Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.113822
Revised: December 16, 2025
Accepted: February 5, 2026
Published online: May 19, 2026
Processing time: 183 Days and 0.6 Hours
Conventional antipsychotic treatment combined with health education shows limited efficacy in patients with schizophrenia, highlighting the need for further optimization of therapeutic strategies.
To evaluate the therapeutic efficacy of exercise-based rehabilitation training in pa
From February 2022 to February 2025, 127 patients with schizophrenia were divided into a control group (n = 59) managed with routine pharmacotherapy and an intervention group (n = 68) receiving additional exercise-based rehabilitation training. Outcomes were assessed before and after the intervention using the Positive and Negative Syndrome Scale, the Nurses’ Observation Scale for Inpa
After the intervention, the intervention group demonstrated significantly greater reductions in Positive and Negative Syndrome Scale scores, NOSIE negative fa
Exercise-based rehabilitation training significantly improves psychiatric symptoms, cognitive performance, social functioning, and biological markers in patients with schizophrenia, supporting its incorporation as an adjunctive component of standard treatment.
Core Tip: This study evaluated exercise-based rehabilitation training in 127 patients with schizophrenia using a comprehensive program integrating motor skills, dynamic cognitive challenges, and structured social interaction. The intervention demonstrated clear advantages in alleviating clinical symptoms and improving cognitive function, information processing speed, and social functioning, supporting its value as an effective adjunctive treatment option.
- Citation: Wang FL, Sima CX, Zhang SJ, Hou TF. Effects of exercise-based rehabilitation training in schizophrenia patients. World J Psychiatry 2026; 16(5): 113822
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/113822.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.113822
Schizophrenia is a complex psychiatric syndrome characterized by abnormalities in mental and motor functioning. It is highly heritable, clinically heterogenous, and typically follows a chronic and relapsing course. Affected individuals display varying degrees of psychiatric symptoms, negative manifestations, cognitive impairments, affective dysregulation, and movement disorders[1]. These deficits impair information processing, attention, working memory, language learning and recall, visuospatial abilities, reasoning, and social cognition, ultimately leading to marked functional disability and occupational instability[2]. Epidemiological data indicate that the prevalence of schizophrenia has gradually increased in countries with a higher Social Demographic Index. Men tend to experience an earlier age at onset and bear a greater disease-related social burden[3]. The neurobiological basis of schizophrenia involves dysregulation of multiple neurotransmitter systems across distinct brain regions, including hyperactivity of glutamate/dopamine signaling in the basal ganglia and hypoactivity of dopamine, glutamate, and gamma-aminobutyric acid in cortical areas, particularly the frontal cortex[4]. Currently, pharmacological treatments primarily rely on dopamine D2 receptor ant
Building upon standard antipsychotic treatment and health education, this study investigates whether adjunctive exercise-based rehabilitation training improves schizophrenia outcomes. Given the limited evidence on this combined approach, our findings contribute important data to an underexplored area.
Participants were eligible if they met all the following criteria: A diagnosis of schizophrenia according to established criteria[8]; stable disease with multiple episodes; good tolerance to antipsychotic medication; cohabitation with at least one caregiver; protocol adherence capability; unimpaired cognition/communication; and complete records. The exclusion criteria included substance-induced psychosis, deaf-mute individuals, cognitive-impairing neurological conditions, administration of any non-antipsychotic drugs that may affect cognitive function (except for trihexyphenidyl or benzo
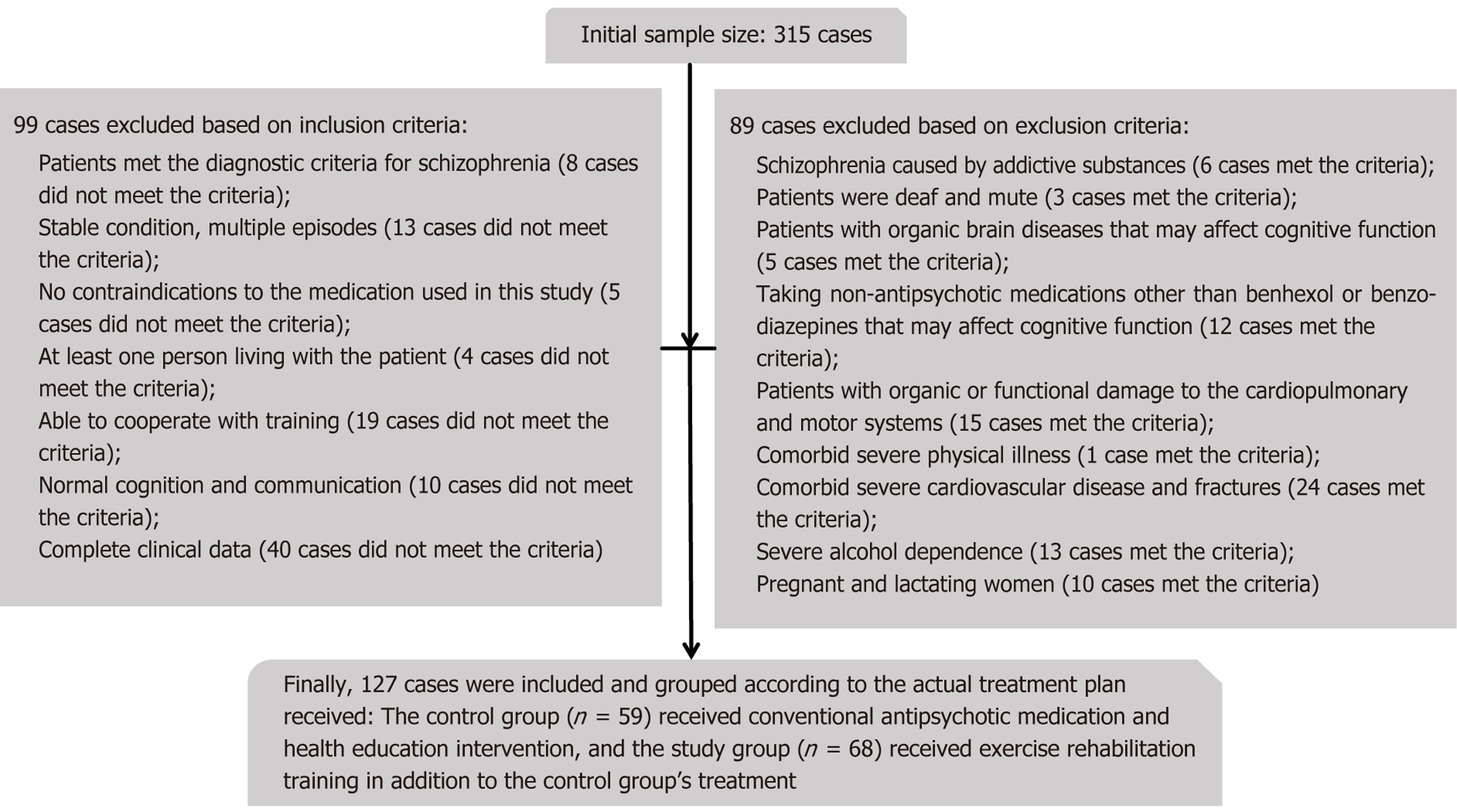
A total of 127 eligible schizophrenia patients treated during a three-year observation window (February 2022 to February 2025) were retrospectively included. Grouping was based on the treatment plan that each patient received. Of these, 59 patients receiving standard pharmacotherapy and health education were allocated to the control group, while 68 patients receiving supplemental exercise-based rehabilitation training were assigned to the intervention group. Baseline demographic and clinical characteristics did not differ significantly between the two groups (P > 0.05). The flowchart for patient inclusion and exclusion is presented in Figure 1.
Patients in the control group were maintained on conventional antipsychotic medication paired with health education. Antipsychotic drug treatment: The patients were administered Risperidone Tablets (Whenzhou KeMiao Biological Technology Co., Ltd., KM11140437), starting at a dose of 1 mg once daily, one tablet at a time. One week later, the dose could be adjusted based on the patient’s condition and adverse reactions, increasing to 2-4 mg, once or twice daily, two tablets each time. The treatment was continued for 8 weeks. Clinical staff implemented the following components: (1) Vigilant monitoring of psychiatric symptoms and stratified safety surveillance, prioritizing high-risk cases for incident prevention; (2) Supervised medication to promote adherence and therapeutic alliance, coupled with structured education on relapse prevention; and (3) Scheduled rehabilitation activities including self-care skill development (personal hygiene, dressing) and group-based activities (radio calisthenics, musical engagement) delivered in 30-45 minutes sessions, 3-5 times per week.
In addition to the above interventions, patients in the intervention group received exercise-based rehabilitation training. Training sessions lasted 50 minutes and were conducted 5 times per week for 8 weeks (total of 40 sessions). All sessions were supervised by both nurses and physical therapists. Each session began with a 10-minute warm-up consisting of 5 minutes of alternating slow and brisk walking followed by 5 minutes of dynamic stretching. The re
Psychiatric symptom improvement assessment: Psychiatric symptoms were compared pre- and post-intervention using the Positive and Negative Syndrome Scale, which assesses positive (7 items), negative (7 items), and general psychiatric symptoms (16 items) on a 7-point scale ranging from 1 to 7[9]. A reduction in total score reflects symptomatic improve
Nurse’s Observation Scale for Inpatient Evaluation scoring: The Nurse’s Observation Scale for Inpatient Evaluation (NOSIE) was administered by nursing personnel at baseline and after the 8-week intervention[10]. Symptom frequency was graded on a 5-point scale ranging from 0 to 4 (0 = none, 1 = occasional, 2 = moderate, 3 = frequent, 4 = persistent). The 30-item scale included 14 positive and 16 negative items. Positive factors (social competence, social interest, and personal hygiene) yielded a subtotal ranging from 0 to 112. Negative factors (agitation, psychotic symptoms, withdrawal, and depression) yielded a subtotal ranging from 0 to 128. The composite illness score was (128 + positive total - negative total), with a maximum score of 240. Higher total scores reflect better overall clinical status.
Stroop Color-Word Test performance: Stroop Color-Word Test (SCWT) outcomes (word/color/total correct responses) were measured pre- and post-8-week intervention[11]. Participants were instructed to read aloud words, colors, or color-words from a test card as quickly as possible within 45 seconds. The number of correct responses in each condition and the total score were recorded, with higher scores indicating better cognitive flexibility, selective attention, and the ability to suppress automatic response tendencies.
Information processing speed: To gauge processing speed, patients completed the Trail Making Test (TMT) and the Brief Assessment of Cognition in Schizophrenia (BACS-SC) before and after the 8-week intervention[12]. For the TMT, participants were instructed to connect numbers from 1 to 25 in ascending order as quickly and accurately as possible. Shorter completion times and fewer errors indicated better processing speed. For the BACS-SC, the number of correctly matched symbols to digits within a 90-second time limit was recorded, with higher scores reflecting faster information processing.
Assessment of social functional deficits: Patient social functioning was evaluated using the Inpatient Psychiatric Rehabilitation Outcome Scale (IPROS) at baseline and after the 8-week intervention. Each item was rated on a 5-point scale ranging from 0 (no symptoms) to 4 (most severe symptoms), with intermediate levels of 1 (mild), 2 (medium), and 3 (significant)[13]. The total score ranged from 0 to 144 and comprised five domains: Occupational therapy performance (maximum 32 points), daily living skills (maximum 32 points), interpersonal skills (maximum 24 points), hygiene practices (maximum 24 points), and engagement/motivation levels (maximum 32 points). Lower total scores indicated better social and functional outcomes.
Serum BDNF quantification: Baseline and post-treatment (8-week) venous blood draws (5 mL, fasting state) were per
Statistical analyses were conducted using SPSS 20.0. Categorical variables were summarized as n (%), and continuous variables as mean ± Standard Deviation. Between-group differences were assessed for statistical significance using the χ2 test for categorical variables and independent samples t-tests for continuous variables. Within-group changes before and after the intervention were analyzed using paired-samples t-tests. A significance threshold of 0.05 was applied.
An analysis of the baseline data showed no significant differences between the control and intervention groups with respect to sex distribution, age, duration of illness, length of hospital stay, educational level, or types of antipsychotic medication used (all P > 0.05), indicating good baseline comparability (Table 1).
| Categories | Control group (n = 59) | Research group (n = 68) | χ2/t value | P value |
| Sex | 1.106 | 0.293 | ||
| Male | 35 (59.32) | 34 (50.00) | ||
| Female | 24 (40.68) | 34 (50.00) | ||
| Age (years), mean ± SD | 43.03 ± 8.72 | 40.03 ± 10.59 | 1.726 | 0.087 |
| Disease course (years), mean ± SD | 8.49 ± 3.91 | 7.94 ± 3.49 | 0.838 | 0.404 |
| Hospital stay duration (months), mean ± SD | 10.31 ± 2.82 | 10.56 ± 3.74 | 0.420 | 0.675 |
| Educational background (years), mean ± SD | 6.75 ± 2.64 | 7.09 ± 3.64 | 0.594 | 0.553 |
| Main antipsychotics | 2.986 | 0.225 | ||
| Clozapine | 42 (71.19) | 40 (58.82) | ||
| Risperidone | 6 (10.17) | 14 (20.59) | ||
| Other | 11 (18.64) | 14 (20.59) | ||
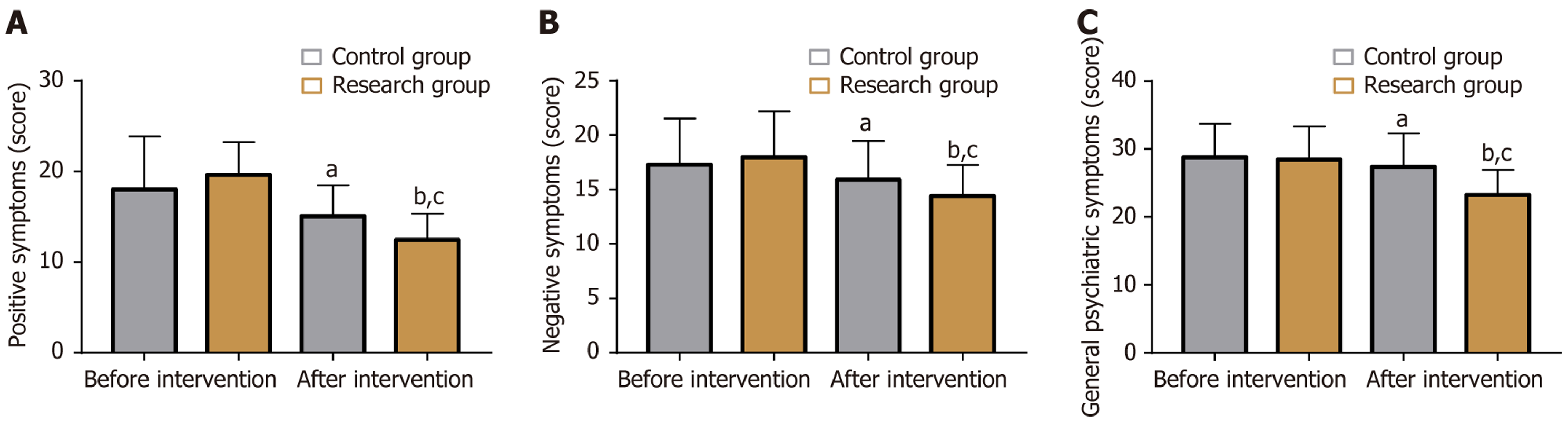
Changes in psychiatric symptoms assessed by the Positive and Negative Syndrome Scale are shown in Figure 2. At baseline, scores for positive, negative, and general psychiatric symptoms were comparable between the two groups (P > 0.05). Following treatment, the intervention group exhibited marked improvement, with significantly reduced scores across all symptom categories compared to controls (P < 0.05).
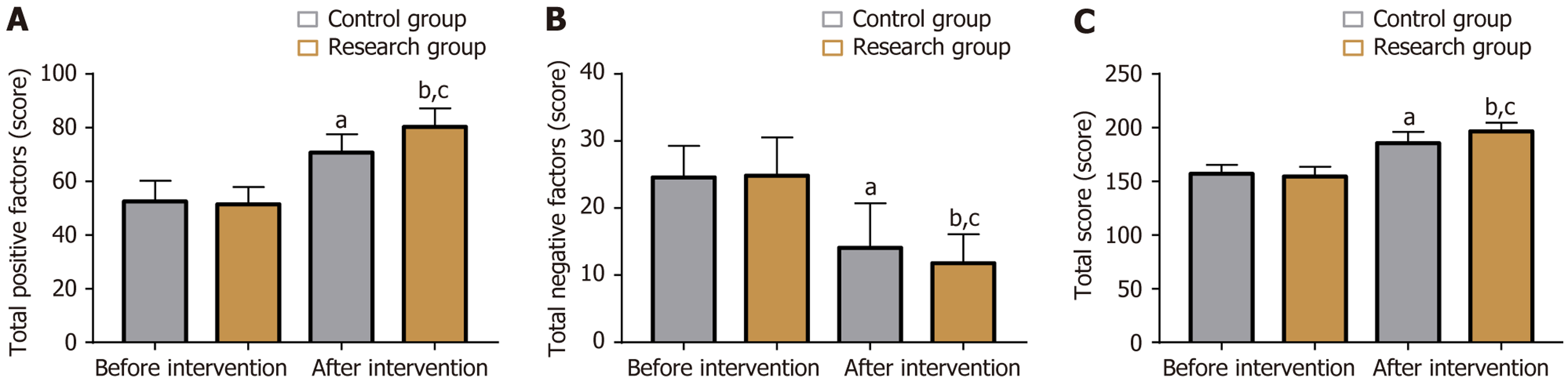
Pretreatment comparisons indicated comparable NOSIE scoring patterns between the two groups (P > 0.05) (Figure 3). Following treatment, both groups showed increases in positive factor and total scores, with the intervention group demonstrating significantly higher post-treatment values than the control group (P < 0.05). Negative factor scores dis
Table 2 compares SCWT outcomes between groups. At baseline, no significant between-group differences were observed in word, color, or total correct responses (P > 0.05). Post-intervention, the intervention group outperformed the control group in word and color tasks (P < 0.05). The control group showed isolated improvement only in word recognition (P < 0.05). Total correct response scores did not change significantly in either group after treatment, and no between-group differences were detected post-intervention (P > 0.05).
| Categories | Control group (n = 59) | Research group (n = 68) | t value | P value |
| SCWT word recognition accuracy | ||||
| Before intervention | 65.53 ± 18.14 | 67.32 ± 19.12 | 0.539 | 0.591 |
| After intervention | 69.93 ± 13.00a | 75.41 ± 15.17b | 2.168 | 0.032 |
| SCWT color recognition accuracy | ||||
| Before intervention | 41.49 ± 14.28 | 39.94 ± 13.53 | 0.628 | 0.532 |
| After intervention | 42.37 ± 13.70 | 49.94 ± 19.25b | 2.517 | 0.013 |
| Total correct responses | ||||
| Before intervention | 21.59 ± 10.91 | 21.50 ± 8.02 | 0.053 | 0.958 |
| After intervention | 23.80 ± 10.31 | 23.56 ± 9.67 | 0.135 | 0.893 |
Table 3 presents the information processing speed comparison. No baseline between-group differences were observed for TMT completion time or BACS-SC scores (P > 0.05). The control group showed no changes in performance after treatment (P > 0.05). Conversely, the intervention group demonstrated significantly shorter TMT completion times and higher BACS-SC accuracy compared with both baseline values and the post-treatment values in the control group (P < 0.05).
| Categories | Control group (n = 59) | Research group (n = 68) | t value | P value |
| TMT completion time (seconds) | ||||
| Before intervention | 82.90 ± 29.66 | 82.41 ± 27.22 | 0.097 | 0.923 |
| After intervention | 83.88 ± 28.14 | 66.15 ± 23.34a | 3.881 | < 0.001 |
| Accuracy count on BACS-SC | ||||
| Before intervention | 27.64 ± 11.35 | 27.76 ± 9.97 | 0.063 | 0.950 |
| After intervention | 30.22 ± 8.93 | 36.85 ± 12.23a | 3.443 | < 0.001 |
Table 4 presents the IPROS scale results for both groups. Baseline scores did not differ significantly between groups across any domain or in total (all P > 0.05). After the intervention, the control group showed significant deterioration in occupational therapy performance, daily living skills, and engagement/motivation, with higher total IPROS scores. In contrast, the intervention group demonstrated significant improvements across all domains, resulting in lower total scores and significantly better post-intervention outcomes compared with the control group (P < 0.05).
| IPROS (points) | Control group (n = 59) | Research group (n = 68) | t value | P value |
| Occupational therapy performance | ||||
| Pre-intervention | 10.24 ± 2.90 | 10.34 ± 3.06 | 0.188 | 0.851 |
| Post-intervention | 8.12 ± 3.18a | 6.54 ± 2.63b | 3.064 | 0.003 |
| Daily living skills | ||||
| Pre-intervention | 9.54 ± 2.73 | 8.66 ± 2.95 | 1.735 | 0.085 |
| Post-intervention | 6.95 ± 2.87a | 5.41 ± 2.39b | 3.299 | 0.001 |
| Interpersonal skills | ||||
| Pre-intervention | 8.29 ± 3.29 | 8.15 ± 2.40 | 0.276 | 0.783 |
| Post-intervention | 7.51 ± 2.65 | 5.54 ± 2.39b | 4.404 | < 0.001 |
| Hygiene practices | ||||
| Pre-intervention | 7.83 ± 2.81 | 7.51 ± 2.92 | 0.627 | 0.532 |
| Post-intervention | 6.41 ± 2.53 | 5.15 ± 2.14b | 3.041 | 0.003 |
| Engagement/motivation levels | ||||
| Pre-intervention | 10.19 ± 3.63 | 10.56 ± 3.72 | 0.565 | 0.573 |
| Post-intervention | 7.44 ± 2.90a | 5.68 ± 2.51b | 3.667 | < 0.001 |
| Total | ||||
| Pre-intervention | 46.10 ± 6.62 | 46.44 ± 5.94 | 0.305 | 0.761 |
| Post-intervention | 36.54 ± 6.33a | 28.12 ± 6.10b | 7.624 | < 0.001 |
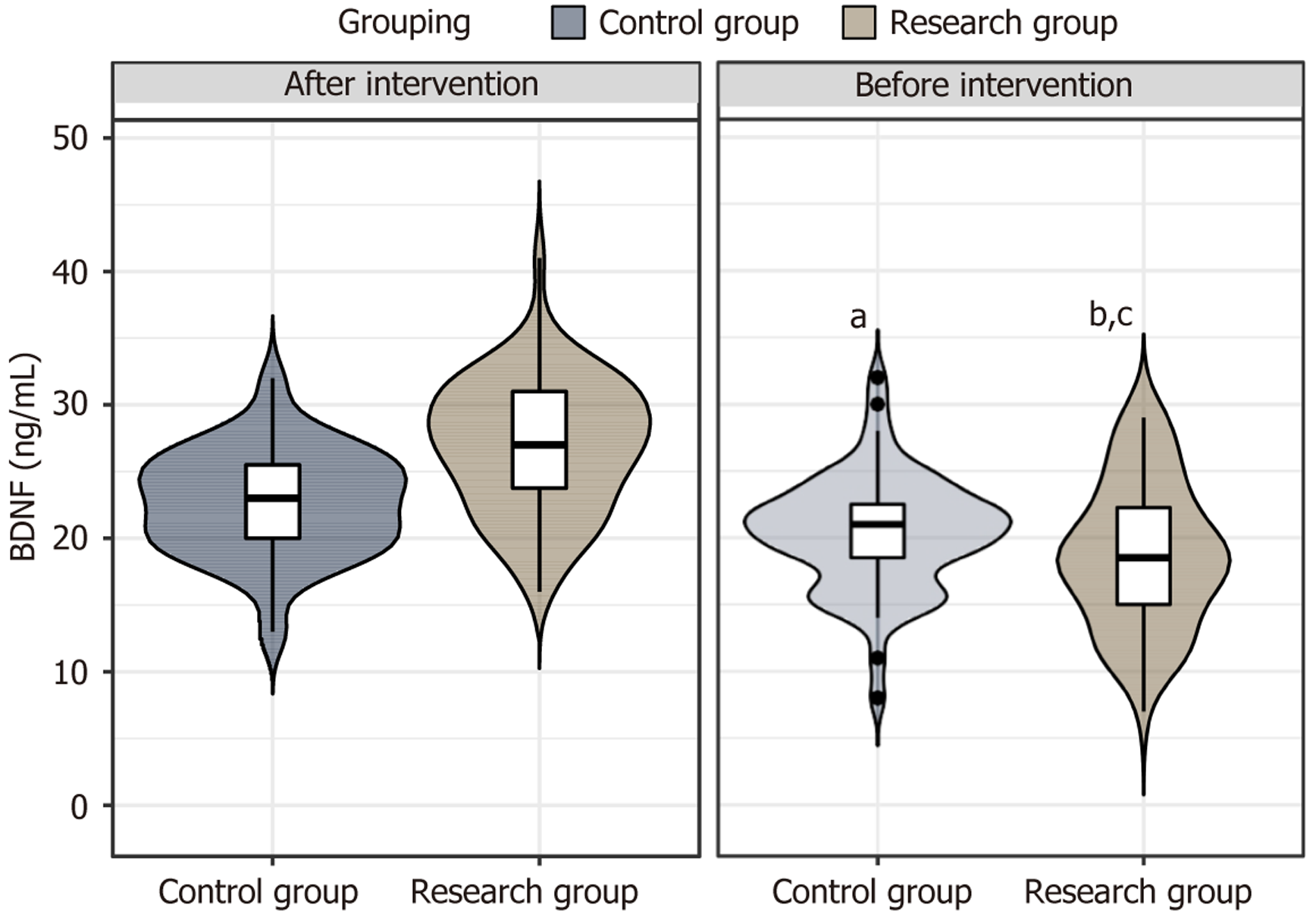
Serum BDNF levels are shown in Figure 4. No baseline differences existed between groups (P > 0.05). After intervention, BDNF levels increased significantly in both groups, with the intervention group exhibiting significantly higher post-treatment levels than the control group (P < 0.05).
Table 5 presents the correlation between the change amplitude of BDNF and the reduction of negative symptoms or the shortening of TMT completion time, further evaluated based on the Pearson correlation coefficient. It was found that the BDNF change amplitude did not correlate significantly with the reduction of negative symptoms or the shortening of TMT completion time (P > 0.05).
| Category | r | P value |
| BDNF change amplitude (points) | 0.033 | 0.709 |
| Reduction of negative symptoms (points) | ||
| BDNF change amplitude (points) | 0.042 | 0.641 |
| Shortening of TMT completion time (second) |
Schizophrenia has a lifetime prevalence of approximately 1% and is characterized by marked clinical heterogeneity[14]. Despite being the cornerstone of treatment, traditional antipsychotics show limited effectiveness against cognitive and negative symptoms[15]. The present study investigated whether adding exercise-based rehabilitation training to standard antipsychotic therapy and health education could enhance clinical outcomes in patients with schizophrenia.
Our findings indicate that exercise-based rehabilitation significantly reduced symptom severity across positive, negative, and general psychiatric domains. These results are consistent with the systematic review and meta-analysis by Kim et al[16], which demonstrated beneficial effects of exercise interventions on both negative and positive symptoms in schizophrenia. These clinical benefits, to a certain extent, are likely linked to exercise-induced hippocampal volumetric increases and serum BDNF upregulation[17]. Additionally, the sense of achievement and self-efficacy improvement brought about by exercise, as well as the naturally increased social interaction in group training, may subtly promote the remission of negative symptoms in patients with schizophrenia.
Furthermore, exercise-based rehabilitation training effectively alleviated symptoms in schizophrenia patients. NOSIE scores showed increased positive factors and total scores with concomitant reductions in negative factors. Cognitive performance improved as evidenced by higher SCWT accuracy in the word and color recognition tasks, along with faster TMT completion and higher BACS-SC correct responses. This might be attributed to the fact that the exercise-based rehabilitation training adopted in this study included route memorization walking and sandbag and table tennis drills, which are high-load cognitive-motor dual tasks, helping to specifically strengthen the executive control function of patients. Therefore, they are more effective for the SCWT sub-tasks that require complex cognitive control, while they perform less well in the relatively simple “total correct score” task. These findings align with the report by Nuechterlein et al[18], demonstrating exercise-related improvements in cognitive and occupational or educational functioning. The observed benefits may be mediated by exercise-induced hippocampal neuroplasticity, including enhanced neurogenesis and synaptogenesis. Expansion of hippocampal neural progenitor cell populations may further support gains in memory retention and learning capacity[19,20]. Moreover, while maintaining the mainstream aerobic and coordination training, the exercise-based rehabilitation training implemented in this study innovatively integrates social interaction, cognitive load, and dynamic task switching, forming a unique social-cognitive-motor integrated rehabilitation model. This rehabilitation model offers a replicable and highly participatory non-pharmacological intervention option for patients with schi
IPROS assessments indicated that exercise-based rehabilitation training improved psychosocial functioning, including occupational therapy performance, daily living skills, interpersonal skills, hygiene practices, and engagement/motivation levels. These findings are consistent with those of Pérez-Romero et al[21], who reported significant improvements in executive functioning following exercise-based interventions in patients with schizophrenia. In addition, Dauwan et al[22] demonstrated that physical activity interventions improve clinical symptoms, psychosocial functioning, and affective states, further supporting the present results.
Furthermore, exercise-based rehabilitation training significantly increased serum BDNF concentrations in patients with schizophrenia, supporting the neuroplasticity hypothesis of exercise-based interventions. BDNF, a protein belonging to the neurotrophin family, is produced in the central nervous system and peripheral tissues. It promotes neuron survival, neurogenesis, and neural plasticity within the central nervous system. Studies suggest its involvement in memory
Several limitations of this research should be acknowledged. First, as a single-center study with a small sample size, future research should expand to multi-center, large-sample analysis to improve the representativeness of the sample and increase the universality of the research results. Second, no causal relationship assessment has been established between the increase in BDNF levels and the functional improvement of patients with schizophrenia. In the future, longitudinal design and mediation analysis should be supplemented to clarify whether BDNF elevation serves as a causal indicator for the functional improvement of patients with schizophrenia.
In this study, exercise-based rehabilitation training was associated with improvements in psychiatric symptoms, cognitive performance, information processing speed, and social functioning. These benefits may be partially mediated by exercise-induced upregulation of serum BDNF.
| 1. | Tandon R, Nasrallah H, Akbarian S, Carpenter WT Jr, DeLisi LE, Gaebel W, Green MF, Gur RE, Heckers S, Kane JM, Malaspina D, Meyer-Lindenberg A, Murray R, Owen M, Smoller JW, Yassin W, Keshavan M. The schizophrenia syndrome, circa 2024: What we know and how that informs its nature. Schizophr Res. 2024;264:1-28. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 119] [Article Influence: 59.5] [Reference Citation Analysis (0)] |
| 2. | McCutcheon RA, Keefe RSE, McGuire PK. Cognitive impairment in schizophrenia: aetiology, pathophysiology, and treatment. Mol Psychiatry. 2023;28:1902-1918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 584] [Cited by in RCA: 462] [Article Influence: 154.0] [Reference Citation Analysis (0)] |
| 3. | Solmi M, Seitidis G, Mavridis D, Correll CU, Dragioti E, Guimond S, Tuominen L, Dargél A, Carvalho AF, Fornaro M, Maes M, Monaco F, Song M, Il Shin J, Cortese S. Incidence, prevalence, and global burden of schizophrenia - data, with critical appraisal, from the Global Burden of Disease (GBD) 2019. Mol Psychiatry. 2023;28:5319-5327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 353] [Article Influence: 117.7] [Reference Citation Analysis (0)] |
| 4. | Howes OD, Bukala BR, Beck K. Schizophrenia: from neurochemistry to circuits, symptoms and treatments. Nat Rev Neurol. 2024;20:22-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 125] [Reference Citation Analysis (0)] |
| 5. | Faden J, Citrome L. Schizophrenia: One Name, Many Different Manifestations. Med Clin North Am. 2023;107:61-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 55] [Article Influence: 18.3] [Reference Citation Analysis (0)] |
| 6. | Maurus I, Roell L, Lembeck M, Papazova I, Greska D, Muenz S, Wagner E, Campana M, Schwaiger R, Schneider-Axmann T, Rosenberger K, Hellmich M, Sykorova E, Thieme CE, Vogel BO, Harder C, Mohnke S, Huppertz C, Roeh A, Keller-Varady K, Malchow B, Walter H, Wolfarth B, Wölwer W, Henkel K, Hirjak D, Schmitt A, Hasan A, Meyer-Lindenberg A, Falkai P. Exercise as an add-on treatment in individuals with schizophrenia: Results from a large multicenter randomized controlled trial. Psychiatry Res. 2023;328:115480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 29] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 7. | Blumenthal JA, Rozanski A. Exercise as a therapeutic modality for the prevention and treatment of depression. Prog Cardiovasc Dis. 2023;77:50-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 45] [Reference Citation Analysis (0)] |
| 8. | Misiak B, Samochowiec J, Kowalski K, Gaebel W, Bassetti CLA, Chan A, Gorwood P, Papiol S, Dom G, Volpe U, Szulc A, Kurimay T, Kärkkäinen H, Decraene A, Wisse J, Fiorillo A, Falkai P. The future of diagnosis in clinical neurosciences: Comparing multiple sclerosis and schizophrenia. Eur Psychiatry. 2023;66:e58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 21] [Reference Citation Analysis (0)] |
| 9. | Šagud M, Madžarac Z, Nedic Erjavec G, Šimunović Filipčić I, Mikulić FL, Rogić D, Bradaš Z, Bajs Janović M, Pivac N. The Associations of Neutrophil-Lymphocyte, Platelet-Lymphocyte, Monocyte-Lymphocyte Ratios and Immune-Inflammation Index with Negative Symptoms in Patients with Schizophrenia. Biomolecules. 2023;13:297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 10. | Qiao C, Zou X, Deng W, Xie M, Zou M, Wang Q. Machine Learning-Based Classifier for Identifying Inpatients With Schizophrenia at High Risk of Suicide. J Adv Nurs. 2025;81:7645-7658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Li C, Delgado-Gómez D, Sujar A, Wang P, Martin-Moratinos M, Bella-Fernández M, Masó-Besga AE, Peñuelas-Calvo I, Ardoy-Cuadros J, Hernández-Liebo P, Blasco-Fontecilla H. Assessment of ADHD Subtypes Using Motion Tracking Recognition Based on Stroop Color-Word Tests. Sensors (Basel). 2024;24:323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 12. | Huang Y, Wang W, Hei G, Shao T, Li L, Yang Y, Wang X, Long Y, Xiao J, Peng X, Song C, Cai J, Song X, Xu X, Gao S, Huang J, Kang D, Wang Y, Zhao J, Pan Y, Wu R. Subgroups of cognitive impairments in schizophrenia characterized by executive function and their morphological features: a latent profile analysis study. BMC Med. 2025;23:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 13. | Lu S, Zhao Y, Liu J, Xu F, Wang Z. Effectiveness of Horticultural Therapy in People with Schizophrenia: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2021;18:964. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Owen MJ, Legge SE, Rees E, Walters JTR, O'Donovan MC. Genomic findings in schizophrenia and their implications. Mol Psychiatry. 2023;28:3638-3647. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 105] [Article Influence: 35.0] [Reference Citation Analysis (0)] |
| 15. | Paul SM, Yohn SE, Brannan SK, Neugebauer NM, Breier A. Muscarinic Receptor Activators as Novel Treatments for Schizophrenia. Biol Psychiatry. 2024;96:627-637. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 42] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 16. | Kim M, Lee Y, Kang H. Effects of Exercise on Positive Symptoms, Negative Symptoms, and Depression in Patients with Schizophrenia: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health. 2023;20:3719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 17. | Girdler SJ, Confino JE, Woesner ME. Exercise as a Treatment for Schizophrenia: A Review. Psychopharmacol Bull. 2019;49:56-69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 18. | Nuechterlein KH, McEwen SC, Ventura J, Subotnik KL, Turner LR, Boucher M, Casaus LR, Distler MG, Hayata JN. Aerobic exercise enhances cognitive training effects in first-episode schizophrenia: randomized clinical trial demonstrates cognitive and functional gains. Psychol Med. 2023;53:4751-4761. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 19. | Fabel K, Wolf SA, Ehninger D, Babu H, Leal-Galicia P, Kempermann G. Additive effects of physical exercise and environmental enrichment on adult hippocampal neurogenesis in mice. Front Neurosci. 2009;3:50. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 193] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 20. | Kempermann G, Fabel K, Ehninger D, Babu H, Leal-Galicia P, Garthe A, Wolf SA. Why and how physical activity promotes experience-induced brain plasticity. Front Neurosci. 2010;4:189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 202] [Cited by in RCA: 214] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 21. | Pérez-Romero N, Campos-Jara C, Pesce C, Araya Sierralta S, Cerda-Vega E, Ramirez-Campillo R, Campos-Jara R, Martínez-Salazar C, Arellano-Roco C, Contreras-Osorio F. Effects of physical exercise on executive functions of individuals with schizophrenia spectrum disorders: Protocol for a systematic review and meta-analysis. PLoS One. 2024;19:e0296273. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 22. | Dauwan M, Begemann MJ, Heringa SM, Sommer IE. Exercise Improves Clinical Symptoms, Quality of Life, Global Functioning, and Depression in Schizophrenia: A Systematic Review and Meta-analysis. Schizophr Bull. 2016;42:588-599. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 333] [Cited by in RCA: 274] [Article Influence: 27.4] [Reference Citation Analysis (0)] |
| 23. | Mosiołek A, Mosiołek J. The Effects of Treatment in Psychotic Disorders-Changes in BDNF Levels and Clinical Outcomes: Systematic Review. Int J Environ Res Public Health. 2023;20:2111. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 24. | Gökçe E, Güneş E, Nalçaci E. Effect of Exercise on Major Depressive Disorder and Schizophrenia: A BDNF Focused Approach. Noro Psikiyatr Ars. 2019;56:302-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 25. | Shen H, Lian A, Wu Y, Zhou J, Liu Y, Zhu L, Zhang Y, Yi Z, Liu X, Fan Q. Shen-based Qigong Exercise improves cognitive impairment in stable schizophrenia patients in rehabilitation wards: a randomized controlled study. BMC Psychiatry. 2024;24:796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 2.0] [Reference Citation Analysis (1)] |
| 26. | Dunleavy C, Elsworthy RJ, Wood SJ, Allott K, Spencer F, Upthegrove R, Aldred S. Exercise4Psychosis: A randomised control trial assessing the effect of moderate-to-vigorous exercise on inflammatory biomarkers and negative symptom profiles in men with first-episode psychosis. Brain Behav Immun. 2024;120:379-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |