Published online May 19, 2026. doi: 10.5498/wjp.v16.i5.113866
Revised: December 11, 2025
Accepted: February 12, 2026
Published online: May 19, 2026
Processing time: 225 Days and 3.3 Hours
Impaired insight is a core feature of schizophrenia and an established predictor of psychosis in individuals at clinical high risk (CHR). However, the neurocognitive mechanisms underlying impaired insight in CHR populations remain unclear.
To investigate the role of neurocognitive deficits in impaired insight and examine their combined influence on psychosis risk over a six-year follow-up.
A total of 312 CHR individuals were assessed for insight using the G12 item of the Positive and Negative Syndrome Scale. Participants were categorized into the low-impairment insight group (n = 151, G12 score < 3) and the high-impairment insight group (n = 161, G12 score ≥ 3). Neurocognition was evaluated using the MATRICS Consensus Cognitive Battery.
Performance on the Brief Visuospatial Memory Test-Revised (BVMT-R) was the only cognitive domain differentiating the insight groups after controlling for positive symptoms. The effect of BVMT-R on insight was most pronounced at moderate symptom levels. Moreover, risk-curve analyses indicated that higher BVMT-R scores were associated with a reduced conversion risk linked to impaired insight.
Incorporating visuospatial memory assessment may improve the identification of those at greatest risk and inform targeted interventions aimed at enhancing insight and reducing conversion to psychosis.
Core Tip: Impaired insight predicts psychosis in individuals at clinical high risk (CHR), yet its neurocognitive underpinnings remain unclear. In a six-year follow-up of 312 CHR participants, visuospatial memory assessed via the Brief Visuospatial Memory Test-Revised (BVMT-R) emerged as the key cognitive domain differentiating low and high impaired insight groups, independent of positive symptoms. Notably, higher BVMT-R scores reduced the risk of conversion to psychosis, moderating the impact of impaired insight. These findings highlight visuospatial memory as a potential marker for identifying CHR individuals at greatest risk and suggest targeted cognitive interventions to enhance insight and mitigate psychosis onset.
- Citation: Xu LH, Cui HR, Wei YY, Tang XC, Qian ZY, Zhang D, Zheng WS, Zhu TY, Hong XF, Zhu JJ, Hu YG, Liu X, Jiao X, Qing Y, Chen XC, Tang YY, Zhang TH, Wang JJ. Visuospatial memory modulates insight and its predictive value for 6-year psychosis risk in clinical high-risk individuals. World J Psychiatry 2026; 16(5): 113866
- URL: https://www.wjgnet.com/2220-3206/full/v16/i5/113866.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i5.113866
Impaired insight, defined as diminished awareness and understanding of one’s own mental health, is a well-documented phenomenon in schizophrenia[1,2] and is increasingly recognized in individuals at clinical high risk (CHR) for psychosis[3]. As a prognostic indicator, impaired insight in individuals with CHR has been associated with an increased risk of transitioning to psychosis[4]. Given that the CHR period presents a critical window for early intervention and etiological clarification[5], understanding the underlying mechanisms of impaired insight is essential to enhance preventive stra
Neurocognitive dysfunction is widely regarded as a core trait-like feature of schizophrenia[6,7], often emerging before the onset of frank psychosis[8-10] and reliably predicting transition outcomes in individuals with CHR[11,12]. The MATRICS Consensus Cognitive Battery (MCCB), developed under the NIMH MATRICS initiative[7], provides a stand
Emerging evidence suggests a potential link between impaired insight and deficits in various neurocognitive domains[14,15]. Specifically, impairments in attention, working memory, and executive function have been proposed as underlying contributors to impaired insight[14], potentially limiting an individual’s ability to recall illness-related experiences and comprehend causal relationships[16]. However, the findings remain inconsistent; some studies report weak or inconclusive associations[17,18], leading to the proposition that intact neurocognition may be necessary but not sufficient for the development of insight[19].
Most prior studies have examined these relationships in individuals with first-episode or chronic schizophrenia. Whether similar neurocognitive patterns are evident during CHR remains unclear. Given that both impaired insight and neurocognitive deficits predate the onset of full-blown psychosis[9,10,12], CHR presents an optimal window for examining whether specific neurocognitive deficits contribute to the exacerbation of impaired insight. Furthermore, these factors may influence clinical outcomes with potential implications for individualized risk prediction.
Despite receiving limited attention, visuospatial memory has shown robust associations with conversion risk in individuals with CHR[12], suggesting that it may influence both insight and prognostic significance during the prodromal phase. For example, individuals with CHR with impaired insight but preserved visuospatial memory may be less likely to convert than those with concurrent impairments, indicating a potential moderating role of neurocognition in the relationship between insight and clinical outcomes.
To address these questions, this study focused on the role of neurocognition in insight and conversion in individuals with CHR. Our objectives were to: (1) Identify specific neurocognitive functions associated with levels of impaired insight; (2) Evaluate whether impaired insight and neurocognition independently predict psychosis conversion over a six-year follow-up period; and (3) Examine whether neurocognition moderates the relationship between impaired insight and the subsequent transition to psychosis. We hypothesized that: (1) Lower performance in visuospatial memory would be significantly associated with greater impairment in insight; (2) Both impaired insight and poorer visuospatial memory would independently predict an increased risk of psychosis conversion; and (3) Visuospatial memory would moderate the impact of impaired insight on conversion risk, such that better performance would attenuate and worse performance would amplify the effect of impaired insight on psychosis transition.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Research Ethics Committee of the Shanghai Mental Health Center (SMHC) (ethical approval No. 2019-41C1). The data were derived from a previously registered longitudinal cohort (ClinicalTrials.gov: No. NCT04444180); however, the specific analyses reported here were not prespecified in the original registration. All participants were drawn from the Shanghai at Risk for Psychosis extended cohort and recruited from the SMHC and its affiliated center, the Shanghai Psychotherapy and Psychological Counseling Center. Written informed consent was obtained from all the participants. For individuals younger than 18 years, informed consent was obtained from both the adolescent and their next of kin or legal guardians.
CHR screening was conducted among outpatients seeking help for the first time, aged between 11 years and 45 years, and who had completed at least six years of primary education. The exclusion criteria were as follows: (1) A history of psychotic disorder; (2) Organic brain disease or severe somatic illness; (3) Intellectual disability (intelligence quotient < 70) or dementia; (4) Prior use of antipsychotic medication for more than two weeks; and (5) Threshold psychotic symptoms attributable to other mental disorders or psychoactive substance use.
Structured interview for psychosis-risk syndromes: The Structured Interview for Psychosis-Risk Syndromes (SIPS), developed by Miller[20-22], was designed to identify CHR individuals. It incorporates the Scale of Psychotic Risk Syndromes (SOPS), with positive symptoms playing a key role in determining the Criteria of Prodromal Syndromes (COPS). These positive symptoms included unusual thought content or delusional ideation, suspiciousness or concept of persecution, grandiose ideas, abnormal perception or hallucinations, and disorganized communication. Our team has translated the SIPS into Chinese and has been using it for approximately 14 years[23]. The Chinese version of the SIPS was used in this study, and previous research has demonstrated its good reliability and validity. The inter-rater reliability coefficient of the Chinese version of the SOPS was 0.96, and the Cronbach’s alpha coefficient was 0.71[24].
Positive and Negative Syndrome Scale: The Positive and Negative Syndrome Scale (PANSS), developed by Kay et al[25], is a widely used tool to evaluate symptom severity across three domains: Positive symptoms, negative symptoms, and general psychopathology. It comprises 30 items, each rated on a seven-point scale, where 1 represents the absence of symptoms and 7 denotes severe symptoms. The Chinese version of the PANSS has demonstrated good reliability and validity with Cronbach’s alpha coefficients ranging from 0.74 to 0.90[26]. Although initially developed for individuals with schizophrenia, the PANSS is also applicable and informative for CHR populations[27]. Insight was assessed using the G12 item (lack of judgment and insight) from the general psychopathology subscale, which has been widely adopted in prior studies[28,29]. Although insight was assessed using a single item on the PANSS, it has been widely adopted as a pragmatic and clinically meaningful measure of insight across psychotic disorders. Previous studies have demonstrated that, despite its brevity, G12 captures multiple dimensions of impaired insight and correlates significantly with more comprehensive self-report tools[30]. In the present study, a cut-off of G12 ≥ 3 was used to define impaired insight, as this threshold reflects at least mild but clinically meaningful deficits in illness awareness and treatment appraisal[29], and distinguishes impaired from intact insight.
MCCB: As mentioned previously, the MCCB was developed by the NIMH[7]. The Chinese version[31], which has been standardized in a Chinese population, was used in the present study. The corresponding normative data were not applied because the available norms do not include individuals below 18 years of age. These subtests are: Trail Making Test; Brief Assessment of Cognition in Schizophrenia: Symbol Coding (BACS-SC); Hopkins Verbal Learning Test-Revised (HVLT-R); Wechsler Memory Scale-3rd Ed: Spatial Span (WMS-III-SS); Neuropsychological Assessment Battery: Mazes; Brief Visuospatial Memory Test-Revised (BVMT-R); Mayer-Salovey-Caruso Emotional Intelligence Test (MSCEIT); Fluency: Animal Naming; and Continuous Performance Test-Identical Pairs (CPT-IP). Given that a considerable proportion of individuals is under 18 years of age and normative data for this age group are limited[12], we prioritized raw scores for all subtests over composite cognitive domain scores and excluded the MSCEIT. A comparison of the MCCB scores between the study sample and age- and sex-matched healthy controls (n = 100) is shown in Supplementary Figure 1. Additionally, we computed the z-scores for each subtest based on the mean and standard deviation of the study sample, which were then used in the logistic and Cox regression models.
This study included individuals with CHR who met the attenuated positive symptom (APS) criteria, meaning that their positive symptom scores ranged from 3 to 5 according to the COPS. The participants in this study were individuals who completed both the MCCB and PANSS assessments at baseline (n = 312) and were subsequently categorized into two groups based on their PANSS G12 item scores. Those with a G12 score below 3 were classified into the low-impairment insight group (IIL) (n = 151), whereas those with a score of 3 or higher were assigned to the high-impairment insight group (IIH) (n = 161). In this study, conversion outcomes at the six-year follow-up were obtained for 266 individuals with APS (85.26%) through face-to-face interviews, telephone interviews, or medical records. Of these, 127 (84.11%) and 139 (86.33%) were in the IIL and IIH group, respectively. To evaluate potential attrition bias, baseline demographic (age, sex, education), clinical [total score of positive symptoms on the Scale of Psychotic-Risk Syndromes (P-Total) and medication status: No treatment, antidepressants, antipsychotics, or combination therapy], and neurocognitive (MCCB domains) characteristics were compared between participants who completed the follow-up and those who did not. No significant differences were detected across any baseline demographic, clinical, or neurocognitive variables, including medication status (Supplementary Table 1), suggesting that attrition was unlikely to introduce systematic bias. Conversion to psychosis was determined based on the presence of at least one psychotic symptom (rated ‘6’) on any of the five positive symptom subscales of the SOPS, provided that the symptom occurred with adequate frequency and duration or reached a level considered disorganizing or dangerous.
All statistical analyses were conducted using RStudio (version 4.4.1). Analytic parameters, thresholds, and model specifications that could influence effect estimates were predefined based on prior literature and clinical interpretability, and are reported explicitly to facilitate reproducibility. To compare the demographic differences between the IIL and IIH groups, a χ2 test was performed for sex and medication status, while independent t-tests were used for age and education. Normality tests for the neurocognitive subsets and the P-Total revealed that only BACS-SC and CPT-IP followed a normal distribution. Robust regression analyses were used to compare the differences between the IIL and IIH groups across these variables. These analyses were conducted using the Laplace function controlling for sex, age, and education. The Holm correction method was used to adjust for multiple comparisons.
For all regression analyses, neurocognitive variables were entered as standardized z-scores to allow direct comparison across domains; thus, these predictors are labeled with the prefix “z” in tables and figures. For the logistic regression models, demographic variables (sex, age, and education) were included in the first layer using the entry method. The second layer consisted of neurocognitive subsets that showed significant differences between the two groups, as deter
Marginal effects were estimated using a slope function[32] to examine the impact of neurocognitive function on impaired insight across different P-Total scores (Supplementary Table 3). To formally test whether positive symptoms linearly moderated the association between neurocognition and impaired insight, interaction terms were included in a supplementary logistic regression model (Supplementary Table 4). To further characterize the regions of significance for these interactions, Johnson-Neyman intervals were calculated, identifying the range of P-Total scores at which the slope of neurocognitive function on impaired insight was statistically significant. This approach complements conventional pointwise marginal effects by locating clinically interpretable thresholds of symptom severity, while acknowledging that pointwise and linear-interaction frameworks rely on different analytic assumptions and may therefore yield different regions of statistical significance. In addition, to enhance clinical interpretability, contrasts were derived by computing predicted probabilities of impaired insight for individuals with high vs low neurocognitive performance (± 1 SD). From these predicted probabilities, absolute risk differences and relative risk ratios were calculated to quantify how much insight risk changed as a function of neurocognition across different P-Total values (Supplementary Table 5).
Kaplan-Meier survival curves were generated to estimate the time to conversion for the IIL and IIH groups, and group differences were evaluated using the log-rank test. To evaluate the impact of impaired insight and neurocognitive function on conversion outcomes, a Cox regression analysis was performed, controlling for sex, age, and education. Participants with missing follow-up data were excluded. The model included neurocognitive subsets, impaired insight, their interaction terms, and P-Total, and variable selection was conducted using the backward LR method. In addition, a full Cox regression model including all demographic, cognitive, and clinical variables was fitted to quantify the joint effects and to provide a pre-specified reference for model uncertainty (Supplementary Table 6).
To further assess predictor stability, penalized sensitivity analyses were conducted using LASSO regression (α = 1) with bootstrap resampling (1000 iterations), which provided inclusion frequencies for each variable. The proportional hazards assumption of the Cox models was examined using Schoenfeld residuals (Supplementary Table 7). Sensitivity analyses also included alternative operationalizations of the PANSS G12 item (Supplementary Table 8): (1) Modeled as a continuous variable; and (2) Using a more lenient threshold of ≥ 2 for impaired insight. Additionally, to evaluate potential treatment-related confounding, Cox models were re-estimated with adjustment for baseline antipsychotic exposure (binary indicator: Treated vs untreated/antidepressant only) (Supplementary Table 9). All sensitivity analyses used the same Cox regression framework as the main analysis.
Furthermore, model performance was evaluated using multiple complementary metrics. Discrimination was assessed using Harrell’s concordance index (C-index) and time-dependent area under the curve (AUC) estimates across 12-72 months. Calibration was examined using bootstrap-corrected calibration plots at multiple time points. Clinical utility was evaluated using decision curve analysis (DCA). To facilitate clinical interpretation, hazard ratios (HR) for the IIL and IIH groups across different z-scores of the neurocognitive variables were derived using the ggpredict function[33], and displayed together with corresponding 95% confidence intervals (CIs). We additionally derived absolute 6-year conversion risk estimates and risk differences between the IIL and IIH groups. Statistical significance was set at P < 0.05 (two-tailed).
When comparing the IIL and IIH groups, no significant differences (P > 0.05) were found in sex distribution, age, or educational level. However, significant differences (P < 0.05, Table 1) were observed in several neurocognitive measures, including BACS-SC, HVLT-R, WMS-III-SS, BVMT-R, and CPT-IP, as well as the total positive symptoms. Compared to the IIL group, the IIH group exhibited significantly greater neurocognitive impairment and symptom severity. However, after Holm correction, only the adjusted P value for BVMT-R remained significant (P = 0.018, Cohen’s f2 = 0.032) for neurocognitive variables, while BACS-SC showed marginal significance (P = 0.068, Cohen’s f2 = 0.030). The P value for the total score of positive symptoms was < 0.001.
| IIL (n = 151) | IIH (n = 161) | χ2/t value | P value | Cramér’s V/Cohen’s d/Cohen’s f2 | ||
| Sex | Male | 72 | 70 | 0.399 | 0.528 | 0.042 |
| Female | 79 | 91 | ||||
| Age | 18.76 ± 4.88 | 19.19 ± 5.41 | 0.727 | 0.468 | 0.082 | |
| Education (years) | 10.44 ± 2.44 | 10.58 ± 3.00 | 0.451 | 0.652 | 0.051 | |
| TMT | 31.58 ± 10.74 | 34.98 ± 14.40 | 0.760 | 0.448 | 0.017 | |
| BACS-SC | 59.06 ± 9.82 | 55.60 ± 10.92 | -2.600 | 0.010 | 0.030 | |
| HVLT-R | 24.31 ± 4.91 | 22.78 ± 5.31 | -2.196 | 0.029 | 0.023 | |
| WMS-III-SS | 15.95 ± 2.96 | 15.21 ± 3.07 | -2.378 | 0.018 | 0.014 | |
| NAB-Mazes | 17.09 ± 6.35 | 15.88 ± 6.44 | -1.519 | 0.130 | 0.007 | |
| BVMT-R | 27.56 ± 6.43 | 25.35 ± 6.00 | -3.090 | 0.002 | 0.032 | |
| Fluency | 19.54 ± 5.16 | 19.33 ± 5.32 | -0.433 | 0.665 | 0.001 | |
| CPT-IP | 2.54 ± 0.78 | 2.38 ± 0.79 | -2.023 | 0.044 | 0.013 | |
| P-Total | 8.27 ± 2.64 | 11.47 ± 3.02 | 9.605 | < 0.001 | 0.325 | |
| Medication | Combination | 26 | 13 | 12.254 | 0.007 | 0.198 |
| Antipsychotics | 92 | 122 | ||||
| Antidepressants | 12 | 4 | ||||
| No medication | 21 | 22 | ||||
Baseline pharmacological treatment differed significantly across insight groups (P < 0.05). Participants in the IIH group were more likely to receive antipsychotic treatment, either as monotherapy or in combination, whereas antidepressant monotherapy was more common in the IIL group.
In the binary logistic regression analysis (Table 2), the first model (using the enter method) showed no significant effects of sex, age, or education on impaired insight, with odds ratios (OR) close to one for all variables. In the second layer of the regression model (backward LR), z-score for the BACS-SC (z-BACS-SC) (OR = 0.767, 95%CI: 0.589-0.991, P = 0.045) and z-score for the BVMT-R (z-BVMT-R) (OR = 0.752, 95%CI: 0.574-0.979, P = 0.036) scores significantly predicted impaired insight, indicating that lower neurocognitive scores were associated with higher odds of impaired insight. However, after adding the total score of positive symptoms in the third layer of regression, the significance of z-BVMT-R increased (OR = 0.695, 95%CI: 0.510-0.939, P = 0.019), while z-BACS-SC became nonsignificant (OR = 0.835, 95%CI: 0.616-1.124, P = 0.237). The total number of positive symptoms was strongly positively associated with impaired insight (OR = 1.503, 95%CI: 1.360-1.679, P < 0.001).
| Model | β | SE | Wald | P value | OR | 95%CI for OR | |
| Lower | Upper | ||||||
| Model 1 (enter) | |||||||
| Sex | -0.165 | 0.229 | 0.520 | 0.471 | 0.848 | 0.540 | 1.328 |
| Age | 0.015 | 0.030 | 0.253 | 0.615 | 1.015 | 0.958 | 1.078 |
| Education | 0.002 | 0.056 | 0.001 | 0.974 | 1.002 | 0.897 | 1.118 |
| Model 2 (backward: LR) | |||||||
| Sex | -0.261 | 0.237 | 1.209 | 0.272 | 0.770 | 0.482 | 1.225 |
| Age | -0.02 | 0.032 | 0.408 | 0.523 | 0.980 | 0.920 | 1.044 |
| Education | 0.054 | 0.059 | 0.846 | 0.358 | 1.056 | 0.941 | 1.186 |
| z-BACS-SC | -0.265 | 0.132 | 4.004 | 0.045 | 0.767 | 0.589 | 0.991 |
| z-BVMT-R | -0.285 | 0.136 | 4.407 | 0.036 | 0.752 | 0.574 | 0.979 |
| Model 3 (backward: LR) | |||||||
| Sex | -0.121 | 0.274 | 0.195 | 0.659 | 0.886 | 0.517 | 1.517 |
| Age | 0.008 | 0.037 | 0.048 | 0.826 | 1.008 | 0.938 | 1.085 |
| Education | 0.041 | 0.067 | 0.377 | 0.539 | 1.042 | 0.914 | 1.189 |
| z-BACS-SC | -0.180 | 0.153 | 1.397 | 0.237 | 0.835 | 0.616 | 1.124 |
| z-BVMT-R | -0.364 | 0.155 | 5.504 | 0.019 | 0.695 | 0.510 | 0.939 |
| P-Total | 0.407 | 0.054 | 57.686 | < 0.001 | 1.503 | 1.360 | 1.679 |
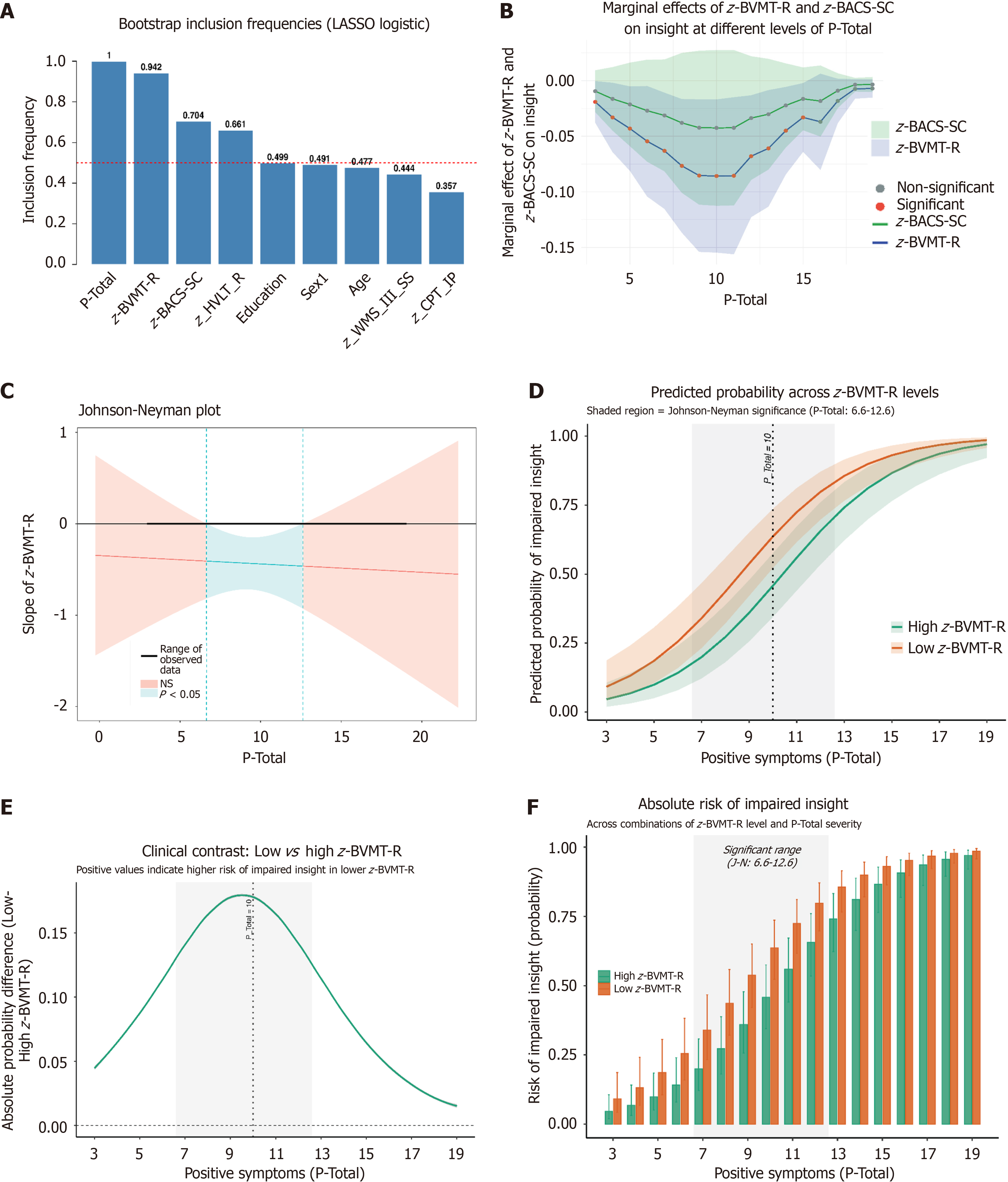
Consistent with these findings, the LASSO logistic model demonstrated that P-Total (inclusion frequency = 1.000) and z-BVMT-R (0.942) were the two most robust predictors across 1000 bootstrap iterations, followed by z-BACS-SC (0.704), whereas other cognitive and demographic variables showed modest or low inclusion frequencies (Figure 1A). These results highlight the central role of positive symptoms and visuospatial memory in predicting impaired insight.
Marginal effects analysis (Figure 1B) indicated that z-BVMT-R had a consistently negative association with impaired insight, meaning poorer visuospatial memory was linked to a higher probability of insight impairment. The effect peaked at mild-to-moderate positive symptom levels (approximately P-Total ≤ 10; -0.019 to -0.086) and diminished at higher symptom severity, becoming nonsignificant beyond P-Total approximately 15.
The Johnson-Neyman analysis (Figure 1C) yielded a more conservative significance range (P-Total = 6.64-12.62), consistent in direction with the marginal effects pattern but narrower owing to its reliance on the linear interaction model. The formal z-BVMT-R × P-Total interaction term was nonsignificant (Supplementary Table 4), indicating that the mode
Predicted probability curves (Figure 1D) further showed that individuals with low visuospatial memory (-1 SD) had substantially higher predicted risk of impaired insight than those with high memory (+1 SD), especially at moderate symptom levels. The corresponding low-high absolute probability difference (Figure 1E) peaked at approximately 18% around P-Total = 9-10 and declined at both lower and higher symptom levels. Figure 1F displays the separate absolute risk curves for low and high BVMT-R groups, illustrating the same pattern: The risk gap is largest at moderate symptom severity and converges when symptoms are severe.
By contrast, BACS-SC did not show significant effects on impaired insight across any P-Total level (Figure 1B, pale green band). This suggests that the BACS-SC does not have a robust relationship with impaired insight within this dataset.
During the 6-year follow-up, 47.5% of the participants in the IIH group (66/139) and 29.9% in the IIL group (38/127) converted. The log-rank test revealed a significant difference between the two groups (χ2 = 8.226, df = 1, P = 0.004). The final Cox proportional hazards regression model identified z-BVMT-R (β = -0.452, P < 0.001, HR = 0.636, 95%CI: 0.518-0.782) and impaired insight (β = 0.456, P = 0.027, HR = 1.577, 95%CI: 1.052-2.366) as significant predictors, whereas their interaction terms and total positive symptoms were excluded. Additionally, male sex remained significant (β = 0.631, P = 0.002, HR = 1.880, 95%CI: 1.266-2.791). The detailed results are presented in Table 3.
| Model | β | SE | Wald | P value | HR | 95%CI for HR | |
| Lower | Upper | ||||||
| Model 1 (enter) | |||||||
| Sex | 0.556 | 0.201 | 7.629 | 0.006 | 1.745 | 1.175 | 2.589 |
| Age | 0.049 | 0.022 | 5.04 | 0.025 | 1.051 | 1.006 | 1.097 |
| Education | -0.085 | 0.045 | 3.549 | 0.060 | 0.918 | 0.841 | 1.003 |
| Model 2 (backward: LR) | |||||||
| Sex | 0.631 | 0.202 | 9.791 | 0.002 | 1.88 | 1.266 | 2.791 |
| Age | 0.017 | 0.023 | 0.516 | 0.473 | 1.017 | 0.972 | 1.064 |
| Education | -0.064 | 0.046 | 1.938 | 0.164 | 0.938 | 0.857 | 1.027 |
| Impaired insight | 0.456 | 0.207 | 4.858 | 0.027 | 1.577 | 1.052 | 2.366 |
| z-BVMT-R | -0.452 | 0.105 | 18.438 | < 0.001 | 0.636 | 0.518 | 0.782 |
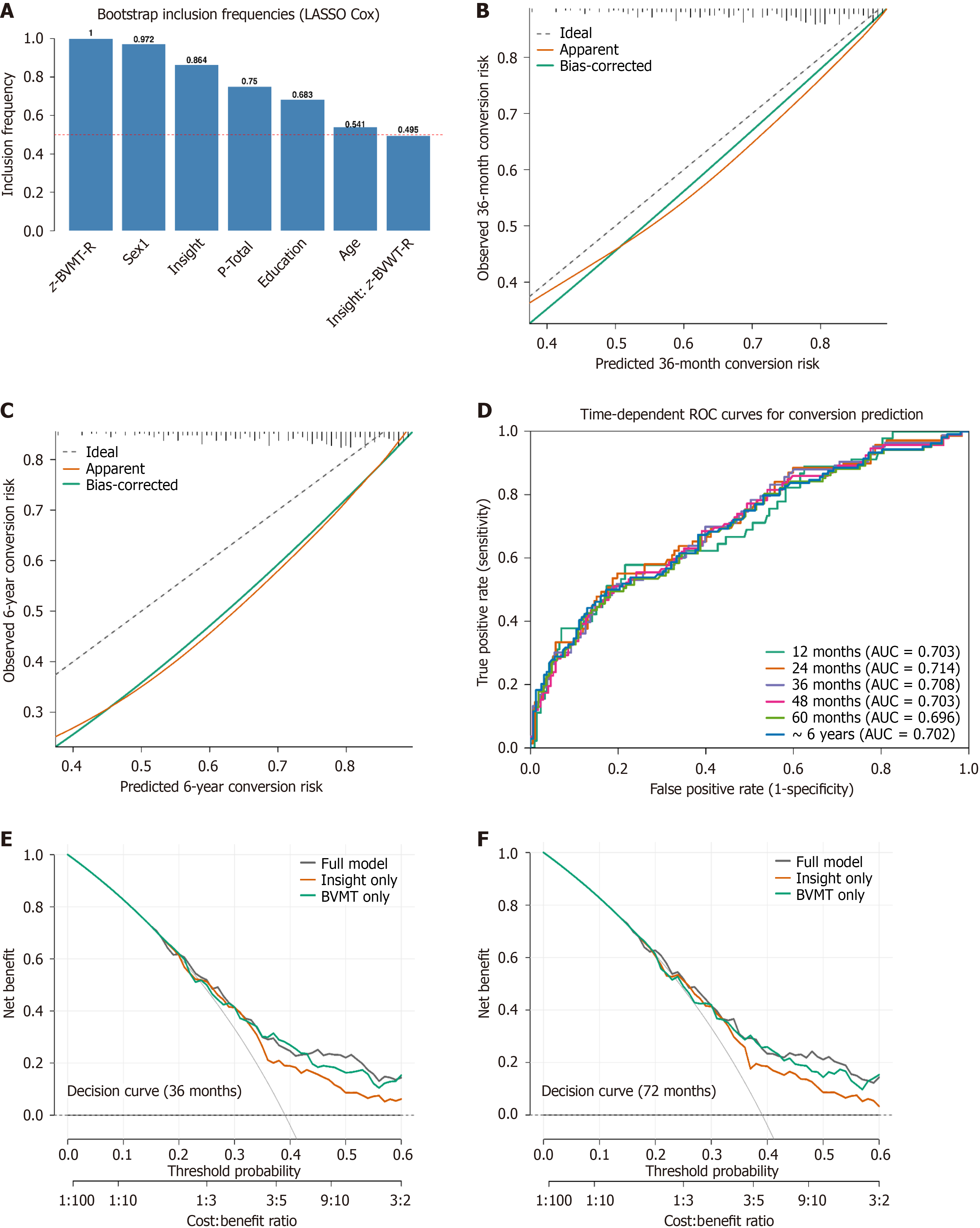
Sensitivity analyses using LASSO regression with bootstrap resampling revealed that z-BVMT-R, sex, and impaired insight were consistently selected across resamples (Figure 2A). Schoenfeld residual tests confirmed that the proportional hazards assumption was met for all covariates (all P > 0.05; Supplementary Table 7). Additional sensitivity analyses using alternative operationalizations of G12 or adjustment for baseline antipsychotic exposure produced comparable results (Supplementary Tables 8 and 9).
Model performance indices further supported the robustness of the final model. Discriminative performance was acceptable: Harrell’s C-index was 0.666, and time-dependent AUCs remained stable from 12 months to 72 months (Figure 2B). Calibration accuracy was highest at mid-term follow-up; representative 36- and 72-month calibration plots are shown in Figure 2C and D, with full calibration curves for all time points provided in Supplementary Figure 2. DCA similarly demonstrated modest but consistent net benefit for the full model over the insight-only model; representative curves at 36 months and 72 months are displayed in Figure 2E and F, with complete 12-72-month DCA results shown in Supplementary Figure 3 and Supplementary Table 10. Although sex was a significant covariate, neither the BVMT-R × sex nor insight × sex interaction reached significance (Supplementary Table 11), with sex-stratified predicted risk curves shown in Supplementary Figure 4.
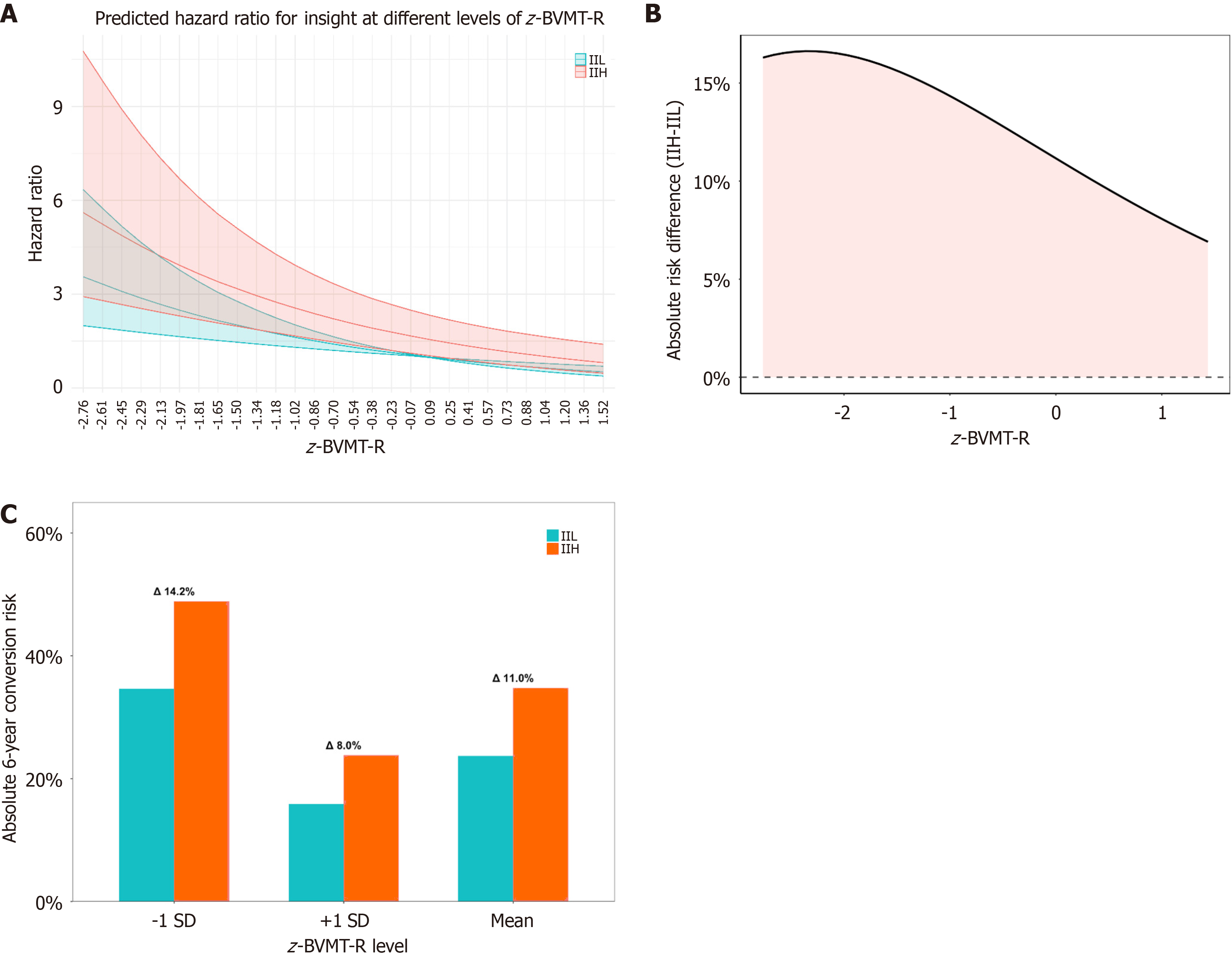
The predicted conversion risk was assessed in both the IIL and IIH groups, and the results indicated that the HR for conversion associated with impaired insight decreased as z-BVMT-R increased. Higher z-BVMT-R values were consistently associated with lower predicted conversion risk. Specifically, for the IIL group, the predicted HR ranged from 3.55 (95%CI: 1.99-6.34) at lower z-BVMT-R values to 0.81 (95%CI: 0.47-1.40) at higher z-BVMT-R values. Similarly, for the IIH group, the predicted HR ranged from 5.61 (95%CI: 2.92-10.77) to 0.81 (95%CI: 0.47-1.40) as z-BVMT-R increased (Figure 3A and Supplementary Table 12).
To enhance clinical interpretability, we additionally computed absolute 6-year conversion risks and the corresponding IIH-IIL risk difference across the full range of visuospatial memory. As shown in Figure 3B, the absolute risk difference was largest at lower z-BVMT-R values, gradually narrowing at higher levels of visuospatial memory, indicating that better visuospatial performance reduces both overall risk and the incremental burden associated with impaired insight. Representative risk differences at -1 SD, +1 SD, and mean z-BVMT-R values are displayed in Figure 3C, demonstrating that the gap between insight groups diminishes as visuospatial memory improves. Together, these results suggest that visuospatial memory not only predicts conversion risk independently but also mitigates the adverse effect of impaired insight on long-term outcomes.
This is the first study to investigate the neurocognitive correlates of impaired insight and how neurocognitive factors condition the clinical impact of impaired insight in CHR individuals with APSs. Across baseline comparisons, impaired insight was associated with broad neurocognitive deficits, with visuospatial memory (BVMT-R) emerging as the only robust difference after multiple comparison correction. Consistent with this pattern, logistic regression and LASSO analyses identified BVMT-R as the most stable neurocognitive predictor of impaired insight, second only to positive symptom burden. Importantly, the influence of visuospatial memory on insight impairment was not uniform across symptom severity: BVMT-R had its strongest association at mild-to-moderate positive symptom levels and diminished when symptoms became severe. Beyond cross-sectional associations, both impaired insight and visuospatial memory independently predicted longitudinal conversion to psychosis, and higher visuospatial memory attenuated the adverse prognostic impact of impaired insight. Together, these findings highlight visuospatial memory as a key cognitive marker shaping both the emergence of impaired insight and its long-term clinical consequences in CHR individuals.
The prominence of visuospatial memory in our findings aligns with, yet also extends, previous literature on the cognitive underpinnings of impaired insight. Previous studies have emphasized the role of neurocognitive deficits, particularly in executive function and working memory, in impaired insight[34-38]. However, visuospatial memory has rarely been highlighted as a key determinant, and existing results have been mixed. For instance, Chan[17] found no significant relationship between immediate visual memory and insight, and the relationship with delayed visual memory did not survive multivariate adjustment. Such inconsistencies may reflect differences in symptom severity and illness stage across samples, factors that our marginal effects analyses indicate critically shape how visuospatial memory relates to insight. Specifically, we found that visuospatial memory exerted its strongest influence when positive symptoms were mild to moderate, whereas this association diminished as symptoms became more severe, suggesting a symptom-dependent contribution that may help explain discordant findings in earlier work.
A key contribution of our study is the demonstration that visuospatial memory does not influence insight uniformly across the spectrum of positive symptoms. Instead of a simple linear moderation effect, the combined evidence from marginal-effects estimation, Johnson-Neyman intervals, and risk-contrast analyses indicates that cognition contributes meaningfully to insight only within a bounded symptom range. Specifically, the strongest cognitive effects emerged within the lower-to-intermediate range of positive symptoms generally before P-Total reached approximately 15, beyond which the influence of visuospatial memory rapidly diminished. A more conservative Johnson-Neyman interval suggested a narrower statistical window (between 7 and 13), but the overall pattern remained the same: Once symptoms exceeded a certain threshold, their impact overshadowed cognitive contributions to insight. This pattern underscores that the cognitive underpinnings of insight are most relevant when psychopathology is not yet overwhelming, offering a more nuanced framework for interpreting how neurocognition interacts with emerging psychotic processes.
Taken together, these findings indicate that visuospatial memory may exert its greatest influence on insight earlier in the illness course or when symptoms are less severe, potentially serving as a cognitive resource that helps maintain accurate self-monitoring. In contrast, at more severe levels of positive symptoms, impaired insight may be driven less by cognitive mechanisms and more directly by symptom-driven disruptions in self-monitoring and illness attribution. This interpretation aligns with evidence that individuals at high-risk state or with milder psychotic symptoms exhibit relatively preserved neural systems supporting metacognition and self-monitoring[39,40], allowing neurocognition to play a more influential role. These findings underscore the clinical relevance of identifying individuals whose insight impairment is more cognitively driven and therefore potentially responsive to cognitive remediation particularly during periods of moderate symptom severity.
Cox regression analysis identified both impaired insight and visuospatial memory as independent predictors of conversion, after adjusting for age, sex, and education level. The fact that P-Total did not enter the final model reinforces the notion that the two factors uniquely contribute to conversion risk, independent of symptom severity[4,11]. Although the visuospatial memory × insight interaction term was not retained, a series of secondary analyses provided important clarification regarding how these predictors jointly operate. Across multiple model performance indices including LASSO stability selection, time-dependent AUCs, and Harrell’s C-index, the combined contribution of impaired insight and visuospatial memory to conversion risk proved highly robust. Furthermore, calibration analyses further indicated good agreement between predicted and observed risk at mid-term follow-up, and DCA showed that models incorporating visuospatial memory consistently yielded greater clinical utility than those relying on insight alone.
Beyond these overall model characteristics, risk curve analyses and absolute 6-year conversion-risk differences offered important clinical nuance. Across the full BVMT-R range, individuals in the IIH group consistently showed a higher predicted conversion risk than those in the IIL group, but the IIH-IIL risk difference was most pronounced at poorer memory levels and narrowed substantially at higher levels. Although this pattern did not manifest as a statistically significant interaction term, it reflects a clear risk gradient: Impaired insight confers the greatest vulnerability when visuospatial memory is poor, whereas stronger visuospatial memory attenuates this vulnerability. This suggests that visuospatial memory may act as a form of cognitive resilience that softens the long-term impact of impaired insight on clinical outcomes[11].
The absence of significant sex interactions despite sex itself being an independent predictor of conversion further indicates that the modifying influence of visuospatial memory on insight-related risk is not sex-specific. Taken together, these findings highlight a dual role for visuospatial memory: It functions both as a direct predictor of psychosis onset and as a factor that shapes the clinical expression of insight-related risk. This pattern suggests a potential therapeutic implication: Early interventions that strengthen visuospatial learning and memory processes may help buffer against the adverse consequences of impaired insight, particularly during the earlier or less severe stages of clinical high-risk progression.
The strengths of this study include the use of complementary analytic approaches including interaction probing, marginal effects modeling, absolute risk estimation, and model performance evaluation to delineate the nuanced role of visuospatial memory in shaping impaired insight and its downstream impact on psychosis risk. By demonstrating that: (1) The influence of neurocognition on insight varies across levels of positive-symptom severity; and (2) The impact of impaired insight on conversion risk differs across levels of neurocognition, our findings offer a more refined and clinically meaningful account of how cognitive and symptomatic vulnerabilities interact in the early course of psychosis.
These insights have important clinical implications for early intervention. Specifically, deficits in visuospatial learning, reflected by poorer BVMT-R performance, may contribute to reduced insight and an increased risk of transition to psychosis. Interventions that enhance visuospatial memory and related cognitive processes could potentially help preserve insight and delay or prevent conversion in high-risk individuals. Moreover, neuromodulation techniques such as transcranial direct current stimulation or repetitive transcranial magnetic stimulation targeting parietal or hippo
However, this study has several limitations that warrant consideration. First, the construct of insight is multidimensional, encompassing awareness of illness, recognition of symptoms, and acknowledgment of the need for treatment. While the PANSS G12 item captures only the clinician-rated global dimension of insight, it remains one of the most widely adopted and validated indicators in both schizophrenia and CHR research[4,41-43]. Previous studies have shown moderate to strong correlations between PANSS item G12 and multidimensional clinician-rated insight scales such as the Scale to Assess Unawareness in Mental Disorder[44], and significant associations with the Schedule for Assessment of Insight and its expanded version[45,46], supporting its criterion validity. Nevertheless, reliance on a single PANSS item may be subject to restricted score variability and potential floor or ceiling effects, particularly in CHR populations where severe impairment is uncommon. To address these limitations, we conducted sensitivity analyses modeling G12 as a continuous predictor as well as using an alternative cut-off (≥ 2), which yielded consistent findings, thereby supporting the robustness of the main results. Hence, although it is a single-item measure, PANSS G12 serves as a practical and valid proxy for overall clinical insight.
Second, although this study had a relatively long follow-up duration, we lacked repeated measurements of neurocognitive functioning and insight over time. This limited our ability to examine the dynamic or bidirectional influences of cognition, insight, and clinical outcomes. Future research should adopt a longitudinal design with multiple assessment points to capture these trajectories better. Third, although most CHR individuals receive pharmacotherapy, our study lacked sufficiently detailed longitudinal treatment records (e.g., medication changes, psychosocial interventions, substance use) to model treatment as a time-varying covariate. Although baseline treatment status did not differ between those retained or lost to follow-up, and adjusting for baseline antipsychotic exposure did not alter the predictive effects of visuospatial memory or insight, the absence of high-resolution treatment data prevents a full assessment of how clinical management may influence symptom trajectories or conversion risk. Future research using high-resolution, time-resolved treatment data will be crucial to clarify whether clinical management modulates neurocognitive predictors of insight and subsequent outcomes. Last, the study was conducted in a single cultural and clinical setting, which may limit the generalizability of the findings. Cultural factors, including variations in help-seeking behaviors, illness perception, and social attitudes toward mental illness in East Asian contexts, may influence both insight and clinical outcomes. Future studies across diverse cultural contexts and healthcare systems are needed to validate and extend these findings.
In conclusion, our findings underscore the intricate interplay between neurocognition, insight, and clinical outcomes in CHR individuals. Visuospatial memory has emerged as a key contributor to impaired insight, with its influence varying systematically across levels of positive-symptom severity. In contrast, processing speed showed no significant association with insight across the symptom spectrum. Both impaired insight and visuospatial memory independently predicted the transition to psychosis over a six-year follow-up. Although no statistically significant interaction was observed in the Cox model, risk-curve analyses consistently showed that higher visuospatial memory was associated with a weaker conver
We thank our participants and their families for their support, and acknowledge the Base for Interdisciplinary Innovative Talent Training at SJTU and the College Students’ Innovative Entrepreneurial Training Plan Program.
| 1. | David AS. Insight and psychosis. Br J Psychiatry. 1990;156:798-808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 755] [Cited by in RCA: 696] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 2. | Mintz AR, Dobson KS, Romney DM. Insight in schizophrenia: a meta-analysis. Schizophr Res. 2003;61:75-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 423] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 3. | Lappin JM, Morgan KD, Valmaggia LR, Broome MR, Woolley JB, Johns LC, Tabraham P, Bramon E, McGuire PK. Insight in individuals with an At Risk Mental State. Schizophr Res. 2007;90:238-244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 39] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 4. | Xu L, Wang Y, Cui H, Wei Y, Tang X, Qian Z, Zhu J, Hong X, Hu Y, Hu H, Guo Q, Qing Y, Tang Y, Zhang T, Wang J. Impaired insight as a predictor of psychotic transition in high-risk individuals with attenuated positive symptoms. Asian J Psychiatr. 2025;108:104504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Yung AR, McGorry PD. The prodromal phase of first-episode psychosis: past and current conceptualizations. Schizophr Bull. 1996;22:353-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 775] [Cited by in RCA: 707] [Article Influence: 23.6] [Reference Citation Analysis (0)] |
| 6. | Bora E, Murray RM. Meta-analysis of cognitive deficits in ultra-high risk to psychosis and first-episode psychosis: do the cognitive deficits progress over, or after, the onset of psychosis? Schizophr Bull. 2014;40:744-755. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 423] [Cited by in RCA: 368] [Article Influence: 30.7] [Reference Citation Analysis (0)] |
| 7. | Kern RS, Green MF, Nuechterlein KH, Deng BH. NIMH-MATRICS survey on assessment of neurocognition in schizophrenia. Schizophr Res. 2004;72:11-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 87] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 8. | Bora E, Lin A, Wood SJ, Yung AR, McGorry PD, Pantelis C. Cognitive deficits in youth with familial and clinical high risk to psychosis: a systematic review and meta-analysis. Acta Psychiatr Scand. 2014;130:1-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 225] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 9. | Zheng W, Zhang QE, Cai DB, Ng CH, Ungvari GS, Ning YP, Xiang YT. Neurocognitive dysfunction in subjects at clinical high risk for psychosis: A meta-analysis. J Psychiatr Res. 2018;103:38-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 42] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 10. | Catalan A, Salazar de Pablo G, Aymerich C, Damiani S, Sordi V, Radua J, Oliver D, McGuire P, Giuliano AJ, Stone WS, Fusar-Poli P. Neurocognitive Functioning in Individuals at Clinical High Risk for Psychosis: A Systematic Review and Meta-analysis. JAMA Psychiatry. 2021;78:859-867. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 178] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 11. | Bang M, Kim KR, Song YY, Baek S, Lee E, An SK. Neurocognitive impairments in individuals at ultra-high risk for psychosis: Who will really convert? Aust N Z J Psychiatry. 2015;49:462-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 12. | Cui H, Giuliano AJ, Zhang T, Xu L, Wei Y, Tang Y, Qian Z, Stone LM, Li H, Whitfield-Gabrieli S, Niznikiewicz M, Keshavan MS, Shenton ME, Wang J, Stone WS. Cognitive dysfunction in a psychotropic medication-naïve, clinical high-risk sample from the ShangHai-At-Risk-for-Psychosis (SHARP) study: Associations with clinical outcomes. Schizophr Res. 2020;226:138-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 39] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 13. | Zhang T, Xu L, Wei Y, Cui H, Tang X, Hu Y, Liu H, Wang Z, Chen T, Yi Z, Li C, Wang J. Symptom Dimensions and Cognitive Impairments in Individuals at Clinical High Risk for Psychosis. Biol Psychiatry Cogn Neurosci Neuroimaging. 2025;10:646-655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 14. | Alkan E, L H Evans S. Relationships between cognitive performance, clinical insight and regional brain volumes in schizophrenia. Schizophrenia (Heidelb). 2022;8:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 15. | Mervis JE, Vohs JL, Lysaker PH. An update on clinical insight, cognitive insight, and introspective accuracy in schizophrenia-spectrum disorders: symptoms, cognition, and treatment. Expert Rev Neurother. 2022;22:245-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 16. | Lysaker PH, Pattison ML, Leonhardt BL, Phelps S, Vohs JL. Insight in schizophrenia spectrum disorders: relationship with behavior, mood and perceived quality of life, underlying causes and emerging treatments. World Psychiatry. 2018;17:12-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 173] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 17. | Chan KK. Associations of symptoms, neurocognition, and metacognition with insight in schizophrenia spectrum disorders. Compr Psychiatry. 2016;65:63-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Pousa E, Brébion G, López-Carrilero R, Ruiz AI, Grasa E, Barajas A, Peláez T, Alfonso-Gutiérrrez-Zotes, Lorente E, Barrigón ML, Ruiz-Delgado I, González-Higueras F, Cid J, Pérez-Solà V; Spanish Metacognition Study Group; Insight Barcelona Work Group, Ochoa S. Clinical insight in first-episode psychosis: Clinical, neurocognitive and metacognitive predictors. Schizophr Res. 2022;248:158-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 19. | Gan J, Fu H, Zhu X. Relationships Between Multiple Dimensions of Insight and Neurocognition, Metacognition, and Social Cognition: A Meta-Analysis. J Nerv Ment Dis. 2022;210:577-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 20. | Miller TJ, McGlashan TH, Rosen JL, Cadenhead K, Cannon T, Ventura J, McFarlane W, Perkins DO, Pearlson GD, Woods SW. Prodromal assessment with the structured interview for prodromal syndromes and the scale of prodromal symptoms: predictive validity, interrater reliability, and training to reliability. Schizophr Bull. 2003;29:703-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1505] [Cited by in RCA: 1411] [Article Influence: 61.3] [Reference Citation Analysis (1)] |
| 21. | Miller TJ, McGlashan TH, Rosen JL, Somjee L, Markovich PJ, Stein K, Woods SW. Prospective diagnosis of the initial prodrome for schizophrenia based on the Structured Interview for Prodromal Syndromes: preliminary evidence of interrater reliability and predictive validity. Am J Psychiatry. 2002;159:863-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 603] [Cited by in RCA: 648] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 22. | Miller TJ, McGlashan TH, Woods SW, Stein K, Driesen N, Corcoran CM, Hoffman R, Davidson L. Symptom assessment in schizophrenic prodromal states. Psychiatr Q. 1999;70:273-287. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 452] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 23. | Zhang T, Li H, Woodberry KA, Seidman LJ, Zheng L, Li H, Zhao S, Tang Y, Guo Q, Lu X, Zhuo K, Qian Z, Chow A, Li C, Jiang K, Xiao Z, Wang J. Prodromal psychosis detection in a counseling center population in China: an epidemiological and clinical study. Schizophr Res. 2014;152:391-399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 124] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 24. | Zheng LN, Wang JJ, Zhang TH, Li H, Li CB, Jiang KD. [Reliability and validity of the Chinese version of Scale of Psychosis-risk Symptoms]. Zhongguo Xinli Weisheng Zazhi. 2012;26:571-576. [DOI] [Full Text] |
| 25. | Kay SR, Fiszbein A, Opler LA. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull. 1987;13:261-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17607] [Cited by in RCA: 16191] [Article Influence: 415.2] [Reference Citation Analysis (0)] |
| 26. | Si TM, Yang JZ, Shu L, Wang XL, Kong QM, Zhou M, Li XN, Liu C. [The Reliability, Validity of PANSS and its Implication]. Zhongguo Xinli Weisheng Zazhi. 2004;1:45-47. [DOI] [Full Text] |
| 27. | Lim K, Peh OH, Yang Z, Rekhi G, Rapisarda A, See YM, Rashid NAA, Ang MS, Lee SA, Sim K, Huang H, Lencz T, Lee J, Lam M. Large-scale evaluation of the Positive and Negative Syndrome Scale (PANSS) symptom architecture in schizophrenia. Asian J Psychiatr. 2021;62:102732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 28. | Lincoln TM, Lüllmann E, Rief W. Correlates and long-term consequences of poor insight in patients with schizophrenia. A systematic review. Schizophr Bull. 2007;33:1324-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 338] [Cited by in RCA: 351] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 29. | Kim J, Ozzoude M, Nakajima S, Shah P, Caravaggio F, Iwata Y, De Luca V, Graff-Guerrero A, Gerretsen P. Insight and medication adherence in schizophrenia: An analysis of the CATIE trial. Neuropharmacology. 2020;168:107634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 71] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 30. | Fischer-Vieler T, Ringen PA, Kvig E, Bell C, Hjell G, Tesli N, Rokicki J, Melle I, Andreassen OA, Friestad C, Haukvik UK. Associations Between Clinical Insight and History of Severe Violence in Patients With Psychosis. Schizophr Bull Open. 2023;4:sgad011. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 31. | Shi C, Kang L, Yao S, Ma Y, Li T, Liang Y, Cheng Z, Xu Y, Shi J, Xu X, Zhang C, Franklin DR, Heaton RK, Jin H, Yu X. The MATRICS Consensus Cognitive Battery (MCCB): Co-norming and standardization in China. Schizophr Res. 2015;169:109-115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 286] [Cited by in RCA: 279] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 32. | Arel-Bundock V, Greifer N, Heiss A. How to Interpret Statistical Models Using marginaleffects for R and Python. J Stat Soft. 2024;111. [DOI] [Full Text] |
| 33. | Lüdecke D. ggeffects: Tidy Data Frames of Marginal Effects from Regression Models. J Open Source Softw. 2018;3:772. [DOI] [Full Text] |
| 34. | Mutsatsa SH, Joyce EM, Hutton SB, Barnes TR. Relationship between insight, cognitive function, social function and symptomatology in schizophrenia: the West London first episode study. Eur Arch Psychiatry Clin Neurosci. 2006;256:356-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 35. | Mysore A, Parks RW, Lee KH, Bhaker RS, Birkett P, Woodruff PW. Neurocognitive basis of insight in schizophrenia. Br J Psychiatry. 2007;190:529-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 36. | Nair A, Palmer EC, Aleman A, David AS. Relationship between cognition, clinical and cognitive insight in psychotic disorders: a review and meta-analysis. Schizophr Res. 2014;152:191-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 129] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 37. | Joseph B, Narayanaswamy JC, Venkatasubramanian G. Insight in schizophrenia: relationship to positive, negative and neurocognitive dimensions. Indian J Psychol Med. 2015;37:5-11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 32] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 38. | Khalil AH, El-Meguid MA, Bastawy M, Rabei S, Ali R, Abd Elmoneam MHE. Correlating cognitive functions to symptom domains and insight in Egyptian patients with schizophrenia. Int J Soc Psychiatry. 2020;66:240-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 39. | Yao Y, Zhang S, Wang B, Lin X, Zhao G, Deng H, Chen Y. Neural dysfunction underlying working memory processing at different stages of the illness course in schizophrenia: a comparative meta-analysis. Cereb Cortex. 2024;34:bhae267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 40. | Zhao Y, Zhang Q, Shah C, Li Q, Sweeney JA, Li F, Gong Q. Cortical Thickness Abnormalities at Different Stages of the Illness Course in Schizophrenia: A Systematic Review and Meta-analysis. JAMA Psychiatry. 2022;79:560-570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 123] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 41. | Lysaker PH, Weiden PJ, Sun X, O'Sullivan AK, McEvoy JP. Impaired insight in schizophrenia: impact on patient-reported and physician-reported outcome measures in a randomized controlled trial. BMC Psychiatry. 2022;22:574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 42. | Bornheimer LA, Cobia DJ, Li Verdugo J, Holzworth J, Smith MJ. Clinical insight and cognitive functioning as mediators in the relationships between symptoms of psychosis, depression, and suicide ideation in first-episode psychosis. J Psychiatr Res. 2022;147:85-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 43. | Hazan H, Ferrara M, Gibbs-Dean T, Tayfur SN, Corbera S, Karmani S, Song Z, Li F, Vlachos I, Selakovic M, Tek C, Srihari VH. Insight and Its Trajectory: Predicting the Risk of Psychiatric Hospitalizations Among First-Episode Psychosis During the First Year of Coordinated Specialty Care. Schizophr Bull. 2026;52:sbaf019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 44. | Michel P, Baumstarck K, Auquier P, Amador X, Dumas R, Fernandez J, Lancon C, Boyer L. Psychometric properties of the abbreviated version of the Scale to Assess Unawareness in Mental Disorder in schizophrenia. BMC Psychiatry. 2013;13:229. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 51] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 45. | Soriano-Barceló J, López-Moríñigo JD, Ramos-Ríos R, Rodríguez-Zanabria EA, David AS. Insight assessment in psychosis and psychopathological correlates: Validation of the Spanish version of the schedule for assessment of insight – Expanded version. Eur J Psychiat. 2016;30. |
| 46. | Ito S, Matsumoto J, Sakai Y, Miura K, Hasegawa N, Yamamori H, Ishimaru K, Kim Y, Hashimoto R. Positive association between insight and attitudes toward medication in Japanese patients with schizophrenia: Evaluation with the Schedule for Assessment of Insight (SAI) and the Drug Attitude Inventory - 10 Questionnaire (DAI-10). Psychiatry Clin Neurosci. 2021;75:187-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |