerburgh F, Weatherall S, Wendland JR, Wolanczyk T, Worbe Y, Zai G, Goes FS, McLaughlin N, Nestadt PS, Grabe HJ, Depienne C, Konkashbaev A, Lanzagorta N, Valencia-Duarte A, Bramon E, Buccola N, Cahn W, Cairns M, Chong SA, Cohen D, Crespo-Facorro B, Crowley J, Davidson M, DeLisi L, Dinan T, Donohoe G, Drapeau E, Duan J, Haan L, Hougaard D, Karachanak-Yankova S, Khrunin A, Klovins J, Kučinskas V, Lee Chee Keong J, Limborska S, Loughland C, Lönnqvist J, Maher B, Mattheisen M, McDonald C, Murphy KC, Nenadic I, van Os J, Pantelis C, Pato M, Petryshen T, Quested D, Roussos P, Sanders AR, Schall U, Schwab SG, Sim K, So HC, Stögmann E, Subramaniam M, Toncheva D, Waddington J, Walters J, Weiser M, Cheng W, Cloninger R, Curtis D, Gejman PV, Henskens F, Mattingsdal M, Oh SY, Scott R, Webb B, Breen G, Churchhouse C, Bulik CM, Daly M, Dichgans M, Faraone SV, Guerreiro R, Holmans P, Kendler KS, Koeleman B, Mathews CA, Price A, Scharf J, Sklar P, Williams J, Wood NW, Cotsapas C, Palotie A, Smoller JW, Sullivan P, Rosand J, Corvin A, Neale BM, Schott JM, Anney R, Elia J, Grigoroiu-Serbanescu M, Edenberg HJ, Murray R. Analysis of shared heritability in common disorders of the brain. Science. 2018;360:eaap8757. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 868] [Cited by in RCA: 1034] [Article Influence: 129.3] [Reference Citation Analysis (0)] |
| 47. | Cattane N, Richetto J, Cattaneo A. Prenatal exposure to environmental insults and enhanced risk of developing Schizophrenia and Autism Spectrum Disorder: focus on biological pathways and epigenetic mechanisms. Neurosci Biobehav Rev. 2020;117:253-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 87] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 48. | Havdahl A, Wootton RE, Leppert B, Riglin L, Ask H, Tesli M, Bugge Askeland R, Hannigan LJ, Corfield E, Øyen AS, Andreassen OA, Tilling K, Davey Smith G, Thapar A, Reichborn-Kjennerud T, Stergiakouli E. Associations Between Pregnancy-Related Predisposing Factors for Offspring Neurodevelopmental Conditions and Parental Genetic Liability to Attention-Deficit/Hyperactivity Disorder, Autism, and Schizophrenia: The Norwegian Mother, Father and Child Cohort Study (MoBa). JAMA Psychiatry. 2022;79:799-810. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 55] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 49. | Matsunaga M, Li Y, He Y, Kishi T, Tanihara S, Iwata N, Tabuchi T, Ota A. Physical, Psychiatric, and Social Comorbidities of Individuals with Schizophrenia Living in the Community in Japan. Int J Environ Res Public Health. 2023;20:4336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 50. | He Y, Matsunaga M, Li Y, Kishi T, Tanihara S, Iwata N, Tabuchi T, Ota A. Classifying Schizophrenia Cases by Artificial Neural Network Using Japanese Web-Based Survey Data: Case-Control Study. JMIR Form Res. 2023;7:e50193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 51. | Sharaev MG, Malashenkova IK, Maslennikova AV, Zakharova NV, Bernstein AV, Burnaev EV, Mamedova GS, Krynskiy SA, Ogurtsov DP, Kondrateva EA, Druzhinina PV, Zubrikhina MO, Arkhipov AY, Strelets VB, Ushakov VL. Diagnosis of Schizophrenia Based on the Data of Various Modalities: Biomarkers and Machine Learning Techniques (Review). Sovrem Tekhnologii Med. 2022;14:53-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 52. | Zhang W, Yang C, Cao Z, Li Z, Zhuo L, Tan Y, He Y, Yao L, Zhou Q, Gong Q, Sweeney JA, Shi F, Lui S. Detecting individuals with severe mental illness using artificial intelligence applied to magnetic resonance imaging. EBioMedicine. 2023;90:104541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 53. | Yang QX, Wang YX, Li FC, Zhang S, Luo YC, Li Y, Tang J, Li B, Chen YZ, Xue WW, Zhu F. Identification of the gene signature reflecting schizophrenia's etiology by constructing artificial intelligence-based method of enhanced reproducibility. CNS Neurosci Ther. 2019;25:1054-1063. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 54. | Coury SM, Lombroso A, Avila-Quintero VJ, Taylor JH, Flores JM, Szejko N, Bloch MH. Systematic review and meta-analysis: Season of birth and schizophrenia risk. Schizophr Res. 2023;252:244-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 55. | Hsu CW, Tseng PT, Tu YK, Lin PY, Hung CF, Liang CS, Hsieh YY, Yang YH, Wang LJ, Kao HY. Month of birth and mental disorders: A population-based study and validation using global meta-analysis. Acta Psychiatr Scand. 2021;144:153-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 56. | Cumming P, Abi-Dargham A, Gründer G. Molecular imaging of schizophrenia: Neurochemical findings in a heterogeneous and evolving disorder. Behav Brain Res. 2021;398:113004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 57. | Dienel SJ, Fish KN, Lewis DA. The Nature of Prefrontal Cortical GABA Neuron Alterations in Schizophrenia: Markedly Lower Somatostatin and Parvalbumin Gene Expression Without Missing Neurons. Am J Psychiatry. 2023;180:495-507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 54] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 58. | Rosenbrock H, Desch M, Wunderlich G. Development of the novel GlyT1 inhibitor, iclepertin (BI 425809), for the treatment of cognitive impairment associated with schizophrenia. Eur Arch Psychiatry Clin Neurosci. 2023;273:1557-1566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 66] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 59. | Davidson M, Saoud J, Staner C, Noel N, Werner S, Luthringer E, Walling D, Weiser M, Harvey PD, Strauss GP, Luthringer R. Efficacy and Safety of Roluperidone for the Treatment of Negative Symptoms of Schizophrenia. Schizophr Bull. 2022;48:609-619. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 51] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 60. | Sommer IE, Kahn RS. The contribution of neuroimaging to understanding schizophrenia; past, present, and future. Schizophr Bull. 2015;41:1-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 61. | Hederih J, Nuninga JO, van Eijk K, van Dellen E, Smit DJA, Oranje B, Luykx JJ. Genetic underpinnings of schizophrenia-related electroencephalographical intermediate phenotypes: A systematic review and meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry. 2021;104:110001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Schijven D, Postema MC, Fukunaga M, Matsumoto J, Miura K, de Zwarte SMC, van Haren NEM, Cahn W, Hulshoff Pol HE, Kahn RS, Ayesa-Arriola R, Ortiz-García de la Foz V, Tordesillas-Gutierrez D, Vázquez-Bourgon J, Crespo-Facorro B, Alnæs D, Dahl A, Westlye LT, Agartz I, Andreassen OA, Jönsson EG, Kochunov P, Bruggemann JM, Catts SV, Michie PT, Mowry BJ, Quidé Y, Rasser PE, Schall U, Scott RJ, Carr VJ, Green MJ, Henskens FA, Loughland CM, Pantelis C, Weickert CS, Weickert TW, de Haan L, Brosch K, Pfarr JK, Ringwald KG, Stein F, Jansen A, Kircher TTJ, Nenadić I, Krämer B, Gruber O, Satterthwaite TD, Bustillo J, Mathalon DH, Preda A, Calhoun VD, Ford JM, Potkin SG, Chen J, Tan Y, Wang Z, Xiang H, Fan F, Bernardoni F, Ehrlich S, Fuentes-Claramonte P, Garcia-Leon MA, Guerrero-Pedraza A, Salvador R, Sarró S, Pomarol-Clotet E, Ciullo V, Piras F, Vecchio D, Banaj N, Spalletta G, Michielse S, van Amelsvoort T, Dickie EW, Voineskos AN, Sim K, Ciufolini S, Dazzan P, Murray RM, Kim WS, Chung YC, Andreou C, Schmidt A, Borgwardt S, McIntosh AM, Whalley HC, Lawrie SM, du Plessis S, Luckhoff HK, Scheffler F, Emsley R, Grotegerd D, Lencer R, Dannlowski U, Edmond JT, Rootes-Murdy K, Stephen JM, Mayer AR, Antonucci LA, Fazio L, Pergola G, Bertolino A, Díaz-Caneja CM, Janssen J, Lois NG, Arango C, Tomyshev AS, Lebedeva I, Cervenka S, Sellgren CM, Georgiadis F, Kirschner M, Kaiser S, Hajek T, Skoch A, Spaniel F, Kim M, Kwak YB, Oh S, Kwon JS, James A, Bakker G, Knöchel C, Stäblein M, Oertel V, Uhlmann A, Howells FM, Stein DJ, Temmingh HS, Diaz-Zuluaga AM, Pineda-Zapata JA, López-Jaramillo C, Homan S, Ji E, Surbeck W, Homan P, Fisher SE, Franke B, Glahn DC, Gur RC, Hashimoto R, Jahanshad N, Luders E, Medland SE, Thompson PM, Turner JA, van Erp TGM, Francks C. Large-scale analysis of structural brain asymmetries in schizophrenia via the ENIGMA consortium. Proc Natl Acad Sci U S A. 2023;120:e2213880120. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 64] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 63. | Haijma SV, Van Haren N, Cahn W, Koolschijn PC, Hulshoff Pol HE, Kahn RS. Brain volumes in schizophrenia: a meta-analysis in over 18 000 subjects. Schizophr Bull. 2013;39:1129-1138. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 829] [Cited by in RCA: 736] [Article Influence: 56.6] [Reference Citation Analysis (0)] |
| 64. | Cattarinussi G, Kubera KM, Hirjak D, Wolf RC, Sambataro F. Neural Correlates of the Risk for Schizophrenia and Bipolar Disorder: A Meta-analysis of Structural and Functional Neuroimaging Studies. Biol Psychiatry. 2022;92:375-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 65. | Cui Y, Li C, Liu B, Sui J, Song M, Chen J, Chen Y, Guo H, Li P, Lu L, Lv L, Ning Y, Wan P, Wang H, Wang H, Wu H, Yan H, Yan J, Yang Y, Zhang H, Zhang D, Jiang T. Consistent brain structural abnormalities and multisite individualised classification of schizophrenia using deep neural networks. Br J Psychiatry. 2022;221:732-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 66. | Howes OD, Murray RM. Schizophrenia: an integrated sociodevelopmental-cognitive model. Lancet. 2014;383:1677-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 594] [Cited by in RCA: 622] [Article Influence: 51.8] [Reference Citation Analysis (2)] |
| 67. | Gao X, Zhang W, Yao L, Xiao Y, Liu L, Liu J, Li S, Tao B, Shah C, Gong Q, Sweeney JA, Lui S. Association between structural and functional brain alterations in drug-free patients with schizophrenia: a multimodal meta-analysis. J Psychiatry Neurosci. 2018;43:131-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 68. | Fišar Z. Biological hypotheses, risk factors, and biomarkers of schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2023;120:110626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 62] [Article Influence: 20.7] [Reference Citation Analysis (0)] |
| 69. | Di Camillo F, Grimaldi DA, Cattarinussi G, Di Giorgio A, Locatelli C, Khuntia A, Enrico P, Brambilla P, Koutsouleris N, Sambataro F. Magnetic resonance imaging-based machine learning classification of schizophrenia spectrum disorders: a meta-analysis. Psychiatry Clin Neurosci. 2024;78:732-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 70. | Horton LE, Smith AA, Haas GL. The nature and timing of social deficits in child and adolescent offspring of parents with schizophrenia: preliminary evidence for precursors of negative symptoms? Schizophr Res. 2014;159:27-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 71. | Bucci P, Galderisi S, Mucci A, Rossi A, Rocca P, Bertolino A, Aguglia E, Amore M, Andriola I, Bellomo A, Biondi M, Cuomo A, dell'Osso L, Favaro A, Gambi F, Giordano GM, Girardi P, Marchesi C, Monteleone P, Montemagni C, Niolu C, Oldani L, Pacitti F, Pinna F, Roncone R, Vita A, Zeppegno P, Maj M; Italian Network for Research on Psychoses. Premorbid academic and social functioning in patients with schizophrenia and its associations with negative symptoms and cognition. Acta Psychiatr Scand. 2018;138:253-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 72. | Giordano GM, Bucci P, Mucci A, Pezzella P, Galderisi S. Gender Differences in Clinical and Psychosocial Features Among Persons With Schizophrenia: A Mini Review. Front Psychiatry. 2021;12:789179. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 72] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 73. | Soygür H, Alptekin K, Atbaşoğlu EC, Herken H. Şizofreni ve diğer psikotik bozukluklar. Türkiye Psikiyatri Derneği Yayınları. 2007;. |
| 74. | Molstrom IM, Nordgaard J, Urfer-Parnas A, Handest R, Berge J, Henriksen MG. The prognosis of schizophrenia: A systematic review and meta-analysis with meta-regression of 20-year follow-up studies. Schizophr Res. 2022;250:152-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 53] [Reference Citation Analysis (0)] |
| 75. | Jääskeläinen E, Juola P, Hirvonen N, McGrath JJ, Saha S, Isohanni M, Veijola J, Miettunen J. A systematic review and meta-analysis of recovery in schizophrenia. Schizophr Bull. 2013;39:1296-1306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 711] [Cited by in RCA: 655] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 76. | Kerna NA, Boulos A, Abreu M, Chigozie IK, Fide-nwoko FU, Arube ER, Eziechi ES, Holets HM, Pruitt KD, Jomsky BM, Chawla S. Current Applications of Artificial Intelligence in Psychiatry. Sci, Technol, Sci Soc. 2025;2:125-143. [DOI] [Full Text] |
| 77. | Stahl SM. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. 5th ed. United Kingdom: Cambridge University Press, 2021. [DOI] [Full Text] |
| 78. | Samara MT, Leucht C, Leeflang MM, Anghelescu IG, Chung YC, Crespo-Facorro B, Elkis H, Hatta K, Giegling I, Kane JM, Kayo M, Lambert M, Lin CH, Möller HJ, Pelayo-Terán JM, Riedel M, Rujescu D, Schimmelmann BG, Serretti A, Correll CU, Leucht S. Early Improvement As a Predictor of Later Response to Antipsychotics in Schizophrenia: A Diagnostic Test Review. Am J Psychiatry. 2015;172:617-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 119] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 79. | Correll CU, Citrome L, Haddad PM, Lauriello J, Olfson M, Calloway SM, Kane JM. The Use of Long-Acting Injectable Antipsychotics in Schizophrenia: Evaluating the Evidence. J Clin Psychiatry. 2016;77:1-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 301] [Article Influence: 33.4] [Reference Citation Analysis (0)] |
| 80. | Del Fabro L, Bondi E, Serio F, Maggioni E, D'Agostino A, Brambilla P. Machine learning methods to predict outcomes of pharmacological treatment in psychosis. Transl Psychiatry. 2023;13:75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 81. | Wu CS, Luedtke AR, Sadikova E, Tsai HJ, Liao SC, Liu CC, Gau SS, VanderWeele TJ, Kessler RC. Development and Validation of a Machine Learning Individualized Treatment Rule in First-Episode Schizophrenia. JAMA Netw Open. 2020;3:e1921660. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 31] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 82. | Barruel D, Hilbey J, Charlet J, Chaumette B, Krebs MO, Dauriac-Le Masson V. Predicting treatment resistance in schizophrenia patients: Machine learning highlights the role of early pathophysiologic features. Schizophr Res. 2024;270:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 84. | Meltzer HY. Treatment of the neuroleptic-nonresponsive schizophrenic patient. Schizophr Bull. 1992;18:515-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 182] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 85. | Öztürk MO, Uluşahin NA. Ruh sağlığı ve bozuklukları. Nobel Tıp Kitabevleri. 2015. |
| 86. | Allesøe RL, Thompson WK, Bybjerg-Grauholm J, Hougaard DM, Nordentoft M, Werge T, Rasmussen S, Benros ME. Deep Learning for Cross-Diagnostic Prediction of Mental Disorder Diagnosis and Prognosis Using Danish Nationwide Register and Genetic Data. JAMA Psychiatry. 2023;80:146-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 26] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 87. | Rivera LA, Costa-dookhan K, Dissanayake J, Siddiqui I, Rotenberg M, Hahn M, Maslej M, Foussias G. Machine Learning Methods and Schizophrenia Spectrum Disorders: A Scoping Review. 2025 Preprint. Available from: JMIR Preprints: 72036. [DOI] [Full Text] |
| 88. | Sun J, Dong QX, Wang SW, Zheng YB, Liu XX, Lu TS, Yuan K, Shi J, Hu B, Lu L, Han Y. Artificial intelligence in psychiatry research, diagnosis, and therapy. Asian J Psychiatr. 2023;87:103705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 76] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 89. | Manamalage DM, Rao RN, Bilal M, Cheung G, Thabrew H, Roop PS, Shahamiri SR, Sundram F. With data in mind: AI applications in depression, autism and dementia care. New Zealand Med Student J. 2025;0:7-11. [DOI] [Full Text] |
| 90. | Rosenfeld A, Benrimoh D, Armstrong C, Mirchi N, Langlois-therrien T, Rollins C, Tanguay-sela M, Mehltretter J, Fratila R, Israel S, Snook E, Perlman K, Kleinerman A, Saab B, Thoburn M, Gabbay C, Yaniv-rosenfeld A. Big Data analytics and artificial intelligence in mental healthcare. In: Khanna A, Gupta D, Dey N, editors. Applications of Big Data in Healthcare. Amsterdam: Academic Press, 2021: 137-171. [DOI] [Full Text] |
| 91. | Kumar TS, Narayanan DM, Kamatham DH. Deep And Machine Learning in Psychology- A Survey of Depression Detection, Diagnosis, and Treatment Progress. Int J Innov Res Eng Manag. 2024;11:22-31. [DOI] [Full Text] |
| 92. | Chandler C, Foltz PW, Elvevåg B. Improving the Applicability of AI for Psychiatric Applications through Human-in-the-loop Methodologies. Schizophr Bull. 2022;48:949-957. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 25] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 93. | Antoniou G, Papadakis E, Baryannis G. Mental Health Diagnosis: A Case for Explainable Artificial Intelligence. Int J Artif Intell Tools. 2022;31:2241003. [DOI] [Full Text] |
| 94. | Eskandar K. Artificial Intelligence in Psychiatry: A Double-Edged Sword? J Psych Insight Rev. 2025;1:1-4. [DOI] [Full Text] |
| 95. | Saha S. AI-driven Treatment Planning for Mental Health Practitioners. Int J Res Appl Sci Eng Technol. 2025. [DOI] [Full Text] |
| 96. | Muslehat A, Samuel O, Oluwakemi A, Ephraim N. A Methodological Review of Artificial Intelligence Technology Application in Mental Health Disorders. 2025 Preprint. Available from: preprints: 202505.2312.v1. [DOI] [Full Text] |
| 97. | Blease C, Torous J. ChatGPT and mental healthcare: balancing benefits with risks of harms. BMJ Ment Health. 2023;26:e300884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 58] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 98. | Volpe U. What can AI do for mental health care. Eur Psychiatr. 2025;68:S61-S62. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 99. | Alemi F, Wojtusiak J, Ursani A, Eklou KP, Lybarger K. Artificial Intelligence for Management of Major Depression: Initial Design, Progress, and Research Plans. Alpha Psychiatry. 2025;26:44608. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 100. | Joshi S, Pandey MK, Solanki B, Rajput PK, Singh MS, Praveen H, Gurav M. Magnetic Resonance Imaging (MRI) Insights into Brain Morphology and Connectivity Disruptions in Schizophrenia. Int Neuropsych Dis J. 2024;21:6-10. [DOI] [Full Text] |
| 101. | Pomarol-Clotet E, Canales-Rodríguez EJ, Salvador R, Sarró S, Gomar JJ, Vila F, Ortiz-Gil J, Iturria-Medina Y, Capdevila A, McKenna PJ. Medial prefrontal cortex pathology in schizophrenia as revealed by convergent findings from multimodal imaging. Mol Psychiatry. 2010;15:823-830. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 132] [Cited by in RCA: 157] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 102. | Kang Y, Huang K, Lin Y, Xu H, Zhang W, Lv Y, Cai S, Huang L. Abnormalities of effective connectivity and white matter microstructure in the triple network in patients with schizophrenia. Psychiatry Res. 2020;290:113019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 103. | Nakamura K, Kawasaki Y, Takahashi T, Furuichi A, Noguchi K, Seto H, Suzuki M. Reduced white matter fractional anisotropy and clinical symptoms in schizophrenia: a voxel-based diffusion tensor imaging study. Psychiatry Res. 2012;202:233-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 68] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 104. | Tanaka-Koshiyama K, Koshiyama D, Miyakoshi M, Joshi YB, Molina JL, Sprock J, Braff DL, Light GA. Abnormal Spontaneous Gamma Power Is Associated With Verbal Learning and Memory Dysfunction in Schizophrenia. Front Psychiatry. 2020;11:832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 105. | Koshiyama D, Miyakoshi M, Tanaka-Koshiyama K, Joshi YB, Sprock J, Braff DL, Light GA. Abnormal phase discontinuity of alpha- and theta-frequency oscillations in schizophrenia. Schizophr Res. 2021;231:73-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 106. | Kim HK, Blumberger DM, Daskalakis ZJ. Neurophysiological Biomarkers in Schizophrenia-P50, Mismatch Negativity, and TMS-EMG and TMS-EEG. Front Psychiatry. 2020;11:795. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 107. | Tamilarasi K, Pavithra S, AnujS. An Effective Detection and Classification of Schizophrenia Patients from Eeg Signals Using Machine Learning Algorithms. 2023 Preprint. Available from: SSRN: 4587644. [DOI] [Full Text] |
| 108. | Shanarova N, Pronina M, Lipkovich M, Ponomarev V, Müller A, Kropotov J. Application of Machine Learning to Diagnostics of Schizophrenia Patients Based on Event-Related Potentials. Diagnostics (Basel). 2023;13:509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 109. | Al Mazroa A, Eltahir MM, Ebad SA, Alotaibi FA, K V, Cho J. EEG-based schizophrenia diagnosis using deep learning with multi-scale and adaptive feature selection. PeerJ Comput Sci. 2025;11:e2811. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 110. | Shoeibi A, Sadeghi D, Moridian P, Ghassemi N, Heras J, Alizadehsani R, Khadem A, Kong Y, Nahavandi S, Zhang YD, Gorriz JM. Automatic Diagnosis of Schizophrenia in EEG Signals Using CNN-LSTM Models. Front Neuroinform. 2021;15:777977. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 85] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 111. | Cecchi M, Adachi M, Basile A, Buhl DL, Chadchankar H, Christensen S, Christian E, Doherty J, Fadem KC, Farley B, Forman MS, Honda S, Johannesen J, Kinon BJ, Klamer D, Marino MJ, Missling C, O'Donnell P, Piser T, Puryear CB, Quirk MC, Rotte M, Sanchez C, Smith DG, Uslaner JM, Javitt DC, Keefe RSE, Mathalon D, Potter WZ, Walling DP, Ereshefsky L. Validation of a suite of ERP and QEEG biomarkers in a pre-competitive, industry-led study in subjects with schizophrenia and healthy volunteers. Schizophr Res. 2023;254:178-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 112. | Alazzawı A, Aljumaili S, Duru AD, Uçan ON, Bayat O, Coelho PJ, Pires IM. Schizophrenia diagnosis based on diverse epoch size resting-state EEG using machine learning. PeerJ Comput Sci. 2024;10:e2170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 113. | Klosterkötter J, Hellmich M, Steinmeyer EM, Schultze-Lutter F. Diagnosing schizophrenia in the initial prodromal phase. Arch Gen Psychiatry. 2001;58:158-164. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 801] [Cited by in RCA: 654] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 114. | Larson MK, Walker EF, Compton MT. Early signs, diagnosis and therapeutics of the prodromal phase of schizophrenia and related psychotic disorders. Expert Rev Neurother. 2010;10:1347-1359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 152] [Cited by in RCA: 128] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 115. | Wikipedia C. Artificial intelligence in mental health. [cited 23 January 2026]. Available from: https://en.wikipedia.org/w/index.php?title=Artificial_intelligence_in_mental_health&oldid=1317814016. |
| 116. | Aine CJ, Bockholt HJ, Bustillo JR, Cañive JM, Caprihan A, Gasparovic C, Hanlon FM, Houck JM, Jung RE, Lauriello J, Liu J, Mayer AR, Perrone-Bizzozero NI, Posse S, Stephen JM, Turner JA, Clark VP, Calhoun VD. Multimodal Neuroimaging in Schizophrenia: Description and Dissemination. Neuroinformatics. 2017;15:343-364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 126] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 117. | Dabiri M, Dehghani Firouzabadi F, Yang K, Barker PB, Lee RR, Yousem DM. Neuroimaging in schizophrenia: A review article. Front Neurosci. 2022;16:1042814. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 118. | Comai S, Manchia M, Bosia M, Miola A, Poletti S, Benedetti F, Nasini S, Ferri R, Rujescu D, Leboyer M, Licinio J, Baune BT, Serretti A. Moving toward precision and personalized treatment strategies in psychiatry. Int J Neuropsychopharmacol. 2025;28:pyaf025. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 119. | Tanaka M. From Serendipity to Precision: Integrating AI, Multi-Omics, and Human-Specific Models for Personalized Neuropsychiatric Care. Biomedicines. 2025;13:167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 120. | Sarisik E, Popovic D, Keeser D, Khuntia A, Schiltz K, Falkai P, Pogarell O, Koutsouleris N. EEG-based Signatures of Schizophrenia, Depression, and Aberrant Aging: A Supervised Machine Learning Investigation. Schizophr Bull. 2025;51:804-817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 121. | Naderi M, Jahanian-Najafabadi A. A systematic review of EEG-based machine learning classifications for obsessive-compulsive disorder: current status and future directions. BMC Psychiatry. 2025;25:854. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 122. | World Health Organization. Artificial intelligence in mental health research: new WHO study on applications and challenges. [cited 14 January 2026]. Available from: https://www.who.int/europe/news-room/06-02-2023-artificial-intelligence-in-mental-health-research--new-who-study-on-applications-and-challenges. |
| 123. | Yap HY, Choo YH, Mohd Yusoh ZI, Khoh WH. An evaluation of transfer learning models in EEG-based authentication. Brain Inform. 2023;10:19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 124. | Zeng H, Zhang J, Zakaria W, Babiloni F, Gianluca B, Li X, Kong W. InstanceEasyTL: An Improved Transfer-Learning Method for EEG-Based Cross-Subject Fatigue Detection. Sensors (Basel). 2020;20:7251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 125. | Liu T, Zhang Y, Wu Y, Li T, Li Z, Chen W, Zhang S. Transfer Learning based Efficient Schizophrenia Classification on Electroencephalogram (EEG) Signals: A Cross-Dataset Study. Proceedings of the 2023 15th International Conference on Bioinformatics and Biomedical Technology; 2023 May 26-28; Xi’an, China. New York: Association for Computing Machinery, 2023: 143-148. [DOI] [Full Text] |
| 126. | Kuntz L. Theta-Band Responses: The EEG Biomarker to Objectively Identify Patients With Cognitive Impairment in Schizophrenia. [cited 14 January 2026]. Available from: https://www.psychiatrictimes.com/view/theta-band-responses-the-eeg-biomarker-to-objectively-identify-patients-with-cognitive-impairment-in-schizophrenia. |
| 127. | Kaya B. Şizofreninin Tanısında Yapay Zekanın Kullanımına Dair Derleme. J Criminol Soc Law. 2024;10:1-19. [DOI] [Full Text] |
| 128. | Just SA, Bröcker AL, Ryazanskaya G, Nenchev I, Schneider M, Bermpohl F, Heinz A, Montag C. Validation of natural language processing methods capturing semantic incoherence in the speech of patients with non-affective psychosis. Front Psychiatry. 2023;14:1208856. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 129. | Byeon H. Advances in Machine Learning and Explainable Artificial Intelligence for Depression Prediction. J Adv Comput Sci Appl. 2023;14. [RCA] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 130. | Joyce DW, Kormilitzin A, Smith KA, Cipriani A. Explainable artificial intelligence for mental health through transparency and interpretability for understandability. NPJ Digit Med. 2023;6:6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 95] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 131. | Avula VCR, Amalakanti S. Applications of Artificial Intelligence in Psychiatry: Current Status, Trends, and Challenges: A Recent Review. Int J Neuropsychopharmacol. 2025;28:i213-i213. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 132. | Muhammad AE, Yow K. Demystifying Canada’s Artificial Intelligence and Data Act (AIDA): The good, the bad and the unclear elements. 2023 IEEE Canadian Conference on Electrical and Computer Engineering (CCECE), Regina, SK, Canada, 2023: 510-515. [DOI] [Full Text] |
| 133. | Brown D. Canada’s Proposed Artificial Intelligence and Data Act (AIDA): A Critical Review. 2024 Preprint. Available from: SSRN: 4687995. [DOI] [Full Text] |
| 134. | Räz T, Pahud De Mortanges A, Reyes M. Explainable AI in medicine: challenges of integrating XAI into the future clinical routine. Front Radiol. 2025;5:1627169. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 135. | Filippis RD, Foysal AA. Interpretable Machine Learning Framework for Predicting Treatment Resistance in Psychiatric Disorders Using Synthetic Pharmacogenomic and Clinical Data. Open Access Libr J. 2025;12:e13870. [DOI] [Full Text] |
| 136. | Rony MKK, Das DC, Khatun MT, Ferdousi S, Akter MR, Khatun MA, Begum MH, Khalil MI, Parvin MR, Alrazeeni DM, Akter F. Artificial intelligence in psychiatry: A systematic review and meta-analysis of diagnostic and therapeutic efficacy. Digit Health. 2025;11:20552076251330528. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |