Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.113723
Revised: December 16, 2025
Accepted: February 9, 2026
Published online: May 19, 2026
Processing time: 240 Days and 0.8 Hours
Clozapine remains the gold therapeutic standard for treatment-resistant schizophrenia, and is associated with a range of adverse effects. Although autonomic dysregulation (e.g., psychosis and cholinergic rebound) during clozapine with
A 60-year-old man with treatment-resistant schizophrenia and metabolic syn
Sudden clozapine withdrawal may precipitate severe hypertension in patients with metabolic syndrome, even in those receiving antihypertensive therapy. It is essential for clinicians to closely monitor BP during clozapine discontinuation, consider a gradual tapering protocol, and recognize the reintroduction of clozapine as a viable the
Core Tip: This case is the first to describe severe, refractory hypertension as a consequence of sudden clozapine withdrawal in a patient with treatment-resistant schizophrenia and metabolic syndrome. Blood pressure remained uncontrolled despite intensive antihypertensive therapy, but normalized rapidly after clozapine reintroduction. These findings demonstrate that hypertension may be an unrecognized manifestation of clozapine withdrawal and highlight the critical importance of vigilant blood pressure monitoring, gradual tapering, and, when necessary, reintroduction of clozapine. This report contributes to the understanding of clozapine withdrawal syndromes and provides valuable clinical guidance for the safe management of aff
- Citation: Liu JZ, Ma T, Liang ZY, Ning XB, Li WJ, Wang JP, Jin YL, Zhou N, Li JF. Hypertension induced by sudden clozapine withdrawal in a schizophrenia patient with metabolic syndrome: A case report. World J Psychiatry 2026; 16(5): 113723
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/113723.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.113723
Clozapine, a dibenzodiazepine derivative and multi-acting receptor-targeted antipsychotic, remains the gold standard for treatment-resistant schizophrenia (TRS) due to its superior efficacy in reducing psychotic symptoms, hospitalization rates, and suicide risk compared with other antipsychotics[1-3]. Despite its clinical benefits, the use of clozapine is often constrained by complex and potentially severe adverse effects, including hematologic (e.g., agranulocytosis), metabolic (e.g., weight gain, insulin resistance), and cardiovascular complications[4-6]. Consequently, clinicians may be required to discontinue clozapine due to the onset of emergent, potentially life-threatening adverse drug reactions.
Abrupt clozapine discontinuation may precipitate withdrawal syndromes characterized by autonomic dysregulation, including withdrawal-associated psychosis, cholinergic rebound, extrapyramidal symptoms, catatonia, and serotonergic discontinuation syndrome[7,8]. Moreover, clozapine discontinuation may induce cardiovascular effects, including tachycardia[9], cardiomyopathy, and myocarditis[10,11]. However, hypertension, as a manifestation of clozapine withdrawal, has not been previously documented. This study described a novel case of severe, refractory hypertension occurring in close association with the abrupt discontinuation of clozapine in a patient with schizophrenia and pre-existing metabolic syndrome (MetS). This case report highlighted a rare but clinically significant adverse effect of clo
A 60-year-old, unmarried, homeless man with a 40-year history of TRS (International Classification of Diseases-10 F20.0) was hospitalized for long-term rehabilitation since 2010 due to marked socio-occupational impairment and the absence of guardianship. His disease was characterized by persistent hallucinatory behavior, persecutory delusions, prominent negative symptoms, severe self-neglect, and significant socio-occupational impairment. Despite several trials of both typical and atypical antipsychotics, the symptoms remained refractory to treatment. Since 2010, the patient had achieved clinical stabilization on clozapine at a dose of 200-225 mg/day.
During hospitalization, the patient presented with severe, intractable constipation (no bowel movement for 72 hours) that was unresponsive to his usual regimen of Maren soft capsules and other supportive treatments. The discomfort was remarkable and significantly impacted his well-being. After excluding other potential causes and considering the known pharmacological properties of clozapine, along with a Naranjo Adverse Drug Reaction Probability Scale score of 8 (Table 1), clozapine was identified as the most likely etiological factor. Consequently, clozapine was abruptly disconti
| Yes | No | Do not know | Score | |
| Are there previous conclusive reports on this reaction? | +1 | 0 | 0 | 1 |
| Did the adverse event appear after the suspected drug was administered? | +2 | -1 | 0 | 2 |
| Did the adverse reaction improve when the drug was discontinued or a specific antagonist was administered? | +1 | 0 | 0 | 1 |
| Did the adverse reaction reappear when the drug was readministered? | +2 | -1 | 0 | 2 |
| Are there alternative causes (other than the drug) that could on their own have caused the reaction? | -1 | +2 | 0 | 0 |
| Did the reaction reappear when a placebo was given? | -1 | +1 | 0 | 0 |
| Was the drug detected in the blood (or other fluids) in concentrations known to be toxic? | +1 | 0 | 0 | 0 |
| Was the reaction more severe when the dose was increased, or less severe when the dose was decreased? | +1 | 0 | 0 | 1 |
| Did the patient have a similar reaction to the same or similar drugs in any previous exposure? | +1 | 0 | 0 | 0 |
| Was the adverse event confirmed by any objective evidence? | +1 | 0 | 0 | 1 |
| Total score | 8 | |||
His medical history included type 2 diabetes mellitus, diagnosed in 2006, and managed with metformin (500 mg, three times a day) and acarbose (50 mg, three times a day). Additionally, he was diagnosed with hypertension in 2011 and treated with amlodipine (5 mg/day) and irbesartan (150 mg/day).
The patient had no significant personal and family history.
Before clozapine withdrawal, physical examination was unremarkable aside from central obesity (waist circumference, 106 cm) and a body mass index of 22.34 kg/m2. Blood pressure (BP) was 124/76 mmHg.
Results of routine laboratory tests (hemogram, liver and renal function tests, serum electrolytes, fasting glucose) and electrocardiogram were within normal limits. The patient met the American Heart Association/National Heart, Lung, and Blood Institute criteria for MetS[12], particularly fulfilling the following criteria: Central obesity (waist > 102 cm), hyperglycemia (known type 2 diabetes mellitus on treatment), and hypertension (known hypertension on treatment) (Table 2).
| Criterion | Definition | Patient portrait | Yes/no |
| Abdominal obesity | Waist circumference ≥ 102 cm for men | 106 cm | Yes |
| Hyperglycemia | Fasting plasma glucose ≥ 100 mg/dL or ongoing treatment | Ongoing treatment | Yes |
| Low HDL-cholesterol | HDL < 40 mg/dL for men or ongoing treatment | 39 mg/dL | Yes |
| Hypertriglyceridemia | Triglycerides ≥ 150 mg/dL or ongoing treatment | 145.14 mg/dL | No |
| Hypertension | Blood pressure ≥ 130/85 mmHg or ongoing treatment | Ongoing treatment | Yes |
No imaging examinations were conducted.
Severe, refractory hypertension occurring in close association with the abrupt discontinuation of clozapine.
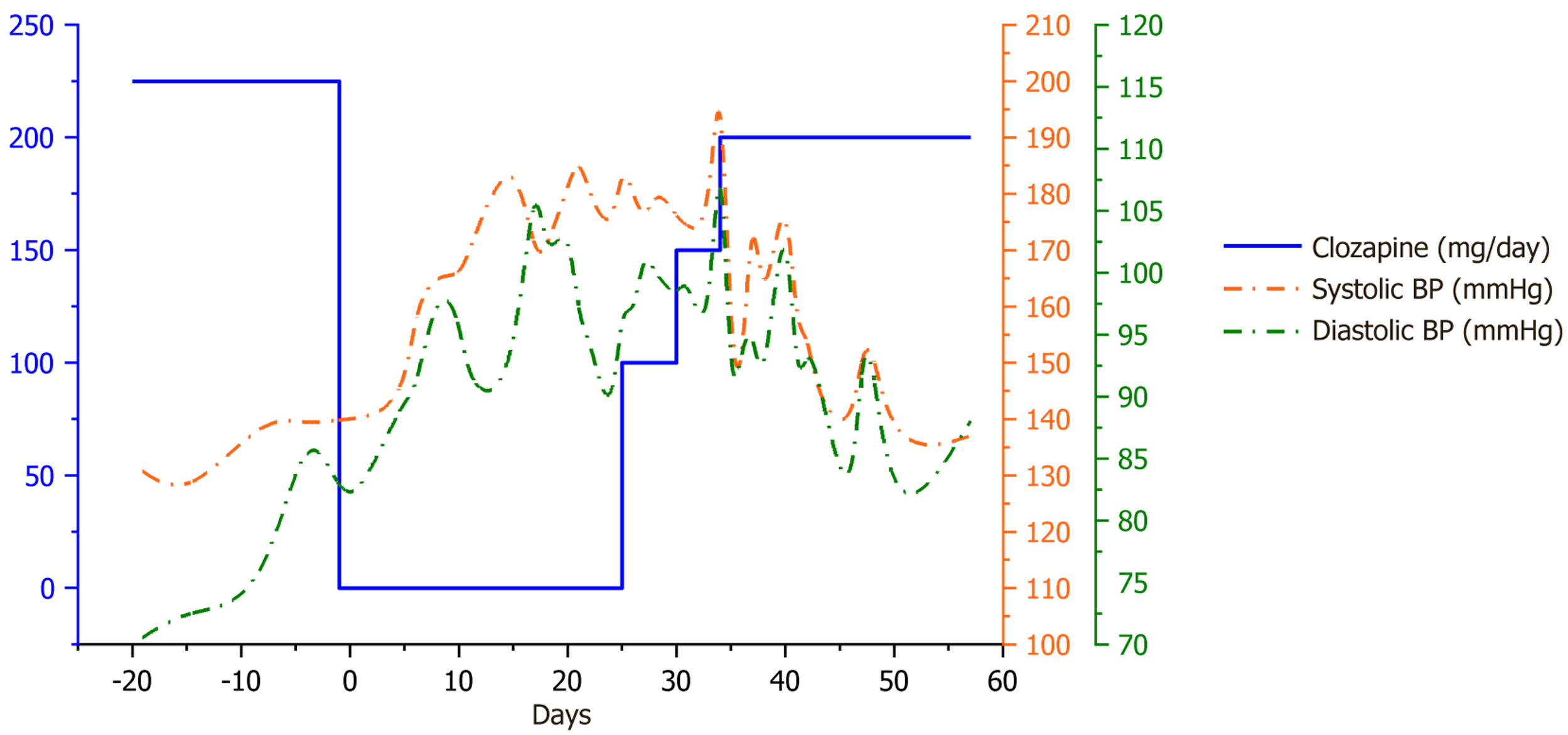
On day 1 of clozapine discontinuation, aripiprazole (5 mg twice daily) was initiated as an alternative antipsychotic. Baseline BP was 140/82 mmHg. Following clozapine withdrawal, frequent BP monitoring (every 8-12 hours) revealed a persistent and significant elevation in both systolic BP (SBP) and diastolic BP (DBP), which sustained for nearly one month (Figure 1 and Table 3). On day 6, amlodipine was replaced with nifedipine (30 mg/day), which was later increased to 60 mg/day on day 21, while BP remained uncontrolled (days 6-25: SBP 160-190 mmHg, DBP 87-110 mmHg; Figure 1 and Table 3). Irbesartan (150 mg/day) treatment was continued. Repeat investigations, including complete hemogram (sodium, potassium), electrolytes, aldosterone (46.9 pg/mL, reference: 28-376 pg/mL), adrenocorticotropic hormone (9 AM) (47.1 pg/mL, reference: 6.0-48 pg/mL), and cortisol (18.80 μg/dL, reference: 7.26-32.28 μg/dL), were within normal ranges.
| Days since discontinuation | Antihypertensive dosage | Clozapine restart | Blood pressure readings | Clinical course |
| 1 | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | No | 140/82 mmHg | No |
| 2 | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | No | NA | No |
| 3 | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | No | NA | No |
| 4 | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | No | NA | No |
| 5 | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | No | 143/90 mmHg | No |
| 6 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + felodipine sustained-release tablets 5 mg | No | 160/90 mmHg | Headache tachycardia |
| 7 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 8 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | 166/99 mmHg | Tachycardia |
| 9 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 10 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 165/97 mmHg | No |
| 11 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 170/90 mmHg | Tachycardia |
| 12 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 13 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 14 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 15 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 190/90 mmHg | Headache tachycardia |
| 16 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 17 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | 168/110 mmHg | No |
| 18 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 170/100 mmHg | No |
| 19 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg | No | NA | No |
| 20 | Nifedipine controlled-release tablets 30 mg + irbesartan tablets 150 mg + nifedipine controlled-release tablets 30 mg | No | 181/105 mmHg | Tachycardia |
| 21 | Nifedipine controlled-release tablets 60 mg + irbesartan tablets 150 mg | No | 187/96 mmHg | No |
| 22 | Nifedipine controlled-release tablets 60 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 180/95 mmHg | No |
| 23 | Nifedipine controlled-release tablets 60 mg + irbesartan tablets 150 mg | No | NA | No |
| 24 | Nifedipine controlled-release tablets 60 mg + irbesartan tablets 150 mg + captopril tablets 25 mg | No | 172/87 mmHg | No |
| 25 | Nifedipine controlled-release tablets 60 mg + irbesartan tablets 150 mg + nifedipine sustained-release tablets 20 mg | No | 186/98 mmHg | No |
| 26 | Nifedipine controlled-release tablets 60 mg + captopril tablets 75 mg + metoprolol succinate sustained-release tablets 23.75 mg + nifedipine sustained-release tablets 20 mg | 100 mg | 180/96 mmHg | Tachycardia |
| 27 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg + nifedipine sustained-release tablets | 100 mg | 175/102 mmHg | Tachycardia |
| 28 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg | 100 mg | 180/100 mmHg | No |
| 29 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg + nifedipine sustained-release tablets | 100 mg | 179/99 mmHg | No |
| 30 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg | 100 mg | 176/98 mmHg | No |
| 31 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg | 150 mg | 174/100 mmHg | No |
| 32 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg | 150 mg | 174/96 mmHg | No |
| 33 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg | 150 mg | 173/93 mmHg | No |
| 34 | Nifedipine sustained-release tablets 40 mg + captopril tablets 75 mg + metoprolol tartrate tablets 100 mg + nitroglycerin injection 10 mg | 150 mg | 210/115 mmHg | Headache tachycardia |
| 35 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + metoprolol tartrate tablets 100 mg | 200 mg | 150/88 mmHg | No |
| 36 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + metoprolol tartrate tablets 100 mg | 200 mg | 145/94 mmHg | No |
| 37 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + metoprolol tartrate tablets 100 mg | 200 mg | 183/96 mmHg | Tachycardia |
| 38 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 155/89 mmHg | No |
| 39 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + metoprolol tartrate tablets 100 mg | 200 mg | NA | No |
| 40 | Nifedipine sustained-release tablets 40 mg + captopril tablets 150 mg + metoprolol tartrate tablets 100 mg + urapidil hydrochloride injection 25 mg | 200 mg | 186/108 mmHg | Headache tachycardia |
| 41 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 155/90 mmHg | No |
| 42 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 156/94 mmHg | No |
| 43 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 143/92 mmHg | No |
| 44 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | NA | No |
| 45 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | NA | No |
| 46 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 137/79 mmHg | No |
| 47 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 152/94 mmHg | No |
| 48 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 154/94 mmHg | No |
| 49 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 137/88 mmHg | No |
| 50 | Nifedipine sustained-release tablets 80 mg + captopril tablets 150 mg + irbesartan tablets 150 mg | 200 mg | 133/78 mmHg | No |
| 3-month follow-up | Nifedipine sustained-release tablets 80 mg + irbesartan tablets 150 mg | 250 mg | 115/74 mmHg | No |
| 6-month follow-up | Amlodipine besylate tablets 5 mg + irbesartan tablets 150 mg | 225 mg | 122/78 mmHg | No |
Given the persistent hypertension and lack of other identifiable causes, clozapine withdrawal was considered as a potential etiological factor. On day 26, clozapine was cautiously reinitiated at 100 mg/day and aripiprazole was discontinued. On day 27, the BP remained elevated at 175/102 mmHg, prompting the addition of captopril (25 mg three times a day). Despite this, the BP remained high (174/100 mmHg) on day 31, necessitating a dose increase of clozapine to 150 mg/day. Further titration of clozapine to 200 mg on day 35 and 200 mg by discharge, alongside intensification of anti
At the 3-month follow-up, BP was 115/74 mmHg. At the 4-month follow-up, nifedipine was replaced with amlodipine (5 mg/day), restoring the pre-withdrawal regimen [clozapine (250 mg/day), amlodipine (5 mg/day), and irbesartan (150 mg/day)]. The patient’s BP was 122/78 mmHg at the 6-month follow-up (Table 3).
To our knowledge, this is the first reported case of severe, refractory hypertension following the abrupt discontinuation of clozapine in a patient with TRS and MetS. Schizophrenia is a chronic psychiatric disorder characterized by positive symptoms, negative symptoms, and cognitive impairment. The prevalence of MetS in patients with schizophrenia is estimated to be as high as 41%, which is 2-3 times greater than in the general population[13]. As a major driver of cardiovascular morbidity, MetS contributes to a shortened life expectancy by 15-20 years in this population[14,15]. Clozapine remains the most effective therapy for TRS, while it is widely recognized for its metabolic adverse effects and cardiovascular risks. Orthostatic hypotension and tachycardia are well-recognized adverse effects; however, clozapine-induced hypertension is increasingly reported, but remains under-recognized[16-18]. In patients with MetS, the risk of BP instability may be particularly remarkable during clozapine initiation, dose titration, or discontinuation.
Clozapine has a complex pharmacologic profile, interacting with dopaminergic (dopamine receptor subtypes 1, 2, 4), serotonergic (5-hydroxytryptamine, 5-hydroxytryptamine receptor subtypes 1C, 2A/C, 3), muscarinic, histaminergic, and adrenergic (α1, α2) receptors[19]. Several mechanisms may contribute to hypertension during clozapine exposure or withdrawal. α2-adrenergic receptor antagonism can increase noradrenaline release, while high affinity for dopamine receptor subtypes 4 receptors, which are implicated in hypertensive phenotypes in animal models, may further exa
The present case report provides clinical insights into this pharmacodynamic complexity. BP gradually improved when clozapine was re-started at doses exceeding 200 mg/day, demonstrating a dose-dependent antihypertensive effect mediated by the restoration of α1-adrenoceptor blockade. MetS likely heightened susceptibility to hypertensive crises by promoting sympathetic overactivity and endothelial dysfunction through insulin resistance[23]. These metabolic disturbances may also explain the patient’s initial refractoriness to standard antihypertensives. Reinstating clozapine restored vascular regulatory balance, highlighting MetS as a critical vulnerability factor and emphasizing the importance of individualized dosing strategies. Gradual tapering of clozapine, such as reducing the dose by increments of 25-50 mg every 2-3 days, is essential to mitigate the risk of withdrawal-induced hypertension. Similar precautions may be required for antipsychotics with comparable receptor-binding profiles, such as olanzapine. Future research should elucidate whether α2-adrenergic receptor-mediated mechanisms contribute to withdrawal-associated hypertension in this pharmacological class.
This case report has several limitations. As a single case study, its generalizability is restricted. Additionally, the absence of clozapine plasma level measurements limits mechanistic interpretation, particularly regarding pharmacokinetic-phar
This case report confirms that abrupt clozapine discontinuation may precipitate severe hypertension, even in patients receiving standard antihypertensive therapy. Clinicians must consider clozapine withdrawal as a potential cause of treatment-resistant hypertension, particularly in patients with pre-existing metabolic or cardiovascular comorbidities. This case report highlights the need for close monitoring of cardiovascular parameters during clozapine discontinuation. Weekly BP monitoring for 4-6 weeks following clozapine discontinuation is recommended for patients with MetS to mitigate risks and promote timely intervention.
| 1. | Murray RM, Egerton A, Gao Y, Grace AA, Howes O, Jauhar S, Leucht S, Chen EYH, MacCabe JH, McCutcheon RA, Natesan S, Taylor D. Why Is Clozapine Uniquely Effective in Treatment-Resistant Schizophrenia? A Review and Proposal. Biol Psychiatry. 2025;S0006-3223(25)01270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 2. | Zhou H, Luo H, Tang JY, Honer WG, Bastiampillai T, Zhou J, Taipale H, Chang WC, Lui SSY, Lee EHM, Chan SKW. Population-based study of long-term mortality risk associated with clozapine use among patients with schizophrenia. Br J Psychiatry. 2025;1-9. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 3. | Land R, Siskind D, McArdle P, Kisely S, Winckel K, Hollingworth SA. The impact of clozapine on hospital use: a systematic review and meta-analysis. Acta Psychiatr Scand. 2017;135:296-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 113] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 4. | De Las Cuevas C. Clozapine and Regulatory Inertia: Revisiting Evidence, Risks, and Reform. Healthcare (Basel). 2025;13:1668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (1)] |
| 5. | Jiang D, Wang R, Ma D, Tang C, Liu Y, Jian N, Luo J, Gong Q, Chen Y. Prevalence and predictors of metabolic abnormalities and metabolic syndrome among patients with schizophrenia in China. Schizophr Res. 2025;282:28-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 6. | Curto M, Girardi N, Lionetto L, Ciavarella GM, Ferracuti S, Baldessarini RJ. Systematic Review of Clozapine Cardiotoxicity. Curr Psychiatry Rep. 2016;18:68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 68] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 7. | Blackman G, Oloyede E. Clozapine discontinuation withdrawal symptoms in schizophrenia. Ther Adv Psychopharmacol. 2021;11:20451253211032053. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 8. | Chiappini S, Schifano F, Corkery JM, Guirguis A. Focus on Clozapine Withdrawal- and Misuse-Related Cases as Reported to the European Medicines Agency (EMA) Pharmacovigilance Database. Brain Sci. 2020;10:105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 9. | Every-Palmer S, Northwood K, Tsakas J, Burrage MK, Siskind D. Clozapine-Related Tachycardia: An Analysis of Incidence. CNS Drugs. 2025;39:597-607. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 10. | Segev A, Iqbal E, McDonagh TA, Casetta C, Oloyede E, Piper S, Plymen CM, MacCabe JH. Clozapine-induced myocarditis: electronic health register analysis of incidence, timing, clinical markers and diagnostic accuracy. Br J Psychiatry. 2021;219:644-651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 11. | Gurrera RJ, Gearin PF, Love J, Li KJ, Xu A, Donaghey FH, Gerace MR. Recognition and management of clozapine adverse effects: A systematic review and qualitative synthesis. Acta Psychiatr Scand. 2022;145:423-441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 35] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 12. | Grundy SM, Cleeman JI, Daniels SR, Donato KA, Eckel RH, Franklin BA, Gordon DJ, Krauss RM, Savage PJ, Smith SC Jr, Spertus JA, Costa F; American Heart Association; National Heart, Lung, and Blood Institute. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735-2752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9267] [Cited by in RCA: 8319] [Article Influence: 396.1] [Reference Citation Analysis (4)] |
| 13. | Salari N, Maghami N, Ammari T, Mosafer H, Abdullahi R, Rasoulpoor S, Babajani F, Mahmodzadeh B, Mohammadi M. Global Prevalence of Metabolic Syndrome in Schizophrenia Patients: A Systematic Review and Meta-Analysis. J Prev (2022). 2024;45:973-986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 14. | Kazlauskienė L, Butnorienė J, Norkus A. Metabolic syndrome related to cardiovascular events in a 10-year prospective study. Diabetol Metab Syndr. 2015;7:102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 15. | Correll CU, Solmi M, Croatto G, Schneider LK, Rohani-Montez SC, Fairley L, Smith N, Bitter I, Gorwood P, Taipale H, Tiihonen J. Mortality in people with schizophrenia: a systematic review and meta-analysis of relative risk and aggravating or attenuating factors. World Psychiatry. 2022;21:248-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 494] [Article Influence: 123.5] [Reference Citation Analysis (0)] |
| 16. | Deepak MB, Deeksha K, Pallavi R, Hemant C, Nidhisha B, Raman D. Clozapine Induced Hypertension and its Association with Autonomic Dysfunction. Psychopharmacol Bull. 2021;51:122-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 17. | Norman SM, Sullivan KM, Liu F, DiPaula BA, Jose PA, Kitchen CA, Feldman SM, Kelly DL. Blood Pressure and Heart Rate Changes During Clozapine Treatment. Psychiatr Q. 2017;88:545-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Tanzer TD, Brouard T, Pra SD, Warren N, Barras M, Kisely S, Brooks E, Siskind D. Treatment strategies for clozapine-induced hypotension: a systematic review. Ther Adv Psychopharmacol. 2022;12:20451253221092931. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 19. | Khokhar JY, Henricks AM, Sullivan EDK, Green AI. Unique Effects of Clozapine: A Pharmacological Perspective. Adv Pharmacol. 2018;82:137-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 143] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 20. | Fehsel K, Schwanke K, Kappel BA, Fahimi E, Meisenzahl-Lechner E, Esser C, Hemmrich K, Haarmann-Stemmann T, Kojda G, Lange-Asschenfeldt C. Activation of the aryl hydrocarbon receptor by clozapine induces preadipocyte differentiation and contributes to endothelial dysfunction. J Psychopharmacol. 2022;36:191-201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 16] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 21. | Alberich S, Fernández-Sevillano J, González-Ortega I, Usall J, Sáenz M, González-Fraile E, González-Pinto A. A systematic review of sex-based differences in effectiveness and adverse effects of clozapine. Psychiatry Res. 2019;280:112506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 35] [Article Influence: 5.0] [Reference Citation Analysis (1)] |
| 22. | Starke K, Docherty JR. Alpha 1- and alpha 2-adrenoceptors: pharmacology and clinical implications. J Cardiovasc Pharmacol. 1981;3 Suppl 1:S14-S23. [PubMed] |
| 23. | Scherer T, Sakamoto K, Buettner C. Brain insulin signalling in metabolic homeostasis and disease. Nat Rev Endocrinol. 2021;17:468-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |