Published online Mar 19, 2026. doi: 10.5498/wjp.v16.i3.114036
Revised: October 21, 2025
Accepted: December 2, 2025
Published online: March 19, 2026
Processing time: 171 Days and 18.9 Hours
Schizophrenia is a severe neuropsychiatric disorder with unclear pathogenesis, although immune-inflammatory pathways are being increasingly implicated. Elevated proinflammatory cytokines are consistently observed in patients with schizophrenia, suggesting a state of chronic low-grade inflammation. Modified el
To examine the association between MECT-induced changes in immunoinflammatory markers and clinical improvement in schizophrenia.
In this prospective study, 619 patients with schizophrenia underwent MECT. Peripheral immunoinflammatory markers, including monocyte-to-lymphocyte ratio (MLR), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio, and systemic immune-inflammatory index (SII), were measured before and after treatment. Clinical symptoms were assessed using the Positive and Negative Syn
After MECT, significant reductions were observed in PANSS scores and most peripheral inflammatory markers (MLR, NLR and SII; all P < 0.05). The decreases in MLR, NLR, and SII showed a significant positive correlation with the PANSS score reduction rate (P < 0.05). Patients with marked clinical improvement showed greater decreases in inflammatory markers. Logistic regression identifies the change in MLR before and after treatment (ΔMLR) as a strong predictor of treatment response, with each 0.1-unit increase associated with a 57% greater probability of clinical symptom improvement (odds ratios = 1.57, P < 0.001). Receiver operating characteristic analysis demonstrated that ΔMLR had sig
MECT modulates peripheral immune inflammation in schizophrenia, and these changes correlate with clinical improvement. ΔMLR may serve as a valuable predictor of MECT treatment response.
Core Tip: This study demonstrates that modified electroconvulsive therapy significantly improves clinical symptoms and reduces peripheral inflammatory markers - including monocyte-to-lymphocyte ratio (MLR), neutrophil-to-lymphocyte ratio, and systemic immune-inflammatory indices - in patients with schizophrenia. Reductions in MLR were strongly correlated with clinical improvement and served as a robust predictor of treatment response. Specifically, each 0.1-unit increase in ΔMLR was associated with a 57% higher likelihood of significant symptom improvement. These findings suggest an immunomodulatory mechanism of modified electroconvulsive therapy and support the use of MLR as a potential biomarker for predicting treatment efficacy.
- Citation: Hou L, Chen R, Huang CB, Shi WJ, Wu LL. Clinical symptom improvement following modified electroconvulsive therapy is associated with modulation of peripheral inflammatory markers in schizophrenia. World J Psychiatry 2026; 16(3): 114036
- URL: https://www.wjgnet.com/2220-3206/full/v16/i3/114036.htm
- DOI: https://dx.doi.org/10.5498/wjp.v16.i3.114036
Schizophrenia is a severe and complex neuropsychiatric disorder with a lifetime prevalence of approximately 1%, contributing substantially to the global disease burden and causing profound personal, familial, and societal challenges[1]. Its clinical presentation is characterized by positive symptoms (e.g., hallucinations and delusions), negative symptoms (e.g., avolition, social withdrawal), and cognitive impairment[2]. Although the pathogenesis of schizophrenia remains unclear, growing evidence suggests that its onset involves interactions among multiple factors, including neurodevelopmental abnormalities, neurodegenerative changes, genetic factors, and environmental factors. Among these, genetic factors play a significant role in the onset of the disease, accounting for approximately 60% to 70% of the total risk[3]. With the development of genomics and bioinformatics technologies, researchers have identified multiple susceptibility genes associated with schizophrenia that play key roles in processes such as neurodevelopment, synaptic function, and immune regulation[4,5]. Despite the considerable advances achieved in the present research, the treatment of schizophrenia continues to represent a significant challenge that necessitates further exploration.
In recent years, immune-inflammatory pathways have attracted increasing attention in schizophrenia research. Meta-analyses have consistently demonstrated elevated levels of proinflammatory cytokines, including tumor necrosis factor alpha (TNF-α), interleukin (IL)-1β, and IL-6, in patients with schizophrenia, suggesting a state of chronic low-grade inflammation[6]. These inflammatory mediators not only are involved in neuroinflammatory processes but also may play a role in emo
Modified electroconvulsive therapy (MECT) remains a widely used biological intervention for severe and treatment-resistant psychiatric conditions, including schizophrenia. It involves the induction of generalized seizures under ane
Recent studies have increasingly emphasized the role of inflammatory markers in the pathophysiological process of schizophrenia[13,14]. Inflammatory markers in peripheral blood, such as monocyte-to-lymphocyte ratio (MLR), the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and systemic immune-inflammatory index (SII), have been increasingly adopted in research due to their ease of assessment, low cost, and ability to accurately reflect the body's inflammatory state. However, there are few reports on the use of peripheral blood immune markers for efficacy analysis in clinical interventions for schizophrenia. This may be because the pathological mechanisms of schizophrenia are more complex, and changes in inflammatory markers may be influenced by multiple factors, including the stage of the disease, treatment methods, and individual differences. Therefore, further research into the role of inflammatory markers in schizophrenia and their relationship with treatment response has significant clinical implications.
Although previous studies have explored the effects of MECT on inflammatory mediators in patients with schizophrenia, these studies have focused mainly on changes in single inflammatory markers and have involved relatively small sample sizes[15]. Furthermore, current studies have not sufficiently explored the relationship between changes in these inflammatory markers and improvements in patients’ clinical symptoms. Therefore, through a single-arm, pre-post observational study with a large sample size, we aim to comprehensively evaluate the regulatory effect of MECT on peripheral blood immune-inflammatory markers in schizophrenia and to analyze the association between changes in these markers and clinical symptom improvement. The results of this study will provide new insights into the immu
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee at Huai’an Third People’s Hospital (No. 2022-23). The study included patients who were diagnosed with schizophrenia and who were treated at Huai'an Third People’s Hospital between July 2022 and December 2023 and who received MECT treatment.
All patients were independently diagnosed by two experienced psychiatrists to ensure diagnostic reliability. The inclusion criteria for the study participants were as follows: (1) Met the diagnostic criteria for schizophrenia according to the International Classification of Diseases, 10th Revision (ICD-10); (2) Had no contraindications for MECT, such as brain tumors or cranial injuries; and (3) Provided informed consent (the patient and guardian), with written consent forms signed. The exclusion criteria were as follows: (1) History of other mental disorders or substance abuse; (2) Severe or unstable physical illnesses; (3) Pregnant or breastfeeding women; and (4) Patients unsuitable for general anaesthesia.
All schizophrenia patients were evaluated and enrolled by experienced psychiatrists, and demographic and clinical information was collected, including age, sex, and duration of illness, was collected. The study used the Positive and Negative Syndrome Scale (PANSS) to assess the severity of symptoms before and after MECT treatment, with the efficacy being evaluated by the PANSS score reduction rate, calculated using the following formula: PANSS score reduction rate = [(pre-treatment score - post-treatment score)/pre-treatment score] × 100%[16]. The PANSS score reduction rate is used as an efficacy evaluation indicator, with ≤ 25% indicating minimal improvement, > 25% and ≤ 50% indicating moderate improvement, > 50% and ≤ 75% indicating marked improvement, and > 75% indicating extensive improvement[17].
MECT therapy was administered using the Thymatron System IV Integrated ECT Instrument (SOMATICS, LLC, United States). Patients fasted for 6 hours and abstained from fluids for 4 hours prior to each session. Pre-treatment management included intravenous administration of 0.5 mg atropine and continuous monitoring of electroencephalogram, electrocardiogram, blood pressure, pulse rate, and blood oxygen saturation. Anaesthesia was induced with intravenous propofol (1.0-2.0 mg/kg), and muscle relaxation was achieved with intravenous succinylcholine (0.5-1.5 mg/kg). Bilateral electrode placement was confirmed on the temporal regions. The initial stimulus dose was determined using the device’s age-based method, set at 50%-75% of the patient’s age. If a seizure failed or was incomplete, the stimulus dose was inc
Peripheral venous blood samples were drawn from all participants after an overnight fast of at least 8 hours during a fixed morning time window (7:00-9:00 AM). Samples were collected at two time points: (1) Before the first MECT session (baseline); and (2) Within 24 hours after the sixth MECT session (post-treatment). Critically, in accordance with standard MECT safety protocols, all patients were clinically assessed prior to each session. The presence of an intercurrent infection (e.g., body temperature > 37.5 °C) was a temporary contraindication for MECT. Therefore, all blood samples in this study were obtained from patients confirmed to be free of acute infection. Blood was collected into vacuum tubes containing the anticoagulant ethylenediaminetetraacetic acid. Complete blood count analysis was performed within 2 hours of collection using a Mindray BC-7500 automated haematology analyser. Quality control was performed daily using the Mindray BC-6D Haematology Control (Cat No. 105-004067-00; Lot: No. MB1125AN; Shenzhen Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China) prior to sample analysis. The laboratory adhered to strict internal quality control procedures and participated in external quality assessment programs. All cell counts are reported in standard units (× 109/L). The clinical reference ranges used for the key parameters in this study were: Neutrophils: 2.00-7.00 × 109/L, lymphocytes: 0.80-4.00 × 109/L, monocytes: 0.12-0.80 × 109/L, platelets: 100-300 × 109/L. The inflammatory indices were calculated from the absolute cell counts as follows: NLR = neutrophil count/Lymphocyte count; MLR = monocyte count/Lymphocyte count; PLR = platelet count/Lymphocyte count; SII = (neutrophil count × platelet count)/Lymphocyte count. The change (Δ) for each marker was defined as the pre-treatment value minus the post-treatment value.
IBM SPSS Statistics Version 26.0 (IBM Corp., Armonk, NY, United States) was used to analyze the data. The normality of the continuous variables was assessed using the Shapiro-Wilk test. Given that all the primary variables (including the PANSS score, PANSS score reduction rate, and peripheral blood inflammatory markers) did not follow a normal distribution, nonparametric statistical methods were employed for subsequent analyses. The Wilcoxon signed-rank test was used to compare changes in PANSS scores and inflammatory markers before and after MECT treatment. Spearman’s rank correlation analysis was used to assess the correlation between PANSS score reduction rates and inflammatory markers. The Mann-Whitney U test was used to compare differences in inflammatory markers between groups with different levels of efficacy. Subsequently, meaningful inflammatory markers were included in a binary logistic regression model to further assess their association with efficacy. Receiver operating characteristic (ROC) curve analysis was also performed to evaluate the value of these inflammatory markers for predicting treatment effects. The threshold for statistical sig
As shown in Table 1, a total of 619 patients, with 323 (52.18%) males and 296 (47.82%) females, were included in this study. Their mean ± SD age was 36.03 ± 10.69 years, and the mean disease duration was 9.24 ± 8.15 years.
| Characteristic | Value |
| Age, years, mean ± SD | 36.03 ± 10.69 |
| Male | 323 (52.18) |
| Female | 296 (47.82) |
| Illness duration, years, mean ± SD | 9.24 ± 8.15 |
| Baseline PANSS total score, mean ± SD | 94.85 ± 14.02 |
| Atypical antipsychotics | 587 (94.83) |
| Typical antipsychotics | 32 (5.17) |
As shown in Table 2, the PANSS scores, MLR, NLR, PLR, and SII were significantly lower after MECT treatment than before treatment, indicating a marked improvement in schizophrenia symptoms and a significant reduction in peripheral blood inflammatory markers.
| Before MECT | After MECT | Statistical analysis | ||
| mean ± SD | mean ± SD | Z | P value | |
| PANSS | 94.85 (14.02) | 57.05 (12.89) | 21.557 | < 0.001 |
| MLR | 0.30 (0.20) | 0.24 (0.18) | 8.486 | < 0.001 |
| NLR | 3.42 (2.27) | 2.59 (2.23) | 10.089 | < 0.001 |
| PLR | 161.67 (116.03) | 167.59 (124.73) | 3.303 | < 0.001 |
| SII | 763.01 (595.37) | 586.36 (470.14) | 8.291 | < 0.001 |
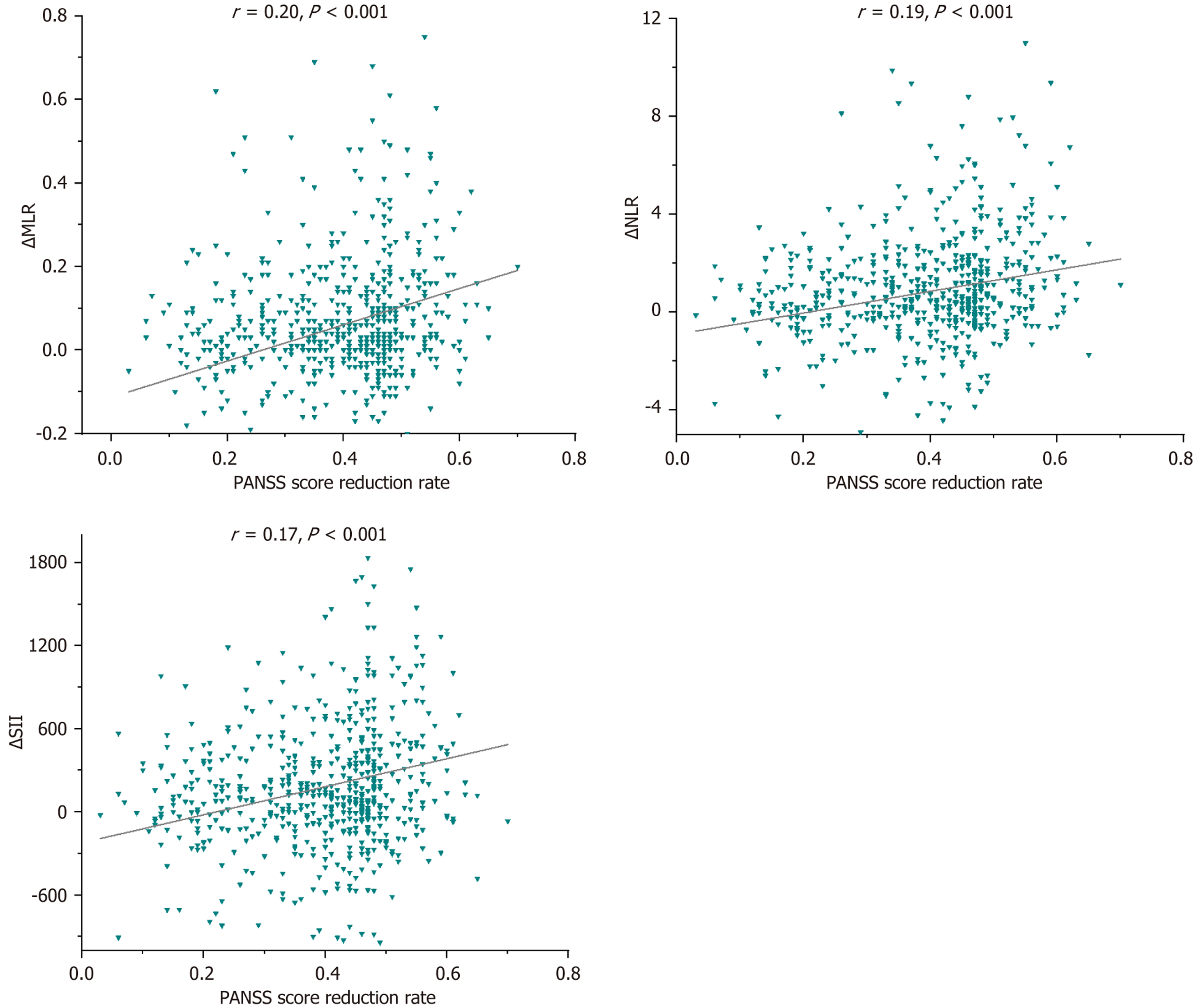
As shown in Figure 1, the ΔMLR showed a significant positive correlation with the PANSS score reduction rate (r = 0.207, P < 0.001). Similarly, the ΔNLR (r = 0.192, P < 0.001) and ΔSII (r = 0.171, P < 0.001) were also significantly correlated with greater clinical improvement. In contrast, the change in ΔPLR was not significantly associated with PANSS score reduction rate (r = -0.032, P = 0.458). These findings indicate that reductions in ΔMLR, ΔNLR, and ΔSII following MECT are significantly, albeit weakly, associated with the degree of clinical symptom improvement, with the change in ΔMLR demonstrating the strongest correlation.
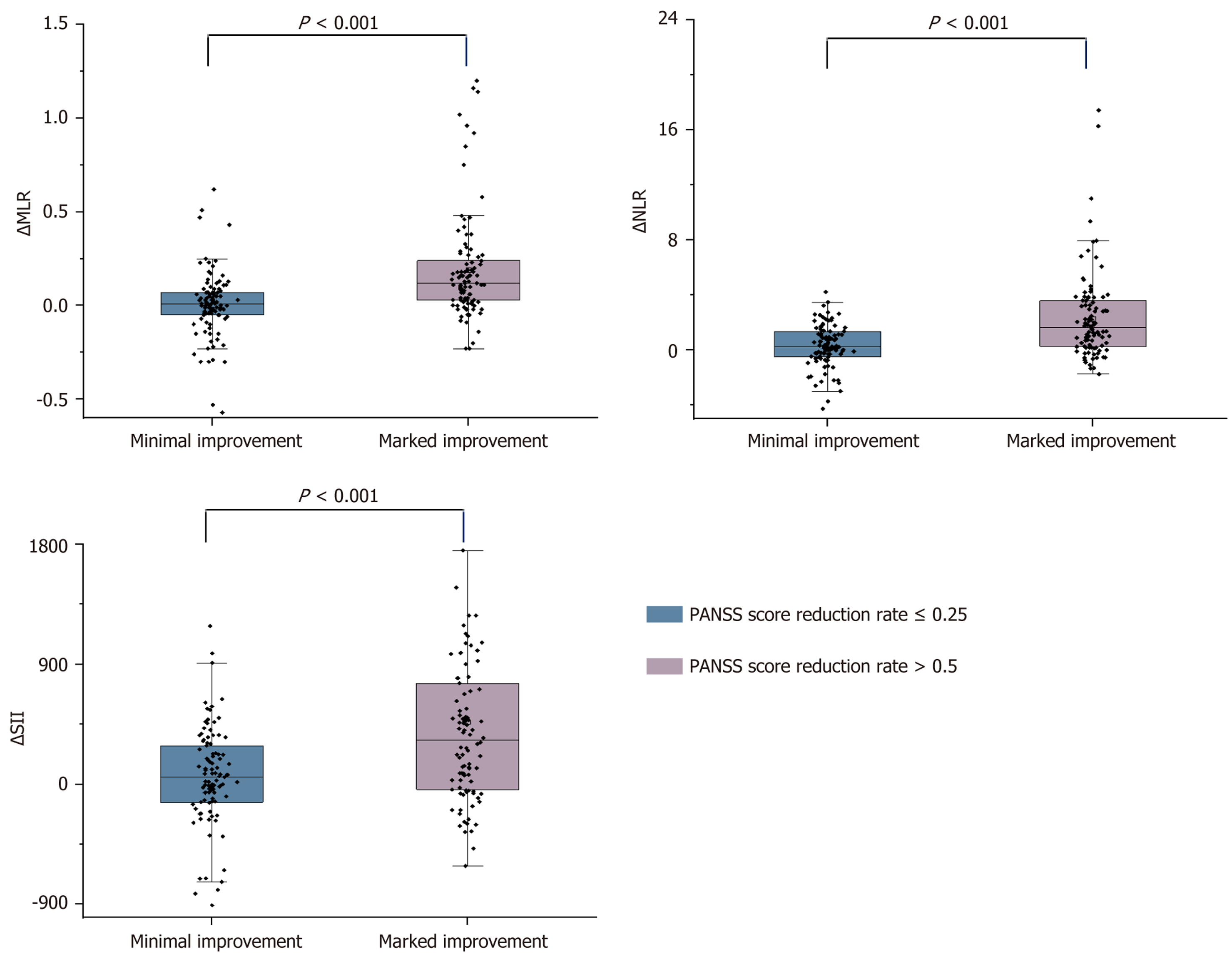
Patients with a PANSS score reduction rate ≤ 0.25 were included in the therapeutic minimal improvement group, while those with a PANSS score reduction rate > 0.5 were included in the therapeutic effective group. Changes in inflammatory markers before and after treatment were subsequently compared between the two groups. As shown in Figure 2, the analysis revealed that the effective group exhibited significantly greater values for all markers compared to the minimal improvement group. Specifically, for ΔMLR, the mean rank was significantly greater in the effective group (124.81, n = 98) than in the minimal improvement group (78.35, n = 103; U = 2714.00, Z = -5.662, P < 0.001). This pattern was consistent for ΔNLR (effective group mean rank = 123.41 vs minimal improvement group = 79.67; U = 2850.50, Z = -5.329, P < 0.001) and ΔSII (effective group mean rank = 119.70 vs minimal improvement group = 83.21; U = 3214.50, Z = -4.446, P < 0.001). These findings indicate that the greater the reduction in these inflammatory markers after treatment, the better the clinical symptom improvement attained through MECT treatment.
To evaluate the predictive value of changes in peripheral blood inflammatory markers for the clinical efficacy of MECT, three univariate logistic regression analyses were conducted. The response to treatment (defined as “effective” for a PANSS score reduction rate > 50% and “minimal improvement” for ≤ 25%) served as the dependent variable, while the ΔMLR, ΔNLR, and ΔSII were used as independent variables. As shown in Table 3, ΔMLR exhibited the most robust pre
| Increment | OR | 95%CI | P value | |
| ΔMLR | per 0.1 unit | 1.572 | 1.300-1.900 | < 0.001 |
| ΔNLR | per 0.1 unit | 1.044 | 1.026-1.062 | < 0.001 |
| ΔSII | per 10 unit | 1.010 | 1.005-1.015 | < 0.001 |
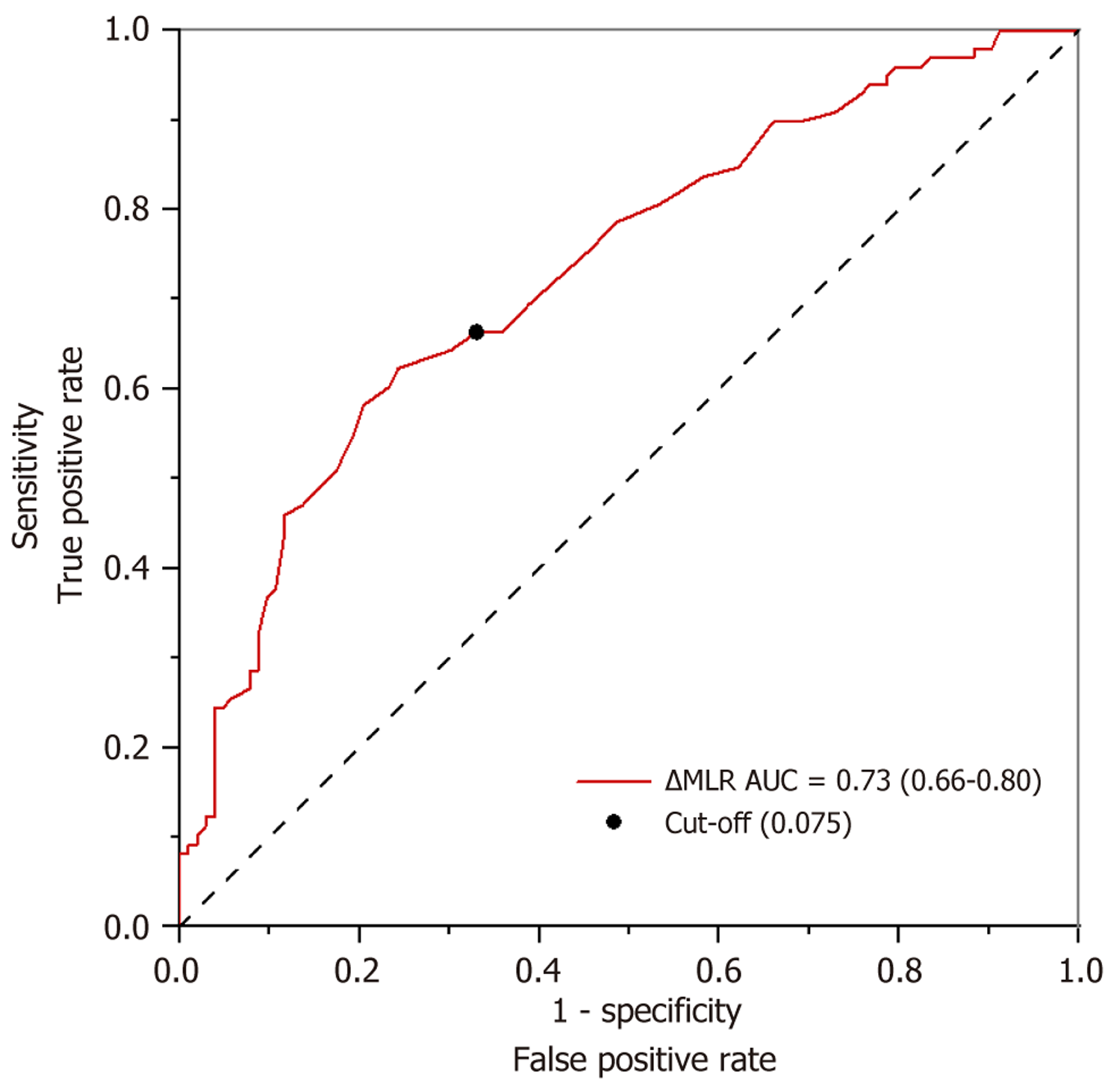
In order to evaluate the predictive efficacy of ΔMLR for MECT treatment outcomes, we conducted further ROC analysis. As shown in Figure 3, the area under the curve (AUC) for the ability of the ΔMLR to predict treatment efficacy was 0.731 (95%CI: 0.662-0.800, P < 0.001). In accordance with the prevailing standards within the academic community (where an AUC between 0.7 and 0.9 is indicative of moderate accuracy), this outcome substantiates the notion that the ΔMLR has considerable predictive value with regard to the clinical efficacy of MECT[18]. Furthermore, the optimal cutoff value for ΔMLR was determined to be 0.075 through calculation using Youden’s index. This value provides a preliminary quan
This study, which was based on large-sample data analysis, revealed that PANSS scores significantly decreased in schizophrenia patients following MECT treatment, which was accompanied by marked reductions in multiple peripheral blood immune-inflammatory markers (including MLR, NLR and SII). Correlation analysis further revealed a significant positive correlation between the degree of decrease in MLR, NLR, and SII with the rate of PANSS score reduction, suggesting that improvements in inflammatory levels are concomitant with clinical symptom improvement. To validate the regulatory effect of MECT on peripheral blood inflammatory markers, patients were divided into two groups based on treatment efficacy: A minimal improvement group and an effective group. The findings demonstrated that the decrease in the afo
To further advance the translation of research findings into clinical practice, we employed logistic regression analysis to evaluate the predictive value of inflammatory marker changes for predicting treatment response. The present study identified ΔMLR as a prominent predictor, with each 0.1-unit increase in ΔMLR being associated with a 57% greater probability of achieving significant clinical improvement. ROC analysis revealed an AUC value of 0.731 for ΔMLR with respect to predicting MECT efficacy, indicating its strong predictive performance. Additionally, the Youden index deter
First, the core findings of this study provide robust human clinical evidence for the anti-inflammatory effects of MECT. The substantial decrease in inflammatory markers after MECT treatment indicates that its therapeutic effects may extend beyond mere neurophysiological modulation. It is hypothesized that this systemic decrease in inflammation is not coincidental, but rather a consequence of MECT intervention within the intricate immune-neuro-endocrine network. The potential mechanisms may involve multiple pathways, such as the regulation of the hypothalamic-pituitary-adrenal (HPA) axis. MECT has been demonstrated to function as a highly efficacious physiological stressor, with the capacity to precisely regulate the HPA axis, thereby suppressing excessive glucocorticoid release[19]. We hypothesize that this effect disrupts the glucocorticoid resistance commonly observed in schizophrenia, thereby restoring the axis's inhibitory function on innate immune responses[20,21]. As a result, the proliferation and activation of proinflammatory cells (e.g. neutrophils and monocytes) are hindered. Additionally, MECT-induced seizures may activate vagal pathways. Acety
Our findings demonstrate that clinical improvement following MECT is associated with a reduction in peripheral inflammation, most reliably reflected by a decrease in ΔMLR. The strong predictive value of ΔMLR establishes it as a robust peripheral marker of treatment response. This association between peripheral immunomodulation and central symptom improvement invites the hypothesis that MECT may engage integrated body-brain pathways. For instance, it has been proposed that peripheral immune changes can influence brain function through various pathways[24,25], and MECT is known to upregulate neurotrophic factors such as BDNF[26,27]. However, these proposed mechanisms are not addressed by our peripheral measures and remain to be tested. Future studies directly assessing central immune activity (e.g., via neuroimaging) alongside peripheral markers are essential for exploring this possibility. The lack of decrease in the PLR after MECT likely reflects platelets’ dual role in inflammation and coagulation. As a physiological stressor, MECT may temporarily alter platelet and lymphocyte kinetics. Consequently, the distinct PLR response may capture a unique, MECT-induced immune-coagulation interaction. This represents an intriguing finding that merits further investigation in future studies.
The most translational medicine-oriented discovery in this study is the advancement of ΔMLR and other indicators from associative biomarkers to predictive biomarkers. Binary logistic regression confirmed ΔMLR as the strongest predictor of treatment response (OR = 1.57 per 0.1 unit increase), while ROC analysis further quantified its predictive value (AUC = 0.731, cutoff value = 0.075). The underlying mechanism for the predictive value of ΔMLR may be attributable to the fact that monocytes are among the fastest-responding and most plastic innate immune cells in the body[28,29]. Alterations in their numbers can serve as a reliable indicator of shifts in the body’s overall inflammatory state. Con
In summary, this study demonstrated that clinical improvement following MECT is associated with a concurrent reduction in peripheral inflammatory activity, as evidenced by decreases in MLR, NLR, and SII. The strong correlation and predictive capacity of ΔMLR establish it as a promising, readily accessible peripheral biomarker for predicting trea
This study has several limitations. First, the single-arm observational design, which lacked a control group (e.g., patients receiving only pharmacological treatment), prevents us from fully attributing the observed clinical imp
In conclusion, our large-sample study demonstrated that MECT significantly improved clinical symptoms and reduced peripheral inflammatory indices (MLR, NLR, PLR, and SII) in patients with schizophrenia. Notably, the reduction in MLR strongly correlated with clinical symptom improvement and served as a robust predictor of treatment response. These findings suggest that the therapeutic effects of MECT may be partially mediated through regulation of the immune-inflammatory system. MLR represents a promising peripheral inflammatory marker for predicting treatment efficacy, highlighting the potential for immune profiling to guide personalized therapeutic strategies in schizophrenia.
We are grateful to all the people who took part in this study.
| 1. | McCutcheon RA, Reis Marques T, Howes OD. Schizophrenia-An Overview. JAMA Psychiatry. 2020;77:201-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1189] [Cited by in RCA: 947] [Article Influence: 157.8] [Reference Citation Analysis (3)] |
| 2. | Mier D, Kirsch P. Social-Cognitive Deficits in Schizophrenia. Curr Top Behav Neurosci. 2017;30:397-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 3. | Guan F, Ni T, Zhu W, Williams LK, Cui LB, Li M, Tubbs J, Sham PC, Gui H. Integrative omics of schizophrenia: from genetic determinants to clinical classification and risk prediction. Mol Psychiatry. 2022;27:113-126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 4. | Sekar A, Bialas AR, de Rivera H, Davis A, Hammond TR, Kamitaki N, Tooley K, Presumey J, Baum M, Van Doren V, Genovese G, Rose SA, Handsaker RE; Schizophrenia Working Group of the Psychiatric Genomics Consortium, Daly MJ, Carroll MC, Stevens B, McCarroll SA. Schizophrenia risk from complex variation of complement component 4. Nature. 2016;530:177-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2231] [Cited by in RCA: 1816] [Article Influence: 181.6] [Reference Citation Analysis (0)] |
| 5. | Schizophrenia Working Group of the Psychiatric Genomics Consortium. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421-427. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7241] [Cited by in RCA: 5859] [Article Influence: 488.3] [Reference Citation Analysis (0)] |
| 6. | Miller BJ, Buckley P, Seabolt W, Mellor A, Kirkpatrick B. Meta-analysis of cytokine alterations in schizophrenia: clinical status and antipsychotic effects. Biol Psychiatry. 2011;70:663-671. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1525] [Cited by in RCA: 1398] [Article Influence: 93.2] [Reference Citation Analysis (1)] |
| 7. | Ortí-Casañ N, Boerema AS, Köpke K, Ebskamp A, Keijser J, Zhang Y, Chen T, Dolga AM, Broersen K, Fischer R, Pfizenmaier K, Kontermann RE, Eisel ULM. The TNFR1 antagonist Atrosimab reduces neuronal loss, glial activation and memory deficits in an acute mouse model of neurodegeneration. Sci Rep. 2023;13:10622. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 8. | Zhu X, Li R, Zhu Y, Zhou J, Huang J, Zhou Y, Tong J, Zhang P, Luo X, Chen S, Li Y, Tian B, Tan SP, Wang Z, Han X, Tian L, Li CR, Tan YL. Changes in Inflammatory Biomarkers in Patients with Schizophrenia: A 3-Year Retrospective Study. Neuropsychiatr Dis Treat. 2023;19:1597-1604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 9. | Rhee TG, Shim SR, Forester BP, Nierenberg AA, McIntyre RS, Papakostas GI, Krystal JH, Sanacora G, Wilkinson ST. Efficacy and Safety of Ketamine vs Electroconvulsive Therapy Among Patients With Major Depressive Episode: A Systematic Review and Meta-analysis. JAMA Psychiatry. 2022;79:1162-1172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 119] [Cited by in RCA: 104] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 10. | Kumar S, Mulsant BH, Liu AY, Blumberger DM, Daskalakis ZJ, Rajji TK. Systematic Review of Cognitive Effects of Electroconvulsive Therapy in Late-Life Depression. Am J Geriatr Psychiatry. 2016;24:547-565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 11. | Kritzer MD, Peterchev AV, Camprodon JA. Electroconvulsive Therapy: Mechanisms of Action, Clinical Considerations, and Future Directions. Harv Rev Psychiatry. 2023;31:101-113. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 12. | van Buel EM, Patas K, Peters M, Bosker FJ, Eisel UL, Klein HC. Immune and neurotrophin stimulation by electroconvulsive therapy: is some inflammation needed after all? Transl Psychiatry. 2015;5:e609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 85] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 13. | Ansari Z, Pawar S, Seetharaman R. Neuroinflammation and oxidative stress in schizophrenia: are these opportunities for repurposing? Postgrad Med. 2022;134:187-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 24] [Article Influence: 6.0] [Reference Citation Analysis (1)] |
| 14. | Altamura AC, Buoli M, Pozzoli S. Role of immunological factors in the pathophysiology and diagnosis of bipolar disorder: comparison with schizophrenia. Psychiatry Clin Neurosci. 2014;68:21-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 15. | 15 Han KY, Wang CM, Du CB, Qiao J, Wang YL, Lv LZ. Treatment outcomes and cognitive function following electroconvulsive therapy in patients with severe depression. World J Psychiatry. 2023;13:949-957. [PubMed] [DOI] [Full Text] |
| 16. | Zhao M, Ma J, Li M, Zhu W, Zhou W, Shen L, Wu H, Zhang N, Wu S, Fu C, Li X, Yang K, Tang T, Shen R, He L, Huai C, Qin S. Different responses to risperidone treatment in Schizophrenia: a multicenter genome-wide association and whole exome sequencing joint study. Transl Psychiatry. 2022;12:173. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 20] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Shi Y, Li M, Song C, Xu Q, Huo R, Shen L, Xing Q, Cui D, Li W, Zhao J, He L, Qin S. Combined study of genetic and epigenetic biomarker risperidone treatment efficacy in Chinese Han schizophrenia patients. Transl Psychiatry. 2017;7:e1170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 18. | Mandrekar JN. Receiver operating characteristic curve in diagnostic test assessment. J Thorac Oncol. 2010;5:1315-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3839] [Cited by in RCA: 2941] [Article Influence: 183.8] [Reference Citation Analysis (3)] |
| 19. | Haskett RF, Loo C. Adjunctive psychotropic medications during electroconvulsive therapy in the treatment of depression, mania, and schizophrenia. J ECT. 2010;26:196-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 20. | Perrin AJ, Pariante CM. Endocrine and immune effects of non-convulsive neurostimulation in depression: A systematic review. Brain Behav Immun. 2020;87:910-920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 21. | Pariante CM. Why are depressed patients inflamed? A reflection on 20 years of research on depression, glucocorticoid resistance and inflammation. Eur Neuropsychopharmacol. 2017;27:554-559. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 340] [Cited by in RCA: 289] [Article Influence: 32.1] [Reference Citation Analysis (0)] |
| 22. | Borovikova LV, Ivanova S, Zhang M, Yang H, Botchkina GI, Watkins LR, Wang H, Abumrad N, Eaton JW, Tracey KJ. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature. 2000;405:458-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3434] [Cited by in RCA: 3068] [Article Influence: 118.0] [Reference Citation Analysis (3)] |
| 23. | Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, Li JH, Wang H, Yang H, Ulloa L, Al-Abed Y, Czura CJ, Tracey KJ. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature. 2003;421:384-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2744] [Cited by in RCA: 2533] [Article Influence: 110.1] [Reference Citation Analysis (0)] |
| 24. | Prinz M, Priller J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nat Rev Neurosci. 2014;15:300-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1166] [Cited by in RCA: 1040] [Article Influence: 86.7] [Reference Citation Analysis (0)] |
| 25. | Saederup N, Cardona AE, Croft K, Mizutani M, Cotleur AC, Tsou CL, Ransohoff RM, Charo IF. Selective chemokine receptor usage by central nervous system myeloid cells in CCR2-red fluorescent protein knock-in mice. PLoS One. 2010;5:e13693. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 521] [Cited by in RCA: 495] [Article Influence: 30.9] [Reference Citation Analysis (0)] |
| 26. | Rocha RB, Dondossola ER, Grande AJ, Colonetti T, Ceretta LB, Passos IC, Quevedo J, da Rosa MI. Increased BDNF levels after electroconvulsive therapy in patients with major depressive disorder: A meta-analysis study. J Psychiatr Res. 2016;83:47-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 97] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 27. | Meshkat S, Alnefeesi Y, Jawad MY, D Di Vincenzo J, B Rodrigues N, Ceban F, Mw Lui L, McIntyre RS, Rosenblat JD. Brain-Derived Neurotrophic Factor (BDNF) as a biomarker of treatment response in patients with Treatment Resistant Depression (TRD): A systematic review & meta-analysis. Psychiatry Res. 2022;317:114857. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 47] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 28. | Yang J, Zhang L, Yu C, Yang XF, Wang H. Monocyte and macrophage differentiation: circulation inflammatory monocyte as biomarker for inflammatory diseases. Biomark Res. 2014;2:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 900] [Cited by in RCA: 807] [Article Influence: 67.3] [Reference Citation Analysis (0)] |
| 29. | Shi C, Pamer EG. Monocyte recruitment during infection and inflammation. Nat Rev Immunol. 2011;11:762-774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2564] [Cited by in RCA: 2370] [Article Influence: 158.0] [Reference Citation Analysis (0)] |
Open Access: This article is an open-access article that was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution NonCommercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: https://creativecommons.org/Licenses/by-nc/4.0/